Effects of Probiotics on Gut Microbiomes of Extremely Preterm Infants in the Neonatal Intensive Care Unit: A Prospective Cohort Study
Abstract
:1. Introduction
2. Methods
2.1. Study Design and Participants
2.2. Sample Collection
2.3. Library Preparation for 16S rRNA Metagenomics Sequencing
2.4. MiSeq-Based High Throughput Sequencing
2.5. Metagenomics Analysis
2.6. Statistical Analyses
3. Results
3.1. Patients Demographics and Clinical Characteristics
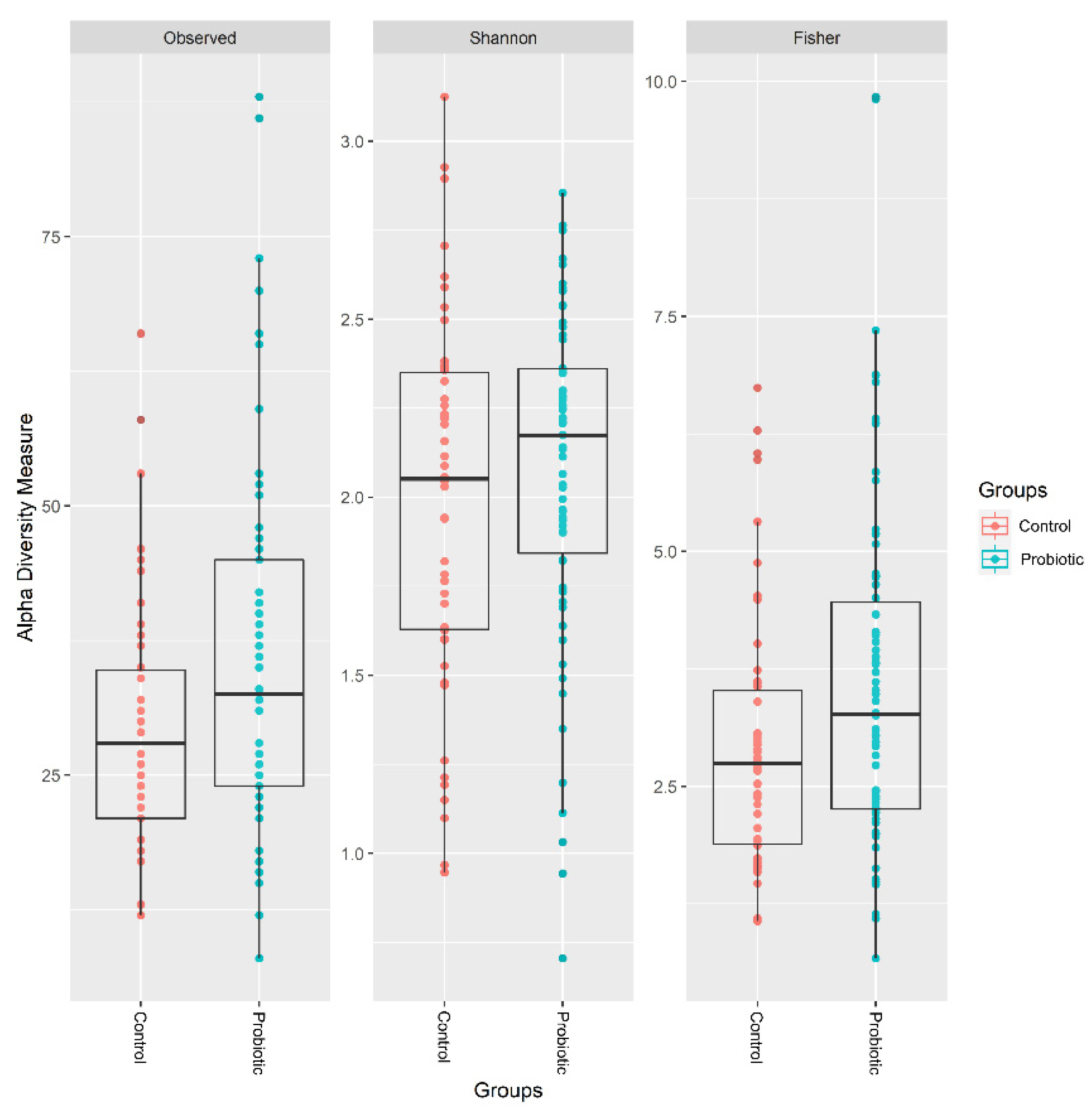
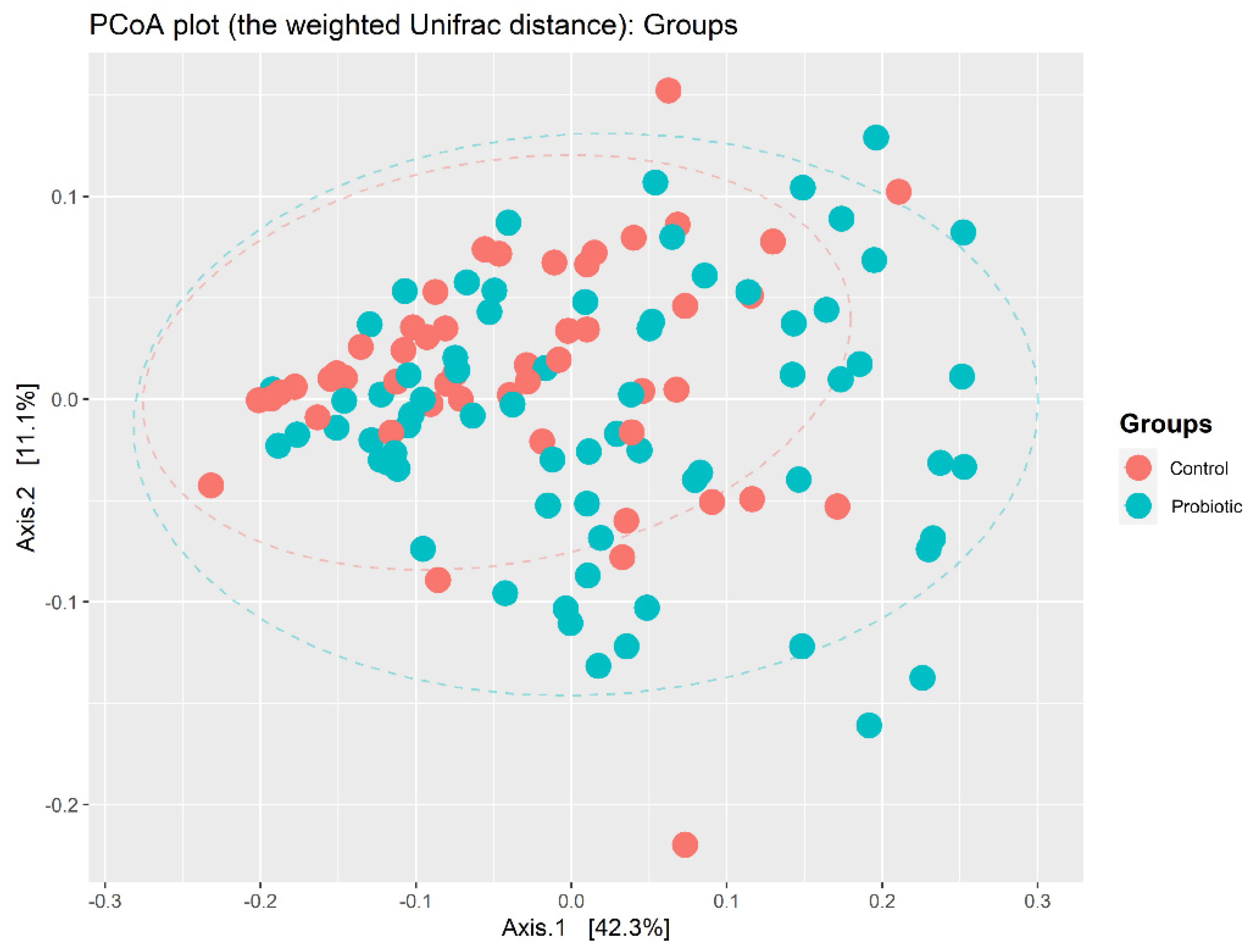
3.2. Summary of Infant Fecal Microbiota Profiling
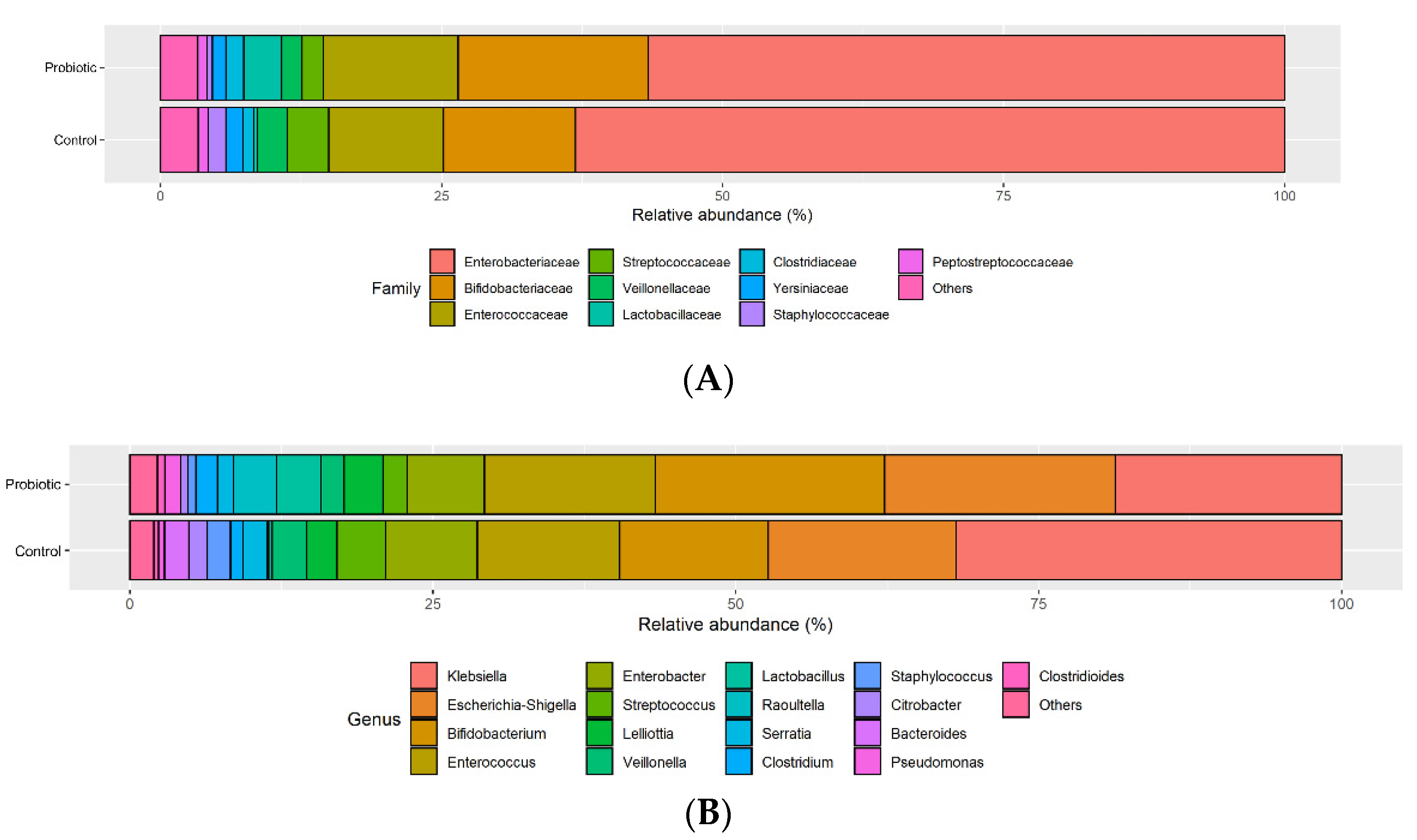
3.3. Changes in the Most Abundant Bacterial Taxa in Infants Supplemented with Probiotics
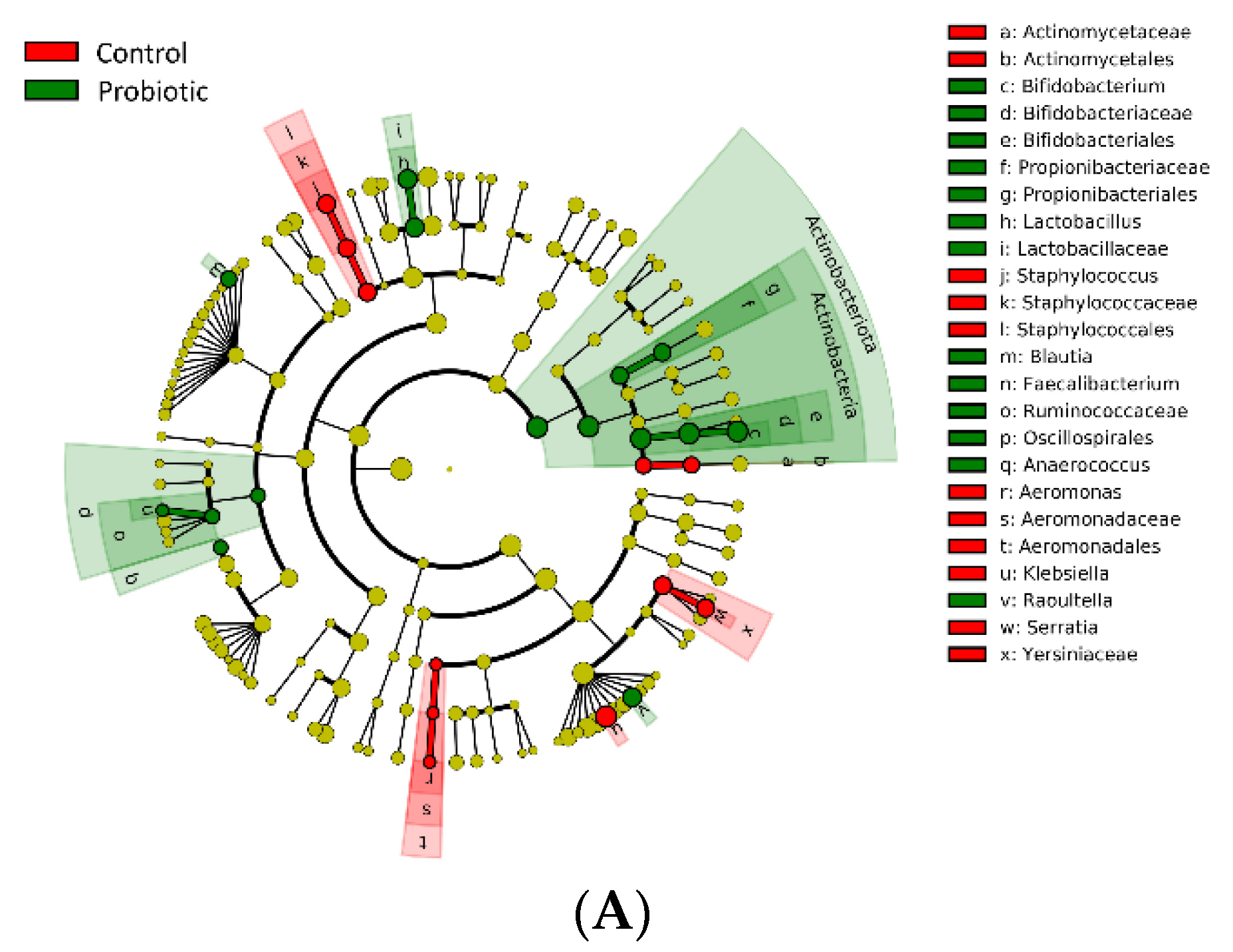
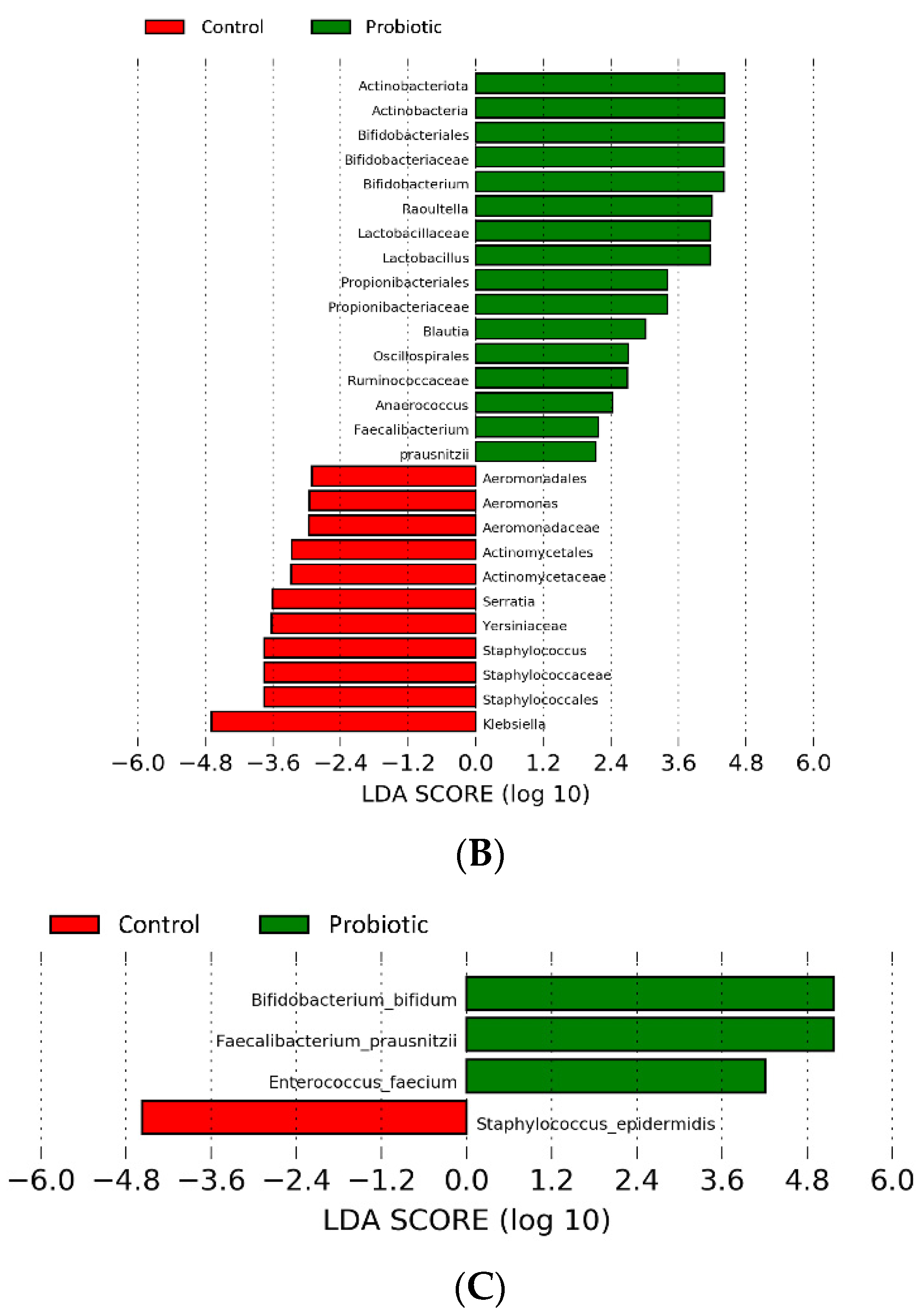
3.4. Differential Taxa in the Microbiome of the Extremely Preterm Neonates
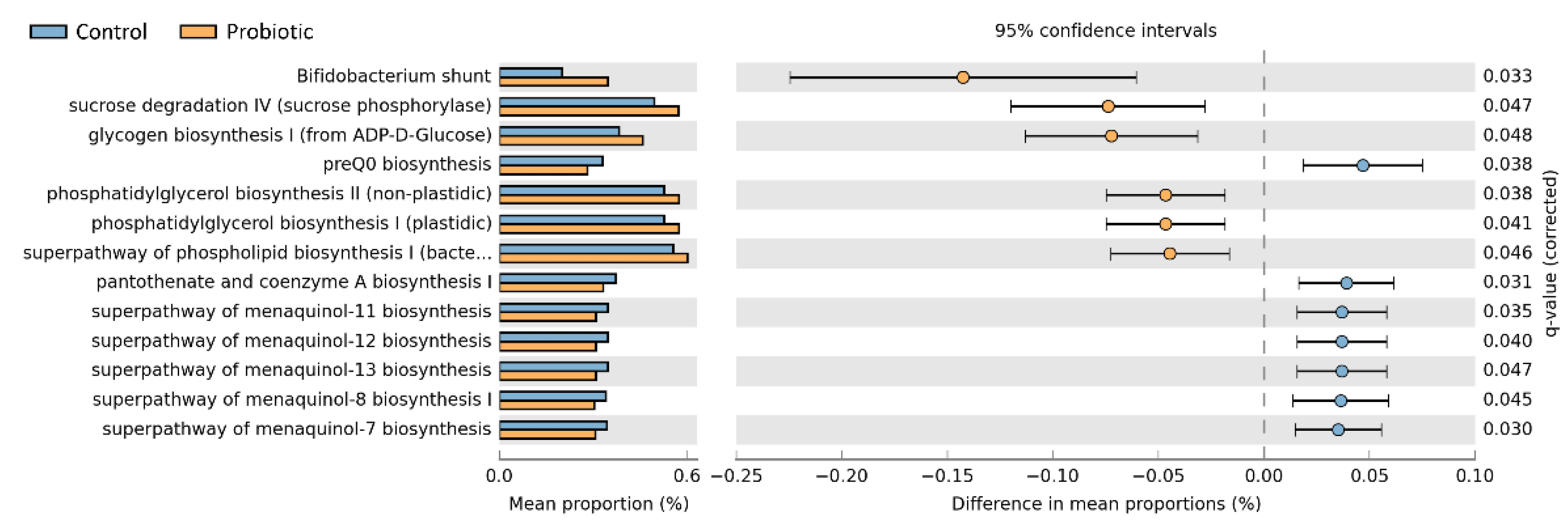
3.5. Functional Prediction of the Microbiome
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chong, C.Y.L.; Bloomfield, F.H.; O’Sullivan, J.M. Factors Affecting Gastrointestinal Microbiome Development in Neonates. Nutrients 2018, 10, 274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujimura, K.E.; Sitarik, A.R.; Havstad, S.; Lin, D.L.; LeVan, S.; Fadrosh, D.; Panzer, A.R.; LaMere, B.; Rackaityte, E.; Lukacs, N.W.; et al. Neonatal gut microbiota associates with childhood multisensitized atopy and T cell differentiation. Nat. Med. 2016, 22, 1187–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piersigilli, F.; Van Grambezen, B.; Hocq, C.; Danhaive, O. Nutrients and Microbiota in Lung Diseases of Prematurity: The Placenta-Gut-Lung Triangle. Nutrients 2020, 12, 469. [Google Scholar] [CrossRef] [Green Version]
- Cukrowska, B.; Bierła, J.B.; Zakrzewska, M.; Klukowski, M.; Maciorkowska, E. The Relationship between the Infant Gut Microbiota and Allergy. The Role of Bifidobacterium breve and Prebiotic Oligosaccharides in the Activation of Anti-Allergic Mechanisms in Early Life. Nutrients 2020, 12, 946. [Google Scholar] [CrossRef] [Green Version]
