Effect of Fructooligosaccharides Supplementation on the Gut Microbiota in Human: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Method
2.1. Eligibility and Search Strategy
2.2. Selection Criteria and Quality Assessment
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
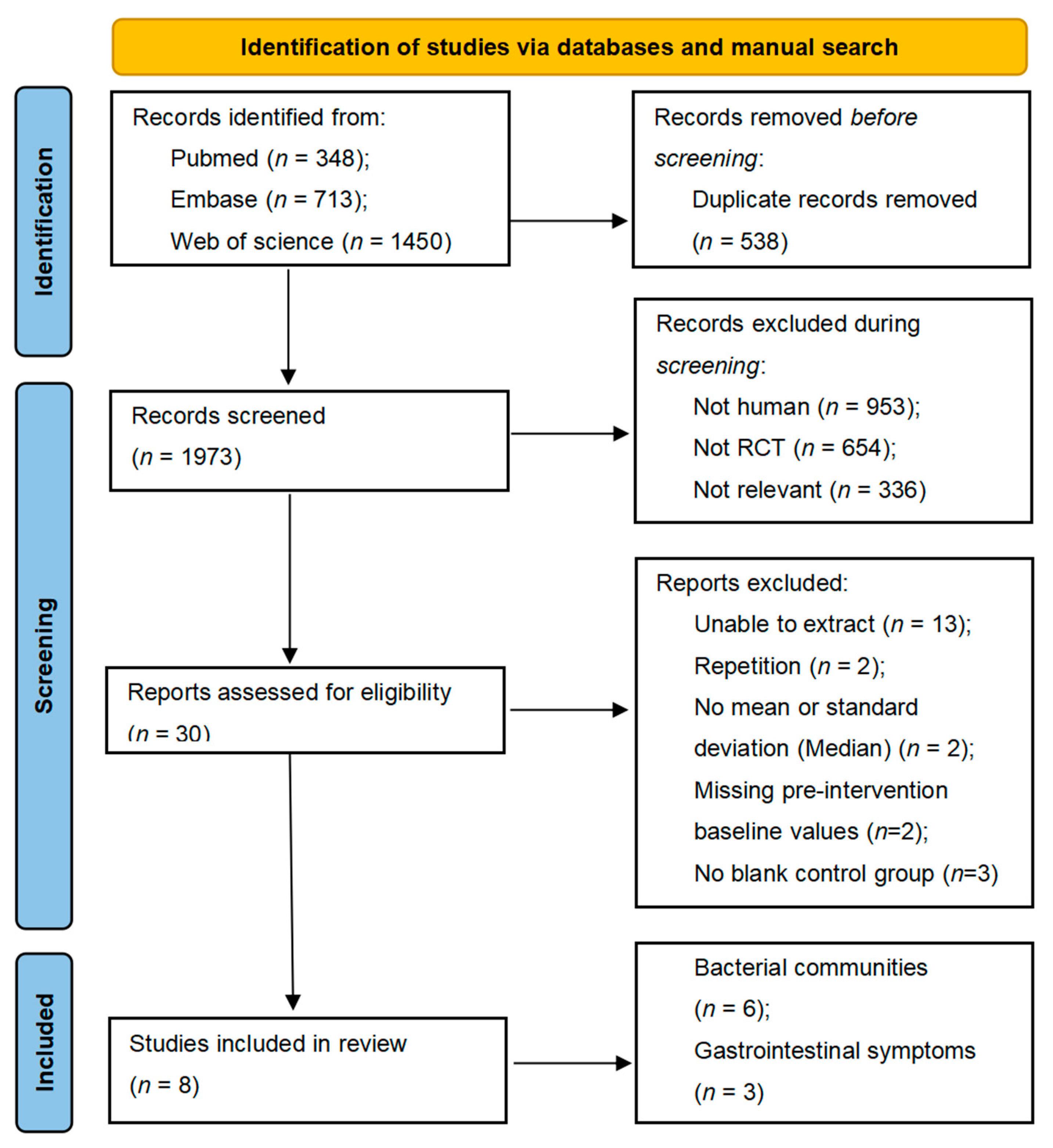
3.1. Literature Search and Study Characteristics
3.2. Changes in Gut Microbiota before and after Supplementation
3.3. Adverse Gastrointestinal Reactions before and after Supplementation
3.4. Meta-Regression
3.5. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Collins, S.; Reid, G. Distant Site Effects of Ingested Prebiotics. Nutrients 2016, 8, 523. [Google Scholar] [CrossRef]
- Scott, K.P.; Antoine, J.; Midtvedt, T.; van Hemert, S. Manipulating the gut microbiota to maintain health and treat disease. Microb. Ecol. Health Dis. 2015, 26, 25877. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Milani, C.; de Giori, G.S.; Sesma, F.; van Sinderen, D.; Ventura, M. Bacteria as vitamin suppliers to their host: A gut microbiota perspective. Curr. Opin. Biotechnol. 2013, 24, 160–168. [Google Scholar] [CrossRef]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.M.; Finlay, B.B. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef]
- Gibson, G.R.; Beatty, E.R.; Wang, X.; Cummings, J.H. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Tojo, R.; Suarez, A.; Clemente, M.G.; de Los Reyes-Gavilan, C.G.; Margolles, A.; Gueimonde, M.; Ruas-Madiedo, P. Intestinal microbiota in health and disease: Role of bifidobacteria in gut homeostasis. World J. Gastroenterol. 2014, 20, 15163–15176. [Google Scholar] [CrossRef]
- Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The Impact of the Gut Microbiota on Human Health: An Integrative View. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef]
- Closa-Monasterolo, R.; Gispert-Llaurado, M.; Luque, V.; Ferre, N.; Rubio-Torrents, C.; Zaragoza-Jordana, M.; Escribano, J. Safety and efficacy of inulin and oligofructose supplementation in infant formula: Results from a randomized clinical trial. Clin. Nutr. 2013, 32, 918–927. [Google Scholar] [CrossRef]
- Fuhren, J.; Schwalbe, M.; Boekhorst, J.; Rosch, C.; Schols, H.A.; Kleerebezem, M. Dietary calcium phosphate strongly impacts gut microbiome changes elicited by inulin and galacto-oligosaccharides consumption. Microbiome 2021, 9, 218. [Google Scholar] [CrossRef]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef]
- Korczak, R.; Slavin, J.L. Fructooligosaccharides and appetite. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 377–380. [Google Scholar] [CrossRef]
