Metabolic-Associated Fatty Liver Disease (MAFLD), Diabetes, and Cardiovascular Disease: Associations with Fructose Metabolism and Gut Microbiota
Abstract
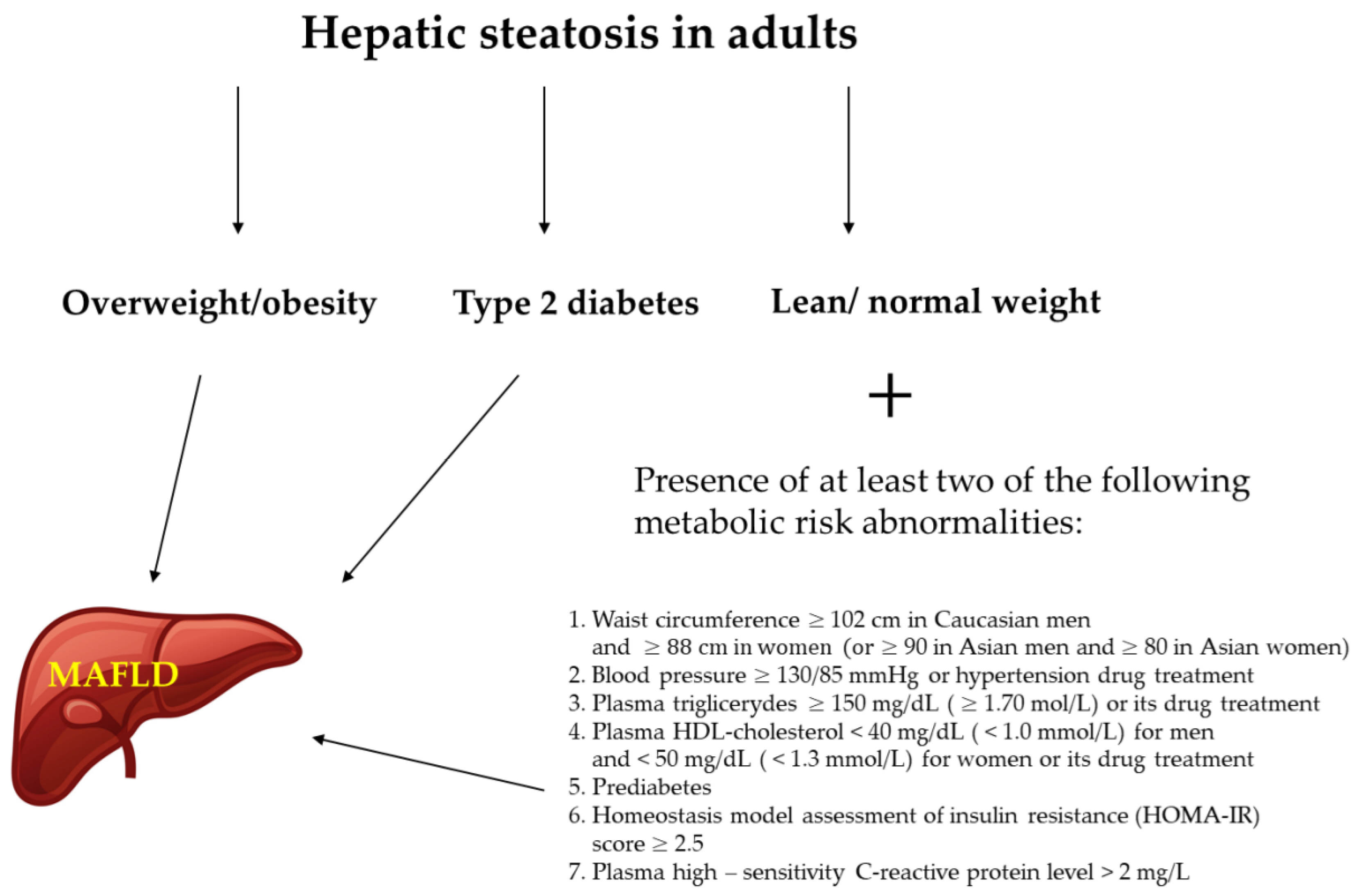
1. Introduction
2. NAFLD and Fructose
3. NAFLD and Gut Microbiota
4. Treatment of NAFLD with Microbiome Alterations
5. NAFLD and T2DM
6. Treatment of NAFLD with Antidiabetic Drugs
7. NAFLD and Cardiovascular Disease
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schaffner, F.; Thaler, H. Nonalcoholic fatty liver disease. Prog. Liver Dis. 1986, 8, 83–298. [Google Scholar]
- Lonardo, A.; Bellini, M.; Tondelli, E.; Frazzoni, M.; Grisendi, A.; Pulvirenti, M.; Della Casa, G. Nonalcoholic steatohepatitis and the “bright liver syndrome”: Should a recently expanded clinical entity be further expanded? Am. J. Gastroenterol. 1995, 90, 2072–2074. [Google Scholar] [PubMed]
- Farrell, G.C.; Chitturi, S.; Lau, G.K.K.; Sollano, J.D. Guidelines for the assessment and management of non-alcoholic fatty liver disease in the Asia–Pacific region: Executive summary. J. Gastroenterol. Hepatol. 2007, 22, 775–777. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.M.; LaVine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef]
- Ratziu, V.; Bellentani, S.; Cortez-Pinto, H.; Day, C.; Marchesini, G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J. Hepatol. 2010, 53, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Addison, T. Observations on fatty degeneration of the liver. Guys Hosp Rep. 1836, 1, 485. [Google Scholar]
- Pepper, W.; Starr, L. A System of Practical Medicine by American Authors. Am. J. Med Sci. 1885, 180, 550–562. [Google Scholar] [CrossRef][Green Version]
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL–EASD–EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Eslam, M.; Newsome, P.N.; Sarin, S.K.; Anstee, Q.M.; Targher, G.; Romero-Gomez, M.; Zelber-Sagi, S.; Wong, V.W.-S.; Dufour, J.-F.; Schattenberg, J.M.; et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 2020, 73, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, L.; Pafundi, P.C.; Galiero, R.; Caturano, A.; Morone, M.V.; Silvestri, C.F.; Giordano, M.; Salvatore, T.; Sasso, F.C. Mechanisms of Non-Alcoholic Fatty Liver Disease in the Metabolic Syndrome. A Narrative Review. Antioxidants 2021, 10, 270. [Google Scholar] [CrossRef] [PubMed]
- Fouad, Y.; Waked, I.; Bollipo, S.; Gomaa, A.; Ajlouni, Y.; Attia, D. What’s in a name? Renaming ‘NAFLD’ to ‘MAFLD’. Liver Int. 2020, 40, 1254–1261. [Google Scholar] [CrossRef]
- Wong, R.J.; Cheung, R.; Ahmed, A. Nonalcoholic steatohepatitis is the most rapidly growing indication for liver transplantation in patients with hepatocellular carcinoma in the U.S. Hepatology 2014, 59, 2188–2195. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Noureddin, M.; Vipani, A.; Bresee, C.; Todo, T.; Kim, I.K.; Alkhouri, N.; Setiawan, V.; Tran, T.; Ayoub, W.S.; Lu, S.C.; et al. NASH Leading Cause of Liver Transplant in Women: Updated Analysis of Indications for Liver Transplant and Ethnic and Gender Variances. Am. J. Gastroenterol. 2018, 113, 1649–1659. [Google Scholar] [CrossRef]
- Cave, M.; Deaciuc, I.; Mendez, C.; Song, Z.; Joshi-Barve, S.; Barve, S.; McClain, C. Nonalcoholic fatty liver disease: Predisposing factors and the role of nutrition. J. Nutr. Biochem. 2007, 18, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Elliott, S.S.; Keim, N.L.; Stern, J.S.; Teff, K.; Havel, P.J. Fructose, weight gain, and the insulin resistance syndrome. Am. J. Clin. Nutr. 2002, 76, 911–922. [Google Scholar] [CrossRef]
- Jensen, T.; Abdelmalek, M.F.; Sullivan, S.; Nadeau, K.J.; Cree-Green, M.; Roncal, C.; Nakagawa, T.; Kuwabara, M.; Sato, Y.; Kang, D.-H.; et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. J. Hepatol. 2018, 68, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Basaranoglu, M.; Basaranoglu, G.; Bugianesi, E. Carbohydrate intake and nonalcoholic fatty liver disease: Fructose as a weapon of mass destruction. Hepatobiliary Surg. Nutr. 2015, 4, 109–116. [Google Scholar] [CrossRef]
- Sánchez-Lozada, L.G.; Mu, W.; Roncal, C.; Sautin, Y.Y.; Abdelmalek, M.; Reungjui, S.; Le, M.; Nakagawa, T.; Lan, H.Y.; Yu, X.; et al. Comparison of free fructose and glucose to sucrose in the ability to cause fatty liver. Eur. J. Nutr. 2009, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aron-Wisnewsky, J.; Vigliotti, C.; Witjes, J.; Le, P.; Holleboom, A.G.; Verheij, J.; Nieuwdorp, M.; Clément, K. Gut microbiota and human NAFLD: Disentangling microbial signatures from metabolic disorders. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 279–297. [Google Scholar] [CrossRef]
- Wong, R.J.; Aguilar, M.; Cheung, R.; Perumpail, R.B.; Harrison, S.A.; Younossi, Z.M.; Ahmed, A. Nonalcoholic Steatohepatitis Is the Second Leading Etiology of Liver Disease among Adults Awaiting Liver Transplantation in the United States. Gastroenterology 2015, 148, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Haldar, D.; Kern, B.; Hodson, J.; Armstrong, M.; Adam, R.; Berlakovich, G.; Fritz, J.; Feurstein, B.; Popp, W.; Karam, V.; et al. Outcomes of liver transplantation for non-alcoholic steatohepatitis: A European Liver Transplant Registry study. J. Hepatol. 2019, 71, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Byrne, C.D.; Tilg, H. NAFLD and increased risk of cardiovascular disease: Clinical associations, pathophysiological mechanisms and pharmacological implications. Gut 2020, 69, 1691–1705. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, V.A. Fructose as a Dietary Sweetener in Diabetes Mellitus. Diabetes Care 1978, 1, 241–246. [Google Scholar] [CrossRef]
- Moorhouse, J.A.; Kark, R.M. Fructose and diabetes. Am. J. Med. 1957, 23, 46–58. [Google Scholar] [CrossRef]
- Pelkonen, R.; Aro, A.; Nikkilä, E.A. Metabolic Effects of Dietary Fructose in Insulin Dependent Diabetes of Adults. Acta Med. Scand. 2009, 192, 187–193. [Google Scholar] [CrossRef]
- Lim, J.S.; Mietus-Snyder, M.; Valente, A.; Schwarz, J.-M.; Lustig, R.H. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 251–264. [Google Scholar] [CrossRef]
- Schwarz, J.-M.; Noworolski, S.M.; Wen, M.J.; Dyachenko, A.; Prior, J.L.; Weinberg, M.E.; Herraiz, L.A.; Tai, V.W.; Bergeron, N.; Bersot, T.P.; et al. Effect of a High-Fructose Weight-Maintaining Diet on Lipogenesis and Liver Fat. J. Clin. Endocrinol. Metab. 2015, 100, 2434–2442. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Schwarz, J.M.; Keim, N.L.; Griffen, S.C.; Bremer, A.A.; Graham, J.L.; Hatcher, B.; Cox, C.L.; Dyachenko, A.; Zhang, W.; et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J. Clin. Investig. 2009, 119, 1322–1334. [Google Scholar] [CrossRef] [PubMed]
- Skenderian, S.; Park, G.; Jang, C. Organismal Fructose Metabolism in Health and Non-Alcoholic Fatty Liver Disease. Biology 2020, 9, 405. [Google Scholar] [CrossRef] [PubMed]
- Jegatheesan, P.; De Bandt, J. Fructose and NAFLD: The Multifaceted Aspects of Fructose Metabolism. Nutrients 2017, 9, 230. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Sanchez-Lozada, L.-G.; Andrews, P.; Lanaspa, M.A. Perspective: A Historical and Scientific Perspective of Sugar and Its Relation with Obesity and Diabetes. Adv. Nutr. 2017, 8, 412–422. [Google Scholar] [CrossRef]
- GBD 2016 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines on Sugar Intake. Available online: https://www.who.int/elena/titles/guidance_summaries/sugars_intake/en/ (accessed on 24 November 2021).