- Milani, C.; Duranti, S.; Bottacini, F.; Casey, E.; Turroni, F.; Mahony, J.; Belzer, C.; Delgado Palacio, S.; Arboleya Montes, S.; Mancabelli, L.; et al. The First Microbial Colonizers of the Human Gut: Composition, Activities, and Health Implications of the Infant Gut Microbiota. Microbiol. Mol. Biol. Rev. 2017, 81, e00036-17. [Google Scholar] [CrossRef] [Green Version]
- Sanidad, K.Z.; Zeng, M.Y. Neonatal gut microbiome and immunity. Curr. Opin. Microbiol. 2020, 56, 30–37. [Google Scholar] [CrossRef]
- Reyman, M.; van Houten, M.A.; van Baarle, D.; Bosch, A.A.T.M.; Man, W.H.; Chu, M.L.J.N.; Arp, K.; Watson, R.L.; Sanders, E.A.M.; Fuentes, S. Impact of delivery mode-associated gut microbiota dynamics on health in the first year of life. Nat. Commun. 2019, 10, 4997. [Google Scholar] [CrossRef] [Green Version]
- Al-Hadidi, A.; Navarro, J.; Goodman, S.D.; Bailey, M.T.; Besner, G.E. Lactobacillus reuteri in its biofilm state improves protection from experimental necrotizing enterocolitis. Nutrients 2021, 10, 918. [Google Scholar] [CrossRef]
- Skonieczna-Zydecka, K.; Janda, K.; Kaczmarczyk, M.; Marlicz, W.; Loniewski, I.; Loniewska, B. The effects of probiotics on symptoms, gut microbiota and inflammatory markers in infantile colic: A systemic review, meta-analysis and meta-regression of randomized controlled trials. J. Clin. Med. 2020, 9, 999. [Google Scholar] [CrossRef] [Green Version]
- Collado, M.C.; Rautava, S.; Aakko, J.; Isolauri, E.; Salminen, S. Human gut colonization may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid. Sci. Rep. 2016, 6, 23129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, T.T.B.; Chung, H.-J.; Kim, H.-J.; Hong, S.-T. Establishment of an ideal gut microbiota to boost healthy growth of neonates. Crit. Rev. Microbiol. 2019, 45, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, L.; Chen, H.; Zhang, S.; Zhuang, J.; Li, Q.; Feng, Z. Intestinal Microbiota in Early Life and Its Implications on Childhood Health. Genom. Proteom. Bioinform. 2019, 17, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Wopereis, H.; Sim, K.; Shaw, A.; Warner, J.O.; Knol, J.; Kroll, J.S. Intestinal microbiota in infants at high risk for allergy: Effects of prebiotics and role in eczema development. J. Allergy Clin. Immunol. 2017, 141, 1334–1342.e5. [Google Scholar] [CrossRef] [Green Version]
