Diet and Maternal Obesity Are Associated with Increased Oxidative Stress in Newborns: A Cross-Sectional Study
Abstract
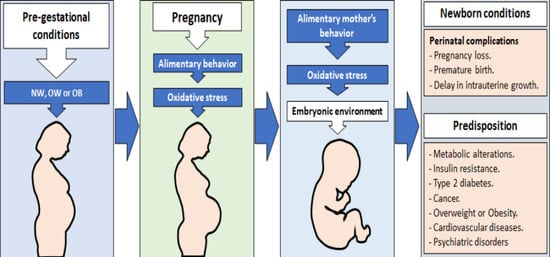
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Food Frequency Questionnaire
2.3. Plasma Samples
2.4. Nitric Oxide (NO)
2.5. Malondialdehyde (MDA)
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics of Mothers and Newborns
3.2. Micronutrients and Macronutrients Consumption
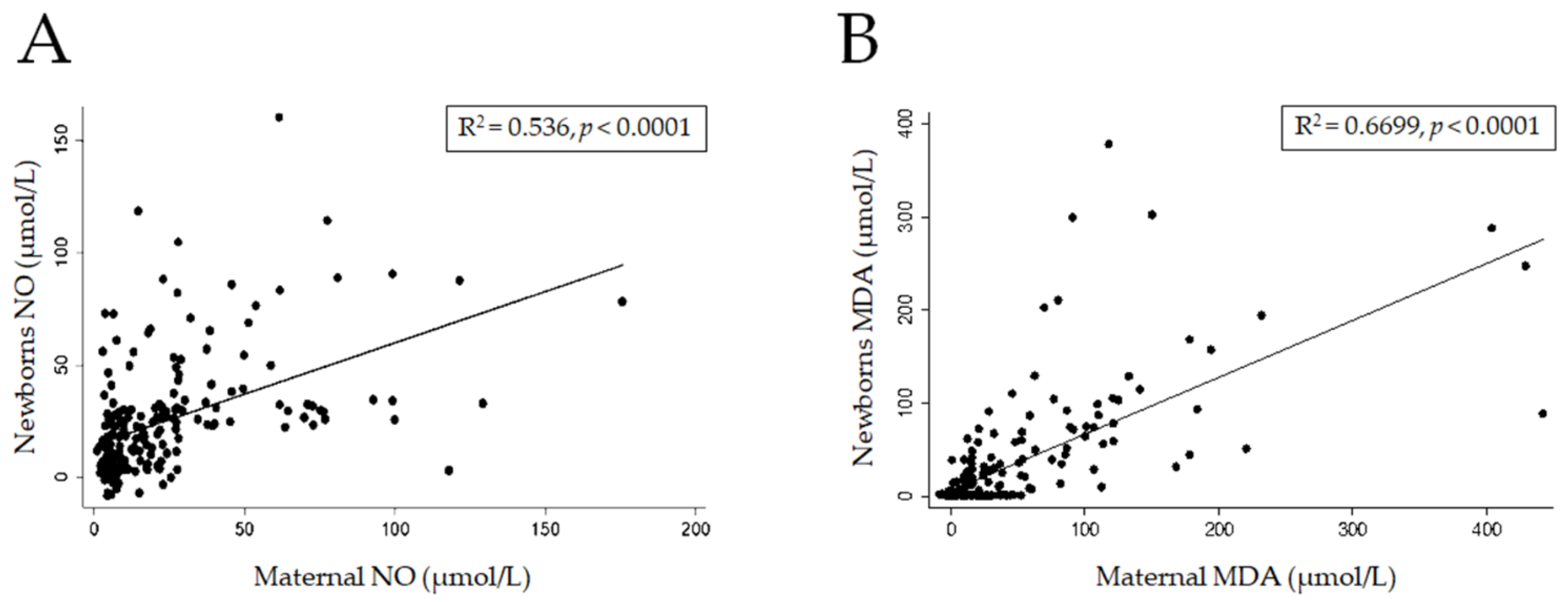
3.3. Levels of Oxidative Stress (MDA and NO)
3.4. Fruit, Vegetables and Vitamins Consumption
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haffner, S.M. Relationship of Metabolic Risk Factors and Development of Cardiovascular Disease and Diabetes. Obesity 2006, 14 (Suppl. 3), 121S–127S. [Google Scholar] [CrossRef] [PubMed]
- Srivaratharajah, K.; Abramson, B.L.; Jairam, J.; Park, A.L.; Berger, H.; Ray, J.G. Ethnic Differences in Visceral Adiposity Measured in Early Pregnancy. J. Obstet. Gynaecol. Can. 2018, 40, 193–198. [Google Scholar] [CrossRef]
- Gutiérrez, J.P.; Rivera-Dommarco, J.; Shamah-Levy, T.; Villalpando-Hernández, S.; Franco, A.; Cuevas-Nasu, L.; Romero-Martínez, M.; Hernández-Ávila, M. Encuesta Nacional de Salud y Nutrición 2012. Result. Nac. 2012, 1, 180–186. [Google Scholar]
- Salsberry, P.J.; Reagan, P.B. Taking the long view: The prenatal environment and early adolescent overweight. Res. Nurs. Health 2007, 30, 297–307. [Google Scholar] [CrossRef]
- Cedergren, M.I. Maternal Morbid Obesity and the Risk of Adverse Pregnancy Outcome. Obstet. Gynecol. 2004, 103, 219–224. [Google Scholar] [CrossRef]
- Gallardo, J.M.; Gómez-López, J.; Medina-Bravo, P.; Juárez-Sánchez, F.; Contreras-Ramos, A.; Galicia-Esquivel, M.; Sánchez-Urbina, R.; Klünder-Klünder, M. Maternal obesity increases oxidative stress in the newborn. Obesity 2015, 23, 1650–1654. [Google Scholar] [CrossRef]
- Carmichael, S.L.; Rasmussen, S.A.; Shaw, G.M. Prepregnancy obesity: A complex risk factor for selected birth defects. Birth Defects Res. Part A Clin. Mol. Teratol. 2010, 88, 804–810. [Google Scholar] [CrossRef]
- Olusi, S. Obesity is an independent risk factor for plasma lipid peroxidation and depletion of erythrocyte cytoprotectic enzymes in humans. Int. J. Obes. 2002, 26, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Elizalde, M.; Rydén, M.; Van Harmelen, V.; Eneroth, P.; Gyllenhammar, H.; Holm, C.; Ramel, S.; Olund, A.; Arner, P.; Andersson, K. Expression of nitric oxide synthases in subcutaneous adipose tissue of nonobese and obese humans. J. Lipid Res. 2000, 41, 1244–1251. [Google Scholar] [CrossRef]
- Łuczaj, W.; Skrzydlewska, E. DNA damage caused by lipid peroxidation products. Cell. Mol. Biol. Lett. 2003, 8, 391–413. [Google Scholar] [PubMed]
