Effects of Probiotic Supplementation on Exercise with Predominance of Aerobic Metabolism in Trained Population: A Systematic Review, Meta-Analysis and Meta-Regression
Abstract
1. Introduction
2. Material and Methods
2.1. Literature Search Strategies
2.2. Inclusion Criteria and Exclusion Criteria
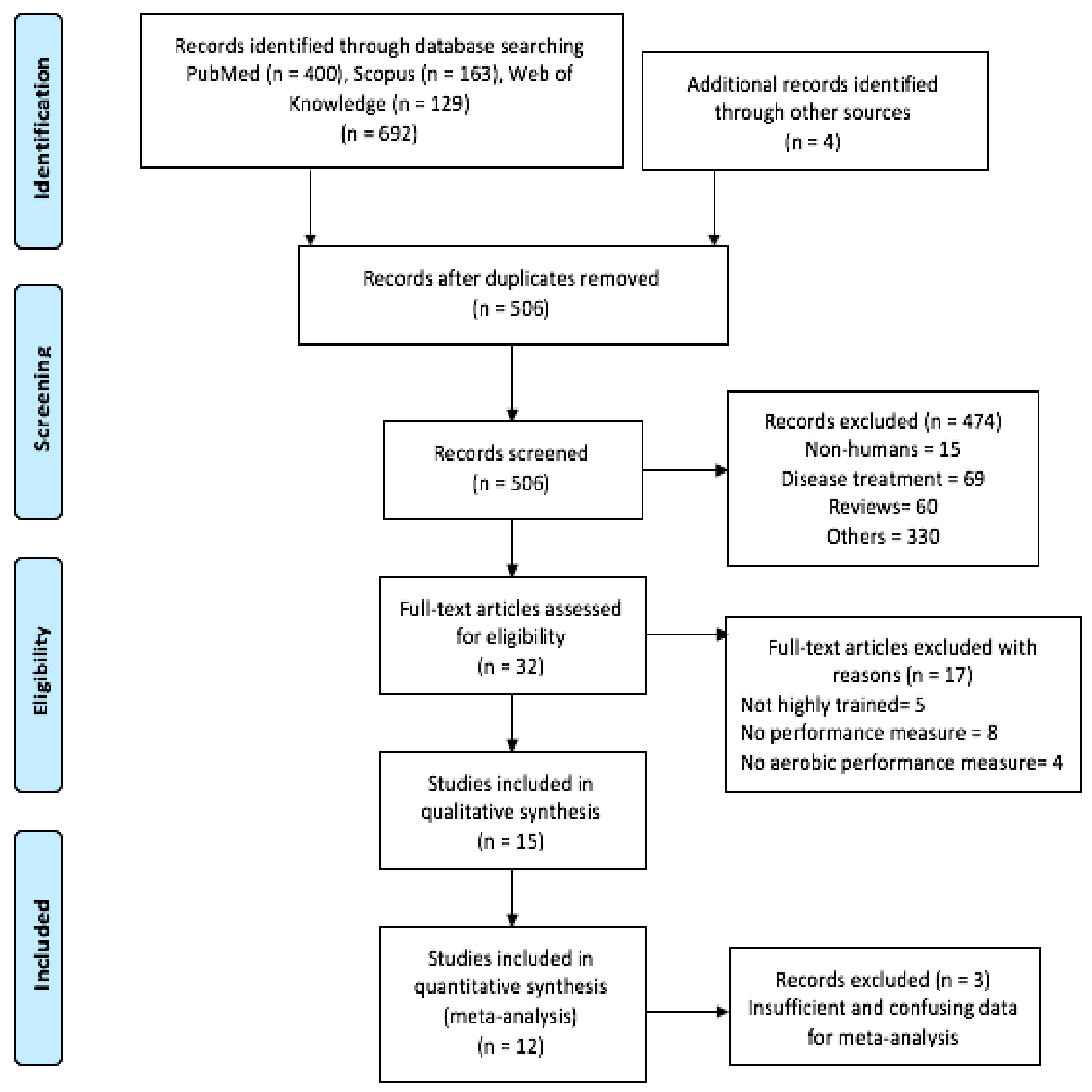
2.3. Text Screening
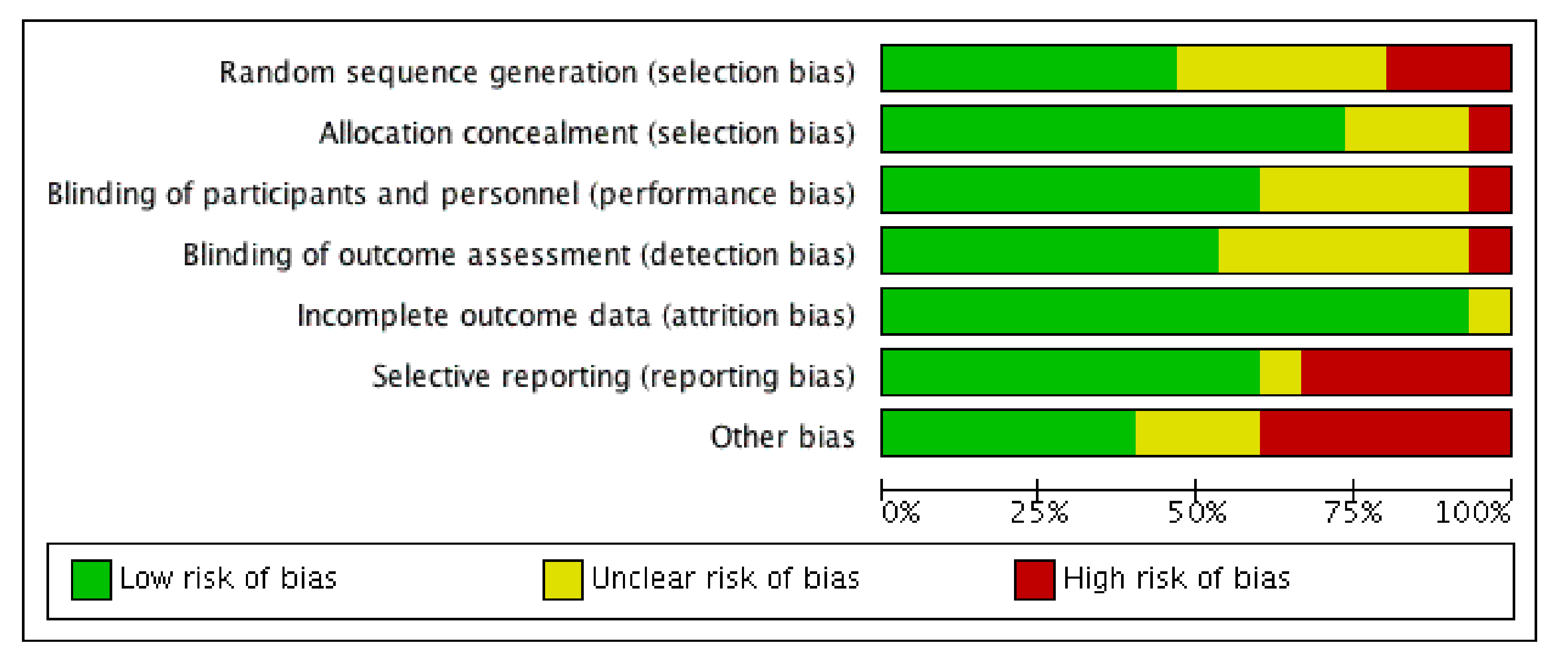
2.4. Data Extraction, Study Coding, and Quality Assessment
2.5. Statistical Analysis
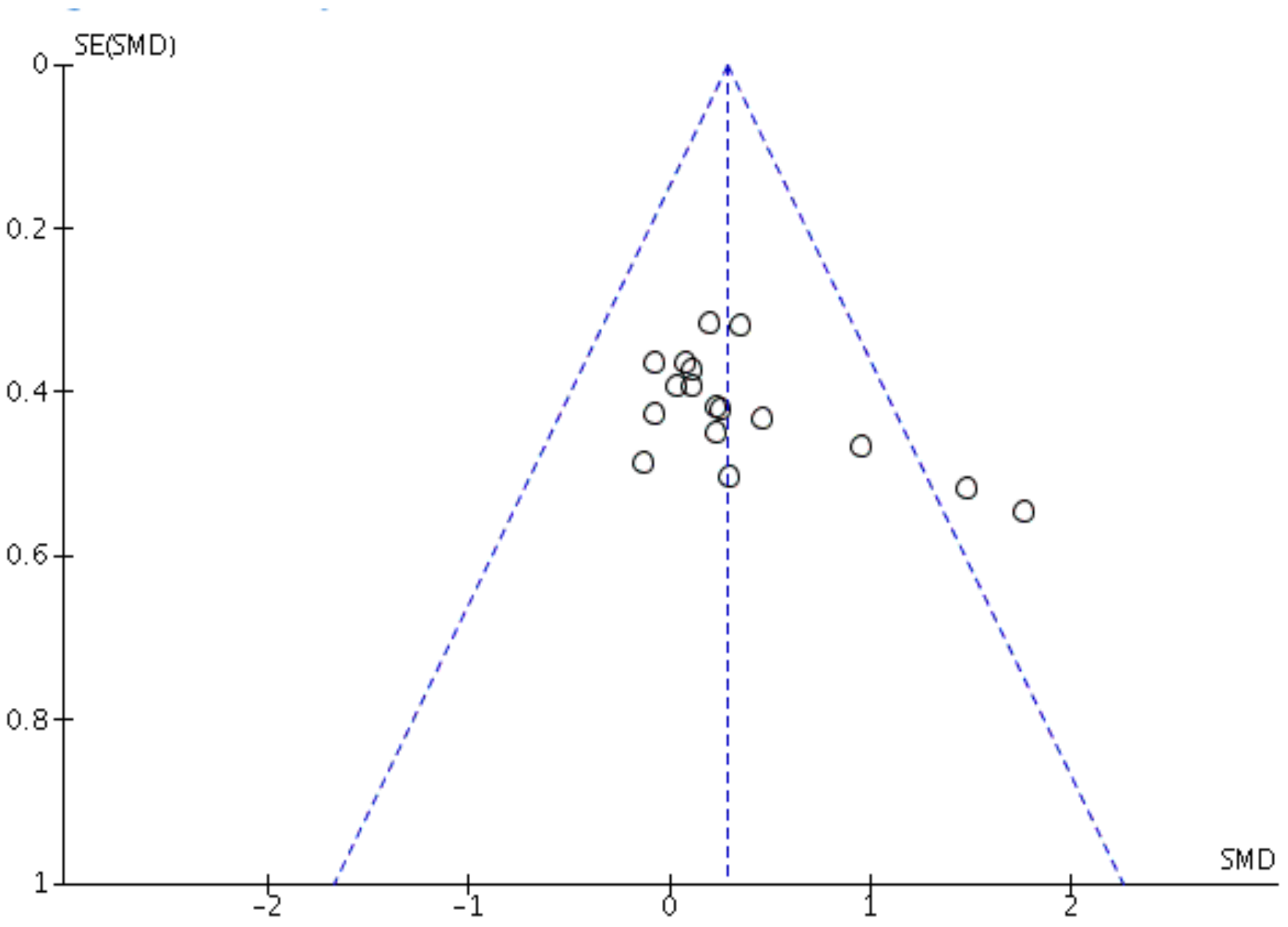
3. Results
3.1. Main Research
3.2. Probiotic Supplementation
3.3. Effect of Probiotics on Exercise in Which Aerobic Metabolism Is Predominant (≥5 min)
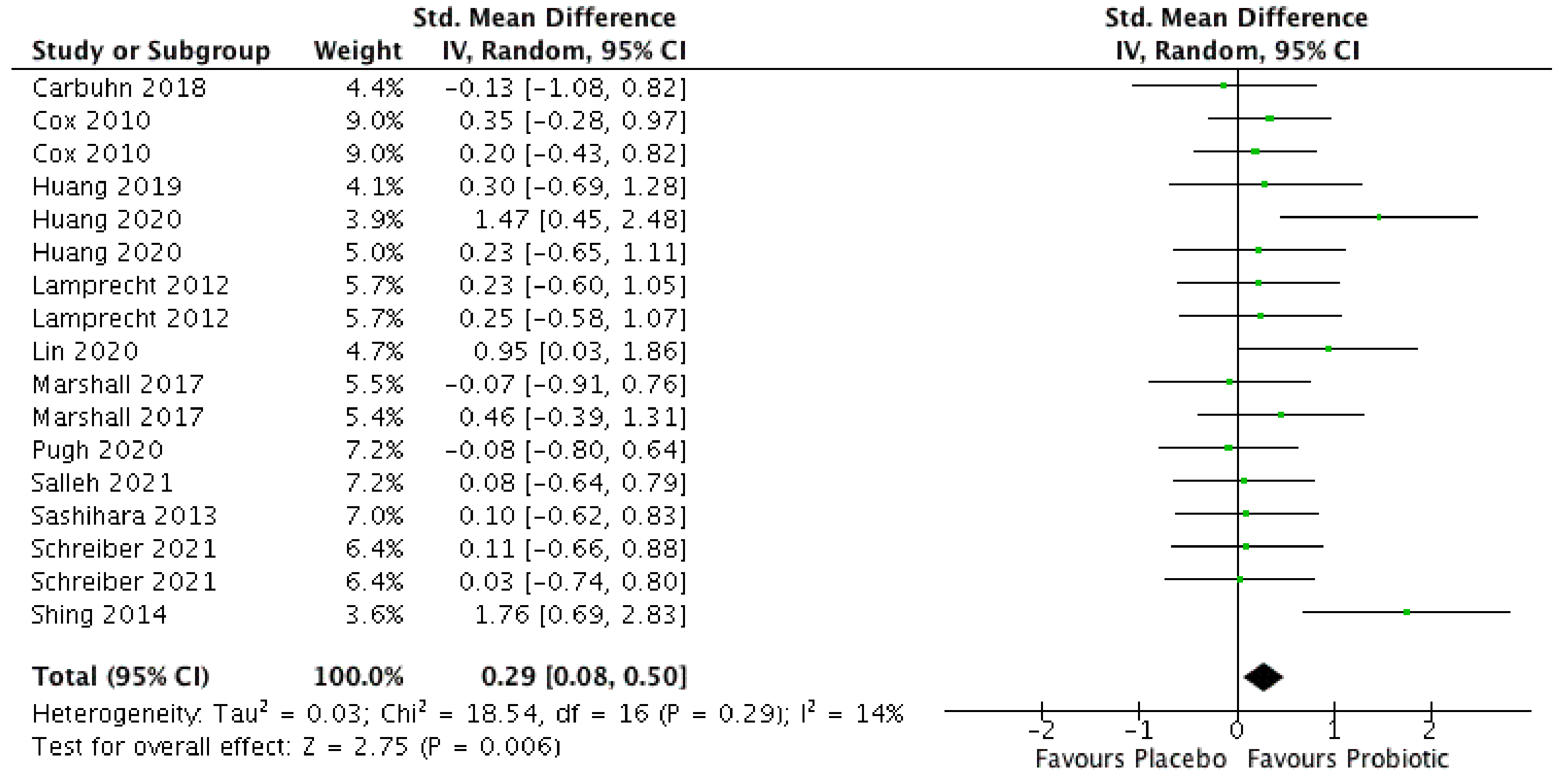
3.4. Effect of Probiotics on Exercise in Which Aerobic Metabolism Is Predominant (≥5 min). Meta-Analysis
3.5. Effect of Different Characteristics of Studies on Exercise in Which Aerobic Metabolism Is Predominant (≥5 min). Meta-Analysis
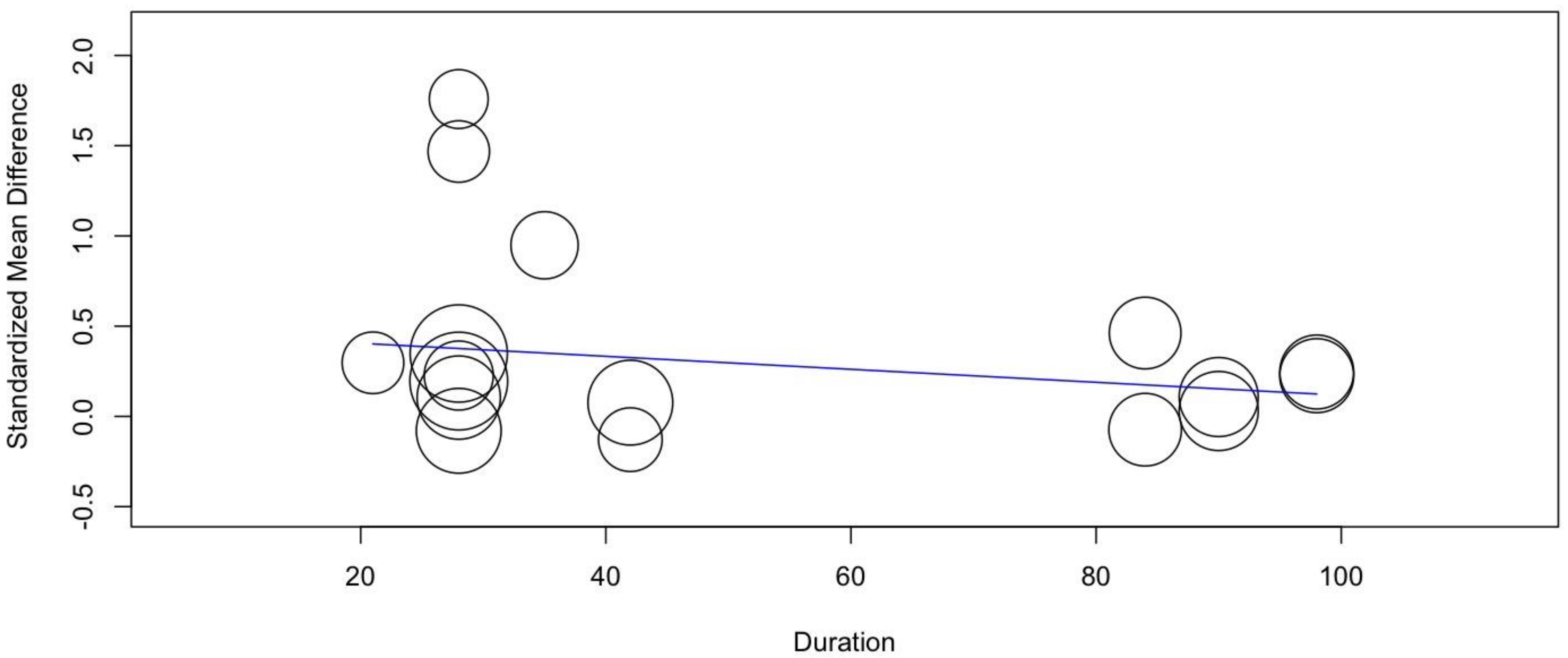
3.6. Results of Meta-Regression
4. Discussion
4.1. Effect of Probiotics on Exercise in Which Aerobic Meatabolism Is Predominant (≥5 min)
4.2. Effects of Different Characteristics of Studies on Exercise with Aerobic Metabolism Is Predominant (≥5 min)
4.3. Limitations, Strengths and Future Research
4.4. Practical Applications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- West, N.P.; Pyne, D.B.; Peake, J.M.; Cripps, A.W. Probiotics, immunity and exercise: A review. Exerc. Immunol. Rev. 2009, 15, 107–126. [Google Scholar] [PubMed]
- McFarland, L.V.; Evans, C.T.; Goldstein, E.J.C. Strain-specificity and disease-specificity of probiotic efficacy: A systematic review and meta-analysis. Front. Med. 2018, 5, 124. [Google Scholar] [CrossRef]
- Jäger, R.; Mohr, A.E.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Moussa, A.; Townsend, J.R.; Lamprecht, M.; West, N.P.; Black, K.; et al. International society of sports nutrition position stand: Probiotics. J. Int. Soc. Sports Nutr. 2019, 16, 1–44. [Google Scholar] [CrossRef] [PubMed]
- Lamprecht, M.; Bogner, S.; Schippinger, G.; Steinbauer, K.; Fankhauser, F.; Hallstroem, S.; Schuetz, B.; Greilberger, J.F. Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2012, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Clancy, R.L.; Gleeson, M.; Cox, A.; Callister, R.; Dorrington, M.; D’Este, C.; Pang, G.; Pyne, D.; Fricker, P.; Henriksson, A. Reversal in fatigued athletes of a defect in interferon γ secretion after administration of Lactobacillus acidophilus. Br. J. Sports Med. 2006, 40, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Inside the microbial and immune labyrinth: Gut microbes: Friends or fiends? Nat. Med. 2010, 16, 1195–1197. [Google Scholar] [CrossRef][Green Version]
- Martarelli, D.; Verdenelli, M.C.; Scuri, S.; Cocchioni, M.; Silvi, S.; Cecchini, C.; Pompei, P. Effect of a probiotic intake on oxidant and antioxidant parameters in plasma of athletes during intense exercise training. Curr. Microbiol. 2011, 62, 1689–1696. [Google Scholar] [CrossRef]
- Liu, J.; Ruckwardt, T.J.; Chen, M.; Nicewonger, J.D.; Johnson, T.R.; Graham, B.S. Epitope-specific regulatory CD4 T cells reduce virus-induced illness while preserving CD8 T-cell effector function at the site of infection. J. Virol. 2010, 84, 10501–10509. [Google Scholar] [CrossRef][Green Version]
- Otczyk, D.C.; Cripps, A.W. Mucosal immunization: A realistic alternative. Hum. Vaccin. 2010, 6, 978–1006. [Google Scholar] [CrossRef]
- Geuking, M.B.; McCoy, K.D.; Macpherson, A.J. Metabolites from intestinal microbes shape treg. Cell Res. 2013, 23, 1339–1340. [Google Scholar] [CrossRef]
- Coqueiro, A.Y.; Garcia, A.B.d.O.; Rogero, M.M.; Tirapegui, J. Probiotic supplementation in sports and physical exercise: Does it present any ergogenic effect? Nutr. Health 2017, 23, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. Effect of probiotics supplementations on health status of athletes. Int. J. Environ. Res. Public Health 2019, 16, E4469. [Google Scholar] [CrossRef] [PubMed]
- Pyne, D.B.; Hopkins, W.G.; Batterham, A.M.; Gleeson, M.; Fricker, P.A. Characterising the individual performance responses to mild illness in international swimmers. Br. J. Sports Med. 2005, 39, 752–756. [Google Scholar] [CrossRef] [PubMed]
