Too Jittery to Sleep? Temporal Associations of Actigraphic Sleep and Caffeine in Adolescents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Actigraphy Device and Scoring
2.3. Daily Alignment of Sleep and Diary and Data Lagging
2.4. Variables
2.4.1. Nightly Actigraphic Sleep Measures
2.4.2. Actigraphic Sleep Measures Calculated per Adolescent
2.4.3. Subjective Sleep Quality
2.5. Caffeinated Beverage Consumption
2.6. Covariates
2.7. Statistical Analyses
2.7.1. Main Analyses
2.7.2. Sensitivity Analyses by Adolescent’s Average Caffeinated Beverage Consumption
3. Results
3.1. Demographic Information
3.2. Nightly Sleep Measures Predicting Next-Day and Average Caffeinated Beverage Consumption (Within- and Between-Person Associations)
3.3. Associations of Sleep Variability with Average Caffeinated Beverage Consumption (Between-Person Associations)
3.4. Caffeinated Beverage Consumption Predicting Sleep Measures That Night and on Average (Within- and Between-Person Associations)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heckman, M.A.; Weil, J.; de Mejia, E.G. Caffeine (1, 3, 7-trimethylxanthine) in foods: A comprehensive review on consumption, functionality, safety, and regulatory matters. J. Food Sci. 2010, 75, R77–R87. [Google Scholar] [CrossRef]
- National Sleep Foundation. 2006 Sleep in America Poll—Teens and Sleep. Sleep Health 2015, 1, e5. [Google Scholar] [CrossRef]
- Temple, J.L. Review: Trends, safety, and recommendations for caffeine use in children and adolescents. J. Am. Acad. Child. Adolesc. Psychiatry 2019, 58, 36–45. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, G.A.; Carroll, M.E.; Thuras, P.D.; Cosgrove, K.P.; Roth, M.E. Caffeine dependence in teenagers. Drug Alcohol Depend. 2002, 66, 1–6. [Google Scholar] [CrossRef]
- Martin, C.A.; Cook, C.; Woodring, J.H.; Burkhardt, G.; Guenthner, G.; Omar, H.A.; Kelly, T.H. Caffeine use: Association with nicotine use, aggression, and other psychopathology in psychiatric and pediatric outpatient adolescents. Sci. World J. 2008, 8, 512–516. [Google Scholar] [CrossRef] [Green Version]
- Dimitriou, D.; Le Cornu, K.F.; Milton, P. The role of environmental factors on sleep patterns and school performance in adolescents. Front. Psychol. 2015, 6, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sampasa-Kanyinga, H.; Hamilton, H.A.; Chaput, J.P. Sleep duration and consumption of sugar-sweetened beverages and energy drinks among adolescents. Nutrition 2018, 48, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Troxel, W.M.; Tucker, J.S.; Ewing, B.; Miles, J.N.V.; D’Amico, E.J. Sleepy teens and energy drink use: Results from an ethnically diverse sample of youth. Behav. Sleep Med. 2018, 16, 223–234. [Google Scholar] [CrossRef] [Green Version]
- Veselska, Z.D.; Husarova, D.; Kosticova, M. Energy drinks consumption associated with emotional and behavioural problems via lack of sleep and skipped breakfast among adolescents. Int. J. Environ. Res. Public Health 2021, 18, 6055. [Google Scholar] [CrossRef]
- Yen, C.F.; Ko, C.H.; Yen, J.Y.; Cheng, C.P. The multidimensional correlates associated with short nocturnal sleep duration and subjective insomnia among Taiwanese adolescents. Sleep 2008, 31, 1515–1525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yilmaz, K.; Kilinçaslan, A.; Aydin, N.; Kul, S. Understanding sleep habits and associated factors can help to improve sleep in high school adolescents. Turk. J. Pediatr. 2011, 53, 430–436. [Google Scholar]