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Blendis, L.; Halpern, Z.; Oren, R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): A population based study. J. Hepatol. 2007, 47, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Assy, N.; Nasser, G.; Kamayse, I.; Nseir, W.; Beniashvili, Z.; Djibre, A.; Grosovski, M. Soft Drink Consumption Linked with Fatty Liver in the Absence of Traditional Risk Factors. Can. J. Gastroenterol. 2008, 22, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Abid, A.; Taha, O.; Nseir, W.; Farah, R.; Grosovski, M.; Assy, N. Soft drink consumption is associated with fatty liver disease independent of metabolic syndrome. J. Hepatol. 2009, 51, 918–924. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.F.; Suzuki, A.; Guy, C.; Unalp-Arida, A.; Colvin, R.; Johnson, R.J.; Diehl, A.M.; Nonalcoholic Steatohepatitis Clinical Research Network. Increased fructose consumption is associated with fibrosis severity in patients with nonalcoholic fatty liver disease. Hepatology 2010, 51, 1961–1971. [Google Scholar] [CrossRef] [PubMed]
- Maersk, M.; Belza, A.; Stødkilde-Jørgensen, H.; Ringgaard, S.; Chabanova, E.; Thomsen, H.; Pedersen, S.B.; Astrup, A.; Richelsen, B. Sucrose-sweetened beverages increase fat storage in the liver, muscle, and visceral fat depot: A 6-mo randomized intervention study. Am. J. Clin. Nutr. 2011, 95, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.; Sievenpiper, J.L.; de Souza, R.; Cozma, A.I.; Mirrahimi, A.; Carleton, A.J.; Ha, V.; Di Buono, M.; Jenkins, A.L.; Leiter, L.A.; et al. Effect of fructose on markers of non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis of controlled feeding trials. Eur. J. Clin. Nutr. 2014, 68, 416–423. [Google Scholar] [CrossRef]
- Chung, M.; Ma, J.; Patel, K.; Berger, S.; Lau, J.; Lichtenstein, A.H. Fructose, high-fructose corn syrup, sucrose, and nonalcoholic fatty liver disease or indexes of liver health: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 833–849. [Google Scholar] [CrossRef]
- Hochuli, M.; Herter-Aeberli, I.; Weiss, A.; Hersberger, M.; Troxler, H.; Gerber, P.; Spinas, G.A.; Berneis, K. Sugar-Sweetened Beverages with Moderate Amounts of Fructose, but Not Sucrose, Induce Fatty Acid Synthesis in Healthy Young Men: A Randomized Crossover Study. J. Clin. Endocrinol. Metab. 2014, 99, 2164–2172. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Welsh, J.A.; Le, N.-A.; Holzberg, J.; Sharma, P.; Martin, D.R.; Vos, M.B. Dietary Fructose Reduction Improves Markers of Cardiovascular Disease Risk in Hispanic-American Adolescents with NAFLD. Nutrients 2014, 6, 3187–3201. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Fox, C.S.; Jacques, P.F.; Speliotes, E.K.; Hoffmann, U.; Smith, C.E.; Saltzman, E.; McKeown, N.M. Sugar-sweetened beverage, diet soda, and fatty liver disease in the Framingham Heart Study cohorts. J. Hepatol. 2015, 63, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Wijarnpreecha, K.; Thongprayoon, C.; Edmonds, P.; Cheungpasitporn, W. Associations of sugar- and artificially sweetened soda with nonalcoholic fatty liver disease: A systematic review and meta-analysis. QJM Int. J. Med. 2016, 109, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wang, J.; Li, Z.; Lam, C.W.K.; Xiao, Y.; Wu, Q.; Zhang, W. Consumption of Sugar-Sweetened Beverages Has a Dose-Dependent Effect on the Risk of Non-Alcoholic Fatty Liver Disease: An Updated Systematic Review and Dose-Response Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 2192. [Google Scholar] [CrossRef] [PubMed]
- Lambertz, J.; Weiskirchen, S.; Landert, S.; Weiskirchen, R. Fructose: A Dietary Sugar in Crosstalk with Microbiota Contributing to the Development and Progression of Non-Alcoholic Liver Disease. Front. Immunol. 2017, 8, 1159. [Google Scholar] [CrossRef]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut Microbiota from Twins Discordant for Obesity Modulate Metabolism in Mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef] [PubMed]
- Aron-Wisnewsky, J.; Gaborit, B.; Dutour, A.; Clement, K. Gut microbiota and non-alcoholic fatty liver disease: New insights. Clin. Microbiol. Infect. 2013, 19, 338–348. [Google Scholar] [CrossRef]
- Wieland, A.; Frank, D.N.; Harnke, B.; Bambha, K. Systematic review: Microbial dysbiosis and nonalcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2015, 42, 1051–1063. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, S.; Selvakumar, P.C.; Cresci, G.A. The Role of the Gut Microbiome in Nonalcoholic Fatty Liver Disease. Med. Sci. 2018, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Henao-Mejia, J.; Elinav, E.; Jin, C.; Hao, L.; Mehal, W.Z.; Strowig, T.; Thaiss, C.A.; Kau, A.L.; Eisenbarth, S.C.; Jurczak, M.J.; et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 2012, 482, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.-C.; Ching, Y.-H.; Li, Y.-P.; Liu, J.-Y.; Huang, Y.-T.; Huang, Y.-W.; Yang, S.-S.; Huang, W.-C.; Chuang, H.-L. Nonalcoholic Fatty Liver Disease Is Exacerbated in High-Fat Diet-Fed Gnotobiotic Mice by Colonization with the Gut Microbiota from Patients with Nonalcoholic Steatohepatitis. Nutrients 2017, 9, 1220. [Google Scholar] [CrossRef] [PubMed]
- Farrell, G.; Schattenberg, J.M.; Leclercq, I.; Yeh, M.M.; Goldin, R.; Teoh, N.; Schuppan, D. Mouse Models of Nonalcoholic Steatohepatitis: Toward Optimization of Their Relevance to Human Nonalcoholic Steatohepatitis. Hepatology 2018, 69, 2241–2257. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.L.A.; Vieira-Silva, S.; Liston, A.; Raes, J. How informative is the mouse for human gut microbiota research? Dis. Model. Mech. 2015, 8, 1–16. [Google Scholar] [CrossRef]
- Brandl, K.; Schnabl, B. Intestinal microbiota and nonalcoholic steatohepatitis. Curr. Opin. Gastroenterol. 2017, 33, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Albillos, A.; de Gottardi, A.; Rescigno, M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J. Hepatol. 2020, 72, 558–577. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Lin, A.; Kong, M.; Yao, X.; Yin, M.; Xia, H.; Ma, J.; Liu, H. Intestinal microbiome and NAFLD: Molecular insights and therapeutic perspectives. J. Gastroenterol. 2019, 55, 142–158. [Google Scholar] [CrossRef]
- Dong, T.S.; Jacobs, J.P. Nonalcoholic fatty liver disease and the gut microbiome: Are bacteria responsible for fatty liver? Exp. Biol. Med. 2019, 244, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Jiang, X.; Cao, M.; Ge, J.; Bao, Q.; Tang, L.; Chen, Y.; Li, L. Altered Fecal Microbiota Correlates with Liver Biochemistry in Nonobese Patients with Non-alcoholic Fatty Liver Disease. Sci. Rep. 2016, 6, 32002. [Google Scholar] [CrossRef]
- Shen, F.; Zheng, R.-D.; Sun, X.-Q.; Ding, W.-J.; Wang, X.-Y.; Fan, J.-G. Gut microbiota dysbiosis in patients with non-alcoholic fatty liver disease. Hepatobiliary Pancreat. Dis. Int. 2017, 16, 375–381. [Google Scholar] [CrossRef]
- Li, F.; Ye, J.; Shao, C.; Zhong, B. Compositional alterations of gut microbiota in nonalcoholic fatty liver disease patients: A systematic review and Meta-analysis. Lipids Health Dis. 2021, 20, 22. [Google Scholar] [CrossRef]
- Lanthier, N.; Rodriguez, J.; Nachit, M.; Hiel, S.; Trefois, P.; Neyrinck, A.M.; Cani, P.D.; Bindels, L.B.; Thissen, J.-P.; Delzenne, N.M. Microbiota analysis and transient elastography reveal new extra-hepatic components of liver steatosis and fibrosis in obese patients. Sci. Rep. 2021, 11, 659. [Google Scholar] [CrossRef]
- Raman, M.; Ahmed, I.; Gillevet, P.M.; Probert, C.S.; Ratcliffe, N.M.; Smith, S.; Greenwood, R.; Sikaroodi, M.; Lam, V.; Crotty, P.; et al. Fecal Microbiome and Volatile Organic Compound Metabolome in Obese Humans with Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2013, 11, 868–875.e3. [Google Scholar] [CrossRef]
- Da Silva, H.E.; Teterina, A.; Comelli, E.M.; Taibi, A.; Arendt, B.M.; Fischer, S.E.; Lou, W.; Allard, J.P. Nonalcoholic fatty liver disease is associated with dysbiosis independent of body mass index and insulin resistance. Sci. Rep. 2018, 8, 1466. [Google Scholar] [CrossRef] [PubMed]
- Arasaradnam, R.P.; Covington, J.; Harmston, C.; Nwokolo, C.U. Review article: Next generation diagnostic modalities in gastroenterology—Gas phase volatile compound biomarker detection. Aliment. Pharmacol. Ther. 2014, 39, 780–789. [Google Scholar] [CrossRef]
- Ma, Y.Y.; Li, L.; Yu, C.H.; Shen, Z.; Chen, L.H.; Li, Y.M. Effects of probiotics on nonalcoholic fatty liver disease: A meta-analysis. World J. Gastroenterol. 2013, 19, 6911–6918. [Google Scholar] [CrossRef] [PubMed]
- Loman, B.; Hernández-Saavedra, D.; An, R.; Rector, R.S. Prebiotic and probiotic treatment of nonalcoholic fatty liver disease: A systematic review and meta-analysis. Nutr. Rev. 2018, 76, 822–839. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.Y.; Mihali, A.B.; Rawala, M.S.; Aslam, A.; Siddiqui, W.J. The promising role of probiotic and synbiotic therapy in aminotransferase levels and inflammatory markers in patients with nonalcoholic fatty liver disease—A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2019, 31, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Sharpton, S.R.; Maraj, B.; Harding-Theobald, E.; Vittinghoff, E.; Terrault, N.A. Gut microbiome–targeted therapies in nonalcoholic fatty liver disease: A systematic review, meta-analysis, and meta-regression. Am. J. Clin. Nutr. 2019, 110, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Wen, S.W.; Kaminga, A.C.; Liu, A. Gut metabolites and inflammation factors in non-alcoholic fatty liver disease: A systematic review and meta-analysis. Sci. Rep. 2020, 10, 8848. [Google Scholar] [CrossRef] [PubMed]
