Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease
Abstract
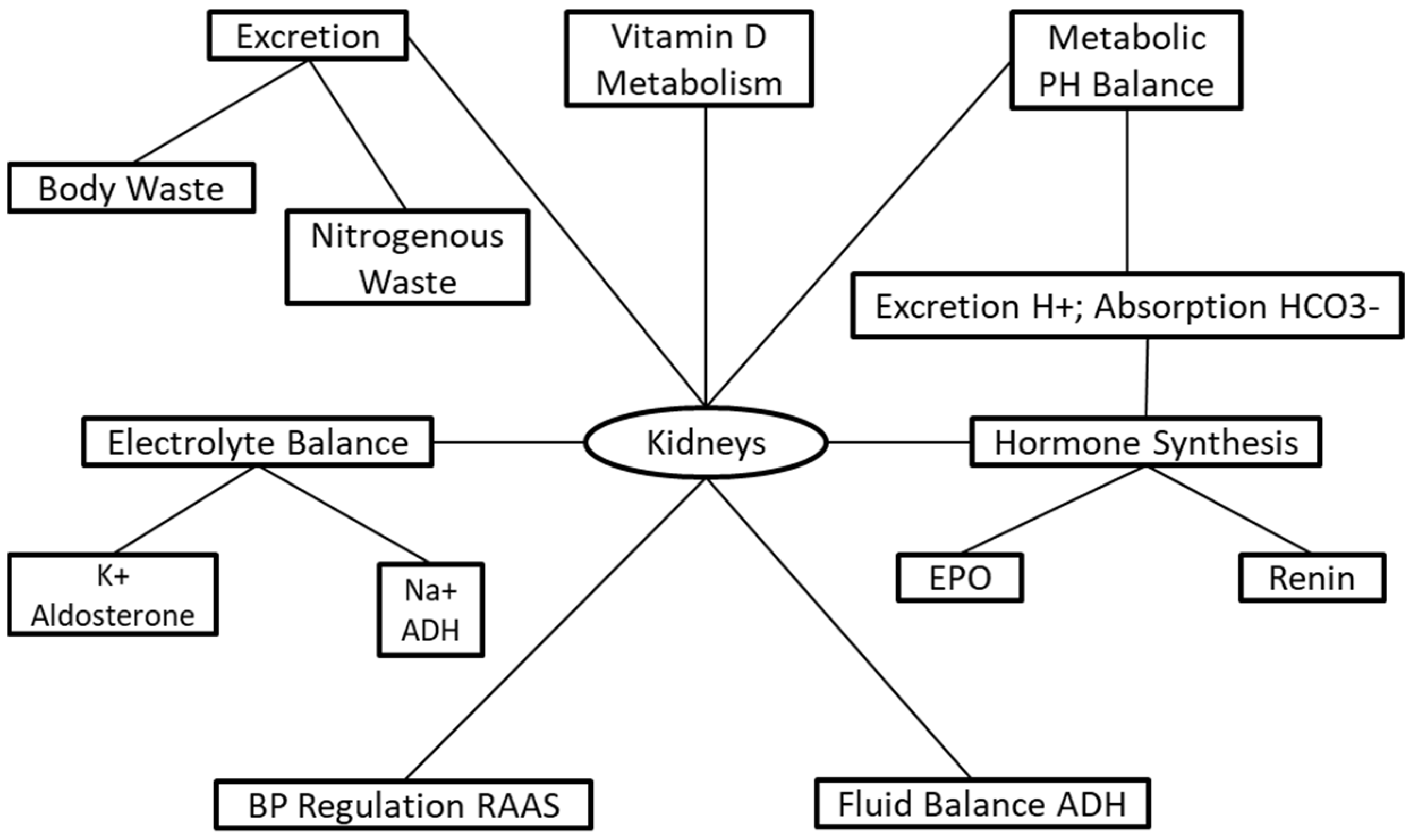
:1. Introduction
2. Medical Nutrition Therapy
2.1. Protein and Renal Function
2.2. Very Low-Protein Diet
2.3. Vitamin D
2.4. Calcium
2.5. Phosphorus
| Electrolytes | Damage in CKD | Recommendation | Outcome | Ref |
|---|---|---|---|---|
| Total calcium CKD 3–4 w/no use of taking active vitamin D analogues | Ca2+ deficiency ↑ risk secondary hyperparathyroidism and bone disorders. Excessive Ca2+ ↑ risk extraosseous calcification and CVD | 800–1000 mg/day | Maintain Ca2+ balance | [18,29,61] |
| CKD 5 w/use of active vitamin D analogues | Ca2+ deficiency ↑ risk secondary hyperparathyroidism and bone disorders. Excessive Ca2+ ↑ risk extraosseous calcification and CVD | Individualize Ca2+ restriction based on the use of vitamin D analogues | Maintain Ca2+ balance and prevent hypercalcemeia | [18,29,62] |
| Dietary Phosphorus * CKD 1–5 | High dietary phosphorus intake associated w/ accelerated progression of disease and greater 5-year mortality risk | adjust dietary phosphorus intake to maintain normal serum phosphate levels between 3.4–4.5 mg/dL | Maintain Ca2+ and PTH balance. ↓ Secondary hyperparathyroidism mineral and bone disorders. Slow progression of CKD | [29,63] |
| Dietary Potassium CKD1–5 or post-transplantation | Hyper/hypokalemia associated w/muscular weakness, hypertension, ventricular arrhythmias, and death. Hypokalemia associated w/peripheral neuropathy. | adjust dietary K+ intake to maintain serum potassium within 3.5–5.5 mEq/L | Slow progression of CKD. Prevention of peripheral neuropathy and other nerve related dysfunction. | [29,64,65] |
| Sodium (Na+) CKD 1–5 or post transplantation | ↑ BP excessive fluid retention/increased weight | <2300 mg/day | ↓ BP and normalize fluid balance/weight reduction/may ↓ proteinuria | [29,66,67,68] |
