Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Camellia japonica Bee Pollen and Extraction (CPE)
2.2. Chemicals and Materials
2.3. Phenolic Compound and Antioxidant Activity of CPE
2.3.1. HPLC Analysis Determination of CPE
2.3.2. Determination of Total Phenolic Content (TPC)
2.3.3. Determination of Total Flavonoid Content (TFC)
2.3.4. DPPH Radical Scavenging Activity
2.3.5. Ferrous Ion-Chelating Activity
2.3.6. Ferric Reducing Antioxidant Power (FRAP)
2.3.7. Effect of CPE on DNA Oxidative Damage Induced by Hydroxyl Radicals
2.4. Animals Experiment
2.4.1. Hyperuricemic Mice Experiment Design
2.4.2. Serum, Liver and Kidney Biochemical Analysis
2.4.3. Histopathological Examination
2.4.4. Quantitative Real-Time PCR Analysis
2.4.5. Gut Microbiota Analysis
2.4.6. SCFA Analysis
2.5. Statistical Analysis
3. Results and Discussion
3.1. The Phenolic Compound and Antioxidant Activity of CPE-E and CPE-H
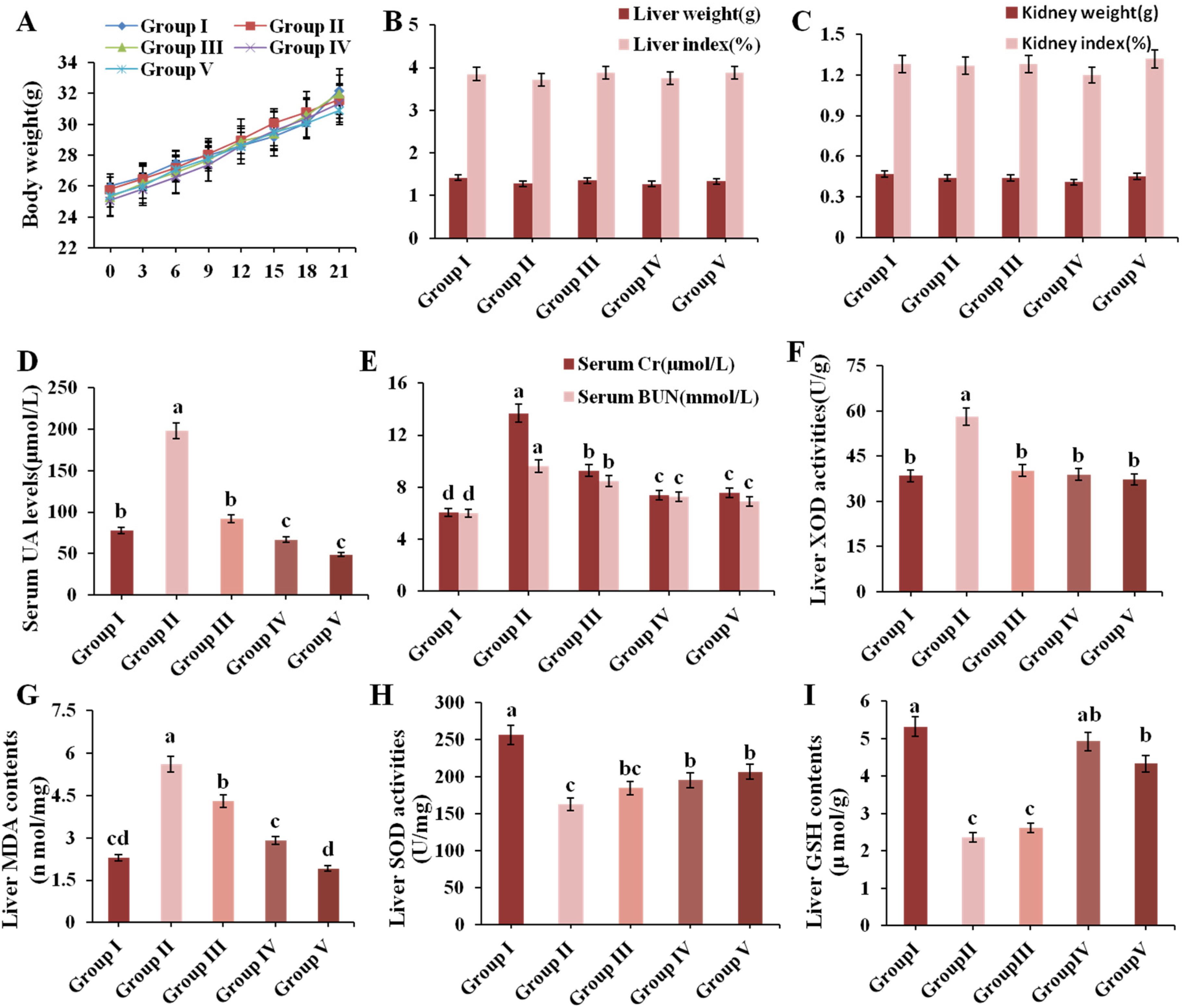
3.2. Effect of CPE-E on Body and Organ Weight and Serum Biochemical Parameters in Mice
3.3. Effect of CPE-E on XOD Activity in Liver
3.4. Effect of CPE-E on Antioxidant Status
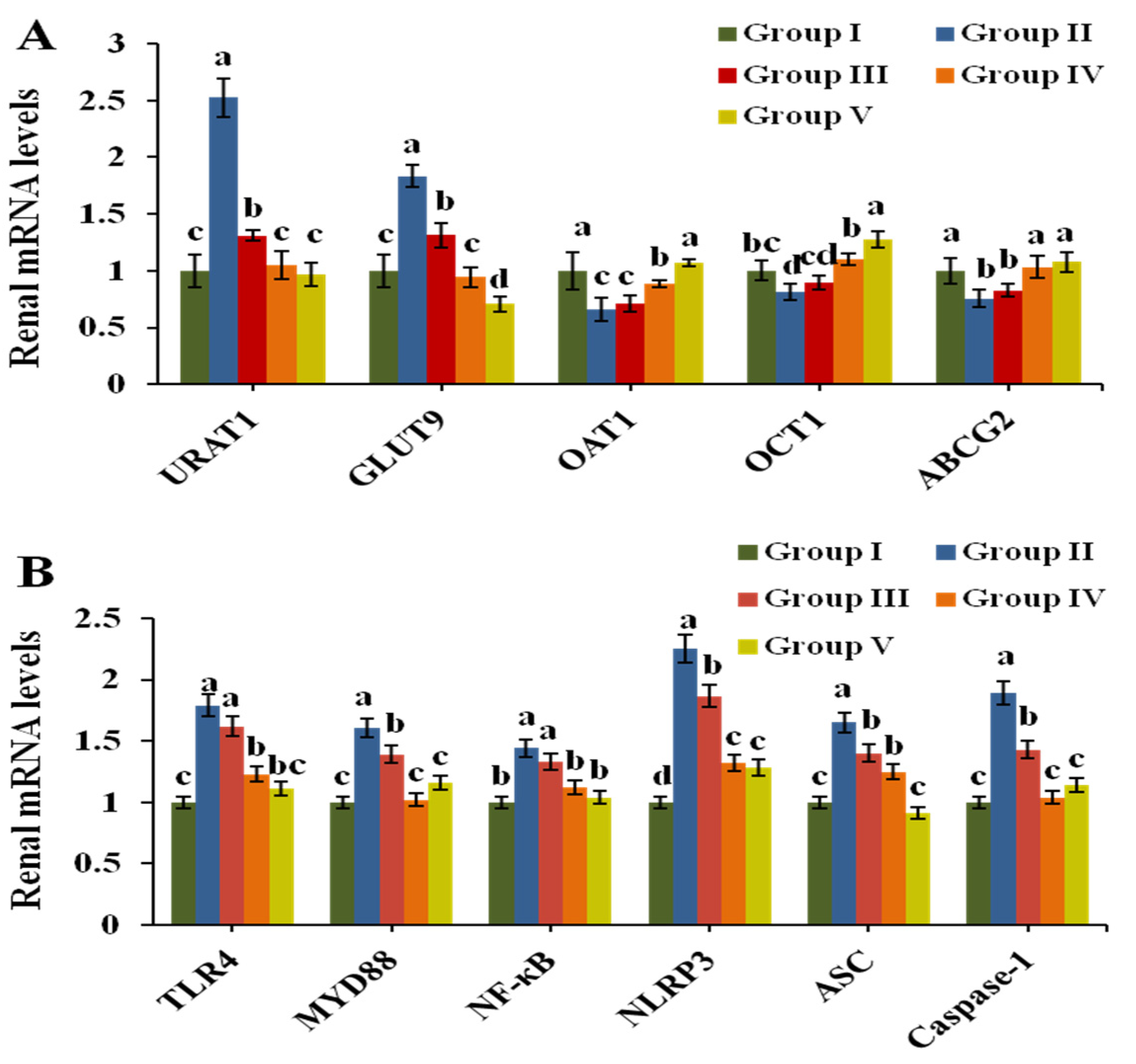
3.5. Effect of CPE-E on the Related Transporters in Renal Tissue
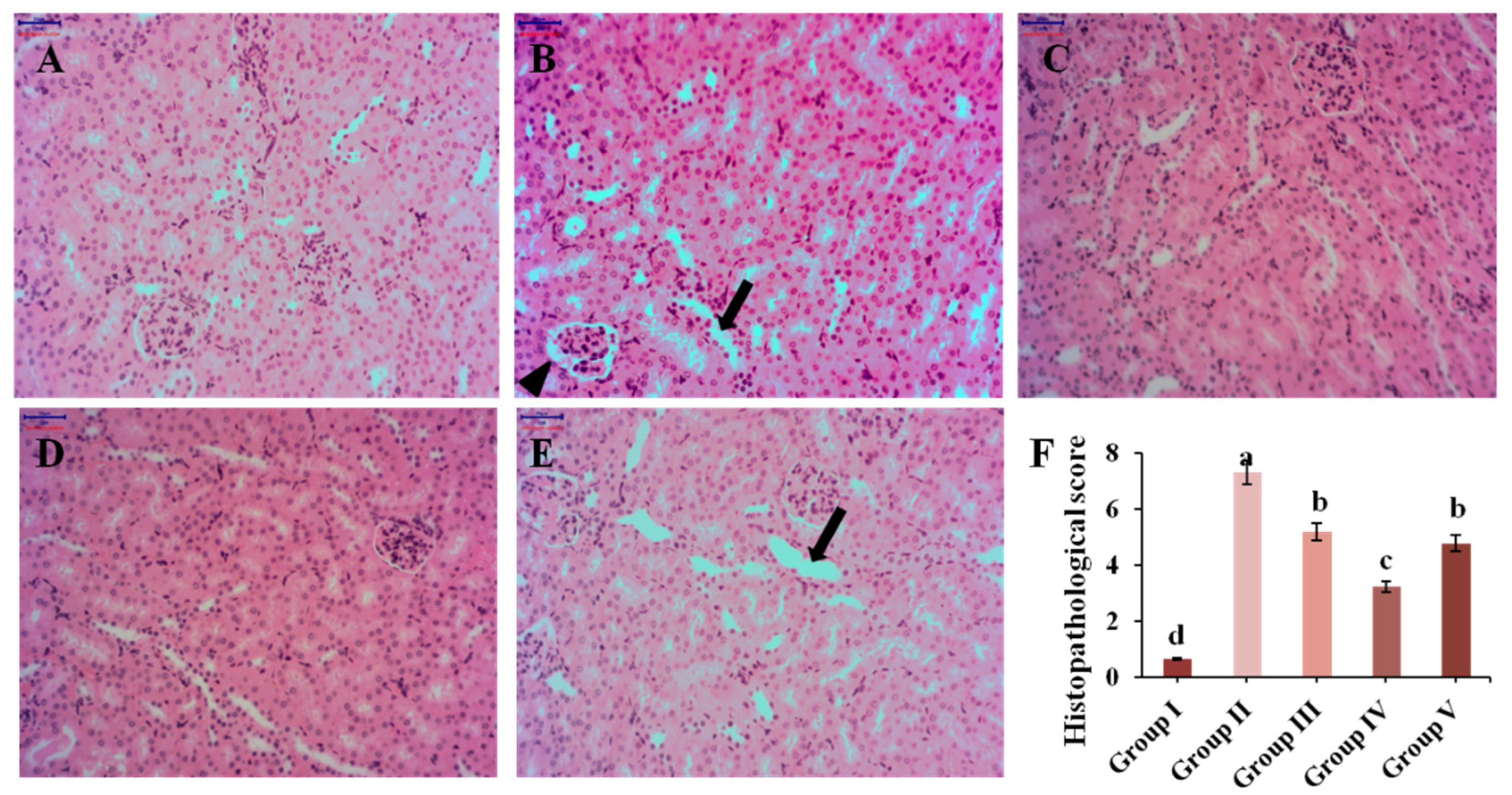
3.6. Histopathological Analysis
3.7. Effect of CPE-E on Inflammatory Cytokines in Renal and Serum
3.8. Effect of CPE-E on TLR4/MyD88/NF-κB and NLRP3/ASC/Caspase-1 Signaling Pathways in Renal
3.9. CPE-E Alter Gut Microbiota
3.10. Effect of CPE-E on the Concentrations of SCFA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kuwabara, M. Hyperuricemia, Cardiovascular Disease, and Hypertension. Pulse 2016, 3, 242–252. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.; Keenan, R.T. Gout, Hyperuricemia, and the Risk of Cardiovascular Disease: Cause and Effect? Curr. Rheumatol. Rep. 2010, 12, 118–124. [Google Scholar] [CrossRef]
- Song, P.; Wang, H.; Xia, W.; Chang, X.; Wang, M.; An, L. Prevalence and correlates of hyperuricemia in the middle-aged and older adults in China. Sci. Rep. 2018, 8, 4314. [Google Scholar] [CrossRef]
- Billiet, L.; Doaty, S.; Katz, J.D.; Velasquez, M.T. Review of Hyperuricemia as New Marker for Metabolic Syndrome. ISRN Rheumatol. 2014, 2014, 852954. [Google Scholar] [CrossRef] [Green Version]
- Jhang, J.J.; Lin, J.H.; Yen, G.C. Beneficial properties of phytochemicals on NLRP3 inflammasome-mediated gout and compli-cation. J. Agric. Food Chem. 2018, 66, 765–772. [Google Scholar] [CrossRef]
