Malnutrition Prevention after Allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): A Prospective Explorative Interventional Study with an Oral Polymeric Formulation Enriched with Transforming Growth Factor Beta 2 (TGF-β2)
Abstract
:1. Introduction
2. Materials and Methods
TE-OPF Treatment Plan
- -
- BMI less than 22: 20% of TDEE;
- -
- BMI between 22.1 and 24.9: 12% of TDEE;
- -
- BMI between 25 and 29.9: 10% of TDEE;
- -
- BMI between 30 and 34.9: 8% of TDEE;
- -
- BMI higher than 35: 5% of TDEE.
3. Results
3.1. Secondary Outcomes
3.1.1. TE-OPF Assumption and GVHD
3.1.2. TE-OPF Assumption and Infectious Complications
3.1.3. TE-OPF Assumption and Relapse-Free Survival
3.2. Biomarker Studies
3.2.1. TE-OPF and IGF-1
3.2.2. TE-OPF, Malnutrition, and Lymphocyte Subsets
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Espinoza, M.; Perelli, J.; Olmos, R.; Bertin, P.; Jara, V.; Ramirez, P. Nutritional Assessment as Predictor of Complications after Hematopoietic Stem Cell Transplantation. Rev. Bras. Hematol. Hemoter. 2016, 38, 7–14. [Google Scholar] [CrossRef]
- Sommacal, H.M.; Gazal, C.H.A.; Jochims, A.M.K.; Beghetto, M.; Paz, A.; da Rocha Silla, L.M.; de Mello, E.D. Clinical Impact of Systematic Nutritional Care in Adults Submitted to Allogeneic Hematopoietic Stem Cell Transplantation. Rev. Bras. Hematol. Hemoter. 2012, 34, 334–338. [Google Scholar] [CrossRef]
- Imataki, O.; Nakatani, S.; Hasegawa, T.; Kondo, M.; Ichihashi, K.; Araki, M.; Ishida, T.; Kim, S.-W.; Mori, S.; Fukuda, T.; et al. Nutritional Support for Patients Suffering from Intestinal Graft-versus-Host Disease after Allogeneic Hematopoietic Stem Cell Transplantation. Am. J. Hematol. 2006, 81, 747–752. [Google Scholar] [CrossRef]
- Gyurkocza, B.; Rezvani, A.; Storb, R.F. Allogeneic Hematopoietic Cell Transplantation: The State of the Art. Expert Rev. Hematol. 2010, 3, 285–299. [Google Scholar] [CrossRef]
- Brotelle, T.; Lemal, R.; Cabrespine, A.; Combal, C.; Hermet, E.; Ravinet, A.; Bay, J.-O.; Bouteloup, C. Prevalence of Malnutrition in Adult Patients Previously Treated with Allogeneic Hematopoietic Stem-Cell Transplantation. Clin. Nutr. 2018, 37, 739–745. [Google Scholar] [CrossRef]
- Rzepecki, P.; Barzal, J.; Sarosiek, T.; Szczylik, C. Biochemical Indices for the Assessment of Nutritional Status during Hematopoietic Stem Cell Transplantation: Are They Worth Using? A Single Center Experience. Bone Marrow Transpl. 2007, 40, 567–572. [Google Scholar] [CrossRef]
- Wang, B.; Yan, X.; Cai, J.; Wang, Y.; Liu, P. Nutritional Assessment with Different Tools in Leukemia Patients after Hematopoietic Stem Cell Transplantation. Chin. J. Cancer Res. 2013, 25, 762–769. [Google Scholar] [CrossRef]