- Chung, K.F.; Cheung, M.M. Sleep-wake patterns and sleep disturbance among Hong Kong Chinese adolescents. Sleep 2008, 31, 185–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drescher, A.A.; Goodwin, J.L.; Silva, G.E.; Quan, S.F. Caffeine and screen time in adolescence: Associations with short sleep and obesity. J. Clin. Sleep Med. 2011, 7, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Halldorsson, T.I.; Kristjansson, A.L.; Thorisdottir, I.; Oddsdóttir, C.; Sveinbjörnsson, J.; Benediktsson, R.; Sigfúsdóttir, I.D.; Jörundsdóttir, H.; Gunnlaugsdóttir, H. Caffeine exposure from beverages and its association with self-reported sleep duration and quality in a large sample of Icelandic adolescents. Food Chem. Toxicol. 2021, 157, 112549. [Google Scholar] [CrossRef]
- Kim, S.Y.; Sim, S.; Choi, H.G. High stress, lack of sleep, low school performance, and suicide attempts are associated with high energy drink intake in adolescents. PLoS ONE 2017, 12, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lodato, F.; Araújo, J.; Barros, H.; Lopes, C.; Agodi, A.; Barchitta, M.; Ramos, E. Caffeine intake reduces sleep duration in adolescents. Nutr. Res. 2013, 33, 726–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nuss, T.; Morley, B.; Scully, M.; Wakefield, M. Energy drink consumption among Australian adolescents associated with a cluster of unhealthy dietary behaviours and short sleep duration. Nutr. J. 2021, 20, 1–10. [Google Scholar] [CrossRef]
- Patte, K.A.; Qian, W.; Leatherdale, S.T. Modifiable predictors of insufficient sleep durations: A longitudinal analysis of youth in the COMPASS study. Prev. Med. 2018, 106, 164–170. [Google Scholar] [CrossRef]
- Pollak, C.P.; Bright, D. Caffeine consumption and weekly sleep patterns in US seventh-, eighth-, and ninth-graders. Pediatrics 2003, 111, 42–46. [Google Scholar] [CrossRef] [Green Version]
- Wheaton, A.G.; Jones, S.E.; Cooper, A.C.; Croft, J.B. Short sleep duration among middle school and high school students—United States, 2015. Morb. Mortal. Wkly. Rep. 2018, 67, 85–90. [Google Scholar] [CrossRef] [Green Version]
- Calamaro, C.J.; Mason, T.B.A.; Ratcliffe, S.J. Adolescents living the 24/7 lifestyle: Effects of caffeine and technology on sleep duration and daytime functioning. Pediatrics 2009, 123, 4–11. [Google Scholar] [CrossRef] [Green Version]
- Merdad, R.A.; Merdad, L.A.; Nassif, R.A.; El-Derwi, D.; Wali, S.O. Sleep habits in adolescents of Saudi Arabia; distinct patterns and extreme sleep schedules. Sleep Med. 2014, 15, 1370–1378. [Google Scholar] [CrossRef]
- Reid, A.; Maldonado, C.; Baker, F. Sleep behavior of South African adolescents. Sleep 2002, 25, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Aepli, A.; Kurth, S.; Tesler, N.; Jenni, O.; Huber, R. Caffeine consuming children and adolescents show altered sleep behavior and deep sleep. Brain Sci. 2015, 5, 441–455. [Google Scholar] [CrossRef] [Green Version]
- Cusick, C.N.; Langberg, J.M.; Breaux, R.; Green, C.D.; Becker, S.P. Caffeine use and associations with sleep in adolescents with and without adhd. J. Pediatr. Psychol. 2020, 45, 643–653. [Google Scholar] [CrossRef]