- Caetano, B.F.; de Moura, N.A.; Almeida, A.P.; Dias, M.C.; Sivieri, K.; Barbisan, L.F. Yacon (Smallanthus sonchifolius) as a Food Supplement: Health-Promoting Benefits of Fructooligosaccharides. Nutrients 2016, 8, 436. [Google Scholar] [CrossRef]
- Costa, G.T.; Vasconcelos, Q.; Aragao, G.F. Fructooligosaccharides on inflammation, immunomodulation, oxidative stress, and gut immune response: A systematic review. Nutr. Rev. 2022, 80, 709–722. [Google Scholar] [CrossRef]
- Adak, A.; Khan, M.R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci. 2019, 76, 473–493. [Google Scholar] [CrossRef]
- Wernimont, S.; Northington, R.; Kullen, M.J.; Yao, M.; Bettler, J. Effect of an α-lactalbumin-enriched infant formula supplemented with oligofructose on fecal microbiota, stool characteristics, and hydration status: A randomized, double-blind, controlled trial. Clin. Pediatr. 2015, 54, 359–370. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Vahedi, K.; Achour, L.; Attar, A.; Salfati, J.; Pochart, P.; Marteau, P.; Flourie, B.; Bornet, F.; Rambaud, J.C. Short-chain fructo-oligosaccharide administration dose-dependently increases fecal bifidobacteria in healthy humans. J. Nutr. 1999, 129, 113–116. [Google Scholar] [CrossRef]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef]
- Wang, X.; Gibson, G.R. Effects of the in vitro fermentation of oligofructose and inulin by bacteria growing in the human large intestine. J. Appl. Bacteriol. 1993, 75, 373–380. [Google Scholar] [CrossRef]
- Euler, A.R.; Mitchell, D.K.; Kline, R.; Pickering, L.K. Prebiotic effect of fructo-oligosaccharide supplemented term infant formula at two concentrations compared with unsupplemented formula and human milk. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 157–164. [Google Scholar] [CrossRef]
- Souza, D.D.S.; Tahan, S.; Weber, T.K.; de Araujo-Filho, H.B.; de Morais, M.B. Randomized, Double-Blind, Placebo-Controlled Parallel Clinical Trial Assessing the Effect of Fructooligosaccharides in Infants with Constipation. Nutrients 2018, 10, 1602. [Google Scholar] [CrossRef]
- Xia, Q.; Williams, T.; Hustead, D.; Price, P.; Morrison, M.; Yu, Z. Quantitative Analysis of Intestinal Bacterial Populations from Term Infants Fed Formula Supplemented with Fructo-oligosaccharides. J. Pediatr. Gastrenterol. Nutr. 2012, 55, 314–320. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Flourié, B.; Riottot, M.; Bisetti, N.; Gailing, M.F.; Guibert, A.; Bornet, F.; Rambaud, J.C. Effects of fructo-oligosaccharides ingestion on fecal bifidobacteria and selected metabolic indexes of colon carcinogenesis in healthy humans. Nutr. Cancer 1996, 26, 21–29. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Vicaut, E.; Neut, C.; Flourié, B.; Brouns, F.; Bornet, F.R. The capacity of nondigestible carbohydrates to stimulate fecal bifidobacteria in healthy humans: A double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am. J. Clin. Nutr. 2004, 80, 1658–1664. [Google Scholar] [CrossRef]
