Chrononutrition—When We Eat Is of the Essence in Tackling Obesity
Abstract
1. Introduction
2. Chronobiology
Genomics and Epigenomics of Chronobiology
3. Chronotypes, Chronodisruption and Energy Homeostasis
4. Chrononutrition
4.1. What to Eat
4.2. When to Eat
5. Time-Restricted Eating—Just Another Approach to Reduce Caloric Intake or a Circadian Alignment Tool
6. Mechanisms of Chrononutrition in Energy Homeostasis and Obesity
6.1. Appetite Control
6.2. Energy Sensors in the Body
6.2.1. AMP-Activated Protein Kinase (AMPK)
6.2.2. Mitochondrial Dynamics
6.2.3. Adipose Tissue
6.2.4. Gut Microbiota
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. World Health Organization. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 16 September 2022).
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and Cardiovascular Disease. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 968–976. [Google Scholar] [CrossRef] [PubMed]
- New WHO Report: Europe Can Reverse Its Obesity “Epidemic”. 2022. Available online: https://www.who.int/europe/news/item/03-05-2022-new-who-report--europe-can-reverse-its-obesity--epidemic (accessed on 16 September 2022.).
- Loos, R.J.F.; Yeo, G.S.H. The genetics of obesity: From discovery to biology. Nat. Rev. Genet. 2021, 23, 1. [Google Scholar] [CrossRef] [PubMed]
- Sandholt, C.H.; Hansen, T.; Pedersen, O. Beyond the fourth wave of genome-wide obesity association studies. Nutr. Diabetes 2012, 2, e37. [Google Scholar] [CrossRef]
- Jiang, P.; Turek, F.W. The endogenous circadian clock programs animals to eat at certain times of the 24-hour day: What if we ignore the clock? Physiol. Behav. 2018, 193, 211–217. [Google Scholar] [CrossRef]
- Stenvers, D.J.; Jongejan, A.; Atiqi, S.; Vreijling, J.P.; Limonard, E.J.; Endert, E.; Baas, F.; Moerland, P.D.; Fliers, E.; Kalsbeek, A.; et al. Diurnal rhythms in the white adipose tissue transcriptome are disturbed in obese individuals with type 2 diabetes compared with lean control individuals. Diabetologia 2019, 62, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Challet, E. Interactions between photic and nonphotic stimuli to synchronize the master circadian clock in mammals. Front. Biosci. 2003, 8, s246–s257. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Takahashi, J.S. Transcriptional architecture of the mammalian circadian clock. Nat. Rev. Genet. 2016, 18, 164–179. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Lazar, M.A. Transcriptional Control of Circadian Rhythms and Metabolism: A Matter of Time and Space. Endocr. Rev. 2020, 41, 707–732. [Google Scholar] [CrossRef]
- Grivas, T.B.; Savvidou, O.D. Melatonin the “light of night” in human biology and adolescent idiopathic scoliosis. Scoliosis 2007, 2, 6–14. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Smits, M.; Spence, W.; Srinivasan, V.; Cardinali, D.P.; Lowe, A.D.; Kayumov, L. Dim light melatonin onset (DLMO): A tool for the analysis of circadian phase in human sleep and chronobiological disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 1–11. [Google Scholar] [CrossRef]
- Zisapel, N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br. J. Pharmacol. 2018, 175, 3190–3199. [Google Scholar] [CrossRef]
- Beccuti, G.; Pannain, S. Sleep and obesity. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Ciccone, G.; Durazzo, M.; Ghinamo, L.; Villois, P.; Canil, S.; Gambino, R.; Cassader, M.; Gentile, L.; Cavallo-Perin, P. Contributors to the obesity and hyperglycemia epidemics. A prospective study in a population-based cohort. Int. J. Obes. 2011, 35, 1442–1449. [Google Scholar] [CrossRef] [PubMed]
- Potter, G.D.M.; Skene, D.J.; Arendt, J.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Circadian Rhythm and Sleep Disruption: Causes, Metabolic Consequences, and Countermeasures. Endocr. Rev. 2016, 37, 584–608. [Google Scholar] [CrossRef] [PubMed]
- Buhr, E.D.; Takahashi, J.S. Molecular components of the Mammalian circadian clock. Handb. Exp. Pharmacol. 2013, 217, 3–27. [Google Scholar]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and Peripheral Circadian Clocks in Mammals. Annu. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef]
- Liu, A.C.; Tran, H.G.; Zhang, E.E.; Priest, A.A.; Welsh, D.K.; Kay, S.A. Redundant Function of REV-ERBα and β and Non-Essential Role for Bmal1 Cycling in Transcriptional Regulation of Intracellular Circadian Rhythms. PLoS Genet. 2008, 4, e1000023. [Google Scholar] [CrossRef]
- Liu, C.; Li, S.; Liu, T.; Borjigin, J.; Lin, J.D. Transcriptional coactivator PGC-1α integrates the mammalian clock and energy metabolism. Nature 2007, 447, 477–481. [Google Scholar] [CrossRef]
- Bass, J.; Takahashi, J.S. Circadian Integration of Metabolism and Energetics. Science 2010, 330, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Canaple, L.; Rambaud, J.; Dkhissi-Benyahya, O.; Rayet, B.; Tan, N.S.; Michalik, L.; Delaunay, F.; Wahli, W.; Laudet, V. Reciprocal Regulation of Brain and Muscle Arnt-Like Protein 1 and Peroxisome Proliferator-Activated Receptor α Defines a Novel Positive Feedback Loop in the Rodent Liver Circadian Clock. Mol. Endocrinol. 2006, 20, 1715–1727. [Google Scholar] [CrossRef] [PubMed]
- Masri, S.; Sassone-Corsi, P. Plasticity and specificity of the circadian epigenome. Nat. Neurosci. 2010, 13, 1324–1329. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.; Reinke, H.; Altmeyer, M.; Gutierrez-Arcelus, M.; Hottiger, M.O.; Schibler, U. Poly(ADP-Ribose) Polymerase 1 Participates in the Phase Entrainment of Circadian Clocks to Feeding. Cell 2010, 142, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, J.; Sahar, S.; Grimaldi, B.; Tamaru, T.; Takamatsu, K.; Nakahata, Y.; Sassone-Corsi, P. CLOCK-mediated acetylation of BMAL1 controls circadian function. Nature 2007, 450, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Doi, M.; Hirayama, J.; Sassone-Corsi, P. Circadian Regulator CLOCK Is a Histone Acetyltransferase. Cell 2006, 125, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Mure, L.S.; Le, H.D.; Benegiamo, G.; Chang, M.W.; Rios, L.; Jillani, N.; Ngotho, M.; Kariuki, T.; Dkhissi-Benyahya, O.; Cooper, H.M.; et al. Diurnal transcriptome atlas of a primate across major neural and peripheral tissues. Science 2018, 359, 6381. [Google Scholar] [CrossRef]
- Fuhr, L.; Abreu, M.; Pett, P.; Relógio, A. Circadian systems biology: When time matters. Comput. Struct. Biotechnol. J. 2015, 13, 417–426. [Google Scholar] [CrossRef]
- Li, J.; Nie, P.; Turck, C.W.; Wang, G.-Z. Gene networks under circadian control exhibit diurnal organization in primate organs. Commun. Biol. 2022, 5, 1–14. [Google Scholar] [CrossRef]
- Eckel-Mahan, K.; Sassone-Corsi, P. Metabolism and the Circadian Clock Converge. Physiol. Rev. 2013, 93, 107–135. [Google Scholar] [CrossRef]
- Kornmann, B.; Schaad, O.; Bujard, H.; Takahashi, J.S.; Schibler, U. System-Driven and Oscillator-Dependent Circadian Transcription in Mice with a Conditionally Active Liver Clock. O’Rahilly, S., editor. PLoS Biol. 2007, 5, e34. [Google Scholar] [CrossRef]
- Peng, X.; Fan, R.; Xie, L.; Shi, X.; Dong, K.; Zhang, S.; Tao, J.; Xu, W.; Ma, D.; Chen, J.; et al. A Growing Link between Circadian Rhythms, Type 2 Diabetes Mellitus and Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 23, 504. [Google Scholar] [CrossRef]
- Ki, Y.; Ri, H.; Lee, H.; Yoo, E.; Choe, J.; Lim, C. Warming Up Your Tick-Tock: Temperature-Dependent Regulation of Circadian Clocks. The Neuroscientist: A Review. J. Bringing Neurobiol. Neurol. Psychiatry 2015, 21, 503–518. [Google Scholar]
- Saini, R.; Jaskolski, M.; Davis, S.J. Circadian oscillator proteins across the kingdoms of life: Structural aspects. BMC Biol. 2019, 17, 13. [Google Scholar]
- Kalmbach, D.A.; Schneider, L.D.; Cheung, J.; Bertrand, S.J.; Kariharan, T.; Pack, A.I.; Gehrman, P.R. Genetic Basis of Chronotype in Humans: Insights from Three Landmark GWAS. Sleep 2016, 40, zsw048. [Google Scholar] [CrossRef] [PubMed]
- Sack, R.L.; Auckley, D.; Auger, R.R.; Carskadon, M.A.; Wright, J.K.P.; Vitiello, M.V.; Zhdanova, I.V. Circadian Rhythm Sleep Disorders: Part II, Advanced Sleep Phase Disorder, Delayed Sleep Phase Disorder, Free-Running Disorder, and Irregular Sleep-Wake Rhythm. Sleep 2007, 30, 1484–1501. [Google Scholar] [CrossRef]
- Adan, A.; Archer, S.N.; Hidalgo, M.P.; Di Milia, L.; Natale, V.; Randler, C. Circadian Typology: A Comprehensive Review. Chronobiol. Int. 2012, 29, 1153–1175. [Google Scholar] [CrossRef] [PubMed]
- Taillard, J.; Philip, P.; Claustrat, B.; Capelli, A.; Coste, O.; Chaumet, G.; Sagaspe, P. Time course of neurobehavioral alertness during extended wakefulness in morning- and evening-type healthy sleepers. Chronobiol. Int. 2011, 28, 520–527. [Google Scholar] [CrossRef]
- Jones, C.R.; Huang, A.L.; Ptáček, L.J.; Fu, Y.-H. Genetic basis of human circadian rhythm disorders. Exp. Neurol. 2013, 243, 28–33. [Google Scholar] [CrossRef]
- Jones, S.E.; Lane, J.M.; Wood, A.R.; van Hees, V.T.; Tyrrell, J.; Beaumont, R.N.; Jeffries, A.R.; Dashti, H.S.; Hillsdon, M.; Ruth, K.S.; et al. Genome-wide association analyses of chronotype in 697,828 individuals provides insights into circadian rhythms. Nat. Commun. 2019, 10, 343. [Google Scholar] [CrossRef]
- Valladares, M.; Obregón, A.M.; Chaput, J.-P. Association between genetic variants of the clock gene and obesity and sleep duration. J. Physiol. Biochem. 2015, 71, 855–860. [Google Scholar] [CrossRef]
- Rijo-Ferreira, F.; Takahashi, J.S. Genomics of circadian rhythms in health and disease. Genome Med. 2019, 11, 82. [Google Scholar] [CrossRef]
- Molina-Montes, E.; Rodríguez-Barranco, M.; Ching-López, A.; Artacho, R.; Huerta, J.M.; Amiano, P.; Lasheras, C.; Moreno-Iribas, C.; Jimenez-Zabala, A.; Chirlaque, M.D.; et al. Circadian clock gene variants and their link with chronotype, chrononutrition, sleeping patterns and obesity in the European prospective investigation into cancer and nutrition (EPIC) study. Clin. Nutr. 2022, 41, 1977–1990. [Google Scholar] [CrossRef] [PubMed]
- Ashbrook, L.H.; Krystal, A.D.; Fu, Y.-H.; Ptáček, L.J. Genetics of the human circadian clock and sleep homeostat. Neuropsychopharmacology 2019, 45, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Reutrakul, S.; Hood, M.M.; Crowley, S.J.; Morgan, M.K.; Teodori, M.; Knutson, K.L.; Van Cauter, E. Chronotype Is Independently Associated with Glycemic Control in Type 2 Diabetes. Diabetes Care 2013, 36, 2523–2529. [Google Scholar] [CrossRef] [PubMed]
- Patterson, F.; Malone, S.K.; Grandner, M.A.; Lozano, A.; Perkett, M.; Hanlon, A. Interactive effects of sleep duration and morning/evening preference on cardiovascular risk factors. Eur. J. Public Health 2017, 28, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, M.; Yuan, F.; Zhang, H. Sugary beverage consumption mediates the relationship between late chronotype, sleep duration, and weight increase among undergraduates: A cross-sectional study. Environ. Health Prev. Med. 2018, 23, 63. [Google Scholar] [CrossRef]
- Anothaisintawee, T.; Lertrattananon, D.; Thamakaison, S.; Thakkinstian, A.; Reutrakul, S. The Relationship Among Morningness-Eveningness, Sleep Duration, Social Jetlag, and Body Mass Index in Asian Patients with Prediabetes. Front. Endocrinol. 2018, 9, 435. [Google Scholar] [CrossRef]
- Culnan, E.; Kloss, J.D.; Grandner, M. A prospective study of weight gain associated with chronotype among college freshmen. Chronobiol. Int. 2013, 30, 682–690. [Google Scholar] [CrossRef]
- Sun, X.; Gustat, J.; Bertisch, S.; Redline, S.; Bazzano, L. The association between sleep chronotype and obesity among black and white participants of the Bogalusa Heart Study. Chronobiol. Int. 2019, 37, 123–134. [Google Scholar] [CrossRef]
- Roenneberg, T.; Wirz-Justice, A.; Merrow, M. Life between Clocks: Daily Temporal Patterns of Human Chronotypes. J. Biol. Rhythm. 2003, 18, 80–90. [Google Scholar] [CrossRef]
- Mishima, K.; Tozawa, T.; Satoh, K.; Saitoh, H.; Mishima, Y. The 3111T/C polymorphism ofhClock is associated with evening preference and delayed sleep timing in a Japanese population sample. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2005, 133, 101–104. [Google Scholar] [CrossRef]
- Garaulet, M.; Corbalán, M.D.; Madrid, J.A.; Morales, E.; Baraza, J.C.; Lee, Y.C.; Ordovas, J.M. CLOCK gene is implicated in weight reduction in obese patients participating in a dietary programme based on the Mediterranean diet. Int. J. Obes. 2010, 34, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, P.; Tortorella, A.; Docimo, L.; Maldonato, M.N.; Canestrelli, B.; De Luca, L.; Maj, M. Investigation of 3111T/C polymorphism of the CLOCK gene in obese individuals with or without binge eating disorder: Association with higher body mass index. Neurosci. Lett. 2008, 435, 30–33. [Google Scholar] [CrossRef]
- López-Guimerà, G.; Dashti, H.S.; Smith, C.E.; Sánchez-Carracedo, D.; Ordovas, J.M.; Garaulet, M. CLOCK 3111 T/C SNP Interacts with Emotional Eating Behavior for Weight-Loss in a Mediterranean Population. Rosenfeld CS, editor. PLoS ONE 2014, 9, e99152. [Google Scholar] [CrossRef] [PubMed]
- Giovaninni, N.P.; Fuly, J.T.; Moraes, L.I.; Coutinho, T.N.; Trarbach, E.B.; de LJorge, A.A.; Costalonga, E.F. Study of the association between 3111T/C polymorphism of the CLOCK gene and the presence of overweight in schoolchildren. J. Pediatr. 2014, 90, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Bandín, C.; Martinez-Nicolas, A.; Ordovás, J.M.; Lucas, J.A.R.; Castell, P.; Silvente, T.; Madrid, J.A.; Garaulet, M. Differences in circadian rhythmicity in CLOCK 3111T/C genetic variants in moderate obese women as assessed by thermometry, actimetry and body position. Int. J. Obes. 2012, 37, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Sánchez-Moreno, C.; Smith, C.E.; Lee, Y.-C.; Nicolás, F.; Ordovás, J.M. Ghrelin, Sleep Reduction and Evening Preference: Relationships to CLOCK 3111 T/C SNP and Weight Loss. Tomé D, editor. PLoS ONE 2011, 6, e17435. [Google Scholar] [CrossRef]
- Garcia-Rios, A.; Gomez-Delgado, F.J.; Garaulet, M.; Alcala-Diaz, J.F.; Delgado-Lista, F.J.; Marin, C.; Rangel-Zuñiga, O.A.; Rodriguez-Cantalejo, F.; Gomez-Luna, P.; Ordovas, J.M.; et al. Beneficial effect of CLOCKgene polymorphism rs1801260 in combination with low-fat diet on insulin metabolism in the patients with metabolic syndrome. Chronobiol. Int. 2013, 31, 401–408. [Google Scholar] [CrossRef]
