Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. RBC Fatty Acid Analysis
2.3. Outcome Measures
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barbaresko, J.; Lellmann, A.W.; Schmidt, A.; Lehmann, A.; Amini, A.M.; Egert, S.; Schlesinger, S.; Nöthlings, U. Dietary Factors and Neurodegenerative Disorders: An Umbrella Review of Meta-Analyses of Prospective Studies. Adv. Nutr. 2020, 11, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teng, E.; Taylor, K.; Bilousova, T.; Weiland, D.; Pham, T.; Zuo, X.; Yang, F.; Chen, P.P.; Glabe, C.G.; Takacs, A.; et al. Dietary DHA supplementation in an APP/PS1 transgenic rat model of AD reduces behavioral and Aβ pathology and modulates Aβ oligomerization. Neurobiol. Dis. 2015, 82, 552–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pifferi, F.; Cunnane, S.C.; Guesnet, P. Evidence of the Role of Omega-3 Polyunsaturated Fatty Acids in Brain Glucose Metabolism. Nutrients 2020, 12, 1382. [Google Scholar] [CrossRef] [PubMed]
- Devassy, J.G.; Leng, S.; Gabbs, M.; Monirujjaman, M.; Aukema, H.M. Omega-3 Polyunsaturated Fatty Acids and Oxylipins in Neuroinflammation and Management of Alzheimer Disease. Adv. Nutr. 2016, 7, 905–916. [Google Scholar] [CrossRef]
- Yassine, H.N.; Braskie, M.N.; Mack, W.J.; Castor, K.J.; Fonteh, A.N.; Schneider, L.S.; Harrington, M.G.; Chui, H.C. Association of Docosahexaenoic Acid Supplementation with Alzheimer Disease Stage in Apolipoprotein E ε4 Carriers: A Review. JAMA Neurol. 2017, 74, 339–347. [Google Scholar] [CrossRef] [Green Version]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef]
- Harris, W.S.; Thomas, R.M. Biological variability of blood omega-3 biomarkers. Clin. Biochem. 2010, 43, 338–340. [Google Scholar] [CrossRef]
- Hooper, C.; De Souto Barreto, P.; Pahor, M.; Weiner, M.; Vellas, B. The Relationship of Omega 3 Polyunsaturated Fatty Acids in Red Blood Cell Membranes with Cognitive Function and Brain Structure: A Review Focussed on Alzheimer’s Disease. J. Prev. Alzheimers Dis. 2018, 5, 78–84. [Google Scholar] [CrossRef]
- Macaron, T.; Giudici, K.V.; Bowman, G.L.; Sinclair, A.; Stephan, E.; Vellas, B.; de Souto Barreto, P. Associations of Omega-3 fatty acids with brain morphology and volume in cognitively healthy older adults: A narrative review. Ageing Res. Rev. 2021, 67, 101300. [Google Scholar] [CrossRef]
- Kröger, E.; Verreault, R.; Carmichael, P.H.; Lindsay, J.; Julien, P.; Dewailly, E.; Ayotte, P.; Laurin, D. Omega-3 fatty acids and risk of dementia: The Canadian Study of Health and Aging. Am. J. Clin. Nutr. 2009, 90, 184–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ammann, E.M.; Pottala, J.V.; Robinson, J.G.; Espeland, M.A.; Harris, W.S. Erythrocyte omega-3 fatty acids are inversely associated with incident dementia: Secondary analyses of longitudinal data from the Women’s Health Initiative Memory Study (WHIMS). Prostaglandins Leukot. Essent. Fatty Acids 2017, 121, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, E.J.; Bongard, V.; Beiser, A.S.; Lamon-Fava, S.; Robins, S.J.; Au, R.; Tucker, K.L.; Kyle, D.J.; Wilson, P.W.; Wolf, P.A. Plasma phosphatidylcholine docosahexaenoic acid content and risk of dementia and Alzheimer disease: The Framingham Heart Study. Arch. Neurol. 2006, 63, 1545–1550. [Google Scholar] [CrossRef] [PubMed]
