Do B Vitamins Enhance the Effect of Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Diseases? A Systematic Review of Clinical Trials
Abstract
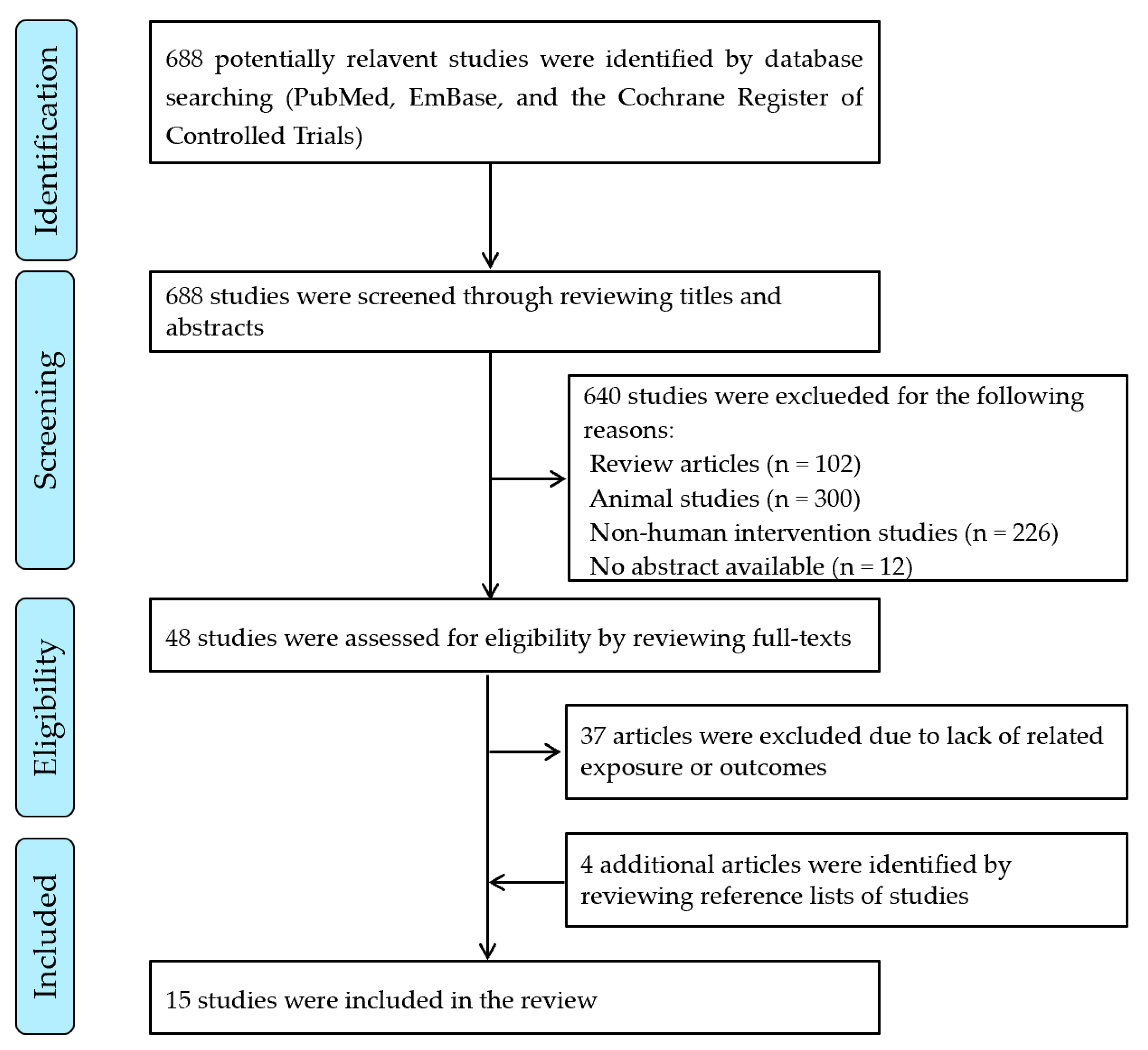
:1. Introduction
2. Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Study Quality Evaluation
3. Results
4. Discussion
4.1. B Vitamins and Omega-3 PUFAs Supplementation and CVD Risk Factors
4.2. B Vitamins and Omega-3 PUFAs Supplementation and CVD Risk
4.3. Possible Mechanisms
4.4. Implications for Practice
4.5. Study Limitations and Methodology Challenges
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
References
- He, K.; Merchant, A.; Rimm, E.B.; Rosner, B.A.; Stampfer, M.J.; Wllett, W.C.; Ascherio, A. Folate, vitamin B6, and B12 intakes in relation to risk of stroke among men. Stroke 2004, 35, 169–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, F.B.; Bronner, L.; Willett, W.C.; Stampfer, M.J.; Rexrode, K.; Albert, C.; Hunter, D.; Manson, J.E. Fish and omega-3 fatty acid intake and risk of coronary heart disease in women. JAMA 2002, 287, 1815–1821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, T.; Li, K.; Asimi, S.; Chen, Q.; Li, D. Effect of vitamin B-12 and n-3 polyunsaturated fatty acids on plasma homocysteine, ferritin, C-reaction protein, and other cardiovascular risk factors: A randomized controlled trial. Asia Pac. J. Clin. Nutr. 2015, 24, 403–411. [Google Scholar] [CrossRef] [PubMed]
- De Natale, C.; Minerva, V.; Patti, L.; Mazzarella, R.; Ciano, O.; Maione, S.; Luongo, D.; Naviglio, D.; Marotta, G.; Turco, S.; et al. Effects of baked products enriched with n-3 fatty acids, folates, beta-glucans, and tocopherol in patients with mild mixed hyperlipidemia. J. Am. Coll. Nutr. 2012, 31, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Garaiova, I.; Muchová, J.; Nagyová, Z.; Mišľanová, C.; Oravec, S.; Dukát, A.; Wang, D.; Plummer, S.F.; Ďuračková, Z. Effect of a plant sterol, fish oil and B vitamin combination on cardiovascular risk factors in hypercholesterolemic children and adolescents: A pilot study. Nutr. J. 2013, 12, 7. [Google Scholar] [CrossRef] [Green Version]
