Energy Balance and Control of Body Weight: Possible Effects of Meal Timing and Circadian Rhythm Dysregulation
Abstract
:1. Introduction
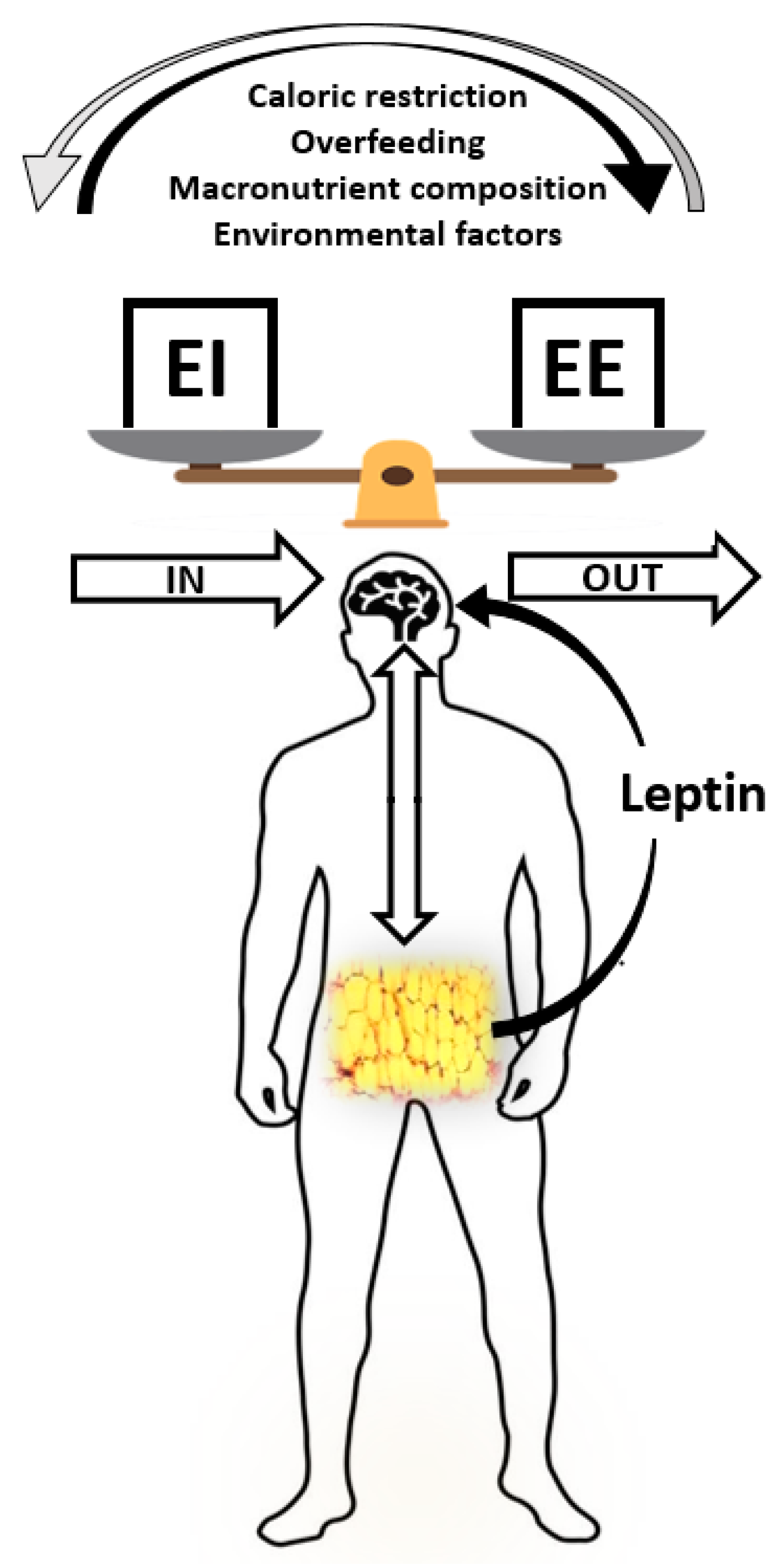
2. The Regulation of Energy Balance: From a Static to a Dynamic Perspective
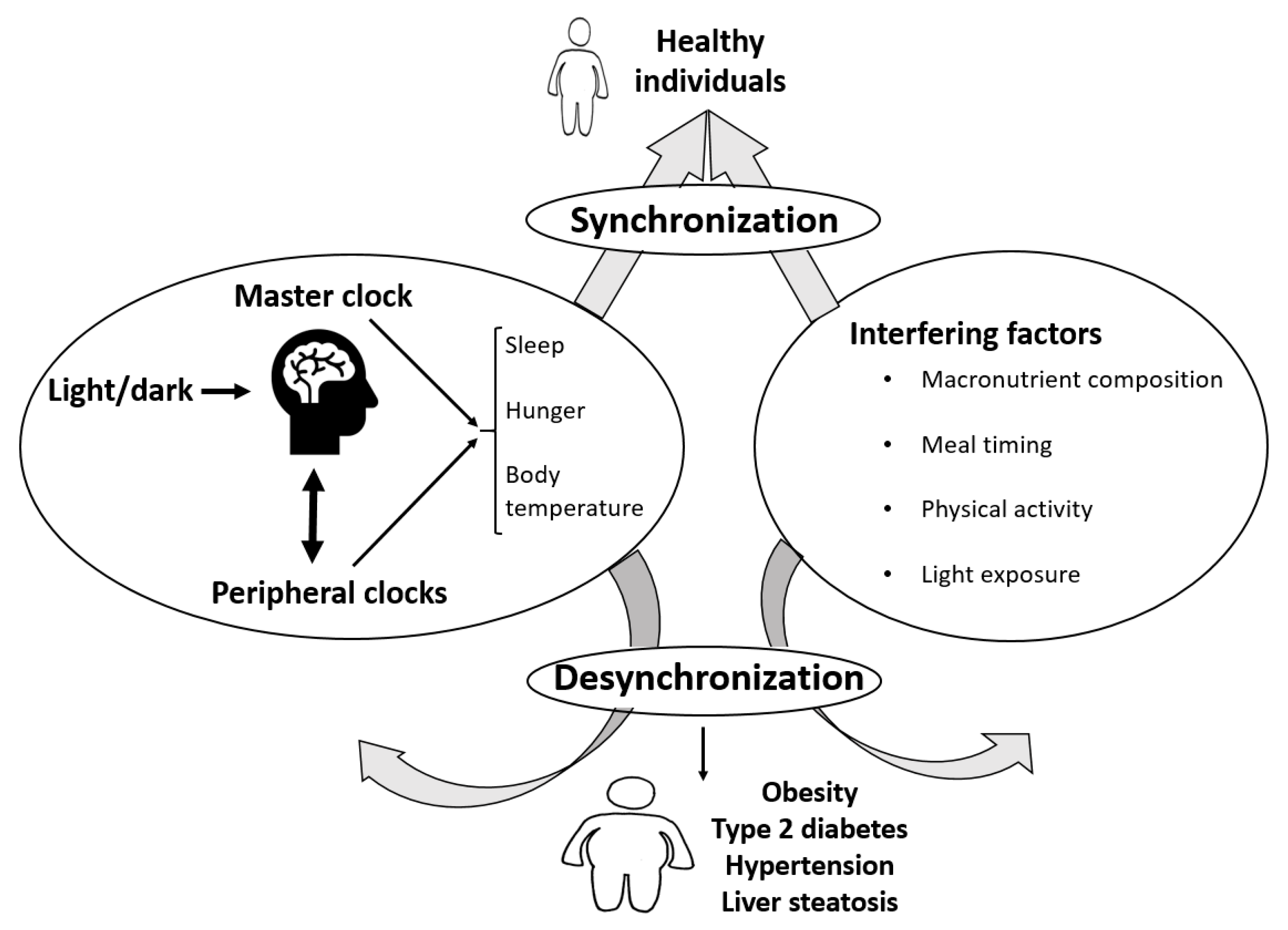
3. Circadian Rhythm and Energy Balance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EI | energy intake |
| EE | energy expenditure |
| PA | physical activity |
| RMR | resting metabolic rate |
| TEF | thermic effect of food |
References
- World Health Organization. World Health Organization Obesity and Overweight Fact Sheet. 2016. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 18 September 2021).
- Kelly, T.; Yang, W.; Chen, C.S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef] [Green Version]
- Schibler, U.; Gotic, I.; Saini, C.; Gos, P.; Curie, T.; Emmenegger, Y.; Sinturel, F.; Gosselin, P.; Gerber, A.; Fleury-Olela, F.; et al. Clock-talk: Interactions between central and peripheral circadian oscillators in mammals. Cold Spring Harb. Symp. Quant. Biol. 2015, 80, 223–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, L.; Zee, P.C. Circadian rhythm sleep disorders. Neurol. Clin. 2012, 30, 1167–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gale, J.E.; Cox, H.I.; Qian, J.; Block, G.D.; Colwell, C.S.; Matveyenko, A.V. Disruption of circadian rhythms accelerates development of diabetes through pancreatic beta-cell loss and dysfunction. J. Biol. Rhythms. 2011, 26, 423–433. [Google Scholar] [CrossRef] [Green Version]
- Garaulet, M.; Gómez-Abellán, P. Timing of food intake and obesity: A novel association. Physiol. Behav. 2014, 134, 44–50. [Google Scholar] [CrossRef]
- Shaw, E.; Leung, G.K.W.; Jong, J.; Coates, A.M.; Davis, R.; Blair, M.; Huggins, C.E.; Dorrian, J.; Banks, S.; Kellow, N.J.; et al. The Impact of time of day on energy expenditure: Implications for long-term energy balance. Nutrients 2019, 11, 2383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, K.D.; Heymsfield, S.B.; Kemnitz, J.W.; Klein, S.; Schoeller, D.A.; Speakman, J.R. Energy balance and its components: Implications for body weight regulation. Am. J. Clin. Nutr. 2012, 95, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Weyer, C.; Snitker, S.; Rising, R.; Bogardus, C.; Ravussin, E. Determinants of energy expenditure and fuel utilization in man: Effects of body composition, age, sex, ethnicity and glucose tolerance in 916 subjects. Int. J. Obes. 1999, 23, 715–722. [Google Scholar] [CrossRef] [Green Version]
- Venti, C.A.; Votruba, S.B.; Franks, P.W.; Krakoff, J.; Salbe, A.D. Reproducibility of ad libitum energy intake with the use of a computerized vending machine system. Am. J. Clin. Nutr. 2010, 91, 343–348. [Google Scholar] [CrossRef]
- Ravussin, E.; Lillioja, S.; Anderson, T.E.; Christin, L.; Bogardus, C. Determinants of 24-hour energy expenditure in man. Methods and results using a respiratory chamber. J. Clin. Investig. 1986, 78, 1568–1578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basolo, A.; Hohenadel, M.; Ang, Q.Y.; Piaggi, P.; Heinitz, S.; Walter, M.; Walter, P.; Parrington, S.; Trinidad, D.D.; von Schwartzenberg, R.J.; et al. Effects of underfeeding and oral vancomycin on gut microbiome and nutrient absorption in humans. Nat. Med. 2020, 26, 589–598. [Google Scholar] [CrossRef]
- Widdowson, E.M. Assessment of the energy value of human foods. Proc. Nutr. Soc. 1955, 14, 142–154. [Google Scholar] [CrossRef] [Green Version]
- Lenard, N.R.; Berthoud, H.-R. Central and peripheral regulation of food intake and physical activity: Pathways and genes. Obesity 2008, 16, S11–S22. [Google Scholar] [CrossRef]
