Survival of Vaccine-Induced Human Milk SARS-CoV-2 IgG, IgA and SIgA Immunoglobulins across Simulated Human Infant Gastrointestinal Digestion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants and Study Design
2.2. SARS-CoV-2 IgA and IgG ELISA Validation
2.3. Determination of Anti-SARS-CoV-2 IgA, IgG, and SIgA Levels in Breast Milk
2.4. In Vitro Infant GI Digestion of Human Milk
2.5. Statistical Analysis
3. Results
3.1. Subject Characteristics
3.2. SARS-CoV-2 IgG and IgA ELISA Method Validation
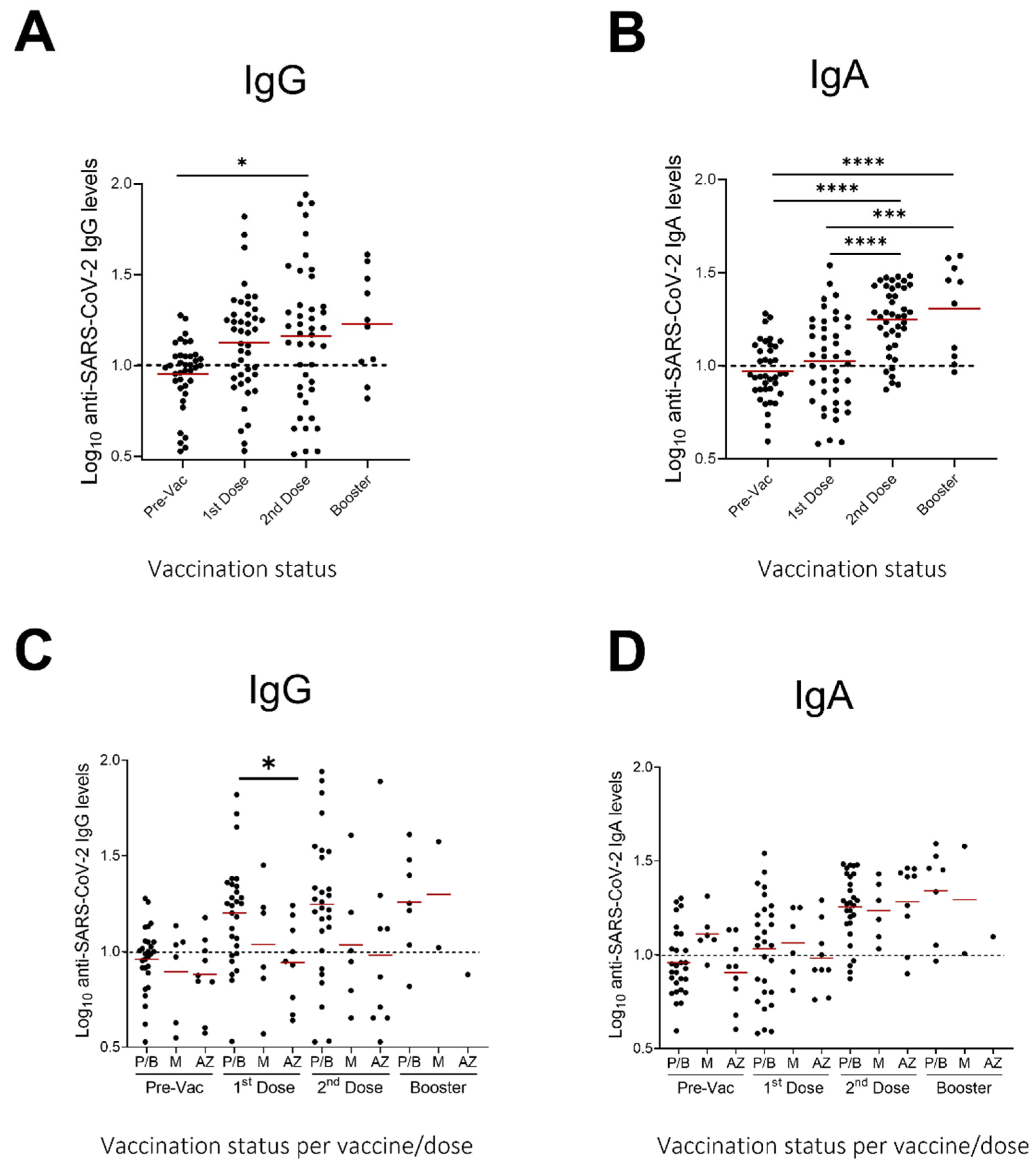
3.3. Evaluation of Anti-SARS-CoV-2 IgA and IgG Levels in Milk Samples after Vaccination
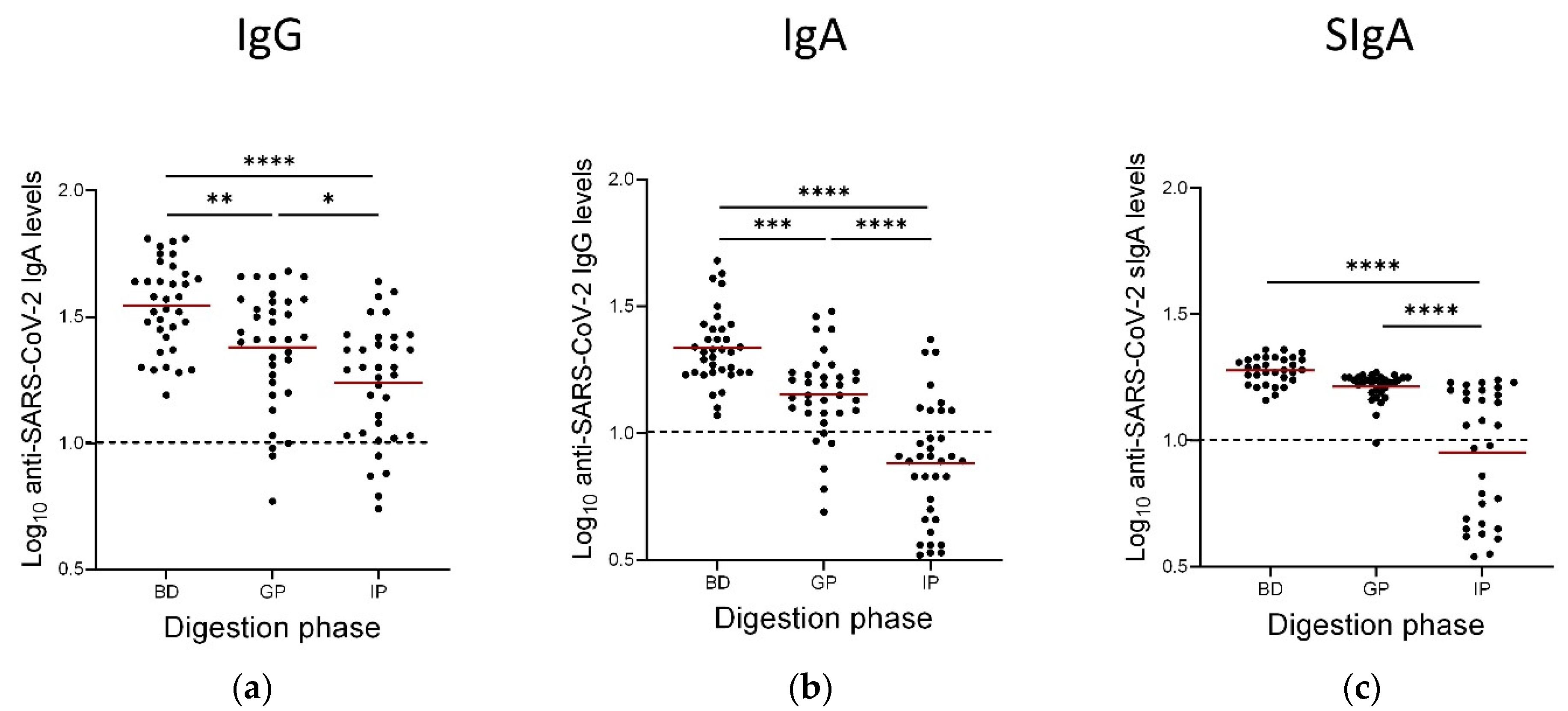
3.4. Persistence of Anti-SARS-CoV-2 IgAs, IgGs and SIgAs Following the Gastric and Intestinal Phases of In Vitro GI Digestion
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharma, A.; Tiwari, S.; Deb, M.K.; Marty, J.L. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2): A global pandemic and treatment strategies. Int. J. Antimicrob. Agents 2020, 56, 106054. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.-P.; Yang, M.; Lai, C.-L. COVID-19 vaccines: A review of the safety and efficacy of current clinical trials. Pharmaceuticals 2021, 14, 406. [Google Scholar] [CrossRef] [PubMed]
- Dufort, E.M.; Koumans, E.H.; Chow, E.J.; Rosenthal, E.M.; Muse, A.; Rowlands, J.; Barranco, M.A.; Maxted, A.M.; Rosenberg, E.S.; Easton, D.; et al. Multisystem inflammatory syndrome in children in New York state. N. Engl. J. Med. 2020, 383, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Abrams, J.Y.; Godfred-Cato, S.E.; Oster, M.E.; Chow, E.J.; Koumans, E.H.; Bryant, B.; Leung, J.W.; Belay, E.D. Multisystem inflammatory syndrome in children associated with severe acute respiratory syndrome coronavirus 2: A systematic review. J. Pediatr. 2020, 226, 45–54.e41. [Google Scholar] [CrossRef]
- Guimaraes, D.; Pissarra, R.; Reis-Melo, A.; Guimaraes, H. Multisystem inflammatory syndrome in children (MISC): A systematic review. Int. J. Clin. Pract. 2021, 75, e14450. [Google Scholar] [CrossRef]
- Hoste, L.; Van Paemel, R.; Haerynck, F. Multisystem inflammatory syndrome in children related to COVID-19: A systematic review. Eur. J. Pediatr. 2021, 180, 2019–2034. [Google Scholar] [CrossRef]
