Nutrition to Optimise Human Health—How to Obtain Physiological Substantiation?
Abstract
1. Introduction
2. Concepts of Maintaining, Improved or Declining Health
2.1. How to Define Health?
2.2. From Normality to Response to Stressors as Proxy for Health
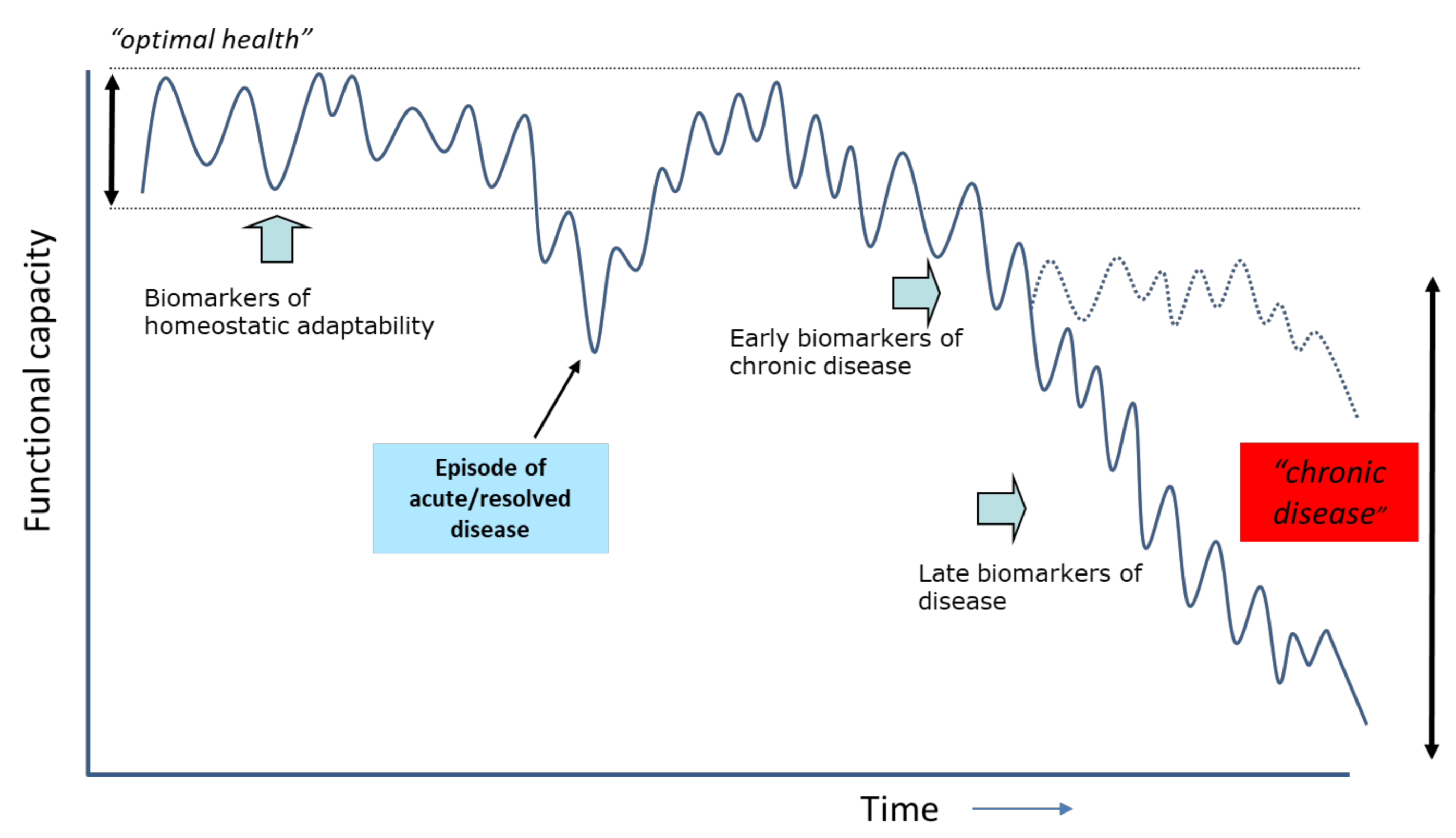
2.3. Loss of Health and Development of Disease
2.4. Effects of Nutrition to Optimise Health—Differences in Starting Points and Goals
2.4.1. Inadequate Nutrition (Section 3)
2.4.2. Modulating Suboptimal Health (Section 4)
2.4.3. Optimising Future Health (Section 5) in Apparently Healthy (‘Normal’) Persons
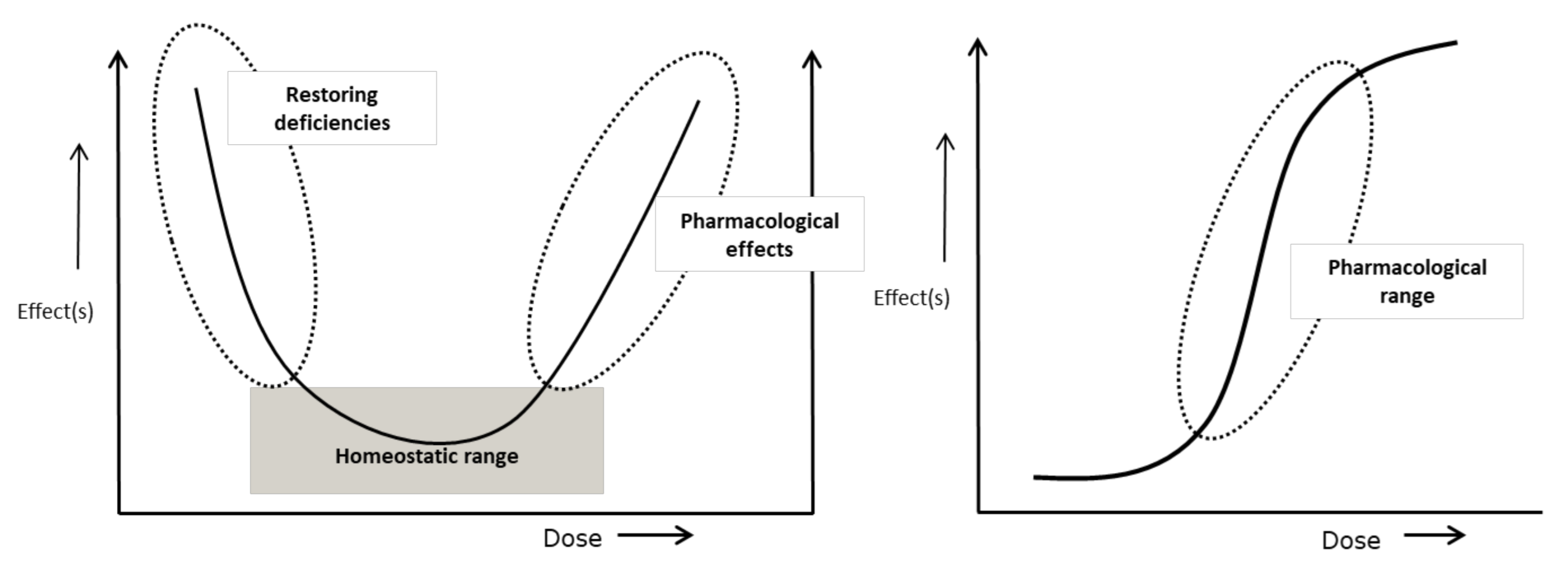
3. Correcting Inadequate Nutrition and Deficiencies
4. Optimising Suboptimal Health with Nutrition
5. Assessing Health Optimisation in Non-Diseased Individuals
5.1. The Current Status of Health Claims—The EU as Example
- ■
- the growth, development and functions of the body;
- ■
- referring to psychological and behavioural functions;
- ■
- slimming or weight-control.
- (1)
- For many of these authorized claims, perhaps in particular those of the 13.1 category, it remains very difficult or even unlikely that they can be easily translated into concrete health benefits that can be understood and/or experienced by consumers.
- (2)
- Only a limited proportion of authorized claims seems to be able to meet the demand that their use contributes to measurable health optimization.
- (3)
- The number of physiological studies that have provided novel, convincing scientific evidence is very small. It seems conceivable that this is due to scientific and also economic reasons.
