Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe?
Abstract
1. Introduction
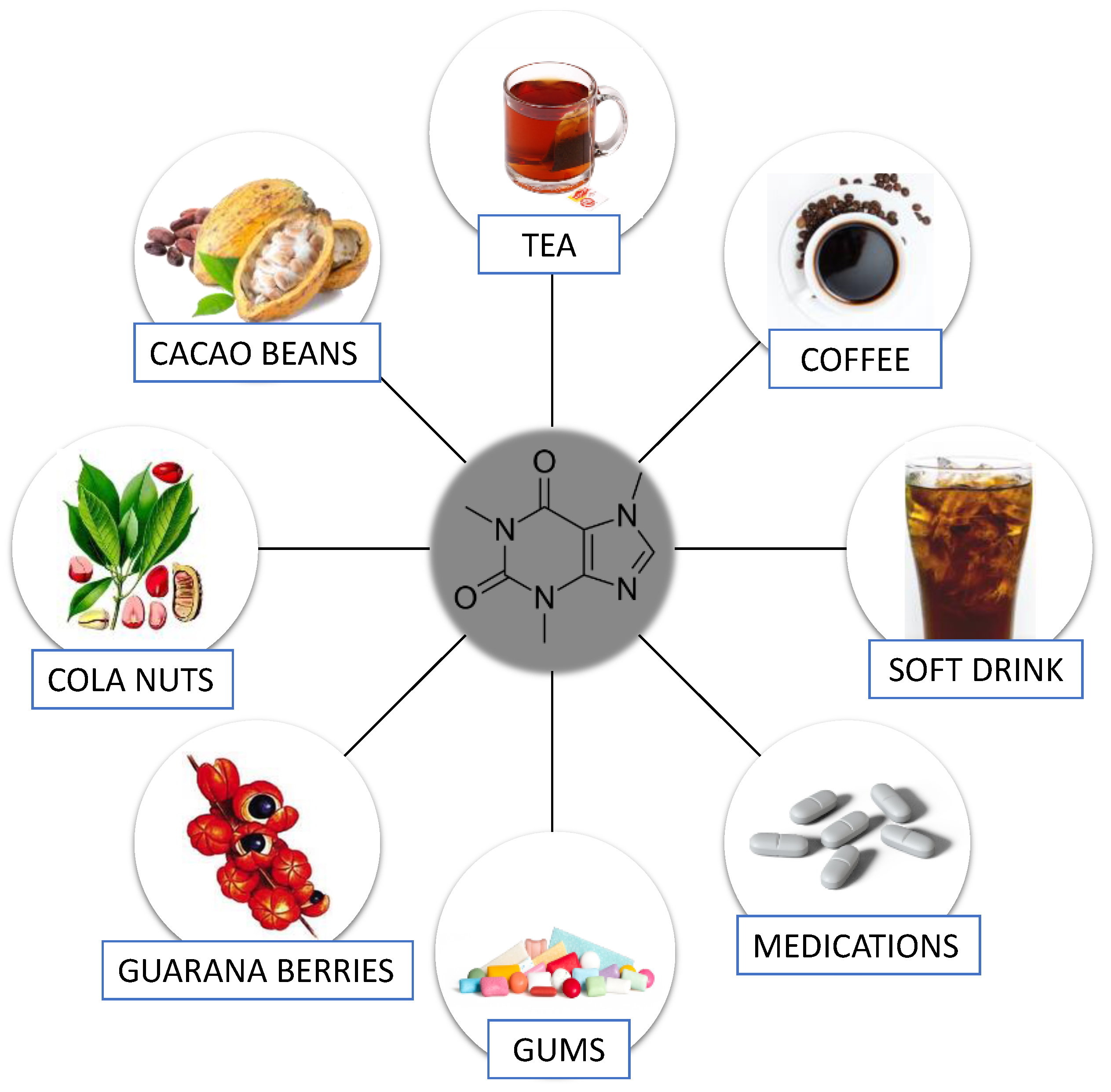
1.1. Caffeine—General Information
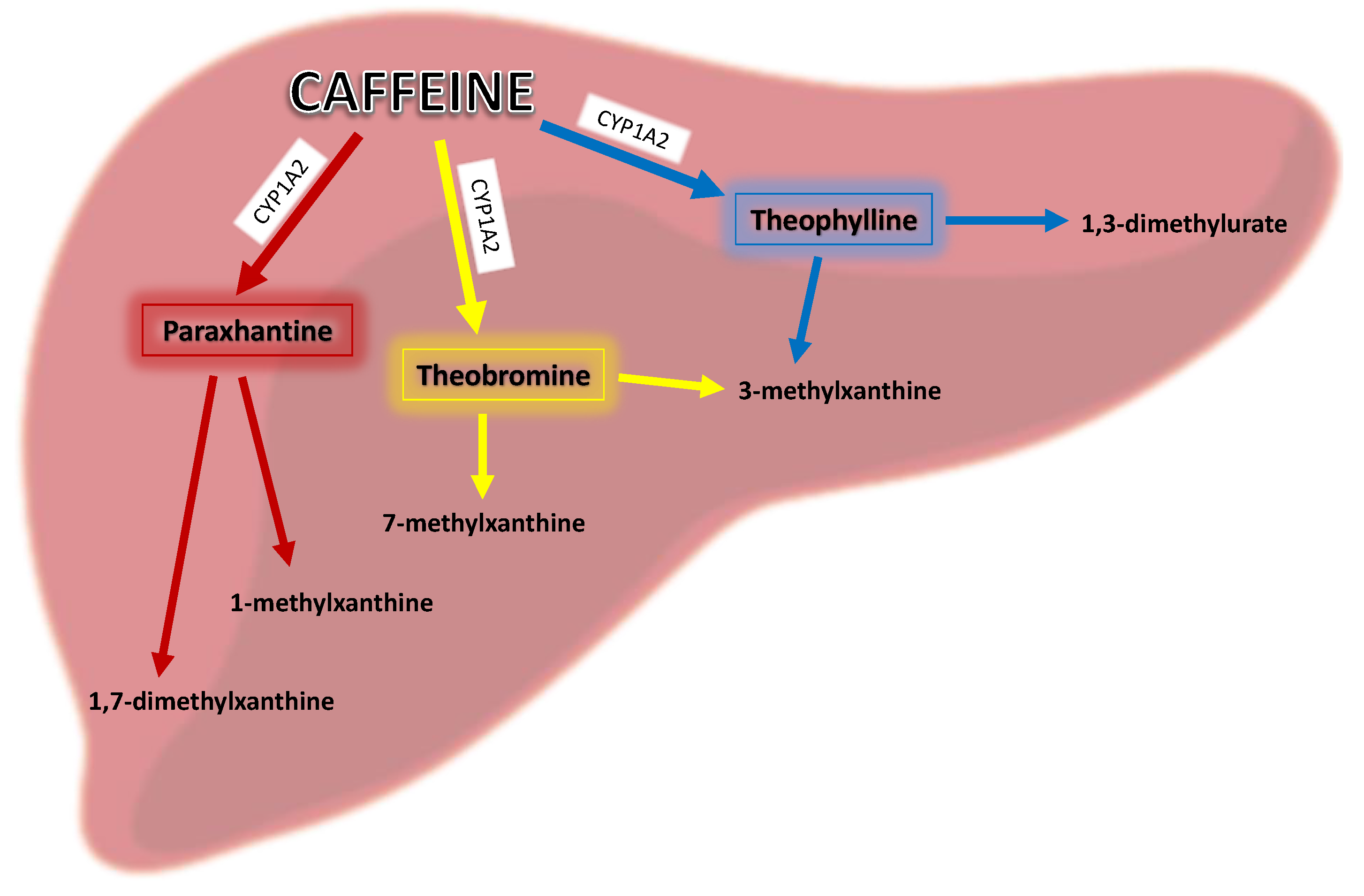
1.2. Metabolism of Caffeine
1.3. Genetics and Caffeine
1.4. Effects on Receptors
1.5. Toxicity of Caffeine
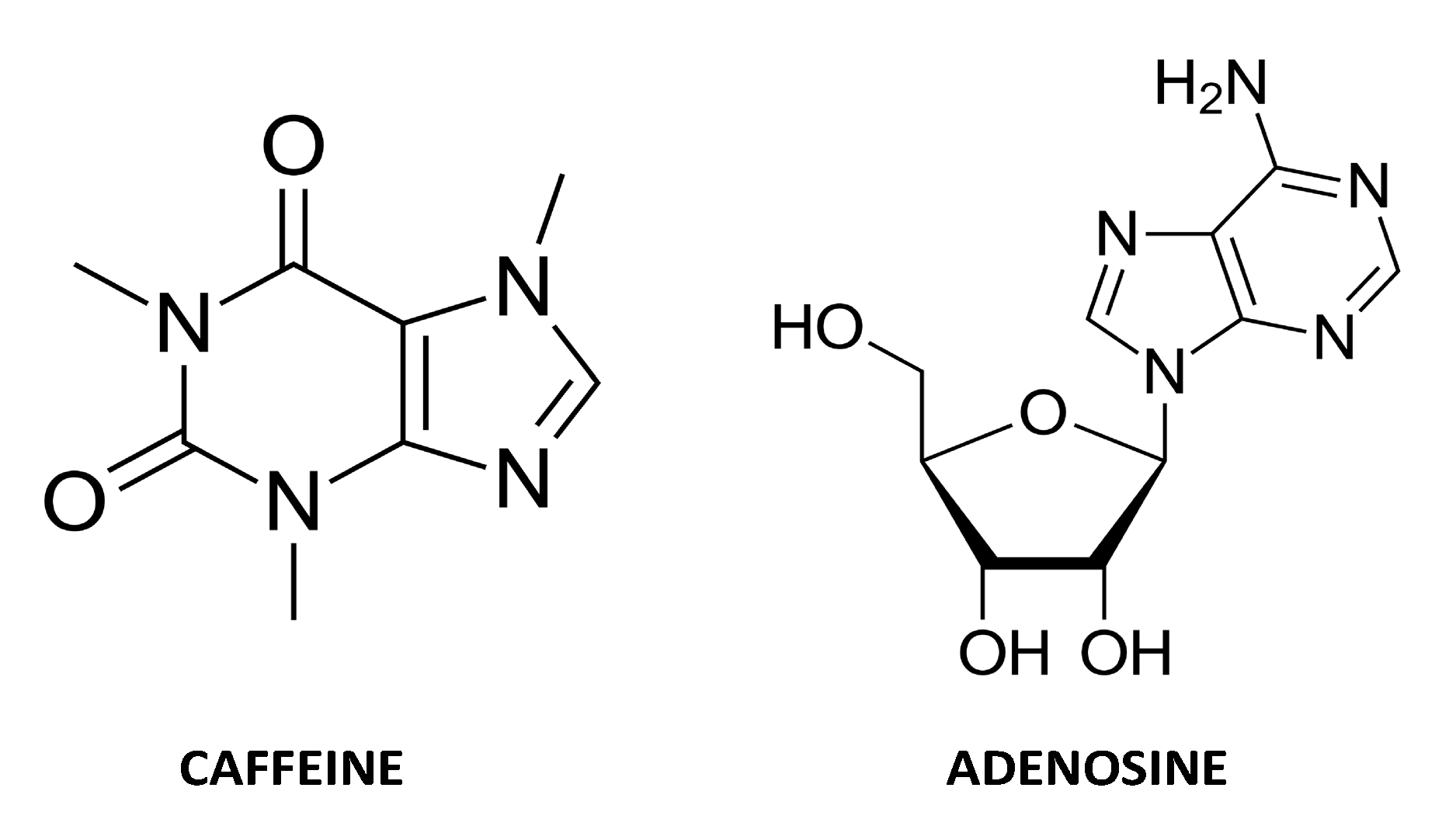
1.6. Adenosine
2. The Role of Caffeine in Various Systems in the Human Body
2.1. Central Nervous System
2.1.1. Caffeine Impact on Children and Adolescent
2.1.2. Caffeine and Taste Perception
2.1.3. Caffeine and Alzheimer’s Disease
2.1.4. Caffeine and Parkinson’s Disease
2.1.5. Caffeine and Huntington’s Disease
2.1.6. Caffeine and Perception of Pain
2.1.7. Caffeine and Mental Health
2.2. The Immune System
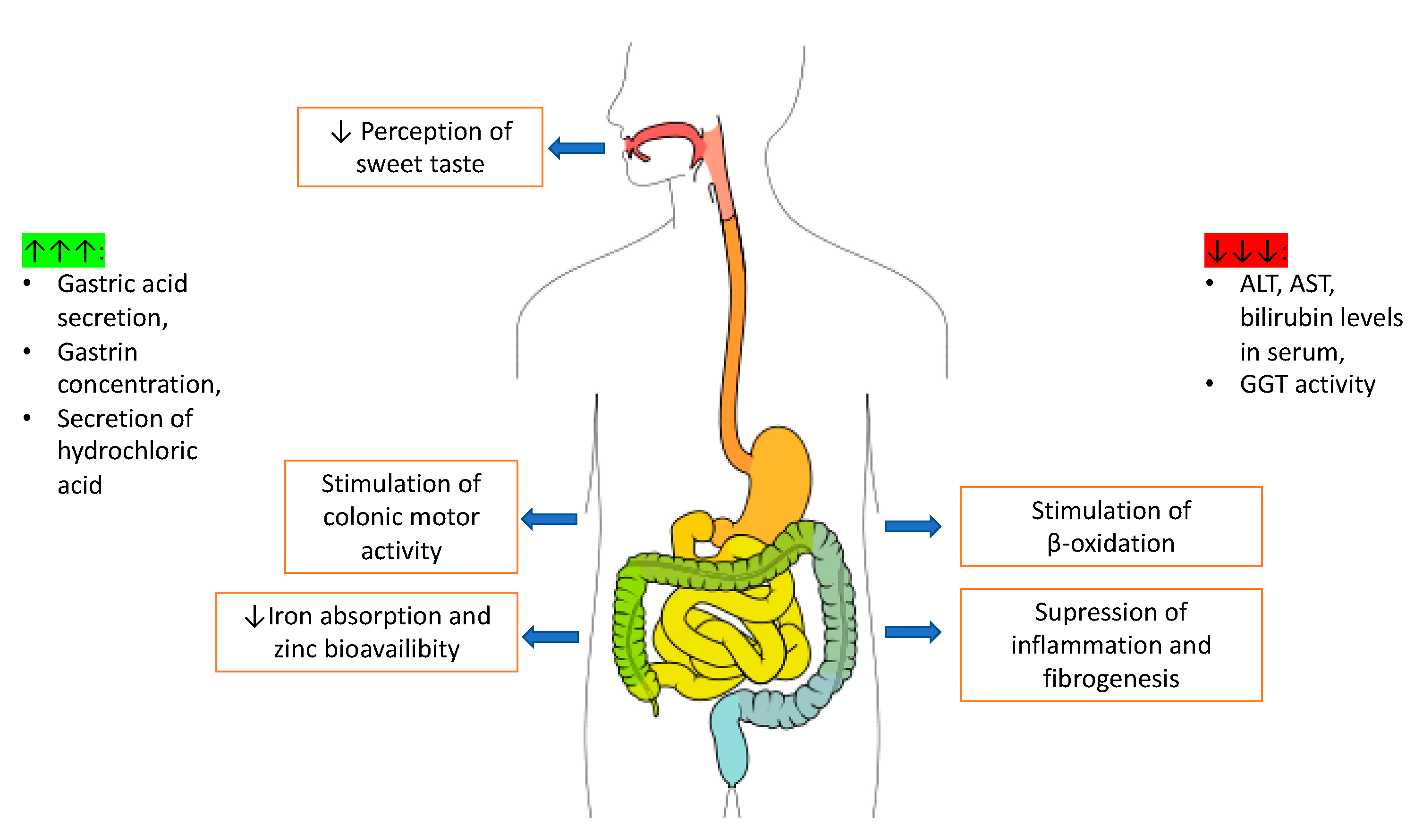
2.3. Digestive System
2.3.1. Caffeine Action on the Small and Large Intestine
2.3.2. Caffeine and the Liver
2.3.3. Caffeine and Glycaemia
2.3.4. Caffeine and Digestive Tract Cancer
2.4. Respiratory System
2.4.1. Caffeine and Asthma and Chronic Obstructive Pulmonary Disease
2.4.2. Caffeine and Breathing Problems
2.4.3. Caffeine and Lung Cancer
2.5. Circulatory System
Caffeine and Arrhythmia
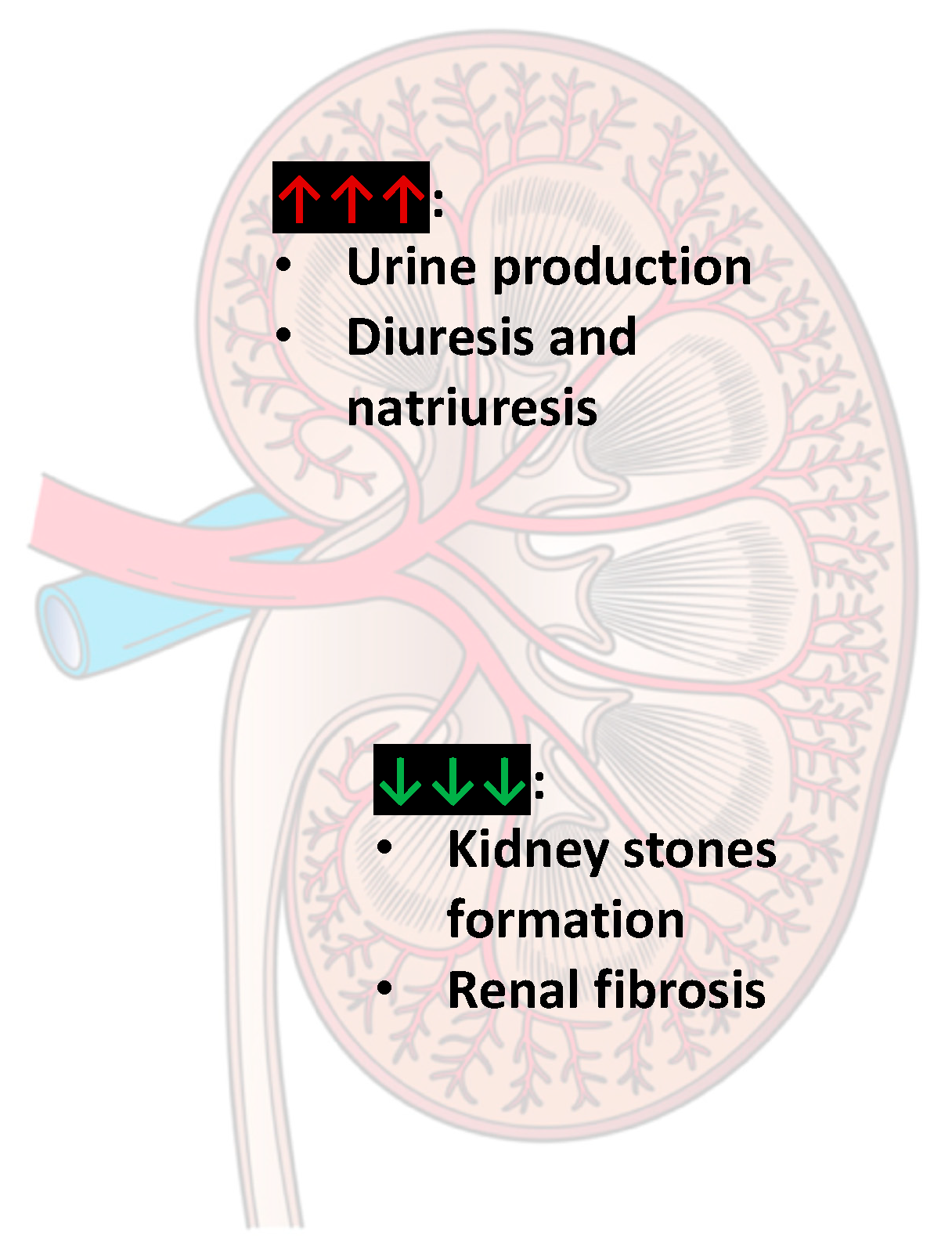