- Hill, C.J.; Lynch, D.B.; Murphy, K.; Ulaszewska, M.; Jeffery, I.; O’Shea, C.A.; Watkins, C.; Dempsey, E.M.; Mattivi, F.; Tuohy, K.; et al. Evolution of gut microbiota composition from birth to 24 weeks in the INFANTMET Cohort. Microbiome 2017, 5, 4. [Google Scholar] [CrossRef] [Green Version]
- AlFaleh, K.; Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2014, 9, CD005496. [Google Scholar] [CrossRef]
- Aceti, A.; Maggio, L.; Beghetti, I.; Gori, D.; Barone, G.; Callegari, M.L.; Fantini, M.P.; Indrio, F.; Meneghin, F.; Morelli, L. Probiotics prevent late-onset sepsis in human milk-fed, vary low birth weight preterm infants: Systemic review and meta-analysis. Nutrients 2017, 9, 904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chi, C.; Li, C.; Buys, N.; Wang, W.; Yin, C.; Sun, J. Effects of Probiotics in Preterm Infants: A Network Meta-analysis. Pediatrics 2021, 147, e20200706. [Google Scholar] [CrossRef]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [Green Version]
- McMurdie, P.J.; Holmes, S. phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [Green Version]
- Kelly, B.J.; Gross, R.; Bittinger, K.; Sherrill-Mix, S.; Lewis, J.D.; Collman, R.; Bushman, F.D.; Li, H. Power and sample-size estimation for microbiome studies using pairwise distances and PERMANOVA. Bioinformatics 2015, 31, 2461–2468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nolan, L.S.; Rimer, J.M.; Good, M. The Role of Human Milk Oligosaccharides and Probiotics on the Neonatal Microbiome and Risk of Necrotizing Enterocolitis: A Narrative Review. Nutrients 2020, 12, 3052. [Google Scholar] [CrossRef] [PubMed]
- Van den Akker, C.H.P.; van Goudoever, J.B.; Szajewska, H.; Embleton, N.D.; Hojsak, I.; Reid, D.; Shamir, R. Probiotics for preterm infants: A strain-specific systematic review and network meta-analysis. J. Pediatr. Gastroenterol. Nutr. 2018, 67, 103–122. [Google Scholar] [CrossRef] [PubMed]