- Lopez, L.B.; Kritz-Silverstein, D.; Barrett Connor, E. High dietary and plasma levels of the omega-3 fatty acid docosahexaenoic acid are associated with decreased dementia risk: The Rancho Bernardo study. J. Nutr. Health Aging 2011, 15, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Baillet, M.; Proust-Lima, C.; Féart, C.; Foubert-Samier, A.; Helmer, C.; Catheline, G.; Samieri, C. Blood polyunsaturated omega-3 fatty acids, brain atrophy, cognitive decline, and dementia risk. Alzheimers Dement. 2021, 17, 407–416. [Google Scholar] [CrossRef]
- Van der Lee, S.J.; Teunissen, C.E.; Pool, R.; Shipley, M.J.; Teumer, A.; Chouraki, V.; Melo van Lent, D.; Tynkkynen, J.; Fischer, K.; Hernesniemi, J.; et al. Circulating metabolites and general cognitive ability and dementia: Evidence from 11 cohort studies. Alzheimers Dement. 2018, 14, 707–722, Erratum in Alzheimers Dement. 2019, 15, 319. [Google Scholar]
- Samieri, C.; Féart, C.; Letenneur, L.; Dartigues, J.F.; Pérès, K.; Auriacombe, S.; Peuchant, E.; Delcourt, C.; Barberger-Gateau, P. Low plasma eicosapentaenoic acid and depressive symptomatology are independent predictors of dementia risk. Am. J. Clin. Nutr. 2008, 88, 714–721. [Google Scholar] [CrossRef] [Green Version]
- Melo van Lent, D.; Egert, S.; Wolfsgruber, S.; Kleineidam, L.; Weinhold, L.; Wagner-Thelen, H.; Maier, W.; Jessen, F.; Ramirez, A.; Schmid, M.; et al. Eicosapentaenoic Acid Is Associated with Decreased Incidence of Alzheimer’s Dementia in the Oldest Old. Nutrients 2021, 13, 461. [Google Scholar] [CrossRef]
- Dawber, T.R.; Meadors, G.F.; Moore, F.E., Jr. Epidemiological approaches to heart disease: The Framingham Study. Am. J. Public Health Nations Health 1951, 41, 279–281. [Google Scholar] [CrossRef]
- Feinleib, M.; Kannel, W.B.; Garrison, R.J.; McNamara, P.M.; Castelli, W.P. The Framingham Offspring Study. Design and preliminary data. Prev. Med. 1975, 4, 518–525. [Google Scholar] [CrossRef]
- Hixson, J.E.; Vernier, D.T. Restriction isotyping of human apolipoprotein E by gene amplification and cleavage with HhaI. J. Lipid Res. 1990, 31, 545–548. [Google Scholar] [CrossRef]
- Harris, W.S.; Pottala, J.V.; Vasan, R.S.; Larson, M.G.; Robins, S.J. Changes in erythrocyte membrane trans and marine fatty acids between 1999 and 2006 in older Americans. J. Nutr. 2012, 142, 1297–1303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachman, D.L.; Wolf, P.A.; Linn, R.; Knoefel, J.E.; Cobb, J.; Belanger, A.; D’Agostino, R.B.; White, L.R. Prevalence of dementia and probable senile dementia of the Alzheimer type in the Framingham Study. Neurology 1992, 42, 115–119. [Google Scholar] [CrossRef] [PubMed]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rimm, E.B.; Appel, L.J.; Chiuve, S.E.; Djoussé, L.; Engler, M.B.; Kris-Etherton, P.M.; Mozaffarian, D.; Siscovick, D.S.; Lichtenstein, A.H.; American Heart Association Nutrition Committee of the Council on Lifestyle and Cardiometabolic Health; et al. Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and Cardiovascular Disease: A Science Advisory from the American Heart Association. Circulation 2018, 138, e35–e47. [Google Scholar] [CrossRef]
- 2022 Alzheimer’s disease facts and figures. Alzheimers Dement. 2022, 18, 700–789. [CrossRef]
- Zissimopoulos, J.; Crimmins, E.; St Clair, P. The Value of Delaying Alzheimer’s Disease Onset. Forum. Health Econ. Policy 2014, 18, 25–39. [Google Scholar] [CrossRef]
- Tan, Z.S.; Harris, W.S.; Beiser, A.S.; Au, R.; Himali, J.J.; Debette, S.; Pikula, A.; Decarli, C.; Wolf, P.A.; Vasan, R.S.; et al. Red blood cell ω-3 fatty acid levels and markers of accelerated brain aging. Neurology 2012, 78, 658–664. [Google Scholar] [CrossRef] [Green Version]