- Blacher, J.; Czernichow, S.; Paillard, F.; Ducimetiere, P.; Hercberg, S.; Galan, P. Cardiovascular effects of B-vitamins and/or N-3 fatty acids: The SU.FOL.OM3 trial. Int. J. Cardiol. 2013, 167, 508–513. [Google Scholar] [CrossRef]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- de Bree, A.; Mennen, L.I.; Hercberg, S.; Galan, P. Evidence for a protective (synergistic?) effect of B-vitamins and omega-3 fatty acids on cardiovascular diseases. Eur. J. Clin. Nutr. 2004, 58, 732–744. [Google Scholar] [CrossRef]
- Cao, Y.; Lu, L.; Liang, J.; Liu, M.; Li, X.; Sun, R.; Zheng, Y.; Zhang, P. Omega-3 Fatty Acids for Primary and Secondary Prevention of Cardiovascular Disease. Cell Biochem. Biophys. 2015, 72, 77–81. [Google Scholar] [CrossRef]
- Martí-Carvajal, A.J.; Solà, I.; Lathyris, D.; Dayer, M. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst. Rev. 2017, 8, CD006612. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, D.J.A.; Spence, J.D.; Giovannucci, E.L.; Kim, Y.I.; Josse, R.G.; Vieth, R.; Sahye-Pudaruth, S.; Paquette, M.; Patel, D.; Mejia, S.; et al. Supplemental Vitamins and Minerals for Cardiovascular Disease Prevention and Treatment. J. Am. Coll. Cardiol. 2021, 77, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Haglund, O.; Hamfelt, A.; Hambraeus, L.; Saldeen, T. Effects of fish oil supplemented with pyridoxine and folic acid on homocysteine, atherogenic index, fibrinogen and plasminogen activator inhibitor-1 in man. Nutr. Res. 1993, 13, 1351–1365. [Google Scholar] [CrossRef]
- Kemse, N.G.; Kale, A.A.; Joshi, S.R. A combined supplementation of omega-3 fatty acids and micronutrients (folic acid, vitamin B12) reduces oxidative stress markers in a rat model of pregnancy induced hypertension. PLoS ONE 2014, 9, e111902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 341, c6273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [Green Version]
- Academy of Nutrition and Dietetics. Evidence Analysis Manual. 2016. Available online: https://www.andeal.org/evidence-analysis-manual (accessed on 21 November 2018).
