Anxiolytic Effect and Improved Sleep Quality in Individuals Taking Lippia citriodora Extract
Abstract
:1. Introduction
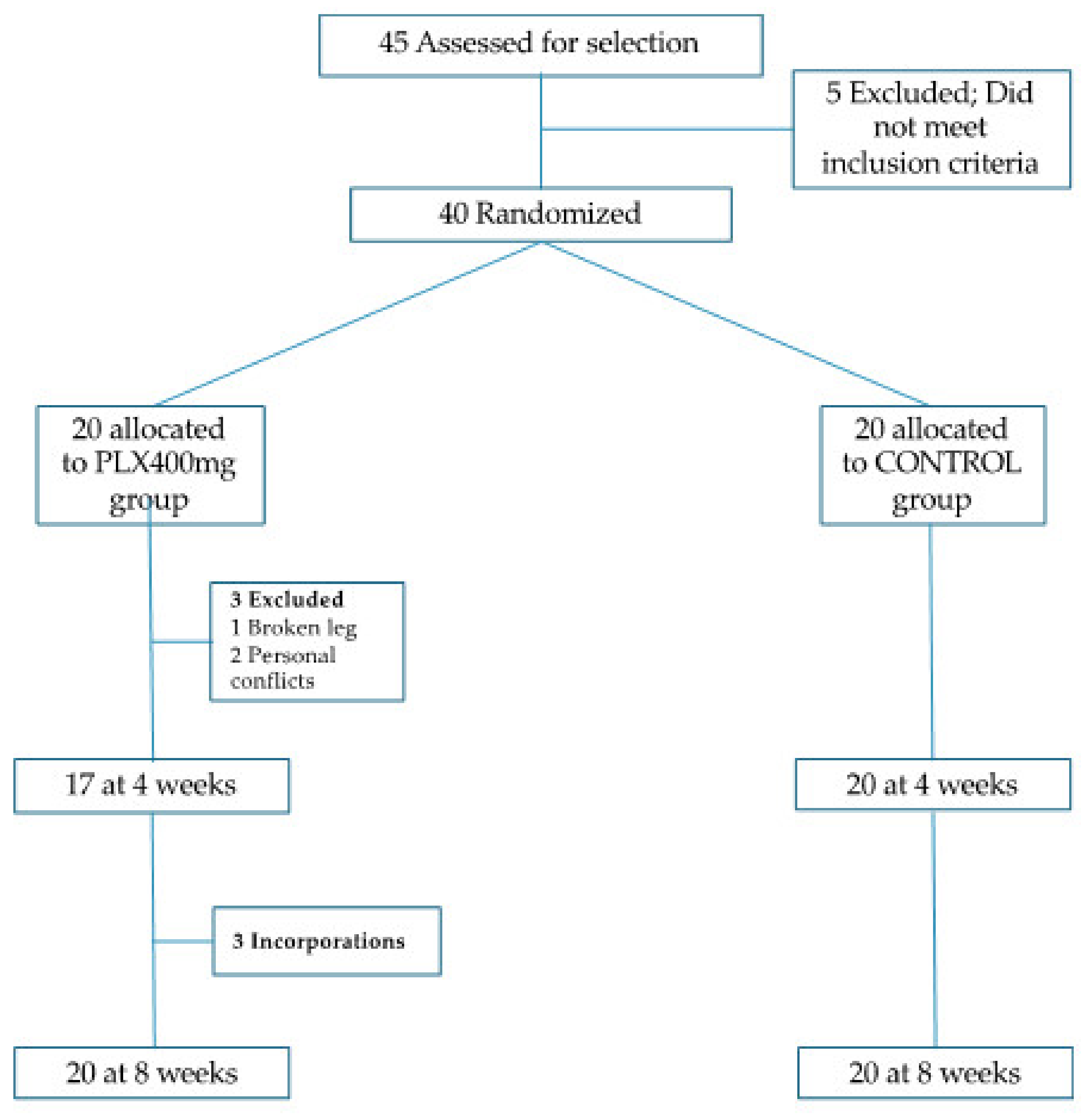
2. Materials and Methods
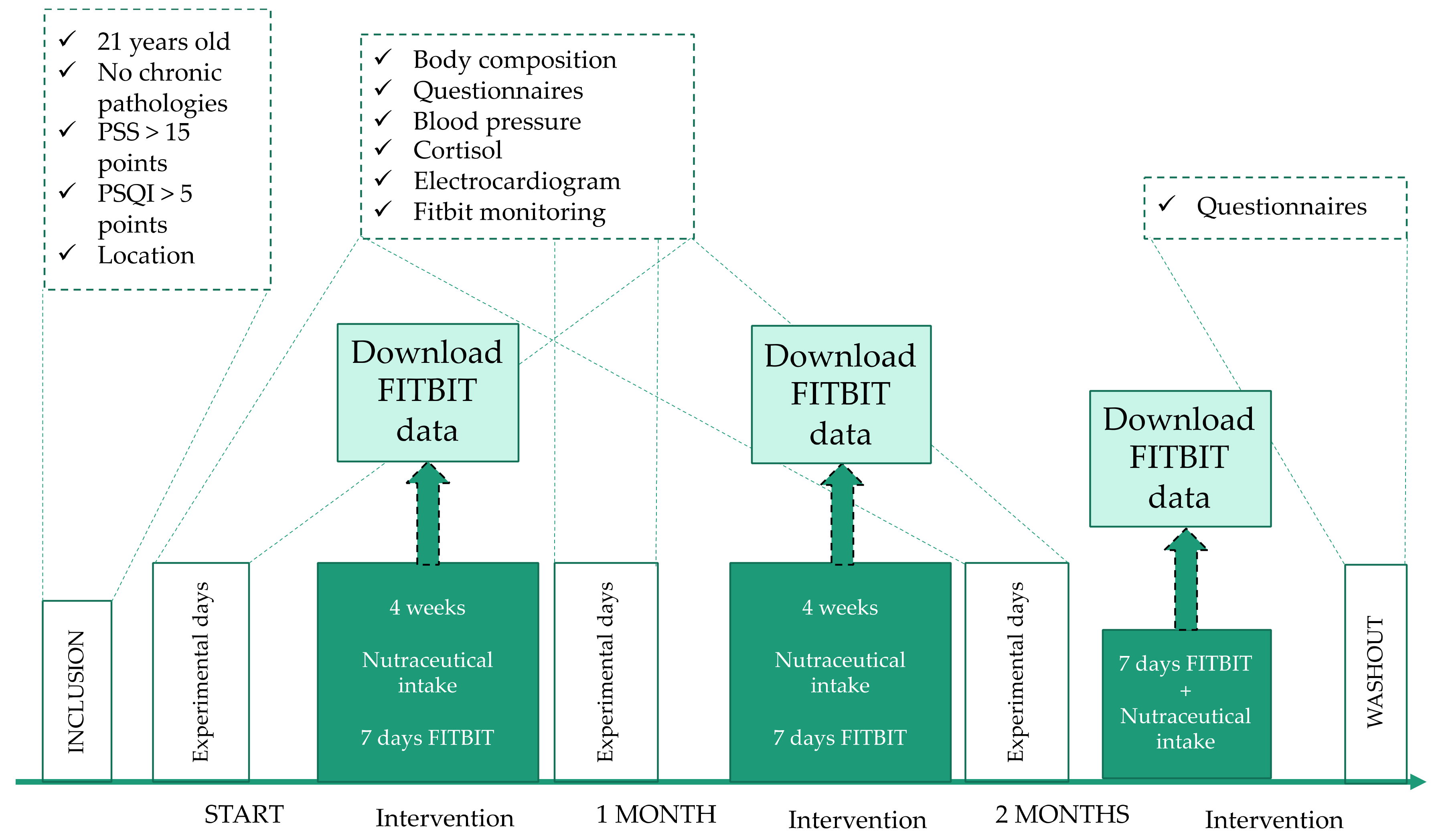
2.1. Study Design
2.2. Lemon Verbena Extract Characterization
2.3. Subjects
2.4. Trial Design
2.5. Declarations: Ethics Approval, Informed Consent
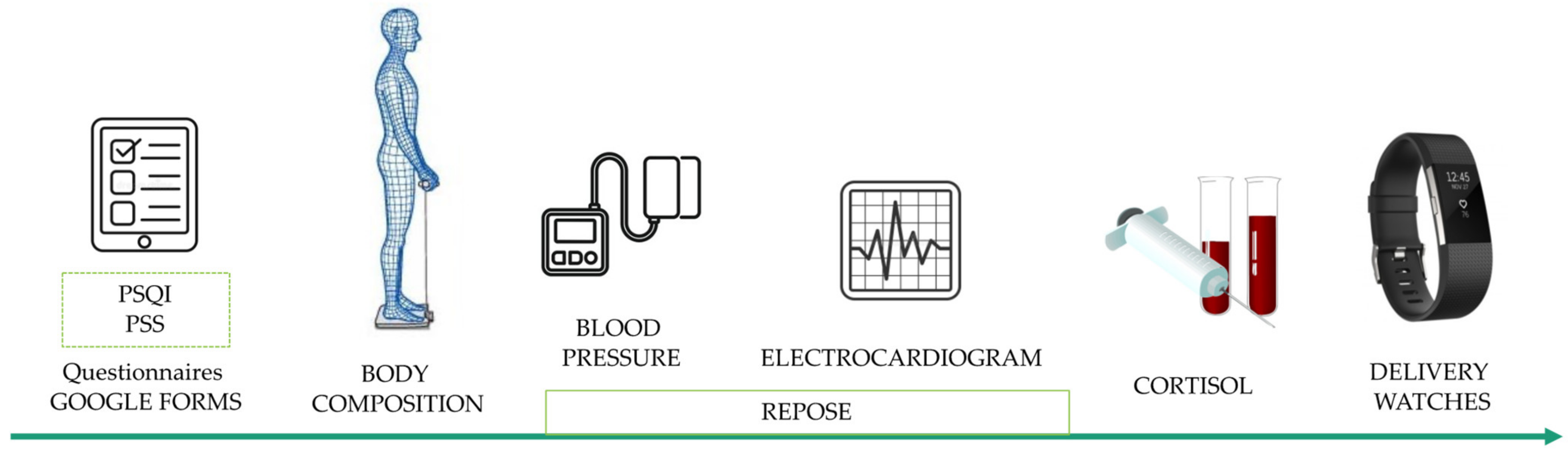
2.6. Data Collection
2.6.1. Body Composition
2.6.2. Sleep Quality Index
2.6.3. Stress
2.6.4. Cortisol Assessment
2.6.5. Fitbit Sleep Tracking
2.7. Statistical Analysis
3. Results
3.1. Population Demographics
3.2. Stress Analysis: Perceived Stress Scale (PSS) and Cortisol Levels
3.3. Sleep Analysis: Perceived Sleep Quality Index (PSQI) and Sleep Activity Tracking
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendlowicz, M.V.; Stein, M.B. Quality of life in individuals with anxiety disorders. Am. J. Psychiatry 2000, 157, 669–682. [Google Scholar] [CrossRef]
- Regier, D.A.; Rae, D.S.; Narrow, W.E.; Kaelber, C.T.; Schatzberg, A.F. Prevalence of anxiety disorders and their comorbidity with mood and addictive disorders. Br. J. Psychiatry 1998, 34, 24–28. [Google Scholar] [CrossRef]
- Trevor, A.J. Sedative-Hypnotic Drugs. In Basic & Clinical Pharmacology, 15e; Katzung, B.G., Vanderah, T.W., Eds.; McGraw-Hill: New York, NY, USA, 2021. [Google Scholar]
- Kamboj, V.P. Herbal medicine. Curr. Sci. 2000, 78, 35. Available online: http://www.sciepub.com/reference/189774 (accessed on 18 November 2021).
- Singh, G.K.; Chauhan, S.K.; Rai, G.; Chatterjee, S.S.; Kumar, V. Potential antianxiety activity of Fumaria indica: A preclinical study. Pharmacogn. Mag. 2013, 9, 14–22. [Google Scholar]
- Jaiswal, A.K.; Bhattacharya, S.K.; Acharya, S.B. Anxiolytic activity of Azadirachta indica leaf extract in rats. Indian J. Exp. Biol. 1994, 32, 489–491. [Google Scholar]
- Saeed, S.A.; Bloch, R.M.; Antonacci, D.J. Herbal and dietary supplements for treatment of anxiety disorders. Am. Fam. Physician 2007, 76, 549–556. [Google Scholar]
- Rabbani, M.; Sajjadi, S.E.; Jalali, A. Hydroalcohol extract and fractions of Stachys lavandulifolia Vahl: Effects on spontaneous motor activity and elevated plus-maze behaviour. Phytother. Res. 2005, 19, 854–858. [Google Scholar] [CrossRef]