- Tan, J.; Wan, L.; Chen, X.; Li, X.; Hao, X.; Li, J.; Ding, H. Conjugated Linoleic Acid Ameliorates High Fructose-Induced Hyperuricemia and Renal Inflammation in Rats via NLRP3 Inflammasome and TLR4 Signaling Pathway. Mol. Nutr. Food Res. 2019, 63, e1801402. [Google Scholar] [CrossRef]
- Bouter, K.E.; van Raalte, D.H.; Groen, A.K.; Nieuwdorp, M. Role of the Gut Microbiome in the Pathogenesis of Obesity and Obesity-Related Metabolic Dysfunction. Gastroenterology 2017, 152, 1671–1678. [Google Scholar] [CrossRef]
- Zhao, H.; Cheng, N.; Zhou, W.; Chen, S.; Wang, Q.; Gao, H.; Xue, X.; Wu, L.; Cao, W. Honey Polyphenols Ameliorate DSS-Induced Ulcerative Colitis via Modulating Gut Microbiota in Rats. Mol. Nutr. Food Res. 2019, 63, e1900638. [Google Scholar] [CrossRef]
- Wang, J.; Chen, Y.; Zhong, H.; Chen, F.; Regenstein, J.; Hu, X.; Cai, L.; Feng, F. The gut microbiota as a target to control hyperuricemia pathogenesis: Potential mechanisms and therapeutic strategies. Crit. Rev. Food Sci. Nutr. 2021, 22, 1–11. [Google Scholar] [CrossRef]
- Liu, G.; Chen, X.; Lu, X.; Zhao, J.; Li, X. Sunflower head enzymatic hydrolysate relives hyperuricemia by inhibiting crucial proteins (xanthine oxidase, adenosine deaminase, uric acid transporter1) and restoring gut microbiota in mice. J. Funct. Foods 2020, 72, 104055. [Google Scholar] [CrossRef]
- Crane, J.K. Role of host xanthine oxidase in infection due to enteropathogenic and Shiga-toxigenic Escherichia coli. Gut Microbes 2013, 4, 388–391. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Yu, Y.; Li, H.; Ding, X.; Li, X.; Jing, X.; Chen, J.; Liu, G.; Lin, Y.; Jiang, C.; et al. Inulin supplementation ameliorates hyperuricemia and modulates gut microbiota in Uox-knockout mice. Eur. J. Nutr. 2021, 60, 2217–2230. [Google Scholar] [CrossRef]
- Pascart, T.; Lioté, F. Gout: State of the art after a decade of developments. Rheumatology 2018, 58, 27–44. [Google Scholar] [CrossRef]
- Pascart, T.; Richette, P. Investigational drugs for hyperuricemia, an update on recent developments. Expert Opin. Investig. Drugs 2018, 27, 437–444. [Google Scholar] [CrossRef]
- Hande, K.; Noone, R.; Stone, W. Severe Allopurinol Toxicity. Description and Guidelines for Prevention in Patients with Renal Insufficiency. J. Urol. 1984, 131, 1258–1259. [Google Scholar] [CrossRef]
