The Inverse Correlation of Isoflavone Dietary Intake and Headache in Peri- and Post-Menopausal Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Menopausal Status
2.3. Physical Assessment
2.4. Lifestyle Characteristics
2.5. Questionnaires
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lauritsen, C.G.; Chua, A.L.; Nahas, S.J. Current Treatment Options: Headache Related to Menopause-Diagnosis and Management. Curr. Treat. Options Neurol. 2018, 20, 7. [Google Scholar] [CrossRef] [PubMed]
- Terauchi, M.; Hiramitsu, S.; Akiyoshi, M.; Owa, Y.; Kato, K.; Obayashi, S.; Matsushima, E.; Kubota, T. Effects of the kampo formula tokishakuyakusan on headaches and concomitant depression in middle-aged women. Evid.-Based Complementary Altern. Med. Ecam 2014, 2014, 593560. [Google Scholar] [CrossRef] [PubMed]
- Lieba-Samal, D.; Wober, C. Sex hormones and primary headaches other than migraine. Curr. Pain Headache Rep. 2011, 15, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Ripa, P.; Ornello, R.; Degan, D.; Tiseo, C.; Stewart, J.; Pistoia, F.; Carolei, A.; Sacco, S. Migraine in menopausal women: A systematic review. Int. J. Women’s Health 2015, 7, 773–782. [Google Scholar] [CrossRef] [Green Version]
- Razeghi Jahromi, S.; Ghorbani, Z.; Martelletti, P.; Lampl, C.; Togha, M.; EHF-SAS. Association of diet and headache. J. Headache Pain 2019, 20, 106. [Google Scholar] [CrossRef] [Green Version]
- Ramsden, C.E.; Zamora, D.; Faurot, K.R.; Macintosh, B.; Horowitz, M.; Keyes, G.S.; Yuan, Z.-X.; Miller, V.; Lynch, C.; Honvoh, G.; et al. Dietary alteration of n-3 and n-6 fatty acids for headache reduction in adults with migraine: Randomized controlled trial. BMJ 2021, 374, n1448. [Google Scholar] [CrossRef]
- Odai, T.; Terauchi, M.; Suzuki, R.; Kato, K.; Hirose, A.; Miyasaka, N. Severity of subjective forgetfulness is associated with high dietary intake of copper in Japanese senior women: A cross-sectional study. Food Sci. Nutr. 2020, 8, 4422–4431. [Google Scholar] [CrossRef]