- Murphy, K.; Kubin, Z.J.; Shepherd, J.N.; Ettinger, R.H. Valeriana officinalis root extracts have potent anxiolytic effects in laboratory rats. Phytomedicine 2010, 17, 674–678. [Google Scholar] [CrossRef]
- Taiwo, A.E.; Leite, F.B.; Lucena, G.M.; Barros, M.; Silveira, D.; Silva, M.V.; Ferreira, V.M. Anxiolytic and antidepressant-like effects of Melissa officinalis (lemon balm) extract in rats: Influence of administration and gender. Indian J. Pharmacol. 2012, 44, 189–192. [Google Scholar]
- Rabbani, M.; Sajjadi, S.E.; Vaezi, A. Evaluation of anxiolytic and sedative effect of essential oil and hydroalcoholic extract of Ocimum basilicum L. and chemical composition of its essential oil. Res. Pharm. Sci. 2015, 10, 535–543. [Google Scholar]
- Pascual, M.E.; Slowing, K.; Carretero, E.; Sánchez Mata, D.; Villar, A. Lippia: Traditional uses, chemistry and pharmacology: A review. J. Ethnopharmacol. 2001, 76, 201–214. [Google Scholar] [CrossRef]
- Ghasempour, M.; Omran, S.M.; Moghadamnia, A.A.; Shafiee, F. Effect of aqueous and ethanolic extracts of Lippia citriodora on candida albicans. Electron. Physician 2016, 8, 2752–2758. [Google Scholar] [CrossRef] [Green Version]
- Montanari, B. Aromatic, Medicinal Plants and Vulnerability of Traditional Herbal Knowledge in a Berber Community of the High Atlas Mountains of Morocco. Plant Divers. Resour. 2014, 36, 388–402. [Google Scholar]
- Carnat, A.P.; Carnat, A.; Fraisse, D.; Lamaison, J.L. The aromatic and polyphenolic composition of lemon balm (Melissa officinalis L. subsp. officinalis) tea. Pharm. Acta Helv. 1998, 72, 301–305. [Google Scholar] [CrossRef]
- Arthur, H.; Joubert, E.; De Beer, D.; Malherbe, C.J.; Witthuhn, R.C. Phenylethanoid glycosides as major antioxidants in Lippia multiflora herbal infusion and their stability during steam pasteurisation of plant material. Food Chem. 2011, 127, 581–588. [Google Scholar] [CrossRef]
- Speranza, L.; Franceschelli, S.; Pesce, M.; Reale, M.; Menghini, L.; Vinciguerra, I.; De Lutiis, M.A.; Felaco, M.; Grilli, A. Antiinflammatory effects in THP-1 cells treated with verbascoside. Phytother. Res. 2010, 24, 1398–1404. [Google Scholar] [CrossRef]
- Avila, J.G.; de Liverant, J.G.; Martínez, A.; Martínez, G.; Muñoz, J.L.; Arciniegas, A.; Romo de Vivar, A. Mode of action of Buddleja cordata verbascoside against Staphylococcus aureus. J. Ethnopharmacol. 1999, 66, 75–78. [Google Scholar] [CrossRef]