- Lee, M.H.H.; Graham, G.G.; Williams, K.M.; Day, R.O. A benefit-risk assessment of benzbromarone in the treatment of gout. Drug Saf. 2008, 31, 643–665. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Qi, S.; Xue, X.; Al Naggar, Y.; Wu, L.; Wang, K. Understanding the Gastrointestinal Protective Effects of Polyphenols using Foodomics-Based Approaches. Front. Immunol. 2021, 12, 671150. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, L.; Lin, D.; Ma, Z.; Deng, X. Lemon fruits lower the blood uric acid levels in humans and mice. Sci. Hortic. 2017, 220, 4–10. [Google Scholar] [CrossRef]
- Zhao, M.; Zhu, D.; Sun-Waterhouse, D.; Su, G.; Lin, L.; Wang, X.; Dong, Y. In Vitro and In Vivo Studies on Adlay-Derived Seed Extracts: Phenolic Profiles, Antioxidant Activities, Serum Uric Acid Suppression, and Xanthine Oxidase Inhibitory Effects. J. Agric. Food Chem. 2014, 62, 7771–7778. [Google Scholar] [CrossRef]
- Soliman, M.M.; Nassan, M.A.; Aldhahrani, A.; Althobaiti, F.; Mohamed, W.A. Molecular and Histopathological Study on the Ameliorative Impacts of Petroselinum Crispum and Apium Graveolens against Experimental Hyperuricemia. Sci. Rep. 2020, 10, 9512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-C.; Su, G.-H.; Luo, C.-L.; Pang, Y.-L.; Wang, L.; Li, X.; Wen, J.-H.; Zhang, J.-L. Effects of anthocyanins from purple sweet potato (Ipomoea batatas L. cultivar Eshu No. 8) on the serum uric acid level and xanthine oxidase activity in hyperuricemic mice. Food Funct. 2015, 6, 3045–3055. [Google Scholar] [CrossRef]
- Jin, T.Y.; Saravanakumar, K.; Wang, M.H. In vitro and in vivo antioxidant properties of water and methanol extracts of linden bee pollen. Biocatal. Agric. Biotechnol. 2018, 13, 186–189. [Google Scholar] [CrossRef]
- Li, Q.; Wang, K.; Marcucci, M.C.; Sawaya, A.C.H.F.; Hu, L.; Xue, X.-F.; Wu, L.-M.; Hu, F.-L. Nutrient-rich bee pollen: A treasure trove of active natural metabolites. J. Funct. Foods 2018, 49, 472–484. [Google Scholar] [CrossRef]
- Nicolson, S.W.; Neves, S.D.S.D.; Human, H.; Pirk, C. Digestibility and nutritional value of fresh and stored pollen for honey bees (Apis mellifera scutellata). J. Insect Physiol. 2018, 107, 302–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freire, K.R.L.; Lins, A.C.S.; Dórea, M.C.; Santos, F.A.R.; Camara, C.A.; Silva, T.M.S. Palynological Origin, Phenolic Content, and Antioxidant Properties of Honeybee-Collected Pollen from Bahia, Brazil. Molecules 2012, 17, 1652–1664. [Google Scholar] [CrossRef] [Green Version]
- Kao, Y.-T.; Lu, M.-J.; Chen, C. Preliminary analyses of phenolic compounds and antioxidant activities in tea pollen extracts. J. Food Drug Anal. 2012, 19, 3. [Google Scholar] [CrossRef]