- Binda, S.; Hill, C.; Johansen, E.; Obis, D.; Pot, B.; Sanders, M.E.; Tremblay, A.; Ouwehand, A.C. Criteria to Qualify Microorganisms as “Probiotic” in Foods and Dietary Supplements. Front. Microbiol. 2020, 11, 1662. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Asp, N.-G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. FAO Technical Meeting on Prebiotics. J. Clin. Gastroenterol. 2008, 42, S156–S159. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 2016, 65, 1038–1048. [Google Scholar] [CrossRef]
- Bakhshimoghaddam, F.; Shateri, K.; Sina, M.; Hashemian, M.; Alizadeh, M. Daily Consumption of Synbiotic Yogurt Decreases Liver Steatosis in Patients with Nonalcoholic Fatty Liver Disease: A Randomized Controlled Clinical Trial. J. Nutr. 2018, 148, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Eslamparast, T.; Poustchi, H.; Zamani, F.; Sharafkhah, M.; Malekzadeh, R.; Hekmatdoost, A. Synbiotic supplementation in nonalcoholic fatty liver disease: A randomized, double-blind, placebo-controlled pilot study. Am. J. Clin. Nutr. 2014, 99, 535–542. [Google Scholar] [CrossRef]
- Van Nood, E.; Vrieze, A.; Nieuwdorp, M.; Fuentes, S.; Zoetendal, E.G.; De Vos, W.M.; Visser, C.E.; Kuijper, E.J.; Bartelsman, J.F.W.M.; Tijssen, J.G.P.; et al. Duodenal Infusion of Donor Feces for Recurrent Clostridium difficile. N. Engl. J. Med. 2013, 368, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Aron-Wisnewsky, J.; Clément, K.; Nieuwdorp, M. Fecal Microbiota Transplantation: A Future Therapeutic Option for Obesity/Diabetes? Curr. Diabetes Rep. 2019, 19, 51. [Google Scholar] [CrossRef] [PubMed]
- Craven, L.; Rahman, A.; Nair Parvathy, S.; Beaton, M.; Silverman, J.; Qumosani, K.; Hramiak, I.; Hegele, R.; Joy, T.; Meddings, J.; et al. Allogenic Fecal Microbiota Transplantation in Patients with Nonalcoholic Fatty Liver Disease Improves Abnormal Small Intestinal Permeability: A Randomized Control Trial. Am. J. Gastroenterol. 2020, 115, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Witjes, J.J.; Smits, L.P.; Pekmez, C.T.; Prodan, A.; Meijnikman, A.S.; Troelstra, M.A.; Bouter, K.E.; Herrema, H.; Levin, E.; Holleboom, A.G.; et al. Donor Fecal Microbiota Transplantation Alters Gut Microbiota and Metabolites in Obese Individuals with Steatohepatitis. Hepatol. Commun. 2020, 4, 1578–1590. [Google Scholar] [CrossRef]
- Blond, E.; Disse, E.; Cuerq, C.; Drai, J.; Valette, P.-J.; Laville, M.; Thivolet, C.; Simon, C.; Caussy, C. EASL–EASD–EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease in severely obese people: Do they lead to over-referral? Diabetologia 2017, 60, 1218–1222. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Byrne, C.D.; Bonora, E.; Targher, G. Nonalcoholic Fatty Liver Disease and Risk of Incident Type 2 Diabetes: A Meta-analysis. Diabetes Care 2018, 41, 372–382. [Google Scholar] [CrossRef]
- Lallukka, S.; Yki-Järvinen, H. Non-alcoholic fatty liver disease and risk of type 2 diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2016, 30, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Cho, Y.K.; Kang, Y.M.; Kim, H.S.; Jung, C.H.; Kim, H.-K.; Park, J.-Y.; Lee, W.J. The Impact of NAFLD and Waist Circumference Changes on Diabetes Development in Prediabetes Subjects. Sci. Rep. 2019, 9, 17258. [Google Scholar] [CrossRef]
- Ballestri, S.; Zona, S.; Targher, G.; Romagnoli, D.; Baldelli, E.; Nascimbeni, F.; Roverato, A.; Guaraldi, G.; Lonardo, A. Nonalcoholic fatty liver disease is associated with an almost twofold increased risk of incident type 2 diabetes and metabolic syndrome. Evidence from a systematic review and meta-analysis. J. Gastroenterol. Hepatol. 2016, 31, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.; Choi, K.C.; Wong, G.L.-H.; Zhang, Y.; Chan, H.L.-Y.; Luk, A.O.-Y.; Shu, S.S.-T.; Chan, A.W.-H.; Yeung, M.-W.; Chan, J.C.-N.; et al. Screening diabetic patients for non-alcoholic fatty liver disease with controlled attenuation parameter and liver stiffness measurements: A prospective cohort study. Gut 2015, 65, 1359–1368. [Google Scholar] [CrossRef]
- Caturano, A.; Acierno, C.; Nevola, R.; Pafundi, P.C.; Galiero, R.; Rinaldi, L.; Salvatore, T.; Adinolfi, L.E.; Sasso, F.C. Non-Alcoholic Fatty Liver Disease: From Pathogenesis to Clinical Impact. Processes 2021, 9, 135. [Google Scholar] [CrossRef]
- Lonardo, A.; Lugari, S.; Ballestri, S.; Nascimbeni, F.; Baldelli, E.; Maurantonio, M. A round trip from nonalcoholic fatty liver disease to diabetes: Molecular targets to the rescue? Acta Diabetol. 2018, 56, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Bril, F.; Cusi, K. Management of Nonalcoholic Fatty Liver Disease in Patients with Type 2 Diabetes: A Call to Action. Diabetes Care 2017, 40, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Valenti, L.; Bugianesi, E.; Pajvani, U.; Targher, G. Nonalcoholic fatty liver disease: Cause or consequence of type 2 diabetes? Liver Int. 2016, 36, 1563–1579. [Google Scholar] [CrossRef] [PubMed]
- Cusi, K.; Sanyal, A.J.; Zhang, S.; Hartman, M.L.; Bue-Valleskey, J.M.; Hoogwerf, B.J.; Haupt, A. Non-alcoholic fatty liver disease (NAFLD) prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes. Metab. 2017, 19, 1630–1634. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Hwang, S.; Park, J.I.; Yang, M.J.; Hwang, J.C.; Yoo, B.M.; Lee, K.M.; Shin, S.J.; Lee, K.J.; Kim, J.H.; et al. Improvement of Nonalcoholic Fatty Liver Disease Reduces the Risk of Type 2 Diabetes Mellitus. Gut Liver 2019, 13, 440–449. [Google Scholar] [CrossRef]
- Mantovani, A.; Petracca, G.; Beatrice, G.; Tilg, H.; Byrne, C.D.; Targher, G. Non-alcoholic fatty liver disease and risk of incident diabetes mellitus: An updated meta-analysis of 501,022 adult individuals. Gut 2020, 70, 962–969. [Google Scholar] [CrossRef]
- Masarone, M.; Rosato, V.; Aglitti, A.; Bucci, T.; Caruso, R.; Salvatore, T.; Sasso, F.C.; Tripodi, M.F.; Persico, M. Liver biopsy in type 2 diabetes mellitus: Steatohepatitis represents the sole feature of liver damage. PLoS ONE 2017, 12, e0178473. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, L.E.; Petta, S.; Fracanzani, A.L.; Nevola, R.; Coppola, C.; Narciso, V.; Rinaldi, L.; Calvaruso, V.; Pafundi, P.C.; Lombardi, R.; et al. Reduced incidence of type 2 diabetes in patients with chronic hepatitis C virus infection cleared by direct-acting antiviral therapy: A prospective study. Diabetes Obes. Metab. 2020, 22, 2408–2416. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, L.E.; Petta, S.; Fracanzani, A.L.; Coppola, C.; Narciso, V.; Nevola, R.; Rinaldi, L.; Calvaruso, V.; Staiano, L.; Di Marco, V.; et al. Impact of hepatitis C virus clearance by direct-acting antiviral treatment on the incidence of major cardiovascular events: A prospective multicentre study. Atherosclerosis 2020, 296, 40–47. [Google Scholar] [CrossRef]
- Sasso, F.C.; Pafundi, P.C.; Caturano, A.; Galiero, R.; Vetrano, E.; Nevola, R.; Petta, S.; Fracanzani, A.L.; Coppola, C.; Di Marco, V.; et al. Impact of direct acting antivirals (DAAs) on cardiovascular events in HCV cohort with pre-diabetes. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 2345–2353. [Google Scholar] [CrossRef] [PubMed]
- Aithal, G.P.; Thomas, J.; Kaye, P.V.; Lawson, A.; Ryder, S.D.; Spendlove, I.; Austin, A.S.; Freeman, J.G.; Morgan, L.; Webber, J. Randomized, Placebo-Controlled Trial of Pioglitazone in Nondiabetic Subjects with Nonalcoholic Steatohepatitis. Gastroenterology 2008, 135, 1176–1184. [Google Scholar] [CrossRef] [PubMed]
- Gastaldelli, A.; Harrison, S.; Belfort-Aguiar, R.; Hardies, J.; Balas, B.; Schenker, S.; Cusi, K. Pioglitazone in the treatment of NASH: The role of adiponectin. Aliment. Pharmacol. Ther. 2010, 32, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Boettcher, E.; Csako, G.; Pucino, F.; Wesley, R.; Loomba, R. Meta-analysis: Pioglitazone improves liver histology and fibrosis in patients with non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2011, 35, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Cusi, K.; Orsak, B.; Bril, F.; Lomonaco, R.; Hecht, J.; Ortiz-Lopez, C.; Tio, F.; Hardies, J.; Darland, C.; Musi, N.; et al. Long-Term Pioglitazone Treatment for Patients with Nonalcoholic Steatohepatitis and Prediabetes or Type 2 Diabetes Mellitus. Ann. Intern. Med. 2016, 165, 305–315. [Google Scholar] [CrossRef]
- Lian, J.; Fu, J. Efficacy of Various Hypoglycemic Agents in the Treatment of Patients with Nonalcoholic Liver Disease with or without Diabetes: A Network Meta-Analysis. Front. Endocrinol. 2021, 12, 649018. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, T.; Fushimi, N.; Kawai, M.; Yoshida, Y.; Hachiya, H.; Ito, S.; Kawai, H.; Ohashi, N.; Mori, A. Luseogliflozin improves liver fat deposition compared to metformin in type 2 diabetes patients with non-alcoholic fatty liver disease: A prospective randomized controlled pilot study. Diabetes Obes. Metab. 2017, 20, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Aso, Y.; Kato, K.; Sakurai, S.; Kishi, H.; Shimizu, M.; Jojima, T.; Iijima, T.; Maejima, Y.; Shimomura, K.; Usui, I. Impact of dapagliflozin, an SGLT2 inhibitor, on serum levels of soluble dipeptidyl peptidase-4 in patients with type 2 diabetes and non-alcoholic fatty liver disease. Int. J. Clin. Pract. 2019, 73, e13335. [Google Scholar] [CrossRef]