5.2. Is Optimisation of Normal Physiology or Risk Factors for Disease via Nutrition Biologically Feasible?
5.3. Biomarkers and Endpoints
5.4. The Nutritional Phenotype and ‘Multi-Omics’ Revolution
5.5. Methodologies: Examples of Studies and Outcome Measures
5.6. Development in Study Designs and Measurement of Effects
5.6.1. The Need for Alternatives to RCTs
5.6.2. Remote and Real-Time Studies
6. Conclusions
Funding
Conflicts of Interest
References
- Canguilhem, G. Le Normal et le Pathologique; Presses Universitaires de France: Paris, France, 1943; Volume 2015. [Google Scholar]
- Wickramasinghe, K.; Mathers, J.C.; Wopereis, S.; Marsman, D.S.; Griffiths, J.C. From lifespan to healthspan: The role of nutrition in healthy ageing. J. Nutr. Sci. 2020, 9, e33. [Google Scholar] [CrossRef]
- Shao, A.; Drewnowski, A.; Willcox, D.C.; Kramer, L.; Lausted, C.; Eggersdorfer, M.; Mathers, J.; Bell, J.D.; Randolph, R.K.; Witkamp, R.; et al. Optimal nutrition and the ever-changing dietary landscape: A conference report. Eur. J. Nutr. 2017, 56, 1–21. [Google Scholar] [CrossRef]
- Michaëlsson, K.; Baron, J.A.; Byberg, L.; Höijer, J.; Larsson, S.C.; Svennblad, B.; Melhus, H.; Wolk, A.; Warensjö Lemming, E. Combined associations of body mass index and adherence to a Mediterranean-like diet with all-cause and cardiovascular mortality: A cohort study. PLoS Med. 2020, 17, e1003331. [Google Scholar] [CrossRef]
- Keys, A. Mediterranean diet and public health: Personal reflections. Am. J. Clin. Nutr. 1995, 61, 1321S–1323S. [Google Scholar] [CrossRef]
- Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.; Naska, A.; Neuhäuser-Berthold, M.; et al. General scientific guidance for stakeholders on health claim applications. EFSA J. 2016, 14, 4367. [Google Scholar] [CrossRef]
- Conti, A.A. Historical evolution of the concept of health in Western medicine. Acta Bio Med. 2018, 89, 352–354. [Google Scholar] [CrossRef]
- Gallagher, A.M.; Meijer, G.W.; Richardson, D.P.; Rondeau, V.; Skarp, M.; Stasse-Wolthuis, M.; Tweedie, G.C.; Witkamp, R.; International Life Sciences Institute Europe Functional Foods Task, F. A standardised approach towards PROving the efficacy of foods and food constituents for health CLAIMs (PROCLAIM): Providing guidance. Br. J. Nutr. 2011, 106, S16–S28. [Google Scholar] [CrossRef]
- WHO. Preamble to the Constitution of the World Health Organization as adopted by the International Health Conference; WHO: Geneva, Switzerland, 1946. [Google Scholar]
- De Carvalho Yokota, R.T.; Van Oyen, H. Operationalization of Concepts of Health and Disability. In International Handbook of Health Expectancies; Jagger, C., Crimmins, E.M., Saito, Y., De Carvalho Yokota, R.T., Van Oyen, H., Robine, J.-M., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 3–18. [Google Scholar]
- Breslow, L. Health Status Measurement in the Evaluation of Health Promotion. Med. Care 1989, 27, S205–S216. [Google Scholar] [CrossRef] [PubMed]
- Lancet. What is health? The ability to adapt. Lancet 2009, 373, 781. [Google Scholar] [CrossRef]
- Huber, M.; Knottnerus, J.A.; Green, L.; van der Horst, H.; Jadad, A.R.; Kromhout, D.; Leonard, B.; Lorig, K.; Loureiro, M.I.; van der Meer, J.W.; et al. How should we define health? BMJ 2011, 343, d4163. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; van Vliet, M.; Giezenberg, M.; Winkens, B.; Heerkens, Y.; Dagnelie, P.C.; Knottnerus, J.A. Towards a ‘patient-centred’ operationalisation of the new dynamic concept of health: A mixed methods study. BMJ Open 2016, 6, e010091. [Google Scholar] [CrossRef]
- Prinsen, C.A.C.; Terwee, C.B. Measuring positive health: For now, a bridge too far. Public Health 2019, 170, 70–77. [Google Scholar] [CrossRef]
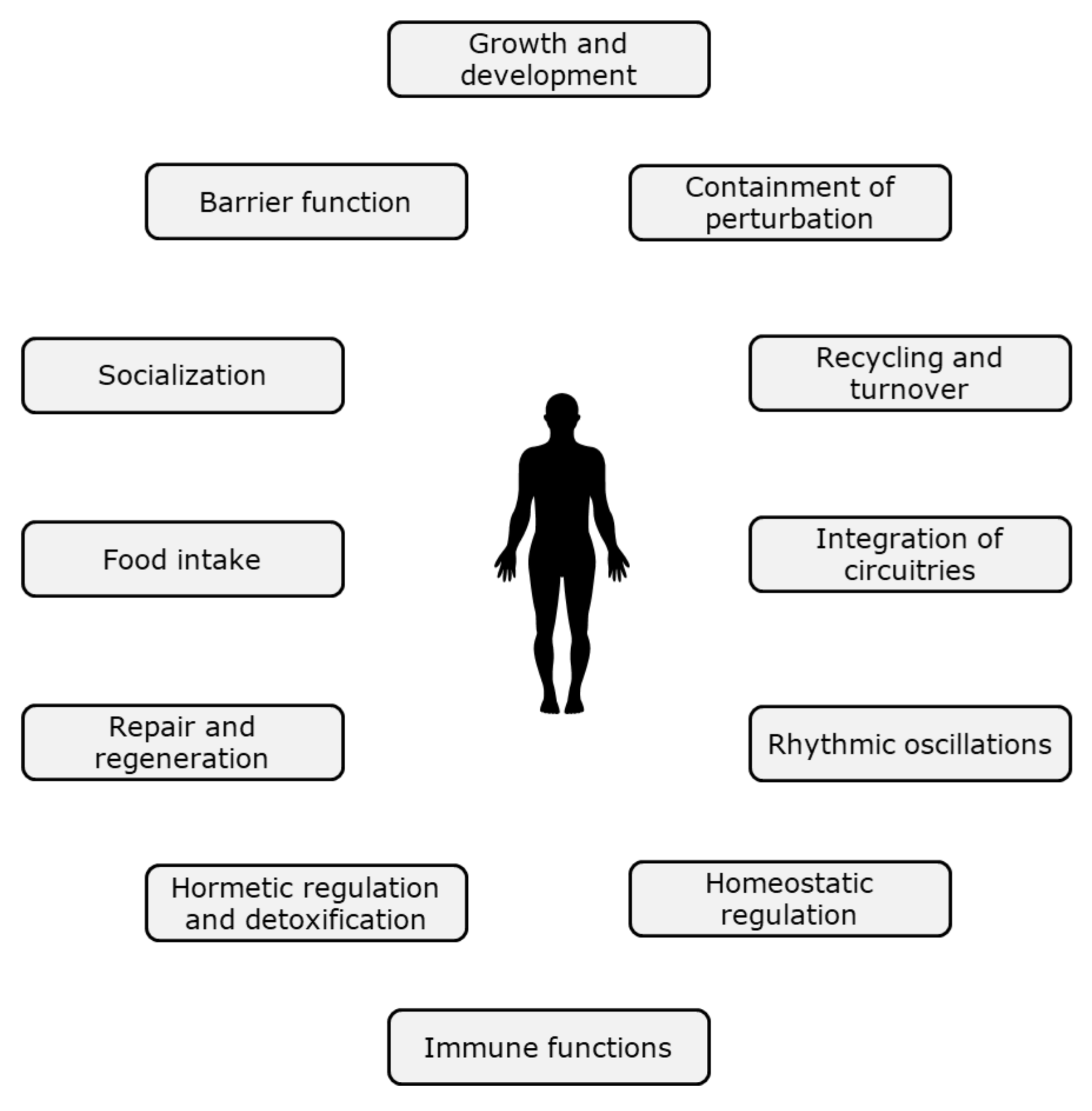
- Ayres, J.S. The Biology of Physiological Health. Cell 2020, 181, 250–269. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Otin, C.; Kroemer, G. Hallmarks of Health. Cell 2021, 184, 33–63. [Google Scholar] [CrossRef]
- Costa-Mattioli, M.; Walter, P. The integrated stress response: From mechanism to disease. Science 2020, 368, eaat5314. [Google Scholar] [CrossRef] [PubMed]
- Russell, G.; Lightman, S. The human stress response. Nat. Rev. Endocrinol. 2019, 15, 525–534. [Google Scholar] [CrossRef]
- Van Ommen, B.; van der Greef, J.; Ordovas, J.M.; Daniel, H. Phenotypic flexibility as key factor in the human nutrition and health relationship. Genes Nutr. 2014, 9, 423. [Google Scholar] [CrossRef] [PubMed]
- Amador, E. Health and Normality. JAMA 1975, 232, 953–955. [Google Scholar] [CrossRef]
- Catita, M.; Aguas, A.; Morgado, P. Normality in medicine: A critical review. Philos. Ethics Humanit. Med. 2020, 15, 3. [Google Scholar] [CrossRef]
- Witkamp, R.F.; van Norren, K. Let thy food be thy medicine....when possible. Eur. J. Pharmacol. 2018, 836, 102–114. [Google Scholar] [CrossRef]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef]
- Debelo, H.; Novotny, J.A.; Ferruzzi, M.G. Vitamin A. Adv. Nutr. 2017, 8, 992–994. [Google Scholar] [CrossRef] [PubMed]
- Wardenaar, F.; Brinkmans, N.; Ceelen, I.; Van Rooij, B.; Mensink, M.; Witkamp, R.; De Vries, J. Micronutrient Intakes in 553 Dutch Elite and Sub-Elite Athletes: Prevalence of Low and High Intakes in Users and Non-Users of Nutritional Supplements. Nutrients 2017, 9, 142. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Webb, D.; Lichtenstein, A.H.; Tucker, K.L.; Akabas, S. Micronutrient Status: Potential Modifiers—Drugs and Chronic Disease. Adv. Nutr. 2018, 9, 509S–510S. [Google Scholar] [CrossRef]
- Wesselink, E.; Koekkoek, W.A.C.; Grefte, S.; Witkamp, R.F.; van Zanten, A.R.H. Feeding mitochondria: Potential role of nutritional components to improve critical illness convalescence. Clin. Nutr. 2019, 38, 982–995. [Google Scholar] [CrossRef] [PubMed]
- Hovinen, T.; Korkalo, L.; Freese, R.; Skaffari, E.; Isohanni, P.; Niemi, M.; Nevalainen, J.; Gylling, H.; Zamboni, N.; Erkkola, M.; et al. Vegan diet in young children remodels metabolism and challenges the statuses of essential nutrients. EMBO Mol. Med. 2021, 13, e13492. [Google Scholar] [CrossRef] [PubMed]
- Peter, S.; Navis, G.; de Borst, M.H.; von Schacky, C.; van Orten-Luiten, A.C.B.; Zhernakova, A.; Witkamp, R.F.; Janse, A.; Weber, P.; Bakker, S.J.L.; et al. Public health relevance of drug-nutrition interactions. Eur. J. Nutr. 2017, 56, 23–36. [Google Scholar] [CrossRef]