- Sharif, S.; Meader, N.; Oddie, S.J.; Rojas-Reyes, M.X.; McGuire, W. Probiotics to prevent necrotizing enterocolitis in very preterm or very low birth weight infants. Cochrane. Database. Syst. Rev. 2020, 10, CD005496. [Google Scholar]
- Tsai, M.-H.; Chu, S.-M.; Lee, C.-W.; Hsu, J.-F.; Huang, H.-R.; Chiang, M.-C.; Fu, R.-H.; Lien, R.; Huang, Y.-C. Recurrent late-onset sepsis in the neonatal intensive care unit: Incidence, clinical characteristics and risk factors. Clin. Microbiol. Infect. 2014, 20, O928–O935. [Google Scholar] [CrossRef] [Green Version]
- Shelby, R.D.; Mar, P.; Janzow, G.E.; Mashburn-Warren, L.; Tengberg, N.; Navarro, J.B.; Allen, J.M.; Wickham, J.; Wang, Y.; Bailey, M.T.; et al. Antibacterial and anti-inflammatory effects of Lactobacillus reuteri in its biofilm state contribute to its beneficial effects in a rat model of experimental necrotizing enterocolitis. J. Pediatr. Surg. 2022, 57, 1382–1390. [Google Scholar] [CrossRef]
- Beghetti, I.; Panizza, D.; Lenzi, J.; Gori, D.; Martini, S.; Corvaglia, L.; Aceti, A. Probiotics for Preventing Necrotizing Enterocolitis in Preterm Infants: A Network Meta-Analysis. Nutrients 2021, 13, 192. [Google Scholar] [CrossRef]
- Murphy, K.; Ross, R.P.; Ryan, C.A.; Dempsey, E.M.; Stanton, C. Probiotics, Prebiotics, and Synbiotics for the Prevention of Necrotizing Enterocolitis. Front. Nutr. 2021, 8, 928–937. [Google Scholar] [CrossRef]
- Plummer, E.L.; Bulach, D.M.; Murray, G.L.; Jacobs, S.E.; Tabrizi, S.N.; Garland, S.M.; ProPrems Study, G. Gut microbiota of preterm infants supplemented with probiotics: Sub-study of the ProPrems trial. BMC Microbiol. 2018, 18, 184. [Google Scholar] [CrossRef] [Green Version]
- Abdulkadir, B.; Nelson, A.; Skeath, T.; Marrs, E.C.; Perry, J.D.; Cummings, S.P.; Embleton, N.D.; Berrington, J.E.; Stewart, C.J. Routine Use of Probiotics in Preterm Infants: Longitudinal Impact on the Microbiome and Metabolome. Neonatology 2016, 109, 239–247. [Google Scholar] [CrossRef]
