Nutrition Interventions for Children with Cerebral Palsy in Low- and Middle-Income Countries: A Scoping Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Database Searching
2.3. Inclusion and Exclusion Criteria
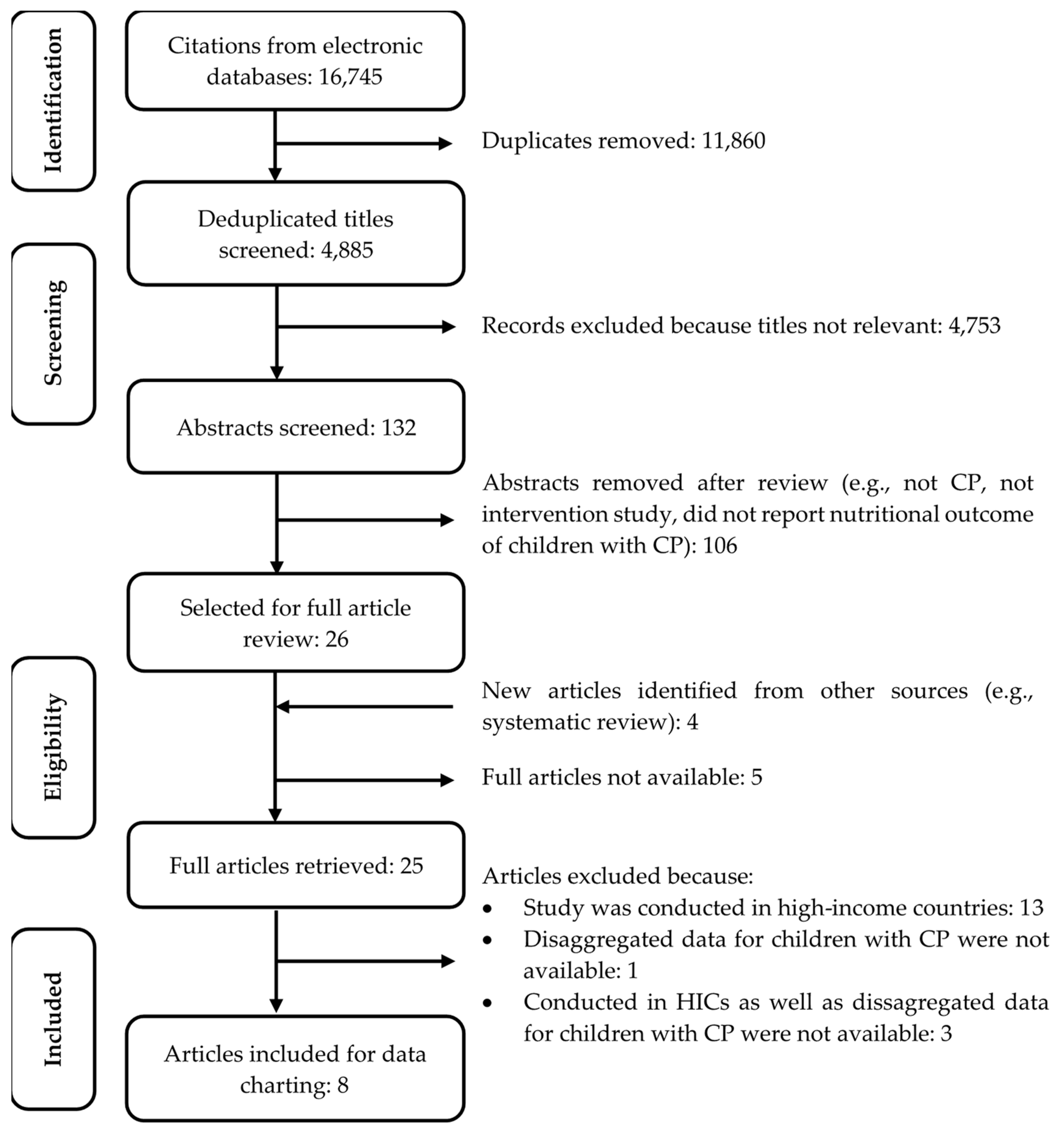
2.4. Data Charting Process
2.5. Assessment of Risk of Bias and Synthesis of Results
2.6. Ethics
3. Results
3.1. Study Characteristics
3.1.1. Study Design
| Study Details | Country | Settings | Study Design | Study Period | Study Participants | Characteristics of Participating Children with CP | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Name | Economic Classification | Number | Age (Range, Mean (SD), Median [IQR]) | Sex | GMFCS Level | |||||
| Donker et al., 2019 [27] | Ghana | Lower MIC | Community-based | Qualitative | July 2015–July 2016 | Children with CP; Primary caregivers of children with CP | 18 | Range: 1 y 6 m–11 y 11 m | F: 10, M: 8 | GMFCS I–II: 4; GMFCS III–V: 14 |
| Caselli et al., 2017 [25] | Brazil | Upper MIC | Hospital/Clinic/Centre-based | Descriptive analytical | Not reported | Children with spastic quadriplegia | 54 | Range: 2 y 1 m–18 y 7 m | F: 20, M: 34 | Not reported |
| Soylu et al., 2008 [23] | Turkey | Upper MIC | Hospital/Clinic/Centre-based | Experimental | May 2007–April 2008 | Children with quadriplegic CP | 45 | Mean ± SD: 5 y 6 m ± 3 y 7 m | F: 16, M: 29 | GMFCS I–II: 3; GMFCS III–V: 42 |
| Zuurmond et al., 2018 [24] | Ghana | Lower MIC | Community-based | Experimental | February 2015–July 2016 | Children with CP; Primary caregivers of children with CP | 64 | Range: 1 y 6 m–12 y 0 m Mean ± SD: 3 y 10 m ± 2 y 8 m | F: 26, M: 38 | GMFCS I–II: 15; GMFCS III: 17; GMFCS IV–V: 32 |
| Adams et al., 2011 [20] | Bangladesh | Lower MIC | Hospital/Clinic/Centre-based | Experimental | Not reported | Children with CP; Primary caregivers of children with CP | 22 | Range: 1 y 7 m–10 y 9 m; Mean ± SD:3 y 11 m ± 2 y 3 m | F: 14, M: 8 | GMFCS III–V: 22 |
| Pike T et al., 2016 [22] | South Africa | Upper MIC | Hospital/Clinic/Centre-based | Experimental | August 2007–January 2009 | Children with CP; Primary caregivers of children with CP | 16 | Range: 1 y 0 m–18 y 0 m | F: 6, M: 10 | GMFCS I–II: 1/16; GMFCS III–V: 15/16 |
| Gracia-Contreras et al., 2014 [26] | Mexico | Upper MIC | Hospital/Clinic/Centre-based | Descriptive analytical | 2010–2011 [month not reported] | Children with quadriplegic CP | 13 | Range: 6 y 11 m–12 y 10 m Mean: 9 y 7 m | F: 8, M: 5 | GMFCS V: 13 |
| Omar et al.,2017 [21] | Egypt | Lower MIC | Hospital/Clinic/Centre-based | Experimental | January 2017–April 2017 | Children with CP; Primary caregivers of children with CP | 20 | Median [IQR]: 2 y 6 m [1 y 1 m, 4 y 10 m] | F: 6, M: 14 | GMFCS III-1/20; GMFCS level IV: 8/20; GMFCS level V: 11/20 |