- Liu, P.; Wang, B.; Yan, X.; Cai, J.; Wang, Y. Comprehensive Evaluation of Nutritional Status before and after Hematopoietic Stem Cell Transplantation in 170 Patients with Hematological Diseases. Chin. J. Cancer Res. 2016, 28, 626–633. [Google Scholar] [CrossRef]
- Hadjibabaie, M.; Iravani, M.; Taghizadeh, M.; Ataie-Jafari, A.; Shamshiri, A.R.; Mousavi, S.A.; Alimoghaddam, K.; Hosseini, S.; Ghavamzadeh, A. Evaluation of Nutritional Status in Patients Undergoing Hematopoietic SCT. Bone Marrow Transpl. 2008, 42, 469–473. [Google Scholar] [CrossRef]
- Morello, E.; Malagola, M.; Bernardi, S.; Pristipino, C.; Russo, D. The Role of Allogeneic Hematopoietic Stem Cell Transplantation in the Four P Medicine Era. Blood Res. 2018, 53, 3–6. [Google Scholar] [CrossRef] [Green Version]
- Falda, M.; Busca, A.; Baldi, I.; Mordini, N.; Bruno, B.; Allione, B.; Rambaldi, A.; Morello, E.; Narni, F.; Santarone, S.; et al. Nonmyeloablative Allogeneic Stem Cell Transplantation in Elderly Patients with Hematological Malignancies: Results from the GITMO (Gruppo Italiano Trapianto Midollo Osseo) Multicenter Prospective Clinical Trial. Am. J. Hematol. 2007, 82, 863–866. [Google Scholar] [CrossRef]
- Jager-Wittenaar, H.; Ottery, F.D. Assessing Nutritional Status in Cancer: Role of the Patient-Generated Subjective Global Assessment. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 322–329. [Google Scholar] [CrossRef]
- Bozzetti, F.; Arends, J.; Lundholm, K.; Micklewright, A.; Zurcher, G.; Muscaritoli, M. ESPEN Guidelines on Parenteral Nutrition: Non-Surgical Oncology. Clin. Nutr. 2009, 28, 445–454. [Google Scholar] [CrossRef]
- August, D.A.; Huhmann, M.B.; American Society for Parenteral and Enteral Nutrition. Clinical Guidelines: Nutrition Support Therapy during Adult Anticancer Treatment and in Hematopoietic Cell Transplantation. JPEN J. Parenter. Enteral. Nutr. 2009, 33, 472–500. [Google Scholar] [CrossRef]
- Arends, J.; Baracos, V.; Bertz, H.; Bozzetti, F.; Calder, P.C.; Deutz, N.E.P.; Erickson, N.; Laviano, A.; Lisanti, M.P.; Lobo, D.N.; et al. ESPEN Expert Group Recommendations for Action against Cancer-Related Malnutrition. Clin. Nutr. 2017, 36, 1187–1196. [Google Scholar] [CrossRef]
- Arfons, L.M.; Lazarus, H.M. Total Parenteral Nutrition and Hematopoietic Stem Cell Transplantation: An Expensive Placebo? Bone Marrow Transpl. 2005, 36, 281–288. [Google Scholar] [CrossRef]
- Verdi Schumacher, M.; Moreira Faulhaber, G.A. Nutritional Status and Hyperglycemia in the Peritransplant Period: A Review of Associations with Parenteral Nutrition and Clinical Outcomes. Rev. Bras. Hematol. Hemoter. 2017, 39, 155–162. [Google Scholar] [CrossRef]
