Omega-3 Supplementation and Heart Disease: A Population-Based Diet by Gene Analysis of Clinical Trial Outcomes
Abstract
:1. Introduction
2. Alterations in Dietary Fatty Acids and Health Consequences
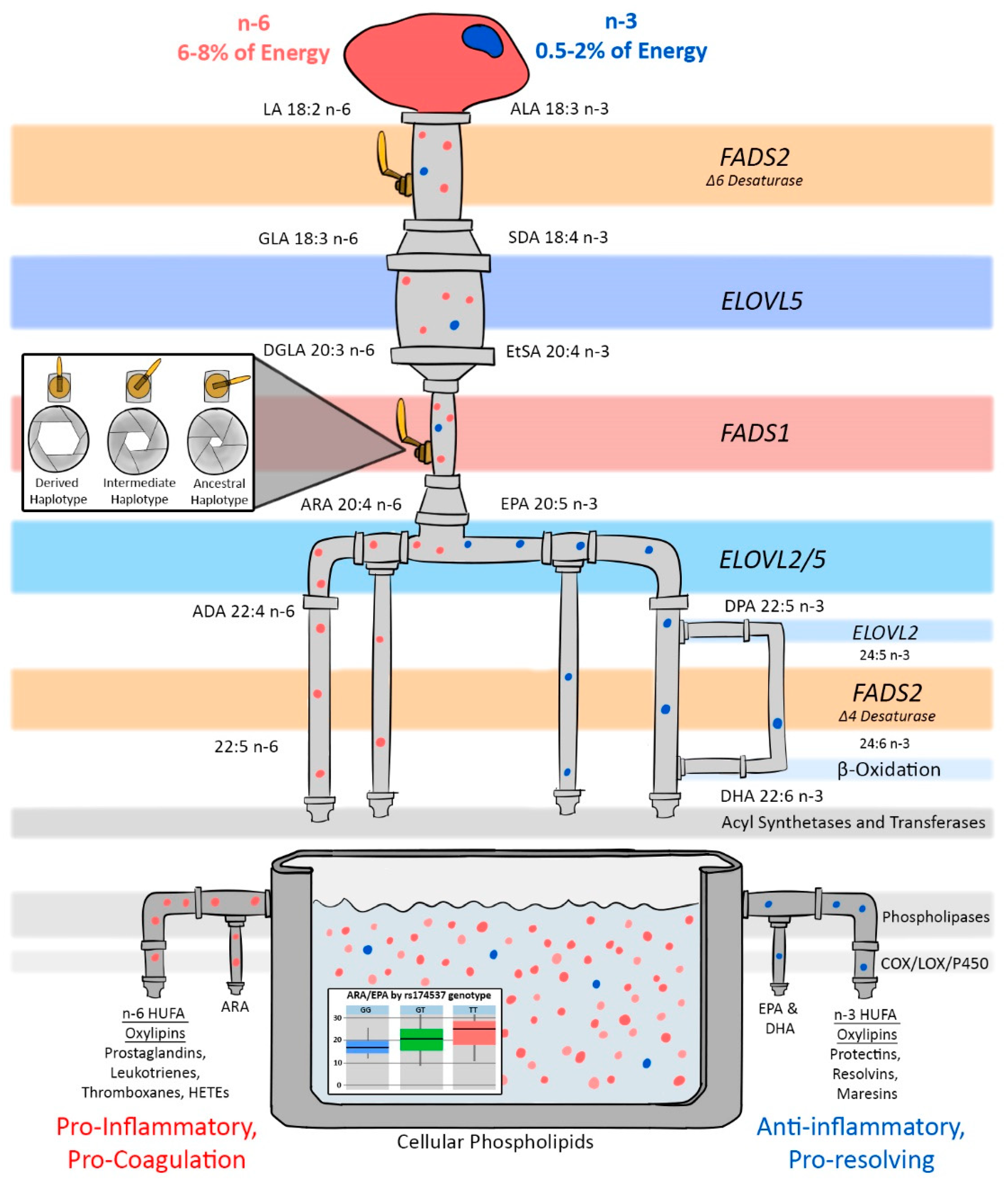
3. Genetic Variation in LC-PUFA Biosynthesis
4. N-3 LC-PUFA and Cardiovascular Risk
5. Summary and Future Recommendations
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Blasbalg, T.L.; Hibbeln, J.-R.; Ramsden, C.E.; Mahchrzak, S.F.; Rawlings, R.R. Changes in the consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century. Am. J. Clin. Nutr. 2011, 93, 950–962. [Google Scholar] [CrossRef] [Green Version]
- Spector, A.A. Plasma free fatty acid and lipoproteins as sources of polyunsaturated fatty acid for the brain. J. Mol. Neurosci. 2001, 16, 159–165. [Google Scholar] [CrossRef]
- Smith, W.L. The eicosanoids and their biochemical mechanisms of action. Biochem. J. 1989, 259, 315–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, J.A.; Kirby, S.S. Eicosanoids, prostacyclin and cyclooxygenase in the cardiovascular system. Br. J. Pharmacol. 2019, 176, 1038–1050. [Google Scholar]
- Schmitz, G.; Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Linderberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet. Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Chilton, F.H.; Murphy, R.C.; Wilson, B.A.; Sergeant, S.; Ainsworth, H.; Seeds, M.C.; Mathias, R.A. Diet-gene interactions and PUFA metabolism. A potential contributor to health disparities and human diseases. Nutrients 2014, 6, 1993–2022. [Google Scholar] [CrossRef] [Green Version]
- Kromhout, D.; de Goede, J. Update on cardiometabolic health effects of ω-3 fatty acids. Curr. Opin. Lipidol. 2014, 25, 85–90. [Google Scholar] [CrossRef]
- Lee, S.; Gura, K.M.; Kim, S.; Arsenault, D.A.; Bistrian, B.R.; Puder, M. Current clinical applications of omega-6 and omega-3 fatty acids. Nutr. Clin. Pract. 2006, 21, 323–341. [Google Scholar] [CrossRef] [Green Version]
- Sheikh, O.; Vande Hei, A.G.; Battisha, A.; Hammad, T.; Pham, S.; Chilton, R. Cardiovascular, electrophysiologic, and hematologic effects of omega-3 fatty acids beyond reducing hypertriglyceridemia: As it pertains to the recently published REDUCE-IT trial. Cardiovasc. Diabetol. 2019, 18, 84. [Google Scholar] [CrossRef] [Green Version]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimt. Biophys. Acta—Mol. Cell. Biol. Lipids 2015, 1851, 469–484. [Google Scholar] [CrossRef]
- Calviello, G.; Serini, S.; Piccioni, E. Alzheimer’s disease and n-3 polyunsaturated fatty acids: Beneficial effects and possible molecular pathways involved. Curr. Signal Transduct. Ther. 2008, 3, 152–157. [Google Scholar] [CrossRef]