- Hopkins, M.; Blundell, J.; Halford, J.; King, N.; Finlayson, G. The Regulation of Food Intake in Humans; Endotext: South Dartmouth, MA, USA, 2016. [Google Scholar]
- Ravussin, E.; Bogardus, C. Energy balance and weight regulation: Genetics versus environment. Br. J. Nutr. 2000, 83, S17–S20. [Google Scholar] [CrossRef]
- Basolo, A.; Ando, T.; Hollstein, T.; Votruba, S.B.; Krakoff, J.; Piaggi, P. Higher urinary dopamine concentration is associated with greater ad libitum energy intake in humans. Obesity 2020, 28, 953–961. [Google Scholar] [CrossRef]
- Basolo, A.; Heinitz, S.; Stinson, E.J.; Begaye, B.; Hohenadel, M.; Piaggi, P.; Krakoff, J.; Votruba, S.B. Fasting glucagon-like peptide 1 concentration is associated with lower carbohydrate intake and increases with overeating. J. Endocrinol. Investig. 2019, 42, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, M.M. Central and peripheral control of food intake. Endocr. Regul. 2017, 51, 52–70. [Google Scholar] [CrossRef] [Green Version]
- Blundell, J.E.; Gillett, A. Control of food intake in the obese. Obes. Res. 2001, 9, 263s–270s. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.Y.; Ravussin, E. Indirect calorimetry: An indispensable tool to understand and predict obesity. Eur. J. Clin. Nutr. 2017, 71, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Schoffelen, P.F.M.; Plasqui, G. Classical experiments in whole-body metabolism: Open-circuit respirometry-diluted flow chamber, hood, or facemask systems. Eur. J. Appl. Physiol. 2018, 118, 33–49. [Google Scholar] [CrossRef] [Green Version]
- Piaggi, P.; Krakoff, J.; Bogardus, C.; Thearle, M.S. Lower “awake and fed thermogenesis” predicts future weight gain in subjects with abdominal adiposity. Diabetes 2013, 62, 4043–4051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravussin, E.; Bogardus, C. A brief overview of human energy metabolism and its relationship to essential obesity. Am. J. Clin. Nutr. 1992, 55, 242s–245s. [Google Scholar] [CrossRef]
- Bogardus, C.; Lillioja, S.; Ravussin, E.; Abbott, W.; Zawadzki, J.K.; Young, A.; Knowler, W.C.; Jacobowitz, R.; Moll, P.P. Familial dependence of the resting metabolic rate. N. Engl. J. Med. 1986, 315, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.A. Non-exercise activity thermogenesis (NEAT). Nutr. Rev. 2004, 62, S82–S97. [Google Scholar] [CrossRef] [PubMed]
- Garland, T.; Schutz, H.; Chappell, M.A.; Keeney, B.K.; Meek, T.H.; Copes, L.E.; Acosta, W.; Drenowatz, C.; Maciel, R.C.; van Dijk, G.; et al. The biological control of voluntary exercise, spontaneous physical activity and daily energy expenditure in relation to obesity: Human and rodent perspectives. J. Exp. Biol. 2011, 214, 206–229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Secor, S.M. Specific dynamic action: A review of the postprandial metabolic response. J. Comp. Physiol. B. 2009, 179, 1–56. [Google Scholar] [CrossRef] [PubMed]
- De Jonge, L.; Bray, G.A. The thermic effect of food and obesity: A critical review. Obes. Res. 1997, 5, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Thearle, M.S.; Pannacciulli, N.; Bonfiglio, S.; Pacak, K.; Krakoff, J. Extent and determinants of thermogenic responses to 24 hours of fasting, energy balance, and five different overfeeding diets in humans. J. Clin. Endocrinol. Metab. 2013, 98, 2791–2799. [Google Scholar] [CrossRef] [Green Version]
- Tappy, L. Thermic effect of food and sympathetic nervous system activity in humans. Reprod. Nutr. Dev. 1996, 36, 391–397. [Google Scholar] [CrossRef] [Green Version]