- Azarnoush, S.; Bruno, B.; Beghin, L.; Guimber, D.; Nelken, B.; Yakoub-Agha, I.; Seguy, D. Enteral Nutrition: A First Option for Nutritional Support of Children Following Allo-SCT? Bone Marrow Transpl. 2012, 47, 1191–1195. [Google Scholar] [CrossRef]
- Mattsson, J.; Westin, S.; Edlund, S.; Remberger, M. Poor Oral Nutrition after Allogeneic Stem Cell Transplantation Correlates Significantly with Severe Graft-versus-Host Disease. Bone Marrow Transpl. 2006, 38, 629–633. [Google Scholar] [CrossRef]
- Couriel, D.; Caldera, H.; Champlin, R.; Komanduri, K. Acute Graft-versus-Host Disease: Pathophysiology, Clinical Manifestations, and Management. Cancer 2004, 101, 1936–1946. [Google Scholar] [CrossRef]
- Sheean, P.; Braunschweig, C. The Incidence and Impact of Dextrose Dose on Hyperglycemia from Parenteral Nutrition (PN) Exposure in Hematopoietic Stem Cell Transplant (HSCT) Recipients. JPEN J Parenter Enteral Nutr 2006, 30, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Fuji, S.; Einsele, H.; Savani, B.N.; Kapp, M. Systematic Nutritional Support in Allogeneic Hematopoietic Stem Cell Transplant Recipients. Biol Blood Marrow Transpl. 2015, 21, 1707–1713. [Google Scholar] [CrossRef] [PubMed]
- Fredricks, D.N. The Gut Microbiota and Graft-versus-Host Disease. J. Clin. Investig. 2019, 129, 1808–1817. [Google Scholar] [CrossRef]
- Nalle, S.C.; Turner, J.R. Intestinal Barrier Loss as a Critical Pathogenic Link between Inflammatory Bowel Disease and Graft-versus-Host Disease. Mucosal. Immunol. 2015, 8, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Bassim, C.W.; Fassil, H.; Dobbin, M.; Steinberg, S.M.; Baird, K.; Cole, K.; Joe, G.; Comis, L.E.; Mitchell, S.A.; Grkovic, L.; et al. Malnutrition in Patients with Chronic GVHD. Bone Marrow Transpl. 2014, 49, 1300–1306. [Google Scholar] [CrossRef] [PubMed]
- Limketkai, B.N.; Wolf, A.; Parian, A.M. Nutritional Interventions in the Patient with Inflammatory Bowel Disease. Gastroenterol. Clin. N. Am. 2018, 47, 155–177. [Google Scholar] [CrossRef]
- Naymagon, S.; Naymagon, L.; Wong, S.-Y.; Ko, H.M.; Renteria, A.; Levine, J.; Colombel, J.-F.; Ferrara, J. Acute Graft-versus-Host Disease of the Gut: Considerations for the Gastroenterologist. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 711–726. [Google Scholar] [CrossRef]
- Hartman, C.; Berkowitz, D.; Weiss, B.; Shaoul, R.; Levine, A.; Adiv, O.E.; Shapira, R.; Fradkin, A.; Wilschanski, M.; Tamir, A.; et al. Nutritional Supplementation with Polymeric Diet Enriched with Transforming Growth Factor-Beta 2 for Children with Crohn’s Disease. Isr. Med. Assoc. J. 2008, 10, 503–507. [Google Scholar]
- Bauche, D.; Marie, J.C. Transforming Growth Factor Beta: A Master Regulator of the Gut Microbiota and Immune Cell Interactions. Clin. Transl. Immunol. 2017, 6, e136. [Google Scholar] [CrossRef]