- Bozzatello, P.; Rocca, P.; Mantelli, E.; Bellino, S. Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders? Int. J. Mol. Sci. 2019, 20, 5257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calviello, G.; Serini, S.; Piccioni, E. n-3 polyunsaturated fatty acids and the prevention of colorectal cancer: Molecular mechanisms involved. Curr. Med. Chem. 2007, 14, 3059–3069. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Chen, S.H.; Chen, Y.Q. Mechanisms of omega-3 polyunsaturated fatty acids in prostate cancer prevention. BioMed. Res. Intern. 2013, 2013, 824563. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Bi, X.; Wang, S.; Zhang, Z.; Li, F.; Zhao, A.Z. Therapeutic Potential of ω-3 Polyunsaturated Fatty Acids in Human Autoimmune Diseases. Front Immunol. 2019, 10, 2241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Globbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wenberg, M.; Yakoob, M.Y.; Chiuve, S.E.; della Cruz, L.; Frazier-Wood, A.C.; et al. Omega-3 polyunsaturated fatty acid biomarkers and coronary heart disease: Pooling project of 19 cohort studies. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef] [Green Version]
- Harris, W.S.; Tintle, N.L.; Etherton, M.R.; Vasan, R.S. Erythrocyte long-chain omega-3 fatty acid levels are inversely associated with mortality and with incident cardiovascular disease: The Framingham Heart Study. J. Clin. Lipidol. 2018, 12, 718–727.e6. [Google Scholar] [CrossRef] [Green Version]
- Glen, A.I.; Glen, E.M.; Horrobin, D.F.; Vaddadi, K.S.; Spellman, M.; Morse-Fisher, N.; Ellis, K.; Skinner, F.S. A red cell membrane abnormality in a subgroup of schizophrenic patients: Evidence for two diseases. Schizophr. Res. 1994, 12, 53–61. [Google Scholar] [CrossRef]
- Reddy, R.D.; Keshavan, M.S.; Yao, J.K. Reduced red blood cell membrane essential polyunsaturated fatty acids in first episode schizophrenia at neuroleptic-naive baseline. Schizophr. Bull. 2004, 30, 901–911. [Google Scholar] [CrossRef] [Green Version]
- Asher, A.; Tintle, N.C.; Myers, M.; Lockshon, L.; Barraza, H.; Harris, W.S. Blood omega-3 fatty acids and death from COVID-19: A pilot study. Prostaglandins Leukot. Essent. Fatty Acids 2021, 166, 102250. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, R.J.; Lee, I.-M.; Christen, W.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Marine Omega-3 Fatty Acids and Prevention of Cardiovascular Disease and Cancer. N. Engl. J. Med. 2019, 380, 23–32. [Google Scholar] [CrossRef] [PubMed]
- ORIGIN Trial Investigators. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N. Eng. J. Med. 2012, 367, 309–318. [Google Scholar] [CrossRef] [Green Version]
