Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Variables
2.2. SARS-CoV-2 Testing
2.3. Statistical Analysis
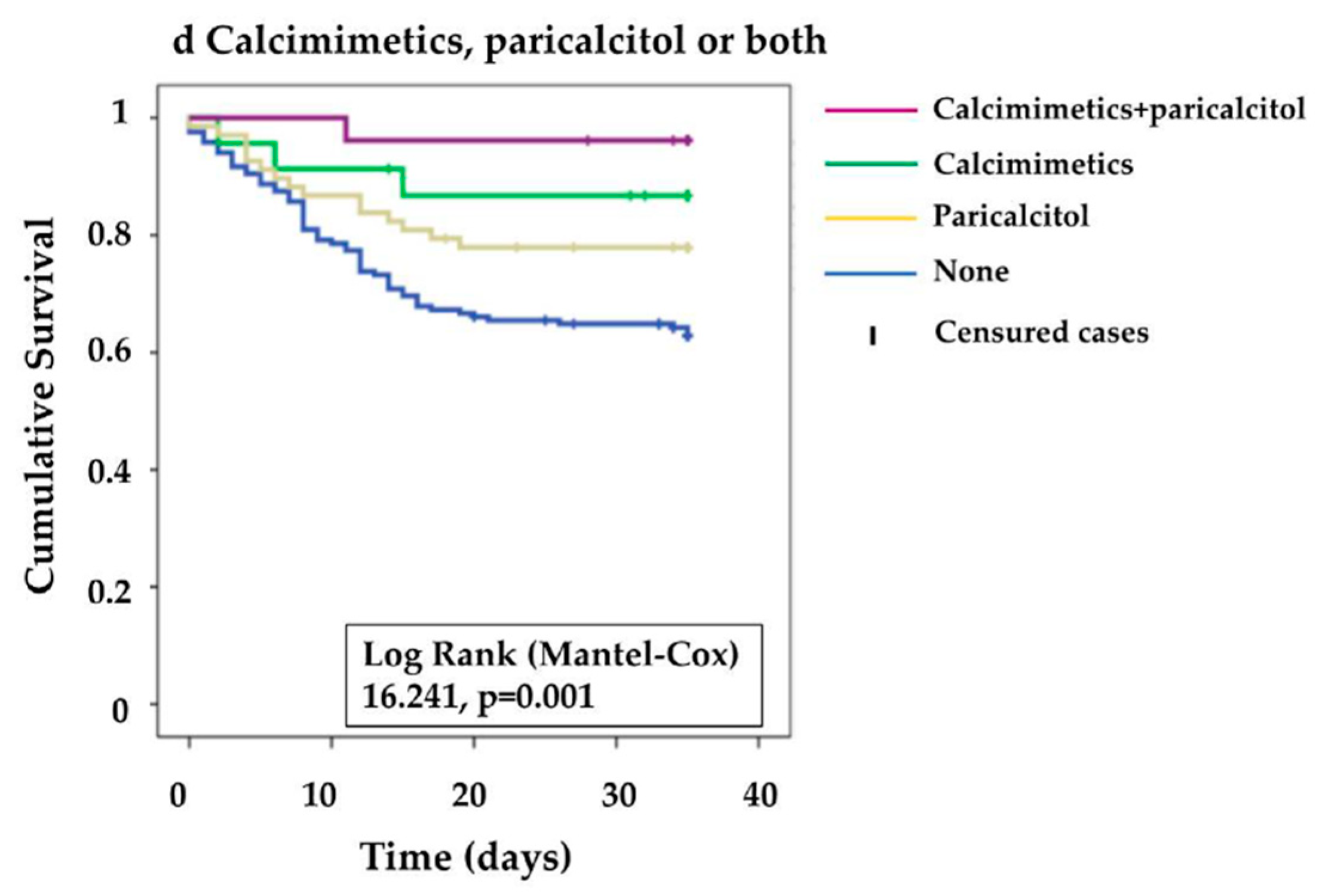
3. Results
Serum VD Levels and Outcomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Wu, J.T.; Leung, K.; Leung, G.M. Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 2020, 395, 689–697. [Google Scholar] [CrossRef]
- Cucinotta, D.; Vanelli, M. WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Peiris, J.S.; Guan, Y.; Yuen, K.Y. Severe acute respiratory syndrome. Nat. Med. 2004, 10 (Suppl. 12), S88–S97. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- McGonagle, D.; Sharif, K.; O’Regan, A.; Bridgewood, C. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun. Rev. 2020, 19, 102537. [Google Scholar] [CrossRef]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar] [CrossRef]
- Arenas, M.D.; Crespo, M.; Pérez-Sáez, M.J.; Collado, S.; Redondo-Pachón, D.; Llinàs-Mallol, L.; Montero, M.M.; Villar-García, J.; Arias-Cabrales, C.; Barbosa, F.; et al. Clinical Profiles in Renal Patients with COVID-19. J. Clin. Med. 2020, 9, 2665. [Google Scholar] [CrossRef]
- Goicoechea, M.; Cámara, L.A.S.; Macías, N.; de Morales, A.M.; Rojas, Á.G.; Bascuñana, A.; Arroyo, D.; Vega, A.; Abad, S.; Verde, E.; et al. COVID-19: Clinical course and outcomes of 36 hemodialysis patients in Spain. Kidney Int. 2020, 98, 27–34. [Google Scholar] [CrossRef]
- Rincón, A.; Moreso, F.; López-Herradón, A.; Fernández-Robres, M.A.; Cidraque, I.; Nin, J.; Méndez, O.; López, M.; Pájaro, C.; Satorra, À.; et al. The keys to control a COVID-19 outbreak in a haemodialysis unit. Clin. Kidney J. 2020, 13, 542–549. [Google Scholar] [CrossRef]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Investig. 2021, 44, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Dancer, R.C.A.; Parekh, D.; Lax, S.; D’Souza, V.; Zheng, S.; Bassford, C.R.; Park, D.; Bartis, D.G.; Mahida, R.; Turner, A.M.; et al. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS). Thorax 2015, 70, 617–624. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Stefanidis, C.; Wang, Z.; Kermani, N.Z.; Dimitrov, V.; White, J.H.; McDonough, J.; Janssens, W.; Pfeffer, P.; Griffiths, C.J.; et al. Vitamin D Metabolism Is Dysregulated in Asthma and Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2020, 202, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; Keppel, M.H.; Grübler, M.R.; März, W.; Pandis, M. Vitamin D testing and treatment: A narrative review of current evidence. Endocr. Connect. 2019, 8, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 1–59. [Google Scholar] [CrossRef]