- Lyons, K.E.; Ryan, C.A.; Dempsey, E.M.; Ross, R.P.; Stanton, C. Breast milk, a source of beneficial microbes and associated benefits for infant health. Nutrients 2020, 12, 1039. [Google Scholar] [CrossRef]
- Rio-Aige, K.; Azagra-Boronat, I.; Castell, M.; Selma-Royo, M.; Collado, M.C.; Rodriguez-Lagunas, M.J.; Perez-Cano, F.J. The breast milk immunoglobulinome. Nutrients 2021, 13, 1810. [Google Scholar] [CrossRef]
- Hurley, W.L.; Theil, P.K. Perspectives on immunoglobulins in colostrum and milk. Nutrients 2011, 3, 442–474. [Google Scholar] [CrossRef]
- Palmeira, P.; Carneiro-Sampaio, M. Immunology of breast milk. Rev. Assoc. Med. Bras. (1992) 2016, 62, 584–593. [Google Scholar] [CrossRef]
- Goldman, A.S.; Chheda, S.; Keeney, S.E.; Schmalstieg, F.C. Immunology of human milk and host immunity. In Fetal and Neonatal Physiology, 4th ed.; Polin, R.A., Fox, W.W., Abman, S.H., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2011; pp. 1690–1701. [Google Scholar]
- Robertson, C.A.; Lowther, S.A.; Birch, T.; Tan, C.; Sorhage, F.; Stockman, L.; McDonald, C.; Lingappa, J.R.; Bresnitz, E. SARS and pregnancy: A case report. Emerg. Infect. Dis. 2004, 10, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Fox, A.; Marino, J.; Amanat, F.; Krammer, F.; Hahn-Holbrook, J.; Zolla-Pazner, S.; Powell, R.L. Robust and specific secretory IgA against SARS-CoV-2 detected in human milk. iScience 2020, 23, 101735. [Google Scholar] [CrossRef] [PubMed]
- van Keulen, B.J.; Romijn, M.; Bondt, A.; Dingess, K.A.; Kontopodi, E.; van der Straten, K.; den Boer, M.A.; Bosch, B.J.; Brouwer, P.J.M.; de Groot, C.J.M.; et al. Breastmilk; a source of SARS-CoV-2 specific IgA antibodies. medRxiv 2020. [Google Scholar] [CrossRef]
- Pace, R.M.; Williams, J.E.; Jarvinen, K.M.; Belfort, M.B.; Pace, C.D.W.; Lackey, K.A.; Gogel, A.C.; Nguyen-Contant, P.; Kanagaiah, P.; Fitzgerald, T.; et al. Characterization of SARS-CoV-2 RNA, antibodies, and neutralizing capacity in milk produced by women with COVID-19. mBio 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- van Keulen, B.J.; Romijn, M.; Bondt, A.; Dingess, K.A.; Kontopodi, E.; van der Straten, K.; den Boer, M.A.; Burger, J.A.; Poniman, M.; Bosch, B.J.; et al. Human milk from previously COVID-19-infected mothers: The effect of pasteurization on specific antibodies and neutralization capacity. Nutrients 2021, 13, 1645. [Google Scholar] [CrossRef]