- Aung, T.; Halsey, J.; Kromhout, D.; Gerstein, H.C.; Marchioli, R.; Tavazzi, L.; Geleijnse, J.M.; Rauch, B.; Ness, A.; Galan, P.; et al. Associations of Omega-3 Fatty Acid Supplement Use With cardiovascular Disease Risks. meta-analysis of 10 trials involving 77 917 individuals. JAMA Cardiol. 2018, 3, 14–22. [Google Scholar] [CrossRef] [Green Version]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; Abdulghafoor, R.K.; Summerbell, C.D.; Worthington, H.V.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, 2018, CD003177. [Google Scholar]
- Bhatt, D.P.; Steg, G.; Miller, M.; Brinton, E.A.; Jacobson, R.A.; Ketchum, S.B.; Doyle, R.T.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Eng. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Stegh, G.; Ketchum, S.B.; Doyler, R.T.; Jualiano, R.A.; Jia, L.; Granowitz, C.; et al. Results from the 3416 Patients randomized in the United States. Circulation 2020, 141, 367–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kris-Etherton, P.M.; Richter, C.K.; Bowen, K.J.; Skulas-Ray, A.C.; Jackson, K.H.; Petersen, K.S.; Harris, W.S. Recent Clinical Trials Shed New Light on the Cardiovascular Benefits of Omega-3 Fatty Acids. Methodist Debakey Cardiovasc. J. 2019, 15, 171–178. [Google Scholar] [PubMed]
- Chilton, F.H.; Dutta, R.; Reynolds, L.M.; Sergeant, S.; Mathias, R.A.; Seeds, M.C. Precision nutrition and Omega-3 Polyunsaturated fatty acids: A case of personalized supplementation approaches for the prevention and management of human diseases. Nutrients 2017, 9, 1165. [Google Scholar] [CrossRef]
- Allison, D.B.; Fontain, K.R.; Manson, J.E.; Severns, J.; Van Itallie, T.B. Annual deaths atrributable to obesity in the United States. JAMA 1999, 282, 1530–1538. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2018, 10, 1069–1091. [Google Scholar] [CrossRef]
- Oparil, S.; Acelajado, M.C.; Bakris, F.L.; Berlowitz, D.R.; Cífková, R.; Dominiczak, A.F.; Grassi, G.; Jordan, J.; Poulter, N.R.; Rodgers, A.; et al. Hypertension. Nat. Rev. Dis. Primers. 2014, 63, 451–458. [Google Scholar]
- Neuenschwander, M.; Ballon, A.; Weber, K.S.; Norat, T.; Aune, D.; Schwingshackl, L.; Schlesinger, S. Role of diet in type 2 diabetes incidence: Umbrella review of meta-analyses of prospective observational studies. BMJ 2019, 366, l2368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cordain, L.; Watkins, B.A.; Florant, G.L.; Kehler, M.; Rogers, L.; Li, Y. Fatty acid analysis of wild ruminant tissues: Evolutionary implications for reducing diet-related chronic disease. Eur. J. Clin. Nutr. 2002, 56, 181–191. [Google Scholar] [CrossRef] [Green Version]
- Popkin, B.M. Global nutrition dynamics: The world is shifting rapidly toward a diet linked with noncommunicable diseases. Am. J. Clin. Nutr. 2006, 84, 289–298. [Google Scholar] [CrossRef] [PubMed]
- USDA Dietary Guidelines. Available online: https://www.dietaryguidelines.gov/ (accessed on 20 February 2021).