- Torregrosa, J.V.; Bover, J.; Andía, J.C.; Lorenzo, V.; de Francisco, A.L.M.; Martínez, I.; Portillo, M.R.; Arenas, L.; Parra, E.G.; Caravaca, F.; et al. Recomendaciones de la Sociedad Española de Nefrología para el manejo de las alteraciones del metabolismo óseo-mineral en los pacientes con enfermedad renal crónica (S.E.N.-MM). Nefrologia 2011, 31 (Suppl. 1), 3–32. [Google Scholar] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Jean, G.; Souberbielle, J.C.; Chazot, C. Vitamin D in Chronic Kidney Disease and Dialysis Patients. Nutrients 2017, 9, 328. [Google Scholar] [CrossRef]
- Sánchez-Álvarez, J.E.; Pérez Fontán, M.; Jiménez Martín, C.; Pelícano, M.B.; Reina, C.J.C.; Prieto, Á.M.S.; Melilli, E.; Barrios, M.C.; Heras, M.M.; del Pino y Pino, M.D. SARS-CoV-2 infection in patients on renal replacement therapy. Report of the COVID-19 Registry of the Spanish Society of Nephrology (SEN). Nefrologia 2020, 40, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Morales, A.J.; Cardona-Ospina, J.A.; Gutiérrez-Ocampo, E.; Villamizar-Peña, R.; Holguin-Rivera, Y.; Antezana, J.P.E.; Alvarado-Arnez, L.E.; Bonilla-Aldana, D.K.; Franco-Paredes, C.; Henao-Martínez, A.; et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med. Infect. Dis. 2020, 34, 101623. [Google Scholar] [CrossRef] [PubMed]
- Chertow, G.M.; Plone, M.; Dillon, M.A.; Burke, S.K.; Slatopolsky, E. Hyperparathyroidism and dialysis vintage. Clin. Nephrol. 2000, 54, 295–300. [Google Scholar]
- Alfieri, C.; Regalia, A.; Zanoni, F.; Vettoretti, S.; Cozzolino, M.; Messa, P. The Importance of Adherence in the Treatment of Secondary Hyperparathyroidism. Blood Purif. 2019, 47, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Darssan, D.; Pascoe, E.M.; Johnson, D.W.; Pi, H.; Dong, J. Vitamin D status and mortality risk among patients on dialysis: A systematic review and meta-analysis of observational studies. Nephrol. Dial. Transplant. 2018, 33, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef]
- Ali, N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J. Infect. Public Health 2020, 13, 1373–1380. [Google Scholar] [CrossRef]
- Hastie, C.E.; Mackay, D.F.; Ho, F.; Celis-Morales, C.A.; Katikireddi, S.V.; Niedzwiedz, C.L.; Jani, B.D.; Welsh, P.; Mair, F.S.; Gray, S.R.; et al. Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab. Syndr. 2020, 14, 561–565. [Google Scholar] [CrossRef]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Cohen, A.G.; Green, I.; Frenkel-Morgenstern, M. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.M.; Solway, J. Association of Vitamin D Levels, Race/Ethnicity, and Clinical Characteristics With COVID-19 Test Results. JAMA Netw. Open 2021, 4, e214117. [Google Scholar] [CrossRef]
- Guerrero, F.; De Oca, A.M.; Aguilera-Tejero, E.; Zafra, R.; Rodriguez, M.; López, I. The effect of vitamin D derivatives on vascular calcification associated with inflammation. Nephrol. Dial. Transplant. 2012, 27, 2206–2212. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Riera, M.; Anguiano, L.; Clotet, S.; Ho, H.R.; Rebull, M.; Pascual, J.; Soler, M.J. Paricalcitol modulates ACE2 shedding and renal ADAM17 in NOD mice beyond proteinuria. Am. J. Physiol. Renal Physiol. 2016, 310, F534–F546. [Google Scholar] [CrossRef]
