Correlates of Calcidiol Deficiency in Adults—Cross-Sectional, Observational, Population-Based Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population Sample
2.2. Variables under Study
2.3. Statistics
3. Results
3.1. Descriptive Statistics
3.2. Linear Regression
3.3. Logistic Regression
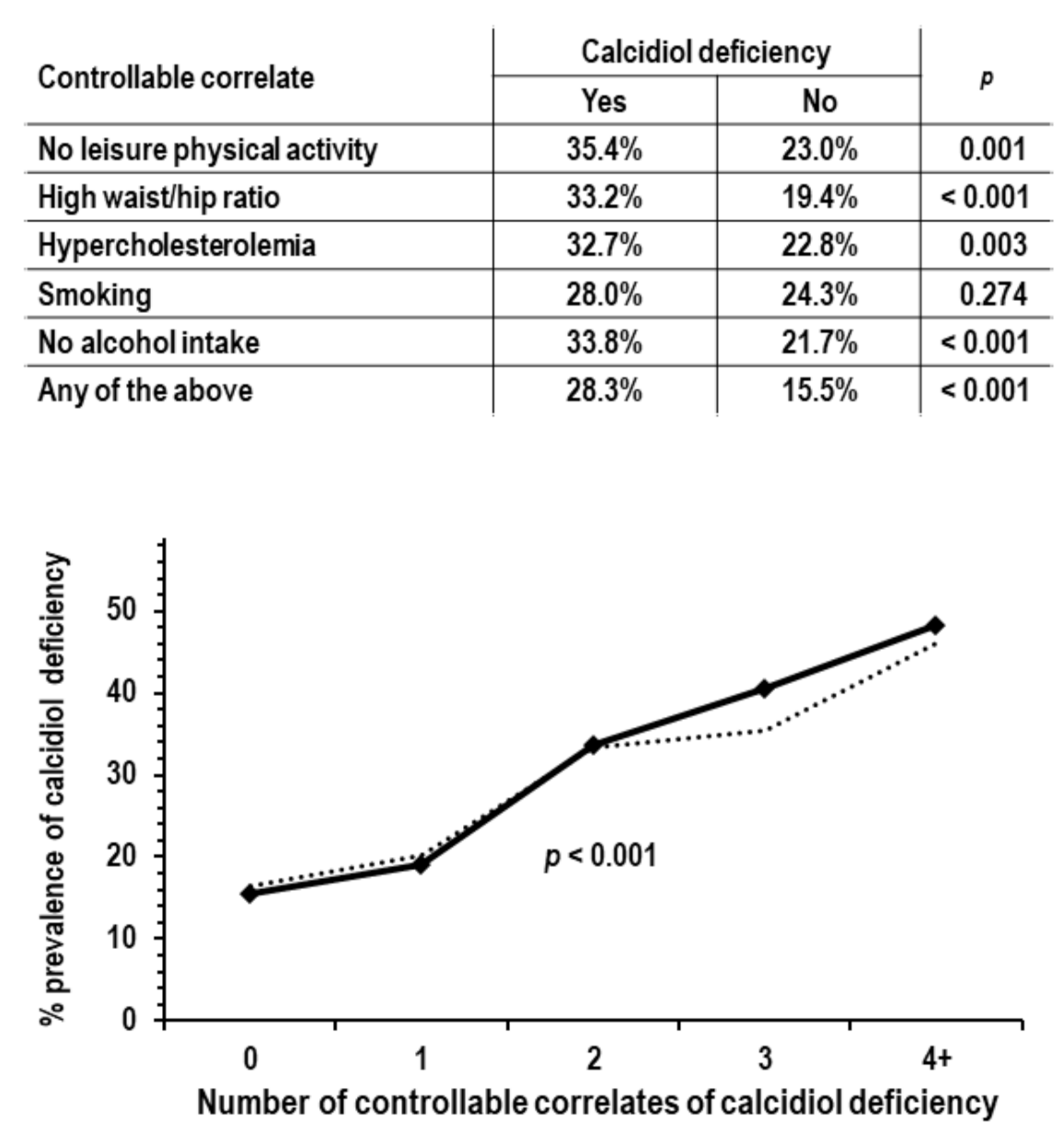
3.4. Controllable Correlates of Calcidiol Deficiency
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Christakos, S.; Ajibade, D.V.; Dhawan, P.; Fechner, A.J.; Mady, L.J. Vitamin D: Metabolism. Endocrinol. Metab. Clin. N. Am. 2010, 39, 243–253. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [Green Version]
- Damasiewicz, M.J.; Magliano, D.J.; Daly, R.M.; Gagnon, C.; Lu, Z.X.; Sikaris, K.A.; Ebeling, P.R.; Chadban, S.J.; Atkins, R.C.; Kerr, P.G.; et al. Serum 25-Hydroxyvitamin D Deficiency and the 5-Year Incidence of CKD. Am. J. Kidney Dis. 2013, 62, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef]
- Rosen, C.J. Vitamin D Insufficiency. N. Engl. J. Med. 2011, 364, 248–254. [Google Scholar] [CrossRef]
- Manson, J.E.; Brannon, P.M.; Rosen, C.J.; Taylor, C.L. Vitamin D Deficiency—Is There Really a Pandemic? N. Engl. J. Med. 2016, 375, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Sempos, C.T.; Heijboer, A.C.; Bikle, D.D.; Bollerslev, J.; Bouillon, R.; Brannon, P.M.; DeLuca, H.F.; Jones, G.; Munns, C.F.; Bilezikian, J.P.; et al. Vitamin D assays and the definition of hypovitaminosis D: Results from the First International Conference on Controversies in Vitamin D. Br. J. Clin. Pharmacol. 2018, 84, 2194–2207. [Google Scholar] [CrossRef] [PubMed]
- Giustina, A.; Adler, R.A.; Binkley, N.; Bollerslev, J.; Bouillon, R.; Dawson-Hughes, B.; Ebeling, P.R.; Feldman, D.; Formenti, A.M.; Lazaretti-Castro, M.; et al. Consensus statement from 2nd International Conference on Controversies in Vitamin D. Rev. Endocr. Metab. Disord. 2020, 21, 89–116. [Google Scholar] [CrossRef] [Green Version]
- Freedman, D.M.; Cahoon, E.K.; Rajaraman, P.; Major, J.M.; Doody, M.M.; Alexander, B.H.; Hoffbeck, R.W.; Kimlin, M.G.; Graubard, B.I.; Linet, M.S. Sunlight and Other Determinants of Circulating 25-Hydroxyvitamin D Levels in Black and White Participants in a Nationwide US Study. Am. J. Epidemiol. 2013, 177, 180–192. [Google Scholar] [CrossRef] [Green Version]
- Parva, N.R.; Tadepalli, S.; Singh, P.; Qian, A.; Joshi, R.; Kandala, H.; Nookala, V.K.; Cheriyath, P. Prevalence of Vitamin D Deficiency and Associated Risk Factors in the US Population (2011–2012). Cureus 2018, 10, e2741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akinlawon, O.J.; Lai, C.