- Yim, S.H.; Yang, K.I.; Kim, J.H.; Hwangbo, Y.; Kim, D.; Hong, S.B. Association between eveningness preference, socio-behavioral factors, and insomnia symptoms in Korean adolescents. Sleep Med. 2021, 82, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Fleig, D.; Randler, C. Association between chronotype and diet in adolescents based on food logs. Eat. Behav. 2009, 10, 115–118. [Google Scholar] [CrossRef]
- Gariépy, G.; Doré, I.; Whitehead, R.D.; Elgar, F.J. More than just sleeping in: A late timing of sleep is associated with health problems and unhealthy behaviours in adolescents. Sleep Med. 2019, 56, 66–72. [Google Scholar] [CrossRef]
- Giannotti, F.; Cortesi, F.; Sebastiani, T.; Ottaviano, S. Circadian preference, sleep and daytime behaviour in adolescence. J. Sleep Res. 2002, 11, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Saxvig, I.W.; Evanger, L.N.; Pallesen, S.; Hysing, M.; Sivertsen, B.; Gradisar, M.; Bjorvatn, B. Circadian typology and implications for adolescent sleep health. Results from a large, cross-sectional, school-based study. Sleep Med. 2021, 83, 63–70. [Google Scholar] [CrossRef]
- Roenneberg, T.; Kuehnle, T.; Pramstaller, P.P.; Ricken, J.; Havel, M.; Guth, A.; Merrow, M. A marker for the end of adolescence. Curr. Biol. 2004, 14, R1038–R1039. [Google Scholar] [CrossRef] [Green Version]
- Shimura, A.; Hideo, S.; Takaesu, Y.; Nomura, R.; Komada, Y.; Inoue, T. Comprehensive assessment of the impact of life habits on sleep disturbance, chronotype, and daytime sleepiness among high-school students. Sleep Med. 2018, 44, 12–18. [Google Scholar] [CrossRef]
- Marino, M.; Li, Y.; Rueschman, M.N.; Winkelman, J.W.; Ellenbogen, J.M.; Solet, J.M.; Dulin, H.; Berkman, L.F.; Buxton, O.M. Measuring sleep: Accuracy, sensitivity, and specificity of wrist actigraphy compared to polysomnography. Sleep 2013, 36, 1747–1755. [Google Scholar] [CrossRef]
- Buysse, D.J. Sleep health: Can we define It? Does it matter? Sleep 2014, 37, 9–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Dea, J.A. Consumption of nutritional supplements among adolescents: Usage and perceived benefits. Health Educ. Res. 2003, 18, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Drake, C.; Roehrs, T.; Shambroom, J.; Roth, T. Caffeine effects on sleep taken 0, 3, or 6 hours before going to bed. J. Clin. Sleep Med. 2013, 9, 1195–1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoffman, L.; Stawski, R.S. Persons as contexts: Evaluating between-person and within-person effects in longitudinal analysis. Res. Hum. Dev. 2009, 6, 97–120. [Google Scholar] [CrossRef]
- Curran, P.J.; Bauer, D.J. Effects in longitudinal models of change. Annu. Rev. Psychol. 2011, 62, 583–619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whalen, D.J.; Silk, J.S.; Semel, M.; Forbes, E.E.; Ryan, N.D.; Axelson, D.A.; Birmaher, B.; Dahl, R.E. Caffeine consumption, sleep, and affect in the natural environments of depressed youth and healthy controls. J. Pediatr. Psychol. 2008, 33, 358–367. [Google Scholar] [CrossRef] [Green Version]
- Lunsford-Avery, J.R.; Kollins, S.H.; Kansagra, S.; Wang, K.W.; Engelhard, M.M. Impact of daily caffeine intake and timing on electroencephalogram-measured sleep in adolescents. J. Clin. Sleep Med. 2021, jcsm-9736. [Google Scholar] [CrossRef]
- Short, M.A.; Gradisar, M.; Lack, L.C.; Wright, H.; Carskadon, M.A. The discrepancy between actigraphic and sleep diary measures of sleep in adolescents. Sleep Med. 2012, 13, 378–384. [Google Scholar] [CrossRef]