- Kobayashi, S.; Honda, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Both comprehensive and brief self-administered diet history questionnaires satisfactorily rank nutrient intakes in Japanese adults. J. Epidemiol. 2012, 22, 151–159. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, S.; Murakami, K.; Sasaki, S.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Comparison of relative validity of food group intakes estimated by comprehensive and brief-type self-administered diet history questionnaires against 16 d dietary records in Japanese adults. Public Health Nutr. 2011, 14, 1200–1211. [Google Scholar] [CrossRef]
- Hanson, B.S.; Isacsson, S.O.; Janzon, L.; Lindell, S.E. Social network and social support influence mortality in elderly men. The prospective population study of “Men born in 1914,” Malmö, Sweden. Am. J. Epidemiol. 1989, 130, 100–111. [Google Scholar] [CrossRef]
- Hunter, M.; Battersby, R.; Whitehead, M. Relationships between psychological symptoms, somatic complaints and menopausal status. Maturitas 1986, 8, 217–228. [Google Scholar] [CrossRef]
- van Keep, P.A.; Kellerhals, J.M. The impact of socio-cultural factors on symptom formation. Some results of a study on ageing women in Switzerland. Psychother. Psychosom. 1974, 23, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Zorita, S.; Gonzalez-Arceo, M.; Fernandez-Quintela, A.; Eseberri, I.; Trepiana, J.; Portillo, M.P. Scientific Evidence Supporting the Beneficial Effects of Isoflavones on Human Health. Nutrients 2020, 12, 3853. [Google Scholar] [CrossRef]
- Wei, H.; Bowen, R.; Cai, Q.; Barnes, S.; Wang, Y. Antioxidant and antipromotional effects of the soybean isoflavone genistein. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 1995, 208, 124–130. [Google Scholar] [CrossRef]
- Chen, L.R.; Chen, K.H. Utilization of Isoflavones in Soybeans for Women with Menopausal Syndrome: An Overview. Int. J. Mol. Sci. 2021, 22, 3212. [Google Scholar] [CrossRef]
- Perna, S.; Peroni, G.; Miccono, A.; Riva, A.; Morazzoni, P.; Allegrini, P.; Preda, S.; Baldiraghi, V.; Guido, D.; Rondanelli, M. Multidimensional Effects of Soy Isoflavone by Food or Supplements in Menopause Women: A Systematic Review and Bibliometric Analysis. Nat. Prod. Commun. 2016, 11, 1733–1740. [Google Scholar] [CrossRef] [Green Version]
- Glazier, M.G.; Bowman, M.A. A review of the evidence for the use of phytoestrogens as a replacement for traditional estrogen replacement therapy. Arch. Intern. Med. 2001, 161, 1161–1172. [Google Scholar] [CrossRef] [Green Version]