-Q.; Noel, S.E.; Flanagan, K.D.; Tucker, K.L. Risk Factors Associated with Vitamin D Status among Older Puerto Rican Adults. J. Nutr. 2021, 151, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Baylin, A.; Levy, P.D. Vitamin D deficiency and insufficiency among US adults: Prevalence, predictors and clinical implications. Br. J. Nutr. 2018, 119, 928–936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Castelnuovo, A.; Costanzo, S.; Persichillo, M.; Olivieri, M.; de Curtis, A.; Zito, F.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. Distribution of short and lifetime risks for cardiovascular disease in Italians. Eur. J. Prev. Cardiol. 2012, 19, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; Persichillo, M.; De Curtis, A.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. MOLI-SANI study Investigators Adherence to the traditional Mediterranean diet and mortality in subjects with diabetes. Prospective results from the MOLI-SANI study. Eur. J. Prev. Cardiol. 2016, 23, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, S.; Mukamal, K.J.; Di Castelnuovo, A.; Bonaccio, M.; Olivieri, M.; Persichillo, M.; De Curtis, A.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; et al. Alcohol consumption and hospitalization burden in an adult Italian population: Prospective results from the Moli-sani study. Addiction 2019, 114, 636–650. [Google Scholar] [CrossRef]
- Iacoviello, L.; De Curtis, A.; Donati, M.B.; de Gaetano, G. Biobanks for cardiovascular epidemiology and prevention. Future Cardiol. 2014, 10, 243–254. [Google Scholar] [CrossRef]
- Zeller, T.; Hughes, M.; Tuovinen, T.; Schillert, A.; Conrads-Frank, A.; Ruijter, H.D.; Schnabel, R.B.; Kee, F.; Salomaa, V.; Siebert, U.; et al. BiomarCaRE: Rationale and design of the European BiomarCaRE project including 300,000 participants from 13 European countries. Eur. J. Epidemiol. 2014, 29, 777–790. [Google Scholar] [CrossRef] [Green Version]
- Cirillo, M.; Bilancio, G.; Guarino, E.; Cavallo, P.; Lombardi, C.; Costanzo, S.; De Curtis, A.; Di Castelnuovo, A.; Iacoviello, L. Vitamin D Status and Indices of Mineral Homeostasis in the Population: Differences between 25-Hydroxyvitamin D and 1,25-Dihydroxyvitamin D. Nutrients 2019, 11, 1777. [Google Scholar] [CrossRef] [Green Version]
- Cirillo, M.; Cavallo, P.; Zulli, E.; Villa, R.; Veneziano, R.; Costanzo, S.; Magnacca, S.; Di Castelnuovo, A.; Iacoviello, L. Sodium intake and proteinuria/albuminuria in the population—Observational, cross-sectional study. Nutrients 2021, 13, 1255. [Google Scholar] [CrossRef]
- Myers, G.L.; Miller, W.G.; Coresh, J.; Fleming, J.; Greenberg, N.; Greene, T.; Hostetter, T.; Levey, A.S.; Panteghini, M.; Welch, M.; et al. Recommendations for Improving Serum Creatinine Measurement: A Report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin. Chem. 2006, 52, 5–18. [Google Scholar] [CrossRef]