- Meuwese, M.C.; Stroes, E.S.; Hazen, S.L.; van Miert, J.N.; Kuivenhoven, J.A.; Schaub, R.G.; Wareham, N.J.; Luben, R.; Kastelein, J.J.; Khaw, K.-T.; et al. Serum Myeloperoxidase Levels Are Associated With the Future Risk of Coronary Artery Disease in Apparently Healthy Individuals: The EPIC-Norfolk Prospective Population Study. J. Am. Coll. Cardiol. 2007, 50, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Dennery, P.A. Effects of oxidative stress on embryonic development. Birth Defects Res. C Embryo Today 2007, 81, 155–162. [Google Scholar] [CrossRef]
- Loeken, M. Free radicals and birth defects. J. Matern. Neonatal Med. 2004, 15, 6–14. [Google Scholar] [CrossRef]
- Carmichael, S.L.; Yang, W.; Gilboa, S.; Ailes, E.; Correa, A.; Botto, L.D.; Feldkamp, M.L.; Shaw, G.M.; The National Birth Defects Prevention Study. Elevated body mass index and decreased diet quality among women and risk of birth defects in their offspring. Birth Defects Res. Part A Clin. Mol. Teratol. 2016, 106, 164–171. [Google Scholar] [CrossRef]
- Shen, C.-L.; Ramamoorthy, S.; Kaur, G.; Dufour, J.; Wang, R.; Mo, H.; Watkins, B. Dietary Annatto-Extracted Tocotrienol Reduces Inflammation and Oxidative Stress, and Improves Macronutrient Metabolism in Obese Mice: A Metabolic Profiling Study. Nutrients 2021, 13, 1267. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, F.; Catapano, A.; Trinchese, G.; Cavaliere, G.; Culurciello, R.; Fogliano, C.; Penna, E.; Lucci, V.; Crispino, M.; Avallone, B.; et al. Dietary Micronutrient Management to Treat Mitochondrial Dysfunction in Diet-Induced Obese Mice. Int. J. Mol. Sci. 2021, 22, 2862. [Google Scholar] [CrossRef] [PubMed]
- Berry, A.; Bellisario, V.; Panetta, P.; Raggi, C.; Magnifico, M.C.; Arese, M.; Cirulli, F. Administration of the Antioxidant N-Acetyl-Cysteine in Pregnant Mice Has Long-Term Positive Effects on Metabolic and Behavioral Endpoints of Male and Female Offspring Prenatally Exposed to a High-Fat Diet. Front. Behav. Neurosci. 2018, 12, 48. [Google Scholar] [CrossRef]
- Ghosh, S.; Sinha, J.K.; Putcha, U.K.; Raghunath, M. Severe but Not Moderate Vitamin B12 Deficiency Impairs Lipid Profile, Induces Adiposity, and Leads to Adverse Gestational Outcome in Female C57BL/6 Mice. Front. Nutr. 2016, 3, 1. [Google Scholar] [CrossRef]
- NORMA Oficial Mexicana NOM-034-SSA2-2013. Para la Prevención y Control de los Defectos al Nacimiento; Diario Oficial de la Federación, Secretaria de Gobernación: Mexico City, Mexico, 2014. [Google Scholar]