- Acheson, K.J.; Ravussin, E.; Wahren, J.; Jéquier, E. Thermic effect of glucose in man. Obligatory and facultative thermogenesis. J. Clin. Investig. 1984, 74, 1572–1580. [Google Scholar] [CrossRef]
- Ravussin, E.; Bogardus, C.; Schwartz, R.S.; Robbins, D.C.; Wolfe, R.R.; Horton, E.S.; Danforth, E.; Sims, E.A. Thermic effect of infused glucose and insulin in man. Decreased response with increased insulin resistance in obesity and noninsulin-dependent diabetes mellitus. J. Clin. Investig. 1983, 72, 893–902. [Google Scholar] [CrossRef] [Green Version]
- Muscogiuri, G.; Barrea, L.; Aprano, S.; Framondi, L.; Di Matteo, R.; Laudisio, D.; Pugliese, G.; Savastano, S.; Colao, A. Chronotype and adherence to the mediterranean diet in obesity: Results from the opera prevention project. Nutrients 2020, 12, 1354. [Google Scholar] [CrossRef] [PubMed]
- McHill, A.W.; Melanson, E.L.; Higgins, J.; Connick, E.; Moehlman, T.M.; Stothard, E.R.; Wright, K.P., Jr. Impact of circadian misalignment on energy metabolism during simulated nightshift work. Proc. Natl. Acad. Sci. USA 2014, 111, 17302–17307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Lorenzo, L.; De Pergola, G.; Zocchetti, C.; L’Abbate, N.; Basso, A.; Pannacciulli, N.; Cignarelli, M.; Giorgino, R.; Soleo, L. Effect of shift work on body mass index: Results of a study performed in 319 glucose-tolerant men working in a Southern Italian industry. Int. J. Obes. 2003, 27, 1353–1358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markwald, R.R.; Melanson, E.L.; Smith, M.R.; Higgins, J.; Perreault, L.; Eckel, R.H.; Wright, K.P., Jr. Impact of insufficient sleep on total daily energy expenditure, food intake, and weight gain. Proc. Natl. Acad. Sci. USA 2013, 110, 5695–5700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segal, K.R.; Gutin, B.; Nyman, A.M.; Pi-Sunyer, F.X. Thermic effect of food at rest, during exercise, and after exercise in lean and obese men of similar body weight. J. Clin. Investig. 1985, 76, 1107–1112. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Ravussin, E. Analysis of energy metabolism in humans: A review of methodologies. Mol. Metab. 2016, 5, 1057–1071. [Google Scholar] [CrossRef]
- Caudwell, P.; Finlayson, G.; Gibbons, C.; Hopkins, M.; King, N.; Näslund, E.; Blundell, J.E. Resting metabolic rate is associated with hunger, self-determined meal size, and daily energy intake and may represent a marker for appetite. Am. J. Clin. Nutr. 2013, 97, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Stinson, E.J.; Graham, A.L.; Thearle, M.S.; Gluck, M.E.; Krakoff, J.; Piaggi, P. Cognitive dietary restraint, disinhibition, and hunger are associated with 24-h energy expenditure. Int. J. Obes. 2019, 43, 1456–1465. [Google Scholar] [CrossRef]
- Weise, C.M.; Hohenadel, M.G.; Krakoff, J.; Votruba, S.B. Body composition and energy expenditure predict ad-libitum food and macronutrient intake in humans. Int. J. Obes. 2014, 38, 243–251. [Google Scholar] [CrossRef] [Green Version]
- Piaggi, P.; Thearle, M.S.; Krakoff, J.; Votruba, S.B. Higher daily energy expenditure and respiratory quotient, rather than fat-free mass, independently determine greater ad libitum overeating. J. Clin. Endocrinol. Metab. 2015, 100, 3011–3020. [Google Scholar] [CrossRef] [Green Version]
- Dulloo, A.G.; Jacquet, J.; Miles-Chan, J.L.; Schutz, Y. Passive and active roles of fat-free mass in the control of energy intake and body composition regulation. Eur. J. Clin. Nutr. 2017, 71, 353–357. [Google Scholar] [CrossRef] [Green Version]
- Ravussin, E.; Lillioja, S.; Knowler, W.C.; Christin, L.; Freymond, D.; Abbott, W.G.; Boyce, V.; Howard, B.V.; Bogardus, C. Reduced rate of energy expenditure as a risk factor for body-weight gain. N. Engl. J. Med. 1988, 318, 467–472. [Google Scholar] [CrossRef]
