Kidney Function-Dependence of Vitamin K-Status Parameters: Results from the TransplantLines Biobank and Cohort Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population—Cohort 1
2.2. Study Population—Cohort 2
2.3. Clinical Assessment
2.4. Biochemical Analyses
2.5. Statistical Analyses
3. Results
3.1. Baseline Characteristics
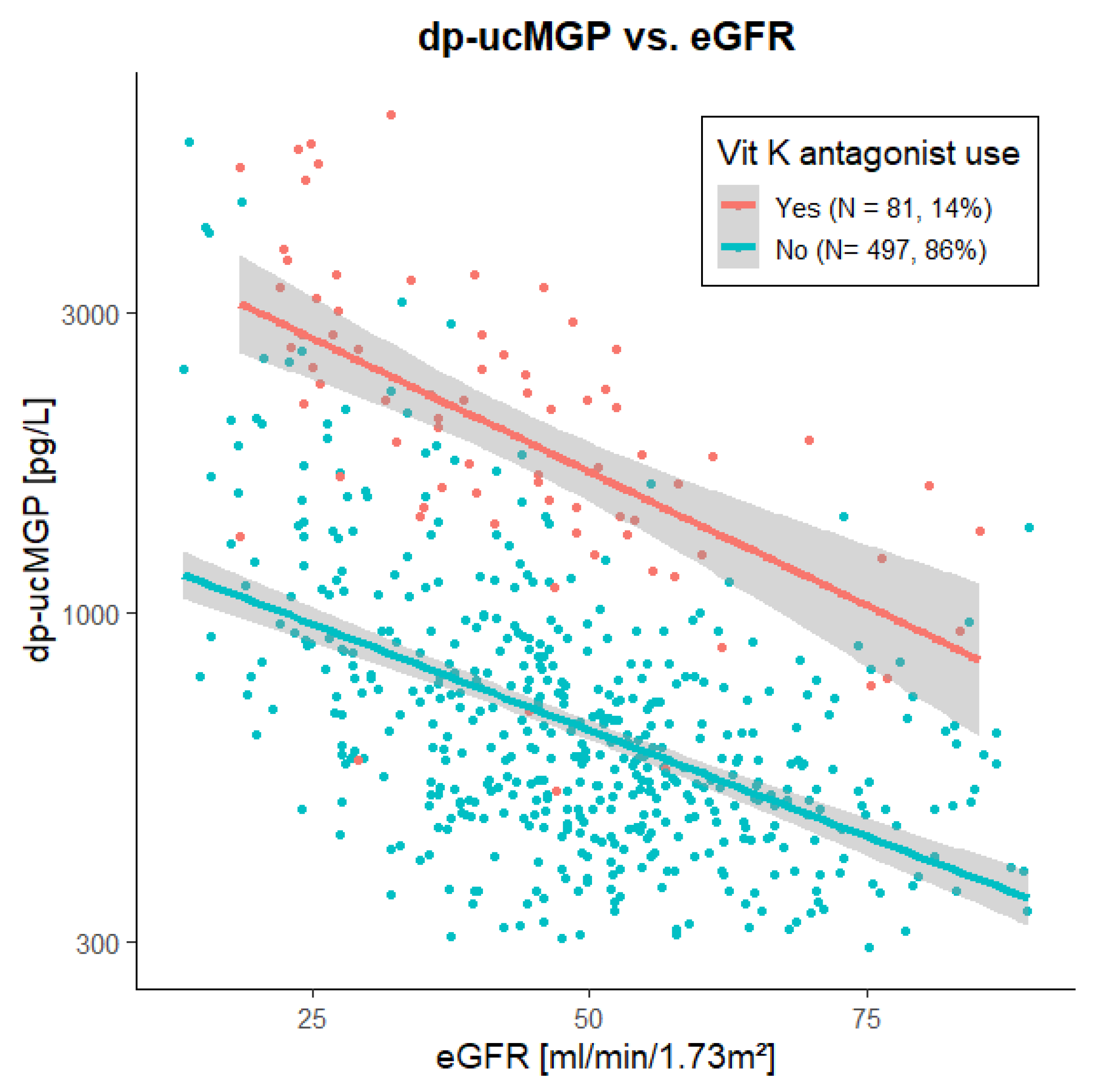
3.2. Association of dp-ucMGP with Kidney Function in Cohort 1
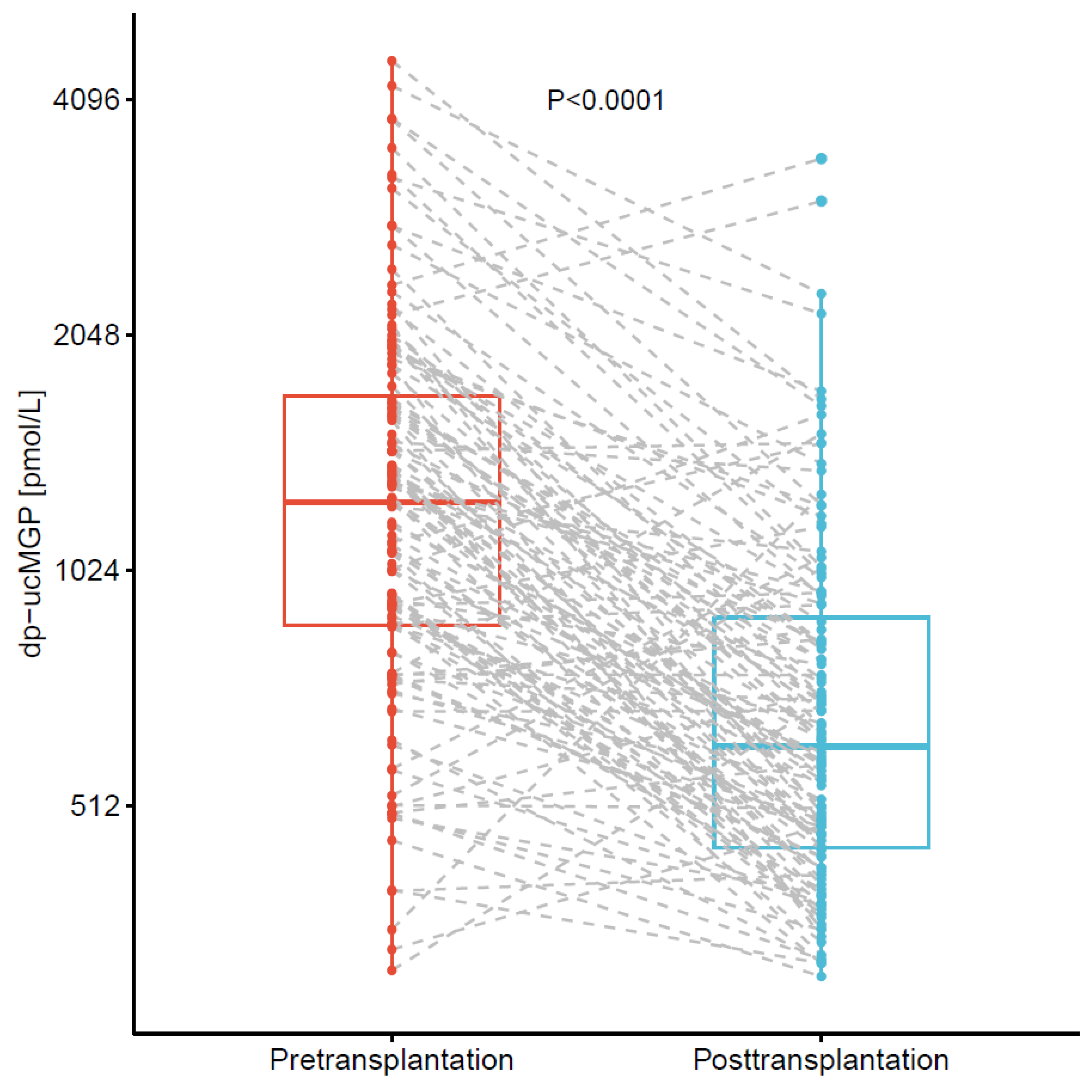
3.3. Changes in of dp-ucMGP after Kidney Transplantation in Cohort 2
3.4. Correlations and Associations of dp-ucMGP, dp-cMGP, ucOC, and cOC in Cohort 1
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schurgers, L.J.; Dissel, P.E.P.; Spronk, H.M.H.; Soute, B.A.M.; Dhore, C.R.; Cleutjens, J.P.M.; Vermeer, C. Role of vitamin K and vitamin K-dependent proteins in vascular calcification. Z. Kardiol. 2001, 90, 57–63. [Google Scholar] [CrossRef]
- Kyla Shea, M.; Holden, R.M. Vitamin K status and vascular calcification: Evidence from observational and clinical studies. Adv. Nutr. 2012, 3, 158–165. [Google Scholar] [CrossRef]
- Villa, J.K.D.; Diaz, M.A.N.; Pizziolo, V.R.; Martino, H.S.D. Effect of vitamin K in bone metabolism and vascular calcification: A review of mechanisms of action and evidences. Crit. Rev. Food Sci. Nutr. 2017, 57, 3959–3970. [Google Scholar] [CrossRef]
- Davie, E.W. Biochemical and molecular aspects of the coagulation cascade. Thromb. Haemost. 1995, 74, 1–6. [Google Scholar] [CrossRef]
- Keyzer, C.A.; Vermeer, C.; Joosten, M.M.; Knapen, M.H.J.; Drummen, N.E.A.; Navis, G.; Bakker, S.J.L.; De Borst, M.H. Vitamin K status and mortality after kidney transplantation: A cohort study. Am. J. Kidney Dis. 2015, 65, 474–483. [Google Scholar] [CrossRef]
