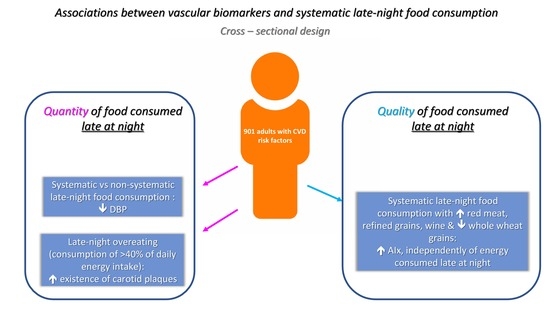
Late-Night Overeating or Low-Quality Food Choices Late at Night Are Associated with Subclinical Vascular Damage in Patients at Increased Cardiovascular Risk
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Study
2.2. Study Population
2.3. Definition of CVD Risk Factors
2.4. Anthropometric Measurements
2.5. Vascular Assessment
2.6. Dietary Intake Assessment and Definition of Late-Night Overeating
2.7. Statistical Analysis
2.7.1. Analyses Based on Food Quantity after 19:00 h
- Model 1: adjusted for age and sex;
- Model 2: adjusted for age, sex, hypertension, diabetes mellitus, dyslipidemia, smoking and BMI;
- Model 3: adjusted for age, sex, hypertension, diabetes mellitus, dyslipidemia, smoking, BMI and dTEI.
2.7.2. Analyses Based on Food Quality after 19:00 h
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gallant, A.; Lundgren, J.; Drapeau, V. Nutritional Aspects of Late Eating and Night Eating. Curr. Obes. Rep. 2014, 3, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kinsey, A.W.; Ormsbee, M.J. The health impact of nighttime eating: Old and new perspectives. Nutrients 2015, 7, 2648–2662. [Google Scholar] [CrossRef] [PubMed]
- Fong, M.; Caterson, I.D.; Madigan, C.D. Are large dinners associated with excess weight, and does eating a smaller dinner achieve greater weight loss? A systematic review and meta-analysis. Br. J. Nutr. 2017, 118, 616–628. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Ard, J.; Baskin, M.L.; Chiuve, S.E.; Johnson, H.M.; Kris-Etherton, P.; Varady, K. Meal Timing and Frequency: Implications for Cardiovascular Disease Prevention: A Scientific Statement from the American Heart Association. Circulation 2017, 135, e96–e121. [Google Scholar] [CrossRef]
- McHill, A.W.; Phillips, A.J.; Czeisler, C.A.; Keating, L.; Yee, K.; Barger, L.K.; Garaulet, M.; Scheer, F.J.L.; Klerman, E.B. Later circadian timing of food intake is associated with increased body fat. Am. J. Clin. Nutr. 2017, 106, 1213–1219. [Google Scholar] [CrossRef]
- Hashemipour, S.; Yazdi, Z.; Mahabad, N. Association of Evening Chronotype with Poor Control of Type 2 Diabetes: Roles of Sleep Duration and Insomnia Level. Int. J. Endocrinol. Metab. 2020, 18, e99701. [Google Scholar] [CrossRef]
- BaHammam, A.S.; Almeneessier, A.S. Recent Evidence on the Impact of Ramadan Diurnal Intermittent Fasting, Mealtime, and Circadian Rhythm on Cardiometabolic Risk: A Review. Front. Nutr. 2020, 7, 28. [Google Scholar] [CrossRef] [Green Version]
- Osman, F.; Haldar, S.; Henry, C.J. Effects of Time-Restricted Feeding during Ramadan on Dietary Intake, Body Composition and Metabolic Outcomes. Nutrients 2020, 12, 2478. [Google Scholar] [CrossRef]
