Curcumin Supplementation (Meriva®) Modulates Inflammation, Lipid Peroxidation and Gut Microbiota Composition in Chronic Kidney Disease
Abstract
:1. Introduction
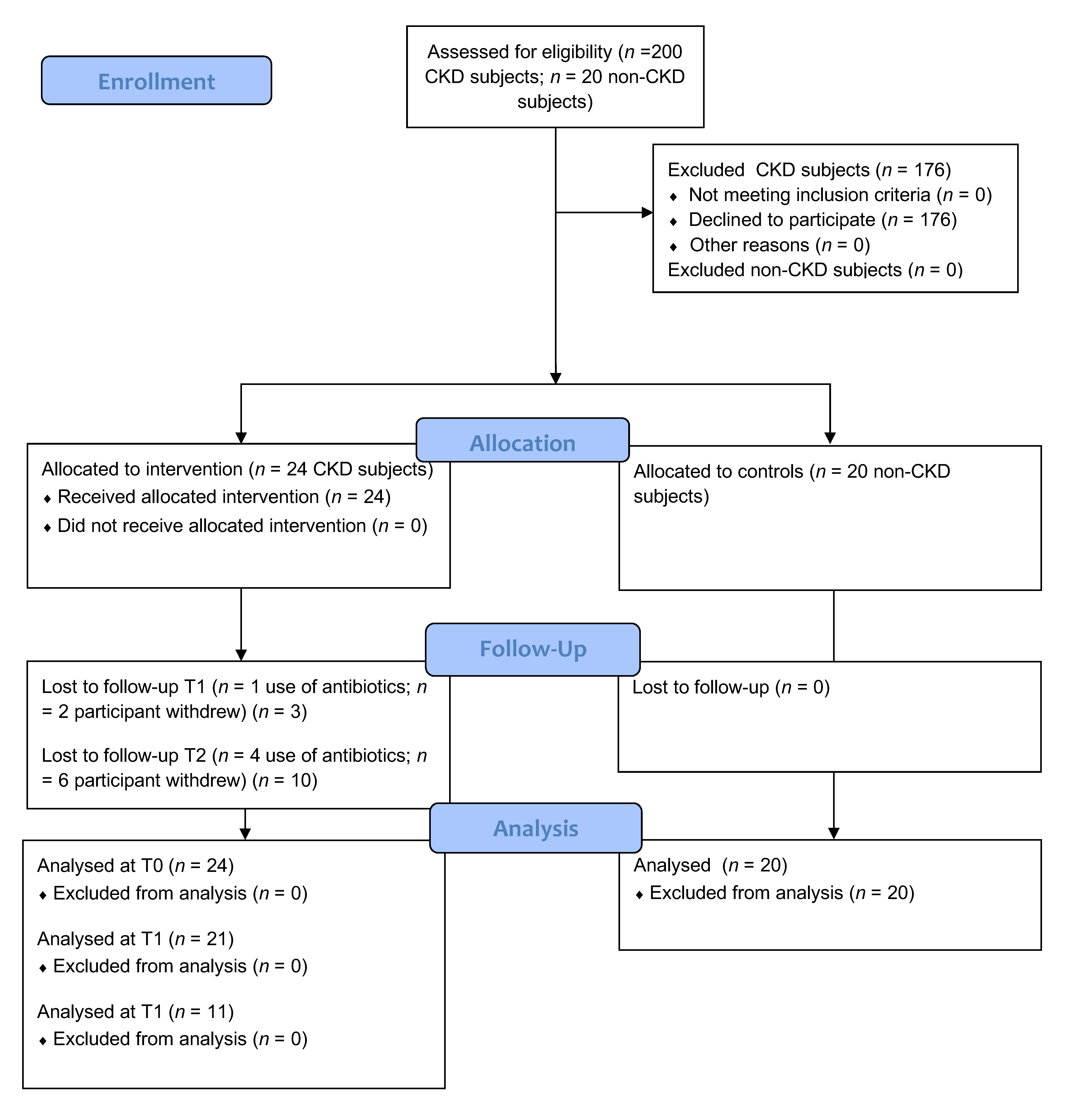
2. Materials and Methods
2.1. Patients
2.2. Study Design and Supplementation
2.3. Clinical Parameters
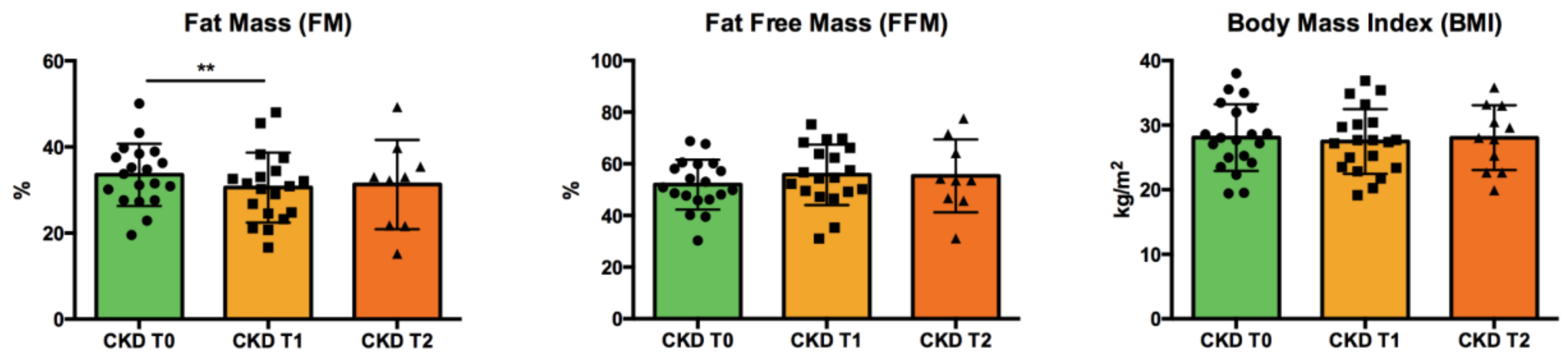
2.4. Body Composition
2.5. Dietary Habits and Nutritional Counseling
2.6. Inflammation and Lipid Peroxidation
2.7. LC–MS/MS Analysis of Uremic Toxins
2.8. Gut Microbiota Sequencing
2.9. Statistical Analysis
3. Results
3.1. Patients’ Characteristics: Anthropometric Data and Clinical Parameters
3.2. Nutrients Intake
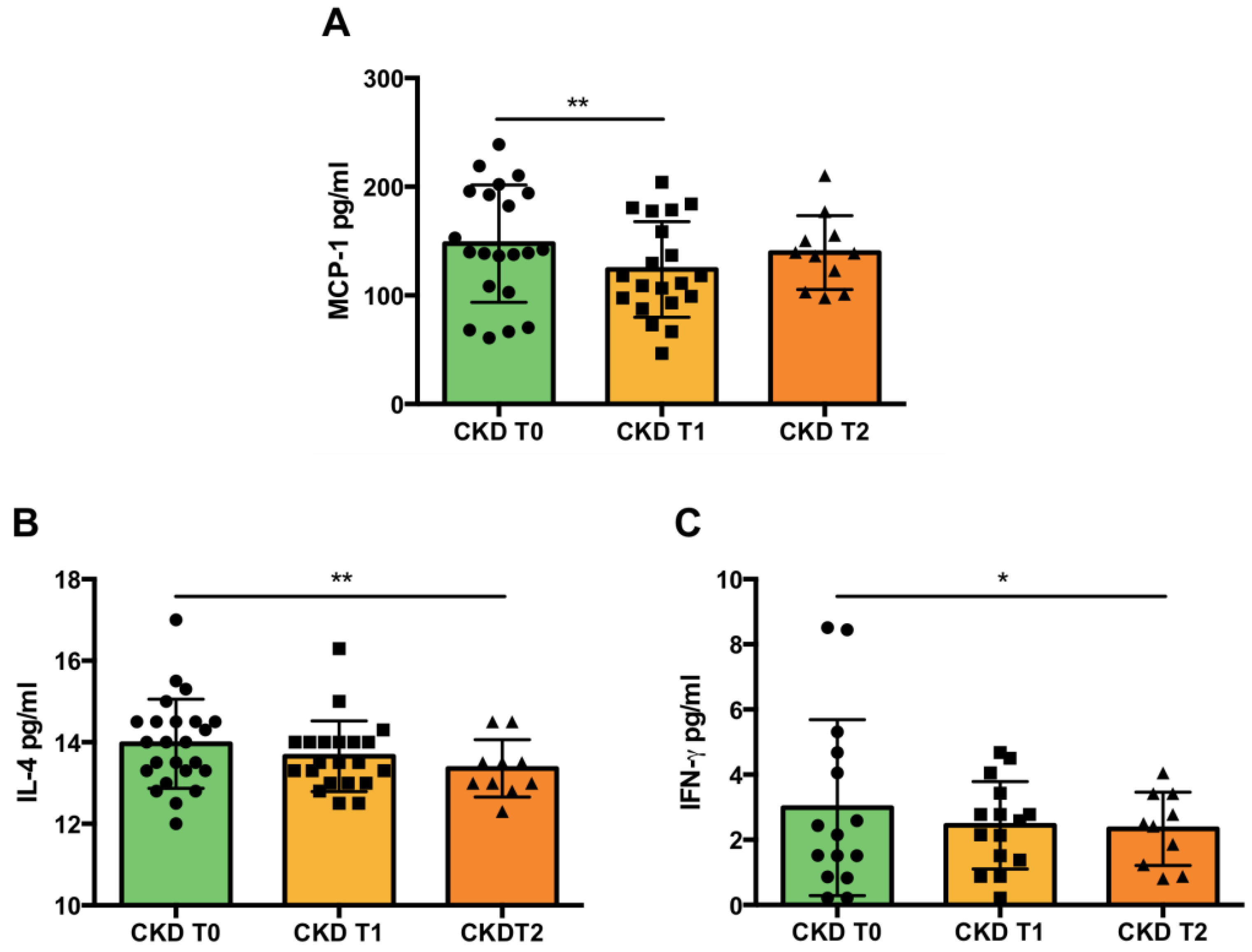
3.3. Inflammation
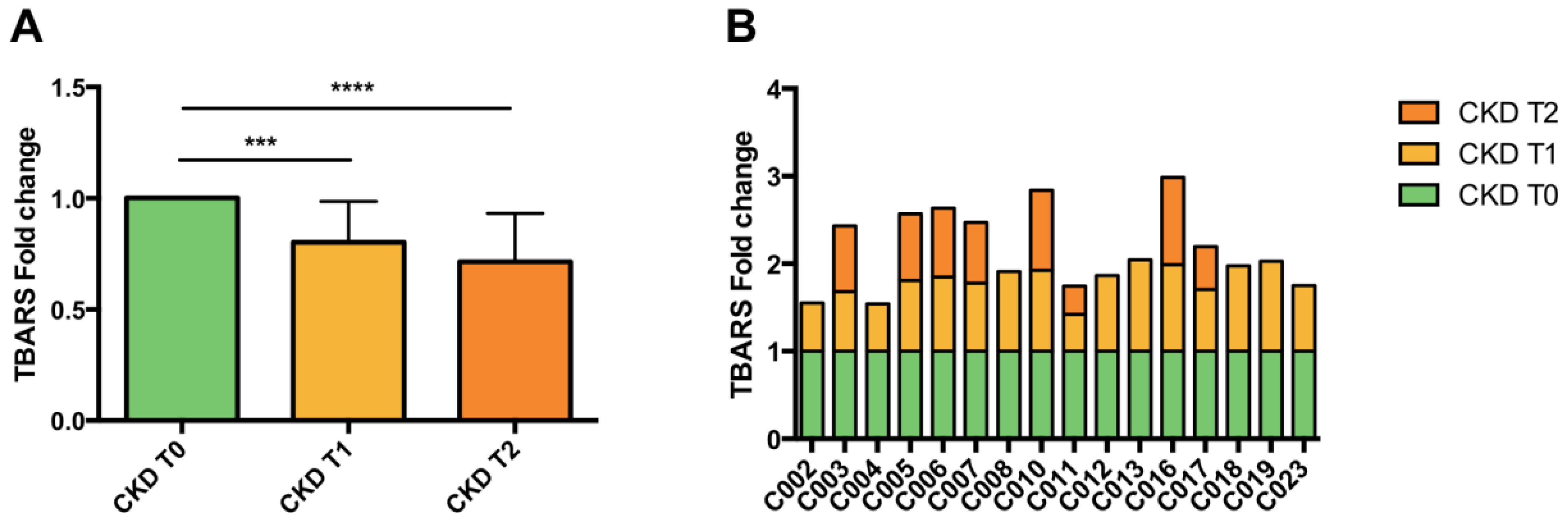
3.4. Lipid Peroxidation
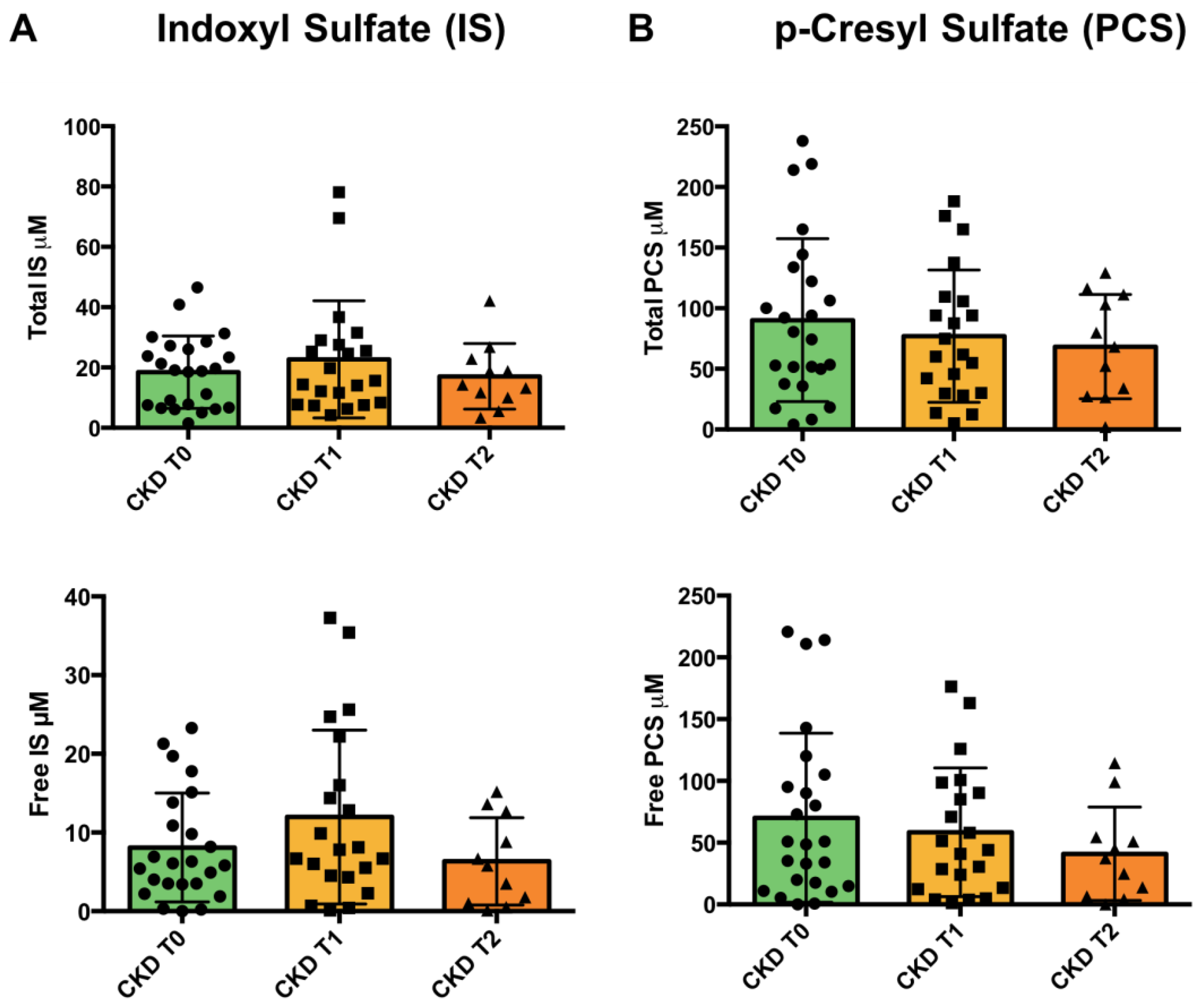
3.5. Uremic Toxins
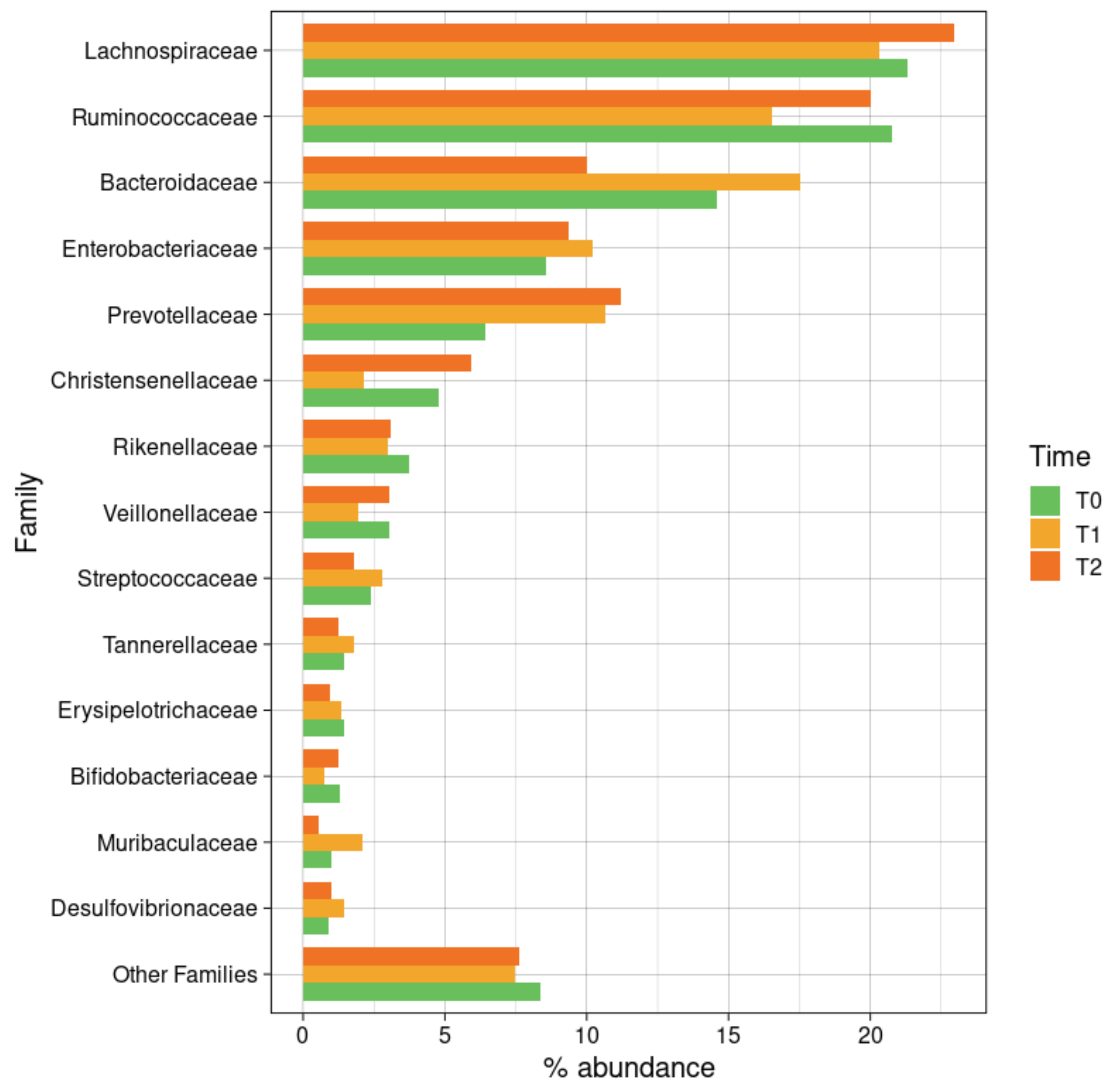
3.6. Gut Microbiota Analysis
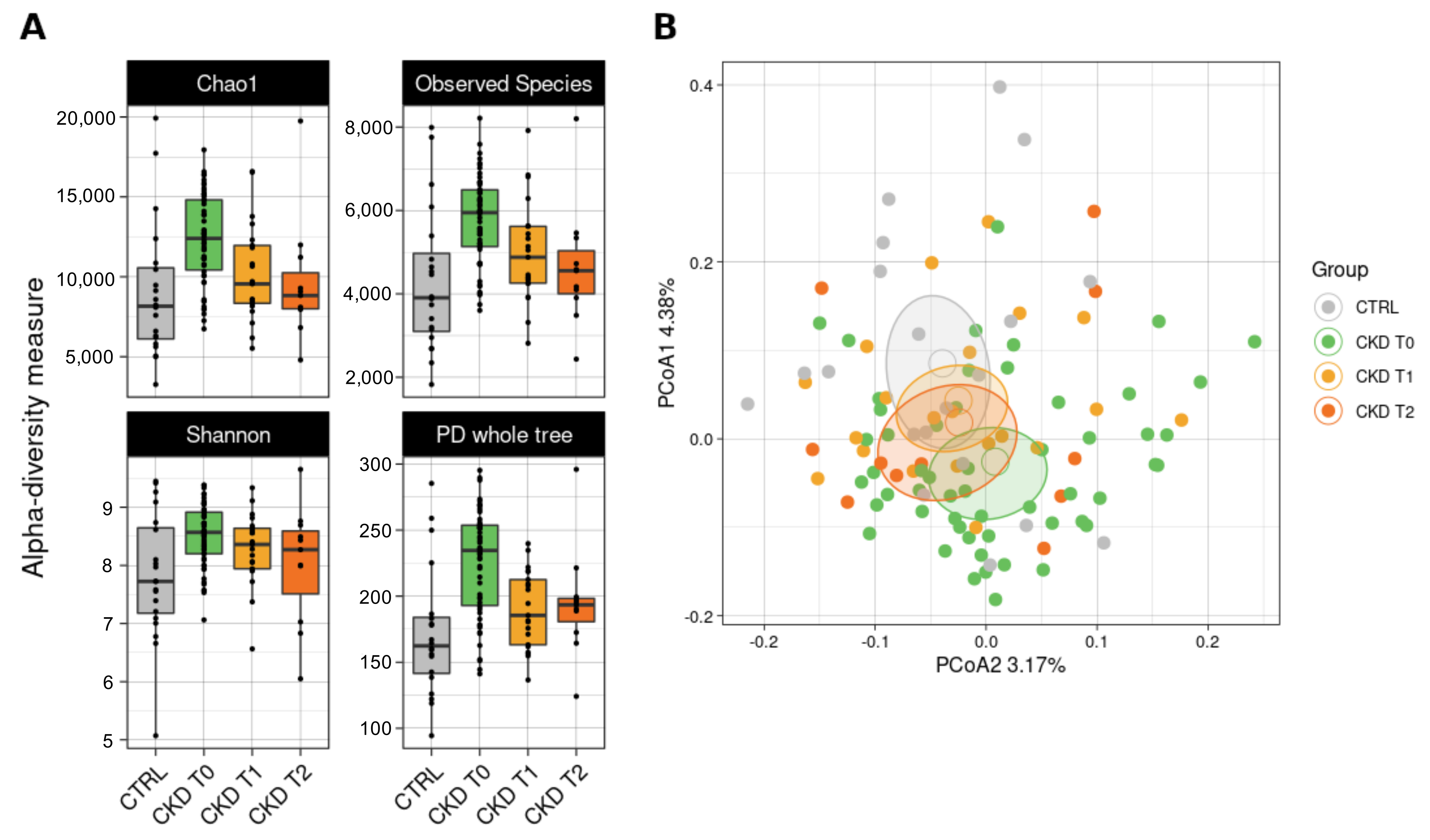
3.6.1. Bacterial Biodiversity in the Dataset
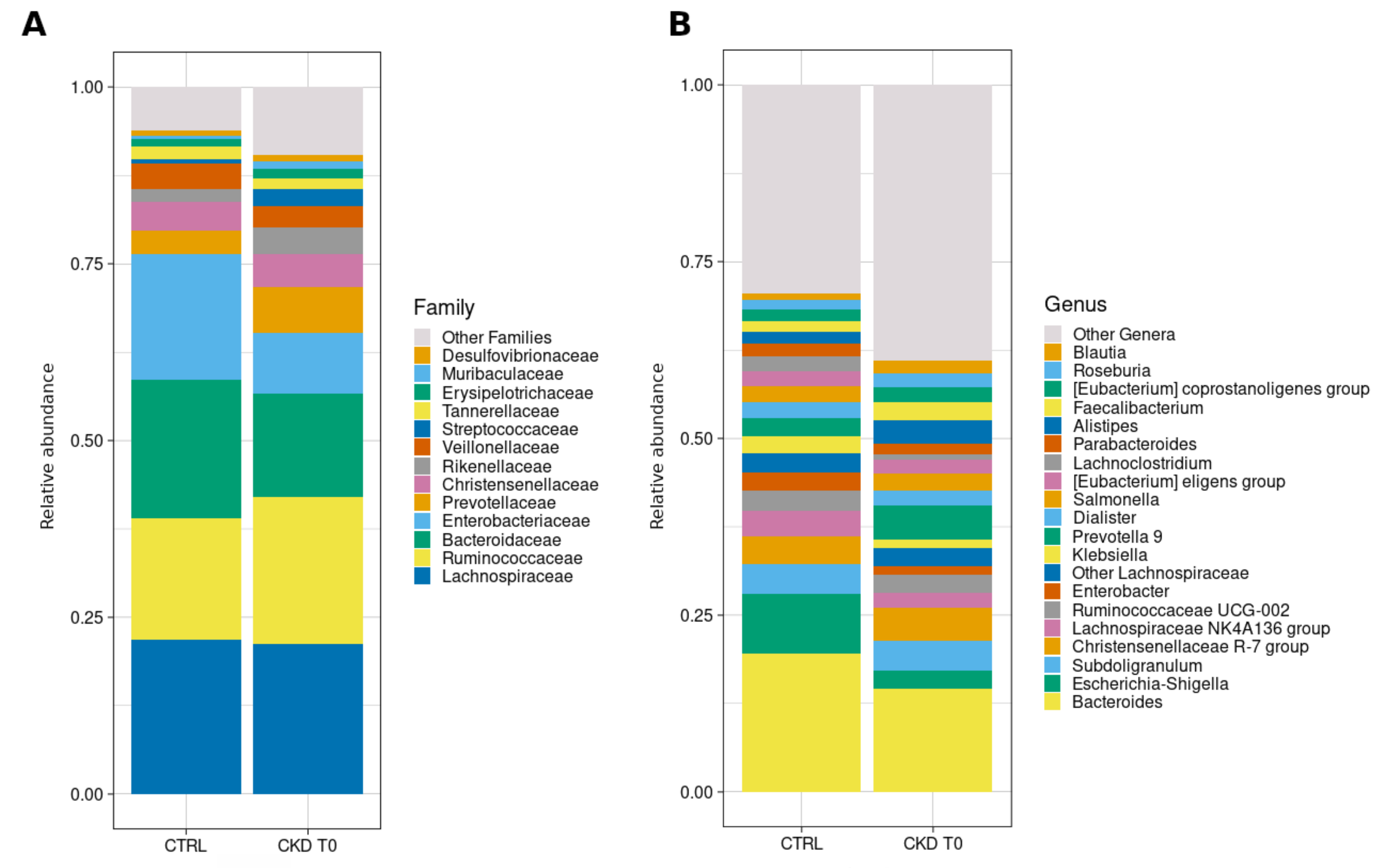
3.6.2. Taxonomic Composition
3.6.3. Comparisons with Healthy Subjects
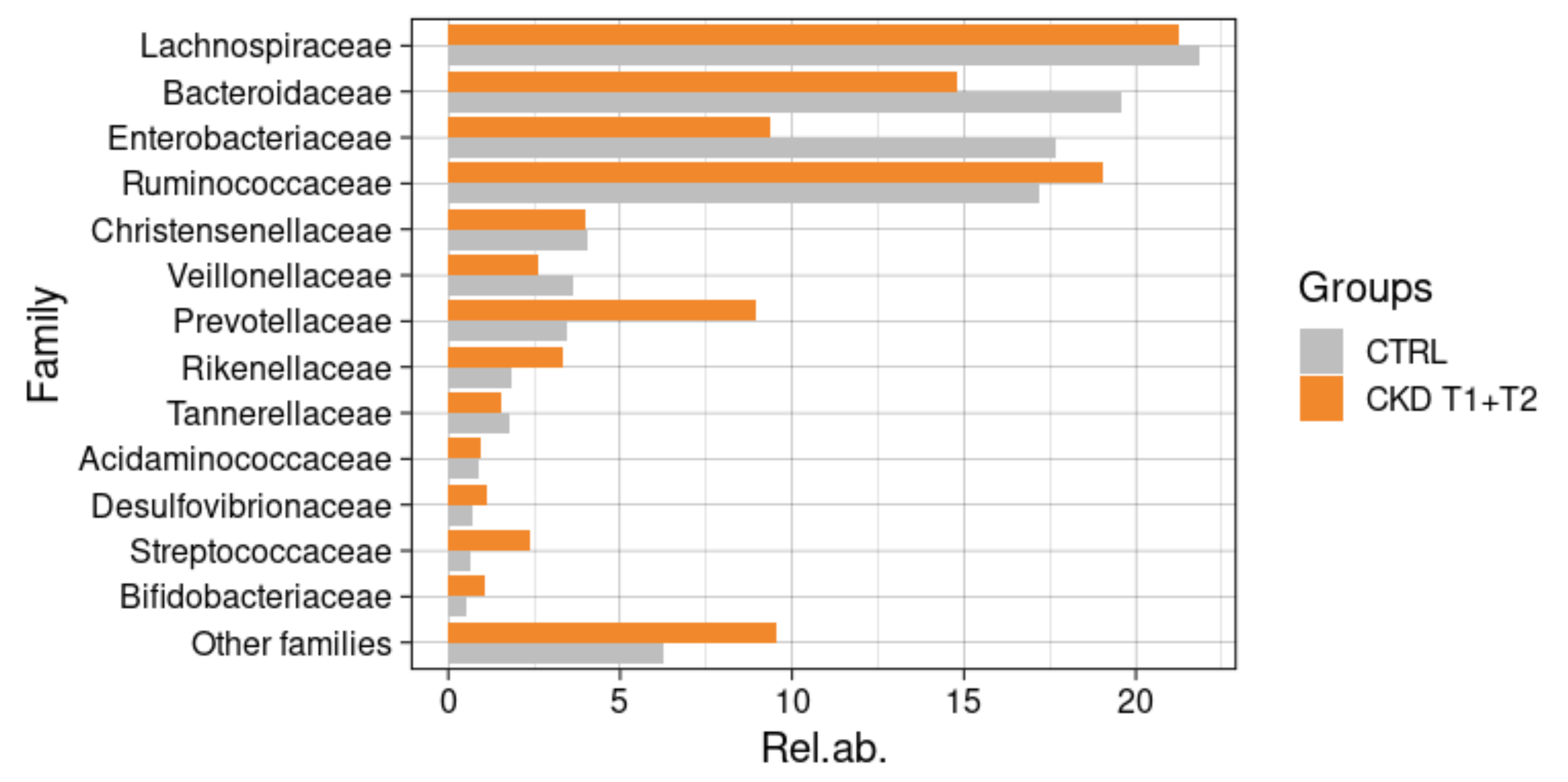
3.6.4. Taxonomic Changes in CKD Patients during Meriva® Supplementation
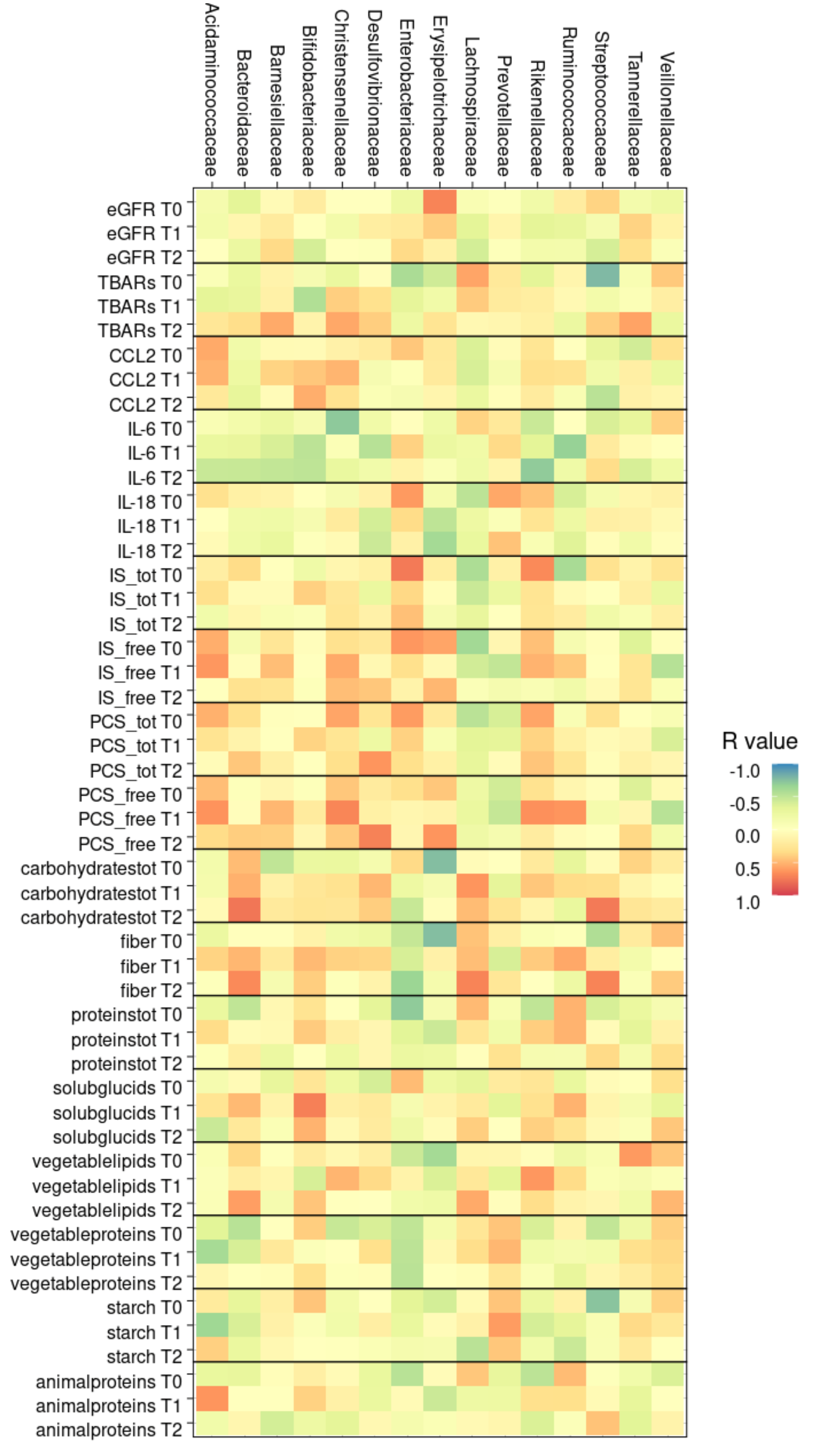
3.6.5. Correlation between Gut Microbiota and Clinical Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global prevalence of chronic kidney disease—A systematic review and meta-analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar]
- Cozzolino, M.; Galassi, A.; Pivari, F.; Ciceri, P.; Conte, F. The Cardiovascular Burden in End-Stage Renal Disease. Contrib. Nephrol. 2017, 191, 44–57. [Google Scholar]
