Associations of Nutritional Behavior and Gut Microbiota with the Risk of COVID-19 in Healthy Young Adults in Poland
Abstract
:1. Introduction
2. Materials and Methods
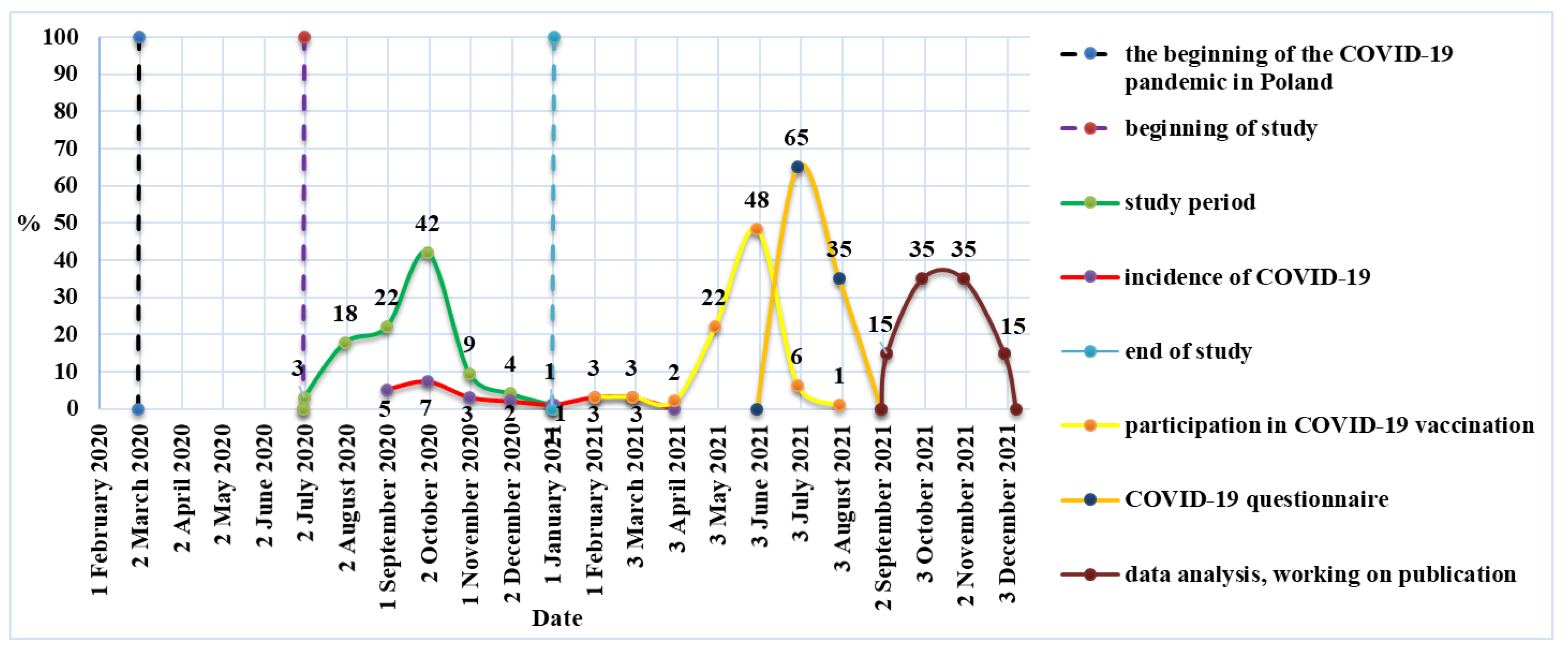
2.1. Study Design and Sample
2.2. Weight, Height, and BMI
2.3. Physical Activity Recording
2.4. Dietary Intake
2.5. Dietary Inflammatory Index (DII)
2.6. Analysis of the Gut Microbiota
2.7. Power Analysis
2.8. Statistical Analysis
3. Results
3.1. Characteristics of Participants
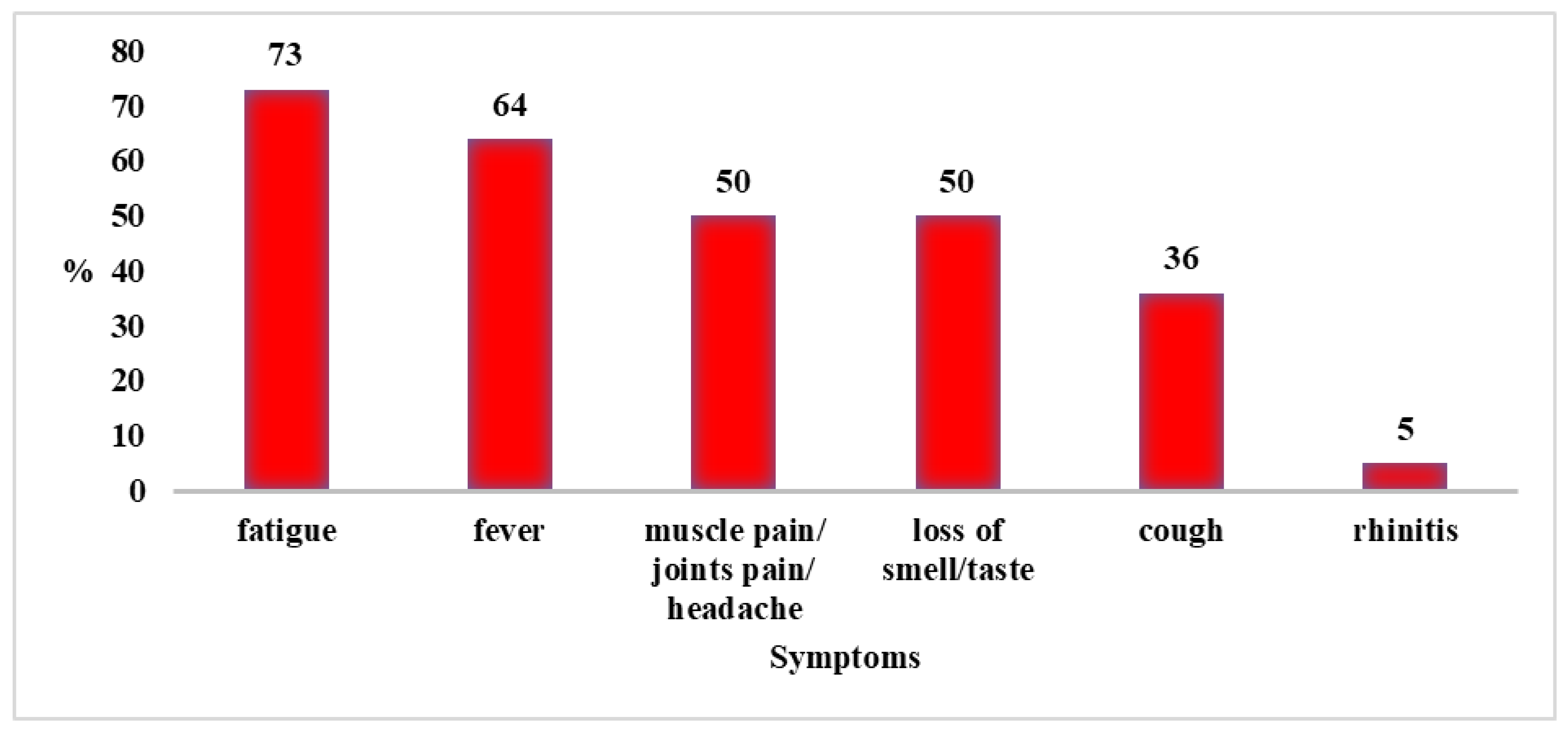
3.2. Findings
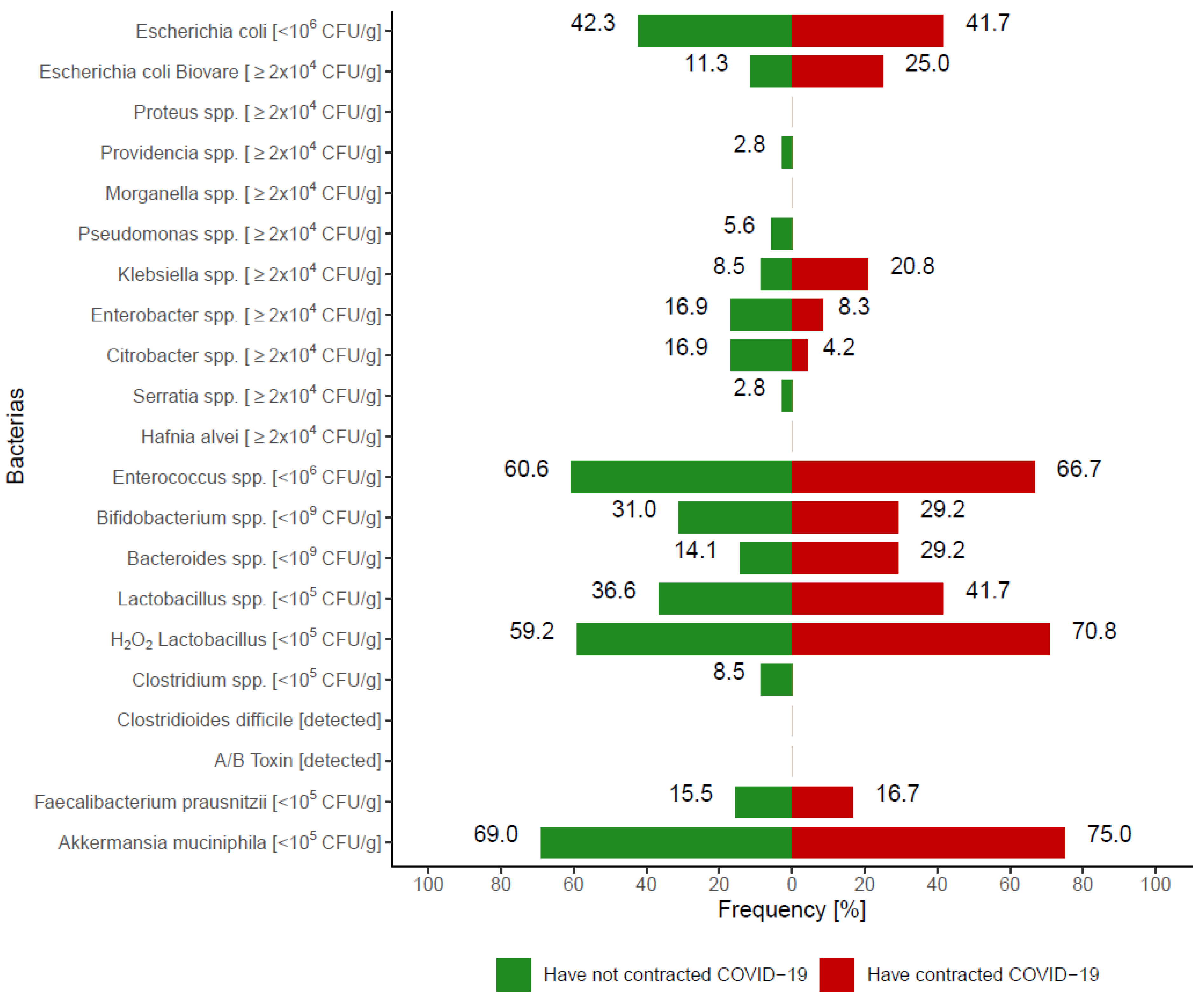
3.2.1. Analysis of the Gut Microbiota
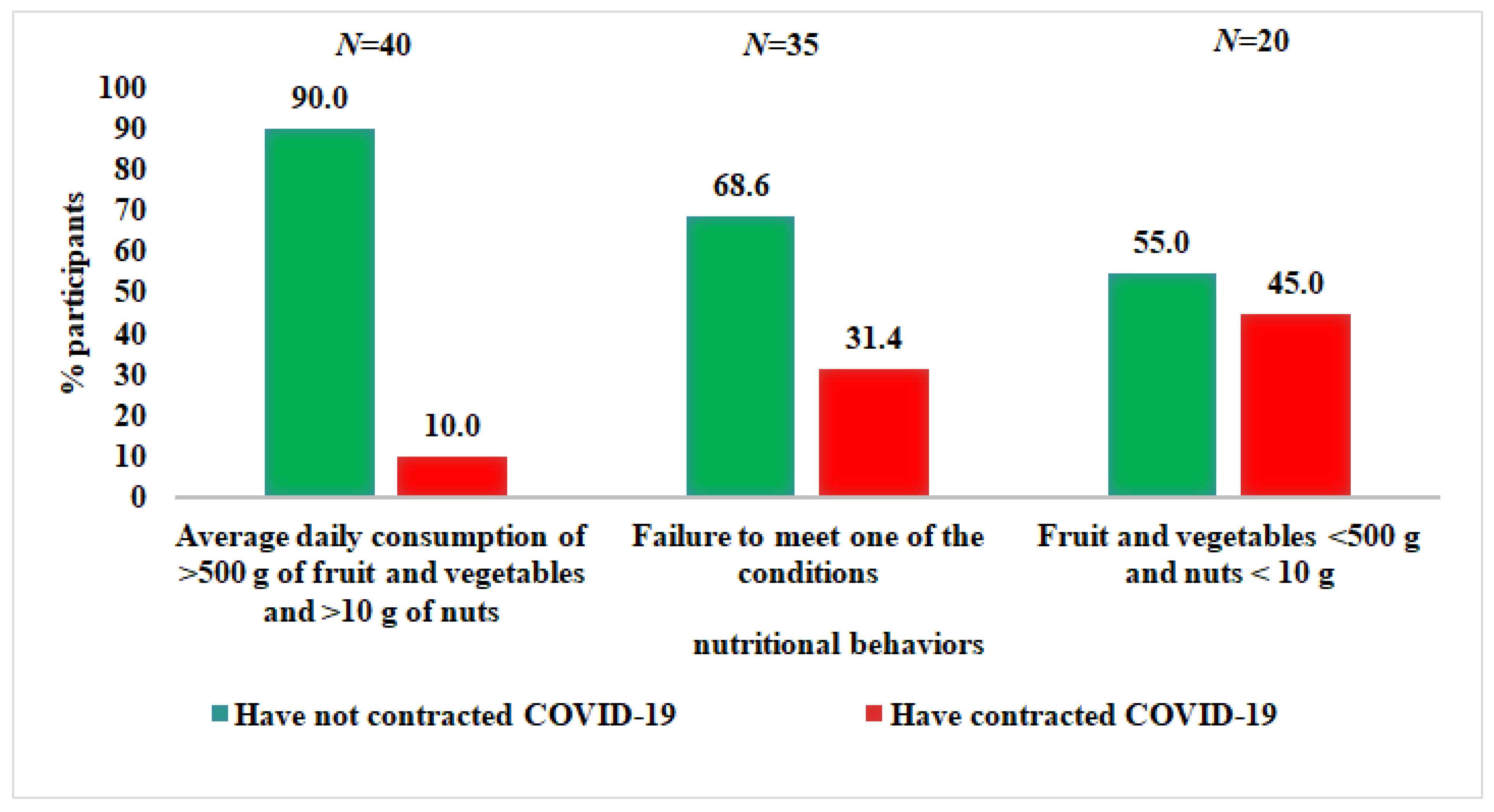
3.2.2. Nutritional Behavior
4. Discussion
4.1. Study Group
4.2. Nutritional Behaviors
4.3. Macronutrients
4.4. Gut Microbiota
4.5. Micronutrients
4.6. Vitamins
4.7. Dietary Intake and Dietary Inflammatory Index
4.8. The Role of Healthy Lifestyle in Prevention of COVID-19
4.9. Healthy Lifestyle Recommendation
4.10. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Cases in the U.S. Website. Available online: https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html (accessed on 12 November 2021).
- Maragakis, L. Coronavirus and COVID-19: Younger Adults Are at Risk, Too. Johns Hopkins Health. 2020. Available online: https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/coronavirus-and-covid-19-younger-adults-are-at-risk-too (accessed on 13 November 2021).
- Severe outcomes among patients with coronavirus disease 2019 (COVID19) d United States, February 12eMarch 16, 2020 website. MMWR Morb. Mortal Wkly. Rep. 2020, 69, 343e6.
