Home-Delivered Meals: Characterization of Food Intake in Elderly Beneficiaries
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Interviews
2.3. Socio-Demographic Data
2.4. Food Intake Measurement
2.4.1. Nutritional Status Characterization
2.4.2. Physical, Psychological, and Cognitive Status Characterization
2.5. Data Analysis
3. Results
3.1. Participant Characterization
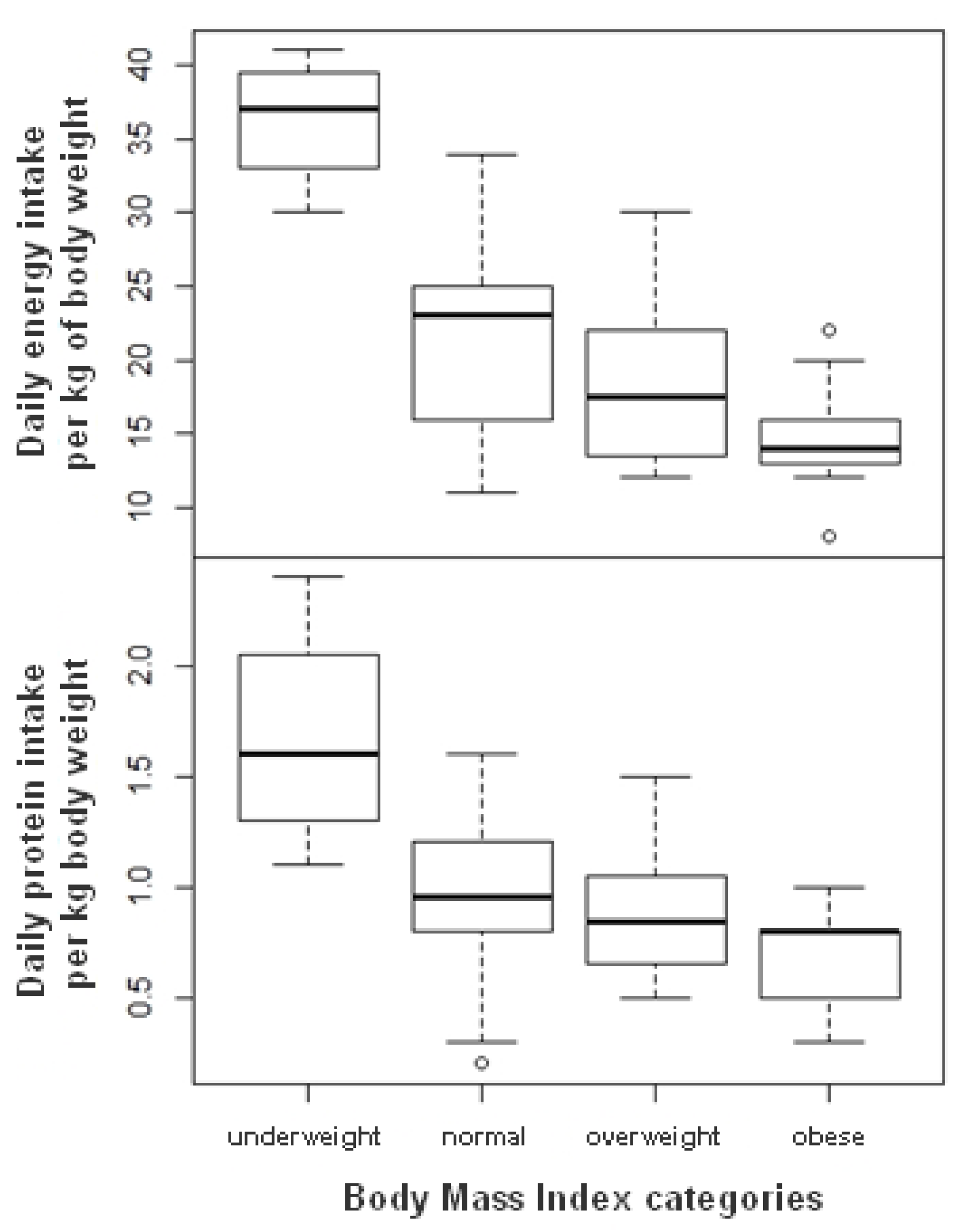
3.2. Nutritional Intakes
3.3. Nutritional Status
3.4. Univariate Regression Results
3.5. Multiple Regression Results
4. Discussion
4.1. Inadequate Dietary Intake in View of Nutritional Recommendations
4.2. A Double Misfortune: Being Simultaneously Overweight and at Risk of Undernutrition
4.3. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| MNA | Mini Nutritional Assessment |
| SPPB | Short Physical Performance Battery |
| MMSE | Mini Mental State Examination |
| GDS | Geriatric Depression Scale |
| M | Mean |
| SD | Standard Deviation |
| TDI | Total Daily nutrient intake |
| DNI | Daily Nutrient Intake relative to body weight |
| RDA | Recommended Dietary Allowances |
| RNI | Recommended Nutrient Intake |
References
- Cardon, P.; Gojard, S. Les personnes âgées face à la dépendance culinaire: Entre délégation et remplacement. La Documentation française. Retraite Soc. 2008, 4, 169–193. [Google Scholar]
- Fleury, S.; Sulmont-Rossé, C.; Tronchon, P.; Wymelbeke, V.V.; Roux, S. Le portage de repas à domicile: Processus d’installation et d’appropriation de ce service par les personnes âgées en perte d’autonomie. Nutr. Clin. Metab. 2021, 35, 107–115. [Google Scholar] [CrossRef]
- An, R. Association of Home-Delivered Meals on Daily Energy and Nutrient Intakes: Findings from the National Health and Nutrition Examination Surveys. J. Nutr. Gerontol. Geriatr. 2015, 34, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Buys, D.R.; Marler, M.L.; O Robinson, C.; Hamlin, C.M.; Locher, J.L. Recruitment of volunteers for a home-delivered meals programme serving homebound older adults: A theoretically derived programme among faith communities. Public Health Nutr. 2010, 14, 1473–1478. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dewar, M.; Dickinson, A.; Smeeton, N. Tracking and treating malnutrition: A retrospective observational study of the nutritional status of vulnerable people accessing a meals-on-wheels (MOW) service. Prim. Health Care Res. Dev. 2020, 21, 19. [Google Scholar] [CrossRef]
- Frongillo, E.A.; Wolfe, W.S. Impact of Participation in Home-Delivered Meals on Nutrient Intake, Dietary Patterns, and Food Insecurity of Older Persons in New York State. J. Nutr. Elder. 2010, 29, 293–310. [Google Scholar] [CrossRef] [PubMed]
- Keller, H.H. Meal Programs Improve Nutritional Risk: A Longitudinal Analysis of Community-Living Seniors. J. Am. Diet. Assoc. 2006, 106, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Lindhardt, T.; Nielsen, M. Older patients’ use of technology for a post-discharge nutritional intervention – A mixed-methods feasibility study. Int. J. Med. Inform. 2017, 97, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Marceaux, S. The Impact of Participation in Meals on Wheels and More (MOWAM) in Austin, TX, on Dietary Intake and Health Status. Ph.D. Thesis, Texas State University-San Marcos, San Marcos, TX, USA, 2012. [Google Scholar]
