Women Taking a Folic Acid Supplement in Countries with Mandatory Food Fortification Programs May Be Exceeding the Upper Tolerable Limit of Folic Acid: A Systematic Review
Abstract
1. Introduction
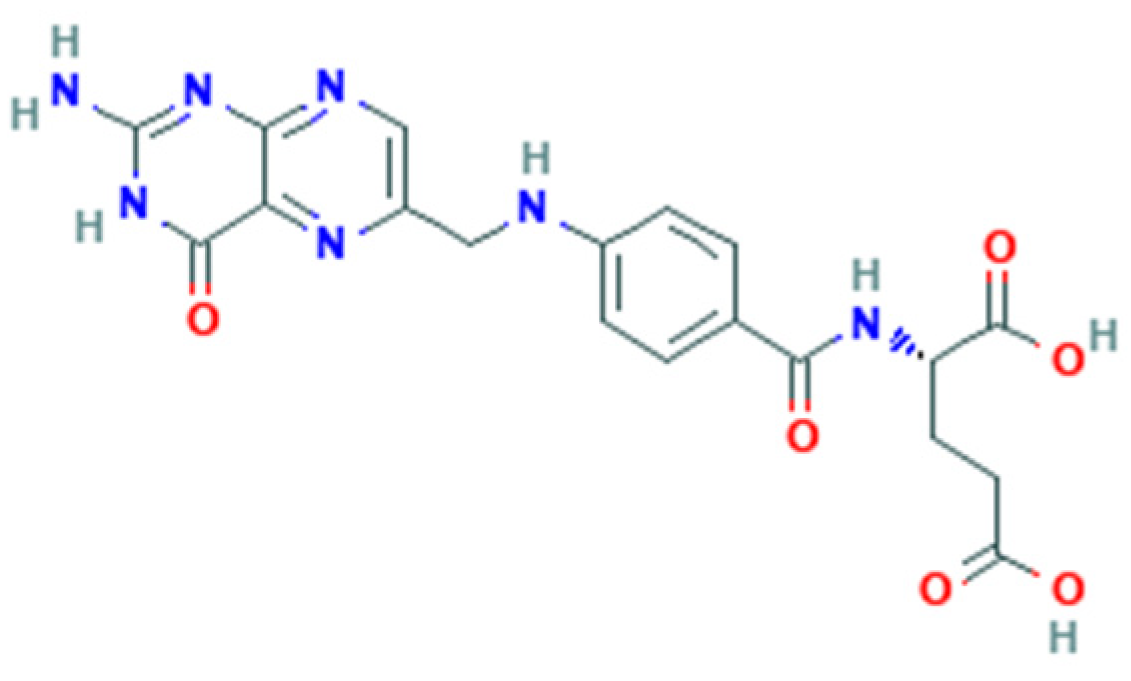
1.1. Folate Metabolism
1.2. Folic Acid Dose
1.3. Upper Tolerable Limit of Folic Acid
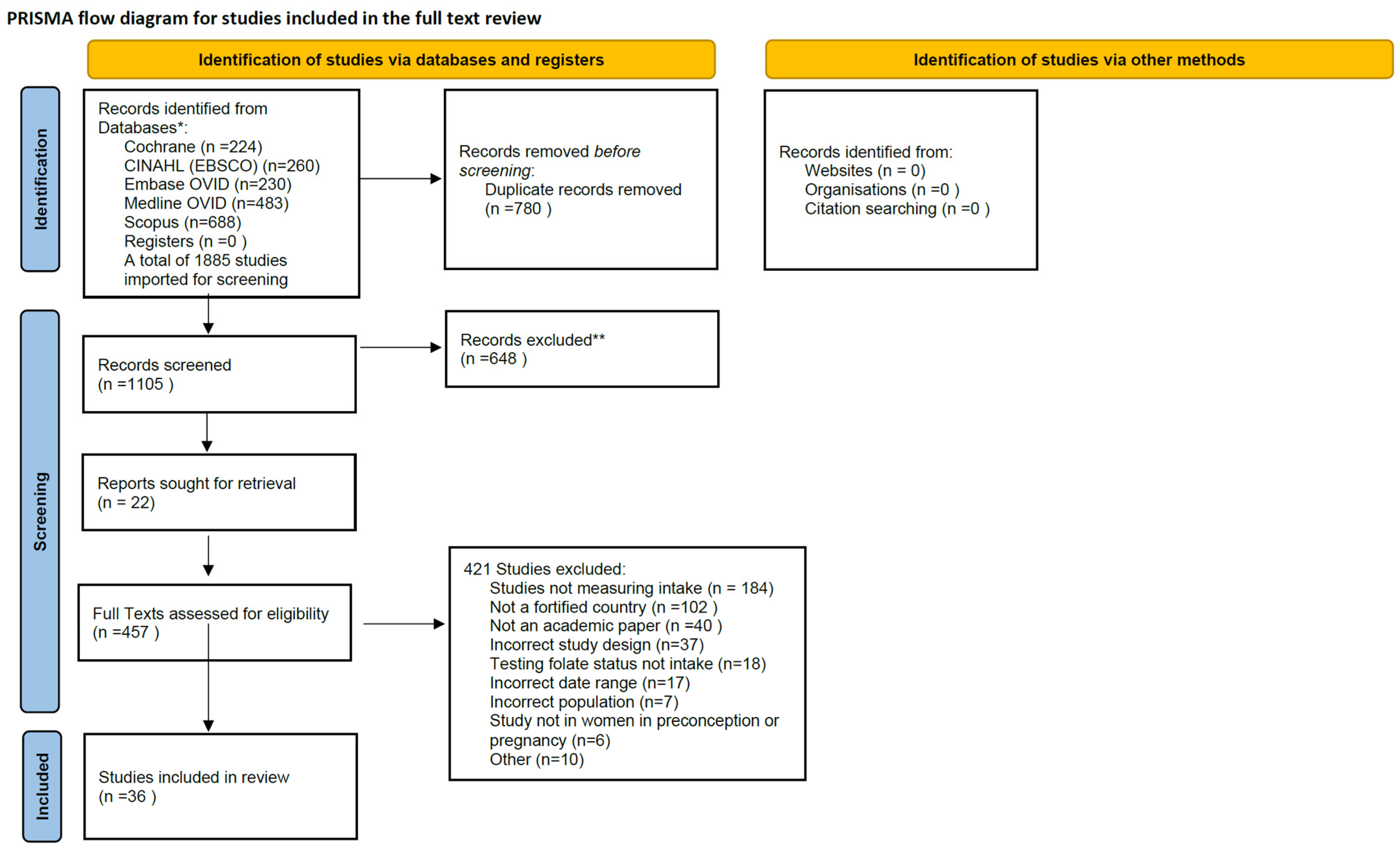
2. Materials and Methods
3. Results
3.1. Study Characteristics
3.1.1. Women of Childbearing Age
3.1.2. Pregnant Women
3.2. Natural Food Folate and Fortification
3.3. Supplementation and Upper Tolerable Limit
4. Discussion
4.1. Natural Food Intake and Fortification
4.2. Folate Measurement
4.3. Folate Metabolism
4.4. Unmetabolized Folic Acid
4.5. Upper Tolerable Limit
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Cena, E.R.; Joy, A.B.; Heneman, K.; Espinosa-Hall, G.; Garcia, L.; Schneider, C.; Wooten Swanson, P.C.; Hudes, M.; Zidenberg-Cherr, S. Folate Intake and Food-Related Behaviors in Nonpregnant, Low-Income Women of Childbearing Age. J. Am. Diet. Assoc. 2008, 108, 1364–1368. [Google Scholar] [CrossRef]
- Tinker, S.C.; Hamner, H.C.; Berry, R.J.; Bailey, L.B.; Pfeiffer, C.M. Does obesity modify the association of supplemental folic acid with folate status among nonpregnant women of childbearing age in the United States? Birth Defects Res. 2012, 94, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, M.; Brown, C.J.P.; Block, G. The effect of folate fortification of cereal-grain products on blood folate status, dietary folate intake, and dietary folate sources among adult non-supplement users in the United States. J. Am. Coll. Nutr. 2005, 24, 266–274. [Google Scholar] [CrossRef] [PubMed]
- French, M.R.; Barr, S.I.; Levy-Milne, R. Folate intakes and awareness of folate to prevent neural tube defects: A survey of women living in Vancouver, Canada. J. Am. Diet. Assoc. 2003, 103, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Mumford, S.L.; Chavarro, J.E.; Zhang, C.; Pollack, A.Z.; Wactawski-Wende, J.; Perkins, N.J.; Schisterman, E.F. The Impact of Dietary Folate Intake on Reproductive Function in Premenopausal Women: A Prospective Cohort Study. PLoS ONE 2012, 7, e46276. [Google Scholar] [CrossRef]
- Department of Health and Ageing. Nutrient Reference Values for Australia and New Zealand; Australian National Health and Medical Research Council, Ed.; Department of Health and Ageing: Canberra, Australia, 2006. [Google Scholar]
- Centers for Disease Control and Prevention. Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects. Morb. Mortal. Wkly. Rep. 1992, 41, 1–7. [Google Scholar]
- Bailey, L.B. Folate and vitamin B12 recommended intakes and status in the United States...including summary. Nutr. Rev. 2004, 62, S14–S21. [Google Scholar] [CrossRef]
- Czeizel, A.E. Primary prevention of neural-tube defects and some other major congenital abnormalities: Recommendations for the appropriate use of folic acid during pregnancy. Paediatr. Drugs 2000, 2, 437–449. [Google Scholar] [CrossRef]
- Institute of Medicine (US); Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press (US): Washington, DC, USA, 1998. [Google Scholar] [CrossRef]
- Food and Drug Administration. Food standards: Amendment of standards of identity for enriched grain products to require addition of folic acid; final rule (21 CFR Parts 136, 137, and 139). Fed. Regist. 1996, 61, 8781–8797. [Google Scholar]
- MRC Vitamin Study Research Group. Prevention of neural tube defects: Results of the Medical Research Council Vitamin Study. Lancet 1991, 338, 131–137. [Google Scholar] [CrossRef]
- Maruvada, P.; Stover, P.J.; Mason, J.B.; Bailey, R.L.; Davis, C.D.; Field, M.S.; Finnell, R.H.; Garza, C.; Green, R.; Gueant, J.-L.; et al. Knowledge gaps in understanding the metabolic and clinical effects of excess folates/folic acid: A summary, and perspectives, from an NIH workshop. Am. J. Clin. Nutr. 2020, 112, 1390–1403. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.R.; McPartlin, J.; Scott, J. Folic acid fortification and public health: Report on threshold doses above which unmetabolised folic acid appear in serum. BMC Public Health 2007, 7, 41. [Google Scholar] [CrossRef]
- Sweeney, M.R.; Staines, A.; Daly, L.; Traynor, A.; Daly, S.; Bailey, S.W.; Alverson, P.B.; Ayling, J.E.; Scott, J.M. Persistent circulating unmetabolised folic acid in a setting of liberal voluntary folic acid fortification. Implications for further mandatory fortification? BMC Public Health 2009, 9, 295. [Google Scholar] [CrossRef]
- Obeid, R.; Herrmann, W. The emerging role of unmetabolized folic acid in human diseases: Myth or reality? Curr. Drug Metab. 2012, 13, 1184–1195. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Kasoha, M.; Kirsch, S.H.; Munz, W.; Herrmann, W. Concentrations of unmetabolized folic acid and primary folate forms in pregnant women at delivery and in umbilical cord blood. Am. J. Clin. Nutr. 2010, 92, 1416–1422. [Google Scholar] [CrossRef] [PubMed]
- Houghton, L.A.; Yang, J.; O’Connor, D.L. Unmetabolized folic acid and total folate concentrations in breast milk are unaffected by low-dose folate supplements. Am. J. Clin. Nutr. 2009, 89, 216–220. [Google Scholar] [CrossRef]
- Page, R.; Robichaud, A.; Arbuckle, T.E.; Fraser, W.D.; MacFarlane, A.J. Total folate and unmetabolized folic acid in the breast milk of a cross-section of Canadian women. Am. J. Clin. Nutr. 2017, 105, 1101–1109. [Google Scholar] [CrossRef]
- Page, R.; Wong, A.; Arbuckle, T.E.; MacFarlane, A.J. The MTHFR 677C>T polymorphism is associated with unmetabolized folic acid in breast milk in a cohort of Canadian women. Am. J. Clin. Nutr. 2019, 110, 401–409. [Google Scholar] [CrossRef]
- Smith, A.D.; Kim, Y.-I.; Refsum, H. Is folic acid good for everyone? Am. J. Clin. Nutr. 2008, 87, 517–533. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, J.O.; Shim, S.H.; Lee, Y.; Kim, J.H.; Jeon, Y.J.; Ko, J.J.; Lee, W.S.; Kim, N.K. Genetic Variation of Methylenetetrahydrofolate Reductase (MTHFR) and Thymidylate Synthase (TS) Genes Is Associated with Idiopathic Recurrent Implantation Failure. PLoS ONE 2016, 11, e0160884. [Google Scholar] [CrossRef]