- Galván-Portillo, M.; Torres-Sánchez, L.; Hernández-Ramírez, R.U.; Anaya-Loyola, M.A. Validity and reproducibility of a food frequency questionnaire to estimate folate intake in a Mexican population. Salud Publica Mex. 2011, 53, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Perez-Lizaur, A.B.; Palacios-Gonzalez, B.; Castro-Becerra, A.L. Sistema Mexicano de Alimentos Equivalentes, 3rd ed.; Fomento de Nutrición y Salud: Mexico City, Mexico, 2008; p. 108. [Google Scholar]
- Institute of Medicine. A report of the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, other B Vitamins, and Choline, and Subcommittee on Upper Reference Levels of Nutrients. In Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B(6), Folate, Vitamin B(12), Pantothenic Acid, Biotin, and Choline; National Academies Press (US): Washington, DC, USA, 1998. Available online: https://www.ncbi.nlm.nih.gov/books/NBK114310/ (accessed on 15 December 2021).
- Adu-Bonsaffoh, K.; Antwi, D.; Obed, S.; Gyan, B. Nitric oxide dysregulation in the pathogenesis of preeclampsia among Ghanaian women. Integr. Blood Press. Control 2015, 8, 1–6. [Google Scholar] [CrossRef][Green Version]
- Malti, N.; Merzouk, H.; Merzouk, S.A.; Loukidi, B.; Karaouzene, N.; Malti, A.; Narce, M. Oxidative stress and maternal obesity: Feto-placental unit interaction. Placenta 2014, 35, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros-Guzmán, A.K.; Carrasco-Legleu, C.E.; Levario-Carrillo, M.; Chávez-Corral, D.V.; Sanchez-Ramirez, B.; Mariñelarena-Carrillo, E.O.; Guerrero-Salgado, F. and Reza-López, S.A. Prepregnancy Obesity, Maternal Dietary Intake, and Oxidative Stress Biomarkers in the Fetomaternal Unit. BioMed Res. Int. 2019, 2019, 5070453. [Google Scholar] [CrossRef]
- MuÑiz, P.; JosÉ, M.; Barchino, G.; Iradi, A.; Mahiques, E.; Marco, V. Age-Related Changes of Liver Antioxidant Enzymes and 8-Hydroxy-2-Deoxyguanosine During Fetal? Neonate Transition and Early Rat Development. IUBMB Life 2000, 49, 497–500. [Google Scholar]
- Akhter, N.; Madhoun, A.; Arefanian, H.; Wilson, A.; Kochumon, S.; Thomas, R.; Shenouda, S.; Al-Mulla, F.; Ahmad, R.; Sindhu, S. Oxidative Stress Induces Expression of the Toll-Like Receptors (TLRs) 2 and 4 in the Human Peripheral Blood Mononuclear Cells: Implications for Metabolic Inflammation. Cell. Physiol. Biochem. 2019, 53, 1–18. [Google Scholar]
- O'Reilly, J.R.; Reynolds, R.M. The risk of maternal obesity to the long-term health of the offspring. Clin. Endocrinol. 2013, 78, 9–16. [Google Scholar] [CrossRef]
- Landete, J.M. Dietary intake of natural antioxidants: Vitamins and polyphenols. Crit. Rev. Food Sci. Nutr. 2013, 53, 706–721. [Google Scholar] [CrossRef]
- Smolková, B.; Dušinská, M.; Rašlová, K.; McNeill, G.; Spustová, V.; Blažíček, P.; Horská, A.; Collins, A. Seasonal changes in markers of oxidative damage to lipids and DNA; correlations with seasonal variation in diet. Mutat. Res. 2004, 551, 135–144. [Google Scholar] [CrossRef]
