Diet, Probiotics and Their Impact on the Gut Microbiota during the COVID-19 Pandemic
Abstract
:1. Introduction
2. Gut Microbiome Alterations and COVID-19 Disease Severity
3. Diet and Probiotics in SARS-CoV-2 infection
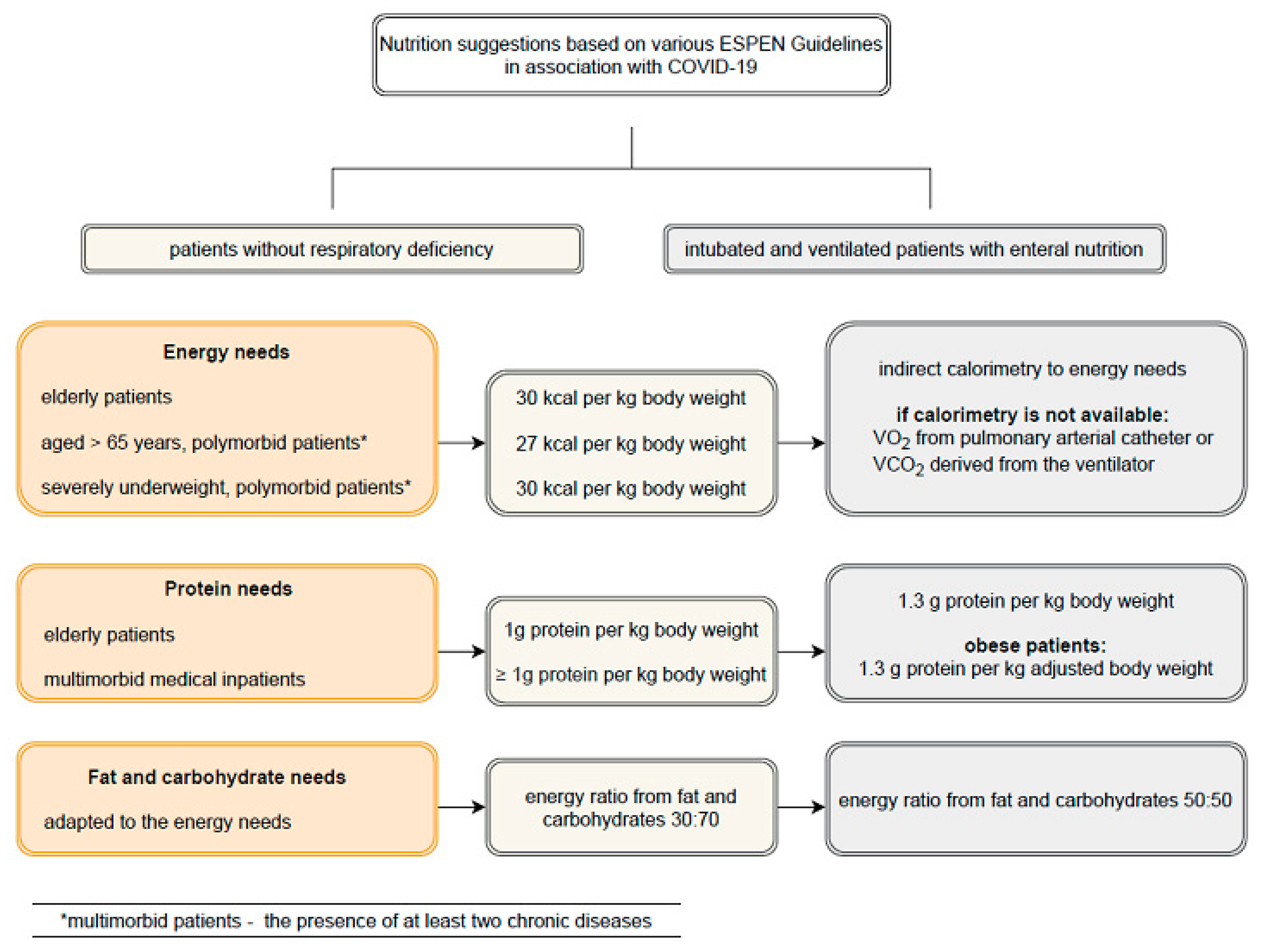
4. Malnutrition in SARS-CoV-2 Infection
5. Probiotic Therapy
- Strengthening of the innate immune responses of the mucosa;
- Decreasing intestinal permeability;
- The effects on the systemically acquired immune response are mediated by regulatory and anti-inflammatory effects.