- Messina, M.J. Legumes and soybeans: Overview of their nutritional profiles and health effects. Am. J. Clin. Nutr. 1999, 70, 439S–450S. [Google Scholar] [CrossRef] [Green Version]
- Allais, G.; Castagnoli Gabellari, I.; Burzio, C.; Rolando, S.; De Lorenzo, C.; Mana, O.; Benedetto, C. Premenstrual syndrome and migraine. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2012, 33 (Suppl. S1), 111. [Google Scholar] [CrossRef]
- Bryant, M.; Cassidy, A.; Hill, C.; Powell, J.; Talbot, D.; Dye, L. Effect of consumption of soy isoflavones on behavioural, somatic and affective symptoms in women with premenstrual syndrome. Br. J. Nutr. 2005, 93, 731–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, P.P.; Li, P.; Zhang, X.H.; Liu, N.; Wang, J.; Chen, D.D.; Sun, W.J.; Zhang, W. Complementary and alternative medicine for natural and treatment-induced vasomotor symptoms: An overview of systematic reviews and meta-analyses. Complementary Ther. Clin. Pract. 2019, 36, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, A.; Wegewitz, U.; Sommerfeld, C.; Grossklaus, R.; Lampen, A. Efficacy of isoflavones in relieving vasomotor menopausal symptoms-A systematic review. Mol. Nutr. Food Res. 2009, 53, 1084–1097. [Google Scholar] [CrossRef] [PubMed]
- Rapkin, A.J. Vasomotor symptoms in menopause: Physiologic condition and central nervous system approaches to treatment. Am. J. Obstet. Gynecol. 2007, 196, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Hirose, A.; Terauchi, M.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Low-dose isoflavone aglycone alleviates psychological symptoms of menopause in Japanese women: A randomized, double-blind, placebo-controlled study. Arch. Gynecol. Obstet. 2016, 293, 609–615. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-de-Las-Penas, C.; Fernandez-Munoz, J.J.; Palacios-Cena, M.; Paras-Bravo, P.; Cigaran-Mendez, M.; Navarro-Pardo, E. Sleep disturbances in tension-type headache and migraine. Ther. Adv. Neurol. Disord. 2017, 11. [Google Scholar] [CrossRef]
