Effects of Selenium on Chronic Kidney Disease: A Mendelian Randomization Study
Abstract
1. Introduction
2. Materials and Methods
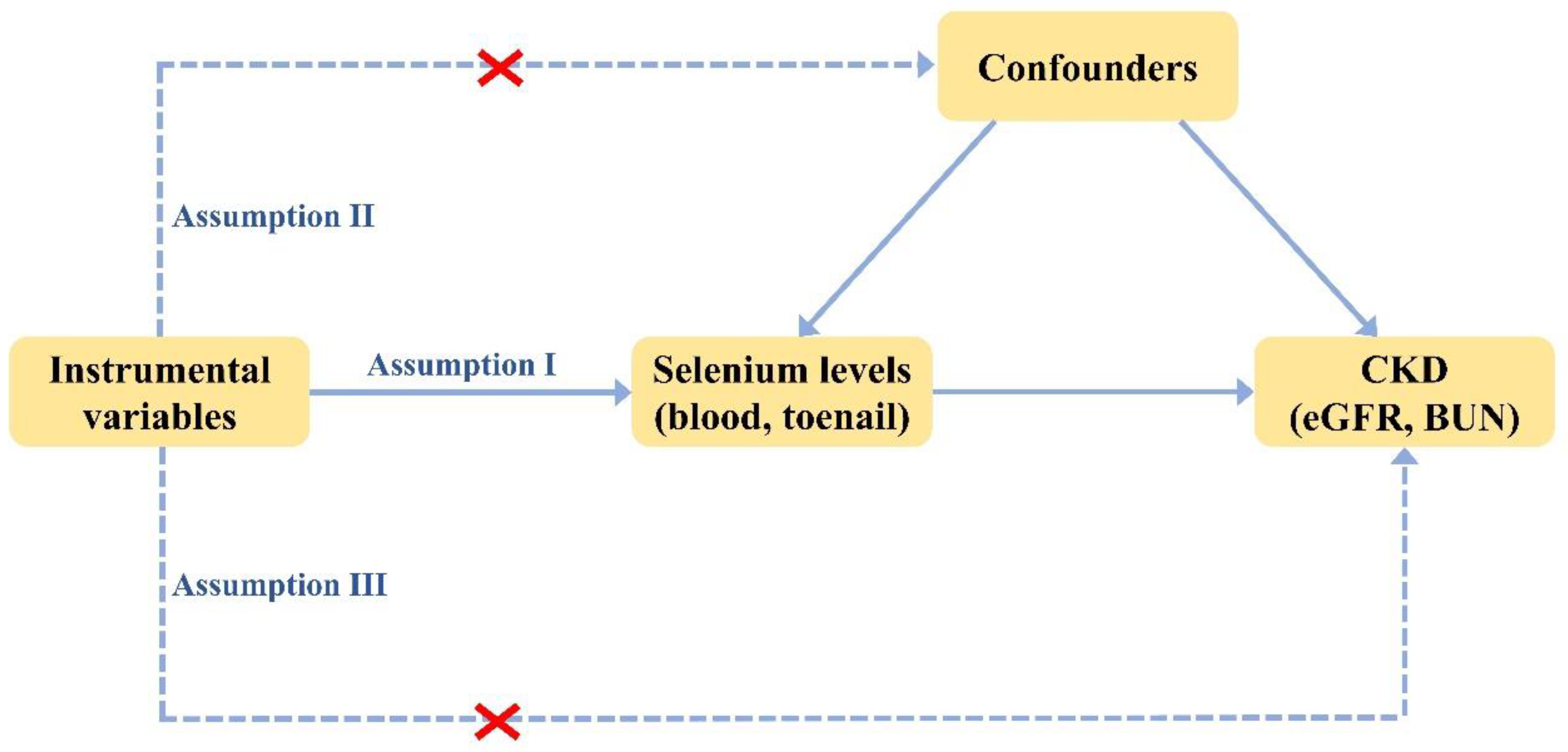
2.1. Study Design
2.2. Selenium Levels Exposure
2.3. Kidney Function Outcome
2.4. Statistical Analysis
2.5. Power Analysis
3. Results
3.1. Development of the Selenium Levels Genetic Instruments
3.2. Association of Selenium with eGFR
3.3. Association of Selenium with BUN
3.4. Power
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Kidney Foundation. K/DOQI Clinical Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and Stratification. Am. J. Kidney Dis. 2002, 39, S1–S266. [Google Scholar]
- Dashtban, A.; Mizani, M.A.; Denaxas, S.; Nitsch, D.; Quint, J.; Corbett, R.; Mamza, J.B.; Morris, T.; Mamas, M.; Lawlor, D.A.; et al. A Retrospective Cohort Study Predicting and Validating Impact of the Covid-19 Pandemic in Individuals with Chronic Kidney Disease. Kidney Int. 2022, 102, 652–660. [Google Scholar] [CrossRef]
- GBD Chronic Kidney Disease Collaboration. Global, Regional, and National Burden of Chronic Kidney Disease, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Xie, Y.; Bowe, B.; Mokdad, A.H.; Xian, H.; Yan, Y.; Li, T.; Maddukuri, G.; Tsai, C.Y.; Floyd, T.; Al-Aly, Z. Analysis of the Global Burden of Disease Study Highlights the Global, Regional, and National Trends of Chronic Kidney Disease Epidemiology from 1990 to 2016. Kidney Int. 2018, 94, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in Human Health and Disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Alexander, J.; Brismar, K.; Larsson, A. Selenium and Coenzyme Q10 Supplementation Improves Renal Function in Elderly Deficient in Selenium: Observational Results and Results from a Subgroup Analysis of a Prospective Randomised Double-Blind Placebo-Controlled Trial. Nutrients 2020, 12, 3780. [Google Scholar] [CrossRef] [PubMed]
- Zitouni, K.; Steyn, M.; Lyka, E.; Kelly, F.J.; Cook, P.; Ster, I.C.; Earle, K.A. Derepression of Glomerular Filtration, Renal Blood Flow and Antioxidant Defence in Patients with Type 2 Diabetes at High-Risk of Cardiorenal Disease. Free Radic. Biol. Med. 2020, 161, 283–289. [Google Scholar] [CrossRef]
- Mojadadi, A.; Au, A.; Salah, W.; Witting, P.; Ahmad, G. Role for Selenium in Metabolic Homeostasis and Human Reproduction. Nutrients 2021, 13, 3526. [Google Scholar] [CrossRef]
- Lönnerdal, B.; Vargas-Fernández, E.; Whitacre, M. Selenium Fortification of Infant Formulas: Does Selenium Form Matter? Food Funct. 2017, 8, 3856–3868. [Google Scholar] [CrossRef]
- Burk, R.F.; Hill, K.E. Regulation of Selenium Metabolism and Transport. Annu. Rev. Nutr. 2015, 35, 109–134. [Google Scholar] [CrossRef]