3.1.2. Study Location
3.1.3. Study Settings
3.1.4. Study Participants and Sample Size
3.2. Intervention Details
3.3. Outcome Measures
3.4. Effect of Different Interventions on Nutritional Status of Children with CP
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jahan, I.; Muhit, M.; Hardianto, D.; Laryea, F.; Amponsah, S.K.; Chhetri, A.B.; Smithers-Sheedy, H.; McIntyre, S.; Badawi, N.; Khandaker, G. Epidemiology of Malnutrition among Children with Cerebral Palsy in Low- and Middle-Income Countries: Findings from the Global LMIC CP Register. Nutrients 2021, 13, 3676. [Google Scholar] [CrossRef]
- Jahan, I.; Muhit, M.; Al Imam, M.H.; Ghose, R.; Chhetri, A.B.; Badawi, N.; Khandaker, G. Nutritional Status of Children with Cerebral Palsy in Gorkha, Nepal: Findings from the Nepal Cerebral Palsy Register. Nutrients 2021, 13, 2537. [Google Scholar] [CrossRef]
- Jahan, I.; Muhit, M.; Hardianto, D.; Karim, T.; Al Imam, M.H.; Das, M.C.; Smithers-Sheedy, H.; Badawi, N.; Khandaker, G. Nutritional status of children with cerebral palsy in remote Sumba Island of Indonesia: A community-based key informants study. Disabil. Rehabil. 2019, 43, 1819–1828. [Google Scholar] [CrossRef]
- Jahan, I.; Muhit, M.; Karim, T.; Smithers-Sheedy, H.; Novak, I.; Jones, C.; Badawi, N.; Khandaker, G. What makes children with cerebral palsy vulnerable to malnutrition? Findings from the Bangladesh cerebral palsy register (BCPR). Disabil. Rehabil. 2019, 41, 2247–2254. [Google Scholar] [CrossRef]
- Karim, T.; Jahan, I.; Dossetor, R.; Giang, N.T.H.; Van Anh, N.T.; Dung, T.Q.; Chau, C.M.; Van Bang, N.; Badawi, N.; Khandaker, G.; et al. Nutritional status of children with cerebral palsy—Findings from prospective hospital-based surveillance in Vietnam indicate a need for action. Nutrients 2019, 11, 2132. [Google Scholar] [CrossRef] [Green Version]
- Scarpato, E.; Staiano, A.; Molteni, M.; Terrone, G.; Mazzocchi, A.; Agostoni, C. Nutritional assessment and intervention in children with cerebral palsy: A practical approach. Int. J. Food Sci. Nutr. 2017, 68, 763–770. [Google Scholar] [CrossRef]
- Aggarwal, S.; Chadha, R.; Pathak, R. Nutritional status and growth in children with cerebral palsy: A review. Int. J. Med. Sci. Public Health 2015, 4, 737–744. [Google Scholar] [CrossRef] [Green Version]
- Kakooza-Mwesige, A.; Tumwine, J.K.; Eliasson, A.-C.; Namusoke, H.K.; Forssberg, H. Malnutrition is common in Ugandan children with cerebral palsy, particularly those over the age of five and those who had neonatal complications. Acta Paediatr. 2015, 104, 1259–1268. [Google Scholar] [CrossRef] [Green Version]