- Buscemi, S.; Verga, S.; Caimi, G.; Cerasola, G. Low relative resting metabolic rate and body weight gain in adult Caucasian Italians. Int. J. Obes. 2005, 29, 287–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luke, A.; Durazo-Arvizu, R.; Cao, G.; Adeyemo, A.; Tayo, B.; Cooper, R. Positive association between resting energy expenditure and weight gain in a lean adult population. Am. J. Clin. Nutr. 2006, 83, 1076–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hales, C.N.; Barker, D.J.P. Type 2 (non-insulin-dependent) diabetes mellitus: The thrifty phenotype hypothesis. Diabetologia 1992, 35, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Basolo, A.; Votruba, S.B.; Heinitz, S.; Krakoff, J.; Piaggi, P. Deviations in energy sensing predict long-term weight change in overweight Native Americans. Metabolism 2018, 82, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, M.; Thearle, M.S.; Ibrahim, M.; Hohenadel, M.G.; Bogardus, C.; Krakoff, J.; Votruba, S.B. A Human Thrifty Phenotype Associated With Less Weight Loss During Caloric Restriction. Diabetes 2015, 64, 2859–2867. [Google Scholar] [CrossRef] [Green Version]
- Weyer, C.; Vozarova, B.; Ravussin, E.; Tataranni, P.A. Changes in energy metabolism in response to 48 h of overfeeding and fasting in Caucasians and Pima Indians. Int. J. Obes. 2001, 25, 593–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlögl, M.; Piaggi, P.; Pannacciuli, N.; Bonfiglio, S.M.; Krakoff, J.; Thearle, M.S. Energy expenditure responses to fasting and overfeeding identify phenotypes associated with weight change. Diabetes 2015, 64, 3680–3689. [Google Scholar] [CrossRef] [Green Version]
- Piaggi, P.; Vinales, K.L.; Basolo, A.; Santini, F.; Krakoff, J. Energy expenditure in the etiology of human obesity: Spendthrift and thrifty metabolic phenotypes and energy-sensing mechanisms. J. Endocrinol. Investig. 2018, 41, 83–89. [Google Scholar] [CrossRef]
- Hollstein, T.; Ando, T.; Basolo, A.; Krakoff, J.; Votruba, S.B.; Piaggi, P. Metabolic response to fasting predicts weight gain during low-protein overfeeding in lean men: Further evidence for spendthrift and thrifty metabolic phenotypes. Am. J. Clin. Nutr. 2019, 110, 593–604. [Google Scholar] [CrossRef]
- Reinhardt, M.; Schlögl, M.; Bonfiglio, S.; Votruba, S.B.; Krakoff, J.; Thearle, M.S. Lower core body temperature and greater body fat are components of a human thrifty phenotype. Int. J. Obes. 2016, 40, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Basolo, A.; Begaye, B.; Hollstein, T.; Vinales, K.L.; Walter, M.; Santini, F.; Krakoff, J.; Piaggi, P. Effects of Short-Term Fasting and Different Overfeeding Diets on Thyroid Hormones in Healthy Humans. Thyroid 2019, 29, 1209–1219. [Google Scholar] [CrossRef]
- Basolo, A.; Hollstein, T.; Walter, M.; Krakoff, J.; Piaggi, P. Urinary dopamine excretion rate decreases during acute dietary protein deprivation and is associated with increased plasma pancreatic polypeptide concentration. Nutrients 2021, 13, 1234. [Google Scholar] [CrossRef] [PubMed]
- Hollstein, T.; Vinales, K.; Chen, K.Y.; Cypess, A.M.; Basolo, A.; Schlögl, M.; Krakoff, J.; Piaggi, P. Reduced brown adipose tissue activity during cold exposure is a metabolic feature of the human thrifty phenotype. Metabolism 2021, 117, 154709. [Google Scholar] [CrossRef] [PubMed]
- Hollstein, T.; Heinitz, S.; Ando, T.; Rodzevik, T.L.; Basolo, A.; Walter, M.; Chang, D.C.; Krakoff, J.; Piaggi, P. Metabolic responses to 24-hour fasting and mild cold exposure in overweight individuals are correlated and accompanied by changes in FGF21 concentration. Diabetes 2020, 69, 1382–1388. [Google Scholar] [CrossRef]