- Cochrane, K.M.; Mayer, C.; Devlin, A.M.; Elango, R.; Hutcheon, J.A.; Karakochuk, C.D. Is natural (6 S)-5-methyltetrahydrofolic acid as effective as synthetic folic acid in increasing serum and red blood cell folate concentrations during pregnancy? A proof-of-concept pilot study. Trials 2020, 21, 380. [Google Scholar] [CrossRef] [PubMed]
- Scaglione, F.; Panzavolta, G. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica 2014, 44, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Holzgreve, W.; Pietrzik, K. Is 5-methyltetrahydrofolate an alternative to folic acid for the prevention of neural tube defects? J. Perinat. Med. 2013, 41, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. The metabolic basis for developmental disorders due to defective folate transport. Biochimie 2016, 126, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Shane, B.; Bailey, L. Folate chemistry and metabolism. Folate Health Dis. 1995, 2, 1–19. [Google Scholar]
- Pietrzik, K.; Bailey, L.; Shane, B. Folic acid and L-5-methyltetrahydrofolate. Clin. Pharmacokinet. 2010, 49, 535–548. [Google Scholar] [CrossRef]
- Qiu, A.; Jansen, M.; Sakaris, A.; Min, S.H.; Chattopadhyay, S.; Tsai, E.; Sandoval, C.; Zhao, R.; Akabas, M.H.; Goldman, I.D. Identification of an intestinal folate transporter and the molecular basis for hereditary folate malabsorption. Cell 2006, 127, 917–928. [Google Scholar] [CrossRef]
- Shane, B. Folate Fortification: Enough Already? Am. J. Clin. Nutr. 2003, 77, 8–9. [Google Scholar] [CrossRef]
- Patanwala, I.; King, M.J.; Barrett, D.A.; Rose, J.; Jackson, R.; Hudson, M.; Philo, M.; Dainty, J.R.; Wright, A.J.; Finglas, P.M. Folic acid handling by the human gut: Implications for food fortification and supplementation. Am. J. Clin. Nutr. 2014, 100, 593–599. [Google Scholar] [CrossRef]
- Steegers-Theunissen, R.P.M.; Twigt, J.; Pestinger, V.; Sinclair, K.D. The periconceptional period, reproduction and long-term health of offspring: The importance of one-carbon metabolism. Hum. Reprod. Update 2013, 19, 640–655. [Google Scholar] [CrossRef]
- Crider, K.S.; Qi, Y.P.; Devine, O.; Tinker, S.C.; Berry, R.J. Modeling the impact of folic acid fortification and supplementation on red blood cell folate concentrations and predicted neural tube defect risk in the United States: Have we reached optimal prevention? Am. J. Clin. Nutr. 2018, 107, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Botto, L.D.; Yang, Q. 5,10-Methylenetetrahydrofolate reductase gene variants and congenital anomalies: A HuGE review. Am. J. Epidemiol. 2000, 151, 862–877. [Google Scholar] [CrossRef] [PubMed]
- Sah, A.K.; Shrestha, N.; Joshi, P.; Lakha, R.; Shrestha, S.; Sharma, L.; Chandra, A.; Singh, N.; Kc, Y.; Rijal, B. Association of parental methylenetetrahydrofolate reductase (MTHFR) C677T gene polymorphism in couples with unexplained recurrent pregnancy loss. BMC Res. Notes 2018, 11, 233. [Google Scholar] [CrossRef] [PubMed]
- Vanilla, S.; Dayanand, C.D.; Kotur, P.F.; Moideen Kutty, A.; Vegi, P.K. Evidence of paternal N5, N10—methylenetetrahydrofolate reductase (MTHFR) C677T gene polymorphism in couples with recurrent spontaneous Abortions (RSAs) in Kolar district—A south west of India. J. Clin. Diagn. Res. 2015, 9, BC15–BC18. [Google Scholar] [CrossRef] [PubMed]
- Altmae, S.; Stavreus-Evers, A.; Ruiz, J.R.; Laanpere, M.; Syvanen, T.; Yngve, A.; Salumets, A.; Nilsson, T.K. Variations in folate pathway genes are associated with unexplained female infertility. Fertil. Steril. 2010, 94, 130–137. [Google Scholar] [CrossRef]
- Gupta, N.; Sarkar, S.; David, A.; Gangwar, P.K.; Gupta, R.; Khanna, G.; Sankhwar, S.N.; Khanna, A.; Rajender, S. Significant impact of the MTHFR polymorphisms and haplotypes on male infertility risk. PLoS ONE 2013, 8, e69180. [Google Scholar] [CrossRef]
- Enciso, M.; Sarasa, J.; Xanthopoulou, L.; Bristow, S.; Bowles, M.; Fragouli, E.; Delhanty, J.; Wells, D. Polymorphisms in the MTHFR gene influence embryo viability and the incidence of aneuploidy. Hum. Genet. 2016, 135, 555–568. [Google Scholar] [CrossRef]
- Gava, M.M.; De Oliveira Chagas, E.; Bianco, B.; Christofolini, D.M.; Pompeo, A.C.L.; Glina, S.; Barbosa, C.P. Methylenetetrahydrofolate reductase polymorphisms are related to male infertility in Brazilian men. Genet. Test. Mol. Biomark. 2011, 15, 153–157. [Google Scholar] [CrossRef]
- Gava, M.M.; Kayaki, E.A.; Bianco, B.; Teles, J.S.; Christofolini, D.M.; Pompeo, A.C.; Glina, S.; Barbosa, C.P. Polymorphisms in folate-related enzyme genes in idiopathic infertile Brazilian men. Reprod. Sci. 2011, 18, 1267–1272. [Google Scholar] [CrossRef]
- Hong, H.H.; Hu, Y.; Yu, X.Q.; Zhou, L.; Lv, M.Q.; Sun, Y.; Ren, W.J.; Zhou, D.X. Associations of C677T polymorphism in methylenetetrahydrofolate reductase (MTHFR) gene with male infertility risk: A meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 212, 101–109. [Google Scholar] [CrossRef]
- Ledowsky, C.; Steel, A.; Schloss, J. Methylenetetrahydrofolate Reductase (MTHFR) genetic polymorphisms and the risk of infertility in couples accessing Assisted Reproductive technologies. Adv. Integr. Med. 2021, 8, 220–229. [Google Scholar] [CrossRef]
- Ferrazzi, E.; Tiso, G.; Di Martino, D. Folic acid versus 5-methyl tetrahydrofolate supplementation in pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 253, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.P.; Savella, G.M.; Church, T.R.; Goez-Mogollon, L.; Sosinsky, A.Z.; Noe, O.B.; Kaimal, A.; Cohen, L.S. A prenatal supplement with methylfolate for the treatment and prevention of depression in women trying to conceive and during pregnancy. Ann. Clin. Psychiatry 2019, 31, 4–16. [Google Scholar]
- Servy, E.J.; Jacquesson-Fournols, L.; Cohen, M.; Menezo, Y.J. MTHFR isoform carriers. 5-MTHF (5-methyl tetrahydrofolate) vs folic acid: A key to pregnancy outcome: A case series. J. Assist. Reprod. Genet. 2018, 35, 1431–1435. [Google Scholar] [CrossRef] [PubMed]