- Crowley, S.J.; van Reen, E.; LeBourgeois, M.K.; Acebo, C.; Tarokh, L.; Seifer, R.; Barker, D.H.; Carskadon, M.A. A longitudinal assessment of sleep timing, circadian phase, and phase angle of entrainment across human adolescence. PLoS ONE 2014, 9, e112199. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, A.M.; Kasprzak, C.M.; Mansouri, T.H.; Gregory, A.M.; Barich, R.A.; Hatzinger, L.A.; Leone, L.A.; Temple, J.L. An ecological perspective of food choice and eating autonomy among adolescents. Front. Psychol. 2021, 12, 654139. [Google Scholar] [CrossRef] [PubMed]
- Reichman, N.E.; Teitler, J. Fragile Families: Sample and design. Child. Youth Serv. Rev. 2001, 23, 303–326. [Google Scholar] [CrossRef]
- Wagmiller, R.L.J. How Representative are the Fragile Study Families? A Comparison of the Early Childhood Longitudinal Study-Birth Cohort and Fragile Families Samples; Working Papers 1216; Princeton University: Princeton, NJ, USA, 2010; Volume 716, pp. 1–28. [Google Scholar]
- Lachat, C.; Hawwash, D.; Ocké, M.C.; Berg, C.; Forsum, E.; Hörnell, A.; Larsson, C.L.; Sonestedt, E.; Wirfält, E.; Åkesson, A.; et al. Strengthening the reporting of observational studies in epidemiology-nutritional epidemiology (STROBE-nut): An extension of the STROBE statement. PLoS Med. 2016, 13, e1002036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lauderdale, D.S.; Knutson, K.L.; Yan, L.L.; Rathouz, P.J.; Hulley, S.B.; Sidney, S.; Liu, K. Objectively measured sleep characteristics among early-middle-aged adults: The CARDIA study. Am. J. Epidemiol. 2006, 164, 5–16. [Google Scholar] [CrossRef] [Green Version]
- Master, L.; Nye, R.T.; Lee, S.; Nahmod, N.G.; Mariani, S.; Hale, L.; Buxton, O.M. Bidirectional, daily temporal associations between sleep and physical activity in adolescents. Sci. Rep. 2019, 9, 1–14. [Google Scholar]
- Buxton, O.M.; Lee, S.; Beverly, C.; Berkman, L.F.; Moen, P.; Kelly, E.L.; Hammer, L.B.; Almeida, D.M. Work-family conflict and employee sleep: Evidence from IT workers in the Work, Family and Health Study. Sleep 2016, 39, 1911–1918. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Wang, R.; Zee, P.; Lutsey, P.L.; Javaheri, S.; Alcántara, C.; Jackson, C.L.; Williams, M.A.; Redline, S. Racial/ethnic differences in sleep disturbances: The Multi-Ethnic Study of Atherosclerosis (MESA). Sleep 2015, 38, 877–888. [Google Scholar] [CrossRef]
- Phillips, A.J.K.; Clerx, W.M.; O’Brien, C.S.; Sano, A.; Barger, L.K.; Picard, R.W.; Lockley, S.W.; Klerman, E.B.; Czeisler, C.A. Irregular sleep/wake patterns are associated with poorer academic performance and delayed circadian and sleep/wake timing. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Wittmann, M.; Dinich, J.; Merrow, M.; Roenneberg, T. Social jetlag: Misalignment of biological and social time. Chronobiol. Int. 2006, 23, 497–509. [Google Scholar] [CrossRef]
- Kuczmarski, R.; Ogden, C.; Guo, S.; Grummer-Strawn, L.; Flegal, K.; Mei, Z.; Wei, R.; Curtin, L.R.; Roche, A.F.; Johnson, C.L. 2000 CDC growth charts for the United States: Methods and development. Vital Health Stat. 2002, 11, 1–190. [Google Scholar]
- Keys, A.; Fidanza, F.; Karvonen, M.J.; Kimura, N.; Taylor, H.L. Indices of relative weight and obesity. J. Chronic Dis. 1972, 25, 329–343. [Google Scholar] [CrossRef]
- Kline, R.B. Principles and Practice of Structural Equation Modeling, 4th ed; Guilford Publications: New York, NY, USA, 2016. [Google Scholar]