- Silaghi, C.N.; Ilyés, T.; Filip, V.P.; Farcaș, M.; van Ballegooijen, A.J.; Crăciun, A.M. Vitamin k dependent proteins in kidney disease. Int. J. Mol. Sci. 2019, 20, 1571. [Google Scholar] [CrossRef]
- Van Den Heuvel, E.G.H.M.; Van Schoor, N.M.; Lips, P.; Magdeleyns, E.J.P.; Deeg, D.J.H.; Vermeer, C.; Heijer, M. Den Circulating uncarboxylated matrix Gla protein, a marker of vitamin K status, as a risk factor of cardiovascular disease. Maturitas 2014, 77, 137–141. [Google Scholar] [CrossRef]
- Lees, J.S.; Chapman, F.A.; Witham, M.D.; Jardine, A.G.; Mark, P.B. Vitamin K status, supplementation and vascular disease: A systematic review and meta-analysis. Heart 2019, 105, 938–945. [Google Scholar] [CrossRef]
- Schlieper, G.; Westenfeld, R.; Krüger, T.; Cranenburg, E.C.; Magdeleyns, E.J.; Brandenburg, V.M.; Djuric, Z.; Damjanovic, T.; Ketteler, M.; Vermeer, C.; et al. Circulating nonphosphorylated carboxylated matrix gla protein predicts survival in ESRD. J. Am. Soc. Nephrol. 2011, 22, 387–395. [Google Scholar] [CrossRef]
- Schurgers, L.J.; Barreto, D.V.; Barreto, F.C.; Liabeuf, S.; Renard, C.; Magdeleyns, E.J.; Vermeer, C.; Choukroun, G.; Massy, Z.A. The circulating inactive form of matrix gla protein is a surrogate marker for vascular calcification in chronic kidney disease: A preliminary report. Clin. J. Am. Soc. Nephrol. 2010, 5, 568–575. [Google Scholar] [CrossRef]
- Schurgers, L.J.; Vermeer, C. Determination of phylloquinone and menaquinones in food: Effect of food matrix on circulating vitamin K concentrations. Haemostasis 2000, 30, 298–307. [Google Scholar] [CrossRef]
- Vermeer, C. γ-Carboxyglutamate-containing proteins and the vitamin K-dependent carboxylase. Biochem. J. 1990, 266, 625–636. [Google Scholar] [CrossRef]