- Go, A.S.; Chertow, G.M.; Fan, D.; Mcculloch, C.E.; Hsu, C.-Y. Chronic Kidney Disease and the Risks of Death, Cardiovascular Events, and Hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef]
- Carrero, J.J.; Stenvinkel, P. Inflammation in End-Stage Renal Disease-What Have We Learned in 10 Years? In Seminars in Dialysis; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2010. [Google Scholar]
- Levey, A.S.; Greene, T.; Kusek, J.W.; Beck, G.J. MDRD Study Group: A simplified equation to predict glomerular filtration rate from serum creatinine. J. Am. Soc. Nephrol. 2000, 11, 155. [Google Scholar]
- Kopple, J.D.; Levey, A.S.; Greene, T.; Chumlea, W.C.; Gassman, J.J.; Hollinger, D.L.; Maroni, B.J.; Merrill, D.; Scherch, L.K.; Schulman, G.; et al. Effect of dietary protein restriction on nutritional status in the Modification of Diet in Renal Disease Study. Kidney Int. 1997, 52, 778–791. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.S.; Coresh, J.; Bolton, K.; Culleton, B.; Harvey, K.S.; Ikizler, T.A.; Johnson, C.A.; Kausz, A.; Kimmel, P.L.; Kusek, J.; et al. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am. J. Kidney Dis. 2002, 39, S1–S266. [Google Scholar]
- Fan, Y.; Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 2021, 19, 55–71. [Google Scholar] [CrossRef]
- Wu, I.W.; Hsu, K.H.; Lee, C.C.; Sun, C.Y.; Hsu, H.J.; Tsai, C.J.; Tzen, C.Y.; Wang, Y.C.; Lin, C.Y.; Wu, M.S. P-cresyl sulphate and indoxyl sulphate predict progression of chronic kidney disease. Nephrol. Dial. Transplant. 2011, 26, 938–947. [Google Scholar] [CrossRef] [Green Version]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; Desantis, T.Z.; Ni, Z.; Nguyen, T.H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef] [Green Version]