- Lai, L.-L.; Vethakkan, S.R.; Mustapha, N.R.N.; Mahadeva, S.; Chan, W.-K. Empagliflozin for the Treatment of Nonalcoholic Steatohepatitis in Patients with Type 2 Diabetes Mellitus. Dig. Dis. Sci. 2019, 65, 623–631. [Google Scholar] [CrossRef]
- Kuchay, M.S.; Krishan, S.; Mishra, S.K.; Farooqui, K.J.; Singh, M.K.; Wasir, J.S.; Bansal, B.; Kaur, P.; Jevalikar, G.; Gill, H.K.; et al. Effect of Empagliflozin on Liver Fat in Patients with Type 2 Diabetes and Nonalcoholic Fatty Liver Disease: A Randomized Controlled Trial (E-LIFT Trial). Diabetes Care 2018, 41, 1801–1808. [Google Scholar] [CrossRef]
- Shimizu, M.; Suzuki, K.; Kato, K.; Jojima, T.; Iijima, T.; Murohisa, T.; Iijima, M.; Takekawa, H.; Usui, I.; Hiraishi, H.; et al. Evaluation of the effects of dapagliflozin, a sodium-glucose co-transporter-2 inhibitor, on hepatic steatosis and fibrosis using transient elastography in patients with type 2 diabetes and non-alcoholic fatty liver disease. Diabetes Obes. Metab. 2018, 21, 285–292. [Google Scholar] [CrossRef]
- Cusi, K.; Sattar, N.; García-Pérez, L.-E.; Pavo, I.; Yu, M.; Robertson, K.E.; Karanikas, C.A.; Haupt, A. Dulaglutide decreases plasma aminotransferases in people with Type 2 diabetes in a pattern consistent with liver fat reduction: A post hoc analysis of the AWARD programme. Diabet. Med. 2018, 35, 1434–1439. [Google Scholar] [CrossRef]
- Mantovani, A.; Petracca, G.; Beatrice, G.; Csermely, A.; Lonardo, A.; Targher, G. Glucagon-Like Peptide-1 Receptor Agonists for Treatment of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis: An Updated Meta-Analysis of Randomized Controlled Trials. Metabolites 2021, 11, 73. [Google Scholar] [CrossRef]
- Newsome, P.N.; Buchholtz, K.; Cusi, K.; Linder, M.; Okanoue, T.; Ratziu, V.; Sanyal, A.J.; Sejling, A.-S.; Harrison, S.A. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2021, 384, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Jia, Y.; Li, Z.; Wang, F.; Ren, L.; Chen, S. Effects of Liraglutide on Nonalcoholic Fatty Liver Disease in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Diabetes Ther. 2021, 12, 1735–1749. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, Y.; Kitajima, Y.; Hyogo, H.; Takahashi, H.; Kojima, M.; Ono, M.; Araki, N.; Tanaka, K.; Yamaguchi, M.; Matsuda, Y.; et al. Pilot study of liraglutide effects in non-alcoholic steatohepatitis and non-alcoholic fatty liver disease with glucose intolerance in Japanese patients (LEAN-J). Hepatol. Res. 2014, 45, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.A.; Patti, A.M.; Giglio, R.V.; Nikolic, D.; Amato, A.; Al-Busaidi, N.; Al-Rasadi, K.; Soresi, M.; Banach, M.; Montalto, G.; et al. Liraglutide improves carotid intima-media thickness in patients with type 2 diabetes and non-alcoholic fatty liver disease: An 8-month prospective pilot study. Expert Opin. Biol. Ther. 2015, 15, 1391–1397. [Google Scholar] [CrossRef]
- Armstrong, M.; Gaunt, P.; Aithal, G.; Barton, D.; Hull, D.; Parker, R.; Hazlehurst, J.M.; Guo, K.; Abouda, G.; Aldersley, M.A.; et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): A multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016, 387, 679–690. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Hull, D.; Guo, K.; Barton, D.; Hazlehurst, J.M.; Gathercole, L.; Nasiri, M.; Yu, J.; Gough, S.C.; Newsome, P.; et al. Glucagon-like peptide 1 decreases lipotoxicity in non-alcoholic steatohepatitis. J. Hepatol. 2015, 64, 399–408. [Google Scholar] [CrossRef]
- Feng, W.; Gao, C.; Bi, Y.; Wu, M.; Li, P.; Shen, S.; Chen, W.; Yin, T.; Zhu, D. Randomized trial comparing the effects of gliclazide, liraglutide, and metformin on diabetes with non-alcoholic fatty liver disease. J. Diabetes 2017, 9, 800–809. [Google Scholar] [CrossRef]
- Seko, Y.; Sumida, Y.; Tanaka, S.; Mori, K.; Taketani, H.; Ishiba, H.; Hara, T.; Okajima, A.; Umemura, A.; Nishikawa, T.; et al. Effect of 12-week dulaglutide therapy in Japanese patients with biopsy-proven non-alcoholic fatty liver disease and type 2 diabetes mellitus. Hepatol. Res. 2016, 47, 1206–1211. [Google Scholar] [CrossRef]
- Yan, J.; Yao, B.; Kuang, H.; Yang, X.; Huang, Q.; Hong, T.; Li, Y.; Dou, J.; Yang, W.; Qin, G.; et al. Liraglutide, Sitagliptin, and Insulin Glargine Added to Metformin: The Effect on Body Weight and Intrahepatic Lipid in Patients with Type 2 Diabetes Mellitus and Nonalcoholic Fatty Liver Disease. Hepatology 2019, 69, 2414–2426. [Google Scholar] [CrossRef]
- Kuchay, M.S.; Krishan, S.; Mishra, S.K.; Choudhary, N.S.; Singh, M.K.; Wasir, J.S.; Kaur, P.; Gill, H.K.; Bano, T.; Farooqui, K.J.; et al. Effect of dulaglutide on liver fat in patients with type 2 diabetes and NAFLD: Randomised controlled trial (D-LIFT trial). Diabetologia 2020, 63, 2434–2445. [Google Scholar] [CrossRef]
- Lee, K.W.; Devaraj, N.K.; Ching, S.M.; Veettil, S.K.; Hoo, F.K.; Deuraseh, I.; Soo, M.J. Effect of SGLT-2 Inhibitors on Non-alcoholic Fatty Liver Disease among Patients with Type 2 Diabetes Mellitus: Systematic Review with Meta-analysis and Trial Sequential Analysis of Randomized Clinical Trials. Oman Med. J. 2021, 36, e273. [Google Scholar] [CrossRef]
- Hartman, M.L.; Sanyal, A.J.; Loomba, R.; Wilson, J.M.; Nikooienejad, A.; Bray, R.; Karanikas, C.A.; Duffin, K.L.; Robins, D.A.; Haupt, A. Effects of Novel Dual GIP and GLP-1 Receptor Agonist Tirzepatide on Biomarkers of Nonalcoholic Steatohepatitis in Patients with Type 2 Diabetes. Diabetes Care 2020, 43, 1352–1355. [Google Scholar] [CrossRef] [PubMed]
- Tokushige, K.; Ikejima, K.; Ono, M.; Eguchi, Y.; Kamada, Y.; Itoh, Y.; Akuta, N.; Yoneda, M.; Iwasa, M.; Yoneda, M.; et al. Evidence-based clinical practice guidelines for nonalcoholic fatty liver disease/nonalcoholic steatohepatitis 2020. J. Gastroenterol. 2021, 56, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, S.; Datta, D.; Sinha, B. A meta-analysis of the effects of glucagon-like-peptide 1 receptor agonist (GLP1-RA) in nonalcoholic fatty liver disease (NAFLD) with type 2 diabetes (T2D). Sci. Rep. 2021, 11, 22063. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.H.; Lin, S.Y.; Chin, Y.H.; Lee, M.H.; Syn, N.; Goh, X.L.; Koh, J.H.; Quek, J.; Tan, D.J.H.; Mok, S.F.; et al. Antidiabetic Medications for Type 2 Diabetics with Nonalcoholic Fatty Liver Disease: Evidence from a Network Meta-Analysis of Randomized Controlled Trials. Endocr. Pract. 2021; in press. [Google Scholar] [CrossRef]
- Paik, J.M.; Golabi, P.; Younossi, Y.; Mishra, A.; Younossi, Z.M. Changes in the Global Burden of Chronic Liver Diseases from 2012 to 2017: The Growing Impact of NAFLD. Hepatology 2020, 72, 1605–1616. [Google Scholar] [CrossRef]
- Francque, S.M.; van der Graaff, D.; Kwanten, W. Non-alcoholic fatty liver disease and cardiovascular risk: Pathophysiological mechanisms and implications. J. Hepatol. 2016, 65, 425–443. [Google Scholar] [CrossRef] [PubMed]
- Kapuria, D.; Takyar, V.K.; Etzion, O.; Surana, P.; O’Keefe, J.H.; Koh, C. Association of Hepatic Steatosis with Subclinical Atherosclerosis: Systematic Review and Meta-Analysis. Hepatol. Commun. 2018, 2, 877–887. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, X.; Wu, S.; Fan, D.; Van Poucke, S.; Chen, Y.; Fu, S.; Zheng, M. Nonalcoholic fatty liver disease contributes to subclinical atherosclerosis: A systematic review and meta-analysis. Hepatol. Commun. 2018, 2, 376–392. [Google Scholar] [CrossRef]
- Mellinger, J.L.; Pencina, K.M.; Massaro, J.M.; Hoffmann, U.; Seshadri, S.; Fox, C.S.; O’Donnell, C.J.; Speliotes, E.K. Hepatic steatosis and cardiovascular disease outcomes: An analysis of the Framingham Heart Study. J. Hepatol. 2015, 63, 470–476. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; LaVine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef] [PubMed]
- Meyersohn, N.M.; Mayrhofer, T.; Corey, K.E.; Bittner, D.O.; Staziaki, P.V.; Szilveszter, B.; Hallett, T.; Lu, M.T.; Puchner, S.B.; Simon, T.G.; et al. Association of Hepatic Steatosis with Major Adverse Cardiovascular Events, Independent of Coronary Artery Disease. Clin. Gastroenterol. Hepatol. 2020, 19, 1480–1488.e14. [Google Scholar] [CrossRef] [PubMed]
- Simon, T.G.; Roelstraete, B.; Hagström, H.; Sundström, J.; Ludvigsson, J.F. Non-alcoholic fatty liver disease and incident major adverse cardiovascular events: Results from a nationwide histology cohort. Gut 2021, 163, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Ye, J.; Li, F.; Lin, Y.; Wu, T.; Wang, W.; Feng, S.; Zhong, B. Early Predictors of Cardiovascular Disease Risk in Nonalcoholic Fatty Liver Disease: Non-obese Versus Obese Patients. Dig. Dis. Sci. 2019, 65, 1850–1860. [Google Scholar] [CrossRef]
- Yoshitaka, H.; Hamaguchi, M.; Kojima, T.; Fukuda, T.; Ohbora, A.; Fukui, M. Nonoverweight nonalcoholic fatty liver disease and incident cardiovascular disease: A post hoc analysis of a cohort study. Medicine 2017, 96, e6712. [Google Scholar] [CrossRef]