2.6. Potassium
2.7. Sodium
2.8. Whole Food Plant-Based Diet
3. The Role of a Registered Dietitian
4. Future Research and Clinical Practice
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Centers for Disease Control, Chronic Kidney Disease in the United States. 2021. Available online: https://www.cdc.gov/kidneydisease/pdf/Chronic-Kidney-Disease-in-the-US-2021-h.pdf (accessed on 23 February 2021).
- Adair, K.E.; Bowden, R.G. Ameliorating Chronic Kidney Disease Using a Whole Food Plant-Based Diet. Nutrients 2020, 12, 1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, M.A. Anatomy and Physiology of the Kidney. Aorn J. 1998, 68, 799–820. [Google Scholar] [CrossRef]
- Regan, M.C.; Young, L.S.; Geraghty, J.; Fitzpatrick, J.M. Regional Renal Blood Flow in Normal and Disease States. Urol. Res. 1995, 23, 1–10. [Google Scholar] [CrossRef]
- McLafferty, E.; Johnstone, C.; Hendry, C.; Farley, A. Fluid and Electrolyte Balance. Nurs. Stand. 2014, 28, 42. [Google Scholar] [CrossRef]
- Shioji, N.; Hayashi, M.; Morimatsu, H. Kidney, Fluid, and Acid-Base Balance. Masui 2016, 65, 503–510. [Google Scholar]
- Drábková, N.; Hojná, S.; Zicha, J.; Vaněčková, I. Contribution of Selected Vasoactive Systems to Blood Pressure Regulation in Two Models of Chronic Kidney Disease. Physiol. Res. 2020, 405–414. [Google Scholar] [CrossRef]
- Kulda, V. Vitamin D metabolism. Vnitr. Lek. 2012, 58, 400–404. [Google Scholar]
- Palmer, S.C.; Saglimbene, V.; Mavridis, D.; Salanti, G.; Craig, J.C.; Tonelli, M.; Wiebe, N.; Strippoli, G.F. Erythropoiesis-stimulating Agents for Anaemia in Adults with Chronic Kidney Disease: A Network Meta-analysis. Cochrane Database Syst. Rev. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- National Kidney Foundation K/DOQI Clinical Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and Stratification. Am. J. Kidney Dis. 2002, 39, S1–S266.