2.6. Urinary Tract
2.6.1. Caffeine and Urinary Incontinence
2.6.2. Caffeine and Kidney Stones
2.6.3. Caffeine and Bladder Cancer
2.7. Skeletal and Muscular System
2.7.1. Caffeine and Bones
2.7.2. Caffeine Action on Muscle Filaments and Muscular Strength
3. Caffeine and Oxidative Stress
4. The Association between Caffeine Consumption and All-Cause and Cause-Specific Mortality
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mahoney, C.R.; Giles, G.E.; Marriott, B.P.; Judelson, D.A.; Glickman, E.L.; Geiselman, P.J.; Lieberman, H.R. Intake of caffeine from all sources and reasons for use by college students. Clin. Nutr. 2019, 38, 668–675. [Google Scholar] [CrossRef]
- Heckman, M.A.; Weil, J.; De Mejia, E.G. Caffeine (1, 3, 7-trimethylxanthine) in Foods: A Comprehensive Review on Consumption, Functionality, Safety, and Regulatory Matters. J. Food Sci. 2010, 75, R77–R87. [Google Scholar] [CrossRef]
- Chawla, J. Neurologic Effects of Caffeine Physiologic Effects of Caffeine. Medscape Ref. Drug Dis. Proced. 2015, 24, 1–8. [Google Scholar]
- Conway, J. Global Coffee Consumption, 2020/21|Statista. Available online: https://www.statista.com/statistics/292595/global-coffee-consumption/ (accessed on 5 May 2021).