- Mann, J.P.; Carter, P.; Armstrong, M.J.; Abdelaziz, H.K.; Uppal, H.; Patel, B.; Chandran, S.; More, R.; Newsome, P.; Potluri, R. Hospital admission with non-alcoholic fatty liver disease is associated with increased all-cause mortality independent of cardiovascular risk factors. PLoS ONE 2020, 15, e0241357. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Wu, F.; Ding, Y.; Hou, J.; Bi, J.; Zhang, Z. Association of non-alcoholic fatty liver disease with major adverse cardiovascular events: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 33386. [Google Scholar] [CrossRef]
- Stepanova, M.; Younossi, Z.M. Independent Association between Nonalcoholic Fatty Liver Disease and Cardiovascular Disease in the US Population. Clin. Gastroenterol. Hepatol. 2012, 10, 646–650. [Google Scholar] [CrossRef]
- Unalp-Arida, A.; Ruhl, C.E. Noninvasive fatty liver markers predict liver disease mortality in the U.S. population. Hepatology 2015, 63, 1170–1183. [Google Scholar] [CrossRef]
- Alexander, M.; Loomis, A.K.; Van Der Lei, J.; Duarte-Salles, T.; Prieto-Alhambra, D.; Ansell, D.; Pasqua, A.; Lapi, F.; Rijnbeek, P.; Mosseveld, M.; et al. Non-alcoholic fatty liver disease and risk of incident acute myocardial infarction and stroke: Findings from matched cohort study of 18 million European adults. BMJ 2019, 367, l5367. [Google Scholar] [CrossRef]
- Lee, H.; Lee, Y.-H.; Kim, S.U.; Kim, H.C. Metabolic Dysfunction-Associated Fatty Liver Disease and Incident Cardiovascular Disease Risk: A Nationwide Cohort Study. Clin. Gastroenterol. Hepatol. 2020, 19, 2138–2147.e10. [Google Scholar] [CrossRef]
- Zhou, Y.-Y.; Zhou, X.-D.; Wu, S.-J.; Hu, X.-Q.; Tang, B.; Van Poucke, S.; Pan, X.-Y.; Wu, W.-J.; Gu, X.-M.; Fu, S.-W.; et al. Synergistic increase in cardiovascular risk in diabetes mellitus with nonalcoholic fatty liver disease: A meta-analysis. Eur. J. Gastroenterol. Hepatol. 2018, 30, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Mu, B.; Guan, Y.; Liu, X.; Zhao, N.; Pan, D.; Wang, S. Assessment of the relationship between non-alcoholic fatty liver disease and diabetic complications. J. Diabetes Investig. 2016, 7, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.K.; Jain, V.; Singla, S.; Baruah, B.P.; Arya, V.; Yadav, R.; Singh, V.P. Prevalence of non-alcoholic fatty liver disease and its correlation with coronary risk factors in patients with type 2 diabetes. J. Assoc. Physicians India 2011, 59, 351–354. [Google Scholar]
- Idilman, I.S.; Akata, D.; Hazirolan, T.; Erdogan, B.D.; Aytemir, K.; Karcaaltincaba, M. Nonalcoholic fatty liver disease is associated with significant coronary artery disease in type 2 diabetic patients: A computed tomography angiography study 2. J. Diabetes 2015, 7, 279–286. [Google Scholar] [CrossRef]
- Guo, K.; Zhang, L.; Lu, J.; Yu, H.; Wu, M.; Bao, Y.; Chen, H.; Jia, W. Non-alcoholic fatty liver disease is associated with late but not early atherosclerotic lesions in Chinese inpatients with type 2 diabetes. J. Diabetes Its Complicat. 2016, 31, 80–85. [Google Scholar] [CrossRef]
- Kim, S.-K.; Choi, Y.J.; Huh, B.W.; Park, S.W.; Lee, E.J.; Cho, Y.-W.; Huh, K.B. Nonalcoholic Fatty Liver Disease Is Associated with Increased Carotid Intima-Media Thickness only in Type 2 Diabetic Subjects with Insulin Resistance. J. Clin. Endocrinol. Metab. 2014, 99, 1879–1884. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Li, X.; Wang, C.; Wang, J.; Wang, F.; Ma, L.; You, W.; Li, C. Association between non-alcoholic fatty liver disease and peripheral artery disease in patients with type 2 diabetes. Intern. Med. J. 2017, 47, 1147–1153. [Google Scholar] [CrossRef]
- Afarideh, M.; Aryan, Z.; Ghajar, A.; Ganji, M.; Ghaemi, F.; Saadat, M.; Heidari, B.; Mechanick, J.I.; Esteghamati, A. Association of non-alcoholic fatty liver disease with microvascular complications of type 2 diabetes. Prim. Care Diabetes 2019, 13, 505–514. [Google Scholar] [CrossRef]
- Targher, G.; Bertolini, L.; Rodella, S.; Zoppini, G.; Lippi, G.; Day, C.; Muggeo, M. Non-alcoholic fatty liver disease is independently associated with an increased prevalence of chronic kidney disease and proliferative/laser-treated retinopathy in type 2 diabetic patients. Diabetologia 2007, 51, 444–450. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, S.; Li, B.; Shao, X.; Liu, X.; Liu, A.; Wu, B.; Zhang, Y.; Wang, H.; Wang, X.; et al. Association between non-alcoholic fatty liver disease and chronic kidney disease in population with prediabetes or diabetes. Int. Urol. Nephrol. 2014, 46, 1785–1791. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; Tabibian, J.H.; Ekstedt, M.; Kechagias, S.; Hamaguchi, M.; Hultcrantz, R.; Hagström, H.; Yoon, S.K.; Charatcharoenwitthaya, P.; et al. Association of Non-alcoholic Fatty Liver Disease with Chronic Kidney Disease: A Systematic Review and Meta-analysis. PLoS Med. 2014, 11, e1001680. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-Y.; Chen, Y.-J.; Chen, W.-L.; Peng, T.-C. The Relationship between Nonalcoholic Fatty Liver Disease and Retinopathy in NHANES III. PLoS ONE 2016, 11, e0165970. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Chonchol, M.; Bertolini, L.; Rodella, S.; Zenari, L.; Lippi, G.; Franchini, M.; Zoppini, G.; Muggeo, M. Increased Risk of CKD among Type 2 Diabetics with Nonalcoholic Fatty Liver Disease. J. Am. Soc. Nephrol. 2008, 19, 1564–1570. [Google Scholar] [CrossRef]
- Koulaouzidis, G.; Charisopoulou, D.; Kukla, M.; Marlicz, W.; Rydzewska, G.; Koulaouzidis, A.; Skonieczna-Żydecka, K. Association of non-alcoholic fatty liver disease with coronary artery calcification progression: A systematic review and meta-analysis. Przegla̜d Gastroenterol. 2021, 16, 196–206. [Google Scholar] [CrossRef]
- Salvatore, T.; Pafundi, P.C.; Galiero, R.; Albanese, G.; Di Martino, A.; Caturano, A.; Vetrano, E.; Rinaldi, L.; Sasso, F.C. The Diabetic Cardiomyopathy: The Contributing Pathophysiological Mechanisms. Front. Med. 2021, 8, 695792. [Google Scholar] [CrossRef]
- Galiero, R.; Caturano, A.; Vetrano, E.; Cesaro, A.; Rinaldi, L.; Salvatore, T.; Marfella, R.; Sardu, C.; Moscarella, E.; Gragnano, F.; et al. Pathophysiological mechanisms and clinical evidence of relationship between Nonalcoholic fatty liver disease (NAFLD) and cardiovascular disease. Rev. Cardiovasc. Med. 2021, 22, 755–768. [Google Scholar] [CrossRef]
- Targher, G.; Byrne, C.D.; Lonardo, A.; Zoppini, G.; Barbui, C. Non-alcoholic fatty liver disease and risk of incident cardiovascular disease: A meta-analysis. J. Hepatol. 2016, 65, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-H.; Cho, Y.; Choi, Y.J.; Huh, B.W.; Lee, B.-W.; Kang, E.S.; Park, S.W.; Cha, B.-S.; Lee, E.J.; Lee, Y.-H.; et al. Non-alcoholic steatohepatitis and progression of carotid atherosclerosis in patients with type 2 diabetes: A Korean cohort study. Cardiovasc. Diabetol. 2020, 19, 81. [Google Scholar] [CrossRef] [PubMed]
- Greco, C.; Nascimbeni, F.; Carubbi, F.; Andreone, P.; Simoni, M.; Santi, D. Association of Nonalcoholic Fatty Liver Disease (NAFLD) with Peripheral Diabetic Polyneuropathy: A Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 4466. [Google Scholar] [CrossRef]
- Santos, R.D.; Valenti, L.; Romeo, S. Does nonalcoholic fatty liver disease cause cardiovascular disease? Current knowledge and gaps. Atherosclerosis 2019, 282, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.D.; Targher, G. Non-alcoholic fatty liver disease is a risk factor for cardiovascular and cardiac diseases: Further evidence that a holistic approach to treatment is needed. Gut 2021. [Google Scholar] [CrossRef]
- Lauridsen, B.K.; Stender, S.; Kristensen, T.S.; Kofoed, K.; Køber, L.; Nordestgaard, B.G.; Tybjærg-Hansen, A. Liver fat content, non-alcoholic fatty liver disease, and ischaemic heart disease: Mendelian randomization and meta-analysis of 279,013 individuals. Eur. Heart J. 2017, 39, 385–393. [Google Scholar] [CrossRef] [PubMed]
- El-Agroudy, N.N.; Kurzbach, A.; Rodionov, R.N.; O’Sullivan, J.; Roden, M.; Birkenfeld, A.L.; Pesta, D.H. Are Lifestyle Therapies Effective for NAFLD Treatment? Trends Endocrinol. Metab. 2019, 30, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, J.R.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
| Authors (Year) | Study Type | Studies/Participants (N) | Average Duration of Follow-Up | Population | Findings |
|---|---|---|---|---|---|
| Zelber-Sagi et al. (2007) [36] | Cross-sectional | 1/349 | - | Patients with or without NAFLD | The group with diagnosed NAFLD consumed almost twice the amount of SSB. Intake of SSB was significantly associated with an increased risk for NAFLD. |
| Assy et al. (2008) [37] | Cross-sectional | 1/61 | 36 months | Patients with NAFLD and healthy control group | 80% of patients with NAFLD consumed an excessive amount of SSB (more than 50 g/day of added sugar) compared with 20% in healthy controls. SSB consumption was the only independent variable that was able to predict the presence of NAFLD. |
| Abid et al. (2009) [38] | Prospective | 1/90 | 6 months | Patients with NAFLD and healthy control group | 80% of patients with NAFLD had an excessive intake of SSB (>500 cm3/day) compared to 17% of healthy controls. Logistic regression analysis showed that SSB consumption is a strong predictor of NAFLD independent of metabolic syndrome and CRP level. |
