Milk Formula Enriched with Sodium Butyrate Influences Small Intestine Contractility in Neonatal Pigs
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
2.2. Sample Preparation
2.3. Intestinal Contractility In Vitro
2.4. Intestinal Morphology
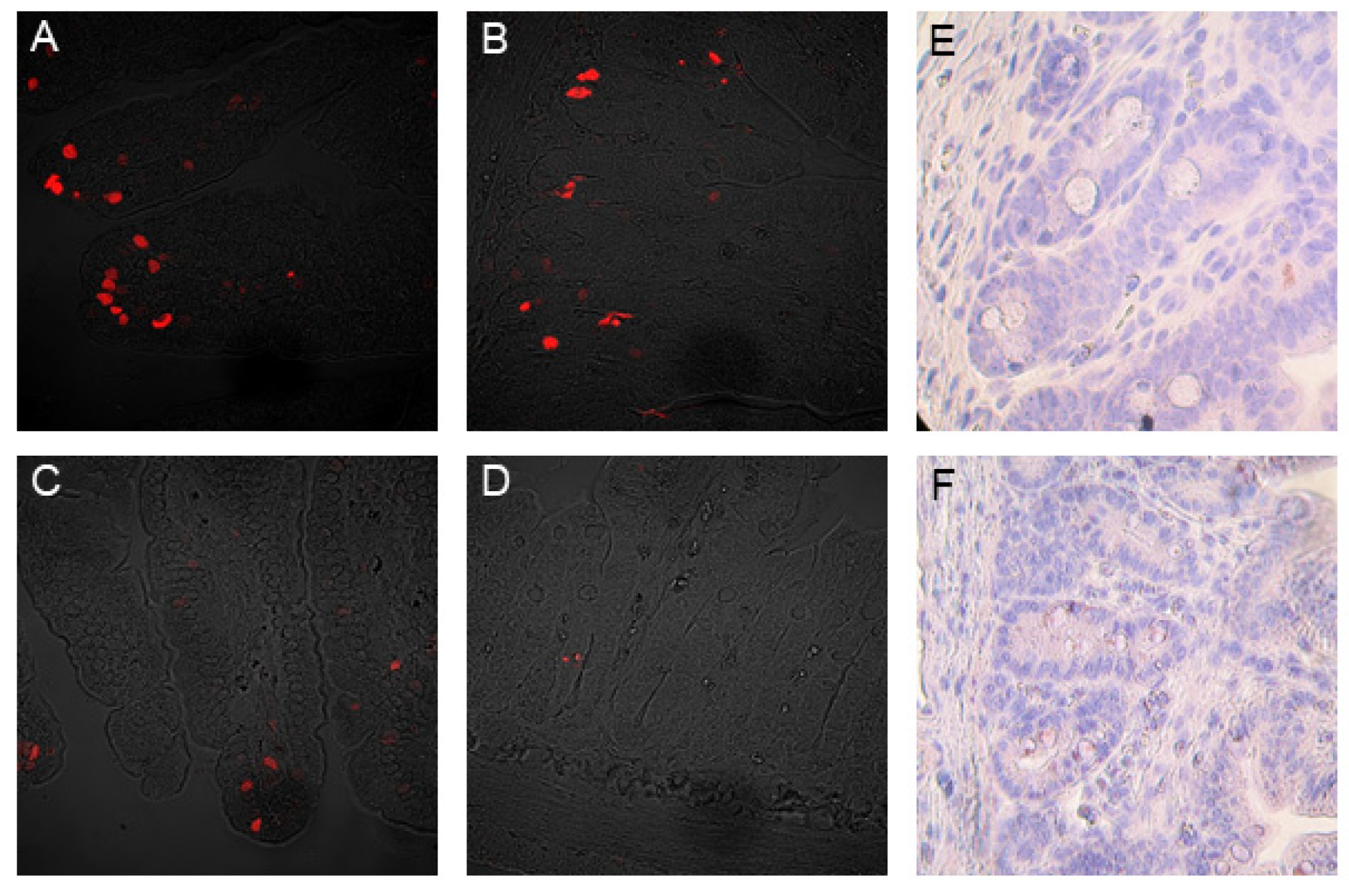
2.5. TUNEL Assay
2.6. Immunohistochemistry
2.7. Western Blotting
2.8. Statistical Analysis
3. Results
3.1. Piglet Growth and Intestinal Morphometry
3.2. Spontaneus Intestinal Contractility
3.3. Electric Field Stimulation
3.4. ACh-Stimulated Intestinal Contractility
3.5. The Mitotic and Apoptotic Indices
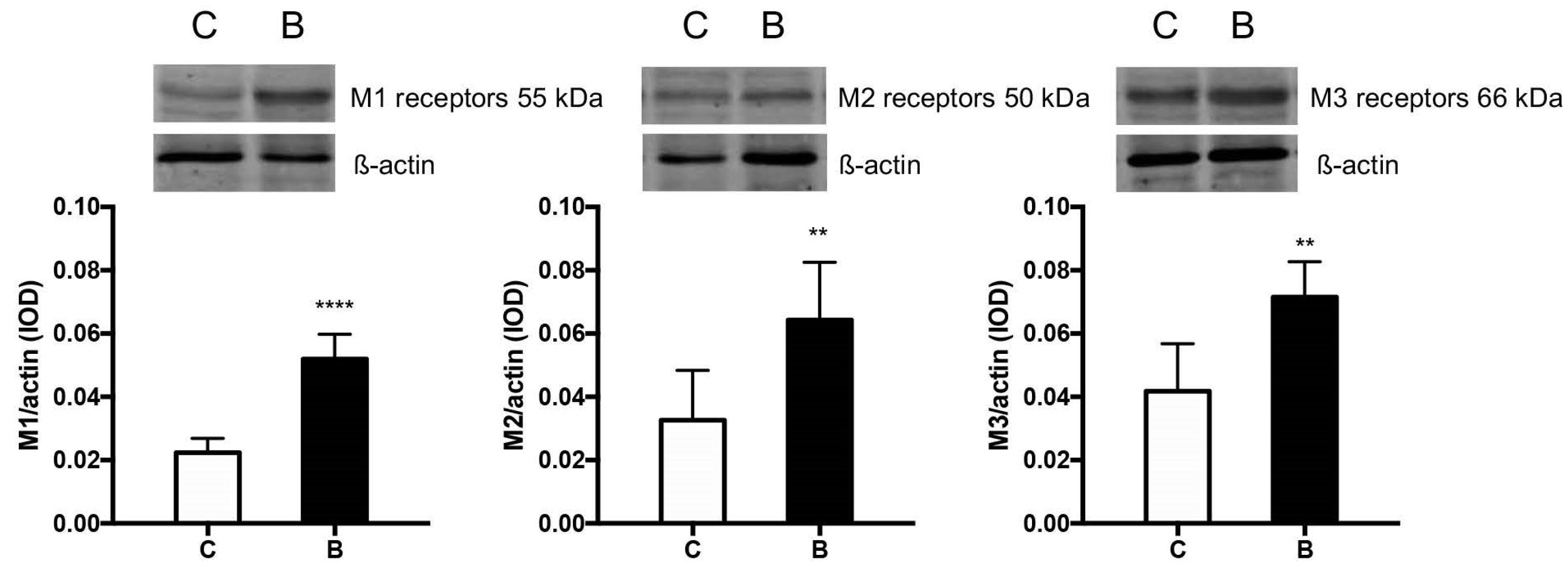
3.6. Muscarinic ACh Receptors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lorijn, F.; Voskuijl, W.P.; Omari, T.I.; Kok, J.H.; Taminiau, J.A.; Benninga, M.A. Assessment of the rectoanal inhibitory reflex in preterm infants with delayed meconium passage. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 434–437. [Google Scholar] [CrossRef] [PubMed]
- Le Huërou-Luron, I.; Blat, S.; Boudry, G. Breast-v. formula-feeding: Impacts on the digestive tract and immediate and long-term health effects. Nutr. Res. Rev. 2010, 23, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Bridgman, S.L.; Azad, M.B.; Field, C.J.; Haqq, A.M.; Becker, A.B.; Mandhane, P.J.; Subbarao, P.; Turvey, S.E.; Sears, M.R.; Scott, J.A.; et al. Fecal short-chain fatty acid variations by breastfeeding status in infants at 4 months: Differences in relative versus absolute concentrations. Front. Nutr. 2017, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- He, X.; M’Clorry, S.; Hernell, O.; Lönnerdal, B.; Slupsky, C.M. Digestion of human milk fat in healthy infants. Nutr. Res. 2020, 83, 15–29. [Google Scholar] [CrossRef]
- Gregg, B.; Ellsworth, L.; Pavela, G.; Shah, K.; Berger, P.K.; Isganaitis, E.; VanOmen, S.; Demerath, E.W.; Field, D.A. Bioactive compounds in mothers milk affecting offspring outcomes: A narrative review. Pediatr. Obes. 2022, 17, e12892. [Google Scholar] [CrossRef]
- Stinson, L.F.; Gay, M.C.L.; Koleva, P.T.; Eggesbø, M.; Johnson, C.C.; Wegienka, G.; du Toit, E.; Shimojo, N.; Munblit, D.; Campbell, D.E.; et al. Human milk from atopic mothers has lower levels of short chain fatty acids. Front. Immunol. 2020, 11, 1427. [Google Scholar] [CrossRef] [PubMed]
- Prentice, P.M.; Schoemaker, M.H.; Vervoort, J.; Hettinga, K.; Lambers, T.T.; van Tol, E.A.F.; Acerini, C.L.; Laurentya, O.; Clive, J.P.; Ieuan, A.H.; et al. Human milk short-chain fatty acids composition is associated with adiposity outcomes in infants. J. Nutr. 2019, 149, 716–722. [Google Scholar] [CrossRef]