- Baird, J.K.; Jensen, S.M.; Urba, W.J.; Fox, B.A.; Baird, J.R. SARS-CoV-2 antibodies detected in human breast milk post-vaccination. medRxiv 2021. [Google Scholar] [CrossRef]
- Esteve-Palau, E.; Gonzalez-Cuevas, A.; Guerrero, M.E.; Garcia-Terol, C.; Alvarez, M.C.; Garcia-Aranda, G.; Casadevall, D.; Diaz-Brito, V. Quantification of specific antibodies against SARS-CoV-2 in breast milk of lactating women vaccinated with an mRNA vaccine. JAMA Netw. Open 2021, 4, e2120575. [Google Scholar] [CrossRef]
- Fox, A.; Norris, C.; Amanat, F.; Zolla-Pazner, S.; Powell, R.L. The vaccine-elicited immunoglobulin profile in milk after COVID-19 mRNA-based vaccination is IgG-dominant and lacks secretory antibodies. medRxiv 2021. [Google Scholar] [CrossRef]
- Friedman, M.R.; Kigel, A.; Bahar, Y.; Yogev, Y.; Dror, Y.; Lubetzky, R.; Many, A.; Wine, Y. BNT162b2 COVID-19 mRNA vaccine elicits a rapid and synchronized antibody response in blood and milk of breastfeeding women. medRxiv 2021. [Google Scholar] [CrossRef]
- Golan, Y.; Prahl, M.; Cassidy, A.; Wu, A.H.B.; Jigmeddagva, U.; Lin, C.Y.; Gonzalez, V.J.; Basilio, E.; Warrier, L.; Buarpung, S.; et al. Immune response during lactation after anti-SARS-CoV2 mRNA vaccine. medRxiv 2021. [Google Scholar] [CrossRef]
- Bertrand, K.; Honerkamp-Smith, G.; Chambers, C. Maternal and child outcomes reported by breastfeeding women following mRNA COVID-19 vaccination. Breastfeed. Med. 2021, 16, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Selma-Royo, M.; Bauerl, C.; Mena-Tudela, D.; Aguilar-Camprubi, L.; Perez-Cano, F.J.; Parra-Llorca, A.; Lerin, C.; Martinez-Costa, C.; Collado, M.C. Anti-SARS-CoV-2 IgA and IgG in human milk after vaccination is dependent on vaccine type and previous SARS-CoV-2 exposure: A longitudinal study. Genome Med. 2022, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Demers-Mathieu, V.; Huston, R.K.; Markell, A.M.; McCulley, E.A.; Martin, R.L.; Spooner, M.; Dallas, D.C. Differences in maternal immunoglobulins within mother’s own breast milk and donor breast milk and across digestion in preterm infants. Nutrients 2019, 11, 920. [Google Scholar] [CrossRef]