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long-COVID. Nat. Med. 2021, 27, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Knight, M.; A’Court, C.; Buxton, M.; Husain, L. Management of post-acute covid-19 in primary care. BMJ 2020, 370, m3026. [Google Scholar] [CrossRef]
- Altonen, B.L.; Arreglado, T.M.; Leroux, O.; Murray-Ramcharan, M.; Engdahl, R. Characteristics, comorbidities and survival analysis of young adults hospitalized with COVID-19 in New York City. PLoS ONE 2020, 15, e0243343. [Google Scholar] [CrossRef]
- Cunningham, J.W.; Vaduganathan, M.; Claggett, B.L.; Jering, K.S.; Bhatt, A.S.; Rosenthal, N.; Solomon, S.D. Clinical Outcomes in Young US Adults Hospitalized With COVID-19. JAMA Intern. Med. 2020, 9, 181, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Li, M.; Zhou, Z.; Guan, X.; Xiang, Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J. Autoimmun. 2020, 111, 102452. [Google Scholar] [CrossRef]
- Lingeswaran, M.; Goyal, T.; Ghosh, R.; Suri, S.; Mitra, P.; Misra, S.; Sharma, P. Inflammation, Immunity and Immunogenetics in COVID-19: A Narrative Review. Indian J. Clin. Biochem. 2020, 35, 260–273. [Google Scholar] [CrossRef]
- Dizdar, O.S.; Baspınar, O.; Kocer, D.; Dursun, Z.B.; Avcı, D.; Karakükcü, C.; Çelik, I.; Gundogan, K. Nutritional Risk, Micronutrient Status and Clinical Outcomes: A Prospective Observational Study in an Infectious Disease Clinic. Nutrients 2016, 8, 124. [Google Scholar] [CrossRef] [Green Version]
- Iddir, M.; Brito, A.; Dingeo, G.; Del Campo, S.S.F.; Samouda, H.; La Frano, M.R.; Bohn, T. Strengthening the Immune System andReducing Inflammation and Oxidative Stress through Diet and Nutrition: Considerations during the COVID-19 Crisis. Nutrients 2020, 12, 1562. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.I.T.D.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM Criteria for the Diagnosis of Malnutrition—A Consensus Report from the Global Clinical Nutrition Community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, Y.; Ye, J.; Chen, M.; Jiang, C.; Lin, W.; Lu, Y.; Ye, H.; Li, Y.; Wang, Y.; Liao, Q.; et al. Malnutrition Prolongs the Hospitalization of Patients with COVID-19 Infection: A Clinical Epidemiological Analysis. J. Nutr. Health Aging 2021, 25, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, Y.; Ge, Y.; Shi, Y.; Lv, P.; Zhang, J.; Fu, G.; Zhou, Y.; Jiang, K.; Lin, N.; et al. Evaluation of Nutrition Risk and Its Association With Mortality Risk in Severely and Critically Ill COVID-19 Patients. J. Parenter. Enter. Nutr. 2021, 45, 32–42. [Google Scholar] [CrossRef]
- Zhang, P.; He, Z.; Yu, G.; Peng, D.; Feng, Y.; Ling, J.; Wang, Y.; Li, S.; Bian, Y. The Modified NUTRIC Score Can Be Used for Nutritional Risk Assessment as Well as Prognosis Prediction in Critically Ill COVID-19 Patients. Clin. Nutr. 2021, 40, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Tinkov, A.; Strand, T.A.; Alehagen, U.; Skalny, A.; Aaseth, J. Early Nutritional Interventions with Zinc, Selenium and Vitamin D for Raising Anti-Viral Resistance against Progressive COVID-19. Nutrients 2020, 12, 2358. [Google Scholar] [CrossRef]
- Im, J.H.; Je, Y.S.; Baek, J.; Chung, M.H.; Kwon, H.Y.; Lee, J.S. Nutritional Status of Patients with COVID-19. Int. J. Infect. Dis. 2020, 100, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.K.Y.; Tao, J.; Chan, O.S.; Li, H.B.; Pang, H. Preventing Respiratory Tract Infections by Synbiotic Interventions: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 979–988. [Google Scholar] [CrossRef]
- Bottari, B.; Castellone, V.; Neviani, E. Probiotics and Covid-19. Int. J. Food Sci. Nutr. 2021, 72, 293–299. [Google Scholar] [CrossRef]
- Kazmierczak-Siedlecka, K.; Roviello, G.; Catalano, M.; Polom, K. Gut Microbiota Modulation in the Context of Immune-Related Aspects of Lactobacillus spp. and Bifidobacterium spp. in Gastrointestinal Cancers. Nutrients 2021, 13, 2674. [Google Scholar] [CrossRef]
- Chattopadhyay, I.; Shankar, E.M. SARS-CoV-2-Indigenous Microbiota Nexus: Does Gut Microbiota Contribute to Inflammation and Disease Severity in COVID-19? Front. Cell. Infect. Microbiol. 2021, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, G.L.V.; Oliveira, C.N.S.; Pinzan, C.F.; de Salis, L.V.V.; Cardoso, C.R.d.B. Microbiota Modulation of the Gut-Lung Axis in COVID-19. Front. Immunol. 2021, 12, 214. [Google Scholar] [CrossRef]
- Louca, P.; Murray, B.; Klaser, K.; Graham, M.S.; Mazidi, M.; Leeming, E.R.; Thompson, E.; Bowyer, R.; Drew, D.A.; Nguyen, L.H.; et al. Modest effects of dietary supplements during the COVID-19 pandemic: Insights from 445 850 users of the COVID-19 Symptom Study app. BMJ Nutr. Prev. Health 2021, 4, 149–157. [Google Scholar] [CrossRef]
- Jarosz, M.; Rychlik, E.; Sto´s, K.; Charzewska, J. (Eds.) Nutrition Standards for the Polish Population and Their Applied; PZH: Warszawa, Poland, 2020. (In Polish) [Google Scholar]
- Shivappa, N.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Hébert, J.R. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 2014, 17, 1689–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janczy, A.; Aleksandrowicz-Wrona, E.; Kochan, Z.; Małgorzewicz, S. Impact of diet and synbiotics on selected gut bacteria and intestinal permeability in individuals with excess body weight—A Prospective, Randomized Study. Acta Biochim. Pol. 2020, 16, 67, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Gałęcka, M.; Basińska, A.; Bartnicka, A. KyberKompaktPro—Modern diagnostics of the gastrointestinal microbiota and its importance in the proper functioning of the human body. Forum Zakażeń. 2017, 8, 111–116. [Google Scholar] [CrossRef]
- W walce z Covid-19 Kluczowa jest nasza Odporność. Available online: https://www.rdn.pl/news/walce-covid-19-kluczowa-nasza-odpornosc (accessed on 22 November 2021).
- Leczmy Alergie, bo Drożny nos to Podstawa Obrony Przed Wirusami. Available online: https://www.medexpress.pl/prof-kuna-leczmy-alergie-bo-drozny-nos-to-podstawa-obrony-przed-wirusami/77395 (accessed on 23 November 2021).
- Koronawirus: Zalecenia Immunologów. Available online: https://informacje.pan.pl/index.php/informacje/materialy-dla-prasy/2909-koronawirus-zalecenia-immunologow?fbclid=IwAR1zCmcbXq7RKpsju4O8NLCRJXa4UVcUXi1Vr8N6LlHobOpHNUjJvAnQz20 (accessed on 22 November 2021).
- Calder, P.C. Nutrition, immunity and COVID-19. BMJ Nutr. Prev. Health 2020, 3, e000085. [Google Scholar] [CrossRef]
- Wądołowska, L.; Drywień, M.; Hamułka, J.; Socha, P.; Borawska, M.; Friedrich, M.; Lange, E.; Members of the Human Nutrition Science Committee of the Polish Academy of Sciences. Dietary recommendations during the COVID-19 pandemic. Statement of the Human Nutrition Science Committee of the Polish Academy of Sciences. Rocz. Panstw. Zakl. Hig. 2021, 72, 209–220. [Google Scholar] [CrossRef]
- Wybory Żywieniowe Niekoniecznie na Zdrowie. Available online: https://www.mp.pl/pacjent/dieta/aktualnosci/285635,wybory-zywieniowe-niekoniecznie-na-zdrowie (accessed on 3 January 2022).
- Health Status of Population in Poland in 2019. Available online: https://stat.gov.pl/files/gfx/portalinformacyjny/pl/defaultaktualnosci/5513/6/7/1/stan_zdrowia_ludnosci_polski_w_2019.pdf (accessed on 30 December 2021).
- Komunikat z Badan. Palimy Papierosy. Available online: https://www.cbos.pl/SPISKOM.POL/2019/K_104_19.PDF (accessed on 3 January 2022).
- Siemieniuk, R.A.; Bartoszko, J.J.; Ge, L.; Zeraatkar, D.; Izcovich, A.; Kum, E.; Pardo-Hernandez, H.; Qasim, A.; Martinez, J.P.D.; Rochwerg, B.; et al. Drug Treatments for Covid-19: Living Systematic Review and Network Meta-Analysis. BMJ 2020, 370, m2980. [Google Scholar] [CrossRef]
- Akhtar, S.; Das, J.K.; Ismail, T.; Wahid, M.; Saeed, W.; Bhutta, Z.A. Nutritional Perspectives for the Prevention and Mitigation of COVID-19. Nutr. Rev. 2021, 79, 289–300. [Google Scholar] [CrossRef]
- National Survey COVID-19 Seroepidemiological Survey: OBSER-CO Report from the First Round of the Survey. Available online: https://www.pzh.gov.pl/projekty-i-programy/obserco/raporty/ (accessed on 22 November 2021).