- Miles, M.P. Probiotics and gut health in athletes. Curr. Nutr. Rep. 2020, 9, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P. Nutrition and athlete immune health: New perspectives on an old paradigm. Sport Med. 2019, 49, 153–168. [Google Scholar] [CrossRef]
- Łagowska, K.; Bajerska, J. Effects of probiotic supplementation on respiratory infection and immune function in athletes: Systematic review and meta-analysis of randomized controlled trials. J. Ahletic Train. 2021, Online ahead of print. [CrossRef]
- Clark, A.; Mach, N. The crosstalk between the gut microbiota and mitochondria during exercise. Front. Physiol. 2017, 8, 319. [Google Scholar] [CrossRef]
- Macarro, M.S.; Ávila-Dandía, V.; Pérez-Piñero, S.; Cánovas, F.; García-Muñoz, A.M.; Abellán-Ruiz, M.S.; Victoria-Montesinos, D.; Luque-Rubia, A.J.; Climent, E.; Genovés, S.; et al. Antioxidant effect of a probiotic product on a model of oxidative stress induced by high-intensity and duration physical exercise. Antioxidants 2021, 10, 323. [Google Scholar] [CrossRef]
- Zamani, B.; Sheikhi, A.; Namazi, N.; Larijani, B.; Azadbakht, L. The effects of supplementation with probiotic on biomarkers of oxidative stress in adult aubjects: A systematic review and meta-analysis of randomized trials. Probiotics Antimicrob. Proteins 2020, 12, 102–111. [Google Scholar] [CrossRef]
- Tong, T.K.; Fu, F.H.; Chow, B.C. Reliability of a 5-min running field test and its accuracy in VO2max evaluation. J. Sports Med. Phys. Fit. 2001, 41, 318–323. [Google Scholar]
- Viribay, A.; Burgos, J.; Fernández-Landa, J.; Seco-Calvo, J.; Mielgo-Ayuso, J. Effects of arginine supplementation on athletic performance based on energy metabolism: A systematic review and meta-analysis. Nutrients 2020, 12, 1300. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, E.P.; Burini, R.C.; Jeukendrup, A. Gastrointestinal complaints during exercise: Prevalence, etiology, and nutritional recommendations. Sport Med. 2014, 44, S79–S85. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Mohr, A.E.; Pugh, J.N. Recent advances in clinical probiotic research for sport. Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 428–436. [Google Scholar] [CrossRef]
- Pugh, J.N.; Wagenmakers, A.J.M.; Doran, D.A.; Fleming, S.C.; Fielding, B.A.; Morton, J.P.; Close, G.L. Probiotic supplementation increases carbohydrate metabolism in trained male cyclists:A randomized, double-blind, placebo-controlled crossover trial. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E504–E513. [Google Scholar] [CrossRef]
- Markowiak-Kopeć, P.; Śliżewska, K. The effect of probiotics on the production of short-chain fatty acids by human intestinal microbiome. Nutrients 2020, 12, 1107. [Google Scholar] [CrossRef]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef]
- Cox, A.J.; Pyne, D.B.; Saunders, P.U.; Fricker, P.A. Oral administration of the probiotic Lactobacillus fermentum VRI-003 and mucosal immunity in endurance athletes. Br. J. Sports Med. 2010, 44, 222–226. [Google Scholar] [CrossRef]
- Huang, W.C.; Wei, C.C.; Huang, C.C.; Chen, W.L.; Huang, H.Y. The beneficial effects of Lactobacillus plantarum PS128 on high-intensity, exercise-induced oxidative stress, inflammation, and performance in triathletes. Nutrients 2019, 11, 353. [Google Scholar] [CrossRef]
- Huang, W.C.; Pan, C.H.; Wei, C.C.; Huang, H.Y. Lactobacillus plantarum ps128 improves physiological adaptation and performance in triathletes through gut microbiota modulation. Nutrients 2020, 12, 2315. [Google Scholar] [CrossRef]
- Marinkovic, D.; Minic, R.; Dikic, N.; Andjelkovic, M.; Kostic-Vucicevic, M.; Stojmenovic, T.; Nikolic, I.; Djordjevic, B. Lactobacillus helveticus Lafti L10 supplementation reduces respiratory infection duration in a cohort of elite athletes: A randomized, double-blind, placebo-controlled trial. Appl. Physiol. Nutr. Metab. 2016, 41, 782–789. [Google Scholar]
- Salleh, R.M.; Kuan, G.; Aziz, M.N.A.; Rahim, M.R.A.; Rahayu, T.; Sulaiman, S.; Kusuma, D.W.Y.; Adikari, A.M.G.C.P.; Razam, M.S.M.; Radhakrishnan, A.K.; et al. Effects of probiotics on anxiety, stress, mood and fitness of badminton players. Nutrients 2021, 13, 1783. [Google Scholar] [CrossRef]