- Satel, S. Is Caffeine Addictive?—A Review of the Literature. Am. J. Drug Alcohol Abus. 2006, 32, 493–502. [Google Scholar] [CrossRef]
- Kolahdouzan, M.; Hamadeh, M.J. The neuroprotective effects of caffeine in neurodegenerative diseases. CNS Neurosci. Ther. 2017, 23, 272–290. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M.; Dobes, P. Caffeine Inhibits Acetylcholinesterase, But Not Butyrylcholinesterase. Int. J. Mol. Sci. 2013, 14, 9873–9882. [Google Scholar] [CrossRef] [PubMed]
- IARC. Coffee, tea, mate, methylxanthines and methylglyoxal. IARC Monogr. Eval. Carcinog. Risks Hum. 1991, 51, 1–513. [Google Scholar]
- Barone, J.J.; Roberts, H. Human Consumption of Caffeine. In Caffeine; Springer: Berlin/Heidelberg, Germany, 1984; pp. 59–73. [Google Scholar]
- De Mejia, E.G.; Ramirez-Mares, M.V. Impact of caffeine and coffee on our health. Trends Endocrinol. Metab. 2014, 25, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, J.; Sawers, S.J.A. The absolute bioavailability of caffeine in man. Eur. J. Clin. Pharmacol. 1983, 24, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Tavares, C.; Sakata, R. Caffeine in the Treatment of Pain. Braz. J. Anesthesiol. 2012, 62, 387–401. [Google Scholar] [CrossRef]
- Fredholm, B.B. Adenosine, Adenosine Receptors and the Actions of Caffeine. Pharmacol. Toxicol. 1995, 76, 93–101. [Google Scholar] [CrossRef]
- Arnaud, M.J. Pharmacokinetics and Metabolism of Natural Methylxanthines in Animal and Man. In Methylxanthines; Springer: Berlin/Heidelberg, Germany, 2010; pp. 33–91. [Google Scholar]
- Pickering, C.; Kiely, J. Are the Current Guidelines on Caffeine Use in Sport Optimal for Everyone? Inter-individual Variation in Caffeine Ergogenicity, and a Move Towards Personalised Sports Nutrition. Sports Med. 2018, 48, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Sachse, C.; Brockmöller, J.; Bauer, S.; Roots, I. Functional significance of a C→A polymorphism in intron 1 of the cytochrome P450 CYP1A2 gene tested with caffeine. Br. J. Clin. Pharmacol. 1999, 47, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Womack, C.J.; Saunders, M.J.; Bechtel, M.K.; Bolton, D.J.; Martin, M.; Luden, N.D.; Dunham, W.; Hancock, M. The influence of a CYP1A2 polymorphism on the ergogenic effects of caffeine. J. Int. Soc. Sports Nutr. 2012, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Pataky, M.W.; Womack, C.J.; Saunders, M.J.; Goffe, J.L.; D’Lugos, A.C.; Elsohemy, A.; Luden, N.D. Caffeine and 3-km cycling performance: Effects of mouth rinsing, genotype, and time of day. Scand. J. Med. Sci. Sports 2015, 26, 613–619. [Google Scholar] [CrossRef]
- Algrain, H.A.; Thomas, R.M.; Carrillo, A.E.; Ryan, E.J.; Kim, C.-H.; Lettan, R.B.; Ryan, E.J. The Effects of a Polymorphism in the Cytochrome P450 CYP1A2 Gene on Performance Enhancement with Caffeine in Recreational Cyclists. J. Caffeine Res. 2016, 6, 34–39. [Google Scholar] [CrossRef]
- Klein, C.S.; Clawson, A.; Martin, M.; Saunders, M.J.; Flohr, J.A.; Bechtel, M.K.; Dunham, W.; Hancock, M.; Womack, C.J. The Effect of Caffeine on Performance in Collegiate Tennis Players. J. Caffeine Res. 2012, 2, 111–116. [Google Scholar] [CrossRef]
- Salinero, J.J.; Lara, B.; Ruiz-Vicente, D.; Areces, F.; Puente-Torres, C.; Gallo-Salazar, C.; Pascual, T.; Del Coso, J. CYP1A2 Genotype Variations Do Not Modify the Benefits and Drawbacks of Caffeine during Exercise: A Pilot Study. Nutrients 2017, 9, 269. [Google Scholar] [CrossRef]
- Nehlig, A. Interindividual Differences in Caffeine Metabolism and Factors Driving Caffeine Consumption. Pharmacol. Rev. 2018, 70, 384–411. [Google Scholar] [CrossRef]
- Carswell, A.T.; Howland, K.; Martinez-Gonzalez, B.; Baron, P.; Davison, G. The effect of caffeine on cognitive performance is influenced by CYP1A2 but not ADORA2A genotype, yet neither genotype affects exercise performance in healthy adults. Graefe’s Arch. Clin. Exp. Ophthalmol. 2020, 120, 1495–1508. [Google Scholar] [CrossRef]
- Loy, B.D.; O’Connor, P.J.; Lindheimer, J.B.; Covert, S.F. Caffeine Is Ergogenic for Adenosine A2A Receptor Gene (ADORA2A) T Allele Homozygotes: A Pilot Study. J. Caffeine Res. 2015, 5, 73–81. [Google Scholar] [CrossRef]
- Ribeiro, J.A.; Sebastio, A.M. Caffeine and adenosine. J. Alzheimer’s Dis. 2010, 20, S3–S15. [Google Scholar] [CrossRef]
- McLellan, T.M.; Caldwell, J.A.; Lieberman, H.R. A review of caffeine’s effects on cognitive, physical and occupational performance. Neurosci. Biobehav. Rev. 2016, 71, 294–312. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.; Jones, P.P.; Koop, A.; Zhang, L.; Duff, H.J.; Chen, S.R.W. Caffeine induces Ca2+ release by reducing the threshold for luminal Ca2+ activation of the ryanodine receptor. Biochem. J. 2008, 414, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Yang, G.; Ren, J.; Zhao, J.; Li, S. Caffeine Suppresses GABA Receptor-Mediated Current in Rat Primary Sensory Neurons via Inhibition of Intracellular Phosphodiesterase. Neurophysiology 2015, 47, 108–114. [Google Scholar] [CrossRef]
- Paiva, C.L.R.S.; Beserra, B.T.S.; Reis, C.E.G.; Dorea, J.G.; Da Costa, T.H.M.; Amato, A.A. Consumption of coffee or caffeine and serum concentration of inflammatory markers: A systematic review. Crit. Rev. Food Sci. Nutr. 2019, 59, 652–663. [Google Scholar] [CrossRef]
- Zampelas, A.; Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Stefanadis, C. Associations between coffee consumption and inflammatory markers in healthy persons: The ATTICA study. Am. J. Clin. Nutr. 2004, 80, 862–867. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the safety of caffeine. EFSA J. 2015, 13. [Google Scholar] [CrossRef]
- Musgrave, I.F.; Farrington, R.L.; Hoban, C.; Byard, R.W. Caffeine toxicity in forensic practice: Possible effects and under-appreciated sources. Forensic Sci. Med. Pathol. 2016, 12, 299–303. [Google Scholar] [CrossRef]
- Boison, D. Methylxanthines, seizures, and excitotoxicity. Handb. Exp. Pharmacol. 2011, 200, 251–266. [Google Scholar]
- Nawrot, P.; Jordan, S.; Eastwood, J.; Rotstein, J.; Hugenholtz, A.; Feeley, M. Effects of caffeine on human health. Food Addit. Contam. 2003, 20, 1–30. [Google Scholar] [CrossRef]
- Seifert, S.M.; Schaechter, J.L.; Hershorin, E.R.; Lipshultz, S.E. Health Effects of Energy Drinks on Children, Adolescents, and Young Adults. Pediatrics 2011, 127, 511–528. [Google Scholar] [CrossRef]
- Rebola, N.; Rodrigues, R.J.; Oliveira, C.R.; Cunha, R.A. Different roles of adenosine A1, A2A and A3 receptors in controlling kainate-induced toxicity in cortical cultured neurons. Neurochem. Int. 2005, 47, 317–325. [Google Scholar] [CrossRef]
- Cunha, R. Adenosine as a neuromodulator and as a homeostatic regulator in the nervous system: Different roles, different sources and different receptors. Neurochem. Int. 2001, 38, 107–125. [Google Scholar] [CrossRef]
- Haskó, G.; Pacher, P.; Deitch, E.A.; Vizi, E.S. Shaping of monocyte and macrophage function by adenosine receptors. Pharmacol. Ther. 2007, 113, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; Arslan, G.; Halldner, L.; Kull, B.; Schulte, G.; Wasserman, W. Structure and function of adenosine receptors and their genes. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2000, 362, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Morelli, M.; Carta, A.R.; Kachroo, A.; Schwarzschild, M.A. Pathophysiological roles for purines. Adenosine, caffeine and urate. Prog. Brain Res. 2010, 183, 183–208. [Google Scholar] [PubMed]
- Polito, C.A.; Cai, Z.-Y.; Shi, Y.-L.; Li, X.-M.; Yang, R.; Shi, M.; Li, Q.-S.; Ma, S.-C.; Xiang, L.-P.; Wang, K.-R.; et al. Association of Tea Consumption with Risk of Alzheimer’s Disease and Anti-Beta-Amyloid Effects of Tea. Nutrients 2018, 10, 655. [Google Scholar] [CrossRef]