- American Heart Association, Dietary Guidelines. Available online: https://www.heart.org/en/healthy-living/healthy-eating/eat-smart/nutrition-basics (accessed on 25 April 2021).
- The American Cancer Society. Available online: https://www.cancer.org/healthy/eat-healthy-get-active/acs-guidelines-nutrition-physical-activity-cancer-prevention.html (accessed on 25 April 2021).
- De Toro-Martin, J.; Arsenault, B.J.; Despres, J.P.; Vohl, M.C. Precision Nutrition: A review of personalized. nutritional approaches for the prevention and management of metabolic syndrome. Nutrients 2017, 9, 913. [Google Scholar] [CrossRef] [Green Version]
- Grundy, S.M.; Greenland, P.; Herd, A.; Huebsch, J.A.; Jones, R.J.; Mitchell, J.H.; Schlant, R.C. Cardiovascular and risk factor evaluation of healthy American adults. A statement for physicians by an Ad Hoc Committee appointed by the Steering Committee, American Heart Association. Circulation 1987, 75, 1340A–1362A. [Google Scholar]
- Upadhyay, R.K. Emerging Risk Biomarkers in Cardiovascular Diseases and Disorders. J. Lipids. 2015, 2015, 971453. [Google Scholar] [CrossRef]
- Tenenbaum, A.; Klempfner, R.; Fisman, E.Z. Hypertriglyceridemia: A too long unfairly neglected major cardiovascular risk factor. J. Lipid Res. 2018, 59, 1266–1275. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An Increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Innis, S.M. Omega-3 fatty acid biochemistry perspectives from human nutrition. Mil. Med. 2014, 179 (Suppl. 11), 82–87. [Google Scholar] [CrossRef] [Green Version]
- Sebastiani, G.; Barbero, A.H.; Borrás-Novell, C.; Casanova, M.A.; Aldecoa-Bilbao, V.; Andreu-Fernández, V.; Tutusaus, M.P.; Ferrero Martínez, S.; Gómez Roig, M.D.; García-Algar, O. The Effects of vegetarian and vegan diet during pregnancy on the health of mothers and offspring. Nutrients 2019, 11, 557. [Google Scholar] [CrossRef] [Green Version]
- MacIntosh, B.A.; Ramsden, C.E.; Faurot, K.R.; Zamora, D.; Mangan, M.; Hibbeln, J.R.; Mann, J.D. n-6 fatty acid-specific and mixed polyunsaturated dietary interventions have different effects on CHD risk: A meta-analysis of randomized controlled trials. Br. J. Nutr. 2010, 104, 1586–1600. [Google Scholar]
- Ramsden, C.E.; Zamora, D.; Majchrzak-Hong, S.; Faurot, K.R.; Broste, S.K.; Frantz, R.P.; Davis, J.M.; Ringel, A.; Suchindran, C.M.; Hibbeln, J.R. Re-evaluation of the traditional diet-heart hypothesis: Analysis of recovered data from Minnesota Coronary Experiment (1968–73). BMJ 2016, 353, i1246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lands, B. Dietary omega-3 and omega-6 fatty acids compete in producing tissue compositions and tissue responses. Mil. Med. 2014, 179 (Suppl. 11), 76–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, K.E.; Mantzioris, E.; Gibson, R.A.; Ramsden, C.E.; Muhkhausler, B.S. The effect of modifying dietary LA and ALA intakes on omega-3 long chain polyunsaturated fatty acid (n-3 LCPUFA) status in human adults: A systematic review and commentary. Prostaglandins Leukot. Essent. Fatty Acids 2015, 95, 47–55. [Google Scholar] [CrossRef] [Green Version]
- MacIntosh, B.A.; Ramsden, C.E.; Faurot, K.R.; Zamora, D.; Mangan, M.; Hibbeln, J.R.; Mann, D. Low-n-6 and low-n-6 plus high-n-3 diets for use in clinical research. Br. J. Nutr. 2013, 110, 559–568. [Google Scholar] [CrossRef] [Green Version]