- Barnes, S.; Chowdhury, S.; Gatto, N.M.; Fraser, G.E.; Lee, G.J. Omega-3 fatty acids are associated with blood-brain barrier integrity in a healthy aging population. Brain Behav. 2021, 11, e2273. [Google Scholar] [CrossRef]
- Yassine, H.N.; Feng, Q.; Azizkhanian, I.; Rawat, V.; Castor, K.; Fonteh, A.N.; Harrington, M.G.; Zheng, L.; Reed, B.R.; DeCarli, C.; et al. Association of Serum Docosahexaenoic Acid with Cerebral Amyloidosis. JAMA Neurol. 2016, 73, 1208–1216. [Google Scholar] [CrossRef]
- Dangour, A.D.; Allen, E.; Elbourne, D.; Fasey, N.; Fletcher, A.E.; Hardy, P.; Holder, G.E.; Knight, R.; Letley, L.; Richards, M.; et al. Effect of 2-y n-3 long-chain polyunsaturated fatty acid supplementation on cognitive function in older people: A randomized, double-blind, controlled trial. Am. J. Clin. Nutr. 2010, 91, 1725–1732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chew, E.Y.; Clemons, T.E.; Agrón, E.; Launer, L.J.; Grodstein, F.; Bernstein, P.S.; Age-Related Eye Disease Study 2 (AREDS2) Research Group. Effect of Omega-3 Fatty Acids, Lutein/Zeaxanthin, or Other Nutrient Supplementation on Cognitive Function: The AREDS2 Randomized Clinical Trial. JAMA 2015, 314, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Vyas, C.M.; Okereke, O.I.; Ogata, S.; Albert, M.; Lee, I.M.; D’Agostino, D.; Buring, J.E.; Cook, N.R.; Grodstein, F.; et al. Marine n-3 fatty acids and cognitive change among older adults in the VITAL randomized trial. Alzheimers Dement. 2022, 8, e12288. [Google Scholar] [CrossRef] [PubMed]
- Andrieu, S.; Guyonnet, S.; Coley, N.; Cantet, C.; Bonnefoy, M.; Bordes, S.; Bories, L.; Cufi, M.N.; Dantoine, T.; Dartigues, J.F.; et al. Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (MAPT): A randomised, placebo-controlled trial. Lancet Neurol. 2017, 16, 377–389. [Google Scholar] [CrossRef]
- Lin, P.Y.; Cheng, C.; Satyanarayanan, S.K.; Chiu, L.T.; Chien, Y.C.; Chuu, C.P.; Lan, T.H.; Su, K.P. Omega-3 fatty acids and blood-based biomarkers in Alzheimer’s disease and mild cognitive impairment: A randomized placebo-controlled trial. Brain Behav. Immun. 2022, 99, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Tintle, N.L.; Pottala, J.V.; Lacey, S.; Ramachandran, V.; Westra, J.; Rogers, A.; Clark, J.; Olthoff, B.; Larson, M.; Harris, W.; et al. A genome-wide association study of saturated, mono- and polyunsaturated red blood cell fatty acids in the Framingham Heart Offspring Study. Prostaglandins Leukot. Essent. Fatty Acids 2015, 94, 65–72. [Google Scholar] [CrossRef] [Green Version]
- Kalsbeek, A.; Veenstra, J.; Westra, J.; Disselkoen, C.; Koch, K.; McKenzie, K.A.; O’Bott, J.; Vander Woude, J.; Fischer, K.; Shearer, G.C.; et al. A genome-wide association study of red-blood cell fatty acids and ratios incorporating dietary covariates: Framingham Heart Study Offspring Cohort. PLoS ONE. 2018, 13, e0194882. [Google Scholar] [CrossRef] [Green Version]
- Hooper, C.; De Souto Barreto, P.; Coley, N.; Cantet, C.; Cesari, M.; Andrieu, S.; Vellas, B. Cognitive Changes with Omega-3 Polyunsaturated Fatty Acids in Non-Demented Older Adults with Low Omega-3 Index. J. Nutr. Health Aging 2017, 21, 988–993. [Google Scholar] [CrossRef]
- Prevention of Cognitive Decline in Older Adults with Low Dha/Epa Index in Red Blood Cells (LO-MAPT). Available online: clinicaltrials.gov/ct2/show/NCT03691519 (accessed on 24 January 2022).