- Mal’tseva, V.N.; Goltyaev, M.V.; Turovsky, E.A.; Varlamova, E.G. Immunomodulatory and Anti-Inflammatory Properties of Selenium-Containing Agents: Their Role in the Regulation of Defense Mechanisms against COVID-19. Int. J. Mol. Sci. 2022, 23, 2360. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Zhang, J.; Xu, J.F.; Pi, J. The Advancing of Selenium Nanoparticles against Infectious Diseases. Front. Pharmacol. 2021, 12, 682284. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wen, X.; Huang, Q.; Zhu, M.; Lu, J. Selenium Status in Diet Affects Nephrotoxicity Induced by Cisplatin in Mice. Antioxidants 2022, 11, 1141. [Google Scholar] [CrossRef] [PubMed]
- Zachara, B.A. Selenium and Selenium-Dependent Antioxidants in Chronic Kidney Disease. Adv. Clin. Chem. 2015, 68, 131–151. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Choi, M.C.; Park, J.M.; Chung, A.S. Antitumor Effects of Selenium. Int. J. Mol. Sci. 2021, 22, 11844. [Google Scholar] [CrossRef] [PubMed]
- Orozco, A.; Valverde, R.C.; Olvera, A.; García, G.C. Iodothyronine Deiodinases: A Functional and Evolutionary Perspective. J. Endocrinol. 2012, 215, 207–219. [Google Scholar] [CrossRef]
- Carrero, J.J.; Qureshi, A.R.; Axelsson, J.; Yilmaz, M.I.; Rehnmark, S.; Witt, M.R.; Bárány, P.; Heimbürger, O.; Suliman, M.E.; Alvestrand, A.; et al. Clinical and Biochemical Implications of Low Thyroid Hormone Levels (Total and Free Forms) in Euthyroid Patients with Chronic Kidney Disease. J. Intern. Med. 2007, 262, 690–701. [Google Scholar] [CrossRef]
- Mostert, V. Selenoprotein P: Properties, Functions, and Regulation. Arch. Biochem. Biophys. 2000, 376, 433–438. [Google Scholar] [CrossRef]
- Zachara, B.A.; Pawluk, H.; Bloch-Boguslawska, E.; Sliwka, K.M.; Korenkiewicz, J.; Skok, Z.; Ryć, K. Tissue Level, Distribution, and Total Body Selenium Content in Healthy and Diseased Humans in Poland. Arch. Environ. Health 2001, 56, 461–466. [Google Scholar] [CrossRef]
- Xie, C.; Zeng, M.; Shi, Z.; Li, S.; Jiang, K.; Zhao, Y. Association between Selenium Status and Chronic Kidney Disease in Middle-Aged and Older Chinese Based on Chns Data. Nutrients 2022, 14, 2695. [Google Scholar] [CrossRef]
- Shen, Y.; Yin, Z.; Lv, Y.; Luo, J.; Shi, W.; Fang, J.; Shi, X. Plasma Element Levels and Risk of Chronic Kidney Disease in Elderly Populations (≥90 years Old). Chemosphere 2020, 254, 126809. [Google Scholar] [CrossRef]
- Zachara, B.A.; Gromadzinska, J.; Zbrog, Z.; Swiech, R.; Wasowicz, W.; Twardowska, E.; Jablonska, E.; Sobala, W. Selenium Supplementation to Chronic Kidney Disease Patients on Hemodialysis Does Not Induce the Synthesis of Plasma Glutathione Peroxidase. Acta Biochim. Pol. 2009, 56, 183–187. [Google Scholar] [CrossRef]