- Moens, A.L.; Vrints, C.J.; Claeys, M.J.; Timmermans, J.-P.; Champion, H.C.; Kass, D.A. Mechanisms and potential therapeutic targets for folic acid in cardiovascular disease. Am. J. Physiol. Circ. Physiol. 2008, 294, H1971–H1977. [Google Scholar] [CrossRef]
- Asbaghi, O.; Ghanavati, M.; Ashtary-Larky, D.; Bagheri, R.; Kelishadi, M.R.; Nazarian, B.; Nordvall, M.; Wong, A.; Dutheil, F.; Suzuki, K.; et al. Effects of Folic Acid Supplementation on Oxidative Stress Markers: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Antioxidants 2021, 10, 871. [Google Scholar] [CrossRef] [PubMed]
- Asbaghi, O.; Ashtary-Larky, D.; Bagheri, R.; Moosavian, S.; Olyaei, H.; Nazarian, B.; Kelishadi, M.R.; Wong, A.; Candow, D.; Dutheil, F.; et al. Folic Acid Supplementation Improves Glycemic Control for Diabetes Prevention and Management: A Systematic Review and Dose-Response Meta-Analysis of Randomized Controlled Trials. Nutrients 2021, 13, 2355. [Google Scholar] [CrossRef] [PubMed]
- Chitayat, D.; Matsui, D.; Amitai, Y.; Kennedy, D.; Vohra, S.; Rieder, M.; Koren, G. Folic acid supplementation for pregnant women and those planning pregnancy: 2015 update. J. Clin. Pharmacol. 2015, 56, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Edlow, A.G. Maternal obesity and neurodevelopmental and psychiatric disorders in offspring. Prenat. Diagn. 2017, 37, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M.; Food and Nutrition Board of the Institute of Medicine, The National Academies. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Kaiser, L.L.; Campbell, C.G. Practice Paper of the Academy of Nutrition and Dietetics Abstract: Nutrition and Lifestyle for a Healthy Pregnancy Outcome. J. Acad. Nutr. Diet. 2014, 114, 1447. [Google Scholar] [CrossRef] [PubMed]
- Barquera, S.; Hernández-Barrera, L.; Campos-Nonato, I.; Espinosa, J.; Flores, M.; Rivera, J.A. Energy and nutrient consumption in adults: Analysis of the Mexican National Health and Nutrition Survey 2006. Salud Pública México 2009, 51, S562–S573. [Google Scholar] [CrossRef]
- Stepananko, I.L.; Smirnova, O.G.; Konstantinov, Y.M. Gene network of redox regulation and the problem of integrating local gene networks. In Proceedings of the Second International Conference On Bioinformatics of Genome Regulation And Structure, Novosibirsk, Russia, 7–11 August 2000; Laboratory of Theoretical Genetics, Institute of Cytology and Genetics of Siberian Branch: Novosibirsk, Russia, 2000. [Google Scholar]
- Domann, F.E.; Hitchler, M.J. Aberrant redox biology and epigenetic reprogramming: Co-conspirators across multiple human diseases. Free Radic. Biol. Med. 2021, 170, 2–5. [Google Scholar] [CrossRef]
