Vitamin D Status and Mortality from SARS CoV-2: A Prospective Study of Unvaccinated Caucasian Adults
Abstract
:1. Introduction
2. Study Design and Methods
3. Statistical Analysis
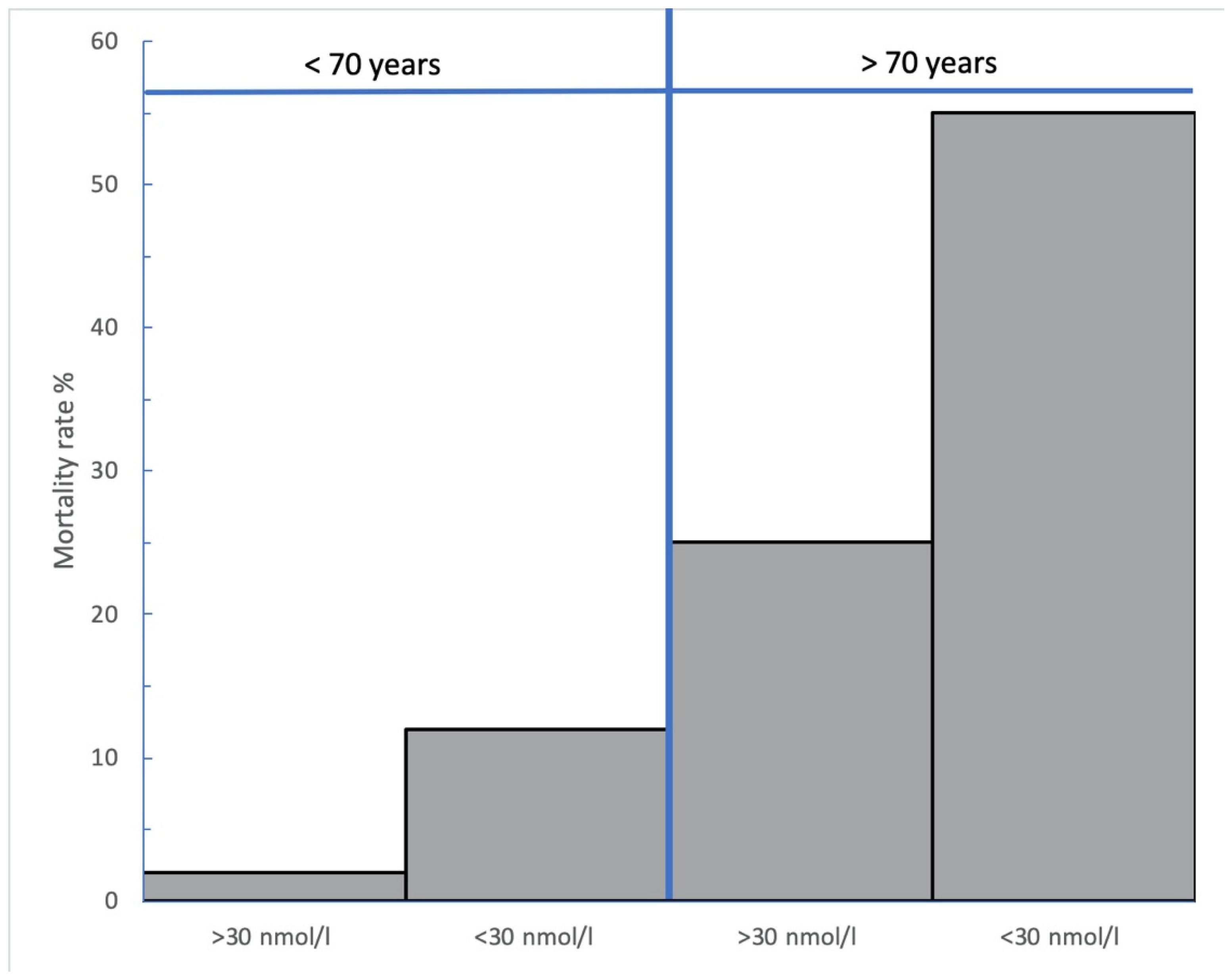
4. Results
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| 25(OH)D | 25-Hydroxyvitamin Vitamin D |
| ARDS | Acute Respiratory Distress Syndrome |
| BMI | Body Mass Index |
| COVID-19 | Coronavirus Disease 2019 |
| CRP | C-Reactive Protein |
| HR | Hazard Ratio |
| ICU | Intensive Care Unit |
| n | Total number of cases |
| O2 | Oxygen |
| OR | Odds Ratio |
| PaO2 RXR | Partial Pressure of Oxygen Retinoid X Receptor |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus-2 |
| WBC | White Blood Cell |
| μg | Microgram |
References
- Israel, A.; Cicurel, A.; Feldhamer, I.; Stern, F.; Dror, Y.; Giveon, S.M.; Gillis, D.; Strich, D.; Lavie, G. Vitamin D deficiency is associated with higher risks for SARS-CoV-2 infection and COVID-19 severity: A retrospective case-control study. Intern. Emerg. Med. 2022, 17, 1053–1063. [Google Scholar] [CrossRef] [PubMed]
- De Smet, D.; De Smet, K.; Herroelen, P.; Gryspeerdt, S.; Martens, G.A. Serum 25(OH)D Level on Hospital Admission Associated With COVID-19 Stage and Mortality. Am. J. Clin. Pathol. 2021, 155, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Van den Abbeele, K.; Mandal, A.K. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med. J. 2021, 97, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef]
- Campi, I.; Gennari, L.; Merlotti, D.; Mingiano, C.; Frosali, A.; Giovanelli, L.; Torlasco, C.; Pengo, M.F.; Heilbron, F.; Soranna, D.; et al. Vitamin D and COVID-19 severity and related mortality: A prospective study in Italy. BMC Infect. Dis. 2021, 21, 566. [Google Scholar] [CrossRef]
- Sunnetcioglu, A.; Sunnetcioglu, M.; Gurbuz, E.; Bedirhanoglu, S.; Erginoguz, A.; Celik, S. Serum 25(OH)D Deficiency and High D-Dimer Levels are Associated with COVID-19 Pneumonia. Clin Lab. 2021, 67. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Griffin, G.; Kenny, R.A. Perspective: Vitamin D deficiency and COVID-19 severity—plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J. Intern. Med. 2021, 289, 97–115. [Google Scholar] [CrossRef]
- Angelidi, A.M.; Belanger, M.J.; Lorinsky, M.K.; Karamanis, D.; Chamorro-Pareja, N.; Ognibene, J.; Palaiodimos, L. Vitamin D Status Is Associated With In-Hospital Mortality and Mechanical Ventilation: A Cohort of COVID-19 Hospitalized Patients. Mayo Clin. Proc. 2021, 96, 875–886. [Google Scholar] [CrossRef]
- Akbar, M.R.; Wibowo, A.; Pranata, R.; Setiabudiawan, B. Low Serum 25-hydroxyvitamin D (Vitamin D) Level Is Associated With Susceptibility to COVID-19, Severity, and Mortality: A Systematic Review and Meta-Analysis. Front. Nutr. 2021, 8, 660420. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Skversky, A.L.; Kumar, J.; Abramowitz, M.K.; Kaskel, F.J.; Melamed, M.L. Association of glucocorticoid use and low 25-hydroxyvitamin D levels: Results from the National Health and Nutrition Examination Survey (NHANES): 2001–2006. J. Clin. Endocrinol. Metab. 2011, 96, 3838–3845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smolders, J.; van den Ouweland, J.; Geven, C.; Pickkers, P.; Kox, M. Letter to the Editor: Vitamin D deficiency in COVID-19: Mixing up cause and consequence. Metabolism 2021, 115, 154434. [Google Scholar] [CrossRef] [PubMed]