- Brunner, M.D.L.M.R.; Cieri, M.E.; Rodriguez Marco, M.P.; Schroeder, A.S.; Cuestas, E. Nutritional status of children with cerebral palsy attending rehabilitation centers. Dev. Med. Child Neurol. 2020, 62, 1383–1388. [Google Scholar] [CrossRef]
- Romano, C.; van Wynckel, M.; Hulst, J.; Broekaert, I.; Bronsky, J.; Dall’Oglio, L.; Mis, N.F.; Hojsak, I.; Orel, R.; Papadopoulou, A.; et al. European Society for Paediatric Gastroenterology, Hepatology and Nutrition guidelines for the evaluation and treatment of gastrointestinal and nutritional complications in children with neurological impairment. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 242–264. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization; World Bank. World Report on Disability 2011; World Health Organization: Geneva, Switzerland, 2011; Available online: https://apps.who.int/iris/handle/10665/44575 (accessed on 6 March 2022).
- Al Imam, M.H.; Jahan, I.; Muhit, M.; Hardianto, D.; Laryea, F.; Chhetri, A.B.; Smithers-Sheedy, H.; McIntyre, S.; Badawi, N.; Khandaker, G. Predictors of Rehabilitation Service Utilisation among Children with Cerebral Palsy (CP) in Low-and Middle-Income Countries (LMIC): Findings from the Global LMIC CP Register. Brain Sci. 2021, 11, 848. [Google Scholar] [CrossRef]
- Al Imam, M.H.; Jahan, I.; Das, M.C.; Muhit, M.; Akbar, D.; Badawi, N.; Khandaker, G. Situation analysis of rehabilitation services for persons with disabilities in Bangladesh: Identifying service gaps and scopes for improvement. Disabil. Rehabil. 2021, 1–14. [Google Scholar] [CrossRef]
- Leal-Martínez, F.; Franco, D.; Peña-Ruiz, A.; Castro-Silva, F.; Escudero-Espinosa, A.A.; Rolón-Lacarrier, O.G.; López-Alarcón, M.; De León, X.; Linares-Eslava, M.; Ibarra, A. Effect of a nutritional support system (diet and supplements) for improving gross motor function in cerebral palsy: An exploratory randomized controlled clinical trial. Foods 2020, 9, 1449. [Google Scholar] [CrossRef]
- Jahan, I.; Karim, T.; Das, M.C.; Muhit, M.; McIntyre, S.; Smithers-Sheedy, H.; Badawi, N.; Khandaker, G. Mortality in children with cerebral palsy in rural Bangladesh: A population-based surveillance study. Dev. Med. Child Neurol. 2019, 61, 1336–1343. [Google Scholar] [CrossRef]
- Power, R.; Muhit, M.; Heanoy, E.; Karim, T.; Badawi, N.; Akhter, R.; Khandaker, G. Health-related quality of life and mental health of adolescents with cerebral palsy in rural Bangladesh. PLoS ONE 2019, 14, e0217675. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.; Horsley, T.; Weeks, L. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Worldbank. World Bank Country and Lending Groups—World Bank Data Help Desk. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed on 6 March 2022).