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rajput, S.; Paliwal, D.; Naithani, M.; Kothari, A.; Meena, K.; Rana, S. COVID-19 and Gut Microbiota: A Potential Connection. Indian J. Clin. Biochem. 2021, 36, 266–277. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, L.; Lin, W.; Tang, W.; Chan, F.K.; Ng, S.C. Review article: Probiotics, prebiotics and dietary approaches during COVID-19 pandemic. Trends Food Sci. Technol. 2021, 108, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Burchill, E.; Lymberopoulos, E.; Menozzi, E.; Budhdeo, S.; McIlroy, J.R.; Macnaughtan, J.; Sharma, N. The Unique Impact of COVID-19 on Human Gut Microbiome Research. Front. Med. 2021, 8, 652464. [Google Scholar] [CrossRef]
- Perlot, T.; Penninger, J.M. ACE2—From the renin-angiotensin system to gut microbiota and malnutrition. Microbes Infect. 2013, 15, 866–873. [Google Scholar] [CrossRef]
- Viana, S.D.; Nunes, S.; Reis, F. ACE2 imbalance as a key player for the poor outcomes in COVID-19 patients with age-related comorbidities-Role of gut microbiota dysbiosis. Ageing Res. Rev. 2020, 62, 101123. [Google Scholar] [CrossRef]
- Lammi, C.; Arnoldi, A. Food-derived antioxidants and COVID-19. J. Food Biochem. 2021, 45, e13557. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Genetic Variation, Diet, Inflammation, and the Risk for COVID-19. Lifestyle Genom. 2021, 14, 37–42. [Google Scholar] [CrossRef]
- Barazzoni, R.; Bischoff, S.C.; Breda, J.; Wickramasinghe, K.; Krznaric, Z.; Nitzan, D.; Pirlich, M.; Singer, P. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin. Nutr. 2020, 39, P1631–P1638. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Rosário, A.; Marques, C.; Pinheiro, H.; Araújo, J.R.; Ribeiro, P.; Rocha, R.; Mota, I.; Pestana, D.; Ribeiro, R.; Pereira, A.; et al. Gut Microbiota Diversity and C-Reactive Protein Are Predictors of Dis-ease Severity in COVID-19 Patients. Front. Microbiol. 2021, 12, 705020. [Google Scholar] [CrossRef] [PubMed]
- Yeoh, Y.K.; Zuo, T.; Lui, G.C.Y.; Zhang, F.; Liu, Q.; Li, A.Y.; Chung, A.C.; Cheung, C.P.; Tso, E.Y.; Fung, K.S.; et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 2021, 70, 698–706. [Google Scholar] [CrossRef]
- Zuo, T.; Zhang, F.; Lui, G.C.Y.; Yeoh, Y.K.; Li, A.Y.; Zhan, H.; Wan, Y.; Chung, A.C.; Cheung, C.P.; Chen, N.; et al. Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization. Gastroenterology 2020, 159, 944–955. [Google Scholar] [CrossRef]
- Groves, H.T.; Cuthberston, L.; James, P.; Moffatt, M.F.; Cox, M.J.; Tregoning, J.S. Respiratory Disease following Viral Lung Infection Alters the Murine Gut Microbiota. Front. Immunol. 2018, 9, 182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartley, J.M.; Zhou, X.; Kuchel, G.A.; Weinstock, G.M.; Haynes, L. Impact of age, caloric restriction, and influenza infection on mouse gut microbiome: An exploratory study of the role of age-related microbiome changes on influenza responses. Front. Immunol. 2017, 8, 1164. [Google Scholar] [CrossRef]
- Zelaya, H.; Alvarez, S.; Kitazawa, H.; Villena, J. Respiratory Antiviral Immunity and Immunobiotics: Beneficial Effects on Inflammation-Coagulation Interaction during Influenza Virus Infection. Front. Immunol. 2016, 7, 633. [Google Scholar] [CrossRef] [Green Version]
- Conte, L.; Toraldo, D.M. Targeting the gut-lung microbiota axis by means of a high-fibre diet and probiotics may have anti-inflammatory effects in COVID-19 infection. Ther. Adv. Respir. Dis. 2020, 14, 1753466620937170. [Google Scholar] [CrossRef]
