Whole Grain Consumption and Inflammatory Markers: A Systematic Literature Review of Randomized Control Trials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Eligibility and Exclusion Criteria
2.2. Search Strategy
2.3. Study Selection, Data Extraction, and Quality Assessment
2.4. Data Analysis
3. Results
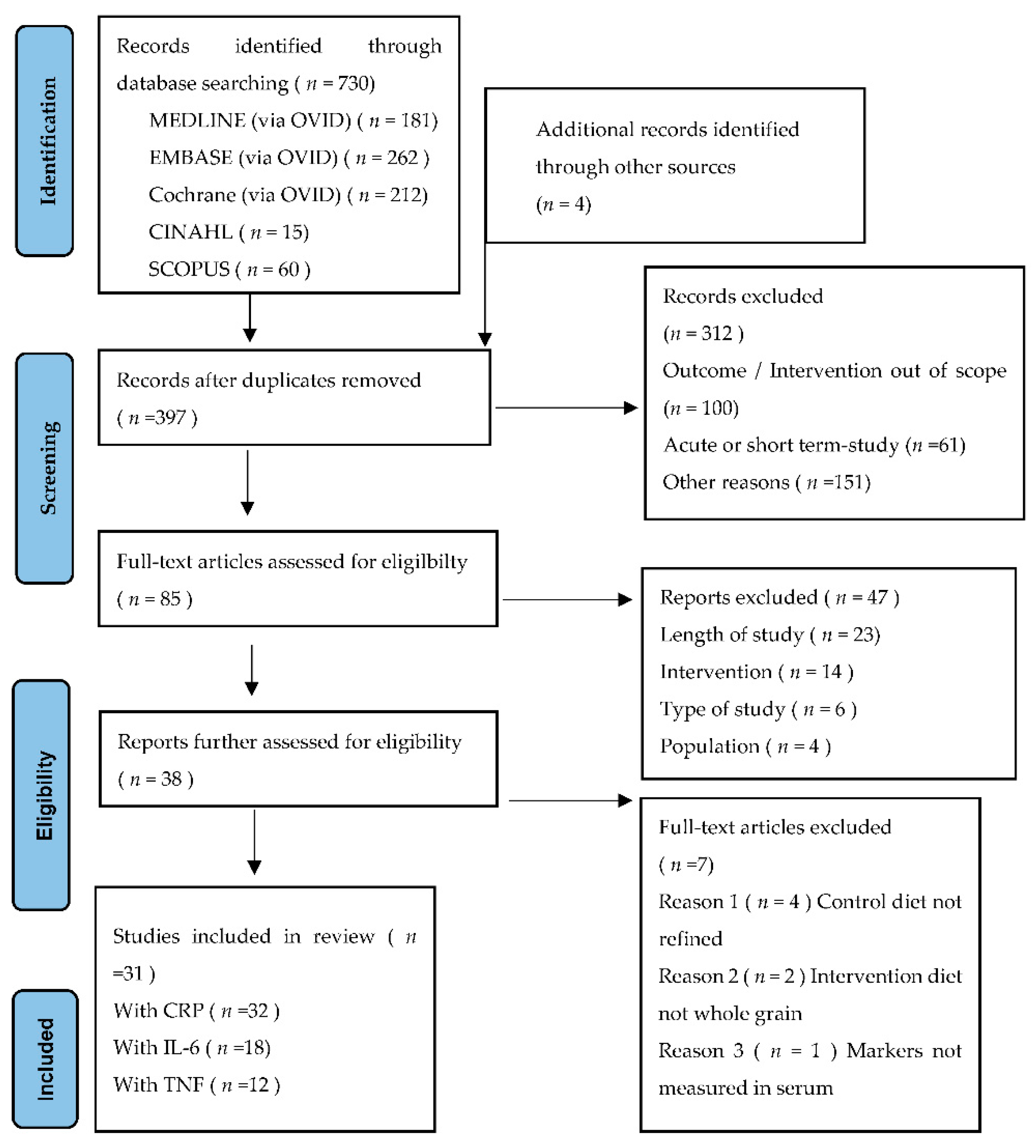
3.1. Search Results and Study Selection
3.2. Study Characteristics
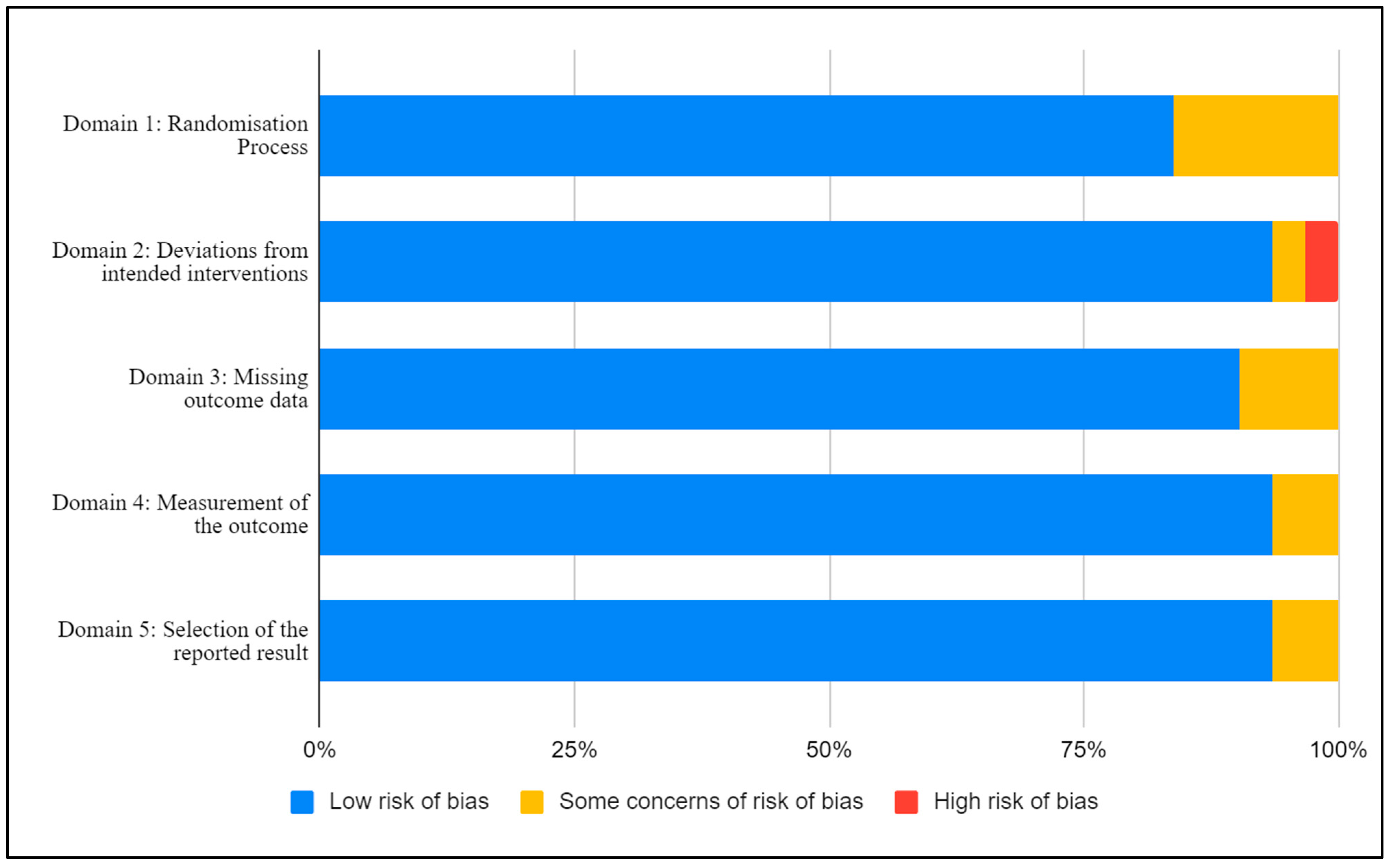
3.3. Risk of Bias
3.4. Effect of the Intervention on the Outcome
3.4.1. Healthy Individuals
3.4.2. Overweight or Obese Individuals
3.4.3. Individuals with Pre-Existing Conditions
3.4.4. Individuals with Other Conditions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Food Standards Australia New Zealand (FSANZ) Wholegrain Definition. Available online: https://www.foodstandards.gov.au/consumer/nutrition/wholegrain/Pages/default.aspx (accessed on 25 August 2021).