- Vlachopoulos, C.; Xaplanteris, P.; Aboyans, V.; Brodmann, M.; Cifkova, R.; Cosentino, F.; De Carlo, M.; Gallino, A.; Landmesser, U.; Laurent, S.; et al. The role of vascular biomarkers for primary and secondary prevention. A position paper from the European Society of Cardiology Working Group on peripheral circulation: Endorsed by the Association for Research into Arterial Structure and Physiology (ARTERY) Society. Atherosclerosis 2015, 241, 507–532. [Google Scholar]
- Vlachopoulos, C.; Aznaouridis, K.; O’Rourke, M.F.; Safar, M.E.; Baou, K.; Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: A systematic review and meta-analysis. Eur. Heart J. 2010, 31, 1865–1871. [Google Scholar] [CrossRef] [Green Version]
- Nambi, V.; Chambless, L.; He, M.; Folsom, A.R.; Mosley, T.; Boerwinkle, E.; Ballantyne, C.M. Common carotid artery intima-media thickness is as good as carotid intima-media thickness of all carotid artery segments in improving prediction of coronary heart disease risk in the Atherosclerosis Risk in Communities (ARIC) study. Eur. Heart J. 2012, 33, 183–190. [Google Scholar] [CrossRef] [Green Version]
- Jakubowicz, D.; Barnea, M.; Wainstein, J.; Froy, O. High caloric intake at breakfast vs. dinner differentially influences weight loss of overweight and obese women. Obesity 2013, 21, 2504–2512. [Google Scholar] [CrossRef]
- Almoosawi, S.; Vingeliene, S.; Karagounis, L.G.; Pot, G.K. Chrono-nutrition: A review of current evidence from observational studies on global trends in time-of-day of energy intake and its association with obesity. Proc. Nutr. Soc. 2016, 75, 487–500. [Google Scholar] [CrossRef] [Green Version]
- Marinac, C.R.; Sears, D.D.; Natarajan, L.; Gallo, L.C.; Breen, C.I.; Patterson, R.E. Frequency and Circadian Timing of Eating May Influence Biomarkers of Inflammation and Insulin Resistance Associated with Breast Cancer Risk. PLoS ONE 2015, 10, e0136240. [Google Scholar] [CrossRef] [Green Version]
- Stamatelopoulos, K.; Karatzi, K.; Sidossis, L.S. Noninvasive methods for assessing early markers of atherosclerosis: The role of body composition and nutrition. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 467–473. [Google Scholar] [CrossRef]
- Ferreira, I.; Huijberts, M.S. Eating at the right time of day: An underappreciated lifestyle therapy for hypertension? J. Hypertens. 2013, 31, 866–869. [Google Scholar] [CrossRef]
- Strasser, B.; Arvandi, M.; Pasha, E.P.; Haley, A.P.; Stanforth, P.; Tanaka, H. Abdominal obesity is associated with arterial stiffness in middle-aged adults. Nutr. Metab. Cardiovasc. Dis. NMCD 2015, 25, 495–502. [Google Scholar] [CrossRef]
- Torres, N.; Guevara-Cruz, M.; Velazquez-Villegas, L.A.; Tovar, A.R. Nutrition and Atherosclerosis. Arch. Med. Res. 2015, 46, 408–426. [Google Scholar] [CrossRef]