- Sookoian, S.; Gemma, C.; Gianotti, T.F.; Burgueño, A.; Castaño, G.; Pirola, C.J. Genetic variants of Clock transcription factor are associated with individual susceptibility to obesity. Am. J. Clin. Nutr. 2008, 87, 1606–1615. [Google Scholar] [CrossRef]
- Garaulet, M.; Lee, Y.-C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.-Q.; Ordovas, J.M. Genetic variants in human CLOCK associate with total energy intake and cytokine sleep factors in overweight subjects (GOLDN population). Eur. J. Hum. Genet. 2009, 18, 364–369. [Google Scholar] [CrossRef]
- Camblor Murube, M.; Borregon-Rivilla, E.; Colmenarejo, G.; Aguilar-Aguilar, E.; Martínez, J.A.; Ramírez De Molina, A.; Reglero, G.; Loria-Kohen, V. Polymorphism of CLOCK Gene rs3749474 as a Modulator of the Circadian Evening Carbohydrate Intake Impact on Nutritional Status in an Adult Sample. Nutrients 2020, 12, 1142. [Google Scholar] [CrossRef]
- Ye, D.; Cai, S.; Jiang, X.; Ding, Y.; Chen, K.; Fan, C.; Jin, M. Associations of polymorphisms in circadian genes with abdominal obesity in Chinese adult population. Obes. Res. Clin. Pract. 2016, 10 (Suppl. 1), S133–S141. [Google Scholar] [CrossRef] [PubMed]
- Scott, E.M.; Carter, A.M.; Grant, P.J. Association between polymorphisms in the Clock gene, obesity and the metabolic syndrome in man. Int. J. Obes. 2008, 32, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Asensio, E.M.; Coltell, O.; Sorlí, J.V.; Estruch, R.; Martínez-González, M.Á.; Salas-Salvadó, J.; Castañer, O.; Arós, F.; Lapetra, J.; et al. CLOCK gene variation is associated with incidence of type-2 diabetes and cardiovascular diseases in type-2 diabetic subjects: Dietary modulation in the PREDIMED randomized trial. Cardiovasc. Diabetol. 2016, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Delgado, F.; Garcia-Rios, A.; Alcala-Diaz, J.F.; Rangel-Zuñiga, O.; Delgado-Lista, J.; Yubero-Serrano, E.M.; Lopez-Moreno, J.; Tinahones, F.J.; Ordovas, J.M.; Garaulet, M.; et al. Chronic consumption of a low-fat diet improves cardiometabolic risk factors according to theCLOCKgene in patients with coronary heart disease. Mol. Nutr. Food Res. 2015, 59, 2556–2564. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Lee, Y.-C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.-Q.; Ordovas, J.M. CLOCK genetic variation and metabolic syndrome risk: Modulation by monounsaturated fatty acids. Am. J. Clin. Nutr. 2009, 90, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Follis, J.L.; Smith, C.E.; Tanaka, T.; Cade, B.E.; Gottlieb, D.J.; Hruby, A.; Jacques, P.F.; Lamon-Fava, S.; Richardson, K.; et al. Habitual sleep duration is associated with BMI and macronutrient intake and may be modified by CLOCK genetic variants. Am. J. Clin. Nutr. 2015, 101, 135–143. [Google Scholar] [CrossRef]
- Allebrandt, K.V.; Teder-Laving, M.; Akyol, M.; Pichler, I.; Müller-Myhsok, B.; Pramstaller, P.; Merrow, M.; Meitinger, T.; Metspalu, A.; Roenneberg, T. CLOCK Gene Variants Associate with Sleep Duration in Two Independent Populations. Biol. Psychiatry 2010, 67, 1040–1047. [Google Scholar] [CrossRef]
- Kim, H.-I.; Lee, H.-J.; Cho, C.-H.; Kang, S.-G.; Yoon, H.-K.; Park, Y.-M.; Lee, S.H.; Moon, J.H.; Song, H.M.; Lee, E.; et al. Association of CLOCK, ARNTL, andNPAS2gene polymorphisms and seasonal variations in mood and behavior. Chronobiol. Int. 2015, 32, 785–791. [Google Scholar] [CrossRef]
- Garaulet, M.; Esteban Tardido, A.; Lee, Y.-C.; Smith, C.E.; Parnell, L.D.; Ordovás, J.M. SIRT1 and CLOCK 3111T>C combined genotype is associated with evening preference and weight loss resistance in a behavioral therapy treatment for obesity. Int. J. Obes. 2012, 36, 1436–1441. [Google Scholar] [CrossRef]
- Garaulet, M.; Corbalán-Tutau, M.D.; Madrid, J.A.; Baraza, J.C.; Parnell, L.D.; Lee, Y.-C.; Ordovas, J.M. PERIOD2 Variants Are Associated with Abdominal Obesity, Psycho-Behavioral Factors, and Attrition in the Dietary Treatment of Obesity. J. Am. Diet. Assoc. 2010, 110, 917–921. [Google Scholar] [CrossRef]
- Partonen, T.; Treutlein, J.; Alpman, A.; Frank, J.; Johansson, C.H.; Depner, M.; Aron, L.; Rietschel, M.; Wellek, S.; Soronen, P.; et al. Three circadian clock genes Per2, Arntl, and Npas2 contribute to winter depression. Ann. Med. 2007, 39, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Smith, C.E.; Gomez-Abellán, P.; Ordovás-Montañés, M.; Lee, Y.-C.; Parnell, L.D.; Arnett, D.K.; Ordovás, J.M. REV-ERB-ALPHA circadian gene variant associates with obesity in two independent populations: Mediterranean and North American. Mol. Nutr. Food Res. 2013, 58, 821–829. [Google Scholar] [CrossRef]
- Reinke, H.; Asher, G. Crosstalk between metabolism and circadian clocks. Nat. Rev. Mol. Cell Biol. 2019, 20, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Longo, V.D.; Panda, S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metab. 2016, 23, 1048–1059. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, M.V.; Booth, F.W. Eating, exercise, and “thrifty” genotypes: Connecting the dots toward an evolutionary understanding of modern chronic diseases. J. Appl. Physiol. 2004, 96, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Challet, E. The circadian regulation of food intake. Nat. Rev. Endocrinol. 2019, 15, 393–405. [Google Scholar] [CrossRef]
- Lane, J.M.; Vlasac, I.; Anderson, S.G.; Kyle, S.D.; Dixon, W.G.; Bechtold, D.A.; Gill, S.; Little, M.A.; Luik, A.; Loudon, A.; et al. Genome-wide association analysis identifies novel loci for chronotype in 100,420 individuals from the UK Biobank. Nat. Commun. 2016, 7, 10889. [Google Scholar] [CrossRef]
- Jones, S.E.; Tyrrell, J.; Wood, A.R.; Beaumont, R.N.; Ruth, K.S.; Tuke, M.A.; Yaghootkar, H.; Hu, T.; Teder-Laving, M.; Hayward, C.; et al. Genome-Wide Association Analyses in 128,266 Individuals Identifies New Morningness and Sleep Duration Loci. Shi, J., editor. PLOS Genet. 2016, 12, e1006125. [Google Scholar] [CrossRef]
- Neves, A.R.; Albuquerque, T.; Quintela, T.; Costa, D. Circadian rhythm and disease: Relationship, new insights, and future perspectives. J. Cell Physiol. 2022, 237, 3239–3256. [Google Scholar] [CrossRef]
- Liu, Q.; Shi, J.; Duan, P.; Liu, B.; Li, T.; Wang, C.; Li, H.; Yang, T.; Gan, Y.; Wang, X.; et al. Is shift work associated with a higher risk of overweight or obesity? A systematic review of observational studies with meta-analysis. Int. J. Epidemiol. 2018, 47, 1956–1971. [Google Scholar] [CrossRef]
- Carriazo, S.; Ramos, A.M.; Sanz, A.B.; Sanchez-Niño, M.D.; Kanbay, M.; Ortiz, A. Chronodisruption: A Poorly Recognized Feature of CKD. Toxins 2020, 12, 151. [Google Scholar] [CrossRef] [PubMed]
- Zerbini, G.; Kantermann, T.; Merrow, M. Strategies to decrease social jetlag: Reducing evening blue light advances sleep and melatonin. Eur. J. Neurosci. 2018, 51, 2355–2366. [Google Scholar] [CrossRef] [PubMed]
- Bonham, M.P.; Kaias, E.; Zimberg, I.; Leung, G.K.W.; Davis, R.; Sletten, T.L.; Windsor-Aubrey, H.; Huggins, C.E. Effect of Night Time Eating on Postprandial Triglyceride Metabolism in Healthy Adults: A Systematic Literature Review. J. Biol. Rhythm. 2019, 34, 119–130. [Google Scholar] [CrossRef]
- Leung, G.K.W.; Huggins, C.E.; Bonham, M.P. Effect of meal timing on postprandial glucose responses to a low glycemic index meal: A crossover trial in healthy volunteers. Clin. Nutr. 2019, 38, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Colles, S.L.; Dixon, J.B.; O’Brien, P.E. Night eating syndrome and nocturnal snacking: Association with obesity, binge eating and psychological distress. Int. J. Obes. 2007, 31, 1722–1730. [Google Scholar] [CrossRef] [PubMed]
- Harb, A.; Levandovski, R.; Oliveira, C.; Caumo, W.; Allison, K.; Stunkard, A.; Hidalgo, M.P. Night eating patterns and chronotypes: A correlation with binge eating behaviors. Psychiatry Res. 2012, 200, 489–493. [Google Scholar] [CrossRef]
- McHill, A.W.; Melanson, E.L.; Higgins, J.; Connick, E.; Moehlman, T.M.; Stothard, E.R.; Wright, K.P., Jr. Impact of circadian misalignment on energy metabolism during simulated nightshift work. Proc. Natl. Acad. Sci. USA 2014, 111, 17302–17307. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L.E.; Chiuve, S.E.; Mekary, R.A.; Jensen, M.K.; Flint, A.J.; Hu, F.B.; Rimm, E.B. A prospective study of breakfast Eating and Incident Coronary Heart Disease in a Cohort of Male, US Health Professionals. Circulation 2013, 128, 337–343. [Google Scholar]
- Pickel, L.; Sung, H.-K. Feeding Rhythms and the Circadian Regulation of Metabolism. Front. Nutr. 2020, 7, 39. [Google Scholar] [CrossRef]
- Kräuchi, K.; Cajochen, C.; Werth, E.; Wirz-Justice, A. Alteration of Internal Circadian Phase Relationships after Morning versus Evening Carbohydrate-Rich Meals in Humans. J. Biol. Rhythm. 2002, 17, 364–376. [Google Scholar] [CrossRef]
- Wehrens, S.M.T.; Christou, S.; Isherwood, C.; Middleton, B.; Gibbs, M.A.; Archer, S.N.; Skene, D.J.; Johnston, J.D. Meal Timing Regulates the Human Circadian System. Curr. Biol. 2017, 27, 1768–1775.e3. [Google Scholar] [CrossRef] [PubMed]
- Damiola, F. Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes Dev. 2000, 14, 2950–2961. [Google Scholar] [CrossRef] [PubMed]
- Baron, K.G.; Reid, K.J.; Kern, A.S.; Zee, P.C. Role of Sleep Timing in Caloric Intake and BMI. Obesity 2011, 19, 1374–1381. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, J.; Eguchi, E.; Nagaoka, K.; Ito, T.; Ogino, K. Association of night eating habits with metabolic syndrome and its components: A longitudinal study. BMC Public Health 2018, 18, 1366. [Google Scholar] [CrossRef] [PubMed]
- Okada, C.; Imano, H.; Muraki, I.; Yamada, K.; Iso, H. The Association of Having a Late Dinner or Bedtime Snack and Skipping Breakfast with Overweight in Japanese Women. J. Obes. 2019, 2019, 2439571. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Lozano, N.; Tvarijonaviciute, A.; Ríos, R.; Barón, I.; Scheer, F.A.J.L.; Garaulet, M. Late Eating Is Associated with Obesity, Inflammatory Markers and Circadian-Related Disturbances in School-Aged Children. Nutrients 2020, 12, 2881. [Google Scholar] [CrossRef]
- Adnan, D.; Trinh, J.; Bishehsari, F. Inconsistent eating time is associated with obesity: A prospective study. EXCLI J. 2022, 21, 300–306. [Google Scholar]
- Olds, T.S.; Maher, C.A.; Matricciani, L. Sleep Duration or Bedtime? Exploring the Relationship between Sleep Habits and Weight Status and Activity Patterns. Sleep 2011, 34, 1299–1307. [Google Scholar] [CrossRef]
- Arora, T.; Taheri, S. Associations among late chronotype, body mass index and dietary behaviors in young adolescents. Int. J. Obes. 2014, 39, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Garaulet, M.; Scheer, F.A.J.L. Meal timing and obesity: Interactions with macronutrient intake and chronotype. Int. J. Obes. 2019, 43, 1701–1711. [Google Scholar] [CrossRef]
- Morris, C.J.; Purvis, T.E.; Hu, K.; Scheer, F.A.J.L. Circadian misalignment increases cardiovascular disease risk factors in humans. Proc. Natl. Acad. Sci. USA 2016, 113, E1402–E1411. [Google Scholar] [CrossRef] [PubMed]
- Scheer, F.A.J.L.; Hilton, M.F.; Mantzoros, C.S.; Shea, S.A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl. Acad. Sci. USA 2009, 106, 4453–4458. [Google Scholar] [CrossRef] [PubMed]
- Zhao, I.; Bogossian, F.; Song, S.; Turner, C. The Association Between Shift Work and Unhealthy Weight. J. Occup. Environ. Med. 2011, 53, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Rong, Y.; Huang, X.; Lai, H.; Luo, X.; Zhang, Z.; Liu, Y.; He, M.; Wu, T.; Chen, W. Shift Work and the Relationship with Metabolic Syndrome in Chinese Aged Workers. PLoS ONE 2015, 10, e0120632. [Google Scholar] [CrossRef]
- Kubo, T.; Oyama, I.; Nakamura, T.; Shirane, K.; Otsuka, H.; Kunimoto, M.; Kadowaki, K.; Maruyama, T.; Otomo, H.; Fujino, Y.; et al. Retrospective cohort study of the risk of obesity among shift workers: Findings from the Industry-based Shift Workers’ Health study, Japan. Occup. Environ. Med. 2010, 68, 327–331. [Google Scholar] [CrossRef]
- Canuto, R.; Pattussi, M.P.; Macagnan, J.B.A.; Henn, R.L.; Olinto, M.T.A. Sleep deprivation and obesity in shift workers in southern Brazil. Public Health Nutr. 2013, 17, 2619–2623. [Google Scholar] [CrossRef] [PubMed]
- Brum, M.C.B.; Dantas Filho, F.F.; Schnorr, C.C.; Bertoletti, O.A.; Bottega, G.B.; da Costa Rodrigues, T. Night shift work, short sleep and obesity. Diabetol. Metab. Syndr. 2020, 12, 13. [Google Scholar] [CrossRef]
- Hulsegge, G.; Proper, K.I.; Loef, B.; Paagman, H.; Anema, J.R.; van Mechelen, W. The mediating role of lifestyle in the relationship between shift work, obesity and diabetes. Int. Arch. Occup. Environ. Health 2021, 94, 1287–1295. [Google Scholar] [CrossRef]
- Halberg, N.; Henriksen, M.; Söderhamn, N.; Stallknecht, B.; Ploug, T.; Schjerling, P.; Dela, F. Effect of intermittent fasting and refeeding on insulin action in healthy men. J. Appl. Physiol. 2005, 99, 2128–2136. [Google Scholar] [CrossRef]
- Cousins, R.J. Nutritional regulation of gene expression. Am. J. Med. 1999, 106, 20S–23S, discussion 50S–51S. [Google Scholar] [CrossRef] [PubMed]
- Haro, D.; Marrero, P.; Relat, J. Nutritional Regulation of Gene Expression: Carbohydrate-, Fat- and Amino Acid-Dependent Modulation of Transcriptional Activity. Int. J. Mol. Sci. 2019, 20, 1386. [Google Scholar] [CrossRef] [PubMed]
- Mierziak, J.; Kostyn, K.; Boba, A.; Czemplik, M.; Kulma, A.; Wojtasik, W. Influence of the Bioactive Diet Components on the Gene Expression Regulation. Nutrients 2021, 13, 3673. [Google Scholar] [CrossRef]
- Shon, J.; Han, Y.; Park, Y.J. Effects of Dietary Fat to Carbohydrate Ratio on Obesity Risk Depending on Genotypes of Circadian Genes. Nutrients 2022, 14, 478. [Google Scholar] [CrossRef] [PubMed]
- Tognini, P.; Samad, M.; Kinouchi, K.; Liu, Y.; Helbling, J.-C.; Moisan, M.-P.; Eckel-Mahan, K.L.; Baldi, P.; Sassone-Corsi, P. Reshaping circadian metabolism in the suprachiasmatic nucleus and prefrontal cortex by nutritional challenge. Proc. Natl. Acad. Sci. USA 2020, 117, 29904–29913. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, J.; Pévet, P.; Challet, E. High-fat feeding alters the clock synchronization to light. J. Physiol. 2008, 586, 5901–5910. [Google Scholar] [CrossRef]
- Petrus, P.; Cervantes, M.; Samad, M.; Sato, T.; Chao, A.; Sato, S.; Koronowski, K.B.; Park, G.; Alam, Y.; Mejhert, N.; et al. Tryptophan metabolism is a physiological integrator regulating circadian rhythms. Mol. Metab. 2022, 64, 101556. [Google Scholar] [CrossRef]