- Van der Kamp, J.-W.; Jones, J.M.; Miller, K.B.; Ross, A.B.; Seal, C.J.; Tan, B.; Beck, E.J. Consensus, global definitions of whole grain as a food ingredient and of whole-grain foods presented on behalf of the whole grain initiative. Nutrients 2022, 14, 138. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole grain and refined grain consumption and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aune, D.; Chan, D.S.M.; Lau, R.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dietary fibre, whole grains, and risk of colorectal cancer: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2011, 343, d6617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vieira, A.R.; Abar, L.; Chan, D.S.M.; Vingeliene, S.; Polemiti, E.; Stevens, C.; Greenwood, D.; Norat, T. Foods and beverages and colorectal cancer risk: A systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR continuous update project. Ann. Oncol. 2017, 28, 1788–1802. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Laure Preterre, A.; Iqbal, K.; Bechthold, A.; de Henauw, S.; Michels, N.; Devleesschauwer, B.; et al. Food groups and risk of colorectal cancer. Int. J. Cancer 2018, 142, 1748–1758. [Google Scholar] [CrossRef] [Green Version]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Morenga, L.T. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef] [Green Version]
- Kissock, K.R.; Neale, E.P.; Beck, E.J. Whole grain food definition effects on determining associations of whole grain intake and body weight changes: A systematic review. Adv. Nutr. 2020, 12, 693–707. [Google Scholar] [CrossRef]
- Marshall, S.; Petocz, P.; Duve, E.; Abbott, K.; Cassettari, T.; Blumfield, M.; Fayet-Moore, F. The effect of replacing refined grains with whole grains on cardiovascular risk factors: A systematic review and meta-analysis of randomized controlled trials with GRADE clinical recommendation. J. Acad. Nutr. Diet. 2020, 120, 1859–1883. [Google Scholar] [CrossRef]
- Curtain, F.; Grafenauer, S. Historical and global perspectives on grains and whole grains within dietary guidelines. Cereal Foods World 2020, 65, 1–7. [Google Scholar]
- Australian Institute of Health and Welfare. Exploring the Definition of Chronic Conditions for Collective Monitoring in Australia; Australian Institute of Health and Welfare: Darlinghurst, Australia, 2021. Available online: https://www.aihw.gov.au/reports/chronic-disease/exploring-the-definition-of-chronic-conditions/summary (accessed on 25 August 2021)ISBN 978-1-76054-803-2.
- Galea, L.; Beck, E.; Probst, Y.; Cashman, C. Whole grain intake of Australians estimated from a cross-sectional analysis of dietary intake data from the 2011–13 Australian Health Survey. Public Health Nutr. 2017, 20, 2166–2172. [Google Scholar] [CrossRef] [Green Version]
- GBD 2017 Risk Factor Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1923–1994. [Google Scholar]
- Browning, L.M.; Krebs, J.D.; Jebb, S.A. Discrimination ratio analysis of inflammatory markers: Implications for the study of inflammation in chronic disease. Metabolism 2004, 53, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Sang, S.; Idehen, E.; Zhao, Y.; Chu, Y. Emerging science on whole grain intake and inflammation. Nutr Rev. 2020, 78 (Suppl. 1), 21–28. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, S.; Sadeghi, O.; Sadeghian, M.; Sadeghi, N.; Larijani, B.; Esmaillzadeh, A. The effect of whole-grain intake on biomarkers of subclinical inflammation: A comprehensive meta-analysis of randomized controlled trials. Adv. Nutr. 2020, 11, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Hajihashemi, P.; Haghighatdoost, F. Effects of whole-grain consumption on selected biomarkers of systematic inflammation: A Systematic review and meta-analysis of randomized controlled trials. J. Am. Coll. Nutr. 2019, 38, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Faulkner, D.A.; Wong, J.M.; de Souza, R.; Emam, A.; Parker, T.L.; Vidgen, E.; Lapsley, K.G.; et al. Effects of a dietary portfolio of cholesterol-lowering foods vs. lovastatin on serum lipids and C-reactive protein. JAMA 2003, 290, 502–510. [Google Scholar] [CrossRef] [Green Version]
- Kristensen, M.; Toubro, S.; Jensen, M.G.; Ross, A.B.; Riboldi, G.; Petronio, M.; Bugel, S.; Tetens, I.; Astrup, A. Whole grain compared with refined wheat decreases the percentage of body fat following a 12-week, energy-restricted dietary intervention in postmenopausal women. J. Nutr. 2012, 142, 710–716. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [Green Version]
- Brownlee, I.A.; Moore, C.; Chatfield, M.; Richardson, D.P.; Ashby, P.; Kuznesof, S.A.; Jebb, S.A.; Seal, C.J. Markers of cardiovascular risk are not changed by increased whole-grain intake: The WHOLEheart study, a randomised, controlled dietary intervention. Br. J. Nutr. 2010, 104, 125–134. [Google Scholar] [CrossRef]