- Mathias, R.A.; Pani, V.; Chilton, F.H. Genetic variance in the FADS gene. Implications for dietary recommendations for fatty acid intake. Curr. Nutr. Rep. 2014, 3, 139–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, D.N.; Rucsink, I.; Yanek, L.R.; Becker, L.C.; Becker, D.M.; Guio, H.; Cui, T.; Chilton, F.H.; Mathias, R.A.; O’Connor, T. Evolution of hominin polyunsaturated fatty acid metabolism: From Africa to the New World. Genome Biol. Evol. 2019, 11, 1417–1430. [Google Scholar] [CrossRef] [Green Version]
- Sergeant, S.; Hugenschmidt, C.E.; Rudock, M.; Ziegler, J.T.; Ivester, P.; Ainsworth, H.C.; Vaidya, D.; Case, L.D.; Langfeld, C.D.; Freedman, B.L.; et al. Differences in arachidonic acid levels and fatty acid desaturase gene variants in African Americans and European Americans with Diabetes/Metabolic syndrome. Br. J. Nutr. 2012, 107, 547–555. [Google Scholar] [CrossRef] [Green Version]
- Ameur, A.; Enroth, S.; Johansson, A.; Zaboli, G.; Igl, W.; Johansson, A.C.V.; Rivas, M.A. Genetic adaptation of fatty-acid metabolism: A human-specific haplotype increasing the biosynthesis of long-chain omega-3 and omega-6 fatty acids. Am. J. Hum. Genet. 2012, 90, 809–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathias, R.A.; Sergeant, S.; Ruczinski, I.; Torgerson, D.G.; Hugenschmidt, C.E.; Kubala, M.; Vaidya, D.; Suktitipat, B.; Ziegler, J.T.; Ivester, P.; et al. The impact of FADS genetic variants on omega6 polyunsaturated fatty acid metabolism in African Americans. BMC Genet. 2011, 12, 50. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Hallmark, B.; Chai, J.C.; O’Connor, T.D.; Reynolds, L.M.; Wood, A.C.; Seeds, M.; Chen, Y.-D.I.; Steffen, L.M.; Tsai, M.Y.; et al. Amerind ancestry predicts the impact of FADS genetic variation on omega-3 PUFA deficiency, cardiometabolic and inflammatory risk in Hispanic populations. MedRxiv 2021. [Google Scholar] [CrossRef]
- Sergeant, S.; Hallmark, B.; Mathias, R.A.; Mustin, T.L.; Ivester, P.; Bohannon, M.; Ruczinski, I.; Johnstone, L.; Seeds, M.; Chilton, F.H. Prospective clinical trial examining the impact of genetic variation in FADS1 on the metabolism of linoleic acid- and ɣ-linolenic acid-containing botanical oils. Am. J. Clin. Nutr. 2020, 111, 1068–1078. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, L.M.; Durra, R.; Seed, M.C.; Lake, K.N.; Hallmark, B.; Mathias, R.A.; Howard, T.D.; Chilton, F.H. FADS genetic and metabolomic analyses identify the ∆5 desaturase (FADS1) step as a critical control point in the formation of biologically important lipids. Sci. Rep. 2020, 10, 15873. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, H.; Kobayashi, T.; Watanabe, S. Dietary fatty acids—The N-6/N-3 balance and chronic elderly diseases. Excess linoleic acid and relative N-3 deficiency syndrome seen in Japan. Prog. Lipid Res. 1996, 35, 409–457. [Google Scholar] [CrossRef]
- Kothapallia, K.S.D.; Parka, H.G.; Brenna, J.T. Polyunsaturated fatty acid biosynthesis pathway and genetics. implications for interindividual variability in prothrombotic, inflammatory conditions such as COVID-19. Prost. Leukot. Essent. Fatty Acids 2020, 162, 102183. [Google Scholar] [CrossRef]