- Omrani, H.; Golmohamadi, S.; Pasdar, Y.; Jasemi, K.; Almasi, A. Effect of Selenium Supplementation on Lipid Profile in Hemodialysis Patients. J. Renal. Inj. Prev. 2016, 5, 179–182. [Google Scholar] [CrossRef]
- Omrani, H.R.; Rahimi, M.; Nikseresht, K. The Effect of Selenium Supplementation on Acute Phase Reactants and Thyroid Function Tests in Hemodialysis Patients. Nephrourol. Mon. 2015, 7, e24781. [Google Scholar] [CrossRef]
- Girelli, D.; Olivieri, O.; Stanzial, A.M.; Azzini, M.; Lupo, A.; Bernich, P.; Menini, C.; Gammaro, L.; Corrocher, R. Low Platelet Glutathione Peroxidase Activity and Serum Selenium Concentration in Patients with Chronic Renal Failure: Relations to Dialysis Treatments, Diet and Cardiovascular Complications. Clin. Sci. 1993, 84, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Ortaç, E.; Ozkaya, O.; Saraymen, R.; Yildiz, N.; Bedir, A.; Buyan, N.; Bek, K.; Okuyucu, A.; Baysal, K. Low Hair Selenium and Plasma Glutathione Peroxidase in Children with Chronic Renal Failure. Pediatr. Nephrol. 2006, 21, 1739–1745. [Google Scholar] [CrossRef]
- Wu, C.Y.; Wong, C.S.; Chung, C.J.; Wu, M.Y.; Huang, Y.L.; Ao, P.L.; Lin, Y.F.; Lin, Y.C.; Shiue, H.S.; Su, C.T.; et al. The Association between Plasma Selenium and Chronic Kidney Disease Related to Lead, Cadmium and Arsenic Exposure in a Taiwanese Population. J. Hazard. Mater. 2019, 375, 224–232. [Google Scholar] [CrossRef]
- Sabé, R.; Rubio, R.; García-Beltrán, L. Reference Values of Selenium in Plasma in Population from Barcelona. Comparison with Several Pathologies. J. Trace Elem. Med. Biol. 2002, 16, 231–237. [Google Scholar] [CrossRef]
- Zachara, B.A.; Trafikowska, U.; Adamowicz, A.; Nartowicz, E.; Manitius, J. Selenium, Glutathione Peroxidases, and Some Other Antioxidant Parameters in Blood of Patients with Chronic Renal Failure. J. Trace Elem. Med. Biol. 2001, 15, 161–166. [Google Scholar] [CrossRef]
- Rayman, M.P.; Winther, K.H.; Pastor-Barriuso, R.; Cold, F.; Thvilum, M.; Stranges, S.; Guallar, E.; Cold, S. Effect of Long-Term Selenium Supplementation on Mortality: Results from a Multiple-Dose, Randomised Controlled Trial. Free Radic. Biol. Med. 2018, 127, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Kiss, I. Importance of Selenium Homeostasis in Chronic and End-Stage Kidney Diseases. Orv. Hetil. 2013, 154, 1641–1647. [Google Scholar] [CrossRef][Green Version]
- Yuan, S.; Mason, A.M.; Carter, P.; Vithayathil, M.; Kar, S.; Burgess, S.; Larsson, S.C. Selenium and Cancer Risk: Wide-Angled Mendelian Randomization Analysis. Int. J. Cancer 2022, 150, 1134–1140. [Google Scholar] [CrossRef]