- Ma, X.; Gu, J.; Zhang, Z.; Jing, L.; Xu, M.; Dai, X.; Jiang, Y.; Bao, L.; Cai, X.; Ding, Y.; et al. Effects of Avena nuda L. on metabolic control and cardiovascular disease risk among Chinese patients with diabetes and meeting metabolic syndrome criteria: Secondary analysis of a randomized clinical trial. Eur. J. Clin. Nutr. 2013, 67, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Ampatzoglou, A.; Williams, C.L.; Atwal, K.K.; Maidens, C.M.; Ross, A.B.; Thielecke, F.; Jonnalagadda, S.S.; Kennedy, O.B.; Yaqoob, P. Effects of increased wholegrain consumption on immune and inflammatory markers in healthy low habitual wholegrain consumers. Eur. J. Nutr. 2016, 55, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Tengblad, S.; Karlstrom, B.; Kamal-Eldin, A.; Landberg, R.; Basu, S.; Aman, P.; Vessby, B. Whole-grain foods do not affect insulin sensitivity or markers of lipid peroxidation and inflammation in healthy, moderately overweight subjects. J. Nutr. 2007, 137, 1401–1407. [Google Scholar] [CrossRef]
- Connolly, M.L.; Tzounis, X.; Tuohy, K.M.; Lovegrove, J.A. Hypocholesterolemic and prebiotic effects of a whole-grain oat-based granola breakfast cereal in a cardio-metabolic “at risk” population. Front. Microbiol. 2016, 7, 1675. [Google Scholar] [CrossRef] [Green Version]
- Giacco, R.; Lappi, J.; Costabile, G.; Kolehmainen, M.; Schwab, U.; Landberg, R.; Uusitupa, M.; Poutanen, K.; Pacini, G.; Rivellese, A.A.; et al. Effects of rye and whole wheat versus refined cereal foods on metabolic risk factors: A randomised controlled two-centre intervention study. Clin. Nutr. 2013, 32, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Harris Jackson, K.; West, S.G.; Vanden Heuvel, J.P.; Jonnalagadda, S.S.; Ross, A.B.; Hill, A.M.; Grieger, J.A.; Lemieux, S.K.; Kris-Etherton, P.M. Effects of whole and refined grains in a weight-loss diet on markers of metabolic syndrome in individuals with increased waist circumference: A randomized controlled-feeding trial. Am. J. Clin. Nutr. 2014, 100, 577–586. [Google Scholar] [CrossRef] [Green Version]
- Hoevenaars, F.P.M.; Esser, D.; Schutte, S.; Priebe, M.G.; Vonk, R.J.; van den Brink, W.J.; van der Kamp, J.W.; Stroeve, J.H.M.; Afman, L.A.; Wopereis, S. Whole grain wheat consumption affects postprandial inflammatory response in a randomized controlled trial in overweight and obese adults with mild hypercholesterolemia in the graandioos study. J. Nutr. 2019, 149, 2133–2144. [Google Scholar] [CrossRef]
- Iversen, K.N.; Carlsson, F.; Andersson, A.; Michaelsson, K.; Langton, M.; Riserus, U.; Hellstrom, P.M.; Landberg, R. A hypocaloric diet rich in high fiber rye foods causes greater reduction in body weight and body fat than a diet rich in refined wheat: A parallel randomized controlled trial in adults with overweight and obesity (the RyeWeight study). Clin. Nutr. ESPEN 2021, 45, 155–169. [Google Scholar] [CrossRef]
- Joo, N.; Han, S.; Kim, K.; Kim, B.; Park, S.; Yeum, K. Black rice with giant embryo ameliorates serum C-reactive protein in adults with metabolic syndrome. J. Clin. Biochem. Nutr. 2020, 67, 344–348. [Google Scholar] [CrossRef]
- Katcher, H.I.; Legro, R.S.; Kunselman, A.R.; Gillies, P.J.; Demers, L.M.; Bagshaw, D.M.; Kris-Etherton, P.M. The effects of a whole grain-enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, 79–90. [Google Scholar] [CrossRef]
- Kazemzadeh, M.; Safavi, S.M.; Nematollahi, S.; Nourieh, Z. Effect of brown rice consumption on inflammatory marker and cardiovascular risk factors among overweight and obese non-menopausal female adults. Int. J. Prev. Med. 2014, 5, 478–488. [Google Scholar] [PubMed]
- Kirwan, J.P.; Malin, S.K.; Scelsi, A.R.; Kullman, E.L.; Navaneethan, S.D.; Pagadala, M.R.; Haus, J.M.; Filion, J.; Godin, J.P.; Kochhar, S.; et al. A whole-grain diet reduces cardiovascular risk factors in overweight and obese adults: A randomized controlled trial. J. Nutr. 2016, 146, 2244–2251. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Morino, K.; Nishio, Y.; Ishikado, A.; Arima, H.; Nakao, K.; Nakagawa, F.; Nikami, F.; Sekine, O.; Nemoto, K.I.; et al. Fiber-rich diet with brown rice improves endothelial function in type 2 diabetes mellitus: A randomized controlled trial. PLoS ONE 2017, 12, e0179869. [Google Scholar] [CrossRef]