- O’Leary, M.F.; Barreto, M.; Bowtell, J.L. Evaluating the Effect of a Home-Delivered Meals Service on the Physical and Psychological Wellbeing of a UK Population of Older Adults - A Pilot and Feasibility Study. J. Nutr. Gerontol. Geriatr. 2019, 39, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.-A.; Payette, H. Meals-on-wheels improves energy and nutrient intake in a frail free-living elderly population. J. Nutr. Health Aging 2006, 10, 554–560. [Google Scholar] [PubMed]
- Ullevig, S.L.; Sosa, E.T.; Crixell, S.; Uc, E.; Greenwald, B.; Marceaux, S.; Friedman, B.J. Impact of Home-Delivered Meals on Nutrition Status and Nutrient Intake among Older Adults in Central Texas. J. Nutr. Health Aging 2018, 22, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Walden, O.; Hayes, P.A.; Lee, D.Y.; Montgomery, D.H. The Provision of Weekend Home Delivered Meals by State and a Pilot Study Indicating the Need for Weekend Home Delivered Meals. J. Nutr. Elder. 1989, 8, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Walton, K.; Charlton, K.; Manning, F.; McMahon, A.T.; Galea, S.; Evans, K. The nutritional status and energy and protein intakes of MOW clients and the need for further targeted strategies to enhance intakes. Appetite 2015, 95, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Wright, L.; Vance, L.; Sudduth, C.; Epps, J.B. The Impact of a Home-Delivered Meal Program on Nutritional Risk, Dietary Intake, Food Security, Loneliness, and Social Well-Being. J. Nutr. Gerontol. Geriatr. 2015, 34, 218–227. [Google Scholar] [CrossRef]
- Charlton, K.E.; Walton, K.; Moon, L.; Smith, K.; McMahon, A.T.; Ralph, F.; Stuckey, M.; Manning, F.; Krassie, J. “It could probably help someone else but not me”: A feasibility study of a snack programme offered to meals on wheels cli-ents. J. Nutr. 2013, 17, 6. [Google Scholar] [CrossRef]
- Frongillo, E.A.; Cantor, M.H.; Macmillan, T.; Issacman, T.D.; Sherrow, R.; Henry, M.; Wethington, E.; Pillemer, K. Who are the recipients of Meals-on-Wheels in New York City? A profile of based on a representative sample of Meals-on-Wheels recipients, part II. Care Manag. J. 2010, 11, 129–139. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoogenboom, M.S.; Spangler, A.A.; Crose, R. Functional Status and Nutrient Intake from the Council on Aging Meal and Total Daily Intake of Congregate, Adult Day Care and Homebound Program Participants. J. Nutr. Elder. 1998, 17, 1–18. [Google Scholar] [CrossRef]
- Krondl, M.; Lau, D.; Coleman, P.; Stocker, G. Tailoring of Nutritional Support for Older Adults in the Community. J. Nutr. Elder. 2003, 23, 17–32. [Google Scholar] [CrossRef] [PubMed]
- MacLellan, D.L. Contribution of Home-Delivered Meals to the Dietary Intake of the Elderly. J. Nutr. Elder. 1997, 16, 17–32. [Google Scholar] [CrossRef]
- Ponza, M. Serving Elders at Risk the Older Americans Act Nutrition Programs: National Evaluation of the Elderly Nutrition Program 1993-199. In Princeton, NJ: Mathematica Policy Research; Title III Evaluation Findings; US Department of Health and Human Services, Office of the Assistant Secretary for Aging: Washington, DC, USA, 1996; Volume I, p. 331. [Google Scholar]