- Leifheit-Nestler, M.; Grabner, A.; Hermann, L.; Richter, B.; Schmitz, K.; Fischer, D.-C.; Yanucil, C.; Faul, C.; Haffner, D. Vitamin D treatment attenuates cardiac FGF23/FGFR4 signaling and hypertrophy in uremic rats. Nephrol. Dial. Transplant. 2017, 32, 1493–1503. [Google Scholar] [CrossRef]
- Panizo, S.; Carrillo-López, N.; Naves-Díaz, M.; Solache-Berrocal, G.; Martínez-Arias, L.; Rodrigues-Díez, R.R.; Fernández-Vázquez, A.; Martínez-Salgado, C.; Ruiz-Ortega, M.; Dusso, A.; et al. Regulation of miR-29b and miR-30c by vitamin D receptor activators contributes to attenuate uraemia-induced cardiac fibrosis. Nephrol. Dial. Transplant. 2017, 32, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.E.; Costa, L.M.E.; Barrios, J.M.V.; Díaz, J.F.A.; Miranda, J.L.; Bouillon, R.; Gomez, J.M.Q. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Gassen, N.C.; Niemeyer, D.; Muth, D.; Corman, V.M.; Martinelli, S.; Gassen, A.; Hafner, K.; Papies, J.; Mösbauer, K.; Zellner, A.; et al. SKP2 attenuates autophagy through Beclin1-ubiquitination and its inhibition reduces MERS-Coronavirus infection. Nat. Commun. 2019, 10, 5770. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yang, J.; Chen, J.; Luo, Q.; Zhang, Q.; Zhang, H. Vitamin D alleviates lipopolysaccharide-induced acute lung injury via regulation of the renin-angiotensin system. Mol. Med. Rep. 2017, 16, 7432–7438. [Google Scholar] [CrossRef]
- Cui, C.; Xu, P.; Li, G.; Qiao, Y.; Han, W.; Geng, C.; Liao, D.; Yang, M.; Chen, D.; Jiang, P. Vitamin D receptor activation regulates microglia polarization and oxidative stress in spontaneously hypertensive rats and angiotensin II-exposed microglial cells: Role of renin-angiotensin system. Redox Biol. 2019, 26, 101295. [Google Scholar] [CrossRef] [PubMed]
- Dijkman, R.; Jebbink, M.F.; Deijs, M.; Milewska, A.; Pyrc, K.; Buelow, E.; Van Der Bijl, A.; Van Der Hoek, L. Replication-dependent downregulation of cellular angiotensin-converting enzyme 2 protein expression by human coronavirus NL63. J. Gen. Virol. 2012, 93 Pt 9, 1924–1929. [Google Scholar] [CrossRef]
- Sodhi, C.P.; Wohlford-Lenane, C.; Yamaguchi, Y.; Prindle, T.; Fulton, W.B.; Wang, S.; McCray, P.B., Jr.; Chappell, M.; Hackam, D.J.; Jia, H.; et al. Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg9 bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration. Am. J. Physiol. Lung Cell. Mol. Physiol. 2018, 314, L17–L31. [Google Scholar] [CrossRef]
- Patel, V.; Clarke, N.; Wang, Z.; Fan, D.; Parajuli, N.; Basu, R.; Putko, B.; Kassiri, Z.; Turner, A.J.; Oudit, G.Y. Angiotensin II induced proteolytic cleavage of myocardial ACE2 is mediated by TACE/ADAM-17: A positive feedback mechanism in the RAS. J. Mol. Cell. Cardiol. 2014, 66, 167–176. [Google Scholar] [CrossRef]