- Madonna, R.; Selvaggio, S.; Selvaggio, G.; Coronelli, M.; Cocco, N. “State-of-Art” paper of the Italian Working Group on Atherosclerosis: Preclinical assessment of early coronary atherosclerosis. Int. J. Cardiol. 2016, 214, 442–447. [Google Scholar] [CrossRef]
- Shirwany, N.A.; Zou, M.H. Arterial stiffness: A brief review. Acta Pharmacol. Sin. 2010, 31, 1267–1276. [Google Scholar] [CrossRef] [Green Version]
- Sacre, J.W.; Jennings, G.L.; Kingwell, B.A. Exercise and dietary influences on arterial stiffness in cardiometabolic disease. Hypertension 2014, 63, 888–893. [Google Scholar] [CrossRef] [Green Version]
- Tsirimiagkou, C.; Karatzi, K.; Argyris, A.; Chalkidou, F.; Tzelefa, V.; Sfikakis, P.P.; Yannakoulia, M.; Protogerou, A.D. Levels of dietary sodium intake: Diverging associations with arterial stiffness and atheromatosis. Hell. J. Cardiol. 2021, 62, 439–446. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, Y.; Na, M.; Lichtenstein, A.H.; Xing, A.; Chen, S.; Wu, S.; Xiang, G. Habitual Night Eating Was Positively Associated With Progress of Arterial Stiffness in Chinese Adults. J. Am. Heart Assoc. 2020, 9, e016455. [Google Scholar] [CrossRef]
- Roman, M.J.; Moeller, E.; Davis, A.; Paget, S.A.; Crow, M.K.; Lockshin, M.D.; Sammaritano, L.; Devereux, R.B.; Schwartz, J.E.; Levine, D.M.; et al. Preclinical carotid atherosclerosis in patients with rheumatoid arthritis. Ann. Intern. Med. 2006, 144, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Reid, K.J.; Baron, K.G.; Zee, P.C. Meal timing influences daily caloric intake in healthy adults. Nutr. Res. 2014, 34, 930–935. [Google Scholar] [CrossRef] [Green Version]
- Kakamu, T.; Hidaka, T.; Kumagai, T.; Masuishi, Y.; Kasuga, H.; Endo, S.; Sato, S.; Takeda, A.; Koizumi, M.; Fukushima, T. Unhealthy changes in eating habits cause acute onset hypertension in the normotensive community-dwelling elderly-3 years cohort study. Medicine 2019, 98, e15071. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Akbari, F.; Qorbani, M.; Esmaeil Motlagh, M.; Ardalan, G.; Heshmat, R.; Daneshzad, E.; Kelishadi, R. Dinner consumption and cardiovascular disease risk factors among a nationally representative sample of Iranian adolescents: The CASPIAN-III Study. J. Cardiovasc. Thorac. Res. 2019, 11, 138–146. [Google Scholar] [CrossRef]
- Benetos, A.; Waeber, B.; Izzo, J.; Mitchell, G.; Resnick, L.; Asmar, R.; Safar, M. Influence of age, risk factors, and cardiovascular and renal disease on arterial stiffness: Clinical applications. Am. J. Hypertens. 2002, 15, 1101–1108. [Google Scholar] [CrossRef]
- Peng, S.; Wang, J.; Xiao, Y.; Yin, L.; Peng, Y.; Yang, L.; Yang, P.; Wang, Y.; Cao, X.; Li, X.; et al. The association of carotid artery atherosclerosis with the estimated excretion levels of urinary sodium and potassium and their ratio in Chinese adults. Nutr. J. 2021, 20, 50. [Google Scholar] [CrossRef]