- Calvo-Lerma, J.; Bueno-Llamoga, P.; Bauerl, C.; Cortes-Macias, E.; Selma-Royo, M.; Perez-Cano, F.; Lerin, C.; Martinez-Costa, C.; Collado, M.C. Persistence of anti SARS-CoV-2 antibodies in breast milk from infected and vaccinated women after In vitro-simulated gastrointestinal digestion. Nutrients 2022, 14, 2117. [Google Scholar] [CrossRef]
- Pieri, M.; Nicolaidou, V.; Paphiti, I.; Pipis, S.; Felekkis, K.; Papaneophytou, C. Survival of vaccine-induced human milk SARS-CoV-2 IgG and IgA immunoglobulins across simulated human infant gastrointestinal digestion. medRxiv 2021. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Andreasson, U.; Perret-Liaudet, A.; van Waalwijk van Doorn, L.J.; Blennow, K.; Chiasserini, D.; Engelborghs, S.; Fladby, T.; Genc, S.; Kruse, N.; Kuiperij, H.B.; et al. A practical guide to immunoassay method validation. Front. Neurol. 2015, 6, 179. [Google Scholar] [CrossRef]
- Lueangsakulthai, J.; Sah, B.N.P.; Scottoline, B.P.; Dallas, D.C. Survival of recombinant monoclonal and naturally-occurring human milk immunoglobulins A and G specific to respiratory syncytial virus F protein across simulated human infant gastrointestinal digestion. J. Funct. Foods 2020, 73, 104115. [Google Scholar] [CrossRef]
- Nguyen, T.T.P.; Bhandari, B.; Cichero, J.; Prakash, S. Gastrointestinal digestion of dairy and soy proteins in infant formulas: An in vitro study. Food Res. Int. 2015, 76, 348–358. [Google Scholar] [CrossRef]
- Leung, H.Y.H.; Leung, B.W.; Gil, M.M.; Rolle, V.; Moungmaithong, S.; Wang, C.C.; Poon, L.C. SARS-CoV-2-specific antibodies and neutralization capacity in breast milk following infection vs vaccination. Ultrasound Obstet. Gynecol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Henle, M.A. Increase in SARS-CoV-2 RBD-specific IgA and IgG antibodies in breast milk from lactating women following the COVID-19 booster vaccination. medRxiv 2022. [Google Scholar] [CrossRef]