- Engelsen, O. The Relationship between Ultraviolet Radiation Exposure and Vitamin D Status. Nutrients 2010, 2, 482–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atlante Italiano della Radiazione Solare. Available online: www.solaritaly.enea.it (accessed on 18 March 2018).
- Ix, J.H.; Wassel, C.L.; Stevens, L.A.; Beck, G.J.; Froissart, M.; Navis, G.; Rodby, R.; Torres, V.E.; Zhang, Y.; Greene, T.; et al. Equations to Estimate Creatinine Excretion Rate: The CKD Epidemiology Collaboration. Clin. J. Am. Soc. Nephrol. 2011, 6, 184–191. [Google Scholar] [CrossRef] [Green Version]
- Heymsfield, S.B.; Arteaga, C.; McManus, C.; Smith, J.; Moffitt, S. Measurement of muscle mass in humans: Validity of the 24-hour urinary creatinine method. Am. J. Clin. Nutr. 1983, 37, 478–494. [Google Scholar] [CrossRef]
- WHO. Waist Circumference and Wais-Hip Ratio, Report of a WHO Expert Consultation; WHO: Geneva, Switzerland, 2008; ISBN 978-92-4-150149-1. [Google Scholar]
- Inker, L.A.; Schmid, C.H.; Tighiouart, H.; Eckfeldt, J.H.; Feldman, H.I.; Greene, T.; Kusek, J.W.; Manzi, J.; Van Lente, F.; Zhang, Y.L.; et al. Estimating Glomerular Filtration Rate from Serum Creatinine and Cystatin C. N. Engl. J. Med. 2012, 367, 20–29. [Google Scholar] [CrossRef] [Green Version]
- Cirillo, M.; Lombardi, C.; Luciano, M.G.; Bilancio, G.; Anastasio, P.; De Santo, N.G. Estimation of GFR: A Comparison of New and Established Equations. Am. J. Kidney Dis. 2010, 56, 802–804. [Google Scholar] [CrossRef]
- Cheung, A.K.; Chang, T.I.; Cushman, W.C.; Furth, S.L.; Hou, F.F.; Ix, J.H.; Knoll, G.A.; Muntner, P.; Pecoits-Filho, R.; Sarnak, M.J.; et al. KDIGO 2021 Clinical Practice Guideline for the Management of Blood Pressure in Chronic Kidney Disease. Kidney Int. 2021, 99 (Suppl. S3), S1–S87. [Google Scholar] [CrossRef]
- Pörsti, I.H. Expanding targets of vitamin D receptor activation: Downregulation of several RAS components in the kidney. Kidney Int. 2008, 74, 1371–1373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spiro, A.; Buttriss, J.L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef] [Green Version]
- Simmons, G.H.; Wong, B.; Holowatz, L.A.; Kenney, W.L. Changes in the control of skin blood flow with exercise training: Where do cutaneous vascular adaptations fit in? Exp. Physiol. 2011, 96, 822–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rybchyn, M.S.; Abboud, M.; Puglisi, D.A.; Gordon-Thomson, C.; Brennan-Speranza, T.C.; Mason, R.S.; Fraser, D.R. Skeletal Muscle and the Maintenance of Vitamin D Status. Nutrients 2020, 12, 3270. [Google Scholar] [CrossRef]
- Earthman, C.P.; Beckman, L.M.; Masodkar, K.; Sibley, S.D. The link between obesity and low circulating 25-hydroxyvitamin D concentrations: Considerations and implications. Int. J. Obes. 2012, 36, 387–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monfrecola, G.; Riccio, G.; Savarese, C.; Posteraro, G.; Procaccini, E. The Acute Effect of Smoking on Cutaneous Microcirculation Blood Flow in Habitual Smokers and Nonsmokers. Dermatology 1998, 197, 115–118. [Google Scholar] [CrossRef] [Green Version]