- Li, P.; Yin, Y.-L.; Li, D.; Kim, S.W.; Wu, G. Amino Acids and Immune Function. Br. J. Nutr. 2007, 98, 237–252. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, L.; Cervantes, E.; Ortiz, R. Malnutrition and Gastrointestinal and Respiratory Infections in Children: A Public Health Problem. Int. J. Environ. Res. Public. Health 2011, 8, 1174–1205. [Google Scholar] [CrossRef] [Green Version]
- Jakulj, F.; Zernicke, K.; Bacon, S.L.; van Wielingen, L.E.; Key, B.L.; West, S.G.; Campbell, T.S. A High-Fat Meal Increases Cardiovascular Reactivity to Psychological Stress in Healthy Young Adults. J. Nutr. 2007, 137, 935–939. [Google Scholar] [CrossRef] [Green Version]
- O’Keefe, J.H.; Bell, D.S.H. Postprandial Hyperglycemia/Hyperlipidemia (Postprandial Dysmetabolism) Is a Cardiovascular Risk Factor. Am. J. Cardiol. 2007, 100, 899–904. [Google Scholar] [CrossRef]
- Hruby, A.; Jacques, P.F. Dietary Protein and Changes in Biomarkers of Inflammation and Oxidative Stress in the Framingham Heart Study Offspring Cohort. Curr. Dev. Nutr. 2019, 3, nzz019. [Google Scholar] [CrossRef] [Green Version]
- Rees, C.A.; Rostad, C.A.; Mantus, G.; Anderson, E.J.; Chahroudi, A.; Jaggi, P.; Wrammert, J.; Ochoa, J.B.; Ochoa, A.; Basu, R.K.; et al. Altered Amino Acid Profile in Patients with SARS-CoV-2 Infection. Proc. Natl. Acad. Sci. USA 2021, 118, e2101708118. [Google Scholar] [CrossRef]
- Gambardella, J.; Khondkar, W.; Morelli, M.B.; Wang, X.; Santulli, G.; Trimarco, V. Arginine and Endothelial Function. Biomedicines 2020, 8, 277. [Google Scholar] [CrossRef]
- Barberis, E.; Timo, S.; Amede, E.; Vanella, V.V.; Puricelli, C.; Cappellano, G.; Raineri, D.; Cittone, M.G.; Rizzi, E.; Pedrinelli, A.R.; et al. Large-Scale Plasma Analysis Revealed New Mechanisms and Molecules Associated with the Host Response to SARS-CoV-2. Int. J. Mol. Sci. 2020, 21, 8623. [Google Scholar] [CrossRef]
- Reizine, F.; Lesouhaitier, M.; Gregoire, M.; Pinceaux, K.; Gacouin, A.; Maamar, A.; Painvin, B.; Camus, C.; Le Tulzo, Y.; Tattevin, P.; et al. SARS-CoV-2-Induced ARDS Associates with MDSC Expansion, Lymphocyte Dysfunction, and Arginine Shortage. J. Clin. Immunol. 2021, 41, 515–525. [Google Scholar] [CrossRef]
- Ponzo, V.; Pellegrini, M.; D’Eusebio, C.; Bioletto, F.; Goitre, I.; Buscemi, S.; Frea, S.; Ghigo, E.; Bo, S. Mediterranean Diet and SARS-COV-2 Infection: Is There Any Association? A Proof-of-Concept Study. Nutrients 2021, 13, 1721. [Google Scholar] [CrossRef]
- Park, Y.; Subar, A.F.; Hollenbeck, A.; Schatzkin, A. Dietary Fiber Intake and Mortality in the NIH-AARP Diet and Health Study. Arch. Intern. Med. 2011, 171, 1061–1068. [Google Scholar] [CrossRef] [Green Version]
- Kan, H.; Stevens, J.; Heiss, G.; Rose, K.M.; London, S.J. Dietary Fiber, Lung Function, and Chronic Obstructive Pulmonary Disease in the Atherosclerosis Risk in Communities Study. Am. J. Epidemiol. 2008, 167, 570–578. [Google Scholar] [CrossRef] [Green Version]
- Lee, E.-S.; Song, E.-J.; Nam, Y.-D. Dysbiosis of Gut Microbiome and Its Impact on Epigenetic Regulation. J. Clin. Epigenet. 2017, 1, 14. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.L.; Erickson, J.M.; Lloyd, B.B.; Slavin, J.L. Health Effects and Sources of Prebiotic Dietary Fiber. Curr. Dev. Nutr. 2018, 2, nzy005. [Google Scholar] [CrossRef] [Green Version]
- Myhrstad, M.C.W.; Tunsjø, H.; Charnock, C.; Telle-Hansen, V.H. Dietary Fiber, Gut Microbiota, and Metabolic Regulation-Current Status in Human Randomized Trials. Nutrients 2020, 12, 859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Munckhof, I.C.L.; Kurilshikov, A.; Ter Horst, R.; Riksen, N.P.; Joosten, L.A.B.; Zhernakova, A.; Fu, J.; Keating, S.T.; Netea, M.G.; de Graaf, J.; et al. Role of Gut Microbiota in Chronic Low-Grade Inflammation as Potential Driver for Atherosclerotic Cardiovascular Disease: A Systematic Review of Human Studies. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2018, 19, 1719–1734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calder, P.C.; Carr, A.C.; Gombart, A.F.; Eggersdorfer, M. Reply to “Comment on: Optimal Nutritional Status for a Well-Functioning Immune System Is an Important Factor to Protect against Viral Infections. Nutrients 2020, 12, 1181. Nutrients 2020, 12, 1181. [Google Scholar] [CrossRef] [Green Version]
- Alfano, G.; Ferrari, A.; Fontana, F.; Perrone, R.; Mori, G.; Ascione, E.; Magistroni, R.; Venturi, G.; Pederzoli, S.; Margiotta, G.; et al. Hypokalemia in Patients with COVID-19. Clin. Exp. Nephrol. 2021, 25, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Li, X.; Song, Q.; Hu, C.; Su, F.; Dai, J.; Ye, Y.; Huang, J.; Zhang, X. Assessment of Hypokalemia and Clinical Characteristics in Patients With Coronavirus Disease 2019 in Wenzhou, China. JAMA Netw. Open 2020, 3, e2011122. [Google Scholar] [CrossRef]
- Macdonald, J.E.; Struthers, A.D. What Is the Optimal Serum Potassium Level in Cardiovascular Patients? J. Am. Coll. Cardiol. 2004, 43, 155–161. [Google Scholar] [CrossRef]
- Lim, J.-H.; Jung, H.-Y.; Choi, J.-Y.; Park, S.-H.; Kim, C.-D.; Kim, Y.-L.; Cho, J.-H. Hypertension and Electrolyte Disorders in Patients with COVID-19. Electrolytes Blood Press. 2020, 18, 23–30. [Google Scholar] [CrossRef]