- Krzizek, E.C.; Brix, J.M.; Stöckl, A.; Parzer, V.; Ludvik, B. Prevalence of Micronutrient Deficiency after Bariatric Surgery. Obes. Facts 2021, 14, 197–204. [Google Scholar] [CrossRef]
- Semba, R.D. The Rise and Fall of Protein Malnutrition in Global Health. Ann. Nutr. Metab. 2016, 69, 79–88. [Google Scholar] [CrossRef]
- Van Ommen, B.; Fairweather-Tait, S.; Freidig, A.; Kardinaal, A.; Scalbert, A.; Wopereis, S. A network biology model of micronutrient related health. Br. J. Nutr. 2008, 99, S72–S80. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Bischoff-Ferrari, H.A.; Bouillon, R.; Feskens, E.J.; Gallagher, C.J.; Hypponen, E.; Llewellyn, D.J.; Stoecklin, E.; Dierkes, J.; Kies, A.K.; et al. Vitamin D: Do we get enough? A discussion between vitamin D experts in order to make a step towards the harmonisation of dietary reference intakes for vitamin D across Europe. Osteoporos. Int. 2013, 24, 1567–1577. [Google Scholar] [CrossRef] [PubMed]
- Balvers, M.G.; Brouwer-Brolsma, E.M.; Endenburg, S.; de Groot, L.C.; Kok, F.J.; Gunnewiek, J.K. Recommended intakes of vitamin D to optimise health, associated circulating 25-hydroxyvitamin D concentrations, and dosing regimens to treat deficiency: Workshop report and overview of current literature. J. Nutr. Sci. 2015, 4, e23. [Google Scholar] [CrossRef] [PubMed]
- Langlois, P.; Lamontagne, F. Vitamin C for the critically ill: Is the evidence strong enough? Nutrition 2019, 60, 185–190. [Google Scholar] [CrossRef]
- Brant, E.B.; Angus, D.C. Is High-Dose Vitamin C Beneficial for Patients With Sepsis? JAMA 2019, 322, 1257–1258. [Google Scholar] [CrossRef]
- Van Soest, A.P.M.; van de Rest, O.; Witkamp, R.F.; de Groot, L. Positive effects of folic acid supplementation on cognitive aging are dependent on omega-3 fatty acid status: A post hoc analysis of the FACIT trial. Am. J. Clin. Nutr. 2021, 113, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Terink, R.; Balvers, M.G.J.; Hopman, M.T.; Witkamp, R.F.; Mensink, M.; Gunnewiek, J. Decrease in Ionized and Total Magnesium Blood Concentrations in Endurance Athletes Following an Exercise Bout Restores within Hours-Potential Consequences for Monitoring and Supplementation. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 264–270. [Google Scholar] [CrossRef]
- Oliai Araghi, S.; van Dijk, S.C.; Ham, A.C.; Brouwer-Brolsma, E.M.; Enneman, A.W.; Sohl, E.; Swart, K.M.; van der Zwaluw, N.L.; van Wijngaarden, J.P.; Dhonukshe-Rutten, R.A.; et al. BMI and Body Fat Mass Is Inversely Associated with Vitamin D Levels in Older Individuals. J. Nutr. Health Aging 2015, 19, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Morley, J.E. Vitamin D: Does the Emperor Have no Clothes? J. Nutr. Health Aging 2019, 23, 316–317. [Google Scholar] [CrossRef]
- Chatelan, A.; Muka, T.; Franco, O.H. Eating to add years of life and life to these years: What should be in the menu? Am. J. Clin. Nutr. 2020, 111, 733–734. [Google Scholar] [CrossRef]
- Downer, S.; Berkowitz, S.A.; Harlan, T.S.; Olstad, D.L.; Mozaffarian, D. Food is medicine: Actions to integrate food and nutrition into healthcare. BMJ 2020, 369, m2482. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Schoufour, J.; Wang, D.D.; Dhana, K.; Pan, A.; Liu, X.; Song, M.; Liu, G.; Shin, H.J.; Sun, Q.; et al. Healthy lifestyle and life expectancy free of cancer, cardiovascular disease, and type 2 diabetes: Prospective cohort study. BMJ 2020, 368, l6669. [Google Scholar] [CrossRef] [PubMed]
- Nyberg, S.T.; Singh-Manoux, A.; Pentti, J.; Madsen, I.E.H.; Sabia, S.; Alfredsson, L.; Bjorner, J.B.; Borritz, M.; Burr, H.; Goldberg, M.; et al. Association of Healthy Lifestyle With Years Lived Without Major Chronic Diseases. JAMA Intern. Med. 2020, 180, 760–768. [Google Scholar] [CrossRef]
- Pot, G.K.; Battjes-Fries, M.C.; Patijn, O.N.; Pijl, H.; Witkamp, R.F.; de Visser, M.; van der Zijl, N.; de Vries, M.; Voshol, P.J. Nutrition and lifestyle intervention in type 2 diabetes: Pilot study in the Netherlands showing improved glucose control and reduction in glucose lowering medication. BMJ Nutr. Prev. Health 2019, 2, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Katsagoni, C.N.; Georgoulis, M.; Papatheodoridis, G.V.; Panagiotakos, D.B.; Kontogianni, M.D. Effects of lifestyle interventions on clinical characteristics of patients with non-alcoholic fatty liver disease: A meta-analysis. Metabolism 2017, 68, 119–132. [Google Scholar] [CrossRef]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef]
- McCombie, L.; Leslie, W.; Taylor, R.; Kennon, B.; Sattar, N.; Lean, M.E.J. Beating type 2 diabetes into remission. BMJ 2017, 358, j4030. [Google Scholar] [CrossRef]
- Van Ommen, B.; Wopereis, S.; van Empelen, P.; van Keulen, H.M.; Otten, W.; Kasteleyn, M.; Molema, J.J.W.; de Hoogh, I.M.; Chavannes, N.H.; Numans, M.E.; et al. From Diabetes Care to Diabetes Cure-The Integration of Systems Biology, eHealth, and Behavioral Change. Front. Endocrinol. 2017, 8, 381. [Google Scholar] [CrossRef]
- Pan, A.; Lin, X.; Hemler, E.; Hu, F.B. Diet and Cardiovascular Disease: Advances and Challenges in Population-Based Studies. Cell Metab. 2018, 27, 489–496. [Google Scholar] [CrossRef]
- Steinberg, D.; Bennett, G.G.; Svetkey, L. The DASH Diet, 20 Years Later. JAMA 2017, 317, 1529–1530. [Google Scholar] [CrossRef]
- Perez-Martinez, P.; Mikhailidis, D.P.; Athyros, V.G.; Bullo, M.; Couture, P.; Covas, M.I.; de Koning, L.; Delgado-Lista, J.; Diaz-Lopez, A.; Drevon, C.A.; et al. Lifestyle recommendations for the prevention and management of metabolic syndrome: An international panel recommendation. Nutr. Rev. 2017, 75, 307–326. [Google Scholar] [CrossRef]
- Webb, V.L.; Wadden, T.A. Intensive Lifestyle Intervention for Obesity: Principles, Practices, and Results. Gastroenterology 2017, 152, 1752–1764. [Google Scholar] [CrossRef]
- Marx, W.; Lane, M.; Hockey, M.; Aslam, H.; Berk, M.; Walder, K.; Borsini, A.; Firth, J.; Pariante, C.M.; Berding, K.; et al. Diet and depression: Exploring the biological mechanisms of action. Mol. Psychiatry 2021, 26, 134–150. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Sanchez-Villegas, A. Food patterns and the prevention of depression. Proc. Nutr. Soc. 2016, 75, 139–146. [Google Scholar] [CrossRef]
