Biological Role of Nutrients, Food and Dietary Patterns in the Prevention and Clinical Management of Major Depressive Disorder
Abstract
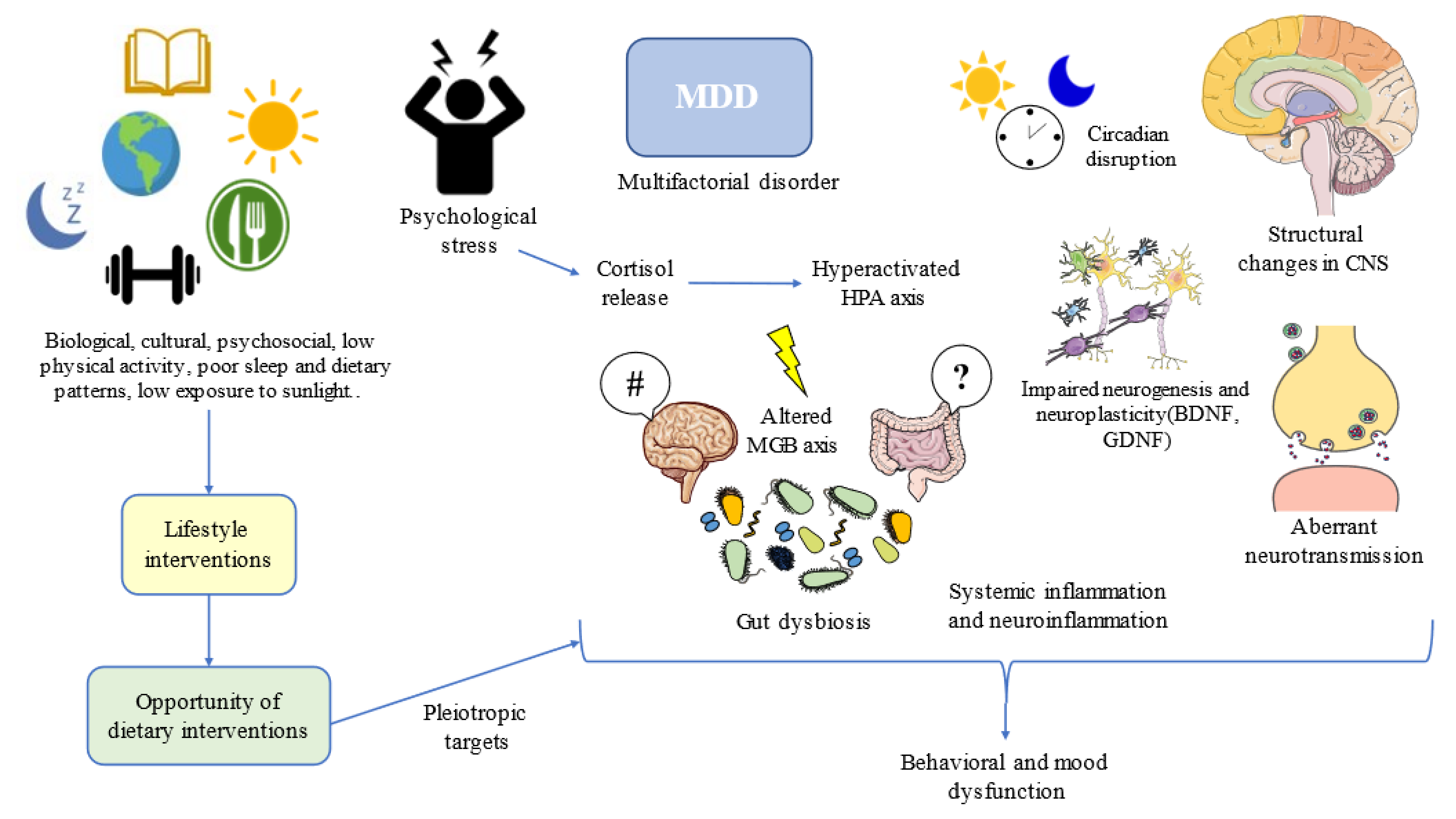
1. Major Depressive Disorder and Diet: What Is the Relationship?
1.1. A General Perspective of Major Depressive Disorder
1.2. Nutritional Status of the Patient with MDD
1.3. Biology of Depression: Is There a Role for Diet?
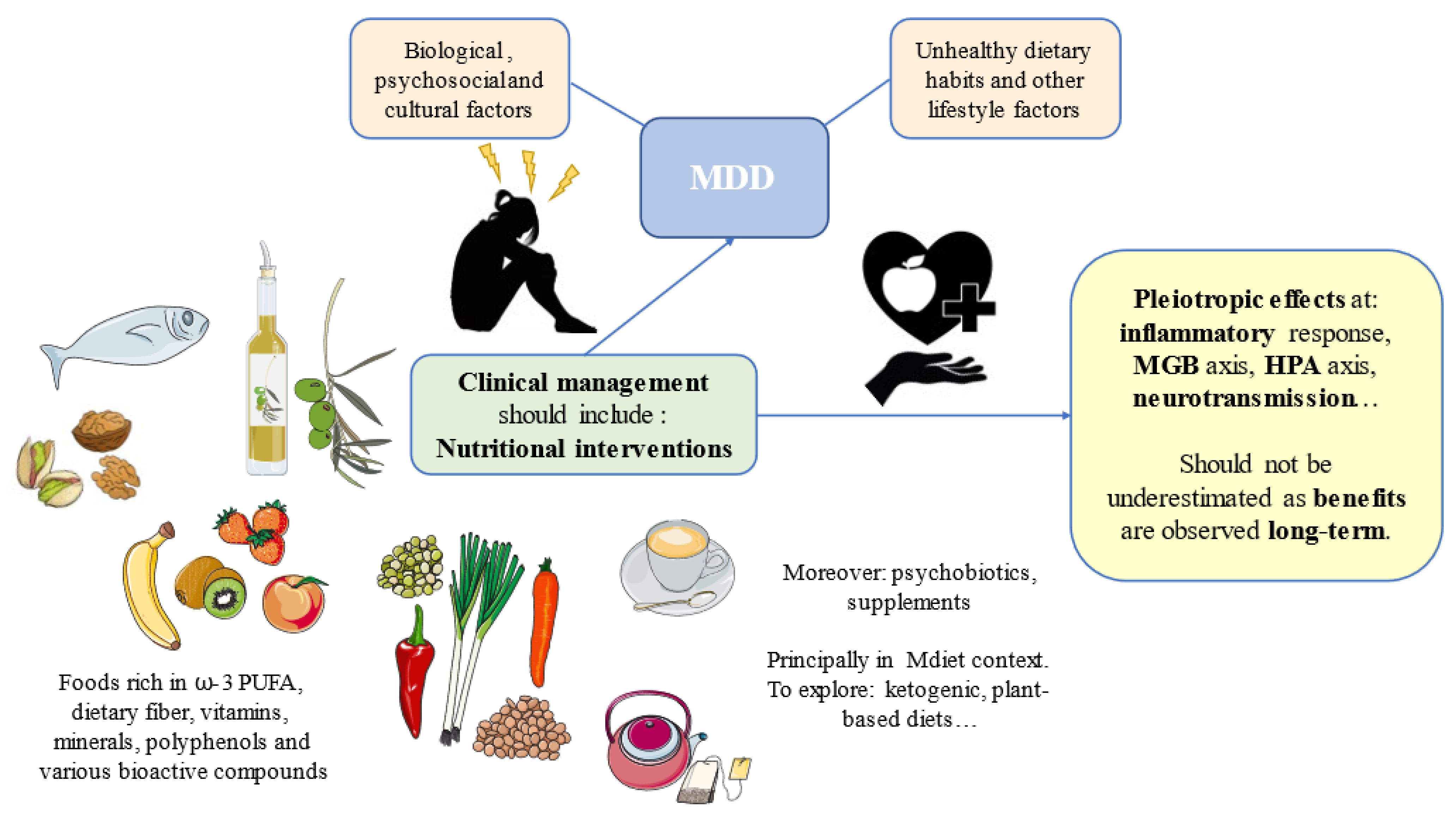
2. Translational Opportunities: Clinical Management of MDD through Diet
2.1. Foods and Nutrients of Interest
2.1.1. Fish, Seeds, and Nuts
2.1.2. Fruits, Vegetables, and Berries
2.1.3. Coffee and Tea: The Role of Caffeine and Specific Bioactive Compounds
2.1.4. Psychobiotics: Relevance of Probiotic Food in MDD
2.1.5. Dietary Supplements
2.2. Dietary Strategies to Implement in Patients with MDD
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACTH | adrenocorticotropin hormone |
| ALA | α-linolenic acid |
| AST | aspartate aminotransferase |
| BBB | blood–brain barrier |
| BDNF | brain-derived neurotrophic factor |
| BUN | blood urea nitrogen |
| CCK | cholecystokinin |
| CNS | central nervous system |
| COVID-19 | coronavirus disease 2019 |
| CRF | cortisol release factor |
| CRP | C-reactive protein |
| DHA | docosahexaenoic acid |
| DHCA | dihydrocaffeic acid |
| DSM-5 | Diagnostic and Statistical Manual of Mental Disorders, 5th Edition |
| EC | epicatechin |
| ECG | epigallocatechin |
| EGCG | epigallocatechin-3-gallate |
| ELS | Early Life Stress |
| EPA | eicosapentaenoic acid |
| EVOO | extra virgin olive oil |
| GABA | gamma-aminobutyric acid |
| GAL | galanin |
| GDNF | glial-derived neurotrophic factor |
| GHDx | Global Health Data Exchange |
| HAM-D | Hamilton Depression Rating Scale |
| HDL-C | high-density lipoprotein cholesterol |
| HPA axis | hypothalamus–pituitary–adrenal |
| IL-1β | interleukin 1β |
| IL-6 | interleukin 6 |
| LAB | lactic acid bacteria |
| LCD | low-carbohydrate diet |
| LPS | lipopolysaccharide |
| MADRS | Montgomery-Åsberg Depression Rating Scale |
| Mal-gluc | malvidin-3′-O-glucoside |
| MAO-A | monoamine oxidase |
| MCH | melanin-concentrating hormone |
| MDA | malondialdehyde |
| MDD | Major Depressive Disorder |
| MDiet | Mediterranean Diet |
| MGB axis | microbiota–gut–brain axis |
| MNA | Mini Nutritional Assessment |
| NCDs | non-communicable diseases |
| NPS | neuropeptide S |
| NPY | neuropeptide Y |
| OXT | oxytocin |
| PREDIMED trials | Prevención con Dieta Mediterránea trials |
| SAMe | S-adenosylmethionine |
| SCFAs | short-chain fatty acids |
| SCOBY | symbiotic culture of bacteria and yeast |
| SD | Sprague-Dawley |
| SMILES trial | Supporting the Modification of lifestyle in Lowered Emotional States trial |
| SUN project | Seguimiento Universidad de Navarra project |
| TNF-α | tumor necrosis factor-α |
| TrkB | tropomyosin receptor kinase B |
| VDBP | vitamin D-binding protein |
| VP | vasopressin |
| WHO | World Health Organization |
| ω-3 PUFA | omega 3 polyunsaturated fatty acids |
References
- World Health Organization. Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 20 July 2022).