- Costeloe, K.; Hardy, P.; Juszczak, E.; Willks, M.; Millar, M.R. Probiotics in preterm infants study collaborate. Bifidobacterium breve BBG-001 in very preterm infants: A randomized controlled phase 3 trial. Lancet 2016, 387, 649–660. [Google Scholar] [CrossRef]
- Sarkar, A.; Yoo, J.Y.; Valeria Ozorio Dutra, S.; Morgan, K.H.; Groer, M. The Association between Early-Life Gut Microbiota and Long-Term Health and Diseases. J. Clin. Med. 2021, 10, 459. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Collado, M.C.; Wopereis, H.; Salminen, S.; Knol, J.; Roeselers, G. The Bifidogenic Effect Revisited—Ecology and Health Perspectives of Bifidobacterial Colonization in Early Life. Microorganisms 2020, 8, 1855. [Google Scholar] [CrossRef] [PubMed]
- Chichlowski, M.; Shah, N.; Wampler, J.L.; Wu, S.S.; Vanderhoof, J.A. Bifidobacterium longum Subspecies infantis (B. infantis) in Pediatric Nutrition: Current State of Knowledge. Nutrients 2020, 12, 1581. [Google Scholar] [CrossRef]
- Robertson, C.; Savva, G.M.; Clapuci, R.; Jones, J.; Maimouni, H.; Brown, E.; Minocha, A.; Hall, L.J.; Clarke, P. Incidence of necrotizing enterocolitis before and after introducing routine prophylactic lactobacillus and Bifidobacterium probiotics. Arch. Dis. Child. Fetal. Neonatal. Ed. 2020, 105, 380–386. [Google Scholar] [CrossRef] [Green Version]
- Juber, B.A.; Boly, T.J.; Pitcher, G.J.; McElroy, S.J. Routine Administration of a Multispecies Probiotic Containing Bifidobacterium and Lactobacillus to Very Low Birth Weight Infants Had No Significant Impact on the Incidence of Necrotizing Enterocolitis. Front. Pediatr. 2021, 9. [Google Scholar] [CrossRef]
- Millar, M.; Seale, J.; Greenland, M.; Hardy, P.; Juszczak, E.; Wilks, M.; Panton, N.; Costeloe, K.; Wade, W.G. The microbiome of infants recruited to a randomized placebo-controlled probiotic trial (PiPS Trial). EBioMedicine 2017, 20, 255–262. [Google Scholar] [CrossRef]
- Deshpande, G.; Rao, S.; Athalye-Jape, G.; Conway, P.; Patole, S. Probiotics in very preterm infants: The PiPS trial. Lancet 2016, 388, 655. [Google Scholar] [CrossRef] [Green Version]