- Arellanes, I.C.; Choe, N.; Solomon, V.; He, X.; Kavin, B.; Martinez, A.E.; Kono, N.; Buennagel, D.P.; Hazra, N.; Kim, G.; et al. Brain delivery of supplemental docosahexaenoic acid (DHA): A randomized placebo-controlled clinical trial. eBioMedicine 2020, 59, 102883. [Google Scholar] [CrossRef]

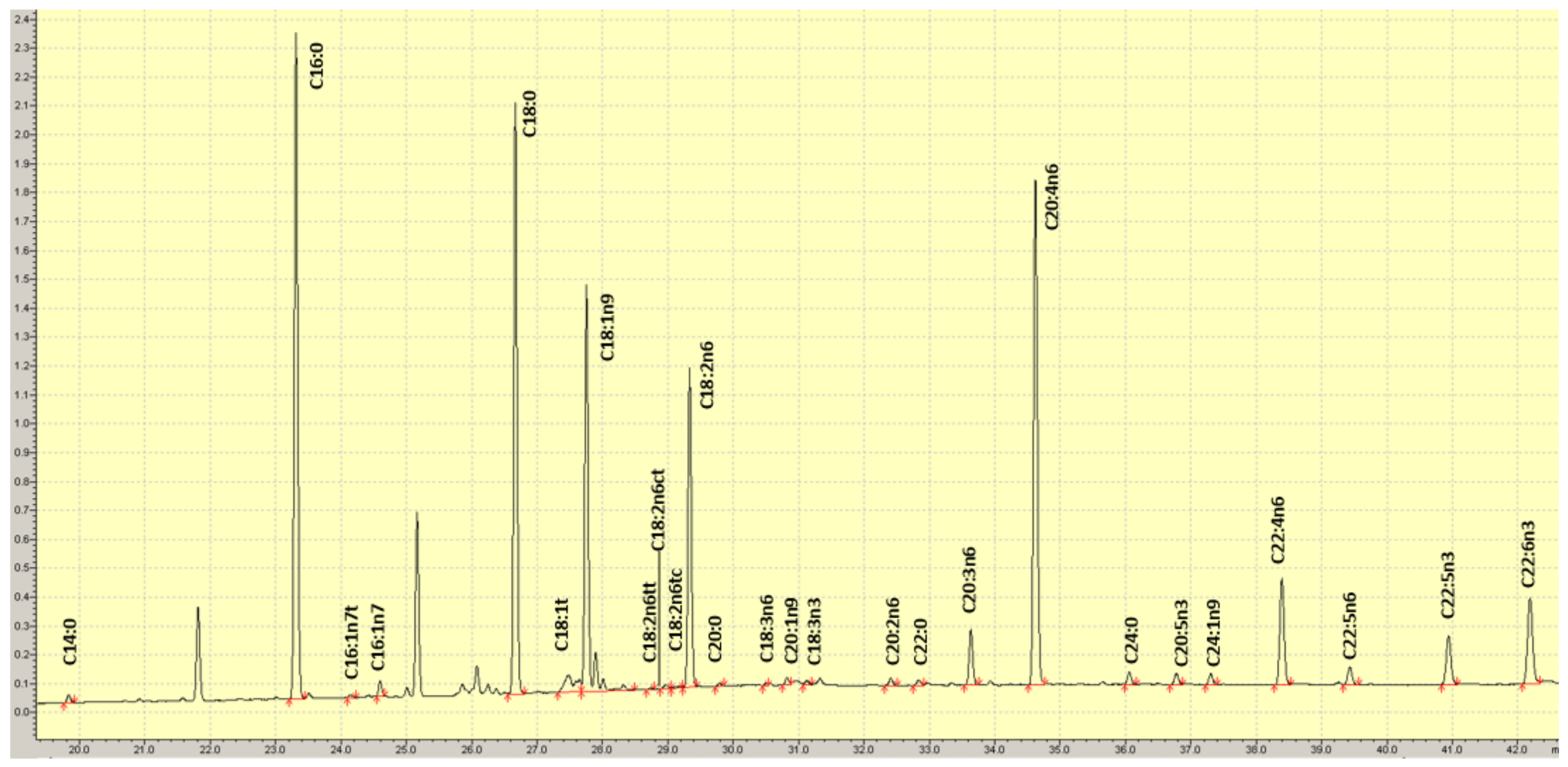


| Variable | All Population (n = 1490) | APOE-ε4 Non-carrier (n = 1155) 1 | APOE-ε4 Carrier (n = 335) 2 | ε3/ε4 + ε4/ε4 (n = 311) |
|---|---|---|---|---|
| Women—No. (%) | 798 (53.6) | 620 (53.7) | 178 (53.1) | 167 (53.7) |
| Age—Mean (SD) | 73.0 (5.7) | 73.2 (5.8) | 72.4 (5.5) 3 | 72.5 (5.5) |
| Education—No. (%) | ||||
| No high school degree | 64 (4.3) | 49 (4.2) | 15 (4.5) | 15 (4.5) |
| High school degree | 341 (22.9) | 275 (23.8) | 66 (19.7) | 62 (20.0) |
| Some years of college | 366 (24.6) | 282 (24.4) | 84 (25.1) | 77 (24.8) |
| College degree | 505 (33.9) | 384 (33.2) | 121 (36.1) | 112 (36.0) |
| Unknown | 214 (14.4) | 165 (14.6) | 49 (14.6) | 45 (14.5) |
| Diabetes—No. (%) | 256 (17.2) | 202 (17.5) | 54 (16.1) | 53 (17.0) |
| Prevalent cardiovascular disease—No. (%) | 330 (22.1) | 252 (21.8) | 78 (23.3) | 74 (23.8) |
| RBC DHA, proportion of total fatty acids—Mean (SD) | 5.00 (1.37) | 5.00 (1.37) | 5.00 (1.39) | 5.04 (1.40) |
| Incident Alzheimer’s disease—No. (%) | 131 (8.8) | 77 (6.7) | 54 (16.1) 3 | 51 (16.4) 3 |
| Incident all-cause dementia—No. (%) | 168 (11.3) | 103 (8.9) | 65 (19.4) 3 | 61 (19.6) 3 |
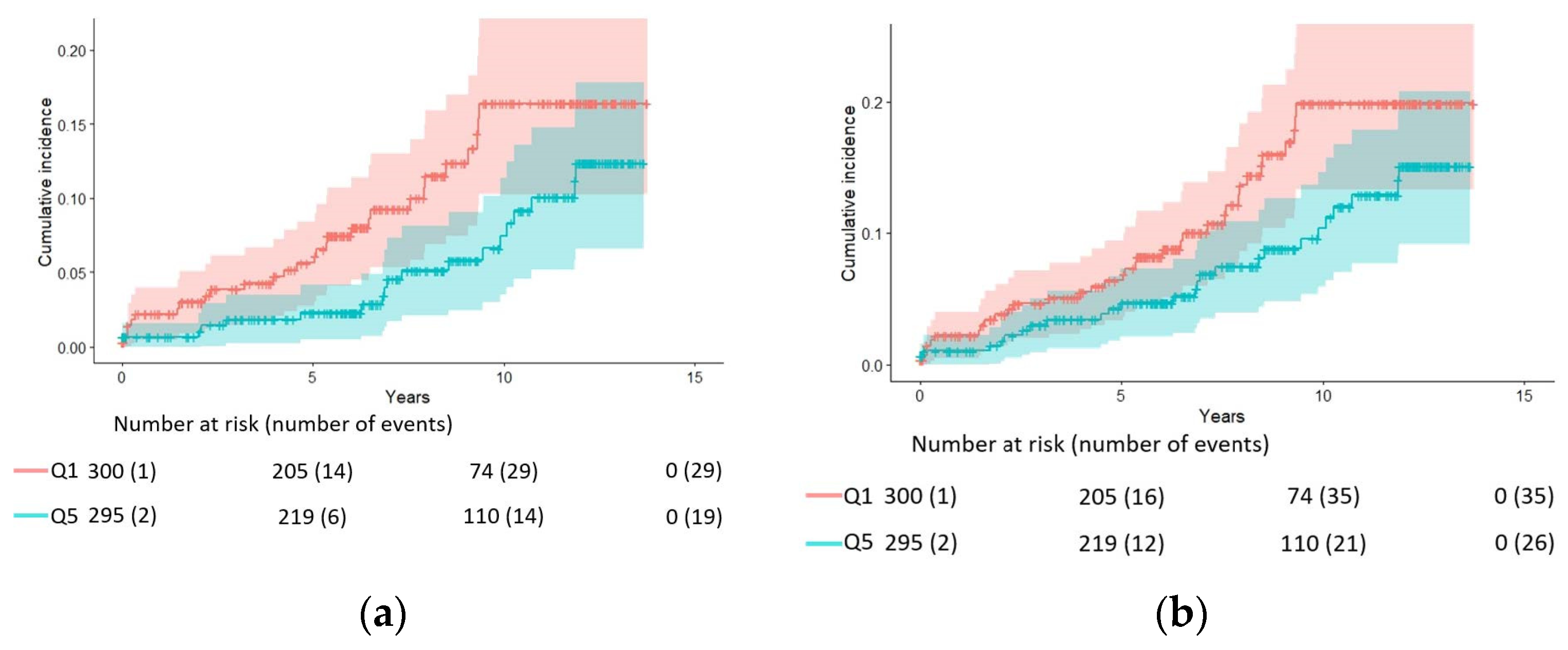
| Endpoint | HR (95% CI) for Quintiles of Red Blood Cell DHA | HR (95% CI) per SD 1 | |||||