- Peng, G.; Zhao, H.; Liu, Q.; Zhang, Y.; Cheng, N.; Cao, W. Antioxidant and hepatoprotective effects of Camellia japonica bee pollen on acute alcohol-induced liver damage in mice. Food Sci. 2018, 39, 127–133. [Google Scholar]
- Medeiros, K.; Figueiredo, C.; Figueredo, T.B.; Freire, K.; Santos, F.; Alcantara-Neves, N.M.; Silva, T.; Piuvezam, M.R. Anti-allergic effect of bee pollen phenolic extract and myricetin in ovalbumin-sensitized mice. J. Ethnopharmacol. 2008, 119, 41–46. [Google Scholar] [CrossRef]
- Zhao, H.; Cheng, N.; He, L.; Peng, G.; Xue, X.; Wu, L.; Cao, W. Antioxidant and hepatoprotective effects of A. cerana honey against acute alcohol-induced liver damage in mice. Food Res. Int. 2017, 101, 35–44. [Google Scholar] [CrossRef]
- Zhou, J.; Li, P.; Cheng, N.; Gao, H.; Wang, B.; Wei, Y.; Cao, W. Protective effects of buckwheat honey on DNA damage induced by hydroxyl radicals. Food Chem. Toxicol. 2012, 50, 2766–2773. [Google Scholar] [CrossRef] [PubMed]
- Blasa, M.; Candiracci, M.; Accorsi, A.; Piacentini, M.P.; Albertini, M.C.; Piatti, E. Raw Millefiori honey is packed full of anti-oxidants. Food Chem. 2006, 97, 217–222. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Byrne, D.H. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Singh, N.; Rajini, P. Free radical scavenging activity of an aqueous extract of potato peel. Food Chem. 2004, 85, 611–616. [Google Scholar] [CrossRef]
- Mărghitaş, L.A.; Stanciu, O.G.; Dezmirean, D.S.; Bobis, O.; Popescu, O.; Bogdanov, S.; Campos, M.G. In vitro antioxidant capacity of honeybee-collected pollen of selected floral origin harvested from Romania. Food Chem. 2009, 115, 878–883. [Google Scholar] [CrossRef]
- Yeung, S.Y.; Lan, W.H.; Huang, C.S.; Lin, C.P.; Chan, C.P.; Chang, M.C.; Jeng, J.H. Scavenging property of three cresol isomers against H2O2, hypochlorite, superoxide and hydroxyl radicals. Food Chem. Toxicol. 2002, 40, 1403–1413. [Google Scholar] [CrossRef]
- Qian, X.; Wang, X.; Luo, J.; Liu, Y.; Pang, J.; Zhang, H.; Xu, Z.; Xie, J.; Jiang, X.; Ling, W. Hypouricemic and nephroprotective roles of anthocyanins in hyperuricemic mice. Food Funct. 2019, 10, 867–878. [Google Scholar] [CrossRef]