- Hekmatdoost, A.; Vahid, F.; Yari, Z.; Sadeghi, M.; Eini-Zinab, H.; Lakpour, N.; Arefi, S. Methyltetrahydrofolate vs folic acid supplementation in idiopathic recurrent miscarriage with respect to methylenetetrahydrofolate reductase C677T and A1298C polymorphisms: A randomized controlled trial. PLoS ONE 2015, 10, e0143569. [Google Scholar] [CrossRef] [PubMed]
- Samaniego-Vaesken Mde, L.; Alonso-Aperte, E.; Varela-Moreiras, G. Contribution of folic acid-fortified foods to fertile women’s folate Recommended Nutrient Intake through breakfast simulation models. Public Health Nutr. 2015, 18, 1960–1968. [Google Scholar] [CrossRef]
- Obeid, R.; Kirsch, S.H.; Kasoha, M.; Eckert, R.; Herrmann, W. Concentrations of unmetabolized folic acid and primary folate forms in plasma after folic acid treatment in older adults. Metabolism 2011, 60, 673–680. [Google Scholar] [CrossRef]
- Murphy, M.; Muldoon, K.A.; Harvey, A.L.J.; Rose, E.G.; Erwin, E.; Rennicks White, R.; MacFarlane, A.J.; Wen, S.W.; Walker, M.C. Gestational Folate and Folic Acid Intake among Women in Canada at Higher Risk of Pre-Eclampsia. J. Nutr. 2021, 151, 1976–1982. [Google Scholar]
- Murphy, M.S.Q.; Muldoon, K.A.; Sheyholislami, H.; Behan, N.; Lamers, Y.; Rybak, N.; White, R.R.; Harvey, A.L.J.; Gaudet, L.M.; Smith, G.N.; et al. Impact of high-dose folic acid supplementation in pregnancy on biomarkers of folate status and 1-carbon metabolism: An ancillary study of the Folic Acid Clinical Trial (FACT). Am. J. Clin. Nutr. 2021, 113, 1361–1371. [Google Scholar] [CrossRef]
- Trivedi, M.K.; Sharma, S.; Rifas-Shiman, S.L.; Camargo, C.A.; Weiss, S.T.; Oken, E.; Gillman, M.W.; Gold, D.R.; DeMeo, D.L.; Litonjua, A.A. Folic Acid in Pregnancy and Childhood Asthma: A US Cohort. Clin. Pediatr. 2018, 57, 421–427. [Google Scholar] [CrossRef]
- Yang, Q.H.; Carter, H.K.; Mulinare, J.; Berry, R.J.; Friedman, J.M.; Erickson, J.D. Race-ethnicity differences in folic acid intake in women of childbearing age in the United States after folic acid fortification: Findings from the National Health and Nutrition Examination Survey, 2001–2002. Am. J. Clin. Nutr. 2007, 85, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control & Prevention. Knowledge and use of folic acid by women of childbearing age—United States, 1995 and 1998. MMWR—Morb. Mortal. Wkly. Rep. 1999, 48, 325–327. [Google Scholar]
- Centers for Disease Control & Prevention. Knowledge and use of folic acid among women of reproductive age—Michigan, 1998. MMWR—Morb. Mortal. Wkly. Rep. 2001, 50, 185–189. [Google Scholar]
- Centers for Disease Control & Prevention. Use of vitamins containing folic acid among women of childbearing age—United States, 2004. MMWR—Morb. Mortal. Wkly. Rep. 2004, 53, 847–850. [Google Scholar]
- Commonwealth of Australia. Reproductive Health; Department of Health, Ed.; Commonwealth of Australia: Canberra, Australia, 2020. [Google Scholar]
- Gómez, M.F.; Field, C.J.; Olstad, D.L.; Loehr, S.; Ramage, S.; McCargar, L.J.; Kaplan, B.J.; Dewey, D.; Bell, R.C.; Bernier, F.P.; et al. Use of micronutrient supplements among pregnant women in Alberta: Results from the Alberta Pregnancy Outcomes and Nutrition (APrON) cohort. Matern. Child Nutr. 2015, 11, 497–510. [Google Scholar] [CrossRef]
- Masih, S.P.; Plumptre, L.; Ly, A.; Berger, H.; Lausman, A.Y.; Croxford, R.; Kim, Y.I.; O’Connor, D.L. Pregnant Canadian Women Achieve Recommended Intakes of One-Carbon Nutrients through Prenatal Supplementation but the Supplement Composition, Including Choline, Requires Reconsideration. J. Nutr. 2015, 145, 1824–1834. [Google Scholar] [CrossRef]
- Kim, Y.I. Will mandatory folic acid fortification prevent or promote cancer? Am. J. Clin. Nutr. 2004, 80, 1123–1128. [Google Scholar] [CrossRef]
- Troen, A.M.; Mitchell, B.; Sorensen, B.; Wener, M.H.; Johnston, A.; Wood, B.; Selhub, J.; McTiernan, A.; Yasui, Y.; Oral, E.; et al. Unmetabolized Folic Acid in Plasma Is Associated with Reduced Natural Killer Cell Cytotoxicity among Postmenopausal Women. J. Nutr. 2006, 136, 189–194. [Google Scholar] [CrossRef]
- Plumptre, L.; Masih, S.P.; Ly, A.; Aufreiter, S.; Kyoung-Jin, S.; Croxford, R.; Lausman, A.Y.; Berger, H.; O’Connor, D.L.; Young-In, K. High concentrations of folate and unmetabolized folic acid in a cohort of pregnant Canadian women and umbilical cord blood. Am. J. Clin. Nutr. 2015, 102, 848–857. [Google Scholar] [CrossRef]
- Quinlivan, E.P.; Gregory, J.F., III. Effect of food fortification on folic acid intake in the United States. Am. J. Clin. Nutr. 2003, 77, 221–225. [Google Scholar] [CrossRef]
- Azaryah, H.; Verdejo-Román, J.; Martin-Pérez, C.; García-Santos, J.A.; Martínez-Zaldívar, C.; Torres-Espínola, F.J.; Campos, D.; Koletzko, B.; Pérez-García, M.; Catena, A.; et al. Effects of Maternal Fish Oil and/or 5-Methyl-Tetrahydrofolate Supplementation during Pregnancy on Offspring Brain Resting-State at 10 Years Old: A Follow-Up Study from the NUHEAL Randomized Controlled Trial. Nutrients 2020, 12, 2701. [Google Scholar] [CrossRef] [PubMed]
- Rozendaal, A.M.; van Essen, A.J.; te Meerman, G.J.; Bakker, M.K.; van der Biezen, J.J.; Goorhuis-Brouwer, S.M.; Vermeij-Keers, C.; de Walle, H.E. Periconceptional folic acid associated with an increased risk of oral clefts relative to non-folate related malformations in the Northern Netherlands: A population based case-control study. Eur. J. Epidemiol. 2013, 28, 875–887. [Google Scholar] [CrossRef] [PubMed]
- Whitrow, M.J.; Moore, V.M.; Rumbold, A.R.; Davies, M.J. Effect of supplemental folic acid in pregnancy on childhood asthma: A prospective birth cohort study. Am. J. Epidemiol. 2009, 170, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- DeVilbiss, E.A.; Magnusson, C.; Gardner, R.M.; Rai, D.; Newschaffer, C.J.; Lyall, K.; Dalman, C.; Lee, B.K. Antenatal nutritional supplementation and autism spectrum disorders in the Stockholm youth cohort: Population based cohort study. BMJ 2017, 359, j4273. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.S.; Jacques, P.F.; Rosenberg, I.H.; Selhub, J. Circulating unmetabolized folic acid and 5-methyltetrahydrofolate in relation to anemia, macrocytosis, and cognitive test performance in American seniors. Am. J. Clin. Nutr. 2010, 91, 1733–1744. [Google Scholar] [CrossRef]