- Koo, T.K.; Li, M.Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [Green Version]
- Horton, N.J.; Lipsitz, S.R. Review of software to fit generalized estimating equation regression models. Am. Stat. 1999, 53, 160–169. [Google Scholar]
- Liang, K.Y.; Zeger, S.L. Longitudinal data analysis using generalized linear models. Biometrika 1986, 73, 13–22. [Google Scholar] [CrossRef]
- Howard, A.L. Leveraging time-varying covariates to test within- and between-person effects and interactions in the multilevel linear model. Emerg. Adulthood 2015, 3, 400–412. [Google Scholar] [CrossRef]
- Van Dyk, T.R.; Zhang, N.; Combs, A.; Howarth, T.; Whitacre, C.; McAlister, S.; Beebe, D.W. Feasibility and impact on daytime sleepiness of an experimental protocol inducing variable sleep duration in adolescents. PLoS ONE 2019, 14, e0218894. [Google Scholar] [CrossRef]
- Fuligni, A.J.; Arruda, E.H.; Krull, J.L.; Gonzales, N.A. Adolescent sleep duration, variability, and peak levels of achievement and mental health. Child. Dev. 2018, 89, e18–e28. [Google Scholar] [CrossRef] [PubMed]
- Zuraikat, F.M.; Makarem, N.; Redline, S.; Aggarwal, B.; Jelic, S.; St-Onge, M.-P. Sleep regularity and cardiometabolic heath: Is variability in sleep patterns a risk factor for excess adiposity and glycemic dysregulation? Curr. Diab. Rep. 2020, 20, 38. [Google Scholar] [CrossRef]
- Asarnow, L.D.; McGlinchey, E.; Harvey, A.G. The effects of bedtime and sleep duration on academic and emotional outcomes in a nationally representative sample of adolescents. J. Adolesc. Health 2014, 54, 350–356. [Google Scholar] [CrossRef] [Green Version]
- Collings, P.J. Independent associations of sleep timing, duration and quality with adiposity and weight status in a national sample of adolescents: The UK Millennium Cohort Study. J. Sleep Res. 2021, 22, e13436. [Google Scholar] [CrossRef]
- Nagare, R.; Woo, M.; Macnaughton, P.; Plitnick, B.; Tinianov, B.; Figueiro, M. Article access to daylight at home improves circadian alignment, sleep, and mental health in healthy adults: A crossover study. Int. J. Environ. Res. Public Health 2021, 18, 9980. [Google Scholar] [CrossRef]
- Lara, T.; Madrid, J.A.; Correa, Á. The vigilance decrement in executive function is attenuated when individual chronotypes perform at their optimal time of day. PLoS ONE 2014, 9, e88820. [Google Scholar] [CrossRef] [PubMed]
- Reichert, C.F.; Veitz, S.; Bühler, M.; Gruber, G.; Deuring, G.; Rehm, S.S.; Rentsch, K.; Garbazza, C.; Meyer, M.; Slawik, H.; et al. Wide awake at bedtime? Effects of caffeine on sleep and circadian timing in male adolescents—A randomized crossover trial. Biochem. Pharmacol. 2021, 191, 114283. [Google Scholar] [CrossRef] [PubMed]
- Zwyghuizen-Doorenbos, A.; Roehrs, T.A.; Lipschutz, L.; Timms, V.; Roth, T. Effects of caffeine on alertness. Psychopharmacology 1990, 100, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, J.; Sawers, S.J.A. The absolute bioavailability of caffeine in man. Eur. J. Clin. Pharmacol. 1983, 24, 93–98. [Google Scholar] [CrossRef]
- Clark, I.; Landolt, H.P. Coffee, caffeine, and sleep: A systematic review of epidemiological studies and randomized controlled trials. Sleep Med. Rev. 2017, 31, 70–78. [Google Scholar] [CrossRef] [Green Version]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; U.S. Department of Health and Human Services and U.S. Department of Agriculture: Washington, DC, USA, 2015; pp. 1–125.