- Tandon, D.; Haque, M.M.; Gote, M.; Jain, M.; Bhaduri, A.; Dubey, A.K.; Mande, S.S. A prospective randomized, double-blind, placebo-controlled, dose-response relationship study to investigate efficacy of fructo-oligosaccharides (FOS) on human gut microflora. Sci. Rep. 2019, 9, 5473. [Google Scholar] [CrossRef]
- Ten, B.S.; Bovee-Oudenhoven, I.M.; Lettink-Wissink, M.L.; Katan, M.B.; van der Meer, R. Dietary fructooligosaccharides affect intestinal barrier function in healthy men. J. Nutr. 2006, 136, 70–74. [Google Scholar]
- Azpiroz, F.; Dubray, C.; Bernalier-Donadille, A.; Cardot, J.M.; Accarino, A.; Serra, J.; Wagner, A.; Respondek, F.; Dapoigny, M. Effects of scFOS on the composition of fecal microbiota and anxiety in patients with irritable bowel syndrome: A randomized, double blind, placebo controlled study. Neurogastroenterol. Motil. 2017, 29, e12911. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Paineau, D.; Bornet, F. The capacity of short-chain fructo-oligosaccharides to stimulate faecal bifidobacteria: A dose-response relationship study in healthy humans. Nutr. J. 2006, 5, 8. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Kapiki, A.; Costalos, C.; Oikonomidou, C.; Triantafyllidou, A.; Loukatou, E.; Pertrohilou, V. The effect of a fructo-oligosaccharide supplemented formula on gut flora of preterm infants. Early Hum. Dev. 2007, 83, 335–339. [Google Scholar] [CrossRef]
- Whelan, K.; Judd, P.A.; Preedy, V.R.; Simmering, R.; Jann, A.; Taylor, M.A. Fructooligosaccharides and fiber partially prevent the alterations in fecal microbiota and short-chain fatty acid concentrations caused by standard enteral formula in healthy humans. J. Nutr. 2005, 135, 1896–1902. [Google Scholar] [CrossRef]
- Buddington, R.K.; Kapadia, C.; Neumer, F.; Theis, S. Oligofructose Provides Laxation for Irregularity Associated with Low Fiber Intake. Nutrients 2017, 9, 1372. [Google Scholar] [CrossRef]
- Reimer, R.A.; Soto-Vaca, A.; Nicolucci, A.C.; Mayengbam, S.; Park, H.; Madsen, K.L.; Menon, R.; Vaughan, E.E. Effect of chicory inulin-type fructan-containing snack bars on the human gut microbiota in low dietary fiber consumers in a randomized crossover trial. Am. J. Clin. Nutr. 2020, 111, 1286–1296. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Luo, Y.; Xiao, Y.; Zhao, J.; Zhang, H.; Chen, W.; Zhai, Q. The role of mucin and oligosaccharides via cross-feeding activities by Bifidobacterium: A review. Int. J. Biol. Macromol. 2021, 167, 1329–1337. [Google Scholar] [CrossRef]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y.; et al. Fructooligosaccharide (FOS) and Galactooligosaccharide (GOS) Increase Bifidobacterium but Reduce Butyrate Producing Bacteria with Adverse Glycemic Metabolism in healthy young population. Sci. Rep. 2017, 7, 11789. [Google Scholar] [CrossRef]