- Yu, Y.; Li, Y.; Hu, Y.; Li, B.; Xu, J. Breastfed 13 month-old infant of a mother with COVID-19 pneumonia: A case report. Int. Breastfeed. J. 2020, 15, 68. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Chi, X.; Hai, H.; Sun, L.; Zhang, M.; Xie, W.-F.; Chen, W. Antibodies in the breast milk of a maternal woman with COVID-19. Emerg Microbes Infect. 2020, 9, 1467–1469. [Google Scholar] [CrossRef]
- Fox, A.; Marino, J.; Amanat, F.; Krammer, F.; Hahn-Holbrook, J.; Zolla-Pazner, S.; Powell, R.L. Evidence of a significant secretory-IgA-dominant SARS-CoV-2 immune response in human milk following recovery from COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Fox, A.; Marino, J.; Amanat, F.; Oguntuyo, K.Y.; Hahn-Holbrook, J.; Lee, B.; Zolla-Pazner, S.; Powell, R.L. The IgA in milk induced by SARS-CoV-2 infection is comprised of mainly secretory antibody that is neutralizing and highly durable over time. PLoS ONE 2022, 17, e0249723. [Google Scholar] [CrossRef]
- Demers-Mathieu, V.; Underwood, M.A.; Beverly, R.L.; Nielsen, S.D.; Dallas, D.C. Comparison of human milk immunoglobulin survival during gastric digestion between preterm and term infants. Nutrients 2018, 10, 631. [Google Scholar] [CrossRef]
- Eibl, M.M.; Wolf, H.M.; Fürnkranz, H.; Rosenkranz, A. Prevention of necrotizing enterocolitis in low-birth-weight infants by IgA-IgG feeding. N. Engl. J. Med. 1988, 319, 1–7. [Google Scholar] [CrossRef]
- Roos, N.; Mahé, S.; Benamouzig, R.; Sick, H.; Rautureau, J.; Tomé, D. 15N-labeled immunoglobulins from bovine colostrum are partially resistant to digestion in human intestine. The Journal of nutrition 1995, 125, 1238–1244. [Google Scholar] [CrossRef]
- Bakker-Zierikzee, A.M.; Tol, E.A.; Kroes, H.; Alles, M.S.; Kok, F.J.; Bindels, J.G. Faecal SIgA secretion in infants fed on pre- or probiotic infant formula. Pediatr. Allergy Immunol. 2006, 17, 134–140. [Google Scholar] [CrossRef]
- Schanler, R.J.; Goldblum, R.M.; Garza, C.; Goldman, A.S. Enhanced fecal excretion of selected immune factors in very low birth weight infants fed fortified human milk. Pediatric Res. 1986, 20, 711–715. [Google Scholar] [CrossRef] [PubMed]