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Ortega, M.A.; Alvarez-Mon, M.A.; García-Montero, C.; Fraile-Martinez, O.; Lahera, G.; Monserrat, J.; Muñoz-Merida, L.; Mora, F.; Rodríguez-Jiménez, R.; Fernandez-Rojo, S.; et al. MicroRNAs as Critical Biomarkers of Major Depressive Disorder: A Comprehensive Perspective. Biomedicines 2021, 9, 1659. [Google Scholar] [CrossRef] [PubMed]
- Santomauro, D.F.; Herrera, A.M.M.; Shadid, J.; Zheng, P.; Ashbaugh, C.; Pigott, D.M.; Abbafati, C.; Adolph, C.; Amlag, J.O.; Aravkin, A.Y.; et al. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet 2021, 398, 1700–1712. [Google Scholar] [CrossRef]
- de la Torre, J.A.; Vilagut, G.; Ronaldson, A.; Dregan, A.; Ricci-Cabello, I.; Hatch, S.L.; Serrano-Blanco, A.; Valderas, J.M.; Hotopf, M.; Alonso, J. Prevalence and age patterns of depression in the United Kingdom. A population-based study. J. Affect. Disord. 2021, 279, 164–172. [Google Scholar] [CrossRef]
- Kessler, R.C.; Bromet, E.J. The Epidemiology of Depression Across Cultures. Annu. Rev. Public Health 2013, 34, 119–138. [Google Scholar] [CrossRef]
- Gutiérrez-Rojas, L.; Porras-Segovia, A.; Dunne, H.; Andrade-González, N.; Cervilla, J.A. Prevalence and correlates of major depressive disorder: A systematic review. Rev. Bras. Psiquiatr. 2020, 42, 657. [Google Scholar] [CrossRef]
- Mullins, N.; Lewis, C. Genetics of Depression: Progress at Last. Curr. Psychiatry Rep. 2017, 19, 43. [Google Scholar] [CrossRef]
- Saveanu, R.V.; Nemeroff, C.B. Etiology of Depression: Genetic and Environmental Factors. Psychiatr. Clin. North Am. 2012, 35, 51–71. [Google Scholar] [CrossRef]
- Saleh, A.; Potter, G.G.; McQuoid, D.R.; Boyd, B.; Turner, R.; MacFall, J.R.; Taylor, W.D. Effects of early life stress on depression, cognitive performance and brain morphology. Psychol. Med. 2017, 47, 171. [Google Scholar] [CrossRef]
- Hammen, C.; Kim, E.Y.; Eberhart, N.K.; Brennan, P.A. Chronic and acute stress and the prediction of major depression in women. Depress. Anxiety 2009, 26, 718–723. [Google Scholar] [CrossRef]
- Yelton, B.; Friedman, D.B.; Noblet, S.; Lohman, M.C.; Arent, M.A.; Macauda, M.M.; Sakhuja, M.; Leith, K.H. Social Determinants of Health and Depression among African American Adults: A Scoping Review of Current Research. Int. J. Environ. Res. Public Health 2022, 19, 1498. [Google Scholar] [CrossRef]
- Assari, S. Social Determinants of Depression: The Intersections of Race, Gender, and Socioeconomic Status. Brain Sci. 2017, 7, 156. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Arlington, TX, USA, 2013. [Google Scholar]
- Tolentino, J.C.; Schmidt, S.L. DSM-5 Criteria and Depression Severity: Implications for Clinical Practice. Front. Psychiatry 2018, 9, 450. [Google Scholar] [CrossRef]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef]
- Carrozzino, D.; Patierno, C.; Fava, G.A.; Guidi, J. The Hamilton Rating Scales for Depression: A Critical Review of Clinimetric Properties of Different Versions. Psychother. Psychosom. 2020, 89, 133–150. [Google Scholar] [CrossRef]
- Zimmerman, M.; Martinez, J.H.; Young, D.; Chelminski, I.; Dalrymple, K. Severity classification on the Hamilton depression rating scale. J. Affect. Disord. 2013, 150, 384–388. [Google Scholar] [CrossRef]
- Grover, S.; Gautam, S.; Jain, A.; Gautam, M.; Vahia, V.N. Clinical Practice Guidelines for the management of Depression. Indian J. Psychiatry 2017, 59, S34–S50. [Google Scholar] [CrossRef]
- Voineskos, D.; Daskalakis, Z.J.; Blumberger, D.M. Management of Treatment-Resistant Depression: Challenges and Strategies. Neuropsychiatr. Dis. Treat. 2020, 16, 221. [Google Scholar] [CrossRef]
- Katon, W.; Unützer, J.; Russo, J. Major depression: The importance of clinical characteristics and treatment response to prognosis. Depress. Anxiety 2010, 27, 19–26. [Google Scholar] [CrossRef]
- Greenberg, P.E.; Fournier, A.-A.; Sisitsky, T.; Simes, M.; Berman, R.; Koenigsberg, S.H.; Kessler, R.C. The Economic Burden of Adults with Major Depressive Disorder in the United States (2010 and 2018). Pharmacoeconomics 2021, 39, 653–665. [Google Scholar] [CrossRef]
- Steiber, A.; Hegazi, R.; Herrera, M.; Zamor, M.L.; Chimanya, K.; Pekcan, A.G.; Redondo-Samin, D.C.D.; Correia, M.I.T.; Ojwang, A.A. Spotlight on Global Malnutrition: A Continuing Challenge in the 21st Century. J. Acad. Nutr. Diet. 2015, 115, 1335–1341. [Google Scholar] [CrossRef]
- Perez-Escamilla, R.; Bermudez, O.; Buccini, G.S.; Kumanyika, S.; Lutter, C.; Monsivais, P.; Victora, C. Nutrition disparities and the global burden of malnutrition. BMJ 2018, 361, k2252. [Google Scholar] [CrossRef]
- Norman, K.; Haß, U.; Pirlich, M. Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients 2021, 13, 2764. [Google Scholar] [CrossRef]
- Ortega, M.A.; Fraile-Martínez, Ó.; García-Montero, C.; Alvarez-Mon, M.A.; Lahera, G.; Monserrat, J.; Llavero-Valero, M.; Mora, F.; Rodríguez-Jiménez, R.; Fernandez-Rojo, S.; et al. Nutrition, Epigenetics, and Major Depressive Disorder: Understanding the Connection. Front. Nutr. 2022, 9, 867150. [Google Scholar] [CrossRef]
- Alam, M.R.; Karmokar, S.; Reza, S.; Kabir, R.; Ghosh, S.; Al Mamun, A. Geriatric malnutrition and depression: Evidence from elderly home care population in Bangladesh. Prev. Med. Rep. 2021, 23, 101478. [Google Scholar] [CrossRef] [PubMed]
- Velázquez-Alva, M.C.; Irigoyen-Camacho, M.E.; Cabrer-Rosales, M.F.; Lazarevich, I.; Arrieta-Cruz, I.; Gutiérrez-Juárez, R.; Zepeda-Zepeda, M.A. Prevalence of Malnutrition and Depression in Older Adults Living in Nursing Homes in Mexico City. Nutrients 2020, 12, 2429. [Google Scholar] [CrossRef] [PubMed]
- Tsai, A.C.; Chou, Y.-T.; Chang, T.-L. Usefulness of the Mini Nutritional Assessment (MNA) in predicting the nutritional status of people with mental disorders in Taiwan. J. Clin. Nurs. 2011, 20, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Kaner, G.; Soylu, M.; Yüksel, N.; Inanç, N.; Ongan, D.; Başmısırlı, E. Evaluation of Nutritional Status of Patients with Depression. BioMed Res. Int. 2015, 2015, 521481. [Google Scholar] [CrossRef] [PubMed]
- Nachane, H.B.; Nayak, A.S. Maternal anthropometric determinants as risk markers of suicidality and severity of illness in women with postnatal depression. J. Postgrad. Med. 2020, 66, 11–16. [Google Scholar] [CrossRef]
- Lee, B.J. Association of depressive disorder with biochemical and anthropometric indices in adult men and women. Sci. Rep. 2021, 11, 13596. [Google Scholar] [CrossRef]
- Silva, D.; Ferriani, L.; Viana, M.C. Depression, anthropometric parameters, and body image in adults: A systematic review. Rev. Assoc. Med. Bras. 2019, 65, 731–738. [Google Scholar] [CrossRef]
- Oh, J.; Yun, K.; Chae, J.-H.; Kim, T.-S. Association Between Macronutrients Intake and Depression in the United States and South Korea. Front. Psychiatry 2020, 11, 207. [Google Scholar] [CrossRef]
- García-Montero, C.; Fraile-Martínez, O.; Gómez-Lahoz, A.; Pekarek, L.; Castellanos, A.; Noguerales-Fraguas, F.; Coca, S.; Guijarro, L.; García-Honduvilla, N.; Asúnsolo, A.; et al. Nutritional Components in Western Diet Versus Mediterranean Diet at the Gut Microbiota–Immune System Interplay. Implications for Health and Disease. Nutrients 2021, 13, 699. [Google Scholar] [CrossRef]
- Sánchez-Villegas, A.; Verberne, L.; De Irala, J.; Ruíz-Canela, M.; Toledo, E.; Serra-Majem, L.; Martínez-González, M.A. Dietary Fat Intake and the Risk of Depression: The SUN Project. PLoS ONE 2011, 6, e16268. [Google Scholar] [CrossRef]
- Vagena, E.; Ryu, J.K.; Baeza-Raja, B.; Walsh, N.M.; Syme, C.; Day, J.P.; Houslay, M.D.; Baillie, G.S. A high-fat diet promotes depression-like behavior in mice by suppressing hypothalamic PKA signaling. Transl. Psychiatry 2019, 9, 141. [Google Scholar] [CrossRef]
- Ciesielski, T.H.; Williams, S.M. Low Omega-3 intake is associated with high rates of depression and preterm birth on the country level. Sci. Rep. 2020, 10, 19749. [Google Scholar] [CrossRef]
- Cuomo, A.; Giordano, N.; Goracci, A.; Fagiolini, A. Depression and Vitamin D Deficiency: Causality, Assessment, and Clinical Practice Implications. Neuropsychiatry 2017, 7, 606–614. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Apostolopoulos, V. The Effects of Vitamin B in Depression. Curr. Med. Chem. 2016, 23, 4317–4337. [Google Scholar] [CrossRef]
- Miki, T.; Kochi, T.; Eguchi, M.; Kuwahara, K.; Tsuruoka, H.; Kurotani, K.; Ito, R.; Akter, S.; Kashino, I.; Pham, N.M.; et al. Dietary intake of minerals in relation to depressive symptoms in Japanese employees: The Furukawa Nutrition and Health Study. Nutrition 2015, 31, 686–690. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Miyagi, S.; Tsujiguchi, H.; Kambayashi, Y.; Hara, A.; Nakamura, H.; Suzuki, K.; Yamada, Y.; Shimizu, Y.; Nakamura, H. Association between Lower Intake of Minerals and Depressive Symptoms among Elderly Japanese Women but Not Men: Findings from Shika Study. Nutrients 2019, 11, 389. [Google Scholar] [CrossRef]