- Vinales, K.L.; Schlögl, M.; Piaggi, P.; Hohenadel, M.; Graham, A.; Bonfiglio, S.; Krakoff, J.; Thearle, M.S. The Consistency in macronutrient oxidation and the role for epinephrine in the response to fasting and overfeeding. J. Clin. Endocrinol. Metab. 2017, 102, 279–289. [Google Scholar] [CrossRef] [Green Version]
- Heinitz, S.; Basolo, A.; Piaggi, P.; Piomelli, D.; von Schwartzenberg, R.J.; Krakoff, J. Peripheral Endocannabinoids associated with energy expenditure in native Americans of Southwestern Heritage. J. Clin. Endocrinol. Metab. 2018, 103, 1077–1087. [Google Scholar] [CrossRef]
- Maury, E.; Navez, B.; Brichard, S.M. Circadian clock dysfunction in human omental fat links obesity to metabolic inflammation. Nat. Commun. 2021, 12, 2388. [Google Scholar] [CrossRef]
- Serin, Y.; Tek, N.A. Effect of circadian rhythm on metabolic processes and the regulation of energy balance. Ann. Nutr. Metab. 2019, 74, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.G.; Sassone-Corsi, P. Clock-in, clock-out: Circadian timekeeping between tissues. Biochem 2020, 42, 6–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harder, L.; Oster, H. The Tissue Clock Network: Driver and gatekeeper of circadian physiology: Circadian rhythms are integrated outputs of central and peripheral tissue clocks interacting in a complex manner—from drivers to gatekeepers. BioEssays 2020, 42, 1900158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Güldür, T.; Otlu, H.G. Circadian rhythm in mammals: Time to eat & time to sleep. Biol. Rhythm Res. 2017, 48, 243–261. [Google Scholar]
- Hastings, M.H.; Reddy, A.B.; Maywood, E.S. A clockwork web: Circadian timing in brain and periphery, in health and disease. Nat. Rev. Neurosci. 2003, 4, 649–661. [Google Scholar] [CrossRef]
- Schibler, U.; Ripperger, J.; Brown, S.A. Peripheral circadian oscillators in mammals: Time and food. J. Biol. Rhythm. 2003, 18, 250–260. [Google Scholar] [CrossRef] [PubMed]
- De Assis, L.V.M.; Oster, H. The circadian clock and metabolic homeostasis: Entangled networks. Cell. Mol. Life Sci. 2021, 78, 4563–4587. [Google Scholar] [CrossRef]
- Richards, J.; Gumz, M.L. Advances in understanding the peripheral circadian clocks. FASEB J. 2012, 26, 3602–3613. [Google Scholar] [CrossRef] [Green Version]
- Roenneberg, T.; Merrow, M. The Circadian clock and human health. Curr. Biol. 2016, 26, R432–R443. [Google Scholar] [CrossRef]
- Rijo-Ferreira, F.; Takahashi, J.S. Genomics of circadian rhythms in health and disease. Genome Med. 2019, 11, 82. [Google Scholar] [CrossRef] [Green Version]
- Scheer, F.A.; Hilton, M.F.; Mantzoros, C.S.; Shea, S.A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl. Acad. Sci. USA 2009, 106, 4453–4458. [Google Scholar] [CrossRef] [Green Version]
- Hawley, J.A.; Sassone-Corsi, P.; Zierath, J.R. Chrono-nutrition for the prevention and treatment of obesity and type 2 diabetes: From mice to men. Diabetologia 2020, 63, 2253–2259. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Lee, Y.-C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.Q.; Ordovas, J.M. Genetic variants in human CLOCK associate with total energy intake and cytokine sleep factors in overweight subjects (GOLDN population). Eur. J. Hum. Genet. 2010, 18, 364–369. [Google Scholar] [CrossRef] [Green Version]
- Sookoian, S.; Gemma, C.; Gianotti, T.F.; Burgueño, A.; Castaño, G.; Pirola, C.J. Genetic variants of Clock transcription factor are associated with individual susceptibility to obesity. Am. J. Clin. Nutr. 2008, 87, 1606–1615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jha, P.K.; Challet, E.; Kalsbeek, A. Circadian rhythms in glucose and lipid metabolism in nocturnal and diurnal mammals. Mol. Cell. Endocrinol. 2015, 418, 74–88. [Google Scholar]