- Lazaros, G.; Oikonomou, E.; Vogiatzi, G.; Christoforatou, E.; Tsalamandris, S.; Goliopoulou, A.; Tousouli, M.; Mystakidou, V.; Chasikidis, C.; Tousoulis, D. The impact of sedentary behavior patterns on carotid atherosclerotic burden: Implications from the Corinthia epidemiological study. Atherosclerosis 2019, 282, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Tisdel, D.M.; Gadberry, J.J.; Burke, S.L.; Carlini, N.A.; Fleenor, B.S.; Campbell, M.S. Dietary fat and alcohol in the prediction of indices of vascular health among young adults. Nutrition 2021, 84, 111120. [Google Scholar] [CrossRef]
- Recio-Rodriguez, J.I.; Gomez-Marcos, M.A.; Patino-Alonso, M.C.; Rodrigo-De Pablo, E.; Cabrejas-Sanchez, A.; Arietaleanizbeaskoa, M.S.; Repiso-Gento, I.; Gonzalez-Viejo, N.; Maderuelo-Fernandez, J.-A.; Agudo-Conde, C.; et al. Glycemic index, glycemic load, and pulse wave reflection in adults. Nutr. Metab. Cardiovasc. Dis. NMCD 2015, 25, 68–74. [Google Scholar] [CrossRef]
| Non-Systematic Late-Night Food Consumers (n = 178) | Systematic Late-Night Food Consumers (n = 723) | p-Value | Systematic Late-Night Eaters (Consumption of <40% of dTEI) (n = 572) | Systematic Late-Night Overeaters (Consumption of >40% of dTEI) (n = 151) | p-Value | |
|---|---|---|---|---|---|---|
| Age (years) | 53.36 ± 13.64 | 52.16 ± 13.85 | 0.300 | 53.05 ± 13.92 | 48.79 ± 13.10 | 0.001 |
| Sex (%) | ||||||
| Male | 48.9 | 44.3 | 0.268 | 39.9 | 60.9 | 0.000 |
| Female | 51.1 | 55.7 | 60.1 | 39.1 | ||
| BMI (Kg/m2) | 28.12 ± 5.56 | 27.93 ± 5.52 | 0.681 | 27.83 ± 5.34 | 28.31 ± 6.15 | 0.344 |
| Smoking (%) | ||||||
| Never smokers | 42.1 | 39.1 | 0.626 | 40.9 | 32.5 | 0.104 |
| Former smokers | 32.6 | 36.4 | 34.6 | 43.0 | ||
| Current smokers | 25.3 | 24.5 | 24.5 | 24.5 | ||
| Biochemical biomarkers | ||||||
| Fasting blood glucose (mg/dL) | 96.62 ± 18.89 | 99.37 ± 32.45 | 0.306 | 99.07 ± 32.70 | 100.53 ± 31.57 | 0.670 |
| Total cholesterol (mg/dL) | 194.10 ± 37.10 | 196.15 ± 36.83 | 0.534 | 195.32 ± 35.19 | 199.37 ± 42.62 | 0.296 |
| LDL-C (mg/dL) | 116.53 ± 28.96 | 118.69 ± 32.15 | 0.451 | 117.04 ± 31.34 | 124.95 ± 34.53 | 0.019 |
| HDL-C (mg/dL) | 56.65 ± 21.11 | 56.18 ± 15.82 | 0.760 | 57.34 ± 15.68 | 51.76 ± 15.66 | 0.001 |
| TG (mg/dL) | 110.99 ± 56.38 | 107.71 ± 66.59 | 0.570 | 105.99 ± 56.31 | 114.35 ± 96.58 | 0.377 |
| Diabetes mellitus (%) | ||||||
| Type 1 diabetes mellitus | 3.4 | 9.0 | 0.040 | 78.4 | 11.3 | 0.351 |
| Type 2 diabetes mellitus | 11.2 | 11.8 | 12.4 | 9.3 | ||
| Hypertension (%) | ||||||
| Yes | 58.9 | 48.6 | 0.022 | 51.2 | 40.2 | 0.037 |
| Dyslipidemia (%) | ||||||
| Yes | 39.3 | 36.2 | 0.444 | 36.2 | 36.4 | 0.957 |
| Subclinical vascular biomarkers | ||||||
| Peripheral SBP (mmHg) | 127.58 ± 15.66 | 124.68 ± 15.84 | 0.029 | 125.12 ± 15.98 | 123.03 ± 15.24 | 0.150 |
| Central SBP (mmHg) | 117.23 ± 15.27 | 114.188 ± 16.59 | 0.027 | 114.49 ± 16.98 | 113.06 ± 15.03 | 0.346 |