|---|---|---|---|---|---|---|---|
| Q1 (<3.8%, Median = 3.4%) (n = 300) | Q2 (3.8% to <4.5%, Median = 4.2%) (n = 298) | Q3 (4.5% to <5.2%, Median = 4.8%) (n = 297) | Q4 (5.2% to 6.1%, Median = 5.6%) (n = 297) | Q5 (>6.1%, Median = 6.97%) (n = 295) | p for Trend | ||
| AD | |||||||
| N. of cases | 29 | 30 | 24 | 29 | 19 | 131 | |
| Model 1 | 1.00 | 0.72 (0.43, 1.21) | 0.61 (0.34, 1.09) | 0.72 (0.41, 1.25) | 0.48 (0.26, 0.87) | 0.04 | 0.80 (0.66, 0.98) |
| Model 2 | 1.00 | 0.78 (0.45, 1.33) | 0.64 (0.35, 1.18) | 0.75 (0.42, 1.35) | 0.51 (0.27, 0.98) | 0.07 | 0.82 (0.67, 1.01) |
| Model 3 | 1.00 | 0.77 (0.45, 1.33) | 0.64 (0.35, 1.18) | 0.75 (0.42, 1.33) | 0.51 (0.27, 0.96) | 0.06 | 0.82 (0.66, 1.00) |
| Dementia | |||||||
| N. of cases | 35 | 38 | 29 | 40 | 26 | 168 | |
| Model 1 | 1.00 | 0.78 (0.49, 1.26) | 0.64 (0.38, 1.08) | 0.87 (0.54, 1.41) | 0.56 (0.33, 0.94) | 0.09 | 0.85 (0.72, 1.01) |
| Model 2 | 1.00 | 0.80 (0.49, 1.29) | 0.65 (0.38, 1.11) | 0.88 (0.54, 1.45) | 0.57 (0.33, 0.99) | 0.12 | 0.86 (0.73, 1.03) |
| Model 3 | 1.00 | 0.79 (0.49, 1.29) | 0.64 (0.37, 1.11) | 0.87 (0.53, 1.44) | 0.56 (0.32, 0.97) | 0.10 | 0.86 (0.72, 1.02) |
| Endpoint | HR (95% CI) for Quintiles of Red Blood Cell DHA | HR (95% CI) per SD 1 | |||||
|---|---|---|---|---|---|---|---|
| Q1 (<3.8%, Median = 3.4%) (n = 284) | Q2 (3.8% to <4.5%, Median = 4.2%) (n = 282) | Q3 (4.5% to <5.2%, Median = 4.8%) (n = 284) | Q4 (5.2% to 6.1%, Median = 5.6%) (n = 280) | Q5 (>6.1%, Median = 6.97%) (n = 283) | p for Trend | ||
| AD | |||||||
| N. of cases | 15 | 18 | 9 | 17 | 13 | 72 | |