- Bergsdorf, T.; Thüroff, S.; Chaussy, C. The Isolated Perfused Kidney: An in Vitro Test System for Evaluation of Renal Tissue Damage Induced by High-Energy Shockwaves Sources. J. Endourol. 2005, 19, 883–888. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Q.; Zhou, M.; Zhu, H.; Lu, G.; Zheng, D.; Li, H.; Hao, K. (E)-2-(4-bromophenyl)-1-(2, 4-dihydroxyphenyl)ethanone oxime is a potential therapeutic agent for treatment of hyperuricemia through its dual inhibitory effects on XOD and URAT1. Biomed. Pharm. 2017, 86, 88–94. [Google Scholar] [CrossRef]
- Eleftheriadis, T.; Golphinopoulos, S.; Pissas, G.; Stefanidis, I. Asymptomatic hyperuricemia and chronic kidney disease: Nar-rative review of a treatment controversial. J. Adv. Res. 2017, 8, 555–560. [Google Scholar] [CrossRef]
- Chen, L.; Li, M.; Wu, J.-L.; Li, J.-X.; Ma, Z.-C. Effect of lemon water soluble extract on hyperuricemia in a mouse model. Food Funct. 2019, 10, 6000–6008. [Google Scholar] [CrossRef]
- Jhang, J.-J.; Ong, J.-W.; Yen, G.-C.; Lu, C.-C.; Hsu, C.-L.; Lin, J.-H.; Liao, J.-W. Hypouricemic effects of Mesona procumbens Hemsl. through modulating xanthine oxidase activity in vitro and in vivo. Food Funct. 2016, 7, 4239–4246. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Liu, X.; Zhao, M. Screening of xanthine oxidase inhibitor from selected edible plants and hypouricemic effect of Rhi-zoma Alpiniae Officinarum extract on hyperuricemic rats. J. Funct. Foods. 2018, 50, 26–36. [Google Scholar] [CrossRef]
- Wang, R.; Ma, C.H.; Zhou, F.; Kong, L.D. Siwu decoction attenuates oxonate-induced hyperuricemia and kidney inflammation in mice. Chin. J. Nat. Med. 2016, 14, 499–507. [Google Scholar] [CrossRef]
- Dalbeth, N.; Merriman, T.R.; Stamp, L.K. Gout. Lancet 2016, 388, 2039–2052. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, X.; Li, C.; Yuan, X.; Han, L.; Li, Z.; Tan, X.; Song, J.; Wang, G.; Jia, X.; et al. Research on the pharmacody-namics and mechanism of Fraxini Cortex on hyperuricemia based on the regulation of URAT1 and GLUT9. Biomed. Pharm. 2018, 106, 434–442. [Google Scholar] [CrossRef]
- Zhao, R.; Chen, D.; Wu, H. Puerh ripened tea resists to hyperuricemia through xanthine oxidase and renal urate transporters in hyperuricemic mice. J. Funct. Foods 2017, 29, 201–207. [Google Scholar] [CrossRef]
- Chen, L.; Lan, Z.; Lin, Q.; Mi, X.; He, Y.; Wei, L.; Lin, Y.; Zhang, Y.; Deng, X. Polydatin ameliorates renal injury by attenuating oxidative stress-related inflammatory responses in fructose-induced urate nephropathic mice. Food Chem. Toxicol. 2013, 52, 28–35. [Google Scholar] [CrossRef]