- Goldblum, R.M.; Schanler, R.J.; Garza, C.; Goldman, A.S. Human milk feeding enhances the urinary excretion of immunologic factors in low birth weight infants. Pediatric Res. 1989, 25, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Jasion, V.S.; Burnett, B.P. Survival and digestibility of orally-administered immunoglobulin preparations containing IgG through the gastrointestinal tract in humans. Nutr. J. 2015, 14, 22. [Google Scholar] [CrossRef]
- Wilks, A.B.; Christian, E.C.; Seaman, M.S.; Sircar, P.; Carville, A.; Gomez, C.E.; Esteban, M.; Pantaleo, G.; Barouch, D.H.; Letvin, N.L.; et al. Robust vaccine-elicited cellular immune responses in breast milk following systemic simian immunodeficiency virus DNA prime and live virus vector boost vaccination of lactating rhesus monkeys. J. Immunol. 2010, 185, 7097–7106. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Fox, A.; DeCarlo, C.; Norris, C.; Griffin, S.; Wedekind, S.; Flanagan, J.M.; Shenker, N.; Powell, R.L. Comparative profiles of SARS-CoV-2 spike-specific human milk antibodies elicited by mRNA- and adenovirus-based COVID-19 vaccines. Breastfeed. Med. 2022, 17, 638–646. [Google Scholar] [CrossRef]
- Rosenberg-Friedman, M.; Kigel, A.; Bahar, Y.; Werbner, M.; Alter, J.; Yogev, Y.; Dror, Y.; Lubetzky, R.; Dessau, M.; Gal-Tanamy, M.; et al. BNT162b2 mRNA vaccine elicited antibody response in blood and milk of breastfeeding women. Nat. Commun. 2021, 12, 6222. [Google Scholar] [CrossRef]
- Czosnykowska-Łukacka, M.; Lis-Kuberka, J.; Królak-Olejnik, B.; Orczyk-Pawiłowicz, M. Changes in human milk immunoglobulin profile during prolonged lactation. Front. Pediatr. 2020, 8, 428. [Google Scholar] [CrossRef]
- Juncker, H.G.; Mulleners, S.J.; Ruhe, E.J.M.; Coenen, E.R.M.; Bakker, S.; van Doesburg, M.; Harinck, J.E.; Rood, R.D.; Bouhuijs, J.H.; Oomen, M.; et al. Comparing the human milk antibody response after vaccination with four COVID-19 vaccines: A prospective, longitudinal cohort study in The Netherlands. EClinicalMedicine 2022, 47, 101393. [Google Scholar] [CrossRef]

| Vaccine | BioNtech/Pfizer n = 28 | Moderna n = 6 | AstraZeneca n = 9 | Total n = 43 |
|---|---|---|---|---|
| Age (years) | 36 ± 3.7 | 37 ± 2.3 | 37 ± 3.1 | 36 ± 3.4 |
| Maternal co-morbidities | ||||
| Chronic hypertension | 0% (0 of 28) | 0% (0 of 6) | 0% (0 of 9) | 0% (0 of 43) |