- Turban, S.; Juraschek, S.P.; Miller, E.R.; Anderson, C.A.M.; White, K.; Charleston, J.; Appel, L.J. Randomized Trial on the Effects of Dietary Potassium on Blood Pressure and Serum Potassium Levels in Adults with Chronic Kidney Disease. Nutrients 2021, 13, 2678. [Google Scholar] [CrossRef]
- Wallace, T.C. Combating COVID-19 and Building Immune Resilience: A Potential Role for Magnesium Nutrition? J. Am. Coll. Nutr. 2020, 39, 685–693. [Google Scholar] [CrossRef]
- Iotti, S.; Wolf, F.; Mazur, A.; Maier, J.A. The COVID-19 Pandemic: Is There a Role for Magnesium? Hypotheses and Perspectives. Magnes. Res. 2020, 33, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Fan, B.E.; Chong, V.C.L.; Chan, S.S.W.; Lim, G.H.; Lim, K.G.E.; Tan, G.B.; Mucheli, S.S.; Kuperan, P.; Ong, K.H. Hematologic Parameters in Patients with COVID-19 Infection. Am. J. Hematol. 2020, 95, E131–E134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; the Northwell COVID-19 Research Consortium; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical Features of Patients Infected with 2019 Novel Coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Keen, C.L.; Gershwin, M.E. Zinc Deficiency and Immune Function. Annu. Rev. Nutr. 1990, 10, 415–431. [Google Scholar] [CrossRef] [PubMed]
- Joachimiak, M.P. Zinc against COVID-19? Symptom Surveillance and Deficiency Risk Groups. PLoS Negl. Trop. Dis. 2021, 15, e0008895. [Google Scholar] [CrossRef] [PubMed]
- Vincent, M.; Duval, R.E.; Hartemann, P.; Engels-Deutsch, M. Contact Killing and Antimicrobial Properties of Copper. J. Appl. Microbiol. 2018, 124, 1032–1046. [Google Scholar] [CrossRef] [Green Version]
- Andreou, A.; Trantza, S.; Filippou, D.; Sipsas, N.; Tsiodras, S. COVID-19: The Potential Role of Copper and N-Acetylcysteine (NAC) in a Combination of Candidate Antiviral Treatments Against SARS-CoV-2. In Vivo 2020, 34, 1567–1588. [Google Scholar] [CrossRef] [PubMed]
- Raha, S.; Mallick, R.; Basak, S.; Duttaroy, A.K. Is Copper Beneficial for COVID-19 Patients? Med. Hypotheses 2020, 142, 109814. [Google Scholar] [CrossRef]
- Percival, S.S. Copper and Immunity. Am. J. Clin. Nutr. 1998, 67, 1064S–1068S. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, C.; Rios, E.; Olivos, J.; Brunser, O.; Olivares, M. Iron, Copper and Immunocompetence. Br. J. Nutr. 2007, 98 (Suppl. S1), S24–S28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghani, S.M.A.; Goon, J.A.; Azman, N.H.E.N.; Zakaria, S.N.A.; Hamid, Z.; Ngah, W.Z.W. Comparing the Effects of Vitamin E Tocotrienol-Rich Fraction Supplementation and α-Tocopherol Supplementation on Gene Expression in Healthy Older Adults. Clinics 2019, 74, e688. [Google Scholar] [CrossRef] [PubMed]
- Neupane, B.; Walter, S.D.; Krueger, P.; Marrie, T.; Loeb, M. Predictors of Inhospital Mortality and Re-Hospitalization in Older Adults with Community-Acquired Pneumonia: A Prospective Cohort Study. BMC Geriatr. 2010, 10, 22. [Google Scholar] [CrossRef] [Green Version]
- Erol, S.A.; Tanacan, A.; Anuk, A.T.; Tokalioglu, E.O.; Biriken, D.; Keskin, H.L.; Moraloglu, O.T.; Yazihan, N.; Sahin, D. Evaluation of Maternal Serum Afamin and Vitamin E Levels in Pregnant Women with COVID-19 and Its Association with Composite Adverse Perinatal Outcomes. J. Med. Virol. 2021, 93, 2350–2358. [Google Scholar] [CrossRef]
- Alkhatib, A. Antiviral Functional Foods and Exercise Lifestyle Prevention of Coronavirus. Nutrients 2020, 12, 2633. [Google Scholar] [CrossRef]
- De Souza, R.G.M.; Schincaglia, R.M.; Pimentel, G.D.; Mota, J.F. Nuts and Human Health Outcomes: A Systematic Review. Nutrients 2017, 9, 1311. [Google Scholar] [CrossRef] [Green Version]
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional Foods and Lifestyle Approaches for Diabetes Prevention and Management. Nutrients 2017, 9, 1310. [Google Scholar] [CrossRef] [Green Version]
- Thomas, S.; Patel, D.; Bittel, B.; Wolski, K.; Wang, Q.; Kumar, A.; Il’Giovine, Z.J.; Mehra, R.; McWilliams, C.; Nissen, S.E.; et al. Effect of High-Dose Zinc and Ascorbic Acid Supplementation vs Usual Care on Symptom Length and Reduction Among Ambulatory Patients With SARS-CoV-2 Infection: The COVID A to Z Randomized Clinical Trial. JAMA Netw. Open 2021, 4, e210369. [Google Scholar] [CrossRef]
- Vatsalya, V.; Li, F.; Frimodig, J.C.; Gala, K.S.; Srivastava, S.; Kong, M.; Ramchandani, V.A.; Feng, W.; Zhang, X.; McClain, C.J. Therapeutic Prospects for Th-17 Cell Immune Storm Syndrome and Neurological Symptoms in COVID-19: Thiamine Efficacy and Safety, In-Vitro Evidence and Pharmacokinetic Profile. MedRxiv Prepr. Serv. Health Sci. 2020. [Google Scholar] [CrossRef]
- Al Sulaiman, K.; Aljuhani, O.; Al Dossari, M.; Alshahrani, A.; Alharbi, A.; Algarni, R.; Al Jeraisy, M.; Al Harbi, S.; Al Katheri, A.; Al Eidan, F.; et al. Evaluation of Thiamine as Adjunctive Therapy in COVID-19 Critically Ill Patients: A Two-Center Propensity Score Matched Study. Crit. Care 2021, 25, 223. [Google Scholar] [CrossRef]
- Chaari, A.; Bendriss, G.; Zakaria, D.; McVeigh, C. Importance of dietary changes during the coronavirus pandemic: How to upgrade your immune response. Front Public Health. 2020, 8, 476. [Google Scholar] [CrossRef]