- Coresh, J.; Selvin, E.; Stevens, L.A.; Manzi, J.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Levey, A.S. Prevalence of Chronic Kidney Disease in the United States. JAMA 2007, 298, 2038–2047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, C.; Ordoñez, J.; Chertow, G.; Fan, D.; McCulloch, C.; Go, A. The Risk of Acute Renal Failure in Patients with Chronic Kidney Disease. Kidney Int. 2008, 74, 101–107. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.S.; Andreoli, S.P.; DuBose, T.; Provenzano, R.; Collins, A.J. Chronic Kidney Disease: Common, Harmful, and Treatable—World Kidney Day 2007. J. Am. Soc. Nephrol. 2007, 18, 374–378. [Google Scholar] [CrossRef] [Green Version]
- McCullough, K.P.; Morgenstern, H.; Saran, R.; Herman, W.H.; Robinson, B.M. Projecting ESRD Incidence and Prevalence in the United States through 2030. J. Am. Soc. Nephrol. 2019, 30, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.D.; Taal, M.W. Epidemiology and Causes of Chronic Kidney Disease. Medicine 2015, 43, 450–453. [Google Scholar] [CrossRef]
- Kovesdy, C.P.; Kopple, J.D.; Kalantar-Zadeh, K. Management of Protein-Energy Wasting in Non-Dialysis-Dependent Chronic Kidney Disease: Reconciling Low Protein Intake with Nutritional Therapy. Am. J. Clin. Nutr. 2013, 97, 1163–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Moe, S.M.; Shroff, R.; Tonelli, M.A.; Toussaint, N.D.; et al. Executive Summary of the 2017 KDIGO Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) Guideline Update: What’s Changed and Why It Matters. Kidney Int. 2017, 92, 26–36. [Google Scholar] [CrossRef] [Green Version]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.-J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. Am. J. Kidney Dis. 2020, 76, S1–S107. [Google Scholar] [CrossRef] [PubMed]
- Delimaris, I. Adverse Effects Associated with Protein Intake above the Recommended Dietary Allowance for Adults. ISRN Nutr. 2013, 2013, 126929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacKay, E.M.; MacKay, L.L.; Addis, T. Factors Which Determine Renal Weight. Am. J. Physiol.-Leg. Content 1928, 86, 466–470. [Google Scholar] [CrossRef] [Green Version]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Meyer, T.W.; Anderson, S.; Brenner, B.M. Dietary Protein Intake and Progressive Glomerular Sclerosis: The Role of Capillary Hypertension and Hyperperfusion in the Progression of Renal Disease. Ann. Intern. Med. 1983, 98, 832–838. [Google Scholar] [CrossRef] [Green Version]
- Modification of Diet in Renal Disease Study Group; Hunsicker, L.G.; Adler, S.; Caggiula, A.; England, B.K.; Greene, T.; Kusek, J.W.; Rogers, N.L.; Teschan, P.E.; Beck, G. Predictors of the Progression of Renal Disease in the Modification of Diet in Renal Disease Study. Kidney Int. 1997, 51, 1908–1919. [Google Scholar] [CrossRef] [Green Version]
- Oba, R.; Kanzaki, G.; Sasaki, T.; Okabayashi, Y.; Haruhara, K.; Koike, K.; Kobayashi, A.; Yamamoto, I.; Tsuboi, N.; Yokoo, T. Dietary Protein Intake and Single-Nephron Glomerular Filtration Rate. Nutrients 2020, 12, 2549. [Google Scholar] [CrossRef]
- Moe, S.M.; Zidehsarai, M.P.; Chambers, M.A.; Jackman, L.A.; Radcliffe, J.S.; Trevino, L.L.; Donahue, S.E.; Asplin, J.R. Vegetarian Compared with Meat Dietary Protein Source and Phosphorus Homeostasis in Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 257–264. [Google Scholar] [CrossRef] [Green Version]
- Bell, R.R.; Draper, H.H.; Tzeng, D.Y.M.; Shin, H.K.; Schmidt, G.R. Physiological Responses of Human Adults to Foods Containing Phosphate Additives. J. Nutr. 1977, 107, 42–50. [Google Scholar] [CrossRef]
- Fouque, D.; Chen, J.; Chen, W.; Garneata, L.; Hwang, S.; Kalantar-Zadeh, K.; Kopple, J.D.; Mitch, W.E.; Piccoli, G.; Teplan, V.; et al. Adherence to Ketoacids/Essential Amino Acids-Supplemented Low Protein Diets and New Indications for Patients with Chronic Kidney Disease. BMC Nephrol. 2016, 17. [Google Scholar] [CrossRef] [Green Version]