- Schurgers, L.J.; Spronk, H.M.H.; Skepper, J.N.; Hackeng, T.M.; Shanahan, C.M.; Vermeer, C.; Weissberg, P.L.; Proudfoot, D. Post-translational modifications regulate matrix Gla protein function: Importance for inhibition of vascular smooth muscle cell calcification. J. Thromb. Haemost. 2007, 5, 2503–2511. [Google Scholar] [CrossRef] [PubMed]
- Hackeng, T.M. Total chemical synthesis of human matrix Gla protein. Protein Sci. 2001, 10, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Inker, L.A.; Okparavero, A. Cystatin C as a marker of glomerular filtration rate: Prospects and limitations. Curr. Opin. Nephrol. Hypertens. 2011, 20, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Abrahamson, M.; Olafsson, I.; Palsdottir, A.; Ulvsback, M.; Lundwall, A.; Jensson, O.; Grubb, A. Structure and expression of the human cystatin C gene. Biochem. J. 1990, 268, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Gundberg, C.M.; Lian, J.B.; Booth, S.L. Vitamin K-dependent carboxylation of osteocalcin: Friend or foe? Adv. Nutr. 2012, 3, 149–157. [Google Scholar] [CrossRef]
- Millar, S.A.; Patel, H.; Anderson, S.I.; England, T.J.; O’Sullivan, S.E. Osteocalcin, vascular calcification, and atherosclerosis: A systematic review and meta-analysis. Front. Endocrinol. 2017, 8, 183. [Google Scholar] [CrossRef]
- Wei, F.F.; Trenson, S.; Thijs, L.; Huang, Q.F.; Zhang, Z.Y.; Yang, W.Y.; Moliterno, P.; Allegaert, K.; Boggia, J.; Janssens, S.; et al. Desphospho-uncarboxylated matrix Gla protein is a novel circulating biomarker predicting deterioration of renal function in the general population. Nephrol. Dial. Transplant. 2018, 33, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Dofferhoff, A.S.M.; Piscaer, I.; Schurgers, L.J.; Visser, M.P.J.; van den Ouweland, J.M.W.; de Jong, P.A.; Gosens, R.; Hackeng, T.M.; van Daal, H.; Lux, P.; et al. Reduced Vitamin K Status as a Potentially Modifiable Risk Factor of Severe Coronavirus Disease 2019. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Groothof, D.; Post, A.; Sotomayor, C.G.; Keyzer, C.A.; Flores-Guerero, J.L.; Hak, E.; Bos, J.H.J.; Schurgers, L.J.; Navis, G.J.; Gans, R.O.B.; et al. Functional vitamin K status and risk of incident chronic kidney disease and microalbuminuria: A prospective general population-based cohort study. Nephrol. Dial. Transplant. 2020, 1, 10. [Google Scholar] [CrossRef]
- Groothof, D.; Post, A.; Gans, R.O.B.; Bakker, S.J.L. Reduced Vitamin K Status and Coronavirus Disease 2019: An Epiphenomenon of Impaired Kidney Function? Clin. Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Van Den Berg, E.; Engberink, M.F.; Brink, E.J.; Van Baak, M.A.; Gans, R.O.B.; Navis, G.; Bakker, S.J.L. Dietary protein, blood pressure and renal function in renal transplant recipients. Br. J. Nutr. 2013, 109, 1463–1470. [Google Scholar] [CrossRef]
- Eisenga, M.F.; Gomes-Neto, A.W.; Van Londen, M.; Ziengs, A.L.; Douwes, R.M.; Stam, S.P.; Osté, M.C.J.; Knobbe, T.J.; Hessels, N.R.; Buunk, A.M.; et al. Rationale and design of TransplantLines: A prospective cohort study and biobank of solid organ transplant recipients. BMJ Open 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Boxma, P.Y.; van den Berg, E.; Geleijnse, J.M.; Laverman, G.D.; Schurgers, L.J.; Vermeer, C.; Kema, I.P.; Muskiet, F.A.; Navis, G.; Bakker, S.J.L.; et al. Vitamin K Intake and Plasma Desphospho-Uncarboxylated Matrix Gla-Protein Levels in Kidney Transplant Recipients. PLoS ONE 2012, 7, e047991. [Google Scholar] [CrossRef]
- American Diabetes Association Standards of Medical Care in Diabetes. Diabetes Care 2017, 40, S11–S24.