- Sharkey, J.R. Risk and Presence of Food Insufficiency Are Associated with Low Nutrient Intakes and Multimorbidity among Homebound Older Women Who Receive Home-Delivered Meals. J. Nutr. 2003, 133, 3485–3491. [Google Scholar] [CrossRef]
- Bresson, J.-L.; Mariotti, F. Actualisation des Repères du PNNS: Elaboration des Références Nutritionnelles. Avis de l’Anses, Rapport D’Expertise Scientifique. Edition Scientifique. 2016. Available online: https://www.anses.fr/fr/system/files/NUT2012SA0103Ra-2.pdf (accessed on 23 October 2020).
- Stratégie de prise en charge en cas de dénutrition protéino-énergétique chez la personne âgée: Recommandations professionnelles (2007). Med. Mal. Metab. 2007, 1, 92–96. [CrossRef]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.C.; Stehle, P.; Teta, D.; et al. Evidence-Based Recommendations for Optimal Dietary Protein Intake in Older People: A Position Paper From the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.J.; Bauer, J.M.; Ms, R.P.S.A.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.S.; Charlton, K.; Maggio, M.; et al. Frequency of Malnutrition in Older Adults: A Multinational Perspective Using the Mini Nutritional Assessment. J. Am. Geriatr. Soc. 2010, 58, 1734–1738. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2004, 19, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Guillet, C.; Salles, J.; Cano, N.; Boirie, Y. Physiopathological Mechanism of Sarcopenia. Clin. Geriatr. Med. 2011, 27, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.I.T.D.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition–A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef]
- Ahmed, T.; Haboubi, N. Assessment and management of nutrition in older people and its importance to health. Clin. Interv. Aging 2010, 5, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Charlton, K.; Nichols, C.; Bowden, S.; Milosavljevic, M.; Lambert, K.; Barone, L.; Mason, M.; Batterham, M. Poor nutritional status of older subacute patients predicts clinical outcomes and mortality at 18 months of follow-up. Eur. J. Clin. Nutr. 2012, 66, 1224–1228. [Google Scholar] [CrossRef]
- Akner, G.; Cederholm, T. Treatment of protein-energy malnutrition in chronic nonmalignant disorders. Am. J. Clin. Nutr. 2001, 74, 6–24. [Google Scholar] [CrossRef]
- Barker, L.A.; Gout, B.S.; Crowe, T.C. Hospital Malnutrition: Prevalence, Identification and Impact on Patients and the Healthcare System. Int. J. Environ. Res. Public Health 2011, 8, 514–527. [Google Scholar] [CrossRef] [PubMed]
- Salminen, K.S.; Suominen, M.H.; Kautiainen, H.; Pitkälä, K.H. Associations between Nutritional Status, Frailty and Health-Related Quality of Life Among Older Long-Term Care Residents in Helsinki. J. Nutr. Health Aging 2020, 24, 319–324. [Google Scholar] [CrossRef]
- Masson, Y. Management et Ingénierie en Restauration Collective. Module 1: Bases sur la Restauration Collective. Master 1 Alimentation MIRC + SSAA. 2015. Available online: http://www.isthia.fr/master-sciences-sociales-mirc-481.html (accessed on 23 October 2020).