- Azizi, S.A. Monoamines: Dopamine, Norepinephrine, and Serotonin, Beyond Modulation, “Switches” That Alter the State of Target Networks. Neuroscientist 2022, 28, 121–143. [Google Scholar] [CrossRef]
- Hindmarch, I. Beyond the monoamine hypothesis: Mechanisms, molecules and methods. Eur. Psychiatry 2002, 17, 294–299. [Google Scholar] [CrossRef]
- Pytka, K.; Dziubina, A.; Młyniec, K.; Dziedziczak, A.; Żmudzka, E.; Furgała, A.; Olczyk, A.; Sapa, J.; Filipek, B. The role of glutamatergic, GABA-ergic, and cholinergic receptors in depression and antidepressant-like effect. Pharmacol. Rep. 2016, 68, 443–450. [Google Scholar] [CrossRef]
- Rana, T.; Behl, T.; Sehgal, A.; Singh, S.; Sharma, N.; Abdeen, A.; Ibrahim, S.F.; Mani, V.; Iqbal, M.S.; Bhatia, S.; et al. Exploring the role of neuropeptides in depression and anxiety. Prog. Neuropsychopharmacol. Biol. Psychiatry 2022, 114, 110478. [Google Scholar] [CrossRef]
- Salva, M.A.Q.; Hartley, S.; Barbot, F.; Alvarez, J.C.; Lofaso, F.; Guilleminault, C. Circadian Rhythms, Melatonin and Depression. Curr. Pharm. Des. 2011, 17, 1459–1470. [Google Scholar] [CrossRef]
- Bourin, M. Neurogenesis and Neuroplasticity in Major Depression: Its Therapeutic Implication. Adv. Exp. Med. Biol. 2021, 1305, 157–173. [Google Scholar] [CrossRef]
- Castrén, E.; Monteggia, L.M. Brain-Derived Neurotrophic Factor Signaling in Depression and Antidepressant Action. Biol. Psychiatry 2021, 90, 128–136. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, L.; Wang, X.; Wang, Y.; Li, C.; Zhu, X. Alterations of DNA Methylation at GDNF Gene Promoter in the Ventral Tegmental Area of Adult Depression-Like Rats Induced by Maternal Deprivation. Front. Psychiatry 2019, 10, 732. [Google Scholar] [CrossRef]
- Keller, J.; Gomez, R.; Williams, G.; Lembke, A.; Lazzeroni, L.; Murphy, G.M., Jr.; Schatzberg, A.F. HPA axis in major depression: Cortisol, clinical symptomatology and genetic variation predict cognition. Mol. Psychiatry 2017, 22, 527. [Google Scholar] [CrossRef]
- Rajkowska, G.; Miguel-Hidalgo, J. Gliogenesis and Glial Pathology in Depression. CNS Neurol. Disord. Drug Targets 2007, 6, 219–233. [Google Scholar] [CrossRef]
- Duman, R.S. Neuronal damage and protection in the pathophysiology and treatment of psychiatric illness: Stress and depression. Dialog. Clin. Neurosci. 2009, 11, 239–255. [Google Scholar] [CrossRef]
- Pandya, M.; Altinay, M.; Malone, D.A.; Anand, A. Where in the Brain Is Depression? Curr. Psychiatry Rep. 2012, 14, 634–642. [Google Scholar] [CrossRef]
- Miller, A.H.; Raison, C.L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2016, 16, 22. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gómez, A.M.; Orozco, A.; Lahera, G.; Sosa, M.D.; Diaz, D.; Auba, E.; Albillos, A.; Monserrat, J.; Alvarez-Mon, M. Abnormal Distribution and Function of Circulating Monocytes and Enhanced Bacterial Translocation in Major Depressive Disorder. Front. Psychiatry 2019, 10, 812. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.; Gómez-Lahoz, A.; Orozco, A.; Lahera, G.; Diaz, D.; Ortega, M.; Albillos, A.; Quintero, J.; Aubá, E.; Monserrat, J.; et al. Expansion of CD4 T Lymphocytes Expressing Interleukin 17 and Tumor Necrosis Factor in Patients with Major Depressive Disorder. J. Pers. Med. 2021, 11, 220. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gomez-Lahoz, A.M.; Orozco, A.; Lahera, G.; Sosa-Reina, M.D.; Diaz, D.; Albillos, A.; Quintero, J.; Molero, P.; Monserrat, J.; et al. Blunted Expansion of Regulatory T Lymphocytes Is Associated With Increased Bacterial Translocation in Patients With Major Depressive Disorder. Front. Psychiatry 2021, 11, 1530. [Google Scholar] [CrossRef]
- Kahl, K.G. Metabolic alterations in patients with depression and their relationship to the etiology of depressive disorders. Ann. Gen. Psychiatry 2010, 9, S34. [Google Scholar] [CrossRef][Green Version]
- Francisco, V.; Gualillo, O.; van den Bossche, J.; Ortega, M.A.; García-Montero, C.; Fraile-Martinez, O.; Angel Alvarez-Mon, M.; Maria Gómez-Lahoz, A.; Lahera, G.; Monserrat, J.; et al. Immune-Mediated Diseases from the Point of View of Psychoneuroimmunoendocrinology. Biology 2022, 11, 973. [Google Scholar] [CrossRef]
- Cepeda, M.S.; Katz, E.G.; Blacketer, C. Microbiome-Gut-Brain Axis: Probiotics and Their Association With Depression. J. Neuropsychiatry Clin. Neurosci. 2016, 29, 39–44. [Google Scholar] [CrossRef]
- Ortega, M.A.; Alvarez-Mon, M.A.; García-Montero, C.; Fraile-Martinez, O.; Guijarro, L.G.; Lahera, G.; Monserrat, J.; Valls, P.; Mora, F.; Rodríguez-Jiménez, R.; et al. Gut Microbiota Metabolites in Major Depressive Disorder—Deep Insights into Their Pathophysiological Role and Potential Translational Applications. Metabolites 2022, 12, 50. [Google Scholar] [CrossRef]
- Xu, Y.; Zeng, L.; Zou, K.; Shan, S.; Wang, X.; Xiong, J.; Zhao, L.; Zhang, L.; Cheng, G. Role of dietary factors in the prevention and treatment for depression: An umbrella review of meta-analyses of prospective studies. Transl. Psychiatry 2021, 11, 478. [Google Scholar] [CrossRef] [PubMed]
- Thomas-Odenthal, F.; Molero, P.; Van Der Does, W.; Molendijk, M. Impact of review method on the conclusions of clinical reviews: A systematic review on dietary interventions in depression as a case in point. PLoS ONE 2020, 15, e0238131. [Google Scholar] [CrossRef] [PubMed]
- Firth, J.; Marx, W.; Dash, S.; Carney, R.; Teasdale, S.; Solmi, M.; Stubbs, B.; Schuch, F.; Carvalho, A.F.; Jacka, F.; et al. The Effects of Dietary Improvement on Symptoms of Depression and Anxiety: A Meta-Analysis of Randomized Controlled Trials. Psychosom. Med. 2019, 81, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Hollon, S.D.; Andrews, P.W.; Thomson, J.A. Cognitive Behavior Therapy for Depression From an Evolutionary Perspective. Front. Psychiatry 2021, 12, 667592. [Google Scholar] [CrossRef]
- Hidaka, B.H. Depression as a disease of modernity: Explanations for increasing prevalence. J. Affect. Disord. 2012, 140, 205–214. [Google Scholar] [CrossRef]
- Durisko, Z.; Mulsant, B.H.; Mckenzie, K.; Andrews, P.W. Using Evolutionary Theory to Guide Mental Health Research. Can. J. Psychiatry 2016, 61, 159–165. [Google Scholar] [CrossRef]
- Marx, W.; Lane, M.; Hockey, M.; Aslam, H.; Berk, M.; Walder, K.; Borsini, A.; Firth, J.; Pariante, C.M.; Berding, K.; et al. Diet and depression: Exploring the biological mechanisms of action. Mol. Psychiatry 2020, 26, 134–150. [Google Scholar] [CrossRef]
- García-Montero, C.; Ortega, M.A.; Alvarez-Mon, M.A.; Fraile-Martinez, O.; Romero-Bazán, A.; Lahera, G.; Montes-Rodríguez, J.M.; Molina-Ruiz, R.M.; Mora, F.; Rodriguez-Jimenez, R.; et al. The Problem of Malnutrition Associated with Major Depressive Disorder from a Sex-Gender Perspective. Nutrients 2022, 14, 1107. [Google Scholar] [CrossRef]
- Aguilera, J.M. The food matrix: Implications in processing, nutrition and health. Crit. Rev. Food Sci. Nutr. 2018, 59, 3612–3629. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Ortega, M.A.; García-Montero, C.; Fraile-Martinez, O.; Monserrat, J.; Lahera, G.; Mora, F.; Rodriguez-Quiroga, A.; Fernandez-Rojo, S.; Quintero, J.; et al. Exploring the Role of Nutraceuticals in Major Depressive Disorder (MDD): Rationale, State of the Art and Future Prospects. Pharmaceuticals 2021, 14, 821. [Google Scholar] [CrossRef]
- Li, F.; Liu, X.; Zhang, D. Fish consumption and risk of depression: A meta-analysis. J. Epidemiol. Community Health 2016, 70, 299–304. [Google Scholar] [CrossRef]
- Yang, Y.; Kim, Y.; Je, Y. Fish consumption and risk of depression: Epidemiological evidence from prospective studies. Asia-Pac. Psychiatry 2018, 10, e12335. [Google Scholar] [CrossRef]
- Bountziouka, V.; Polychronopoulos, E.; Zeimbekis, A.; Papavenetiou, E.; Ladoukaki, E.; Papairakleous, N.; Gotsis, E.; Metallinos, G.; Lionis, C.; Panagiotakos, D. Long-Term Fish Intake Is Associated With Less Severe Depressive Symptoms Among Elderly Men and Women: The MEDIS (MEDiterranean ISlands Elderly) Epidemiological Study. J. Aging Health 2009, 21, 864–880. [Google Scholar] [CrossRef]
- Chen, J.; Jayachandran, M.; Bai, W.; Xu, B. A critical review on the health benefits of fish consumption and its bioactive constituents. Food Chem. 2022, 369, 130874. [Google Scholar] [CrossRef]
- Persson, M.; Fagt, S.; Nauta, M.J. Personalised fish intake recommendations: The effect of background exposure on optimisation. Br. J. Nutr. 2018, 120, 946–957. [Google Scholar] [CrossRef]