- Rosenbaum, P.L.; Palisano, R.J.; Bartlett, D.J.; Galuppi, B.E.; Russell, D.J. Development of the gross motor function classification system for cerebral palsy. Dev. Med. Child Neurol. 2008, 50, 249–253. [Google Scholar] [CrossRef]
- Adams, M.S.; Khan, N.; Begum, S.; Wirz, S.; Hesketh, T.; Pring, T. Feeding difficulties in children with cerebral palsy: Low-cost caregiver training in Dhaka, Bangladesh. Child Care Health Dev. 2012, 38, 878–888. [Google Scholar] [CrossRef]
- Omar, O.M.; Khalil, M.; Abd El-Ghany, H.M.; Abdallah, A.M.; Omar, T.E. Intervention program to improve feeding, swallowing, and nutritional problems in children with cerebral palsy. Alex. J. Pediatr. 2017, 30, 85. [Google Scholar] [CrossRef]
- Pike, T. Feeding Intervention in Children with Cerebral Palsy over an 18-Month Period in Rural Eastern Cape; University of Pretoria: Pretoria, South Africa, 2016. [Google Scholar]
- Soylu, O.B.; Unalp, A.; Uran, N.; Dizdarer, G.; Ozgonul, F.O.; Conku, A.; Ataman, H.; Ozturk, A.A. Effect of nutritional support in children with spastic quadriplegia. Pediatr. Neurol. 2008, 39, 330–334. [Google Scholar] [CrossRef]
- Zuurmond, M.; O’Banion, D.; Gladstone, M.; Carsamar, S.; Kerac, M.; Baltussen, M.; Tann, C.J.; Gyamah Nyante, G.; Polack, S. Evaluating the impact of a community-based parent training programme for children with cerebral palsy in Ghana. PLoS ONE 2018, 13, e0202096. [Google Scholar] [CrossRef]
- Caselli, T.B.; Lomazi, E.A.; Montenegro, M.A.S.; Bellomo-Brandão, M.A. Comparative study on gastrostomy and orally nutrition of children and adolescents with tetraparesis cerebral palsy. Arq. Gastroenterol. 2017, 54, 292–296. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Contreras, A.A.; Vasquez-Garibay, E.M.; Romero-Velarde, E.; Ibarra-Gutierrez, A.I.; Troyo-Sanroman, R.; Sandoval-Montes, I.E. Intensive nutritional support improves the nutritional status and body composition in severely malnourished children with cerebral palsy. Nutr. Hosp. 2014, 29, 838–843. [Google Scholar] [CrossRef]
- Donkor, C.M.; Lee, J.; Lelijveld, N.; Adams, M.; Baltussen, M.M.; Nyante, G.G.; Kerac, M.; Polack, S.; Zuurmond, M. Improving nutritional status of children with Cerebral palsy: A qualitative study of caregiver experiences and community-based training in Ghana. Food Sci. Nutr. 2019, 7, 35–43. [Google Scholar] [CrossRef] [Green Version]
- de Romaña, D.L.; Greig, A.; Thompson, A.; Arabi, M. Successful delivery of nutrition programs and the sustainable development goals. Curr. Opin. Biotechnol. 2021, 70, 97–107. [Google Scholar] [CrossRef]
- Mollah, A.; Dio, A.; Haque, M.M.; Alim, M.; Rashida, S.A. Sustainable Development Goals and Disability: A Baseline of Bangladesh; The Disability Alliance on SDGs: Dhaka, Bangladesh, 2019. [Google Scholar]
- Khalid, H.; Gill, S.; Fox, A.M. Global aid for nutrition-specific and nutrition-sensitive interventions and proportion of stunted children across low- and middle-income countries: Does aid matter? Health Policy Plan. 2019, 34, ii18–ii27. [Google Scholar] [CrossRef]
- Micha, R.; Mannar, V.; Afshin, A.; Allemandi, L.; Baker, P.; Battersby, J.; Bhutta, Z.; Chen, K.; Corvalan, C.; Di Cesare, M.; et al. 2020 Global Nutrition Report: Action on Equity to End Malnutrition; Development Initiatives: Bristol, UK, 2020. [Google Scholar]
- Al Imam, M.H.; Jahan, I.; Das, M.C.; Muhit, M.; Smithers-Sheedy, H.; McIntyre, S.; Badawi, N.; Khandaker, G. Rehabilitation status of children with cerebral palsy in Bangladesh: Findings from the Bangladesh Cerebral Palsy Register. PLoS ONE 2021, 16, e0250640. [Google Scholar] [CrossRef]
- Karim, T.; Muhit, M.; Jahan, I.; Galea, C.; Morgan, C.; Smithers-Sheedy, H.; Badawi, N.; Khandaker, G. Outcome of Community-Based Early Intervention and Rehabilitation for Children with Cerebral Palsy in Rural Bangladesh: A Quasi-Experimental Study. Brain Sci. 2021, 11, 1189. [Google Scholar] [CrossRef]