- Sashihara, T.; Nagata, M.; Mori, T.; Ikegami, S.; Gotoh, M.; Okubo, K.; Uchida, M.; Itoh, H. Effects of lactobacillus gasseri OLL2809 and α-lactalbumin on university-student athletes: A randomized, double-blind, placebo-controlled clinical trial. Appl. Physiol. Nutr. Metab. 2013, 38, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- West, N.P.; Pyne, D.B.; Cripps, A.W.; Hopkins, W.G.; Eskesen, D.C.; Jairath, A.; Christophersen, C.T.; Conlon, M.A.; Fricker, P.A. Lactobacillus fermentum (PCC®) supplementation and gastrointestinal and respiratory-tract illness symptoms: A randomised control trial in athletes. Nutr. J. 2011, 10, 1–11. [Google Scholar] [CrossRef]
- Carbuhn, A.F.; Reynolds, S.M.; Campbell, C.W.; Bradford, L.A.; Deckert, J.A.; Kreutzer, A.; Fry, A.C. Effects of probiotic (Bifidobacterium longum 35624) supplementation on exercise performance, immune modulation, and cognitive outlook in division I female swimmers. Sports 2018, 6, 116. [Google Scholar] [CrossRef]
- Lin, C.-L.; Hsu, Y.-J.; Ho, H.-H.; Chang, Y.-C.; Kuo, Y.-W.; Yeh, Y.T.; Tsai, S.-Y.; Chen, C.-W.; Chen, J.-F.; Huang, C.-C.; et al. Bifidobacterium longum subsp. longum OLP-01 supplementation during endurance running training improves exercise performance in middle- and long-distance runners: A double-blind controlled trial. Nutrients 2020, 12, 1972. [Google Scholar] [CrossRef]
- Marshall, H.; Chrismas, B.C.R.; Suckling, C.A.; Roberts, J.D.; Foster, J.; Taylor, L. Chronic probiotic supplementation with or without glutamine does not influence the eHsp72 response to a multi-day ultra-endurance exercise event. Appl. Physiol. Nutr. Metab. 2017, 42, 876–883. [Google Scholar] [CrossRef]
- Shing, C.M.; Peake, J.M.; Lim, C.L.; Briskey, D.; Walsh, N.P.; Fortes, M.B.; Ahuja, K.D.; Vitetta, L. Effects of probiotics supplementation on gastrointestinal permeability, inflammation and exercise performance in the heat. Eur. J. Appl. Physiol. 2014, 114, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Strasser, B.; Geiger, D.; Schauer, M.; Gostner, J.M.; Gatterer, H.; Burtscher, M.; Fuchs, D. Probiotic supplements beneficially affect tryptophan–kynurenine metabolism and reduce the incidence of upper respiratory tract infections in trained athletes: A randomized, double-blinded, placebo-controlled trial. Nutrients 2016, 8, 752. [Google Scholar] [CrossRef]
- Schreiber, C.; Tamir, S.; Golan, R.; Weinstein, A.; Weinstein, Y. The effect of probiotic supplementation on performance, inflammatory markers and gastro-intestinal symptoms in elite road cyclists. J. Int. Soc. Sports Nutr. 2021, 18, 36. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Academia and clinic annals of internal medicin preferred reporting items for systematic reviews and meta-analyses. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Sumathipala, A.; Siribaddana, S.; Hewage, S.; Lekamwattage, M.; Athukorale, M.; Siriwardhana, C.; Munasinghe, K.; Sumathipala, K.; Murray, J.; Prince, M. Understanding of research: A Sri Lankan perspective. BMC Med. Ethics 2010, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Higgings, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Chichester, UK, 2008. [Google Scholar]
- Morton, N.A. De The PEDro scale is a valid measure of the methodological quality of clinical trials: A demographic study. Aust. J. Physiother. 2009, 55, 129–133. [Google Scholar] [CrossRef]
- Hedges, L. V Distribution theory for Glass’s estimator of effect size and related estimators. J. Educ. Behav. Stat. 1981, 6, 107–128. [Google Scholar] [CrossRef]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Academic Press: Cambridge, MA, USA, 2013; ISBN 1483276481. [Google Scholar]
- Higgings, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 237, 557–560. [Google Scholar] [CrossRef]
- Fernandez-Landa, J.; Calleja-Gonzalez, J.; León-Guereño, P.; Caballero-Garcia, A.; Córdova, A.; Mielgo-Ayuso, J. Effect of the combination of creatine monohydrate plus HMB supplementation on sports performance, body composition, markers of muscle damage and hormone status: A systematic review. Nutrients 2019, 11, 2528. [Google Scholar] [CrossRef]
- Hawley, J.A.; Leckey, J.J. Carbohydrate dependence during prolonged, intense endurance exercise. Sport Med. 2015, 45, 5–12. [Google Scholar] [CrossRef]
- Jeukendrup, A.E. Training the gut for athletes. Sport Med. 2017, 47, 101–110. [Google Scholar] [CrossRef]