- Kopf, J.C.; Suhr, M.J.; Clarke, J.; Eyun, S.I.; Riethoven, J.J.M.; Ramer-Tait, A.E.; Rose, D.J. Role of whole grains versus fruits and vegetables in reducing subclinical inflammation and promoting gastrointestinal health in individuals affected by overweight and obesity: A randomized controlled trial. Nutr. J. 2018, 17, 72. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Lietz, G.; Bal, W.; Watson, A.; Morfey, B.; Seal, C. Effects of quinoa (Chenopodium quinoa Willd.) consumption on markers of CVD risk. Nutrients 2018, 10, 777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malik, V.S.; Sudha, V.; Wedick, N.M.; RamyaBai, M.; Vijayalakshmi, P.; Lakshmipriya, N.; Gayathri, R.; Kokila, A.; Jones, C.; Hong, B.; et al. Substituting brown rice for white rice on diabetes risk factors in India: A randomised controlled trial. Br. J. Nutr. 2019, 121, 1389–1397. [Google Scholar] [CrossRef]
- Meng, H.; Matthan, N.R.; Fried, S.K.; Berciano, S.; Walker, M.E.; Galluccio, J.M.; Lichtenstein, A.H. Effect of Dietary Carbohydrate Type on Serum Cardiometabolic Risk Indicators and Adipose Tissue Inflammatory Markers. J Clin. Endocrinol. Metab. 2018, 103, 3430–3438. [Google Scholar] [CrossRef] [PubMed]
- Munch Roager, H.; Vogt, J.K.; Kristensen, M.; Hansen, L.B.S.; Ibrugger, S.; Maerkedahl, R.B.; Bahl, M.I.; Lind, M.V.; Nielsen, R.L.; Frokiaer, H.; et al. Whole grain-rich diet reduces body weight and systemic low-grade inflammation without inducing major changes of the gut microbiome: A randomised cross-over trial. Gut 2019, 68, 83–93. [Google Scholar] [CrossRef] [Green Version]
- Navarro, S.L.; Tarkhan, A.; Shojaie, A.; Randolph, T.W.; Gu, H.; Djukovic, D.; Osterbauer, K.J.; Hullar, M.A.; Kratz, M.; Neuhouser, M.L.; et al. Plasma metabolomics profiles suggest beneficial effects of a low-glycemic load dietary pattern on inflammation and energy metabolism. Am. J. Clin. Nutr. 2019, 110, 984–992. [Google Scholar] [CrossRef]
- Pavadhgul, P.; Bumrungpert, A.; Harjani, Y.; Kurilich, A. Oat porridge consumption alleviates markers of inflammation and oxidative stress in hypercholesterolemic adults. Asia Pac. J. Clin. Nutr. 2019, 28, 260–265. [Google Scholar]
- Pavithran, N.; Kumar, H.; Menon, A.S.; Pillai, G.K.; Sundaram, K.R.; Ojo, O. South Indian cuisine with low glycemic index ingredients reduces cardiovascular risk factors in subjects with type 2 diabetes. Int. J. Environ. Res. Public Health 2020, 17, 6232. [Google Scholar] [CrossRef]
- Pourshahidi, L.K.; Caballero, E.; Osses, A.; Hyland, B.W.; Ternan, N.G.; Gill, C.I.R. Modest improvement in CVD risk markers in older adults following quinoa (Chenopodium quinoa Willd.) consumption: A randomized-controlled crossover study with a novel food product. Eur. J. Nutr. 2020, 59, 3313–3323. [Google Scholar] [CrossRef]
- Saglam, D.; Saka, M.; Sayaca, N. The effect of consumption of low glycemic index, high fat content bread on anthropometric measurement and cardiometabolic risk factors in women with type 2 diabetes mellitus. Int. J. Diabetes Dev. Ctries. 2019, 39, 166–172. [Google Scholar] [CrossRef]
- Schutte, S.; Esser, D.; Hoevenaars, F.P.M.; Hooiveld, G.J.E.J.; Priebe, M.G.; Vonk, R.J.; Wopereis, S.; Afman, L.A. A 12-wk whole-grain wheat intervention protects against hepatic fat: The Graandioos study, a randomized trial in overweight subjects. Am. J. Clin. Nutr. 2018, 108, 1264–1274. [Google Scholar] [CrossRef] [Green Version]
- Tighe, P.; Duthie, G.; Vaughan, N.; Brittenden, J.; Simpson, W.G.; Duthie, S.; Mutch, W.; Wahle, K.; Horgan, G.; Thies, F. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 92, 733–740. [Google Scholar] [CrossRef]
- Vetrani, C.; Costabile, G.; Luongo, D.; Naviglio, D.; Rivellese, A.A.; Riccardi, G.; Giacco, R. Effects of whole-grain cereal foods on plasma short chain fatty acid concentrations in individuals with the metabolic syndrome. Nutrition 2016, 32, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Mennella, I.; Ferracane, R.; Rivellese, A.A.; Giacco, R.; Ercolini, D.; Gibbons, S.M.; La Storia, A.; Gilbert, J.A.; Jonnalagadda, S.; et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: Role of polyphenols bound to cereal dietary fiber. Am. J. Clin. Nutr. 2015, 101, 251–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whittaker, A.; Sofi, F.; Luisi, M.L.; Rafanelli, E.; Fiorillo, C.; Becatti, M.; Abbate, R.; Casini, A.; Gensini, G.F.; Benedettelli, S. An organic khorasan wheat-based replacement diet improves risk profile of patients with acute coronary syndrome: A randomized crossover trial. Nutrients 2015, 7, 3401–3415. [Google Scholar] [CrossRef] [Green Version]