| Variable | M or % | SD or n |
|---|---|---|
| Demographic and household | ||
| Age | 15.39 | 0.52 |
| Sex a | ||
| Female | 53% | 311 |
| Male | 47% | 278 |
| Race/ethnicity | ||
| Black/African American | 41% | 240 |
| Hispanic and/or Latino | 25% | 149 |
| White/Caucasian | 19% | 112 |
| Other, b mixed, or none | 15% | 88 |
| Body mass index percentile c | 73.87 | 25.21 |
| Annual household income (USD) | $64,906 | $57,879 |
| Primary caregiver’s highest education level | ||
| Did not graduate high school | 14% | 85 |
| High school graduate | 18% | 106 |
| Completed some college | 47% | 276 |
| College graduate | 21% | 122 |
| Youth living arrangements | ||
| Lives with 2 married/cohabiting biological parents | 32% | 187 |
| Lives with <2 biological parents | 68% | 402 |
| School attendance | ||
| Attended school (proportion of days) | 0.44 | 0.34 |
| Nightly sleep measures | ||
| Sleep duration (h) | 7.79 | 1.08 |
| Sleep onset (clock time) | 0:28 | 1:44 |
| Sleep midpoint (clock time) | 4:21 | 1:42 |
| Sleep offset (clock time) | 8:20 | 1:47 |
| Sleep maintenance efficiency (%) | 90.70 | 3.43 |
| Subjective sleep quality d | 2.36 | 0.50 |
| Sleep variability measures e | ||
| Variability (SD) of sleep duration (h) | 1.57 | 0.80 |
| Variability (SD) of sleep onset (h) | 1.30 | 0.74 |
