Intermittent Fasting and the Possible Benefits in Obesity, Diabetes, and Multiple Sclerosis: A Systematic Review of Randomized Clinical Trials
Abstract
:1. Introduction
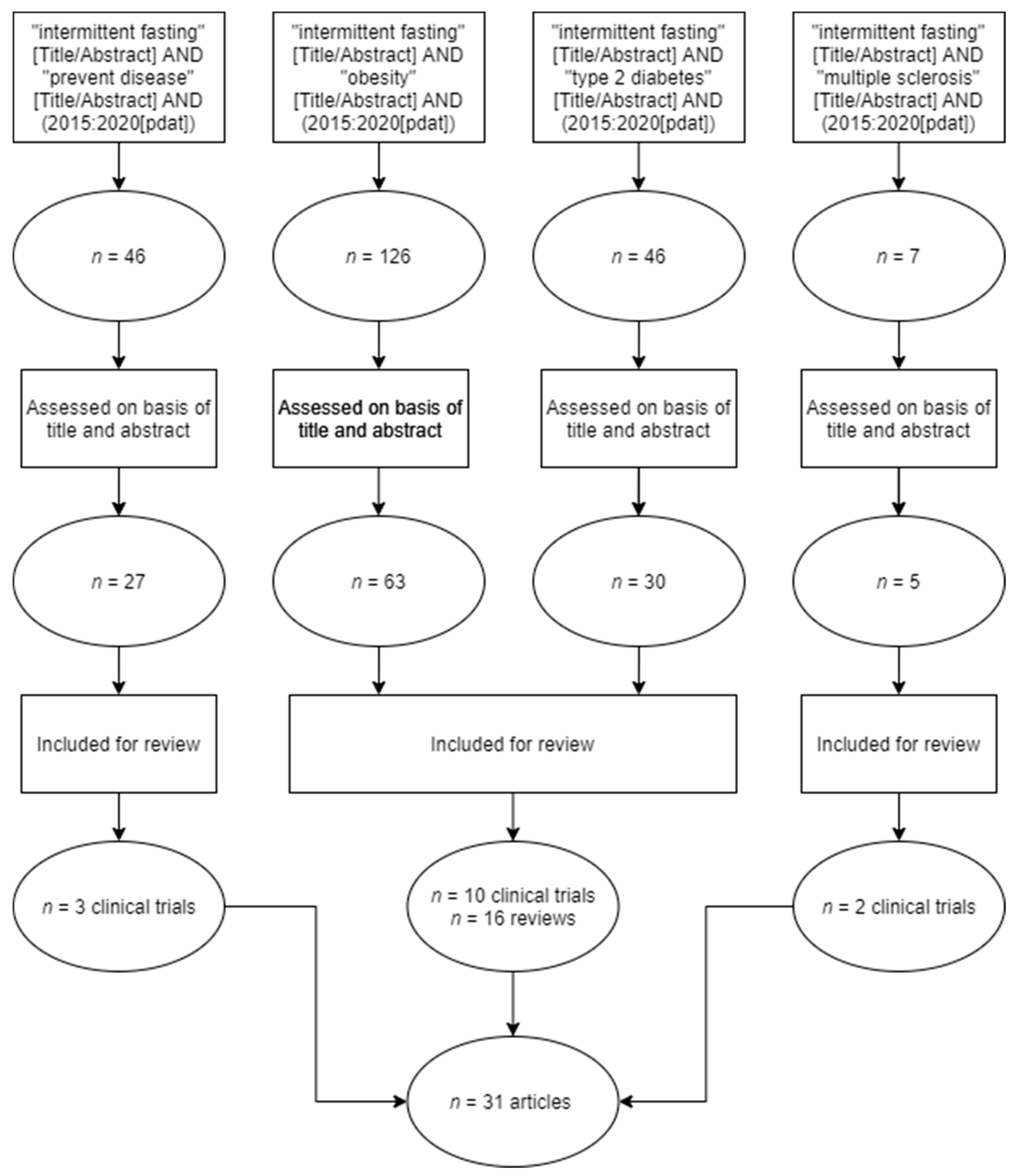
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Correia, J.M.; Santos, I.; Pezarat-Correia, P.; Minderico, C.; Mendonca, G.V. Effects of intermittent fasting on specific exercise performance outcomes: A systematic review including meta-analysis. Nutrients 2020, 12, 1390. [Google Scholar] [CrossRef] [PubMed]
- Welton, S.; Minty, R.; O’Driscoll, T.; Willms, H.; Poirier, D.; Madden, S.; Kelly, L. Intermittent fasting and weight loss: Systematic review. Can. Fam. Physician 2020, 66, 117–125. [Google Scholar]
- Wilhelmi de Toledo, F.; Grundler, F.; Sirtori, C.R.; Ruscica, M. Unravelling the health effects of fasting: A long road from obesity treatment to healthy life span increase and improved cognition. Ann. Med. 2020, 52, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Vitale, R.; Kim, Y. The effects of intermittent fasting on glycemic control and body composition in adults with obesity and type 2 diabetes: A systematic review. Metab. Syndr. Relat. Disord 2020, 18, 450–461. [Google Scholar] [CrossRef]
- Hawley, J.A.; Sassone-Corsi, P.; Zierath, J.R. Chrono-nutrition for the prevention and treatment of obesity and type 2 diabetes: From mice to men. Diabetologia 2020, 63, 2253–2259. [Google Scholar] [CrossRef]
- Liu, B.; Hutchison, A.T.; Thompson, C.H.; Lange, K.; Heilbronn, L.K. Markers of adipose tissue inflammation are transiently elevated during intermittent fasting in women who are overweight or obese. Obes. Res. Clin. Pract. 2019, 13, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Golbidi, S.; Daiber, A.; Korac, B.; Li, H.; Essop, M.F.; Laher, I. Health benefits of fasting and caloric restriction. Curr. Diab. Rep. 2017, 17, 123. [Google Scholar] [CrossRef]
- Borgundvaag, E.; Mak, J.; Kramer, C.K. Metabolic impact of intermittent fasting in patients with type 2 diabetes mellitus: A systematic review and meta-analysis of interventional studies. J. Clin. Endocrinol. Metab. 2021, 106, 902–911. [Google Scholar] [CrossRef]
- Cignarella, F.; Cantoni, C.; Ghezzi, L.; Salter, A.; Dorsett, Y.; Chen, L.; Phillips, D.; Weinstock, G.M.; Fontana, L.; Cross, A.H.; et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. 2018, 27, 1222–1235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Scottish Intercollegiate Guidelines Network (SIGN). SIGN 50: A Guideline Developer’s Handbook; Scottish Intercollegiate Guidelines Network: Edinburgh, UK, 2015. [Google Scholar]
- Washburn, R.L.; Cox, J.E.; Muhlestein, J.B.; May, H.T.; Carlquist, J.F.; Le, V.T.; Anderson, J.L.; Horne, B.D. Pilot study of novel intermittent fasting effects on metabolomic and trimethylamine N-oxide changes during 24-hour water-only fasting in the FEELGOOD Trial. Nutrients 2019, 11, 246. [Google Scholar] [CrossRef] [Green Version]
- Stekovic, S.; Hofer, S.J.; Tripolt, N.; Aon, M.A.; Royer, P.; Pein, L.; Madeo, F. Alternate day fasting improves physiological and molecular markers of aging in healthy, non-obese humans. Cell Metab. 2019, 30, 462–476. [Google Scholar] [CrossRef] [PubMed]
- McAllister, M.J.; Pigg, B.L.; Renteria, L.I.; Waldman, H.S. Time-restricted feeding improves markers of cardiometabolic health in physically active college-age men: A 4-week randomized pre-post pilot study. Nutr. Res. 2020, 75, 32–43. [Google Scholar] [CrossRef]
- Sutton, E.F.; Beyl, R.; Early, K.S.; Cefalu, W.T.; Ravussin, E.; Peterson, C.M. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab. 2018, 27, 1212–1221. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Sadraie, B.; Steckhan, N.; Kessler, C.; Stange, R.; Jeitler, M.; Michalsen, A. Effects of a one-week fasting therapy in patients with type-2 diabetes mellitus and metabolic syndrome–A randomized controlled explorative study. Exp. Clin. Endocrinol. Diabetes 2017, 125, 618–624. [Google Scholar] [CrossRef]