- Van der Meulen, R.; Adriany, T.; Verbrugghe, K.; De Vuyst, L. Kinetic analysis of bifidobacterial metabolism reveals a minor role for succinic acid in the regeneration of NAD(+) through its growth-associated production. Appl. Environ. Microb. 2006, 72, 5204–5210. [Google Scholar] [CrossRef]
- Gaenzle, M.G.; Follador, R. Metabolism of oligosaccharides and starch in lactobacilli: A review. Front. Microbiol. 2012, 3, 340. [Google Scholar] [CrossRef]
- Langlands, S.J.; Hopkins, M.J.; Coleman, N.; Cummings, J.H. Prebiotic carbohydrates modify the mucosa associated microflora of the human large bowel. Gut 2004, 53, 1610–1616. [Google Scholar] [CrossRef]
- Kelly, S.M.; Munoz-Munoz, J.; van Sinderen, D. Plant Glycan Metabolism by Bifidobacteria. Front. Microbiol. 2021, 12, 609418. [Google Scholar] [CrossRef] [PubMed]
- Lawson, M.; O’Neill, I.J.; Kujawska, M.; Gowrinadh, J.S.; Wijeyesekera, A.; Flegg, Z.; Chalklen, L.; Hall, L.J. Breast milk-derived human milk oligosaccharides promote Bifidobacterium interactions within a single ecosystem. ISME J. 2020, 14, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Louis, P.; Flint, H.J. Cultivable bacterial diversity from the human colon. Lett. Appl. Microbiol. 2007, 44, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Wexler, H.M. Bacteroides: The good, the bad, and the nitty-gritty. Clin. Microbiol. Rev. 2007, 20, 593–621. [Google Scholar] [CrossRef]
- Dempsey, M. Microbiology: An Evolving Science. Brit. J. Biomed. Sci. 2011, 68, 51–52. [Google Scholar] [CrossRef]
- Yusuf, E. A Book Review on: Sherris Medical Microbiology—International Edition, 6th Edition. Front. Cell. Infect. Microbiol. 2015, 5, 34. [Google Scholar] [CrossRef]
- Michiko, M.N.; Peter, Z. Strict and Facultative Anaerobes:Medical and Environmental Aspects; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Le Bastard, Q.; Chapelet, G.; Javaudin, F.; Lepelletier, D.; Batard, E.; Montassier, E. The effects of inulin on gut microbial composition: A systematic review of evidence from human studies. Eur. J. Clin. Microbiol. 2020, 39, 403–413. [Google Scholar] [CrossRef]
- McClean, K.L.; Sheehan, G.J.; Harding, G.K. Intraabdominal infection: A review. Clin. Infect. Dis. 1994, 19, 100–116. [Google Scholar] [CrossRef]