- Razavi, B.M.; Zargarani, N.; Hosseinzadeh, H. Anti-anxiety and hypnotic effects of ethanolic and aqueous extracts of Lippia citriodora leaves and verbascoside in mice. Avicenna J. Phytomed. 2017, 7, 353–365. [Google Scholar]
- Saghaei, M. An overview of randomization and minimization programs for randomized clinical trials. J. Med. Signals Sens. 2011, 1, 55–61. [Google Scholar] [CrossRef] [Green Version]
- Quirantes-Piné, R.; Funes, L.; Micol, V.; Segura-Carretero, A.; Fernández-Gutiérrez, A. High-performance liquid chromatography with diode array detection coupled to electrospray time-of-flight and ion-trap tandem mass spectrometry to identify phenolic compounds from a lemon verbena extract. J. Chromatogr. A 2009, 1216, 5391–5397. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Remor, E. Scientific Information System Psychometric Properties of a European Spanish Version of the Perceived Stress Scale (PSS). Spanish J. Psychol. 2006, 9, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Sousa-Lima, J.; Moreira, P.S.; Raposo-Lima, C.; Sousa, N.; Morgado, P. Relationship between obsessive compulsive disorder and cortisol: Systematic review and meta-analysis. Eur. Neuropsychopharmacol. 2019, 29, 1185–1198. [Google Scholar] [CrossRef]
- Funes, L.; Fernández-Arroyo, S.; Laporta, O.; Pons, A.; Roche, E.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Micol, V. Correlation between plasma antioxidant capacity and verbascoside levels in rats after oral administration of lemon verbena extract. Food Chem. 2009, 117, 589–598. [Google Scholar] [CrossRef]
- Funes, L.; Carrera-Quintanar, L.; Cerdán-Calero, M.; Ferrer, M.D.; Drobnic, F.; Pons, A.; Roche, E.; Micol, V. Effect of lemon verbena supplementation on muscular damage markers, proinflammatory cytokines release and neutrophils’ oxidative stress in chronic exercise. Eur. J. Appl. Physiol. 2011, 111, 695–705. [Google Scholar] [CrossRef]
- Martínez-Rodríguez, A.; Moya, M.; Vicente-Salar, N.; Brouzet, T.; Carrera-Quintanar, L.; Cervelló, E.; Micol, V.; Roche, E. Changes in biochemical and psychological parameters in university students performing aerobic exercise and consuming lemon verbena extracts. Curr. Top. Nutraceutical Res. 2015, 13, 95–102. [Google Scholar]