- Antoni, R.; Johnston, K.L.; Collins, A.L.; Robertson, M.D. Investigation into the acute effects of total and partial energy restriction on postprandial metabolism among overweight/obese participants. Br. J. Nutr. 2016, 115, 951–959. [Google Scholar] [CrossRef] [Green Version]
- Sundfør, T.; Svendsen, M.; Tonstad, S. Effect of intermittent versus continuous energy restriction on weight loss, maintenance and cardiometabolic risk: A randomized 1-year trial. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 698–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamshed, H.; Beyl, R.A.; Della Manna, D.L.; Yang, E.S.; Ravussin, E.; Peterson, C.M. Early time-restricted feeding improves 24-hour glucose levels and affects markers of the circadian clock, aging, and autophagy in humans. Nutrients 2019, 11, 1234. [Google Scholar] [CrossRef] [Green Version]
- Anton, S.D.; Lee, S.A.; Donahoo, W.T.; McLaren, C.; Manini, T.; Leeuwenburgh, C.; Pahor, M. The Effects of Time Restricted Feeding on Overweight, Older Adults: A Pilot Study. Nutrients 2019, 11, 1500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jospe, M.R.; Roy, M.; Brown, R.C.; Haszard, J.J.; Meredith-Jones, K.; Fangupo, L.J.; Osborne, H.; Fleming, E.A.; Taylor, R.W. Intermittent fasting, paleolithic, or mediterranean diets in the real world: Exploratory secondary analyses of a weight-loss trial that included choice of diet and exercise. Am. J. Clin. Nutr. 2020, 111, 503–514. [Google Scholar] [CrossRef]
- Harney, D.J.; Hutchison, A.T.; Hatchwell, L.; Humphrey, S.J.; James, D.E.; Hocking, S.; Heilbronn, L.K.; Larance, M. Proteomic analysis of human plasma during intermittent fasting. J. Proteome Res. 2019, 18, 2228–2240. [Google Scholar] [CrossRef]
- Kim, C.; Pinto, A.M.; Bordoli, C.; Buckner, L.P.; Kaplan, P.C.; del Arenal, I.M.; Jeffcock, E.J.; Hall, W.L.; Thuret, S. Energy Restriction Enhances Adult Hippocampal Neurogenesis-Associated Memory after Four Weeks in an Adult Human Population with Central Obesity; a Randomized Controlled Trial. Nutrients 2020, 12, 638. [Google Scholar] [CrossRef] [Green Version]
- Fitzgerald, K.C.; Vizthum, D.; Henry-Barron, B.; Vizthum, D.; Henry-Barron, B.; Schweitzer, A.; Cassard, S.D.; Kossoff, E.; Hartman, A.L.; Kapogiannis, D.; et al. Effect of intermittent vs. daily calorie restriction on changes in weight and patient-reported outcomes in people with multiple sclerosis. Mult. Scler. Relat. Dis. 2018, 23, 33–39. [Google Scholar] [CrossRef]
- Harris, L.; Hamilton, S.; Azevedo, L.B.; Olajide, J.; De Brún, C.; Waller, G.; Whittaker, V.; Sharp, T.; Lean, M.; Hankey, C.; et al. Intermittent fasting interventions for treatment of overweight and obesity in adults: A systematic review and meta-analysis. JBI Database Syst. Rev. Implement. Rep. 2018, 16, 507–547. [Google Scholar] [CrossRef] [Green Version]
- Zubrzycki, A.; Cierpka-Kmiec, K.; Kmiec, Z.; Wronska, A. The role of low-calorie diets and intermittent fasting in the treatment of obesity and type-2 diabetes. J. Physiol. Pharmacol. 2018, 69, 663–683. [Google Scholar]
- Malinowski, B.; Zalewska, K.; Węsierska, A.; Sokołowska, M.M.; Socha, M.; Liczner, G.; Pawlak-Osińska, K.; Wiciński, M. Intermittent fasting in cardiovascular disorders—An overview. Nutrients 2019, 11, 673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.; Seo, Y.G.; Paek, Y.J.; Song, H.J.; Park, K.H.; Noh, H.M. Effect of alternate-day fasting on obesity and cardiometabolic risk: A systematic review and meta-analysis. Metabolism 2020, 111, 154336. [Google Scholar] [CrossRef] [PubMed]