- Wolf, M.; Shah, A.; Gutierrez, O.; Ankers, E.; Monroy, M.; Tamez, H.; Steele, D.; Chang, Y.; Camargo, C.; Tonelli, M.; et al. Vitamin D levels and early mortality among incident hemodialysis patients. Kidney Int. 2007, 72, 1004–1013. [Google Scholar] [CrossRef]
- Teng, M.; Wolf, M.; Lowrie, E.; Ofsthun, N.; Lazarus, J.M.; Thadhani, R. Survival of Patients Undergoing Hemodialysis with Paricalcitol or Calcitriol Therapy. N. Engl. J. Med. 2003, 349, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Arcidiacono, M.V.; Yang, J.; Fernandez, E.; Dusso, A. The induction of C/EBPβ contributes to vitamin D inhibition of ADAM17 expression and parathyroid hyperplasia in kidney disease. Nephrol. Dial. Transplant. 2014, 30, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Palau, V.; Riera, M.; Soler, M.J. ADAM17 inhibition may exert a protective effect on COVID-19. Nephrol. Dial. Transplant. 2020, 35, 1071–1072. [Google Scholar] [CrossRef] [PubMed]
- Ostroverkhova, D.S.; Hu, J.; Tarasov, V.V.; Melnikova, T.I.; Porozov, Y.B.; Mutig, K. Calcium-Sensing Receptor and Regulation of WNK Kinases in the Kidney. Cells 2020, 9, 1644. [Google Scholar] [CrossRef]
- Atchison, D.K.; Ortiz-Capisano, M.C.; Beierwaltes, W.H. Acute activation of the calcium-sensing receptor inhibits plasma renin activity in vivo. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R1020–R1026. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moe, S.M.; Chertow, G.M.; Parfrey, P.S.; Kubo, Y.; Block, G.A.; Correa-Rotter, R.; Drüeke, T.B.; Herzog, C.A.; London, G.M.; Mahaffey, K.W.; et al. Cinacalcet, Fibroblast Growth Factor-23, and Cardiovascular Disease in Hemodialysis: The Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) Trial. Circulation 2015, 132, 27–39. [Google Scholar] [CrossRef]
- Isakova, T.; Wahl, P.; Vargas, G.S.; Gutiérrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; Chen, J.; et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Li, L.; Yang, J.; King, G.; Xiao, Z.; Quarles, L.D. Counter-regulatory paracrine actions of FGF-23 and 1,25(OH)2D in macrophages. FEBS Lett. 2016, 590, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Rossaint, J.; Oehmichen, J.; Van Aken, H.; Reuter, S.; Pavenstädt, H.J.; Meersch, M.; Unruh, M.; Zarbock, A. FGF23 signaling impairs neutrophil recruitment and host defense during CKD. J. Clin. Investig. 2016, 126, 962–974. [Google Scholar] [CrossRef]

| Demography | ||
| Age (years, mean ± SD) | 72.4 ± 12.6 | |
| Body mass index (kg/m2, mean ± SD) | 25.6 ± 5.5 | |
| Time on Hemodialysis (years, median [IQR]) | 2.5 [1.2–5.2] | |
| Sex | ||
| Female | 84 (29.2) | |
| Race | ||
| Caucasian | 208 (72.2) | |
| Other | 11 (3.8) | |
| Unknown | 69 (23.9) | |
| Vascular access (N: 261) ** | ||
| Arteriovenous fistulae | 154 (59) | |
| Central venous catheter | 107 (41) | |