- Sánchez-Lozada, L.G.; Soto, V.; Tapia, E.; Avila-Casado, C.; Sautin, Y.Y.; Nakagawa, T.; Franco, M.; Rodríguez-Iturbe, B.; Johnson, R.J. Role of oxidative stress in the renal abnormalities induced by experimental hyperuricemia. Am. J. Physiol. Physiol. 2008, 295, F1134–F1141. [Google Scholar] [CrossRef] [Green Version]
- Ulusoy, E.; Kolayli, S. Phenolic Composition and Antioxidant Properties of Anzer Bee Pollen. J. Food Biochem. 2013, 38, 73–82. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Russa, D.L.; Pellegrino, D.; Montesanto, A.; Gigliotti, P.; Perri, A.; Russa, A.L.; Bonofiglio, R. Oxidative balance and inflammation in hemodialysis patients: Biomarkers of cardiovascular risk? Oxid. Med. Cell Longev. 2019, 2019, 8567275. [Google Scholar] [CrossRef]
- Strazzullo, P.; Puig, J.G. Uric acid and oxidative stress: Relative impact on cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 409–414. [Google Scholar] [CrossRef]
- Zhao, H.; Cheng, N.; Wang, Q.; Zhou, W.; Liu, C.; Liu, X.; Chen, S.; Fan, D.; Cao, W. Effects of honey-extracted polyphenols on serum antioxidant capacity and metabolic phenotype in rats. Food Funct. 2019, 10, 2347–2358. [Google Scholar] [CrossRef]
- Yu, M.A.; Sánchez-Lozada, L.G.; Johnson, R.J.; Kang, D.H. Oxidative stress with an activation of the renin-angiotensin system in human vascular endothelial cells as a novel mechanism of uric acid-induced endothelial dysfunction. J. Hypertens 2010, 28, 1234. [Google Scholar] [CrossRef]
- Han, J.; Wang, X.; Tang, S.; Lu, C.; Wan, H.; Zhou, J.; Li, Y.; Ming, T.; Wang, Z.; Su, X. Protective effects of tuna meat oligopeptides (TMOP) supplementation on hyperuricemia and associated renal inflammation mediated by gut microbiota. FASEB J. 2020, 34, 5061–5076. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Li, C.; Duan, S.; Yuan, X.; Liang, J.; Hou, S. Curcumin attenuates potassium oxonate-induced hyperuricemia and kidney inflammation in mice. Biomed. Pharmacother. 2019, 118, 109195. [Google Scholar] [CrossRef]
- Wan, H.; Han, J.; Tang, S.; Bao, W.; Lu, C.; Zhou, J.; Ming, T.; Li, Y.; Su, X. Comparisons of protective effects between two sea cucumber hydrolysates against diet induced hyperuricemia and renal inflammation in mice. Food Funct. 2020, 11, 1074–1086. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Lu, X.; Lu, C.; Shen, N.; Jiang, Y.; Chen, M.; Wu, H. Soluble uric acid increases PDZK1 and ABCG2 expression in human intestinal cell lines via the TLR4-NLRP3 inflammasome and PI3K/Akt signaling pathway. Arthritis Res. Ther. 2018, 20, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finco, D.R.; Duncan, J.R. Evaluation of blood urea nitrogen and serum creatinine concentrations as indicators of renal dysfunction: A study of 111 cases and a review of related literature. J. Am. Vet. Med. Assoc. 1976, 168, 593–601. [Google Scholar] [PubMed]