- Khandaker, G.; Muhit, M.; Karim, T.; Smithers-Sheedy, H.; Novak, I.; Jones, C.; Badawi, N. Epidemiology of cerebral palsy in Bangladesh: A population-based surveillance study. Dev. Med. Child Neurol. 2019, 61, 601–609. [Google Scholar] [CrossRef] [Green Version]
- Benfer, K.A.; Weir, K.A.; Bell, K.L.; Ware, R.S.; Davies, P.S.; Boyd, R.N. Oropharyngeal dysphagia and gross motor skills in children with cerebral palsy. Pediatrics 2013, 131, e1553–e1562. [Google Scholar] [CrossRef]
- Snider, L.; Majnemer, A.; Darsaklis, V. Feeding interventions for children with cerebral palsy: A review of the evidence. Phys. Occup. Ther. Pediatr. 2011, 31, 58–77. [Google Scholar] [CrossRef]
- Ferluga, E.D.; Archer, K.R.; Sathe, N.A.; Krishnaswami, S.; Klint, A.; Lindegren, M.L.; McPheeters, M.L. Interventions for Feeding and Nutrition in Cerebral Palsy; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2013. [Google Scholar]
- Jadi, J.; Hyder, S.; Rodriguez Ormaza, N.P.; Twer, E.; Phillips, M.; Akinkuotu, A.; Reid, T.D. Evaluation of Complications and Weight Outcomes in Pediatric Cerebral Palsy Patients with Gastrostomy Tubes. Am. Surg. 2021, 00031348211034753. [Google Scholar] [CrossRef] [PubMed]
- Haberfellner, H.; Schwartz, S.; Gisel, E.G. Feeding skills and growth after one year of intraoral appliance therapy in moderately dysphagic children with cerebral palsy. Dysphagia 2001, 16, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Vernon-Roberts, A.; Wells, J.; Grant, H.; Alder, N.; Vadamalayan, B.; Eltumi, M.; Sullivan, P.B. Gastrostomy feeding in cerebral palsy: Enough and no more. Dev. Med. Child Neurol. 2010, 52, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.B.; Juszczak, E.; Bachlet, A.M.; Lambert, B.; Vernon-Roberts, A.; Grant, H.W.; Eltumi, M.; McLean, L.; Alder, N.; Thomas, A.G. Gastrostomy tube feeding in children with cerebral palsy: A prospective, longitudinal study. Dev. Med. Child Neurol. 2005, 47, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Henderson, R.C.; Grossberg, R.I.; Matuszewski, J.; Menon, N.; Johnson, J.; Kecskemethy, H.H.; Vogel, L.; Ravas, R.; Wyatt, M.; Bachrach, S.J. Growth and nutritional status in residential center versus home-living children and adolescents with quadriplegic cerebral palsy. J. Pediatrics 2007, 151, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Campanozzi, A.; Capano, G.; Miele, E.; Romano, A.; Scuccimarra, G.; Del Giudice, E.; Strisciuglio, C.; Militerni, R.; Staiano, A. Impact of malnutrition on gastrointestinal disorders and gross motor abilities in children with cerebral palsy. Brain Dev. 2007, 29, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Kernizan, D.; Mintz, D.; Colin, M.; Lee, M.; Yoakam, L.; Chen, Y.P.; Iofel, E.; Koniaris, S.; Weidner, M. Outcomes and Safety of Blenderized Tube Feedings in Pediatric Patients: A Single Center's Experience. J. Pediatric Gastroenterol. Nutr. 2020, 71, e124–e128. [Google Scholar] [CrossRef] [PubMed]
- Dipasquale, V.; Catena, M.A.; Cardile, S.; Romano, C. Standard polymeric formula tube feeding in neurologically impaired children: A five-year retrospective study. Nutrients 2018, 10, 684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clawson, E.; Kuchinski, K.; Bach, R. Use of behavioral interventions and parent education to address feeding difficulties in young children with spastic diplegic cerebral palsy. NeuroRehabilitation 2007, 22, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Corwin, D.; Isaacs, J.; Georgeson, K.; Bartolucci, A.A.; Cloud, H.H.; Craig, C.D. Weight and length increases in children after gastrostomy placement. J. Am. Diet. Assoc. 1996, 96, 874–879. [Google Scholar] [CrossRef]