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Inker, L.A.; Eckfeldt, J.; Levey, A.S.; Leiendecker-Foster, C.; Rynders, G.; Manzi, J.; Waheed, S.; Coresh, J. Expressing the CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) cystatin C equations for estimating GFR with standardized serum cystatin C Values. Am. J. Kidney Dis. 2011, 58, 682–684. [Google Scholar] [CrossRef]
- Jaminon, A.M.G.; Dai, L.; Qureshi, A.R.; Evenepoel, P.; Ripsweden, J.; Söderberg, M.; Witasp, A.; Olauson, H.; Schurgers, L.J.; Stenvinkel, P. Matrix Gla protein is an independent predictor of both intimal and medial vascular calcification in chronic kidney disease. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cranenburg, E.C.M.; Schurgers, L.J.; Uiterwijk, H.H.; Beulens, J.W.J.; Dalmeijer, G.W.; Westerhuis, R.; Magdeleyns, E.J.; Herfs, M.; Vermeer, C.; Laverman, G.D. Vitamin K intake and status are low in hemodialysis patients. Kidney Int. 2012, 82, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Holden, R.M.; Morton, A.R.; Garland, J.S.; Pavlov, A.; Day, A.G.; Booth, S.L. Vitamins K and D status in stages 3-5 chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 590–597. [Google Scholar] [CrossRef]
- Rennenberg, R.J.M.W.; Schurgers, L.J.; Vermeer, C.; Scholte, J.B.J.; Houben, A.J.H.M.; De Leeuw, P.W.; Kroon, A.A. Renal Handling of Matrix Gla-Protein in Humans with Moderate to Severe Hypertension. Hypertens. Res. 2008, 31, 1745–1751. [Google Scholar] [CrossRef][Green Version]
- Parker, B.D.; Ix, J.H.; Cranenburg, E.C.M.; Vermeer, C.; Whooley, M.A.; Schurgers, L.J. Association of kidney function and uncarboxylated matrix Gla protein: Data from the Heart and Soul Study. Nephrol. Dial. Transplant. 2009, 24, 2095–2101. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.F.; Drummen, N.E.A.; Schutte, A.E.; Thijs, L.; Jacobs, L.; Petit, T.; Yang, W.Y.; Smith, W.; Zhang, Z.Y.; Gu, Y.M.; et al. Vitamin K Dependent Protection of Renal Function in Multi-ethnic Population Studies. EBioMedicine 2016, 4, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Roumeliotis, S.; Dounousi, E.; Eleftheriadis, T.; Liakopoulos, V. Association of the inactive circulating matrix Gla protein with vitamin K intake, calcification, mortality, and cardiovascular disease: A review. Int. J. Mol. Sci. 2019, 20, 628. [Google Scholar] [CrossRef] [PubMed]
- Hoang, Q.Q.; Sicheri, F.; Howard, A.J.; Yang, D.S.C. Bone recognition mechanism of porcine osteocalcin from crystal structure. Nature 2003, 425, 977–980. [Google Scholar] [CrossRef]
- Card, D.J.; Gorska, R.; Harrington, D.J. Laboratory assessment of vitamin K status. J. Clin. Pathol. 2020, 73, 70–75. [Google Scholar] [CrossRef] [PubMed]


| Cohort 1 N = 578 | Cohort 2 N = 124 with Serial Measurements | ||
|---|---|---|---|
| Time of Blood Sampling | 7.5 (3.2 to 13.7) Years after Transplantation | At Transplantation | ~3 Months after Transplantation |
| dp-ucMGP, pmol/L | 671 (495 to 1076) | 1252 (868 to 1744) | 609 (451 to 914) |
| Clinical characteristics | |||
| Female sex, n (%) | 237 (41) | 45 (36) | - |
| Age, years | 56 (13) | 53 (14) | - |
| Height, cm | 173 (10) | 175 (10) | - |
| Weight, kg | 82 (16) | 81 (15) | 82 (15) |