- Krishna Rao, R.; Samak, G. Protection and restitution of gut barrier by probiotics: Nutritional and clinical implications. Curr. Nutr. Food Sci. 2013, 9, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Dokladny, K.; Zuhl, M.N.; Moseley, P.L. Intestinal epithelial barrier function and tight junction proteins with heat and exercise. J. Appl. Physiol. 2016, 120, 692–701. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Lauretani, F.; Tana, C.; Nouvenne, A.; Ridolo, E.; Meschi, T. Exercise and immune system as modulators of intestinal microbiome: Implications for the gut-muscle axis hypothesis. Exerc. Immunol. Rev. 2019, 25, 84–95. [Google Scholar] [PubMed]
- De Oliveira, E.P.; Burini, R.C. Carbohydrate-dependent, exercise-induced gastrointestinal distress. Nutrients 2014, 6, 4191–4199. [Google Scholar] [CrossRef] [PubMed]
- Van Wijck, K.; Lenaerts, K.; van Loon, L.J.C.; Peters, W.H.M.; Buurman, W.A.; Dejong, C.H.C. Exercise-induced splanchnic hypoperfusion results in gut dysfunction in healthy men. PLoS ONE 2011, 6, e223. [Google Scholar] [CrossRef] [PubMed]
- Pugh, J.N.; Phelan, M.M.; Caamaño-Gutiérrez, E.; Sparks, S.A.; Morton, J.P.; Close, G.L.; Owens, D.J. Four weeks of probiotic supplementation alters the metabolic perturbations induced by marathon running: Insight from metabolomics. Metabolites 2021, 11, 535. [Google Scholar] [CrossRef]
- Currell, K.; Jeukendrup, A.E. Superior endurance performance with ingestion of multiple transportable carbohydrates. Med. Sci. Sports Exerc. 2008, 40, 275–281. [Google Scholar] [CrossRef]
- Børsheim, E.; Cree, M.G.; Tipton, K.D.; Elliott, T.A.; Aarsland, A.; Wolfe, R.R. Effect of carbohydrate intake on net muscle protein synthesis during recovery from resistance exercise. J. Appl. Physiol. 2004, 96, 674–678. [Google Scholar] [CrossRef]
- Jäger, R.; Shields, K.A.; Lowery, R.P.; De Souza, E.O.; Partl, J.M.; Hollmer, C.; Purpura, M.; Wilson, J.M. Probiotic Bacillus coagulans GBI-30, 6086 reduces exercise-induced muscle damage and increases recovery. PeerJ 2016, 4, e2276. [Google Scholar] [CrossRef]
- Jäger, R.; Zaragoza, J.; Purpura, M.; Iametti, S.; Marengo, M.; Tinsley, G.M.; Anzalone, A.J.; Oliver, J.M.; Fiore, W.; Biffi, A.; et al. Probiotic administration increases amino acid absorption from plant protein—A placebo-controlled, randomized, double-blind, multicenter, crossover study. Probiotics Antimicrob. Proteins 2020, 12, 1330–1339. [Google Scholar] [CrossRef]
- Boirivant, M.; Strober, W. The mechanism of action of probiotics. Curr. Opin. Gastroenterol. 2007, 23, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef]
- Blad, C.C.; Tang, C.; Offermanns, S. G protein-coupled receptors for energy metabolites as new therapeutic targets. Nat. Rev. Drug Discov. 2012, 11, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Offermanns, S. Free fatty acid (FFA) and hydroxy carboxylic acid (HCA) receptors. Annu. Rev. Pharmacol. Toxicol. 2014, 54, 407–434. [Google Scholar] [CrossRef]
- Cheng, Z.; Ristow, M. Mitochondria and metabolic homeostasis. Antioxid. Redox Signal. 2013, 19, 240–242. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Den Besten, G.; Gerding, A.; van Dijk, T.H.; Ciapaite, J.; Bleeker, A.; van Eunen, K.; Havinga, R.; Groen, A.K.; Reijngoud, D.J.; Bakker, B.M. Protection against the metabolic syndrome by guar gum-derived short-chain fatty acids depends on peroxisome proliferator-activated receptor γ and Glucagon-like peptide-1. PLoS ONE 2015, 10, e0136364. [Google Scholar] [CrossRef]
- Hood, D.A.; Uguccioni, G.; Vainshtein, A.; D’Souza, D. Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle: Implications for health and disease. Compr. Physiol. 2011, 1, 1119–1134. [Google Scholar]
- Chen, Y.M.; Wei, L.; Chiu, Y.S.; Hsu, Y.J.; Tsai, T.Y.; Wang, M.F.; Huang, C.C. Lactobacillus plantarum TWK10 supplementation improves exercise performance and increases muscle mass in mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef]
- Przewłócka, K.; Folwarski, M.; Kaźmierczak-Siedlecka, K.; Skonieczna-żydecka, K.; Kaczor, J.J. Gut-muscle axisexists and may affect skeletal muscle adaptation to training. Nutrients 2020, 12, 1451. [Google Scholar] [CrossRef] [PubMed]
- Peternelj, T.T.; Coombes, J.S. Antioxidant supplementation during exercise training: Beneficial or detrimental? Sport Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef]
- Borges, L.D.S.; Dermargos, A.; Junior, E.P.D.S.; Weimann, E.; Lambertucci, R.H.; Hatanaka, E. Melatonin decreases muscular oxidative stress and inflammation induced by strenuous exercise and stimulates growth factor synthesis. J. Pineal Res. 2015, 58, 166–172. [Google Scholar] [CrossRef]