| Model 1 | 1.00 | 0.82 (0.41, 1.66) | 0.41 (0.17, 0.99) | 0.81 (0.38, 1.73) | 0.61 (0.28, 1.32) | 0.29 | 0.84 (0.64, 1.11) |
| Model 2 | 1.00 | 0.82 (0.40, 1.69) | 0.40 (0.16, 0.95) | 0.82 (0.37, 1.82) | 0.57 (0.24. 1.34) | 0.29 | 0.84 (0.62, 1.12) |
| Model 3 | 1.00 | 0.80 (0.39, 1.65) | 0.38 (0.16, 0.92) | 0.77 (0.35, 1.73) | 0.53 (0.22, 1.26) | 0.23 | 0.81 (0.61, 1.09) |
| Dementia | |||||||
| N. of cases | 19 | 22 | 13 | 23 | 14 | 91 | |
| Model 1 | 1.00 | 0.81 (0.43, 1.53) | 0.49 (0.24, 1.03) | 0.91 (0.48, 1.76) | 0.53 (0.26, 1.09) | 0.18 | 0.83 (0.66, 1.05) |
| Model 2 | 1.00 | 0.77 (0.41, 1.46) | 0.47 (0.23, 0.98) | 0.89 (0.45, 1.76) | 0.49 (0.23, 1.04) | 0.17 | 0.82 (0.64, 1.05) |
| Model 3 | 1.00 | 0.75 (0.39, 1.42) | 0.45 (0.21, 0.95) | 0.84 (0.43, 1.67) | 0.45 (0.21, 0.97) | 0.11 | 0.81 (0.63, 1.02) |
| Endpoint | HR (95% CI) for Quintiles of Red Blood Cell EPA | HR (95% CI) per SD 1 | |||||
|---|---|---|---|---|---|---|---|
| Q1 (<0.44%, Median = 0.37%) (n = 303) | Q2 (0.44% to <0.55%, Median = 0.49%) (n = 294) | Q3 (0.55% to <0.70%, Median = 0.61%) (n = 299) | Q4 (0.70% to 0.95%, Median = 0.78%) (n = 297) | Q5 (>0.95%, Median = 1.21%) (n = 297) | p for Trend | ||
| AD | |||||||
| N. of cases | 39 | 31 | 42 | 27 | 29 | 168 | |
| Model 1 | 1.00 | 0.68 (0.42, 1.10) | 0.97 (0.62, 1.51) | 0.53 (0.32, 0.91) | 0.74 (0.46, 1.20) | 0.10 | 0.97 (0.81, 1.16) |
| Model 2 | 1.00 | 0.69 (0.42, 1.12) | 0.99 (0.63, 1.55) | 0.57 (0.33, 0.98) | 0.76 (0.46, 1.25) | 0.19 | 0.98 (0.81, 1.16) |
| Model 3 | 1.00 | 0.69 (0.42, 1.12) | 0.98 (0.63, 1.55) | 0.56 (0.32, 0.96) | 0.74 (0.45, 1.23) | 0.17 | 0.97 (0.81, 1.16) |