- Carrera-Quintanar, L.; Funes, L.; Vicente-Salar, N.; Blasco-Lafarga, C.; Pons, A.; Micol, V.; Roche, E. Effect of polyphenol supplements on redox status of blood cells: A randomized controlled exercise training trial. Eur. J. Nutr. 2015, 54, 1081–1093. [Google Scholar] [CrossRef]
- Caturla, N.; Funes, L.; Pérez-Fons, L.; Micol, V. A Randomized, Double-Blinded, Placebo-Controlled Study of the Effect of a Combination of Lemon Verbena Extract and Fish Oil Omega-3 Fatty Acid on Joint Management. J. Altern. Complement. Med. 2011, 17, 1051. [Google Scholar] [CrossRef]
- Carrera-Quintanar, L.; Funes, L.; Viudes, E.; Tur, J.; Micol, V.; Roche, E.; Pons, A. Antioxidant effect of lemon verbena extracts in lymphocytes of university students performing aerobic training program. Scand. J. Med. Sci. Sports 2012, 22, 454–461. [Google Scholar] [CrossRef]
- Afrasiabian, F.; Mirabzadeh Ardakani, M.; Rahmani, K.; Azadi, N.A.; Alemohammad, Z.B.; Bidaki, R.; Karimi, M.; Emtiazy, M.; Hashempur, M.H. Aloysia citriodora Palau (lemon verbena) for insomnia patients: A randomized, double-blind, placebo-controlled clinical trial of efficacy and safety. Phytother. Res. 2019, 33, 350–359. [Google Scholar] [CrossRef]
- Peach, H.D.; Gaultney, J.F.; Ruggiero, A.R. Direct and Indirect Associations of Sleep Knowledge and Attitudes With Objective and Subjective Sleep Duration and Quality via Sleep Hygiene. J. Prim. Prev. 2018, 39, 555–570. [Google Scholar] [CrossRef]
- Weatherall, J.; Paprocki, Y.; Meyer, T.M.; Kudel, I.; Witt, E.A. Sleep Tracking and Exercise in Patients with Type 2 Diabetes Mellitus (Step-D): Pilot Study to Determine Correlations between Fitbit Data and Patient-Reported Outcomes. JMIR mHealth uHealth 2018, 6, e8122. [Google Scholar] [CrossRef]
- Liang, Z.; Ploderer, B.; Liu, W.; Nagata, Y.; Bailey, J.; Kulik, L.; Li, Y. SleepExplorer: A visualization tool to make sense of correlations between personal sleep data and contextual factors. Pers. Ubiquitous Comput. 2016, 20, 985–1000. [Google Scholar] [CrossRef]
- Yurkiewicz, I.R.; Simon, P.; Liedtke, M.; Dahl, G.; Dunn, T. Effect of Fitbit and iPad Wearable Technology in Health-Related Quality of Life in Adolescent and Young Adult Cancer Patients. J. Adolesc. Young Adult Oncol. 2018, 7, 579–583. [Google Scholar] [CrossRef]