- Waldman, H.S.; Renteria, L.I.; McAllister, M.J. Time-restricted feeding for the prevention of cardiometabolic diseases in high-stress occupations: A mechanistic review. Nutr. Rev. 2020, 78, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, Q.; Liao, Q.; Li, M.; Zhang, P.; Santos, H.O.; Kord-Varkaneh, H.; Abshirini, M. Effects of intermittent fasting diets on plasma concentrations of inflammatory biomarkers: A systematic review and meta-analysis of randomized controlled trials: Fasting and inflammation. Nutrition 2020, 79–80, 110974. [Google Scholar] [CrossRef]
- Meng, H.; Zhu, L.; Kord-Varkaneh, H.; Santos, H.O.; Tinsley, G.M.; Fu, P. Effects of intermittent fasting and energy-restricted diets on lipid profile: A systematic review and meta-analysis. Nutrition 2020, 77, 110801. [Google Scholar] [CrossRef] [PubMed]
- Pureza, I.R.O.M.; Macena, M.L.; da Silva Junior, A.E.; Praxedes, D.R.S.; Vasconcelos, L.G.L.; Bueno, N.B. Effect of early time-restricted feeding on the metabolic profile of adults with excess weight: A systematic review with meta-analysis. Clin. Nutr. 2021, 40, 1788–1799. [Google Scholar] [CrossRef] [PubMed]
- Asif, S.; Morrow, N.M.; Mulvihill, E.E.; Kim, K. Understanding dietary intervention-mediated epigenetic modifications in metabolic diseases. Front. Genet. 2020, 11, 590369. [Google Scholar] [CrossRef] [PubMed]
- Adafer, R.; Messaadi, W.; Meddahi, M.; Patey, A.; Haderbache, A.; Bayen, S.; Messaadi, N. Food timing, circadian rhythm and chrononutrition: A systematic review of time-restricted eating’s effects on human health. Nutrients 2020, 12, 3770. [Google Scholar] [CrossRef] [PubMed]
| Authors, Year and Country | Study Duration | Sample Size | Type of Study | Exhibition Time | The Purpose of the Study | Main Effects Observed | LE | GR |
|---|---|---|---|---|---|---|---|---|
| Washburn et al., 2019 [13] Utah, USA | - | n = 30 | Randomized clinical trial | 2 days | To elucidate some of the mechanisms for the different benefits attributed to IF in relation to disease, through secondary analyzes of fasting and longevity gene expression during food abstinence trial | A decrease in mean trimethylamine N-oxide was observed, although levels returned to baseline on refeeding. Acute alterations in levels of proline, tyrosine, galactitol, and urea plasma levels were observed along with changes in 24 other metabolites during the fasting period. | 1++ | A |
| Stekovic et al., 2019 [14] Graz, Austria | 2015–2017 | n = 60 | Randomized clinical trial | 4 weeks | To clarify to what extent IF influences human physiology in healthy individuals both in the short and long term | Improved cardiovascular markers, reduced fat mass, and increased β-hydroxybutyrate. On fasting days, the pro-aging amino-acid methionine, among others, was periodically depleted, while polyunsaturated fatty acids were elevated. Reduced levels of age-associated inflammatory marker, low-density lipoprotein, and triiodothyronine. | 1++ | A |
| McAllister et al., 2019 [15] Texas, USA | - | n = 23 22 patients finished the trial | Randomized clinical trial | 28 days | To examine the impact of time-restricted eating on markers of cardio-metabolic health and antioxidant status and to determine whether adaptations of this diet would differ ad libitum compared to isocaloric conditions. | IF was associated with significant reductions in body fat, blood pressure, and significant increases in adiponectin and HDL-cholesterol. No changes in caloric intake were detected. | 1++ | A |
| Authors, Year and Country | Study Duration | Sample Size | Type of Study | Exhibition Time | The Purpose of the Study | Main Effects Observed | LE | GR |
|---|---|---|---|---|---|---|---|---|