- Ratajczak, W.; Rył, A.; Mizerski, A.; Walczakiewicz, K.; Sipak, O.; Laszczyńska, M. Immunomodulatory potential of gut microbiome-derived short-chain fatty acids (SCFAs). Acta Biochim. Pol. 2019, 66, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rishi, P.; Thakur, K.; Vij, S.; Rishi, L.; Singh, A.; Kaur, I.P.; Patel, S.K.S.; Lee, J.-K.; Kalia, V.C. Diet, Gut Microbiota and COVID-19. Indian J. Microbiol. 2020, 60, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Root, M.M.; Houser, S.M.; Anderson, J.J.B.; Dawson, H.R. Healthy Eating Index 2005 and selected macronutrients are correlated with improved lung function in humans. Nutr. Res. 2014, 34, 277–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Q.; Liang, Q.; Balakrishnan, B.; Belobrajdic, D.P.; Feng, Q.J.; Zhang, W. Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients 2020, 12, 381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarpellini, E.; Fagoonee, S.; Rinninella, E.; Rasetti, C.; Aquila, I.; LaRussa, T.; Ricci, P.; Luzza, F.; Abenavoli, L. Gut Microbiota and Liver Interaction through Immune System Cross-Talk: A Comprehensive Review at the Time of the SARS-CoV-2 Pandemic. J. Clin. Med. 2020, 9, 2488. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [Green Version]
- Jabczyk, M.; Nowak, J.; Hudzik, B.; Zubelewicz-Szkodzińska, B. Curcumin and its potential impact on microbiota. Nutrients 2021, 13, 2004. [Google Scholar] [CrossRef]
- Vázquez-Calvo, Á.; de Oya, N.J.; Martín-Acebes, M.A.; Garcia-Moruno, E.; Saiz, J.C. Antiviral Properties of the Natural Polyphenols Delphinidin and Epigallocatechin Gallate against the Flaviviruses West Nile Virus, Zika Virus, and Dengue Virus. Front. Microbiol. 2017, 8, 1314. [Google Scholar] [CrossRef]
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Xiaofeng, L.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastronterology 2020, 158, 1831–1833. [Google Scholar] [CrossRef]
- Infusino, F.; Marazzato, M.; Mancone, M.; Fedele, F.; Mastroianni, C.M.; Severino, P.; Ceccarelli, G.; Santinelli, L.; Cavarretta, E.; Marullo, A.G.M.; et al. Diet Supplementation, Probiotics, and Nutraceuticals in SARS-CoV-2 Infection: A Scoping Review. Nutrients 2020, 12, 1718. [Google Scholar] [CrossRef] [PubMed]
- Utomo, R.Y.; Ikawati, M.; Meiyanto, E. Revealing the Potency of Citrus and Galangal Constituents to Halt SARS-CoV-2 Infection. Preprints 2020, 2020030214. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Hu, C.; Hood, M.; Zhang, X.; Zhang, L.; Kan, J.; Du, J. A Novel Combination of Vitamin C, Curcumin and Glycyrrhizic Acid Potentially Regulates Immune and Inflammatory Response Associated with Coronavirus Infections: A Perspective from System Biology Analysis. Nutrietns 2020, 12, 1193. [Google Scholar] [CrossRef]
- Biancatelli, R.M.L.C.; Berril, M.; Catravas, J.D.; Msrik, P.E. Quercetin and Vitamin C: An Experimental, Synergistic Therapy for the Prevention and Treatment of SARS-CoV-2 Related Disease (COVID-19). Front. Immunol. 2020, 11, 1451. [Google Scholar] [CrossRef] [PubMed]
- Hemila, H. Vitamin C and Infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef] [Green Version]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, F. Vitamin-D and COVID-19: Do deficient risk a poorer outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef]