- De Zambotti, M.; Claudatos, S.; Inkelis, S.; Colrain, I.M.; Baker, F.C. Evaluation of a consumer fitness-tracking device to assess sleep in adults. Chronobiol. Int. 2015, 32, 1024–1028. [Google Scholar] [CrossRef] [Green Version]
- Cook, J.D.; Prairie, M.L.; Plante, D.T. Utility of the Fitbit Flex to evaluate sleep in major depressive disorder: A comparison against polysomnography and wrist-worn actigraphy. J. Affect. Disord. 2017, 217, 299–305. [Google Scholar] [CrossRef]
- Shi, Y.; Dong, J.-W.; Zhao, J.-H.; Tang, L.-N.; Zhang, J.-J. Herbal Insomnia Medications that Target GABAergic Systems: A Review of the Psychopharmacological Evidence. Curr. Neuropharmacol. 2014, 12, 289–302. [Google Scholar] [CrossRef]
- Ragone, M.; Sella, M.; Pastore, A.; Consolini, A. Sedative and Cardiovascular Effects of Aloysia citriodora Palau, on Mice and Rats. Lat. Am. J. Pharm. 2010, 29, 79–86. [Google Scholar]
- Sabti, M.; Sasaki, K.; Gadhi, C.; Isoda, H. Elucidation of the molecular mechanism underlying lippia citriodora(Lim.)-Induced relaxation and Anti-Depression. Int. J. Mol. Sci. 2019, 20, 3556. [Google Scholar] [CrossRef] [Green Version]
- Carmona, F.; Coneglian, F.S.; Batista, P.A.; Aragon, D.C.; Angelucci, M.A.; Martinez, E.Z.; Pereira, A.M.S. Aloysia polystachya (Griseb.) Moldenke (Verbenaceae) powdered leaves are effective in treating anxiety symptoms: A phase-2, randomized, placebo-controlled clinical trial. J. Ethnopharmacol. 2019, 242, 112060. [Google Scholar] [CrossRef]
- Costa de Melo, N.; Sánchez-Ortiz, B.L.; dos Santos Sampaio, T.I.; Matias Pereira, A.C.; Pinheiro da Silva Neto, F.L.; Ribeiro da Silva, H.; Alves Soares Cruz, R.; Keita, H.; Soares Pereira, A.M.; Tavares Carvalho, J.C. Anxiolytic and Antidepressant Effects of the Hydroethanolic Extract from the Leaves of Aloysia polystachya (Griseb.) Moldenke: A Study on Zebrafish (Danio rerio). Pharmaceuticals 2019, 12, 106. [Google Scholar] [CrossRef] [Green Version]
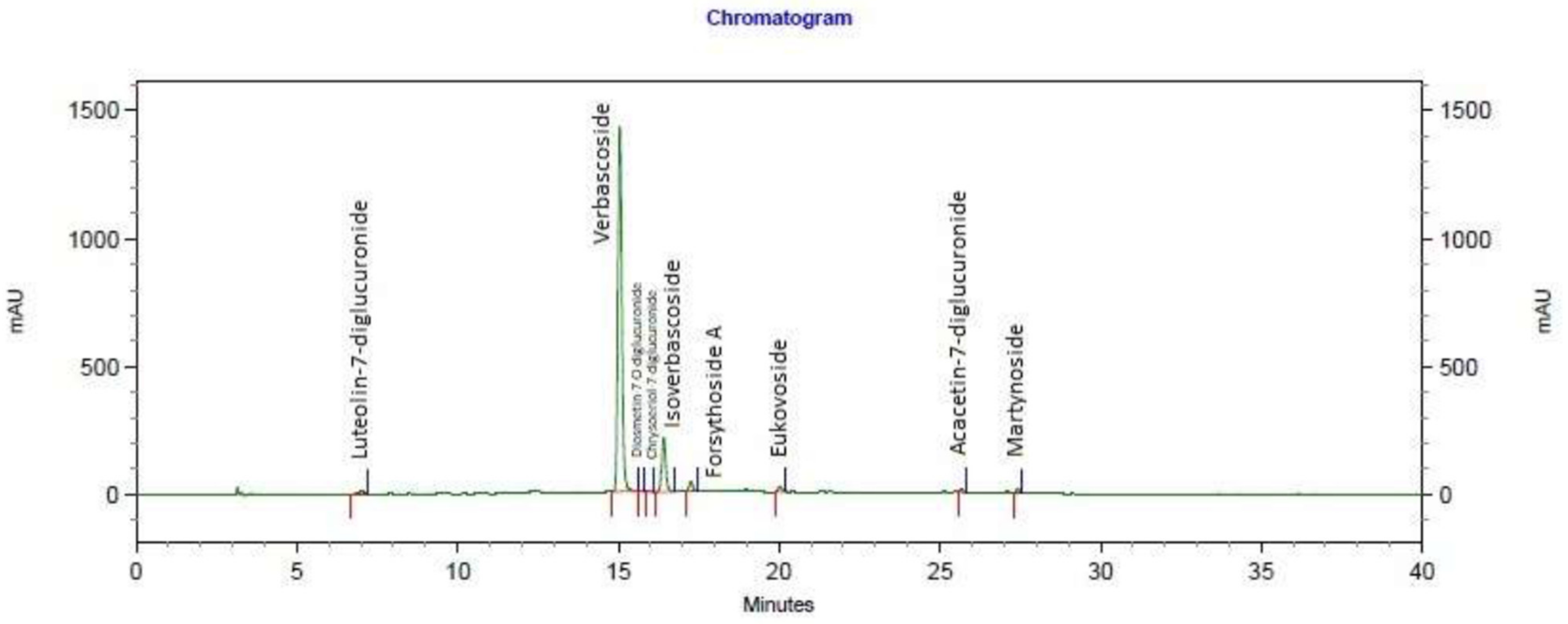
| Name | Retention Time |
|---|---|
| Luteolin-7-diglucuronide | 7.013 |
| Verbascoside | 15.040 |
| Diosmetin-7-O-diglucuronide | 15.720 |
| Chrysoeriol-7-diglucuronide | 15.987 |
| Isoverbascoside | 16.413 |
| Forsythoside A | 17.253 |
| Eukovoside | 20.027 |
| Acacetin-7-diglucuronide | 25.667 |
| Martynoside | 27.427 |
| Variable | Placebo Group (n = 20) | Experimental Group (n = 20) |