- Briskey, D.; Tucker, P.; Johnson, D.W.; Coombes, J.S. The role of the gastrointestinal tract and microbiota on uremic toxins and chronic kidney disease development. Clin. Exp. Nephrol. 2017, 21, 7–15. [Google Scholar] [CrossRef]
- Fulop, T.; Witkowski, J.M.; Pawelec, G.; Alan, C.; Larbi, A. On the immunological theory of aging. Interdiscip. Top. Gerontol. 2014, 39, 163–176. [Google Scholar]
- Tsai, Y.L.; Lin, T.L.; Chang, C.J.; Wu, T.R.; Lai, W.F.; Lu, C.C.; Lai, H.C. Probiotics, prebiotics and amelioration of diseases. J. Biomed. Sci. 2019, 26, 1–8. [Google Scholar] [CrossRef]
- Quigley, E.M.M. Nutraceuticals as modulators of gut microbiota: Role in therapy. Br. J. Pharmacol. 2020, 177, 1351–1362. [Google Scholar] [CrossRef] [Green Version]
- Ramezani, A.; Raj, D.S. The gut microbiome, kidney disease, and targeted interventions. J. Am. Soc. Nephrol. 2014, 25, 657–670. [Google Scholar] [CrossRef] [Green Version]
- Pivari, F.; Mingione, A.; Brasacchio, C.; Soldati, L. Curcumin and type 2 diabetes mellitus: Prevention and treatment. Nutrients 2019, 11, 1837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.S.; Krieg, R.; Massey, H.D.; Sica, D.A.; Fakhry, I.; Ghosh, S.; Gehr, T.W.B. Curcumin and enalapril ameliorate renal failure by antagonizing inflammation in 5/6 nephrectomized rats: Role of phospholipase and cyclooxygenase. Am. J. Physiol.-Ren. Physiol. 2012, 302, F439–F454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez-Osorio, A.S.; García-Niño, W.R.; González-Reyes, S.; Álvarez-Mejía, A.E.; Guerra-León, S.; Salazar-Segovia, J.; Falcón, I.; Montes de Oca-Solano, H.; Madero, M.; Pedraza-Chaverri, J. The effect of dietary supplementation with curcumin on redox status and Nrf2 activation in patients with nondiabetic or diabetic proteinuric chronic kidney disease: A pilot study. J. Ren. Nutr. 2016, 26, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Cuomo, J.; Appendino, G.; Dern, A.S.; Schneider, E.; McKinnon, T.P.; Brown, M.J.; Togni, S.; Dixon, B.M. Comparative absorption of a standardized curcuminoid mixture and its lecithin formulation. J. Nat. Prod. 2011, 74, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, L.; Favari, C.; Calani, L.; Francinelli, V.; Riva, A.; Petrangolini, G.; Allegrini, P.; Mena, P.; Del Rio, D. The effect of formulation of curcuminoids on their metabolism by human colonic microbiota. Molecules 2020, 25, 940. [Google Scholar] [CrossRef] [Green Version]
- Teng, C.F.; Yu, C.H.; Chang, H.Y.; Hsieh, W.C.; Wu, T.H.; Lin, J.H.; Wu, H.C.; Jeng, L.B.; Su, I.J. Chemopreventive Effect of Phytosomal Curcumin on Hepatitis B Virus-Related Hepatocellular Carcinoma in A Transgenic Mouse Model. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tenero, L.; Piazza, M.; Zanoni, L.; Bodini, A.; Peroni, D.; Piacentini, G.L. Antioxidant supplementation and exhaled nitric oxide in children with asthma. Allergy Asthma Proc. 2016, 37, e8–e13. [Google Scholar] [CrossRef]
- Hu, S.; Belcaro, G.; Dugall, M.; Peterzan, P.; Hosoi, M.; Ledda, A.; Riva, A.; Giacomelli, L.; Togni, S.; Eggenhoffner, R.; et al. Interaction study between antiplatelet agents, anticoagulants, thyroid replacement therapy and a bioavailable formulation of curcumin (Meriva®). Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5042–5046. [Google Scholar] [PubMed]
- Panahi, Y.; Kianpour, P.; Mohtashami, R.; Jafari, R.; Simental-Mendía, L.E.; Sahebkar, A. Efficacy and Safety of Phytosomal Curcumin in Non-Alcoholic Fatty Liver Disease: A Randomized Controlled Trial. Drug Res. 2017, 67, 244–251. [Google Scholar] [CrossRef] [Green Version]