- World Bank. List of High Income Countries. Available online: https://data.worldbank.org/country/XD (accessed on 21 October 2021).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Boeke, C.E.; Gillman, M.W.; Hughes, M.D.; Rifas-Shiman, S.L.; Villamor, E.; Oken, E. Choline intake during pregnancy and child cognition at age 7 years. Am. J. Epidemiol. 2013, 177, 1338–1347. [Google Scholar] [CrossRef]
- Mojtabai, R. Body mass index and serum folate in childbearing age women. Eur. J. Epidemiol. 2004, 19, 1029–1036. [Google Scholar] [CrossRef]
- Bailey, R.L.; Pac, S.G.; Fulgoni, V.L., III; Reidy, K.C.; Catalano, P.M. Estimation of Total Usual Dietary Intakes of Pregnant Women in the United States. JAMA Netw. Open 2019, 2, e195967. [Google Scholar] [CrossRef]
- Dorise, B.; Byth, K.; McGee, T.; Wood, A.; Blumenthal, C. A low intensity dietary intervention for reducing excessive gestational weight gain in an overweight and obese pregnant cohort. Eat. Weight Disord. 2020, 25, 257–263. [Google Scholar] [CrossRef]
- Dubois, L.; Diasparra, M.; Bedard, B.; Colapinto, C.K.; Fontaine-Bisson, B.; Morisset, A.S.; Tremblay, R.E.; Fraser, W.D. Adequacy of nutritional intake from food and supplements in a cohort of pregnant women in Québec, Canada: The 3D Cohort Study (Design, Develop, Discover). Am. J. Clin. Nutr. 2017, 106, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Furness, D.; Fenech, M.; Dekker, G.; Khong, T.Y.; Roberts, C.; Hague, W. Folate, Vitamin B12, Vitamin B6 and homocysteine: Impact on pregnancy outcome. Matern. Child Nutr. 2013, 9, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Hamner, H.C.; Tinker, S.C.; Flores, A.L.; Mulinare, J.; Weakland, A.P.; Dowling, N.F. Modelling fortification of corn masa flour with folic acid and the potential impact on Mexican-American women with lower acculturation. Public Health Nutr. 2013, 16, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Hure, A.; Young, A.; Smith, R.; Collins, C.; Hure, A.; Young, A.; Smith, R.; Collins, C. Diet and pregnancy status in Australian women. Public Health Nutr. 2009, 12, 853–861. [Google Scholar] [CrossRef]
- Jun, S.; Gahche, J.J.; Potischman, N.; Dwyer, J.T.; Guenther, P.M.; Sauder, K.A.; Bailey, R.L. Dietary Supplement Use and Its Micronutrient Contribution During Pregnancy and Lactation in the United States. Obstet. Gynecol. 2020, 135, 623–633. [Google Scholar] [CrossRef]
- Livock, M.; Anderson, P.J.; Lewis, S.; Bowden, S.; Muggli, E.; Halliday, J. Maternal micronutrient consumption periconceptionally and during pregnancy: A prospective cohort study. Public Health Nutr. 2017, 20, 294–304. [Google Scholar] [CrossRef]
- Marchetta, C.M.; Hamner, H.C. Blood folate concentrations among women of childbearing age by race/ethnicity and acculturation, NHANES 2001–2010. Matern. Child Nutr. 2016, 12, 39–50. [Google Scholar] [CrossRef]
- Rai, D.; Bird, J.K.; McBurney, M.I.; Chapman-Novakofski, K.M. Nutritional status as assessed by nutrient intakes and biomarkers among women of childbearing age-is the burden of nutrient inadequacies growing in America? Public Health Nutr. 2015, 18, 1658–1669. [Google Scholar] [CrossRef]
- Shin, D.; Lee, K.W.; Song, W.O. Pre-Pregnancy Weight Status Is Associated with Diet Quality and Nutritional Biomarkers during Pregnancy. Nutrients 2016, 8, 162. [Google Scholar] [CrossRef]
- Tinker, S.C.; Cogswell, M.E.; Hamner, H.C.; Berry, R.J. Usual folic acid intakes: A modelling exercise assessing changes in the amount of folic acid in foods and supplements, National Health and Nutrition Examination Survey, 2003–2008. Public Health Nutr. 2012, 15, 1216–1227. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Afeiche, M.C.; Wright, D.L.; Toth, T.L.; Williams, P.L.; Gillman, M.W.; Hauser, R.; Chavarro, J.E. Dietary folate and reproductive success among women undergoing assisted reproduction. Obstet. Gynecol. 2014, 124, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Mínguez-Alarcón, L.; Fong, K.C.; Abu Awad, Y.; Di, Q.; Chavarro, J.E.; Ford, J.B.; Coull, B.A.; Schwartz, J.; Kloog, I.; et al. Supplemental Folate and the Relationship between Traffic-Related Air Pollution and Livebirth among Women Undergoing Assisted Reproduction. Am. J. Epidemiol. 2019, 188, 1595–1604. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Rich-Edwards, J.W.; Hauser, R.; Williams, P.L.; Gillman, M.W.; Ginsburg, E.S.; Missmer, S.A.; Chavarro, J.E. Maternal prepregnancy folate intake and risk of spontaneous abortion and stillbirth. Obstet. Gynecol. 2014, 124, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Pick, M.E.; Edwards, M.; Moreau, D.; Ryan, E.A. Assessment of diet quality in pregnant women using the Healthy Eating Index. J. Am. Diet. Assoc. 2005, 105, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Sotres-Alvarez, D.; Siega-Riz, A.M.; Herring, A.H.; Carmichael, S.L.; Feldkamp, M.L.; Hobbs, C.A.; Olshan, A.F. Maternal dietary patterns are associated with risk of neural tube and congenital heart defects. Am. J. Epidemiol. 2013, 177, 1279–1288. [Google Scholar] [CrossRef]
- Beringer, M.; Schumacher, T.; Keogh, L.; Sutherland, K.; Knox, P.; Herden, J.; Brown, L.; Rae, K. Nutritional adequacy and the role of supplements in the diets of Indigenous Australian women during pregnancy. Midwifery 2021, 93, 102886. [Google Scholar] [CrossRef]
- Hromi-Fiedler, A.; Bermúdez-Millán, A.; Segura-Pérez, S.; Pérez-Escamilla, R. Nutrient and food intakes differ among Latina subgroups during pregnancy. Public Health Nutr. 2012, 15, 341–351. [Google Scholar] [CrossRef]
- Martinussen, M.P.; Risnes, K.R.; Jacobsen, G.W.; Bracken, M.B. Folic acid supplementation in early pregnancy and asthma in children aged 6 years. Am. J. Obstet. Gynecol. 2012, 206, e71–e77. [Google Scholar] [CrossRef]
- Roy, A.; Evers, S.E.; Campbell, M.K. Dietary supplement use and iron, zinc and folate intake in pregnant women in London, Ontario. Chronic Dis. Inj. Can. 2012, 32, 76–83. [Google Scholar] [CrossRef]
- Standard 2.1.1; Australian New Zealand Food Standards Code—Cereals and Cereal Products. Australian Government, Federal Register of Legislation: Canberra, Australia, 2010.