- Kaplan, G.B.; Greenblatt, D.J.; Ehrenberg, B.L.; Goddard, J.E.; Bs, M.M.C.; Ba, J.S.H.; Shader, R. Dose-Dependent Pharmacokinetics and Psychomotor Effects of Caffeine in Humans. J. Clin. Pharmacol. 1997, 37, 693–703. [Google Scholar] [CrossRef]
- Watson, E.J.; Coates, A.M.; Kohler, M.; Banks, S. Caffeine Consumption and Sleep Quality in Australian Adults. Nutrients 2016, 8, 479. [Google Scholar] [CrossRef]
- Watson, E.J.; Banks, S.; Coates, A.M.; Kohler, M.J. The Relationship between Caffeine, Sleep, and Behavior in Children. J. Clin. Sleep Med. 2017, 13, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Richards, G.; Smith, A. Caffeine consumption and self-assessed stress, anxiety, and depression in secondary school children. J. Psychopharmacol. 2015, 29, 1236–1247. [Google Scholar] [CrossRef] [PubMed]
- Dando, R.; Dvoryanchikov, G.; Pereira, E.; Chaudhari, N.; Roper, S.D. Adenosine Enhances Sweet Taste through A2B Receptors in the Taste Bud. J. Neurosci. 2012, 32, 322–330. [Google Scholar] [CrossRef]
- Holtzman, S.G.; Mante, S.; Minneman, K.P. Role of adenosine receptors in caffeine tolerance. J. Pharmacol. Exp. Ther. 1991, 256, 62–68. [Google Scholar] [PubMed]
- Choo, E.; Picket, B.; Dando, R. Caffeine May Reduce Perceived Sweet Taste in Humans, Supporting Evidence That Adenosine Receptors Modulate Taste. J. Food Sci. 2017, 82, 2177–2182. [Google Scholar] [CrossRef]
- Pickering, C. Are caffeine’s performance-enhancing effects partially driven by its bitter taste? Med. Hypotheses 2019, 131, 109301. [Google Scholar] [CrossRef] [PubMed]
- Gramling, L.; Kapoulea, E.; Murphy, C. Taste Perception and Caffeine Consumption: An fMRI Study. Nutrients 2018, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Lashley, T.; Schott, J.M.; Weston, P.; Murray, C.E.; Wellington, H.; Keshavan, A.; Foti, S.C.; Foiani, M.; Toombs, J.; Rohrer, J.D.; et al. Molecular biomarkers of Alzheimer’s disease: Progress and prospects. Dis. Model. Mech. 2018, 11. [Google Scholar] [CrossRef]
- Li, S.; Geiger, N.H.; Soliman, M.L.; Hui, L.; Geiger, J.D.; Chen, X. Caffeine, Through Adenosine A3 Receptor-Mediated Actions, Suppresses Amyloid-β Protein Precursor Internalization and Amyloid-β Generation. J. Alzheimer’s Dis. 2015, 47, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A. The Role of Adenosine in Alzheimers Disease. Curr. Neuropharmacol. 2009, 7, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Cunha, R.A. Neuroprotection by adenosine in the brain: From A1 receptor activation to A2A receptor blockade. Purinergic Signal. 2005, 1, 111–134. [Google Scholar] [CrossRef] [PubMed]
- Kaster, M.P.; Machado, N.J.; Silva, H.B.; Nunes, A.M.; Ardais, A.P.; Santana, M.; Baqi, Y.; Müller, C.E.; Rodrigues, A.L.; Porciúncula, L.O.; et al. Caffeine acts through neuronal adenosine A2A receptors to prevent mood and memory dysfunction triggered by chronic stress. Proc. Natl. Acad. Sci. USA 2015, 112, 7833–7838. [Google Scholar] [CrossRef]
- Maia, L.; De Mendonca, A. Does caffeine intake protect from Alzheimer’s disease? Eur. J. Neurol. 2002, 9, 377–382. [Google Scholar] [CrossRef]
- Ritchie, K.; Carriere, I.; de Mendonça, A.; Portet, F.; Dartigues, J.-F.; Rouaud, O.; Barberger-Gateau, P.; Ancelin, M.-L. The neuroprotective effects of caffeine: A prospective population study (the Three City Study). Neurology 2007, 69, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Ngandu, T.; Tuomilehto, J.; Soininen, H.; Kivipelto, M. Midlife Coffee and Tea Drinking and the Risk of Late-Life Dementia: A Population-Based CAIDE Study. J. Alzheimer’s Dis. 2009, 16, 85–91. [Google Scholar] [CrossRef]
- Lindsay, J.; Laurin, D.; Verreault, R.; Hébert, R.; Helliwell, B.; Hill, G.B.; McDowell, I. Risk Factors for Alzheimer’s Disease: A Prospective Analysis from the Canadian Study of Health and Aging. Am. J. Epidemiol. 2002, 156, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Li, S.; Sun, J.; Li, Y.; Zhang, D. Association of Coffee, Decaffeinated Coffee and Caffeine Intake from Coffee with Cognitive Performance in Older Adults: National Health and Nutrition Examination Survey (NHANES) 2011–2014. Nutrients 2020, 12, 840. [Google Scholar] [CrossRef] [PubMed]
- Camfield, D.A.; Silber, B.Y.; Scholey, A.B.; Nolidin, K.; Goh, A.; Stough, C. A Randomised Placebo-Controlled Trial to Differentiate the Acute Cognitive and Mood Effects of Chlorogenic Acid from Decaffeinated Coffee. PLoS ONE 2013, 8, e82897. [Google Scholar] [CrossRef] [PubMed]
- Haskell-Ramsay, C.F.; Jackson, P.A.; Forster, J.S.; Dodd, F.L.; Bowerbank, S.L.; Kennedy, D.O. The Acute Effects of Caffeinated Black Coffee on Cognition and Mood in Healthy Young and Older Adults. Nutrients 2018, 10, 1386. [Google Scholar] [CrossRef]
- Fujimaki, M.; Saiki, S.; Li, Y.; Kaga, N.; Taka, H.; Hatano, T.; Ishikawa, K.-I.; Oji, Y.; Mori, A.; Okuzumi, A.; et al. Serum caffeine and metabolites are reliable biomarkers of early Parkinson disease. Neurology 2018, 90, e404–e411. [Google Scholar] [CrossRef]
- Ross, G.W.; Abbott, R.D.; Petrovitch, H.; Morens, D.M.; Grandinetti, A.; Tung, K.-H.; Tanner, C.M.; Masaki, K.H.; Blanchette, P.L.; Curb, J.D.; et al. Association of Coffee and Caffeine Intake With the Risk of Parkinson Disease. JAMA 2000, 283, 2674–2679. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Guo, X.; Park, Y.; Huang, X.; Sinha, R.; Freedman, N.D.; Hollenbeck, A.R.; Blair, A.; Chen, H. Caffeine Intake, Smoking, and Risk of Parkinson Disease in Men and Women. Am. J. Epidemiol. 2012, 175, 1200–1207. [Google Scholar] [CrossRef]
- Palacios, N.; Gao, X.; ScD, M.L.M.; Schwarzschild, M.A.; Shah, R.; Gapstur, S.; Ascherio, A. Caffeine and risk of Parkinson’s disease in a large cohort of men and women. Mov. Disord. 2012, 27, 1276–1282. [Google Scholar] [CrossRef]
- Postuma, R.B.; Anang, J.; Pelletier, A.; Joseph, L.; Moscovich, M.; Grimes, D.; Furtado, S.; Munhoz, R.P.; Appel-Cresswell, S.; Moro, A.; et al. Caffeine as symptomatic treatment for Parkinson disease (Café-PD). Neurology 2017, 89, 1795–1803. [Google Scholar] [CrossRef]
- Roshan, M.H.K.; Tambo, A.; Pace, N.P. Potential Role of Caffeine in the Treatment of Parkinson’s Disease. Open Neurol. J. 2016, 10, 42–58. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.K.; Wu, C.; Tilley, B.C.; Wills, A.-M.; Aminoff, M.J.; Bainbridge, J.; Hauser, R.A.; Schneider, J.S.; Sharma, S.; Singer, C.; et al. Caffeine and Progression of Parkinson Disease: A deleterious interaction with creatine. Clin. Neuropharmacol. 2015, 38, 163–169. [Google Scholar] [CrossRef][Green Version]
- Hespel, P.; Eijnde, B.O.; Van Leemputte, M. Opposite actions of caffeine and creatine on muscle relaxation time in humans. J. Appl. Physiol. 2002, 92, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Vandenberghe, K.; Gillis, N.; Van Leemputte, M.; Van Hecke, P.; Vanstapel, F.; Hespel, P. Caffeine counteracts the ergogenic action of muscle creatine loading. J. Appl. Physiol. 1996, 80, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Simonin, C.; Duru, C.; Salleron, J.; Hincker, P.; Charles, P.; Delval, A.; Youssov, K.; Burnouf, S.; Azulay, J.-P.; Verny, C.; et al. Association between caffeine intake and age at onset in Huntington’s disease. Neurobiol. Dis. 2013, 58, 179–182. [Google Scholar] [CrossRef]
- MacDonald, M.E.; Ambrose, C.M.; Duyao, M.P.; Myers, R.H.; Lin, C.; Srinidhi, L.; Barnes, G.; Taylor, S.A.; James, M.; Groot, N.; et al. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 1993, 72, 971–983. [Google Scholar] [CrossRef]
- Blum, D.; Chern, Y.; Domenici, M.R.; Buee, L.; Lin, C.-Y.; Rea, W.; Ferré, S.; Popoli, P. The Role of Adenosine Tone and Adenosine Receptors in Huntington’s Disease. J. Caffeine Adenosine Res. 2018, 8, 43–58. [Google Scholar] [CrossRef]