- Thota, R.N.; Dias, C.B.; Abbott, K.A.; Acharya, S.H.; Garg, M.L. Curcumin alleviates postprandial glycaemic response in healthy subjects: A cross-over, randomized controlled study. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Pastorelli, D.; Fabricio, A.S.C.; Giovanis, P.; D’Ippolito, S.; Fiduccia, P.; Soldà, C.; Buda, A.; Sperti, C.; Bardini, R.; Da Dalt, G.; et al. Phytosome complex of curcumin as complementary therapy of advanced pancreatic cancer improves safety and efficacy of gemcitabine: Results of a prospective phase II trial. Pharmacol. Res. 2018, 132, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Ledda, A.; Belcaro, G.; Feragalli, B.; Hosoi, M.; Cacchio, M.; Luzzi, R.; Dugall, M.; Cotellese, R. Temporary kidney dysfunction: Supplementation with Meriva® in initial, transient kidney micro-macro albuminuria. Panminerva Med. 2019, 61, 444–448. [Google Scholar] [CrossRef]
- Thompson, F.E.; Subar, A.F. Dietary Assessment Methodology. In Nutrition in the Prevention and Treatment of Disease; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 5–46. ISBN 9780123918840. [Google Scholar]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. PANDAseq: Paired-end assembler for illumina sequences. BMC Bioinform. 2012, 13, 31. [Google Scholar] [CrossRef] [Green Version]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [Green Version]
- Pruesse, E.; Quast, C.; Knittel, K.; Fuchs, B.M.; Ludwig, W.; Peplies, J.; Glöckner, F.O. SILVA: A comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 2007, 35, 7188–7196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B. Vegan: Community Ecology Package. R Package Version 2.0 ± 10. 2013. Available online: http://CRAN.R-project.org/package=vegan (accessed on 7 December 2021).