- De Edelenyi, F.S.; Vergnaud, A.-C.; Ahluwalia, N.; Julia, C.; Hercberg, S.; Blacher, J.; Galán, P. Effect of B-vitamins and n-3 PUFA supplementation for 5 years on blood pressure in patients with CVD. Br. J. Nutr. 2012, 107, 921–927. [Google Scholar] [CrossRef] [Green Version]
- Fonollá, J.; Lopez-Huertas, E.; Machado, F.J.; Molina, D.; Álvarez, I.; Mármol, E.; Navas, M.; Palacín, E.; García-Valls, M.J.; Remón, B.; et al. Milk enriched with "healthy fatty acids" improves cardiovascular risk markers and nutritional status in human volunteers. Nutrition 2009, 25, 408–414. [Google Scholar] [CrossRef]
- Earnest, C.P.; Kupper, J.S.; Thompson, A.M.; Guo, W.; Church, T. Complementary effects of multivitamin and omega-3 fatty acid supplementation on indices of cardiovascular health in individuals with elevated homocysteine. Int. J. Vitam. Nutr. Res. 2012, 82, 41–52. [Google Scholar] [CrossRef]
- Carrero, J.J.; López-Huertas, E.; Salmerón, L.M.; Baró, L.; Ros, E. Daily supplementation with (n-3) PUFAs, oleic acid, folic acid, and vitamins B-6 and E increases pain-free walking distance and improves risk factors in men with peripheral vascular disease. J. Nutr. 2005, 135, 1393–1399. [Google Scholar] [CrossRef]
- Benito, P.; Caballero-Villarraso, J.; Moreno, J.; Gutiérrez-Alcántara, C.; Muñoz, C.; Rojo-Martínez, G.; Garcia, S.; Soriguer, F.C. Effects of milk enriched with omega-3 fatty acid, oleic acid and folic acid in patients with metabolic syndrome. Clin. Nutr. 2006, 25, 581–587. [Google Scholar] [CrossRef]
- Carrero, J.J.; Fonollá, J.; Marti, J.L.; Jiménez, J.; Boza, J.J.; López-Huertas, E. Intake of fish oil, oleic acid, folic acid, and vitamins B-6 and E for 1 year decreases plasma C-reactive protein and reduces coronary heart disease risk factors in male patients in a cardiac rehabilitation program. J. Nutr. 2007, 137, 384–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrero, J.J.; López-Huertas, E.; Salmerón, L.M.; Ramosc, V.E.; Barób, L.; Rosc, E. Simvastatin and supplementation with ω-3 polyunsaturated fatty acids and vitamins improves claudication distance in a randomized PILOT study in patients with peripheral vascular disease. Nutr. Res. 2006, 26, 637–643. [Google Scholar] [CrossRef]
- Baró, L.; Fonollá, J.; Peña, J.L.; Martínez-Férez, A.; Lucena, A.; Jiménez, J.; Boza, J.J.; López-Huertas, E. n-3 Fatty acids plus oleic acid and vitamin supplemented milk consumption reduces total and LDL cholesterol, homocysteine and levels of endothelial adhesion molecules in healthy humans. Clin. Nutr. 2003, 22, 175–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrero, J.J.; Baró, L.; Fonollá, J.; González-Santiago, M.; Martinez-Ferez, A.; Castillo, R.; Jiménez, J.; Boza, J.J.; López-Huertas, Y.E. Cardiovascular effects of milk enriched with omega-3 polyunsaturated fatty acids, oleic acid, folic acid, and vitamins E and B6 in volunteers with mild hyperlipidemia. Nutrition 2004, 20, 521–527. [Google Scholar] [CrossRef]
- Ji, X.; Grandner, M.A.; Liu, J. The relationship between micronutrient status and sleep patterns: A systematic review. Public Health Nutr. 2017, 20, 687–701. [Google Scholar] [CrossRef] [Green Version]