- Whittaker, A.; Dinu, M.; Cesari, F.; Gori, A.M.; Fiorillo, C.; Becatti, M.; Casini, A.; Marcucci, R.; Benedettelli, S.; Sofi, F. A khorasan wheat-based replacement diet improves risk profile of patients with type 2 diabetes mellitus (T2DM): A randomized crossover trial. Eur. J. Nutr. 2017, 56, 1191–1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zamaratskaia, G.; Mhd Omar, N.A.; Brunius, C.; Hallmans, G.; Johansson, J.E.; Andersson, S.O.; Larsson, A.; Aman, P.; Landberg, R. Consumption of whole grain/bran rye instead of refined wheat decrease concentrations of TNF-R2, e-selectin, and endostatin in an exploratory study in men with prostate cancer. Clin. Nutr. 2020, 39, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Koh-Banerjee, P.; Hu, F.B.; Franz, M.; Sampson, L.; Grønbæk, M.; Rimm, E.B. Intakes of whole grains, bran, and germ and the risk of coronary heart disease in men. Am. J. Clin. Nutr. 2004, 80, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.; Beck, E.; Hughes, J.; Grafenauer, S. Whole grains and consumer understanding: Investigating consumers’ identification, knowledge and attitudes to whole grains. Nutrients 2020, 12, 2170. [Google Scholar] [CrossRef] [PubMed]
- Landberg, R.; Kamal-Eldin, A.; Andersson, A.; Vessby, B.; Åman, P. Alkylresorcinols as biomarkers of whole-grain wheat and rye intake: Plasma concentration and intake estimated from dietary records. Am. J. Clin. Nutr. 2008, 87, 832–838. [Google Scholar] [CrossRef] [Green Version]
- Ross, A.B.; Kristensen, M.; Seal, C.J.; Jacques, P.; McKeown, N.M. Recommendations for reporting whole-grain intake in observational and intervention studies. Am. J. Clin. Nutr. 2015, 101, 903–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galland, L. Diet and Inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef]
- Hu, Y.; Ding, M.; Sampson, L.; Willett, W.C.; Manson, J.E.; Wang, M.; Rosner, B.; Hu, F.B.; Sun, Q. Intake of whole grain foods and risk of type 2 diabetes: Results from three prospective cohort studies. BMJ 2020, 370, m2206. [Google Scholar] [CrossRef] [PubMed]
- Wyczalkowska-Tomasik, A.; Czarkowska-Paczek, B.; Zielenkiewicz, M.; Paczek, L. Inflammatory markers change with age, but do not fall beyond reported normal ranges. Arch. Immunol. Ther. Exp. 2016, 64, 249–254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdullah, M.M.H.; Hughes, J.; Grafenauer, S. Whole grain intakes are associated with healthcare cost savings following reductions in risk of colorectal cancer and total cancer mortality in Australia: A cost-of-illness model. Nutrients 2021, 13, 2982. [Google Scholar] [CrossRef]
- Abdullah, M.M.H.; Hughes, J.; Grafenauer, S. Healthcare cost savings associated with increased whole grain consumption among Australian adults. Nutrients 2021, 13, 1855. [Google Scholar] [CrossRef]
| Study | Design and Duration | N (I/C) | Characteristics | (M/F) | Age (Years) | Intervention Diet | Control Diet |
|---|---|---|---|---|---|---|---|
| Ampatzoglou et al. 2016 [24] | Cr 6 weeks | 33 (33/33) | Healthy | (12/21) | 48.8 ± 1.1 | WG > 80 g/day | RG diet; <16 g/day WG |
| Andersson et al. 2007 [25] | Cr 6 weeks | 30 (30/30) | Overweight | (8/22) | 59 ± 5 | Various WGs = 112 g/day | Various RGs-111 g/day |
| Brownlee et al. 2010 [22] | P 16 weeks | 266 (85/81/100) | Overweight | (133/133) | G1: 45.9 ± 10.1; G2: 45.7 ± 9.9; G3: 45.6 ± 1.0 | G1: WG 60g/day; G2: 60 g/day 8 weeks + 120 g/day 8 weeks | Same diet as prior WG < 30 g/day |
| Connolly et al. 2011 [26] | Cr 16 weeks | 32 (16/16) | Glucose intolerant or mild to moderate hypercholesterolamic | (12/20) | 23–64 | WG: 45 g WG/day as breakfast cereal | RG: 45 g/day as breakfast cereal |
| Giacco et al. 2013 [27] | P 12 weeks | 123 (61/62) | Metabolic syndrome | N/A | 40–65 | WG or WW foods to replace RG | RG foods only for breads, pastas, cereals |
| Harris Jackson et al. 2014 [28] | P 12 weeks | 50 (25/25) | Metabolic syndrome | (25/25) | 35–45 | 187 g WG/day | RG, WG = 0 g/day |
| Hoevenaars et al. 2019 [29] | P 12 weeks | 50 (25/25) | Overweight and obese | (19/31) | 45–70 | 98 g WG/day | 98 g RG/day |
| Iversen et al. 2021 [30] | P 12 weeks | 242 (121/121) | Overweight and obese | (95/147) | 30–70 | Rye 53–60 g/day | Wheat 66 g/day |
| Joo et al. 2020 [31] | P 12 weeks | 49 (26/23) | Metabolic syndrome | (38/11) | 44.3 ± 6.1 | Black rice powder 60 g/day | White rice powder 60 g/day |
| Katcher et al. 2008 [32] | P 12 weeks | 50 (25/25) | Obese with metabolic syndrome | (25/25) | WG 45.4 ± 8; RG 46.6 ± 9.7 | WG: 5, 6, 7 serves on hypocaloric diet | No WG foods in hypocaloric diet |
| Kazemzadeh et al. 2014 [33] | Cr 14 weeks | 35 (20/15) | Overweight and obese | (0/35) | 32.6 ± 6 | Brown rice 150 g/day | White rice 150 g/day |
| Kirwan et al. 2016 [34] | Cr 8 weeks | 33 (33/33) | Overweight and obese | (6/27) | 39 ± 7 | WG 93 ± 19 g/day | RG, WG = 0 g |
| Kondo et al. 2017 [35] | P 8 weeks | 28 (14/14) | Type 2 Diabetes | (18/10) | 40–80 | Brown rice (250 cal = 182 g) to replace 10/21 meals/week | White rice (250 cal = 153 g) to replace 10/21 meals/week |