- Costeloe, K.; Bowler, U.; Brocklehurst, P.; Hardy, P.; Heal, P.; Juszczak, E.; King, A.; Panton, N.; Stacey, F.; Whiley, A. A randomized controlled trial of the probiotic Bifidobacterium breve BBG-001 in preterm babies to prevent sepsis, necrotizing enterocolitis and death: The Probiotics in Preterm infants (PiPS) trial. Health Technol. Assess. 2016, 20, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, Y.; Forster, S.C.; Tsaliki, E.; Vervier, K.; Strang, A.; Simpson, N.; Kumar, N.; Stares, M.D.; Rodger, A.; Brocklehurst, P.; et al. Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature 2019, 574, 117–121. [Google Scholar] [CrossRef]
- Elgin, T.G.; Kern, S.L.; McElroy, S.J. Development of the Neonatal Intestinal Microbiome and Its Association With Necrotizing Enterocolitis. Clin. Ther. 2016, 38, 706–715. [Google Scholar] [CrossRef] [Green Version]
- Puopolo, K.M.; Benitz, W.E.; Zaoutis, T.E. Management of neonates born at ≤34 6/7 weeks’ gestation with suspected or proven early-onset bacterial sepsis. Pediatrics 2018, 142, e20182896. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poindexter, B.; Cummings, J.; Hand, I.; Adams-Chapman, I.; Aucott, S.W.; Puopolo, K.M.; Goldsmith, J.P.; Kaufman, D.; Martin, C.; Mowitz, M.; et al. Use of Probiotics in Preterm Infants. Pediatrics 2021, 147, e2021051485. [Google Scholar] [CrossRef] [PubMed]
| The Probiotic Group (Total n = 70) | The Control Group (Total n = 50) | p Values | |
|---|---|---|---|
| Patients demographics | |||
| Birth body weight (g), median (IQR) | 780.0 (689.3–915.0) | 815.0 (757.5–920.0) | 0.511 |
| Gestational age (weeks), median (IQR) | 26.0 (25.0–27.0) | 26.0 (25.0–27.0) | 0.621 |
| Gender (male/female) | 36 (51.2)/34 (48.6) | 32 (64.0)/18 (36.0) | 0.194 |
| NSD/Cesarean section | 18 (25.7)/52 (74.3) | 19 (38.0)/31 (62.0) | 0.166 |
| Inborn/outborn | 12 (17.1) | 10 (20.0) | 0.812 |
| 5 minutes Apgar score ≤ 7, n (%) | 25 (35.7) | 11 (22.0) | 0.157 |
| Perinatal asphyxia, n (%) | 7 (10.0) | 5 (10.0) | 1.000 |
| Premature rupture of membrane, n (%) | 27 (38.6) | 25 (50.0) | 0.263 |
| Intraventricular hemorrhage (≥ Stage II), n (%) | 10 (14.3) | 8 (16.0) | 0.801 |