| DBP (mmHg) | 77.88 ± 9.77 | 75.50 ± 8.97 | 0.002 | 75.42 ± 8.87 | 75.82 ± 9.37 | 0.631 |
| PWV (m/s) | 8.39 ± 1.78 | 8.27 ± 2.00 | 0.491 | 8.36 ± 2.07 | 7.94 ± 1.69 | 0.023 |
| AIx@75 (%) | 26.76 ± 11.85 | 25.78 ± 13.16 | 0.367 | 26.61 ± 12.96 | 22.63 ± 13.46 | 0.001 |
| right IMT (mm) | 0.68 ± 0.15 | 0.68 ± 0.15 | 0.789 | 0.68 ± 0.15 | 0.67 ± 0.15 | 0.252 |
| left IMT (mm) | 0.73 ± 0.17 | 0.71 ± 0.16 | 0.182 | 0.72 ± 0.17 | 0.70 ± 0.16 | 0.253 |
| Existence of plaques (%) | ||||||
| Yes | 57.9 | 48.1 | 0.020 | 47.7 | 49.7 | 0.671 |
| Existence of carotid plaques (%) | ||||||
| Yes | 42.7 | 36.4 | 0.119 | 35.7 | 39.1 | 0.439 |
| Existence of femoral plaques (%) | ||||||
| Yes | 43.3 | 35.1 | 0.044 | 34.8 | 36.4 | 0.708 |
| Non-Systematic Late-Night Food Consumers (n = 178) | Systematic Late-Night Food Consumers (n = 723) | p-Value | Systematic Late-Night Eaters (n = 572) | Systematic Late-Night Overeaters (n = 151) | p-Value | |
|---|---|---|---|---|---|---|
| Total energy consumption after 19:00 (Kcal) | 415.75 ± 340.63 | 523.00 ± 375.74 | 0.001 | 405.31 ± 227.45 | 968.84 ± 478.90 | 0.000 |
| Energy from CHO after 19:00 (%) | 41.77 ± 9.89 | 42.33 ± 9.69 | 0.490 | 42.71 ± 9.36 | 40.91 ± 10.77 | 0.043 |
| Energy from PRO after 19:00 (%) | 16.51 ± 4.96 | 17.30 ± 4.96 | 0.058 | 17.35 ± 5.06 | 17.08 ± 4.59 | 0.549 |
| Energy from FAT after 19:00 (%) | 40.68 ± 10.36 | 39.64 ± 8.78 | 0.174 | 39.78 ± 8.74 | 39.12 ± 8.95 | 0.414 |
| dTEI (Kcal) | 1767.79 ± 742.20 | 1694.95 ± 608.26 | 0.172 | 1644.39 ± 558.02 | 1886.45 ± 740.84 | 0.000 |
| Food groups | ||||||
| Low fat dairy products (250 mL milk or soy milk, 1 cup of yogurt) | 0.39 ± 0.62 | 0.37 ± 0.55 | 0.712 | 0.25 ± 0.40 | 0.20 ± 0.42 | 0.001 |
| Full fat dairy products (250 mL milk, 1 cup of yogurt) | 0.18 ± 0.36 | 0.19 ± 0.40 | 0.695 | 0.20 ± 0.42 | 0.15 ± 0.33 | 0.146 |
| Vegetables (e.g., 1 medium tomato or pepper, ½ cup of cabbage or peas or lettuce or spinach) | 2.21 ± 1.87 | 2.44 ± 1.88 | 0.139 | 2.44 ± 1.77 | 2.45 ± 2.25 | 0.933 |
| Legumes cooked and drained (½ cup) | 0.41 ± 0.83 | 0.26 ± 0.59 | 0.006 | 0.27 ± 0.60 | 0.21 ± 0.51 | 0.244 |
| Fruits (e.g., 1 medium apple or peach or orange or pear, 1 cup of strawberries or melon) and fresh juice (½ cup) | 1.62 ± 1.89 | 1.46 ± 1.45 | 0.228 | 1.53 ± 1.45 | 1.20 ± 1.46 | 0.014 |
| Processed juices (½ cup) | 0.06 ± 0.24 | 0.08 ± 0.34 | 0.512 | 0.08 ± 0.35 | 0.08 ± 0.32 | 0.950 |
| Refined grains (e.g., 30 g of white bread, ½ cup rice or pasta, 2 rusks) | 2.93 ±2.40 | 2.98 ± 2.35 | 0.810 | 2.80 ± 2.26 | 3.66 ± 2.57 | 0.000 |
| Whole grains (e.g., 30 g of whole wheat bread, ½ cup brown or wild rice or whole wheat pasta, 2 rusks) | 1.00 ± 1.45 | 1.07 ± 1.39 | 0.559 | 1.08 ± 1.40 | 1.01 ± 1.31 | 0.563 |