| Abdelmalek et al. (2010) [39] | Cross-sectional | 1/427 | 3 months | Patients with NAFLD with none, minimum to moderate, and daily SSB and fruit juices consumption | Increased fructose consumption was associated with hypertriglyceridemia, low HDL-c levels. Daily fructose consumption was associated with lower steatosis grade and higher fibrosis stage. In older adults (age > or = 48 years), daily fructose consumption was associated with increased hepatic inflammation and hepatocyte ballooning. |
| Maersk et al. (2012) [40] | RCT | 1/47 | 6 months | Overweight patients for 6 months consuming water, milk, diet cola, and regular cola (SSSD) | Milk and diet cola reduced systolic blood pressure by 10–15% compared with regular cola. Daily intake of SSSDs increased accumulation of: liver fat, skeletal muscle fat, visceral fat, blood triglycerides, and total cholesterol, compared with milk, diet cola, and water. |
| Chiu et al. (2014) [41] | Meta-analysis, systematic review of controlled trials | 13/260 | More than 7 days | Healthy participants | There was no effect of fructose in isocaloric trials. Increased consumption of fructose in hypercaloric trials increased both IHCL and ALT. |
| Chung et al. (2014) [42] | Meta-analysis, systematic review of observational and interventional studies | 27/1670 | 6 days to 6 months | Patients with or without NAFLD | Observational studies were rated insufficient because of the high risk of biases and inconsistent study findings. Hypercaloric fructose diet (supplemented by pure fructose) increased liver fat and AST concentrations in healthy men compared with the consumption of a weight-maintenance diet with a low level of evidence. Hypercaloric fructose and glucose diets have similar effects on liver fat and liver enzymes in healthy adults, also with a low level of evidence. |
| Hochuli et al. (2014) [43] | Randomized crossover | 1/34 | 3 weeks | Healthy young men with medium fructose, high fructose, high sucrose, and high glucose consumption for 3 weeks | Fatty acid synthesis was increased after high fructose consumption and medium fructose consumption compared with high sucrose consumption, high glucose consumption, or baseline. Fasting palmitoylcarnitine was significantly increased after high fructose and high sucrose consumption. |
| Jin et al. (2014) [44] | RCT | 1/24 | 4 weeks | Overweight patients with average self-reported consumption of at least 3 servings of SSB or fruit juice divided into 2 groups: consuming glucose only beverages and fructose only beverages | There was no significant change in hepatic fat or body weight in the group consuming glucose only or fructose only beverages. In the glucose beverage group, there was significantly improved adipose insulin sensitivity, CRP, and LDL-c oxidation. |
| Ma et al. (2015) [45] | Cross-sectional | 1/2634 | 3 years | Patients consuming SSB vs. patients who did not consume SSB | Increased incidence of NAFLD was observed in patients with daily consumption of SSB. SSB consumption was positively associated with increased ALT levels. |
| Schwarz et al. (2015) [29] | Prospective | 1/8 | 9 days | Healthy men on weight-maintaining diets: high in fructose vs. isocaloric diet with complex carbohydrate substituted for fructose | Participants’ weight remained stable. A high fructose diet was associated with higher DNL and higher liver fat in all participants. |
| Wijarnpreecha et al. (2015) [46] | Meta-analysis, systematic review of cross-sectional and cohort studies | 7/4639 | 6 months to 7 years | Patients consuming a significant amount of either sugar or SSB vs. patients who did not consume SSB | Patients consuming a significant amount of either sugar or SSB have an increased risk of NAFLD. |
| Chen et al. (2019) [47] | Meta-analysis, systematic review of cross-sectional, case-control and cohort studies | 12/35,705 | - | Patients consuming low, middle, and high doses of SSB | Consumption of SSB was associated with an increased risk of NAFLD. Consumption of SSB had a dose-dependent effect on the risk of NAFLD. |
| Authors (Year) | Study Type | Studies/Participants (N) | Average Duration of Follow-Up | Population | Findings |
|---|---|---|---|---|---|
| Raman et al. (2013) [65] | Observational case-control | 1/60 | - | Obese patients with NAFLD vs. healthy control | In the fecal microbiome of NAFLD patients, there was an over-representation of Lactobacillus species and selected members of phylum Firmicutes and increased fecal ester VOC. |
| Wang et al. (2016) [61] | Cross-sectional | 1/126 | - | Non-obese patients with NAFLD vs. healthy control | In non-obese patients with NAFLD, there was lower diversity and a phylum-level change in microbiota compared to healthy control. NAFLD patients had 20% more phylum Bacteroidetes and 24% fewer Firmicutes compared to healthy control. |
| Shen et al. (2017) [62] | Cross-sectional | 1/47 | - | Patients with NAFLD vs. healthy control | NAFLD patients had lower gut microbiota diversity than healthy control. In stools of patients with NASH, there were decreased levels of Prevotella, increased levels of Blautia, Lachnospiraceae, Escherichia, Shigella, and Enterobacteriacae compared to patients with NAFLD. |
| Da Silva et al. (2018) [66] | Cross-sectional | 1/67 | 7 days | Patients with NAFLD, NASH vs. healthy control | In stools of NAFLD patients, Firmicutes, Bacteroidetes were less abundant and Lactobacillaceae more abundant compared to healthy control. NAFLD patients had higher concentrations of fecal propionate and isobutyric acid and serum 2-hydroxybutyrate and L-lactic acid. |
| Lanthier et al. (2021) [64] | Prospective | 1/52 | 3 months | Obese adults with NAFLD: patients with severe liver steatosis vs. patients with fibrosis | Abundance of fecal Clostridium was significantly decreased with the presence of liver fibrosis and was negatively associated with liver stiffness measurement. Escherichia and Shigella increased fecal abundance was observed in patients with fibrosis compared to patients with severe steatosis without fibrosis. |
| Li et al. (2021) [63] | Meta-analysis, systematic review | 15/1265 | - | Adults with NAFLD vs. healthy control group | Stools of patients with NAFLD exhibited an increased abundance of Escherichia, Prevotella, Streptococcus and exhibited a decreased abundance of Coprococcus, Faecalibacterium and Ruminococcus. No significant difference in the abundance of Bacteroides, Bifidobacterium, Blautia, Clostridium, Dorea, Lactobacillus, Parabacteroides, or Roseburia. |
| Authors (Year) | Study Type | Studies/Participants (N) | Duration of Treatment | Population | Findings |
|---|---|---|---|---|---|
| Ma et al. (2013) [68] | Meta-analysis of RCT (probiotic vs. placebo) | 4/132 | 8–24 weeks | Adults with NAFLD | Probiotic therapy was associated with a significant decrease in levels of ALT, AST, total cholesterol, HDL-c, TNF-α, and HOMA-IR. |
| Eslamparast et al. (2014) [78] | RCT (synbiotic vs. placebo) | 1/52 | 28 weeks | Adults with NAFLD | In patients with NAFLD using synbiotic compared to the placebo group, significantly decreased levels of ALT, AST, GGT, CRP, TNF-α, and fibrosis scores were observed. |
| Loman et al. (2018) [69] | Meta-analysis of RCT (probiotic or synbiotic or prebiotic vs. placebo) | 25/1309 | 2–28 weeks | Adults with NAFLD | Probiotic/synbiotic/prebiotic therapies were associated with significantly reduced levels of ALT, AST, GGT, total cholesterol, LDL-c, and TAG, but no significant difference in TNF-α and CRP levels. |
| Bakhshimoghaddam et al. (2018) [77] | RCT (synbiotic vs. control group) | 1/102 | 24 weeks | Adults with NAFLD | Grades of NAFLD determined in ultrasound examination significantly decreased in patients with NAFLD consuming synbiotic, compared to conventional and control groups. |
| Khan et al. (2019) [70] | Meta-analysis, systematic review of RCT (probiotic or synbiotic vs. placebo) | 12/624 | 8–24 weeks | Adults with NAFLD | Probiotic/synbiotic therapies were associated with a significant reduction in levels of ALT, AST, CRP, and significant improvement in liver fibrosis score. |
| Sharpton et al. (2019) [71] | Meta-analysis of RCT (probiotic or synbiotic vs. placebo) | 21/1252 | 8–28 weeks | Adults with NAFLD | Probiotic/synbiotic therapies were associated with a significant reduction in levels of ALT and LSM. Usage of probiotics/synbiotics was associated with increased odds of improvement in hepatic steatosis. |
| Craven et al. (2020) [81] | RCT (allogenic FMT vs. autologous FMT) | 1/21 | 6 months | Adults with NAFLD | There were no significant differences between patients with NAFLD after allogenic FMT and autologous FMT in HOMA-IR or hepatic PDFF. Allogenic FMT in patients with NAFLD with elevated small intestine permeability at baseline caused a significant reduction of small intestine permeability 6 weeks after allogenic FMT. |
| Pan et al. (2020) [72] | Meta-analysis, systematic review of RCT (probiotic vs. placebo) | 19/954 | Adults with NAFLD | Probiotic supplementation significantly decreased TNF-α and CRP levels. | |