| Cause of renal failure | ||
| Diabetic Nephropathy | 85 (29.5) | |
| Vascular | 37 (12.8) | |
| Glomerular | 34 (11.8) | |
| Hereditary | 18 (6.2) | |
| Tubulointerstitial | 15 (5.2) | |
| Unknown | 78 (27.1) | |
| Other | 21 (7.2) | |
| Toxic habits (N: 117) ** | ||
| Current smoking | 102 (49) | |
| Alcohol abuse | 17 (9.6) | |
| Risk factors and comorbidities (N: 288) | ||
| Diabetes mellitus | 137 (47.6) | |
| Hypertension | 129 (44.8) | |
| Dyslipemia | 106 (36.8) | |
| Ischemic heart disease | 94 (32.6) | |
| Lung disease | 66 (22.9) | |
| Previous trasplantation | 20 (6.9) | |
| Tuberculosis | 5 (1.7) | |
| Previous treatment for secondary hyperparathyroidism (N: 288) | ||
| No treatment | 128 (44.4) | |
| Any treatment (Vitamin D and/or calcimetics) | 160 (55.5) | |
| Any calcimimetic | 49 (17.0) | |
| Cinacalcet | 33 (11.5) | |
| Etelcalcetide | 16 (5.6) | |
| No vitamin D treatments | 151 (52.4) | |
| Any vitamin D treatments | 137 (47.6) | |
| Calcifediol (25-OH-Vitamin D) | 33 (11.5) | |
| Calcitriol | 22 (7.6) | |
| Paricalcitol | 94 (32.6) | |
| Two kind of Vitamin D | 12(4.2) | |
| One kind of vitamin D | 125 (43.4) | |
| Calcimimetics or paricalcitol | 117 (40.6) | |
| Both calcimimetics and paricalcitol | 26 (9.0) | |
| Calcidiol dose (mcg/month, median, [IQR]) | 399 [399–798] | |
| Calcitriol dose (mcg/week median, [IQR]) | 1.75 [0.99–1.83] | |
| Paricalcitol dose (mcg/week median, [IQR]) | 4.10 [2.25–5.60] | |
| Other concomitant medication | ||
| ACEIs | 63 (21.9) | |
| ARBs | 32 (11.1) | |
| NSAIDs | 21 (7.3) | |
| Baseline laboratory data *** | ||
| Time from baseline Blood test and PCR positive (days, median [p25–75]) | 20.0 [12.5–25] | |
| Hemoglobin (gr/dL, mean (SD)) | 11.0 (1.3) | |
| White blood cells (×10 3/uL, median [p25–75]) | 6.4 (5–8.3) | |
| Lymphocytes (×10 3/uL, median [p25–75]) | 1.07 (0.76–1.4) | |
| Neutrophils (×10 3/uL, median [p25–75]) | 4.26 (3.2–5.5) | |
| Platelets (×10 3/uL, median [p25–75]) | 190 (146–227) | |
| Fibrinogen (UI/L, mean (SD)) | 433 (139) | |
| AST (UI/L. median [p25–75]) | 16 (12–20) | |
| Albumin (g/dL. mean (SD)) | 3.8 (0.3) | |
| C-reactive protein (mg/L, median [p25–75]) | 7.9 (3.1–19.4) | |
| Ferritin (mcg/mL. median [p25–75]) | 436 (265–586) | |
| Transferrin Saturation Index (%. median [p25–75]) | 26 (17.8–36) | |
| Calcium (mg/dL. mean (SD)) | 8.8 (0.7) | |
| Phosphate (mg/dL, mean (SD)) | 4.1 (1.1) | |
| PTH (pg/dL, mean (SD)) | 240 (133–398) | |
| Hemoglobin (gr/dL, mean (SD)) | 10.9 (1.6) | |
| White blood cells (×10 3/uL, median [p25–75]) | 5.5 (4.2–7.7) | |
| Lymphocytes (×10 3/uL, median [p25–75]) | 0.70 (0.4–1.0) | |
| Neutrophils (×10 3/uL, median [p25–75]) | 4.0 (2.8–5.6) | |
| Platelets (×10 3/uL, median [p25–75]) | 166 (120–204) | |
| D-dimer (mcg/L, median [p25–75]) | 982 (580–2000) | |
| Fibrinogen (UI/L, mean (SD)) | 593 (194) | |