- Favre, A.; Szylit, O.; Popot, F.; Catala, I.; Rondeau, C.; Maurage, C.; Gold, F.; Borderon, J.C.; Butel, M.J. Diet, length of gestation, and fecal short chain fatty acids in healthy premature neonates. J. Parenter. Enter. Nutr. 2002, 26, 51–56. [Google Scholar] [CrossRef]
- Pourcyrous, M.; Nolan, V.G.; Goodwin, A.; Davis, S.L.; Buddington, R.K. Fecal short-chain fatty acids of very-low-birth-weight preterm infants fed expressed breast milk or formula. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 725–731. [Google Scholar] [CrossRef]
- Guilloteau, P.; Martin, L.; Eeckhaut, V.; Ducatelle, R.; Zabielski, R. From the gut to the peripheral tissues: The multiple effects of butyrate. Nutr. Res. Rev. 2010, 23, 366–384. [Google Scholar] [CrossRef]
- Canani, R.B.; Di Costanzo, M.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519. [Google Scholar] [CrossRef]
- Leonel, A.J.; Alvarez-Leite, J.I. Butyrate: Implications for intestinal function. Curr. Opin. Clin. Nutr. Metab. Care. 2012, 15, 474–479. [Google Scholar] [CrossRef]
- Soret, R.; Chevalier, J.; De Coppet, P.; Poupeau, G.; Derkinderen, P.; Sagain, J.P.; Neunlist, M. Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats. Gastroenterology 2010, 138, 1772–1782. [Google Scholar] [CrossRef]
- McManus, C.M.; Michel, K.E.; Simon, D.M.; Washabau, R.J. Effect of short-chain fatty acids on contraction of smooth muscle in the canine colon. Am. J. Vet. Res. 2020, 63, 295–300. [Google Scholar] [CrossRef]
- Rondeau, M.P.; Meltzer, K.; Michel, K.E. Short chain fatty acids stimulate feline colonic smooth muscle contraction. J. Feline Med. Surg. 2003, 5, 167–173. [Google Scholar] [CrossRef]
- Bajka, B.H.; Clarke, J.M.; Topping, D.L.; Cobiac, L.; Abeywardena, M.Y.; Patten, G.S. Butyrylated starch increases large bowel butyrate levels and lowers colonic smooth muscle contractility in rats. Nutr. Res. 2010, 30, 427–434. [Google Scholar] [CrossRef]
- Cuche, G.; Malbert, C.H. Short-chain fatty acids present in the ileum inhibit fasting gastrointestinal motility in conscious pigs. J. Neurogastroenterol. Motil. 1999, 11, 219–225. [Google Scholar] [CrossRef]
- Suply, E.; de Vries, P.; Soret, R.; Cossais, F.; Neunlist, M. Butyrate enemas enhance both cholinergic and nitrergic phenotype of myenteric neurons and neuromuscular transmission in newborn rat colon. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G1373–G1380. [Google Scholar] [CrossRef]