- Kong, C.-K.; Wong, H.-S.S. Weight-for-height values and limb anthropometric composition of tube-fed children with quadriplegic cerebral palsy. Pediatrics 2005, 116, e839–e845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rempel, G.R.; Colwell, S.O.; Nelson, R.P. Growth in children with cerebral palsy fed via gastrostomy. Pediatrics 1988, 82, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Arrowsmith, F.; Allen, J.; Gaskin, K.; Somerville, H.; Clarke, S.; O’loughlin, E. The effect of gastrostomy tube feeding on body protein and bone mineralization in children with quadriplegic cerebral palsy. Dev. Med. Child Neurol. 2010, 52, 1043–1047. [Google Scholar] [CrossRef] [PubMed]
- Mahant, S.; Friedman, J.N.; Connolly, B.; Goia, C.; Macarthur, C. Tube feeding and quality of life in children with severe neurological impairment. Arch. Dis. Child. 2009, 94, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Pinnington, L.; Hegarty, J. Effects of consistent food presentation on efficiency of eating and nutritive value of food consumed by children with severe neurological impairment. Dysphagia 1999, 14, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Gisel, E.G.; Tessier, M.-J.; Lapierre, G.; Seidman, E.; Drouin, E.; Filion, G. Feeding management of children with severe cerebral palsy and eating impairment: An exploratory study. Phys. Occup. Ther. Pediatrics 2003, 23, 19–44. [Google Scholar] [CrossRef]
- Brant, C.Q.; Stanich, P.; Ferrari, A.P., Jr. Improvement of children’s nutritional status after enteral feeding by PEG: An interim report. Gastrointest. Endosc. 1999, 50, 183–188. [Google Scholar] [CrossRef]
| Study Details | Intervention Provided | ||||
|---|---|---|---|---|---|
| Type | Settings | Contents | Number of Sessions | F/U Period | |
| Donker et al., 2019 [27] | Training to parents/caregivers | Community | Group support sessions followed the “Getting to Know Cerebral Palsy” (GTKCP) manual comprised of 11 modules including a module on “Eating and Drinking” | Not reported | 12 months |
| Caselli et al., 2017 [25] | Gastrostomy tube placement/feeding/nasogastric tube feeding | Hospital/clinic/centre | A comparison between children fed by gastrostomy (GTF) at least for 6 months versus children fed orally (PO) | Not applicable | Not applicable |
| Soylu et al., 2008 [23] | Nutritional rehabilitation/therapy | Hospital/clinic/centre | Dietary modification | Not reported | 6 months |
| Zuurmond et al., 2018 [24] | Training to parents/caregivers | Community | Evaluation of the child, positioning and carrying, communication, everyday activities, feeding child, play, disability in the local community, running an own parent support group, assistive devices and resources | Monthly 3 h sessions, total 11 sessions for each participant | 11 months |
| Adams et al., 2012 [20] | Training to parents/caregivers | Hospital/clinic/centre | (i) Dietary modification (calorie density, balanced, frequency, portion size), (ii) Feeding skills (posture/positioning, responsive, self-feeding) | 6 fortnightly training sessions each pair | 4–6 months |
| Pike T et al., 2016 [22] | Neurodevelopmental therapy (NDT) feeding intervention | Hospital/clinic/centre | NDT: speech-language therapy, physiotherapy, occupational therapy, assistive device | 90 min sessions for 5 consecutive days | 18 months |
| Training to parents/caregivers | Caregiver training on the recommended therapies as part of NDT | ||||
| Dietary modification | |||||
| Gracia-Contreras et al., 2014 [26] | Gastrostomy tube placement/feeding/nasogastric tube feeding | Hospital/clinic/centre | Not applicable | Not applicable | 4 weeks |
| Dietary modification | Calorie and nutrient (e.g., protein adjustments) | ||||
