Magnesium Metabolism in Chronic Alcohol-Use Disorder: Meta-Analysis and Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Searches
2.2. Selection Criteria
2.3. Definitions
2.4. Data Extraction
2.5. Analysis
3. Results
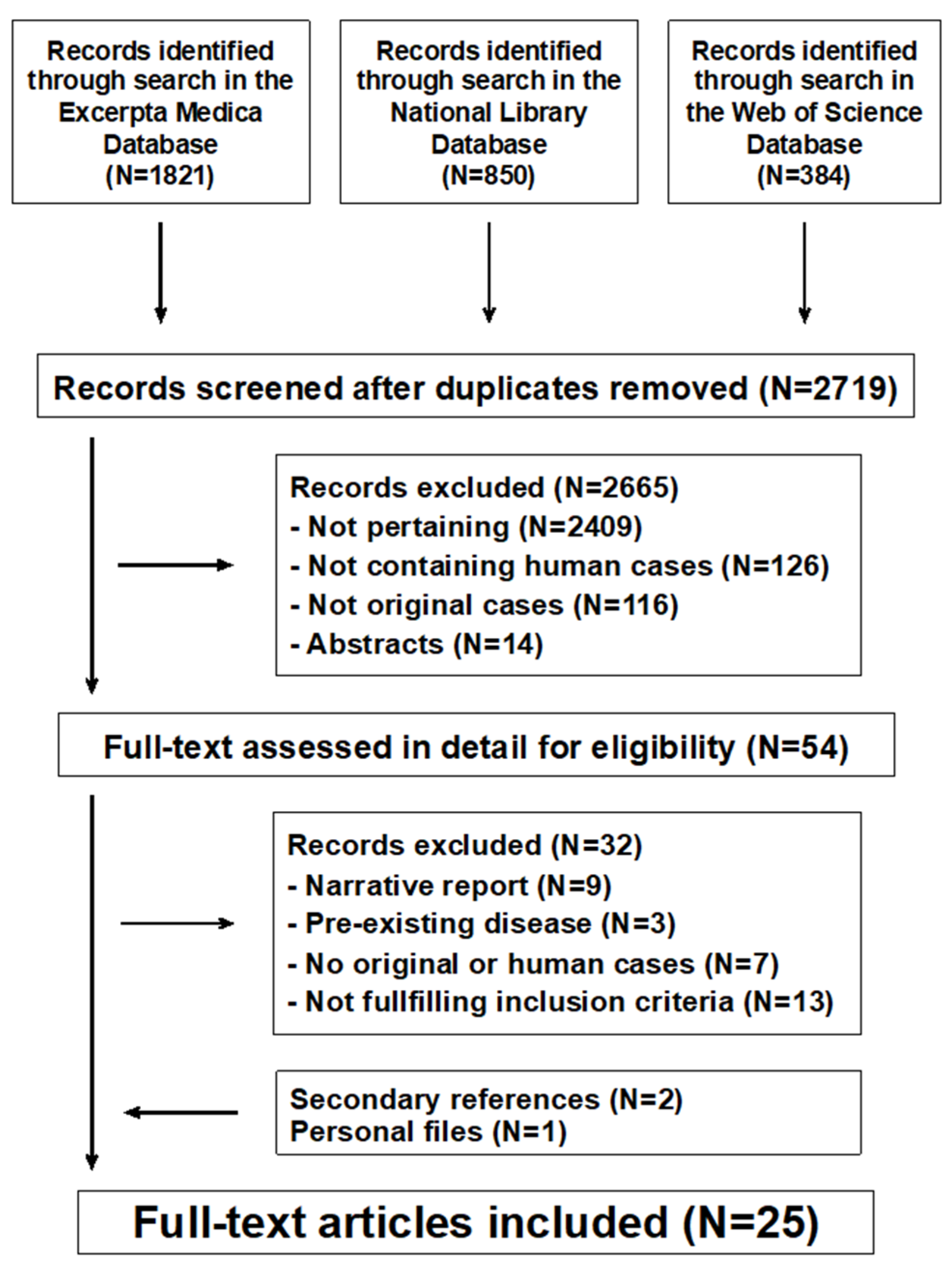
3.1. Search Results
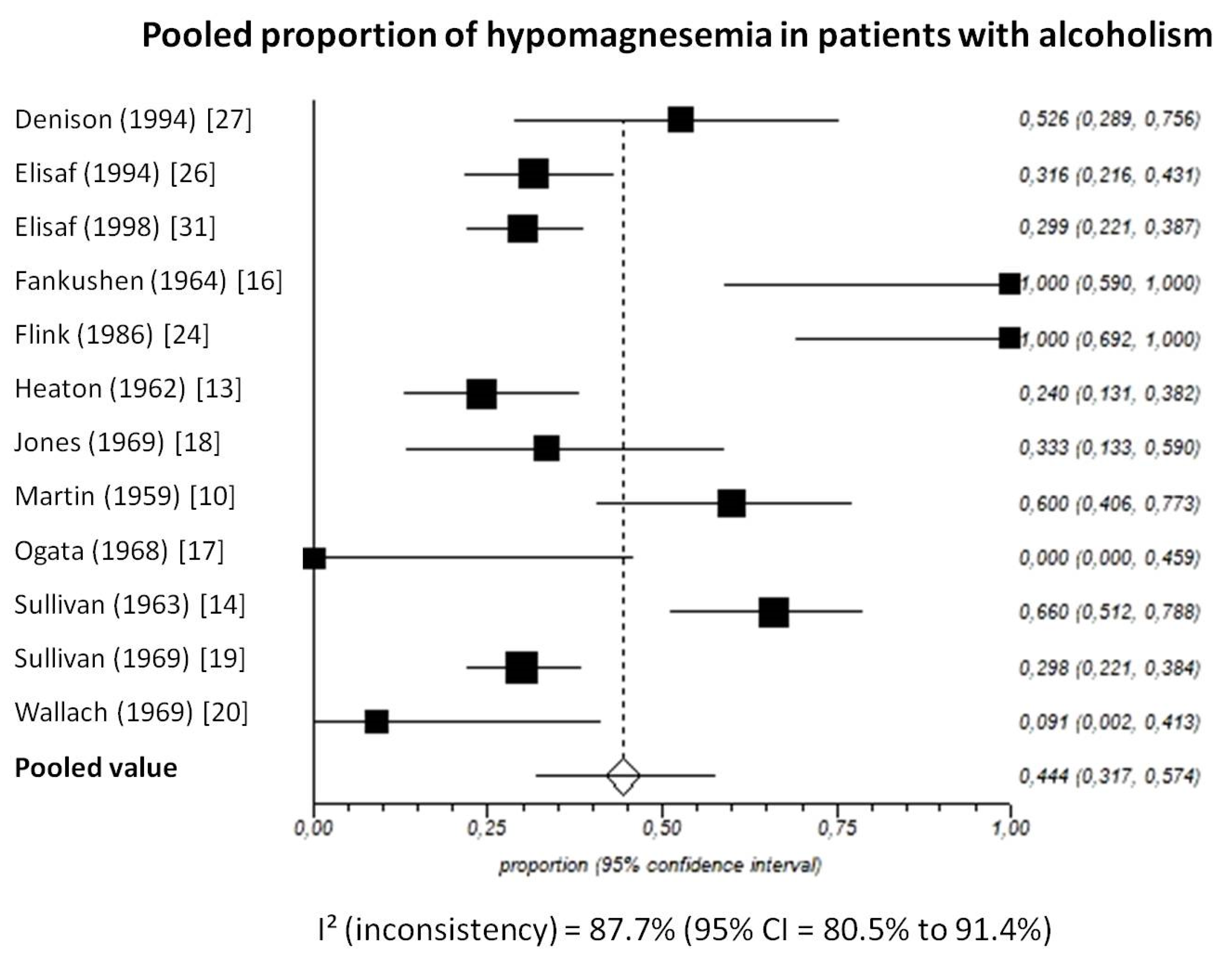
3.2. Extracellular Magnesium Concentration
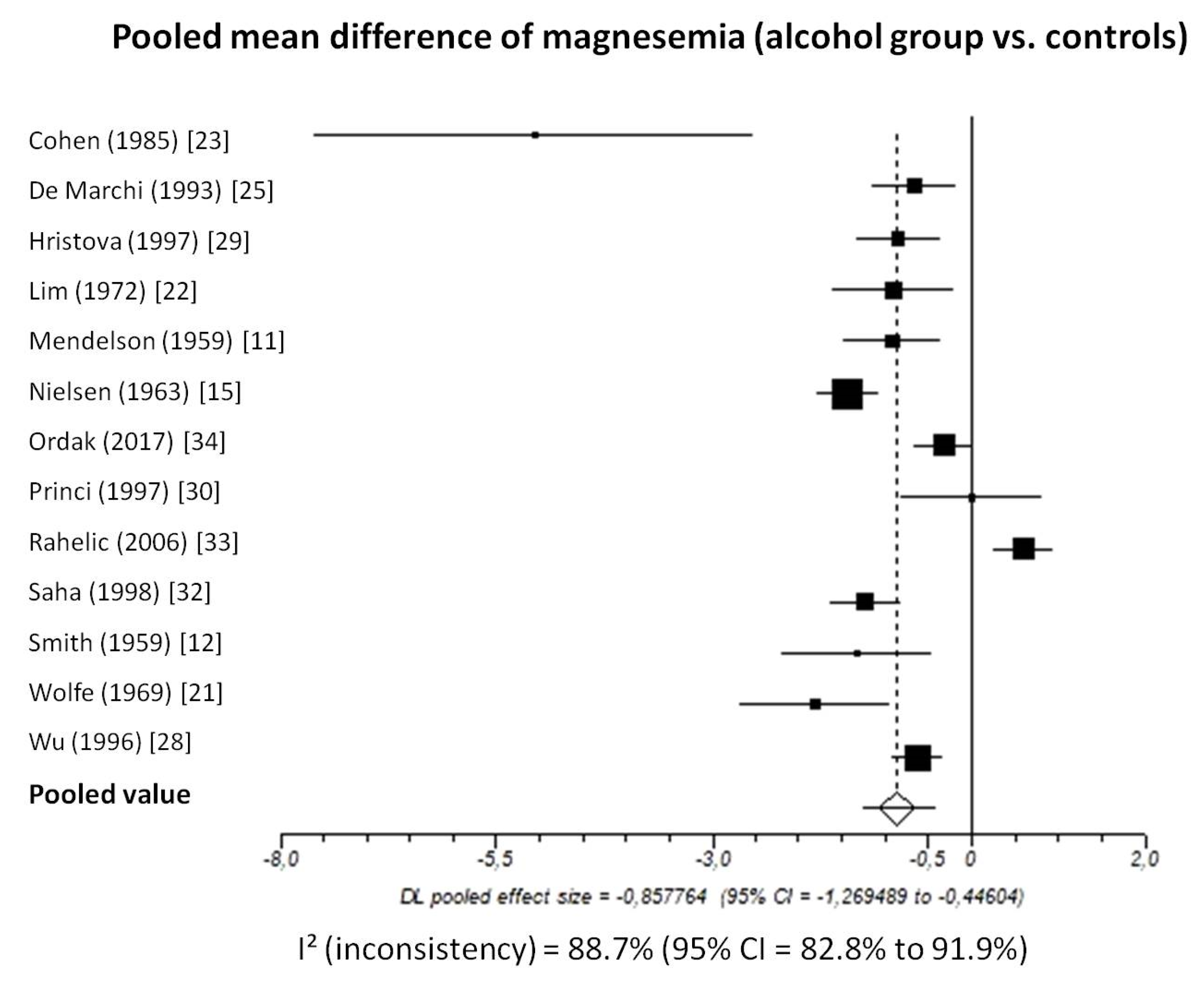
3.2.1. Total Magnesium
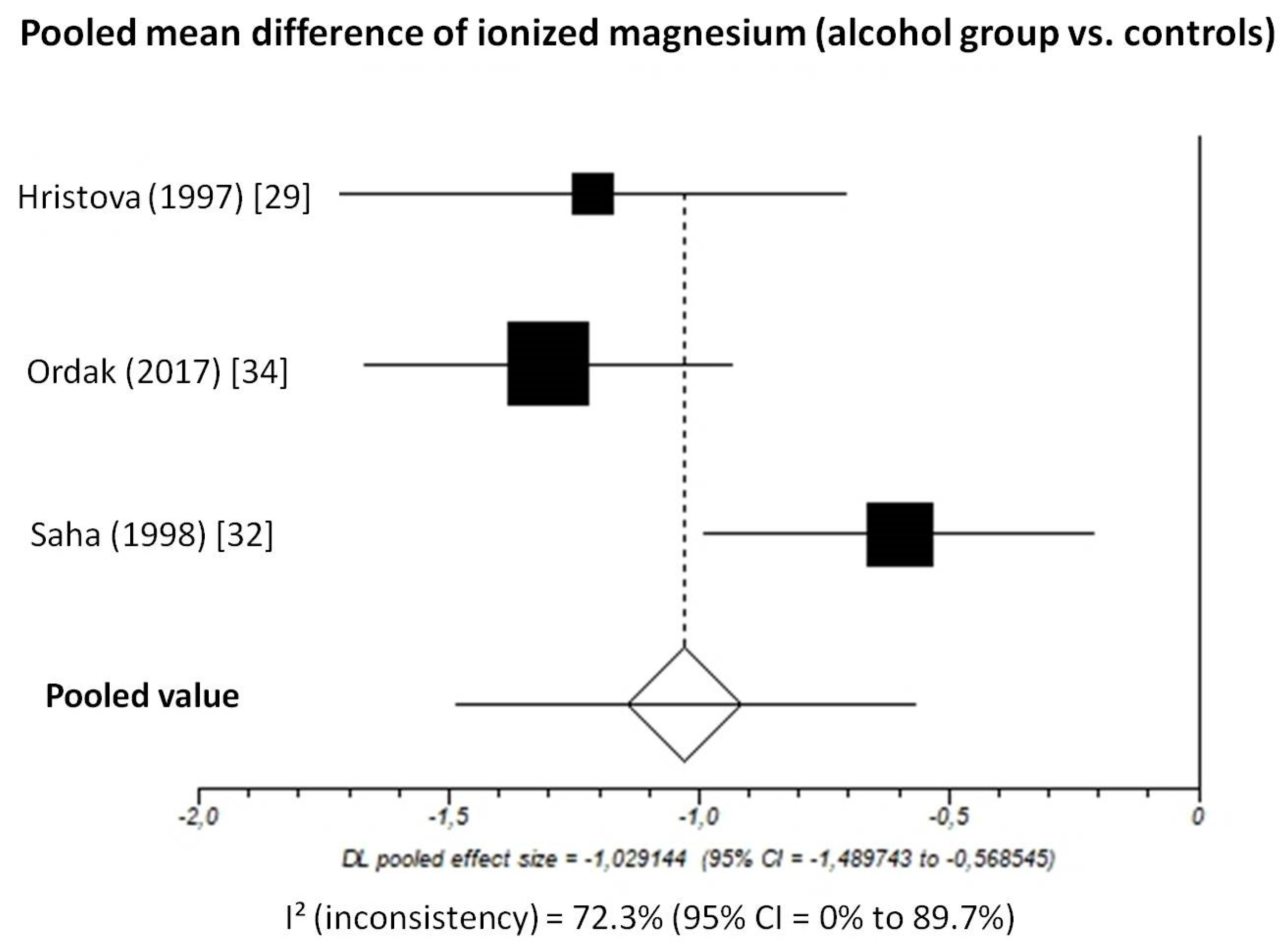
3.2.2. Ionized Magnesium
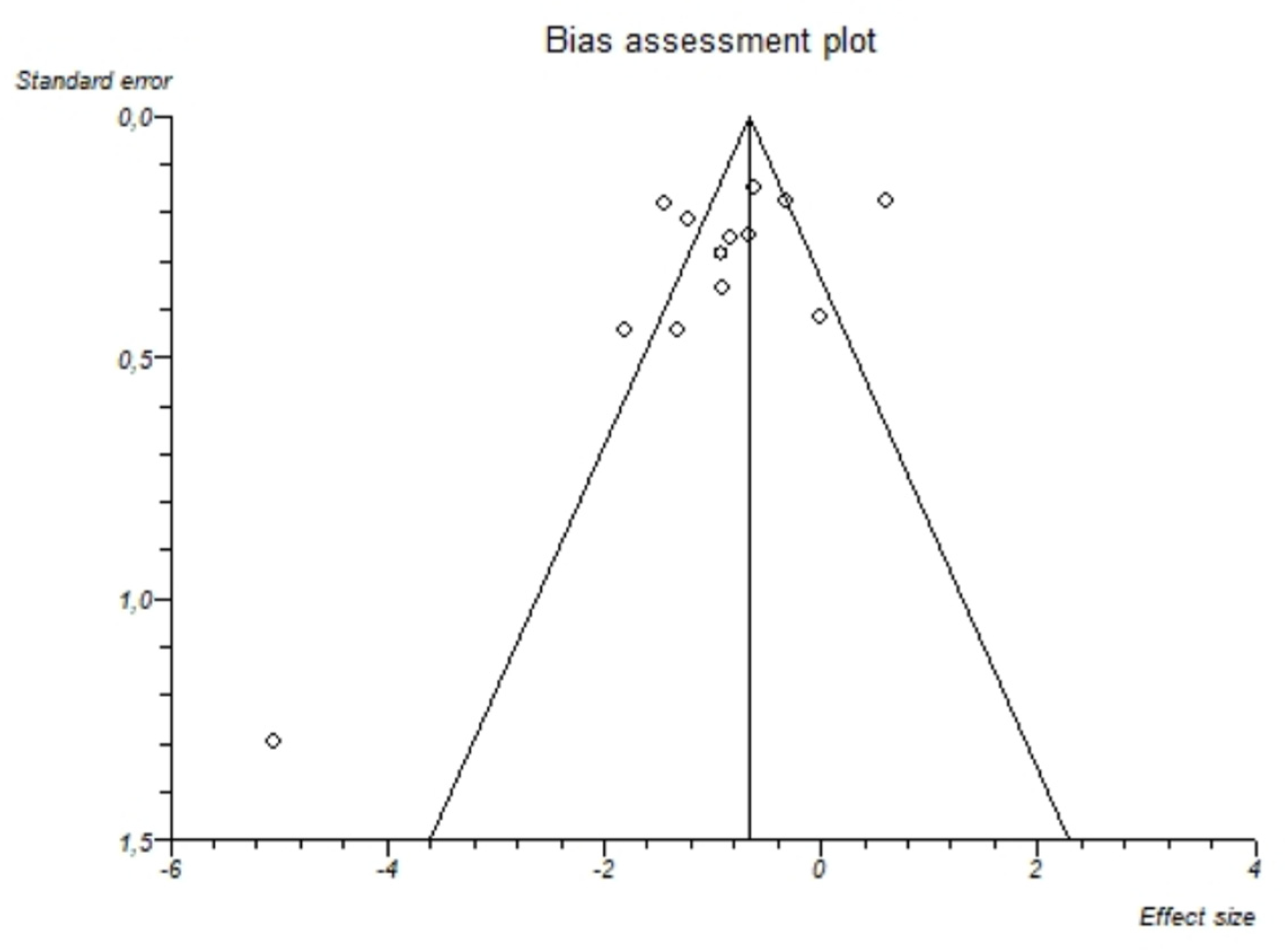
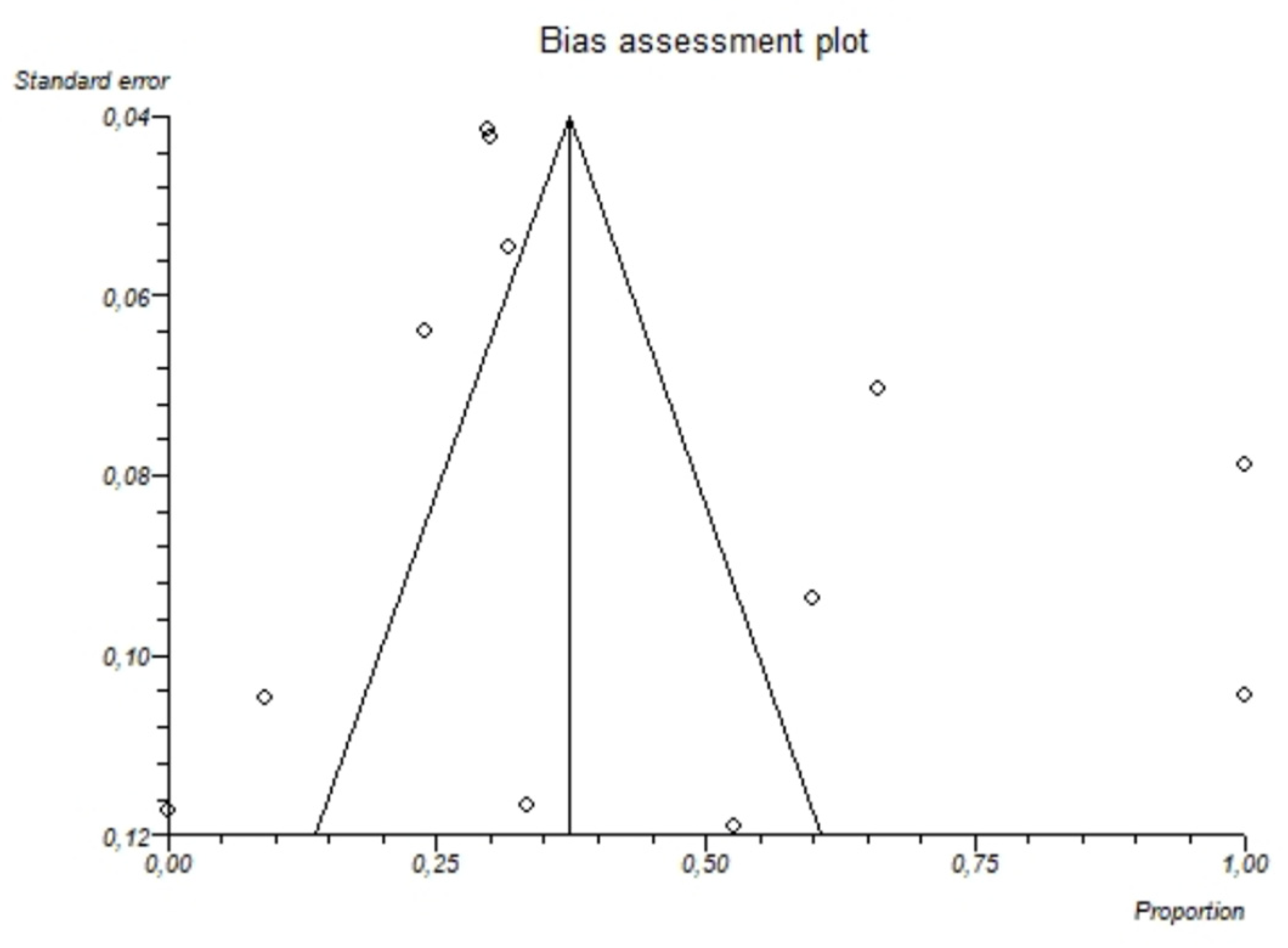
3.2.3. Pooled Analyses