- Cossais, F.; Durand, T.; Chevalier, J.; Boudaud, M.; Kermarrec, L.; Aubert, P.; Neveu, I.; Naveilhan, P.; Neunlist, M. Postnatal development of the myenteric glial network and its modulation by butyrate. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G941–G951. [Google Scholar] [CrossRef]
- Kotunia, A.; Woliński, J.; Laubitz, D.; Jurkowska, M.; Romé, V.; Gouilloteau, P.; Zabielski, R. Effect of sodium butyrate on the small intestine development in neonatal piglets feed by artificial sow. J. Physiol. Pharmacol. 2004, 55 (Suppl. S2), 59–68. [Google Scholar]
- Bartholome, A.L.; Albin, D.M.; Baker, D.H.; Holst, J.J.; Tappenden, K.A. Supplementation of total parenteral nutrition with butyrate acutely increases structural aspects of intestinal adaptation after an 80% jejunoileal resection in neonatal piglets. J. Parenter. Enter. Nutr. 2004, 28, 210–222. [Google Scholar] [CrossRef]
- Słupecka, M.; Woliński, J.; Pierzynowski, S.G. The effects of enteral ghrelin administration on the remodeling of the small intestinal mucosa in neonatal piglets. Regul. Pept. 2012, 174, 38–45. [Google Scholar] [CrossRef]
- Słupecka-Ziemilska, M.; Szczurek, P.; Boryczka, M.; Gajewska, M.; Wychowański, P.; Kuwahara, A.; Kato, I.; Dzięgelewska, Ż.; Woliński, J. The effects of intra-stomach obestatin administration on intestinal contractility in neonatal piglets fed milk formula. PLoS ONE 2020, 15, e0230190. [Google Scholar] [CrossRef]
- Zhang, X. Influence of Food Intervention on Enteric Nervous System and Enteroendocrine Cells Development in A Small for Gestational Age Piglet Model. Master’s Thesis, University of Illinois, Urbana-Champaign, IL, USA, 2015. [Google Scholar]
- Woliński, J.; Leśniewska, V.; Biernat, M.; Bąblewska, M.; Korczyński, W.; Zabielski, R. Exogenous leptin influences gastrointestinal growth and in vitro small intestinal motility in neonatal piglets—Preliminary results. J. Anim. Feed Sci. 2001, 10 (Suppl. S2), 249–254. [Google Scholar] [CrossRef]
- Riezzo, G.; Castellana, R.M.; De Bellis, T.; Laforgia, F.; Indrio, F.; Chiloiro, M. Gastric electrical activity in normal neonates during the first year of life: Effect of feeding with breast milk and formula. J. Gastroenterol. 2003, 38, 836–843. [Google Scholar] [CrossRef]
- Lu, H.; Su, S.; Ajuwom, K.M. Butyrate supplementation to gestating sows and piglets induces muscle and adipose tissue oxidative genes and improves growth performance. Sci. J. Anim. Sci. 2012, 90 (Suppl. S4), 430–432. [Google Scholar] [CrossRef]