| Initial use of antibiotics | |||
| Ampicillin plus gentamicin, n (%) | 38 (54.3) | 25 (50.0) | 0.712 |
| Ampicillin plus cefotaxime, n (%) | 32 (45.7) | 25 (50.0) | 0.712 |
| Early-onset sepsis, n (%) | 5 (7.1) | 5 (10.0) | 0.740 |
| Duration of initial empiric antibiotics (days), median (IQR) | 3.0 (1.0–7.0) | 2.0 (1.0–5.0) | |
| Feeding, n (%) | 0.286 | ||
| Breast feeding | 5 (7.1) | 4 (8.0) | |
| Regular formula feeding | 4 (5.7) | 7 (14.0) | |
| Mixed (breast feeding plus regular formula feeding) | 61 (87.1) | 39 (78.0) | |
| Day of feeding initiation (day), median (IQR) | 5.0 (3.0–9.0) | 4.0 (3.8–7.0) | 0.555 |
| Day of stool sample collection (day), median (IQR) | 14.0 (11.0–19.0) | 14.0 (11.0–20.0) | 0.487 |
| Final outcomes, n (%) | |||
| Necrotizing enterocolitis (≥ stage II) | 5 (7.1) | 2 (4.0) | 0.469 |
| Late-onset sepsis | 33 (47.1) | 35 (70.0) | 0.015 |
| Duration of total parenteral nutrition/Intrafat (days), median (IQR) | 29.0 (26.8–35.0) | 35.5 (27.8–45.0) | 0.004 |
| Duration of hospitalization (days), median (IQR) | 96.5 (88.0–112.0) | 98.0 (89.0–116.8) | 0.269 |
| In-hospital mortality | 3 (4.3) | 3 (6.0) | 0.535 |
| Genus * | Probiotic (n = 70) | Control (n = 50) | AOR ** (95% CI) | p Value | Adjusted p Value | ||
|---|---|---|---|---|---|---|---|
| Prevalence n (%) | Relative Abundance Mean% (SD) | Prevalence n (%) | Relative Abundance Mean% (SD) | ||||
| Bifidobacterium | 61 (87) | 18.9 (20.5) | 23 (46) | 12.2 (19.0) | 1.71 (0.83–4.03) | 0.22 | 0.32 |
| Enterobacter | 45 (64) | 6.4 (16.2) | 42 (84) | 7.6 (18.7) | 0.42 (0.19–0.93) | 0.031 | 0.124 |
| Escherichia/Shigella | 48 (69) | 19.1 (23.8) | 29 (58) | 15.5 (24.2) | 1.99 (0.90–4.43) | 0.091 | 0.183 |
| Klebsiella | 62 (89) | 18.7 (25.6) | 46 (92) | 31.8 (30.2) | 0.32 (0.14–0.76) | 0.01 | 0.055 |
| Staphylococcus | 51 (73) | 0.6 (1.5) | 41 (82) | 1.9 (4.4) | 0.35 (0.15–0.78) | 0.01 | 0.055 |
| Enterococcus | 69 (99) | 14.1 (14.7) | 48 (96) | 11.8 (14.7) | 1.51 (0.73–3.14) | 0.27 | 0.329 |
| Streptococcus | 50 (71) | 2 (5.6) | 35 (70) | 4 (9.3) | 0.62 (0.28–1.38) | 0.24 | 0.327 |