- Božović, I.B.; Vraneković, J. Folate and folic acid: Current knowledge and gaps. Med. Flum. 2014, 50, 169–175. [Google Scholar]
- Czeizel, A.E.; Dudás, I.; Vereczkey, A.; Bánhidy, F. Folic acid and pregnancy: Properties, medical uses and health benefits. In Folic Acid: Properties, Medical Uses and Health Benefits; Nova Science Publishers, Inc.: Happauge, NY, USA, 2012; pp. 1–37. [Google Scholar]
- Fitzpatrick, A. Folate (folic acid): Implications for health and disease. Agro Food Ind. Hi-Tech 2003, 14, 45–48. [Google Scholar]
- Haggarty, P.; Campbell, D.M.; Duthie, S.; Andrews, K.; Hoad, G.; Piyathilake, C.; Fraser, I.; McNeill, G. Folic acid use in pregnancy and embryo selection. BJOG Int. J. Obstet. Gynaecol. 2008, 115, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Imbard, A.; Benoist, J.F.; Blom, H.J. Neural tube defects, folic acid and methylation. Int. J. Environ. Res. Public Health 2013, 10, 4352–4389. [Google Scholar] [CrossRef] [PubMed]
- Rossokha, Z.; Gorovenko, N. Assessment of the individual folic acid doses requirement for patients with reproductive disorders. J. Perinat. Med. 2017, 45, 493. [Google Scholar]
- Bjork, M.; Riedel, B.; Spigset, O.; Veiby, G.; Kolstad, E.; Daltveit, A.K.; Gilhus, N.E. Association of Folic Acid Supplementation during Pregnancy with the Risk of Autistic Traits in Children Exposed to Antiepileptic Drugs In Utero. JAMA Neurol. 2018, 75, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Sauberlich, H.; Kretsch, M.; Skala, J.; Johnson, H.; Taylor, P. Folate requirement and metabolism in nonpregnant women. Am. J. Clin. Nutr. 1987, 46, 1016–1028. [Google Scholar] [CrossRef]
- Winkels, R.M.; Brouwer, I.A.; Siebelink, E.; Katan, M.B.; Verhoef, P. Bioavailability of food folates is 80% of that of folic acid. Am. J. Clin. Nutr. 2007, 85, 465–473. [Google Scholar] [CrossRef]
- Brouwer, I.A.; van Dusseldorp, M.; West, C.E.; Meyboom, S.; Thomas, C.M.; Duran, M.; van het Hof, K.H.; Eskes, T.K.; Hautvast, J.G.; Steegers-Theunissen, R.P. Dietary folate from vegetables and citrus fruit decreases plasma homocysteine concentrations in humans in a dietary controlled trial. J. Nutr. 1999, 129, 1135–1139. [Google Scholar] [CrossRef]
- Hannon-Fletcher, M.P.; Armstrong, N.C.; Scott, J.M.; Pentieva, K.; Bradbury, I.; Ward, M.; Strain, J.; Dunn, A.A.; Molloy, A.M.; Kerr, M.A. Determining bioavailability of food folates in a controlled intervention study. Am. J. Clin. Nutr. 2004, 80, 911–918. [Google Scholar] [CrossRef]
- McNulty, H.; Pentieva, K. Folate bioavailability. Proc. Nutr. Soc. 2004, 63, 529–536. [Google Scholar] [CrossRef]
- Pietrzik, K.; Remer, T. Zur Bioverfügbarkeitsprüfung von Mikronährstoffen. Z. Ernährungswiss. 1989, 28, 130–141. [Google Scholar] [CrossRef]
- Kelly, P.; McPartlin, J.; Goggins, M.; Weir, D.G.; Scott, J.M. Unmetabolized folic acid in serum: Acute studies in subjects consuming fortified food and supplements. Am. J. Clin. Nutr. 1997, 65, 1790–1795. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.R.; McPartlin, J.; Weir, D.G.; Scott, J.M. Measurements of sub-nanomolar concentrations of unmetabolised folic acid in serum. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2003, 788, 187–191. [Google Scholar] [CrossRef]
- Bailey, S.W.; Ayling, J.E. The pharmacokinetic advantage of 5-methyltetrahydrofolate for minimization of the risk for birth defects. Sci. Rep. 2018, 8, 4096. [Google Scholar] [CrossRef]
- Prinz-Langenohl, R.; Brämswig, S.; Tobolski, O.; Smulders, Y.; Smith, D.; Finglas, P.; Pietrzik, K. [6S]-5-methyltetrahydrofolate increases plasma folate more effectively than folic acid in women with the homozygous or wild-type 677C → T polymorphism of methylenetetrahydrofolate reductase. Br. J. Pharmacol. 2009, 158, 2014–2021. [Google Scholar] [CrossRef] [PubMed]
- Niederberger, K.E.; Dahms, I.; Broschard, T.H.; Boehni, R.; Moser, R. Safety evaluation of calcium L-methylfolate. Toxicol. Rep. 2019, 6, 1018–1030. [Google Scholar] [CrossRef]
- Bostom, A.G.; Shemin, D.; Bagley, P.; Massy, Z.A.; Zanabli, A.; Christopher, K.; Spiegel, P.; Jacques, P.F.; Dworkin, L.; Selhub, J. Controlled comparison of L-5-methyltetrahydrofolate versus folic acid for the treatment of hyperhomocysteinemia in hemodialysis patients. Circulation 2000, 101, 2829–2832. [Google Scholar] [CrossRef]
- Bentley, S.; Hermes, A.; Phillips, D.; Daoud, Y.A.; Hanna, S. Comparative Effectiveness of a Prenatal Medical Food to Prenatal Vitamins on Hemoglobin Levels and Adverse Outcomes: A Retrospective Analysis. Clin. Ther. 2011, 33, 204–210. [Google Scholar] [CrossRef]
- Fava, M.; Shelton, R.C.; Zajecka, J.M. Evidence for the use of l-methylfolate combined with antidepressants in MDD. J. Clin. Psychiatry 2011, 72, 27693. [Google Scholar] [CrossRef]
- Papakostas, G.I.; Shelton, R.C.; Zajecka, J.M.; Etemad, B.; Rickels, K.; Clain, A.; Baer, L.; Dalton, E.D.; Sacco, G.R.; Schoenfeld, D. L-methylfolate as adjunctive therapy for SSRI-resistant major depression: Results of two randomized, double-blind, parallel-sequential trials. Am. J. Psychiatry 2012, 169, 1267–1274. [Google Scholar] [CrossRef]
- Beard, C.M.; Panser, L.A.; Katusic, S.K. Is excess folic acid supplementation a risk factor for autism? Med. Hypotheses 2011, 77, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Czeizel, A.E.; Vereczkey, A.; Bánhidy, F. Cleft lip and palate: Etiology and prevention. In Cleft Lip and Palate: Etiology, Surgery and Repair and Sociopsychological Consequences; Nova Science Publishers, Inc.: Happauge, NY, USA, 2013; pp. 1–53. [Google Scholar]
- Ray, J.G.; Meier, C.; Vermeulen, M.J.; Wyatt, P.R.; Cole, D.E. Association between folic acid food fortification and congenital orofacial clefts. J. Pediatr. 2003, 143, 805–807. [Google Scholar] [CrossRef]
- Christensen, K.E.; Mikael, L.G.; Leung, K.-Y.; Lévesque, N.; Deng, L.; Wu, Q.; Malysheva, O.V.; Best, A.; Caudill, M.A.; Greene, N.D.; et al. High folic acid consumption leads to pseudo-MTHFR deficiency, altered lipid metabolism, and liver injury in mice. Am. J. Clin. Nutr. 2015, 101, 646–658. [Google Scholar] [CrossRef] [PubMed]
- Blount, B.C.; Mack, M.M.; Wehr, C.M.; MacGregor, J.T.; Hiatt, R.A.; Wang, G.; Wickramasinghe, S.N.; Everson, R.B.; Ames, B.N. Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: Implications for cancer and neuronal damage. Proc. Natl. Acad. Sci. USA 1997, 94, 3290–3295. [Google Scholar] [CrossRef] [PubMed]
- Crider, K.S.; Bailey, L.B.; Berry, R.J. Folic acid food fortification—Its history, effect, concerns, and future directions. Nutrients 2011, 3, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Pickell, L.; Brown, K.; Li, D.; Wang, X.L.; Deng, L.; Wu, Q.; Selhub, J.; Luo, L.; Jerome-Majewska, L.; Rozen, R. High intake of folic acid disrupts embryonic development in mice. Birth Defects Res. Part A—Clin. Mol. Teratol. 2011, 91, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Canovas, S.; Ross, P.J.; Kelsey, G.; Coy, P. DNA Methylation in Embryo Development: Epigenetic Impact of ART (Assisted Reproductive Technologies). Bioessays 2017, 39, 1700106. [Google Scholar] [CrossRef]
- Cisneros, F.J. DNA methylation and male infertility. Front. Biosci. 2004, 9, 1189–1200. [Google Scholar] [CrossRef]
- Santi, D.; De Vincentis, S.; Magnani, E.; Spaggiari, G. Impairment of sperm DNA methylation in male infertility: A meta-analytic study. Andrology 2017, 5, 695–703. [Google Scholar] [CrossRef]
- Kelly, T.L.J.; Trasler, J.M. Reproductive epigenetics. Clin. Genet. 2004, 65, 247–260. [Google Scholar] [CrossRef]