- Cormier, H.; Rudkowska, I.; Lemieux, S.; Couture, P.; Julien, P.; Vohl, M.-C. Effects of FADS and ELOVL polymorphisms on indexes of desaturase and elongase activities: Results from a pre-post fish oil supplemenatation. Genet. Nutr. 2014, 9, 437. [Google Scholar] [CrossRef] [Green Version]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef]
- Filion, K.B.; El Khoury, F.; Bielinski, M.; Schiller, I.; Dendukuri, N.; Brophy, J.M. Omega-3 fatty acids in high-risk cardiovascular patients: A meta-analysis of randomized controlled trials. BMC Cardiovasc. Disord. 2010, 10, 24. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Chen, L.-Q.; Xiao, T.-H.; Zhang, Y.-C.; Zhu, M.; Zhang, R.; Li, K.; Wang, Y.; Li, Y. Effects of omega-3 fatty acid for sudden cardiac death prevention in patients with cardiovascular disease: A contemporary meta-analysis of randomized, controlled trials. Cardiovasc. Drugs Ther. 2011, 25, 259–265. [Google Scholar] [CrossRef]
- Zhao, Y.T.; Chen, Q.; Sum, Y.-X.; Li, X.-B.; Zhang, P.; Xu, Y.; Guo, J.-H. Prevention of sudden cardiac death with omega-3 fatty acids in patients with coronary heart disease: A meta-analysis of randomized controlled trials. Ann. Med. 2009, 41, 301–310. [Google Scholar] [CrossRef]
- World Heatlh Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death# (accessed on 20 February 2021).
- Steven, S.; Frenis, K.; Oelze, M.; Kalinovic, S.; Kuntic, M.; Bayo Jimenez, M.T.; Vujacic-Mirski, K.; Helmstädter, J.; Kröller-Schön, S.; Münzel, T.; et al. Vascular Inflammation and Oxidative Stress: Major Triggers for Cardiovascular Disease. J. Am. Heart Assoc. 2018, 7, e007809. [Google Scholar] [CrossRef] [Green Version]
- Van Dyke, M.; Greer, S.; Odom, E.; Schieb, L.; Vaughan, A.; Kramer, M.; Casper, M. Heart Disease Death Rates Among Blacks and Whites Aged ≥35 Years—United States, 1968–2015. MMWR Surveill. Summ. 2018, 67, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turkson-Ocran, R.-A.N.; Nmezi, N.A.; Botchway, M.O.; Szanton, S.L.; Golden, S.H.; Cooper, L.A.; Commodore-Mensah, Y. Comparison of cardiovascular disease risk factors among African immigrants and African Americans: An analysis of the 2010 to 2016 National Health Interview Surveys. J. Am. Heart Assoc. 2020, 9, e013220. [Google Scholar] [CrossRef] [PubMed]
- Burns, S.F.; Lee, S.J.; Arslanian, S.A. In vivo insulin sensitivity and lipoprotein particle size and concentration in black and white children. Diabetes Care 2009, 32, 2087–2093. [Google Scholar] [CrossRef] [Green Version]
- Zilbermint, M.; Hannah-Shmouni, F.; Stratakis, C.A. Genetics of hypertension in African Americans and others of African descent. Int. J. Mol. Sci. 2019, 20, 1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khera, A.; McGuire, D.K.; Murphy, S.A. Race and gender differences in C-reactive protein levels. J. Am. Coll. Cardiol. 2005, 46, 464–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albert, M.A.; Glynn, R.J.; Buring, J. C -Reactive protein levels among women of various ethnic groups living in the United States (from the Women’s Health Study). Am. J. Cardiol. 2004, 93, 1238–1242. [Google Scholar] [CrossRef]
- U.S. Census-Bureau. Profile America Facts for Features: Hispanic Heritage Month 2015; Department of Commerce, Economics and Statistics Administration: Washington, DC, USA, 2015; pp. 1–6. [Google Scholar]