- Ernster, V.L.; Grady, D.; Miike, R.; Black, D.; Selby, J.; Kerlikowske, K. Facial wrinkling in men and women, by smoking status. Am. J. Public Health 1995, 85, 78–82. [Google Scholar] [CrossRef] [Green Version]
- Holick, M. Environmental factors that influence the cutaneous production of vitamin D. Am. J. Clin. Nutr. 1995, 61 (Suppl. S3), 638S–645S. [Google Scholar] [CrossRef] [PubMed]
- Mahabir, S.; Baer, D.J.; Pfeiffer, R.M.; Li, Y.; A Watkins, B.; Taylor, P.R. Low to moderate alcohol consumption on serum vitamin D and other indicators of bone health in postmenopausal women in a controlled feeding study. Eur. J. Clin. Nutr. 2014, 68, 1267–1270. [Google Scholar] [CrossRef]
- Teschke, R. Alcoholic Liver Disease: Alcohol Metabolism, Cascade of Molecular Mechanisms, Cellular Targets, and Clinical Aspects. Biomedicines 2018, 6, 106. [Google Scholar] [CrossRef] [Green Version]
- Zhu, T.; Zhao, J.; Zhuo, S.; Hu, Z.; Ouyang, S.; Wunier, Y.S.; Chen, Y.; Li, Y.; Le, Y. High Fat Diet and High Cholesterol Diet Reduce Hepatic Vitamin D-25-Hydroxylase Expression and Serum 25-Hydroxyvitamin D 3 Level through Elevating Circulating Cholesterol, Glucose, and Insulin Levels. Mol. Nutr. Food Res. 2021, 65, 2100220. [Google Scholar] [CrossRef]
- Freisling, H.; Fahey, M.; Moskal, A.; Ocké, M.C.; Ferrari, P.; Jenab, M.; Norat, T.; Naska, A.; Welch, A.A.; Navarro, C.; et al. Region-Specific Nutrient Intake Patterns Exhibit a Geographical Gradient within and between European Countries. J. Nutr. 2010, 140, 1280–1286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D Intake: A Global Perspective of Current Status. J. Nutr. 2005, 135, 310–316. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Vaes, A.M.; van der Zwaluw, N.L.; van Wijngaarden, J.P.; Swart, K.M.; Ham, A.C.; van Dijk, S.C.; Enneman, A.W.; Sohl, E.; van Schoor, N.; et al. Relative importance of summer sun exposure, vitamin D intake, and genes to vitamin D status in Dutch older adults: The B-PROOF study. J. Steroid Biochem. Mol. Biol. 2015, 164, 168–176. [Google Scholar] [CrossRef]
- Scragg, R.; Holdaway, I.; Jackson, R.; Lim, T. Plasma 25-hydroxyvitamin D3 and its relation to physical activity and other heart disease risk factors in the general population. Ann. Epidemiol. 1992, 2, 697–703. [Google Scholar] [CrossRef]
- Arabi, A.; Chamoun, N.; Nasrallah, M.P.; Tamim, H.M. Vitamin D Deficiency in Lebanese Adults: Prevalence and Predictors from a Cross-Sectional Community-Based Study. Int. J. Endocrinol. 2021, 2021, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.C.; Aucott, L.S.; Duthie, G.G.; Macdonald, H.M. An application of partial least squares for identifying dietary patterns in bone health. Arch. Osteoporos. 2017, 12, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Sex, men/women = 0/1 | 501/478 |