- Morris, T.T.; Heron, J.; Sanderson, E.C.M.; Davey Smith, G.; Didelez, V.; Tilling, K. Interpretation of Mendelian Randomization Using a Single Measure of an Exposure That Varies over Time. Int. J. Epidemiol. 2022, 1–11. [Google Scholar] [CrossRef]
- Davey Smith, G.; Holmes, M.V.; Davies, N.M.; Ebrahim, S. Mendel’s Laws, Mendelian Randomization and Causal Inference in Observational Data: Substantive and Nomenclatural Issues. Eur. J. Epidemiol. 2020, 35, 99–111. [Google Scholar] [CrossRef]
- Cornelis, M.C.; Fornage, M.; Foy, M.; Xun, P.; Gladyshev, V.N.; Morris, S.; Chasman, D.I.; Hu, F.B.; Rimm, E.B.; Kraft, P.; et al. Genome-Wide Association Study of Selenium Concentrations. Hum. Mol. Genet. 2015, 24, 1469–1477. [Google Scholar] [CrossRef]
- Friedman, G.D.; Cutter, G.R.; Donahue, R.P.; Hughes, G.H.; Hulley, S.B.; Jacobs, D.R.; Liu, K.; Savage, P.J. Cardia: Study Design, Recruitment, and Some Characteristics of the Examined Subjects. J. Clin. Epidemiol. 1988, 41, 1105–1116. [Google Scholar] [CrossRef]
- Jordan, J.M.; Helmick, C.G.; Renner, J.B.; Luta, G.; Dragomir, A.D.; Woodard, J.; Fang, F.; Schwartz, T.A.; Abbate, L.M.; Callahan, L.F.; et al. Prevalence of Knee Symptoms and Radiographic and Symptomatic Knee Osteoarthritis in African Americans and Caucasians: The Johnston County Osteoarthritis Project. J. Rheumatol. 2007, 34, 172–180. [Google Scholar]
- Colditz, G.A.; Hankinson, S.E. The Nurses’ Health Study: Lifestyle and Health among Women. Nat. Rev. Cancer 2005, 5, 388–396. [Google Scholar] [CrossRef]
- Chu, N.F.; Spiegelman, D.; Yu, J.; Rifai, N.; Hotamisligil, G.S.; Rimm, E.B. Plasma Leptin Concentrations and Four-Year Weight Gain among US Men. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 346–353. [Google Scholar] [CrossRef]
- Evans, D.M.; Zhu, G.; Dy, V.; Heath, A.C.; Madden, P.A.; Kemp, J.P.; McMahon, G.; St Pourcain, B.; Timpson, N.J.; Golding, J.; et al. Genome-Wide Association Study Identifies Loci Affecting Blood Copper, Selenium and Zinc. Hum. Mol. Genet. 2013, 22, 3998–4006. [Google Scholar] [CrossRef]
- Burgess, S.; Thompson, S.G. Avoiding Bias from Weak Instruments in Mendelian Randomization Studies. Int. J. Epidemiol. 2011, 40, 755–764. [Google Scholar] [CrossRef]
- Kamat, M.A.; Blackshaw, J.A.; Young, R.; Surendran, P.; Burgess, S.; Danesh, J.; Butterworth, A.S.; Staley, J.R. Phenoscanner V2: An Expanded Tool for Searching Human Genotype-Phenotype Associations. Bioinformatics 2019, 35, 4851–4853. [Google Scholar] [CrossRef]
- Stanzick, K.J.; Li, Y.; Schlosser, P.; Gorski, M.; Wuttke, M.; Thomas, L.F.; Rasheed, H.; Rowan, B.X.; Graham, S.E.; Vanderweff, B.R.; et al. Discovery and Prioritization of Variants and Genes for Kidney Function in >1.2 Million Individuals. Nat. Commun. 2021, 12, 4350. [Google Scholar] [CrossRef]