- Persson, M.; Fagt, S.; Nauta, M.J. Optimising healthy and safe fish intake recommendations: A trade-off between personal preference and cost. Br. J. Nutr. 2019, 122, 206–219. [Google Scholar] [CrossRef]
- Sikka, P.; Behl, T.; Sharma, S.; Sehgal, A.; Bhatia, S.; Al-Harrasi, A.; Singh, S.; Sharma, N.; Aleya, L. Exploring the therapeutic potential of omega-3 fatty acids in depression. Environ. Sci. Pollut. Res. 2021, 28, 43021–43034. [Google Scholar] [CrossRef]
- Kalkman, H.; Hersberger, M.; Walitza, S.; Berger, G. Disentangling the Molecular Mechanisms of the Antidepressant Activity of Omega-3 Polyunsaturated Fatty Acid: A Comprehensive Review of the Literature. Int. J. Mol. Sci. 2021, 22, 4393. [Google Scholar] [CrossRef]
- Martins, J.G. EPA but not DHA Appears to be Responsible for the Efficacy of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation in Depression: Evidence from a Meta-Analysis of Randomized Controlled Trials. J. Am. Coll. Nutr. 2009, 28, 525–542. [Google Scholar] [CrossRef]
- Reeves, J.L.; Otahal, P.; Magnussen, C.G.; Dwyer, T.; Kangas, A.J.; Soininen, P.; Ala-Korpela, M.; Venn, A.J.; Smith, K.J. DHA mediates the protective effect of fish consumption on new episodes of depression among women. Br. J. Nutr. 2017, 118, 743–749. [Google Scholar] [CrossRef]
- Smith, K.J.; Sanderson, K.; McNaughton, S.; Gall, S.; Dwyer, T.; Venn, A.J. Longitudinal Associations Between Fish Consumption and Depression in Young Adults. Am. J. Epidemiol. 2014, 179, 1228–1235. [Google Scholar] [CrossRef]
- Hussey, B.; Lindley, M.R.; Mastana, S.S. Omega 3 fatty acids, inflammation and DNA methylation: An overview. Clin. Lipidol. 2017, 12, 24–32. [Google Scholar] [CrossRef]
- Vijay, A.; Astbury, S.; Le Roy, C.; Spector, T.D.; Valdes, A.M. The prebiotic effects of omega-3 fatty acid supplementation: A six-week randomised intervention trial. Gut Microbes 2021, 13, 1–11. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Jain, A.; Aggarwal, K.; Zhang, P. Omega-3 Fatty Acids and Cardiovascular Disease. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 441–445. [Google Scholar]
- Lu, X.; Ce, Q.; Jin, L.; Zheng, J.; Sun, M.; Tang, X.; Li, D.; Sun, J. Deoiled sunflower seeds ameliorate depression by promoting the production of monoamine neurotransmitters and inhibiting oxidative stress. Food Funct. 2021, 12, 573–586. [Google Scholar] [CrossRef]
- Bikri, S.; Aboussaleh, Y.; Berrani, A.; Louragli, I.; Hafid, A.; Chakib, S.; Ahami, A. Effects of date seeds administration on anxiety and depressive symptoms in streptozotocin-induced diabetic rats: Biochemical and behavioral evidences. J. Basic Clin. Physiol. Pharmacol. 2021, 32, 1031–1040. [Google Scholar] [CrossRef]
- Arab, L.; Guo, R.; Elashoff, D. Lower Depression Scores among Walnut Consumers in NHANES. Nutrients 2019, 11, 275. [Google Scholar] [CrossRef]
- Opie, R.S.; Itsiopoulos, C.; Parletta, N.; Sanchez-Villegas, A.; Akbaraly, T.N.; Ruusunen, A.; Jacka, F.N. Dietary recommendations for the prevention of depression. Nutr. Neurosci. 2017, 20, 161–171. [Google Scholar] [CrossRef]
- Patrick, R.P.; Ames, B.N. Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: Relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. FASEB J. 2015, 29, 2207–2222. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Walther, B. Natural Vitamin D Content in Animal Products. Adv. Nutr. 2013, 4, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Hossein-Nezhad, A.; Holick, M.F. Vitamin D for Health: A Global Perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Kar, S.; Suthar, N.; Nebhinani, N. Vitamin D and Depression: A Critical Appraisal of the Evidence and Future Directions. Indian J. Psychol. Med. 2020, 42, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Guzek, D.; Kołota, A.; Lachowicz, K.; Skolmowska, D.; Stachoń, M.; Głąbska, D. Association between Vitamin D Supplementation and Mental Health in Healthy Adults: A Systematic Review. J. Clin. Med. 2021, 10, 5156. [Google Scholar] [CrossRef]
- Tomé, A.L.; Cebriá, M.J.R.; González-Teruel, A.; Carbonell-Asíns, J.A.; Nicolás, C.C.; Hernández-Viadel, M. Efficacy of vitamin D in the treatment of depression: A systematic review and meta-analysis. Actas. Esp. Psiquiatr. 2021, 49, 12–23. [Google Scholar]
- Fakhoury, H.M.A.; Kvietys, P.R.; AlKattan, W.; Al Anouti, F.A.; Elahi, M.A.; Karras, S.N.; Grant, W.B. Vitamin D and intestinal homeostasis: Barrier, microbiota, and immune modulation. J. Steroid Biochem. Mol. Biol. 2020, 200, 105663. [Google Scholar] [CrossRef]
- Patrick, R.P.; Ames, B.N. Vitamin D hormone regulates serotonin synthesis. Part 1: Relevance for autism. FASEB J. 2014, 28, 2398–2413. [Google Scholar] [CrossRef]
- Singh, P.; Rawat, A.; Alwakeel, M.; Sharif, E.; Al Khodor, S. The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals. Sci. Rep. 2020, 10, 21641. [Google Scholar] [CrossRef]
- Knippenberg, S.; Damoiseaux, J.; Bol, Y.; Hupperts, R.; Taylor, B.V.; Ponsonby, A.-L.; Dwyer, T.; Simpson, S.; Van Der Mei, I.A.F.; Simpson, J.S. Higher levels of reported sun exposure, and not vitamin D status, are associated with less depressive symptoms and fatigue in multiple sclerosis. Acta Neurol. Scand. 2014, 129, 123–131. [Google Scholar] [CrossRef]
- Pooyan, S.; Rahimi, M.H.; Mollahosseini, M.; Khorrami-Nezhad, L.; Nasir, Y.; Maghbooli, Z.; Mirzaei, K. A High-Protein/Low-Fat Diet May Interact with Vitamin D-Binding Protein Gene Variants to Moderate the Risk of Depression in Apparently Healthy Adults. Lifestyle Genom. 2018, 11, 64–72. [Google Scholar] [CrossRef]
- Vashum, K.P.; McEvoy, M.; Milton, A.H.; McElduff, P.; Hure, A.; Byles, J.; Attia, J. Dietary zinc is associated with a lower incidence of depression: Findings from two Australian cohorts. J. Affect. Disord. 2014, 166, 249–257. [Google Scholar] [CrossRef]
- Ulloa, A.C.; Gliga, A.; Love, T.M.; Pineda, D.; Mruzek, D.W.; Watson, G.E.; Davidson, P.W.; Shamlaye, C.F.; Strain, J.; Myers, G.J.; et al. Prenatal methylmercury exposure and DNA methylation in seven-year-old children in the Seychelles Child Development Study. Environ. Int. 2021, 147, 106321. [Google Scholar] [CrossRef]
- Wallace, T.C.; Bailey, R.L.; Blumberg, J.B.; Burton-Freeman, B.; Chen, C.-Y.O.; Crowe-White, K.M.; Drewnowski, A.; Hooshmand, S.; Johnson, E.; Lewis, R.; et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 2020, 60, 2174–2211. [Google Scholar] [CrossRef]
- Głąbska, D.; Guzek, D.; Groele, B.; Gutkowska, K. Fruit and Vegetable Intake and Mental Health in Adults: A Systematic Review. Nutrients 2020, 12, 115. [Google Scholar] [CrossRef]
- Saghafian, F.; Malmir, H.; Saneei, P.; Milajerdi, A.; Larijani, B.; Esmaillzadeh, A. Fruit and vegetable consumption and risk of depression: Accumulative evidence from an updated systematic review and meta-analysis of epidemiological studies. Br. J. Nutr. 2018, 119, 1087–1101. [Google Scholar] [CrossRef]
- Grases, G.; Colom, M.A.; Sanchis, P.; Grases, F. Possible relation between consumption of different food groups and depression. BMC Psychol. 2019, 7, 14. [Google Scholar] [CrossRef]
- Swann, O.G.; Kilpatrick, M.; Breslin, M.; Oddy, W.H. Dietary fiber and its associations with depression and inflammation. Nutr. Rev. 2020, 78, 394–411. [Google Scholar] [CrossRef]
- Kim, Y.; Hong, M.; Kim, S.; Shin, W.-Y.; Kim, J.-H. Inverse association between dietary fiber intake and depression in premenopausal women: A nationwide population-based survey. Menopause 2020, 28, 150–156. [Google Scholar] [CrossRef]
- Obeid, R.; Heil, S.G.; Verhoeven, M.M.A.; van den Heuvel, E.G.H.M.; de Groot, L.; Eussen, S.J.P.M. Vitamin B12 Intake From Animal Foods, Biomarkers, and Health Aspects. Front. Nutr. 2019, 6, 93. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Valtueña, J.; Huybrechts, I.; Breidenassel, C.; Cuenca-García, M.; De Henauw, S.; Stehle, P.; Kafatos, A.; Kersting, M.; Widhalm, K.; et al. Fruit and vegetables consumption is associated with higher vitamin intake and blood vitamin status among European adolescents. Eur. J. Clin. Nutr. 2017, 71, 458–467. [Google Scholar] [CrossRef]
- Carazo, A.; Macáková, K.; Matoušová, K.; Krčmová, L.; Protti, M.; Mladěnka, P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients 2021, 13, 1703. [Google Scholar] [CrossRef]
- Jäpelt, R.B.; Jakobsen, J. Vitamin D in plants: A review of occurrence, analysis, and biosynthesis. Front. Plant Sci. 2013, 4, 136. [Google Scholar] [CrossRef]
- Pham, V.T.; Dold, S.; Rehman, A.; Bird, J.K.; Steinert, R.E. Vitamins, the gut microbiome and gastrointestinal health in humans. Nutr. Res. 2021, 95, 35–53. [Google Scholar] [CrossRef]