| Systolic blood pressure, mmHg | 135 (17) | 140 (19) | 133 (14) |
| Diabetes, n (%) | 146 (25) | 17 (14) | 32 (26) |
| Laboratory measurements | |||
| Hemoglobin, mmol/L | 8.5 (1.1) | 7.5 (1.0) | 7.9 (1.1) |
| Sodium, mmol/L | 140.1 (2.7) | 139.0 (2.5) | 140.0 (2.4) |
| Potassium, mmol/L | 4.0 (0.4) | 4.6 (0.7) | 4.0 (0.4) |
| Creatinine, µmol/L | 129 (108 to 163) | 595 (441 to 776) | 130 (111 to 161) |
| eGFR, mL/min/1.73 m2 | 49 (17) | - | 49 (14) |
| Urea, mmol/L | 8.8 (6.7 to 12.0) | 22.3 (16.8 to 28.3) | 8.4 (6.4 to 10.4) |
| HbA1c, mmol/mol | 39 (36 to 45) | 36 (34 to 40) | 40 (36 to 46) |
| Medication use | |||
| Vitamin K-antagonists, n (%) | 81 (14) | 12 (10) Discontinued in 9 (7) prior to planned surgery | 17 (14) |
| Prednisolone, n (%) | 559 (97) | 24 (19) | 123 (99) |
| Calcineurin inhibitor, n (%) | 414 (72) | - | 123 (99) |
| Proliferation inhibitor, n (%) | 476 (82) | - | 117 (94) |
| mTOR inhibitor, n (%) | 20 (3) | - | 4 (3) |
| Other vit. K-associated parameters | Subgroup (N = 60) | ||
| dp-cMGP, pmol/L | 2787 (2169 to 3688) | ||
| Proportion uncarboxylated MGP, % | 21 (16 to 25) | ||
| ucOC, pmol/L | 1.07 (0.49 to 3.95) | ||
| cOC, pmol/L | 6.2 (3.2 to 9.9) | ||
| Proportion uncarboxylated OC, % | 15 (9 to 31) | ||
| Variable | Change in dp-ucMGP | T-Value | p-Value | Model R2 | |
|---|---|---|---|---|---|
| Model 1 | Vitamin K-antagonist use, yes vs. no | +181.3% | - | <0.001 | 0.348 |
| Model 2 | eGFR, per 10 mL/min/1.73 m2 increase | −15.4% | - | <0.001 | 0.224 |
| Model 3 | Vitamin K-antagonist use, yes vs. no | +165.1% | 19.4 | <0.001 | 0.531 |
| eGFR, per 10 mL/min/1.73 m2 increase | −14.0% | −15.0 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kremer, D.; Groothof, D.; Keyzer, C.A.; Eelderink, C.; Knobbe, T.J.; Post, A.; van Londen, M.; Eisenga, M.F.; TransplantLines Investigators; Schurgers, L.J.; et al. Kidney Function-Dependence of Vitamin K-Status Parameters: Results from the TransplantLines Biobank and Cohort Studies. Nutrients 2021, 13, 3069. https://doi.org/10.3390/nu13093069
Kremer D, Groothof D, Keyzer CA, Eelderink C, Knobbe TJ, Post A, van Londen M, Eisenga MF, TransplantLines Investigators, Schurgers LJ, et al. Kidney Function-Dependence of Vitamin K-Status Parameters: Results from the TransplantLines Biobank and Cohort Studies. Nutrients. 2021; 13(9):3069. https://doi.org/10.3390/nu13093069
Chicago/Turabian StyleKremer, Daan, Dion Groothof, Charlotte A. Keyzer, Coby Eelderink, Tim J. Knobbe, Adrian Post, Marco van Londen, Michele F. Eisenga, TransplantLines Investigators, Leon J. Schurgers, and et al. 2021. "Kidney Function-Dependence of Vitamin K-Status Parameters: Results from the TransplantLines Biobank and Cohort Studies" Nutrients 13, no. 9: 3069. https://doi.org/10.3390/nu13093069
APA StyleKremer, D., Groothof, D., Keyzer, C. A., Eelderink, C., Knobbe, T. J., Post, A., van Londen, M., Eisenga, M. F., TransplantLines Investigators, Schurgers, L. J., Berger, S. P., de Borst, M. H., & Bakker, S. J. L. (2021). Kidney Function-Dependence of Vitamin K-Status Parameters: Results from the TransplantLines Biobank and Cohort Studies. Nutrients, 13(9), 3069. https://doi.org/10.3390/nu13093069