- Kopple, J.D.; Shinaberger, J.H.; Coburn, J.W.; Sorensen, M.K.; Rubini, M.E. Optimal Dietary Protein Treatment during Chronic Hemodialysis. Trans. Am. Soc. Artif. Intern. Organs 1969, 15, 302–308. [Google Scholar] [PubMed]
- Lynch, K.E.; Lynch, R.; Curhan, G.C.; Brunelli, S.M. Prescribed Dietary Phosphate Restriction and Survival among Hemodialysis Patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 620–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutiérrez, O.M. Sodium- and Phosphorus-Based Food Additives: Persistent but Surmountable Hurdles in the Management of Nutrition in Chronic Kidney Disease. Adv. Chronic Kidney Dis. 2013, 20, 150–156. [Google Scholar] [CrossRef] [Green Version]
- Fouque, D.; Aparicio, M. Eleven Reasons to Control the Protein Intake of Patients with Chronic Kidney Disease. Nat. Rev. Nephrol. 2007, 3, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-Supplemented Vegetarian Very Low–Protein Diet and CKD Progression. JASN 2016, 27, 2164–2176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elliott, P.; Stamler, J.; Dyer, A.R.; Appel, L.; Dennis, B.; Kesteloot, H.; Ueshima, H.; Okayama, A.; Chan, Q.; Garside, D.B.; et al. Association between Protein Intake and Blood Pressure: The INTERMAP Study. Arch. Intern. Med. 2006, 166, 79–87. [Google Scholar] [CrossRef]
- Evenepoel, P.; Meijers, B.K. Dietary Fiber and Protein: Nutritional Therapy in Chronic Kidney Disease and Beyond. Kidney Int. 2012, 81, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.H.; Lima, P.R.; Almerinda, M.; Alves, V.F.; Bottini, P.V.; de Faria, J.B. Renal Haemodynamic Responses to a Chicken or Beef Meal in Normal Individuals. Nephrol. Dial. Transpl. 1998, 13, 2261–2264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, H.; Takasawa, M.; Kashara, S.; Tsuda, A.; Momotsu, T.; Ito, S.; Shibata, A. Effects of Acute Protein Loads of Different Sources on Renal Function of Patients with Diabetic Nephropathy. Tohoku J. Exp. Med. 1989, 159, 153–162. [Google Scholar] [CrossRef] [Green Version]
- Attini, R.; Leone, F.; Parisi, S.; Fassio, F.; Capizzi, I.; Loi, V.; Colla, L.; Rossetti, M.; Gerbino, M.; Maxia, S.; et al. Vegan-Vegetarian Low-Protein Supplemented Diets in Pregnant CKD Patients: Fifteen Years of Experience. BMC Nephrol. 2016, 17, 132. [Google Scholar] [CrossRef] [Green Version]
- Cases, A.; Cigarrán-Guldrís, S.; Mas, S.; Gonzalez-Parra, E. Vegetable-Based Diets for Chronic Kidney Disease? It Is Time to Reconsider. Nutrients 2019, 11, 1263. [Google Scholar] [CrossRef] [Green Version]
- Moorthi, R.N.; Vorland, C.J.; Gallant, K.M.H. Diet and Diabetic Kidney Disease: Plant versus Animal Protein. Curr. Diab. Rep. 2017, 17, 15. [Google Scholar] [CrossRef] [Green Version]
- Kahleova, H.; Levin, S.; Barnard, N. Cardio-Metabolic Benefits of Plant-Based Diets. Nutrients 2017, 9, 848. [Google Scholar] [CrossRef]
- Klahr, S.; Levey, A.S.; Beck, G.J.; Caggiula, A.W.; Hunsicker, L.; Kusek, J.W.; Striker, G. The Effects of Dietary Protein Restriction and Blood-Pressure Control on the Progression of Chronic Renal Disease. Modification of Diet in Renal Disease Study Group. N. Engl. J. Med. 1994, 330, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Kasiske, B.L.; Lakatua, J.D.; Ma, J.Z.; Louis, T.A. A Meta-Analysis of the Effects of Dietary Protein Restriction on the Rate of Decline in Renal Function. Am. J. Kidney Dis. 1998, 31, 954–961. [Google Scholar] [CrossRef]
- Shah, B.V.; Patel, Z.M. Role of Low Protein Diet in Management of Different Stages of Chronic Kidney Disease—Practical Aspects. BMC Nephrol. 2016, 17, 156. [Google Scholar] [CrossRef] [Green Version]
- Yan, B.; Su, X.; Xu, B.; Qiao, X.; Wang, L. Effect of Diet Protein Restriction on Progression of Chronic Kidney Disease: A Systematic Review and Meta-Analysis. PLoS ONE 2018, 13, e0206134. [Google Scholar] [CrossRef]
- Lips, P.; Goldsmith, D.; de Jongh, R. Vitamin D and Osteoporosis in Chronic Kidney Disease. J. Nephrol. 2017, 30, 671–675. [Google Scholar] [CrossRef]