| Sutton et al., 2018 [16] Los Angeles, USA | 2013–2016 | n =12 8 patients finished the trial | Randomized clinical trial | 5 weeks | To find out the effects of early time-restricted feeding on insulin sensitivity, blood pressure, and oxidative stress in men with prediabetes. | Improved insulin sensitivity, β-cell responsiveness, blood pressure, oxidative stress, and appetite. | 1++ | A |
| Li et al., 2017 [17] Berlin, Germany | 2015 | n = 46 36 patients finished the trial | Randomized clinical trial | 7 days | To investigate the effects of a one-week fasting period compared to usual care in type 2 diabetes mellitus using a pilot trial. | Decreased mean weight, reduction of abdominal circumference, decrease of systolic/diastolic blood pressure, and increased quality of life. No improvement in HbA1c, insulin, and Homeostatic Model Assessment for Insulin Resistance index. | 1++ | A |
| Antoni et al., 2016 [18] Guilford, UK | - | n = 14 10 patients finished the trial | Randomized clinical trial | 3 days | To investigate the early metabolic response to various degrees of energy restriction, which occurs acutely and before weight loss. | Increased postprandial glucose responses, reductions in postprandial triacylglycerol responses, and 3-day energy intake deficits. | 1++ | A |
| Sundfør et al., 2018 [19] Oslo, Norway | 2015–2017 | n = 112 | Randomized clinical trial | 12 months | To compare the effects of intermittent versus continuous energy restriction in relation to weight loss, maintenance, and cardiometabolic risk factors in adults with abdominal obesity and ≥1 additional component of metabolic syndrome. | Improvement in weight loss, maintenance, and cardiovascular risk factors (waist circumference, blood pressure, triglycerides, and HDL-cholesterol) after one year but with no differences between intermittent and continuous energy restriction. | 1++ | A |
| Harney et al., 2019 [23] Adelaida, Australia | 2013–2015 | n = 88 85 patients finished the trial | Randomized clinical trial | 10 weeks (2 weeks with normal diet + 8 weeks with intermittent fasting) | To perform a proteomic analysis of human plasma during IF in sedentary people. | Increased apolipoprotein A4 and clusterin and decreased apolipoprotein C2, apolipoprotein A2, C3, and plasma triglycerides. | 1++ | A |
| Jamshed et al., 2019 [20] Birmingham, USA | - | n = 11 | Randomized clinical trial | 4 days | To determine how time-restricted feeding affects gene expression, circulating hormones, and diurnal patterns in cardiometabolic risk factors in humans. | Decreased mean 24-h glucose and glycemic excursions, altered lipid metabolism, and circadian clock gene expression. | 1++ | A |
| Anton et al., 2019 [21] Florida USA | - | n = 10 | Randomized clinical trial | 4 weeks | To assess the safety and feasibility of time-restricted feeding in an overweight sedentary older adult population. | Decreased body weight, no significant changes in other outcome (waist circumference, cognitive and physical function, health-related quality of life, and adverse events) except for clinically meaningful changes in walking speed and improvements in quality of life, with few reported adverse events. | 1++ | A |
| Liu et al., 2019 [6] Adelaida, Australia | - | n = 76 | Randomized clinical trial | 8 weeks | To compare the effects of daily caloric restriction vs. IF on markers of inflammation and extracellular matrix deposition in adipose tissue and skeletal muscle in a controlled feeding trial in overweight or obese women. | Markers of inflammation in serum, subcutaneous adipose tissue, and skeletal muscle were unchanged after fed days. After fasting, non-esterified fatty acids (NEFA), M1-macrophages in adipose tissue and M2-macrophages in muscle were increased, and the changes in NEFA and mRNA of pan-macrophage marker CD68 in adipose tissue were positively correlated. | 1++ | A |