- Almassinokiani, F.; Kashanian, M.; Akbari, P.; Mossayebi, E.; Sadeghian, E. Folic acid supplementation reduces plasma homocysteine in postmenopausal women. J. Obstet. Gynaecol. 2016, 36, 492–495. [Google Scholar] [CrossRef]
- Dawson, S.L.; Bowe, S.J.; Crowe, T.C. A combination of omega-3 fatty acids, folic acid and B-group vitamins is superior at lowering homocysteine than omega-3 alone: A meta-analysis. Nutr. Res. 2016, 36, 499–508. [Google Scholar] [CrossRef]
- Liu, M.; Wang, Z.; Liu, S.; Liu, Y.; Ma, Y.; Liu, Y.; Xue, M.; Li, Q.; Zhang, X.; Zhang, S.; et al. Effect of B vitamins supplementation on cardio-metabolic factors in patients with stable coronary artery disease: A randomized double-blind trial. Asia Pac. J. Clin. Nutr. 2020, 29, 245–252. [Google Scholar] [CrossRef]
- Micronutrient Information Center, Oregon State University. Essential Fatty Acids. 2014. Available online: http://lpi.oregonstate.edu/mic/other-nutrients/essential-fatty-acids (accessed on 27 December 2021).
- Pooya, S.H.; Jalali, M.D.; Jazayery, A.D.; Saedisomeolia, A.; Eshraghian, M.R.; Toorang, F. The efficacy of omega-3 fatty acid supplementation on plasma homocysteine and malondialdehyde levels of type 2 diabetic patients. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 326–331. [Google Scholar] [CrossRef]
- Grundt, H.; Nilsen, D.W.; Hetland, O.; Mansoor, M.A.; Aarsland, T.; Woie, L. Atherothrombogenic risk modulation by n-3 fatty acids was not associated with changes in homocysteine in subjects with combined hyperlipidaemia. Thromb. Haemost. 1999, 81, 561–565. [Google Scholar] [CrossRef]
- Sara, J.D.; Prasad, M.; Zhang, M.; Lennon, R.J.; Herrmann, J.; Lerman, L.O.; Lerman, A. High-sensitivity C-reactive protein is an independent marker of abnormal coronary vasoreactivity in patients with non-obstructive coronary artery disease. Am. Heart J. 2017, 190, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; AlAbdulghafoor, F.K.; Summerbell, C.D.; Worthington, H.V.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, 7, CD003177. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhou, Y.; Chen, S.-H.; Zhao, X.-L.; Ran, L.; Zeng, X.-L.; Wu, Y.; Chen, J.-L.; Kang, C.; Shu, F.-R.; et al. Fish Oil Supplements Lower Serum Lipids and Glucose in Correlation with a Reduction in Plasma Fibroblast Growth Factor 21 and Prostaglandin E2 in Nonalcoholic Fatty Liver Disease Associated with Hyperlipidemia: A Randomized Clinical Trial. PLoS ONE 2015, 10, e0133496. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Mayneris-Perxachs, J.; Lovegrove, J.A.; Todd, S.; Yaqoob, P. Fish-oil supplementation alters numbers of circulating endothelial progenitor cells and microparticles independently of eNOS genotype. Am. J. Clin. Nutr. 2014, 100, 1232–1243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masuda, D.; Miyata, Y.; Matsui, S.; Yamashita, S. Omega-3 fatty acid ethyl esters improve low-density lipoprotein subclasses without increasing low-density lipoprotein-cholesterol levels: A phase 4, randomized study. Atherosclerosis 2020, 292, 163–170. [Google Scholar] [CrossRef] [Green Version]