- Bardez, C.; Cressens, M.; Hilaire, A.; Lyannaz, S.; Montignies, J.; Costa, B.; Etchetto, C.; Kohler, C.; Michelot, J. Personnes Âgées Vivant à Domicile, le Portage de Repas: Élément Essentiel de Lutte Contre la Dénutrition? Module Interpro-Fessionnel de Santé Publique, Ecole des Hautes Etudes en Santé Publique (EHESP); EHESP (Ecole Des Hautes Etudes En Santé Publique): Rennes, France, 2008. [Google Scholar]
- Fleury, S.; Tronchon, P.; Rota, J.; Meunier, C.; Mardiros, O.; Van Wymelbeke-Delannoy, V.; Sulmont-Rossé, C. The Nutritional Issue of Older People Receiving Home-Delivered Meals: A Systematic Review. Front. Nutr. 2021, 8. [Google Scholar] [CrossRef] [PubMed]
- Borkent, J.W.; Beelen, J.; Linschooten, J.O.; Roodenburg, A.J.C.; De Van Der Schueren, M.A.E. The ConsuMEER study: A randomised trial towards the effectiveness of protein-rich ready-made meals and protein-rich dairy products in increasing protein intake of community-dwelling older adults after switching from self-prepared meals towards ready-made meals. J. Nutr. Sci. 2019, 8, e30. [Google Scholar] [CrossRef] [PubMed]
- Arjuna, T.; Miller, M.; Soenen, S.; Chapman, I.; Visvanathan, R.; Luscombe-Marsh, N.D. Serve Size and Estimated Energy and Protein Contents of Meals Prepared by ‘Meals on Wheels’ South Australia Inc.: Findings from a Meal Audit Study. Foods 2018, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Gollub, E.A.; Weddle, D.O. Improvements in nutritional intake and quality of life among frail homebound older adults receiving home-delivered breakfast and lunch. J. Am. Diet. Assoc. 2004, 104, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Ziylan, C.; Haveman-Nies, A.; Kremer, S.; de Groot, L.C. Protein-Enriched Bread and Readymade Meals Increase Community-Dwelling Older Adults’ Protein Intake in a Double-Blind Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2017, 18, 145–151. [Google Scholar] [CrossRef]
- Ciqual: Table de Composition Nutritionnelle des Aliments. Available online: https://ciqual.anses.fr/ (accessed on 23 October 2020).
- Guide Pratique du Poids des Vêtements et Tissus en Machine: Jean, Serviette, t-Shirt, (n.d.). IDEAL. Available online: https://www.ideal.fr/guide/poids-et-quantite-de-linge/ (accessed on 19 October 2020).
- Chumlea, W.C.; Roche, A.F.; Steinbaugh, M.L. Estimating Stature from Knee Height for Persons 60 to 90 Years of Age. J. Am. Geriatr. Soc. 1985, 33, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Guigoz, Y.; Lauque, S.; Vellas, B.J. Identifying the elderly at risk for malnutrition The Mini Nutritional Assess-ment. Clin. Geriatr. Med. 2002. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A Short Physical Performance Battery Assessing Lower Extremity Function: Association with Self-Reported Disability and Prediction of Mortality and Nursing Home Admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Bird, V.; Rizzo, M.; Meader, N. Diagnostic validity and added value of the geriatric depression scale for depression in primary care: A meta-analysis of GDS30 and GDS15. J. Affect. Disord. 2010, 125, 10–17. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Sheikh, J. 9/Geriatric Depression Scale (GDS). Clin. Gerontol. 1986, 5, 165–173. [Google Scholar] [CrossRef]
- Lipschitz, D.A.; Mitchell, C.O.; Steele, R.W.; Milton, K.Y. Nutritional Evaluation and Supplementation of Elderly Subjects Participating in a "Meals on Wheels" Program. J. Parenter. Enter. Nutr. 1985, 9, 343–347. [Google Scholar] [CrossRef] [PubMed]
- GEM-RCN. Recommandations Nutritionnelles Pour Les Personnes Agées. Version 1.0 Juillet 2015. Available online: http://www.gemrcn.fr/ (accessed on 10 July 2020).
- CLCV. Enquête sur L’alimentation des Personnes Âgées. 2012. Available online: http://www.clcv.org/nos-enquetes/enquete-sur-l-alimentation-des-personnes-agees-02-02-2012.html (accessed on 15 December 2020).