| Danishes (1 medium slice) | 0.13 ± 0.28 | 0.11 ± 0.29 | 0.545 | 0.11 ± 0.28 | 0.13 ± 0.32 | 0.462 |
| Low fat cheese (30 g) | 0.13 ± 0.34 | 0.23 ± 0.51 | 0.012 | 0.21 ± 0.45 | 0.31 ± 0.70 | 0.037 |
| Full fat cheese (30 g or 1 slice) | 0.78 ± 0.97 | 0.99 ± 1.15 | 0.027 | 0.96 ± 1.15 | 1.10 ± 1.15 | 0.185 |
| Dressings, full fat or light (1 tbs) | 0.12 ± 0.38 | 0.12 ± 0.40 | 0.994 | 0.11 ± 0.36 | 0.19 ± 0.50 | 0.023 |
| Light soft drinks (250 mL) | 0.03 ± 0.16 | 0.07 ± 0.29 | 0.057 | 0.07 ± 0.28 | 0.09 ± 0.36 | 0.337 |
| Soft drinks with sugar (250 mL) | 0.08 ± 0.26 | 0.07 ± 0.25 | 0.676 | 0.06 ± 0.23 | 0.11 ± 0.32 | 0.026 |
| Low fat sweets (e.g., 1 ice cream 0%, 1 cup of fruit gel) | 0.06 ± 0.32 | 0.07 ± 0.28 | 0.881 | 0.06 ± 0.25 | 0.09 ± 0.42 | 0.334 |
| Sweets rich in sugar and saturated fat (e.g., 1 slice of cake, 1 ice cream) | 0.65 ± 1.01 | 0.69 ± 0.94 | 0.639 | 0.71 ± 0.93 | 0.62 ± 0.98 | 0.344 |
| Sugar (1 tsp) | 0.94 ± 1.32 | 0.78 ± 1.27 | 0.137 | 0.76 ± 1.27 | 0.84 ± 1.27 | 0.462 |
| Sugar substitutes, honey, jam (1 tsp) | 0.37 ± 0.93 | 0.62 ± 1.10 | 0.005 | 0.65 ± 1.13 | 0.53 ± 0.97 | 0.238 |
| Snacks (1 cup of chips, crackers or popcorn) | 0.05 ± 0.22 | 0.04 ± 0.24 | 0.787 | 0.04 ± 0.23 | 0.07 ± 0.26 | 0.131 |
| High alcohol drinks (e.g., 30 mL of gin, vodka, whiskey, rum) | 0.18 ± 0.74 | 0.14 ± 0.65 | 0.487 | 0.13 ± 0.69 | 0.17 ± 0.50 | 0.477 |
| A. Systematic vs. Non-Systematic Consumption of Food after 19:00 h | B. Late-Night Food Consumption of >40% of dTEI vs. Late-Night Eating | |||||
|---|---|---|---|---|---|---|
| Model 1 | Model 2 | Model 3 | Model 1 | Model 2 | Model 3 | |
| B (95% CI) | B (95% CI) | B (95% CI) | B (95% CI) | B (95% CI) | B (95% CI) | |
| Peripheral SBP | −2.20 (−4.61, 0.21) | −1.22 (−3.27, 0.83) | −1.15 (−3.20, 0.90) | −1.23 (−3.92, 1.47) | −1.66 (−3.96, 0.64) | −1.83 (−4.14, 0.48) |
| Central SBP | −2.29 (−4.68, 0.12) | −1.57 (−3.73, 0.59) | −1.52 (−3.69, 0.64) | 0.81 (−1.89, 3.51) | 0.40 (−2.04, 2.83) | 0.32 (−2.13, 2.77) |
| DBP | −2.03 (−3.47, −0.59) | −1.45 (−2.76, −0.13) | −1.44 (−2.76, −0.12) | −0.06 (−1.64, 1.52) | −0.25 (−1.70, 1.20) | −0.25 (−1.71, 1.20) |
| AIx@75 | −0.83 (−2.48, 0.81) | −0.65 (−2.28, 0.98) | −0.75 (−2.37, 0.88) | 0.19 (−1.64, 2.02) | −0.04 (−1.85, 1.78) | 0.19 (−1.63, 2.01) |
| PWV | 0.08 (−0.18, 0.35) | 0.09 (−0.15, 0.33) | 0.09 (−0.15, 0.33) | −0.17 (−0.47, 0.13) | −0.16 (−0.44, 0.11) | −0.16 (−0.43, 0.12) |
| right IMT | 0.00 (−0.02, 0.02) | 0.00 (−0.02, 0.02) | 0.00 (−0.02, 0.02) | 0.01 (−0.01, 0.03) | 0.01 (−0.01, 0.03) | 0.01 (−0.01, 0,03) |
| left IMT | −0.01 (−0.03, 0.01) | −0.01 (−0.03, 0.01) | −0.01 (−0.03, 0.01) | 0.01 (−0.01, 0.04) | 0.01 (−0.02, 0.03) | 0.01 (−0.02/0.03) |
| Exp(B) (95%CI) | Exp(B) (95%CI) | Exp(B) (95%CI) | Exp(B) (95%CI) | Exp(B) (95%CI) | Exp(B) (95%CI) | |