- Zuccato, C.; Valenza, M.; Cattaneo, E. Molecular Mechanisms and Potential Therapeutical Targets in Huntington’s Disease. Physiol. Rev. 2010, 90, 905–981. [Google Scholar] [CrossRef] [PubMed]
- Stack, E.C.; Dedeoglu, A.; Smith, K.M.; Cormier, K.; Kubilus, J.K.; Bogdanov, M.; Matson, W.R.; Yang, L.; Jenkins, B.G.; Luthi-Carter, R.; et al. Neuroprotective Effects of Synaptic Modulation in Huntington’s Disease R6/2 Mice. J. Neurosci. 2007, 27, 12908–12915. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schwarzschild, M.A.; Agnati, L.; Fuxe, K.; Chen, J.-F.; Morelli, M. Targeting adenosine A2A receptors in Parkinson’s disease. Trends Neurosci. 2006, 29, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Blum, D.; Hourez, R.; Galas, M.-C.; Popoli, P.; Schiffmann, S.N. Adenosine receptors and Huntington’s disease: Implications for pathogenesis and therapeutics. Lancet Neurol. 2003, 2, 366–374. [Google Scholar] [CrossRef]
- Tanner, C.; Marder, K.; Eberly, S.; Biglan, K.; Oakes, D.; Shoulson, I. Selected health and lifestyle factors, cytosine-adenine-guanine status, and phenoconversion in Huntington’s disease. Mov. Disord. 2018, 33, 472–478. [Google Scholar] [CrossRef]
- Temple, J.L.; Bernard, C.; Lipshultz, S.E.; Czachor, J.D.; Westphal, J.A.; Mestre, M.A. The Safety of Ingested Caffeine: A Comprehensive Review. Front. Psychiatry 2017, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, D.S.; Penn, T.M.; Cable, S.T.; Aroke, E.N.; Goodin, B.R. Higher habitual dietary caffeine consumption is related to lower experimental pain sensitivity in a community-based sample. Psychopharmacology 2018, 235, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Sawynok, J. Adenosine receptor activation and nociception. Eur. J. Pharmacol. 1998, 347, 1–11. [Google Scholar] [CrossRef]
- Sawynok, J.; Liu, X.J. Adenosine in the spinal cord and periphery: Release and regulation of pain. Prog. Neurobiol. 2003, 69, 313–340. [Google Scholar] [CrossRef]
- Latini, S.; Pedata, F. Adenosine in the central nervous system: Release mechanisms and extracellular concentrations. J. Neurochem. 2001, 79, 463–484. [Google Scholar] [CrossRef] [PubMed]
- Rogers, N.L.; Dinges, D.F. Caffeine: Implications for Alertness in Athletes. Clin. Sports Med. 2005, 24, e1–e13. [Google Scholar] [CrossRef]
- Sollevi, A. Adenosine for pain control. Acta Anaesthesiol. Scand. 1997, 41, 135–136. [Google Scholar] [CrossRef]
- Baratloo, A.; Rouhipour, A.; Forouzanfar, M.M.; Safari, S.; Amiri, M.; Negida, A. The Role of Caffeine in Pain Management: A Brief Literature Review. Anesthesiol. Pain Med. 2016, 6, e33193. [Google Scholar] [CrossRef] [PubMed]
- Derry, C.J.; Derry, S.; Moore, R.A. Caffeine as an analgesic adjuvant for acute pain in adults. Cochrane Database Syst. Rev. 2014, 2014, CD009281. [Google Scholar] [CrossRef]
- Nikolajsen, L.; Haroutiunian, S. Koffein som adjuverende analgetikum til behandling af akutte smerter-en gennemgang af et Cochranereview. Ugeskr Læger 2013, 175, 2486–2488. [Google Scholar]
- Zwilling, M.; Theiss, C.; Matschke, V. Caffeine and NAD+Improve Motor Neural Integrity of Dissociated Wobbler Cells in Vitro. Antioxidants 2020, 9, 460. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.; O’Reilly, E.J.; Pan, A.; Mirzaei, F.; Willett, W.C.; Okereke, O.I.; Ascherio, A. Coffee, caffeine, and risk of completed suicide: Results from three prospective cohorts of American adults. World J. Biol. Psychiatry 2014, 15, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Botella, P.; Parra, A. Coffee increases state anxiety in males but not in females. Hum. Psychopharmacol. Clin. Exp. 2003, 18, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Iranpour, S.; Sabour, S. Inverse association between caffeine intake and depressive symptoms in US adults: Data from National Health and Nutrition Examination Survey (NHANES) 2005–2006. Psychiatry Res. 2019, 271, 732–739. [Google Scholar] [CrossRef]
- Kendler, K.S.; Myers, J.; Gardner, C.O. Caffeine intake, toxicity and dependence and lifetime risk for psychiatric and substance use disorders: An epidemiologic and co-twin control analysis. Psychol. Med. 2006, 36, 1717–1725. [Google Scholar] [CrossRef]
- Bertasi, R.A.O.; Humeda, Y.; Bertasi, T.G.O.; Zins, Z.; Kimsey, J.; Pujalte, G. Caffeine Intake and Mental Health in College Students. Cureus 2021, 13. [Google Scholar] [CrossRef]
- Hedström, A.K.; Mowry, E.M.; Gianfrancesco, M.A.; Shao, X.A.; Schaefer, C.; Shen, L.; Olsson, T.; Barcellos, L.F.; Alfredsson, L. High consumption of coffee is associated with decreased multiple sclerosis risk; results from two independent studies. J. Neurol. Neurosurg. Psychiatry 2016, 87, 454–460. [Google Scholar] [CrossRef]
- Konishi, Y.; Hori, H.; Ide, K.; Katsuki, A.; Atake, K.; Igata, R.; Kubo, T.; Tominaga, H.; Beppu, H.; Asahara, T.; et al. Effect of single caffeine intake on neuropsychological functions in healthy volunteers: A double-blind placebo-controlled study. PLoS ONE 2018, 13, e0202247. [Google Scholar] [CrossRef] [PubMed]
- Yu, N.Y.; Bieder, A.; Raman, A.; Mileti, E.; Katayama, S.; Einarsdottir, E.; Fredholm, B.B.; Falk, A.; Tapia-Páez, I.; Daub, C.O.; et al. Acute doses of caffeine shift nervous system cell expression profiles toward promotion of neuronal projection growth. Sci. Rep. 2017, 7, 11458. [Google Scholar] [CrossRef]
- Kawachi, I.; Willett, W.C.; Colditz, G.A.; Stampfer, M.J.; Speizer, F.E. A prospective study of coffee drinking and suicide in women. Arch. Intern. Med. 1996, 156, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, S.; Piacentino, D.; Fineschi, V.; Frati, P.; Cipolloni, L.; Aromatario, M. Caffeine-Related Deaths: Manner of Deaths and Categories at Risk. Nutrients 2018, 10, 611. [Google Scholar] [CrossRef]
- Tanskanen, A.; Tuomilehto, J.; Viinamäki, H.; Vartiainen, E.; Lehtonen, J.; Puska, P. Heavy coffee drinking and the risk of suicide. Eur. J. Epidemiol. 2000, 16, 789–791. [Google Scholar] [CrossRef]
- Lucas, M.; Mirzaei, F.; Pan, A.; Okereke, O.I.; Willett, W.C.; O’Reilly, É.J.; Koenen, K.; Ascherio, A. Coffee, Caffeine, and Risk of Depression Among Women. Arch. Intern. Med. 2011, 171, 1571–1578. [Google Scholar] [CrossRef]
- Tomar, N.; De, R.K. A Brief Outline of the Immune System. In Immunoinformatics; Humana Press: New York, NY, USA, 2014; Volume 1184, pp. 3–12. [Google Scholar] [CrossRef]
- Venter, C.; Eyerich, S.; Sarin, T.; Klatt, K.C. Nutrition and the Immune System: A Complicated Tango. Nutrients 2020, 12, 818. [Google Scholar] [CrossRef]
- Gibbs, B.F.; Silva, I.G.; Prokhorov, A.; Abooali, M.; Yasinska, I.; A Casely-Hayford, M.; Berger, S.M.; Fasler-Kan, E.; Sumbayev, V.V. Caffeine affects the biological responses of human hematopoietic cells of myeloid lineage via downregulation of the mTOR pathway and xanthine oxidase activity. Oncotarget 2015, 6, 28678–28692. [Google Scholar] [CrossRef]
- Chavez-Valdez, R.; Wills-Karp, M.; Ahlawat, R.; Cristofalo, E.A.; Nathan, A.; Gauda, E.B. Caffeine Modulates TNF-α Production by Cord Blood Monocytes: The Role of Adenosine Receptors. Pediatr. Res. 2009, 65, 203–208. [Google Scholar] [CrossRef]
- Chavez-Valdez, R.; Ahlawat, R.; Wills-Karp, M.; Gauda, E.B. Mechanisms of modulation of cytokine release by human cord blood monocytes exposed to high concentrations of caffeine. Pediatr. Res. 2016, 80, 101–109. [Google Scholar] [CrossRef]
- Steck, R.P.; Hill, S.L.; Weagel, E.; Weber, S.; Robison, R.A.; O’Neill, K.L. Pharmacologic immunosuppression of mononuclear phagocyte phagocytosis by caffeine. Pharmacol. Res. Perspect. 2015, 3, e00180. [Google Scholar] [CrossRef]