- Jeong, K.; He, B.; Nohara, K.; Park, N.; Shin, Y.; Kim, S.; Shimomura, K.; Koike, N.; Yoo, S.H.; Chen, Z. Dual attenuation of proteasomal and autophagic BMAL1 degradation in ClockΔ19/+ mice contributes to improved glucose homeostasis. Sci. Rep. 2015, 5, 12801. [Google Scholar] [CrossRef] [PubMed]
- Kohsaka, A.; Laposky, A.D.; Ramsey, K.M.; Estrada, C.; Joshu, C.; Kobayashi, Y.; Turek, F.W.; Bass, J. High-Fat Diet Disrupts Behavioral and Molecular Circadian Rhythms in Mice. Cell Metab. 2007, 6, 414–421. [Google Scholar] [CrossRef]
- Barnea, M.; Madar, Z.; Froy, O. High-Fat Diet Delays and Fasting Advances the Circadian Expression of Adiponectin Signaling Components in Mouse Liver. Endocrinology 2009, 150, 161–168. [Google Scholar] [CrossRef]
- Cha, M.C.; Chou, C.J.; Boozer, C.N. High-fat diet feeding reduces the diurnal variation of plasma leptin concentration in rats. Metabolism 2000, 49, 503–507. [Google Scholar] [CrossRef]
- Pivovarova, O.; Jürchott, K.; Rudovich, N.; Hornemann, S.; Ye, L.; Möckel, S.; Murahovschi, V.; Kessler, K.; Seltmann, A.C.; Maser-Gluth, C.; et al. Changes of Dietary Fat and Carbohydrate Content Alter Central and Peripheral Clock in Humans. J. Clin. Endocrinol. Metab. 2015, 100, 2291–2302. [Google Scholar] [CrossRef] [PubMed]
- Sherman, H.; Gutman, R.; Chapnik, N.; Meylan, J.; le Coutre, J.; Froy, O. Caffeine alters circadian rhythms and expression of disease and metabolic markers. Int. J. Biochem. Cell Biol. 2011, 43, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.M.; Markwald, R.R.; McHill, A.W.; Chinoy, E.D.; Snider, J.A.; Bessman, S.C.; Jung, C.M.; O’Neill, J.S.; Wright, K.P. Effects of caffeine on the human circadian clock in vivo and in vitro. Sci. Transl. Med. 2015, 7, 305ra146. [Google Scholar] [CrossRef] [PubMed]
- Oishi, K.; Uchida, D.; Ohkura, N.; Doi, R.; Ishida, N.; Kadota, K.; Horie, S. Ketogenic Diet Disrupts the Circadian Clock and Increases Hypofibrinolytic Risk by Inducing Expression of Plasminogen Activator Inhibitor-1. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1571–1577. [Google Scholar] [CrossRef] [PubMed]
- Speed, J.S.; Hyndman, K.A.; Roth, K.J.; Heimlich, J.B.; Kasztan, M.; Fox, B.M.; Johnston, J.G.; Becker, B.K.; Jin, C.; Gamble, K.L.; et al. High dietary sodium causes dyssynchrony of the renal molecular clock in rats. Am. J. Physiol.-Ren. Physiol. 2018, 314, F89–F98. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J.; Skene, D.J. Melatonin as a chronobiotic. Sleep Med. Rev. 2005, 9, 25–39. [Google Scholar] [CrossRef]
- Guan, Q.; Wang, Z.; Cao, J.; Dong, Y.; Chen, Y. Mechanisms of Melatonin in Obesity: A Review. Int. J. Mol. Sci. 2021, 23, 218. [Google Scholar] [CrossRef]
- Delpino, F.M.; Figueiredo, L.M. Melatonin supplementation and anthropometric indicators of obesity: A systematic review and meta-analysis. Nutrition 2021, 91–92, 111399. [Google Scholar] [CrossRef]
- Gomes Domingos, A.L.; Hermsdorff, H.H.M.; Bressan, J. Melatonin intake and potential chronobiological effects on human health. Crit. Rev. Food Sci. Nutr. 2017, 59, 133–140. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2016, 20, 1689–1699. [Google Scholar] [CrossRef]
- Huang, J.; Lu, M.; Ho, C.-T. Health benefits of dietary chronobiotics: Beyond resynchronizing internal clocks. Food Funct. 2021, 12, 6136–6156. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W. Possible anti-obesity therapeutics from nature—A review. Phytochemistry 2010, 71, 1625–1641. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ávila-Román, J.; Soliz-Rueda, J.R.; Bravo, F.I.; Aragonès, G.; Suárez, M.; Arola-Arnal, A.; Mulero, M.; Salvadó, M.-J.; Arola, L.; Torres-Fuentes, C.; et al. Phenolic compounds and biological rhythms: Who takes the lead? Trends Food Sci. Technol. 2021, 113, 77–85. [Google Scholar] [CrossRef]
- Man, A.W.C.; Xia, N.; Daiber, A.; Li, H. The roles of gut microbiota and circadian rhythm in the cardiovascular protective effects of polyphenols. Br. J. Pharmacol. 2020, 177, 1278–1293. [Google Scholar] [CrossRef]
- Arola-Arnal, A.; Cruz-Carrión, Á.; Torres-Fuentes, C.; Ávila-Román, J.; Aragonès, G.; Mulero, M.; Bravo, F.I.; Muguerza, B.; Arola, L.; Suárez, M. Chrononutrition and Polyphenols: Roles and Diseases. Nutrients 2019, 11, 2602. [Google Scholar] [CrossRef] [PubMed]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; da Silva Pinto, M. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef] [PubMed]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Ana, F. Analytical Methods for Determining Bioavailability and Bioaccessibility of Bioactive Compounds from Fruits and Vegetables: A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, S.; Garau, C.; Esteban, S.; Nicolau, M.C.; Rivero, M.; Rial, R.V. Chrononutrition: Use of dissociated day/night infant milk formulas to improve the development of the wake–sleep rhythms. Eff. Tryptophan. Nutr. Neurosci. 2007, 10, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Li, L.; Wang, J.; Zhao, B.; Pan, J.; Wang, L.; Liu, X.; Liu, X.; Liu, Z. Resveratrol Prevents Acrylamide-Induced Mitochondrial Dysfunction and Inflammatory Responses via Targeting Circadian Regulator Bmal1 and Cry1 in Hepatocytes. J. Agric. Food Chem. 2019, 67, 8510–8519. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, Y.; Song, Y.; Cheng, X.-R.; Xia, S.; Rahman, M.R.T.; Shi, Y.; Le, G. Resveratrol restores the circadian rhythmic disorder of lipid metabolism induced by high-fat diet in mice. Biochem. Biophys. Res. Commun. 2015, 458, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Mi, Y.; Liu, Z.; Fan, R.; Qiao, Q.; Sun, Y.; Ren, B.; Liu, X. Dietary tea polyphenols ameliorate metabolic syndrome and memory impairment via circadian clock related mechanisms. J. Funct. Foods 2017, 34, 168–180. [Google Scholar] [CrossRef]
- Oike, H.; Kobori, M. Resveratrol Regulates Circadian Clock Genes in Rat-1 Fibroblast Cells. Biosci. Biotechnol. Biochem. 2008, 72, 3038–3040. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.; Portillo, M.P.; Madrid, J.A.; Arias, N.; Macarulla, M.T.; Garaulet, M. Effects of resveratrol on changes induced by high-fat feeding on clock genes in rats. Br. J. Nutr. 2013, 110, 1421–1428. [Google Scholar] [CrossRef]
- Li, J.; Wei, L.; Zhao, C.; Li, J.; Liu, Z.; Zhang, M.; Wang, Y. Resveratrol Maintains Lipid Metabolism Homeostasis via One of the Mechanisms Associated with the Key Circadian Regulator Bmal1. Molecules 2019, 24, 2916. [Google Scholar] [CrossRef]
- Okada, Y.; Okada, M. Quercetin, caffeic acid and resveratrol regulate circadian clock genes and aging-related genes in young and old human lung fibroblast cells. Mol. Biol. Rep. 2020, 47, 1021–1032. [Google Scholar] [CrossRef]
- Ribas-Latre, A.; Baselga-Escudero, L.; Casanova, E.; Arola-Arnal, A.; Salvadó, M.-J.; Bladé, C.; Arola, L. Dietary proanthocyanidins modulate BMAL1 acetylation, Nampt expression and NAD levels in rat liver. Sci. Rep. 2015, 5, 10954. [Google Scholar] [CrossRef]
- Jiang, P.; Xu, C.; Zhang, P.; Ren, J.; Mageed, F.; Wu, X.; Chen, L.; Zeb, F.; Feng, Q.; Li, S. Epigallocatechin-3-gallate inhibits self-renewal ability of lung cancer stem-like cells through inhibition of CLOCK. Int. J. Mol. Med. 2020, 46, 2216–2224. [Google Scholar] [CrossRef]
- Li, H.; Kek, H.C.; Lim, J.; Gelling, R.W.; Han, W. Green tea (-)-epigallocatechin-3-gallate counteracts daytime overeating induced by high-fat diet in mice. Mol. Nutr. Food Res. 2016, 60, 2565–2575. [Google Scholar] [CrossRef]
- Mi, Y.; Qi, G.; Fan, R.; Ji, X.; Liu, Z.; Liu, X. EGCG ameliorates diet-induced metabolic syndrome associating with the circadian clock. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2017, 1863, 1575–1589. [Google Scholar] [CrossRef]
- He, B.; Nohara, K.; Park, N.; Park, Y.-S.; Guillory, B.; Zhao, Z.; Garcia, J.M.; Koike, N.; Lee, C.C.; Takahashi, J.S.; et al. The small molecule Nobiletin targets the molecular oscillator to enhance circadian rhythms and protect against metabolic syndrome. Cell Metab. 2016, 23, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Guo, R.; Tian, H.; Li, L.; Liu, H.; Mi, Y.; Liu, X. Nobiletin protects against insulin resistance and disorders of lipid metabolism by reprogramming of circadian clock in hepatocytes. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2018, 1863, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, A.; Misawa, K.; Ikeda, Y.; Haraguchi, A.; Kamagata, M.; Tahara, Y.; Shibata, S. Potent Effects of Flavonoid Nobiletin on Amplitude, Period, and Phase of the Circadian Clock Rhythm in PER2::LUCIFERASE Mouse Embryonic Fibroblasts. PLoS ONE 2017, 12, e0170904. [Google Scholar] [CrossRef] [PubMed]
- Lellupitiyage Don, S.S.; Robertson, K.L.; Lin, H.-H.; Labriola, C.; Harrington, M.E.; Taylor, S.R.; Farkas, M.E. Nobiletin affects circadian rhythms and oncogenic characteristics in a cell-dependent manner. PLoS ONE 2020, 15, e0236315. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Nohara, K.; Wirianto, M.; Escobedo, G.; Lim, J.; Morales, R.; Yoo, S.-H.; Chen, Z. Effects of the Clock Modulator Nobiletin on Circadian Rhythms and Pathophysiology in Female Mice of an Alzheimer’s Disease Model. Biomolecules 2021, 11, 1004. [Google Scholar] [CrossRef]
- Larion, S.; Padgett, C.A.; Butcher, J.T.; Mintz, J.D.; Fulton, D.J.; Stepp, D.W. The biological clock enhancer nobiletin ameliorates steatosis in genetically obese mice by restoring aberrant hepatic circadian rhythm. Am. J. Physiol.-Gastrointest. Liver Physiol. 2022, 323, G387–G400. [Google Scholar] [CrossRef]
- Wirianto, M.; Wang, C.; Kim, E.; Koike, N.; Gomez-Gutierrez, R.; Nohara, K.; Escobedo, G., Jr.; Choi, J.M.; Han, C.; Yagita, K.; et al. The clock modulator Nobiletin mitigates astrogliosis-associated neuroinflammation and disease hallmarks in an Alzheimer’s disease model. FASEB J. 2022, 36, e22186. [Google Scholar] [CrossRef]
- Oyama, Y.; Bartman, C.M.; Gile, J.; Sehrt, D.; Eckle, T. The Circadian PER2 Enhancer Nobiletin Reverses the Deleterious Effects of Midazolam in Myocardial Ischemia and Reperfusion Injury. Curr. Pharm. Des. 2018, 24, 3376–3383. [Google Scholar] [CrossRef]
- Nohara, K.; Mallampalli, V.; Nemkov, T.; Wirianto, M.; Yang, J.; Ye, Y.; Sun, Y.; Han, L.; Esser, K.A.; Mileykovskaya, E.; et al. Nobiletin fortifies mitochondrial respiration in skeletal muscle to promote healthy aging against metabolic challenge. Nat. Commun. 2019, 10, 3923. [Google Scholar] [CrossRef]
- Petrenko, V.; Gandasi, N.R.; Sage, D.; Tengholm, A.; Barg, S.; Dibner, C. In pancreatic islets from type 2 diabetes patients, the dampened circadian oscillators lead to reduced insulin and glucagon exocytosis. Proc. Natl. Acad. Sci. USA 2020, 117, 2484–2495. [Google Scholar] [CrossRef]
- Guo, T.; Ho, C.-T.; Zhang, X.; Cao, J.; Wang, H.; Shao, X.; Pan, D.; Wu, Z. Oolong Tea Polyphenols Ameliorate Circadian Rhythm of Intestinal Microbiome and Liver Clock Genes in Mouse Model. J. Agric. Food Chem. 2019, 67, 11969–11976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yan, R.; Wu, Z. Metagenomics analysis of intestinal flora modulatory effect of green tea polyphenols by a circadian rhythm dysfunction mouse model. J. Food Biochem. 2020, 44, e13430. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Zhao, B.; Wang, Y.; Wu, D.; Wang, Y.; Yu, Y.; Yan, Y.; Zhang, W.; Liu, Z.; Liu, X. Cichoric Acid Prevents Free-Fatty-Acid-Induced Lipid Metabolism Disorders via Regulating Bmal1 in HepG2 Cells. J. Agric. Food Chem. 2018, 66, 9667–9678. [Google Scholar] [CrossRef]
- Numata, M.; Hirano, A.; Yamamoto, Y.; Yasuda, M.; Miura, N.; Sayama, K.; Shibata, M.A.; Asai, T.; Oku, N.; Miyoshi, N.; et al. Metastasis of Breast Cancer Promoted by Circadian Rhythm Disruption due to Light/Dark Shift and its Prevention by Dietary Quercetin in Mice. J. Circadian Rhythm. 2021, 19, 2. [Google Scholar] [CrossRef] [PubMed]
- Al Abdi, T.; Andreou, E.; Papageorgiou, A.; Heraclides, A.; Philippou, E. Personality, Chrono-nutrition and Cardiometabolic Health: A Narrative Review of the Evidence. Adv. Nutr. 2020, 11, 1201–1210. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, M.S.; Lee, J.S.; Halberg, F.E.; Halberg, J.; Schwartzkopff, O.; Cornélissen, G. A Tribute to Franz Halberg, MD. Hypertension 2015, 66, 1090–1092. [Google Scholar] [CrossRef]
- Cornelissen, G. When You Eat Matters: 60 Years of Franz Halberg’s Nutrition Chronomics. Open Nutraceuticals J. 2012, 5, 16–44. [Google Scholar] [CrossRef]
- Doi, R.; Oishi, K.; Ishida, N. CLOCK Regulates Circadian Rhythms of Hepatic Glycogen Synthesis through Transcriptional Activation ofGys2. J. Biol. Chem. 2010, 285, 22114–22121. [Google Scholar] [CrossRef]
- Ruddick-Collins, L.C.; Morgan, P.J.; Johnstone, A.M. Mealtime: A circadian disruptor and determinant of energy balance? J. Neuroendocrinol. 2020, 32, e12886. [Google Scholar] [CrossRef]
- Gill, S.; Panda, S. A Smartphone App Reveals Erratic Diurnal Eating Patterns in Humans that Can Be Modulated for Health Benefits. Cell Metab. 2015, 22, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Eating patterns of US adults: Meals, snacks, and time of eating. Physiol. Behav. 2018, 193, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.J.; Kumar, V.; Panda, S. A camera-phone based study reveals erratic eating pattern and disrupted daily eating-fasting cycle among adults in India. PLoS ONE 2017, 12, e0172852. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Rodríguez, V.A.; Rijo-Ferreira, F.; Green, C.B.; Takahashi, J.S. Importance of circadian timing for aging and longevity. Nat. Commun. 2021, 12, 2862. [Google Scholar] [CrossRef] [PubMed]
- Cienfuegos, S.; Gabel, K.; Kalam, F.; Ezpeleta, M.; Wiseman, E.; Pavlou, V.; Lin, S.; Oliveira, M.L.; Varady, K.A. Effects of 4- and 6-h Time-Restricted Feeding on Weight and Cardiometabolic Health: A Randomized Controlled Trial in Adults with Obesity. Cell Metab. 2020, 32, 366–378.e3. [Google Scholar] [CrossRef] [PubMed]
- Parr, E.B.; Devlin, B.L.; Radford, B.E.; Hawley, J.A. A Delayed Morning and Earlier Evening Time-Restricted Feeding Protocol for Improving Glycemic Control and Dietary Adherence in Men with Overweight/Obesity: A Randomized Controlled Trial. Nutrients 2020, 12, 505. [Google Scholar] [CrossRef]
- Parr, E.B.; Devlin, B.L.; Lim, K.H.C.; Moresi, L.N.Z.; Geils, C.; Brennan, L.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Time-Restricted Eating as a Nutrition Strategy for Individuals with Type 2 Diabetes: A Feasibility Study. Nutrients 2020, 12, 3228. [Google Scholar] [CrossRef]