- Levey, A.S.; Eckardt, K.U.; Dorman, N.M.; Christiansen, S.L.; Cheung, M.; Jadoul, M.; Winkelmayer, W.C. Nomenclature for Kidney Function and Disease: Executive Summary and Glossary from a Kidney Disease: Improving Global Outcomes Consensus Conference. Transplantation 2020, 104, 1986–1994. [Google Scholar] [CrossRef]
- Nankivell, B.J.; Nankivell, L.F.J.; Elder, G.J.; Gruenewald, S.M. How Unmeasured Muscle Mass Affects Estimated GFR and Diagnostic Inaccuracy. EClinicalMedicine 2020, 29-30, 100662. [Google Scholar] [CrossRef]
- Wuttke, M.; Li, Y.; Li, M.; Sieber, K.B.; Feitosa, M.F.; Gorski, M.; Tin, A.; Wang, L.; Chu, A.Y.; Hoppmann, A.; et al. A Catalog of Genetic Loci Associated with Kidney Function from Analyses of a Million Individuals. Nat. Genet. 2019, 51, 957–972. [Google Scholar] [CrossRef]
- Pattaro, C.; Riegler, P.; Stifter, G.; Modenese, M.; Minelli, C.; Pramstaller, P.P. Estimating the Glomerular Filtration Rate in the General Population Using Different Equations: Effects on Classification and Association. Nephron Clin. Pract. 2013, 123, 102–111. [Google Scholar] [CrossRef]
- Mian, A.N.; Schwartz, G.J. Measurement and Estimation of Glomerular Filtration Rate in Children. Adv. Chronic Kidney Dis. 2017, 24, 348–356. [Google Scholar] [CrossRef]
- Hemani, G.; Zheng, J.; Elsworth, B.; Wade, K.H.; Haberland, V.; Baird, D.; Laurin, C.; Burgess, S.; Bowden, J.; Langdon, R.; et al. The MR-Base Platform Supports Systematic Causal Inference across the Human Phenome. elife 2018, 7, e34408. [Google Scholar] [CrossRef]
- Taylor, A.E.; Burgess, S.; Ware, J.J.; Gage, S.H.; Richards, J.B.; Davey Smith, G.; Munafò, M.R. Investigating Causality in the Association between 25(OH)D and Schizophrenia. Sci. Rep. 2016, 6, 26496. [Google Scholar] [CrossRef]
- Rees, J.M.B.; Wood, A.M.; Dudbridge, F.; Burgess, S. Robust Methods in Mendelian Randomization Via Penalization of Heterogeneous Causal Estimates. PLoS ONE 2019, 14, e0222362. [Google Scholar] [CrossRef]
- Burgess, S.; Thompson, S.G. Multivariable Mendelian Randomization: The Use of Pleiotropic Genetic Variants to Estimate Causal Effects. Am. J. Epidemiol. 2015, 181, 251–260. [Google Scholar] [CrossRef]
- Bowden, J.; Del Greco, M.F.; Minelli, C.; Davey Smith, G.; Sheehan, N.; Thompson, J. A Framework for the Investigation of Pleiotropy in Two-Sample Summary Data Mendelian Randomization. Stat. Med. 2017, 36, 1783–1802. [Google Scholar] [CrossRef]
- Verbanck, M.; Chen, C.Y.; Neale, B.; Do, R. Detection of Widespread Horizontal Pleiotropy in Causal Relationships Inferred from Mendelian Randomization between Complex Traits and Diseases. Nat. Genet. 2018, 50, 693–698. [Google Scholar] [CrossRef]