3.3. Intracellular Magnesium
3.3.1. Intra-Erythrocytic Magnesium
3.3.2. Intra-Lymphocytic Magnesium
3.3.3. Bone Magnesium Content
3.3.4. Skeletal Muscle Magnesium Content
3.4. Kidney Magnesium Handling
4. Discussion
- Definition of alcohol-use disorder: The DSM-5 criteria for alcohol-use disorders, its severity and its various subtypes should be used to enroll patients in future investigations.
- Nutritional status: Nutritional status is often impaired in alcohol-use disorder [37,38]. In fact, affected subjects often ingest too little of essential nutrients. Furthermore, alcohol may prevent the body from properly absorbing and using nutrients [37,38]. The determination of mid-arm muscle circumference, triceps skinfold thickness, mid-arm circumference, and subscapular skinfold thickness is imperative for the clinical evaluation of nutritional status. The gold standard for the diagnosis of poor intestinal absorption is the determination for fat on 72-h stool collection. Near-infrared analysis is superior because it allows for simultaneous measurement of fecal fat, nitrogen, and carbohydrates in a single sample. Finally, stable isotopes represent a useful technique for determining intestinal absorption of magnesium [41].
- Dietary magnesium intake: The strategy to collect information on dietary magnesium intake is nowadays an image-assisted dietary food record [42].
- Liver disease: Alcohol-associated liver disease might play a role in modulating the severity of magnesium depletion [39]. Magnesium metabolism should be investigated separately in patients without and with liver diseases.
- Pancreatitis: Attacks of pancreatitis are common in chronic alcohol-use disorder. Since hypomagnesemia can occur in this condition, we advise the determination of lipase in future studies [40].
- Drug-induced magnesium depletion: The reports selected for this review did not include subjects with drug-induced hypomagnesemia. Since an association between hypomagnesemia and proton pump inhibitors was first suspected in 2006 [7], we speculate that these agents were sometimes not specifically imputed as a cause of hypomagnesemia. As a consequence, the possible role of drug-induced hypomagnesemia should be carefully addressed in future studies.
- Magnesium status: Ideally, total circulating magnesium level should be measured by atomic absorption spectrometry. It is true, however, that many colorimetric methods have been validated. Potentiometric sensors are employed for the measurement of blood-ionized magnesium. Since the selectivity of these sensors for magnesium over calcium is unsatisfactory, the results are adjusted for calcium interference. It has been suggested that magnesium content in red blood cells, peripheral lymphocytes, bones, and skeletal muscles might be an index of magnesium status. However, the available evidence does not support the use of intracellular magnesium levels as an indicator of overall bodily magnesium status [3,4,43].