| Mothers | Normal Weight Mean ± SD (n = 133) | Overweight Mean ± SD (n = 74) | Obesity Mean ± SD (n = 35) | p |
|---|---|---|---|---|
| Age (y) | 23.2 ± 5.2 | 25.9 ± 5.9 | 25.5 ± 5.7 | 0.002 † |
| Height (cm) | 157.1 ± 5.6 | 156.1 ± 5.8 | 155.6 ± 7.1 | 0.377 † |
| Weight before pregnancy (kg) | 54.5 ± 8.0 | 66.3 ± 6.0 | 77.1 ± 8.4 | <0.001 † |
| BMI (kg/m2) | 22.0 ± 2.0 | 27.2 ± 1.3 | 31.8 ± 2.0 | <0.001 † |
| Weight gain (kg) | 11.7 ± 4.8 | 10.7 ± 5.9 | 7.9 ± 5.1 | 0.002 † |
| Perinatal folic acid supplementation &, n (%) | 127 (97.0) | 70 (94.6) | 32 (91.4) | 0.340 ‡ |
| 0–4 weeks of gestation, n (%) 5–20 weeks of gestation, n (%) >20 weeks of gestation, n (%) | 50 (39.4) 67 (52.8) 10 (7.9) | 15 (21.4) 46 (65.7) 9 (12.9) | 5 (14.3) 25 (71.4) 5 (14.3) | 0.016 ‡ |
| Newborns | ||||
| Weight (kg) | 3.1 ± 0.4 | 3.2 ± 0.4 | 3.2 ± 0.5 | 0.159 † |
| Length (cm) | 48.9 ± 2.0 | 49.5 ± 1.8 | 49.7 ± 2.0 | 0.015 † |
| Weeks of gestation | 38.8 ± 1.5 | 39.0 ± 1.2 | 38.4 ± 1.3 | 0.153 † |
| Type of birth/caesarean, n (%) | 32 (26.5) | 29 (40.3) | 12 (35.3) | 0.126 ‡ |
| Apgar score at 1 min (≤ 8) | 5 (3.8) | 6 (8.1) | 5 (14.3) | 0.069 ‡ |
| Sex, male, n (%) | 62 (48.1) | 34 (46.0) | 19 (54.3) | 0.716 ‡ |
| Daily Nutrient Intake | Normal Weight Median (p25, p75) (n = 131) | Overweight Median (p25, p75) (n = 73) | Obesity Median (p25, p75) (n = 34) | p † |
|---|---|---|---|---|
| Energy, (kcal/d) | 2282 (1737, 3053) | 1793 (1590, 2393) | 1711 (1514, 2420) | 0.002 |
| Protein, (g/d) % kcal/d | 101 (68, 139) 17.0 (15.4, 18.3) | 76 (61, 113) 15.7 (14.5, 18.1) | 74 (58, 99) 17.0 (13.8, 20.3) | 0.005 0.275 |
| Carbohydrates, (g/d) % kcal/d | 325 (257, 456) 56.2 (51.9, 59.6) | 284 (246, 369) 59.0 (54.3, 61.1) | 276 (217, 374) 59.9 (56.3, 64.0) | 0.045 0.001 |
| Lipids, (g/d) % kcal/d | 72 (53, 96) 27.2 (24.3, 29.8) | 55 (45, 75) 25.1 (23.1, 27.3) | 49 (40, 63) 23.5 (21.3, 25.8) | <0.001 <0.001 |
| Folate (μg/d) % adequacy ‡ | 255 (184, 379) 79.8 (57.6, 118.5) | 199 (169, 267) 62.3 (52.7, 83.5) | 190 (153, 251) 59.4 (47.7, 78.4) | 0.002 0.002 |
| Vitamin B12 (μg/d) % adequacy ‡ | 4.1 (2.7, 7.4) 206 (134, 370) | 3.1 (2.2, 5.1) 156 (109, 257) | 2.5 (1.9, 5.4) 127 (96, 271) | 0.005 0.005 |
| Vitamin A (RE/d) % adequacy ‡ | 799 (545, 1129) 160 (109, 226) | 578 (453, 982) 116 (91, 196) | 511 (370, 840) 102 (74, 168) | <0.001 <0.001 |
| Vitamin C (mg/d) % adequacy ‡ | 171 (119, 238) 258 (198, 397) | 163 (130, 201) 270 (215, 334) | 154 (125, 204) 257 (207, 340) | 0.540 0.540 |
| Vitamin E (mg/d) % adequacy ‡ | 4.5 (3.4, 5.5) 37 (28, 46) | 4.0 (3.4, 4.9) 34 (28, 41) | 3.8 (2.9, 4.8) 32 (24, 40) | 0.069 0.069 |
| Consumption of fruits and vegetables | ||||
| Fruit (g/d) | 288 (173, 445) | 272 (197, 395) | 258 (175, 399) | 0.557 |