- Laposky, A.D.; Bass, J.; Kohsaka, A.; Turek, F.W. Sleep and circadian rhythms: Key components in the regulation of energy metabolism. FEBS Lett. 2008, 582, 142–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Potter, G.D.M.; Skene, D.J.; Arendt, J.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Circadian rhythm and sleep disruption: Causes, metabolic consequences, and countermeasures. Endocr. Rev. 2016, 37, 584–608. [Google Scholar] [CrossRef] [Green Version]
- Buxton, O.M.; Cain, S.W.; O’Connor, S.P.; Porter, J.H.; Duffy, J.F.; Wang, W.; Czeisler, C.A.; Shea, S.A. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci. Trans. Med. 2012, 4, 129ra43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.B.; Patterson, R.E.; Ang, A.; Emond, J.A.; Shetty, N.; Arab, L. Timing of energy intake during the day is associated with the risk of obesity in adults. J. Hum. Nutr. Diet. 2014, 27, 255–262. [Google Scholar] [CrossRef]
- Hermenegildo, Y.; López-García, E.; García-Esquinas, E.; Pérez-Tasigchana, R.F.; Rodríguez-Artalejo, F.; Guallar-Castillón, P. Distribution of energy intake throughout the day and weight gain: A population-based cohort study in Spain. Br. J. Nutr. 2016, 115, 2003–2010. [Google Scholar] [CrossRef] [Green Version]
- Bo, S.; Musso, G.; Beccuti, G.; Fadda, M.; Fedele, D.; Gambino, R.; Gentile, L.; Durazzo, M.; Ghigo, E.; Cassader, M. Consuming more of daily caloric intake at dinner predisposes to obesity. A 6-year population-based prospective cohort study. PLoS ONE. 2014, 9, e108467. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Barnea, M.; Wainstein, J.; Froy, O. High caloric intake at breakfast vs. dinner differentially influences weight loss of overweight and obese women. Obesity 2013, 21, 2504–2512. [Google Scholar] [CrossRef]
- Ruiz-Lozano, T.; Vidal, J.; de Hollanda, A.; Scheer, F.; Garaulet, M.; Izquierdo-Pulido, M. Timing of food intake is associated with weight loss evolution in severe obese patients after bariatric surgery. Clin. Nutr. 2016, 35, 1308–1314. [Google Scholar] [CrossRef] [Green Version]
- Thomas, E.A.; Zaman, A.; Cornier, M.-A.; Catenacci, V.A.; Tussey, E.J.; Grau, L.; Arbet, J.; Broussard, J.L.; Rynders, C.A. Later Meal and Sleep Timing Predicts Higher Percent Body Fat. Nutrients 2020, 13, 73. [Google Scholar] [CrossRef]
- Xiao, Q.; Garaulet, M.; Scheer, F.A.J.L. Meal timing and obesity: Interactions with macronutrient intake and chronotype. Int. J. Obes. 2019, 43, 1701–1711. [Google Scholar] [CrossRef] [PubMed]
- Jakubowicz, D.; Froy, O.; Wainstein, J.; Boaz, M. Meal timing and composition influence ghrelin levels, appetite scores and weight loss maintenance in overweight and obese adults. Steroids 2012, 77, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K.; Graubard, B.I. Secular trends in patterns of self-reported food consumption of adult Americans: NHANES 1971–1975 to NHANES 1999–2002. Am. J. Clin. Nutr. 2006, 84, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Aljuraiban, G.S.; Chan, Q.; Griep, L.M.O.; Brown, I.J.; Daviglus, M.L.; Stamler, J.; Van Horn, L.; Elliott, P.; Frost, G.S. The impact of eating frequency and time of intake on nutrient quality and Body Mass Index: The INTERMAP Study, a Population-Based Study. J. Acad. Nutr. Diet. 2015, 115, 528–536.e1. [Google Scholar] [CrossRef] [Green Version]