- Gidding, S.S.; Prospero, C.; Hossain, J.; Zappalla, F.; Balagopal, P.; Falkner, B.; Kwiterovich, P. A double-blind randomized trial of fish oil to lower triglycerides and improve cardiometabolic risk in adolescents. J. Pediatr. 2014, 165, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Alves Luzia, L.; Mendes Aldrighi, J.; Teixeira Damasceno, N.R.; Sampaio, G.R.; Aparecida, R.; Soares, R.A.M.; Silva, I.T.; De Queiroz Mello, A.P.; Carioca, A.A.F.; da Silva Torres, A.F. Fish Oil and Vitamin E Change Lipid Profiles and Anti-Ldl-Antibodies in Two Different Ethnic Groups of Women Transitioning through Menopause. Nutr. Hosp. 2015, 32, 165–174. [Google Scholar] [CrossRef]
- Allaire, J.; Vors, C.; Harris, W.S.; Jackson, K.H.; Tchernof, A.; Couture, P.; Lamarche, B. Comparing the serum TAG response to high-dose supplementation of either DHA or EPA among individuals with increased cardiovascular risk: The ComparED study. Br. J. Nutr. 2019, 121, 1223–1234. [Google Scholar] [CrossRef] [Green Version]
- Tani, S.; Matsuo, R.; Yagi, T.; Matsumoto, N. Administration of eicosapentaenoic acid may alter high-density lipoprotein heterogeneity in statin-treated patients with stable coronary artery disease: A 6-month randomized trial. J. Cardiol. 2020, 75, 282–288. [Google Scholar] [CrossRef] [Green Version]
- Yokote, K.; Niwa, K.; Hakoda, T.; Oh, F.; Kajimoto, Y.; Fukui, T.; Kim, H.; Noda, Y.; Lundström, T.; Yajima, T. Short-Term Efficacy (at 12 Weeks) and Long-Term Safety (up to 52 Weeks) of Omega-3 Free Fatty Acids (AZD0585) for the Treatment of Japanese Patients With Dyslipidemia―A Randomized, Double-Blind, Placebo-Controlled, Phase III Study. Circ. J. 2020, 84, 994–1003. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Q.; Zhang, Z.; Wang, P.; Zhang, B.; Chen, C.; Zhang, C.; Su, Y. EPA+DHA, but not ALA, Improved Lipids and Inflammation Status in Hypercholesterolemic Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. Mol. Nutr. Food Res. 2019, 63, e1801157. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Briskey, D.; Nalley, J.O.; Ganuza, E. Omega-3 Eicosapentaenoic Acid (EPA) Rich Extract from the Microalga Nannochloropsis Decreases Cholesterol in Healthy Individuals: A Double-Blind, Randomized, Placebo-Controlled, Three-Month Supplementation Study. Nutrients 2020, 12, 1869. [Google Scholar] [CrossRef] [PubMed]
- Sunkara, A.; Raizner, A. Supplemental Vitamins and Minerals for Cardiovascular Disease Prevention and Treatment. Methodist Debakey Cardiovasc. J. 2019, 15, 179–184. [Google Scholar] [CrossRef]
- Manson, J.E.; Bassuk, S.S.; Cook, N.R.; Lee, I.M.; Mora, S.; Albert, C.M.; Buring, J.E. VITAL Research Group. Vitamin D, Marine n-3 Fatty Acids, and Primary Prevention of Cardiovascular Disease: Current Evidence. Circ. Res. 2020, 126, 112–128. [Google Scholar] [CrossRef] [PubMed]
- Sable, P.S.; Kale, A.A.; Joshi, S.R. Prenatal omega 3 fatty acid supplementation to a micronutrient imbalanced diet protects brain neurotrophins in both the cortex and hippocampus in the adult rat offspring. Metabolism 2013, 62, 1607–1622. [Google Scholar] [CrossRef] [PubMed]
- Dhobale, M.; Joshi, S. Altered maternal micronutrients (folic acid, vitamin B 12) and omega 3 fatty acids through oxidative stress may reduce neurotrophic factors in preterm pregnancy. J. Matern. Fetal Neonatal Med. 2012, 25, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Bergami, R.; Maranesi, M.; Marchetti, M.; Sangiorgi, Z.; Tolomelli, B. Influence of dietary n-3 polyunsaturated fatty acids on plasma lipemic effect of vitamin B6 deficiency. Int. J. Vitam. Nutr. Res. 1999, 69, 315–321. [Google Scholar] [CrossRef]