| Kopf et al. 2018 [36] | P 6 weeks | 31 (17/14) | Overweight and obese | N/A | WG:39.2 ± 13.5 RG:27.6 ± 5.9 | Whole grains 3.4 ± 0.2 serves/day | Refined grains 7.1 ± 0.7 serves/day |
| Li et al. 2018 [37] | Cr 8 weeks | 30 (15/15) | Overweight and obese | (30/0) | 36–70 | 20 g quinoa flour/day in form of 160 g bread roll | 20 g refined flour/day in form of 160 g bread roll |
| Ma et al. 2013 [23] | P 30 days | 199 (65/71/63) | Type 2 Diabetes & Metabolic Syndrome | (84/115) | 20–65 | WG1: 50 g oat germ/day | Usual diet |
| WG2: 100 g oat germ/day | Usual diet | ||||||
| Malik et al. 2019 [38] | Cr 14 weeks | 113 (55/58) | Overweight BMI > 23 | (62/51) | 25–65 | Brown rice 182 g/day | White rice 175 g/day |
| Meng et al. 2018 [39] | Cr 13 weeks | 11 | Overweight and obese | (4/7) | 50–80 | Unrefined carbohydrate 19.5 g fiber/day | Refined carbohydrate 9.6 g fiber/day |
| Munch Roager et al. 2019 [40] | Cr 16 weeks | 50 (25/25) | Overweight and obese | (18/32) | 20–65 | WG 157.9 ± 35 g/day | RG diet; WG 6 ± 4.8 g/day |
| Navarro et al. 2018 [41] | Cr 4 weeks | 80 | Healthy | (40/40) | 18–45 | Whole grain foods 55 g fiber/day | Refined grain foods 28 g fiber/day |
| Pavadhgul et al. 2019 [42] | Cr 8 weeks | 24 | Hypercholesterolamic | (12/12) | 30–60 | Whole grain oat porridge 70 g/day | Rice porridge 70 g/day |
| Pavithran et al. 2020 [43] | P 24 weeks | 80 (40/40) | Type 2 diabetes | (52/28) | LGI: 54.43 ± 7.57 Control: 51.93 ± 7.43 | LGI: whole wheat, red rice | Usual diet |
| Pourshahidi et al. 2020 [44] | Cr 12 weeks | 40 | Overweight and obese | (12/28) | 57.68 ± 6.15 | 15g quinoa biscuits (60 g flour/100 g) | Control iso-energetic biscuits |
| Saglam et al. 2018 [45] | P 4 weeks | 24 (12/12) | Type 2 Diabetes | (0/24) | 40.29 ± 6.81 | Whole grain bread 270 cal/ 35.32 g fiber/day | Whole wheat bread 227 cal/7.39 g fiber/day |
| Schutte et al. 2018 [46] | P 12 weeks | 50 (25/25) | Overweight | (31/19) | WG: 61 [51–70] RG: 61 [4–69] | WG 98 g/day | RG 98 g/day |
| Tighe et al. 2010 [47] | P 12 weeks | 136 (73/63) | Overweight | (68/68) | WG1: 51.6 ± 0.8; RG: 51.8 ± 0.8 | WG1: 3 servings (70-8 0g WG bread + 30-40 g WG cereal) | Refined cereals and white bread |
| WG2: 52.1 ± 0.9; RG: 51.8 ± 0.8 | WG2: 1 serve of whole wheat foods + 2 serving of oats | Refined cereals and white bread | |||||
| Vetrani et al. 2016 [48] | P 12 weeks | 40 (21/19) | Metabolic syndrome | (16/24) | WG 57.2 ± 1.9; RG 58.4 ± 1.6 | WG products plus a small portion of endosperm rye bread 40.2 ± 1.2 g fiber/day | Commercial refined grain cereal products 22.1 ± 0.9g fiber/day |
| Vitaglione et al. 2015 [49] | P 8 weeks | 68 (36/32) | Overweight and obese | (0/68) | WG 40 ± 2; RG 37 ± 2 | 100% WG, 70 g/day | RG products, 60 g/day |
| Whittaker et al. 2015 [50] | Cr 24 weeks | 22 | Acute Coronary Syndrome | (13/9) | 61 (47-75) | Khosoran Semolina 62 g/day Khosoran flour 140 g/day | Control Semolina 62 g/day Control Flour 140 g/day |
| Whittaker et al. 2017 [51] | Cr 24 weeks | 21 | Type 2 Diabetes | (7/14) | 64.4 ± 10.9 w | Khosoran Semolina 62 g/day Khosoran flour 140 g/day | Control Semolina 62 g/day Control Flour 140 g/day |
| Zamaratskaia et al. 2020 [52] | Cr 24 weeks | 17 | Prostate cancer | (17/0) | 73.5 ± 4.6 | WG foods 485 g/day | RG foods 485 g/day |
| Study | N (I/C) | CRP Baseline | CRP Endpoint | p-Value |
| Ampatzoglou et al. 2016 [24] | I (n = 33) | 2.2 (0.5) ng/L | 1.6 (0.4) ng/L | 0.099 |
| C (n = 33) | 1.7 (0.3) ng/L | 1.8 (0.3) ng/L | ||
| Navarro et al. 2019 [41] | I (n = 40) | 1.5 ± 2.7 mg/L | n.d | 0.19 |
| C (n = 40) | 1.5 ± 2.7 mg/L | n.d | ||
| Study | N (I/C) | IL-6 Baseline | IL-6 Endpoint | p-Value |
| Ampatzoglou et al. 2016 [24] | I (n = 33) | 1.2 (0.2) ng/L | 1.6 (0.1) ng/L | 0.702 |
| C (n = 33) | 1.3 (0.2) ng/L | 1.4 (0.2) ng/L | ||
| Study | N (I/C) | TNF Baseline | TNF Endpoint | p-Value |
| Ampatzoglou et al. 2016 [24] | I (n = 33) | 10.8 (0.4) ng/L | 10.8 (0.6) ng/L | 0.381 |
| C (n = 33) | 10.5 (0.5) ng/L | 10.7 (0.5) ng/L |
| Study | N (I/C) | CRP Baseline | CRP Endpoint | p-Value |
| Andersson et al. 2007 [25] | I (n = 30) | 2.03 ± 1.62 mg/L | 2.38 ± 2.29 mg/L | 0.55 |
| C (n = 30) | 2.86 ± 2.96 mg/L | 2.34 ± 1.57 mg/L | ||
| Brownlee et al. 2010 [22] | I1 (n = 85) | 2.4 ± 9.9 mg/L | 3.1 ± 4.3 mg/L | >0.05 |
| C (n = 100) | 2.4 ± 2.3 mg/L | 2.9 ± 3.5 mg/L | ||
| Brownlee et al. 2010 [22] | I2 (n = 81) | 3.2 ± 4.6 mg/L | 3.2 ± 5.9 mg/L | >0.05 |
| C (n = 100) | 2.4 ± 2.3 mg/L | 2.9 ± 3.5 mg/L | ||
| Hoevenaars et al. 2019 [29] | I (n = 20) | 5.29 ± 8.14 μg/mL | 2.16 ± 1.82 μg/mL | 0.03 ** |
| C (n = 20) | 2.58 ± 2.70 μg/mL | 5.24 ± 14.1 μg/mL | ||