| Omar et al., 2017 [21] | Training to parents/caregivers | Hospital/clinic/centre | (i) Feeding skills (position, mode of feeding, utensils, associated feeding problems), (ii) responsive feeding, (iii) food consistency and adequacy. | 10 sessions over 5 days, 3 h each day | 3 months |
| Study Details | Nutritional Outcome Measures | Other Outcome Measures | Adverse Outcome Reported | Change in Nutritional Indicators | Nutritional Status Improved | ||
|---|---|---|---|---|---|---|---|
| Anthropometric Measurements | Indicator | Pre-Intervention/Intervention Group | Post Intervention/Control Group | ||||
| Donker et al., 2019 [27] | Weight | N/A | (i) Changes in positioning, utensils, and feeding skills, (ii) Dietary intake | Not reported | N/A | N/A | No 1 |
| Length/Height | HAZ | Mean: −2.13 | Mean: −2.62 | ||||
| Stunting | 50% | 50% | |||||
| Weight/Height | WHZ | Mean: −2.47 | Mean: −2.24 | ||||
| Wasting | 50% | 50% | |||||
| MUAC | Mean | Mean: 151 mm | Mean: 149 mm | ||||
| Caselli et al., 2017 [25] | Weight | WA (percentiles) | (i) Dietary intake | Not reported | GTF: <25th 20.0%; 25–90th 68.0%; >90th 12.0% | PO: <25th 24.1%; 25–90th 72.4%, 90th 3.4% | N/A 2 |
| Length/Height, KH | HA (percentiles) | GTF: 10–90th 10.0% | PO: 10–90th 100.0% | ||||
| Weight/Height | BA (percentiles) | GTF: <5th 20.0%; 10–90th 72.0%; >90th 8.0% | PO: <5th 24.1%; 10–90th 72.4%; >90th 3.4% | ||||
| AC | Percentile | GTF: <10th 40%; 10–90th and >95th 60% | PO: <10th 75.8%; 10–90th and >95th 24.1% | ||||
| TSF | Percentile | GTF: <10th 12%; 10–90th 64%; >95th 24% | PO: <10th 62.7%; 10–90th 37.9%; >95th 0% | ||||
| AMC | Percentile | GTF: <10th 52%; 10–90th and >95th 48% | PO: <10th 68.9%; 10–90th and >95th 31% | ||||
| AMA | Percentile | GTF: <10th 60%; 10–90th and >95th 40% | PO: <10th 72.4%; 10–90th and >95th 27.5% | ||||
| AAA | Percentile | GTF: <5th 16%; 10–90th and >95th 84% | PO: <5th 55.1%; 10–90th and >95th 83% | ||||
| Soylu et al., 2008 [23] | Weight | WAZ | None | Number of infections | Mean ± SD: −2.1 ± 0.9 | Mean ± SD: −1.8 ± 0.9 | Yes 3 |
| Length/Height | HAZ | Mean ± SD: −2.4 ± 2.1 | Mean ± SD: −2.3 ± 2.0 | ||||
| Weight/Height | BMI | Mean ± SD: 13.6 ± 2.1 kg/m2 | Mean ± SD: 14.4 ± 2.0 kg/m2 | ||||
| WH (NCHS percentiles) | <10th 18/31; 10–50th 9/31;50–90th 0/31; >90th 4/31 | <10th 14/31; 10–50th 9/31;50–90th 5/31; >90th 3/31 | |||||
| MUAC | Mean, SD | Mean ± SD: 14.5 ± 2.2 cm | Mean ± SD: 15.2 ± 2.2 cm | ||||
| TSF | Mean, SD | Mean ± SD: 9.8 ± 3.5 mm | Mean ± SD: 10.4 ± 4.1 mm | ||||
| Not applicable | Not applicable | They also compared the findings with CP growth chart. Significant improvement after therapies was observed for weight, height, MUAC, TSF, WAZ, WHZ, BMI | |||||
| Zuurmond et al., 2018 [24] | Weight | WAZ; Underweight | (i) Caregiver’s quality of life, (ii) Caregiver’s knowledge, confidence about providing care to their child, (iii) Caregiver’s perception about child’s physical and emotional health | (i) Serious health problem/illness in preceding 6 months, (ii) Mortality | Mean (95% CI): −2.6 (−2.9, −2.2);Underweight: 24.5%, Severely underweight: 38.5% | Mean (95% CI): −2.8 (−3.1, −2.3);Underweight: 17.2%, Severely underweight: 48.2% | No 4 |
| Length/Height | HAZ; Stunting | Mean (95% CI): −2.3 (−2.6, −1.9);Stunted: 26.9%, Severely stunted: 26.9% | Mean (95% CI): −2.7 (−3.0, −2.4);Stunted: 25.0%, Severely stunted: 39.1% | ||||
| Weight/height | WHZ; wasting | Mean (95% CI): −2.1 (−2.5, −1.6);Wasted: 40.0%, severely wasted: 20.0% | Mean (95% CI): −1.9 (−2.5, −1.3);Wasted: 12.9%, severely wasted: 32.3% | ||||
| MUAC | Mean, 95% CI; Wasting | 144.8 (139.3, 150.4)Wasted: 15.2%, Severely wasted: 0% | 144.4 (137.9, 150.8); Wasted: 13.0%, Severely wasted: 8.7% | ||||