- Spyropoulos, B.G.; Misiakos, E.P.; Fotiadis, C.; Stoidis, C.N. Antioxidant properties of probiotics and their protective effects in the pathogenesis of radiation-induced enteritis and colitis. Dig. Dis. Sci. 2011, 56, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Jandhyala, S.M.; Talukdar, R.; Subramanyam, C.; Vuyyuru, H.; Sasikala, M.; Reddy, D.N. Role of the normal gut microbiota. World J. Gastroenterol. 2015, 21, 8787. [Google Scholar] [CrossRef]
- Bermudez-Brito, M.; Plaza-Díaz, J.; Muñoz-Quezada, S.; Gómez-Llorente, C.; Gil, A. Probiotic mechanisms of action. Ann. Nutr. Metab. 2012, 61, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Jara, D.-J.; Eduardo, S.-S.; Ordoñez, F.J.; Rosety, I.; Díaz, A.J.; Rosety-Rodriguez, M.; Rosety, M.Á.; Brenes, F. Impact of probiotics on the performance of endurance athletes: A systematic review. Int. J. Environ. Res. Public Health 2021, 18, 11576. [Google Scholar]
- Park, C.; Brietzke, E.; Rosenblat, J.D.; Musial, N.; Zuckerman, H.; Ragguett, R.M.; Pan, Z.; Rong, C.; Fus, D.; McIntyre, R.S. Probiotics for the treatment of depressive symptoms: An anti-inflammatory mechanism? Brain. Behav. Immun. 2018, 73, 115–124. [Google Scholar] [CrossRef]
- McFarland, L. V Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease. Am. J. Gastroenterol. 2006, 101, 812–822. [Google Scholar] [CrossRef]
- Khalesi, S.; Sun, J.; Buys, N.; Jayasinghe, R. Effect of probiotics on blood pressure: A systematic review and meta-analysis of randomized, controlled trials. Hypertension 2014, 64, 897–903. [Google Scholar] [CrossRef]
- Markowiak, P.; Ślizewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Butel, M.J. Probiotics, gut microbiota and health. Med. Mal. Infect. 2014, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wieërs, G.; Belkhir, L.; Enaud, R.; Leclercq, S.; Philippart de Foy, J.M.; Dequenne, I.; de Timary, P.; Cani, P.D. How probiotics affect the microbiota. Front. Cell. Infect. Microbiol. 2020, 9, 454. [Google Scholar] [CrossRef] [PubMed]
- Kechagia, M.; Basoulis, D.; Konstantopoulou, S.; Dimitriadi, D.; Gyftopoulou, K.; Skarmoutsou, N.; Fakiri, E.M. Health benefits of probiotics: A review. ISRN Nutr. 2013, 2013, 481651. [Google Scholar] [CrossRef] [PubMed]
- Pyne, D.B.; West, N.P.; Cripps, A.W. Probiotics and immune eesponse to exercise. Am. J. Lifestyle Med. 2012, 7, 51–59. [Google Scholar] [CrossRef]
- Haywood, B.A.; Black, K.E.; Baker, D.; McGarvey, J.; Healey, P.; Brown, R.C. Probiotic supplementation reduces the duration and incidence of infections but not severity in elite rugby union players. J. Sci. Med. Sport 2014, 17, 356–360. [Google Scholar] [CrossRef]
- Lleo, A.; Battezzati, P.M.; Selmi, C.; Gershwin, M.E.; Podda, M. Is autoimmunity a matter of sex? Autoimmun. Rev. 2008, 7, 626–630. [Google Scholar] [CrossRef]
- He, C.S.; Bishop, N.C.; Handzlik, M.K.; Muhamad, A.S.; Gleeson, M. Sex differences in upper respiratory symptoms prevalence and oral-respiratory mucosal immunity in endurance athletes. Exerc. Immunol. Rev. 2014, 20, 8–22. [Google Scholar]
- Mccabe, L.R.; Irwin, R.; Schaefer, L.; Britton, R.A. Probiotic use decreases intestinal inflammation and increases bone density in healthy male but not female mice. J. Cell. Physiol. 2013, 228, 1793–1798. [Google Scholar] [CrossRef]
- Floch, M.H. Gender in probiotic use. J. Clin. Gastroenterol. 2015, 49, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Frei, R.; Akdis, M.; O’Mahony, L. Prebiotics, probiotics, synbiotics, and the immune system: Experimental data and clinical evidence. Curr. Opin. Gastroenterol. 2015, 31, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Jirillo, E.; Jirillo, F.; Magrone, T. Healthy effects exerted by prebiotics, probiotics, and symbiotics with special reference to their impact on the immune system. Int. J. Vitam. Nutr. Res. 2012, 82, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.H.; Huang, Y.T.; Lu, Y.C.; Lee, S.Y.; Tsai, M.F.; Hung, S.P.; Hsu, T.Y. Effects of gender and age on immune responses of human peripheral blood mononuclear cells to probiotics: A large scale pilot study. J. Nutr. Health Aging 2017, 21, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Stilling, R.M.; Kennedy, P.J.; Stanton, C.; Cryan, J.F.; Dinan, T.G. Minireview: Gut microbiota: The neglected endocrine organ. Mol. Endocrinol. 2014, 28, 1221–1238. [Google Scholar] [CrossRef]