- Shushtari, N.; Froushani, S.M.A. Caffeine Augments the Instruction of Anti-Inflammatory Macrophages by the Conditioned Medium of Mesenchymal Stem Cells. Cell J. 2017, 19, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Bojarowicz, H.; Przygoda, M. Powszechność stosowania kofeiny oraz jej działanie na organizm Caffeine. Part, I. Common use of caffeine and its effect on human organism. Probl. Hig. Epidemiol. 2012, 93, 8–13. [Google Scholar]
- Lohsiriwat, S.; Puengna, N.; Leelakusolvong, S. Effect of caffeine on lower esophageal sphincter pressure in Thai healthy volunteers. Dis. Esophagus 2006, 19, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Helal, M.G.; Ayoub, S.E.; Elkashefand, W.F.; Ibrahim, T.M. Caffeine affects HFD-induced hepatic steatosis by multifactorial intervention. Hum. Exp. Toxicol. 2018, 37, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, N.; Nakamura, K.; Nakajima, K.; Suzuki, K.; Tatara, K. Coffee consumption and decreased serum γ-glutamyltransferase: A study of middle-aged Japanese men. Eur. J. Epidemiol. 2000, 16, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhou, Y.; Tang, L. Caffeine induces sustained apoptosis of human gastric cancer cells by activating the caspase-9/caspase-3 signalling pathway. Mol. Med. Rep. 2017, 16, 2445–2454. [Google Scholar] [CrossRef] [PubMed]
- Van Dyck, K.; Tas, S.; Robberecht, H.; Deelstra, H. The influence of different food components on thein vitroavailability of iron, zinc and calcium from a composed meal. Int. J. Food Sci. Nutr. 1996, 47, 499–506. [Google Scholar] [CrossRef]
- González, S.; Salazar, N.; Ruiz-Saavedra, S.; Gómez-Martín, M.; de los Reyes-Gavilán, C.G.; Gueimonde, M. Long-Term Coffee Consumption is Associated with Fecal Microbial Composition in Humans. Nutrients 2020, 12, 1287. [Google Scholar] [CrossRef]
- Salomone, F.; Galvano, F.; Volti, G.L. Molecular Bases Underlying the Hepatoprotective Effects of Coffee. Nutrients 2017, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Welsh, C.; Pan, J.; Belik, J. Caffeine impairs gastrointestinal function in newborn rats. Pediatr. Res. 2015, 78, 24–28. [Google Scholar] [CrossRef]
- Boekema, P.J.; Samsom, M.; Van Berge Henegouwen, G.P.; Smout, A.J.P.M. Coffee and gastrointestinal function: Facts and fiction: A review. Scand. J. Gastroenterol. Suppl. 1999, 33, 35–39. [Google Scholar]
- Rao, S.S.C.; Welcher, K.; Zimmerman, B.; Stumbo, P. Is coffee a colonie stimulant? Eur. J. Gastroenterol. Hepatol. 1998, 10, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.A.; Farah, B.L.; Singh, B.K.; Siddique, M.M.; Gili, L.M.; Wu, Y.; Ilkayeva, O.R.; Gooding, J.; Ching, J.; Zhou, J.; et al. Caffeine stimulates hepatic lipid metabolism by the autophagy-lysosomal pathway in mice. Hepatology 2014, 59, 1366–1380. [Google Scholar] [CrossRef]
- Murosaki, S.; Lee, T.R.; Muroyama, K.; Shin, E.S.; Cho, S.Y.; Yamamoto, Y.; Lee, S.J. A Combination of Caffeine, Arginine, Soy Isoflavones, and l-Carnitine Enhances Both Lipolysis and Fatty Acid Oxidation in 3T3-L1 and HepG2 Cells In Vitro and in KK Mice In Vivo. J. Nutr. 2007, 137, 2252–2257. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gallus, S.; Tavani, A.; Negri, E.; La Vecchia, C. Does Coffee Protect Against Liver Cirrhosis? Ann. Epidemiol. 2002, 12, 202–205. [Google Scholar] [CrossRef]
- Inoue, M.; Yoshimi, I.; Sobue, T.; Tsugane, S. Influence of Coffee Drinking on Subsequent Risk of Hepatocellular Carcinoma: A Prospective Study in Japan. J. Natl. Cancer Inst. 2005, 97, 293–300. [Google Scholar] [CrossRef]
- Modi, A.A.; Feld, J.J.; Park, Y.; Kleiner, D.; Everhart, J.E.; Liang, T.J.; Hoofnagle, J.H. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology 2009, 51, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, Y.; Huang, H.; Shi, M.; Yang, W.; Kuang, J.; Haiyan, H. Autophagy mediated by endoplasmic reticulum stress enhances the caffeine-induced apoptosis of hepatic stellate cells. Int. J. Mol. Med. 2017, 40, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
- Özgün, G.S.; Özgün, E.; Tabakçıoğlu, K.; Gökmen, S.S.; Eskiocak, S.; Çakır, E. Caffeine Increases Apolipoprotein A-1 and Paraoxonase-1 but not Paraoxonase-3 Protein Levels in Human-Derived Liver (HepG2) Cells. Balk. Med. J. 2017, 34, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Graham, T.E.; Sathasivam, P.; Rowland, M.; Marko, N.; Greer, F.; Battram, D. Caffeine ingestion elevates plasma insulin response in humans during an oral glucose tolerance test. Can. J. Physiol. Pharmacol. 2001, 79, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Keijzers, G.B.; De Galan, B.E.; Tack, C.J.; Smits, P. Caffeine Can Decrease Insulin Sensitivity in Humans. Diabetes Care 2002, 25, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.L.; Lopez-Garcia, E.; Li, T.Y.; Hu, F.B.; Van Dam, R.M. Coffee consumption and risk of cardiovascular events and all-cause mortality among women with type 2 diabetes. Diabetologia 2009, 52, 810–817. [Google Scholar] [CrossRef]
- Van Dam, R.M.; Feskens, E.J. Coffee consumption and risk of type 2 diabetes mellitus. Lancet 2002, 360, 1477–1478. [Google Scholar] [CrossRef]
- Carlsson, S.; Hammar, N.; Grill, V.; Kaprio, J. Coffee consumption and risk of type 2 daibetes in Finnish twins. Int. J. Epidemiol. 2004, 33, 616–617. [Google Scholar] [CrossRef]
- Rosengren, A.; Dotevall, A.; Wilhelmsen, L.; Thelle, D.; Johansson, S. Coffee and incidence of diabetes in Swedish women: A prospective 18-year follow-up study. J. Intern. Med. 2004, 255, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Tuomilehto, J.; Hu, G.; Bidel, S.; Lindström, J.; Jousilahti, P. Coffee Consumption and Risk of Type 2 Diabetes Mellitus among Middle-aged Finnish Men and Women. JAMA 2004, 291, 1213–1219. [Google Scholar] [CrossRef]
- Gilboe, D.P. The mechanism of caffeine-enhanced glucose stimulation of liver glycogen synthase phosphatase activity. Biochem. Pharmacol. 1986, 35, 2097–2105. [Google Scholar] [CrossRef]
- Van Dam, R.M. Coffee consumption and risk of type 2 diabetes, cardiovascular diseases, and cancer. Appl. Physiol. Nutr. Metab. 2008, 33, 1269–1283. [Google Scholar] [CrossRef]
- Temple, J.L. Caffeine use in children: What we know, what we have left to learn, and why we should worry. Neurosci. Biobehav. Rev. 2009, 33, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Ma, D.; Zhang, Y.; Zheng, W.; Wang, P. Coffee consumption and risk of colorectal cancer: A meta-analysis of observational studies. Public Health Nutr. 2013, 16, 346–357. [Google Scholar] [CrossRef]
- Tian, C.; Wang, W.; Hong, Z.; Zhang, X. Coffee consumption and risk of colorectal cancer: A dose–response analysis of observational studies. Cancer Causes Control. 2013, 24, 1265–1268. [Google Scholar] [CrossRef]
- Sang, L.-X.; Chang, B.; Li, X.-H.; Jiang, M. Consumption of coffee associated with reduced risk of liver cancer: A meta-analysis. BMC Gastroenterol. 2013, 13, 34. [Google Scholar] [CrossRef]
- Bravi, F.; Bosetti, C.; Tavani, A.; Gallus, S.; La Vecchia, C. Coffee Reduces Risk for Hepatocellular Carcinoma: An Updated Meta-analysis. Clin. Gastroenterol. Hepatol. 2013, 11, 1413–1421.e1. [Google Scholar] [CrossRef]
- Smith, A.P.; Nutt, D.J. Effects of upper respiratory tract illnesses, ibuprofen and caffeine on reaction time and alertness. Psychopharmacology 2014, 231, 1963–1974. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, C.; Lara, B.; de Souza, D.B.; Gutiérrez-Hellín, J.; Romero-Moraleda, B.; Cuéllar-Rayo, Á; Del Coso, J. Acute caffeine intake increases muscle oxygen saturation during a maximal incremental exercise test. Br. J. Clin. Pharmacol. 2020, 86, 861–867. [Google Scholar] [CrossRef]