- Bryc, K.; Durand, E.Y.; Macpherson, J.M.; Reich, D.; Mountain, J.L. The Genetic Ancestry of African Americans, Latinos, and European Americans across the United States. Am. J. Hum. Gen. 2015, 96, 37–53. [Google Scholar] [CrossRef] [Green Version]
- Fleischman, M.W.; Budoff, M.; Zeb, I.; Li, D.; Foste, T. NAFLD prevalence differs among hispanic subgroups: The Multi-Ethnic Study of Atherosclerosis. World J. Gastroenterol. 2014, 20, 4987–4999. [Google Scholar] [CrossRef] [PubMed]
- Howard, B.V.; Lee, E.T.; Cowan, L.D.; Deveraux, R.B.; Galloway, J.M.; Gom, O.T.; Howard, W.J.; Rhoades, E.R.; Robbins, D.C.; Sievers, M.L.; et al. Rising tide of cardiovascular disease in American Indians. The Strong Heart Study. Circulation 1999, 99, 2389–2395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Zhang, Y.; Lee, E.T.; Howard, B.V.; Deveraux, R.B.; Cole, S.A.; Best, L.G. Risk Factors and Prediction of Stroke in a Population with High Prevalence of Diabetes: The Strong Heart Study. World J. Cardiovasc. Dis. 2017, 7, 145–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Vital Statistics System, LCWK1_2013. In Deaths, 15 Leading Causes of Death by Race; U.S. Department of Health & Human Services: Washington, DC, USA, 2013; p. 100.
- Bang, H.O.; Dyerberg, J.; Nielsen, B. Plasma lipid and lipoprotein pattern in greenlandic west-coast Eskimos. Lancet 1971, 7710, 1143–1146. [Google Scholar] [CrossRef]
- Dyerberg, J.; Bang, H.O. Hæmostatic function and platelet polyunsaturated fatty acids in Eskimos. Lancet 1979, 8140, 433–435. [Google Scholar] [CrossRef]
- Hutchinson, A.; Tingö, L.; Brummer, R.J. The potential effects of probiotics and ω-3 fatty acids on chronic low-grade inflammation. Nutrients 2020, 12, 2402. [Google Scholar] [CrossRef] [PubMed]
- Harris, N.S.; Tintle, N.L.; Imamura, F.; Qian, F.; Ardisson Korat, A.V.; Marklund, M.; D’Jousee, L.; Bassett, J.K. Blood n-3 fatty acid levels and total and cause-specific mortality from 17 prospective studies. Nat. Commun. 2021, 12, 1–9. [Google Scholar] [CrossRef]
- GISSI-Prevenzione Trial. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E. after myocardial infarction: Results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’infarto miocardico. Lancet 1999, 354, 447–455. [Google Scholar] [CrossRef]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R. GISSI-HF Investigators. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsusawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomized open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M. Alpha Omega Trial Group. N-3 fatty. acids and cardiovascular events after myocardial infarction. N. Eng. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef]
- Rauch, B.; Schiele, R.; Schneider, S.; Diller, F.; Victor, N.; Gohlke, H.; Gottwik, M.; Steinbeck, G.; del Castillo, U.; Sack, R.; et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010, 122, 2152–2159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S.; SU. FOL.OM3 Collaborative Group. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 29, 341-c6273. [Google Scholar]