- Morello, E.; Arena, F.; Malagola, M.; Turra, A.; Polverelli, N.; Zollner, T.; Cavagna, E.; Farina, M.; Cancelli, V.; Gandolfi, L. Malnutrition and gvhd in allogeneic bone marrow transplantation: A prospective study. In Haematologica; Ferrata Storti Foundation: Pavia, Italy, 2019; Volume 104, p. 97. [Google Scholar]
- Morello, E.; Guarinoni, M.G.; Arena, F.; Andreoli, M.; Bernardi, S.; Malagola, M.; Turra, A.; Polverelli, N.; Russo, D. A Systematic Review of the Literature and Perspectives on the Role of Biomarkers in the Management of Malnutrition After Allogeneic Hematopoietic Stem Cell Transplantation. Front. Immunol. 2020, 11, 535890. [Google Scholar] [CrossRef]
- Caocci, G.; Baccoli, R.; Vacca, A.; Mastronuzzi, A.; Bertaina, A.; Piras, E.; Littera, R.; Locatelli, F.; Carcassi, C.; La Nasa, G. Comparison between an Artificial Neural Network and Logistic Regression in Predicting Acute Graft-vs-Host Disease after Unrelated Donor Hematopoietic Stem Cell Transplantation in Thalassemia Patients. Exp. Hematol. 2010, 38, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Pigneur, B.; Ruemmele, F.M. Nutritional Interventions for the Treatment of IBD: Current Evidence and Controversies. Therap Adv. Gastroenterol. 2019, 12, 1756284819890534. [Google Scholar] [CrossRef] [PubMed]
- Moreno, D.F.; Cid, J. Graft-versus-host disease. Med. Clin. (Barc.) 2019, 152, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Shono, Y.; van den Brink, M.R.M. Gut Microbiota Injury in Allogeneic Haematopoietic Stem Cell Transplantation. Nat. Rev. Cancer 2018, 18, 283–295. [Google Scholar] [CrossRef]
- Romick-Rosendale, L.E.; Haslam, D.B.; Lane, A.; Denson, L.; Lake, K.; Wilkey, A.; Watanabe, M.; Bauer, S.; Litts, B.; Luebbering, N.; et al. Antibiotic Exposure and Reduced Short Chain Fatty Acid Production after Hematopoietic Stem Cell Transplant. Biol. Blood Marrow Transpl. 2018, 24, 2418–2424. [Google Scholar] [CrossRef]
- Whangbo, J.; Ritz, J.; Bhatt, A. Antibiotic-Mediated Modification of the Intestinal Microbiome in Allogeneic Hematopoietic Stem Cell Transplantation. Bone Marrow Transpl. 2017, 52, 183–190. [Google Scholar] [CrossRef]
- Sun, M.; Wu, W.; Liu, Z.; Cong, Y. Microbiota Metabolite Short Chain Fatty Acids, GPCR, and Inflammatory Bowel Diseases. J Gastroenterol. 2017, 52, 1–8. [Google Scholar] [CrossRef]
- Yoshioka, K.; Kakihana, K.; Doki, N.; Ohashi, K. Gut Microbiota and Acute Graft-versus-Host Disease. Pharmacol. Res. 2017, 122, 90–95. [Google Scholar] [CrossRef]
- Chen, Z.; Nilsson, E.; Lindholm, B.; Heimburger, O.; Barany, P.; Stenvinkel, P.; Qureshi, A.R.; Chen, J. Low Plasma Insulin-like Growth Factor-1 Associates with Increased Mortality in Chronic Kidney Disease Patients with Reduced Muscle Strength. J. Ren. Nutr. 2022; in press. [Google Scholar] [CrossRef]