| Kim et al., 2020 [24] London, UK | 2016 | n = 43 | Randomized clinical trial | 4 weeks | To investigate the effects of intermittent and continuous energy restriction on cognition related to the neurogenesis of the human hippocampus. | Significantly improved pattern separation and significant deterioration in recognition memory. | 1++ | A |
| Jospe et al., 2020 [22] Dunedin, New Zealand | 2014–2015 | n = 250 | Randomized clinical trial | 12 weeks | To investigate the implication of dietary intake, weight loss, and metabolic outcomes in overweight adults who could choose to follow Mediterranean diets, intermittent fasting, and standard exercise or high-intensity interval training (HIIT) programs. | Weight loss and reduced systolic blood pressure. | 1++ | A |
| Authors, Year and Country | Study Duration | Sample Size | Type of Study | Exhibition Time | The Purpose of the Study | Main Effects Observed | LE | GR |
|---|---|---|---|---|---|---|---|---|
| Cignarella et al., 2018 [9] Washington, USA | 2014–2016 | Animal study: 10 mice. Study with patients: 17 subjects, and 1 did not complete it | Randomized clinical trial | In mice (4 weeks) In patients (2 weeks) | To assess the safety, feasibility, and compliance of IF in patients with MS. | Altered blood adipokines and the gut flora resembling protective changes observed in mice. | 1++ | A |
| Fitzgerald et al., 2018 [25] Baltimore, USA | 2015–2016 | n = 36 31 patients finished the trial | Randomized clinical trial | 8 weeks | To assess the safety and feasibility of different types of diets related to IF in patients with MS. | Weight loss and significant improvements in emotional well-being/depression scores relative to control. | 1++ | A |
| Authors, Year and Country | Type of Study | Analyzed Study | The Purpose of the Study | Main Effects Observed | LE | GR |
|---|---|---|---|---|---|---|
| Golbidi et al., 2017 [7] Vancouver, Canada | Review | Non-analytical studies, such as a case report or case series | To review different mechanisms related to fasting, aimed at controlling metabolic diseases, particularly diabetes. | IF is associated increased transcription of stress-induced proteins, cellular autophagy, reduced advance glycation end-products, increased adiponectin levels, reduced adipocyte size, lower body weight, and better diabetes control. | 3 | D |
| Harris et al., 2018 [26] Glasgow, UK | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with high risk of bias | To examine the effectiveness of IF in treating overweight and obesity in adults compared to usual care treatment or no treatment. | Intermittent energy restriction was more effective than no treatment for weight loss although no significant difference in weight loss was observed in comparison to continuous energy restriction. | 1− | A |
| Zubrzycki et al., 2018 [27] Gdansk, Poland | Systematic review | Non-analytical studies, such as a case report or case series | To find out the role of low-calorie diets and IF in the treatment of obesity and type 2 diabetes. | Low-calorie diets and IF in patients with obesity (including those with coexisting type 2 diabetes) can lead to a reduction in body fat mass and metabolic parameter improvements. | 3 | D |