- Gabel, K.; Hoddy, K.K.; Haggerty, N.; Song, J.; Kroeger, C.M.; Trepanowski, J.F.; Panda, S.; Varady, K.A. Effects of 8-hour time restricted feeding on body weight and metabolic disease risk factors in obese adults: A pilot study. Nutr. Healthy Aging 2018, 4, 345–353. [Google Scholar] [CrossRef]
- Moro, T.; Tinsley, G.; Bianco, A.; Marcolin, G.; Pacelli, Q.F.; Battaglia, G.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. J. Transl. Med. 2016, 14, 290. [Google Scholar] [CrossRef]
- Anton, S.D.; Lee, S.A.; Donahoo, W.T.; McLaren, C.; Manini, T.; Leeuwenburgh, C.; Pahor, M. The Effects of Time Restricted Feeding on Overweight, Older Adults: A Pilot Study. Nutrients 2019, 11, 1500. [Google Scholar] [CrossRef]
- Sutton, E.F.; Beyl, R.; Early, K.S.; Cefalu, W.T.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metab. 2018, 27, 1212–1221.e3. [Google Scholar] [CrossRef]
- Wilkinson, M.J.; Manoogian, E.N.C.; Zadourian, A.; Lo, H.; Fakhouri, S.; Shoghi, A.; Wang, X.; Fleischer, J.G.; Navlakha, S.; Panda, S.; et al. Ten-Hour Time-Restricted Eating Reduces Weight, Blood Pressure, and Atherogenic Lipids in Patients with Metabolic Syndrome. Cell Metab. 2020, 31, 92–104.e5. [Google Scholar] [CrossRef] [PubMed]
- Jamshed, H.; Beyl, R.A.; Della Manna, D.L.; Yang, E.S.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves 24-Hour Glucose Levels and Affects Markers of the Circadian Clock, Aging, and Autophagy in Humans. Nutrients 2019, 11, 1234. [Google Scholar] [CrossRef] [PubMed]
- Ravussin, E.; Beyl, R.A.; Poggiogalle, E.; Hsia, D.S.; Peterson, C.M. Early Time-Restricted Feeding Reduces Appetite and Increases Fat Oxidation but Does Not Affect Energy Expenditure in Humans. Obesity 2019, 27, 1244–1254. [Google Scholar] [CrossRef] [PubMed]
- Kotarsky, C.J.; Johnson, N.R.; Mahoney, S.J.; Mitchell, S.L.; Schimek, R.L.; Stastny, S.N.; Hackney, K.J. Time-restricted eating and concurrent exercise training reduces fat mass and increases lean mass in overweight and obese adults. Physiol. Rep. 2021, 9, e14868. [Google Scholar] [CrossRef] [PubMed]
- LeCheminant, J.; Christenson, E.; Bailey, B.; Tucker, L.A. Restricting night-time eating reduces daily energy intake in healthy young men:a short-term cross-over study. Br. J. Nutr. 2013, 110, 2108–2113. [Google Scholar] [CrossRef]
- Tinsley, G.; Forsse, J.S.; Butler, N.K.; Paoli, A.; Bane, A.A.; La Bounty, P.M.; Morgan, G.B.; Grandjean, P.W. Time-restricted feeding in young men performing resistance training: A randomized controlled trial. Eur. J. Sport Sci. 2016, 17, 200–207. [Google Scholar] [CrossRef]
- Antoni, R.; Johnston, K.L.; Collins, A.L.; Robertson, M.D. Intermittent v. continuous energy restriction: Differential effects on postprandial glucose and lipid metabolism following matched weight loss in overweight/obese participants. Br. J. Nutr. 2018, 119, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; Moore, M.L.; Graybeal, A.J.; Paoli, A.; Kim, Y.; Gonzales, J.U.; Harry, J.R.; VanDusseldorp, T.A.; Kennedy, D.N.; Cruz, M.R. Time-restricted feeding plus resistance training in active females: A randomized trial. Am. J. Clin. Nutr. 2019, 110, 628–640. [Google Scholar] [CrossRef]
- Kesztyüs, D.; Cermak, P.; Gulich, M.; Kesztyüs, T. Adherence to Time-Restricted Feeding and Impact on Abdominal Obesity in Primary Care Patients: Results of a Pilot Study in a Pre–Post Design. Nutrients 2019, 11, 2854. [Google Scholar] [CrossRef]
- McAllister, M.J.; Pigg, B.L.; Renteria, L.I.; Waldman, H.S. Time-restricted feeding improves markers of cardiometabolic health in physically active college-age men: A 4-week randomized pre-post pilot study. Nutr. Res. 2020, 75, 32–43. [Google Scholar] [CrossRef]
- Chow, L.S.; Manoogian, E.N.C.; Alvear, A.; Fleischer, J.G.; Thor, H.; Dietsche, K.; Wang, Q.; Hodges, J.S.; Esch, N.; Malaeb, S.; et al. Time-Restricted Eating Effects on Body Composition and Metabolic Measures in Humans who are Overweight: A Feasibility Study. Obesity 2020, 28, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.A.; Wu, N.; Rohdin-Bibby, L.; Moore, A.H.; Kelly, N.; Liu, Y.E.; Philip, E.; Vittinghoff, E.; Heymsfield, S.B.; Olgin, J.E.; et al. Effects of Time-Restricted Eating on Weight Loss and Other Metabolic Parameters in Women and Men with Overweight and Obesity: The TREAT Randomized Clinical Trial. JAMA Intern. Med. 2020, 180, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Qin, Y.-L.; Shi, Z.-Y.; Chen, J.-H.; Zeng, M.-J.; Zhou, W.; Chen, R.-Q.; Chen, Z.-Y. Effects of alternate-day fasting on body weight and dyslipidaemia in patients with non-alcoholic fatty liver disease: A randomised controlled trial. BMC Gastroenterol. 2019, 19, 219. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Xing, C.; Zhang, J.; Zhao, H.; Shi, W.; He, B. Eight-hour time-restricted feeding improves endocrine and metabolic profiles in women with anovulatory polycystic ovary syndrome. J. Transl. Med. 2021, 19, 148. [Google Scholar] [CrossRef]
- Martens, C.R.; Rossman, M.J.; Mazzo, M.R.; Jankowski, L.R.; Nagy, E.E.; Denman, B.A.; Richey, J.J.; Johnson, S.A.; Ziemba, B.P.; Wang, Y.; et al. Short-term time-restricted feeding is safe and feasible in non-obese healthy midlife and older adults. GeroScience 2020, 42, 667–686. [Google Scholar] [CrossRef]
- Peeke, P.M.; Greenway, F.L.; Billes, S.K.; Zhang, D.; Fujioka, K. Effect of time restricted eating on body weight and fasting glucose in participants with obesity: Results of a randomized, controlled, virtual clinical trial. Nutr. Diabetes 2021, 11, 6. [Google Scholar] [CrossRef]
- Phillips, N.; Mareschal, J.; Schwab, N.; Manoogian, E.; Borloz, S.; Ostinelli, G.; Gauthier-Jaques, A.; Umwali, S.; Rodriguez, E.G.; Aeberli, D.; et al. The Effects of Time-Restricted Eating versus Standard Dietary Advice on Weight, Metabolic Health and the Consumption of Processed Food: A Pragmatic Randomised Controlled Trial in Community-Based Adults. Nutrients 2021, 13, 1042. [Google Scholar] [CrossRef]
- Zeb, F.; Wu, X.; Chen, L.; Fatima, S.; Haq, I.-U.; Chen, A.; Majeed, F.; Feng, Q.; Li, M. Effect of time-restricted feeding on metabolic risk and circadian rhythm associated with gut microbiome in healthy males. Br. J. Nutr. 2020, 123, 1216–1226. [Google Scholar] [CrossRef]
- Jones, R.; Pabla, P.; Mallinson, J.; Nixon, A.; Taylor, T.; Bennett, A.; Tsintzas, K. Two weeks of early time-restricted feeding (eTRF) improves skeletal muscle insulin and anabolic sensitivity in healthy men. Am. J. Clin. Nutr. 2020, 112, 1015–1028. [Google Scholar] [CrossRef]
- Karras, S.N.; Koufakis, T.; Adamidou, L.; Antonopoulou, V.; Karalazou, P.; Thisiadou, K.; Mitrofanova, E.; Mulrooney, H.; Petróczi, A.; Zebekakis, P.; et al. Effects of orthodox religious fasting versus combined energy and time restricted eating on body weight, lipid concentrations and glycaemic profile. Int. J. Food Sci. Nutr. 2020, 72, 82–92. [Google Scholar] [CrossRef]
- Hutchison, A.T.; Regmi, P.; Manoogian, E.N.; Fleischer, J.G.; Wittert, G.A.; Panda, S.; Heilbronn, L.K. Time-Restricted Feeding Improves Glucose Tolerance in Men at Risk for Type 2 Diabetes: A Randomized Crossover Trial. Obesity 2019, 27, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Sun, Y.; Ye, Y.; Hu, D.; Zhang, H.; He, Z.; Zhao, H.; Yang, H.; Mao, Y. Randomized controlled trial for time-restricted eating in healthy volunteers without obesity. Nat. Commun. 2022, 13, 1003. [Google Scholar] [CrossRef] [PubMed]
- Jakubowicz, D.; Barnea, M.; Wainstein, J.; Froy, O. High Caloric intake at breakfast vs. dinner differentially influences weight loss of overweight and obese women. Obesity 2013, 21, 2504–2512. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Musso, G.; Beccuti, G.; Fadda, M.; Fedele, D.; Gambino, R.; Gentile, L.; Durazzo, M.; Ghigo, E.; Cassader, M. Consuming More of Daily Caloric Intake at Dinner Predisposes to Obesity. A 6-Year Population-Based Prospective Cohort Study. Claret M, editor. PLoS ONE 2014, 9, e108467. [Google Scholar] [CrossRef] [PubMed]
- do Nascimento Queiroz, J.; Macedo, R.C.O.; Tinsley, G.M.; Reischak-Oliveira, A. Time-restricted eating and circadian rhythms: The biological clock is ticking. Crit. Rev. Food Sci. Nutr. 2020, 61, 2863–2875. [Google Scholar] [CrossRef]
- Froy, O. Metabolism and Circadian Rhythms—Implications for Obesity. Endocr. Rev. 2010, 31, 1–24. [Google Scholar] [CrossRef]
- Most, J.; Tosti, V.; Redman, L.M.; Fontana, L. Calorie restriction in humans: An update. Ageing Res. Rev. 2017, 39, 36–45. [Google Scholar] [CrossRef]
- Wing, R.R.; Blair, E.H.; Bononi, P.; Marcus, M.D.; Watanabe, R.; Bergman, R.N. Caloric Restriction Per Se Is a Significant Factor in Improvements in Glycemic Control and Insulin Sensitivity During Weight Loss in Obese NIDDM Patients. Diabetes Care 1994, 17, 30–36. [Google Scholar] [CrossRef]
- Fontana, L.; Meyer, T.E.; Klein, S.; Holloszy, J.O. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 6659–6663. [Google Scholar] [CrossRef]
- Colman, R.J.; Beasley, T.M.; Kemnitz, J.W.; Johnson, S.C.; Weindruch, R.; Anderson, R.M. Caloric restriction reduces age-related and all-cause mortality in rhesus monkeys. Nat. Commun. 2014, 5, 3557. [Google Scholar] [CrossRef]
- Mattison, J.A.; Colman, R.J.; Beasley, T.M.; Allison, D.B.; Kemnitz, J.W.; Roth, G.S.; Ingram, D.K.; Weindruch, R.; de Cabo, R.; Anderson, R. Caloric restriction improves health and survival of rhesus monkeys. Nat. Commun. 2017, 8, 14063. [Google Scholar] [CrossRef] [PubMed]
- Rynders, C.A.; Thomas, E.A.; Zaman, A.; Pan, Z.; Catenacci, V.A.; Melanson, E.L. Effectiveness of Intermittent Fasting and Time-Restricted Feeding Compared to Continuous Energy Restriction for Weight Loss. Nutrients 2019, 11, 2442. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.J.; VanWormer, J.J.; Crain, A.L.; Boucher, J.L.; Histon, T.; Caplan, W.; Bowman, J.D.; Pronk, N.P. Weight-Loss Outcomes: A Systematic Review and Meta-Analysis of Weight-Loss Clinical Trials with a Minimum 1-Year Follow-Up. J. Am. Diet. Assoc. 2007, 107, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Xie, C.; Lu, S.; Nichols, R.G.; Tian, Y.; Li, L.; Patel, D.; Ma, Y.; Brocker, C.N.; Yan, T.; et al. Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota. Cell Metab. 2017, 26, 672–685.e4. [Google Scholar] [CrossRef]
- Patterson, R.E.; Sears, D.D. Metabolic Effects of Intermittent Fasting. Annu. Rev. Nutr. 2017, 37, 371–393. [Google Scholar] [CrossRef]
- Flanagan, A.; Bechtold, D.A.; Pot, G.K.; Johnston, J.D. Chrono-nutrition: From molecular and neuronal mechanisms to human epidemiology and timed feeding patterns. J. Neurochem. 2020, 157, 53–72. [Google Scholar] [CrossRef]
- Lessan, N.; Ali, T. Energy Metabolism and Intermittent Fasting: The Ramadan Perspective. Nutrients 2019, 11, 1192. [Google Scholar] [CrossRef]
- Anton, S.D.; Moehl, K.; Donahoo, W.T.; Marosi, K.; Lee, S.A.; Mainous, A.G., 3rd; Leeuwenburgh, C.; Mattson, M.P. Flipping the Metabolic Switch: Understanding and Applying the Health Benefits of Fasting. Obesity 2018, 26, 254–268. [Google Scholar] [CrossRef]
- Vasim, I.; Majeed, C.N.; DeBoer, M.D. Intermittent Fasting and Metabolic Health. Nutrients 2022, 14, 631. [Google Scholar] [CrossRef]
- Carter, S.; Clifton, P.M.; Keogh, J.B. Effect of Intermittent Compared with Continuous Energy Restricted Diet on Glycemic Control in Patients with Type 2 Diabetes. JAMA Netw. Open 2018, 1, e180756. [Google Scholar] [CrossRef]
- Catenacci, V.A.; Pan, Z.; Ostendorf, D.; Brannon, S.; Gozansky, W.S.; Mattson, M.P.; Martin, B.; MacLean, P.S.; Melanson, E.L.; Donahoo, W.T. A randomized pilot study comparing zero-calorie alternate-day fasting to daily caloric restriction in adults with obesity. Obesity 2016, 24, 1874–1883. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.N.; Pegington, M.; Mattson, M.P.; Frystyk, J.; Dillon, B.; Evans, G.; Cuzick, J.; Jebb, S.A.; Martin, B.; Cutler, R.G.; et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. Int. J. Obes. 2010, 35, 714–727. [Google Scholar] [CrossRef] [PubMed]
- Headland, M.L.; Clifton, P.M.; Keogh, J.B. Effect of intermittent compared to continuous energy restriction on weight loss and weight maintenance after 12 months in healthy overweight or obese adults. Int. J. Obes. 2018, 43, 2028–2036. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, A.T.; Liu, B.; Wood, R.E.; Vincent, A.D.; Thompson, C.H.; O’Callaghan, N.J.; Wittert, G.A.; Heilbronn, L.K. Effects of Intermittent Versus Continuous Energy Intakes on Insulin Sensitivity and Metabolic Risk in Women with Overweight. Obesity 2018, 27, 50–58. [Google Scholar] [CrossRef]
- Trepanowski, J.F.; Kroeger, C.M.; Barnosky, A.; Klempel, M.C.; Bhutani, S.; Hoddy, K.K.; Gabel, K.; Freels, S.; Rigdon, J.; Rood, J.; et al. Effect of Alternate-Day Fasting on Weight Loss, Weight Maintenance, and Cardioprotection Among Metabolically Healthy Obese Adults. JAMA Intern. Med. 2017, 177, 930. [Google Scholar] [CrossRef]
- Schübel, R.; Nattenmüller, J.; Sookthai, D.; Nonnenmacher, T.; Graf, M.E.; Riedl, L.; Schlett, C.L.; von Stackelberg, O.; Johnson, T.; Nabers, D.; et al. Effects of intermittent and continuous calorie restriction on body weight and metabolism over 50 wk: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 933–945. [Google Scholar] [CrossRef]
- Di Francesco, A.; Di Germanio, C.; Bernier, M.; de Cabo, R. A time to fast. Science 2018, 362, 770–775. [Google Scholar] [CrossRef]
- Mitchell, S.J.; Bernier, M.; Mattison, J.A.; Aon, M.A.; Kaiser, T.A.; Anson, R.M.; Ikeno, Y.; Anderson, R.M.; Ingram, D.K.; de Cabo, R. Daily Fasting Improves Health and Survival in Male Mice Independent of Diet Composition and Calories. Cell Metab. 2019, 29, 221–228.e3. [Google Scholar] [CrossRef]
- Chaix, A.; Manoogian, E.N.C.; Melkani, G.C.; Panda, S. Time-Restricted Eating to Prevent and Manage Chronic Metabolic Diseases. Annu. Rev. Nutr. 2019, 39, 291–315. [Google Scholar] [CrossRef]
- Hatori, M.; Vollmers, C.; Zarrinpar, A.; DiTacchio, L.; Bushong, E.A.; Gill, S.; Leblanc, M.; Chaix, A.; Joens, M.; Fitzpatrick, J.A.; et al. Time-Restricted Feeding without Reducing Caloric Intake Prevents Metabolic Diseases in Mice Fed a High-Fat Diet. Cell Metab. 2012, 15, 848–860. [Google Scholar] [CrossRef]
- Regmi, P.; Heilbronn, L.K. Time-Restricted Eating: Benefits, Mechanisms, and Challenges in Translation. iScience 2020, 23, 101161. [Google Scholar] [CrossRef] [PubMed]