- Hou, Y.-C.; Lu, C.-L.; Lu, K.-C. Mineral Bone Disorders in Chronic Kidney Disease. Nephrology 2018, 23, 88–94. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.M.; Choi, H.J.; Lee, J.P.; Kim, D.K.; Oh, Y.K.; Kim, Y.S.; Lim, C.S. Prevalence of Vitamin D Deficiency and Effects of Supplementation with Cholecalciferol in Patients with Chronic Kidney Disease. J. Ren. Nutr. 2014, 24, 20–25. [Google Scholar] [CrossRef]
- Filipov, J.J.; Zlatkov, B.K.; Dimitrov, E.P.; Svinarov, D. Relationship between Vitamin D Status and Immunosuppressive Therapy in Kidney Transplant Recipients. Biotechnol. Biotechnol. Equip. 2015, 29, 331–335. [Google Scholar] [CrossRef]
- Berdanier, C.D.; Berdanier, L.A. Advanced Nutrition Macronutrients, Micronutrients, and Metabolism, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2015; ISBN 978-1-4822-0517-6. [Google Scholar]
- Garofalo, C.; Provenzano, M.; Andreucci, M.; Pisani, A.; De Nicola, L.; Conte, G.; Borrelli, S. Predictive Effect of Salt Intake on Patient and Kidney Survival in Non-Dialysis CKD: Competing Risk Analysis in Older versus Younger Patients under Nephrology Care. Nephrol. Dial. Transplant. 2020, gfaa252. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Román-Viñas, B.; Sanchez-Villegas, A.; Guasch-Ferré, M.; Corella, D.; La Vecchia, C. Benefits of the Mediterranean Diet: Epidemiological and Molecular Aspects. Mol. Asp. Med. 2019, 67, 1–55. [Google Scholar] [CrossRef] [PubMed]
- Kandula, P.; Dobre, M.; Schold, J.D.; Schreiber, M.J.; Mehrotra, R.; Navaneethan, S.D. Vitamin D Supplementation in Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Observational Studies and Randomized Controlled Trials. Clin. J. Am. Soc. Nephrol. 2011, 6, 50–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jean, G.; Souberbielle, J.C.; Chazot, C. Vitamin D in Chronic Kidney Disease and Dialysis Patients. Nutrients 2017, 9, 328. [Google Scholar] [CrossRef]
- Massart, A.; Debelle, F.D.; Racapé, J.; Gervy, C.; Husson, C.; Dhaene, M.; Wissing, K.M.; Nortier, J.L. Biochemical Parameters After Cholecalciferol Repletion in Hemodialysis: Results From the VitaDial Randomized Trial. Am. J. Kidney Dis. 2014, 64, 696–705. [Google Scholar] [CrossRef]
- Umut, S. Relationship of Dietary Phosphate Intake with Risk of End-Stage Renal Disease and Mortality in Chronic Kidney Disease Stages 3–5: The Modification of Diet in Renal Disease Study|Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S0085253815000241?token=91355844158B638748824241A0450698282B3693E30793102456034ADB82312BE8F55F454D23184C440C0F6760D9B40D (accessed on 23 February 2021).
- Malluche, H.; Monier-Faugère, M. Hyperphosphatemia: Pharmacologic Intervention Yesterday, Today and Tomorrow—Abstract—Europe PMC. Available online: https://europepmc.org/article/med/11076107 (accessed on 23 February 2021).
- Carrero, J.J.; Stenvinkel, P.; Cuppari, L.; Ikizler, T.A.; Kalantar-Zadeh, K.; Kaysen, G.; Mitch, W.E.; Price, S.R.; Wanner, C.; Wang, A.Y.M.; et al. Etiology of the Protein-Energy Wasting Syndrome in Chronic Kidney Disease: A Consensus Statement From the International Society of Renal Nutrition and Metabolism (ISRNM). J. Ren. Nutr. 2013, 23, 77–90. [Google Scholar] [CrossRef] [Green Version]
- Kalantar-Zadeh, K. Patient Education for Phosphorus Management in Chronic Kidney Disease. PPA 2013, 7, 379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melse-Boonstra, A. Bioavailability of Micronutrients From Nutrient-Dense Whole Foods: Zooming in on Dairy, Vegetables, and Fruits. Front. Nutr. 2020, 7, 101. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Gutekunst, L.; Mehrotra, R.; Kovesdy, C.P.; Bross, R.; Shinaberger, C.S.; Noori, N.; Hirschberg, R.; Benner, D.; Nissenson, A.R.; et al. Understanding Sources of Dietary Phosphorus in the Treatment of Patients with Chronic Kidney Disease. CJASN 2010, 5, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Sigrist, M.K.; Taal, M.W.; Bungay, P.; McIntyre, C.W. Progressive Vascular Calcification over 2 Years Is Associated with Arterial Stiffening and Increased Mortality in Patients with Stages 4 and 5 Chronic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2007, 2, 1241–1248. [Google Scholar] [CrossRef] [Green Version]