|---|---|---|
| Mean ± SD | Mean ± SD | |
| Age (years) | 43.4 ± 13.9 | 34.0 ± 6.28 |
| Height (cm) | 164 ± 8.08 | 173 ± 9.07 |
| Weight (kg) | 63.2 ± 10.7 | 80.8 ± 10.7 |
| BMI (kg/m2) | 23.5 ± 3.0 | 26.7 ± 4.5 |
| Body fat (%) | 23.8 ± 8.17 | 25.7 ± 8.81 |
| Visceral fat | 4.47 ± 2.60 | 5.11 ± 2.84 |
| Variable | Group | Baseline | Month 2 |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| Weight | Placebo | 63.2 ± 10.9 | 63.3 ± 11.0 |
| Experimental | 80.8 ± 16.2 | 80.0 ± 15.8 | |
| BMI | Placebo | 23.5 ± 3.1 | 23.5 ± 3.0 |
| Experimental | 26.7 ± 4.5 | 26.5 ± 4.3 | |
| Fat Mass | Placebo | 24.5 ± 8.2 | 23.5 ± 9.1 |
| Experimental | 25.4 ± 10.3 | 24.9 ± 10.0 |
| Variable | Group | Baseline | Month 1 | Month 2 | Washout |
|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | ||
| PSS Score | Placebo | 22.3 ± 7.1 | 21.5 ± 8.3 | 20.7 ± 7.6 | 20.2 ± 6.6 |
| Experimental | 20.8 ± 7.1 | 20.3 ± 6.6 | 18.7 ± 5.7* | 17.1 ± 7.6 * | |
| Cortisol | Placebo | 16.6 ± 5.6 | 14.3 ± 4.8 *** | 15.7 ± 4.9 | NA |
| Experimental | 15.1 ± 6.0 | 13.7 ± 5.7 ** | 12.6 ± 6.0*** | NA |
| Variable | Group | Month 1 vs. Baseline | Month 2 vs. Baseline | Washout vs. Baseline |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| PSS Score | Placebo | 0.96 ± 0.24 | 0.93 ± 0.27 | 0.91 ± 0.41 |
| Experimental | 0.98 ± 0.24 | 0.90 ± 0.27 * | 0.82 ± 0.25 * | |
| Cortisol | Placebo | 0.87 ± 0.11 *** | 0.94 ± 0.31 | NA |
| Experimental | 0.91 ± 0.08 ** | 0.83 ± 0.31 *** | NA |
| Variable | Group | Baseline | Month 1 | Month 2 | Washout |
|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | ||
| PSQI Score (Total) | Placebo | 8.5 ± 2.9 | 7.8 ± 2.3 | 7.7 ± 2.6 | 7.3 ± 2.8 |
| Experimental | 6.7 ± 2.0 | 6.2 ± 2.2 * | 5.9 ± 2.6 * | 5.0 ± 2.4 ** | |
| PSQI Score Males | Placebo | 5.5 ± 0.6 | 5.38 ± 2.6 | 6.4 ± 4.6 | 4.9 ± 2.8 |
| Experimental | 8.7 ± 3.2 | 8.7 ± 1.9 | 8.0 ± 2.5 | 7.8 ± 3.1* | |
| PSQI Score Females | Placebo | 8.3 ± 2.7 | 8.1 ± 2.4 | 8.3 ± 2.4 | 7.9 ± 2.5 |
| Experimental | 7.3 ± 2.6 | 6.5 ± 2.4 * | 5.7 ±2.5 *† | 5.6 ± 2.7 **† |
| Variable | Group | Month 1 vs. Baseline | Month 2 vs. Baseline | Washout vs. Baseline |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| PSQI Score (Total) | Placebo | 0.92 ± 0.24 | 0.91 ± 0.42 | 0.86 ± 0.48 |