- Lai, S.W.; Kuo, Y.H.; Liao, K.F. Allopurinol and the risk of prostate cancer. Postgrad. Med. J. 2019, 96, 102. [Google Scholar] [CrossRef]
- Zhang, R.; Zhan, S.; Li, S.; Zhu, Z.; He, J.; Lorenzo, J.M.; Barba, F.J. Anti-hyperuricemia and nephroprotective effects of extracts from Chaenomeles sinensis (Thouin) Koehne in hyperuricemic mice. Food Funct. 2018, 9, 5778–5790. [Google Scholar] [CrossRef]
- Anna, R.S.; Jerzy, S.; Anna, K.G.; Michal, G.; Agata, K.D.; Robert, K.; Aleksandra, M.; Ewa, B. Polyphenols from bee pollen: Structure, absorption, metabolism and biological activity. Molecules 2015, 20, 21732–21749. [Google Scholar]
- Wang, K.; Jin, X.; Li, Q.; Sawaya, A.C.H.F.; Le Leu, R.K.; Conlon, M.A.; Wu, L.; Hu, F. Propolis from Different Geographic Origins Decreases Intestinal Inflammation and Bacteroides spp. Populations in a Model of DSS-Induced Colitis. Mol. Nutr. Food Res. 2018, 62, 1800080. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomes, A.C.; Bueno, A.A.; De Souza, R.G.M.; Mota, J.F. Gut microbiota, probiotics and diabetes. Nutr. J. 2014, 13, 60. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Lv, Q.; Ren, H.; Gao, L.; Zhao, P.; Yang, X.; Yang, G.; Xu, D.; Wang, G.; Yang, W.; et al. The altered gut microbiota of high-purine-induced hyperuricemia rats and its correlation with hyperuricemia. PeerJ 2020, 8, e8664. [Google Scholar] [CrossRef] [PubMed]
- Garcia, F.; Gonzaga, G.; Muñoz-Jiménez, I.; Blas-Marron, M.G.; Silverio, O.; Tapia, E.; Soto, V.; Ranganathan, N.; Ranganathan, P.; Vyas, U.; et al. Probiotic supplements prevented oxonic acid-induced hyperuricemia and renal damage. PLoS ONE 2018, 13, e0202901. [Google Scholar] [CrossRef] [Green Version]
- Xing, S.-C.; Meng, D.-M.; Chen, Y.; Jiang, G.; Liu, X.-S.; Li, N.; Yan, Y.-Y.; Li, C.-G. Study on the Diversity of Bacteroides and Clostridium in Patients with Primary Gout. Cell Biophys. 2014, 71, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Nieuwdorp, M.; Gilijamse, P.W.; Pai, N.; Kaplan, L.M. Role of the Microbiome in Energy Regulation and Metabolism. Gastroenterology 2014, 146, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Marzocco, S.; Fazeli, G.; Di Micco, L.; Autore, G.; Adesso, S.; Piaz, F.D.; Heidland, A.; Di Iorio, B. Supplementation of Short-Chain Fatty Acid, Sodium Propionate, in Patients on Maintenance—“Beneficial Effects on Inflammatory Parameters and Gut-Derived Uremic Toxins”—A Pilot Study (PLAN Study). J. Clin. Med. 2018, 7, 315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