| Diabetes/gestational Diabetes | 0% (0 of 28) | 0% (0 of 6) | 0% (0 of 9) | 0% (0 of 43) |
| BMI (kg/m2) > 30 | 3.6% (1 of 28) | 0% (0 of 6) | 0% (0 of 9) | 2.3% (1 of 43) |
| Asthma | 2% (0 of 28) | 0% (0 of 6) | 1% (0 of 9) | 7.0% (3 of 43) |
| Immunosuppression/cancer | 10.7% (3 of 28) | 0% (0 of 6) | 22.2% (2 of 9) | 11.6% (5 of 43) |
| Gestational age (weeks) | 84 ± 47.9 1 | 82 ± 35.9 | 81 ± 19.7 | 83 ± 40.3 |
| Timing between two doses (days) | 21 | 28 | 28–84 | |
| Timing of booster dose (months after 2nd dose) | 6 | 6 | 6 | |
| Participants that provided milk after booster dose | 7 | 2 | 1 | 10 |
| Parameter | IgA | IgG | sIgA |
|---|---|---|---|
| Error (%) | 5.17 | 18.25 | 6.21 |
| Precision (% CV) | 8.51 | 16.73 | 10.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pieri, M.; Maniori, M.-A.; Shahabian, L.; Kanaan, E.; Paphiti-Demetriou, I.; Pipis, S.; Felekkis, K.; Nicolaidou, V.; Papaneophytou, C. Survival of Vaccine-Induced Human Milk SARS-CoV-2 IgG, IgA and SIgA Immunoglobulins across Simulated Human Infant Gastrointestinal Digestion. Nutrients 2022, 14, 3368. https://doi.org/10.3390/nu14163368
Pieri M, Maniori M-A, Shahabian L, Kanaan E, Paphiti-Demetriou I, Pipis S, Felekkis K, Nicolaidou V, Papaneophytou C. Survival of Vaccine-Induced Human Milk SARS-CoV-2 IgG, IgA and SIgA Immunoglobulins across Simulated Human Infant Gastrointestinal Digestion. Nutrients. 2022; 14(16):3368. https://doi.org/10.3390/nu14163368
Chicago/Turabian StylePieri, Myrtani, Maria-Arsenia Maniori, Lucy Shahabian, Elie Kanaan, Irene Paphiti-Demetriou, Spyros Pipis, Kyriakos Felekkis, Vicky Nicolaidou, and Christos Papaneophytou. 2022. "Survival of Vaccine-Induced Human Milk SARS-CoV-2 IgG, IgA and SIgA Immunoglobulins across Simulated Human Infant Gastrointestinal Digestion" Nutrients 14, no. 16: 3368. https://doi.org/10.3390/nu14163368
APA StylePieri, M., Maniori, M.-A., Shahabian, L., Kanaan, E., Paphiti-Demetriou, I., Pipis, S., Felekkis, K., Nicolaidou, V., & Papaneophytou, C. (2022). Survival of Vaccine-Induced Human Milk SARS-CoV-2 IgG, IgA and SIgA Immunoglobulins across Simulated Human Infant Gastrointestinal Digestion. Nutrients, 14(16), 3368. https://doi.org/10.3390/nu14163368