- Supinski, G.S.; Deal, E.C.; Kelsen, S.G. The effects of caffeine and theophylline on diaphragm contractility. Am. Rev. Respir. Dis. 1984, 130, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Bruce, C.; Yates, D.H.; Thomas, P.S. Caffeine decreases exhaled nitric oxide. Thorax 2002, 57, 361–363. [Google Scholar] [CrossRef]
- Welsh, E.J.; Bara, A.; Barley, E.; Cates, C.J. Caffeine for asthma. Cochrane Database Syst. Rev. 2010, 2010, CD001112. [Google Scholar] [CrossRef]
- Hirayama, F.; Lee, A.H.; Yasukawa, K.; Ishihara, Y.; Shinjo, M. Caffeine Intake and the Risk of Chronic Obstructive Pulmonary Disease in Japanese Adults. J. Caffeine Res. 2012, 2, 176–179. [Google Scholar] [CrossRef]
- Lopes, P.O.; Alfaro, T.M.; Lopes, P.; Cunha, R.A.; Robalo Cordeiro, C. Caffeine consumption and exacerbations of chronic obstructive pulmonary disease: Retrospective study. Rev. Port. Pneumol. 2015, 21, 271–275. [Google Scholar] [CrossRef]
- Aranda, J.V.; Turmen, T.; Davis, J.; Trippenbach, T.; Grondin, D.; Zinman, R.; Watters, G. Effect of caffeine on control of breathing in infantile apnea. J. Pediatr. 1983, 103, 975–978. [Google Scholar] [CrossRef]
- Kassim, Z.; Greenough, A.; Rafferty, G. Effect of caffeine on respiratory muscle strength and lung function in prematurely born, ventilated infants. Eur. J. Nucl. Med. Mol. Imaging 2009, 168, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.M.; Bhutani, V.K.; Stefano, J.L.; Fox, W.W.; Spitzer, A.R. Changes in pulmonary mechanics following caffeine administration in infants with bronchopulmonary dysplasia. Pediatr. Pulmonol. 1989, 6, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.A.; McCann, S.E.; Reid, M.E.; Nowell, S.; Beehler, G.P.; Moysich, K.B. Associations Between Black Tea and Coffee Consumption and Risk of Lung Cancer Among Current and Former Smokers. Nutr. Cancer 2005, 52, 15–21. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, X.; Wu, Y.; Zhang, D. Coffee and tea consumption and risk of lung cancer: A dose–response analysis of observational studies. Lung Cancer 2012, 78, 169–170. [Google Scholar] [CrossRef] [PubMed]
- Noordzij, M.; Uiterwaal, C.S.P.M.; Arends, L.R.; Kok, F.J.; E Grobbee, D.; Geleijnse, J.M. Blood pressure response to chronic intake of coffee and caffeine: A meta-analysis of randomized controlled trials. J. Hypertens. 2005, 23, 921–928. [Google Scholar] [CrossRef]
- Robertson, D.; Frölich, J.C.; Carr, R.K.; Watson, J.T.; Hollifield, J.W.; Shand, D.G.; Oates, J.A. Effects of Caffeine on Plasma Renin Activity, Catecholamines and Blood Pressure. N. Engl. J. Med. 1978, 298, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, M.J. The pharmacology of caffeine. Prog. Drug Res. 1987, 31, 273–313. [Google Scholar] [CrossRef] [PubMed]
- Van Dusseldorp, M.; Smits, P.; Thien, T.; Katan, M.B. Effect of decaffeinated versus regular coffee on blood pressure. A 12-week, double-blind trial. Hypertension 1989, 14, 563–569. [Google Scholar] [CrossRef]
- Riksen, N.P.; Rongen, G.A.; Smits, P. Acute and long-term cardiovascular effects of coffee: Implications for coronary heart disease. Pharmacol. Ther. 2009, 121, 185–191. [Google Scholar] [CrossRef]
- Chaban, R.; Kornberger, A.; Branski, N.; Buschmann, K.; Stumpf, N.; Beiras-Fernandez, A.; Vahl, C. In-vitro examination of the positive inotropic effect of caffeine and taurine, the two most frequent active ingredients of energy drinks. BMC Cardiovasc. Disord. 2017, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jee, S.H.; He, J.; Appel, L.J.; Whelton, P.K.; Suh, I.; Klag, M.J. Coffee Consumption and Serum Lipids: A Meta-Analysis of Randomized Controlled Clinical Trials. Am. J. Epidemiol. 2001, 153, 353–362. [Google Scholar] [CrossRef]
- Melik, Z.; Princi, T.; Grill, V.; Cankar, K. The effect of caffeine on cutaneous postocclusive reactive hyperaemia. PLoS ONE 2019, 14, e0214919. [Google Scholar] [CrossRef]
- Ferre, S.; Fuxe, K.; Fredholm, B.B.; Morelli, M.; Popoli, P. Adenosine–dopamine receptor–receptor interactions as an integrative mechanism in the basal ganglia. Trends Neurosci. 1997, 20, 482–487. [Google Scholar] [CrossRef]
- Cappelletti, S.; Daria, P.; Sani, G.; Aromatario, M. Caffeine: Cognitive and Physical Performance Enhancer or Psychoactive Drug? Curr. Neuropharmacol. 2014, 13, 71–88. [Google Scholar] [CrossRef]
- Thames, M.D.; Kinugawa, T.; Dibner-Dunlap, M.E. Reflex sympathoexcitation by cardiac sympathetic afferents during myocardial ischemia. Role of adenosine. Circulation 1993, 87, 1698–1704. [Google Scholar] [CrossRef]
- Seitz, A.; Kaesemann, P.; Chatzitofi, M.; Löbig, S.; Tauscher, G.; Bekeredjian, R.; Sechtem, U.; Mahrholdt, H.; Greulich, S. Impact of caffeine on myocardial perfusion reserve assessed by semiquantitative adenosine stress perfusion cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2019, 21, 1–10. [Google Scholar] [CrossRef]
- Greenland, S. A Meta-Analysis of Coffee, Myocardial Infarction, and Coronary Death on JSTOR. Available online: https://www.jstor.org/stable/3702235?seq=1 (accessed on 5 January 2021).
- Marx, B.; Scuvée, É.; Scuvée-Moreau, J.; Seutin, V.; Jouret, F. Mécanismes de l’effet diurétique de la caféine. Médecine Sci. 2016, 32, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Arya, L.A.; Myers, D.L.; Jackson, N.D. Dietary caffeine intake and the risk for detrusor instability: A case-control study. Obstet. Gynecol. 2000, 96, 85–89. [Google Scholar] [CrossRef]
- Rieg, T.; Steigele, H.; Schnermann, J.; Richter, K.; Osswald, H.; Vallon, V. Requirement of Intact Adenosine A1Receptors for the Diuretic and Natriuretic Action of the Methylxanthines Theophylline and Caffeine. J. Pharmacol. Exp. Ther. 2005, 313, 403–409. [Google Scholar] [CrossRef]
- Wu, S.E.; Chen, W.-L. Exploring the Association between Urine Caffeine Metabolites and Urine Flow Rate: A Cross-Sectional Study. Nutrients 2020, 12, 2803. [Google Scholar] [CrossRef]
- Lohsiriwat, S.; Hirunsai, M.; Chaiyaprasithi, B. Effect of caffeine on bladder function in patients with overactive bladder symptoms. Urol. Ann. 2011, 3, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Jura, Y.H.; Townsend, M.K.; Curhan, G.C.; Resnick, N.M.; Grodstein, F. Caffeine Intake, and the Risk of Stress, Urgency and Mixed Urinary Incontinence. J. Urol. 2011, 185, 1775–1780. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, F.; Lee, A.H. Is Caffeine Intake Associated With Urinary Incontinence in Japanese Adults? J. Prev. Med. Public Health 2012, 45, 204–208. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Townsend, M.K.; Resnick, N.M.; Grodstein, F. Caffeine Intake and Risk of Urinary Incontinence Progression among Women. Obstet. Gynecol. 2012, 119, 950–957. [Google Scholar] [CrossRef]
- Gleason, J.L.; Richter, H.E.; Redden, D.T.; Goode, P.S.; Burgio, K.L.; Markland, A. Caffeine and urinary incontinence in US women. Int. Urogynecology J. 2013, 24, 295–302. [Google Scholar] [CrossRef]
- Curhan, G.C.; Willett, W.C.; Speizer, F.E.; Stampfer, M.J. Beverage Use and Risk for Kidney Stones in Women. Ann. Intern. Med. 1998, 128, 534–540. [Google Scholar] [CrossRef]
- Peerapen, P.; Thongboonkerd, V. Caffeine prevents kidney stone formation by translocation of apical surface annexin A1 crystal-binding protein into cytoplasm: In vitro evidence. Sci. Rep. 2016, 6, 38536. [Google Scholar] [CrossRef] [PubMed]
- Nilnumkhum, A.; Kanlaya, R.; Yoodee, S.; Thongboonkerd, V. Caffeine inhibits hypoxia-induced renal fibroblast activation by antioxidant mechanism. Cell Adhes. Migr. 2019, 13, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tian, C.; Jia, C. A dose–response meta-analysis of coffee consumption and bladder cancer. Prev. Med. 2012, 55, 14–22. [Google Scholar] [CrossRef]