- Rayman, M.P. Diet, nutrition and osteoarthritis. BMC Musculoskelet. Disord. 2015, 16, S7. [Google Scholar] [CrossRef]
- Tuck, C.J.; Vanner, S.J. Dietary therapies for functional bowel symptoms: Recent advances, challenges, and future directions. Neurogastroenterol. Motil. 2018, 30, e13238. [Google Scholar] [CrossRef]
- Fitzgerald, K.C.; Tyry, T.; Salter, A.; Cofield, S.S.; Cutter, G.; Fox, R.; Marrie, R.A. Diet quality is associated with disability and symptom severity in multiple sclerosis. Neurology 2017, 90, e1–e11. [Google Scholar] [CrossRef]
- Verhagen, H.; van Loveren, H. Status of nutrition and health claims in Europe by mid 2015. Trends Food Sci. Technol. 2016, 56, 39–45. [Google Scholar] [CrossRef]
- EU Register of Nutrition and Health Claims Made on Food. Available online: https://ec.europa.eu/food/safety/labelling_nutrition/claims/register/public/?event=search (accessed on 3 April 2021).
- Netea, M.G.; Dominguez-Andres, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining trained immunity and its role in health and disease. Nat. Rev. Immunol. 2020, 20, 375–388. [Google Scholar] [CrossRef]
- JanssenDuijghuijsen, L.M.; Keijer, J.; Mensink, M.; Lenaerts, K.; Ridder, L.; Nierkens, S.; Kartaram, S.W.; Verschuren, M.C.; Pieters, R.H.; Bas, R.; et al. Adaptation of exercise-induced stress in well-trained healthy young men. Exp. Physiol. 2017, 102, 86–99. [Google Scholar] [CrossRef]
- McGee, S.L.; Hargreaves, M. Exercise adaptations: Molecular mechanisms and potential targets for therapeutic benefit. Nat. Rev. Endocrinol. 2020, 16, 495–505. [Google Scholar] [CrossRef]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef]
- Dai, X.; Yan, X.; Wintergerst, K.A.; Cai, L.; Keller, B.B.; Tan, Y. Nrf2: Redox and Metabolic Regulator of Stem Cell State and Function. Trends Mol. Med. 2020, 26, 185–200. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Kozumbo, W.J. The hormetic dose-response mechanism: Nrf2 activation. Pharmacol. Res. 2021, 167, 105526. [Google Scholar] [CrossRef]
- Li, X.; Yang, T.; Sun, Z. Hormesis in Health and Chronic Diseases. Trends Endocrinol. Metab. 2019, 30, 944–958. [Google Scholar] [CrossRef]
- Martucci, M.; Ostan, R.; Biondi, F.; Bellavista, E.; Fabbri, C.; Bertarelli, C.; Salvioli, S.; Capri, M.; Franceschi, C.; Santoro, A. Mediterranean diet and inflammaging within the hormesis paradigm. Nutr. Rev. 2017, 75, 442–455. [Google Scholar] [CrossRef]
- Rothhammer, V.; Quintana, F.J. The aryl hydrocarbon receptor: An environmental sensor integrating immune responses in health and disease. Nat. Rev. Immunol. 2019, 19, 184–197. [Google Scholar] [CrossRef]
- Shinde, R.; McGaha, T.L. The Aryl Hydrocarbon Receptor: Connecting Immunity to the Microenvironment. Trends Immunol. 2018, 39, 1005–1020. [Google Scholar] [CrossRef]
- Hubbard, B.P.; Sinclair, D.A. Small molecule SIRT1 activators for the treatment of aging and age-related diseases. Trends Pharmacol. Sci. 2014, 35, 146–154. [Google Scholar] [CrossRef]
- Egger, G.; Dixon, J. Obesity and chronic disease: Always offender or often just accomplice? Br. J. Nutr. 2009, 102, 1238–1242. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation, metaflammation and immunometabolic disorders. Nature 2017, 542, 177. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Balkwill, F.; Chonchol, M.; Cominelli, F.; Donath, M.Y.; Giamarellos-Bourboulis, E.J.; Golenbock, D.; Gresnigt, M.S.; Heneka, M.T.; Hoffman, H.M.; et al. A guiding map for inflammation. Nat. Immunol. 2017, 18, 826–831. [Google Scholar] [CrossRef]
- Lee, Y.S.; Wollam, J.; Olefsky, J.M. An Integrated View of Immunometabolism. Cell 2018, 172, 22–40. [Google Scholar] [CrossRef]
- Tilg, H.; Zmora, N.; Adolph, T.E.; Elinav, E. The intestinal microbiota fuelling metabolic inflammation. Nat. Rev. Immunol. 2020, 20, 40–54. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Lee, S.K.; Zhang, D.; Frenette, P.S. The Gut Microbiome Regulates Psychological-Stress-Induced Inflammation. Immunity 2020, 53, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.D.; Nguyen, L.H.; Li, Y.; Yan, Y.; Ma, W.; Rinott, E.; Ivey, K.L.; Shai, I.; Willett, W.C.; Hu, F.B. The gut microbiome modulates the protective association between a Mediterranean diet and cardiometabolic disease risk. Nat. Med. 2021, 27, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Zmora, N.; Suez, J.; Elinav, E. You are what you eat: Diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 35–56. [Google Scholar] [CrossRef]
- Stockinger, B.; Shah, K.; Wincent, E. AHR in the intestinal microenvironment: Safeguarding barrier function. Nat. Rev. Gastroenterol. Hepatol. 2021, 1–12. [Google Scholar] [CrossRef]
- Weseler, A.R.; Bast, A. Pleiotropic-acting nutrients require integrative investigational approaches: The example of flavonoids. J. Agric. Food Chem. 2012, 60, 8941–8946. [Google Scholar] [CrossRef]
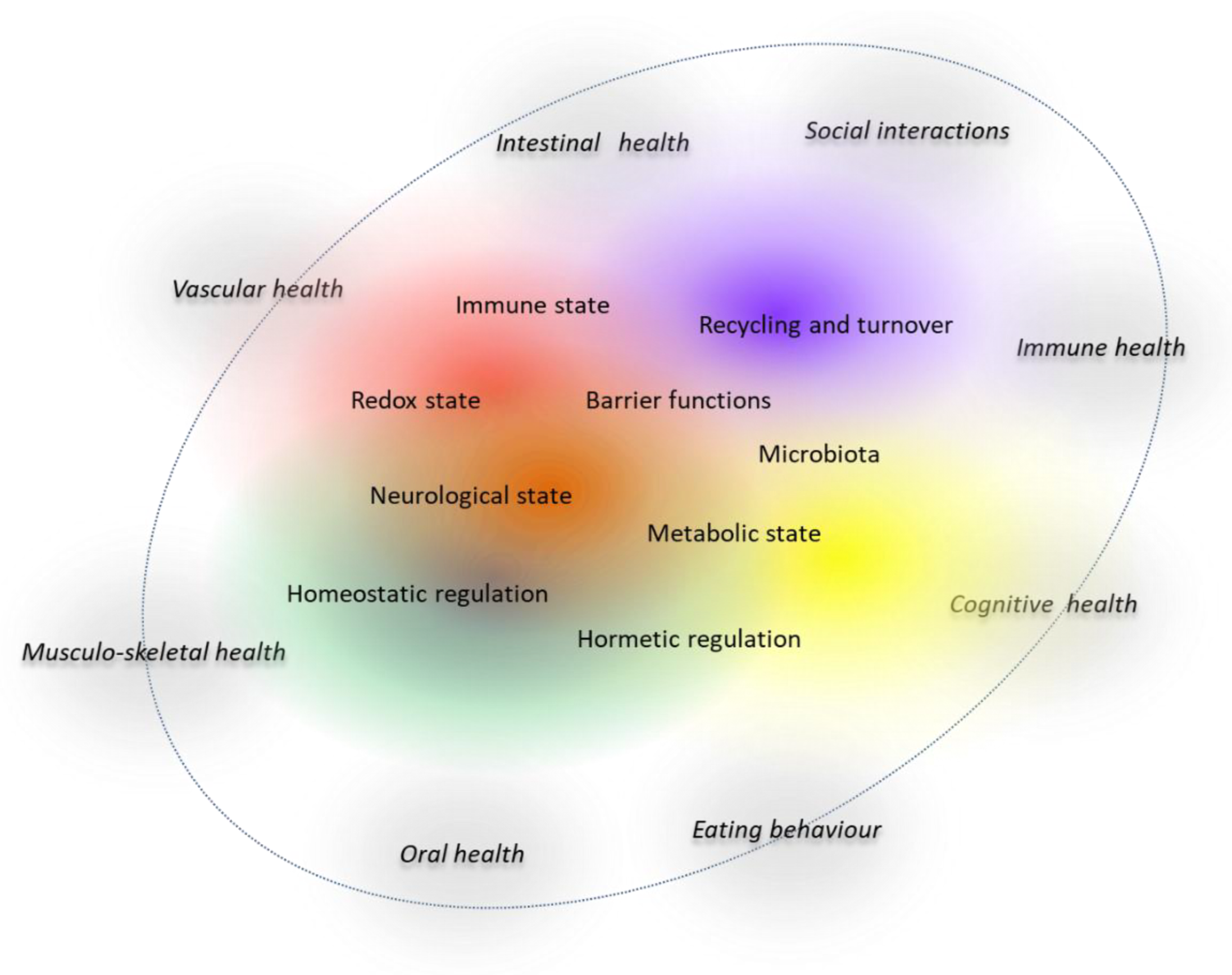
- Bouwman, J.; Vogels, J.T.; Wopereis, S.; Rubingh, C.M.; Bijlsma, S.; Ommen, B. Visualization and identification of health space, based on personalized molecular phenotype and treatment response to relevant underlying biological processes. BMC Med. Genom. 2012, 5, 1. [Google Scholar] [CrossRef]
- Van den Broek, T.J.; Bakker, G.C.M.; Rubingh, C.M.; Bijlsma, S.; Stroeve, J.H.M.; van Ommen, B.; van Erk, M.J.; Wopereis, S. Ranges of phenotypic flexibility in healthy subjects. Genes Nutr. 2017, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Ball, J.R.; Micheel, C.M. Evaluation of Biomarkers and Surrogate Endpoints in Chronic Disease; The National Academies Press: Washington, DC, USA, 2010; p. 334. [Google Scholar]
- Boobis, A.; Chiodini, A.; Hoekstra, J.; Lagiou, P.; Przyrembel, H.; Schlatter, J.; Schutte, K.; Verhagen, H.; Watzl, B. Critical appraisal of the assessment of benefits and risks for foods, ’BRAFO Consensus Working Group’. Food Chem. Toxicol. 2013, 55, 659–675. [Google Scholar] [CrossRef]
- Yetley, E.A.; DeMets, D.L.; Harlan, W.R., Jr. Surrogate disease markers as substitutes for chronic disease outcomes in studies of diet and chronic disease relations. Am. J. Clin. Nutr. 2017, 106, 1175–1189. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Pratico, G.; Scalbert, A.; Vergeres, G.; Kolehmainen, M.; Manach, C.; Brennan, L.; Afman, L.A.; Wishart, D.S.; Andres-Lacueva, C.; et al. A scheme for a flexible classification of dietary and health biomarkers. Genes Nutr. 2017, 12, 34. [Google Scholar] [CrossRef]