- Boyd, P.; O’Connor, S.G.; Heckman-Stoddard, B.M.; Sauter, E.R. Time-Restricted Feeding Studies and Possible Human Benefit. JNCI Cancer Spectr. 2022, 6, pkac032. [Google Scholar] [CrossRef] [PubMed]
- Chaix, A.; Zarrinpar, A.; Miu, P.; Panda, S. Time-Restricted Feeding Is a Preventative and Therapeutic Intervention against Diverse Nutritional Challenges. Cell Metab. 2014, 20, 991–1005. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M.K.; Choi, M.H.; Kulseng, B.; Zhao, C.-M.; Chen, D. Time-restricted feeding on weekdays restricts weight gain: A study using rat models of high-fat diet-induced obesity. Physiol. Behav. 2017, 173, 298–304. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, S.G.; Boyd, P.; Bailey, C.P.; Shams-White, M.M.; Agurs-Collins, T.; Hall, K.; Reedy, J.; Sauter, E.R.; Czajkowski, S.M. Perspective: Time-Restricted Eating Compared with Caloric Restriction: Potential Facilitators and Barriers of Long-Term Weight Loss Maintenance. Adv. Nutr. 2021, 12, 325–333. [Google Scholar] [CrossRef]
- Parr, E.B.; Devlin, B.L.; Hawley, J.A. Time-Restricted Eating: Integrating the What with the When. Adv. Nutr. 2022, 13, 699–711. [Google Scholar] [PubMed]
- Isenmann, E.; Dissemond, J.; Geisler, S. The Effects of a Macronutrient-Based Diet and Time-Restricted Feeding (16:8) on Body Composition in Physically Active Individuals—A 14-Week Randomised Controlled Trial. Nutrients 2021, 13, 3122. [Google Scholar] [CrossRef]
- Adafer, R.; Messaadi, W.; Meddahi, M.; Patey, A.; Haderbache, A.; Bayen, S.; Messaadi, N. Food Timing, Circadian Rhythm and Chrononutrition: A Systematic Review of Time-Restricted Eating’s Effects on Human Health. Nutrients 2020, 12, 3770. [Google Scholar] [CrossRef]
- Lanfray, D.; Richard, D. Emerging Signaling Pathway in Arcuate Feeding-Related Neurons: Role of the Acbd7. Front. Neurosci. 2017, 11, 328. [Google Scholar] [CrossRef]
- Varela, L.; Horvath, T.L. Leptin and insulin pathways in POMC and AgRP neurons that modulate energy balance and glucose homeostasis. EMBO Rep. 2012, 13, 1079–1086. [Google Scholar] [CrossRef]
- Silva, A.D.; Bloom, S.R. Gut Hormones and Appetite Control: A Focus on PYY and GLP-1 as Therapeutic Targets in Obesity. Gut Liver 2012, 6, 10–20. [Google Scholar] [CrossRef]
- Yanagi, S.; Sato, T.; Kangawa, K.; Nakazato, M. The Homeostatic Force of Ghrelin. Cell Metab. 2018, 27, 786–804. [Google Scholar] [CrossRef] [PubMed]
- LeSauter, J.; Hoque, N.; Weintraub, M.; Pfaff, D.W.; Silver, R. Stomach ghrelin-secreting cells as food-entrainable circadian clocks. Proc. Natl. Acad. Sci. USA 2009, 106, 13582–13587. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, H.; Tong, J.; Tschöp, M.H.; Pfluger, P.T. Ghrelin and PYY in the regulation of energy balance and metabolism: Lessons from mouse mutants. Am. J. Physiol.-Endocrinol. Metab. 2010, 298, E909–E919. [Google Scholar] [CrossRef] [PubMed]
- Bodosi, B.; Gardi, J.; Hajdu, I.; Szentirmai, E.; Obal, F.; Krueger, J.M. Rhythms of ghrelin, leptin, and sleep in rats: Effects of the normal diurnal cycle, restricted feeding, and sleep deprivation. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2004, 287, R1071–R1079. [Google Scholar] [CrossRef]
- Sinha, M.K.; Ohannesian, J.P.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Magosin, S.; Marco, C.; Caro, J.F. Nocturnal rise of leptin in lean, obese, and non-insulin-dependent diabetes mellitus subjects. J. Clin. Investig. 1996, 97, 1344–1347. [Google Scholar] [CrossRef]
- Suriagandhi, V.; Nachiappan, V. Protective Effects of Melatonin against Obesity-Induced by Leptin Resistance. Behav. Brain Res. 2022, 417, 113598. [Google Scholar] [CrossRef]
- Kettner Nicole, M.; Mayo Sara, A.; Hua, J.; Lee, C.; Moore David, D.; Fu, L. Circadian Dysfunction Induces Leptin Resistance in Mice. Cell Metab. 2015, 22, 448–459. [Google Scholar] [CrossRef]
- Shapiro, A.; Mu, W.; Roncal, C.; Cheng, K.-Y.; Johnson, R.J.; Scarpace, P.J. Fructose-induced leptin resistance exacerbates weight gain in response to subsequent high-fat feeding. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2008, 295, R1370–R1375. [Google Scholar] [CrossRef]
- Lin, S.; Thomas, T.; Storlien, L.; Huang, X. Development of high fat diet-induced obesity and leptin resistance in C57Bl/6J mice. Int. J. Obes. 2000, 24, 639–646. [Google Scholar] [CrossRef]
- Chan, J.L.; Heist, K.; DePaoli, A.M.; Veldhuis, J.D.; Mantzoros, C.S. The role of falling leptin levels in the neuroendocrine and metabolic adaptation to short-term starvation in healthy men. J. Clin. Investig. 2003, 111, 1409–1421. [Google Scholar] [CrossRef] [PubMed]
- Spezani, R.; da Silva, R.R.; Martins, F.F.; de Souza Marinho, T.; Aguila, M.B.; Mandarim-de-Lacerda, C.A. Intermittent fasting, adipokines, insulin sensitivity, and hypothalamic neuropeptides in a dietary overload with high-fat or high-fructose diet in mice. J. Nutr. Biochem. 2020, 83, 108419. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Hong, N.; Kim, K.-W.; Cho, S.J.; Lee, M.; Lee, Y.-H.; Lee, Y.H.; Kang, E.S.; Cha, B.S.; Lee, B.W. The Effectiveness of Intermittent Fasting to Reduce Body Mass Index and Glucose Metabolism: A Systematic Review and Meta-Analysis. J. Clin. Med. 2019, 8, 1645. [Google Scholar] [CrossRef] [PubMed]
- Albosta, M.; Bakke, J. Intermittent fasting: Is there a role in the treatment of diabetes? A review of the literature and guide for primary care physicians. Clin. Diabetes Endocrinol. 2021, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Kahn, B.B.; Alquier, T.; Carling, D.; Hardie, D.G. AMP-activated protein kinase: Ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 2005, 1, 15–25. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Alquier, T.; Furukawa, N.; Kim, Y.-B.; Lee, A.; Xue, B.; Mu, J.; Foufelle, F.; Ferré, P.; Birnbaum, M.J.; et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 2004, 428, 569–574. [Google Scholar] [CrossRef]
- Scarpace, P.J.; Matheny, M.; Pollock, B.H.; Tümer, N. Leptin increases uncoupling protein expression and energy expenditure. Am. J. Physiol. 1997, 273 Pt 1, E226–E230. [Google Scholar] [CrossRef]
- Biancolin, A.D.; Martchenko, A.; Mitova, E.; Gurges, P.; Michalchyshyn, E.; Chalmers, J.A.; Doria, A.; Mychaleckyj, J.C.; Adriaenssens, A.E.; Reimann, F.; et al. The core clock gene, Bmal1, and its downstream target, the SNARE regulatory protein secretagogin, are necessary for circadian secretion of glucagon-like peptide-1. Mol. Metab. 2020, 31, 124–137. [Google Scholar] [CrossRef]
- Hill, B.R.; De Souza, M.J.; Williams, N.I. Characterization of the diurnal rhythm of peptide YY and its association with energy balance parameters in normal-weight premenopausal women. Am. J. Physiol.-Endocrinol. Metab. 2011, 301, E409–E415. [Google Scholar] [CrossRef]
- Wever, M.C.; van Meer, F.; Charbonnier, L.; Crabtree, D.R.; Buosi, W.; Giannopoulou, A.; Androutsos, O.; Johnstone, A.M.; Manios, Y.; Meek, C.L.; et al. Associations between ghrelin and leptin and neural food cue reactivity in a fasted and sated state. NeuroImage 2021, 240, 118374. [Google Scholar] [CrossRef]
- Cummings, D.E.; Weigle, D.S.; Frayo, R.S.; Breen, P.A.; Ma, M.K.; Dellinger, E.P.; Purnell, J.Q. Plasma Ghrelin Levels after Diet-Induced Weight Loss or Gastric Bypass Surgery. N. Engl. J. Med. 2002, 346, 1623–1630. [Google Scholar] [CrossRef] [PubMed]
- Sumithran, P.; Prendergast, L.A.; Delbridge, E.; Purcell, K.; Shulkes, A.; Kriketos, A.; Proietto, J. Long-Term Persistence of Hormonal Adaptations to Weight Loss. N. Engl. J. Med. 2011, 365, 1597–1604. [Google Scholar] [CrossRef] [PubMed]
- Lean, M.E.J.; Malkova, D. Altered gut and adipose tissue hormones in overweight and obese individuals: Cause or consequence? Int. J. Obes. 2015, 40, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Al-Rawi, N.; Madkour, M.; Jahrami, H.; Salahat, D.; Alhasan, F.; BaHammam, A.; Faris, M.A.-I. Effect of diurnal intermittent fasting during Ramadan on ghrelin, leptin, melatonin, and cortisol levels among overweight and obese subjects: A prospective observational study. PLoS ONE 2020, 15, e0237922. [Google Scholar] [CrossRef] [PubMed]
- Alzoghaibi, M.A.; Pandi-Perumal, S.R.; Sharif, M.M.; BaHammam, A.S. Diurnal Intermittent Fasting during Ramadan: The Effects on Leptin and Ghrelin Levels. PLoS ONE 2014, 9, e92214. [Google Scholar] [CrossRef]
- Omisade, A.; Buxton, O.M.; Rusak, B. Impact of acute sleep restriction on cortisol and leptin levels in young women. Physiol. Behav. 2010, 99, 651–656. [Google Scholar] [CrossRef]
- Nedeltcheva, A.V.; Kilkus, J.M.; Imperial, J.; Schoeller, D.A.; Penev, P.D. Insufficient Sleep Undermines Dietary Efforts to Reduce Adiposity. Ann. Intern. Med. 2010, 153, 435. [Google Scholar] [CrossRef]
- Hayes, A.L.; Xu, F.; Babineau, D.; Patel, S.R. Sleep Duration and Circulating Adipokine Levels. Sleep 2011, 34, 147–152. [Google Scholar] [CrossRef]
- Ali, R.; Tariq, S.; Kareem, O.; Fayaz, F.; Aziz, T.; Pottoo, F.H.; Siddiqui, N. Nutraceuticals for Sleep Disorders. Comb. Chem. High Throughput Screen. 2021, 24, 1583–1592. [Google Scholar] [CrossRef]
- Godos, J.; Ferri, R.; Castellano, S.; Angelino, D.; Mena, P.; Del Rio, D.; Caraci, F.; Galvano, F.; Grosso, G. Specific Dietary (Poly)phenols Are Associated with Sleep Quality in a Cohort of Italian Adults. Nutrients 2020, 12, 1226. [Google Scholar] [CrossRef]
- Cantó, C.; Auwerx, J. Calorie Restriction: Is AMPK a Key Sensor and Effector? Physiology 2011, 26, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.-J.; Logunova, L.; Maywood, E.S.; Gallego, M.; Lebiecki, J.; Brown, T.M.; Sládek, M.; Semikhodskii, A.S.; Glossop, N.R.; Piggins, H.D.; et al. Setting Clock Speed in Mammals: The CK1ε tau Mutation in Mice Accelerates Circadian Pacemakers by Selectively Destabilizing PERIOD Proteins. Neuron 2008, 58, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Lamia, K.A.; Sachdeva, U.M.; DiTacchio, L.; Williams, E.C.; Alvarez, J.G.; Egan, D.F.; Vasquez, D.S.; Juguilon, H.; Panda, S.; Shaw, R.J.; et al. AMPK Regulates the Circadian Clock by Cryptochrome Phosphorylation and Degradation. Science 2009, 326, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Sherman, H.; Genzer, Y.; Cohen, R.; Chapnik, N.; Madar, Z.; Froy, O. Timed high-fat diet resets circadian metabolism and prevents obesity. FASEB J. 2012, 26, 3493–3502. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B. NAMPT-Mediated NAD Biosynthesis as the Internal Timing Mechanism: In NAD+ World, Time Is Running in Its Own Way. Rejuvenation Res. 2018, 21, 210–224. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Yoshino, J. Adipose tissue NAD+biology in obesity and insulin resistance: From mechanism to therapy. BioEssays 2017, 39, 1600227. [Google Scholar] [CrossRef]
- Levine, D.C.; Ramsey, K.M.; Bass, J. Circadian NAD(P)(H) cycles in cell metabolism. Semin. Cell Dev. Biol. 2022, 126, 15–26. [Google Scholar] [CrossRef]
- Ramsey, K.M.; Yoshino, J.; Brace, C.S.; Abrassart, D.; Kobayashi, Y.; Marcheva, B.; Hong, H.-K.; Chong, J.L.; Buhr, E.D.; Lee, C.; et al. Circadian Clock Feedback Cycle Through NAMPT-Mediated NAD+ Biosynthesis. Science 2009, 324, 651–654. [Google Scholar] [CrossRef]
- Fulco, M.; Cen, Y.; Zhao, P.; Hoffman, E.P.; McBurney, M.W.; Sauve, A.A.; Sartorelli, A. Glucose Restriction Inhibits Skeletal Myoblast Differentiation by Activating SIRT1 through AMPK-Mediated Regulation of Nampt. Dev. Cell 2008, 14, 661–673. [Google Scholar] [CrossRef]
- Yoshino, J.; Mills Kathryn, F.; Yoon, M.; Imai, S. Nicotinamide Mononucleotide, a Key NAD+ Intermediate, Treats the Pathophysiology of Diet- and Age-Induced Diabetes in Mice. Cell Metab. 2011, 14, 528–536. [Google Scholar] [CrossRef]
- Canto, C.; Jiang, L.Q.; Deshmukh, A.S.; Mataki, C.; Coste, A.; Lagouge, M.; Zierath, J.R.; Auwerx, J. Interdependence of AMPK and SIRT1 for Metabolic Adaptation to Fasting and Exercise in Skeletal Muscle. Cell Metab. 2010, 11, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Nakahata, Y.; Sahar, S.; Astarita, G.; Kaluzova, M.; Sassone-Corsi, P. Circadian Control of the NAD+ Salvage Pathway by CLOCK-SIRT1. Science 2009, 324, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B.; Kovač, V.; Milisav, I. Healthy Lifestyle Recommendations: Do the Beneficial Effects Originate from NAD+ Amount at the Cellular Level? Myers JN, editor. Oxidative Med. Cell. Longev. 2020, 2020, 8819627. [Google Scholar] [CrossRef]
- Houtkooper, R.H.; Pirinen, E.; Auwerx, J. Sirtuins as regulators of metabolism and healthspan. Nat. Rev. Mol. Cell Biol. 2012, 13, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Bordone, L.; Guarente, L. Calorie restriction, SIRT1 and metabolism: Understanding longevity. Nat. Rev. Mol. Cell Biol. 2005, 6, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Guarente, L. Sirtuins and calorie restriction. Nat. Rev. Mol. Cell Biol. 2012, 13, 207. [Google Scholar] [CrossRef]
- Guarente, L. Calorie restriction and sirtuins revisited. Genes Dev. 2013, 27, 2072–2085. [Google Scholar] [CrossRef]
- Yu, W.; Qin, J.; Chen, C.; Fu, Y.; Wang, W. Moderate calorie restriction attenuates age-associated alterations and improves cardiac function by increasing SIRT1 and SIRT3 expression. Mol. Med. Rep. 2018, 18, 4087–4094. [Google Scholar] [CrossRef]
- Cantó, C. Dietary restriction and Sirtuin 1 in metabolic health: Connections and divergences. Proc. Nutr. Soc. 2015, 75, 30–37. [Google Scholar] [CrossRef]
- Watroba, M.; Szukiewicz, D. Sirtuins at the Service of Healthy Longevity. Front. Physiol. 2021, 12, 724506. [Google Scholar] [CrossRef]
- Asher, G.; Gatfield, D.; Stratmann, M.; Reinke, H.; Dibner, C.; Kreppel, F.; Mostoslavsky, R.; Alt, F.W.; Schibler, U. SIRT1 Regulates Circadian Clock Gene Expression through PER2 Deacetylation. Cell 2008, 134, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Nakahata, Y.; Kaluzova, M.; Grimaldi, B.; Sahar, S.; Hirayama, J.; Chen, D.; Guarente, L.P.; Sassone-Corsi, P. The NAD+-Dependent Deacetylase SIRT1 Modulates CLOCK-Mediated Chromatin Remodeling and Circadian Control. Cell 2008, 134, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-C.; Guarente, L. SIRT1 Mediates Central Circadian Control in the SCN by a Mechanism that Decays with Aging. Cell 2013, 153, 1448–1460. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.-H.; Zhao, T.; Cui, K.; Hu, G.; Chen, Q.; Chen, W.; Wang, X.-W.; Soto-Gutierrez, A.; Zhao, K.; Deng, C.-X. Negative reciprocal regulation between Sirt1 and Per2 modulates the circadian clock and aging. Sci. Rep. 2016, 6, 28633. [Google Scholar] [CrossRef]