| Adams et al., 2011 [20] | Weight | WAZ | (i) Dietary intake, (ii) Child’s feeding skills and mood, (iii) Caregivers’ feelings about child’s feeding difficulties, (iv) Caregiver’s compliance with training recommendations, (v) Child and caregiver’s behavior during mealtime | (i) Chest health, (ii) Mortality | Mean ± SD: −4.83 ± 1.84 | Mean ± SD: −4.07 ± 2.45 | Yes 5 |
| MUAC | Mean, SD | Mean ± SD: 14.75 ± 1.41 cm | Mean ± SD: 15.46 ± 1.57) | ||||
| Pike T et al., 2016 [22] | Weight | Mean, SD, median | (i) Child’s health related quality of life, (ii) Feeding profile | Not reported | Mean: 16.0 (4.0) kg; Median: 15.5 kg | Mean: 18.3 (5.1) kg; Median: 17.8 kg | Yes 6 |
| Length/Height | Mean: 101.9 (15.2) cm; Median: 100.6 cm | Mean: 106.7 (13.5) cm; Median: 105.1 cm | |||||
| MUAC | Mean: 17.5 (1.6) cm; Median: 17.8 cm | Mean: 18.3 (2.1) cm; Median: 18.1 cm | |||||
| TSF | Mean: 7.7 (3.0) mm; Median: 7.3 mm | Mean: 8.6 (3.7) mm; Median: 7.8 mm | |||||
| Gracia-Contreras et al., 2014 [26] | Weight | Mean, SD | (i) Bioelectrical Impedance analysis-fat mass and fat free mass, (ii) Feeding type and energy intake | Not reported | Mean ± SD: 11.9 ± 2.3 kg | Mean ± SD: 14.6 ± 2.6 kg | Yes 7 |
| Weight/ Height | BMI | Mean ± SD: 10.0 ± 1.0 kg/m2 | Mean ± SD: 12 ± 0.9 kg/m2 | ||||
| BAZ | Mean ± SD: −2.8 ± 0.5 | Mean ± SD: −1.9 ± 0.3 | |||||
| MUAC | MUACZ | Mean ± SD: −3.5 ± 0.3 | Mean ± SD: −2.8 ± 0.4 | ||||
| TSF | Mean, SD | Mean ± SD: 4.0 ± 1.8 mm | Mean ± SD: 7.4 ± 2.7 mm | ||||
| TSFZ | Mean ± SD: 7.4 ± 2.7 | Mean ± SD: −0.9 ± 0.4 | |||||
| SSF | Mean, SD | Mean ± SD: 3.7 ± 0.5 mm | Mean ± SD: 6.4 ± 1.9 mm | ||||
| SSFZ | Mean ± SD: −0.8 ± 0.1 | Mean ± SD: −0.3 ± 0.5 | |||||
| THSF | Mean, SD | Mean ± SD: 7.3 ± 5.3 mm | Mean ± SD: 13.5 ± 7.7 mm | ||||
| CSF | Mean, SD | Mean ± SD: 5.8 ± 3.4 mm | Mean ± SD: 9.4 ± 4.9 mm | ||||
| AMA | Mean, SD | Mean ± SD: 8.0 ± 1.2 cm2 | Mean ± SD: 9.4 ± 1.4 cm2 | ||||
| AFA | Mean, SD | Mean ± SD: 2.2 ± 1.1 cm2 | Mean ± SD: 4.5 ± 1.8 cm2 | ||||
| Omar et al., 2017 [21] | Weight | Mean, SD | (i) Feeding problem, (ii) feeding practices | Not reported | Mean ± SD: 10.1 ± 2.4 kg | Mean ± SD: 10 ± 2.5 kg | No change 8 |
| WAZ | Mean ± SD: −3.36 ± 1.26 | Mean ± SD: −3.36 ± 1.26 | |||||
| Length/Height | Mean, SD | Mean ± SD: 84.80 ± 10.26 cm | Mean ± SD: 84.80 ± 10.26 cm | ||||
| BMI | Mean ± SD: 14.77 ± 2.08 kg/m2 | Mean ± SD: 14.77 ± 2.08 kg/m2 | |||||
| MUAC | Mean, SD | Mean ± SD: 14.75 ± 1.81 cm | Mean ± SD: 14.75 ± 1.81 cm | ||||
| TL | Mean, SD | Mean ± SD: 16.8 ± 3.03 cm | Mean ± SD: 16.8 ± 3.03 cm | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jahan, I.; Sultana, R.; Muhit, M.; Akbar, D.; Karim, T.; Al Imam, M.H.; Das, M.C.; Smithers-Sheedy, H.; McIntyre, S.; Badawi, N.; et al. Nutrition Interventions for Children with Cerebral Palsy in Low- and Middle-Income Countries: A Scoping Review. Nutrients 2022, 14, 1211. https://doi.org/10.3390/nu14061211
Jahan I, Sultana R, Muhit M, Akbar D, Karim T, Al Imam MH, Das MC, Smithers-Sheedy H, McIntyre S, Badawi N, et al. Nutrition Interventions for Children with Cerebral Palsy in Low- and Middle-Income Countries: A Scoping Review. Nutrients. 2022; 14(6):1211. https://doi.org/10.3390/nu14061211
Chicago/Turabian StyleJahan, Israt, Risad Sultana, Mohammad Muhit, Delwar Akbar, Tasneem Karim, Mahmudul Hassan Al Imam, Manik Chandra Das, Hayley Smithers-Sheedy, Sarah McIntyre, Nadia Badawi, and et al. 2022. "Nutrition Interventions for Children with Cerebral Palsy in Low- and Middle-Income Countries: A Scoping Review" Nutrients 14, no. 6: 1211. https://doi.org/10.3390/nu14061211
APA StyleJahan, I., Sultana, R., Muhit, M., Akbar, D., Karim, T., Al Imam, M. H., Das, M. C., Smithers-Sheedy, H., McIntyre, S., Badawi, N., & Khandaker, G. (2022). Nutrition Interventions for Children with Cerebral Palsy in Low- and Middle-Income Countries: A Scoping Review. Nutrients, 14(6), 1211. https://doi.org/10.3390/nu14061211