- Committee, E.S.; Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; et al. Guidance on the assessment of the biological relevance of data in scientific assessments. EFSA J. 2017, 15, e04970. [Google Scholar] [CrossRef]
- Van Ommen, B.; Keijer, J.; Kleemann, R.; Elliott, R.; Drevon, C.A.; McArdle, H.; Gibney, M.; Muller, M. The challenges for molecular nutrition research 2: Quantification of the nutritional phenotype. Genes Nutr. 2008, 3, 51–59. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Freake, H.C.; Bauman, D.E.; Bier, D.M.; Burrin, D.G.; German, J.B.; Klein, S.; Marquis, G.S.; Milner, J.A.; Pelto, G.H.; et al. The nutritional phenotype in the age of metabolomics. J. Nutr. 2005, 135, 1613–1616. [Google Scholar] [CrossRef]
- Van Ommen, B.; Keijer, J.; Heil, S.G.; Kaput, J. Challenging homeostasis to define biomarkers for nutrition related health. Mol. Nutr. Food Res. 2009, 53, 795–804. [Google Scholar] [CrossRef]
- Vis, D.J.; Westerhuis, J.A.; Jacobs, D.M.; van Duynhoven, J.P.M.; Wopereis, S.; van Ommen, B.; Hendriks, M.M.W.B.; Smilde, A.K. Analyzing metabolomics-based challenge tests. Metabolomics 2014, 11, 50–63. [Google Scholar] [CrossRef]
- Stroeve, J.H.; Wietmarschen, H.; Kremer, B.H.; Ommen, B.; Wopereis, S. Phenotypic flexibility as a measure of health: The optimal nutritional stress response test. Genes Nutr. 2015, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Kardinaal, A.F.; van Erk, M.J.; Dutman, A.E.; Stroeve, J.H.; van de Steeg, E.; Bijlsma, S.; Kooistra, T.; van Ommen, B.; Wopereis, S. Quantifying phenotypic flexibility as the response to a high-fat challenge test in different states of metabolic health. FASEB J. 2015, 29, 4600–4613. [Google Scholar] [CrossRef]
- Janssen Duijghuijsen, L.M.; Mensink, M.; Lenaerts, K.; Fiedorowicz, E.; Protege Study Group; van Dartel, D.A.M.; Mes, J.J.; Luiking, Y.C.; Keijer, J.; Wichers, H.J.; et al. The effect of endurance exercise on intestinal integrity in well-trained healthy men. Physiol. Rep. 2016, 4, e12994. [Google Scholar] [CrossRef]
- Kartaram, S.W.; van Norren, K.; Schoen, E.; Teunis, M.; Mensink, M.; Verschuren, M.; M’Rabet, L.; Besseling-van der Vaart, I.; Mohrmann, K.; Wittink, H.; et al. Kinetics of Physiological Responses as a Measure of Intensity and Hydration Status During Experimental Physical Stress in Human Volunteers. Front. Physiol. 2020, 11, 1006. [Google Scholar] [CrossRef]
- Ten Bruggencate, S.J.; Frederiksen, P.D.; Pedersen, S.M.; Floris-Vollenbroek, E.G.; Lucas-van de Bos, E.; van Hoffen, E.; Wejse, P.L. Dietary Milk-Fat-Globule Membrane Affects Resistance to Diarrheagenic Escherichia coli in Healthy Adults in a Randomized, Placebo-Controlled, Double-Blind Study. J. Nutr. 2016, 146, 249–255. [Google Scholar] [CrossRef]
- Schrieks, I.C.; Joosten, M.M.; Klopping-Ketelaars, W.A.; Witkamp, R.F.; Hendriks, H.F. Moderate alcohol consumption after a mental stressor attenuates the endocrine stress response. Alcohol 2016, 57, 29–34. [Google Scholar] [CrossRef]
- McCrea, C.E.; West, S.G.; Kris-Etherton, P.M.; Lambert, J.D.; Gaugler, T.L.; Teeter, D.L.; Sauder, K.A.; Gu, Y.; Glisan, S.L.; Skulas-Ray, A.C. Effects of culinary spices and psychological stress on postprandial lipemia and lipase activity: Results of a randomized crossover study and in vitro experiments. J. Transl. Med. 2015, 13, 7. [Google Scholar] [CrossRef]
- Hasin, Y.; Seldin, M.; Lusis, A. Multi-omics approaches to disease. Genome Biol. 2017, 18, 83. [Google Scholar] [CrossRef]
- Pinu, F.R.; Beale, D.J.; Paten, A.M.; Kouremenos, K.; Swarup, S.; Schirra, H.J.; Wishart, D. Systems Biology and Multi-Omics Integration: Viewpoints from the Metabolomics Research Community. Metabolites 2019, 9, 76. [Google Scholar] [CrossRef]
- Tavassoly, I.; Goldfarb, J.; Iyengar, R. Systems biology primer: The basic methods and approaches. Essays Biochem. 2018, 62, 487–500. [Google Scholar] [CrossRef]
- Nielsen, J. Systems Biology of Metabolism: A Driver for Developing Personalized and Precision Medicine. Cell Metab. 2017, 25, 572–579. [Google Scholar] [CrossRef]
- Schultze, J.L.; consortium, S.; Rosenstiel, P. Systems Medicine in Chronic Inflammatory Diseases. Immunity 2018, 48, 608–613. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA); Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; et al. Carbohydrate solutions and contribute to the improvement of physical performance during a high-intensity and long-lasting physical exercise: Evaluation of a health claim pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J. 2018, 16, e05191. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (EFSA NDA Panel); Turck, D.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Kearney, J.; Knutsen, H.K.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; et al. Guidance on the scientific requirements for health claims related to muscle function and physical performance: (Revision 1). EFSA J. 2018, 16, e05434. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Guidance on the scientific requirements for health claims related to appetite ratings, weight management, and blood glucose concentrations. EFSA J. 2012, 10. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Guidance on the scientific requirements for health claims related to bone, joints, skin, and oral health. EFSA J. 2012, 10. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (EFSA NDA Panel). Guidance on the scientific requirements for health claims related to antioxidants, oxidative damage and cardiovascular health. EFSA J. 2011, 9. [Google Scholar] [CrossRef]
- EFSA. EFSA Public Consultation on the Draft Guidance for the Scientific Requirements for Health Claims Related to Antioxidants, Oxidative Damage and Cardiovascular Health. 2017. Available online: https://www.efsa.europa.eu/en/consultations/call/170712 (accessed on 15 April 2021).