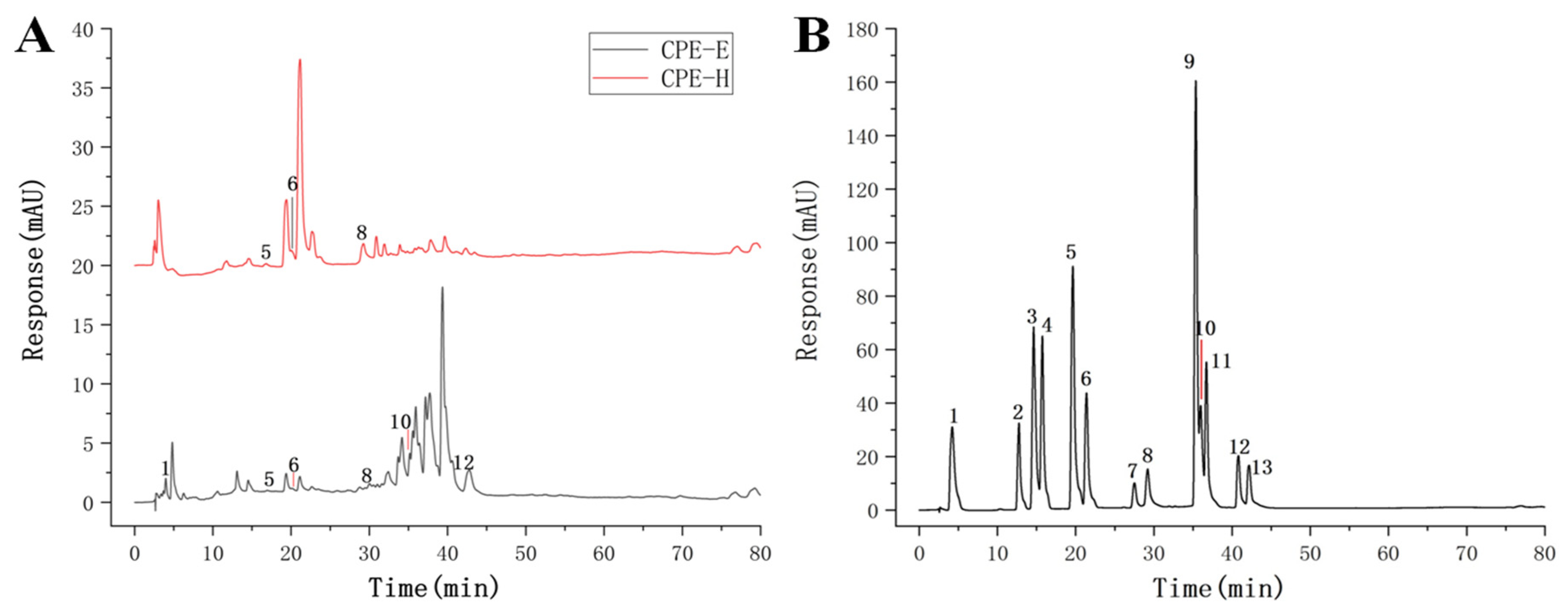




| Peak No. | Retention Time (min) | Phenolic Compound | Content (mg/g) | |
|---|---|---|---|---|
| CPE-E | CPE-H | |||
| 1 | 4.21 | gallic acid | 5.03 ± 0.21 | nd |
| 5 | 19.64 | p-hydroxybenzoic acid | 1.28 ± 0.13 | 1.54 ± 0.09 |
| 6 | 21.39 | ferulic acid | 1.90 ± 0.15 | 2.40 ± 0.13 |
| 8 | 29.20 | ellagic acid | 2.26 ± 0.12 | 4.35 ± 0.21 |
| 10 | 36.98 | quercetin | 6.85 ± 0.23 | nd |
| 12 | 40.81 | kaempferol | 7.29 ± 0.19 | nd |
| Samples | TPC (mg GAE/g) | TFC (mg RE/g) | FRAP (mg Trolox/mg) | DPPH (IC50 mg/mL) | Chelating Activity (mg Na2EDTA/g) |
|---|---|---|---|---|---|
| CPE-E | 136.63 ± 4.32 a | 67.49 ± 4.64 a | 0.49 ± 0.01 a | 0.20 ± 0.02 a | 29.10 ± 8.49 a |
| CPE-H | 31.74 ± 0.21 b | 15.26 ± 0.38 b | 0.07 ± 0.01 b | 5.03 ± 0.03 b | 12.93 ± 1.03 b |
| Groups | Chao1 | Shannon | Simpson | Observed Species |
|---|---|---|---|---|
| Group I | 1426.14 ± 165.01 a | 6.89 ± 0.54 a | 0.97 ± 0.015 a | 1429.87 ± 164.75 a |
| Group II | 1333.45 ± 175.10 a | 6.08 ± 0.39 b | 0.90 ± 0.039 b | 1322.59 ± 189.73 a |
| Group III | 1491.26 ± 303.42 a | 6.79 ± 0.59 a | 0.95 ± 0.018 a | 1402.30 ± 220.19 a |
| Group IV | 1508.36 ± 234.15 a | 6.95 ± 0.43 a | 0.98 ± 0.013 a | 1494.26 ± 303.43 a |
| Group V | 1463.56 ± 275.42 a | 6.91 ± 0.78 a | 0.96 ± 0.038 a | 1470.93 ± 245.26 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Cao, X.; Zhao, H.; Yang, E.; Wang, Y.; Cheng, N.; Cao, W. Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice. Nutrients 2021, 13, 2665. https://doi.org/10.3390/nu13082665
Xu Y, Cao X, Zhao H, Yang E, Wang Y, Cheng N, Cao W. Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice. Nutrients. 2021; 13(8):2665. https://doi.org/10.3390/nu13082665
Chicago/Turabian StyleXu, Yuanyuan, Xirong Cao, Haoan Zhao, Erlin Yang, Yue Wang, Ni Cheng, and Wei Cao. 2021. "Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice" Nutrients 13, no. 8: 2665. https://doi.org/10.3390/nu13082665
APA StyleXu, Y., Cao, X., Zhao, H., Yang, E., Wang, Y., Cheng, N., & Cao, W. (2021). Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice. Nutrients, 13(8), 2665. https://doi.org/10.3390/nu13082665