- Faul, J.L.; Kerley, C.P.; Love, B.; O’Neill, E.; Cody, C.; Tormey, W.; Hutchinson, K.; Cormican, L.W. Vitamin D Deficiency and ARDS after SARS-CoV-2 Infection. Ir. Med. J. 2020, 113, 84. [Google Scholar] [PubMed]
- Cashman, K.D.; Muldowney, S.; McNulty, B.; Nugent, A.; FitzGerald, A.P.; Kiely, M.; Walton, J.; Gibney, M.J.; Flynn, A. Vitamin D status of Irish adults: Findings from the National Adult Nutrition Survey. Br. J. Nutr. 2013, 109, 1248–1256. [Google Scholar] [CrossRef] [Green Version]
- Bian, P.; Jin, X.; Shou, Z. Effects of Monthly Intramuscular High-Dose Vitamin D2 on Serum 25-Hydroxyvitamin D and Immune Parameters in Very Elderly Chinese Patients with Vitamin D Deficiency. Int. J. Endocrinol. 2021, 2021, 1343913. [Google Scholar] [CrossRef]
- Benskin, L. A Basic Review of the Preliminary Evidence That COVID-19 Risk and Severity Is Increased in Vitamin D Deficiency. Front. Public Health 2020, 8, 513. [Google Scholar] [CrossRef]
- Vassiliou, A.G.; Jahaj, E.; Pratikaki, M.; Orfanos, S.E.; Dimopoulou, I.; Kotanidou, A. Low 25-Hydroxyvitamin D Levels on Admission to the Intensive Care Unit May Predispose COVID-19 Pneumonia Patients to a Higher 28-Day Mortality Risk: A Pilot Study on a Greek ICU Cohort. Nutrients 2020, 12, 3773. [Google Scholar] [CrossRef]
- The RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- Efird, J.T.; Anderson, E.J.; Jindal, C.; Redding, T.S.; Thompson, A.D.; Press, A.M.; Upchurch, J.; Williams, C.D.; Choi, Y.M.; Suzuki, A. The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2. Int. J. Environ. Res. Public Health 2021, 19, 447. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, J.X. Administration with corticosteroid relieving pain following total knee arthroplasty: A meta-analysis. Medicine 2020, 99, e23567. [Google Scholar] [CrossRef]
- Reid, I.R.; Bolland, M.J.; Grey, A. Effects of vitamin D supplements on bone mineral density: A systematic review and meta-analysis. Lancet 2014, 383, 146–155. [Google Scholar] [CrossRef]
- Black, L.J.; Walton, J.; Flynn, A.; Cashman, K.D.; Kiely, M. Small Increments in Vitamin D Intake by Irish Adults over a Decade Show That Strategic Initiatives to Fortify the Food Supply Are Needed. J. Nutr. 2015, 145, 969–976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romero-Ortuno, R.; Cogan, L.; Browne, J.; Healy, M.; Casey, M.C.; Cunningham, C.; Walsh, J.B.; Kenny, R.A. Seasonal variation of serum vitamin D and the effect of vitamin D supplementation in Irish community-dwelling older people. Age Ageing 2011, 40, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Griffin, T.P.; Wall, D.; Blake, L.; Griffin, D.G.; Robinson, S.; Bell, M.; Mulkerrin, E.C.; O’Shea, P.M. Higher risk of vitamin D insufficiency/deficiency for rural than urban dwellers. J. Steroid. Biochem. Mol. Biol. 2020, 197, 105547. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; O’Halloran, A.M.; Carey, D.; Healy, M.; O’Connor, D.; Moore, P.; Shannon, T.; Molloy, A.M. The prevalence of vitamin D deficiency and the determinants of 25(OH)D concentration in older Irish adults: Data from the Irish Longitudinal Study on Ageing (TILDA). J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubée, V.; Legrand, E.; Sacco, G. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef]
- Kazemi, A.; Mohammadi, V.; Aghababaee, S.K.; Golzarand, M.; Clark, C.C.T.; Babajafari, S. Association of Vitamin D Status with SARS-CoV-2 Infection or COVID-19 Severity: A Systematic Review and Meta-analysis. Adv. Nutr. 2021, 12, 1636–1658. [Google Scholar] [CrossRef]
- Petrelli, F.; Luciani, A.; Perego, G.; Dognini, G.; Colombelli, P.L.; Ghidini, A. Therapeutic and prognostic role of vitamin D for COVID-19 infection: A systematic review and meta-analysis of 43 observational studies. J. Steroid Biochem. Mol. Biol. 2021, 211, 105883. [Google Scholar] [CrossRef]
- Jain, A.; Chaurasia, R.; Sengar, N.S.; Singh, M.; Mahor, S.; Narain, S. Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci. Rep. 2020, 10, 20191. [Google Scholar] [CrossRef]
- Jevalikar, G.; Mithal, A.; Singh, A.; Sharma, R.; Farooqui, K.J.; Mahendru, S.; Dewan, A. Lack of association of baseline 25-hydroxyvitamin D levels with disease severity and mortality in Indian patients hospitalized for COVID-19. Sci. Rep. 2021, 11, 6258. [Google Scholar] [CrossRef]
- Patchen, B.K.; Clark, A.G.; Gaddis, N.; Hancock, D.B.; Cassano, P.A. Genetically predicted serum vitamin D and COVID-19: A Mendelian randomisation study. BMJ Nutr. Prev. Health 2021, 4, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Wang, L. C-reactive protein levels in the early stage of COVID-19. Médecine Mal. Infect. 2020, 50, 332–334. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Cao, J.; Wang, Q.; Shi, Q.; Liu, K.; Luo, Z.; Chen, X.; Chen, S.; Yu, K.; Huang, Z. D-dimer as a biomarker for disease severity and mortality in COVID-19 patients: A case control study. J. Intensive Care 2020, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Rostami, M.; Mansouritorghabeh, H. D-dimer level in COVID-19 infection: A systematic review. Expert Rev. Hematol. 2020, 13, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Hyppönen, E.; Berry, D.; Cortina-Borja, M.; Power, C. 25-Hydroxyvitamin D and pre-clinical alterations in inflammatory and hemostatic markers: A cross sectional analysis in the 1958 British Birth Cohort. PLoS ONE 2010, 5, e10801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lippi, G.; Plebani, M.; Henry, B.M. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin. Chim. Acta 2020, 506, 145–148. [Google Scholar] [CrossRef]