- Aarabi, M.; San Gabrie, M.C.; Chan, D.; Behan, N.A.; Caron, M.; Pastinen, T.; Bourque, G.; MacFarlane, A.J.; Zini, A.; Trasler, J. High-dose folic acid supplementation alters the human sperm methylome and is influenced by the MTHFR C677T polymorphism. Hum. Mol. Genet. 2015, 24, 6301–6313. [Google Scholar] [CrossRef] [PubMed]
- Lambrot, R.; Xu, C.; Saint-Phar, S.; Chountalos, G.; Cohen, T.; Paquet, M.; Suderman, M.; Hallett, M.; Kimmins, S. Low paternal dietary folate alters the mouse sperm epigenome and is associated with negative pregnancy outcomes. Nat. Commun. 2013, 4, 2889. [Google Scholar] [CrossRef] [PubMed]
- Dauglait, K.; Serapinas, D. The importance of MTHFR gene mutation detection in patient with recurrent miscarriages. Genetika 2015, 47, 609–616. [Google Scholar] [CrossRef]
- Eloualid, A.; Abidi, O.; Charif, M.; El Houate, B.; Benrahma, H.; Louanjli, N.; Chadli, E.; Ajjemami, M.; Barakat, A.; Bashamboo, A.; et al. Association of the MTHFR A1298C variant with unexplained severe male infertility. PLoS ONE 2012, 7, e34111. [Google Scholar] [CrossRef]
- Khaleghparast, A.; Khaleqparast, S.; Khaleghparast, H. Association between the A1298C polymorphism of the methylenetetrahydrofolate reductase gene and recurrent spontaneous abortion. Iran. J. Neonatol. 2014, 5, 7–11. [Google Scholar] [CrossRef]
- Ledowsky, C. The different forms and dosages of folate that practitioners specialising in infertility support prescribe to patients with the MTHFR genetic polymorphisms: A case series. Aust. J. Herb. Naturop. Med. 2021, 33, 20. [Google Scholar]



| Author/ Year | Location | Study Design | Study Period | Population Description | Total Number of Participants | Objective of the Study | Summary of Key Findings in Relation to Folate |
|---|---|---|---|---|---|---|---|
| Cena 2008 [1] | United States | Cross-sectional study | Prior to 2008 | Women 18–45 years | 157 | To assess folate intake among low-income, non-pregnant women of childbearing age | * ~85% met RDA for folate * 4 exceeded UL of 1000 mcg FA/day food, fortified foods and supplements |
| Crider 2018 [33] | United States | Cross-sectional study | 2007–2012 | Women 12–49 years | 4783 | To estimate the usual daily FA and RBC to prevent NTDs | * If women only have fortified foods > risk of NTDs. * Require additional FA intake to achieve 400 mcg/day |
| Dietrich 2005 [3] | United States | Cross-sectional study | 1999–2000 | Women 20–39 years | 2260 NHANES III 356 NHANES 1999–2000 | To explore the changes in serum and erythrocyte folate following FA fortification | * Fortification increased the serum folate levels to acceptable levels * <10% reach RBC folate to reduce NTD risk |
| French 2003 [4] | Canada | Cross-sectional study | 2001–2001 | Women 18–45 years | 148 | To estimate folate intake and knowledge in women of childbearing age, in relation to risk of NTDs | * 7% exceeded the UL * Fortification does not cause > UL, supplements do |
| Gaskins 2012 [5] | United States | Cohort study | 2005–2007 | Women 18–44 years | 259 | To evaluate the association between dietary FA intake and hormones in healthy, women | * Mean dietary intake of 500 mcg achieved without supplementation (FA 50.8%/49.2% natural folate * 29.1% of dietary folate came from fortified cereals 41.1% from fortified grains * 18.1% from vegetables and 11.7% from beans. |
| Gaskins 2014 [85] | United States | Cohort study | 2006–2013 | Women 18–46 years | 316 | To evaluate the association of folate with ART outcomes | * ~57% women’s folate came from supplements * 78% took supplements with 400 mcg FA, 19% 1000 mcg * 43% from foods (both natural and fortified) |
| Gaskins 2019 [86] | United States | Cohort study | 2004–2017 | Women 18–46 years | 513 ART cycles from 304 women. | To evaluate folate intake air pollution and livebirth in women using ART | * 20% of women consumed 1000 mcg of supplemental FA |
| Hamner 2013 [77] | United States | Cohort study | 2001–2008 | Women 15–44 years | 5369 | FA fortification to increase folate levels in Mexican women with lower acculturation | * 24.0% women had FA intake of 400 mcg |
| Hure 2008 [78] | Australia | Cross-sectional study | 2003–2003 | Women 25–30 years | 9076 (606 pregnant) | To investigate and report the diet quality of young Australian women by pregnancy status | * Folate < EAR * Dietary deficit * No supplements recorded |
| Jun 2020 [79] | United States | Cross-sectional study | 1999–2014 | Women 20–44 years | 8096 | To estimate dietary supplements use and prevalence | * 44.8% used dietary supplements * Only supplements measured |
| Marchetta 2016 [81] | United States | Cross-sectional study | 2001–2010 | Women 15–44 years | 4985 NHW and MA non-pregnant women | To assess the differences in serum and RBC folate concentrations by acculturation factors | * Supplements with FA impact blood folate status * Acculturation factors impact folate levels * RBC folate concentrations indicate long-term status |
| Mojtabai 2004 [72] | United States | Cohort study | 1999–2000 | Women 17–49 years | 1351 | To discover the impact of BMI on serum folate levels | * Fortification increased FA levels from mean 228.5 to 324.3 mcg/day * Serum folate levels increased due to FDA mandate of fortification. |
| Pick 2005 [88] | Canada | Case control study | Prior to 2004 | Women 20–40 years | 112 | To examine the diets of healthy women | * FA + natural folate in food does not achieve recommended 400 mcg/day * 80% of women did not meet the RDA for folate * Supplements not included |
| Rai 2014 [82] | United States | Cohort study | 2003–2008 | Women 19–50 years | 3641 | To evaluate nutritional status in women of childbearing age and ethnicity | * RBC folate cut off for deficiency is 200 nmol/L * Most women achieved minimum in relation to folate deficiency but few reached 906 nmol/L for prevention of NTDs * RDA of 400 mcg folate 90% did not achieve RBC folate levels to prevent NTDs * At 1000 mcg/d, less than 25% of women had RBC folate level that prevent NTDs |
| Sotres-Alvarez 2012 [89] | United States | Case control study | 1997–2005 | Women childbearing age | 1047 cases with NTDs 6641 CHD 6123 nonmalformed controls | Dietary intake and NTDs and CHDs | * Non-users of FA/multivitamin supplements who ate more fruits and vegetables significantly less likely to have NTDs * Supplements not included |
| Tinker 2012 [84] | United States | Cohort study | 2003–2008 | Women 15 to 44 years | 4272 | BMI supplemental FA intake and folate status | * Women 25–44 years age more likely to use FA supplements than 15–24 year olds * Nonusers and users of dietary supplements similar FA intakes of DFEs * BMI may affect body distribution of folate |
| Tinker 2012 [2] | United States | Cross-sectional study | 2003–2004, 2005–2006, 2007–2008 NHANES-2008 | Women childbearing age | 4272 | We sought to model FA intake under various fortification and supplementation scenarios | * UL influenced by supplements * Median intake influenced by enriched cereal grain products * 23% of women achieve RDA of FA * 2.4% exceed the UL |
| Yang 2007 [53] | United States | Cohort study | 2001–2002 | Women 14–49 years | 1685 | To examine FA intake in women of childbearing age in the United States | * Average serum folate concentrations show 50% increase since fortification * RBC concentrations show 59% increase since fortification * 47.5% of FA intake from supplements * 5.7% women consuming supplements and fortified foods exceeded the UL * Underreporting of FA from fortified foods |