| Vegetables (g/d) | 229 (158, 313) | 185 (140, 254) | 157 (116, 194) | 0.004 |
| Maternal NO (µmol/L)† | p | Maternal MDA (µmol/L)† | p | ||||||
| Normal weight | Overweight | Obesity | Normal weight | Overweight | Obesity | ||||
| p25 | 5.1 | 6.3 | 15.0 | <0.001 | p25 | 5.3 | 3.7 | 8.4 | <0.001 |
| Median | 7.9 | 17.4 | 27.9 | Median | 16.8 | 16.4 | 104.1 | ||
| p75 | 21.1 | 49.9 | 51.5 | p75 | 29.4 | 47.9 | 129.1 | ||
| Newborn NO (µmol/L) † | p | Newborn MDA (µmol/L) † | p | ||||||
| Normal weight | Overweight | Obesity | Normal weight | Overweight | Obesity | ||||
| p25 | 5.5 | 9.6 | 15.9 | 0.001 | p25 | 0.6 | 0.7 | 2.3 | <0.001 |
| Median | 14.2 | 22.1 | 53.5 | Median | 0.8 | 15.3 | 73.3 | ||
| p75 | 30.0 | 30.9 | 84.3 | p75 | 19.9 | 51.6 | 114.7 | ||
| Nitric Oxide (µmol/L) † | ||||||
| Mothers | Newborns | |||||
| Tertile 1 (1.0 to 6.5) p50 (p25, p75) | Tertile 2 (6.8 to 22.8) p50 (p25, p75) | Tertile 3 (23.0 to 384.4) p50 (p25, p75) | Tertile 1 (0 to 9.6) p50 (p25, p75) | Tertile 2 (10.2 to 29.1) p50 (p25, p75) | Tertile 3 (29.2 to 231.9) p50 (p25, p75) | |
| Fruits (g/d) | 336 (272, 419) | 293 (234, 369) | 242 (215, 271) | 328 (253, 398) | 297 (234, 363) | 252 (214, 310) |
| Vegetables (g/d) | 235 (198, 299) | 207 (162, 262) | 166 (150, 193) | 224 (183, 271) | 211 (164, 262) | 177 (150, 217) |
| Folate (μg/d) | 296 (224, 391) | 247 (178, 334) | 189 (157, 221) | 285 (201, 367) | 253 (178, 328) | 201 (157, 267) |
| Vitamin A (ER/d) | 784 (635, 1045) | 675 (493, 902) | 508 (448, 615) | 741 (572, 934) | 699 (497, 886) | 554 (448, 722) |
| Vitamin C (mg/d) | 187 (153, 220) | 167 (144, 202) | 149 (135, 163) | 184 (149, 210) | 167 (147, 197) | 155 (135, 179) |
| Vitamin B12 (μg/d) | 4.2 (3.4, 5.7) | 3.6 (2.6, 4.9) | 2.6 (2.3, 3.2) | 4.0 (3.0, 5.0) | 3.7 (2.6, 4.8) | 2.9 (2.3, 3.9) |
| Vitamin E (mg/d) | 4.7 (3.9, 5.6) | 4.2 (3.5, 5.0) | 3.6 (3.3, 3.9) | 4.6 (3.7, 5.4) | 4.2 (3.5, 4.9) | 3.8 (3.3, 4.3) |
| Perinatal folic acid supplementation | ||||||
| 0–4 weeks of gestation, n (%) | 32 (47.8) | 30 (44.8) | 5 (7.4) | 33 (56.9) | 17 (29.3) | 8 (13.8) |
| 5–20 weeks of gestation, n (%) | 32 (25.2) | 39 (30.7) | 56 (44.1) | 25 (21.6) | 44 (37.9) | 47 (40.5) |
| >20 weeks of gestation, n (%) | 4 (17.4) | 6 (26.1) | 13 (56.5) | 5 (21.8) | 7 (30.4) | 11 (47.8) |
| Malondialdehyde (µmol/L) † | ||||||
| Mothers | Newborns | |||||
| Tertile 1 (0 to 11.8) p50 (p25, p75) | Tertile 2 (11.9 to 30.3) p50 (p25, p75) | Tertile 3 (30.8 to 707.9) p50 (p25, p75) | Tertile 1 (0 to 0.72) p50 (p25, p75) | Tertile 2 (0.74 to 26.1) p50 (p25, p75) | Tertile 3 (28.4 to 700.8) p50 (p25, p75) | |
| Fruits (g/d) | 325 (246, 422) | 398 (257, 366) | 245 (209, 275) | 339 (283, 419) | 297 (237, 366) | 247 (216, 276) |