| Variability (SD) of sleep midpoint (h) | 1.22 | 0.65 |
| Variability (SD) of sleep offset (h) | 1.56 | 0.90 |
| SRI f | 48.35 | 13.38 |
| Social jetlag (h) g | 1.79 | 1.15 |
| Dietary intake | ||
| Consumed ≥1 cup caffeinated beverage (proportion of days) h | 0.61 | 0.34 |
| Consumed ≥1 caffeinated beverage 0–24% of the days | 19% | 112 |
| Consumed ≥1 caffeinated beverage 25–49% of the days | 15% | 88 |
| Consumed ≥1 caffeinated beverage 50–74% of the days | 22% | 129 |
| Consumed ≥1 caffeinated beverage 75–100% of the days | 44% | 260 |
| Model Predictor | OR | 95%CI OR | |
|---|---|---|---|
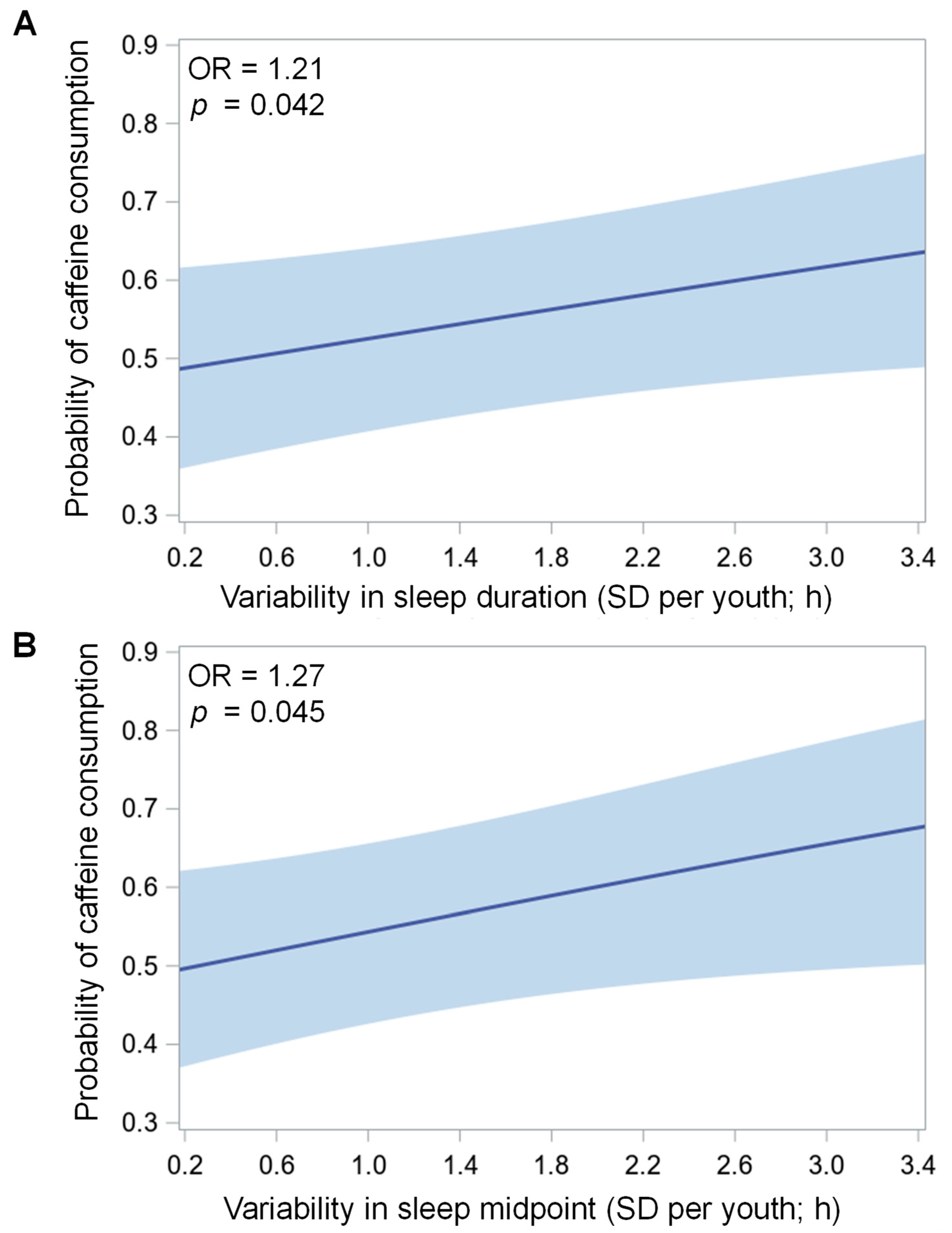
| Sleep duration (SD, h) | 1.21 * | 1.01 | 1.45 |
| Sleep onset (SD, h) | 1.19 † | 0.97 | 1.46 |
| Sleep midpoint (SD, h) | 1.27 * | 1.01 | 1.59 |
| Sleep offset (SD, h) | 1.17 † | 1.00 | 1.38 |
| SRI a | 0.99 | 0.98 | 1.00 |
| Social jetlag (h) b | 1.07 | 0.91 | 1.25 |
| Model Outcome | Within-Person | Between-Person | ||||
|---|---|---|---|---|---|---|
| b | 95% CI | b | 95% CI | |||
| Sleep duration (h) | <0.01 | −0.22 | 0.23 | 0.04 | −0.24 | 0.32 |
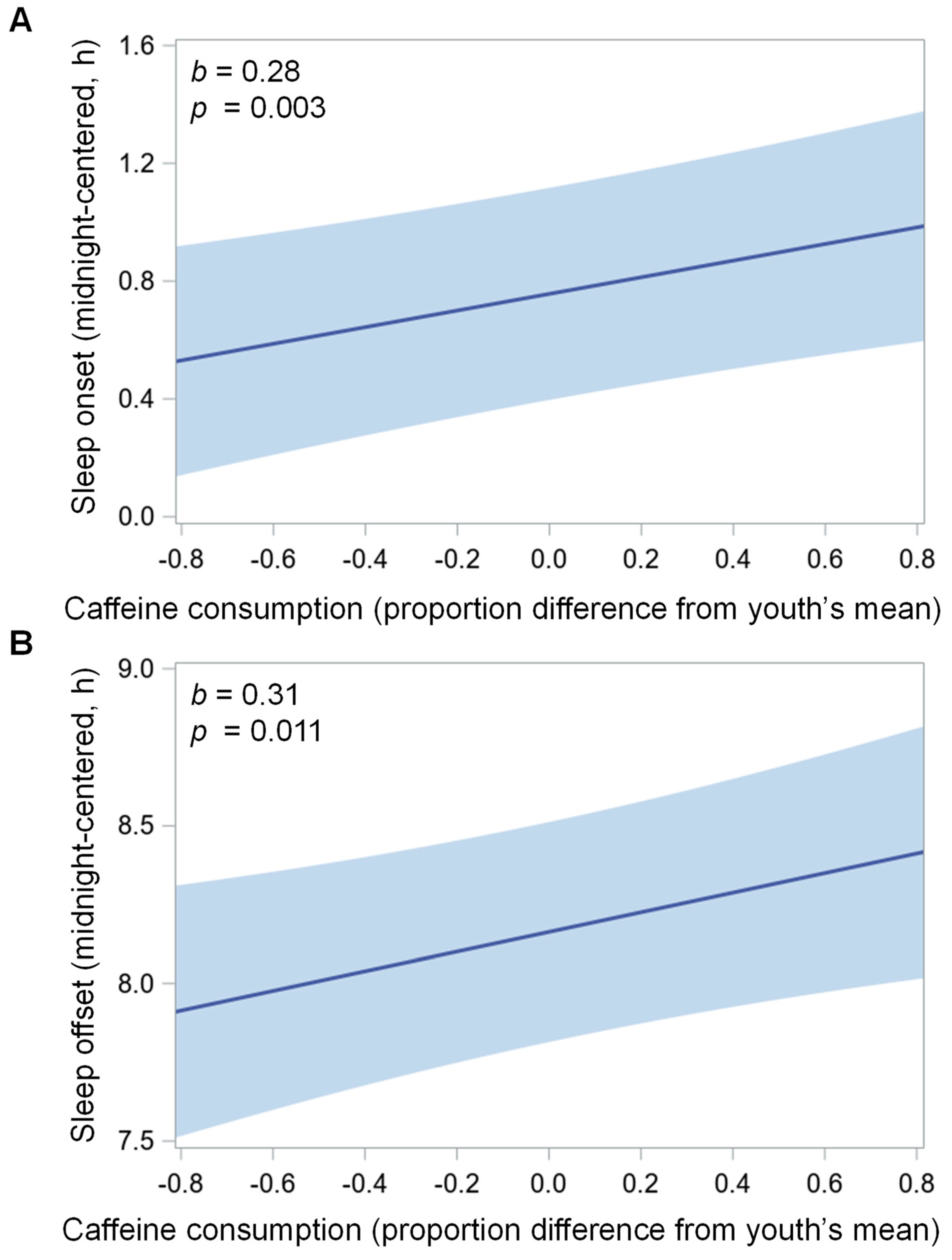
| Sleep onset (h) | 0.28 ** | 0.10 | 0.47 | −0.08 | −0.46 | 0.30 |
| Sleep midpoint (h) | 0.28 ** | 0.11 | 0.46 | −0.06 | −0.41 | 0.30 |