| Malinowski et al., 2019 [28] Bidgostia, Poland | Review | Non-analytical studies, such as a case report or case series | To study the effects of IF on the cardiovascular system, including the progression of atherosclerosis, the benefits for type 2 diabetes mellitus, a decrease in blood pressure, and the exploration of other cardiovascular risk factors (such as lipid profile and inflammation). | IF inhibits the development of atherosclerotic plaque. The IF diet causes an increase of brain-derived neurotrophic factor (BDNF), which results in lowering the systolic and diastolic blood pressure by activating the parasympathetic system. The reduced amount of food consumed when using the IF diet results in a decrease in body weight. It also improves glucose metabolism and increases the sensitivity of tissues to insulin by increasing the β cells of the pancreatic islets. The IF diet also limits cardiac hypertrophy. | 3 | D |
| Welton et al., 2020 [2] Canada | Systematic review | Randomized clinical trials or randomized clinical trials with low risk of bias | To examine the evidence for intermittent fasting, an alternative to calorie restricted diets, in the treatment of obesity. | Weight loss of 0.8% to 13.0% of baseline weight with no serious adverse events and improved glycemic control. | 1+ | A |
| Correia et al., 2020 [1] Lisboa, Portugal | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with low risk of bias | To summarize the current evidence on the interaction between IF during Ramadan vs time-restricted eating and specific physical performance parameters, such as VO2max, vertical jump height, distance in 30 min of running, power output. Wingate maximum and mean. | Maximum oxygen uptake is significantly enhanced with time-restricted eating but reduced with Ramadan intermittent fasting. Additional effects may be observed in body mass and fat mass. Non-significant effects were observed for muscle strength and anaerobic capacity. While Ramadan may lead to impairments in aerobic capacity, time-restricted eating may be effective for improving it. | 1+ | A |
| Vitale et al., 2020 [4] Michigan, USA | Systematic review | Randomized clinical trials or randomized clinical trials with low risk of bias | To assess the effects of IF on glycemic control and body composition in adults with obesity and type 2 diabetes. | Strong evidence to support IF as a feasible diet to improve glycemia and body composition measures within 12–24 weeks. Follow-up 12–18 months after IF did not show promising results for continued weight loss and improved glycemic control. | 1+ | A |
| Park et al., 2020 [29] Anyang, South Korea | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with low risk of bias | To assess the effects of IF on obesity-related factors and cardiometabolic risk factors in adults. | Significant improvements in BMI, body weight, body fat mass, and total cholesterol. | 1+ | A |
| Waldman et al., 2020 [30] Alabama, USA | Review | Non-analytical studies, such as a case report or case series | To research the benefits of time-restricted eating in relation to the prevention of cardiometabolic diseases in high-stress occupations, such as police, firefighters, and the military. | The timing of the feeding-fasting window, with feeding taking place in the waking hours and fasting in the evening hours, might offer the greatest benefit for improving cardiometabolic markers. | 3 | D |
| Wang et al., 2020 [31] Zhejiang, China | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with high risk of bias | To assess the effects of IF or diets with dietary restriction on plasma concentrations of inflammatory biomarkers. | Significantly reduced C-reactive protein concentrations but did not significantly reduce tumor necrosis factor-α and interleukin-6 concentrations. | 1− | A |
| Meng et al., 2020 [32] Shandog, China | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with high risk of bias | To summarize the effects of controlled clinical trials examining the influence of IF and dietary restriction diets on lipid profiles. | Significant changes in total cholesterol, low-density lipoprotein cholesterol, and triacylglycerols concentrations and no change in high-density lipoprotein cholesterol. | 1− | A |