- He, D.; Cui, L. Assessing the Causal Role of Selenium in Amyotrophic Lateral Sclerosis: A Mendelian Randomization Study. Front. Genet. 2021, 12, 724903. [Google Scholar] [CrossRef]
- Brion, M.J.; Shakhbazov, K.; Visscher, P.M. Calculating Statistical Power in Mendelian Randomization Studies. Int. J. Epidemiol. 2013, 42, 1497–1501. [Google Scholar] [CrossRef]
- Burgess, S.; Davey Smith, G.; Davies, N.M.; Dudbridge, F.; Gill, D.; Glymour, M.M.; Hartwig, F.P.; Holmes, M.V.; Minelli, C.; Relton, C.L.; et al. Guidelines for Performing Mendelian Randomization Investigations. Wellcome Open Res. 2019, 4, 186. [Google Scholar] [CrossRef]
- Qian, Y.; Ye, D.; Huang, H.; Wu, D.J.H.; Zhuang, Y.; Jiang, X.; Mao, Y. Coffee Consumption and Risk of Stroke: A Mendelian Randomization Study. Ann. Neurol. 2020, 87, 525–532. [Google Scholar] [CrossRef]
- Robinson, P.C.; Choi, H.K.; Do, R.; Merriman, T.R. Insight into Rheumatological Cause and Effect through the Use of Mendelian Randomization. Nat. Rev. Rheumatol. 2016, 12, 486–496. [Google Scholar] [CrossRef]
- Larsson, S.C.; Traylor, M.; Burgess, S.; Boncoraglio, G.B.; Jern, C.; Michaëlsson, K.; Markus, H.S. Serum Magnesium and Calcium Levels in Relation to Ischemic Stroke: Mendelian Randomization Study. Neurology 2019, 92, e944–e950. [Google Scholar] [CrossRef]
- Batai, K.; Trejo, M.J.; Chen, Y.; Kohler, L.N.; Lance, P.; Ellis, N.A.; Cornelis, M.C.; Chow, H.S.; Hsu, C.H.; Jacobs, E.T. Genome-Wide Association Study of Response to Selenium Supplementation and Circulating Selenium Concentrations in Adults of European Descent. J. Nutr. 2021, 151, 293–302. [Google Scholar] [CrossRef]
- Kotur, N.; Skakic, A.; Klaassen, K.; Gasic, V.; Zukic, B.; Skodric-Trifunovic, V.; Stjepanovic, M.; Zivkovic, Z.; Ostojic, O.; Stevanovic, G.; et al. Association of Vitamin D, Zinc and Selenium Related Genetic Variants with COVID-19 Disease Severity. Front. Nutr. 2021, 8, 689419. [Google Scholar] [CrossRef]
- Maclean, K.N.; Jiang, H.; Phinney, W.N.; Keating, A.K.; Hurt, K.J.; Stabler, S.P. Taurine Alleviates Repression of Betaine-Homocysteine S-Methyltransferase and Significantly Improves the Efficacy of Long-Term Betaine Treatment in a Mouse Model of Cystathionine Β-Synthase-Deficient Homocystinuria. FASEB J. 2019, 33, 6339–6353. [Google Scholar] [CrossRef]
- Bekaert, B.; Cooper, M.L.; Green, F.R.; McNulty, H.; Pentieva, K.; Scott, J.M.; Molloy, A.M.; Rayman, M.P. Effect of Selenium Status and Supplementation with High-Selenium Yeast on Plasma Homocysteine and B Vitamin Concentrations in the Uk Elderly. Mol. Nutr. Food Res. 2008, 52, 1324–1333. [Google Scholar] [CrossRef]