| Author/ Year | Location | Study Design | Study Period | Population Description | Total Number of Participants | Objective of the Study | Summary of Key Findings in Relation to Folate |
|---|---|---|---|---|---|---|---|
| Bailey 2019 [73] | United States | Cross-sectional study | 2001–2014 | Pregnant women 20–40 years at all stages of gestation | 1003 | To estimate nutrient intakes (from foods and dietary supplements) and prevalence of meeting or exceeding RDA among pregnant U.S. women | * Supplements responsible for UL being exceeded * Supplements required for pregnant women to meet RDA * 33.4% exceed UL FA |
| Beringer 2021 [74] | Australia | Cross-sectional study | 2009–2019 | Pregnant Indigenous women in all stages of gestation | 152 | To determine sources of key nutrients contributing towards nutritional adequacy during pregnancy | * 75% met EAR * 55% met folate EAR form diet alone * ~51% of women took a supplement containing folate |
| Boeke 2013 [71] | United States | Cohort study | 1999–2006 | Pregnant women in T1/T2 | 1896 | To examine maternal T1/T2 dietary intake of methyl donor nutrients during pregnancy in relation to child visual memory | * Second trimester folate intake average was 1268 mcg/day |
| Dorise 2020 [74] | Australia | retrospective study | 2015–2015 | 10–18 weeks gestation | 231 | To evaluate the effectiveness of a group-based outpatient dietary intervention in pregnancy to reduce excessive gestational weight gain | * Supplement required to meet EAR * 20% met with folate EAR from diet alone * Mean folate intake 533 mcg/day NB: measuring food (adjusted for supplementation) |
| Dubois 2017 [75] | Canada | Cohort study | 2010–2012 | Pregnant women T1 | 1533 | To assess nutritional intakes during pregnancy by examining dietary sources and supplements and comparing to RDI | * 70% did not meet EAR with diet * With supplements 87% exceeded the UL (diet and supplement sources) |
| Furness 2013 [76] | Australia | Prospective observational study | Prior to 2009 | Pregnant women < 20 weeks gestation | 46 low risk and 91 high risk women = total of 137 | To determine if methyl donor nutrients < 18–20 weeks gestation are associated with subsequent adverse pregnancy outcomes. To investigate maternal B vitamin concentrations with DNA damage markers | * Older women had increased RBC folate, serum folate * FA supplementation > 1000 mg/day resulted in the highest RBC folate * 100% of the high risk women exceeded the UL of FA (2116 mcg mean) range 1615–2617 mcg NB: study conducted before mandatory FA fortification from 18 September 2009 in Australia |
| Gomez 2015 [58] | Canada | Cohort study | 2009–2010 | Pregnant women < 27 weeks gestation | 599 | To describe the use of natural health products (NHP) by pregnant women in each trimester of pregnancy | * Average FA intake was 200% above RDA in each trimester * 25% pregnant women exceeded UL in each trimester * IOM guidelines met by 97% in first trimester, decreasing to 91% in third trimester NB: supplement use only recorded |
| Hromi-Fiedler 2011 [91] | United States | Cross-sectional study | 2004–2006 | Pregnant Latinas between 16 and 32 weeks gestation | 241 | To document nutrient and food intakes from food sources among Latina subgroups living in the same geographical area | * Mean FA intake 768 mcg from diet alone NB supplement use not recorded |
| Hure 2008 [78] | Australia | Cross-sectional study | 2003–2003 | Women aged 25–30 years at any stage of pregnancy | 606 | To investigate and report the diet quality of young Australian women by pregnancy status | * Folate was consistently below EAR * Only food reported/no supplements |
| Jun 2020 [79] | United States | Cross-sectional study | 1999–2014 | 1314 women 20–44 years of age at all stages of pregnancy | 1314 | To estimate the prevalence of use and the micronutrient contribution of dietary supplements among pregnant, lactating, and non-pregnant and non-lactating women | * 77% used dietary supplements * More than 60% of pregnant women used supplements with FA * Mean intakes of FA from supplements alone were at or above the RDI * Dietary supplements contributed a mean daily intake of 787 mcg of FA * >40% supplement users exceeded the UL of FA |
| Livock 2016 [80] | Australia | Cohort study | 2011–2012 | Women < 19 weeks gestation | 2146 | To examine overall micronutrient intake periconceptionally and throughout pregnancy | * Many women failed to meet RDI for folate in periconceptional period. (fortified food not included) * UL exceeded through diet and supplemental sources esp. late in T1 * 80% of folate for overconsumers coming from supplements NB: food fortification not included |
| Martinussen 2011 [92] | United States | Cohort study | 1997–2000 | Pregnant women < 24 weeks gestational age | 1499 | To assess whether FA intake during T1 of pregnancy is related to asthma in the offspring by the age of 6 years | * Mean intake FA ↑ from 303 mcg preconceptionally, to 404 mcg in 1st, 605 mcg in 2nd and 676 mcg in the 3rd month of pregnancy * Mean supplementation in T1 was 497 mcg * 51% women used FA supplements before pregnancy * 61% used FA supplements in first month of pregnancy * 81% in second and 88% in third month of pregnancy 92% women used FA in first trimester > 800 mcg FA NB: only supplements measured. No food or fortification |
| Masih 2015 [59] | Canada | Cohort study | 2010–2012 | pregnant women at < 16 weeks gestation | 353 | To determine dietary and supplemental intakes and major dietary sources of one-carbon nutrients | * 85% women exceeded UL FA through supplements alone * Typical dose FA in supplements 1000 mcg * Pregnant women exposed to FA 2.5-fold RDI of 400 mcg/day |
| Murphy 2021 [51] | Canada | Case control study | 2013–2015 | Pregnant women 24–26 weeks gestational age 18–44 years of age | 51 | This was an ancillary study within the Folic Acid Clinical Trial (FACT), a randomized, double-blinded, placebo-controlled, phase III trial designed to assess the efficacy of high-dose FA to prevent preeclampsia | * All women exceeded the WHO RBC total folate 906 nmol/L cut off for NTDs * ~80% of women were above the 97th percentile for RBC folate concentrations * Folate status for all women > WHO cut off for NTD risk reduction * UMFA measurable in all women and some at high levels * High-dose FA is unwarranted for this clinical population |
| Rose/Murphy 2021 [50] | Canada | Case control study | 2011–2015 | Pregnant women 8–16 weeks gestation | 1198 | To evaluate the dietary and supplemental intakes of FA and to determine the proportions of pregnant women exceeding the estimated average requirement (EAR) and tolerable upper intake level (UL) | * FA intake from diet (food and fortification) insufficient to achieve 400 mcg FA/day * Median food/fortification 333 mcg DFE/day * If FA supplements are added mean folate 2167 mcg DFE/day * 89.2% of participants > EAR if supplements taken * 96% exceeded UL 1000 mcg/day, 0.4% below RDA (400 mcg) |
| Pick 2005 [88] | Canada | Case control study | Prior 2004 | Women 20–38 weeks gestation aged 20–40 years | 112 | The objectives of this pilot study were to examine the diets of pregnant women and healthy women of child-bearing age | * Daily dietary folate intake for pregnant women was 331 mcg/day * 98% of pregnant women did not meet minimum RDI for folate from food Note: Supplements not included in study |