- Terada, S.; Goto, M.; Kato, M.; Kawanaka, K.; Shimokawa, T.; Tabata, I. Effects of low-intensity prolonged exercise on PGC-1 mRNA expression in rat epitrochlearis muscle. Biochem. Biophys. Res. Commun. 2002, 296, 350–354. [Google Scholar] [CrossRef]
- Cantó, C.; Gerhart-Hines, Z.; Feige, J.N.; Lagouge, M.; Noriega, L.; Milne, J.C.; Elliott, P.J.; Puigserver, P.; Auwerx, J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 2009, 458, 1056–1060. [Google Scholar] [CrossRef]
- Jäger, S.; Handschin, C.; St-Pierre, J.; Spiegelman, B.M. AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1alpha. Proc. Natl. Acad. Sci. USA 2007, 104, 12017–12022. [Google Scholar] [CrossRef]
- Borra, M.T.; Smith, B.C.; Denu, J.M. Mechanism of human SIRT1 activation by resveratrol. J. Biol. Chem. 2005, 280, 17187–17195. [Google Scholar] [CrossRef]
- Gertz, M.; Nguyen, G.T.T.; Fischer, F.; Suenkel, B.; Schlicker, C.; Fränzel, B.; Tomaschewski, J.; Aladini, F.; Becker, C.; Wolters, D.; et al. A Molecular Mechanism for Direct Sirtuin Activation by Resveratrol. PLoS ONE 2012, 7, e49761. [Google Scholar] [CrossRef]
- Iside, C.; Scafuro, M.; Nebbioso, A.; Altucci, L. SIRT1 Activation by Natural Phytochemicals: An Overview. Front. Pharmacol. 2020, 11, 1225. [Google Scholar] [CrossRef]
- Akan, O.D.; Qin, D.; Guo, T.; Lin, Q.; Luo, F. Sirtfoods: New Concept Foods, Functions, and Mechanisms. Foods 2022, 11, 2955. [Google Scholar] [CrossRef] [PubMed]
- Pallauf, K.; Giller, K.; Huebbe, P.; Rimbach, G. Nutrition and Healthy Ageing: Calorie Restriction or Polyphenol-Rich “MediterrAsian” Diet? Oxidative Med. Cell. Longev. 2013, 2013, 707421. [Google Scholar] [CrossRef]
- Chung, S.; Yao, H.; Caito, S.; Hwang, J.; Arunachalam, G.; Rahman, I. Regulation of SIRT1 in cellular functions: Role of polyphenols. Arch. Biochem. Biophys. 2010, 501, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Sarubbo, F.; Esteban, S.; Miralles, A.; Moranta, D. Effects of Resveratrol and other Polyphenols on Sirt1: Relevance to Brain Function During Aging. Curr. Neuropharmacol. 2018, 16, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Xu, H.; Xie, Z.; Wang, L.; Sun, Y.; Yang, H.; Hu, D.; Mao, Y. Time-Restricted Feeding Reduces the Detrimental Effects of a High-Fat Diet, Possibly by Modulating the Circadian Rhythm of Hepatic Lipid Metabolism and Gut Microbiota. Front. Nutr. 2020, 7, 596285. [Google Scholar] [CrossRef]
- Cantó, C.; Menzies Keir, J.; Auwerx, J. NAD+ Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Yang, Y.; Sauve, A.A. NAD+ metabolism: Bioenergetics, signaling and manipulation for therapy. Biochim. Biophys. Acta 2016, 1864, 1787–1800. [Google Scholar] [CrossRef]
- Bai, P.; Cantó, C.; Oudart, H.; Brunyánszki, A.; Cen, Y.; Thomas, C.; Yamamoto, H.; Huber, A.; Kiss, B.; Houtkooper, R.H.; et al. PARP-1 Inhibition Increases Mitochondrial Metabolism through SIRT1 Activation. Cell Metab. 2011, 13, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Szántó, M.; Bai, P. The role of ADP-ribose metabolism in metabolic regulation, adipose tissue differentiation, and metabolism. Genes Dev. 2020, 34, 321–340. [Google Scholar] [CrossRef]
- Vernochet, C.; Peres, S.B.; Davis, K.E.; McDonald, M.E.; Qiang, L.; Wang, H.; Scherer, P.E.; Farmer, S.R. C/EBPα and the Corepressors CtBP1 and CtBP2 Regulate Repression of Select Visceral White Adipose Genes during Induction of the Brown Phenotype in White Adipocytes by Peroxisome Proliferator-Activated Receptor γ Agonists. Mol. Cell. Biol. 2009, 29, 4714–4728. [Google Scholar] [CrossRef]
- Kajimura, S. Promoting brown and beige adipocyte biogenesis through the PRDM16 pathway. Int. J. Obes. Suppl. 2015, 5 (Suppl. 1), S11–S14. [Google Scholar] [CrossRef] [PubMed]
- Bournat, J.C.; Brown, C.W. Mitochondrial dysfunction in obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2010, 17, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Breininger, S.P.; Malcomson, F.C.; Afshar, S.; Turnbull, D.M.; Greaves, L.; Mathers, J.C. Effects of obesity and weight loss on mitochondrial structure and function and implications for colorectal cancer risk. Proc. Nutr. Soc. 2019, 78, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.T.; Puigserver, P. Fasting-dependent glucose and lipid metabolic response through hepatic sirtuin 1. Proc. Natl. Acad. Sci. USA 2007, 104, 12861–12866. [Google Scholar] [CrossRef] [PubMed]
- Gerhart-Hines, Z.; Rodgers, J.T.; Bare, O.; Lerin, C.; Kim, S.-H.; Mostoslavsky, R.; Alt, F.W.; Wu, Z.; Puigserver, P. Metabolic control of muscle mitochondrial function and fatty acid oxidation through SIRT1/PGC-1α. EMBO J. 2007, 26, 1913–1923. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.T.; Lerin, C.; Haas, W.; Gygi, S.P.; Spiegelman, B.M.; Puigserver, P. Nutrient control of glucose homeostasis through a complex of PGC-1α and SIRT1. Nature 2005, 434, 113–118. [Google Scholar] [CrossRef]
- Popov, L. Mitochondrial biogenesis: An update. J. Cell. Mol. Med. 2020, 24, 4892–4899. [Google Scholar] [CrossRef]
- Higashida, K.; Kim, S.H.; Jung, S.R.; Asaka, M.; Holloszy, J.O.; Han, D.-H. Effects of Resveratrol and SIRT1 on PGC-1α Activity and Mitochondrial Biogenesis: A Reevaluation. PLoS Biol. 2013, 11, e1001603. [Google Scholar] [CrossRef] [PubMed]
- Cantó, C.; Auwerx, J. PGC-1α, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Curr. Opin. Lipidol. 2009, 20, 98–105. [Google Scholar] [CrossRef]
- Gurd, B.J. Deacetylation of PGC-1α by SIRT1: Importance for skeletal muscle function and exercise-induced mitochondrial biogenesis. Appl. Physiol. Nutr. Metab. 2011, 36, 589–597. [Google Scholar] [CrossRef]
- Tang, B. Sirt1 and the Mitochondria. Mol. Cells 2016, 39, 87–95. [Google Scholar] [PubMed]
- Sanchis-Gomar, F.; Garcia-Gimenez, J.; Gomez-Cabrera, M.; Pallardo, F. Mitochondrial Biogenesis in Health and Disease. Molecular and Therapeutic Approaches. Curr. Pharm. Des. 2014, 20, 5619–5633. [Google Scholar] [CrossRef] [PubMed]
- Onyango, I.G.; Lu, J.; Rodova, M.; Lezi, E.; Crafter, A.B.; Swerdlow, R.H. Regulation of neuron mitochondrial biogenesis and relevance to brain health. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2010, 1802, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, R.M.; Corum, D.; Beeson, C.C.; Schnellmann, R.G. Mitochondrial Biogenesis as a Pharmacological Target: A New Approach to Acute and Chronic Diseases. Annu. Rev. Pharmacol. Toxicol. 2016, 56, 229–249. [Google Scholar] [CrossRef] [PubMed]
- Goede, P.; Wüst, R.C.I.; Schomakers, B.V.; Denis, S.; Vaz, F.M.; Pras-Raves, M.L.; Weeghel, M.; Yi, C.; Kalsbeek, A.; Houtkooper, R.H. Time-restricted feeding during the inactive phase abolishes the daily rhythm in mitochondrial respiration in rat skeletal muscle. FASEB J. 2022, 36, e22133. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; De Stefani, D.; De Vivo, I.; Scapagnini, G. Polyphenols as Caloric Restriction Mimetics Regulating Mitochondrial Biogenesis and Mitophagy. Trends Endocrinol. Metab. 2020, 31, 536–550. [Google Scholar] [CrossRef]
- Chodari, L.; Aytemir, M.D.; Vahedi, P.; Alipour, M.; Vahed, S.Z.; Khatibi, S.M.H.; Ahmadian, E.; Ardalan, M.; Eftekhari, A. Targeting Mitochondrial Biogenesis with Polyphenol Compounds. de Oliveira FL, editor. Oxidative Med. Cell. Longev. 2021, 2021, 4946711. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, Z.; Liu, J.; Hao, Y.; Sun, B.; Wang, J. Potential Health Benefits of Whole Grains: Modulation of Mitochondrial Biogenesis and Energy Metabolism. J. Agric. Food Chem. 2021, 69, 14065–14074. [Google Scholar] [CrossRef]
- Cypess, A.M.; Kahn, C.R. The role and importance of brown adipose tissue in energy homeostasis. Curr. Opin. Pediatr. 2010, 22, 478–484. [Google Scholar] [CrossRef]
- Lo, K.; Sun, L. Turning WAT into BAT: A review on regulators controlling the browning of white adipocytes. Biosci. Rep. 2013, 33, 711–719. [Google Scholar] [CrossRef]
- Van der Veen, D.R.; Laing, E.E.; Bae, S.-E.; Johnston, J.D.; Dijk, D.-J.; Archer, S.N. A Topological Cluster of Differentially Regulated Genes in Mice Lacking PER3. Front. Mol. Neurosci. 2020, 13, 15. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Rosen, C.J. PPARγ: A circadian transcription factor in adipogenesis and osteogenesis. Nat. Rev. Endocrinol. 2010, 6, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yu, Q.; Wang, G.-X.; Lin, J.D. The Biological Clock is Regulated by Adrenergic Signaling in Brown Fat but is Dispensable for Cold-Induced Thermogenesis. PLoS ONE 2013, 8, e70109. [Google Scholar] [CrossRef]
- Heyde, I.; Begemann, K.; Oster, H. Contributions of white and brown adipose tissues to the circadian regulation of energy metabolism. Endocrinology 2021, 162, bqab009. [Google Scholar] [CrossRef] [PubMed]
- Seale, P.; Kajimura, S.; Yang, W.; Chin, S.; Rohas, L.M.; Uldry, M.; Tavernier, G.; Langin, D.; Spiegelman, B.M. Transcriptional Control of Brown Fat Determination by PRDM16. Cell Metab. 2007, 6, 38–54. [Google Scholar] [CrossRef]
- Ohno, H.; Shinoda, K.; Spiegelman Bruce, M.; Kajimura, S. PPARγ agonists Induce a White-to-Brown Fat Conversion through Stabilization of PRDM16 Protein. Cell Metab. 2012, 15, 395–404. [Google Scholar] [CrossRef]
- Seale, P.; Bjork, B.; Yang, W.; Kajimura, S.; Chin, S.; Kuang, S.; Scimè, A.; Devarakonda, S.; Conroe, H.M.; Erdjument-Bromage, H.; et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature 2008, 454, 961–967. [Google Scholar] [CrossRef]
- Monnier, C.; Auclair, M.; Le Cam, G.; Garcia, M.; Antoine, B. The nuclear retinoid-related orphan receptor ROR α controls circadian thermogenic programming in white fat depots. Physiol. Rep. 2018, 6, e13678. [Google Scholar] [CrossRef]
- Weger, M.; Diotel, N.; Dorsemans, A.-C.; Dickmeis, T.; Weger, B.D. Stem cells and the circadian clock. Dev. Biol. 2017, 431, 111–123. [Google Scholar] [CrossRef]
- Nam, D.; Chatterjee, S.; Yin, H.; Liu, R.; Lee, J.; Yechoor, V.K.; Ma, K. Novel Function of Rev-erbα in Promoting Brown Adipogenesis. Sci. Rep. 2015, 5, 11239. [Google Scholar] [CrossRef]
- Lettieri Barbato, D.; Baldelli, S.; Pagliei, B.; Aquilano, K.; Ciriolo, M.R. Caloric Restriction and the Nutrient-Sensing PGC-1αin Mitochondrial Homeostasis: New Perspectives in Neurodegeneration. Int. J. Cell Biol. 2012, 2012, 759583. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.N.; Clark, J.P.; Martin, S.A.; Howell, P.R.; Burhans, M.S.; Haws, S.A.; Johnson, N.B.; Rhoads, T.W.; Pavelec, D.M.; Eliceiri, K.W.; et al. PGC-1a integrates a metabolism and growth network linked to caloric restriction. Aging Cell 2019, 18, e12999. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Deguchi, Y.; Nozaki, Y.; Higami, Y. Contribution of PGC-1α to Obesity- and Caloric Restriction-Related Physiological Changes in White Adipose Tissue. Int. J. Mol. Sci. 2021, 22, 6025. [Google Scholar] [CrossRef] [PubMed]
- Lira, V.A.; Benton, C.R.; Yan, Z.; Bonen, A. PGC-1α regulation by exercise training and its influences on muscle function and insulin sensitivity. Am. J. Physiol.-Endocrinol. Metab. 2010, 299, E145–E161. [Google Scholar] [CrossRef]
- Summermatter, S.; Handschin, C. PGC-1α and exercise in the control of body weight. Int. J. Obes. 2012, 36, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
- Vidal, P.; Stanford, K.I. Exercise-Induced Adaptations to Adipose Tissue Thermogenesis. Front. Endocrinol. 2020, 11, 270. [Google Scholar] [CrossRef]
- Aouichat, S.; Chayah, M.; Bouguerra-Aouichat, S.; Agil, A. Time-Restricted Feeding Improves Body Weight Gain, Lipid Profiles, and Atherogenic Indices in Cafeteria-Diet-Fed Rats: Role of Browning of Inguinal White Adipose Tissue. Nutrients 2020, 12, 2185. [Google Scholar] [CrossRef]
- Goede, P.; Hellings, T.P.; Coopmans, T.V.; Ritsema, W.I.G.R.; Kalsbeek, A. After-Effects of Time-Restricted Feeding on Whole-Body Metabolism and Gene Expression in Four Different Peripheral Tissues. Obesity 2020, 28, S68–S80. [Google Scholar] [CrossRef]
- Persinger, A.; Butawan, M.; Faietti, M.; Pryke, A.; Rose, K.; van der Merwe, M.; Bloomer, R.; Puppa, M. Effects of Feeding Time on Markers of Muscle Metabolic Flexibility Following Acute Aerobic Exercise in Trained Mice Undergoing Time Restricted Feeding. Nutrients 2021, 13, 1717. [Google Scholar] [CrossRef]
- Vieira, R.F.L.; Muñoz, V.R.; Junqueira, R.L.; Oliveira, F.; Gaspar, R.C.; Nakandakari, S.C.B.R.; Costa, S.D.O.; Torsoni, M.A.; da Silva, A.S.; Cintra, D.E.; et al. Time-restricted feeding combined with aerobic exercise training can prevent weight gain and improve metabolic disorders in mice fed a high-fat diet. J. Physiol. 2021, 600, 797–813. [Google Scholar] [CrossRef]
- Rodrigues, L.G.F.; de Araujo, L.D.; Roa, S.L.R.; Bueno, A.C.; Uchoa, E.T.; Antunes-Rodrigues, J.; Moreira, A.C.; Elias, L.L.K.; de Castro, M.; Martins, C.S. Restricted feeding modulates peripheral clocks and nutrient sensing pathways in rats. Arch. Endocrinol. Metab. 2021, 65, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Wood dos Santos, T.; Cristina Pereira, Q.; Teixeira, L.; Gambero, A.; Villena, J.A.; Lima Ribeiro, M. Effects of Polyphenols on Thermogenesis and Mitochondrial Biogenesis. Int. J. Mol. Sci. 2018, 19, 2757. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Sapere, N.; Visentin, M.; Zella, D.; Scapagnini, G. Enhancement of mitochondrial biogenesis with polyphenols: Combined effects of resveratrol and equol in human endothelial cells. Immun. Ageing 2013, 10, 28. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.M.; Forsyth, C.B.; Green, S.J.; Mutlu, E.; Engen, P.; Vitaterna, M.H.; Turek, F.W.; Keshavarzian, A. Circadian Disorganization Alters Intestinal Microbiota. PLoS ONE 2014, 9, e97500. [Google Scholar] [CrossRef]
- Voigt, R.M.; Summa, K.C.; Forsyth, C.B.; Green, S.J.; Engen, P.; Naqib, A.; Vitaterna, M.H.; Turek, F.W.; Keshavarzian, A. The CircadianClockMutation Promotes Intestinal Dysbiosis. Alcohol. Clin. Exp. Res. 2016, 40, 335–347. [Google Scholar] [CrossRef]
- Thaiss Christoph, A.; Zeevi, D.; Levy, M.; Zilberman-Schapira, G.; Suez, J.; Tengeler Anouk, C.; Abramson, L.; Katz, M.N.; Korem, T.; Zmora, N.; et al. Transkingdom Control of Microbiota Diurnal Oscillations Promotes Metabolic Homeostasis. Cell 2014, 159, 514–529. [Google Scholar] [CrossRef]
- Zarrinpar, A.; Chaix, A.; Yooseph, S.; Panda, S. Diet and Feeding Pattern Affect the Diurnal Dynamics of the Gut Microbiome. Cell Metab. 2014, 20, 1006–1017. [Google Scholar] [CrossRef]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef]