- Guijarro, L.G.; Cano-Martinez, D.; Toledo-Lobo, M.V.; Salinas, P.S.; Chaparro, M.; Gomez-Lahoz, A.M.; Zoullas, S.; Rodriguez-Torres, R.; Roman, I.D.; Monasor, L.S.; et al. Relationship between IGF-1 and Body Weight in Inflammatory Bowel Diseases: Cellular and Molecular Mechanisms Involved. Biomed. Pharmacother. 2021, 144, 112239. [Google Scholar] [CrossRef]
- Papadopoulou, S.K.; Voulgaridou, G.; Kondyli, F.S.; Drakaki, M.; Sianidou, K.; Andrianopoulou, R.; Rodopaios, N.; Pritsa, A. Nutritional and Nutrition-Related Biomarkers as Prognostic Factors of Sarcopenia, and Their Role in Disease Progression. Diseases 2022, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Shimabukuro, M. IGF-1 and Cardiovascular and Non-Cardiovascular Mortality Risk in Patients with Chronic Kidney Disease: A Model of “Malnutrition- Inflammation-Atherosclerosis Syndrome”. J. Atheroscler. Thromb. 2022, 29, 1138–1139. [Google Scholar] [CrossRef] [PubMed]
- Shokouhifar, A.; Anani Sarab, G.; Yazdanifar, M.; Fereidouni, M.; Nouri, M.; Ebrahimi, M. Overcoming the UCB HSCs -Derived NK Cells Dysfunction through Harnessing RAS/MAPK, IGF-1R and TGF-Beta Signaling Pathways. Cancer Cell Int. 2021, 21, 298. [Google Scholar] [CrossRef] [PubMed]




| Sex (n; Female/Male) | 22/29 (43.1%/56.9%) |
|---|---|
| Age (Median) | 55 (range 20–72) |
| Median follow-up (days) | 279 (29–564) |
| Diagnosis (Acute leukemias, Myelodysplastic syndrome or Myeloproliferative disease vs. Lymphoma or Myeloma) | 40/11 (78.5%/21.5%) |
| Disease status at admission (Complete remission/Minimal residual disease/Advanced disease) | 22/21/8 (43.2%/41.1%/15.7%) |
| Donor type (Match-related donor/MUD/Haploidentical) | 21/20/10 (41.2%/39.2%/15.7%) |
| Stem cell source (Peripheral blood /Bone marrow) | 48/3 (94.1%/5.9%) |
| Nutritional status at admission following PG-SGA (Score A/B/C) | 38 A/12 B/1 C (74.5%/23.5%/2%) |
| GROUP A | Group B | p Value | |
|---|---|---|---|
| Total (n) | 24/51 (47%) | 27/51 (53%) | |
| Sex (F/M) | 9/15 | 13/14 | ns |
| Age (median) | 54 | 54 | ns |
| Diagnosis (AL-MDS-MPD/LPD) | 19/5 | 21/6 | ns |
| Disease status (AD/CR) | 10/14 | 12/15 | ns |
| Donor (MRD/MUD/Haplo) | 10/11/3 | 11/9/7 | ns |
| Source (PB/BM) | 22/2 | 26/1 | ns |
| Conditioning (MA/RIC) | 18/6 | 22/5 | ns |
| PG-SGA (A/B/C) | 19/5/0 | 19/7/1 | ns |
| GROUP A | GROUP B | p Value | |
|---|---|---|---|
| Total (n) | 24/51 (47%) | 27/51 (53%) | |
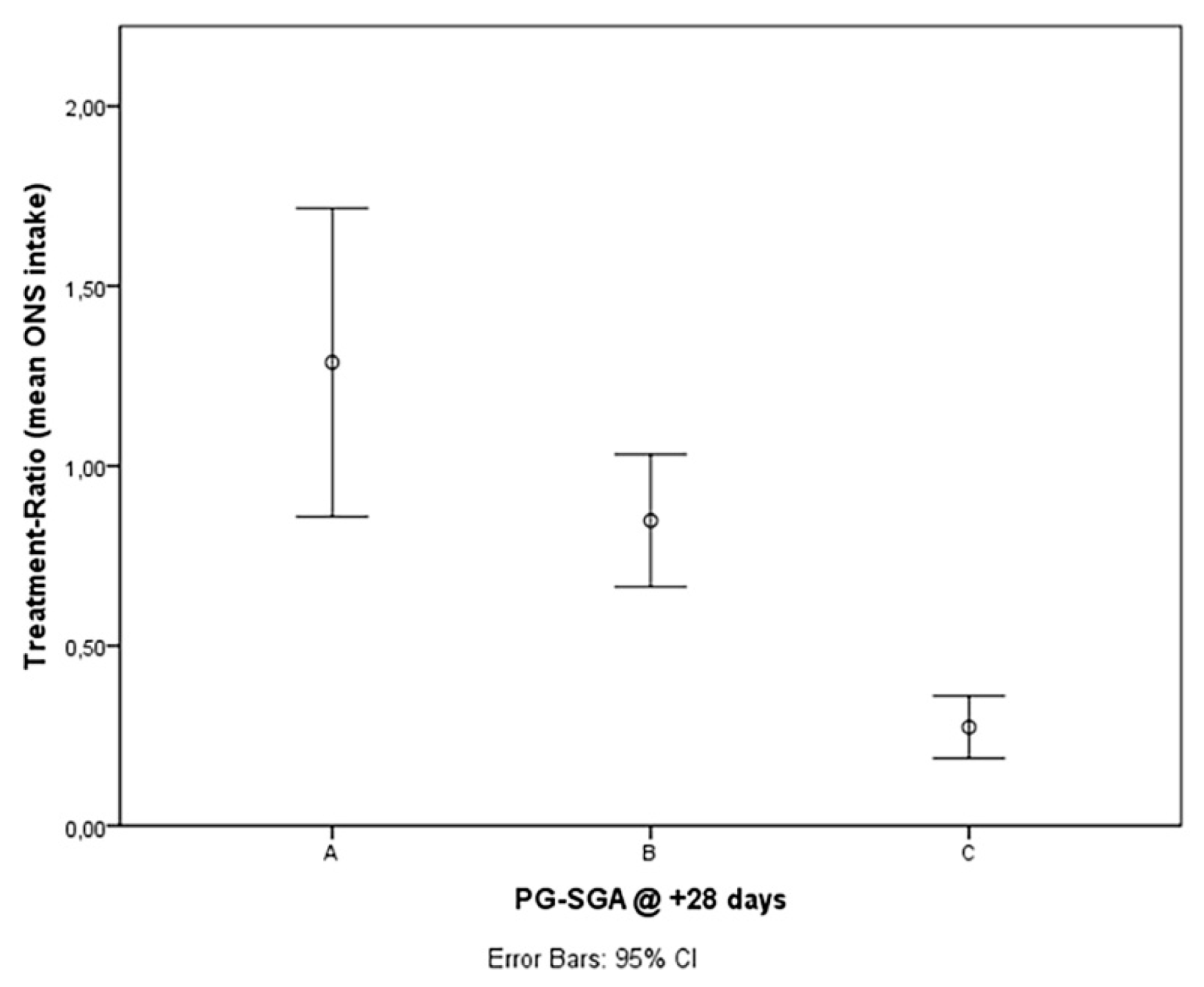
| PG-SGA Score A + B at +28 days (n) (%) | 21/24 (87.5%) | 3/27 (11.1%) | 0.000 |
| PG-SGA Score C at +28 days (n) (%) | 3/24 (12.5%) | 24/27 (88.9%) | 0.000 |
| Prevalence of aGVHD (%) | 7/24 (29.1%) | 14/27 (51.8%) | Ns |