- Minen, M.T.; Begasse De Dhaem, O.; Kroon Van Diest, A.; Powers, S.; Schwedt, T.J.; Lipton, R.; Silbersweig, D. Migraine and its psychiatric comorbidities. J. Neurol. Neurosurg. Psychiatry 2016, 87, 741–749. [Google Scholar] [CrossRef]
- Messina, M. Soy and Health Update: Evaluation of the Clinical and Epidemiologic Literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef] [Green Version]
- Burger, H.G.; Dudley, E.C.; Hopper, J.L.; Groome, N.; Guthrie, J.R.; Green, A.; Dennerstein, L. Prospectively measured levels of serum follicle-stimulating hormone, estradiol, and the dimeric inhibins during the menopausal transition in a population-based cohort of women. J. Clin. Endocrinol. Metab. 1999, 84, 4025–4030. [Google Scholar] [CrossRef]
- MacGregor, E.A. Migraine headache in perimenopausal and menopausal women. Curr. Pain Headache Rep. 2009, 13, 399–403. [Google Scholar] [CrossRef]
- Chai, N.C.; Peterlin, B.L.; Calhoun, A.H. Migraine and estrogen. Curr. Opin. Neurol. 2014, 27, 315–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allais, G.; Chiarle, G.; Sinigaglia, S.; Airola, G.; Schiapparelli, P.; Benedetto, C. Estrogen, migraine, and vascular risk. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2018, 39, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Dahlman-Wright, K.; Gustafsson, J.A. Estrogen receptor alpha and beta in health and disease. Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.D.; Lampe, J.W.; Qu, C.; Gundersen, G.; Fuller, S.; Copeland, W.K.; Newton, K.M. Self-reported menopausal symptoms in a racially diverse population and soy food consumption. Maturitas 2013, 75, 152–158. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Headache | p-Value | |
|---|---|---|---|
| Frequent (N = 60) | Not So Frequent (N = 349) | ||
| Age and Menopausal Status | |||
| Age (years), mean (SD) | 49.4 (3.9) | 50.2 (3.8) | 0.128 a |
| Menopausal status, premenopausal/peri-menopausal/postmenopausal (%) | 37.3/27.1/35.6 | 32.4/23.1/44.5 | 0.441 b |
| Body composition, mean (SD) | |||
| Height (cm) | 157.4 (5.0) | 157.6 (5.4) | 0.839 c |
| Weight (kg) | 54.0 (10.8) | 54.4 (9.6) | 0.691 a |
| Body mass index (kg/m2) | 21.8 (4.1) | 21.9 (3.8) | 0.616 a |
| Waist–hip ratio | 0.87 (0.07) | 0.87 (0.06) | 0.762 c |
| Fat mass (kg) | 15.9 (8.2) | 15.8 (7.1) | 0.899 a |