- Borsche, L.; Glauner, B.; von Mendel, J. COVID-19 Mortality Risk Correlates Inversely with Vitamin D3 Status, and a Mortality Rate Close to Zero Could Theoretically Be Achieved at 50 ng/mL 25(OH)D3: Results of a Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3596. [Google Scholar] [CrossRef]
- Walsh, J.B.; McCartney, D.M.; Laird, É.; McCarroll, K.; Byrne, D.G.; Healy, M.; O’Shea, P.M.; Kenny, R.A.; Faul, J.L. Understanding a Low Vitamin D State in the Context of COVID-19. Front. Pharmacol. 2022, 13, 835480. [Google Scholar] [CrossRef]
- Haussler, M.R.; Jurutka, P.W.; Mizwicki, M.; Norman, A.W. Vitamin D receptor (VDR)-mediated actions of 1α,25(OH)₂vitamin D₃: Genomic and non-genomic mechanisms. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 543–559. [Google Scholar] [CrossRef]
- Hii, C.S.; Ferrante, A. The Non-Genomic Actions of Vitamin, D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef] [Green Version]
- Jeong, H.; Vacanti, N.M. Systemic vitamin intake impacting tissue proteomes. Nutr. Metab. 2020, 17, 73. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef] [PubMed]
| Male | 60 (%) | |
|---|---|---|
| BMI (kg/m2) (%) <18.5 kg/m2 18.5–24.9 kg/m2 25–29.9 kg/m2 ≥30 kg/m2 | (%) 4 36 22 38 | |
| Current Smoker | 2% | |
| Comorbidity Diabetes Mellitus Hypertension Hyperlipidaemia Renal Disease Respiratory Disease Malignancy Other Comorbidity | n (%) 16 35 15 5 23 10 49 | |
| Serum 25(OH)D (nmol·L−1) (%) Deficient (<30 nmol·L−1) Insufficient (30–49.9 nmol·L−1) Sufficient (>50 nmol·L−1) | (%) 38 26 36 | |
| Mortality (%) Survivors Non-survivors | n (%) 85 15 | |
| ICU Admission (%) Admitted to ICU > 24 h | n (%) 19 | |
| Extended O2 Requirement > 24 h (%) Yes | n (%) 69 |
| Parameter Biochemistry | 25(OH)D Concentration < 30 nmol·L−1 (Deficient) | 25(OH)D Concentration 30–49.9 nmol·L−1 (Insufficient) | 25(OH)D Concentration > 50 nmol·L−1 (Sufficient) | p Value |
|---|---|---|---|---|
| CRP (mg/L) (median (IQR)) | 114 (182) | 158 (153) | 91 (148) | 0.390 |
| WBC (C/mL) (mean (SD)) | 7.43 × 109 (3.89 × 109) | 6.42 × 109 (2.84 × 109) | 7.74 × 109 (2.74 × 109) | 0.409 |
| Lymphocytes (C/mL) (median (IQR)) | 1.00 × 109 (0.63 × 109) | 0.68 × 109 (0.47 × 109) | 1.00 × 109 (0.56 × 109) | 0.036 |
| Eosinophils (C/mL) (median (IQR)) | 0.00 × 109 (0.30 × 109) | 0.00 × 109 (0.01 × 109) | 0.00 × 109 (0.05 × 109) | 0.554 |
| Neutrophils (C/mL) (mean (SD)) | 5.62 × 109 (3.5 × 109) | 5.12 × 109 (2.76 × 109) | 5.83 × 109 (2.49 × 109) | 0.739 |
| Ferritin (ng/mL) (median (IQR)) | 894 (1250) | 814 (1944) | 1068 (1663) | 0.929 |
| D-Dimer (ng/mL) (median (IQR)) | 895 (3039) | 902 (806) | 768 (1513) | 0.453 |
| Platelets (109/L) (mean SD)) | 216 (88) | 219 (119) | 222 (70) | 0.965 |
| Troponin (ng/mL) (median (IQR)) | 6.5 (13.2) | 6.7 (11.2) | 7.0 (9.7) | 0.923 |
| ACE-2 (ng/mL) (median (IQR)) | 35 (25) | 32 (12) | 35 (48) | 0.788 |
| Calcium (mmol/L) (median (IQR)) | 2.19 (0.18) | 2.20 (0.19) | 2.20 (0.2) | 0.180 |