- Tripathi, S.; Tecle, T.; Verma, A.; Crouch, E.; White, M.; Hartshorn, K.L. The human cathelicidin LL-37 inhibits influenza a viruses through a mechanism distinct from that of surfactant protein D or defensins. J. Gen. Virol. 2013, 94, 40–49. [Google Scholar] [CrossRef]
- Monlezun, D.J.; Bittner, E.A.; Christopher, K.B.; Camargo, C.A.; Quraishi, S.A. Vitamin D status and acute respiratory infection: Cross sectional results from the United States national health and nutrition examination survey, 2001–2006. Nutrients 2015, 7, 1933–1944. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; A Ginde, A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [Green Version]
- Teymoori-Rad, M.; Shokri, F.; Salimi, V.; Marashi, S.M. The interplay Vit D and viral infections. Rev. Med. Virol. 2019, 29, e2032. [Google Scholar] [CrossRef] [PubMed]
- Teymoori-Rad, M.; Marashi, S.M. Vitamin D and Covid-19: From potential therapeutic effects to unanswered questions. Rev. Med. Virol. 2020, 31, e2159. [Google Scholar] [CrossRef] [PubMed]
- Caccialanza, R.; Laviano, A.; Lobascio, F.; Montagna, E.; Bruno, R.; Ludovisi, S.; Corsico, A.G.; Di Sabatino, A.; Belliato, M.; Calvi, M.; et al. Early nutritional supplementation in non-critically ill patients hospitalized for the 2019 novel coronavirus disease (COVID-19): Rationale and feasibility of a shared pragmatic protocol. Nutrition 2020, 74, 110835. [Google Scholar] [CrossRef] [PubMed]
- Hiemstra, I.H.; Beijer, M.R.; Veninga, H.; Vrijland, K.; Borg, E.G.F.; Olivier, B.J.; Mebius, R.E.; Kraal, G.; Haan, J.M.M.D. The identification and developmental requirements of colonic CD169(+) macrophages. Immunology 2014, 142, 269–278. [Google Scholar] [CrossRef]
- Shrestha, S.; Kim, S.Y.; Yun, Y.J.; Kim, J.-K.; Lee, J.M.; Shin, M.; Song, D.-K.; Hong, C.-W. Retinoic acid induces hypersegmentation and enhances cytotoxicity of neutrophils against cancer cells. Immunol. Lett. 2017, 182, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Iddir, M.; Brito, A.; Dingeo, G.; Del Campo, S.S.F.; Samouda, H.; La Frano, M.R.; Bohn, T. Strengthening the Immune System and Reducing Inflammation and Oxidative Stress through Diet and Nutrition: Considerations during the COVID-19 Crisis. Nutrients 2020, 12, 1562. [Google Scholar] [CrossRef]
- Pironi, L.; Sasdelli, A.S.; Ravaioli, F.; Baracco, B.; Battaiola, C.; Bocedi, G.; Brodosi, L.; Leoni, L.; Mari, G.A.; Musio, A. Malnutrition and nutritional therapy in patients with SARS-CoV-2 disease. Clin. Nutr. 2021, 40, 1330–1337. [Google Scholar] [CrossRef]
- Baud, D.; Agri, D.; Gibson, G.R.; Gionnoni, E. Using probiotics to flatten the curve of coronavirus disease COVID-2019 pandemic. Front. Public Health 2020, 8, 186. [Google Scholar] [CrossRef]
- D’Amelio, P.; Sassi, F. Gut Microbiota, Immune System, and Bone. Calcif. Tissue Int. 2018, 102, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Olaimat, A.N.; Aolymat, I.; Al-Holy, M.; Ayyash, M.; Abu Ghoush, M.; Al-Nabulsi, A.A.; Osaili, T.; Apostolopoulos, V.; Liu, S.-Q.; Shah, N.P. The potential application of probiotics and prebiotics for the prevention and treatment of COVID-19. NPJ Sci. Food 2020, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- King, S.; Glanville, J.; Sanders, M.E.; Fitzgerald, A.; Varley, D. Effectiveness of probiotics on the duration of illness in healthy children and adults who develop common acute respiratory infectious conditions: A systematic review and meta-analysis. Br. J. Nutr. 2014, 112, 41–54. [Google Scholar] [CrossRef]