- Goraya, N.; Simoni, J.; Jo, C.-H.; Wesson, D.E. Treatment of Metabolic Acidosis in Patients with Stage 3 Chronic Kidney Disease with Fruits and Vegetables or Oral Bicarbonate Reduces Urine Angiotensinogen and Preserves Glomerular Filtration Rate. Kidney Int. 2014, 86, 1031–1038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawasaki, T.; Maeda, Y.; Matsuki, H.; Matsumoto, Y.; Akazawa, M.; Kuyama, T. Urinary Phosphorus Excretion per Creatinine Clearance as a Prognostic Marker for Progression of Chronic Kidney Disease: A Retrospective Cohort Study. BMC Nephrol. 2015, 16, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnold, R.; Pianta, T.J.; Pussell, B.A.; Kirby, A.; O’Brien, K.; Sullivan, K.; Holyday, M.; Cormack, C.; Kiernan, M.C.; Krishnan, A.V. Randomized, Controlled Trial of the Effect of Dietary Potassium Restriction on Nerve Function in CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 1569–1577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cupisti, A.; Kovesdy, C.P.; D’Alessandro, C.; Kalantar-Zadeh, K. Dietary Approach to Recurrent or Chronic Hyperkalaemia in Patients with Decreased Kidney Function. Nutrients 2018, 10, 261. [Google Scholar] [CrossRef] [Green Version]
- McMahon, E.J.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Johnson, D.W.; Campbell, K.L. A Randomized Trial of Dietary Sodium Restriction in CKD. JASN 2013, 24, 2096–2103. [Google Scholar] [CrossRef] [Green Version]
- Vogt, L.; Waanders, F.; Boomsma, F.; de Zeeuw, D.; Navis, G. Effects of Dietary Sodium and Hydrochlorothiazide on the Antiproteinuric Efficacy of Losartan. J. Am. Soc. Nephrol. 2008, 19, 999–1007. [Google Scholar] [CrossRef] [Green Version]
- Campbell, K.L.; Johnson, D.W.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Whitehead, J.P.; Dimeski, G.; McMahon, E. A Randomized Trial of Sodium-Restriction on Kidney Function, Fluid Volume and Adipokines in CKD Patients. BMC Nephrol. 2014, 15, 57. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, R.; Watanabe, R. Hyperkalemia in Chronic Kidney Disease. Rev. Assoc. Médica Bras. 2020, 66, s31–s36. [Google Scholar] [CrossRef] [Green Version]
- Hansrivijit, P.; Oli, S.; Khanal, R.; Ghahramani, N.; Thongprayoon, C.; Cheungpasitporn, W. Mediterranean Diet and the Risk of Chronic Kidney Disease: A Systematic Review and Meta-Analysis. Nephrology 2020, 25, 913–918. [Google Scholar] [CrossRef]
- Mirabelli, M.; Chiefari, E.; Arcidiacono, B.; Corigliano, D.M.; Brunetti, F.S.; Maggisano, V.; Russo, D.; Foti, D.P.; Brunetti, A. Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases. Nutrients 2020, 12, 1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raphael, K.L. The Dietary Approaches to Stop Hypertension (DASH) Diet in Chronic Kidney Disease: Should We Embrace It? Kidney Int. 2019, 95, 1296–1298. [Google Scholar] [CrossRef] [PubMed]
- Tyson, C.C.; Lin, P.-H.; Corsino, L.; Batch, B.C.; Allen, J.; Sapp, S.; Barnhart, H.; Nwankwo, C.; Burroughs, J.; Svetkey, L.P. Short-Term Effects of the DASH Diet in Adults with Moderate Chronic Kidney Disease: A Pilot Feeding Study. Clin. Kidney J. 2016, 9, 592–598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean Diet Pyramid: A Cultural Model for Healthy Eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Tomaino, L.; Dernini, S.; Berry, E.M.; Lairon, D.; de la Cruz, J.N.; Bach-Faig, A.; Donini, L.M.; Medina, F.-X.; Belahsen, R.; et al. Updating the Mediterranean Diet Pyramid towards Sustainability: Focus on Environmental Concerns. Int. J. Environ. Res. Public Health 2020, 17, 8758. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean Diet and Multiple Health Outcomes: An Umbrella Review of Meta-Analyses of Observational Studies and Randomised Trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef]