- Gougeon, L.; Payette, H.; Morais, J.A.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K. Intakes of folate, vitamin B6 and B12 and risk of depression in community-dwelling older adults: The Quebec Longitudinal Study on Nutrition and Aging. Eur. J. Clin. Nutr. 2016, 70, 380–385. [Google Scholar] [CrossRef]
- Lewis, J.E.; Tiozzo, E.; Melillo, A.B.; Leonard, S.; Chen, L.; Mendez, A.; Woolger, J.M.; Konefal, J. The Effect of Methylated Vitamin B Complex on Depressive and Anxiety Symptoms and Quality of Life in Adults with Depression. ISRN Psychiatry 2013, 2013, 621453. [Google Scholar] [CrossRef]
- Markun, S.; Gravestock, I.; Jäger, L.; Rosemann, T.; Pichierri, G.; Burgstaller, J. Effects of Vitamin B12 Supplementation on Cognitive Function, Depressive Symptoms, and Fatigue: A Systematic Review, Meta-Analysis, and Meta-Regression. Nutrients 2021, 13, 923. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The effects of vitamin B on the immune/cytokine network and their involvement in depression. Maturitas 2017, 96, 58–71. [Google Scholar] [CrossRef]
- Amr, M.; El-Mogy, A.; Shams, T.; Vieira, K.; Lakhan, S.E. Efficacy of vitamin C as an adjunct to fluoxetine therapy in pediatric major depressive disorder: A randomized, double-blind, placebo-controlled pilot study. Nutr. J. 2013, 12, 31. [Google Scholar] [CrossRef]
- Bolzetta, F.; Veronese, N.; Stubbs, B.; Noale, M.; Vaona, A.; Demurtas, J.; Celotto, S.; Cacco, C.; Cester, A.; Caruso, M.G.; et al. The Relationship between Dietary Vitamin K and Depressive Symptoms in Late Adulthood: A Cross-Sectional Analysis from a Large Cohort Study. Nutrients 2019, 11, 787. [Google Scholar] [CrossRef]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P. Polyphenols and human health: Prevention of disease and mechanisms of action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef]
- Williamson, G. The role of polyphenols in modern nutrition. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Trebatická, J.; Ďuračková, Z. Psychiatric Disorders and Polyphenols: Can They Be Helpful in Therapy? Oxidative Med. Cell. Longev. 2015, 2015, 248529. [Google Scholar] [CrossRef] [PubMed]
- Fusar-Poli, L.; Vozza, L.; Gabbiadini, A.; Vanella, A.; Concas, I.; Tinacci, S.; Petralia, A.; Signorelli, M.; Aguglia, E. Curcumin for depression: A meta-analysis. Crit. Rev. Food Sci. Nutr. 2020, 60, 2643–2653. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.; Beidler, J.; Hong, M.Y. Resveratrol and Depression in Animal Models: A Systematic Review of the Biological Mechanisms. Molecules 2018, 23, 2197. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hodes, G.; Zhang, H.; Zhang, S.; Zhao, W.; Golden, S.A.; Bi, W.; Menard, C.; Kana, V.; Leboeuf, M.; et al. Epigenetic modulation of inflammation and synaptic plasticity promotes resilience against stress in mice. Nat. Commun. 2018, 9, 477. [Google Scholar] [CrossRef] [PubMed]
- Ke, F.; Li, H.R.; Chen, X.X.; Gao, X.R.; Huang, L.L.; Du, A.Q.; Jiang, C.; Li, H.; Ge, J.F. Quercetin Alleviates LPS-Induced Depression-like Behavior in Rats via Regulating BDNF-Related Imbalance of Copine 6 and TREM1/2 in the Hippocampus and PFC. Front. Pharmacol. 2020, 10, 1544. [Google Scholar] [CrossRef]
- Fang, J.-L.; Luo, Y.; Jin, S.-H.; Yuan, K.; Guo, Y. Ameliorative effect of anthocyanin on depression mice by increasing monoamine neurotransmitter and up-regulating BDNF expression. J. Funct. Foods 2020, 66, 103757. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef]
- Bhatti, S.K.; O’Keefe, J.; Lavie, C.J. Coffee and tea. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 688–697. [Google Scholar] [CrossRef]
- LIczbiński, P.; Bukowska, B. Tea and coffee polyphenols and their biological properties based on the latest in vitro investigations. Ind. Crop. Prod. 2022, 175, 114265. [Google Scholar] [CrossRef]
- Samanta, S. Potential Bioactive Components and Health Promotional Benefits of Tea (Camellia sinensis). J. Am. Nutr. Assoc. 2022, 41, 65–93. [Google Scholar] [CrossRef]
- Socała, K.; Szopa, A.; Serefko, A.; Poleszak, E.; Wlaź, P. Neuroprotective Effects of Coffee Bioactive Compounds: A Review. Int. J. Mol. Sci. 2020, 22, 107. [Google Scholar] [CrossRef]
- Brandt, P.A.V.D. Coffee or Tea? A prospective cohort study on the associations of coffee and tea intake with overall and cause-specific mortality in men versus women. Eur. J. Epidemiol. 2018, 33, 183–200. [Google Scholar] [CrossRef]
- Grosso, G.; Micek, A.; Castellano, S.; Pajak, A.; Galvano, F. Coffee, tea, caffeine and risk of depression: A systematic review and dose-response meta-analysis of observational studies. Mol. Nutr. Food Res. 2016, 60, 223–234. [Google Scholar] [CrossRef]
- Tsai, A.C.; Chi, S.-H.; Wang, J.-Y. Cross-sectional and longitudinal associations of lifestyle factors with depressive symptoms in ≥53-year old Taiwanese—Results of an 8-year cohort study. Prev. Med. 2013, 57, 92–97. [Google Scholar] [CrossRef]
- Wasim, S.; Kukkar, V.; Awad, V.M.; Sakhamuru, S.; Malik, B.H. Neuroprotective and Neurodegenerative Aspects of Coffee and Its Active Ingredients in View of Scientific Literature. Cureus 2020, 12, e9578. [Google Scholar] [CrossRef]
- Bastianetto, S.; Yao, Z.-X.; Papadopoulos, V.; Quirion, R. Neuroprotective effects of green and black teas and their catechin gallate esters against β-amyloid-induced toxicity. Eur. J. Neurosci. 2006, 23, 55–64. [Google Scholar] [CrossRef]
- Kang, D.; Kim, Y.; Je, Y. Non-alcoholic beverage consumption and risk of depression: Epidemiological evidence from observational studies. Eur. J. Clin. Nutr. 2018, 72, 1506–1516. [Google Scholar] [CrossRef]
- Wang, L.; Shen, X.; Wu, Y.; Zhang, D. Coffee and caffeine consumption and depression: A meta-analysis of observational studies. Aust. N. Z. J. Psychiatry 2016, 50, 228–242. [Google Scholar] [CrossRef]
- Liu, Q.-S.; Deng, R.; Fan, Y.; Li, K.; Meng, F.; Li, X.; Liu, R. Low dose of caffeine enhances the efficacy of antidepressants in major depressive disorder and the underlying neural substrates. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Szopa, A.; Poleszak, E.; Wyska, E.; Serefko, A.; Wośko, S.; Wlaź, A.; Pieróg, M.; Wróbel, A.; Wlaź, P. Caffeine enhances the antidepressant-like activity of common antidepressant drugs in the forced swim test in mice. Naunyn Schmiedebergs Arch. Pharmacol. 2016, 389, 211–221. [Google Scholar] [CrossRef]
- Chuang, Y.-H.; Quach, A.; Absher, D.; Assimes, T.; Horvath, S.; Ritz, B. Coffee consumption is associated with DNA methylation levels of human blood. Eur. J. Hum. Genet. 2017, 25, 608. [Google Scholar] [CrossRef]
- Marques, S.; Batalha, V.L.; Lopes, L.V.; Outeiro, T.F. Modulating Alzheimer’s Disease Through Caffeine: A Putative Link to Epigenetics. J. Alzheimer’s Dis. 2011, 24, 161–171. [Google Scholar] [CrossRef]
- Lopez-Cruz, L.; Salamone, J.D.; Correa, M. Caffeine and Selective Adenosine Receptor Antagonists as New Therapeutic Tools for the Motivational Symptoms of Depression. Front. Pharmacol. 2018, 9, 526. [Google Scholar] [CrossRef]
- Tenore, G.; Daglia, M.; Orlando, V.; D’Urso, E.; Saadat, S.H.; Novellino, E.; Nabavi, S. Coffee and Depression: A Short Review of Literature. Curr. Pharm. Des. 2015, 21, 5034–5040. [Google Scholar] [CrossRef]
- Islam, M.T.; Tabrez, S.; Jabir, N.R.; Ali, M.; Kamal, M.A.; da Silva Araújo, L.; de Oliveira Santos, J.V.; Oliveira Ferreira da Mata, A.M.; Sousa de Aguiar, R.P.; De Carvalho Melo Cavalcante, A.A. An Insight into the Therapeutic Potential of Major Coffee Components. Curr. Drug Metab. 2018, 19, 544–556. [Google Scholar] [CrossRef]
- Li, G.; Yang, J.; Wang, X.; Zhou, C.; Zheng, X.; Lin, W. Effects of EGCG on depression-related behavior and serotonin concentration in a rat model of chronic unpredictable mild stress. Food Funct. 2020, 11, 8780–8787. [Google Scholar] [CrossRef]
- Rothenberg, D.O.; Zhang, L. Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients 2019, 11, 1361. [Google Scholar] [CrossRef]
- Monteiro, Á.B.; Rodrigues, C.K.D.S.; Nascimento, E.P.D.; Sales, V.D.S.; Delmondes, G.D.A.; da Costa, M.H.N.; de Oliveira, V.A.P.; de Morais, L.P.; Boligon, A.A.; Barbosa, R.; et al. Anxiolytic and antidepressant-like effects of Annona coriacea (Mart.) and caffeic acid in mice. Food Chem. Toxicol. 2020, 136, 111049. [Google Scholar] [CrossRef]
- Grzelczyk, J.; Budryn, G.; Peña-García, J.; Szwajgier, D.; Gałązka-Czarnecka, I.; Oracz, J.; Pérez-Sánchez, H. Evaluation of the inhibition of monoamine oxidase A by bioactive coffee compounds protecting serotonin degradation. Food Chem. 2021, 348, 129108. [Google Scholar] [CrossRef]