| Vegetables (g/d) | 217 (171, 294) | 223 (186, 262) | 172 (149, 193) | 239 (198, 294) | 211 (166, 263) | 173 (149, 191) |
| Folate intake (μg/d) | 283 (1934, 393) | 277 (207, 332) | 191 (152, 228) | 300 (235, 391) | 253 (183, 332) | 192 (157, 229) |
| Vitamin A intake (REd) | 733 (531, 1034) | 731 (586, 898) | 529 (432, 614) | 815 (636, 1032) | 706 (506, 896) | 549 (442, 615) |
| Vitamin C intake (mg/d) | 183 (149, 224) | 177 (152, 197) | 151 (135, 167) | 184 (156, 221) | 166 (146, 202) | 151 (135, 167) |
| Vitamin B12 intake (μg/d) | 3.9 (2.8, 5.6) | 3.9 (3.1, 4.9) | 2.6 (2.2, 3.2) | 4.4 (3.4, 5.6) | 3.8 (2.6, 4.8) | 2.9 (2.3, 3.2) |
| Vitamin E intake (mg/d) | 4.5 (3.6, 5.7) | 4.4 (3.7, 5.0) | 3.6 (3.2, 3.9) | 4.6 (4.0, 5.6) | 4.2 (3.5, 5.0) | 3.7 (3.3, 4.0) |
| Perinatal folic acid supplementation | ||||||
| 0–4 weeks of gestation, n (%) | 30 (44.8) | 28 (41.8) | 9 (13.4) | 29 (50.0) | 23 (39.7) | 6 (10.3) |
| 5–20 weeks of gestation, n (%) | 35 (28.7) | 40 (32.8) | 47 (38.5) | 35 (31.0) | 28 (24.8) | 50 (44.2) |
| >20 weeks of gestation, n (%) | 4 (18.2) | 5 (22.7) | 13 (59.1) | 4 (17.4) | 9 (39.1) | 10 (43.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopez-Yañez Blanco, A.; Díaz-López, K.M.; Vilchis-Gil, J.; Diaz-Garcia, H.; Gomez-Lopez, J.; Medina-Bravo, P.; Granados-Riveron, J.T.; Gallardo, J.M.; Klünder-Klünder, M.; Sánchez-Urbina, R. Diet and Maternal Obesity Are Associated with Increased Oxidative Stress in Newborns: A Cross-Sectional Study. Nutrients 2022, 14, 746. https://doi.org/10.3390/nu14040746
Lopez-Yañez Blanco A, Díaz-López KM, Vilchis-Gil J, Diaz-Garcia H, Gomez-Lopez J, Medina-Bravo P, Granados-Riveron JT, Gallardo JM, Klünder-Klünder M, Sánchez-Urbina R. Diet and Maternal Obesity Are Associated with Increased Oxidative Stress in Newborns: A Cross-Sectional Study. Nutrients. 2022; 14(4):746. https://doi.org/10.3390/nu14040746
Chicago/Turabian StyleLopez-Yañez Blanco, Arturo, Keyla M Díaz-López, Jenny Vilchis-Gil, Hector Diaz-Garcia, Jacqueline Gomez-Lopez, Patricia Medina-Bravo, Javier T Granados-Riveron, Juan M Gallardo, Miguel Klünder-Klünder, and Rocío Sánchez-Urbina. 2022. "Diet and Maternal Obesity Are Associated with Increased Oxidative Stress in Newborns: A Cross-Sectional Study" Nutrients 14, no. 4: 746. https://doi.org/10.3390/nu14040746
APA StyleLopez-Yañez Blanco, A., Díaz-López, K. M., Vilchis-Gil, J., Diaz-Garcia, H., Gomez-Lopez, J., Medina-Bravo, P., Granados-Riveron, J. T., Gallardo, J. M., Klünder-Klünder, M., & Sánchez-Urbina, R. (2022). Diet and Maternal Obesity Are Associated with Increased Oxidative Stress in Newborns: A Cross-Sectional Study. Nutrients, 14(4), 746. https://doi.org/10.3390/nu14040746