- Deng, Y.; Liu, W.; Wang, J.; Yu, J.; Yang, L. Intermittent Fasting Improves Lipid Metabolism Through Changes in Gut Microbiota in Diet-Induced Obese Mice. Med. Sci. Monit. 2020, 26, e926789-1–e926789-11. [Google Scholar] [CrossRef]
- Patterson, R.E.; Laughlin, G.A.; LaCroix, A.Z.; Hartman, S.J.; Natarajan, L.; Senger, C.M.; Martínez, M.E.; Villaseñor, A.; Sears, D.D.; Marinac, C.R.; et al. Intermittent Fasting and Human Metabolic Health. J. Acad. Nutr. Diet. 2015, 115, 1203–1212. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2008, 457, 480–484. [Google Scholar] [CrossRef]
- Cui, Y.; Li, S.; Yin, Y.; Li, X.; Li, X. Daytime restricted feeding promotes circadian desynchrony and metabolic disruption with changes in bile acids profiles and gut microbiota in C57BL/6 Male Mice. J. Nutr. Biochem. 2022, 109, 109121. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Qi, Y.; Zheng, H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants 2022, 11, 1212. [Google Scholar] [CrossRef]
- Molinari, R.; Merendino, N.; Costantini, L. Polyphenols as modulators of pre-established gut microbiota dysbiosis: State-of-the-art. BioFactors 2021, 48, 255–273. [Google Scholar] [CrossRef] [PubMed]
- Hills, R.D.; Pontefract, B.A.; Mishcon, H.R.; Black, C.A.; Sutton, S.C.; Theberge, C.R. Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 2019, 11, 1613. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Liwinski, T.; Elinav, E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020, 30, 492–506. [Google Scholar] [CrossRef] [PubMed]
- De Vadder, F.; Kovatcheva-Datchary, P.; Goncalves, D.; Vinera, J.; Zitoun, C.; Duchampt, A.; Bäckhed, F.; Mithieux, G. Microbiota-Generated Metabolites Promote Metabolic Benefits via Gut-Brain Neural Circuits. Cell 2014, 156, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Kumar Singh, A.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Lauro, M.R.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients 2019, 11, 2216. [Google Scholar] [CrossRef]
- Leone, V.; Gibbons, S.M.; Martinez, K.; Hutchison, A.L.; Huang, E.Y.; Cham, C.M.; Pierre, J.F.; Heneghan, A.F.; Nadimpalli, A.; Hubert, N.; et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe 2015, 17, 681–689. [Google Scholar] [CrossRef]
- Effects of alternate-day fasting, time-restricted fasting and intermittent energy restriction DSS-induced on colitis and behavioral disorders. Redox Biol. 2020, 32, 101535. [CrossRef] [PubMed]
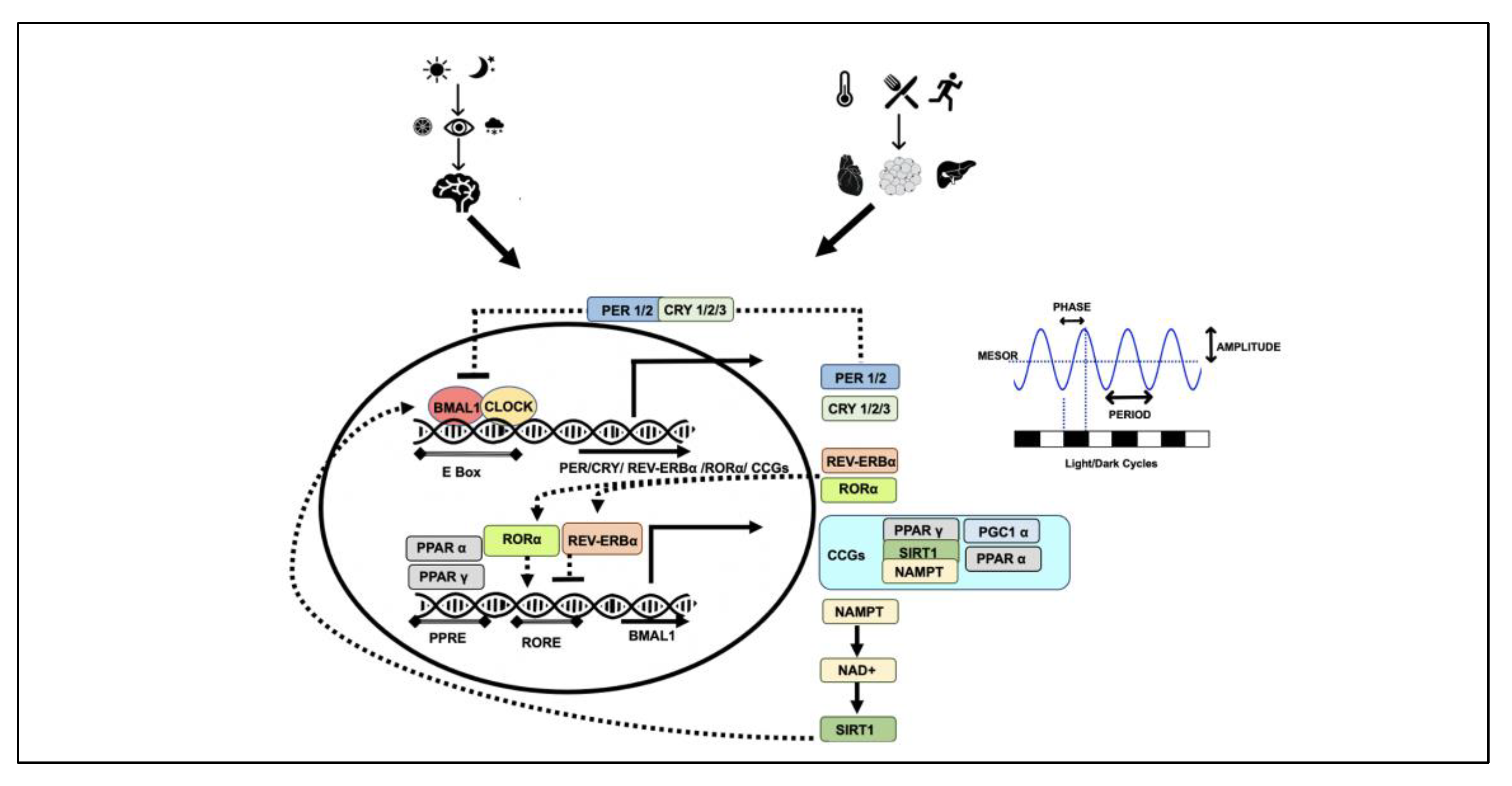

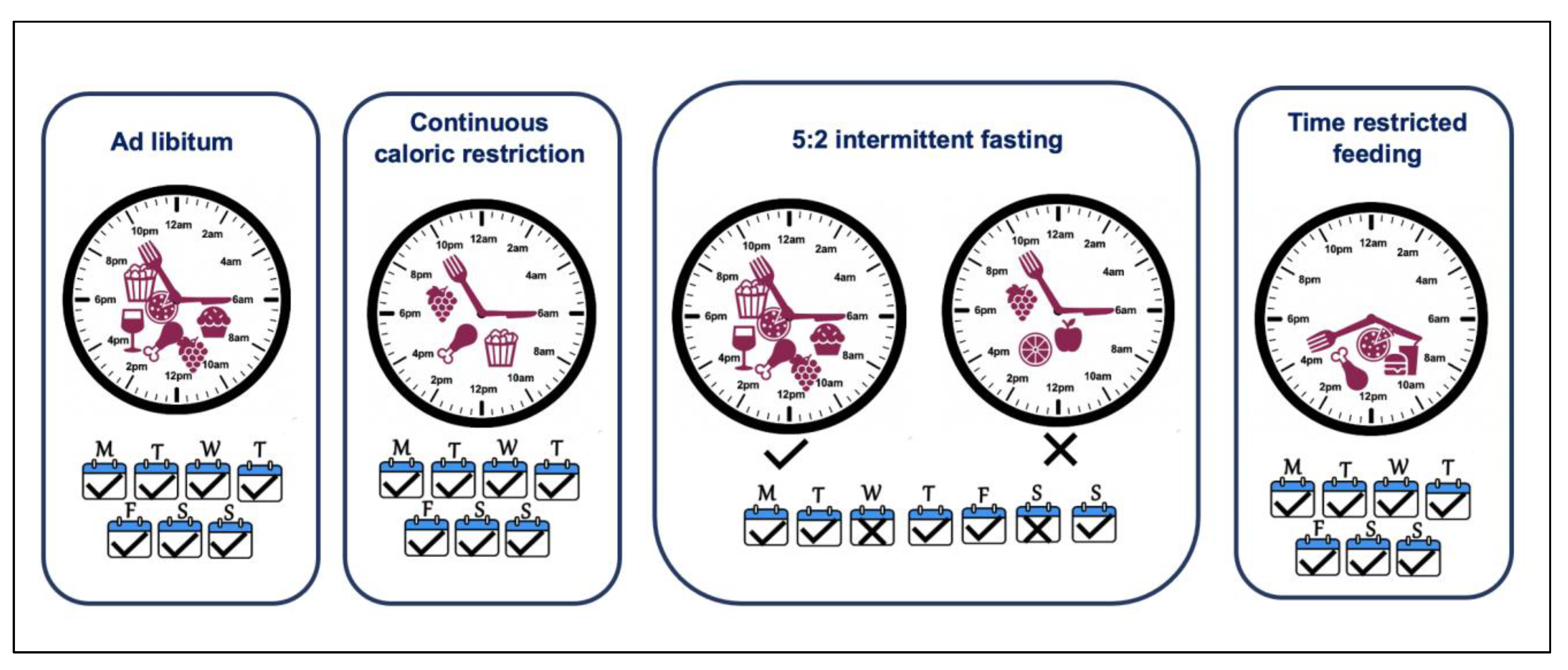
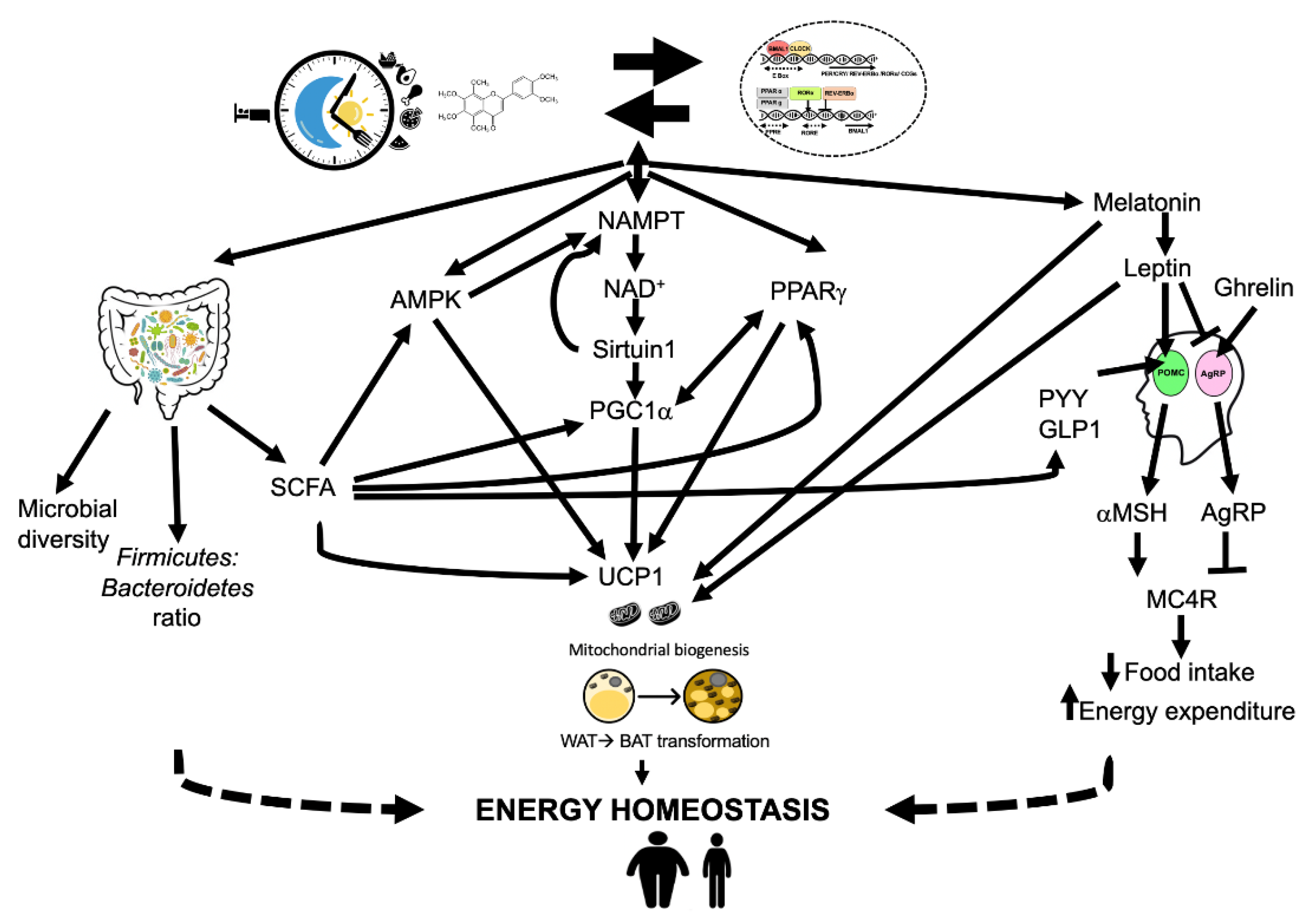
| Genetic Variant/Haplotypes | Population | Observations | Ref |
|---|---|---|---|
| CLOCK rs1801260 | 421 Japanese subjects | C allele associated with an evening chronotype with significant delayed onset of sleep, shorter sleep times, greater daytime sleepiness | [52] |
| 500 overweight and obese subjects during 28-week weight-loss program | C allele carriers more emotional eaters and more resistant to weight loss | [53] | |
| 284, 92 controls vs. 192 overweight and obese with or without binge eating disorder | C allele predisposes obese individuals to a higher BMI | [54] | |
| 1272 overweight and obese participants attending 30-week weight-loss program | C allele carriers were emotional eaters and lost less weight and at a lower rate compared to non-emotional eaters with the same risk allele or non-risk allele | [55] | |
| 370 children aged 6–13 years | No association between the genotype and sleep duration. However, there seems to be a trend between sleep duration and overweight | [56] | |
| 85 overweight women | C allele carriers with significant circadian abnormalities: lower amplitude and greater fragmentation of the rhythm, and an evening chronotype | [57] | |
| 1495 overweight/obese subjects participating in a Mediterranean-diet-based weight-reduction program for 28 weeks | C allele carriers are more resistant to weight loss, shorter sleep duration, higher plasma ghrelin concentrations, delayed breakfast time, evening preference and less compliance with a Mediterranean diet plan | [58] | |
| 475 metabolic syndrome subjects participating in the CORDIOPREV clinical trial for 1 year | Gene diet interactions; C carriers showed non-significant improved insulin sensitivity while T carriers have significantly improved insulin sensitivity on low-fat diet | [59] | |
| CLOCK haplotype rs1554483 and rs4864548 | 715 lean and 391 overweight or obese | GA haplotype associated with a 1.8-fold risk of overweight or obesity | [60] |
| CLOCK rs3749474 | 500 overweight/obese subjects | T allele carriers have significantly higher weight, BMI and waist circumference | [53] |
| CLOCK rs3749474 | 1100 individual participants in the Genetics of Lipid Lowering Drugs and Diet Network (GOLDN) | T allele carriers have significantly higher energy, total fat, protein and carbohydrate intakes | [61] |
| CLOCK rs3749474 | 898 subjects | T allele associated positively with higher BMI and evening carbohydrate intake | [62] |
| CLOCK rs6850524 | 260 cases with abdominal obesity and 260 controls Chinese population | CC genotype have a lower risk of overweight or obesity than those with GG genotype | [63] |
| CLOCK haplotype rs4864548-rs3736544-rs1801260 | 537 individuals from 89 families characterized for inflammatory, atherothrombotic and metabolic risk associated with insulin resistance. | CGC haplotype protective for the development of obesity and the CAT haplotype is associated with the presence of the metabolic syndrome | [64] |
| CLOCK rs4580704 | 7098 PREvención con DIeta MEditerránea (PREDIMED) trial over a median duration of 4.8 years | G allele with decreased incidence of type 2 diabetes and associated stroke. Mediterranean diet provides further protection in G allele carriers. | [65] |
| 897 patients from the CORDIOPREV clinical trial, low-fat diet and Mediterranean diet for 12 months | C allele carriers showed significant reduction in CRP levels and an improvement in HDL/ApoA1 ratio after a low-fat diet for 12 months | [66] | |
| 1100 individual participants in the Genetics of Lipid Lowering Drugs and Diet Network (GOLDN) | G allele show lower blood pressure and higher erythrocyte membrane oleic acid (MUFA) and improved insulin sensitivity in high-MUFA intake | [67] | |
| CLOCK haplotype rs3749474-rs4580704-rs1801260 | 1100 individual participants in the Genetics of Lipid Lowering Drugs and Diet Network (GOLDN) | The haplotype CGA was associated with lower BMI, weight, waist circumference, adiponectin concentration, blood pressure and with oleic acid (MUFA) RBC membrane composition. | [67] |
| CLOCK rs12649507, rs6858749 | 14,906 from the CHARGE (Cohorts for Heart and Aging Research in Genomic Epidemiology) Consortium | Longer habitual sleep duration is associated with lower BMI and a favourable dietary behaviour. rs12649507 G allele: Higher PUFA intake and more sleep.rs6858749 T allele: Lower protein intake with each additional hour of sleep | [68] |
| CLOCK rs10002541 and rrs6850524 | 260 cases with abdominal obesity and 260 controls, Chinese population | Significant associations between CG and TG haplotypes and abdominal obesity. rs10002541 C allele is protective for abdominal obesity. | [63] |
| CLOCK rs12649507 and rs11932595 | 1011 individuals from Tyrol and Estonia | The haplotype GGGG in Tyrolean and GGAA in Estonian population associated with longer sleep | [69] |
| CLOCK rs1801260 BMAL1 rs2278749 | 507 healthy young adults | CLOCK rs1801260 associated with seasonal affective disorder and it synergistically interacts with BMAL1 rs2278749. Seasonal variation in body weight and appetite also associated with BMAL1 rs2278749. | [70] |
| CLOCK rs1801260 + SIRT1 rs1467568 | 1465 Overweight/obese subjects over 30 weeks of Mediterranean-diet-based weight-loss program | SIRT1 (A allele) ad CLOCK (C allele) are more resistant to lose weight and have more of an evening preference | [71] |
| PER2 gene rs4663302 rs2304672 | 454 obese subjects, weight reduction program based on Mediterranean diet | PER2 gene rs4663302 rs2304672 associated with abdominal obesity. rs4663302 T allele and rs2304672 G allele carriers associated with not completing the weight-loss programme. rs2304672 G also linked to extreme snacking, experiencing stress with dieting, eating when bored, and skipping breakfast than noncarriers. | [72] |
| PER2, BMAL1, and NPAS2 | 189 patients with winter depression and 189 matched controls | PER2 rs10870, BMAL1 rs2290035 and NPAS2 rs11541353 significantly association with SAD | [73] |
| CRY1 rs10861688 | 260 cases with abdominal obesity and 260 controls, Chinese population | CRY1 rs10861688 T allele negatively associated with the risk of abdominal obesity. | [63] |
| REV-ERBα rs2314339 | 2212 subjects from two independent populations (1402 from Spanish Mediterranean and 810 North American) | Minor allele carriers (AA + AG) have lower probability of abdominal obesity than noncarriers. A allele carriers on low MUFA lead to high BMI while A carriers on high MUFA reduce BMI and BMI was low in A carriers in high-PUFA intake | [74] |
| Late Night Eating, BMI and Metabolic Health | ||
|---|---|---|
| 180 bariatric surgery candidates, 93 non-surgical weight-loss intervention and 158 general community candidates | Night-eating syndrome associated with binge eating, higher BMI and male gender. Night-eating syndrome, consuming nocturnal snacks leads to more hunger and depressive symptoms. | [87] |
| 26,902 men over 16 years follow-up | Late night eaters have a 55% higher risk of CHD, are more likely to have baseline hypertension and men who did not eat breakfast have a 27% higher risk of CHD than those who ate breakfast. | [90] |
| 8153 adults over an average of 3.9 yrs. | Night-time eating associated with dyslipidaemia in both men and women but metabolic syndrome and an increase in the risk of obesity only in women | [96] |
| 10 participants on glucose solution at 8 am and 8 pm and 9 participants taking a low-glycaemic-index meal at 8 am, 8 pm and midnight. | Even low-glycaemic-index meals late at night disturb glucose metabolism | [86] |
| 19,687 Japanese women | Skipping breakfast, late dinner and bedtime snack associated with overweight and obesity in Japanese women | [97] |
| 397, 8–12 yr old children | Late dinner eaters (after 21.07 h) were more likely to be overweight and obese, with higher waist circumference and inflammatory markers | [98] |
| 49 participants | Inconsistent meal time, especially late, eaters have significantly higher BMI | [99] |
| 100 subjects | Correlation between night-time eating and binge eating and BMI | [88] |
| Later chronotype, BMI and metabolic health | ||
| 2200 9–16 yr old in Australia | The later chronotype more likely to be overweight and obese | [100] |
| 54 college freshmen | An evening chronotype associated with higher BMI as compared to morning or neutral chronotypes | [49] |
| 511 UK 11–13 yr old children | An evening chronotype was associated with higher BMI, higher frequency of unhealthy food choices. Sleep duration is an independent risk factor for BMI | [101] |
| 194 participants | Later chronotype and larger dinner are associated with poorer glycaemic control in patients with type 2 diabetes independently of sleep disturbances. | [45] |
| 439,933 adults from a UK biobank | Evening preference associated with high risk of cardiovascular disease | [46] |
| 800 undergraduate students | Evening chronotype associated with BMI that can be negated by a decrease in sugary beverage intake, increases in physical activity | [47] |
| 2133 prediabetic patients | More evening preference is directly associated with higher BMI | [48] |
| 872 adults | Later chronotypes with higher percentage of daily energy intake during the night are associated with overweight and obesity while earlier chronotypes consuming more energy in the morning are at a lower risk of weight gain. | [102] |
| 1197 middle-aged men and women | An evening chronotype associated with obesity | [50] |
| Circadian misalignment, BMI and metabolic health | ||
| 14 healthy participants on 8-day protocol for short-term misalignment and cross over | Short-term circadian misalignment leads to increased systolic and diastolic blood pressure and serum inflammatory markers | [103] |
| 14 adults in a 6-day simulated shift-work environment | Eating during the biological night, e.g., for shift workers, decreases total daily energy expenditure and increases the risk of weight gain and obesity | [89] |
| 10 adults underwent a 10-day protocol with eating and sleeping in all phases | Forced desynchrony protocols cause circadian misalignment and disturb postprandial glucose response typical of prediabetes | [104] |
| 2494 participants (1259 day and 1235 shift workers) | Shift work is associated with higher risk of being overweight/obese | [105] |
| 26,382 participants (9088 shift workers) | Long-term shift work is associated with metabolic syndrome | [106] |
| 9912 male employees (8892 daytime workers and 920 rotating three-shift workers | High risk of obesity among male shift workers | [107] |
| 905 shift workers | Strong association between sleep deprivation and obesity in shift workers | [108] |
| 200 shift workers | Night work is a risk factor for abdominal obesity, social jetlag is higher in night shift workers and it was associated with the presence of obesity. | [109] |
| 3188 shift workers and 6395 non-shift workers | Shift work associated with obesity, lower physical activity, poor dietary choices | [110] |
| Polyphenol | Interactions with Circadian Clocks | Reference |
|---|---|---|
| Resveratrol | Improvement in rhythmic expression of core clock and various clock-controlled genes including NAMPT, SIRT1, PPARα | [142,143,144,145,146,147,148] |
| Improvement of insulin, glucose, lipid metabolism impairments caused by chronodisruption | ||
| Prevention of neuron damage and memory impairment caused by circadian disruption | ||
| Enhanced expression of PER1, PER2 and BMAL1 in rat fibroblasts | ||
| Downregulation of high-fat diet induced REV-ERBα in adipose tissue | ||
| Reversal of free-fatty-acid-induced loss of oscillation amplitude in core clock genes in HEPG2 cells | ||
| Amelioration of acrylamide suppressed amplitude and phase of oscillations in core clock genes and increased expression of SIRT1 and PGC1α | ||