- Meeusen, R. Exercise, nutrition and the brain. Sport Med. 2014, 44, S47–S56. [Google Scholar] [CrossRef]
 low risk of bias.
low risk of bias.  unknown risk of bias.
unknown risk of bias.  high risk of bias.
high risk of bias.
 low risk of bias.
low risk of bias.  unknown risk of bias.
unknown risk of bias.  high risk of bias.
high risk of bias.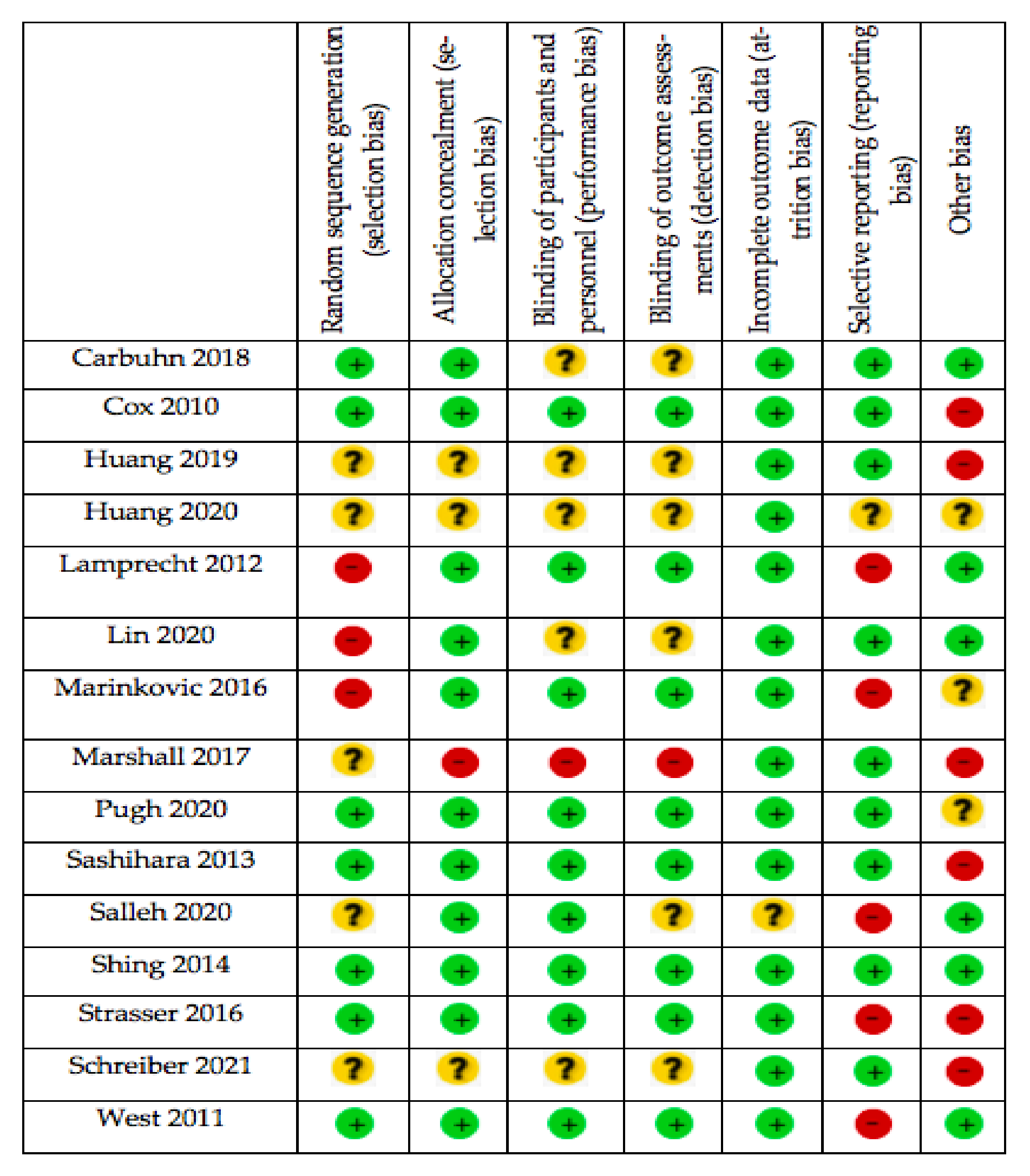

| Inclusion Criteria | Exclusion Criteria | |
|---|---|---|
| P (Population): “athletes and/or Division I and/or trained population (≥8 h/week and/or ≥5 workouts/week)” | Participants had to be athletes and/or Division I and/or trained population (≥8 h/week and/or ≥5 workouts/week clinical trial | Participants had previous health problems or injuries leading to drug intake |
| I (Intervention) “effects of probiotic supplementation on test with predominance on aerobic metabolism” | Clear information concerning supplementation administration | Unclear information concerning probiotic supplementation |
| C (Comparators) “similar experimental conditions in the placebo or control group compared with the probiotic group” | - | - |
| O (Outcome) “performance test with aerobic metabolism dominance” | Used test in which aerobic metabolism is primary | - |
| S (Study design): “double-blind controlled clinical trial” | Well-designed experiment, a clinical trial, peer-reviewed and original articles written in the English Language; and clear information about funding sources | - |
| Study | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | TOTAL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbuhn et al., (2018) [35] | Yes | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 6 |
| Cox et al., (2010) [28] | Yes | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 9 |
| Huang et al., (2019) [29] | Yes | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 5 |
| Huang et al., (2020) [30] | Yes | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 5 |
| Lamprecht et al., (2012) [5] | Yes | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 5 |
| Lin et al., (2020)[36] | Yes | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 5 |
| Marinkovic et al., (2016) [31] | Yes | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 4 |
| Marshall et al., (2017) [37] | Yes | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 6 |
| Pugh et al., (2020) [25] | Yes | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Salleh et al., (2021) [32] | Yes | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 9 |
| Sashihara et al., (2013) [33] | Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 9 |
| Schreiber et al., (2021) [40] | Yes | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 3 |
| Shing et al., (2014) [38] | Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 |
| Strasser et al., (2016) [39] | Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 9 |
| West et al., (2011) [34] | Yes | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 8 |
| Author/s | Population | Supplementation protocol | Duration | Training protocol | Test | Outcomes | Effect |
|---|---|---|---|---|---|---|---|
| Carbuhn et al., (2018) [35] | 17 female swimmers from Division I | 1 × 109 CFU of Bifidobacterium longum 35624 daily (1 capsule per day) | 42 days | 8–20 h/week, 5 times a week | -500 m freestyle aerobic swim test | -Time trial (s) | - ↔ |
| Cox et al., (2010) [28] | 20 highly trained distance male runners (27.3 ± 6.4 years) | 1.2 × 1010 CFU of Lactobacillus fermentum VRI-003 PCC daily (3 capsules twice a day) | 28 days | 8.2 ± 2.8 h/week endurance training | -Treadmill running test | -Treadmill time (min) | - ↔ |
| -VO2max (mL/kg/min) | - ↔ | ||||||
| Huang et al., (2019) [29] | 16 triathletes (Ni) (EG: 22.3 ± 1.2 years; PLA: 20.1 ± 0.3 years) | 3 × 1010 CFU of Lactobacillus plantarum PS128 daily (1 capsule twice a day) | 21 days | Specialized training | -VO2max endurance cycling test (48 h after a triathlon championship) | -Time trial (s) | - ↑ |
| Huang et al., (2020) [30] | 20 male triathletes (EG: 21.6 ± 1.3 years; PLA: 21.9 ± 1.4 years) | 3 × 1010 CFU of Lactobacillus plantarum PS128 daily (1 capsule twice a day) | 28 days | Usual training | -Treadmill running test | -Treadmill time (s) | - ↑ |