- Lesourd, B.; Dadet, S. Portage de repas à domicile: Quel rôle et quelle efficacité ? Cah. L’année Gerontol. 2013, 5, 318–325. [Google Scholar] [CrossRef]
- Inzitari, M.; Doets, E.; Bartali, B.; Benetou, V.; Di Bari, M.; Visser, M.; Volpato, S.; Gambassi, G.; Topinkova, E.; De Groot, L.; et al. Nutrition in the age-related disablement process. J. Nutr. Health Aging 2011, 15, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Rosenbloom, C.A.; Whittington, F.J. The Effects of Bereavement on Eating Behaviors and Nutrient Intakes in Elderly Widowed Persons. J. Gerontol. 1993, 48, S223–S229. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pols-Vijlbrief, R.; Wijnhoven, H.; Visser, M. Perspectives on the causes of undernutrition of community-dwelling older adults: A qualitative study. J. Nutr. Health Aging 2017, 21, 1200–1209. [Google Scholar] [CrossRef]
- Locher, J.L.; Robinson, C.O.; Roth, D.L.; Ritchie, C.S.; Burgio, K.L. The Effect of the Presence of Others on Caloric Intake in Homebound Older Adults. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2005, 60, 1475–1478. [Google Scholar] [CrossRef]
- Kimura, Y.; Wada, T.; Okumiya, K.; Ishimoto, Y.; Fukutomi, E.; Kasahara, Y.; Chen, W.; Sakamoto, R.; Fujisawa, M.; Otsuka, K.; et al. Eating alone among community-dwelling Japanese elderly: Association with depression and food diversity. J. Nutr. Health Aging 2012, 16, 728–731. [Google Scholar] [CrossRef]
- Berry, E.M.; Marcus, E.-L. Disorders of Eating in the Elderly. J. Adult Dev. 2000, 7, 87–99. [Google Scholar] [CrossRef]
- Murphy, M.; Brooks, C.; New, S.; Lumbers, M. The use of the Mini-Nutritional Assessment (MNA) tool in elderly orthopaedic patients. Eur. J. Clin. Nutr. 2000, 54, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Vellas, B.; Guigoz, Y.; Baumgartner, M.; Garry, P.J.; Lauque, S.; Albarede, J.-L. Relationships between nutritional markers and the mini-nutritional assessment in 155 older persons. J. Am. Geriatr. Soc. 2000, 48, 1300–1309. [Google Scholar] [CrossRef] [PubMed]
- Herrera, J.C.M.; Martinsantos, F.J.; Carobautista, J.; Figueredo, M.C.S.; Garciamayor, S.; Morales-Asencio, J.M. Effectiveness of food-based fortification in older people a systematic review and meta-analysis. J. Nutr. Health Aging 2016, 20, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Sulmont-Rossé, C.; Maître, I.; Feyen, V.; Vandenberghe-Descamps, M.; Labouré, H.; Feron, G.; Van Wymelbeke, V. Quels aliments pour maintenir la prise alimentaire chez les personnes âgées et prévenir la dénutrition? Innov. Agron. 2018, 65, 99–111. [Google Scholar] [CrossRef]
- Van Wymelbeke, V.; Sulmont-Rossé, C.; Feyen, V.; Issanchou, S.; Manckoundia, P.; Maître, I. Optimizing sensory quality and variety: An effective strategy for increasing meal enjoyment and food intake in older nursing home residents. Appetite 2020, 153, 104749. [Google Scholar] [CrossRef] [PubMed]
- Desai, J.; Winter, A.; Young, K.W.; Greenwood, C.E. Changes in Type of Foodservice and Dining Room Environment Preferentially Benefit Institutionalized Seniors with Low Body Mass Indexes. J. Am. Diet. Assoc. 2007, 107, 808–814. [Google Scholar] [CrossRef]
- Elmståhl, S.; Blabolil, V.; Fex, G.; Küller, R.; Steen, B. Hospital nutrition in geriatric long-term care medicine. I. Effects of a changed meal environment. Compr. Gerontol. Sect. A Clin. Lab. Sci. 1987, 1, 29–33. [Google Scholar]
- Kremer, S.; Derks, J.; De Vries, M.N.; Boer, E.P.J.; Gorselink, M. Effect of a holistic meal and ambiance concept on main meal enjoyment and food intake of dutch nursing home residents: A pilot study. J. Aging Res. Clin. Pract. 2012, 1, 237–244. [Google Scholar]