| Lean mass (kg) | 38.1 (3.3) | 38.5 (3.5) | 0.355 c |
| Muscle mass (kg) | 35.9 (3.1) | 36.3 (3.2) | 0.355 c |
| Body fat percentage (%) | 27.9 (8.8) | 28.0 (7.7) | 0.927 c |
| Basal metabolism (MJ/day) | 1107 (125) | 1118 (122) | 0.491 a |
| Resting energy expenditure (MJ/day) | 1664 (400) | 1578 (428) | 0.171 a |
| Body temperature (℃) | 36.2 (0.5) | 36.3 (0.5) | 0.765 a |
| Physical fitness test, mean (SD) | |||
| Hand-grip strength (kg) | 25.2 (4.8) | 25.3 (4.8) | 0.834 c |
| Ruler-drop test (cm) | 22.9 (4.8) | 22.8 (4.0) | 0.895 a |
| Sit-and-reach test (cm) | 35.9 (10.0) | 36.1 (9.7) | 0.794 a |
| Cardiovascular parameters, mean (SD) | |||
| Systolic blood pressure (mmHg) | 128 (16) | 126.5 (17) | 0.433 a |
| Diastolic blood pressure (mmHg) | 82.0 (12.5) | 80.8 (12.2) | 0.350 a |
| Heart rate (per min) | 65.0 (11.3) | 63.7 (11.7) | 0.441 a |
| Cardio-ankle vascular index | 7.47 (1.12) | 7.42 (0.69) | 0.148 a |
| Ankle-brachial pressure index | 1.11 (0.08) | 1.10 (0.06) | 0.077 c |
| Lifestyle factors (%) | |||
| Smoking (cigarettes per day) | |||
| None/fewer than 20/20 or more | 88.3/8.3/3.3 | 89.7/7.5/2.9 | 0.953 b |
| Drinking | |||
| Never/sometimes/daily | 63.3/25.0/11.7 | 56.9/31.0/12.1 | 0.609 b |
| Caffeine (per day) | |||
| Never/1–2 times/3 or more times | 13.3/36.7/50.0 | 6.9/34.5/58.6 | 0.179 b |
| Exercise | |||
| Moderate exercise Yes/no | 51.7/48.3 | 43.0/57.0 | 0.260 d |
| Regular exercise Yes/no | 40.0/60.0 | 40.3/60.0 | >0.999 d |
| Physical symptoms, mean (SD) | |||
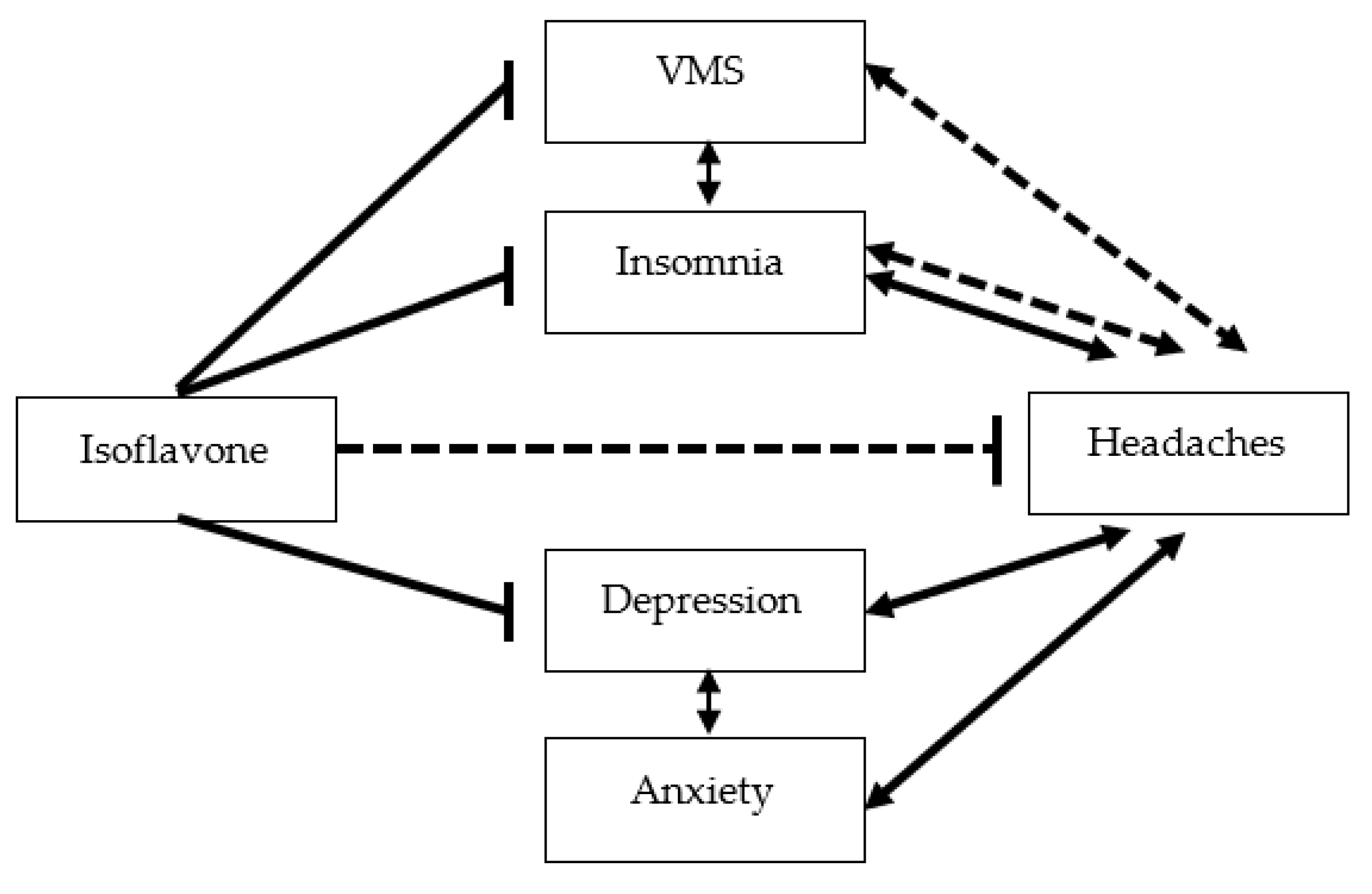
| MHR-QOL vasomotor symptom score (0–6) | 3.4 (2.3) | 2.3 (2.1) | 0.001 a |
| Psychological symptoms, mean (SD) | |||
| MHR-QOL insomnia symptom score (0–6) | 3.7 (2.2) | 2.3 (2.1) | <0.001 a |
| Hospital Anxiety and Depression scale Anxiety subscale score (0–21) | 9.2 (4.3) | 7.8 (3.7) | 0.029 a |
| Hospital Anxiety and Depression scale Depression subscale score (0–21) | 8.7 (4.4) | 7.3 (3.7) | 0.024 a |
| Characteristic | Headache | p-Value | |
|---|---|---|---|
| Frequent (N = 60) | Not So Frequent (N = 349) | ||
| Nutrition, Mean (SD) | |||
| Protein (%E) | 15.5 (3.3) | 15.8 (3.1) | 0.244 a |
| Animal protein (%E) | 8.8 (3.3) | 9.0 (3.1) | 0.585 a |