| AST (UI/L, median [p25–75]) | 21 (16–28) | |
| LDH (UI/L, mean (SD)) | 353 (664) | |
| IL6 (ng/L, median [p25–75]) | 18 (8.9–59.9) | |
| Albumin (g/dL, mean (SD)) | 3.3 (0.5) | |
| C-reactive protein (mg/L, median [p25–75]) | 14.7 (4.3–61) | |
| Ferritin (mcg/mL, median [p25–75]) | 1000 (547–1566) | |
| Transferrin Saturation Index (%, median [p25–75]) | 20 (13.8–31) | |
| Calcium (mg/dL, mean (SD)) | 8.6 (0.7) | |
| Phosphate (mg/dL, mean (SD) | 4.6 (1.5) | |
| Hospital admission N, % | |
| Admission required (N: 285) | 228 (80) |
| ICU admission (N: 257) | 15 (5.8) |
| Mechanical ventilation (N: 254) | 10 (3.9) |
| Outcome | |
| Resolution and/or discharged (N: 285) | 169 (59.2) |
| Exitus (N: 285) | 81 (28.4) |
| Admission to discharge (time elapsed in days, median [p25–75]) | 16.0 [10.8–28.0] |
| Admission to exitus (time elapsed in days days, median [p25–75]) | 8.0 [ 4.7–14.2] |
| Approached COVID-19+ contact | |
| Nursing home or relatives (N: 285) | 52 (18.2) |
| Symptoms | |
| Asymptomatic (N: 276) | 35 (12.6) |
| Fever (N: 265) | 180 (67.9) |
| Cough (N: 265) | 139 (52.4) |
| Dyspnea (N: 265) | 85(32.07) |
| Other (sore throat, discomfort, muscle pain, fatigue) (N: 265) | 66 (24.9) |
| Diarrhea (N: 265) | 28 (10.5) |
| Ageusia (N: 93) | 4 (4.2) |
| Anosmia (N: 93) | 5 (5.2) |
| Lung disease (N: 265) | |
| Pneumonia | 194 (73.2) |
| Covid-19 treatment | |
| Hydroxychloroquine (N: 238) | 176 (73.9) |
| Azithromycin (N: 239) | 94 (39.3) |
| Lopinavir/Ritonavir (N: 239) | 21(8.7) |
| Steroids (N:284) | 92 (32.3) |
| Tocilizumab (N: 239) | 21 (8.7) |
| Beta-Interferon (N: 239) | 12 (5) |
| Remdesevir (N: 239) | 1 (0.4) |
| Other (Antibiotics.anticoagulants..etc) (N: 239) | 137 (57.3) |
| No treatment (N: 239) | 24 (10) |
| HR [CI 95%] | p | |
|---|---|---|
| Demographics and Comorbidities | ||
| Age (years) < 70 (Ref.) | ||
| 70–80 | 2.889 [1.569–5.321] | 0.001 |
| >80 | 2.817 [1.529–5.187] | 0.001 |
| Gender (female) | 1.195 [0.749–1.905] | 0.455 |
| Race (caucasian) | 0.957 [0.738–1.241] | 0.742 |
| Body mass index (kg/m2) | 0.929 [0.882–0.979] | 0.006 |
| Dialysis vintage (years) | 1.011 [0.974–1.050] | 0.563 |
| Renal etiology | 0.983 [0.908–1.065] | 0.682 |
| Current Alcohol status | 0.347 [0.084–1.429] | 0.143 |
| Current Smoking status | 1.019 [0.617–1.683] | 0.941 |
| Native fistulae as vascular access | 0.679 [0.428–1.078] | 0.101 |
| Previous transplant | 0.915 [0.369–2.271] | 0.848 |
| Diabetes Mellitus | 1.080 [0.698–1.670] | 0.730 |
| Arterial hypertension | 0.974 [0.629–1.510] | 0.908 |
| Tuberculosis | 0.649 [0.090–4.672] | 0.668 |
| Dyslipemia | 0.839 [0.529–1.332] | 0.456 |
| Lung disease | 1.537 [0.959–2.463] | 0.074 |
| Ischemic cardiopahty | 1.181 [0.750–1.859] | 0.474 |
| Mineral Metabolism | ||
| 25-OH-Vitamin D. (ng/mL) | 0.994 [0.975–1.012] | 0.491 |
| 25-OH Vitamin D (ng/mL) | ||