| Veillonella | 26 (33) | 1.9 (5.2) | 14 (28) | 2.9 (8.9) | 1.14 (0.47–2.75) | 0.77 | 0.767 |
| Clostridium | 19 (27) | 1.8 (6.6) | 15 (30) | 1 (2.8) | 0.38 (0.13–1.10) | 0.073 | 0.167 |
| Lactobacillus | 45 (64) | 3.5 (7.3) | 14 (28) | 0.3 (1.3) | 4.33 (1.89–9.96) | 0.001 | 0.009 |
| Citrobacter | 23 (33) | 0.6 (1.6) | 15 (30) | 1.5 (4.6) | 0.71 (0.28–1.83) | 0.48 | 0.514 |
| Bacteroides | 26 (37) | 0 (0) | 11 (22) | 2 (12.6) | 2.02 (0.85–4.75) | 0.11 | 0.195 |
| Serratia | 26 (37) | 1.3 (6.5) | 28 (56) | 2 (7.1) | 0.46 (0.22–0.97) | 0.042 | 0.132 |
| Raoultella | 19 (27) | 3.6 (12.8) | 6 (12) | 0.1 (0.4) | 2.73 (1.00–7.45) | 0.049 | 0.132 |
| Lelliottia | 41 (59) | 3.2 (9.9) | 21 (42) | 2.5 (7.1) | 1.74 (0.83–3.62) | 0.14 | 0.224 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-M.; Tsai, M.-H.; Liao, W.-C.; Yang, P.-H.; Li, S.-W.; Chu, S.-M.; Huang, H.-R.; Chiang, M.-C.; Hsu, J.-F. Effects of Probiotics on Gut Microbiomes of Extremely Preterm Infants in the Neonatal Intensive Care Unit: A Prospective Cohort Study. Nutrients 2022, 14, 3239. https://doi.org/10.3390/nu14153239
Chang C-M, Tsai M-H, Liao W-C, Yang P-H, Li S-W, Chu S-M, Huang H-R, Chiang M-C, Hsu J-F. Effects of Probiotics on Gut Microbiomes of Extremely Preterm Infants in the Neonatal Intensive Care Unit: A Prospective Cohort Study. Nutrients. 2022; 14(15):3239. https://doi.org/10.3390/nu14153239
Chicago/Turabian StyleChang, Ching-Min, Ming-Horng Tsai, Wei-Chao Liao, Peng-Hong Yang, Shiao-Wen Li, Shih-Ming Chu, Hsuan-Rong Huang, Ming-Chou Chiang, and Jen-Fu Hsu. 2022. "Effects of Probiotics on Gut Microbiomes of Extremely Preterm Infants in the Neonatal Intensive Care Unit: A Prospective Cohort Study" Nutrients 14, no. 15: 3239. https://doi.org/10.3390/nu14153239
APA StyleChang, C.-M., Tsai, M.-H., Liao, W.-C., Yang, P.-H., Li, S.-W., Chu, S.-M., Huang, H.-R., Chiang, M.-C., & Hsu, J.-F. (2022). Effects of Probiotics on Gut Microbiomes of Extremely Preterm Infants in the Neonatal Intensive Care Unit: A Prospective Cohort Study. Nutrients, 14(15), 3239. https://doi.org/10.3390/nu14153239