- Borrelli, S.; De Nicola, L.; Minutolo, R.; Conte, G.; Chiodini, P.; Cupisti, A.; Santoro, D.; Calabrese, V.; Giannese, D.; Garofalo, C.; et al. Current Management of Hyperkalemia in Non-Dialysis CKD: Longitudinal Study of Patients Receiving Stable Nephrology Care. Nutrients 2021, 13, 942. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F. Potassium Binders for Hyperkalemia in Chronic Kidney Disease—Diet, Renin-Angiotensin-Aldosterone System Inhibitor Therapy, and Hemodialysis. Mayo Clin. Proc. 2020, 95, 339–354. [Google Scholar] [CrossRef] [Green Version]
- Murphy, D.; Ster, I.C.; Kaski, J.-C.; Anderson, L.; Banerjee, D. The LIFT Trial: Study Protocol for a Double-Blind, Randomised, Placebo-Controlled Trial of K+-Binder Lokelma for Maximisation of RAAS Inhibition in CKD Patients with Heart Failure. BMC Nephrol. 2021, 22, 254. [Google Scholar] [CrossRef]
- Mattson, D.L. Immune Mechanisms of Salt-Sensitive Hypertension and Renal End-Organ Damage. Nat. Rev. Nephrol. 2019, 15, 290–300. [Google Scholar] [CrossRef]
- Borrelli, S.; Provenzano, M.; Gagliardi, I.; Ashour, M.; Liberti, M.E.; De Nicola, L.; Conte, G.; Garofalo, C.; Andreucci, M. Sodium Intake and Chronic Kidney Disease. Int. J. Mol. Sci. 2020, 21, 4744. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Borrelli, S.; Provenzano, M.; De Stefano, T.; Vita, C.; Chiodini, P.; Minutolo, R.; Nicola, L.; Conte, G. Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials. Nutrients 2018, 10, 732. [Google Scholar] [CrossRef] [Green Version]
- Chang, A.R.; Lóser, M.; Malhotra, R.; Appel, L.J. Blood Pressure Goals in Patients with CKD. Clin. J. Am. Soc. Nephrol. 2019, 14, 161–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotchen, T.A.; Cowley, A.W.; Frohlich, E.D. Salt in Health and Disease—A Delicate Balance. N. Engl. J. Med. 2013, 368, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Kempner, W. Some Effects of the Rice Diet Treatment of Kidney Disease and Hypertension. Bull. N. Y. Acad. Med. 1946, 22, 358–370. [Google Scholar]
- Garofalo, C.; Borrelli, S.; Pacilio, M.; Minutolo, R.; Chiodini, P.; De Nicola, L.; Conte, G. Hypertension and Prehypertension and Prediction of Development of Decreased Estimated GFR in the General Population: A Meta-Analysis of Cohort Studies. Am. J. Kidney Dis. 2016, 67, 89–97. [Google Scholar] [CrossRef]
- Tuso, P.J.; Ismail, M.H.; Ha, B.P.; Bartolotto, C. Nutritional Update for Physicians: Plant-Based Diets. Perm J. 2013, 17, 61–66. [Google Scholar] [CrossRef] [Green Version]
- Barnard, N.D.; Cohen, J.; Jenkins, D.J.A.; Turner-McGrievy, G.; Gloede, L.; Green, A.; Ferdowsian, H. A Low-Fat Vegan Diet and a Conventional Diabetes Diet in the Treatment of Type 2 Diabetes: A Randomized, Controlled, 74-Wk Clinical Trial. Am. J. Clin. Nutr. 2009, 89, 1588S–1596S. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Mirrahimi, A.; Sievenpiper, J.L.; Jenkins, D.J.A.; Darling, P.B. Dietary Fiber Effects in Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Controlled Feeding Trials. Eur. J. Clin. Nutr. 2015, 69, 761–768. [Google Scholar] [CrossRef] [Green Version]
- Biörklund, M.; van Rees, A.; Mensink, R.P.; Önning, G. Changes in Serum Lipids and Postprandial Glucose and Insulin Concentrations after Consumption of Beverages with β-Glucans from Oats or Barley: A Randomised Dose-Controlled Trial. Eur. J. Clin. Nutr. 2005, 59, 1272–1281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, M.-J.; Chang, C.-S.; Cheng, C.-H.; Chen, C.-H.; Lee, W.-C.; Hsu, Y.-H.; Shu, K.-H.; Tang, M.-J. Colonic Transit Time in Long-Term Dialysis Patients. Am. J. Kidney Dis. 2004, 44, 322–327. [Google Scholar] [CrossRef]
- Rebello, C.J.; O’Neil, C.E.; Greenway, F.L. Dietary Fiber and Satiety: The Effects of Oats on Satiety. Nutr. Rev. 2016, 74, 131–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bowden, R.G.; Wilson, R.L.; Deike, E.; Gentile, M. Fish Oil Supplementation Lowers C-Reactive Protein Levels Independent of Triglyceride Reduction in Patients with End-Stage Renal Disease. Nutr. Clin. Pract. 2009, 24, 508–512. [Google Scholar] [CrossRef]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of Omega-3 Fatty Acids on Serum Markers of Cardiovascular Disease Risk: A Systematic Review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J.; American Heart Association. Nutrition Committee Fish Consumption, Fish Oil, Omega-3 Fatty Acids, and Cardiovascular Disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef]