| Existence of total plaques | 0.72 (0.48, 1.07) | 0.71 (0.46, 1.09) | 0.68 (0.45, 1.05) | 1.40 (0.90, 2.17) | 1.27 (0.79, 2.04) | 3.14 (0.83, 2.16) |
| Existence of carotid plaques | 0.82 (0.56, 1.20) | 0.84 (0.57, 1.24) | 0.81 (0.54, 1.19) | 1.69 (1.09/2.61) | 1.61 (1.02, 2.53) | 1.70 (1.07, 2.68) |
| Existence of femoral plaques | 0.78 (0.53, 1.15) | 0.74 (0.48, 1.13) | 0.73 (0.47, 1.12) | 1.28 (0.82, 1.99) | 1.09 (0.67, 1.78) | 1.11 (0.68, 1.80) |
| Food Groups | Dietary Pattern 1 | Dietary Pattern 2 | Dietary Pattern 3 |
|---|---|---|---|
| Consumption of olive oil | 0.770 | ||
| Consumption of vegetables | 0.740 | ||
| Consumption of fish | 0.553 | ||
| Consumption of cold cuts | 0.686 | ||
| Consumption of full-fat cheese | 0.676 | ||
| Consumption of fruits and juices | −0.338 | ||
| Consumption of sweets rich in sugar and saturated fat | 0.306 | ||
| Consumption of red meat | 0.658 | ||
| Consumption of refined grains | 0.558 | ||
| Consumption of whole wheat grains | −0.530 | ||
| Consumption of wine | 0.407 | ||
| Explained variance % | 15.661 | 13.484 | 12.425 |
| Dietary Pattern 1 | Dietary Pattern 2 | Dietary Pattern 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Model 1 B (95% CI) p-Value | Model 2 B (95% CI) p-Value | Model 3 B (95% CI) p-Value | Model 1 B (95% CI) p-Value | Model 2 B (95% CI) p-Value | Model 3 B (95% CI) p-Value | Model 1 B (95% CI) p-Value | Model 2 B (95% CI) p-Value | Model 3 B (95% CI) p-Value | |
| Peripheral SBP | −0.55 (−1.62, 0.53) 0.320 | −0.23 (−1.16, 0.69) 0.618 | −0.34 (−1.27, 0.60) 0.480 | 0.15 (−0.94, 1.23) 0.790 | 0.40 (−0.53, 1.32) 0.400 | 0.23 (−0.75, 1.21) 0.645 | 0.04 (−1.06, 1.15) 0.938 | 0.31 (−0.63, 1.25) 0.512 | 0.20 (−0.76, 1.16) 0.684 |
| Central SBP | −0.42 (−1.50, 0.66) 0.447 | −0.23 (−1.21, 0.74) 0.641 | −0.29 (−1.27, 0.70) 0.570 | 0.08 (−1.00, 1.17) 0.879 | 0.32 (−0.66, 1.30) 0.523 | 0.25 (−0.79, 1.29) 0.638 | 0.26 (−0.84, 1.36) 0.645 | 0.51 (−0.48, 1.50) 0.313 | 0.47 (−0.55, 1.48) 0.365 |
| DBP | −0.12 (−0.75, 0.51) 0.706 | 0.04 (−0.54, 0.62) 0.888 | 0.04 (−0.54, 0.63) 0.886 | 0.21 (−0.42, 0.85) 0.513 | 0.31 (−0.27, 0.89) 0.294 | 0.35 (−0.27, 0.96) 0.267 | 0.03 (−0.62, 0.67) 0.939 | 0.12 (−0.47, 0.71) 0.690 | 0.13 (−0.48, 0.73) 0.683 |
| AIx@75 | −0.70 (−1.43, 0.03) 0.059 | −0.71 (−1.43, 0.02) 0.056 | −0.59 (−1.32, 0.15) 0.117 | −0.28 (−1.02,0.46) 0.458 | −0.21 (−0.94, 0.52) 0.570 | 0.07 (−0.70, 0.85) 0.850 | 0.60 (−0.15, 1.34) 0.117 | 0.63 (−0.11, 1.37) 0.094 | 0.84 (0.09, 1.59) 0.028 |
| PWV | −0.04 (−0.16, 0.09) 0.585 | −0.03 (−0.14, 0.09) 0.621 | −0.03 (−0.14, 0.09) 0.646 | −0.02 (−0.14,0.10) 0.778 | 0.01 (−0.10, 0.11) 0.926 | 0.01 (−0.10, 0.13) 0.845 | −0.03 (−0.15,0.09) 0.633 | 0.03 (−0.09, 0.14) 0.638 | 0.03 (−0.08, 0.15) 0.588 |