| -VO2max (mL/kg/min) | - ↔ | ||||||
| Lin et al., (2020)[36] | 21 (14 males and 7 females, aged 20–30 years) well-trained runners | 1.5 × 1010 CFU of OLP-01, a human strain probiotic derived the Bifidobacterium longum subsp. Longum (3 capsules per day) | 35 days | Usual training | -12-min running/walking distance | -Distance (m) | - ↑ |
| Marinkovic et al., (2016) [31] | 39 male and females’ elite athletes (EG: 23.5 ± 2.7 years; PLA: 22.8 ± 2.5 years) | 2 × 1010 CFU of Lactobacillus helveticus Lafti® L10 daily (capsules) | 98 days | >11 h/week | -Graded cardiopulmonary test (treadmill) | -VO2max (mL/kg/min) | - ↔ |
| -Time (min) | - ↔ | ||||||
| Salleh et al., (2021) [32] | 30 males badminton players (18–30 years) | 3 × 1010 CFU of Lactobacillus casei daily (commercial probiotic drink) mixed with commercial orange juice (in total 200 mL) | 42 days | Usual training | -20 m multi-stage shuttle run | -VO2max (mL/kg/min) | - ↑ |
| Sashihara et al., (2013) [33] | 29 male soccer players (EG: 19.8 ± 0.9 years; PLA: 20.2 ± 1.1 years) | 3 × 1010 CFU of heat-killed cells of Lactobacillus gasseri OLL2809 daily (2 tablets 3 times a day) | 28 days | Minimum of 5 days/week high intensity training | -Cycle ergometer exercise (1 h at 70% of heart rate reserve) | -Workload (kW/h) | - ↔ |
| West et al., (2011) [34] | 88 male and female cyclists (EG: 35.2 ± 10.3 years; PLA: 36.4 ± 8.9 years) | 1 × 109 CFU of Lactobacillus fermentum VRI-003 PCC® daily (1 capsule per day) | 77 days | Usual training | -Incremental performance test (cycle ergometer) | -VO2max (mL/kg/min) | - ↔ |
| Author/s | Population | Supplementation Protocol | Duration | Training Protocol | Test | Outcomes | Effect |
|---|---|---|---|---|---|---|---|
| Lamprecht et al., (2012) [5] | 23 endurance trained men (EG: 37.6 ± 4.7 years; PLA: 38.2 ± 4.4 years) | 1010 CFU of Bifidobacterium lactis W51, Bifidobacterium bifidum W23, Enterococcus faecium W54, Lactobacillus brevis W63, Lactobacillus acidophilus W22 and Lactococcus lactis W58 daily (2 powder sachets twice a day) | 98 days | Usual training | -Triple cycle step test ergometry | -VO2max (mL/kg/min) | - ↔ |
| -Performance (W/kg) | - ↔ | ||||||
| Marshall et al., (2017) [37] | 22 male and female marathon runners (EG: 25–50 years; PLA: 23–60 years) | 1 × 109 CFU of Lactobacillus acidophilus CUL-60 [NCIMB 30157], 9.5 × 109 of Bifidobacterium bifidum CUL-20 [NCIMB 30172], 1 × 109 CFU of Lactobacillus acidophilus CUL-61 [NCIMB 30156], 0.5 × 109 of Bifidobacterium animalis subspecies lactis CUL-34 [NCIMB 30153] and 55.8 mg. d-1 fructooligosaccharides daily (1 capsule per day) | 84 days | Not reported | -Graded exercise test to exhaustion (treadmill) | -VO2max (mL/kg/min) | - ↔ |
| -Marathon des Sables | -Time to completion (min) | - ↔ | |||||
| Pugh et al., (2020) [25] | 7 male trained cyclists (23 ± 4 years) | 2.5 × 1010 CFU of Lactobacillus acidophilus (CUL60), Bifidobacterium bifidum (CUL20), Lactobacillus acidophilus (CUL21), and Bifidobacterium animalis subsp. Lactis (CUL34) daily (1 capsule per day) | 28 days | Usual training | -120 min of cycling at 55% Wmax | -VO2max (mL/kg/min) | - ↔ |
| Schreiber et al., (2021) [40] | 27 male elite cyclists (19–49 years) | 15 × 109 CFU of (≥) ≥4.3 × 109 CFU Bifidobacterium animalis ssp. lactis Lafti B94 (28.6 %), ≥4.3 × 109 CFU Lactobacillus helveticus Lafti L10 (28.6 %), ≥2.1 × 109 CFU Bifidobacterium longum R0175 (14.3 %), ≥3.9 × 109 CFU Enterococcus faecium R0026 (25.7 %), and ≥0.4 × 109 CFU Bacillus subtilis R0179 (2.8 %) (1 capsule per day) | 90 days | Usual training | -Time to fatigue (85% maximal power) | -Time to fatigue (min:s) | - ↔ |
| -Graded exercise test to exhaustion (cycle ergometer) | -VO2max (mL/kg/min) | - ↔ | |||||
| Shing et al., (2014) [38] | 10 male runners (27 ± 2 years) | 4.5 × 1010 CFU of Lactobacillus rhamnosus, Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus fermentum, Lactobacillus plantarum, Bifidobacterium bifidum, Bifidobacterium breve, Bifidobacterium lactis, and Streptococcus thermophilus daily (1 capsule per day) | 28 days | Not reported | -Time-to-fatigue run at 80 % of ventilatory threshold (treadmill) | -Time to fatigue (s) | - ↑ |