- Sherman, R.A.; Mehta, O. Phosphorus and Potassium Content of Enhanced Meat and Poultry Products: Implications for Patients Who Receive Dialysis. CJASN 2009, 4, 1370–1373. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.M.; Leon, J.B.; Sehgal, A.R. Phosphorus-Containing Food Additives and the Accuracy of Nutrient Databases: Implications for Renal Patients. J. Ren. Nutr. 2007, 17, 350–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siener, R. Dietary Treatment of Metabolic Acidosis in Chronic Kidney Disease. Nutrients 2018, 10, 512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kramer, H. Diet and Chronic Kidney Disease. Adv. Nutr. 2019, 10, S367–S379. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Hashmi, S.; Shah, S.; Kalantar-Zadeh, K. Plant-Based Diets for Prevention and Management of Chronic Kidney Disease. Curr. Opin. Nephrol. Hypertens. 2020, 29, 16–21. [Google Scholar] [CrossRef]
- Levey, A.S.; Adler, S.; Caggiula, A.W.; England, B.K.; Greene, T.; Hunsicker, L.G.; Kusek, J.W.; Rogers, N.L.; Teschan, P.E. Effects of Dietary Protein Restriction on the Progression of Advanced Renal Disease in the Modification of Diet in Renal Disease Study. Am. J. Kidney Dis. 1996, 27, 652–663. [Google Scholar] [CrossRef]
- Zeller, K.; Whittaker, E.; Sullivan, L.; Raskin, P.; Jacobson, H.R. Effect of Restricting Dietary Protein on the Progression of Renal Failure in Patients with Insulin-Dependent Diabetes Mellitus. N. Engl. J. Med. 1991, 324, 78–84. [Google Scholar] [CrossRef]
- Levey, A.S.; Greene, T.; Beck, G.J.; Caggiula, A.W.; Kusek, J.W.; Hunsicker, L.G.; Klahr, S. Dietary Protein Restriction and the Progression of Chronic Renal Disease: What Have All of the Results of the MDRD Study Shown? Modification of Diet in Renal Disease Study Group. J. Am. Soc. Nephrol. 1999, 10, 2426–2439. [Google Scholar] [CrossRef]
| Nutrients | Damage in CKD | Recommendation | Outcome | Ref |
|---|---|---|---|---|
| Protein CKD 3–5 patients. Not on Dialysis/Without Diabetes | Proteinuria/glomerular sclerosis/hyperfiltration/intraglomerular hypertension and hyperperfusion | 0.55–0.6 g/kg body wt/day | Reduce uremia, uremic toxins, and hyperfiltration Improve organ function and renal hemodynamics | [18,21,22] |
| CKD 3–5 Pts Not on Dialysis and w/Diabetes | Proteinuria/glomerular sclerosis/hyperfiltration/intraglomerular hypertension and hyperperfusion | 0.6–0.8 g/kg body wt/day | Reduce uremia, uremic toxins, and hyperfiltration Improve organ function, and renal hemodynamics | [18,25,26] |
| HD and PD Pts w/ and without Diabetes | Proteinuria/glomerular sclerosis/hyperfiltration/intraglomerular hypertension and hyperperfusion | 1.0–1.2 g/kg body wt/day | Reduce uremia, uremic toxins, and hyperfiltration Improve organ function, and renal hemodynamics. | [18,25,26] |
| Energy Intake CKD 1–5D or post transplantation | Inadequate intake ↑ risk PEW, ↑ risk malnutrition. Excessive intake ↑ risk CVD, ↑ risk diabetes | 25–35 kcals/kg body wt/day | Maintain neutral nitrogen balance and body composition. | [18,28] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naber, T.; Purohit, S. Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease. Nutrients 2021, 13, 3277. https://doi.org/10.3390/nu13093277
Naber T, Purohit S. Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease. Nutrients. 2021; 13(9):3277. https://doi.org/10.3390/nu13093277
Chicago/Turabian StyleNaber, Tania, and Sharad Purohit. 2021. "Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease" Nutrients 13, no. 9: 3277. https://doi.org/10.3390/nu13093277
APA StyleNaber, T., & Purohit, S. (2021). Chronic Kidney Disease: Role of Diet for a Reduction in the Severity of the Disease. Nutrients, 13(9), 3277. https://doi.org/10.3390/nu13093277