- Cheng, L.-H.; Liu, Y.-W.; Wu, C.-C.; Wang, S.; Tsai, Y.-C. Psychobiotics in mental health, neurodegenerative and neurodevelopmental disorders. J. Food Drug Anal. 2019, 27, 632–648. [Google Scholar] [CrossRef] [PubMed]
- Del Toro-Barbosa, M.; Hurtado-Romero, A.; Garcia-Amezquita, L.E.; García-Cayuela, T. Psychobiotics: Mechanisms of Action, Evaluation Methods and Effectiveness in Applications with Food Products. Nutrients 2020, 12, 3896. [Google Scholar] [CrossRef] [PubMed]
- Saez-Lara, M.J.; Gomez-Llorente, C.; Plaza-Diaz, J.; Gil, A. The Role of Probiotic Lactic Acid Bacteria and Bifidobacteria in the Prevention and Treatment of Inflammatory Bowel Disease and Other Related Diseases: A Systematic Review of Randomized Human Clinical Trials. BioMed. Res. Int. 2015, 2015, 505878. [Google Scholar] [CrossRef] [PubMed]
- Casertano, M.; Fogliano, V.; Ercolini, D. Psychobiotics, gut microbiota and fermented foods can help preserving mental health. Food Res. Int. 2022, 152, 110892. [Google Scholar] [CrossRef] [PubMed]
- Kok, C.R.; Hutkins, R. Yogurt and other fermented foods as sources of health-promoting bacteria. Nutr. Rev. 2018, 76, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, S. Yogurt Production. Methods Mol. Biol. 2019, 1887, 45–54. [Google Scholar] [CrossRef]
- Le Roy, C.I.; Kurilshikov, A.; Leeming, E.R.; Visconti, A.; Bowyer, R.C.E.; Menni, C.; Falchi, M.; Koutnikova, H.; Veiga, P.; Zhernakova, A.; et al. Yoghurt consumption is associated with changes in the composition of the human gut microbiome and metabolome. BMC Microbiol. 2022, 22, 39. [Google Scholar] [CrossRef]
- Rosa, D.D.; Dias, M.M.S.; Grześkowiak, Ł.M.; Reis, S.A.; Conceição, L.L.; Peluzio, M.D.C.G. Milk Kefir: Nutritional, Microbiological and Health Benefits. Nutr. Res. Rev. 2017, 30, 82–96. [Google Scholar] [CrossRef]
- Bourrie, B.C.T.; Ju, T.; Fouhse, J.M.; Forgie, A.J.; Sergi, C.; Cotter, P.D.; Willing, B.P. Kefir microbial composition is a deciding factor in the physiological impact of kefir in a mouse model of obesity. Br. J. Nutr. 2020, 125, 129–138. [Google Scholar] [CrossRef]
- Benton, D.; Williams, C.; Brown, A. Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur. J. Clin. Nutr. 2007, 61, 355–361. [Google Scholar] [CrossRef]
- Mohammadi, A.A.; Jazayeri, S.; Khosravi-Darani, K.; Solati, Z.; Mohammadpour, N.; Asemi, Z.; Adab, Z.; Djalali, M.; Tehrani-Doost, M.; Hosseini, M.; et al. The effects of probiotics on mental health and hypothalamic–pituitary–adrenal axis: A randomized, double-blind, placebo-controlled trial in petrochemical workers. Nutr. Neurosci. 2016, 19, 387–395. [Google Scholar] [CrossRef]
- Pérez-Cornago, A.; Sánchez-Villegas, A.; Bes-Rastrollo, M.; Gea, A.; Molero, P.; Lahortiga-Ramos, F.; Martínez-González, M.A. Intake of High-Fat Yogurt, but Not of Low-Fat Yogurt or Prebiotics, Is Related to Lower Risk of Depression in Women of the SUN Cohort Study. J. Nutr. 2016, 146, 1731–1739. [Google Scholar] [CrossRef]
- Kim, H.-K.; Kim, S.-H.; Jang, C.-S.; Kim, S.-I.; Kweon, C.-O.; Kim, B.-W.; Ryu, J.-K. The combined effects of yogurt and exercise in healthy adults: Implications for biomarkers of depression and cardiovascular diseases. Food Sci. Nutr. 2018, 6, 1968–1974. [Google Scholar] [CrossRef]
- van de Wouw, M.; Walsh, A.M.; Crispie, F.; Van Leuven, L.; Lyte, J.M.; Boehme, M.; Clarke, G.; Dinan, T.G.; Cotter, P.D.; Cryan, J.F. Distinct actions of the fermented beverage kefir on host behaviour, immunity and microbiome gut-brain modules in the mouse. Microbiome 2020, 8, 67. [Google Scholar] [CrossRef]
- Chen, H.-L.; Lan, Y.-W.; Tu, M.-Y.; Tung, Y.-T.; Chan, M.N.-Y.; Wu, H.-S.; Yen, C.-C.; Chen, C.-M. Kefir peptides exhibit antidepressant-like activity in mice through the BDNF/TrkB pathway. J. Dairy Sci. 2021, 104, 6415–6430. [Google Scholar] [CrossRef]
- Jang, C.; Oh, J.; Lim, J.; Kim, H.; Kim, J.-S. Fermented Soy Products: Beneficial Potential in Neurodegenerative Diseases. Foods 2021, 10, 636. [Google Scholar] [CrossRef]
- Cao, Z.-H.; Green-Johnson, J.M.; Buckley, N.D.; Lin, Q.-Y. Bioactivity of soy-based fermented foods: A review. Biotechnol. Adv. 2019, 37, 223–238. [Google Scholar] [CrossRef]
- Selhub, E.M.; Logan, A.C.; Bested, A.C. Fermented foods, microbiota, and mental health: Ancient practice meets nutritional psychiatry. J. Physiol. Anthr. 2014, 33, 2. [Google Scholar] [CrossRef]
- Perini, G.; Ramusino, M.C.; Sinforiani, E.; Bernini, S.; Petrachi, R.; Costa, A. Cognitive impairment in depression: Recent advances and novel treatments. Neuropsychiatr. Dis. Treat. 2019, 15, 1249. [Google Scholar] [CrossRef]
- Hwang, Y.-H.; Park, S.; Paik, J.-W.; Chae, S.-W.; Kim, D.-H.; Jeong, D.-G.; Ha, E.; Kim, M.; Hong, G.; Park, S.-H.; et al. Efficacy and Safety of Lactobacillus Plantarum C29-Fermented Soybean (DW2009) in Individuals with Mild Cognitive Impairment: A 12-Week, Multi-Center, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2019, 11, 305. [Google Scholar] [CrossRef]
- Ko, C.Y.; Lin, H.-T.V.; Tsai, G.J. Gamma-aminobutyric acid production in black soybean milk by Lactobacillus brevis FPA 3709 and the antidepressant effect of the fermented product on a forced swimming rat model. Process Biochem. 2013, 48, 559–568. [Google Scholar] [CrossRef]
- Lynch, K.M.; Wilkinson, S.; Daenen, L.; Arendt, E.K. An update on water kefir: Microbiology, composition and production. Int. J. Food Microbiol. 2021, 345, 109128. [Google Scholar] [CrossRef]
- de Miranda, J.F.; Ruiz, L.F.; Silva, C.B.; Uekane, T.M.; Silva, K.A.; Gonzalez, A.G.M.; Fernandes, F.F.; Lima, A.R. Kombucha: A review of substrates, regulations, composition, and biological properties. J. Food Sci. 2022, 87, 503–527. [Google Scholar] [CrossRef]
- Abaci, N.; Deniz, F.S.S.; Orhan, I.E. Kombucha—An ancient fermented beverage with desired bioactivities: A narrowed review. Food Chem. X 2022, 14, 100302. [Google Scholar] [CrossRef]
- Wang, P.; Feng, Z.; Sang, X.; Chen, W.; Zhang, X.; Xiao, J.; Chen, Y.; Chen, Q.; Yang, M.; Su, J. Kombucha ameliorates LPS-induced sepsis in a mouse model. Food Funct. 2021, 12, 10263–10280. [Google Scholar] [CrossRef]
- O’Connor, J.C.; Lawson, M.A.; André, C.; Moreau, M.; Lestage, J.; Castanon, N.; Kelley, K.W.; Dantzer, R. Lipopolysaccharide-induced depressive-like behavior is mediated by indoleamine 2,3-dioxygenase activation in mice. Mol. Psychiatry 2009, 14, 511–522. [Google Scholar] [CrossRef]
- Kabiri, N.; Setorki, M. Protective effect of Kombucha tea on brain damage induced by transient cerebral ischemia and reperfusion in rat. Bangladesh J. Pharmacol. 2016, 11, 675–683. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Ortega, M.A.; García-Montero, C.; Fraile-Martinez, O.; Lahera, G.; Monserrat, J.; Gomez-Lahoz, A.M.; Molero, P.; Gutierrez-Rojas, L.; Rodriguez-Jimenez, R.; et al. Differential malondialdehyde (MDA) detection in plasma samples of patients with major depressive disorder (MDD): A potential biomarker. J. Int. Med. Res. 2022, 50, 30006052210949. [Google Scholar] [CrossRef]
- Dudek, K.A.; Dion-Albert, L.; Lebel, M.; LeClair, K.; Labrecque, S.; Tuck, E.; Perez, C.F.; Golden, S.A.; Tamminga, C.; Turecki, G.; et al. Molecular Adaptations of the Blood–Brain Barrier Promote Stress Resilience vs. Depression. Proc. Natl. Acad. Sci. USA 2020, 117, 3326–3336. [Google Scholar] [CrossRef]
- Kapp, J.M.; Sumner, W. Kombucha: A systematic review of the empirical evidence of human health benefit. Ann. Epidemiol. 2019, 30, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.N.S.; Ryu, J.-K.; Kim, Y.; Jeon, B.H. The Effects of Fermented Laminaria japonica on Short-Term Working Memory and Physical Fitness in the Elderly. Evid. Based Complement. Altern. Med. 2018, 2018, 8109621. [Google Scholar] [CrossRef]
- Sarris, J.; Murphy, J.; Mischoulon, D.; Papakostas, G.I.; Fava, M.; Berk, M.; Ng, C.H. Adjunctive Nutraceuticals for Depression: A Systematic Review and Meta-Analyses. Am. J. Psychiatry 2016, 173, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Stubbs, B.; Solmi, M.; Ajnakina, O.; Carvalho, A.F.; Maggi, S. Acetyl-l-Carnitine Supplementation and the Treatment of Depressive Symptoms: A Systematic Review and Meta-Analysis. Psychosom. Med. 2018, 80, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Byrne, G.J.; Stough, C.; Bousman, C.; Mischoulon, D.; Murphy, J.; Macdonald, P.; Adams, L.; Nazareth, S.; Oliver, G.; et al. Nutraceuticals for major depressive disorder- more is not merrier: An 8-week double-blind, randomised, controlled trial. J. Affect. Disord. 2019, 245, 1007–1015. [Google Scholar] [CrossRef]