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Guidance on the scientific requirements for health claims related to functions of the nervous system, including psychological functions. EFSA J. 2012, 10. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Guidance on the scientific requirements for health claims related to the immune system, the gastrointestinal tract and defence against pathogenic microorganisms. EFSA J. 2016, 14. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA); Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; et al. Guidance for the scientific requirements for health claims related to antioxidants, oxidative damage and cardiovascular health: (Revision 1). EFSA J. 2018, 16, e05136. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Sadler, M.J. Authorised EU Health Claim for Vitamin D and Reduced Risk of Falls. In Foods, Nutrients and Food Ingredients with Authorised EU Health Claims; Woodhead Publishing: Duxford, UK, 2018; Volume 3, pp. 49–63. [Google Scholar]
- Ellins, E.A.; Halcox, J.P. Where are we heading with noninvasive clinical vascular physiology? Why and how should we assess endothelial function? Cardiol. Res. Pract. 2011, 2011, 870132. [Google Scholar] [CrossRef]
- Van Mil, A.; Pouwels, S.; Wilbrink, J.; Warle, M.C.; Thijssen, D.H.J. Carotid Artery Reactivity Predicts Events in Peripheral Arterial Disease Patients. Ann. Surg. 2019, 269, 767–773. [Google Scholar] [CrossRef]
- Covic, A.; Siriopol, D. Pulse wave velocity ratio: The new “gold standard” for measuring arterial stiffness. Hypertension 2015, 65, 289–290. [Google Scholar] [CrossRef] [PubMed]
- Struijker-Boudier, H.; Wilkinson, I.; Vlachopoulos, C.; Pannier, B.; Hayoz, D.; Giannattasio, C.; Boutouyrie, P.; Van Bortel, L.; Cockcroft, J.; Laurent, S. Abridged version of the expert consensus document on arterial stiffness☆. Artery Res. 2007, 1, 2–12. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Ikram, M.K.; Sabanayagam, C.; Wong, T.Y. Retinal microvasculature as a model to study the manifestations of hypertension. Hypertension 2012, 60, 1094–1103. [Google Scholar] [CrossRef]
- Camara, M.; Fernandez-Ruiz, V.; Sanchez-Mata, M.C.; Dominguez Diaz, L.; Kardinaal, A.; van Lieshout, M. Evidence of antiplatelet aggregation effects from the consumption of tomato products, according to EFSA health claim requirements. Crit. Rev. Food Sci. Nutr. 2020, 60, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Duttaroy, A.K. Regulation of functional foods in European Union: Assessment of health claim by the European food safety authority. In Nutraceutical and Functional Food Regulations in the United States and Around the World; Academic Press: Cambridge, MA, USA, 2019; pp. 267–276. [Google Scholar]
- Berretz, G.; Packheiser, J.; Kumsta, R.; Wolf, O.T.; Ocklenburg, S. The brain under stress-A systematic review and activation likelihood estimation meta-analysis of changes in BOLD signal associated with acute stress exposure. Neurosci. Biobehav. Rev. 2021, 124, 89–99. [Google Scholar] [CrossRef]
- Biasini, B.; Marchi, L.; Angelino, D.; Bedogni, G.; Zavaroni, I.; Pruneti, C.; Galli, D.; Mirandola, P.; Vitale, M.; Dei Cas, A.; et al. Claimed effects, outcome variables and methods of measurement for health claims on foods related to the gastrointestinal tract proposed under regulation (EC) 1924/2006. Int. J. Food Sci. Nutr. 2018, 69, 771–804. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of a health claim related to hydroxyanthracene derivatives and improvement of bowel function pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J. 2013, 11, 3412. [Google Scholar] [CrossRef]
- Van Trijp, M.P.H.; Schutte, S.; Esser, D.; Wopereis, S.; Hoevenaars, F.P.M.; Hooiveld, G.; Afman, L.A. Minor Changes in the Composition and Function of the Gut Microbiota During a 12-Week Whole Grain Wheat or Refined Wheat Intervention Correlate with Liver Fat in Overweight and Obese Adults. J. Nutr. 2021, 151, 491–502. [Google Scholar] [CrossRef]
- Van Wijck, K.; Lenaerts, K.; Grootjans, J.; Wijnands, K.A.; Poeze, M.; van Loon, L.J.; Dejong, C.H.; Buurman, W.A. Physiology and pathophysiology of splanchnic hypoperfusion and intestinal injury during exercise: Strategies for evaluation and prevention. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G155–G168. [Google Scholar] [CrossRef]
- Janssen Duijghuijsen, L.M.; van Norren, K.; Grefte, S.; Koppelman, S.J.; Lenaerts, K.; Keijer, J.; Witkamp, R.F.; Wichers, H.J. Endurance Exercise Increases Intestinal Uptake of the Peanut Allergen Ara h 6 after Peanut Consumption in Humans. Nutrients 2017, 9, 84. [Google Scholar] [CrossRef]
- Troost, F.J.; Saris, W.H.; Brummer, R.J. Recombinant human lactoferrin ingestion attenuates indomethacin-induced enteropathy in vivo in healthy volunteers. Eur. J. Clin. Nutr. 2003, 57, 1579–1585. [Google Scholar] [CrossRef]
- Albers, R.; Bourdet-Sicard, R.; Braun, D.; Calder, P.C.; Herz, U.; Lambert, C.; Lenoir-Wijnkoop, I.; Meheust, A.; Ouwehand, A.; Phothirath, P.; et al. Monitoring immune modulation by nutrition in the general population: Identifying and substantiating effects on human health. Br. J. Nutr. 2013, 110, S1–S30. [Google Scholar] [CrossRef]
- Van den Brink, W.; van Bilsen, J.; Salic, K.; Hoevenaars, F.P.M.; Verschuren, L.; Kleemann, R.; Bouwman, J.; Ronnett, G.V.; van Ommen, B.; Wopereis, S. Current and Future Nutritional Strategies to Modulate Inflammatory Dynamics in Metabolic Disorders. Front. Nutr. 2019, 6, 129. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Ten Bruggencate, S.J.M.; Schonewille, A.J.; Alhoniemi, E.; Forssten, S.D.; Bovee-Oudenhoven, I.M.J. Lactobacillus acidophilus supplementation in human subjects and their resistance to enterotoxigenic Escherichia coli infection. Br. J. Nutr. 2014, 111, 465–473. [Google Scholar] [CrossRef]
- Dillingh, M.R.; van Poelgeest, E.P.; Malone, K.E.; Kemper, E.M.; Stroes, E.S.G.; Moerland, M.; Burggraaf, J. Characterization of inflammation and immune cell modulation induced by low-dose LPS administration to healthy volunteers. J. Inflamm. 2014, 11, 28. [Google Scholar] [CrossRef]
- Carol, A.; Witkamp, R.F.; Wichers, H.J.; Mensink, M. Bovine colostrum supplementation’s lack of effect on immune variables during short-term intense exercise in well-trained athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 135–145. [Google Scholar] [CrossRef]
- Wyatt, P.; Berry, S.E.; Finlayson, G.; O’Driscoll, R.; Hadjigeorgiou, G.; Drew, D.A.; Khatib, H.A.; Nguyen, L.H.; Linenberg, I.; Chan, A.T.; et al. Postprandial glycaemic dips predict appetite and energy intake in healthy individuals. Nat. Metab. 2021, 3, 523–529. [Google Scholar] [CrossRef]