- Durand, P.; Prost, M.; Blache, D. Pro-thrombotic effects of a folic acid deficient diet in rat platelets and macrophages related to elevated homocysteine and decreased n-3 polyunsaturated fatty acids. Atherosclerosis 1996, 121, 231–243. [Google Scholar] [CrossRef]
- Starkebaum, G.; Harlan, J.M. Endothelial cell injury due to copper-catalyzed hydrogen peroxide generation from homocysteine. J. Clin. Investig. 1986, 77, 1370–1376. [Google Scholar] [CrossRef] [Green Version]
- Hayden, M.S.; Ghosh, S. Regulation of NF-kappaB by TNF family cytokines. Semin. Immunol. 2014, 26, 253–266. [Google Scholar] [CrossRef] [Green Version]
- Jones, M.L.; Mark, P.J.; Mori, T.A.; Keelan, J.A.; Waddell, B.J. Maternal dietary omega-3 fatty acid supplementation reduces placental oxidative stress and increases fetal and placental growth in the rat. Biol. Reprod. 2013, 88, 37. [Google Scholar] [CrossRef] [PubMed]
- Loscalzo, J. Homocysteine trials--clear outcomes for complex reasons. N. Engl. J. Med. 2006, 354, 1629–1632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steffen, L.M.; Folsom, A.R.; Cushman, M.; Jacobs, D.R., Jr.; Rosamond, W.D. Greater fish, fruit, and vegetable intakes are related to lower incidence of venous thromboembolism: The Longitudinal Investigation of Thromboembolism Etiology. Circulation 2007, 115, 188–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, J.; Wilmot, K.A.; Ghasemzadeh, N.; Molloy, D.L.; Burkman, G.; Mekonnen, G.; Gongora, M.C.; Quyyumi, A.A.; Sperling, L.S. Mediterranean Dietary Patterns and Cardiovascular Health. Annu. Rev. Nutr. 2015, 35, 425–449. [Google Scholar] [CrossRef]
- Rosato, V.; Temple, N.J.; La Vecchia, C.; Castellan, G.; Tavani, A.; Guercio, V. Mediterranean diet and cardiovascular disease: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2019, 58, 173–191. [Google Scholar] [CrossRef]
- Zhao, M.; Wu, G.; Li, Y.; Wang, X.; Hou, F.F.; Xu, X.; Qin, X.; Cai, Y. Meta-analysis of folic acid efficacy trials in stroke prevention: Insight into effect modifiers. Neurology 2017, 88, 1830–1838. [Google Scholar] [CrossRef]
- Chilton, F.H.; Dutta, R.; Reynolds, L.M.; Sergeant, S.; Mathias, R.A.; Seeds, M.C. Precision Nutrition and Omega-3 Polyunsaturated Fatty Acids: A Case for Personalized Supplementation Approaches for the Prevention and Management of Human Diseases. Nutrients 2017, 9, 1165. [Google Scholar] [CrossRef]
| Reference | Year | Location | Participants | Design | Intervention | Duration | Outcomes |
|---|---|---|---|---|---|---|---|
| Haglund et al. [12] | 1993 | Sweden | n = 12, male volunteers, healthy or with slightly to moderately increased blood lipids | A double-blind cross-over study | 30 mL of fish oil with 30 mL of orange juice/day for 4 weeks followed by a 5-week washout, and then 30 mL of fish oil with 30 mL of orange juice/day supplemented with vitamin B6 (80 mg/day) and folic acid (10 mg/day) for 4 weeks. Fish oil contained 13% DHA and 19% EPA. | Fish oil- supplemented orange juice for 4 weeks followed by a 5-week washout period and then B vitamins + fish oil-supplemented orange juice for 4 weeks |
|