- Bot, M.; Brouwer, I.; Roca, M.; Kohls, E.; Penninx, B.W.J.H.; Watkins, E.; van Grootheest, G.; Cabout, M.; Hegerl, U.; Gili, M.; et al. Effect of Multinutrient Supplementation and Food-Related Behavioral Activation Therapy on Prevention of Major Depressive Disorder Among Overweight or Obese Adults with Subsyndromal Depressive Symptoms. JAMA 2019, 321, 858–868. [Google Scholar] [CrossRef]
- Haller, H.; Anheyer, D.; Cramer, H.; Dobos, G. Complementary therapies for clinical depression: An overview of systematic reviews. BMJ Open 2019, 9, e028527. [Google Scholar] [CrossRef]
- Ng, Q.X.; Venkatanarayanan, N.; Ho, C.Y.X. Clinical use of Hypericum perforatum (St John’s wort) in depression: A meta-analysis. J. Affect. Disord. 2017, 210, 211–221. [Google Scholar] [CrossRef]
- Kholghi, G.; Arjmandi-Rad, S.; Zarrindast, M.-R.; Vaseghi, S. St. John’s wort (Hypericum perforatum) and depression: What happens to the neurotransmitter systems? Naunyn-Schmiedeberg’s Arch. Pharmacol. 2022, 395, 629–642. [Google Scholar] [CrossRef]
- Zirak, N.; Shafiee, M.; Soltani, G.; Mirzaei, M.; Sahebkar, A. Hypericum perforatum in the treatment of psychiatric and neurodegenerative disorders: Current evidence and potential mechanisms of action. J. Cell. Physiol. 2019, 234, 8496–8508. [Google Scholar] [CrossRef]
- Liu, L.; Liu, C.; Wang, Y.; Wang, P.; Li, Y.; Li, B. Herbal Medicine for Anxiety, Depression and Insomnia. Curr. Neuropharmacol. 2015, 13, 481. [Google Scholar] [CrossRef]
- Silvers, K.M.; Woolley, C.C.; Hedderley, D. Dietary Supplement Use in People Being Treated for Depression. Asia Pac. J. Clin. Nutr. 2006, 15, 30–34. [Google Scholar]
- Booker, A.; Agapouda, A.; Frommenwiler, D.A.; Scotti, F.; Reich, E.; Heinrich, M. St John’s wort (Hypericum perforatum) products—An assessment of their authenticity and quality. Phytomedicine 2018, 40, 158–164. [Google Scholar] [CrossRef]
- Simon, L.V.; Keenaghan, M. Serotonin Syndrome. Pain Med. Essent. Rev. 2022, 11, 201–202. [Google Scholar]
- Patel, Y.A.; Marzella, N. Dietary Supplement-Drug Interaction-Induced Serotonin Syndrome Progressing to Acute Compartment Syndrome. Am. J. Case Rep. 2017, 18, 926. [Google Scholar] [CrossRef]
- Ronis, M.J.; Pedersen, K.B.; Watt, J. Adverse Effects of Nutraceuticals and Dietary Supplements. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 583–601. [Google Scholar] [CrossRef]
- Li, Y.; Lv, M.-R.; Wei, Y.-J.; Sun, L.; Zhang, J.-X.; Zhang, H.-G.; Li, B. Dietary patterns and depression risk: A meta-analysis. Psychiatry Res. 2017, 253, 373–382. [Google Scholar] [CrossRef]
- Tolkien, K.; Bradburn, S.; Murgatroyd, C. An anti-inflammatory diet as a potential intervention for depressive disorders: A systematic review and meta-analysis. Clin. Nutr. 2019, 38, 2045–2052. [Google Scholar] [CrossRef]
- Dominguez, L.; Di Bella, G.; Veronese, N.; Barbagallo, M. Impact of Mediterranean Diet on Chronic Non-Communicable Diseases and Longevity. Nutrients 2021, 13, 2028. [Google Scholar] [CrossRef]
- Arpón, A.; Milagro, F.I.; Razquin, C.; Corella, D.; Estruch, R.; Fitó, M.; Marti, A.; Martínez-González, M.A.; Ros, E.; Salas-Salvadó, J.; et al. Impact of Consuming Extra-Virgin Olive Oil or Nuts within a Mediterranean Diet on DNA Methylation in Peripheral White Blood Cells within the PREDIMED-Navarra Randomized Controlled Trial: A Role for Dietary Lipids. Nutrients 2017, 10, 15. [Google Scholar] [CrossRef]
- Sánchez-Villegas, A.; Álvarez-Pérez, J.; Toledo, E.; Salas-Salvadó, J.; Ortega-Azorín, C.; Zomeño, M.D.; Vioque, J.; Martínez, J.A.; Romaguera, D.; Pérez-López, J.; et al. Seafood Consumption, Omega-3 Fatty Acids Intake, and Life-Time Prevalence of Depression in the PREDIMED-Plus Trial. Nutrients 2018, 10, 2000. [Google Scholar] [CrossRef]
- Shafiei, F.; Moghaddam, A.S.; Larijani, B.; Esmaillzadeh, A. Adherence to the Mediterranean diet and risk of depression: A systematic review and updated meta-analysis of observational studies. Nutr. Rev. 2019, 77, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Sergentanis, T.N.; Panagiotakos, D.B.; Sergentanis, I.N.; Kosti, R.; Scarmeas, N. Mediterranean diet, stroke, cognitive impairment, and depression: A meta-analysis. Ann. Neurol. 2013, 74, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Hershey, M.S.; Sanchez-Villegas, A.; Sotos-Prieto, M.; Fernandez-Montero, A.; Pano, O.; Lahortiga-Ramos, F.; Martínez-González, M.; Ruiz-Canela, M. The Mediterranean Lifestyle and the Risk of Depression in Middle-Aged Adults. J. Nutr. 2022, 152, 227–234. [Google Scholar] [CrossRef]
- Jacka, F.N.; O’Neil, A.; Opie, R.; Itsiopoulos, C.; Cotton, S.; Mohebbi, M.; Castle, D.; Dash, S.; Mihalopoulos, C.; Chatterton, M.L.; et al. A randomised controlled trial of dietary improvement for adults with major depression (the ‘SMILES’ trial). BMC Med. 2017, 15, 23. [Google Scholar] [CrossRef]
- Opie, R.; O’Neil, A.; Jacka, F.N.; Pizzinga, J.; Itsiopoulos, C. A modified Mediterranean dietary intervention for adults with major depression: Dietary protocol and feasibility data from the SMILES trial. Nutr. Neurosci. 2018, 21, 487–501. [Google Scholar] [CrossRef]
- Foshati, S.; Ghanizadeh, A.; Akhlaghi, M. Extra-Virgin Olive Oil Improves Depression Symptoms Without Affecting Salivary Cortisol and Brain-Derived Neurotrophic Factor in Patients with Major Depression: A Double-Blind Randomized Controlled Trial. J. Acad. Nutr. Diet. 2022, 122, 284–297.e1. [Google Scholar] [CrossRef] [PubMed]
- Oh, R.; Gilani, B.; Uppaluri, K.R. Low Carbohydrate Diet. In StatPearls; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Dąbek, A.; Wojtala, M.; Pirola, L.; Balcerczyk, A. Modulation of Cellular Biochemistry, Epigenetics and Metabolomics by Ketone Bodies. Implications of the Ketogenic Diet in the Physiology of the Organism and Pathological States. Nutrients 2020, 12, 788. [Google Scholar] [CrossRef] [PubMed]
- Brietzke, E.; Mansur, R.B.; Subramaniapillai, M.; Balanzá-Martínez, V.; Vinberg, M.; González-Pinto, A.; Rosenblat, J.D.; Ho, R.; McIntyre, R.S. Ketogenic diet as a metabolic therapy for mood disorders: Evidence and developments. Neurosci. Biobehav. Rev. 2018, 94, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Winwood-Smith, H.S.; Franklin, C.E.; White, C.R. Low-carbohydrate diet induces metabolic depression: A possible mechanism to conserve glycogen. Am. J. Physiol. Integr. Comp. Physiol. 2017, 313, R347–R356. [Google Scholar] [CrossRef]
- Igwe, O.; Sone, M.; Matveychuk, D.; Baker, G.B.; Dursun, S.M. A review of effects of calorie restriction and fasting with potential relevance to depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 111, 110206. [Google Scholar] [CrossRef]
- Berthelot, E.; Etchecopar-Etchart, D.; Thellier, D.; Lancon, C.; Boyer, L.; Fond, G. Fasting Interventions for Stress, Anxiety and Depressive Symptoms: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3947. [Google Scholar] [CrossRef]
- Alcorta, A.; Porta, A.; Tárrega, A.; Alvarez, M.D.; Pilar Vaquero, M. Foods for Plant-Based Diets: Challenges and Innovations. Foods 2021, 10, 293. [Google Scholar] [CrossRef]
- Medawar, E.; Huhn, S.; Villringer, A.; Witte, A.V. The effects of plant-based diets on the body and the brain: A systematic review. Transl. Psychiatry 2019, 9, 226. [Google Scholar] [CrossRef]
- Tiffon, C. The Impact of Nutrition and Environmental Epigenetics on Human Health and Disease. Int. J. Mol. Sci. 2018, 19, 3425. [Google Scholar] [CrossRef]
- Iguacel, I.; Huybrechts, I.; Moreno, L.A.; Michels, N. Vegetarianism and veganism compared with mental health and cognitive outcomes: A systematic review and meta-analysis. Nutr. Rev. 2021, 79, 361–381. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Borawski, J. Vegetarian diet and depression scores: A meta-analysis. J. Affect. Disord. 2021, 294, 813–815. [Google Scholar] [CrossRef]
- Guzek, D.; Gła¸bska, D.; Groele, B.; Gutkowska, K. Fruit and Vegetable Dietary Patterns and Mental Health in Women: A Systematic Review. Nutr. Rev. 2022, 80, 1357–1370. [Google Scholar] [CrossRef]
- Barnard, N.D.; Leroy, F. Children and adults should avoid consuming animal products to reduce risk for chronic disease: YES. Am. J. Clin. Nutr. 2020, 112, 926–930. [Google Scholar] [CrossRef]
- Lee, M.F.; Eather, R.; Best, T. Plant-based dietary quality and depressive symptoms in Australian vegans and vegetarians: A cross-sectional study. BMJ Nutr. Prev. Health 2021, 4, e000332. [Google Scholar] [CrossRef]
- Sangsefidi, Z.S.; Mirzaei, M.; Hosseinzadeh, M. The relation between dietary intakes and psychological disorders in Iranian adults: A population-based study. BMC Psychiatry 2020, 20, 257. [Google Scholar] [CrossRef]