| Iversen et al. 2021 [30] | I (n = 121) | 1.45 (1.21; 1.73) mg/L | 1.12 (0.93; 1.36) mg/L | 0.001 ** |
| C (n = 121) | 1.44 (1.19; 1.74) mg/L | 1.58 (1.29; 1.92) mg/L | ||
| Katcher et al. 2008 [32] | I (n = 121) | 1.45 (1.21; 1.73) | 1.12 (0.93; 1.36) mg/L | 0.001 ** |
| C (n = 121) | 1.44 (1.19; 1.74) | 1.58 (1.29; 1.92) mg/L | ||
| Kazemzadeh et al. 2014 [33] | I (n = 20) | G1: 2.0 ± 1.3 mg/L G2: 1.5 ± 1.2 mg/L | G1: 1.9 ± 1.9 mg/L G2: 0.9 ± 1.1 mg/L | 0.012 ** |
| C (n = 15) | G1: 2.0 ± 1.3 mg/L G2: 1.5 ± 1.2 mg/L | G1: 1.9 ± 1.9 mg/L G2: 0.9 ± 1.1 mg/L | ||
| Kirwan et al. 2016 [34] | I (n = 33) | 3.7 ± 3.3 mg/L | 0.8 (−1.1, 2.6) mg/L | 0.06 |
| C (n = 33) | 5.9 ± 7.1 mg/L | −2.3 (−4.8, 0.1) mg/L | ||
| Kopf et al. 2018 [36] | I (n = 17) | 0.8 ± 0.6 mg/mL | 0.8 ± 0.4 mg/mL | 0.89 |
| C (n = 14) | 0.6 ± 0.4 mg/mL | 0.7 ± 0.5 mg/mL | ||
| Li et al. 2018 [37] | I (n = 28) | 3.7 ± 3.3 mg/L | 3.7 ± 3.3 mg/L | 0.197 |
| C (n = 28) | 3.7 ± 3.3 mg/L | 3.7 ± 3.3 mg/L | ||
| Malik et al. 2019 [38] | I (n = 55) | 4.1 ± 2.8 mg/L | 0.03 ± 2.12 mg/L | 0.04 ** |
| C (n = 58) | 4.1 ± 2.8 mg/L | 0.63 ± 2.35 mg/L | ||
| Meng et al. 2019 [39] | I (n = 11) | n.d | 2.1 (0.7–4.7) mg/L | 0.84 |
| C (n = 11) | n.d | 2.0 (0.6–4.6) mg/L | ||
| Munch Roager et al. 2019 [40] | I (n = 25) | 6.3 ± 14.0 mg/L | 4.2 ± 6.8 mg/L | 0.003 ** |
| C (n = 25) | 3.1 ± 2.6 mg/L | 5.0 ± 5.8 mg/L | ||
| Pourshahidi et al. 2020 [44] | I (n = 20) | 156 ± 195 μg/dL | 142 ± 115 μg/dL | 0.265 |
| C (n = 20) | 156 ± 195 μg/dL | 171 ± 254 μg/dL | ||
| Schutte et al. 2018 [46] | I (n = 25) | 5294 ± 8140 ng/mL | 2162 ± 7260 ng/mL | 0.064 |
| C (n = 25) | 2575 ± 2702 ng/mL | 2555 ± 1658 ng/mL | ||
| Tighe et al. 2010 [47] | I1 (n = 85) | 3.3 (0.5, 2.3) mg/L | 0.9 (0.5, 1.9) mg/L | 0.349 |
| C (n = 100) | 1.4 (0.7, 2.7) mg/L | 1.1 (0.6, 3.0) mg/L | ||
| Tighe et al. 2010 [47] | I2 (n = 81) | 1.0 (0.4, 1.6) mg/L | 1.0 (0.6, 2.3) mg/L | 0.349 |
| C (n = 100) | 1.4 (0.7, 2.7) mg/L | 1.1 (0.6, 3.0) mg/L | ||
| Study | N (I/C) | IL-6 Baseline | IL-6 Endpoint | p-Value |
| Andersson et al. 2007 | I (n = 30) | 14.8 ± 32.2 mg/L | 15.2 ± 33.2 mg/L | 0.79 |
| [25] | C (n = 30) | 15.9 ± 32.4 mg/L | 15.8 ± 30.9 mg/L | |
| Hoevenaars et al. 2019 | I (n = 20) | 1.17 ± 1.26 pg/mL | 1.13 ± 0.89 pg/mL | 0.73 |
| [29] | C (n = 20) | 1.09 ± 0.81 pg/mL | 1.46 ± 1.58 pg/mL | |
| Katcher et al. 2008 [32] | I (n = 121) | 3.2 ± 6.3 pg/mL^6 | 2.3 ± 3.6 pg/mL^6 | Group 0.94 ^ |
| C (n = 121) | 2.2 ± 1.3 pg/mL^6 | 2.1 ± 0.4 pg/mL^6 | Time 0.57 | |
| Kopf et al. 2018 [36] | I (n = 17) | 4.4 ± 1.9 mg/mL | 5.2 ± 1.3 mg/mL | 0.89 |
| C (n = 14) | 2.9 ± 1.5 mg/mL | 3.2 ± 1.7 mg/mL | ||
| Meng et al. 2019 [39] | I (n = 11) | n.d | 0.6 (0.4–0.8) pg/L | 0.77 |
| C (n = 11) | n.d | 0.6 (0.4–0.8) pg/L | ||
| Munch Roager et al. 2019 [40] | I (n = 20) | 1.6 ± 1.2 mg/L | 1.4 ± 1.1 mg/L | 0.009 ** |
| C (n = 15) | 1.2 ± 0.7 mg/L | 2.0 ± 2.0 mg/L | ||
| Tighe et al. 2010 [47] | I1 (n = 85) | 1.3 (0.8, 2.3) pg/L | 1.4 (1.0, 2.4) pg/L | >0.05 |
| C (n = 100) | 1.1 (0.8, 1.7) pg/L | 1.1 (0.8, 1.6) pg/L | ||
| Tighe et al. 2010 [47] | I2 (n = 81) | 1.2 (0.9, 1.9) pg/L | 0.9 (0.5, 1.9) pg/L | >0.05 |
| C (n = 100) | 1.1 (0.8, 1.7) pg/L | 1.1 (0.8, 1.6) pg/L | ||
| Vitaglione et al. 2015 [49] | I (n = 36) | 57.5 ± 7.5 pg/mL | 46.9 ± 4.0 pg/mL | 0.06 |
| C (n = 32) | 65.5 ± 11.4 pg/mL | 60.2 ± 7.2 pg/mL | ||
| Study | N (I/C) | TNF Baseline | TNF Endpoint | p-Value |
| Hoevenaars et al. 2019 | I (n = 20) | 3.07 ± 1.85 pg/mL | 2.90 ± 1.89 pg/mL | 0.26 |
| [29] | C (n = 20) | 2.26 ± 1.43 pg/mL | 2.29 ± 1.38 pg/mL | |
| Katcher et al. 2008 [32] | I (n = 121) | 1.2 ± 0.3 pg/mL^6 | 1.1 ± 0.3 pg/mL^6 | Group 0.04 **^ |
| C (n = 121) | 1.3 ± 0.4 pg/mL^6 | 1.2 ± 0.2 pg/mL^6 | Time 0.80 | |
| Kopf et al. 2018 [36] | I (n = 17) | 26.7 ± 4.17 pg/mL | 21.4 ± 2.9 pg/mL | 0.11 |
| C (n = 14) | 23.8 ± 5.9 pg/mL | 23.4 ± 6.6 pg/mL | ||
| Munch Roager et al. 2019 [40] | I (n = 20) | 1.7 ± 0.8 pg/mL | 1.7 ± 0.08 pg/mL | 0.87 |
| C (n = 15) | 1.7 ± 0.9 pg/mL | 1.7 ± 0.9 pg/mL | ||
| Vitaglione et al. 2015 [49] | I (n = 36) | 341.9 ± 25.5 pg/mL | 26.8 ± 3.2 pg/mL | 0.04 ** |
| C (n = 32) | 321.9 ± 52.1 pg/mL | 329.8 ± 5.06 pg/mL |
| Study | N (I/C) | CRP Baseline | CRP Endpoint | p-Value |
| Connolly et al. 2011 [26] | I (n = 16) | 1.69 ± 0.35 mg/L | 2.45 ± 0.92mg/L | 0.934 |
| C (n = 16) | 1.8 ± 0.47 mg/L | 2.36 ± 0.49 mg/L | ||
| Giacco et al. 2013 [27] | I (n = 61) | 1.95 (0.74; 4.12) mg/dl | 1.36 (0.62; 3.34) mg/dl | 0.16 |
| C (n = 62) | 1.95 (0.96; 2.56) mg/dl | 1.74 (1.04; 2.95) mg/dl | ||
| Harris Jackson et al. 2014 [28] | I (n = 17) | 3.0 (2.0, 4.6) mg/L | 2.4 ± 0.5 mg/L | >0.05 |
| C (n = 25) | 2.1 (1.4, 3.1) mg/L | 1.5 ± 0.4 mg/L | ||