- Lekawanvijit, S.; Kompa, A.R.; Wang, B.H.; Kelly, D.J.; Krum, H. Cardiorenal syndrome: The emerging role of protein-bound uremic toxins. Circ. Res. 2012, 111, 1470–1483. [Google Scholar] [CrossRef] [Green Version]
- Fouque, D.; Kalantar-Zadeh, K.; Kopple, J.; Cano, N.; Chauveau, P.; Cuppari, L.; Franch, H.; Guarnieri, G.; Ikizler, T.A.; Kaysen, G.; et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. In Proceedings of the Kidney International; Nature Publishing Group: Berlin, Germany, 2008; Volume 73, pp. 391–398. [Google Scholar]
- De Nicola, L.; Minutolo, R.; Chiodini, P.; Zoccali, C.; Castellino, P.; Donadio, C.; Strippoli, M.; Casino, F.; Giannattasio, M.; Petrarulo, F.; et al. Global approach to cardiovascular risk in chronic kidney disease: Reality and opportunities for intervention. Kidney Int. 2006, 69, 538–545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansen, K.L.; Lee, C. Body composition in chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2015, 24, 268–275. [Google Scholar] [CrossRef] [Green Version]
- Cupisti, A.; Brunori, G.; Di Iorio, B.R.; D’Alessandro, C.; Pasticci, F.; Cosola, C.; Bellizzi, V.; Bolasco, P.; Capitanini, A.; Fantuzzi, A.L.; et al. Nutritional treatment of advanced CKD: Twenty consensus statements. J. Nephrol. 2018, 31, 457–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interf. Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Hashimoto, S.; Horie, T. Curcumin inhibition of inflammatory cytokine production by human peripheral blood monocytes and alveolar macrophages. Pharmacol. Res. 1999, 39, 41–47. [Google Scholar] [CrossRef]
- Bereswill, S.; Muñoz, M.; Fischer, A.; Plickert, R.; Haag, L.M.; Otto, B.; Kühl, A.A.; Loddenkemper, C.; Göbel, U.B.; Heimesaat, M.M. Anti-inflammatory effects of Resveratrol, Curcumin and Simvastatin in acute small intestinal inflammation. PLoS ONE 2010, 5, e15099. [Google Scholar] [CrossRef] [Green Version]
- Jacob, A.; Chaves, L.; Eadon, M.T.; Chang, A.; Quigg, R.J.; Alexander, J.J. Curcumin alleviates immune-complex-mediated glomerulonephritis in factor-H-deficient mice. Immunology 2013, 139, 328–337. [Google Scholar] [CrossRef]
- Hewlings, S.; Kalman, D. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Gadani, S.P.; Cronk, J.C.; Norris, G.T.; Kipnis, J. IL-4 in the Brain: A Cytokine To Remember. J. Immunol. 2012, 189, 4213–4219. [Google Scholar] [CrossRef]
- Gour, N.; Wills-Karp, M. IL-4 and IL-13 signaling in allergic airway disease. Cytokine 2015, 75, 68–78. [Google Scholar] [CrossRef] [Green Version]
- Babaei, E.; Sadeghizadeh, M.; Hassan, Z.M.; Feizi, M.A.H.; Najafi, F.; Hashemi, S.M. Dendrosomal curcumin significantly suppresses cancer cell proliferation in vitro and in vivo. Int. Immunopharmacol. 2012, 12, 226–234. [Google Scholar] [CrossRef]
- Chung, S.H.; Choi, S.H.; Choi, J.A.; Chuck, R.S.; Joo, C.K. Curcumin suppresses ovalbumin-induced allergic conjunctivitis. Mol. Vis. 2012, 18, 1966–1972. [Google Scholar]
- Ganjali, S.; Sahebkar, A.; Mahdipour, E.; Jamialahmadi, K.; Torabi, S.; Akhlaghi, S.; Ferns, G.; Parizadeh, S.M.R.; Ghayour-Mobarhan, M. Investigation of the effects of curcumin on serum cytokines in obese individuals: A randomized controlled trial. Sci. World J. 2014. [Google Scholar] [CrossRef] [PubMed]
- Law, B.M.P.; Wilkinson, R.; Wang, X.; Kildey, K.; Lindner, M.; Rist, M.J.; Beagley, K.; Healy, H.; Kassianos, A.J. Interferon-γ production by tubulointerstitial human CD56bright natural killer cells contributes to renal fibrosis and chronic kidney disease progression. Kidney Int. 2017, 92, 79–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Sánchez, O.; López-Novoa, J.M.; López-Hernández, F.J. Interferon-γ Reduces the Proliferation of Primed Human Renal Tubular Cells. Nephron Extra 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ullah, F.; Liang, H.; Niedermayer, G.; Münch, G.; Gyengesi, E. Evaluation of Phytosomal Curcumin as an Anti-inflammatory Agent for Chronic Glial Activation in the GFAP-IL6 Mouse Model. Front. Neurosci. 2020, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Valizadeh, H.; Abdolmohammadi-Vahid, S.; Danshina, S.; Gencer, M.Z.; Ammari, A.; Sadeghi, A.; Roshangar, L.; Aslani, S.; Esmaeilzadeh, A.; Ghaebi, M.; et al. Nano-curcumin therapy, a promising method in modulating inflammatory cytokines in COVID-19 patients. Int. Immunopharmacol. 2020, 89, 107088. [Google Scholar] [CrossRef]
- Belcaro, G.; Cesarone, M.R.; Dugall, M.; Pellegrini, L.; Ledda, A.; Grossi, M.G.; Togni, S.; Appendino, G. Efficacy and safety of Meriva®, a curcumin-phosphatidylcholine complex, during extended administration in osteoarthritis patients. Altern Med. Rev. 2010, 15, 337–344. [Google Scholar] [PubMed]
- Mahfouz, M.M.; Zhou, S.Q.; Kummerow, F.A. Curcumin prevents the oxidation and lipid modification of LDL and its inhibition of prostacyclin generation by endothelial cells in culture. Prostaglandins Other Lipid Mediat. 2009, 90, 13–20. [Google Scholar] [CrossRef]
- Drion, C.M.; van Scheppingen, J.; Arena, A.; Geijtenbeek, K.W.; Kooijman, L.; van Vliet, E.A.; Aronica, E.; Gorter, J.A. Effects of rapamycin and curcumin on inflammation and oxidative stress in vitro and in vivo—In search of potential anti-epileptogenic strategies for temporal lobe epilepsy. J. Neuroinflamm. 2018, 15, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Sadeghi, A.; Rostamirad, A.; Seyyedebrahimi, S.; Meshkani, R. Curcumin ameliorates palmitate-induced inflammation in skeletal muscle cells by regulating JNK/NF-kB pathway and ROS production. Inflammopharmacology 2018, 26, 1265–1272. [Google Scholar] [CrossRef]
- Lin, X.; Bai, D.; Wei, Z.; Zhang, Y.; Huang, Y.; Deng, H.; Huang, X. Curcumin attenuates oxidative stress in RAW264.7 cells by increasing the activity of antioxidant enzymes and activating the Nrf2-Keap1 pathway. PLoS ONE 2019, 14, e0216711. [Google Scholar]