- Writing Group for the AREDS2 Research Group; Bonds, D.E.; Harrington, M.; Worral, B.B.; Bertoni, A.G.; Eaton, C.B.; Hsia, J.; Robinson, J.; Clemons, T.E.; Fine, L.J.; et al. Effect of long-chain ω-3 fatty acids and lutein + zeaxanthin supplements on cardiovascular outcomes: Results of the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA Intern. Med. 2014, 174, 763–771. [Google Scholar] [CrossRef]
- White, M.N.; Shrubsole, M.J.; Cai, Q.; Su, T.; Hardee, J.; Coppola, J.-A.; Cai, S.S. Effects of Fish Oil Supplementation on Eicosanoid Production in Subjects at Higher Risk for Colorectal Cancer. Eur. J. Cancer Prev. 2019, 28, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Mathias, R.A.; Vergara, C.; Gao, L.; Rafaels, W.; Hand, T.; Campbell, M.; Bickel, C. FADS genetic variants and omega-6 polyunsaturated fatty acid metabolism in a homogeneous island population. J. Lipid Res. 2010, 51, 2766–2774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Study | Participant Characteristics | Type and Duration of Intervention | Populations | Primary Outcomes: | Other Outcomes |
|---|---|---|---|---|---|
| ASCEND [23] | 15,840 diabetics | Median of 7.4 Years, 1 g/day of n-3 (0. 41 EPA and 0.34 DHA) | 97% White | No difference with Placebo in cardiovascular death | 7% reduction in the rate of non-fatal myocardial infarction and 20% reduction in vascular death |
| REDUCE IT [26] | 8179, on statin therapy and TG = 135 to 499 and LD L= 41–100 mg/dL | Median 4.9 years, 2 g of icosapentenyl twice a day or placebo | 11 Countries Primarily European Ancestry | 26% reduction in rate of composite primary cardiovascular endpoints | Rates of individual end points (except death from any cause lowered by ≥20% |
| REDUCE-IT, USA [27] | 3146 patients | Median 4.9 years, 2 g of icosapentenyl twice a day or placebo | 32.3 % women, 9 % Hispanic. 4% African American and 2% Asian | 31% reduction in rate of composite primary cardiovascular endpoints | Rates of individual end points (except death from any cause lowered by ≥28% |
| VITAL [22] | 29,581. 50 y old (5,106 subset of AA) | Median of 5.3 years 840 mg/day n-3 FA | All Participants African Americans (20%) Subjects who ate less than 1.5 portions of fish per week | No differences in rate of combined major cardiovascular events No significant difference 19% reduction in rate | 28% reduction in rate of myocardial infarction 71% reduction in myocardial infarction |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandez, M.L.; Blomquist, S.A.; Hallmark, B.; Chilton, F.H. Omega-3 Supplementation and Heart Disease: A Population-Based Diet by Gene Analysis of Clinical Trial Outcomes. Nutrients 2021, 13, 2154. https://doi.org/10.3390/nu13072154
Fernandez ML, Blomquist SA, Hallmark B, Chilton FH. Omega-3 Supplementation and Heart Disease: A Population-Based Diet by Gene Analysis of Clinical Trial Outcomes. Nutrients. 2021; 13(7):2154. https://doi.org/10.3390/nu13072154
Chicago/Turabian StyleFernandez, Maria Luz, Sarah A. Blomquist, Brian Hallmark, and Floyd H. Chilton. 2021. "Omega-3 Supplementation and Heart Disease: A Population-Based Diet by Gene Analysis of Clinical Trial Outcomes" Nutrients 13, no. 7: 2154. https://doi.org/10.3390/nu13072154
APA StyleFernandez, M. L., Blomquist, S. A., Hallmark, B., & Chilton, F. H. (2021). Omega-3 Supplementation and Heart Disease: A Population-Based Diet by Gene Analysis of Clinical Trial Outcomes. Nutrients, 13(7), 2154. https://doi.org/10.3390/nu13072154