- Dobersek, U.; Wy, G.; Adkins, J.; Altmeyer, S.; Krout, K.; Lavie, C.J.; Archer, E. Meat and mental health: A systematic review of meat abstention and depression, anxiety, and related phenomena. Crit. Rev. Food Sci. Nutr. 2021, 61, 622–635. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Fogelholm, M.; Jalo, E.; Poppitt, S.D.; Silvestre, M.P.; Møller, G.; Huttunen-Lenz, M.; Stratton, G.; Sundvall, J.; Macdonald, I.A.; et al. Animal-based food choice and associations with long-term weight maintenance and metabolic health after a large and rapid weight loss: The PREVIEW study. Clin. Nutr. 2022, 41, 817–828. [Google Scholar] [CrossRef] [PubMed]
| Nutrients | Food | Intake Recommendations (Food) | Antidepressant Mechanisms | Studies in MDD | References |
|---|---|---|---|---|---|
| Omega 3 PUFA | Fish and seafood (DHA and EPA), seeds and nuts (ALA) | Two servings of fish per week or 350 g weekly including in which 200 g may be from oily fishes. 30 g/day of mixed nuts (15 g walnuts, 7.5 g hazelnuts, and 7.5 g almonds) comprised in a healthy dietary pattern appears to be an important recommendation to prevent MDD. | Epigenetic modulation; Anti-inflammatory; Prebiotic; Increase in membrane fluidity; serotonin transport; enhanced dopamine concentration and dopamine 2 receptor in the frontal cortex; cellular signaling. | DHA and EPA either by supplementation or contained in a high intake of fish (≥2 times per week exerted relevant protective and antidepressant effects. ALA contained in seeds and nuts (especially walnuts) can ameliorate depressive symptoms and prevent their onset. | [78,79,80,81,82,83,84,85,86,87,88,89,90] |
| Vitamin D | Fish and animal products; mushrooms; fortified products | Two servings of fish per week or 350 g weekly including in which 200 g may be from oily fishes. | Vitamin D influences the immune and gut microbiota modulation; Serotonin synthesis; Circadian clock regulation and augmented BDNF production, exerting synergic effects with omega 3 PUFA. | Controversial results have been obtained regarding the possible antidepressant effect of vitamin D intake; although, its low consumption worldwide can be involved in the high prevalence of MDD. | [91,92,93,94,95,96,97,98,99] |
| Vitamins from the B complex | Fruits and vegetables/Animal products or supplementation (B12) | 800 g per day of fruits and vegetables are recommended as part of a healthy dietary pattern. | Anti-inflammatory and pleiotropic actions. | Low levels of B1, B2, B3, B6, B9, and B12 vitamins can be related to a proinflammatory status of the immune system. Some studies have found slight benefits from supplementing with B6 or B12 alone or integrated into the B complex. However, daily intake above 400 g of fruits or vegetables is the most adequate approach to elevate the serum levels of vitamins | [114,115,116,117,118] |
| Other vitamins (A, C, E, K) | Fruits and vegetables, animal products | 800 g per day of fruits and vegetables are recommended as part of a healthy dietary pattern. | Antioxidant, anti-inflammatory, and pleiotropic effects | There is some preclinical evidence supporting the supplementation with provitamin A and E as antidepressants; Vitamin C has been explored as an adjuvant with fluoxetine in children but not in adults; High intake of vitamin K (>232 μg/day) had significantly lower odds of developing depressive symptoms at baseline and each per 100 μg/day the odds of this condition decreased by12%. Daily intake above 400 g of fruits or vegetables is the most adequate approach to elevate the serum levels of vitamins. | [114,118,119,120] |
| Dietary fiber | Fruits and vegetables | 800 g per day of fruits and vegetables are recommended as part of a healthy dietary pattern. | Prebiotic and modulatory effects in the MGB axis; SCFA modulation. | 1 g of fiber per 1000 kcal exerts protective actions of fiber against depression in premenopausal, but not postmenopausal women. | [108,109] |
| Polyphenols | Fruits and vegetables/Coffee and tea | 800 g per day of fruits and vegetables are recommended as part of a healthy dietary pattern. A nonlinear J shape curve with a peak on 400 mL of coffee/day and more than 3 cups of tea per week. | Pleiotropic effects | 1000 mg/day of curcumin but not 500 mg/day may be used as adjunctive therapy for patients with MDD Preclinical models have shown that doses between 10 and 80 mg/kg/day, of resveratrol, provide antidepressant effects; although, higher doses had the most significant benefits. Quercetin has provided a potential antidepressant role in vivo, ameliorating lipopolysaccharide (LPS)-induced depressive rats, leading to the upregulation of BDNF and other molecular markers. Anthocyanins, mainly found in berries, are being investigated in mice models as prominent antidepressants, upregulating monoamines and neurotrophic factors such as BDNF. EGCG has proven to have some critical antidepressant effects in preclinical models, leading to an augmented BDNF, serotonin, and reduced stress hormones. Caffeic acid also provides significant antidepressant effects in vivo, especially when combined with antidepressants. Chlorogenic acid found in green coffee appears to be a major inhibitor of the monoamine oxidase A (MAO-A), upregulating serotonin levels and providing antidepressant actions. | [121,122,123,124,125,126,127,128,129] |
| Caffeine | Coffee and tea | A nonlinear J shape curve with a peak on 400 mL of coffee/day and more than 3 cups of tea per week. | Neuroprotective; Epigenetic modulation of neurons, immune and glial cells; Targeting of the dopaminergic system through the non-selective adenosine antagonist action. | A nonlinear response between caffeine consumption and depression was found, showing their most benefits above 68 mg/day and below 509 mg/day. | [137,138,139,140,141,142,143,144,145,146,147] |
| Psychobiotics | Fermented foods and beverages (Yogurt, kefir, soy-derived products, kombucha, Laminaria japonica, etc.) | Fermented foods can be included in a healthy dietary pattern daily. | Modulatory effects on HPA and MGB axis; inflammation; neurotransmitter production (monoamines, GABA, glutamate, acetylcholine); BDNF levels; host metabolism. | 3 weeks with daily consumption of probiotic yogurt improved the mood in people with poor mood and other significant benefits on mental health by positively modulating the HPA axis; Whole-fat yogurt, but not low fat, was related to a decreased risk of depression in women. Kefir and kefir peptides appear to be a promising antidepressant tested in vivo, displaying a major modulatory role in the MGB axis, influencing host behavior, serotonin synthesis, immune system, BDNF/TrkB signaling, and GABA levels High adherence to soy-based fermented products in traditional Japanese regimes is associated with lower rates of depressive symptoms, aiding in the clinical management of depression and cognitive impairment; Soy-based milk with Lactobacillus brevis FPA 3709 (1 × 106 CFU/mL) demonstrated antidepressant efficacy in SD rats similar to fluoxetine. Kombucha can attenuate LPS-mediated neuroinflammation and oxidative stress markers, but direct evidence of its antidepressant role is needed. | [152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega, M.A.; Fraile-Martínez, Ó.; García-Montero, C.; Alvarez-Mon, M.A.; Lahera, G.; Monserrat, J.; Llavero-Valero, M.; Gutiérrez-Rojas, L.; Molina, R.; Rodríguez-Jimenez, R.; et al. Biological Role of Nutrients, Food and Dietary Patterns in the Prevention and Clinical Management of Major Depressive Disorder. Nutrients 2022, 14, 3099. https://doi.org/10.3390/nu14153099
Ortega MA, Fraile-Martínez Ó, García-Montero C, Alvarez-Mon MA, Lahera G, Monserrat J, Llavero-Valero M, Gutiérrez-Rojas L, Molina R, Rodríguez-Jimenez R, et al. Biological Role of Nutrients, Food and Dietary Patterns in the Prevention and Clinical Management of Major Depressive Disorder. Nutrients. 2022; 14(15):3099. https://doi.org/10.3390/nu14153099
Chicago/Turabian StyleOrtega, Miguel A., Óscar Fraile-Martínez, Cielo García-Montero, Miguel Angel Alvarez-Mon, Guillermo Lahera, Jorge Monserrat, Maria Llavero-Valero, Luis Gutiérrez-Rojas, Rosa Molina, Roberto Rodríguez-Jimenez, and et al. 2022. "Biological Role of Nutrients, Food and Dietary Patterns in the Prevention and Clinical Management of Major Depressive Disorder" Nutrients 14, no. 15: 3099. https://doi.org/10.3390/nu14153099
APA StyleOrtega, M. A., Fraile-Martínez, Ó., García-Montero, C., Alvarez-Mon, M. A., Lahera, G., Monserrat, J., Llavero-Valero, M., Gutiérrez-Rojas, L., Molina, R., Rodríguez-Jimenez, R., Quintero, J., & De Mon, M. A. (2022). Biological Role of Nutrients, Food and Dietary Patterns in the Prevention and Clinical Management of Major Depressive Disorder. Nutrients, 14(15), 3099. https://doi.org/10.3390/nu14153099