| Experimental | 0.93 ± 0.21 * | 0.88 ± 0.34 * | 0.75± 0.25 ** | |
| PSQI Score Males | Placebo | 0.98 ± 0.18 | 1.18 ± 0.65 | 0.86 ± 0.36 |
| Experimental | 0.99 ± 0.33 | 0.92 ± 0.65 | 0.89 ± 0.36 | |
| PSQI Score Females | Placebo | 0.98 ± 0.23 | 1.00 ± 0.41 | 0.95 ± 0.48 |
| Experimental | 0.88 ± 0.21 * | 0.78 ± 0.28*† | 0.76 ± 0.22 **† |
| Variable | Group | Baseline | Month 1 | Month 2 |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| % Minutes Asleep | Placebo | 87.7 ± 1.7 | 87.6 ± 2.2 | 88.6 ± 3.0 |
| Experimental | 84.9 ± 1.0 | 87.8 ± 1.8 | 88.4 ± 2.3 | |
| % Minutes Awake | Placebo | 12.3 ± 1.7 | 12.4 ± 2.2 | 11.3 ± 2.9 |
| Experimental | 12.1 ± 2.1 | 12.2 ± 1.7 | 11.6 ± 2.3 | |
| Number of Times Awakened | Placebo | 24.2 ± 4.9 | 20.9 ± 5.2 * | 20.6 ± 6.7 |
| Experimental | 28.8 ± 8.7 | 29.9 ± 7.8 | 22.9 ± 7.2 * | |
| % REM | Placebo | 20.2 ± 4.7 | 23.2 ± 8.1 | 19.0 ± 5.2 |
| Experimental | 21.4 ± 5.0 | 21.2 ± 5.3 | 23.0 ± 5.1 † | |
| % Light Sleep | Placebo | 65.3 ± 6.8 | 70.4 ± 12.2 | 65.1 ± 10.1 |
| Experimental | 64.6 ± 9.9 | 62.9 ± 6.5 | 61.3 ± 7.1 | |
| % Deep Sleep | Placebo | 15.2 ± 5.4 | 16.9 ± 7.1 | 16.8 ± 3.6 |
| Experimental | 16.7 ± 3.4 | 16.6 ± 3.4 | 18.6 ± 3.7 * |
| Variable | Group | Month 1 vs. Baseline | Month 2 vs. Baseline |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| % Minutes Asleep | Placebo | 0.99 ± 0.13 | 1.01 ± 0.10 |
| Experimental | 1.03 ± 0.10 | 1.04 ± 0.07 | |
| % Minutes Awake | Placebo | 1.01 ± 0.13 | 0.92 ± 0.21 |
| Experimental | 1.01 ± 0.13 | 0.96 ± 0.16 | |
| Number of Times Awakened | Placebo | 0.86 ± 0.19 * | 0.85 ± 0.28 |
| Experimental | 1.04 ± 0.36 | 0.79 ± 0.29 * | |
| % REM | Placebo | 1.05 ± 0.09 | 0.94 ± 0.18 |
| Experimental | 1.08 ± 0.29 | 1.07 ± 0.25 † | |
| % Light Sleep | Placebo | 1.08 ± 0.06 | 0.99 ± 0.13 |
| Experimental | 0.97 ± 0.15 | 0.95 ± 0.13 | |
| % Deep Sleep | Placebo | 1.11 ± 0.06 | 1.11 ± 0.28 |
| Experimental | 0.99 ± 0.13 | 1.11 ± 0.27 * |
| Variable | Group | Baseline | Month 1 | Month 2 |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| % Minutes Asleep | Placebo | 87.7 ± 1.8 | 87.3 ± 2.2 | 88.0 ± 2.3 |
| Experimental | 88.1 ± 2.4 | 87.6 ± 1.9 | 89.2 ± 1.7 | |
| % Minutes Awake | Placebo | 12.3 ± 1.8 | 12.6 ± 2.2 | 11.8 ± 2.1 |
| Experimental | 11.8 ± 2.2 | 12.3 ± 1.9 | 10.7 ± 1.8 | |