- Ali, B.H.; Al-Salam, S.; Al Suleimani, Y.; Al Kalbani, J.; Al Bahlani, S.; Ashique, M.; Manoj, P.; Al Dhahli, B.; Al Abri, N.; Naser, H.T.; et al. Curcumin Ameliorates Kidney Function and Oxidative Stress in Experimental Chronic Kidney Disease. Basic Clin. Pharmacol. Toxicol. 2018, 122, 65–73. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T.; Samini, F. Anti-oxidative effects of curcumin on immobilization-induced oxidative stress in rat brain, liver and kidney. Biomed. Pharmacother. 2017, 87, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Usharani, P.; Mateen, A.A.; Naidu, M.U.R.; Raju, Y.S.N.; Chandra, N. Effect of NCB-02, Atorvastatin and Placebo on Endothelial Function, Oxidative Stress and Inflammatory Markers in Patients with Type 2 Diabetes Mellitus: A Randomized, Parallel-Group, Placebo-Controlled, 8-Week Study. Drugs R D 2008, 9, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Durgaprasad, S.; Ganesh Pai, C.; Vasanthkumar; Alvres, J.F.; Namitha, S. A pilot study of the antioxidant effect of curcumin in tropical pancreatitis. Indian J. Med. Res. 2005, 122, 315–318. [Google Scholar]
- Barreto, F.C.; Barreto, D.V.; Liabeuf, S.; Meert, N.; Glorieux, G.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A. Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1551–1558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salarolli, R.T.; Alvarenga, L.; Cardozo, L.F.M.F.; Teixeira, K.T.R.; de SG Moreira, L.; Lima, J.D.; Rodrigues, S.D.; Nakao, L.S.; Fouque, D.; Mafra, D. Can curcumin supplementation reduce plasma levels of gut-derived uremic toxins in hemodialysis patients? A pilot randomized, double-blind, controlled study. Int. Urol. Nephrol. 2021, 53, 1231–1238. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Zhao, Y.Y.; Pahl, M.V. Altered intestinal microbial flora and impaired epithelial barrier structure and function in CKD: The nature, mechanisms, consequences and potential treatment. Nephrol. Dial. Transplant. 2016, 31, 737–746. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Ning, X.; Liu, B.; Dong, R.; Bai, M.; Sun, S. Specific alterations in gut microbiota in patients with chronic kidney disease: An updated systematic review. Ren Fail. 2021, 43, 102–112. [Google Scholar] [CrossRef]
- Margiotta, E.; Miragoli, F.; Callegari, M.L.; Vettoretti, S.; Caldiroli, L.; Meneghini, M.; Zanoni, F.; Messa, P. Gut microbiota composition and frailty in elderly patients with Chronic Kidney Disease. PLoS ONE 2020, 15, e0228530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canani, R.B.; Di Costanzo, M.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Nishida, A.; Sugitani, Y.; Nishino, K.; Inatomi, O.; Sugimoto, M.; Kawahara, M.; Andoh, A. Nanoparticle curcumin ameliorates experimental colitis via modulation of gut microbiota and induction of regulatory T cells. PLoS ONE 2017, 12, e0185999. [Google Scholar]
- Feng, W.; Wang, H.; Zhang, P.; Gao, C.; Tao, J.; Ge, Z.; Zhu, D.; Bi, Y. Modulation of gut microbiota contributes to curcumin-mediated attenuation of hepatic steatosis in rats. Biochim. Biophys. Acta-Gen. Subj. 2017, 1861, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
| CTRL (n = 20) | CKD T0 (n = 24) | CKD T1 (n = 21) | CKD T2 (n = 11) | p-Value | |
|---|---|---|---|---|---|
| Gender | M: 60% (n = 12) F: 40% (n = 8) | M: 58% (n = 14) F: 42% (n = 10) | M: 57% (n = 12) F: 43% (n = 9) | M: 45.5% (n = 5) F: 54.5% (n = 6) | n.s. |
| Age (Y) | 74 (68.5–78.7) | 72 (67.5–78.8) | 72 (66–77) | 72 (67–81) | n.s. |
| Height (m) | 1.68 (1.58–1.75) | 1.65 (1.62–1.71) | 1.65 (1.62–1.70) | 1.64 (1.60–1.71) | n.s. |
| Weight (kg) | 75 (66–80) | 77.5 (63.3–83.0) | 79.0 (64.7–81.0) | 80.0 (64.0–85.0) | n.s. |
| BMI (kg/m2) | 25.20 (22.50–30.70) | 27.40 (22.62–31.50) | 27.53 (23.42–30.35) | 28.04 (22.68–33.06) | n.s. |
| Waist circumference (cm) | 99.0 (90.5–111.0) | 102.0 (91.0–110.5) | 103.0 (97.0–108.0) | 99.5 (93.0–110.0) | n.s. |
| Bioimpedance | |||||
| TBW (l) | 37.7 (29.9–44.1) | 36.8 (33.5–42.2) | 38.1 (34.8–40.6) | 37.2 (35.3–45.0) | *** CKD T0 vs. CKD T1 |
| ECW (I) | 17.7 (14.2–19.9) | 17.7 (15.5–19.5) | 18.0 (16.1–20.0) | 17.0 (16.1–19.2) | n.s. |
| ICW (I) | 19.8 (15.8–23.0) | 19.5 (17.5–21.6) | 20.0 (18.0–22.1) | 20.0 (19.1–22.9) | n.s. |
| FFM (%) | 54.8 (46.0–60.3) | 52.7 (46.6–59.5) | 54.6 (49.2–66.2) | 53.6 (46.2–67.9) | n.s. |
| FM (%) | 31.4 (25.1–38.4) | 31.8 (28.2–37.3) | 30.9 (24.5–34.4) | 33.0 (21.7–37.6) | *** CKD T0 vs. CKD T1 |
| Clinical parameters | |||||
| Creatininemia (mg/dL) | 0.80 (0.70–1.00) | 1.96 (1.46–2.73) | 1.90 (1.57–2.80) | 1.45 (1.48–2.50) | *** CTRL vs. CKD T0 *** CTRL vs. CKD T1 *** CTRL vs. CKD T2 |
| GFR (ml/min/1.73 m2) | 83.0 (70.0–91.0) | 33.5 (22.1–39.8) | 27.2 (21.0–45.5) | 35.7 (24.0–45.0) | *** CTRL vs. CKD T0 *** CTRL vs. CKD T1 *** CTRL vs. CKD T2 |
| Azotemia (mg/dL) | n.d. | 60.0 (30.0–90.0) | 46.5 (32.5–63.5) | 35.0 (30.0–40.0) | n.s. |
| Sodium (mEq/L) | 140.0 (138.5–141.5) | 142.3 (139.3–143.8) | 141.9 (140.0–143.5) | 143.0 (140.7–143.8) | n.s. |
| Potassium (mEq/L) | 4.40 (4.25–5.10) | 4.60 (4.46–5.08) | 4.60 (4.25–4.80) | 4.55 (4.37–4.77) | n.s. |
| Phosphorus (mg/dL) | n.d. | 3.65 (3.20–4.00) | 3.50 (3.00–4.00) | 3.30 (3.10–4.10) | n.s. |
| Calcium (mg/dL) | n.d. | 9.60 (9.31–9.99) | 9.60 (9.45–9.99) | 9.48 (9.26–9.59) | n.s. |
| CTRL (n = 20) | CKD T0 (n = 24) | CKD T1 (n = 21) | CKD T2 (n = 11) | p-Value | |
|---|---|---|---|---|---|
| Caloric intake (kcal/day) | 1788 (1438–2226) | 1839 (1399–2520) | 1540 (1195–1871) | 1270 (1131–2612) | * CKD T0 vs. CKD T1 |
| Total carbohydrates (g/day) | 154.4 (105.8–230.2) | 215.9 (169.4–267.7) | 160.6 (136.7–235.6) | 128.0 (109.9–200.1) | * CTRL vs. CKD T0 ** CKD T0 vs. CKD T1 *** CKD T0 vs. CKD T2 |
| Starch (g/day) | 93.34 (62.51–156.00) | 118.80 (67.72–163.20) | 89.56 (58.20–120.0) | 64.77 (27.36–85.33) | * CKD T0 vs. CKD T1 ** CKD T0 vs. CKD T2 |
| Soluble glucids (g/day) | 52.06 (36.70–72.27) | 64.62 (46.90–92.12) | 56.32 (36.50–79.68) | 50.47 (36.03–60.06) | * CKD T0 vs. CKD T2 |
| Fibers (g/day) | 12.85 (9.80–18.58) | 16.97 (14.49–20.35) | 13.95 (11.41–16.07) | 13.91 (10.89–19.64) | * CKD T0 vs. CKD T1 |
| Total lipids (g/day) | 52.72 (43.05–64.90) | 57.15 (41.01–76.42) | 49.29 (42.35–69.62) | 49.82 (36.49–71.93) | n.s. |
| Animal lipids (g/day) | 23.33 (16.59–33.67) | 28.95 (18.07–38.84) | 28.84 (19.47–35.98) | 21.57 (14.24–30.28) | n.s. |