| Prevalence of Gastrointestinal aGVHD (n) (%) | 0/24 | 8/27 (29.6%) | 0.005 |
| Incidence of Sepsis (%) | 7/24 (29.1%) | 15/27 (55.5%) | Ns |
| Incidence of Pneumonia (%) | 3/24 (12.5%) | 13/27 (48.1%) | 0.006 |
| Survival after alloHSCT (median, days) | 734 (580–881) | 424 (347–501) | Ns |
| Biomarker | Group A Mean (C.I.) | Group B Mean (C.I.) | |
|---|---|---|---|
| IGF1_14 | 165.56 (45–300) | 109.16 (36–228) | 0.021 |
| IGF1_28 | 153.64 (80.2–256) | 109.52 (34–211) | 0.032 |
| Lymphocytes_28 | 820 (70–2160) | 490 (10–1970) | NS |
| CD3+/CD4+_28 | 121.23 (3–424) | 63.08 (1–260) | 0.025 |
| CD19+_28 | 197.86 (0–199) | 136.17 (0–418) | NS |
| CD56+_28 | 230.74 (37–1431) | 138.22 (0–446) | NS |
| CD16+_28 | 155.61 (30–501) | 80.87 (0–218) | 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morello, E.; Arena, F.; Malagola, M.; Farina, M.; Polverelli, N.; Cavagna, E.; Colnaghi, F.; Donna, L.; Zollner, T.; Accorsi Buttini, E.; et al. Malnutrition Prevention after Allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): A Prospective Explorative Interventional Study with an Oral Polymeric Formulation Enriched with Transforming Growth Factor Beta 2 (TGF-β2). Nutrients 2022, 14, 3589. https://doi.org/10.3390/nu14173589
Morello E, Arena F, Malagola M, Farina M, Polverelli N, Cavagna E, Colnaghi F, Donna L, Zollner T, Accorsi Buttini E, et al. Malnutrition Prevention after Allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): A Prospective Explorative Interventional Study with an Oral Polymeric Formulation Enriched with Transforming Growth Factor Beta 2 (TGF-β2). Nutrients. 2022; 14(17):3589. https://doi.org/10.3390/nu14173589
Chicago/Turabian StyleMorello, Enrico, Francesco Arena, Michele Malagola, Mirko Farina, Nicola Polverelli, Elsa Cavagna, Federica Colnaghi, Lorenzo Donna, Tatiana Zollner, Eugenia Accorsi Buttini, and et al. 2022. "Malnutrition Prevention after Allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): A Prospective Explorative Interventional Study with an Oral Polymeric Formulation Enriched with Transforming Growth Factor Beta 2 (TGF-β2)" Nutrients 14, no. 17: 3589. https://doi.org/10.3390/nu14173589
APA StyleMorello, E., Arena, F., Malagola, M., Farina, M., Polverelli, N., Cavagna, E., Colnaghi, F., Donna, L., Zollner, T., Accorsi Buttini, E., Andreoli, M., Ricci, C., Leoni, A., Samarani, E., Bertulli, A., Leali, D., Bernardi, S., & Russo, D. (2022). Malnutrition Prevention after Allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): A Prospective Explorative Interventional Study with an Oral Polymeric Formulation Enriched with Transforming Growth Factor Beta 2 (TGF-β2). Nutrients, 14(17), 3589. https://doi.org/10.3390/nu14173589