- Le Gall, M.; Gallois, M.; Seve, B.; Louveau, I.; Holst, J.J.; Oswald, I.P.; Lallès, J.; Guilloteau, P. Comparative effect of orally administered sodium butyrate before or after weaning on growth and several indices of gastrointestinal biology of piglets. Br. J. Nutr. 2009, 102, 1285–1296. [Google Scholar] [CrossRef]
- Manzanilla, E.G.; Nofrarias, M.; Anguita, M.; Castillo, M.; Perez, J.F.; Martín-Orúe, S.M.; Kamel, C.; Gasa, J. Effects of butyrate, avilamycin, and a plant extract combination on the intestinal equilibrium of early-weaned pigs. Sci. J. Anim. Sci. 2006, 84, 2743–2751. [Google Scholar] [CrossRef]
- Górka, P.; Śliwiński, B.; Flaga, J.; Olszewski, J.; Wojciechowski, M.; Krupa, K.; Godlewski, M.M.; Zabielski, R.; Kowalski, Z.M. Effect of exogenous butyrate on gastrointestinal tract of sheep. I. Structure and function of the rumen, omasum, and abomasum. Sci. J. Anim. Sci. 2018, 96, 5311–5324. [Google Scholar] [CrossRef]
- Yajima, T. Contractile effect of short-chain fatty acids on the isolated colon of the rat. Physiol. J. 1985, 368, 667–678. [Google Scholar] [CrossRef]
- Hurst, N.R.; Kendig, D.M.; Murthy, K.S.; Grider, J.R. The short chain fatty acids, butyrate and propionate, have differential effects on the motility of the guinea pig colon. Neurogastroenterol. Motil. 2014, 26, 1586–1596. [Google Scholar] [CrossRef] [PubMed]
- Yajima, T. Effect of sodium propionate on the contractile response of the rat ileum in situ. Jpn. J. Pharmacol. 1984, 35, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Frankel, W.L.; Zhang, W.; Singh, A.; Klurfeld, D.M.; Don, S.; Sakata, T.; Modlin, I.; Rombeau, J.L. Mediation of the trophic effects of short-chain fatty acids on rat jejunum and colon. Gastroenterology 1994, 106, 375–380. [Google Scholar] [CrossRef]
- Matini, P.; Mayer, B.; Faussone-Pellegrini, M. Neurochemical differentiation of rat enteric neurons during pre- and postnatal life. Cell Tissue Res. 1997, 288, 11–23. [Google Scholar] [CrossRef]
- Nøhr, M.K.; Pedersen, M.H.; Gille, A.; Egerod, K.L.; Engelstoft, M.S.; Husted, A.S.; Sichlau, R.M.; Grunddal, K.V.; Poulsen, S.S.; Han, S. GPR41/FFAR3 and GPR43/FFAR2 as cosensors for short-chain fatty acids in enteroendocrine cells vs. FFAR3 in enteric neurons and FFAR2 in enteric leukocytes. Endocrinology 2013, 154, 3552–3564. [Google Scholar] [CrossRef]