| Pureza et al., 2020 [33] São Paulo, Brasil | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with high risk of bias | To assess the effect of early time-restricted feeding (eTRF) on the metabolic profile of overweight adults. | Significant effects on the fasting blood glucose and Homeostatic Model Assessment for Insulin Resistance index. | 1− | A |
| Hawley et al., 2020 [5] Melbourne, Australia | Review | Non-analytical studies, such as a case report or case series | To find out the effects of chrononutrition for the prevention and treatment of obesity and type 2 diabetes in mice and humans. | While data from studies of time-restricted eating in animals and a small number of clinical populations are encouraging, there is a need for multicenter, randomized, clinical trials of comparisons between different eating patterns across a range of human cohorts to determine the most efficacious intervention. | 3 | D |
| Borgundvaag et al., 2020 [8] Toronto, Canada | Systematic review with meta-analysis | Randomized clinical trials or randomized clinical trials with high risk of bias | To assess the metabolic impact of IF compared to a standard diet in patients with type 2 diabetes mellitus. | IF induced a greater decrease in body weight especially in heavier patients and in studies of shorter duration but was not associated with reduction in HbA1c. | 1− | A |
| Asif et al., 2020 [34] Ottawa, Canada | Review | Non-analytical studies, such as a case report or case series | To review the epigenetic changes implicated in both metabolic diseases and dietary interventions in primary metabolic tissues (i.e., adipose tissue, liver, and pancreas) with the hope of pinpointing potential biomarkers and therapeutic targets for prevention and treatment of disease. | The global metabolic changes seen in several tissues upon disease and dietary intervention make it difficult to not only characterize the adaptive or pathological role of these epigenetic events but to also pinpoint the primary insult that triggers secondary, systemic aspects of these responses. | 3 | D |
| Adafer et al., 2020 [35] Lille, France | Systematic review | Randomized clinical trials or randomized clinical trials with high risk of bias | To review how time-restricted eating affects human health. | Decreased body weight and fat mass with beneficial metabolic effects independent of weight loss. | 1− | A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales-Suarez-Varela, M.; Collado Sánchez, E.; Peraita-Costa, I.; Llopis-Morales, A.; Soriano, J.M. Intermittent Fasting and the Possible Benefits in Obesity, Diabetes, and Multiple Sclerosis: A Systematic Review of Randomized Clinical Trials. Nutrients 2021, 13, 3179. https://doi.org/10.3390/nu13093179
Morales-Suarez-Varela M, Collado Sánchez E, Peraita-Costa I, Llopis-Morales A, Soriano JM. Intermittent Fasting and the Possible Benefits in Obesity, Diabetes, and Multiple Sclerosis: A Systematic Review of Randomized Clinical Trials. Nutrients. 2021; 13(9):3179. https://doi.org/10.3390/nu13093179
Chicago/Turabian StyleMorales-Suarez-Varela, María, Ester Collado Sánchez, Isabel Peraita-Costa, Agustín Llopis-Morales, and José M. Soriano. 2021. "Intermittent Fasting and the Possible Benefits in Obesity, Diabetes, and Multiple Sclerosis: A Systematic Review of Randomized Clinical Trials" Nutrients 13, no. 9: 3179. https://doi.org/10.3390/nu13093179
APA StyleMorales-Suarez-Varela, M., Collado Sánchez, E., Peraita-Costa, I., Llopis-Morales, A., & Soriano, J. M. (2021). Intermittent Fasting and the Possible Benefits in Obesity, Diabetes, and Multiple Sclerosis: A Systematic Review of Randomized Clinical Trials. Nutrients, 13(9), 3179. https://doi.org/10.3390/nu13093179