| Dementia | |||||||
| N. of cases | 32 | 24 | 31 | 22 | 22 | 131 | |
| Model 1 | 1.00 | 0.61 (0.35, 1.06) | 0.84 (0.51, 1.38) | 0.50 (0.28, 0.89) | 0.66 (0.38, 1.14) | 0.13 | 0.96 (0.78, 1.19) |
| Model 2 | 1.00 | 0.62 (0.35, 1.09) | 0.90 (0.54, 1.50) | 0.55 (0.30, 1.01) | 0.69 (0.39, 1.24) | 0.20 | 0.98 (0.79, 1.21) |
| Model 3 | 1.00 | 0.62 (0.35, 1.09) | 0.90 (0.54, 1.50) | 0.54 (0.29, 0.99) | 0.68 (0.38, 1.21) | 0.17 | 0.97 (0.78, 1.21) |
| Endpoint | HR (95% CI) for Quintiles of Omega-3 Index | HR (95% CI) per SD 1 | |||||
|---|---|---|---|---|---|---|---|
| Q1 (<4.3%, Median = 3.9%) (n = 300) | Q2 (4.3% to <5.1%, Median = 4.7%) (n = 300) | Q3 (5.1% to <5.9%, Median = 5.4%) (n = 298) | Q4 (5.9% to 7.1%, Median = 6.3%) (n = 293) | Q5 (>7.1%, Median = 8.0%)(n = 299) | p for Trend | ||
| AD | |||||||
| N. of cases | 29 | 31 | 24 | 27 | 20 | 131 | |
| Model 1 | 1.00 | 0.81 (0.49, 1.36) | 0.60 (0.34, 1.08) | 0.74 (0.43, 1.29) | 0.52 (0.28, 0.93) | 0.04 | 0.83 (0.67, 1.02) |
| Model 2 | 1.00 | 0.90 (0.52, 1.54) | 0.62 (0.34, 1.15) | 0.81 (0.45, 1.45) | 0.55 (0.29, 1.05) | 0.08 | 0.85 (0.68, 1.05) |
| Model 3 | 1.00 | 0.90 (0.52, 1.54) | 0.61 (0.33, 1.14) | 0.81 (0.45, 1.45) | 0.55 (0.29, 1.03) | 0.07 | 0.84 (0.68, 1.04) |
| Dementia | |||||||
| N. of cases | 34 | 39 | 32 | 36 | 27 | 168 | |
| Model 1 | 1.00 | 0.90 (0.56, 1.44) | 0.72 (0.43, 1.22) | 0.88 (0.54, 1.44) | 0.61 (0.36, 1.03) | 0.09 | 0.87 (0.73, 1.04) |
| Model 2 | 1.00 | 0.93 (0.57, 1.50) | 0.73 (0.42, 1.25) | 0.93 (0.56, 1.54) | 0.63 (0.36, 1.09) | 0.14 | 0.88 (0.74, 1.05) |