- Fiamoncini, J.; Rundle, M.; Gibbons, H.; Thomas, E.L.; Geillinger-Kastle, K.; Bunzel, D.; Trezzi, J.P.; Kiselova-Kaneva, Y.; Wopereis, S.; Wahrheit, J.; et al. Plasma metabolome analysis identifies distinct human metabotypes in the postprandial state with different susceptibility to weight loss-mediated metabolic improvements. FASEB J. 2018, 32, 5447–5458. [Google Scholar] [CrossRef]
- Wopereis, S.; Rubingh, C.M.; van Erk, M.J.; Verheij, E.R.; van Vliet, T.; Cnubben, N.H.; Smilde, A.K.; van der Greef, J.; van Ommen, B.; Hendriks, H.F. Metabolic profiling of the response to an oral glucose tolerance test detects subtle metabolic changes. PLoS ONE 2009, 4, e4525. [Google Scholar] [CrossRef]
- Tanemura, N.; Ito, K.; Hamadate, N.; Koshizaka, M.; Urushihara, H. Differences in efficacy evaluation endpoints in clinical trials for claiming reduction of post-prandial glycemic response between Japan and the European Union. Crit. Rev. Food Sci. Nutr. 2020, 60, 3589–3593. [Google Scholar] [CrossRef] [PubMed]
- Martini, D.; Galli, C.; Guareschi, C.; Angelino, D.; Bedogni, G.; Biasini, B.; Zavaroni, I.; Pruneti, C.; Ventura, M.; Galli, D.; et al. Claimed effects, outcome variables and methods of measurement for health claims on foods proposed under Regulation (EC) 1924/2006 in the area of oral health. NFS J. 2018, 10, 10–25. [Google Scholar] [CrossRef]
- Martini, D.; Angelino, D.; Cortelazzi, C.; Zavaroni, I.; Bedogni, G.; Musci, M.; Pruneti, C.; Passeri, G.; Ventura, M.; Galli, D.; et al. Claimed Effects, Outcome Variables and Methods of Measurement for Health Claims Proposed Under European Community Regulation 1924/2006 in the Framework of Maintenance of Skin Function. Nutrients 2018, 10, 7. [Google Scholar] [CrossRef]
- Martini, D.; Biasini, B.; Rossi, S.; Zavaroni, I.; Bedogni, G.; Musci, M.; Pruneti, C.; Passeri, G.; Ventura, M.; Galli, D.; et al. Claimed effects, outcome variables and methods of measurement for health claims on foods proposed under European Community Regulation 1924/2006 in the area of appetite ratings and weight management. Int. J. Food Sci. Nutr. 2018, 69, 389–409. [Google Scholar] [CrossRef]
- Bothwell, L.E.; Greene, J.A.; Podolsky, S.H.; Jones, D.S. Assessing the Gold Standard--Lessons from the History of RCTs. N. Engl. J. Med. 2016, 374, 2175–2181. [Google Scholar] [CrossRef]
- Penders, B.; Wolters, A.; Feskens, E.F.; Brouns, F.; Huber, M.; Maeckelberghe, E.L.M.; Navis, G.; Ockhuizen, T.; Plat, J.; Sikkema, J.; et al. Capable and credible? Challenging nutrition science. Eur. J. Nutr. 2017, 56, 2009–2012. [Google Scholar] [CrossRef]
- Weaver, C.M.; Miller, J.W. Challenges in conducting clinical nutrition research. Nutr. Rev. 2017, 75, 491–499. [Google Scholar] [CrossRef]
- Heaney, R.P. Nutrients, endpoints, and the problem of proof. J. Nutr. 2008, 138, 1591–1595. [Google Scholar] [CrossRef]
- Blumberg, J.; Heaney, R.P.; Huncharek, M.; Scholl, T.; Stampfer, M.; Vieth, R.; Weaver, C.M.; Zeisel, S.H. Evidence-based criteria in the nutritional context. Nutr. Rev. 2010, 68, 478–484. [Google Scholar] [CrossRef]
- Biesalski, H.K.; Aggett, P.J.; Anton, R.; Bernstein, P.S.; Blumberg, J.; Heaney, R.P.; Henry, J.; Nolan, J.M.; Richardson, D.P.; van Ommen, B.; et al. 26th Hohenheim Consensus Conference, September 11, 2010 Scientific substantiation of health claims: Evidence-based nutrition. Nutrition 2011, 27, S1–S20. [Google Scholar] [CrossRef]
- Tufford, A.R.; Calder, P.C.; Van’t Veer, P.; Feskens, E.F.; Ockhuizen, T.; Kraneveld, A.D.; Sikkema, J.; de Vries, J. Is nutrition science ready for the twenty-first century? Moving towards transdisciplinary impacts in a changing world. Eur. J. Nutr. 2020, 59, 1–10. [Google Scholar] [CrossRef]
- Verjans-Janssen, S.R.B.; Van Kann, D.H.H.; Gerards, S.; Vos, S.B.; Jansen, M.W.J.; Kremers, S.P.J. Study protocol of the quasi-experimental evaluation of “KEIGAAF”: A context-based physical activity and nutrition intervention for primary school children. BMC Public Health 2018, 18, 842. [Google Scholar] [CrossRef]
- Abidi, L.; Nagelhout, G.E.; Spruijt, R.; Schutte, H.; De Vries, H. A quasi-experimental study evaluating a health promotion program targeting healthy nutrition, physical activity and social network enhancement for low-income multi-problem households: Study protocol. Int. J. Clin. Trials 2018, 5, 132. [Google Scholar] [CrossRef]
- Walach, H.; Falkenberg, T.; Fonnebo, V.; Lewith, G.; Jonas, W.B. Circular instead of hierarchical: Methodological principles for the evaluation of complex interventions. BMC Med. Res. Methodol. 2006, 6, 29. [Google Scholar] [CrossRef]
- Barker, D.; McElduff, P.; D’Este, C.; Campbell, M.J. Stepped wedge cluster randomised trials: A review of the statistical methodology used and available. BMC Med. Res. Methodol. 2016, 16, 69. [Google Scholar] [CrossRef]
- Pot, G.K.; Battjes-Fries, M.C.; Patijn, O.N.; van der Zijl, N.; Pijl, H.; Voshol, P. Lifestyle medicine for type 2 diabetes: Practice-based evidence for long-term efficacy of a multicomponent lifestyle intervention (Reverse Diabetes2 Now). BMJ Nutr. Prev. Health 2020. [Google Scholar] [CrossRef] [PubMed]
- Mirza, R.D.; Punja, S.; Vohra, S.; Guyatt, G. The history and development of N-of-1 trials. J. R. Soc. Med. 2017, 110, 330–340. [Google Scholar] [CrossRef]
- Stunnenberg, B.C.; Deinum, J.; Nijenhuis, T.; Huysmans, F.; van der Wilt, G.J.; van Engelen, B.G.M.; van Agt, F. N-of-1 Trials: Evidence-Based Clinical Care or Medical Research that Requires IRB Approval? A Practical Flowchart Based on an Ethical Framework. Healthcare 2020, 8, 49. [Google Scholar] [CrossRef]
- Duan, N.; Eslick, I.; Gabler, N.; Kaplan, H.; Kravitz, R.; Larson, E.; Pace, W.; Schmid, C.; Sim, I.; Vohra, S. Design and implementation of N-of-1 trials: A user’s guide. Agency Health Care Res. Qual. 2014, 110, 330–340. [Google Scholar]
- Duan, N.; Kravitz, R.L.; Schmid, C.H. Single-patient (n-of-1) trials: A pragmatic clinical decision methodology for patient-centered comparative effectiveness research. J. Clin. Epidemiol. 2013, 66, S21–S28. [Google Scholar] [CrossRef]
- Krone, T.; Boessen, R.; Bijlsma, S.; van Stokkum, R.; Clabbers, N.D.S.; Pasman, W.J. The possibilities of the use of N-of-1 and do-it-yourself trials in nutritional research. PLoS ONE 2020, 15, e0232680. [Google Scholar] [CrossRef] [PubMed]