| <20 (Ref.) | 1 | |
| 20–30 | 1.185 [0.663–2.115] | 0.567 |
| >30 | 1.077 [0.586–1.979] | 0.811 |
| PTH (ng/L) | 0.998 [0.997–0.999] | 0.002 |
| Calcium (mg/dL) | 0.875 [0.765–1.000] | 0.051 |
| Phosphate (mg/dL) | 0.879 [0.758–1.020] | 0.089 |
| Vitamin D derivatives use | 0.493 [0.310–0.782] | 0.003 |
| Calcidiol use | 1.140 [0.591–2.222] | 0.686 |
| Calcidiol dose (mcg/month) | 0.999 [0.999–1.000] | 0.653 |
| Calcitriol use | 0.587 [0.215–1.605] | 0.260 |
| Calcitriol dose (mcg/week) | 0.489 [0.110–2.175] | 0.348 |
| Paricalcitol use | 0.455 [0.263 -0.787] | 0.002 |
| Paricalcitol dose (mcg/week) | 0.915 [0.749–1.118] | 0.385 |
| Calcimimetics use | 0.220 [0.080–0.601] | 0.003 |
| Other Treatments | ||
| ACEIs | 0.903 [0.523–1.560] | 0.715 |
| ARBs | 1.050 [0.525–2.099] | 0.891 |
| NSAIDs | 0.821 [0.332–2.029] | 0.669 |
| COVID-19 Clinical symptoms | ||
| Ageusia | 2.112 [0.499–8.945] | 0.310 |
| Anosmia | 1.565 [0.370–6.625] | 0.543 |
| Cough | 0.813 [0.515–1.283] | 0.374 |
| Fever | 1.665 [0.998–2.779] | 0.051 |
| Diarrhea | 0.832 [0.382–1.810] | 0.642 |
| Dyspnea | 1.755 [1.110–2.774] | 0.016 |
| Asthenia | 1.867 [1.165–2.993] | 0.009 |
| Asymptomatic | 0.259 [0.082–0.821] | 0.022 |
| Pneumonia | 4.705 [2.037–10.867] | <0.001 |
| X-Ray compatible with COVID-19 | 5.629 [1.735–18.265] | 0.004 |
| Pre-hospital admission for COVID-19–Laboratory findings | ||
| Hemoglobin (g/dL) | 0.937 [0.842–1.043] | 0.617 |
| White blood cells (n/mm3) | . | |
| <4500 (Ref.) | 1 | |
| 4500–7000 | 0.751 [0.443–1.272] | 0.287 |
| >7000 | 0.797 [0.458–1.388] | 0.423 |
| Lymphocytes (n/mm3), | . | |
| <700 (Ref.) | 1 | |
| 700–1200 | 0.832 [0.507–1.367] | 0.468 |
| >1200 | 0.634 [0.357–1.125] | 0.120 |
| Platelets (n/mm3) (×100) | 1.000 [0.998–1.003] | 0.927 |
| Neutrophils (n/mm3) (×100) | 0.988 [0.912–1.071] | 0.770 |
| Albumin (g/dL) | 0.762 [0.630–0.923] | 0.005 |
| C-reactive protein (mg/L) | ||
| <5.6 (Ref.) | 1 | |
| 5.6–24 | 2.492 [1.349–4.602] | 0.004 |
| >24 | 2.746 [1.495–5.043] | 0.001 |
| D-dimer (ng/mL) | . | |
| <700 (Ref.) | 1 | |
| 700–1500 | 1.02 [0.433–2.403] | 0.963 |
| >1500 | 2.175 [1.049–4.513] | 0.037 |
| Fibrinogen (mg/dL) | . | |
| <400 (Ref.) | ||
| 400–600 | 1.37 [0.613–3.063] | 0.444 |
| >600 | 1.192 [0.51–2.786] | 0.685 |
| AST (IU/L) | 0.999 [0.995–1.003] | 0.678 |
| Alanin aminotransferase (ALT) (IU/L) | 0.998 [0.988–1.009] | 0.681 |
| GGT (IU/L) | 0.997 [0.992–1.002] | 0.870 |
| LDH (IU/L) | . | |
| <200 (Ref.) | 1 | |
| 200–300 | 1.998 [ 0.922–4.331] | 0.079 |
| >300 | 1.798 [0.813–3.976] | 0.147 |
| Troponin T (ng/mL) | 0.996 [0.982–1.01] | 0.336 |
| Procalcitonin (ng/mL) | . | |
| <0.5 (Ref.) | 1 | |
| 0.5–1 | 1.207 [0.479–3.04] | 0.690 |
| >1 | 2.48 [1.085–5.672] | 0.031 |
| IL-6 (ng/L) | 0.998 [0.988–1.008] | 0.744 |
| Ferritin (μg/L) | . | |
| <400 (Ref.) | 1 | |
| 400–800 | 0.321 [1.092–0] | 0.093 |