- Nijs, K.A.N.D.; De Graaf, C.; Kok, F.J.; A Van Staveren, W. Effect of family style mealtimes on quality of life, physical performance, and body weight of nursing home residents: Cluster randomised controlled trial. BMJ 2006, 332, 1180–1184. [Google Scholar] [CrossRef]
- Oehlschlaeger, M.H.K.; Pastore, C.A.; Cavalli, A.S.; Gonzalez, M.C. Nutritional status, muscle mass and strength of elderly in southern Brazil. Nutr. Hosp. 2014, 31, 363–370. [Google Scholar] [CrossRef]
- Özkaya, I.; Gürbüz, M. Malnourishment in the overweight and obese elderly. Nutr. Hosp. 2019, 36, 39–42. [Google Scholar] [CrossRef]
- Roubenoff, R. Sarcopenic Obesity: The Confluence of Two Epidemics. Obes. Res. 2004, 12, 887–888. [Google Scholar] [CrossRef]
- Cetin, D.C.; Nasr, G. Obesity in the elderly: More complicated than you think. Clevel. Clin. J. Med. 2014, 81, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Oreopoulos, A.; McAlister, F.A.; Kalantar-Zadeh, K.; Padwal, R.; Ezekowitz, J.; Sharma, A.M.; Kovesdy, C.P.; Fonarow, G.; Norris, C.M. The relationship between body mass index, treatment, and mortality in patients with established coronary artery disease: A report from APPROACH. Eur. Heart J. 2009, 30, 2584–2592. [Google Scholar] [CrossRef] [PubMed]
- Batsis, J.A.; Mackenzie, T.A.; Barre, L.K.; LopezJimenez, F.; Bartels, S.J. Sarcopenia, sarcopenic obesity and mortality in older adults: Results from the National Health and Nutrition Examination Survey III. Eur. J. Clin. Nutr. 2014, 68, 1001–1007. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Human Basal Metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Waters, D.L.; Ward, A.L.; Villareal, D.T. Weight loss in obese adults 65years and older: A review of the controversy. Exp. Gerontol. 2013, 48, 1054–1061. [Google Scholar] [CrossRef]
- Eurostat. Eurostat, hlth_ehis_bm1i, Données. 2014. Available online: https://appsso.eurostat.ec.europa.eu (accessed on 3 December 2020).
- Starr, K.N.P.; McDonald, S.R.; Bales, C.W. Obesity and Physical Frailty in Older Adults: A Scoping Review of Lifestyle Intervention Trials. J. Am. Med. Dir. Assoc. 2014, 15, 240–250. [Google Scholar] [CrossRef]
- Bouchonville, M.F.; Villareal, D.T. Sarcopenic obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 412–419. [Google Scholar] [CrossRef]
- Houston, D.K.; Tooze, J.A.; Demons, J.L.; Davis, B.L.; Shertzer-Skinner, R.; Kearsley, L.B.; Kritchevsky, S.B.; Williamson, J.D. Delivery of a Vitamin D Intervention in Homebound Older Adults Using a Meals-on-Wheels Program: A Pilot Study. J. Am. Geriatr. Soc. 2015, 63, 1861–1867. [Google Scholar] [CrossRef] [PubMed]
- Darmon, N.; Lacroix, A.; Ruffieux, B. Experimental Economics Shows How Food Price Policies May Improve Diet While Increasing Socioeconomic Inequalities in Nutrition; Working Paper INRA; UMPF. 2011. Available online: https://hal.archives-ouvertes.fr/hal-02805543/ (accessed on 10 December 2020).
- Ruffieux, B. Évaluation par L’économie Expérimentale de Politiques Nutritionnelles de Prix et D’information. In Proceedings of the Internal Workshop, Dijon France, May 2010. [Google Scholar]
- Biró, G.; Hulshof, K.F.A.M.; Ovesen, L.; Cruz, J.A.A. For the EFCOSUM Group; Selection of methodology to assess food intake. Eur. J. Clin. Nutr. 2002, 56, S25–S32. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Roth, D.L.; Ritchie, C.S.; Burgio, K.L.; Locher, J.L. Reliability and Predictive Validity of Energy Intake Measures from the 24-Hour Dietary Recalls of Homebound Older Adults. J. Am. Diet. Assoc. 2010, 110, 773–778. [Google Scholar] [CrossRef] [PubMed]
| SPPB Initial Scores | Modified Scores |
|---|---|
| Chair lift a | |