- Zuhra, K.; Augsburger, F.; Majtan, T.; Szabo, C. Cystathionine-Β-Synthase: Molecular Regulation and Pharmacological Inhibition. Biomolecules 2020, 10, 697. [Google Scholar] [CrossRef]
- Seale, L.A.; Ha, H.Y.; Hashimoto, A.C.; Berry, M.J. Relationship between Selenoprotein P and Selenocysteine Lyase: Insights into Selenium Metabolism. Free Radic. Biol. Med. 2018, 127, 182–189. [Google Scholar] [CrossRef]
- Andrade, I.G.A.; Suano-Souza, F.I.; Fonseca, F.L.A.; Lago, C.S.A.; Sarni, R.O.S. Selenium Levels and Glutathione Peroxidase Activity in Patients with Ataxia-Telangiectasia: Association with Oxidative Stress and Lipid Status Biomarkers. Orphanet J. Rare Dis. 2021, 16, 83. [Google Scholar] [CrossRef]
- Rayman, M.P. The Importance of Selenium to Human Health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Zhang, L.; Zeng, H.; Cheng, W.H. Beneficial and Paradoxical Roles of Selenium at Nutritional Levels of Intake in Healthspan and Longevity. Free Radic. Biol. Med. 2018, 127, 3–13. [Google Scholar] [CrossRef]
- Zhou, J.; Huang, K.; Lei, X.G. Selenium and Diabetes--Evidence from Animal Studies. Free Radic. Biol. Med. 2013, 65, 1548–1556. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium Intake, Status, and Health: A Complex Relationship. Hormones 2020, 19, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The Use of High-Selenium Yeast to Raise Selenium Status: How Does It Measure Up? Br. J. Nutr. 2004, 92, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F. Biomarkers of Selenium Status. Nutrients 2015, 7, 2209–2236. [Google Scholar] [CrossRef]
- Rahmanto, A.S.; Davies, M.J. Selenium-Containing Amino Acids as Direct and Indirect Antioxidants. IUBMB Life 2012, 64, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Ganio, K.; Roberts, B.R. Expanding Beyond ICP-MS to Better Understand Selenium Biochemistry. Metallomics 2019, 11, 1974–1983. [Google Scholar] [CrossRef]
- Burk, R.F.; Hill, K.E.; Motley, A.K. Plasma Selenium in Specific and Non-Specific Forms. BioFactors 2001, 14, 107–114. [Google Scholar] [CrossRef]
- Zachara, B.A.; Gromadzińska, J.; Wasowicz, W.; Zbróg, Z. Red Blood Cell and Plasma Glutathione Peroxidase Activities and Selenium Concentration in Patients with Chronic Kidney Disease: A Review. Acta Biochim. Pol. 2006, 53, 663–677. [Google Scholar] [CrossRef] [PubMed]

| SNP | Nearby Gene | Chr | E/O Allele | EAF | Association with the Exposure | Association with the Outcome: eGFR | Association with the Outcome: BUN | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta | SE | p Value | Z-Score | K | R2 | Beta | SE | p Value | Beta | SE | p Value | |||||
| rs672413 | ARSB | 5 | A/G | 0.32 | 0.164418 | 0.021835 | 5.21 × 10−14 | 7.53 | 114.728 | 0.011765 | −0.0012 | 3.00 × 10−4 | 6.91 × 10−5 | 6.00 × 10−4 | 5.00 × 10−4 | 0.2719 |