| right ΙΜΤ | 0.00 (−0.01, 0.01) 0.707 | 0.00 (−0.01, 0.01) 0.619 | 0.00 (−0.01, 0.01) 0.721 | −0.01 (−0.01,0.00) 0.274 | 0.00 (−0.01, 0.01) 0.535 | 0.00 (−0.01, 0.01) 0.735 | 0.00 (−0.01, 0.01) 0.354 | 0.01 (0.00, 0.02) 0.146 | 0.01 (0.00, 0.02) 0.093 |
| left ΙΜΤ | 0.00 (−0.01, 0.01) 0.752 | 0.00 (−0.01, 0.01) 0.858 | 0.00 (−0.01, 0.01) 0.933 | −0.01 (−0.02,0.00) 0.087 | −0.01 (−0.02, 0.00) 0.245 | −0.01 (−0.02, 0.00) 0.150 | 0.00 (−0.01, 0.01) 0.960 | 0.00 (−0.01, 0.01) 0.616 | 0.00 (−0.01, 0.01) 0.702 |
| Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | Exp(B) (95% CI) p-value | |
| Existence of total plaques | 1.00 (0.83, 1.19) 0.965 | 0.99 (0.82/1.20) 0.906 | 1.04 (0.85, 1.27) 0.697 | 0.94 (0.79, 1.13) 0.523 | 0.99 (0.82, 1.20) 0.919 | 1.08 (0.88, 1.32) 0.459 | 1.13 (0.95, 1.36) 0.170 | 1.11 (0.91, 1.34) 0.298 | 1.18 (0.96, 1.44) 0.116 |
| Existence of carotid plaques | 0.87 (0.73, 1.04) 0.128 | 0.87 (0.72, 1.04) 0.125 | 0.90 (0.75, 1.09) 0.298 | 0.92 (0.76, 1.11) 0.380 | 0.96 (0.79, 1.17) 0.682 | 1.04 (0.85, 1.28) 0.712 | 1.13 (0.94, 1.35) 0.196 | 1.12 (0.92, 1.34) 0.256 | 1.18 (0.97, 1.43) 0.103 |
| Existence of femoral plaques | 1.12 (0.94, 1.34) 0.220 | 1.14 (0.93, 1.39) 0.207 | 1.17 (0.95, 1.43) 0.143 | 0.96 (0.80, 1.16) 0.677 | 1.01 (0.81, 1.24) 0.964 | 1.04 (0.83, 1.30) 0.756 | 1.12 (0.93, 1.35) 0.233 | 1.08 (0.88, 1.32) 0.450 | 1.10 (0.90, 1.36) 0.347 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basdeki, E.D.; Koumi, K.; Tsirimiagkou, C.; Argyris, A.; Chrysostomou, S.; Sfikakis, P.P.; Protogerou, A.D.; Karatzi, K. Late-Night Overeating or Low-Quality Food Choices Late at Night Are Associated with Subclinical Vascular Damage in Patients at Increased Cardiovascular Risk. Nutrients 2022, 14, 470. https://doi.org/10.3390/nu14030470
Basdeki ED, Koumi K, Tsirimiagkou C, Argyris A, Chrysostomou S, Sfikakis PP, Protogerou AD, Karatzi K. Late-Night Overeating or Low-Quality Food Choices Late at Night Are Associated with Subclinical Vascular Damage in Patients at Increased Cardiovascular Risk. Nutrients. 2022; 14(3):470. https://doi.org/10.3390/nu14030470
Chicago/Turabian StyleBasdeki, Eirini D., Konstantina Koumi, Christiana Tsirimiagkou, Antonios Argyris, Stavri Chrysostomou, Petros P. Sfikakis, Athanase D. Protogerou, and Kalliopi Karatzi. 2022. "Late-Night Overeating or Low-Quality Food Choices Late at Night Are Associated with Subclinical Vascular Damage in Patients at Increased Cardiovascular Risk" Nutrients 14, no. 3: 470. https://doi.org/10.3390/nu14030470
APA StyleBasdeki, E. D., Koumi, K., Tsirimiagkou, C., Argyris, A., Chrysostomou, S., Sfikakis, P. P., Protogerou, A. D., & Karatzi, K. (2022). Late-Night Overeating or Low-Quality Food Choices Late at Night Are Associated with Subclinical Vascular Damage in Patients at Increased Cardiovascular Risk. Nutrients, 14(3), 470. https://doi.org/10.3390/nu14030470