| 0: test not performed 1: test duration ≥ 16.7 seconds 2: test duration [16.6, 13.7] seconds 3: test duration [13.6, 11.2] seconds 4: test duration ≤ 11.1 seconds | 0: no chair lift 1: 1 or 2 chair lifts 2: 3 or 4 chair lifts 3: test duration ≥ 16.7 seconds with help 4: test duration [16.6, 13.6] seconds with help 5: test duration [13.6, 11.2] seconds with help 6: test duration ≤ 11.1 seconds with help 7: test duration ≥ 16.7 seconds without help 8: test duration [16.6, 13.6] seconds without help 9: test duration [13.6, 11.2] seconds without help 10: test duration ≤ 11.1 seconds without help |
| Walking speed b | |
| 0: test not performed 1: test duration ≥ 9.3 seconds 2: test duration [6.7; 9.2] seconds 3: test duration [5.2; 6.6] seconds 4: test duration ≤ 5.1 seconds | 0: test not performed 1: test duration ≥ 9.3 seconds with help 2: test duration [6.7, 9.2] seconds with help 3: test duration [5.2, 6.6] seconds with help 4: test duration ≤ 5.1 seconds with help 5: test duration ≥ 9.3 seconds without help 6: test duration [6.7, 9.2] seconds without help 7: test duration [5.2, 6.6] seconds without help 8: test duration ≤ 5.1 seconds without help |
| Variables | Participants (n = 64) |
|---|---|
| % of women | 75% |
| Age | 83.4 (7.5) (70–97) |
| Number of meals delivered per week | |
| ≤7 meals per week | 69% |
| Between 13 et 21 meals per week | 31% |
| Marital status | |
| Alone a | 51% |
| Couple | 44% |
| Widow | 5% |
| Education level | |
| No | 14% |
| Primary | 27% |
| Secondary | 33% |
| Graduate | 26% |
| Self-perception of financial resources | |
| low | 60% |
| Fair | 31% |
| Good | 9% |
| Nutritional statut | |
| Body Mass Index (BMI) | 26.1 (6.1) (15–46) |
| Underweight | 6% |
| Normal body weight | 39% |
| Overweight | 33% |
| Obese | 22% |
| Mini Nutritional Assessment (MNA) | 20.1 (3.8) (10–26) |
| Normal | 22% |
| Risk of malnourishment | 61% |
| Malnourishment | 17% |
| Physical, psychological, and cognitive status | |
| Number of comorbidities | 3.4 (1.6) [0–8] |
| Short Physical Performance Battery (SPPB) | 7.2 (5.2) [0–14] |
| Chair lift | 3.0 (2.4) [0–8] |
| Walking test | 4.2 (3.4) [0–8] |
| Mini-Mental State Examination (MMSE) | 25.9 (3.9) [14–30] |
| Geriatric Depression Scale (GDS) | 4.9 (3.4) [0–14] |
| Nutrients | TDI | DNI | RNI per kg of Body Weight | % Deficient 1 |
|---|---|---|---|---|
| Energy (kcal) | 1306 (369) | 20.0 (7.0) | 30.0 | 83% |
| Proteins (g) | 58 (21) | 0.9 (0.4) | 1.2 | 72% |
| Carbohydrates (g) | 152 (48) | 2.4 (1.1) | 3.5 | 81% |
| Lipids (g) | 48 (18) | 0.8 (0.3) | 1.1 | 75% |
| Body Mass Index (BMI) | |||||
|---|---|---|---|---|---|
| Nutritional Status (MNA) | Underweight <21 | Normal 21–24 | Overweight 25–29 | Obese >30 | Total |
| Normal > 23.5 n % | 0 0% | 3 4.5% | 10 16% | 1 1.5% | 14 22% |
| At risk of malnutrition 23.5–17 n % | 4 6% | 13 20% | 10 16% | 12 19% | 39 61% |
| Malnutrition < 17 n % | 5 8% | 4 6% | 1 1.5% | 1 1.5% | 11 17% |
| Total n % | 9 14% | 20 31% | 21 33% | 14 22% | 64 100% |
| Energy | Protein | |||||||
|---|---|---|---|---|---|---|---|---|
| TDI | DNI | TDI | DNI | |||||
| β | CI | β | CI | β | CI | β | CI | |
| Sex (reference: man) | ||||||||
| Woman | −77.73 | [−299.36; 143.90] | 1.02 | [−3.34; 5.39] | −9.37 | [−22.09; 3.35] | −0.04 | [−0.28; 0.19] |
| Age | −8.91 | [−22.04; 4.21] | −0.10 | [−0.36; 0.16] | −0.53 | [−1.30; 0.23] | −0.01 | [−0.02; 0.01] |
| Marital status (reference: single) | ||||||||
| Couple | 141.92 | [−310.18; 594.02] | 2.75 | [−6.11; 11.61] | 14.36 | [−11.52; 40.25] | 0.31 | [−0.16; 0.79] |
| Widow | −42.41 | [−242.47; 157.64] | 1.77 | [−2.15; 5.69] | −5.38 | [−16.83; 6.08] | 0.03 | [−0.18; 0.24] |
| Education level (reference: no) | ||||||||