| Baró et al. [24] | 2003 | Spain | n = 30, healthy volunteers (50% man) | CS without control | 500 mL/day of semi-skimmed milk for 4 weeks and then 500 mL/day of the enriched milk (containing omega-3 PUFAs, oleic acid, vitamins E, B6, and folic acid) for 8 further weeks. | 8 weeks |
|
| Carrero et al. [26] | 2004 | Spain | n = 30, volunteers (50% man) with mild hyperlipidemia | CS without control | 500 mL/day of semi-skimmed milk for 4 weeks and then 500 mL/day of the enriched milk (containing omega-3 PUFAs, oleic acid, vitamins E, B6, and folic acid) for 8 weeks. | 8 weeks |
|
| Carrero et al. [20] | 2005 | Spain | n = 60, patients with PVD and intermittent claudication (100% man) | RCT with parallel- group design | Supplemented group: 500 mL/day of a fortified dairy product containing EPA, DHA, oleic acid, folic acid, and vitamins B6, D, A, and E; Control group: 500 mL/day of semi-skimmed milk with added vitamins A and D. | 12 months |
|
| Benito et al. [21] | 2006 | Spain | n = 72, patients with metabolic syndrome | A randomized, placebo- controlled and open clinical trial of parallel design | Control group: 500 mL semi-skimmed milk/day; Test group: 36 patients consumed 500 mL enriched milk (5.7 g of oleic acid, 0.2 g of omega-3 PUFAs, 150 μg of folic acid and 7.5 mg of vitamin E) /day. | 3 months |
|
| Carrero et al. [23] | 2006 | Spain | n = 40, male PVD patients | A longitudinal, randomized, controlled study | Group 1: 500 mL/day fortified dairy product containing fish oil, oleic acid, folic acid, vitamins A, D, E, and B6; Group 2: 500 mL/day fortified dairy product + simvastatin (20 mg/day); Group 3: 500 mL/day semi-skimmed milk; Group 4: 500 mL/day semi-skimmed milk + simvastatin (20 mg/day). | 12 months |
|
| Carrero et al. [22] | 2007 | Spain | n = 40, patients suffered from MI (100% man) | RCT with parallel- group design | Supplemented group: 500 mL/day of a fortified dairy product containing EPA, DHA, oleic acid, folic acid, and vitamins B6, D, A, and E; Control group: 500 mL/day of semi-skimmed milk with added vitamins A and D. | 12 months |
|
| Fonollá et al. [18] | 2009 | Spain | n = 297, participants (84.5% man) with moderate cardiovascular risk | RCT with parallel- group design | Group 1: 500 mL/day enriched milk (containing omega-3 PUFAs, oleic acid, vitamins E, B6, and folic acid); Group 2: 500 mL/day skimmed milk; Group 3 (control): 500 mL/day semi-skimmed milk. | 12 months |
|
| Galan et al. [14] | 2010 | France | n = 2501 patients (79.5% man) with CVD (MI, stroke and unstable angina) | RCT with a 2 × 2 factorial design | Group 1: B-vitamins [5-methyl-THF (560 μg), vitamin B6 (3 mg) and vitamin B12 (20 μg)] and a placebo capsule for omega-3 PUFAs; Group 2: omega-3 PUFAs (600 mg of EPA and DHA at a ratio of 2:1) and a placebo capsule for B-vitamins; Group 3: both B-vitamins and omega-3 PUFAs; Group 4: placebo capsules for both treatments. | 4.7 years |
|
| Szabo et al. [17] | 2012 | France | n = 2501, patients (79.5% man) with a history of CVD (MI, stroke and unstable angina) | RCT with a 2 × 2 factorial design | Group 1: B-vitamins [5-methyl-THF (560 μg), vitamin B6 (3 mg) and vitamin B12 (20 μg)] and a placebo capsule for omega-3 PUFAs; Group 2: omega-3 PUFAs (600 mg of EPA and DHA at a ratio of 2:1) and a placebo capsule for B-vitamins; Group 3: both B-vitamins and omega-3 PUFAs; Group 4: placebo capsules for both treatments. | 4.7 years |