- Wierzejska, R. Coffee Consumption vs. Cancer Risk-a Review of Scientific Data. Roczniki Państwowego Zakładu 2015, 66, 293–298. [Google Scholar]
- Xu, H.; Hu, L.; Liu, T.; Chen, F.; Li, J.; Xu, J.; Jiang, L.; Xiang, Z.; Wang, X.; Sheng, J. Caffeine Targets G6PDH to Disrupt Redox Homeostasis and Inhibit Renal Cell Carcinoma Proliferation. Front. Cell Dev. Biol. 2020, 8, 556162. [Google Scholar] [CrossRef] [PubMed]
- Del Coso, J.; Muñoz, G.; Muñoz-Guerra, J. Prevalence of caffeine use in elite athletes following its removal from the World Anti-Doping Agency list of banned substances. Appl. Physiol. Nutr. Metab. 2011, 36, 555–561. [Google Scholar] [CrossRef]
- Bazzucchi, I.; Felici, F.; Montini, M.; Figura, F.; Sacchetti, M. Caffeine improves neuromuscular function during maximal dynamic exercise. Muscle Nerve 2011, 43, 839–844. [Google Scholar] [CrossRef]
- Daly, J.W. Caffeine analogs: Biomedical impact. Cell. Mol. Life Sci. 2007, 64, 2153–2169. [Google Scholar] [CrossRef]
- Warren, G.L.; Park, N.D.; Maresca, R.D.; Mckibans, K.I.; Millard-Stafford, M.L. Effect of Caffeine Ingestion on Muscular Strength and Endurance: A meta-analysis. Med. Sci. Sports Exerc. 2010, 42, 1375–1387. [Google Scholar] [CrossRef] [PubMed]
- Astorino, T.A.; Terzi, M.N.; Roberson, D.W.; Burnett, T.R. Effect of caffeine intake on pain perception during high-intensity exercise. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Domaszewski, P.; Pakosz, P.; Konieczny, M.; Bączkowicz, D.; Sadowska-Krępa, E. Caffeine-Induced Effects on Human Skeletal Muscle Contraction Time and Maximal Displacement Measured by Tensiomyography. Nutrients 2021, 13, 815. [Google Scholar] [CrossRef]
- Shin, J.; Choi, Y.; Kim, J.; Yu, A.-R.; Shin, J.-S.; Choi, Y.-Y.; Roh, J. High doses of caffeine reduce in vivo osteogenic activity in prepubertal rats. J. Anat. 2015, 227, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R. Effects of caffeine on bone and the calcium economy. Food Chem. Toxicol. 2002, 40, 1263–1270. [Google Scholar] [CrossRef]
- Yokokawa, T.; Hashimoto, T.; Iwanaka, N. Caffeine increases myoglobin expression via the cyclic AMP pathway in L6 myotubes. Physiol. Rep. 2021, 9, e14869. [Google Scholar] [CrossRef]
- Tazzeo, T.; Bates, G.; Roman, H.N.; Lauzon, A.-M.; Khasnis, M.D.; Eto, M.; Janssen, L.J. Caffeine relaxes smooth muscle through actin depolymerization. Am. J. Physiol. Cell. Mol. Physiol. 2012, 303, L334–L342. [Google Scholar] [CrossRef] [PubMed]
- Wilk, M.; Krzysztofik, M.; Filip, A.; Zajac, A.; Del Coso, J. The Effects of High Doses of Caffeine on Maximal Strength and Muscular Endurance in Athletes Habituated to Caffeine. Nutrients 2019, 11, 1912. [Google Scholar] [CrossRef]
- Cesareo, K.R.; Mason, J.R.; Saracino, P.G.; Morrissey, M.C.; Ormsbee, M.J. The effects of a caffeine-like supplement, TeaCrine®, on muscular strength, endurance and power performance in resistance-trained men. J. Int. Soc. Sports Nutr. 2019, 16, 1–11. [Google Scholar] [CrossRef]
- Tan, B.L.; Norhaizan, M.E.; Liew, W.-P. Nutrients and Oxidative Stress: Friend or Foe? Oxidative Med. Cell. Longev. 2018, 2018, 9719584. [Google Scholar] [CrossRef]
- Martini, D.; Del Bo’, C.; Tassotti, M.; Riso, P.; Del Rio, D.; Brighenti, F.; Porrini, M. Coffee consumption and oxidative stress: A review of human intervention studies. Molecules 2016, 21, 979. [Google Scholar] [CrossRef]
- Zeidán-Chuliá, F.; Gelain, D.P.; Kolling, E.A.; Filho, J.L.R.; Ambrosi, P.; Terra, S.R.; Pires, A.S.; da Rocha, J.B.T.; Behr, G.A.; Moreira, J.C.F. Major Components of Energy Drinks (Caffeine, Taurine, and Guarana) Exert Cytotoxic Effects on Human Neuronal SH-SY5Y Cells by Decreasing Reactive Oxygen Species Production. Oxidative Med. Cell. Longev. 2013, 2013, 1–22. [Google Scholar] [CrossRef]
- Kaczmarczyk-Sedlak, I.; Folwarczna, J.; Sedlak, L.; Zych, M.; Wojnar, W.; Szumińska, I.; Wyględowska-Promieńska, D.; Mrukwa-Kominek, E. Effect of caffeine on biomarkers of oxidative stress in lenses of rats with streptozotocin-induced diabetes. Arch. Med. Sci. 2019, 15, 1073–1080. [Google Scholar] [CrossRef]
- Chu, Y.-F.; Brown, P.H.; Lyle, B.J.; Chen, Y.; Black, R.M.; Williams, C.E.; Lin, Y.-C.; Hsu, C.-W.; Cheng, I.H. Roasted Coffees High in Lipophilic Antioxidants and Chlorogenic Acid Lactones Are More Neuroprotective than Green Coffees. J. Agric. Food Chem. 2009, 57, 9801–9808. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-F.; Ouyang, S.-H.; Tu, L.-F.; Wang, X.; Yuan, W.-L.; Wang, G.-E.; Wu, Y.-P.; Duan, W.-J.; Yu, H.-M.; Fang, Z.-Z.; et al. Caffeine Protects Skin from Oxidative Stress-Induced Senescence through the Activation of Autophagy. Theranostics 2018, 8, 5713–5730. [Google Scholar] [CrossRef]
- Xu, H.; Gan, C.; Gao, Z.; Huang, Y.; Wu, S.; Zhang, D.; Wang, X.; Sheng, J. Caffeine Targets SIRT3 to Enhance SOD2 Activity in Mitochondria. Front. Cell Dev. Biol. 2020, 8, 822. [Google Scholar] [CrossRef]
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, Caffeine, and Health Outcomes: An Umbrella Review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Kajio, H.; Sugiyama, T. Association between Caffeine Intake and All-Cause and Cause-Specific Mortality: A Population-Based Prospective Cohort Study. Mayo Clin. Proc. 2017, 92, 1190–1202. [Google Scholar] [CrossRef] [PubMed]
- Neves, J.S.; Leitão, L.; Magriço, R.; Vieira, M.B.; Dias, C.V.; Oliveira, A.; Carvalho, D.; Claggett, B. Caffeine Consumption and Mortality in Diabetes: An Analysis of NHANES 1999–2010. Front. Endocrinol. 2018, 9, 547. [Google Scholar] [CrossRef]
- Vieira, M.B.; Magriço, R.; Dias, C.V.; Leitão, L.; Neves, J.S. Caffeine consumption and mortality in chronic kidney disease: A nationally representative analysis. Nephrol. Dial. Transplant. 2019, 34, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Liu, Y.; Sun, X.; Yin, Z.; Li, H.; Cheng, C.; Liu, L.; Zhang, R.; Liu, F.; Zhou, Q.; et al. Caffeinated and decaffeinated coffee consumption and risk of all-cause mortality: A dose–response meta-analysis of cohort studies. J. Hum. Nutr. Diet. 2019, 32, 279–287. [Google Scholar] [CrossRef]
| Disease/Disorder | Mechanism | Effect |
|---|---|---|
| Alzheimer’s disease | ↓ Aβ generation, activation of ARs, antioxidant activity | ↓ risk of disease [45,50,97] |
| Parkinson’s disease | Activation of ARs | ↓ risk of disease [47,51,98] |
| Huntington’s disease | Unknown | ↑ risk of disease [57] |
| Depression | Antagonism of ARs | ↓ risk of disease [45,96,102] |
| Mood | Antagonism of ARs, inhibition of AChE, impact on RyRs | stimulation at low doses [8,27] ↑ restlessness, excitement, tremor, tinnitus, headache, and insomnia at high doses [30,34] ↑ anxiety [94,95,96] |
| Disease | Effect |
|---|---|
| Breathing problems | ↑ ventilation [150,151] ↑ tidal volume [150,151] ↑ inspiratory flow [150] ↓ total lung resistance [151] |
| Asthma COPD | ↑ airways functions [146] ↑ risk of COPD [147] |
| Lung cancer | ↑ risk of lung cancer [152] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodak, K.; Kokot, I.; Kratz, E.M. Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe? Nutrients 2021, 13, 3088. https://doi.org/10.3390/nu13093088
Rodak K, Kokot I, Kratz EM. Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe? Nutrients. 2021; 13(9):3088. https://doi.org/10.3390/nu13093088
Chicago/Turabian StyleRodak, Kamil, Izabela Kokot, and Ewa Maria Kratz. 2021. "Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe?" Nutrients 13, no. 9: 3088. https://doi.org/10.3390/nu13093088
APA StyleRodak, K., Kokot, I., & Kratz, E. M. (2021). Caffeine as a Factor Influencing the Functioning of the Human Body—Friend or Foe? Nutrients, 13(9), 3088. https://doi.org/10.3390/nu13093088