- Potter, T.; Vieira, R.; de Roos, B. Perspective: Application of N-of-1 Methods in Personalized Nutrition Research. Adv. Nutr. 2021, 12, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Soldevila-Domenech, N.; Boronat, A.; Langohr, K.; de la Torre, R. N-of-1 Clinical Trials in Nutritional Interventions Directed at Improving Cognitive Function. Front. Nutr. 2019, 6, 110. [Google Scholar] [CrossRef] [PubMed]
- Berry, S.E.; Valdes, A.M.; Drew, D.A.; Asnicar, F.; Mazidi, M.; Wolf, J.; Capdevila, J.; Hadjigeorgiou, G.; Davies, R.; Al Khatib, H.; et al. Human postprandial responses to food and potential for precision nutrition. Nat. Med. 2020, 26, 964–973. [Google Scholar] [CrossRef]
- Bianchi, M.T. Sleep devices: Wearables and nearables, informational and interventional, consumer and clinical. Metabolism 2018, 84, 99–108. [Google Scholar] [CrossRef]
- Kim, J.; Campbell, A.S.; de Avila, B.E.; Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 2019, 37, 389–406. [Google Scholar] [CrossRef]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M.; et al. Personalized Nutrition by Prediction of Glycemic Responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef]
| Health Domain | Study Set-Up |
|---|---|
| Bone and joint health | Maintenance of bone mass or bone mineral density [111]. |
| Maintenance of joint function [111]. | |
| Reduced falls and fractures [117]. | |
| Cardiovascular health | Beneficial changes in the blood lipid profile [113]. |
| Reduction in arterial (systolic) blood pressure (SBP) [113]. | |
| Flow mediated dilatation [118]. | |
| Carotid-artery reactivity (CAR) measurement. Effect of a cold pressor test (CPT) [119]. | |
| Arterial stiffness via carotid-to-femoral pulse wave velocity (PWVc-f) [120]. | |
| Arterial stiffness via the augmentation index (AIx) [121]. | |
| Retinal microvascular structure [122]. | |
| Decreased platelet aggregation [123,124]. | |
| Maintenance of normal blood homocysteine concentrations [113]. | |
| Cognitive performance, stress, psychological functions and other CNS domains | Improvement or maintenance of cognitive functions [114]. |
| Improvement of alertness and/or attention [114]. | |
| Improvement of mood/affect [114]. | |
| Psychological stress tests [114,125]. | |
| Anxiety [114]. | |
| Improvement or maintenance of vision [114]. | |
| Improvement of sleep [114]. | |
| Gastro-intestinal functionality | Breath hydrogen levels, gas volume assessed by imaging (i.e., MRI). [115,126]. |
| Transit time, frequency of bowel movements, stool bulk [115,126,127]. | |
| Validated subjective global symptom severity questionnaires [115,126]. | |
| (Changes in) microbiota composition of the gut accompanied by evidence of a beneficial physiological effect or clinical outcome and/or including pathogenic and toxicogenic microorganisms [115,126,128]. | |
| Changes in short chain fatty acid production in the gut [115]. | |
| Changes in digestion or (and) absorption [115,126]. | |
| Changes in structure of intestinal epithelium [115]. | |
| Changes in barrier function, using physical exercise challenge [129,130] or NSAIDs [131]. | |
| Immune function and defense against pathogens | Changes in immune markers, e.g., numbers of various lymphoid subpopulations in the Circulation * [115,132]. |
| Changes in markers of inflammation 1 [115,132]. | |
| Metabolic challenge tests 2 [132,133]. | |
| Immune-training effects [64]. | |
| An increase in the number of responders to vaccination [115]. | |
| Microbiological data at specific sites [115]. | |
| Improved protection of groups at increased risk [115]. | |
| Beneficial response to allergens [115]. | |
| Response to experimental infections [100,115,132,134]. | |
| LPS challenge [135]. | |
| Response to exercise challenge (indirectly) 2 [136]. | |
| Metabolic health | Metabolic challenge tests, including mixed meal tests and post-prandial glycemic responses [96,97,116,133,137,138,139,140]. |
| Protection against oxidative damage (DNA, proteins, Lipids) and DNA breaks [116]. | |
| Oral health | Saliva flow or measurement of self-perceived oral dryness by validated questionnaires. |
| Maintenance of gum function [111,141]. | |
| Reduction of dental plaque, acid production and/or dental calculus [111,141]. | |
| Maintenance of tooth mineralization [111,141]. | |
| Reduction of oral dryness [111,141]. | |
| Specific colonisation with Streptococcus mutans, decrease of caries [111,141]. | |
| Physical performance | Improvement of physical performance (the ability to complete certain physical tasks) at certain (high) intensity and with a certain (long) duration in a physical exercise trial [108,109]. |
| Physical capacity (exercise time to fatigue at predefined conditions) [109]. | |
| Muscle function (i.e., change in muscle structure) 3 [109]. | |
| Skin health | Trans epidermal water loss [142]. |
| Skin Hydration [142]. | |
| Skin Dryness [142]. | |
| Skin Elasticity [142]. | |
| Corneocyte Adhesion [142]. | |
| Oxidative Damage to Lipids [142]. | |
| Protection of the skin against oxidative (including UV-induced) damage [111,142]. | |
| Protection of the skin from UV-induced (other than oxidative) damage [111,142]. | |
| Weight management | Appetite ratings [110,143]. |
| Behavioral assessment (energy intake etc) 4 [110,143]. | |
| Body fat (different methods of assessment) and lean body mass (idem) [110,143]. | |
| Bodyweight regain/maintenance (prolonged time period, 6 months) [110,143]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Witkamp, R.F. Nutrition to Optimise Human Health—How to Obtain Physiological Substantiation? Nutrients 2021, 13, 2155. https://doi.org/10.3390/nu13072155
Witkamp RF. Nutrition to Optimise Human Health—How to Obtain Physiological Substantiation? Nutrients. 2021; 13(7):2155. https://doi.org/10.3390/nu13072155
Chicago/Turabian StyleWitkamp, Renger F. 2021. "Nutrition to Optimise Human Health—How to Obtain Physiological Substantiation?" Nutrients 13, no. 7: 2155. https://doi.org/10.3390/nu13072155
APA StyleWitkamp, R. F. (2021). Nutrition to Optimise Human Health—How to Obtain Physiological Substantiation? Nutrients, 13(7), 2155. https://doi.org/10.3390/nu13072155