| Model 3 | 1.00 | 0.93 (0.57, 1.51) | 0.72 (0.42, 1.24) | 0.92 (0.56, 1.53) | 0.62 (0.36, 1.07) | 0.12 | 0.88 (0.73, 1.05) |
| Variable | Alzheimer’s Disease | All-Cause Dementia | ||
|---|---|---|---|---|
| Hazard Ratio | Scaled β | Hazard Ratio | Scaled β | |
| Age, 1 year | 1.16 | 1.00 | 1.15 | 1.00 |
| APOE-ε4 carriership, yes | 3.12 | −7.59 | 2.78 | −7.30 |
| RBC DHA, Q5 to Q1 | 2.01 | −4.65 | 1.76 | −4.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sala-Vila, A.; Satizabal, C.L.; Tintle, N.; Melo van Lent, D.; Vasan, R.S.; Beiser, A.S.; Seshadri, S.; Harris, W.S. Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study. Nutrients 2022, 14, 2408. https://doi.org/10.3390/nu14122408
Sala-Vila A, Satizabal CL, Tintle N, Melo van Lent D, Vasan RS, Beiser AS, Seshadri S, Harris WS. Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study. Nutrients. 2022; 14(12):2408. https://doi.org/10.3390/nu14122408
Chicago/Turabian StyleSala-Vila, Aleix, Claudia L. Satizabal, Nathan Tintle, Debora Melo van Lent, Ramachandran S. Vasan, Alexa S. Beiser, Sudha Seshadri, and William S. Harris. 2022. "Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study" Nutrients 14, no. 12: 2408. https://doi.org/10.3390/nu14122408
APA StyleSala-Vila, A., Satizabal, C. L., Tintle, N., Melo van Lent, D., Vasan, R. S., Beiser, A. S., Seshadri, S., & Harris, W. S. (2022). Red Blood Cell DHA Is Inversely Associated with Risk of Incident Alzheimer’s Disease and All-Cause Dementia: Framingham Offspring Study. Nutrients, 14(12), 2408. https://doi.org/10.3390/nu14122408