- Moludi, J.; Qaisar, S.; Alizadeh, M.; Jafari Vayghan, H.; Naemi, M.; Rahimi, A.; Mousavi, R. The relationship between Dietary Inflammatory Index and disease severity and inflammatory status: A case–control study of COVID-19 patients. Br. J. Nutr. 2021, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Perez-Araluce, R.; Martinez-Gonzalez, M.A.; Fern, C.I.; Fernández-Lázaro, C.; Bes-Rastrollo, M.; Gea, A.; Carlos, S. Mediterranean diet and the risk of COVID-19 in the ‘Seguimiento Universidad de Navarra’ cohort. Clin. Nutr. 2021. [Google Scholar] [CrossRef]
- Deschasaux-Tanguy, M.; Srour, B.; Bourhis, L.; Arnault, N.; Druesne-Pecollo, N.; Esseddik, Y.; de Edelenyi, F.S.; Allègre, J.; Allès, B.; Andreeva, V.A.; et al. Nutritional risk factors for SARS-CoV-2 infection: A prospective study within the NutriNet-Santé cohort. BMC Med. 2021, 19, 290. [Google Scholar] [CrossRef]
- Vu, T.-H.T.; Rydland, K.J.; Achenbach, C.J.; Van Horn, L.; Cornelis, M.C. Dietary behaviors and incident COVID-19 in the UK Biobank. Nutrients. 2021, 13, 2114. [Google Scholar] [CrossRef]
- Rouf, R.; Uddin, S.J.; Sarker, D.K.; Islam, M.T.; Ali, E.S.; Shilpi, J.A.; Nahar, L.; Tiralongo, E.; Sarker, S.D. Antiviral potential of garlic (Allium sativum) and its organosulfur compounds: A systematic update of pre-clinical and clinical data. Trends Food Sci. Technol. 2020, 104, 219. [Google Scholar] [CrossRef]
- Donmaa, M.M.; Donmab, O. The effects of allium sativum on immunity within the scope of COVID-19 infection. Med. Hypotheses 2020, 144, 109934. [Google Scholar] [CrossRef] [PubMed]
- Mösbauer, K.; Fritsch, V.N.; Adrian, L.; Bernhardt, J.; Gruhlke, M.C.H.; Slusarenko, A.J.; Niemeyer, D.; Antelmann, H. The Effect of Allicin on the Proteome of SARS-CoV-2 Infected Calu-3 Cells. Front. Microbiol. 2021, 12, 746795. [Google Scholar] [CrossRef] [PubMed]
- Jandaghi, P.; Hosseini, Z.; Chilibeck, P.; Hanley, A.J.; Deguire, J.R.; Bandy, B.; Pahwa, P.; Vatanparast, H. The Role of Immunomodulatory Nutrients in Alleviating Complications Related to SARS-CoV-2: A Scoping Review. Adv. Nutr. 2021, nmab128. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Kunugi, H. Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies. Molecules. 2021, 25, 1232. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Cheng, C.-S.; Zhang, C.; Tang, G.-Y.; Tan, H.-Y.; Chen, H.-Y.; Wang, N.; Lai, A.Y.-K.; Feng, Y. Edible and Herbal Plants for the Prevention and Management of COVID-19. Front. Pharmacol. 2021, 12, 656103. [Google Scholar] [CrossRef]
- Saakre, M.; Mathew, D.; Ravisankar, V. Perspectives on plant flavonoid quercetin-based drugs for novel SARS-CoV-2. Beni-Suef Univ. J. Basic Appl. Sci. 2021, 10, 21. [Google Scholar] [CrossRef]
- Moreb, N.A.; Albandary, A.; Jaiswal, S.; Jaiswal, A.K. Fruits and Vegetables in the Management of Underlying Conditions for COVID-19 High-Risk Groups. Foods 2021, 10, 389. [Google Scholar] [CrossRef]
- Ghidoli, M.; Colombo, F.; Sangiorgio, S.; Landoni, M.; Giupponi, L.; Nielsen, E.; Pilu, R. Food Containing Bioactive Flavonoids and Other Phenolic or Sulfur Phytochemicals with Antiviral Effect: Can We Design a Promising Diet Against COVID-19? Front. Nutr. 2021, 8, 661331. [Google Scholar] [CrossRef] [PubMed]

| Variable | Total N = 95 | Have Not Contracted COVID-19 N = 71 | Have Contracted COVID-19 N = 24 | |||||
|---|---|---|---|---|---|---|---|---|
| Student’s t-test p | ||||||||
| Age (years) | 34.66 ± 5.76 | 33.90 ± 5.72 | 36.92 ± 5.40 | 0.0212 | ||||
| Body weight (kg) | 73.39 ± 12.27 | 73.81 ± 12.31 | 72.17 ± 12.36 | 0.6921 | ||||
| Height (cm) | 176.97 ± 8.90 | 177.74 ± 8.88 | 174.71 ± 8.75 | 0.2202 | ||||
| BMI (kg/m2) | 23.32 ± 2.76 | 23.26 ± 2.82 | 23.51 ± 2.63 | 0.6431 | ||||
| TEE (kcal) | 2547 ± 445 | 2581 ± 459 | 2445 ± 393 | 0.3167 | ||||
| PAL | 1.50 ± 0.17 | 1.50 ± 0.18 | 1.47 ± 0.12 | 0.5443 | ||||
| Sleep duration (h) | 7:22 ± 0:49 | 7:26 ± 0:50 | 7:10 ± 0:45 | 0.1348 | ||||
| Steps | 12,080 ± 4526 | 12,226 ± 4768 | 11,648 ± 3778 | 0.6067 | ||||
| Variable | Category | N | % | N | % | N | % | Chi2 p |
| Sex | Men | 73 | 76.8 | 55 | 77.5 | 18 | 75.0 | 0.8046 |
| Women | 22 | 23.2 | 16 | 22.5 | 6 | 25.0 | ||
| Marital status | Single/divorced | 48 | 50.5 | 39 | 54.9 | 9 | 37.5 | 0.1398 |
| Married/cohabiting | 47 | 49.5 | 32 | 45.1 | 15 | 62.5 | ||
| Diet | Traditional | 54 | 56.8 | 38 | 53.5 | 16 | 66.7 | 0.2610 |
| Vegetarian | 41 | 43.2 | 33 | 46.5 | 8 | 33.3 | ||
| BMI (kg/m2) | Normal body weight | 64 | 67.4 | 48 | 67.6 | 16 | 66.7 | 0.9324 |
| Overweight | 31 | 32.6 | 23 | 32.4 | 8 | 33.3 | ||
| BF (%) | Under fat | 9 | 9.6 | 8 | 11.4 | 1 | 4.2 | 0.5676 |
| Normal | 69 | 73.4 | 50 | 71.4 | 19 | 79.2 | ||
| Overfat | 16 | 17.0 | 12 | 17.1 | 4 | 16.7 | ||
| Smoking | No | 83 | 87.4 | 61 | 85.9 | 22 | 91.7 | 0.5009 |
| Yes | 12 | 12.6 | 10 | 14.1 | 2 | 8.3 | ||
| Education | Secondary | 7 | 7.4 | 7 | 9.9 | 0 | 0.0 | 0.2393 |
| Higher | 88 | 92.6 | 64 | 90.1 | 24 | 90.0 | ||
| How do you rate your physical activity in your free time? | Low | 19 | 20.0 | 16 | 22.5 | 3 | 12.5 | 0.4448 |
| Moderate | 52 | 54.7 | 36 | 50.7 | 16 | 66.7 | ||
| High | 24 | 25.3 | 19 | 26.8 | 5 | 20.8 | ||
| Vitamin supplementation | I don’t use | 30 | 31.6 | 23 | 32.4 | 7 | 29.2 | 0.8841 |
| Periodically | 21 | 22.1 | 15 | 21.1 | 6 | 25.0 | ||
| Regular | 44 | 46.3 | 33 | 46.5 | 11 | 45.8 | ||