- Hao, Q.; Dong, B.R.; Wu, T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst. Rev. 2015, 2, CD006895. [Google Scholar] [CrossRef]
- Mak, J.W.Y.; Chan, F.K.L.; Ng, S.C. Probiotics and COVID-19: One size does not fit all. Lancet Gastroenterol. Hepatol. 2020, 5, 644–645. [Google Scholar] [CrossRef]
- Giannoni, E.; Baud, D.; Agri, V.D.; Gibson, G.R.; Reid, G. Probiotics and COVID-19. Lancet Gastroenterol. Hepatol. 2020, 5, 720–721. [Google Scholar] [CrossRef]
- Morrow, L.E.; Kollef, M.H.; Casale, T.B. Probiotic prophylaxis of ventilator-associated pneumonia: A blinded, randomized, controlled trial. Am. J. Respir Care Med. 2010, 182, 1058–1064. [Google Scholar] [CrossRef] [Green Version]
- Zeng, J.; Wang, C.T.; Zhang, F.S.; Qi, F.; Wang, S.-F.; Ma, S.; Wu, T.-J.; Tian, H.; Tian, Z.-T.; Zhang, S.-L.; et al. Effect of probiotics on the incidence of ventilator-associated pneumonia in critically ill patients: A randomized controlled multicenter trial. Intensive Care Med. 2016, 42, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Cai, H.; Shen, Y.; Ni, Q.; Chen, Y.; Hu, S.; Li, J.; Wang, H.; Yu, L.; Huang, H.; et al. Management of corona virus disease-19 (COVID-19): The Zhejiang experience. Zhejiang Da Xue Xue Bao Yi Xue Ban 2020, 49, 147–157. [Google Scholar] [CrossRef]
- Feng, Z.; Wang, Y.; Qi, W. The small intestine, an underestimated site of SARS-CoV-2 infection: From Red Queen Effect to Probiotics. Preprints 2020, 2020030161. [Google Scholar] [CrossRef] [Green Version]
- Dermyshi, E.; Wang, Y.; Yan, C.; Hong, W.; Qiu, G.; Gong, X.; Zhang, T. The “Golden Age” of Probiotics: A Systematic Review and Meta-Analysis of Randomized and Observational Studies in Preterm Infants. Neonatology 2017, 112, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Merenstein, D.J.; Ouwehand, A.C.; Reid, G.; Salminen, S.; Cabana, M.D.; Paraskevakos, G.; Leyer, G. Probiotic use in at-risk populations. J. Am. Pharm. Assoc. 2016, 56, 680–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, C.K.Y.; Chan, O.S.; Li, H.B.; Pang, H. Preventing respiratory tract infections by synbiotic interventions: A systematic review and meta-analysis of randomized controlled trials. Adv. Nutr. 2020, 111, 979–988. [Google Scholar] [CrossRef]
- D’Amico, F.; Baumgart, D.C.; Danese, S.; Peyrin-Biroulet, L. Diarrhea During COVID-19 Infection: Pathogenesis, Epidemiology, Prevention, and Management. Clin. Gastroenterol. Hepatol. 2020, 18, 1663–1672. [Google Scholar] [CrossRef]
- Gao, Q.Y.; Chen, Y.X.; Fang, J.Y. 2019 Novel coronavirus infection and gastrointestinal tract. J. Dig. Dis. 2020, 21, 125–126. [Google Scholar] [CrossRef] [Green Version]
- Saleh, J.; Peyssonnaux, C.; Singh, K.K.; Edeas, M. Mitochondria and microbiota dysfunction in COVID-19 pathogenesis. Mitochondrion 2020, 54, 1–7. [Google Scholar] [CrossRef]
- Belojevic, G.; Prasher, D. Music of microbiota against SARS CoV-2. Noise Health 2019, 21, 97. [Google Scholar] [CrossRef]
- De Souza Monnerat, J.A.; de Souza, P.R.; Cardoso, L.M.; Mattos, J.D.; de Souza Rocha, G.; Medeiros, R.F. Micronutrients and bioactive compounds in the immunological pathways related to SARS-CoV-2 (adults and elderly). Eur. J. Nutr. 2021, 60, 559–579. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. The Microbiome-Gut-Brain Axis in Health and Disease. Gastroenterol. Clin. N. Am. 2017, 46, 77–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Toole, P.W.; Jeffery, I.B. Microbiome-health interactions in older people. Cell Mol. Life Sci. 2018, 75, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Kulczyński, B.; Gramza-Michałowska, A. Health benefits of inulin-type fructans. Med. Rodz. 2016, 2, 86–90. [Google Scholar]