| Strasser et al., (2016) [39] | 29 male and female athletes (EG: 25.7 ± 3.5 years; PLA: 26.6 ± 3.5 years) | 1 × 1010 CFU of Bifidobacterium lactis W51, Bifidobacterium bifidum W23, Lactobacillus acidophilus W22, Lactobacillus Brevis W63 and Lactococcus lactis W58 daily Enterococcus faecium W54 (1 sachet per day) | 84 days | Usual training | -Exercise test untilexhaustion (cycle ergometer) | -VO2max (mL/kg/min) | - ↔ |
| Study | Study Design | GRADE Approach | Food Record | Prohibited Foods and Supplements |
|---|---|---|---|---|
| Carbuhn et al., (2018) [35] | RD-BP-C | High | 3 days dietary food record | Nutritional supplements Ergogenic supplements Antibiotics and anti-inflammatories |
| Cox et al., (2010) [28] | RD-BP-CC | High | - | Yoghurt Yoghurt based products |
| Huang et al., (2019) [29] | D-B | High | - | Fermented food products Probiotics Prebiotics Vitamins Materials and herbal extracts Antibiotics |
| Huang et al., (2020) [30] | D-BP | High | Dietary record (undefined). | Fermented food Probiotics Prebiotics Alcohol and smoking Antibiotics |
| Lamprecht et al., (2012) [5] | RD-BP-C | High | 7 days food record | - |
| Lin et al., (2020)[36] | D-B | High | - | - |
| Marinkovic et al., (2016) [31] | RD-BP-CP | High | - | Yoghurt Fermented milk products Supplements for enhancing the immune system |
| Marshall et al., (2017) [37] | RIM | Moderate | - | Any other supplements |
| Pugh et al., (2020) [25] | RD-BP-CC | High | 24 h food record | Probiotic foods Alcohol Spicy food Caffeine |
| Salleh et al., (2021) [32] | RP-C | High | 3 days dietary record | Other additional probiotic supplements |
| Sashihara et al., (2013) [33] | RD-BP-CP | High | - | - |
| Schreiber et al., (2021) [40] | RD-BP-C | High | Liquid or solid food consumed | Probiotcs supplements Ergogenic supplements Antibiotics and medications |
| Shing et al., (2014) [38] | RD-BP-CC | High | - | Probiotic supplements Antibiotics and non-steroidal anti-inflammatory drugs |
| Strasser et al., (2016) [39] | RD-BP-C | High | 3 days food record | Fermented dairy products Probiotics Dietary supplements Minerals Vitamins Alcohol Medicines |
| West et al., (2011) [34] | RD-BP-CP | High | 4 days food record | Probiotic enriched yoghurt Supplements/foods containing probiotics Foods or supplements fortify with prebiotics Antibiotics |
| Subgroups | SMD | 95% CI | p Value |
|---|---|---|---|
| Dose (CFU) | |||
| <30 × 109 (n = 6) | 0.20 | −0.05 to 0.45 | 0.12 |
| ≥30 × 109 (n = 6) | 0.47 | 0.04 to 0.89 | <0.05 * |
| Duration | |||
| ≤4 weeks (n = 6) | 0.44 | 0.05 to 0.84 | <0.05 * |
| >4 weeks (n = 6) | 0.19 | −0.08 to 0.47 | 0.16 |
| Strain | |||
| Multi strain (n = 5) | 0.26 | −0.08 to 0.60 | 0.14 |
| Single strain (n = 7) | 0.33 | 0.06 to 0.60 | <0.05 * |
| Sex | |||
| Males (n = 8) | 0.30 | 0.04 to 0.56 | <0.05 * |
| Females + mix (males + females) (n = 3) | 0.30 | −0.19 to 0.79 | 0.23 |
| Test | |||
| To fatigue (n = 7) | 0.45 | 0.03 to 0.86 | <0.05 * |
| VO2max (n = 7) | 0.21 | −0.11 to 0.52 | 0.21 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santibañez-Gutierrez, A.; Fernández-Landa, J.; Calleja-González, J.; Delextrat, A.; Mielgo-Ayuso, J. Effects of Probiotic Supplementation on Exercise with Predominance of Aerobic Metabolism in Trained Population: A Systematic Review, Meta-Analysis and Meta-Regression. Nutrients 2022, 14, 622. https://doi.org/10.3390/nu14030622
Santibañez-Gutierrez A, Fernández-Landa J, Calleja-González J, Delextrat A, Mielgo-Ayuso J. Effects of Probiotic Supplementation on Exercise with Predominance of Aerobic Metabolism in Trained Population: A Systematic Review, Meta-Analysis and Meta-Regression. Nutrients. 2022; 14(3):622. https://doi.org/10.3390/nu14030622
Chicago/Turabian StyleSantibañez-Gutierrez, Asier, Julen Fernández-Landa, Julio Calleja-González, Anne Delextrat, and Juan Mielgo-Ayuso. 2022. "Effects of Probiotic Supplementation on Exercise with Predominance of Aerobic Metabolism in Trained Population: A Systematic Review, Meta-Analysis and Meta-Regression" Nutrients 14, no. 3: 622. https://doi.org/10.3390/nu14030622
APA StyleSantibañez-Gutierrez, A., Fernández-Landa, J., Calleja-González, J., Delextrat, A., & Mielgo-Ayuso, J. (2022). Effects of Probiotic Supplementation on Exercise with Predominance of Aerobic Metabolism in Trained Population: A Systematic Review, Meta-Analysis and Meta-Regression. Nutrients, 14(3), 622. https://doi.org/10.3390/nu14030622