| Age, years | 59.9 ± 9.8 |
| High education, no/yes = 0/1 | 561/418 |
| Daily solar irradiance, MJ/m2 | 14.5 ± 6.8 |
| Leisure physical activity, MET-h/day log MET-h/day | 2.27 (0.73/4.91) 0.029 ± 0.995 |
| Urinary creatinine, g/24-h | 1.27 ± 0.32 |
| Body mass index, kg/m2 | 28.7 ± 4.9 |
| Waist/hip ratio | 0.927 ± 0.075 |
| Diabetes, no/yes = 0/1 | 124/855 |
| eGFR, mL/min × 1.73 m2 | 83 ± 16 |
| Urinary albumin/creatinine ratio, mg/g log mg/g | 8.8 (3.9/20.5) 0.95 ± 0.54 |
| Systolic pressure, mm Hg | 146 ± 20 |
| Diastolic pressure, mm Hg | 83 ± 9 |
| Serum total cholesterol, mg/dL | 213 ± 40 |
| Serum HDL cholesterol, mg/dL | 57 ± 14 |
| Serum non-HDL cholesterol, mg/dL | 156 ± 38 |
| Smoking, no/yes = 0/1 | 210/769 |
| Alcohol intake, g/day log g/day | 8.7 (0.0/27.2) 0.55 ± 1.08 |
| Calorie intake, kcal/day | 2062 ± 664 |
| Dietary vitamin D, IU/day log IU/day | 83.8 (62.6/110.1) 1.91 ± 0.20 |
| Vitamin D supplement, no/yes = 0/1 | 952/27 |
| Beta (95% CI) | ||
|---|---|---|
| Single-Variable Regression | Multi-Variable Regression | |
| Sex, men/women = 0/1 | −0.069 (−0.132/−0.006) | −0.013 (−0.239/0.213) |
| Age, years | −0.115 (−0.178/−0.052) | −0.121 (−0.229/−0.013) |
| High education, no/yes = 0/1 | 0.050 (−0.013/0.113) | −0.003 (−0.066/0.059) |
| Daily solar irradiance, MJ/m2 | 0.255 (0.194/0.316) | 0.229 (0.170/0.287) |
| Leisure physical activity, log MET-h/day | 0.158 (0.095/0.0.221) | 0.115 ((0.054/0.177) |
| Urinary creatinine, g/24-h | 0.046 (−0.017/0.109) | 0.123 (−0.136/0.381) |
| Body mass index, kg/m2 | −0.112 (−0.175/−0.049) | 0.093 (−0.232/0.045) |
| Waist/hip ratio | −0.136 (−0.199/−0.073) | −0.126 (−0.193/−0.059) |
| Diabetes, no/yes = 0/1 | −0.075 (−0.138/−0.012) | −0.012 (−0.074/0.049) |
| eGFR, mL/min × 1.73 m2 | −0.008 (−0.071/0.055) | −0.129 (−0.204/−0.054) |
| Urinary albumin/creatinine ratio, log mg/g | −0.102 (−0.165/−0.039) | −0.041 (−0.101/0.020) |
| Systolic pressure, mm Hg | −0.142 (−0.205/−0.079) | −0.120 (−0.207/−0.034) |
| Diastolic pressure, mm Hg | −0.038 (−0.101/0.025) | 0.047 (−0.034/0.128) |
| Serum total cholesterol, mg/dL | −0.170 (−0.233/−0.107) | −0.110 (−0.169/−0.050) |
| Serum HDL cholesterol, mg/dL | −0.073 (−0.136/−0.010) | not included |
| Serum non-HDL cholesterol, mg/dL | −0.150 (−0.213/−0.087) | not included |
| Smoking, no/yes = 0/1 | −0.066 (−0.129/−0.003) | −0.092 (−0.150/−0.033) |
| Alcohol intake, log g/day | 0.071 (0.008/0.134) | 0.075 (0.008/0.142) |
| Calorie intake, kcal/day | 0.029 (−0.034/0.092) | −0.043 (−0.120/0.034) |
| Dietary vitamin D, log IU/day | 0.040 (−0.023/0.103) | 0.027 (−0.043/0.097) |
| Vitamin D supplement, no/yes = 0/1 | 0.150 (0.087/0.213) | 0.200 (0.142/0.258) |