| Witjes et al. (2020) [82] | RCT (allogenic FMT vs. autologous FMT) | 1/21 | 24 weeks | Adults with NAFLD | Allogenic FMT was associated with modified gut microbiota composition (increased abundance of ruminococcus, eubacterium hallii, faecalibacterium, and prevotella copri), decreased levels of GGT, a trend toward improvement in the necro-inflammation score (consisting of both lobular inflammation and hepatocellular ballooning). There was no significant difference in fibrosis score after allogenic FMT. |
| Authors (Year) | Study Type | Studies/Participants (N) | Average Duration of Follow-Up | Population | Findings |
|---|---|---|---|---|---|
| Lalukka et al. (2016) [85] | Meta-analysis of systematic review, prospective studies | 20/122,517 | 2–20 years | Adults with NAFLD without T2DM | NAFLD predicted the risk of T2DM in all studies. NAFLD predicted the risk of T2DM in all studies with NAFLD diagnosis based on ultrasonography independently of age. NAFLD predicted the risk of T2DM in 12 of 14 studies with NAFLD diagnosis based on liver function tests independently of age or BMI. |
| Cusi et al. (2017) [93] | Observational | 1/204 | - | Adults with T1DM and T2DM with or without NAFLD | The prevalence of NAFLD in T1DM patients was low (8.8%) but high in T2DM patients not treated with insulin (75.6%) and treated with insulin (61.7%). |
| Mantovani et al. (2018) [84] | Meta-analysis of observational studies | 19/296,439 | at least 5 years | Adults with NAFLD, without T2DM | Patients with NAFLD had a greater risk of T2DM incidence. Patients with advanced NAFLD with fibrosis had an even greater risk of T2DM incidence. |
| Cho et al. (2019) [94] | Cohort | 1/2726 | 12–135 months | Adults with or without NAFLD or T2DM | Incident and persistent NAFLD increased risk of T2DM development. |
| Lee et al. (2019) [86] | Cohort | 1/6240 | 4.30 ± 1.91 years | Adults with prediabetes with or without NAFLD from Korea | The prevalence of NAFLD was 45.4%. During follow-up, the incidence of T2DM was 8.1%. Subjects with prediabetes and NAFLD had a higher prevalence of T2DM. |
| Younossi et al. (2019) [14] | Meta-analysis, systematic review of cross-sectional and longitudinal studies | 80/49,419 | median 3 years | Adults with T2DM with or without NAFLD and NASH | The global prevalence of NAFLD was 55.5%. The highest prevalence of NAFLD reported in studies from Europe was 68%. The global prevalence of NASH was 37.3%. The prevalence of advanced fibrosis in patients with NAFLD and T2DM was 17%. |
| Mantovani et al. (2021) [95] | Meta-analysis of prospective studies | 33/501,022 | at least 1 year | Adults with NAFLD | Patients with NAFLD had a higher risk of incident DM. The risk increased across the severity of NAFLD. |
| Authors (Year) | Study Type | Studies/Participants (N) | Duration of Treatment | Population | Findings |
|---|---|---|---|---|---|
| Boettcher et al. (2012) [102] | Meta-analysis of RCT (pioglitazone vs. placebo) | 4/334 | 24–96 weeks | T2DM patients with NASH | Pioglitazone treatment was associated with histological improvement of ballooning degeneration, lobular inflammation, and steatosis compared to placebo. |
| Eguchi et al. (2015) [114] | Prospective (liraglutide vs. lifestyle modification) | 1/26 | 24 weeks | Adults with NASH, BMI ≥ 25 kg/m2, with or without T2DM | Liraglutide treatment improved histological features of steatohepatitis and fibrosis in 80% of patients and aminotransferase levels in 78.9% of patients. |
| Rizvi et al. (2015) [115] | Prospective (liraglutide and metformin therapy vs. metformin therapy) | 1/58 | 8 months | Two groups of T2DM patients with or without NAFLD | Carotid IMT decreased significantly in T2DM patients with NAFLD but not in T2DM patients without NAFLD. |
| Armstrong et al. (2016) [116] | RCT (liraglutide vs. placebo) | 1/52 | 48 weeks | Adults with NASH, BMI ≥ 25 kg/m2, with or without T2DM | Treatment with liraglutide was associated with histological improvement of steatohepatitis. |
| Armstrong et al. (2016) [117] | RCT (liraglutide vs. placebo) | 1/14 | 12 weeks | Adults with NASH, BMI ≥ 25 kg/m2, with or without T2DM | Liraglutide treatment was associated with significant reduction of ALT, increased hepatic insulin sensitivity, suppression of hepatic endogenous glucose production with low-dose insulin, a decrease of hepatic de novo lipogenesis. |
| Cusi et al. (2016) [103] | RCT (pioglitazone vs. placebo) | 1/101 | 18 months | Patients with prediabetes or T2DM and NASH | Pioglitazone treatment was associated with histological improvement of fibrosis score, reduced hepatic triglyceride content and improved insulin sensitivity of adipose tissue, liver, and muscles. |
| Feng et al. (2017) [109] | RCT (liraglutide vs. metformin and gliclazide) | 1/85 | 24 weeks | T2DM patients with NAFLD | Liraglutide or metformin monotherapy was associated with greater weight loss, reduction in body fat mass, and improved glucose control compared to gliclazide. Weight loss, fat mass, and waist reduction affected favorably hepatic function IHF decreased significantly after liraglutide. |
| Seko et al. (2017) [110] | Retrospective study (all patients dulaglutide) | 1/15 | 12 weeks | T2DM patients with biopsy-proven NAFLD | Dulaglutide treatment was associated with significantly decreased BMI, ALT, AST, HbA1c levels. |
| Cusi et al. (2018) [110] | A post hoc analysis of AWARD program (dulaglutide vs. placebo) | 4/1499 | 6 months | T2DM patients with NAFLD | Dulaglutide treatment was associated with a significant decrease in ALT, AST, and GGT consistent with liver fat reduction. |
| Kuchay et al. (2018) [108] | RCT (empaglifozin vs. standard treatment) | 1/50 | 20 weeks | T2DM patients with NAFLD | Empagliflozin treatment was associated with significant liver fat reduction and ALT activity improvement compared to the control group. |
| Shibuya et al. (2018) [105] | RCT (luseogliflozin vs. metformin) | 1/32 | 24 weeks | T2DM patients with NAFLD | Luseogliflozin was associated with significantly greater liver fat reduction than metformin and a significantly greater decrease in VAT and BMI. |
| Shimizu et al. (2019) [109] | RCT (dapagliflozin vs. control group) | 1/57 | 24 weeks | T2DM patients with NAFLD | Dapagliflozin treatment was associated with a significant decrease in CAP and with a greater significant decrease in ALT and VAT. |
| Aso et al. (2019) [106] | RCT (dapagliflozin vs. control group) | 1/57 | 24 weeks | T2DM patients with NAFLD | Dapagliflozin was associated with a significant decrease in VAT, SAT, ALT, AST, and GGT. |
| Yan et al. (2019) [120] | RCT (liraglutide vs. sitagliptin vs. insulin glargine) | 1/75 | 26 weeks | T2DM patients with NAFLD under inadequate glycemic control by metformin | Liraglutide treatment was associated with a significant decrease in MRI-PDFF, VAT, SAT, and body weight. Sitagliptin treatment was associated with a significant decrease in MRI-PDFF, VAT, and body weight. |
| Hartman et al. (2020) [123] | RCT (tirzepatide vs. dulaglutide vs. placebo) | 1/316 | 26 weeks | T2DM patients with NASH and fibrosis | Tirzepatide treatment was associated with a greater decrease in ALT level than dulaglutide treatment. Adiponectin level increased significantly compared to placebo, but not with dulaglutide therapy. |
| Kuchay et al. (2020) [121] | RCT (dulaglutide vs. control group) | 1/88 | 24 weeks | T2DM patients with NAFLD | Dulaglutide treatment was associated with a 2.6-fold reduction of LFC and reduction of GGT levels. |
| Lai et al. (2020) [107] | Prospective, pilot study (empagliflozin vs. placebo) | 1/39 | 24 weeks | T2DM patients with or without NASH | Empagliflozin treatment improved steatosis, ballooning, and fibrosis. |
| Ghosal et al. (2021) [125] | Meta-analysis, systematic review of RCT (GLP-1 RA vs. placebo) | 8/615 | 12–72 weeks | T2DM patients with NAFLD | GLP-1 RA significantly reduced ALT, AST, GGT levels, LFC, HbA1c levels, and body weight. GLP-1 RA caused significant improvement of NAFLD in biopsy. |
| Lee et al. (2021) [122] | Meta-analysis, systematic review of RCT (canagliflozin or dapagliflozin vs. placebo) | 8/5984 | 12–18 weeks | T2DM patients with NAFLD | Canagliflozin significantly reduced GGT levels. Dapagliflozin significantly reduced HbA1c levels and HOMA-IR. |
| Lian et al. (2021) [104] | Meta-analysis of RCT (metformin or liraglutide or pioglitazone vs. placebo) | 26/?? | 12–96 weeks | Patients with NAFLD and with or without T2DM | Pioglitazone had a significant effect on the levels of ALT and AST but was also associated with an increased risk of weight gain and increased BMI. Liraglutide and metformin had significant effects on reducing ALT and AST. |
| Mantovani et al. (2021) [111] | Meta-analysis of RCT (liraglutide or semaglutide vs. placebo) | 22/936 | median 26 weeks | Overweight or obese patients with NASH or NAFLD with or without T2DM | Treatment with GLP-1 RA decreased LFC measured by MRI, decreased ALT, GGT, but not AST levels, and greater histological resolution without worsening of liver fibrosis. |
| Newsome et al. (2021) [112] | RCT (semaglutide vs. placebo) | 1/320 | 72 weeks | Patients with NASH and biopsy confirmed fibrosis | After semaglutide treatment, NASH resolution was achieved in 36–59% of patients with improvement in fibrosis stage in 43% of them. |