| Primary | −113.28 | [−428.94; 202.37] | −1.59 | [−8.03; 4.89] | −5.74 | [−24.30; 12.82] | −0.08 | [−0.41; 0.26] |
| Secondary | 142.80 | [−158.81; 444.42] | −0.50 | [−6.70; 5.70] | 1.62 | [−16.12; 19.35] | −0.08 | [−0.41; 0.24] |
| Graduate | 154.55 | [−157.65; 466.76] | 0.50 | [−5.92; 6.91] | 10.53 | [−7.83; 28.89] | 0.16 | [−0.17; 0.50] |
| Self-perception of financial resources (reference: low) | ||||||||
| Fair | −52.80 | [−263.89; 158.29] | 2.29 | [−1.83; 6.42] | −2.69 | [−15.05; 9.67] | 0.06 | [−0.16; 0.29] |
| good | −190.44 | [−582.78; 201.90] | −1.68 | [−9.35; 5.98] | −6.43 | [−29.40; 16.54] | 0.22 | [−0.19; 0.63] |
| BMI | 12.09 | [−3.34; 27.52] | −0.60 *** | [−0.87; −0.34] | 1.11 ** | [0.23; 1.97] | −0.02 ** | [−0.04; −0.01] |
| Number of meals delivered per week (reference: ≤1 meal per day) | ||||||||
| >1 meal/day | 335.76 *** | [151.39; 520.13] | 5.64 ** | [1.91; − 9.38] | 16.50 ** | [5.41; 27.59] | 0.31 ** | [0.11; 0.51] |
| MNA | 27.42 * | [2.73; 52.11] | −0.09 | [−0.59; 0.42] | 1.48 * | [0.04; 2.93] | −0.01 | [−0.04; 0.01] |
| Comorbidities | 65.64 * | [7.13; 124.15] | −0.06 | [−1.26; 1.14] | 5.56 *** | [2.32; 8.79] | 0.04 | [−0.02; 0.10] |
| SPPB | −0.84 | [−19.55; 17.87] | 0.43 ** | [0.08; 0.78] | −0.17 | [−1.26; 0.92] | 0.02 (*) | [0.00; 0.04] |
| MMSE | 3.20 | [−22.60; 29.00] | −0.04 | [−0.55; 0.47] | −0.09 | [−1.60; 1.41] | −0.01 | [−0.03; 0.03] |
| GDS | 1.12 | [−27.77; 30.02] | −0.07 | [−0.64; 0.49] | 0.84 | [−0.83; 2.51] | 0.01 | [−0.02; 0.04] |
| Author (s), Year Country | Population | Energy Intakes | Protein Intakes |
|---|---|---|---|
| Our Study France | 64 beneficiaries of a home-delivery meals service (social services center of the city of Paris) | 83% did not meet the recommendations of 30 kcal per day per kg of body weight | 72% did not meet the recommendations of 1.2 g of protein per day per kg of body weight |
| Lipschitz et al, 1985 USA | 33 beneficiaries of a home-delivery meals service (not OAA) | 35% did not reach 80% of the energy and protein recommendations *. | |
| Ponza, 1996 USA | 818 beneficiaries of a home-delivery meals service (OAA) | 44% did not reach 2/3 of the energy recommendations (1900–2300 kcal/d) | 14% did not reach 2/3 of the protein recommendations (50–63 g/d) |
| Sharkey, 2003 USA | 279 beneficiaries of a home-delivery meals service (OAA) | 25% did not reach 2/3 of the energy recommendations * | 25% did not meet the protein recommendations * |
| Walden et al, 1998 USA | 16 beneficiaries of a home-delivery meals service (OAA) | 56% did not meet the energy recommendations * | 6% did not meet the protein recommendations * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fleury, S.; Van Wymelbeke-Delannoy, V.; Lesourd, B.; Tronchon, P.; Maître, I.; Sulmont-Rossé, C. Home-Delivered Meals: Characterization of Food Intake in Elderly Beneficiaries. Nutrients 2021, 13, 2064. https://doi.org/10.3390/nu13062064
Fleury S, Van Wymelbeke-Delannoy V, Lesourd B, Tronchon P, Maître I, Sulmont-Rossé C. Home-Delivered Meals: Characterization of Food Intake in Elderly Beneficiaries. Nutrients. 2021; 13(6):2064. https://doi.org/10.3390/nu13062064
Chicago/Turabian StyleFleury, Ségolène, Virginie Van Wymelbeke-Delannoy, Bruno Lesourd, Paul Tronchon, Isabelle Maître, and Claire Sulmont-Rossé. 2021. "Home-Delivered Meals: Characterization of Food Intake in Elderly Beneficiaries" Nutrients 13, no. 6: 2064. https://doi.org/10.3390/nu13062064
APA StyleFleury, S., Van Wymelbeke-Delannoy, V., Lesourd, B., Tronchon, P., Maître, I., & Sulmont-Rossé, C. (2021). Home-Delivered Meals: Characterization of Food Intake in Elderly Beneficiaries. Nutrients, 13(6), 2064. https://doi.org/10.3390/nu13062064