| Supplementation with minerals | I don’t use | 50 | 55.6 | 38 | 56.7 | 12 | 52.2 | 0.8236 |
| Periodically | 23 | 25.6 | 16 | 23.9 | 7 | 30.4 | ||
| Regular | 17 | 18.9 | 13 | 19.4 | 4 | 17.4 | ||
| Variable | Total N = 95 | Have Not Contracted COVID-19 N = 71 | Have Contracted COVID-19 N = 24 | |
|---|---|---|---|---|
| Me (Q1–Q3) | Me (Q1–Q3) | Me (Q1–Q3) | U Mann-Whitney Test p | |
| Energy (kcal) | 2209 (1897–2478) | 2274 (2001–2549) | 2120 (1731–2246) | 0.0173 |
| Water (mL) | 2500 (2064–3132) | 2680 (2232–3203) | 2118 (1671–2681) | 0.0015 |
| Total protein (g) | 82.1 (69.1–103.5) | 89 (70.9–109) | 77.3 (67.2–88.6) | 0.1454 |
| Animal protein (g) | 44.7 (24.7–61.8) | 44.7 (16.7–64.9) | 44.4 (32.6–56.5) | 0.6194 |
| Plant protein (g) | 37.9 (29.6–47.2) | 39.3 (33.3–54.5) | 30.3 (25–38.8) | 0.0015 |
| Arginine (mg) | 4621 (3342–5650) | 4850 (3650–5716) | 3699 (3147–4907) | 0.0540 |
| Fat (g) | 69.7 (56–85.7) | 72.1 (56.4–88.8) | 66 (52.8–78.4) | 0.2843 |
| Linoleic acid LA (C18:2) | 9.8 (7.3–13.7) | 10.3 (7.5–14) | 8.4 (6.9–12) | 0.1576 |
| α-Linolenic acid ALA (C18:3) | 1.6 (1.2–2.4) | 1.6 (1.3–2.5) | 1.5 (1.1–2.4) | 0.5041 |
| Omega−3 fatty acids | 1.7 (1.1–2.5) | 1.7 (1.2–2.3) | 1.6 (1–2.6) | 0.6561 |
| Omega−6 fatty acids | 8 (6.4–11.5) | 8.3 (6.5–11.6) | 7.6 (5.4–10.2) | 0.3639 |
| Total carbohydrates (g) | 296.3 (246.1–335.7) | 307.9 (265.4–344.7) | 272 (220.2–301.3) | 0.0177 |
| Saccharose (g) | 40.1 (30–55.9) | 42 (30–55.9) | 36 (29–56) | 0.7578 |
| Dietary fiber (g) | 24.6 (19.1–34.5) | 26.8 (22.2–38.1) | 18.8 (17–27) | 0.0004 |
| Alcohol (g) | 6 (0–13.2) | 6.2 (0–16.5) | 5 (0–11.3) | 0.6358 |
| Potassium (mg) | 3646 (3008–4409) | 3758 (3166–4572) | 3131 (2509–3777) | 0.0015 |
| Calcium (mg) | 843 (686–1007) | 888 (686–1073) | 758 (687–884) | 0.1252 |
| Magnesium (mg) | 410.3 (338.9–548.1) | 439.4 (352.8–581.2) | 346.9 (285.1–429.7) | 0.0003 |
| Iron (mg) | 15.7 (12.4–19.6) | 17 (12.8–20.8) | 12.7 (11.1–16.4) | 0.0010 |
| Zinc (mg) | 12.2 (9.5–14.5) | 12.6 (9.9–15.2) | 10.1 (8.8–12.9) | 0.0248 |
| Copper (mg) | 1.7 (1.3–2.3) | 1.8 (1.4–2.4) | 1.4 (1.1–1.7) | 0.0004 |
| Manganese (mg) | 6.4 (4.5–8.9) | 6.8 (4.8–9.3) | 4.8 (3.9–6.3) | 0.0072 |
| Vitamin A (µg) | 1190 (804.3–1532.1) | 1209 (863.1–1593.4) | 938 (705.3–1496.2) | 0.1055 |
| Beta-carotene (µg) | 4170 (2889–6481) | 4375 (3186–6802) | 2924 (2017–5976) | 0.0607 |
| Vitamin E (alpha-tocopherol equivalent) (mg) | 12.5 (9–16.2) | 13.4 (10.3–16.3) | 9.1 (7.8–11.9) | 0.0034 |
| Thiamin (mg) | 1.4 (1–1.7) | 1.4 (1.2–1.8) | 1.2 (0.9–1.4) | 0.0099 |
| Riboflavin (mg) | 1.8 (1.4–2.1) | 1.9 (1.5–2.2) | 1.6 (1.4–1.9) | 0.0821 |
| Niacin (mg) | 18.7 (14.9–24.7) | 19.2 (15.4–26.6) | 16.8 (14.2–20.1) | 0.1074 |
| Vitamin B6 (mg) | 2 (1.5–2.6) | 2.1 (1.6–2.8) | 1.8 (1.4–2.2) | 0.0112 |
| Vitamin B12 (µg) | 3.5 (2.6–5.8) | 3.6 (2.5–6.1) | 3.1 (2.7–4.7) | 0.5096 |
| Vitamin C (mg) | 109.8 (71.1–162.5) | 120 (79.3–171) | 107 (47.8–141.7) | 0.1111 |
| Folates (µg) | 370.1 (274.9–477.8) | 392 (297.8–494.1) | 279.9 (221.7–378.5) | 0.0024 |
| Vitamin D (µg) Dietary Inflammatory Index | 3.6 (2–8) −0.82 (−2.1–0.93) | 3.5 (2–7.8) −1.08 (−2.28–0.45) | 4.2 (1.8–11.2) 0.98 (−1.78–1.83) | 0.7063 0.0042 |
| Variable | Total N = 95 | Have Not Contracted COVID-19 N = 71 | Have Contracted COVID-19 N = 24 | |
|---|---|---|---|---|
| Me (Q1–Q3) [%] | Me (Q1–Q3) [%] | Me (Q1–Q3) [%] | U Mann-Whitney Test p | |
| Water | 100.4 (83.2–131.7) | 110.9 (89.7–143.9) | 85.8 (73.2–107.2) | 0.0011 |
| Total protein | 120.2 (102.6–147.4) | 124.9 (105.1–154.3) | 112.5 (102.6–129.8) | 0.2174 |
| Digestible carbohydrates | 206.2 (174.7–231.2) | 212.5 (187.8–239.3) | 187.5 (153.1–214.1) | 0.0323 |
| Potassium | 104.2 (86–126) | 107.4 (90.5–130.6) | 89.5 (71.7–107.9) | 0.0015 |
| Calcium | 84.3 (68.7–100.7) | 88.8 (68.7–107.3) | 75.8 (68.7–88.4) | 0.1252 |
| Magnesium | 105.9 (87.2–135.7) | 110.2 (93.4–147) | 86.2 (74.8–105.1) | 0.0001 |
| Iron | 151.7 (108.4–183.2) | 156.9 (113.8–202.4) | 122.3 (101.3–135.9) | 0.0052 |
| Zinc | 115.2 (93.3–137.6) | 123 (97.8–143.9) | 94.4 (89.4–118.5) | 0.0088 |
| Copper | 187.6 (147.7–250.3) | 198.1 (156.4–270.4) | 152.9 (124–189.1) | 0.0004 |
| Manganese | 280 (207.1–405.8) | 309.9 (218.5–478.5) | 215.6 (191.1–275.1) | 0.0065 |
| Vitamin A | 136.8 (97.9–177) | 143.9 (106.3–180.7) | 104.2 (88.9–169) | 0.0883 |
| Vitamin E (alpha-tocopherol equivalent) | 126.5 (93.7–169.4) | 139.2 (103.2–178.2) | 99.8 (86–118.8) | 0.0042 |
| Thiamin | 107.1 (87.6–138) | 112.5 (91.1–143.3) | 91.2 (73.9–111.2) | 0.0112 |
| Riboflavin | 137.4 (118.8–169.1) | 143.3 (119.7–180.8) | 130 (108.5–147.6) | 0.0734 |
| Niacin | 117.4 (97–159.5) | 131.3 (101.2–166.3) | 108 (90.8–125.7) | 0.0966 |
| Vitamin B6 | 152.8 (118.3–196.8) | 163.2 (123.6–213.3) | 135.8 (105.4–170.9) | 0.0112 |
| Vitamin C | 132.9 (79–210.3) | 138.9 (90.5–215.3) | 118.9 (60.9–160.5) | 0.1150 |
| Saturated fatty acids | 141.5 (98–180.6) | 142.1 (97.9–180.6) | 139.1 (109.9–181.6) | 0.7906 |
| Linoleic acid LA (C18:2) | 78.3 (59.3–102.5) | 82.9 (61.9–103.9) | 68.7 (59.1–95.9) | 0.2441 |
| α-Linolenic acid ALA (C18:3) | 101.3 (79.7–159) | 103.1 (80.1–159.5) | 101.2 (74–155.4) | 0.7906 |
| Dietary fiber | 98.3 (76.2–137.8) | 107.3 (88.7–152.4) | 75.3 (68–108.1) | 0.0004 |