| Ng et al. (2021) [126] | Meta-analysis of RCT (PPARγ agonists or SGLT-2i vs. placebo) | 14/?? | - | T2DM patients with NAFLD | PPARγ agonists and SGLT-2i significantly reduced steatosis. SGLT-2i resulted in a significantly greater reduction of fibrosis compared to PPARγ. |
| Song et al. (2021) [13] | Meta-analysis of RCT (liraglutide vs. pioglitazone vs. insulin vs. placebo) | 11/535 | 12–24 weeks | T2DM patients with NAFLD | Liraglutide decreased LFC, BMI, HDL-c, LDL-c, HbA1c, TC, and TAG. |
| Authors (Year) | Study Type | Studies/Participants (N) | Average Duration of Follow-Up | Population | Findings |
|---|---|---|---|---|---|
| Targher et al. (2008) [151] | Cross-sectional | 1/2103 | - | T2DM patients with or without CKD | NAFLD was associated with increased rates of CKD and proliferative/laser-treated retinopathy. |
| Agarwal et al. (2011) [145] | Retrospective | 1/124 | - | T2DM adults with or without NAFLD | The prevalence of NAFLD in T2DM patients was 57.2%. In T2DM patients with NAFLD, CAD was more prevalent compared to T2DM patients without NAFLD. |
| Stepanowa et al. (2012) [139] | Prospective | 1/11,613 | 14 years | Adults with or without NAFLD | NAFLD was associated with a higher risk of CVD. NAFLD was not significantly associated with higher CVD mortality. |
| Idilman et al. (2014) [146] | Observational | 1/273 | - | T2DM adults without previous known liver disease | In T2DM patients, NAFLD was associated with significant CAD (≥50 stenosis in CTA). |
| Kim et al. (2014) [148] | Observational | 1/4437 | - | T2DM patients with or without NAFLD | The prevalence of NAFLD in T2DM patients was 72.7%. Carotid IMT was significantly higher in T2DM patients with NAFLD and insulin resistance compared to insulin-sensitive T2DM patients without NAFLD and insulin-sensitive T2DM patients with NAFLD. |
| Li et al. (2014) [152] | Cross-sectional | 1/190 | - | Adults with diabetes and prediabetes with or without NAFLD | Patients with NAFLD had a higher albumin-to-creatinine ratio. CKD had a higher prevalence in T2DM patients with NAFLD. |
| Musso et al.(2014) [153] | Meta-analysis of cross-sectional, longitudinal studies | 20/29,282 | - | T2DM patients with or without NAFLD | NAFLD was associated with an increased risk of prevalent and incident CKD. NASH was associated with a higher prevalence and incidence of CKD than simple steatosis. Advanced fibrosis was associated with a higher prevalence and incidence of CKD than non-advanced fibrosis. |
| Mellinger et al. (2015) [131] | Prospective cohort | 1/3014 | 3 years | Adults with or without NAFLD | There was no significant association between NAFLD and CVD. NAFLD was associated with CAC and AAC. |
| Lin et al. (2016) [154] | Cross-sectional | 1/5963 | - | Adults with NAFLD with or without T2DM | NAFLD was not significantly associated with retinopathy in T2DM patients. |
| Targher et al. (2016) [159] | Meta-analysis of prospective, retrospective, and observational studies | 16/34,043 | median period 6.9 years | Adults with or without NAFLD | Patients with NAFLD had a higher risk of MACE than patients without NAFLD. |
| Unalp-Arida et al. (2016) [140] | Retrospective cohort | 1/12,216 | 6 years | Adults with or without NAFLD | NAFLD was not independently associated with mortality from all causes, including CVD, cancer, or diabetes. |
| Wu et al. (2016) [138] | Meta-analysis, systematic review of cross-sectional and cohort studies | 34/164,494 | 1.6–26.4 years | Adults with or without NAFLD | NAFLD was associated with increased risk of prevalent and incident CVD, prevalent atherosclerosis, prevalent and incident hypertension, prevalent and incident CAD. NAFLD was not associated with overall and CVD mortality. |
| Yan et al. (2016) [144] | Observational, retrospective | 1/212 | - | T2DM patients with or without NAFLD | Patients with NAFLD diagnosed earlier than T2DM had a significantly higher prevalence of CAD and hypertension and lower prevalence of diabetic retinopathy and diabetic peripheral neuropathy compared to T2DM patients with NAFLD diagnosed later than T2DM and T2DM patients without NAFLD. There was no significant difference in the prevalence of diabetic kidney disease. |
| Zou et al. (2016) [149] | Cross-sectional | 1/2646 | - | T2DM patients ≥ 40 years old with or without NAFLD | T2DM patients with NAFLD had a significantly higher prevalence of PAD compared with those without NAFLD. The prevalence of NAFLD among T2DM patients was 10.3%. NAFLD was associated with an increased risk of PAD. |
| Guo et al. (2017) [147] | Cross-sectional | 1/8571 | - | T2DM patients with or without NAFLD | The prevalence of carotid and lower limb plaque, as well as carotid and lower limb stenosis, was significantly higher in T2DM patients with NAFLD than in T2DM patients without NAFLD. There was no significant difference between T2DM patients with or without NAFLD in carotid IMT. |
| Yoshitaka et al. (2017) [136] | Cohort | 1/1674 | 6 years | Overweight and non-overweight patients with or without NAFLD | NAFLD was associated with a higher risk of CVD incidents in non-overweight patients with NAFLD. |
| Kapuria et al. (2018) [129] | Meta-analysis, systematic review of cross-sectional and cohort studies | 12/42,410 | - | Adults with or without NAFLD | NAFLD was associated with a higher CAC score compared to adults without NAFLD. |
| Zhou et al. (2018) [130] | Meta-analysis, systematic review of cross-sectional, case-control, and cohort studies | 26/83,395 | - | Adults with or without NAFLD | NAFLD was associated with a higher risk of increased carotid IMT, arterial stiffness, coronary artery calcification, and endothelial disfunction. |
| Zhou et al. (2018) [143] | Meta-analysis of cross-sectional, cohort studies | 11/8346 | At least 5 years | T1DM and T2DM adults with or without NAFLD | T2DM patients with diagnosed NAFLD had a 2 times higher risk for CVD compared with patients without NAFLD. |
| Afarideh et al. (2019) [150] | Case-control | 1/935 | - | T2DM patients with at least one microvascular complication vs. T2DM patients control group | Diabetic retinopathy and DKD were inversely associated with the presence of NAFLD. The subgroup of NAFLD with elevated liver enzymes had lower odds of having diabetic peripheral neuropathy. |
| Alexander et al. (2019) [141] | Cohort | 1/120,795 | mean 2.1–5.5 years | Adults with NAFLD | After adjustment for established cardiovascular risk factors, NAFLD was not associated with AMI or stroke risk. |
| Lee et al. (2020) [160] | Cohort | 1/1120 | 6–8 years | T2DM patients with or without NAFLD | NAFLD was significantly associated with atherosclerosis progression. |
| Mann et al. (2020) [137] | Retrospective cohort | 1/26,539 | 14 years after discharge | Patients with or without NAFLD | Patients with NAFLD without cirrhosis and NAFLD with cirrhosis had higher mortality compared to controls. |
| Shao et al. (2020) [135] | Cross-sectional | 1/543 | - | Obese patients with NAFLD vs. non-obese patients with NAFLD | Predictive factors of subclinical atherosclerosis in all patients with NAFLD were age increased per 10 years and liver stiffness. LFC was an additional predictor in obese patients with NAFLD. |
| Greco et al. (2021) [161] | Meta-analysis, systematic review of cross-sectional studies | 13/9614 | - | T1DM and T2DM patients with or without NAFLD | Diabetic peripheral neuropathy prevalence was significantly higher in T2DM patients with NAFLD compared to T2DM patients without NAFLD, but not in T1DM patients with NAFLD. |
| Meyersohn et al. (2021) [133] | Cohort | 1/3756 | 25 months | Symptomatic patients without previous diagnosed CAD | NAFLD was associated with MACE independently of other cardiovascular risk factors or extent of CAD. |
| Lee et al. (2021) [142] | Cohort | 1/8,962,813 | median 10.1 years | Adults with or without NAFLD | NAFLD and MAFLD were associated with significantly higher risk for CVD events. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drożdż, K.; Nabrdalik, K.; Hajzler, W.; Kwiendacz, H.; Gumprecht, J.; Lip, G.Y.H. Metabolic-Associated Fatty Liver Disease (MAFLD), Diabetes, and Cardiovascular Disease: Associations with Fructose Metabolism and Gut Microbiota. Nutrients 2022, 14, 103. https://doi.org/10.3390/nu14010103
Drożdż K, Nabrdalik K, Hajzler W, Kwiendacz H, Gumprecht J, Lip GYH. Metabolic-Associated Fatty Liver Disease (MAFLD), Diabetes, and Cardiovascular Disease: Associations with Fructose Metabolism and Gut Microbiota. Nutrients. 2022; 14(1):103. https://doi.org/10.3390/nu14010103
Chicago/Turabian StyleDrożdż, Karolina, Katarzyna Nabrdalik, Weronika Hajzler, Hanna Kwiendacz, Janusz Gumprecht, and Gregory Y. H. Lip. 2022. "Metabolic-Associated Fatty Liver Disease (MAFLD), Diabetes, and Cardiovascular Disease: Associations with Fructose Metabolism and Gut Microbiota" Nutrients 14, no. 1: 103. https://doi.org/10.3390/nu14010103
APA StyleDrożdż, K., Nabrdalik, K., Hajzler, W., Kwiendacz, H., Gumprecht, J., & Lip, G. Y. H. (2022). Metabolic-Associated Fatty Liver Disease (MAFLD), Diabetes, and Cardiovascular Disease: Associations with Fructose Metabolism and Gut Microbiota. Nutrients, 14(1), 103. https://doi.org/10.3390/nu14010103