5. Management
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spanagel, R. Alcoholism: A systems approach from molecular physiology to addictive behavior. Physiol. Rev. 2009, 89, 649–705. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F.; Clegg, D.J. Electrolyte disturbances in patients with chronic alcohol-use disorder. N. Engl. J. Med. 2017, 377, 1368–1377. [Google Scholar] [CrossRef]
- Elin, R.J. Magnesium metabolism in health and disease. Dis. Mon. 1988, 34, 161–218. [Google Scholar] [CrossRef]
- Agus, Z.S. Mechanisms and causes of hypomagnesemia. Curr. Opin. Nephrol. Hypertens. 2016, 25, 301–307. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Ann. Intern. Med. 2009, 151, W65–W94. [Google Scholar] [CrossRef] [PubMed]
- Krupski, T.L.; Dahm, P.; Fesperman, S.F.; Schardt, C.M. How to perform a literature search. J. Urol. 2008, 179, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Janett, S.; Camozzi, P.; Peeters, G.G.; Lava, S.A.; Simonetti, G.D.; Goeggel Simonetti, B.; Bianchetti, M.G.; Milani, G.P. Hypomagnesemia induced by long-term treatment with proton-pump inhibitors. Gastroenterol. Res. Pract. 2015, 2015, 951768. [Google Scholar] [CrossRef] [PubMed]
- Santi, M.; Milani, G.P.; Simonetti, G.D.; Fossali, E.F.; Bianchetti, M.G.; Lava, S.A.G. Magnesium in cystic fibrosis—Systematic review of the literature. Pediatr. Pulmonol. 2016, 51, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Scoble, J.E.; Sweny, P.; Varghese, Z.; Moorhead, J. Investigating hypomagnesaemia. Lancet 1987, 329, 276. [Google Scholar] [CrossRef]
- Martin, H.E.; McCuskey, C., Jr.; Tupikova, N. Electrolyte disturbance in acute alcoholism: With particular reference to magnesium. Am. J. Clin. Nutr. 1959, 7, 191–196. [Google Scholar] [CrossRef]
- Mendelson, J.; Wexler, D.; Kubzansky, P.; Leiderman, H.; Solomon, P. Serum magnesium in delirium tremens and alcoholic hallucinosis. J. Nerv. Ment. Dis. 1959, 128, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.O.; Hammarsten, J.F. Intracellular magnesium in delirium tremens and uremia. Am. J. Med. Sci. 1959, 237, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Heaton, F.W.; Pyrah, L.N.; Beresford, C.C.; Bryson, R.W.; Martin, D.F. Hypomagnesæmia in chronic alcoholism. Lancet 1962, 280, 802–805. [Google Scholar] [CrossRef]
- Sullivan, J.F.; Lankford, H.G.; Swartz, M.J.; Farrell, C. Magnesium metabolism in alcoholism. Am. J. Clin. Nutr. 1963, 13, 297–303. [Google Scholar] [CrossRef]
- Nielsen, J. Magnesium metabolism in acute alcoholics. Dan. Med. Bull. 1963, 10, 225–233. [Google Scholar] [CrossRef]
- Fankushen, D.; Raskin, D.; Dimich, A.; Wallach, S. The significance of hypomagnesemia in alcoholic patients. Am. J. Med. 1964, 37, 802–812. [Google Scholar] [CrossRef]
- Ogata, M.; Mendelson, J.H.; Mello, N.K. Electrolyte and osmolality in alcoholics during experimentally induced intoxication. Psychosom. Med. 1968, 30, 463–488. [Google Scholar] [CrossRef]
- Jones, J.E.; Shane, S.R.; Jacobs, W.H.; Flink, E.B. Magnesium balance studies in chronic alcoholism. Ann. N. Y. Acad. Sci. 1969, 162, 934–946. [Google Scholar] [CrossRef]
- Sullivan, J.F.; Wolpert, P.W.; Williams, R.; Egan, J.D. Serum magnesium in chronic alcoholism. Ann. N. Y. Acad. Sci. 1969, 162, 947–962. [Google Scholar] [CrossRef]
- Wallach, S.; Dimich, A. Radiomagnesium turnover studies in hypomagnesemic states. Ann. N. Y. Acad. Sci. 1969, 162, 963–972. [Google Scholar] [CrossRef]
- Wolfe, S.M.; Victor, M. The relationship of hypomagnesemia and alkalosis to alcohol withdrawal symptoms. Ann. N. Y. Acad. Sci. 1969, 162, 973–984. [Google Scholar] [CrossRef]
- Lim, P.; Jacob, E. Magnesium status of alcoholic patients. Metabolism 1972, 21, 1045–1051. [Google Scholar] [CrossRef]
- Cohen, L.; Laor, A.; Kitzes, R. Lymphocyte and bone magnesium in alcohol-associated osteoporosis. Magnesium 1985, 4, 148–152. [Google Scholar]
- Flink, E.B. Magnesium deficiency in alcoholism. Alcohol. Clin. Exp. Res. 1986, 10, 590–594. [Google Scholar] [CrossRef] [PubMed]