| Plumptre 2015 [62] | Canada | Cohort study | 2010–2012 | Pregnant women aged 18–45 yers between 10 and 22 weeks gestation | 368 | Determine maternal and cord blood concentrations of folate and unmetabolized folic acid (UMFA) and examine effect of maternal intakes of folate and FA and fetal genetic variants in folate metabolism on folate status | * Folate intake (natural folate and fortified foods) mean 483 mcg DFE/day early T1 and 465 mcg DFE/day late preg * 83% > UL FA * Median maternal folic acid intake was 1000 mcg early T1 * Early T1, maternal plasma UMFA detected in 97% of women * Maternal serum folate concentrations significantly decreased during pregnancy, whereas RBC folate significantly ↑ * UMFA detectable in 93% of cord blood samples |
| Roy 2012 [93] | Canada | Cohort study | 2002–2005 | Pregnant women between 10 and 22 weeks gestation | 2019 | Examine dietary intake of iron, zinc and folate, from food and supplement sources | * Mean food intake of FA 473 mcgDFE/day * Mean FA supplement intake 1338 mcg DFE/day, mean total folate 1811 mcg DFE/day * Only 16% did not reach RDA of 600 mcg DFE/day * Supplement intake > UL 1000 mcg |
| Shin 2016 [83] | United States | Cross-sectional study | 2003–2012 | 795 pregnant women at all stages of gestation | 856 | To examine relationship between pre-pregnancy weight status/diet quality and nutritional status | * Normal-weight women had a mean dietary intake of 282.2 mcgDFE/day, and dietary supplemental intake 1329 mcgDFE/day |
| Trivedi 2018 [52] | United States | Cohort study | 1999–2002 | Pregnant women 1–26 weeks gestation—T1 and T2 | 1279 mother-child pairs | To examine this association in the United States, where the food supply is generally fortified with FA | * Mean intake of FA first trimester 930 mcg, second trimester 1238 mcg * ~75% exceeded 400 mcg * 94% women took FA supplement |
| Whitrow 2009 [66] | Australia | Cohort study | 1998–2005 | Pregnant women < 16 weeks gestation | 557 | To investigate the effect of the timing, dose, and source of folate during pregnancy on childhood asthma | * Median intake of FA from supplements was 2948 mcg/day * Supplements contributed to 84% of FA in early pregnancy and 63% in late pregnancy NB: Study conducted before fortification in Australia implemented |
| Author/ Year | Natural Food Folate Intake Dietary | Food Folate and FA Fortified Foods Combined Intake | Synthetic FA Intake—from Fortified Foods | Synthetic FA Intake from Supplements | Folate Intake Total (Food and Supplements) | Supplement Use during Study | Stage of Pregnancy/ Weeks Gestation |
|---|---|---|---|---|---|---|---|
| Women of childbearing age/Not pregnant | |||||||
| Cena 2008 [1] | 180.7 mcg | n/a | 253.4 mcg | 148.6 mcg | 402.0 mcg, 864.0 mcg DFE | yes | NIL |
| Crider 2018 [33] | 236 mcg | n/a | 239 mcg, 582 mcg DFE | 461 mcg | 661 mcg, 1341 mcg DFE | yes | NIL |
| Dietrich 2005 [3] | n/a | 294 mcg DFE | n/a | n/a | n/a | no | NIL |
| French 2003 [4] | 259 mcg DFE | n/a | 470 mcg DFE | n/a | 812 mcg DFE | yes | NIL |
| Gaskins 2012 [5] | 181.7 mcg | 500.5 mcg, 368.9 mcg DFE | 181.7 mcg | n/a | n/a | no | NIL |
| Gaskins 2014 [87] | n/a | 764.54 mcg DFE | n/a | 1013.46 mcg | 1778 mcg DFE | yes | NIL |
| Gaskins 2019 [86] | n/a | 459.1 mcg | n/a | 338.4 mcg | 797.5 mcg | yes | NIL |
| Hamner 2013 [77] | n/a | 244 mcg | n/a | 380 mcg | n/a | yes | NIL |
| Hure 2008 [78] | n/a | 265.9 mcg | n/a | n/a | n/a | no | NIL |
| Jun 2020 [79] | n/a | n/a | n/a | 375 mcg | n/a | yes, no food | NIL |
| Marchetta 2016 [81] | n/a | n/a | n/a | 381 mcg | n/a | yes | NIL |
| Mojtabai 2004 [72] | n/a | 332.1 mcg | n/a | n/a | n/a | no | NIL |
| Pick 2005 [88] | n/a | 300 mcg | n/a | n/a | n/a | no | NIL |
| Rai 2015 [82] | n/a | 456.4 mcg DFE | n/a | n/a | n/a | no | NIL |
| Sotres-Alvarez 2013 [89] | 170.92 mcg, 430.70 mcg DFE | 430.70 mcg DFE | 130.76 mcg DFE | n/a | n/a | no | NIL |
| Tinker 2012 [2] | n/a | 434 mcg DFE | n/a | >400 mcg, 475 mcg DFE | n/a | yes | NIL |
| Tinker 2012 [84] | n/a | n/a | n/a | n/a | n/a | no | NIL |
| Yang 2007 [53] | 151 mcg | 221 mcg | 128 mcg | n/a | n/a | no | NIL |
| Pregnancy Studies | |||||||
| Bailey 2019 [73] | n/a | 630 mcg DFE | n/a | n/a | 1451 mcg DFE | yes | All |
| Beringer 2021 [90] | n/a | 502.6 mcg DFE | n/a | 833.4 mcg DFE | 996.6 mcg DFE | yes | All |
| Boeke 2013 [71] | n/a | n/a | n/a | n/a | T1 972 mcg T2 1268 mcg | yes | T1 |
| Dorise 2020 [74] | n/a | 533 mcg | n/a | n/a | n/a | no | 10–22 weeks |
| Dubois 2017 [75] | n/a | 463 mcg | n/a | n/a | 2181 mcg DFE | no | T1 |
| Furness 2013 [76] | 283 mcg | n/a | n/a | 668–2116 mcg | n/a | yes | 18–20 |
| Gomez 2015 [58] | n/a | n/a | n/a | T1: 1225 mcg T2: 1353 mcg T3: 1228 mcg | n/a | yes, no food | <27 |
| Hromi-Fiedler 2011 [91] | 284.9 mcg | 610 mcg | 768 mcg | n/a | n/a | no | 16–32 |
| Hure 2008 [78] | n/a | 284.4 mcg | n/a | n/a | n/a | no | All |
| Jun 2020 [79] | n/a | n/a | n/a | 787 mcg DFE | n/a | yes, no food. | All |
| Livock 2017 [80] | n/a | n/a | T1: 247 mcg | T1: 522 mcg | T1: 840 mcg | yes | |
| T2: 245 mcg | T2: 527 mcg | T2: 760 mcg | |||||
| T3: 251 mcg | T3: 518 mcg | T3: 690 mcg | <19 | ||||
| Martinussen 2012 [92] | n/a | n/a | n/a | month 1 402 mcg month 2 605 mcg month 3 676 mcg | n/a | yes, no Food | <24 |
| Masih 2015 [59] | T1: 313 ± 140 mcg DFE T3: 297 ± 131 mcg DFE | T1: 483 mcg DFE T3: 465 mcg DFE | T1: 96 ± 54 mcg T3: 96 ± 50 mcg | 1000 mcg | n/a | yes | <16 |
| Murphy 2021 [51] | 148.2 mcg DFE | 346.7 mcg 512.5 mcg DFE | 226.6 mcg | 1100 mcg | n/a | yes | 24–26 |
| Murphy/Rose 2021 [50] | 140 mcg DFE | 485 mcg DFE | 333 mcg DFE | 1000 mcg | 2167 mcg DFE | yes | 8–16 weeks |
| Pick 2005 [88] | - | 331 mcg | n/a | n/a | n/a | no | 20–38 |
| Plumptre 2015 [62] | T1: 483 ± 203 mcg DFE T3: 465 ± 186 mcg DFE | n/a | n/a | 1000 mcg | n/a | yes | 10–22 weeks |
| Roy 2012 [93] | n/a | 473 mcg DFE | n/a | 1338 mcg | 1811 mcg DFE | yes | 10–22 weeks |
| Shin 2016 [83] | n/a | 627.6 mcg DFE | n/a | 781.8, 1329 mcg DFE | n/a | yes | All |
| Trivedi 2018 [52] | n/a | n/a | n/a | n/a | T1: 930 mcg T2: 1238 mcg | yes | <25 weeks |
| Whitrow 2009 [66] | T1: 224.7 mcg T3: 208.4 mcg | n/a | n/a | T1: 658.3 mcg T3: 300 mcg | n/a | yes | <16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ledowsky, C.; Mahimbo, A.; Scarf, V.; Steel, A. Women Taking a Folic Acid Supplement in Countries with Mandatory Food Fortification Programs May Be Exceeding the Upper Tolerable Limit of Folic Acid: A Systematic Review. Nutrients 2022, 14, 2715. https://doi.org/10.3390/nu14132715
Ledowsky C, Mahimbo A, Scarf V, Steel A. Women Taking a Folic Acid Supplement in Countries with Mandatory Food Fortification Programs May Be Exceeding the Upper Tolerable Limit of Folic Acid: A Systematic Review. Nutrients. 2022; 14(13):2715. https://doi.org/10.3390/nu14132715
Chicago/Turabian StyleLedowsky, Carolyn, Abela Mahimbo, Vanessa Scarf, and Amie Steel. 2022. "Women Taking a Folic Acid Supplement in Countries with Mandatory Food Fortification Programs May Be Exceeding the Upper Tolerable Limit of Folic Acid: A Systematic Review" Nutrients 14, no. 13: 2715. https://doi.org/10.3390/nu14132715
APA StyleLedowsky, C., Mahimbo, A., Scarf, V., & Steel, A. (2022). Women Taking a Folic Acid Supplement in Countries with Mandatory Food Fortification Programs May Be Exceeding the Upper Tolerable Limit of Folic Acid: A Systematic Review. Nutrients, 14(13), 2715. https://doi.org/10.3390/nu14132715