| Vegetable protein (%E) | 6.7 (1.3) | 6.8 (1.2) | 0.511 b |
| Carbohydrate (%E) | 53.5 (10.1) | 52.2 (8.6) | 0.235 a |
| Ash (g/1000 kcal/day) | 10.7 (2.5) | 10.6 (2.0) | 0.815 a |
| Sodium (mg/1000 kcal/day) | 2405 (551) | 2316 (478) | 0.173 a |
| Potassium (mg/1000 kcal/day) | 1525 (588) | 1594 (439) | 0.054 a |
| Calcium (mg/1000 kcal/day) | 325 (127) | 336 (112) | 0.142 a |
| Magnesium (mg/1000 kcal/day) | 147 (41) | 153 (35) | 0.072 a |
| Phosphorus (mg/1000 kcal/day) | 589 (133) | 608 (129) | 0.174 a |
| Iron (mg/1000 kcal/day) | 4.6 (1.5) | 4.7 (1.1) | 0.218 a |
| Zinc (mg/1000 kcal/day) | 4.53 (0.80) | 4.58 (0.74) | 0.432 a |
| Copper (mg/1000 kcal/day) | 0.65 (0.14) | 0.66 (0.12) | 0.344 a |
| Manganese (mg/1000 kcal/day) | 1.79 (0.64) | 1.78 (0.61) | 0.909 a |
| Fat (%E) | 27.2 (6.1) | 28.0 (5.8) | 0.330 b |
| Animal fat (%E) | 12.4 (4.5) | 12.8 (4.2) | 0.496 a |
| Vegetable fat (%E) | 14.8 (4.0) | 15.2 (4.0) | 0.500 b |
| Saturated fatty acid (%E) | 7.4 (2.1) | 7.7 (1.9) | 0.095 a |
| Monounsaturated fatty acid (%E) | 9.6 (2.4) | 9.9 (2.3) | 0.224 b |
| Polyunsaturated fatty acid (%E) | 6.6 (1.7) | 6.7 (1.6) | 0.350 a |
| Cholesterol (mg/1000 kcal/day) | 201 (74) | 208 (73) | 0.320 a |
| N-3 fatty acid (%E) | 1.3 (0.5) | 1.4 (0.4) | 0.295 a |
| N-6 fatty acid (%E) | 5.2 (1.4) | 5.3 (1.3) | 0.399 a |
| Soluble dietary fiber (g/1000 kcal/day) | 1.86 (0.81) | 1.99 (0.66) | 0.054 a |
| Insoluble dietary fiber (g/1000 kcal/day) | 5.18 (2.46) | 5.38 (1.65) | 0.064 a |
| Dietary fiber (g/1000 kcal/day) | 7.31 (3.56) | 7.59 (2.37) | 0.065 a |
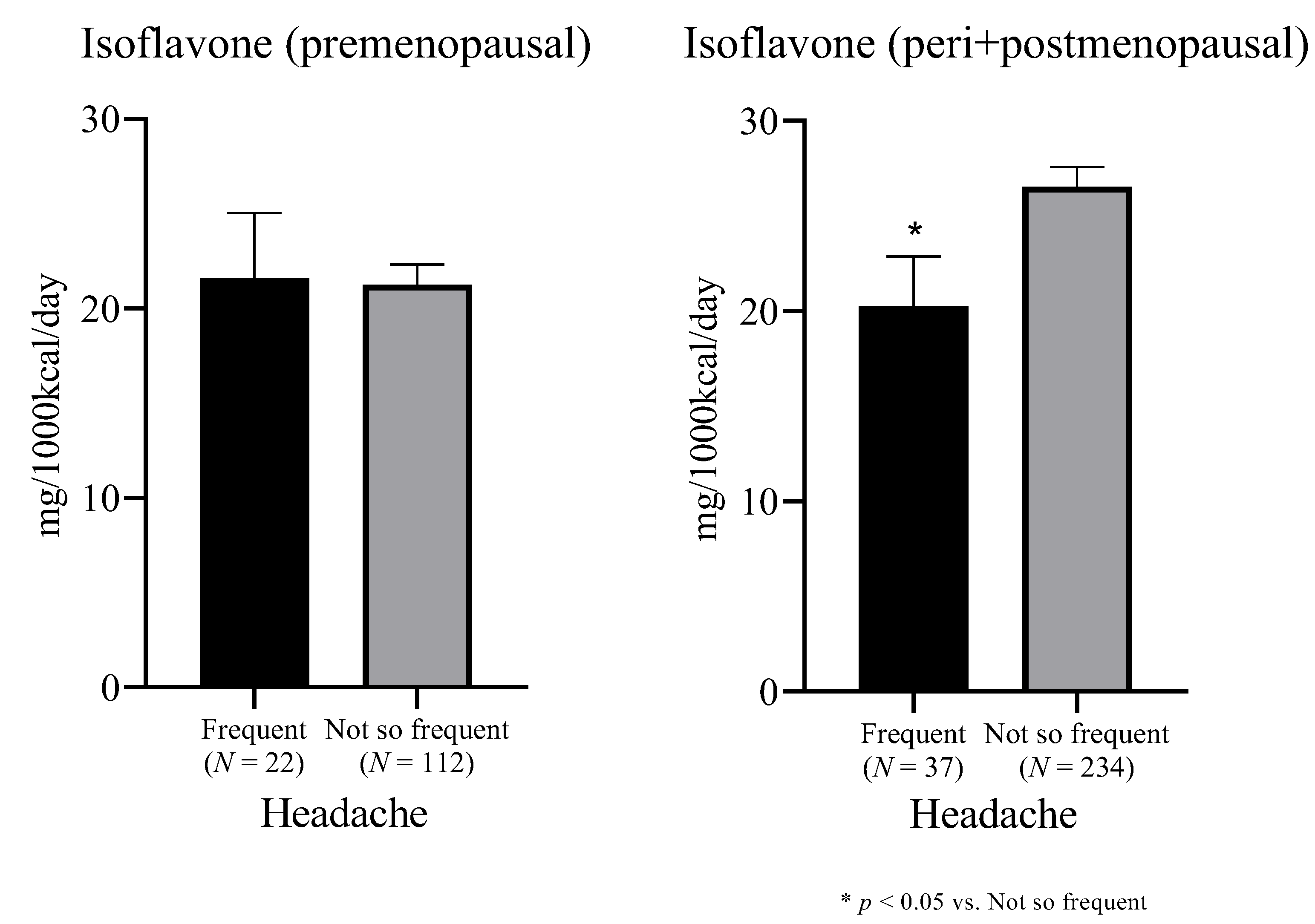
| Daidzein (mg/1000 kcal/day) | 7.7 (5.8) | 9.2 (5.4) | 0.009 a |
| Genistein (mg/1000 kcal/day) | 13.0 (9.9) | 15.6 (9.1) | 0.009 a |
| Isoflavone (mg/1000 kcal/day) | 20.7 (15.7) | 24.8 (14.5) | 0.009 a |
| Retinol (μg/1000 kcal/day) | 254 (171) | 223 (147) | 0.360 a |
| β-carotene (μg/1000 kcal/day) | 2495 (2492) | 2459 (1522) | 0.108 a |
| Retinol equivalent (μg/1000 kcal/day) | 463 (262) | 430 (202) | 0.779 a |
| Vitamin D (μg/1000 kcal/day) | 6.92 (4.87) | 7.29 (4.75) | 0.294 a |
| α-tocopherol (mg/1000 kcal/day) | 4.45 (1.60) | 4.42 (1.15) | 0.417 a |
| Vitamin K (μg/1000 kcal/day) | 198 (149) | 209 (101) | 0.044 a |