| Magnesium (mmol/L) (median (IQR)) | 0.86 (0.16) | 0.82 (0.11) | 0.83 (0.13) | 0.119 |
| Parameter | 25(OH)D Concentration <30 nmol/L (n = 88) | 25(OH)D Concentration 30–50 nmol/L (n = 60) | 25(OH)D Concentration >50 nmol/L (n = 84) | p Value |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | ||
| O2 Requirement >24 h | 36 (75) | 14 (70) | 14 (56.0) | 0.249 |
| ICU Admission >24 h | 19 (21.6) | 17 (28.3) | 8 (9.5) | 0.013 |
| Mortality Non-survivors | 19 (22) | 4 (7) | 11 (13) | 0.037 |
| Category | Beta Coefficient | Standard Error (SE) | OR (95% CI) | p Value |
|---|---|---|---|---|
| Gender | 0.069 | 0.277 | 1.071 (0.622–1.844) | 0.804 |
| Age | −0.653 | 0.308 | 0.520 (0.285–0.951) | 0.034 |
| Diabetes Mellitus | 0.053 | 0.373 | 1.055 (0.508–2.189) | 0.886 |
| Renal Disease | 0.338 | 0.937 | 1.402 (0.223–8.801) | 0.718 |
| BMI (≥30 kg/m2) | 0.382 | 0.358 | 1.465 (0.726–2.959) | 0.286 |
| Outcome Measure | Covariate | OR (95% CI) | p Value |
|---|---|---|---|
| Mortality | 25(OH)D Concentration <30 nmol/L | 4.63 (1.53–13.97) | 0.006 |
| Age ≥70 years Diabetes Mellitus Male Gender Obesity | 13.32 (4.24–41.81) 1.59 (0.43–5.86) 0.90 (0.33–2.41) 2.03 (0.744–5.57) | <0.001 0.484 0.836 0.166 | |
| ICU Admission | 25(OH)D Concentration < 30 nmol/L | 1.18 (0.58–2.39) | 0.632 |
| Age ≥70 years | 0.29 (0.11–0.73) | 0.009 | |
| Male Gender | 2.90 (1.29–6.51) | 0.010 | |
| Diabetes Mellitus | 2.82 (1.21–6.53) | 0.016 | |
| Extended O2 Requirement >24 h | 25(OH)D Concentration < 30 nmol/L | 2.11 (0.83–5.39) | 0.116 |
| Male Gender | 3.22 (1.26–8.22) | 0.014 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrett, R.; Youssef, M.; Shah, I.; Ioana, J.; Lawati, A.A.; Bukhari, A.; Hegarty, S.; Cormican, L.J.; Judge, E.; Burke, C.M.; et al. Vitamin D Status and Mortality from SARS CoV-2: A Prospective Study of Unvaccinated Caucasian Adults. Nutrients 2022, 14, 3252. https://doi.org/10.3390/nu14163252
Barrett R, Youssef M, Shah I, Ioana J, Lawati AA, Bukhari A, Hegarty S, Cormican LJ, Judge E, Burke CM, et al. Vitamin D Status and Mortality from SARS CoV-2: A Prospective Study of Unvaccinated Caucasian Adults. Nutrients. 2022; 14(16):3252. https://doi.org/10.3390/nu14163252
Chicago/Turabian StyleBarrett, Robert, Modar Youssef, Irfan Shah, Julia Ioana, Abdullah Al Lawati, Abdullah Bukhari, Suzanne Hegarty, Liam J. Cormican, Eoin Judge, Conor M. Burke, and et al. 2022. "Vitamin D Status and Mortality from SARS CoV-2: A Prospective Study of Unvaccinated Caucasian Adults" Nutrients 14, no. 16: 3252. https://doi.org/10.3390/nu14163252
APA StyleBarrett, R., Youssef, M., Shah, I., Ioana, J., Lawati, A. A., Bukhari, A., Hegarty, S., Cormican, L. J., Judge, E., Burke, C. M., Cody, C., Feely, J., Hutchinson, K., Tormey, W., Neill, E. O., Shea, A. O., Connolly, M., McCartney, D. M. A., & Faul, J. L. (2022). Vitamin D Status and Mortality from SARS CoV-2: A Prospective Study of Unvaccinated Caucasian Adults. Nutrients, 14(16), 3252. https://doi.org/10.3390/nu14163252