- Almoosawi, S.; Prynne, C.J.; Hardy, R.; Stephen, A.M. Time-of-day of energy intake: Association with hypertension and blood pressure 10 years later in the 1946 British Birth Cohort. J. Hypertens. 2013, 31, 882–892. [Google Scholar] [CrossRef]
- Summerbell, C.D.; Moody, R.C.; Shanks, J.; Stock, M.J.; Geissler, C. Relationship between feeding pattern and body mass index in 220 free-living people in four age groups. Eur. J. Clin. Nutr. 1996, 50, 513–519. [Google Scholar]
- Kant, A.K.; Ballard-Barbash, R.; Schatzkin, A. Evening eating and its relation to self-reported body weight and nutrient intake in women, CSFII 1985–86. J. Am. Coll. Nutr. 1995, 14, 358–363. [Google Scholar] [CrossRef]
- Striegel-Moore, R.H.; Franko, D.L.; Thompson, D.; Affenito, S.; Kraemer, H.C. Night eating: Prevalence and demographic correlates. Obesity 2006, 14, 139–147. [Google Scholar] [CrossRef] [Green Version]
- Fong, M.; Caterson, I.D.; Madigan, C.D. Are large dinners associated with excess weight, and does eating a smaller dinner achieve greater weight loss? A systematic review and meta-analysis. Br. J. Nutr. 2017, 118, 616–628. [Google Scholar] [CrossRef]
- Spengler, C.M.; Czeisler, C.A.; Shea, S.A. An endogenous circadian rhythm of respiratory control in humans. J. Physiol. 2000, 526, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Wirz-Justice, A. Circadian rhythm of heat production, heart rate, and skin and core temperature under unmasking conditions in men. Am. J. Physiol. 1994, 267, R819–R829. [Google Scholar] [CrossRef] [PubMed]
- Haugen, H.A.; Melanson, E.L.; Tran, Z.V.; Kearney, J.T.; Hill, J.O. Variability of measured resting metabolic rate. Am. J. Clin. Nutr. 2003, 78, 1141–1144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bandín, C.; Scheer, F.A.J.L.; Luque, A.J.; Ávila-Gandía, V.; Zamora, S.; Madrid, J.A.; Gómez-Abellán, P.; Garaulet, M. Meal timing affects glucose tolerance, substrate oxidation and circadian-related variables: A randomized, crossover trial. Int. J. Obes. 2015, 39, 828–833. [Google Scholar] [CrossRef]
- Sato, M.; Nakamura, K.; Ogata, H.; Miyashita, A.; Nagasaka, S.; Omi, N.; Yamaguchi, S.; Hibi, M.; Umeda, T.; Nakaji, S.; et al. Acute effect of late evening meal on diurnal variation of blood glucose and energy metabolism. Obes. Res. Clin. Pract. 2011, 5, e220–e228. [Google Scholar] [CrossRef]
- Betts, J.A.; Richardson, J.D.; Chowdhury, E.A.; Holman, G.D.; Tsintzas, K.; Thompson, D. The causal role of breakfast in energy balance and health: A randomized controlled trial in lean adults. Am. J. Clin. Nutr. 2014, 100, 539–547. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, E.A.; Richardson, J.D.; Holman, G.D.; Tsintzas, K.; Thompson, D.; Betts, J.A. The causal role of breakfast in energy balance and health: A randomized controlled trial in obese adults. Am. J. Clin. Nutr. 2016, 103, 747–756. [Google Scholar] [CrossRef]
- Farshchi, H.R.; Taylor, M.A.; Macdonald, I.A. Deleterious effects of omitting breakfast on insulin sensitivity and fasting lipid profiles in healthy lean women. Am. J. Clin. Nutr. 2005, 81, 388–396. [Google Scholar] [CrossRef] [Green Version]
- Romon, M.; Edme, J.L.; Boulenguez, C.; Lescroart, J.L.; Frimat, P. Circadian variation of diet-induced thermogenesis. Am. J. Clin. Nutr. 1993, 57, 476–480. [Google Scholar] [CrossRef]