- Rumessen, J.J.; Bode, S.; Hamberg, O.; Gudmand-Hoyer, E. Fructans of Jerusalem artichokes: Intestinal transport, absorption, fermentation, and influence on blood glucose, insulin, and C-peptide responses in healthy subjects. Am. J. Clin. Nutr. 1990, 52, 675–681. [Google Scholar] [CrossRef]
- Roberfroid, M.; Slavin, J. Nondigestible oligosaccharides. Crit. Rev. Food Sci. Nutr. 2000, 40, 461–480. [Google Scholar] [CrossRef] [PubMed]


| Study | Participants (N) | Interventions | RCT Design | Index of Analysis | |||||
|---|---|---|---|---|---|---|---|---|---|
| Compound Studied and Dose | Comparator | Design | Duration (d) | Run-in | Washout | Analysis (Microbiota) | |||
| Azpiroz, 2017 [28] | France; IBS patients (34 scFOS group and 34 Placebo group); 18–60 years age | scFOS (Actilight 950P; Beghin_x0002_Meiji, Marckolsheim, France), 5 g per day | Maltodextrins (Maldex 120; Tereos Syral, Marckolsheim, France) | Parallel | 28 | × | × | real-time PCR; 16S rDNA | Intestinal flora |
| Bouhnik, 1996 [24] | France; Healthy volunteers (10 Fructo-Oligosaccharides and 10 Placebo); 22–39 years age | FOS (Actilight™, Eridania-Beghin Say, Paris, France), 12.5 g/day | Saccharose | Parallel | 36 | × | × | Culture | Intestinal flora |
| Bouhnik, 2006 [29] | France; Healthy volunteers (32 FOS and 8 placebo); 29 ± 1.3 years age | scFOS (Actilight™, Beghin Meiji, Paris, France); 2.5, 5.0, 7.5 or 10 g/d. | 50% sucrose—50% fully digestible waxy maize-derived maltodextrins (DE6.5) (Cerestar, Vil_x0002_voorde, Belgium) | Parallel | 7 | √ | × | Culture | Intestinal flora, gastrointestinal symptoms |
| Buddington, 2017 [34] | USA; Adults (49 OF and 48 placebo) having a body mass index ≤ 35; age: from 18 to 65 years | OF (Orafti® P95 Oligofructose), 5 g/day, 10 g/day and 15 g/day | Maltodextrin | Parallel | 28 | √ | × | / | Gastrointestinal symptoms |
| Gibson, 1995 [7] | UK; Eight healthy volunteers with a mean body mass index of 22.4; age: 21–48 years | Oligofructose (Orafti, Tienen, Belgium), 15 g/day | Sucrose | Crossover | 15 | × | √ | Culture | Intestinal flora |
| Kapiki, 2007 [32] | Greece; Preterm infants (36 FOS and 20 placebo) with a maximum gestational age of 36 weeks | FOS, 0.4 g/100 mL | Maltodextrins | Parallel | 7 | × | × | Culture | Intestinal flora |
| Reimer, 2020 [35] | Canada; Healthy adults without obesity (BMI: 18.5–29.9) (11 Moderate dose ITF and 14 placebo or 11 Low dose ITF and 12 placebo); age: 18–65 years | Trial 1—Moderate Dose ITF snack bar, 7 g/d Trial 2—Low Dose ITF snack bar, 3 g/d | Control 1 snack bar Control 2 snack bar | Crossover | 28 | √ | × | q-PCR; 16S rRNA | Gastrointestinal symptoms |
| Wernimont, 2015 [17] | USA; Infants (20 CF and 19 EF); 11.2 ± 2.3 | OF (Orafti® P95, Tienen, Belgium), 3.0 g/L | α-lactalbumin-enriched control formula (CF) | Parallel | 56 | × | × | FISH | Intestinal flora |