| rs705415 | DMGDH | 5 | T/C | 0.14 | −0.20006 | 0.032113 | 4.64 × 10−10 | −6.23 | 78.36179 | 0.009638 | 8.00 × 10−4 | 4.00 × 10−4 | 0.0541 | −4.00 × 10−4 | 8.00 × 10−4 | 0.6377 |
| rs3797535 | DMGDH | 5 | T/C | 0.08 | 0.298102 | 0.037544 | 2.05 × 10−15 | 7.94 | 127.7319 | 0.013081 | −8.00 × 10−4 | 5.00 × 10−4 | 0.1122 | 9.00 × 10−4 | 0.001 | 0.3606 |
| rs11951068 | DMGDH | 5 | A/G | 0.07 | 0.268264 | 0.03992 | 1.86 × 10−11 | 6.72 | 91.15215 | 0.00937 | −0.0014 | 5.00 × 10−4 | 0.008477 | −2.00 × 10−4 | 8.00 × 10−4 | 0.7902 |
| rs921943 | DMGDH | 5 | T/C | 0.29 | 0.294952 | 0.022447 | 1.90 × 10−39 | 13.14 | 358.0757 | 0.035825 | −0.0021 | 3.00 × 10−4 | 2.62 × 10−12 | 0.001 | 5.00 × 10−4 | 0.05763 |
| rs10944 | BHMT2 | 5 | T/G | 0.49 | 0.257746 | 0.020375 | 1.13 × 10−36 | 12.65 | 330.9678 | 0.033203 | −0.0012 | 3.00 × 10−4 | 6.42 × 10−6 | 0.0012 | 5.00 × 10−4 | 0.01758 |
| rs567754 | BHMT | 5 | T/C | 0.34 | −0.19588 | 0.021502 | 8.38 × 10−20 | −9.11 | 168.8575 | 0.01722 | 8.00 × 10−4 | 3.00 × 10−4 | 0.005016 | −0.0016 | 5.00 × 10−4 | 3.21 × 10−3 |
| rs6859667 | HOMER1 | 5 | T/C | 0.96 | −0.35969 | 0.051978 | 4.40 × 10−12 | −6.92 | 96.71387 | 0.009936 | 0.001 | 7.00 × 10−4 | 0.1318 | −0.0017 | 0.0013 | 0.1955 |
| rs6586282 | CBS | 21 | T/C | 0.17 | −0.15971 | 0.027116 | 3.96 × 10−9 | −5.89 | 69.87277 | 0.007198 | 0.0011 | 4.00 × 10−4 | 0.00323 | 4.00 × 10−4 | 7.00 × 10−4 | 0.5388 |
| rs1789953 | CBS | 21 | T/C | 0.14 | 0.162035 | 0.029354 | 3.40 × 10−8 | 5.52 | 61.31581 | 0.006322 | −2.00 × 10−4 | 4.00 × 10−4 | 0.6217 | −0.0018 | 8.00 × 10−4 | 0.02094 |
| rs234709 | CBS | 21 | T/C | 0.45 | −0.11957 | 0.020474 | 5.23 × 10−9 | −5.84 | 68.68309 | 0.007077 | 1.00 × 10−4 | 3.00 × 10−4 | 0.7589 | 4.00 × 10−4 | 5.00 × 10−4 | 0.4208 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, S.; Zhang, L.; Ma, F.; Xue, S.; Sun, T.; Xu, Z. Effects of Selenium on Chronic Kidney Disease: A Mendelian Randomization Study. Nutrients 2022, 14, 4458. https://doi.org/10.3390/nu14214458
Fu S, Zhang L, Ma F, Xue S, Sun T, Xu Z. Effects of Selenium on Chronic Kidney Disease: A Mendelian Randomization Study. Nutrients. 2022; 14(21):4458. https://doi.org/10.3390/nu14214458
Chicago/Turabian StyleFu, Shaojie, Li Zhang, Fuzhe Ma, Shuai Xue, Tao Sun, and Zhonggao Xu. 2022. "Effects of Selenium on Chronic Kidney Disease: A Mendelian Randomization Study" Nutrients 14, no. 21: 4458. https://doi.org/10.3390/nu14214458
APA StyleFu, S., Zhang, L., Ma, F., Xue, S., Sun, T., & Xu, Z. (2022). Effects of Selenium on Chronic Kidney Disease: A Mendelian Randomization Study. Nutrients, 14(21), 4458. https://doi.org/10.3390/nu14214458