| Group of Compounds | Therapeutic Effects | References |
|---|---|---|
| Curcumin | Binding to the SARS-CoV-2 target receptors | [25,26] |
| Polyphenols | Modulation of cytokine production and the expression of proinflammatory genes. Antiviral activities against several viruses (SARS-CoV-2 has not been proven) | [21,23] |
| Vitamin C | Alleviating the course of upper respiratory tract infections. Participation in the processes of phagocytosis and chemotaxis. Participation in the production, differentiation, and proliferation of T cells (resulting in the synthesis of antibodies) | [24,25,60] |
| Vitamin D | Participation in the macrophage synthesis of cathelicidin. Regulation of the activity and levels of NF-kB, IL-6, IL1-β, and TNF-α, and the production of GM-CSF, IL-4, IL-5, VCAM-1, ICAM-1, and E-selectin. An adequate supply of 25(OH)D3 protects against acute respiratory infections | [24,25,30,31,32,33,34,37] |
| Combination of vitamin C, curcumin, and licoric acid | The production of interferons and the regulation of inflammatory responses | [25,27] |
| Probiotics | Bifidobacterium and Lactobacillus have beneficial effects against infections (e.g., the influenza virus), including an increase in the number of helper T cells in the lung parenchyma. Lactobacillus plantarum and Lactobacillus reuteri reduce the recruitment of granulocytes and the expression of the proinflammatory cytokines that inhibit the development of pneumonia virus. Lactobacillus rhamnosus increases interferon-γ and interleukin-2. Bifidobacterium infantis inhibits IL-17. Bifidobacterium animalis prevents virus replication. Bifidobacterium lactis increases the proportions of total, helper (CD4+), and activated (CD25+) T cells, and NK cells in the blood. Lactococcus lactis activates plasmacytoid dendritic cells. Lactobacillus plantarum reduces the tissue damage caused by inflammation in TGEV (gastroenteritis coronavirus). Bacillus subtilis inhibits the adherence of TGEV | [14,18,61,62] |
| Prebiotics | Inulin decreases the abundance of Bifidobacterium, Lactobacillus, and Eubacteria. Oligofructose decreases the number of bacteria of the Clostridium, Bacteroides, and Fusobacterium genera | [63] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jabczyk, M.; Nowak, J.; Hudzik, B.; Zubelewicz-Szkodzińska, B. Diet, Probiotics and Their Impact on the Gut Microbiota during the COVID-19 Pandemic. Nutrients 2021, 13, 3172. https://doi.org/10.3390/nu13093172
Jabczyk M, Nowak J, Hudzik B, Zubelewicz-Szkodzińska B. Diet, Probiotics and Their Impact on the Gut Microbiota during the COVID-19 Pandemic. Nutrients. 2021; 13(9):3172. https://doi.org/10.3390/nu13093172
Chicago/Turabian StyleJabczyk, Marzena, Justyna Nowak, Bartosz Hudzik, and Barbara Zubelewicz-Szkodzińska. 2021. "Diet, Probiotics and Their Impact on the Gut Microbiota during the COVID-19 Pandemic" Nutrients 13, no. 9: 3172. https://doi.org/10.3390/nu13093172
APA StyleJabczyk, M., Nowak, J., Hudzik, B., & Zubelewicz-Szkodzińska, B. (2021). Diet, Probiotics and Their Impact on the Gut Microbiota during the COVID-19 Pandemic. Nutrients, 13(9), 3172. https://doi.org/10.3390/nu13093172