- Miletta, M.C.; Petkovic, V.; Eblé, A.; Ammann, R.A.; Flück, C.E.; Mullis, P. Butyrate increases intracellular calcium levels and enhances growth hormone release from rat anterior pituitary cells via the G-protein-coupled receptors GPR41 and 43. PLoS ONE 2014, 9, e107388. [Google Scholar] [CrossRef]
- Biernat, M.; Zabielski, R.; Sysa, P.; Sosak-Świderska, B.; Le Huërou-Lurone, I.; Guilloteau, P. Small intestinal and pancreatic microstructures are modified by an intraduodenal CCK-A receptor antagonist administration in neonatal calves. Regul. Pept. 1999, 85, 77–85. [Google Scholar] [CrossRef]
- Guilloteau, P.; Zabielski, R.; David, J.C.; Blum, J.W.; Morisset, J.A.; Biernat, M.; Woliński, J.; Laubitz, D.; Hamon, Y. Sodium-butyrate as a growth promoter in milk replacer formula for young calves. J. Dairy Sci. 2009, 92, 1038–1049. [Google Scholar] [CrossRef]
- Wang, J.F.; Chen, Y.X.; Xang, Z.X. Effect of sodium butyrate on the structure of the small intestine mucous epithelium of weaning piglets. Chin. J. Vet. Sci. Technol. 2005, 35, 298–301. [Google Scholar]
- Burns, A.J.; Roberts, R.R.; Bornstein, J.C.; Young, H.M. Development of the enteric nervous system and its role in intestinal motility during fetal and early postnatal stages. Semin. Pediatr Surg. 2009, 18, 196–205. [Google Scholar] [CrossRef]
- Woliński, J.; Słupecka-Ziemilska, M.; Boryczka, M.; Grzesiak, P.; Kwiatkowski, J.; Kotarba, G. Small intestine motility development in newborn mammals. Dev. Period. Med. 2016, 20, 53–60. [Google Scholar]


| Amplitude of EFS-Induced Contraction (mm) | ||||||
|---|---|---|---|---|---|---|
| No Additional Treatment | TTX | |||||
| 0.5 Hz | 5 Hz | 50 Hz | 0.5 Hz | 5 Hz | 50 Hz | |
| Duodenum | ||||||
| C | 1.67 ± 0.57 | 1.89 ± 0.76 | 2.08 ± 0.58 | 0.79 ± 0.32 §§ | 0.84 ± 0.34 §§ | 1.38 ± 0.25 § |
| B | 2.90 ± 0.50 *** | 3.30 ± 0.59 *** | 3.53 ± 0.74 *** | 1.15 ± 0.17 *§§§ | 1.55 ± 0.23 ***§§§ | 1.73 ± 0.24 *§§§ |
| p value | 0.0004 | 0.0009 | 0.0007 | 0.014 | 0.0002 | 0.014 |
| Middle jejunum | ||||||
| C | 1.23 ± 0.24 | 1.73 ± 0.66 | 1.91 ± 0.36 | 0.59 ± 0.10 §§§ | 0.61 ± 0.14 §§§ | 1.11 ± 0.20 §§§ |
| B | 1.91 ± 0.36 *** | 2.34 ± 0.26 * | 2.79 ± 0.54 * | 0.74 ± 0.08 **§§§ | 0.96 ± 0.19 ***§§§ | 1.38 ± 0.25 *§§§ |
| p value | 0.0006 | 0.030 | 0.011 | 0.003 | 0.001 | 0.030 |
| ACh−9 | ACh−8 | ACh−7 | ACh−6 | ACh−5 | ACh−4 | ATR−6 | |
|---|---|---|---|---|---|---|---|
| Duodenum | |||||||
| C | 4.5 ± 0.5 | 4.5 ± 0.5 | 4.6 ± 0.5 | 4.8 ± 0.5 | 6.1 ± 1.2 | 8.2 ± 1.0 | 0.9 ± 0.2 |