| Number of Times Awakened | Placebo | 23.5 ± 5.3 | 20.6 ± 5.5 | 21.0 ± 5.4 |
| Experimental | 25.6 ± 9.4 | 28.1 ± 7.6 | 19.1 ± 7.6 | |
| % REM | Placebo | 20.6 ± 5.2 | 22.9 ± 8.3 | 19.3 ± 5.8 |
| Experimental | 21.5 ± 6.0 | 21.1 ± 6.5 | 26.9 ± 2.7 † | |
| % Light Sleep | Placebo | 66.3 ± 7.1 | 70.9 ± 13.0 | 66.4 ± 11.1 |
| Experimental | 67.7 ± 12.5 | 65.1 ± 8.1 | 58.8 ± 8.6 | |
| % Deep Sleep | Placebo | 14.1 ± 5.4 | 16.2 ± 7.3 | 16.0 ± 3.6 |
| Experimental | 15.8 ± 3.9 | 15.2 ± 3.5 | 19.1 ± 4.5 * |
| Variable | Group | Month 1 vs. Baseline | Month 2 vs. Baseline |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| % Minutes Asleep | Placebo | 0.99 ± 0.14 | 1.00 ± 0.02 |
| Experimental | 0.99 ± 0.07 | 1.01 ± 0.01 | |
| % Minutes Awake | Placebo | 1.02 ± 0.15 | 0.96 ± 0.13 |
| Experimental | 1.04 ± 0.12 | 0.91 ± 0.05 | |
| Number of Times Awakened | Placebo | 0.88 ± 0.49 | 0.89 ± 0.24 |
| Experimental | 1.10 ± 0.19 | 0.75 ± 0.41 | |
| % REM | Placebo | 1.11 ± 0.21 | 0.94 ± 0.21 |
| Experimental | 0.98 ± 0.07 | 1.25 ± 0.24 † | |
| % Light Sleep | Placebo | 1.07 ± 0.14 | 1.00 ± 0.14 |
| Experimental | 0.96 ± 0.08 | 0.87 ± 0.17 | |
| % Deep Sleep | Placebo | 1.15 ± 0.42 | 1.13 ± 0.28 |
| Experimental | 0.96 ± 0.08 | 1.22 ± 0.29 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Rodríguez, A.; Martínez-Olcina, M.; Mora, J.; Navarro, P.; Caturla, N.; Jones, J. Anxiolytic Effect and Improved Sleep Quality in Individuals Taking Lippia citriodora Extract. Nutrients 2022, 14, 218. https://doi.org/10.3390/nu14010218
Martínez-Rodríguez A, Martínez-Olcina M, Mora J, Navarro P, Caturla N, Jones J. Anxiolytic Effect and Improved Sleep Quality in Individuals Taking Lippia citriodora Extract. Nutrients. 2022; 14(1):218. https://doi.org/10.3390/nu14010218
Chicago/Turabian StyleMartínez-Rodríguez, Alejandro, María Martínez-Olcina, Juan Mora, Pau Navarro, Nuria Caturla, and Jonathan Jones. 2022. "Anxiolytic Effect and Improved Sleep Quality in Individuals Taking Lippia citriodora Extract" Nutrients 14, no. 1: 218. https://doi.org/10.3390/nu14010218
APA StyleMartínez-Rodríguez, A., Martínez-Olcina, M., Mora, J., Navarro, P., Caturla, N., & Jones, J. (2022). Anxiolytic Effect and Improved Sleep Quality in Individuals Taking Lippia citriodora Extract. Nutrients, 14(1), 218. https://doi.org/10.3390/nu14010218