| Sleep offset (h) | 0.31 * | 0.07 | 0.55 | −0.05 | −0.41 | 0.32 |
| Sleep maintenance efficiency (%) | −0.05 | −0.45 | 0.35 | −0.13 | −0.98 | 0.72 |
| Subjective sleep quality a | 0.01 | −0.05 | 0.08 | −0.07 | −0.20 | 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mathew, G.M.; Reichenberger, D.A.; Master, L.; Buxton, O.M.; Chang, A.-M.; Hale, L. Too Jittery to Sleep? Temporal Associations of Actigraphic Sleep and Caffeine in Adolescents. Nutrients 2022, 14, 31. https://doi.org/10.3390/nu14010031
Mathew GM, Reichenberger DA, Master L, Buxton OM, Chang A-M, Hale L. Too Jittery to Sleep? Temporal Associations of Actigraphic Sleep and Caffeine in Adolescents. Nutrients. 2022; 14(1):31. https://doi.org/10.3390/nu14010031
Chicago/Turabian StyleMathew, Gina Marie, David A. Reichenberger, Lindsay Master, Orfeu M. Buxton, Anne-Marie Chang, and Lauren Hale. 2022. "Too Jittery to Sleep? Temporal Associations of Actigraphic Sleep and Caffeine in Adolescents" Nutrients 14, no. 1: 31. https://doi.org/10.3390/nu14010031
APA StyleMathew, G. M., Reichenberger, D. A., Master, L., Buxton, O. M., Chang, A.-M., & Hale, L. (2022). Too Jittery to Sleep? Temporal Associations of Actigraphic Sleep and Caffeine in Adolescents. Nutrients, 14(1), 31. https://doi.org/10.3390/nu14010031