| Folates | 92.5 (68.7–119.4) | 98 (74.4–123.5) | 70 (55.4–94.6) | 0.0024 |
| Vitamin B12 | 146 (109.4–242.2) | 149.7 (106.2–254.9) | 128.8 (111–194.1) | 0.5096 |
| Vitamin D | 23.8 (13.2–53.5) | 23.3 (13.2–51.9) | 28.1 (12.3–74.7) | 0.7063 |
| Variable | Total N = 95 | Have Not Contracted COVID-19 N = 71 | Have Contracted COVID-19 N = 24 | |||||
|---|---|---|---|---|---|---|---|---|
| Me (Q1–Q3) | Me (Q1–Q3) | Me (Q1–Q3) | U Mann-Whitney Test p | |||||
| Groats and rice (g) | 14.3 (6.5–29.6) | 18 (9.9–33.3) | 8.8 (0–22.8) | 0.0129 | ||||
| Seeds (g) | 2 (0–6.7) | 2.5 (0–6.9) | 1.4 (0–4.3) | 0.2686 | ||||
| Nuts (g) | 13.7 (2.2–30.8) | 16.3 (6.7–35.6) | 1.9 (0.5–16.1) | 0.0035 | ||||
| Fruit (g) | 204.6 (111.4–400.4) | 231.4 (110.8–419.5) | 154 (115.8–282.8) | 0.2511 | ||||
| Vegetable (g) | 343.3 (236.1–470.9) | 361 (262.5–476.5) | 240.8 (167.6–414.8) | 0.0136 | ||||
| Fruit and vegetables (g) Legumes (g) | 554.9 (409.9–781.8) 10 (0–50.9) | 579,1 (455.3–839.4) 16 (1.5–72.8) | 431.8 (262.6–670.3) 2.4 (0–11.4) | 0.0177 0.0125 | ||||
| Variable | Category | N | % | N | % | N | % | Chi2 p |
| Garlic | <1 g on average daily | 78 | 82.1 | 55 | 77.5 | 23 | 95.8 | 0.0424 |
| ≥1 g on average daily | 17 | 17.9 | 16 | 22.5 | 1 | 4.2 | ||
| Onion | <10 g on average daily | 70 | 73.7 | 50 | 70.4 | 20 | 83.3 | 0.2143 |
| ≥10g on average daily | 25 | 26.3 | 21 | 29.6 | 4 | 16.7 | ||
| Fruit and vegetables | 500 g and more per day | 59 | 62.1 | 48 | 67.6 | 11 | 45.8 | 0.0573 |
| Less than 500 g per day | 36 | 37.9 | 23 | 32.4 | 13 | 54.2 | ||
| Nuts | 10 g and more per day | 56 | 58.9 | 48 | 67.6 | 8 | 33.3 | 0.0032 |
| Up to 10 g per day | 39 | 41.1 | 23 | 32.4 | 16 | 66.7 | ||
| Model | FV ≥ 500 g and nuts ≥ 10 g | Failure to Meet One of the Conditions | FV< 500 g and Nuts < 10 g | ptrend |
|---|---|---|---|---|
| Model 1 a | 1 (ref.) | 4.13 (1.25–16.30) | 7.36 (2.00–31.81) | 0.003 |
| Model 2 b | 1 (ref.) | 5.03 (1.39–21.97) | 9.01 (1.99–49.31) | 0.005 |
| Model 3 c | 1 (ref.) | 5.73 (1.49–27.82) | 12.22 (2.47–78.12) | 0.003 |
| Variable | Average Daily Consumption of >500 g of Fruit and Vegetables and >10 g of Nuts N = 40 | Failure to Meet One of the Conditions (Daily Consumption of <500 g of Fruit and Vegetables or <10 g of Nuts) N = 35 | Failure to Meet any of the Conditions (Daily Consumption of <500 g of Fruit and Vegetables and <10 g of Nuts) N = 20 | |||||
|---|---|---|---|---|---|---|---|---|
| Me (Q1–Q3) | Me (Q1–Q3) | Me (Q1–Q3) | Kruskal–Wallis Test p | |||||
| Groats and rice (g) | 21.6 (8.6–42.5) | 13 (1–28) | 12.2 (7.1–22.4) | 0.1655 | ||||
| Seeds (g) | 4.1 (1.5–9.6) | 1.7 (0–8.7) | 0 (0.0–1.3) | 0.0001 | ||||
| Nuts (g) | 28.7 (17.8–46.9) | 9.2 (2.2–17.3) | 0.9 (0.0–2.1) | <0.0001 | ||||
| Fruit (g) | 332.4 (237.8–442.4) | 167.8 (110.8–347.9) | 105.3 (39.8–124) | <0.0001 | ||||
| Vegetable (g) | 427 (354.3–509.7) | 305.2 (212.2–454.5) | 233.9 (130.3–297.5) | <0.0001 | ||||
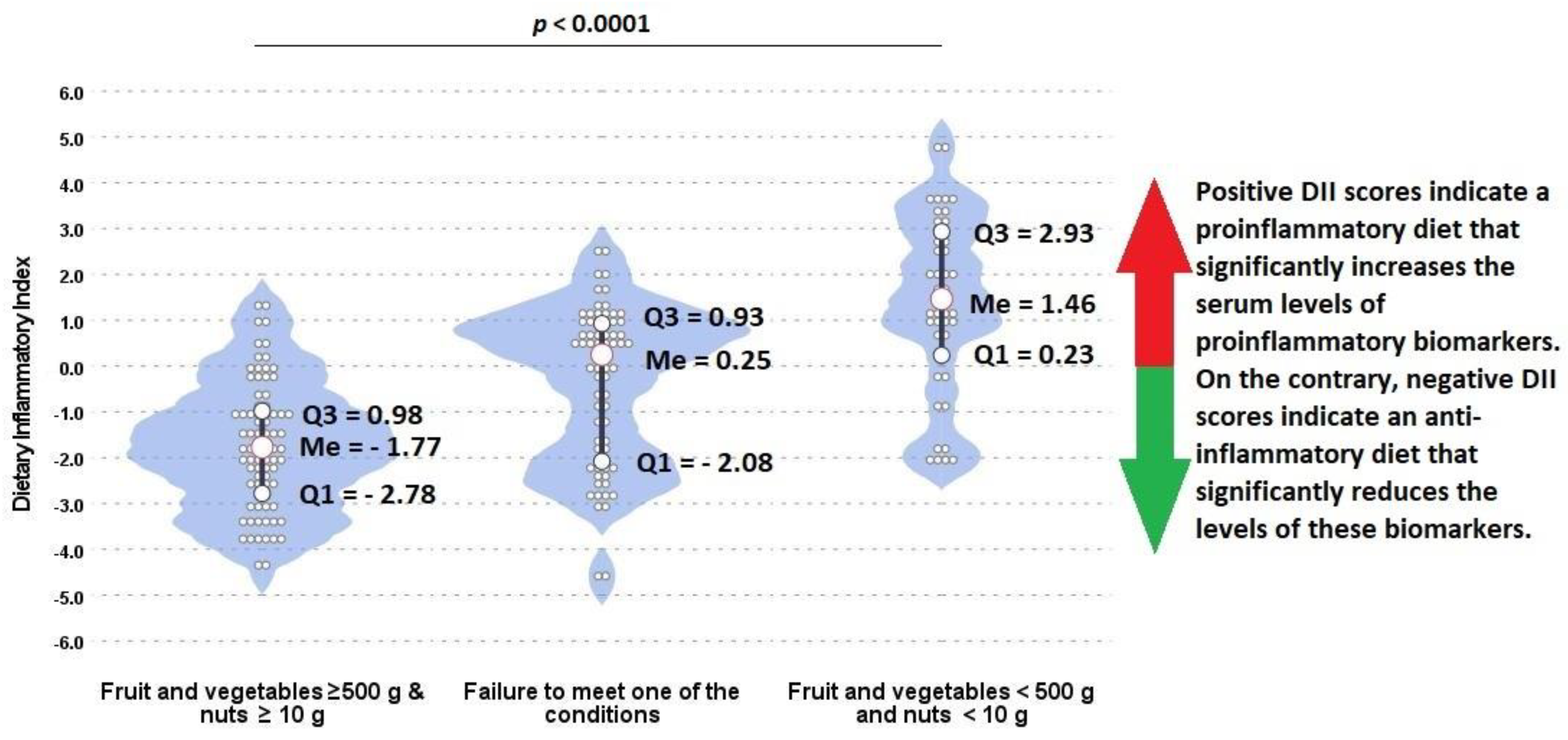
| Fruit and vegetables (g) Legumes (g) Dietary Inflammatory Index | 722.6 (587.1–971.6) 25.8 (6.2–78.9) −1.77 (−2.78–0.98) | 521.2 (420.5–698.5) 6.3 (0.0–30.7) 0.25 (−2.08–0.93) | 354.9 (231.2–386.5) 2.1 (0.0–14.9) 1.46 (0.23–2.93) | <0.0001 0.0137 <0.0001 | ||||
| Variable | Category | N | % | N | % | N | % | Chi2 p |
| Garlic | <1 g on average daily | 29 | 72.5 | 30 | 85.7 | 19 | 95.0 | 0.0787 |
| ≥1 g on average daily | 11 | 27.5 | 5 | 14.3 | 1 | 5.0 | ||
| Onion | <10 g on average daily | 26 | 65.0 | 26 | 74.3 | 18 | 90.0 | 0.1160 |
| ≥10 g on average daily | 14 | 35.0 | 9 | 25.7 | 2 | 10.0 | ||
| Fruit and vegetables | 500 and more g per day | 40 | 100.0 | 19 | 54.3 | 0 | 0.0 | <0.0001 |
| <500 g per day | 0 | 0.0 | 16 | 45.7 | 20 | 100.0 | ||
| Nuts | 10 and more g per day | 40 | 100.0 | 16 | 45.7 | 0 | 0.0 | <0.0001 |
| Up to 10 g per day | 0 | 0.0 | 19 | 54.3 | 20 | 100.0 | ||
| Diet | Traditional | 18 | 45.0 | 20 | 57.1 | 16 | 80.0 | 0.0358 |
| Vegetarian | 22 | 55.0 | 15 | 42.9 | 4 | 20.0 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jagielski, P.; Łuszczki, E.; Wnęk, D.; Micek, A.; Bolesławska, I.; Piórecka, B.; Kawalec, P. Associations of Nutritional Behavior and Gut Microbiota with the Risk of COVID-19 in Healthy Young Adults in Poland. Nutrients 2022, 14, 350. https://doi.org/10.3390/nu14020350
Jagielski P, Łuszczki E, Wnęk D, Micek A, Bolesławska I, Piórecka B, Kawalec P. Associations of Nutritional Behavior and Gut Microbiota with the Risk of COVID-19 in Healthy Young Adults in Poland. Nutrients. 2022; 14(2):350. https://doi.org/10.3390/nu14020350
Chicago/Turabian StyleJagielski, Paweł, Edyta Łuszczki, Dominika Wnęk, Agnieszka Micek, Izabela Bolesławska, Beata Piórecka, and Paweł Kawalec. 2022. "Associations of Nutritional Behavior and Gut Microbiota with the Risk of COVID-19 in Healthy Young Adults in Poland" Nutrients 14, no. 2: 350. https://doi.org/10.3390/nu14020350
APA StyleJagielski, P., Łuszczki, E., Wnęk, D., Micek, A., Bolesławska, I., Piórecka, B., & Kawalec, P. (2022). Associations of Nutritional Behavior and Gut Microbiota with the Risk of COVID-19 in Healthy Young Adults in Poland. Nutrients, 14(2), 350. https://doi.org/10.3390/nu14020350