| Independent Variables | Reference Interval | Odds Ratio (95% CI) of Calcidiol Deficiency | |
|---|---|---|---|
| Single-Variable Regression | Multiple-Variable Regression | ||
| Sex | women vs. men | 1.78 (1.32/2.39) | 1.35 (0.38/4.77) |
| Age, years | +1SD | 1.32 (1.13/1.53) | 1.37 (1.01/1.87) |
| High education | No vs. yes | 1.33 (0.99/1.79) | 1.04 (0.73/1.47) |
| Daily solar irradiance, MJ/m2 | −1SD | 1.53 (1.31/1.78) | 1.53 (1.30/1.81) |
| Leisure physical activity, log MET-h/day | −1SD | 1.36 (1.18/1.56) | 1.24 (1.06/1.46) |
| Urinary creatinine, g/24-h | +1SD | 1.30 (1.12/1.51) | 1.20 (0.58/2.50) |
| Body mass index, kg/m2 | +1SD | 1.03 (0.99/1.06) | 1.12 (0.76/1.64) |
| Waist/hip ratio | +1SD | 1.12 (0.97/1.30) | 1.21 (1.12/1.44) |
| Diabetes | Yes vs. no | 1.49 (0.99/2.25) | 1.17 (0.72/1.90) |
| eGFR, mL/min × 1.73 m2 | +1SD | 0.95 (0.82/1.10) | 1.23 (0.99/1.52) |
| Urinary albumin/creatinine ratio, log mg/g | +1SD | 1.24 (1.07/1.44) | 1.08 (0.92/1.28) |
| Systolic pressure, mm Hg | +1SD | 1.25 (1.08/1.44) | 1.29 (1.03/1.63) |
| Diastolic pressure, mm Hg | +1SD | 1.11 (1.04/1.18) | 0.88 (0.72/1.09) |
| Serum total cholesterol, mg/dL | +1SD | 1.33 (1.14/1.54) | 1.24 (1.05/1.46) |
| Smoking | Yes vs. no | 1.23 (0.87/1.73) | 1.48 (1.01/2.19) |
| Alcohol intake, log g/day | −1SD | 1.23 (1.11/1.36) | 1.21 (1.06/1.38) |
| Dietary calorie, kcal/day | +1SD | 1.20 (1.03/1.40) | 0.90 (0.73/1.12) |
| Dietary vitamin D, log IU/day | −1SD | 1.17 (1.01/1.36) | 1.09 (0.90/1.32) |
| Vitamin D supplement | No vs. yes | 8.81 (1.20/64.64) | 24.32 (3.12/189.62) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cirillo, M.; Bilancio, G.; Cavallo, P.; Costanzo, S.; De Curtis, A.; Di Castelnuovo, A.; Iacoviello, L. Correlates of Calcidiol Deficiency in Adults—Cross-Sectional, Observational, Population-Based Study. Nutrients 2022, 14, 459. https://doi.org/10.3390/nu14030459
Cirillo M, Bilancio G, Cavallo P, Costanzo S, De Curtis A, Di Castelnuovo A, Iacoviello L. Correlates of Calcidiol Deficiency in Adults—Cross-Sectional, Observational, Population-Based Study. Nutrients. 2022; 14(3):459. https://doi.org/10.3390/nu14030459
Chicago/Turabian StyleCirillo, Massimo, Giancarlo Bilancio, Pierpaolo Cavallo, Simona Costanzo, Amalia De Curtis, Augusto Di Castelnuovo, and Licia Iacoviello. 2022. "Correlates of Calcidiol Deficiency in Adults—Cross-Sectional, Observational, Population-Based Study" Nutrients 14, no. 3: 459. https://doi.org/10.3390/nu14030459
APA StyleCirillo, M., Bilancio, G., Cavallo, P., Costanzo, S., De Curtis, A., Di Castelnuovo, A., & Iacoviello, L. (2022). Correlates of Calcidiol Deficiency in Adults—Cross-Sectional, Observational, Population-Based Study. Nutrients, 14(3), 459. https://doi.org/10.3390/nu14030459