| Vitamin B1 (mg/1000 kcal/day) | 0.45 (0.13) | 0.46 (0.10) | 0.183 a |
| Vitamin B2 (mg/1000 kcal/day) | 0.77 (0.23) | 0.78 (0.20) | 0.256 a |
| Niacin (mgNE/1000 kcal/day) | 9.64 (3.06) | 9.85 (2.68) | 0.425 a |
| Vitamin B6 (mg/1000 kcal/day) | 0.72 (0.25) | 0.75 (0.20) | 0.068 a |
| Vitamin B12 (μg/1000 kcal/day) | 5.14 (3.06) | 5.06 (2.57) | 0.677 a |
| Folic acid (μg/1000 kcal/day) | 210 (104) | 210 (76) | 0.356 a |
| Pantothenic acid (mg/1000 kcal/day) | 3.67 (0.84) | 3.82 (0.77) | 0.081 b |
| Vitamin C (mg/1000 kcal/day) | 73.6 (41.7) | 73.8 (34.0) | 0.499 a |
| Nutrient | OR | 95% CI | p-Value | |
|---|---|---|---|---|
| Model 1 | Vitamin K (μg/1000 kcal/day) | 1.000 | 0.998–1.004 | 0.547 |
| Isoflavone (mg/1000 kcal/day) | 0.976 | 0.952–1.000 | 0.048 | |
| Model 2 | Vitamin K (μg/1000 kcal/day) | 1.000 | 0.998–1.004 | 0.625 |
| Isoflavone (mg/1000 kcal/day) | 0.974 | 0.950–0.999 | 0.036 | |
| MHR-QOL VMS score (0–6) | 1.160 | 1.012–1.329 | 0.033 | |
| MHR-QOL insomnia score (0–6) | 1.334 | 1.152–1.546 | <0.001 | |
| HADS depression subscale score (0–21) | 1.067 | 0.967–1.177 | 0.199 | |
| HADS anxiety subscale score (0–21) | 0.994 | 0.897–1.101 | 0.902 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazama, M.; Terauchi, M.; Odai, T.; Kato, K.; Miyasaka, N. The Inverse Correlation of Isoflavone Dietary Intake and Headache in Peri- and Post-Menopausal Women. Nutrients 2022, 14, 1226. https://doi.org/10.3390/nu14061226
Kazama M, Terauchi M, Odai T, Kato K, Miyasaka N. The Inverse Correlation of Isoflavone Dietary Intake and Headache in Peri- and Post-Menopausal Women. Nutrients. 2022; 14(6):1226. https://doi.org/10.3390/nu14061226
Chicago/Turabian StyleKazama, Mayuko, Masakazu Terauchi, Tamami Odai, Kiyoko Kato, and Naoyuki Miyasaka. 2022. "The Inverse Correlation of Isoflavone Dietary Intake and Headache in Peri- and Post-Menopausal Women" Nutrients 14, no. 6: 1226. https://doi.org/10.3390/nu14061226
APA StyleKazama, M., Terauchi, M., Odai, T., Kato, K., & Miyasaka, N. (2022). The Inverse Correlation of Isoflavone Dietary Intake and Headache in Peri- and Post-Menopausal Women. Nutrients, 14(6), 1226. https://doi.org/10.3390/nu14061226