| Increase in expression of BMAL1, PER1, SIRT6, SIRT1 and REV-ERBα mRNA in fibroblasts | ||
| Proanthocyanidins | Modulation BMAL acetylation, increase in PER2 expression and inhibition of REV-ERBα and RORα in rat models of diet-induced obesity | [149] |
| Modulation of NAMPT expression and NAD+ levels in rat liver | ||
| Epigallocatechin-3-gallate | Altered circadian expression patterns of CLOCK, BMAL1 and key appetite-regulating genes in mice | [150,151,152] |
| Ameliorated diet-induced metabolic misalignment by regulating the rhythmic expression of the circadian clock genes in the liver and adipose tissue in mice | ||
| Repressed CLOCK expression in lung cancer cell lines and reduced the self-renewal capacity of the cells | ||
| Nobiletin | Affects amplitude, period and phase of mutant mice cells with weaker rhythmic amplitude. | [153,154,155,156,157,158,159,160,161,162] |
| Activation of RORs and protection against metabolic syndrome in a clock-dependent manner. | ||
| Enhances BMAL1, reverses the loss of oscillation amplitude observed in metabolic disease state | ||
| Induction of AMPK-SIRT1 signalling and lipogenesis | ||
| Ability to induce circadian rhythmicity and inhibit oncogenicity in MDA-MB-231 cells | ||
| Modulates expression of core clock and clock-controlled genes in the cortex | ||
| Restores endogenous rhythm of clock genes in steatosis liver | ||
| Modulates clock and Alzheimer’s-disease-related genes in cortex of AD model mice | ||
| Cardioprotective role in ischemia reperfusion injury by upregulating midazolam-inhibited PER2 | ||
| Improves metabolic fitness in naturally aged mice and promotes healthy aging in high-fat diet by activating genes to promote mitochondrial function | ||
| Improves insulin secretion by enhancing the amplitude of circadian gene expression in T2D islets | ||
| Oolong tea polyphenols | Improves Firmicutes:Bacteroidetes ratio in the intestinal flora in mice | [163,164] |
| Promotes the growth of strains of gut microbiota and positively regulates the production of SCFA | ||
| Restoration of CLOCK, BMAL1, PER and CRY expression disturbed by constant day feeding in mice | ||
| Chichoric acid | Regulation of rhythmic expression of clock genes | [165] |
| Quercetin | Upregulation of BMAL1, SIRT1, SIRT6, REV-ERBα and reduction in PER1 expression in fibroblasts | [148,166] |
| Suppresses breast cancer metastasis to lymph nodes promoted by circadian disruption |
| Time-Restricted Feeding (TRF) | |||||
|---|---|---|---|---|---|
| Participants | Eating Restrictions | Study Type/Duration | Health Outcomes | Energy Intake | Reference |
| n = 49 obese subjects BMI 30–50 kg/m2 | Eating window of 4 h (3 pm–7 pm) vs. 6 h (1 pm–7 pm) vs. controls (7 am–7 pm) | Randomized parallel-arm trial over 8 weeks | Both TRF regimens showed reduction in body weight, insulin resistance, oxidative stress levels. Four-hour TRE did not result in greater weight loss compared to six-hour TRE. | Reduction in energy intake by 550 kcal/day in both cases without calorie counting | [176] |
| n = 11 Obese sedentary males BMI: 30.2–34.2 kg/m2 | Eating window 10 am–5 pm vs. 7 am–9 pm | Randomized crossover trial; 3 weeks each intervention of 5 days with 10 days washout period | Improved glycaemic control and decrease in evening hunger | Isocaloric intake | [177] |
| n = 19 with T2D BMI: 29–39 kg/m2 | 4-week TRE 10 am–7 pm | non-randomised 2-week baseline, 4-week intervention | Compliance 72 ± 24%, no improvement in glycaemic control or body mass | Isocaloric intake | [178] |
| n = 23 obese subjects BMI 30 and 45 kg/m2 | Eating over 8-h window (10 am–6 pm) vs. ad libitum eating | 2-week baseline intake, 12-week intervention | Time-restricted eating showed reduction in body weight and systolic blood pressure | Decreases caloric intake by ~300 kcal/d | [179] |
| n = 34 resistance-trained weight 84.6 ± 6.2 kg | TRF (1 pm–8 pm) vs. control (8 am–8 pm) | Randomized parallel-arm trial over 8 weeks | TRF only showed a reduction in fat mass but no other metabolic parameters were altered. Fat free mass and muscle mass area in arm and thigh remain unchanged | Isocaloric intake | [180] |
| n = 9 overweight sedentary older adults BMI 25–40 kg/m2 | 16 h fast (14–18 h range) | Baseline assessment followed by 4-week intervention | TRE resulted in short-term weight loss and improved waist circumference, cognitive and physical function and health-related quality of life | No data available | [181] |
| n = 8, prediabetic BMI 32.2 ± 4.4 kg/m2 | eTRF; 6-h eating period and dinner before 3 pm for 5 weeks, vs. 12-h eating period | Randomized crossover trial for 17 weeks, each intervention 5 weeks | eTRF reduced insulin levels and improved insulin sensitivity, lowered blood pressure; reduction in oxidative stress and appetite in the evening. | Isocaloric intake | [182] |
| n = 19 with metabolic syndrome | Eating over self-selected 10-h window | 2-week baseline intake, 12-week intervention | TRE improves cardiometabolic health (reduction in weight, BMI, waist circumference, percentage body fat, systolic and diastolic blood pressure, improved lipid parameters, glucose and insulin homeostasis | Decreases caloric intake | [183] |
| n = 8 overweight BMI > 25 kg/m2 | Eating over self-selected 10-h window | 3-week baseline intake, 16-week | Reduction in body weight. Significant improvement in sleep, hunger at bedtime, energy levels | Reduced estimated energy intake by 20–26% | [170] |
| n = 11, BMI 25.0 and 35.0 kg/m2 | eTRF (8 am to 2 pm) vs. control (8 am to 8 pm) | Randomized crossover 4-day intervention, 3.5–5 weeks’ washout period between interventions | eTRF improves 24-hour glucose levels, alters lipid metabolism and expression of SIRT1 and LC3A (autophagy gene), BDNF (a neurotrophic factor promoting neuronal growth) and mTOR | Isocaloric intake | [184] |
| n = 11 overweight BMI 25–35 kg/m2 | eTRF (8 am–2 pm) vs. control (8 am–8 pm) | Randomized crossover 4-day intervention, 3.5–5 weeks’ washout period between interventions | Meal-timing interventions facilitate weight loss primarily by decreasing appetite rather than by increasing energy expenditure. eTRF may also increase fat loss by increasing fat oxidation. | Isocaloric | [185] |
| n = 21 healthy adults BMI 29.6 ± 2.6 kg/m2 | TRE (12 pm to 8 pm) vs. control eating habits with concomitant aerobic exercise for 8 weeks | Randomized, controlled trial | TRF individuals lost significantly more body mass (3.3% vs. 0.2%) and fat mass (9% vs. 3.3%). Lean mass increased but no significant difference between the groups. | Reduction in caloric intake in TRE (~300 kCal/day) | [186] |
| n = 27 BMI 21.9–26.9 kg/m2 | TRE included an elimination of caloric intake between 7 pm and 6 am vs. controls | Crossover 2-week intervention with one-week washout period | TRE led to a loss in small amount of body weight | Reduction in energy (~240 Kcal) and fat intake un TRE group | [187] |
| n = 18 Body weight 79.0 ± 13.5 kg in control group and 87.4 ± 19.2 in TRE group | TRE (eating over any 4-h window between 4 pm and 12 pm on the four days a week when they exercised but ad libitum on days without exercise) vs. control without any restrictions | Randomized controlled trial 8 weeks | No significant loss of body weight, no adverse effect on lean mass retention or muscular improvements. | TRF reduced caloric intake by ~667 kCal a day | [188] |
| n = 13 BMI 20–39 kg/m2 | TRF with delayed breakfast and advanced dinner by 1.5 h vs. controls with habitual eating patterns | 2-week baseline, 10 weeks’ intervention | No significant reduction in weight, but reduction in adiposity, fasting glucose observed | Reduction in energy intake in TRE group | [189] |
| n = 40 resistance-trained females Body weight 57.1 to 73.4 Kg | Control diet vs. TRF (12 pm–8 pm) vs. TRF+ a leucine metabolite β-hydroxy β-methyl butyrate (HMB) supplementation | randomized, placebo-controlled for 8 weeks | TRF did not produce changes in physiological variables including resting metabolic rate, substrate utilization, blood lipids, glucose and insulin, blood pressure, arterial stiffness, or cortisol responses. No significant difference in physical performance. | No significant variation between the groups | [190] |
| n = 40 with abdominal obesity BMI 25.1–37.6 kg/m2 | TRF eating window 8–9 h | 3-month single arm trial | Moderate weight loss, improved waist circumference, HbA1C | No data available | [191] |
| n = 22 men BMI: 28.5 ± 8.3 kg/m2 | Isocaloric TRF (8-h eating window, caloric intake within 300 Kcal of habitual intake) vs. ad libitum TRF (8-h eating window but no restriction on calories) | 28 days randomised control trial | Decrease in body mass, decrease in fat body mass, decrease in BP and increase in HDLC in both groups. | No significant difference in caloric intake | [192] |
| n = 20 obese BMI 34.1± 7.5 kg/m2 | TRE (self-selected 8-h eating window) vs. control on ad libitum | 12 weeks | Decrease in eating frequency, weight, lean mass, visceral fat | No data reported | [193] |
| n = 116 overweight and obese BMI 27.4–35.4 kg/m2 | TRE (12–8 pm) eating vs. ad libitum | 12 weeks’ randomised control trial | Loss of body weight in TRE group | No significant difference in caloric intake | [194] |
| n = 271 NAFLD BMI > 24 kg/m2 | Control vs. ADF (25% energy intake on fast days) vs. TRF (self-directed 8-hour window) | 12 weeks’ randomised control trial | Significant weight loss and fat mass loss, reduction in cholesterol and triglycerides both in ADF and TRF with ADF achieving better outcomes | No significant difference in caloric intake | [195] |
| n = 15 PCOS women BMI ≥ 24 kg/m2 | TRE (8 am–4 pm) | Non-randomized 1 week baseline, 5 weeks’ intervention | Reduction in body weight, BMI, body fat mass, body fat percentage, improved insulin sensitivity | Isocaloric | [196] |
| n = 22 BMI = 24.7 ± 0.6 kg/m2 | TRE (eating within 8-h window but first meal between 10–11 am) vs. controls with normal feeding patterns | Randomized controlled trial, 1 week baseline, 6-week intervention | No weight loss or improvement in cardiovascular function with modest improvement in functional endurance and glucose tolerance, 84–95% adherence | Isocaloric | [197] |
| n = 60 BMI ≥ 30 kg/m2 | 14:10 TRE (14-h metabolic fast with snack with 200 kcal mixed nuts 12 h after the fast started) vs. 12:12 TRE (12-h fast without any snack) | Randomized controlled trial 8 weeks intervention | Weight loss observed in both cases but more in 14 h metabolic fast group, improved fasting blood glucose. Fasting snack decreased hunger | 500–1000 kcal deficit each day | [198] |
| n = 45 with at least one metabolic syndrome component and usual eating window of 14 h. BMI ≥ 20 kg/m2 | TRE (self-selected window of 12 h) vs. no restriction | Randomised control trial 4 weeks’ baseline, 6-month intervention | No significant difference in weight loss | No difference reported | [199] |
| n = 80 males | TRF (8-h eating window, 7.30 pm–3.30 am) vs. normal diet daily fasting for 16 h for 25 days | 25 Days | TRF improved lipid parameters, reduced inflammatory markers, enhanced gut microbial richness with enrichment of Prevotellaceae and Bacteroideaceae; activated SIRT1 | No data available | [200] |
| Time-restricted feeding (TRF) vs. Continuous caloric restriction (CR) | |||||
| n = 16 BMI 24.0 ± 0.6 kg/m2 | eTRF (8 am till 4 pm) vs. control on caloric restriction | Non-randomised 1-week baseline, 2 weeks’ intervention | eTRF improved insulin sensitivity, glucose uptake, reduction in energy intake and weight loss | Isocaloric | [201] |
| n = 37 overweight BMI 26.4–28.55 kg/m2 | TRE (8 am–4 pm) vs. BMI matched participants on hypocaloric diet based on orthodox fasting | Both groups showed reduction in BMI and fasting group also showed a reduction in total and LDL cholesterol | Isocaloric | [202] | |
| Early time-restricted feeding (eTRF) vs. late time-restricted feeding (lTRF) | |||||
| n = 15 men at risk of T2D BMI 33.9 ± 0.8 kg/m2 | eTRF (8 am–5 pm) vs. late TRF (12 pm–9 pm) over 2-time 7-day TRF with 2 weeks’ washout period | Randomised crossover trial, 1-week baseline, 1-week intervention, 2 weeks’ washout period | Both TRF regimens showed reductions in body weight, glycaemic responses to a test meal, triglycerides. | No data available | [203] |
| n = 82 BMI 18.6–25.8 kg/m2 | Early TRF (6 am–3 pm) vs. mid-day TRF (11 an–8 pm) vs. controls | Randomised control trial, 5 weeks’ intervention | Early TRF was more effective in improving insulin sensitivity, fasting glucose, reduction in body mass and adiposity, reduction in inflammation and increased gut microbial diversity | Reduction in caloric intake in both TRF groups vs. control | [204] |
| n = 8 prediabetic BMI 32.2 ± 4.4 kg/m2 | eTRF (6-h eating window and dinner before 3 pm) vs. 12-h eating period | Randomised crossover trial, 5 weeks’ intervention with a 7-week washout period | eTRF reduced insulin levels and improved insulin sensitivity, lowered blood pressure, reduction in oxidative stress and appetite in the evening. | Isocaloric | [181] |
| Breakfast vs. dinner calories | |||||
| n = 93 Overweight and obese women BMI 32.4 ± 1.8 kg/m2 | 1440 KCal consumed over breakfast/lunch/dinner 700, 500, 200 kcal vs. 200,500, 700 kcal | Randomized parallel-arm study for 12 weeks. | High caloric breakfast group showed greater weight loss and waist circumference, fasting glucose, insulin, triglycerides, HOMA-IR. Ghrelin, hunger vs satiety improved. | [205] | |
| n = 1245 non-obese, non-diabetic middle-aged adults | Daily caloric intake at dinner (<33% vs. 33–48 vs. ≥48% of daily kcal) | 6 years | Consuming more calories at dinner is associated with an increased risk of obesity, metabolic syndrome and NAFLD | [206] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahluwalia, M.K. Chrononutrition—When We Eat Is of the Essence in Tackling Obesity. Nutrients 2022, 14, 5080. https://doi.org/10.3390/nu14235080
Ahluwalia MK. Chrononutrition—When We Eat Is of the Essence in Tackling Obesity. Nutrients. 2022; 14(23):5080. https://doi.org/10.3390/nu14235080
Chicago/Turabian StyleAhluwalia, Maninder Kaur. 2022. "Chrononutrition—When We Eat Is of the Essence in Tackling Obesity" Nutrients 14, no. 23: 5080. https://doi.org/10.3390/nu14235080
APA StyleAhluwalia, M. K. (2022). Chrononutrition—When We Eat Is of the Essence in Tackling Obesity. Nutrients, 14(23), 5080. https://doi.org/10.3390/nu14235080