|
| Earnest et al. [19] | 2012 | UK | n = 100, participants with elevated Hcy (>8.0 umol/L) | RCT with 2 × 2 factorial design | Group 1: placebo; Group 2: MVit (vit C: 200 mg; vitE: 400 IU; vit B6: 25 mg; folic acid: 400 ug; vit B12: 400 ug) + placebo; Group 3: omega-3 PUFAs (2 g omega-3 PUFAs, 760 mg EPA, 440 mg DHA) + placebo; Group 4: MVit + omega-3 PUFAs. | 12 weeks |
|
| De Natale C et al. [4] | 2012 | Italy | n = 16, participants (43.8% man) with mild plasma lipid abnormalities | a randomized crossover design | Group 1: a diet containing baked products enriched with active nutrients [β-glucans (3.6 g/day), folic acid (1620 μg/day), long-chain (800 mg/day) and short-chain (400 mg/day) omega-3 PUFAs, and tocopherols (120 mg/day); Group 2: a diet containing the same products without active nutrients (control diet). | 1 month for each of the control and enriched diets and then cross over to the other diet |
|
| Blacher et al. [6] | 2013 | France | n = 2501, patients (79.5% man) with a past history of cardio- or cerebrovascular diseases | RCT with a 2 × 2 factorial design | Group 1: B-vitamins [(5-methyl-THF (560 μg), vitamin B6 (3 mg) and vitamin B12 (20 μg)] and a placebo capsule for omega-3 PUFAs; Group 2: omega-3 PUFAs (600 mg of EPA and DHA at a ratio of 2:1) and a placebo capsule for B-vitamins; Group 3: both B-vitamins and omega-3 PUFAs; Group 4: placebo capsules for both treatments. | 4.2 (± 1.0) years |
|
| Garaiova et al. [5] | 2013 | Slovakia | n = 25, hypercholesterolmic children and adolescents, mean age 16.4 ± 3.8 years | CS without control | A combination of plant sterols esters (1300 mg), fish oil (1000 mg EPA plus DHA) and vitamins B12 (50 μg), B6 (2.5 mg), folic acid (800 μg) and coenzyme Q10 (3 mg). | 16 weeks |
|
| Huang et al. [3] | 2015 | China | n = 38, healthy individuals, 57% man, 23 ± 3 years of old | RCT with parallel- group design | Group 1: vitamin B12 (1000 μg); Group 2: fish oil (2 g); Group 3: vitamin B12 (1000 μg) + fish oil (2 g) (each 1 g capsule provided 490 mg of 22:6 omega-3, and 98 mg of 20:5 omega-3). | 8 weeks |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, J.; Xun, P.-C.; Kolencik, M.; Yang, K.-F.; Fly, A.D.; Kahe, K. Do B Vitamins Enhance the Effect of Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Diseases? A Systematic Review of Clinical Trials. Nutrients 2022, 14, 1608. https://doi.org/10.3390/nu14081608
Zhu J, Xun P-C, Kolencik M, Yang K-F, Fly AD, Kahe K. Do B Vitamins Enhance the Effect of Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Diseases? A Systematic Review of Clinical Trials. Nutrients. 2022; 14(8):1608. https://doi.org/10.3390/nu14081608
Chicago/Turabian StyleZhu, Jie, Peng-Cheng Xun, Marissa Kolencik, Ke-Feng Yang, Alyce D. Fly, and Ka Kahe. 2022. "Do B Vitamins Enhance the Effect of Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Diseases? A Systematic Review of Clinical Trials" Nutrients 14, no. 8: 1608. https://doi.org/10.3390/nu14081608
APA StyleZhu, J., Xun, P.-C., Kolencik, M., Yang, K.-F., Fly, A. D., & Kahe, K. (2022). Do B Vitamins Enhance the Effect of Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Diseases? A Systematic Review of Clinical Trials. Nutrients, 14(8), 1608. https://doi.org/10.3390/nu14081608