- Morris, C.J.; Garcia, J.I.; Myers, S.; Yang, J.N.; Trienekens, N.; Scheer, F.A. The human circadian system has a dominating role in causing the morning/evening difference in diet-induced thermogenesis. Obesity 2015, 23, 2053–2058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bo, S.; Fadda, M.; Castiglione, A.; Ciccone, G.; De Francesco, A.; Fedele, D.; Guggino, A.; Parasiliti Caprino, M.; Ferrara, S.; Vezio Boggio, M.; et al. Is the timing of caloric intake associated with variation in diet-induced thermogenesis and in the metabolic pattern? A randomized cross-over study. Int. J. Obes. 2015, 39, 1689–1695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richter, J.; Herzog, N.; Janka, S.; Baumann, T.; Kistenmacher, A.; Oltmanns, K.M. Twice as high diet-induced thermogenesis after breakfast vs dinner on high-calorie as well as low-calorie meals. J. Clin. Endocrinol. Metab. 2020, 105, e211–e221. [Google Scholar] [CrossRef]
- Carpentier, A.C.; Blondin, D.P.; Virtanen, K.A.; Richard, D.; Haman, F.; Turcotte, É.E. Brown adipose tissue energy metabolism in humans. Front. Endocrinol. 2018, 9, 447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsushita, M.; Nirengi, S.; Hibi, M.; Wakabayashi, H.; Lee, S.I.; Domichi, M.; Sakane, N.; Saito, M. Diurnal variations of brown fat thermogenesis and fat oxidation in humans. Int. J. Obes. 2021, 1–7, Online ahead of print. [Google Scholar]
- Weststrate, J.A.; Weys, P.J.; Poortvliet, E.J.; Deurenberg, P.; Hautvast, J.G. Diurnal variation in postabsorptive resting metabolic rate and diet-induced thermogenesis. Am. J. Clin. Nutr. 1989, 50, 908–914. [Google Scholar] [CrossRef]
- Zwiauer, K.F.; Mueller, T.; Widhalm, K. Effect of daytime on resting energy expenditure and thermic effect of food in obese adolescents. J. Am. Coll. Nutr. 1992, 11, 267–271. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Lo, S.K.; Westerterp, K.R.; Rush, E.C.; Rosenbaum, M.; Luke, A.; Schoeller, D.A.; DeLany, J.P.; Butte, N.F.; et al. Estimating the changes in energy flux that characterize the rise in obesity prevalence. Am. J. Clin. Nutr. 2009, 89, 1723–1728. [Google Scholar] [CrossRef]
- Zitting, K.-M.; Vujovic, N.; Yuan, R.K.; Isherwood, C.M.; Medina, J.E.; Wang, W.; Buxton, O.M.; Williams, J.S.; Czeisler, C.A.; Duffy, J.F. Human resting energy expenditure varies with circadian phase. Curr. Biol. 2018, 28, 3685–3690.e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmbäck, U.; Forslund, A.; Forslund, J.; Hambraeus, L.; Lennernäs, M.; Lowden, A.; Stridsberg, M.; Akerstedt, T. Metabolic responses to nocturnal eating in men are affected by sources of dietary energy. J. Nutr. 2002, 132, 1892–1899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravussin, E.; Beyl, R.A.; Poggiogalle, E.; Hsia, D.S.; Peterson, C.M. Early time-restricted feeding reduces appetite and increases fat oxidation but does not affect energy expenditure in humans. Obesity 2019, 27, 1244–1254. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basolo, A.; Bechi Genzano, S.; Piaggi, P.; Krakoff, J.; Santini, F. Energy Balance and Control of Body Weight: Possible Effects of Meal Timing and Circadian Rhythm Dysregulation. Nutrients 2021, 13, 3276. https://doi.org/10.3390/nu13093276
Basolo A, Bechi Genzano S, Piaggi P, Krakoff J, Santini F. Energy Balance and Control of Body Weight: Possible Effects of Meal Timing and Circadian Rhythm Dysregulation. Nutrients. 2021; 13(9):3276. https://doi.org/10.3390/nu13093276
Chicago/Turabian StyleBasolo, Alessio, Susanna Bechi Genzano, Paolo Piaggi, Jonathan Krakoff, and Ferruccio Santini. 2021. "Energy Balance and Control of Body Weight: Possible Effects of Meal Timing and Circadian Rhythm Dysregulation" Nutrients 13, no. 9: 3276. https://doi.org/10.3390/nu13093276
APA StyleBasolo, A., Bechi Genzano, S., Piaggi, P., Krakoff, J., & Santini, F. (2021). Energy Balance and Control of Body Weight: Possible Effects of Meal Timing and Circadian Rhythm Dysregulation. Nutrients, 13(9), 3276. https://doi.org/10.3390/nu13093276