| >800 | 1.218 [0.731–2.029] | 0.449 |
| Transferrin Saturation Index (%) | 0.999 [0.984–1.015] | 0.958 |
| COVID-19–Outcomes | ||
| Hospital admission | 5.295 [1.937–14.470] | 0.001 |
| Oxygen saturation | 0.857 [0.809–0.908] | <0.001 |
| Non-invasive mechanical ventilation | 2.177 [1.342–3.529] | 0.002 |
| ICU admission | 1.560 [0.718–3.392] | 0.262 |
| Endotracheal intubation | 1.727 [0.697–4.276] | 0.238 |
| COVID-19–Treatments | ||
| Hydroxychloroquine | 1.251 [0.692–2.263] | 0.459 |
| Azithromycin | 1.268 [0.775–2.075] | 0.344 |
| Lopinavir/Ritonavir | 1.938 [1.185–3.167] | 0.008 |
| Steroids | 1.162 [0.738–1.831] | 0.516 |
| Beta-Interferon | 3.663 [1.738–7.719] | 0.001 |
| Tocilizumab | 0.872 [0.350–2.174] | 0.769 |
| Antibiotics | 1.402 [0.841–2.336] | 0.195 |
| No Treatment | 0.599 [0.218–1.647] | 0.320 |
| HR [IC95%] | p | |
|---|---|---|
| Age (years) | ||
| ≤70 (Ref) | 1 | |
| 70–80 | 2.911 [1.532–5.532] | 0.001 |
| ≥80 | 2.372 [1.195–4.707] | 0.014 |
| Serum albumin (g/dL) | 0.668 [0.496–0.900] | 0.008 |
| Paricalcitol use (Yes) | 0.499 [0.278–0.898] | 0.020 |
| Calcimimetics use (Yes) | 0.332 [0.12–0.921] | 0.034 |
| C-reactive protein (mg/dL) | ||
| <5.6 (Ref) | 1 | |
| 5.6–24 | 1.576 [0.808–3.072] | 0.182 |
| >24 | 2.417 [1.284–4.55] | 0.006 |
| PTH (ng/L) | 0.999 [0.998–1.000] | 0.167 |
| Serum calcium (mg/dL) | 1.046 [0.837–1.306] | 0.695 |
| Serum phosphorus (mg/dL) | 1.037 [0.863–1.246] | 0.697 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arenas Jimenez, M.D.; González-Parra, E.; Riera, M.; Rincón Bello, A.; López-Herradón, A.; Cao, H.; Hurtado, S.; Collado, S.; Ribera, L.; Barbosa, F.; et al. Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics. Nutrients 2021, 13, 2559. https://doi.org/10.3390/nu13082559
Arenas Jimenez MD, González-Parra E, Riera M, Rincón Bello A, López-Herradón A, Cao H, Hurtado S, Collado S, Ribera L, Barbosa F, et al. Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics. Nutrients. 2021; 13(8):2559. https://doi.org/10.3390/nu13082559
Chicago/Turabian StyleArenas Jimenez, María Dolores, Emilio González-Parra, Marta Riera, Abraham Rincón Bello, Ana López-Herradón, Higini Cao, Sara Hurtado, Silvia Collado, Laura Ribera, Francesc Barbosa, and et al. 2021. "Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics" Nutrients 13, no. 8: 2559. https://doi.org/10.3390/nu13082559
APA StyleArenas Jimenez, M. D., González-Parra, E., Riera, M., Rincón Bello, A., López-Herradón, A., Cao, H., Hurtado, S., Collado, S., Ribera, L., Barbosa, F., Dapena, F., Torregrosa, V., Broseta, J.-J., Soto Montañez, C., Navarro-González, J. F., Ramos, R., Bover, J., Nogués-Solan, X., Crespo, M., ... Pascual, J. (2021). Mortality in Hemodialysis Patients with COVID-19, the Effect of Paricalcitol or Calcimimetics. Nutrients, 13(8), 2559. https://doi.org/10.3390/nu13082559