| B | 5.4 ± 1.1 * | 5.6 ± 0.9 ** | 6.1 ± 0.9 *** | 6.2 ± 1.0 *** | 7.4 ± 1.4 * | 11.0 ± 1.9 *** | 2.2 ± 0.4 ** |
| p value | 0.022 | 0.002 | 0.0001 | 0.0005 | 0.025 | 0.0004 | 0.001 |
| Middle jejunum | |||||||
| C | 3.3 ± 0.8 | 3.4 ± 0.9 | 3.4 ± 1.0 | 3.6 ± 0.9 | 4.8 ± 1.6 | 8.9 ± 2.4 | 0.6 ± 0.1 |
| B | 5.3 ± 1.5 *** | 5.4 ± 1.5 *** | 5.9 ± 1.5 *** | 6.4 ± 2.1 *** | 7.6 ± 2.3 ** | 11.9 ± 3.4 * | 2.0 ± 1.2 |
| p value | 0.0004 | 0.0009 | 0.0001 | 0.0002 | 0.003 | 0.034 | 0.342 |
| Group/Segment | Duodenum | Middle Jejunum | p Value |
|---|---|---|---|
| Mitotic index | |||
| C | 1.11 ± 0.48 | 0.88 ± 0.35 | 0.181 |
| B | 1.37 ± 0.26 | 1.32 ± 0.20 ** | 0.622 |
| p value | 0.271 | 0.001 | |
| Apoptotic index—crypts | |||
| C | 3.19 ± 0.45 | 3.11 ± 0.69 | 0.722 |
| B | 2.87 ± 0.61 | 3.43 ± 0.93 | 0.105 |
| p value | 0.153 | 0.358 | |
| Apoptotic index—villi | |||
| C | 3.42 ± 0.41 | 3.28 ± 0.58 | 0.487 |
| B | 3.13 ± 0.57 | 2.51 ± 0.51 **§ | 0.011 |
| p value | 0.165 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Słupecka-Ziemilska, M.; Pierzynowski, S.G.; Szczurek, P.; Pierzynowska, K.; Wychowański, P.; Seklecka, B.; Koperski, M.; Starzyńska, A.; Szkopek, D.; Donaldson, J.; et al. Milk Formula Enriched with Sodium Butyrate Influences Small Intestine Contractility in Neonatal Pigs. Nutrients 2022, 14, 4301. https://doi.org/10.3390/nu14204301
Słupecka-Ziemilska M, Pierzynowski SG, Szczurek P, Pierzynowska K, Wychowański P, Seklecka B, Koperski M, Starzyńska A, Szkopek D, Donaldson J, et al. Milk Formula Enriched with Sodium Butyrate Influences Small Intestine Contractility in Neonatal Pigs. Nutrients. 2022; 14(20):4301. https://doi.org/10.3390/nu14204301
Chicago/Turabian StyleSłupecka-Ziemilska, Monika, Stefan Grzegorz Pierzynowski, Paulina Szczurek, Kateryna Pierzynowska, Piotr Wychowański, Blanka Seklecka, Maciej Koperski, Anna Starzyńska, Dominika Szkopek, Janine Donaldson, and et al. 2022. "Milk Formula Enriched with Sodium Butyrate Influences Small Intestine Contractility in Neonatal Pigs" Nutrients 14, no. 20: 4301. https://doi.org/10.3390/nu14204301
APA StyleSłupecka-Ziemilska, M., Pierzynowski, S. G., Szczurek, P., Pierzynowska, K., Wychowański, P., Seklecka, B., Koperski, M., Starzyńska, A., Szkopek, D., Donaldson, J., Andrzejewski, K., & Woliński, J. (2022). Milk Formula Enriched with Sodium Butyrate Influences Small Intestine Contractility in Neonatal Pigs. Nutrients, 14(20), 4301. https://doi.org/10.3390/nu14204301