| Whelan, 2005 [33] | UK; Healthy men and women (n = 10) between 21 and 34 y old | FOS (Nutren fiber, Nestle’ Switzerland), 9.5 ± 1.5 g/d | Standard (FOS and fiber-free) enteral formula (Nutren 1.0, Nestle´ Switzerland) | Crossover | 28 | × | √ | FISH | Intestinal flora |
| Gut Microbiota | Subgroup | WMD (95% CI) | Heterogeneity | ||
|---|---|---|---|---|---|
| I2 | p Value | ||||
| Bifidobacterium spp. | dose | ≤5 g | 0.52 (0.38, 0.66 ) | 0 | 0.522 |
| >5 g | 1.12 (0.69, 1.55) | 9.3% | 0.347 | ||
| duration | ≤4 weeks | 0.53 (0.37, 0.69) | 3.8% | 0.374 | |
| >4 weeks | 0.84 (0.44, 1.25) | 55.% | 0.062 | ||
| intervention group | adult | 0.86 (0.61, 1.11) | 0 | 0.460 | |
| infant | 0.46 (0.30, 0.62) | 0 | 0.615 | ||
| Lactobacillus spp. | dose | ≤5 g | −0.01 (−0.35, 0.33) | 0 | 0.550 |
| >5 g | −0.49 (−1.38, 0.41) | 0 | 0.784 | ||
| Enterobacteriaceae | dose | ≤5 g | 0.21 (−0.02, 0.45) | 0 | 0.923 |
| >5 g | 0.26 (−0.38, 0.90) | 0 | 0.786 | ||
| Bacteroides | dose | ≤5 g | 0.40 (0.24, 0.56) | 44.4% | 0.145 |
| >5 g | 0.14 (−0.07, 0.36) | 40.6% | 0.186 | ||
| duration | ≤4 weeks | 0.31 (−0.09, 0.70) | 86% | 0.008 | |
| >4 weeks | 0.19 (−0.10, 0.47) | 5.1% | 0.377 | ||
| intervention group | adult | 0.17 (−0.04, 0.38) | 0 | 0.408 | |
| infant | 0.29 (−0.17, 0.75) | 81.4 | 0.020 | ||
| Bacteroides after removing the study of Kapiki et al. | dose | ≤5 g | 0.12 (−0.19, 0.43) | 0 | 0.592 |
| >5 g | 0.14 (−0.07, 0.36) | 40.6% | 0.186 | ||
| Total anaerobes | dose | ≤5 g | 0.25 (−0.13, 0.64) | 0 | 0.902 |
| >5 g | 0.23 (−0.17, 0.63) | 64.1% | 0.062 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dou, Y.; Yu, X.; Luo, Y.; Chen, B.; Ma, D.; Zhu, J. Effect of Fructooligosaccharides Supplementation on the Gut Microbiota in Human: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 3298. https://doi.org/10.3390/nu14163298
Dou Y, Yu X, Luo Y, Chen B, Ma D, Zhu J. Effect of Fructooligosaccharides Supplementation on the Gut Microbiota in Human: A Systematic Review and Meta-Analysis. Nutrients. 2022; 14(16):3298. https://doi.org/10.3390/nu14163298
Chicago/Turabian StyleDou, Yuqi, Xue Yu, Yuanli Luo, Botian Chen, Defu Ma, and Jing Zhu. 2022. "Effect of Fructooligosaccharides Supplementation on the Gut Microbiota in Human: A Systematic Review and Meta-Analysis" Nutrients 14, no. 16: 3298. https://doi.org/10.3390/nu14163298
APA StyleDou, Y., Yu, X., Luo, Y., Chen, B., Ma, D., & Zhu, J. (2022). Effect of Fructooligosaccharides Supplementation on the Gut Microbiota in Human: A Systematic Review and Meta-Analysis. Nutrients, 14(16), 3298. https://doi.org/10.3390/nu14163298