| Plant lipids (g/day) | 27.58 (24.24–38.82) | 28.49 (20.14–33.55) | 22.04 (17.95–26.55) | 21.73 (15.73–32.62) | n.s. |
| Total proteins (g/day) | 59.18 (43.96–75.47) | 60.08 (49.39–70.34) | 49.17 (42.92–59.74) | 40.66 (34.31–73.92) | * CKD T0 vs. CKD T1 |
| Animal proteins (g/day) | 38.79 (31.37–50.88) | 35.60 (24.17–47.05) | 33.21 (24.43–40.70) | 31.60 (22.14–40.58) | n.s. |
| Plant proteins (g/day) | 19.01 (12.54–27.40) | 23.16 (14.80–28.23) | 16.37 (13.28–21.52) | 13.67 (10.09–17.09) | *** CKD T0 vs. CKD T1 *** CKD T0 vs. CKD T2 |
| Phosphorus (mg/day) | 997.4 (718.1–1216.0) | 862.4 (756.2–1047.0) | 813.8 (665.2–894.3) | 684.2 (609.5–874.2) | * CTRL vs. CKD T1 ** CKD T0 vs. CKD T1 |
| Potassium (g/day) | 2.38 (1.71–3.66) | 2.27 (1.78–2.75) | 1.89 (1.60–2.39) | 1.98 (1.58–2.28) | * CTRL vs. CKD T1 ** CKD T0 vs. CKD T1 |
| Phylum | Family | Genus | CTRL | CKD T0 | CKD T1 | CKD T2 | p-Value |
|---|---|---|---|---|---|---|---|
| Firmicutes | 50.43 (20.54) | 56.67 (15.24) | 47.46 (12.01) | 57.46 (18.61) | 0.0428 (T0-T1) | ||
| Lachnospiraceae | 21.84 (10.43) | 21.30 (10.00) | 20.32 (8.38) | 22.96 (15.40) | 0.014 (CTRL-T2); 0.033 (T1-T2) | ||
| Other Lachnospiraceae | 2.62 (2.54) | 2.51 (2.82) | 3.24 (2.26) | 1.42 (1.40) | |||
| Lachnospiraceae NK4A136 group | 3.65 (4.96) | 2.19 (2.64) | 3.46 (3.65) | 2.75 (2.35) | |||
| Roseburia | 1.43 (1.55) | 2.04 (2.90) | 1.57 (2.43) | 4.41 (3.03) | |||
| [Eubacterium] eligens group | 2.13 (2.31) | 2.00 (1.40) | 1.41 (1.67) | 1.72 (2.34) | |||
| Blautia | 0.93 (0.80) | 1.83 (0.57) | 1.11 (0.82) | 0.35 (3.71) | |||
| Ruminococcaceae | 17.20 (12.30) | 20.75 (10.80) | 16.53 (8.06) | 20.00 (10.11) | |||
| Subdoligranulum | 4.14 (5.40) | 4.23 (2.45) | 5.38 (7.41) | 3.71 (7.10) | |||
| Ruminococcaceae UCG-002 | 2.84 (5.02) | 2.48 (3.30) | 2.76 (2.65) | 3.12 (3.13) | |||
| Faecalibacterium | 1.58 (2.26) | 2.46 (2.12) | 2.63 (2.87) | 2.01 (4.77) | |||
| Ruminococcus 2 | 0.48 (0.57) | 2.39 (1.66) | 2.26 (3.35) | 2.33 (4.10) | |||
| [Eubacterium] coprostanoligenes group | 1.57 (1.92) | 2.07 (1.12) | 1.19 (0.76) | 1.04 (2.08) | 0.0453 (T0-T1) | ||
| Christensenellaceae | 4.03 (5.35) | 4.77 (6.02) | 2.14 (2.24) | 5.95 (12.73) | |||
| Christensenellaceae R-7 group | 3.93 (5.26) | 4.66 (2.04) | 5.66 (12.08) | 2.20 (5.97) | |||
| Veillonellaceae | 3.61 (7.28) | 3.02 (4.39) | 1.93 (2.66) | 3.02 (2.59) | |||
| Dialister | 2.31 (6.57) | 2.10 (1.15) | 2.24 (2.64) | 2.35 (4.23) | |||
| Streptococcaceae | 0.65 (0.85) | 2.37 (3.85) | 2.77 (4.97) | 1.78 (2.64) | |||
| Streptococcus | 0.64 (0.84) | 2.36 (2.76) | 1.76 (2.64) | 4.97 (3.84) | |||
| Erysipelotrichaceae | 1.00 (1.83) | 1.44 (1.41) | 1.36 (1.55) | 0.97 (0.84) | |||
| Acidaminococcaceae | 0.88 (1.44) | 1.11 (1.69) | 1.02 (1.46) | 0.48 (0.71) | |||
| Bacteroidetes | 28.01 (15.40) | 28.90 (14.45) | 36.80 (12.08) | 27.68 (16.73) | |||
| Muribaculaceae | 0.47 (1.06) | 0.97 (2.55) | 2.09 (6.01) | 0.57 (1.64) | |||
| Barnesiellaceae | 0.62 (0.80) | 0.93 (1.28) | 1.03 (0.95) | 1.07 (1.65) | |||
| Bacteroidaceae | 19.58 (15.17) | 14.59 (11.04) | 17.55 (11.52) | 10.01 (7.27) | 0.037 (CTRL-T2) | ||
| Bacteroides | 19.57 (15.17) | 14.586 (17.55) | 10.01 (7.27) | 11.52 (11.04) | |||
| Prevotellaceae | 3.44 (7.46) | 6.42 (10.04) | 10.68 (13.90) | 11.19 (18.77) | |||
| Prevotella 9 | 2.48 (7.54) | 4.76 (7.20) | 9.32 (17.86) | 12.22 (9.25) | |||
| Rikenellaceae | 1.83 (1.40) | 3.75 (3.92) | 2.97 (3.58) | 3.09 (4.04) | |||
| Alistipes | 1.67 (1.34) | 3.44 (2.24) | 2.07 (1.99) | 1.69 (3.85) | |||
| Tannerellaceae | 1.79 (1.83) | 1.46 (0.93) | 1.78 (1.08) | 1.25 (1.11) | |||
| Parabacteroides | 1.79 (1.83) | 1.46 (1.78) | 1.25 (1.10) | 1.081 (0.93) | |||
| Proteobacteria | 19.56 (23.55) | 10.48 (11.26) | 13.27 (14.16) | 11.66 (18.12) | |||
| Enterobacteriaceae | 17.68 (24.35) | 8.58 (11.54) | 10.21 (14.29) | 9.34 (18.94) | |||
| Escherichia-Shigella | 8.44 (15.15) | 2.54 (3.41) | 5.20 (10.86) | 5.64 (5.12) | |||
| Salmonella | 2.25 (3.62) | 2.44 (2.85) | 2.09 (4.28) | 3.88 (2.83) | |||
| Klebsiella | 2.52 (5.55) | 1.32 (1.70) | 0.53 (1.30) | 3.88 (2.17) | |||
| Desulfovibrionaceae | 0.72 (0.93) | 0.89 (0.66) | 1.45 (1.24) | 0.99 (1.10) | 0.020 (CTRL-T1) | ||
| Actinobacteria | 1.00 (1.02) | 2.36 (2.90) | 1.35 (1.24) | 1.95 (2.85) | 0.0361 (CTRL-T0) | ||
| Bifidobacteriaceae | 0.51 (0.67) | 1.30 (1.88) | 0.75 (1.08) | 1.27 (2.73) | |||
| Coriobacteriaceae | 0.15 (0.25) | 0.52 (1.11) | 0.27 (0.31) | 0.25 (0.42) | |||
| Verrucomicrobia | 0.12 (0.12) | 0.03 (0.09) | 0.09 (0.04) | 0.11 (0.01) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pivari, F.; Mingione, A.; Piazzini, G.; Ceccarani, C.; Ottaviano, E.; Brasacchio, C.; Dei Cas, M.; Vischi, M.; Cozzolino, M.G.; Fogagnolo, P.; et al. Curcumin Supplementation (Meriva®) Modulates Inflammation, Lipid Peroxidation and Gut Microbiota Composition in Chronic Kidney Disease. Nutrients 2022, 14, 231. https://doi.org/10.3390/nu14010231
Pivari F, Mingione A, Piazzini G, Ceccarani C, Ottaviano E, Brasacchio C, Dei Cas M, Vischi M, Cozzolino MG, Fogagnolo P, et al. Curcumin Supplementation (Meriva®) Modulates Inflammation, Lipid Peroxidation and Gut Microbiota Composition in Chronic Kidney Disease. Nutrients. 2022; 14(1):231. https://doi.org/10.3390/nu14010231
Chicago/Turabian StylePivari, Francesca, Alessandra Mingione, Giada Piazzini, Camilla Ceccarani, Emerenziana Ottaviano, Caterina Brasacchio, Michele Dei Cas, Margherita Vischi, Mario Gennaro Cozzolino, Paolo Fogagnolo, and et al. 2022. "Curcumin Supplementation (Meriva®) Modulates Inflammation, Lipid Peroxidation and Gut Microbiota Composition in Chronic Kidney Disease" Nutrients 14, no. 1: 231. https://doi.org/10.3390/nu14010231
APA StylePivari, F., Mingione, A., Piazzini, G., Ceccarani, C., Ottaviano, E., Brasacchio, C., Dei Cas, M., Vischi, M., Cozzolino, M. G., Fogagnolo, P., Riva, A., Petrangolini, G., Barrea, L., Di Renzo, L., Borghi, E., Signorelli, P., Paroni, R., & Soldati, L. (2022). Curcumin Supplementation (Meriva®) Modulates Inflammation, Lipid Peroxidation and Gut Microbiota Composition in Chronic Kidney Disease. Nutrients, 14(1), 231. https://doi.org/10.3390/nu14010231