| Joo et al. 2020 [31] | I (n = 26) | 0.205 (0.183) mg/dL | 0.101 (0.028) mg/dL | 0.03 ** |
| C (n = 23) | 0.137 (0.165) mg/dL | 0.154 (0.025) mg/dL | ||
| Kondo et al. 2017 [35] | I (n = 14) | 0.09 ± 0.12 μg/L | 0.05 ± 0.05 μg/L | 0.063 |
| C (n = 14) | 0.04 ± 0.03 μg/L | 0.05 ± 0.06 μg/L | ||
| Ma et al. 2013 [23] | I1 (n = 65) | 3.65 (2.45) mg/L | 3.13 (2.61) mg/L | >0.05 |
| C (n = 63) | 3.76 (1.99) mg/L | 3.81 (2.21) mg/L | ||
| Ma et al. 2013 [23] | I2 (n = 71) | 3.46 (2.55) mg/L | 2.26 (2.12) mg/L | <0.05 ** |
| C (n = 63) | 3.76 (1.99) mg/L | 3.81 (2.21) mg/L | ||
| Pavadhgul et al. 2019 [42] | I (n = 24) | 2.7 ± 2.1 mg/L | 2.2 ± 2.1 mg/L | <0.05 ** |
| C (n = 24) | 2.7 ± 2.1 mg/L | 2.9 ± 2.9 mg/L | ||
| Pavithran et al. 2020 [43] | I (n = 40) | 3.38 ± 3.83 mg/L | 1.46 ± 1.04 mg/L | 0.026 ** |
| C (n = 40) | 2.79 ± 4.20 mg/L | 3.16 ± 4.61 mg/L | ||
| Saglam et al. 2019 [45] | I (n = 12) | n.d | n.d | >0.05 |
| C (n = 12) | n.d | n.d | ||
| Vetrani et al. 2016 [48] | I (n = 21) | 2.52 ± 0.5 mg/dL | 2.44 ± 0.5 mg/dL | 0.693 |
| C (n = 19) | 2.27 ± 0.4 mg/dL | 2.39 ± 0.4 mg/dL | ||
| Study | N (I/C) | IL-6 Baseline | IL-6 Endpoint | p-Value |
| Connolly et al. 2011 [26] | I (n = 16) | 4.13 ± 1.47 pg/mL | 5.88 ± 1.78 pg/mL | 0.925 |
| C (n = 16) | 4.09 ± 1.71 pg/mL | 7.16 ± 3.46 pg/mL | ||
| Giacco et al. 2013 [27] | I (n = 61) | 1.42 (1.01; 2.32) pg/mL | 1.54 (1.12; 2.23) pg/mL | 0.52 |
| C (n = 62) | 1.41 (0.84; 2.21) pg/mL | 1.43 (1.07; 2.11) pg/mL | ||
| Harris Jackson et al. 2014 [28] | I (n = 23) | 1.8 (1.5, 2.2) pg/mL | 2.1 ± 0.2 pg/mL | >0.05 |
| C (n = 23) | 1.7 (1.4, 2.0) pg/mL | 1.8 ± 0.2 pg/mL | ||
| Pavadhgul et al. 2019 [42] | I (n = 24) | 150 ± 57.9 pg/L | 123 ± 44.5 pg/L | <0.01 ** |
| C (n = 24) | 150 ± 57.9 pg/L | 145 ± 54.0 pg/L | ||
| Vetrani et al. 2016 [48] | I (n = 21) | 1.84 ± 0.2 pg/mL | 2.23 ± 0.3 pg/mL | 0.161 |
| C (n = 19) | 1.69 ± 0.3 pg/mL | 1.7 ± 0.3 pg/mL | ||
| Whittaker et al. 2015 [50] | I (n = 22) | 2.26 (1.50–3.03) pg/mL | 1.53 (1.16–1.90) pg/mL | 0.698 |
| C (n = 22) | 3.16 (1.51–4.81) pg/mL | 3.30 (1.24–6.37) pg/mL | ||
| Whittaker et al. 2017 [51] | I (n = 21) | 2.76 ± 2.01 pg/mL | 2.16 ± 1.21 pg/mL | 0.9 |
| C (n = 21) | 2.15 ± 1.57 pg/mL | 1.70 ± 1.24 pg/mL | ||
| Study | N (I/C) | TNF Baseline | TNF Endpoint | p-Value |
| Connolly et al. 2011 [26] | I (n = 16) | 20.2 ± 4.0 pg/mL | 36.5 ± 15.7 pg/mL | 0.519 |
| C (n = 16) | 46.3 ± 26.0 pg/mL | 42.2 ± 14.8 pg/mL | ||
| Giacco et al. 2013 [27] | I (n = 61) | 0.73 (0.50; 0.96) pg/mL | 0.68 (0.50; 0.94) pg/mL | 0.84 |
| C (n = 62) | 0.62 (0.43; 1.05) pg/mL | 0.63 (0.41; 0.90) pg/mL | ||
| Harris Jackson et al. 2014 [28] | I (n = 24) | 1.2 (1.0, 1.3) pg/mL | 1.2 ± 0.1 pg/mL | <0.05 ** |
| C (n = 24) | 1.4 (1.2, 1.7) pg/mL | 1.3 ± 0.1^5 pg/mL | ||
| Pavadhgul et al. 2019 [42] | I (n = 24) | 49.5 ± 26.4 pg/L | 39.83 ± 15.9 pg/L | <0.01 ** |
| C (n = 24) | 49.5 ± 26.4 pg/L | 47.4 ± 24.1 pg/L | ||
| Vetrani et al. 2016 [48] | I (n = 21) | 1.71 ± 0.6 pg/mL | 1.50 ± 0.6 pg/mL | 0.232 |
| C (n = 19) | 1.07 ± 0.4μg/mL | 1.31 ± 0.5 pg/mL | ||
| Whittaker et al. 2015 [50] | I (n = 22) | 4.54 ± 3.32 pg/mL | 3.9 (1.4–6.4) pg/mL | 0.798 |
| C (n = 22) | 6.5 (2.9–9.9) pg/mL | 4.6 (0.9–8.2) pg/mL | ||
| Whittaker et al. 2017 [51] | I (n = 21) | 4.54 ± 3.32 pg/mL | 4.74 ± 3.09 pg/mL | 0.04 ** |
| C (n = 21) | 4.36 ± 4.09 pg/mL | 4.84 ± 4.07 pg/mL |
| Study | N (I/C) | CRP Baseline | CRP Endpoint | p-Value |
| Zamaratskaia et al. 2020 | I (n = 17) | n.d | n.d | >0.05 |
| [52] | C (n = 17) | |||
| Study | N (I/C) | IL-6 Baseline | IL-6 Endpoint | p-Value |
| Zamaratskaia et al. 2020 | I (n = 17) | 6.3 (5.3–7.5) pg/mL | n.d | >0.05 |
| [52] | C (n = 17) | 5.8 (4.8–6.9) pg/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milesi, G.; Rangan, A.; Grafenauer, S. Whole Grain Consumption and Inflammatory Markers: A Systematic Literature Review of Randomized Control Trials. Nutrients 2022, 14, 374. https://doi.org/10.3390/nu14020374
Milesi G, Rangan A, Grafenauer S. Whole Grain Consumption and Inflammatory Markers: A Systematic Literature Review of Randomized Control Trials. Nutrients. 2022; 14(2):374. https://doi.org/10.3390/nu14020374
Chicago/Turabian StyleMilesi, Genevieve, Anna Rangan, and Sara Grafenauer. 2022. "Whole Grain Consumption and Inflammatory Markers: A Systematic Literature Review of Randomized Control Trials" Nutrients 14, no. 2: 374. https://doi.org/10.3390/nu14020374
APA StyleMilesi, G., Rangan, A., & Grafenauer, S. (2022). Whole Grain Consumption and Inflammatory Markers: A Systematic Literature Review of Randomized Control Trials. Nutrients, 14(2), 374. https://doi.org/10.3390/nu14020374