- De Marchi, S.; Cecchin, E.; Basile, A.; Bertotti, A.; Nardini, R.; Bartoli, E. Renal tubular dysfunction in chronic alcohol abuse—Effects of abstinence. N. Engl. J. Med. 1993, 329, 1927–1934. [Google Scholar] [CrossRef] [PubMed]
- Elisaf, M.; Merkouropoulos, M.; Tsianos, E.V.; Siamopoulos, K.C. Acid-base and electrolyte abnormalities in alcoholic patients. Miner. Electrolyte Metab. 1994, 20, 274–281. [Google Scholar]
- Denison, H.; Jern, S.; Jagenburg, R.; Wendestam, C.; Wallerstedt, S. Influence of increased adrenergic activity and magnesium depletion on cardiac rhythm in alcohol withdrawal. Br. Heart J. 1994, 72, 554–560. [Google Scholar] [CrossRef]
- Wu, C.; Kenny, M.A. Circulating total and ionized magnesium after ethanol ingestion. Clin. Chem. 1996, 42, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Hristova, E.N.; Rehak, N.N.; Cecco, S.; Ruddel, M.; Herion, D.; Eckardt, M.; Linnoila, M.; Elin, R.J. Serum ionized magnesium in chronic alcoholism: Is it really decreased? Clin. Chem. 1997, 43, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Princi, T.; Artero, M.; Malusà, N.; Uxa, L.; Livia, V.; Reina, G. Serum and intracellular magnesium concentrations in intoxicated chronic alcoholic and control subjects. Drug Alcohol. Depend. 1997, 46, 119–122. [Google Scholar] [CrossRef]
- Elisaf, M.; Bairaktari, E.; Kalaitzidis, R.; Siamopoulos, K.C. Hypomagnesemia in alcoholic patients. Alcohol. Clin. Exp. Res. 1998, 22, 134. [Google Scholar] [CrossRef]
- Saha, H.; Harmoinen, A.; Karvonen, A.L.; Mustonen, J.; Pasternack, A. Serum ionized versus total magnesium in patients with intestinal or liver disease. Clin. Chem. Lab. Med. 1998, 36, 715–718. [Google Scholar] [CrossRef]
- Rahelić, D.; Kujundzić, M.; Romić, Z.; Brkić, K.; Petrovecki, M. Serum concentration of zinc, copper, manganese and magnesium in patients with liver cirrhosis. Coll. Antropol. 2006, 30, 523–528. [Google Scholar]
- Ordak, M.; Maj-Zurawska, M.; Matsumoto, H.; Bujalska-Zadrozny, M.; Kieres-Salomonski, I.; Nasierowski, T.; Muszynska, E.; Wojnar, M. Ionized magnesium in plasma and erythrocytes for the assessment of low magnesium status in alcohol dependent patients. Drug Alcohol. Depend. 2017, 178, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Rooney, M.R.; Rudser, K.D.; Alonso, A.; Harnack, L.; Saenger, A.K.; Lutsey, P.L. Circulating ionized magnesium: Comparisons with circulating total magnesium and the response to magnesium supplementation in a randomized controlled trial. Nutrients 2020, 12, 263. [Google Scholar] [CrossRef]
- Khaderi, S.A. Alcohol and alcoholism. Clin. Liver Dis. 2019, 23, 1–10. [Google Scholar] [CrossRef]
- Green, P.H. Alcohol, nutrition and malabsorption. Clin. Gastroenterol. 1983, 12, 563–574. [Google Scholar] [CrossRef]
- Glória, L.; Cravo, M.; Camilo, M.E.; Resende, M.; Cardoso, J.N.; Oliveira, A.G.; Leitão, C.N.; Mira, F.C. Nutritional deficiencies in chronic alcoholics: Relation to dietary intake and alcohol consumption. Am. J. Gastroenterol. 1997, 92, 485–489. [Google Scholar]
- Kourkoumpetis, T.; Sood, G. Pathogenesis of alcoholic liver disease: An update. Clin. Liver Dis. 2019, 23, 71–80. [Google Scholar] [CrossRef]
- Haber, P.S.; Kortt, N.C. Alcohol use disorder and the gut. Addiction 2021, 116, 658–667. [Google Scholar] [CrossRef]
- Nikaki, K.; Gupte, G.L. Assessment of intestinal malabsorption. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 225–325. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on dietary reference values for magnesium. EFSA J. 2015, 13, 4186. [Google Scholar] [CrossRef]
- Reddy, S.T.; Soman, S.S.; Yee, J. Magnesium balance and measurement. Adv. Chronic Kidney Dis. 2018, 25, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Airagnes, G.; Ducoutumany, G.; Laffy-Beaufils, B.; Le Faou, A.L.; Limosin, F. Alcohol withdrawal syndrome management: Is there anything new? Rev. Med. Interne 2019, 40, 373–379. [Google Scholar] [CrossRef]
- Maguire, D.; Talwar, D.; Burns, A.; Catchpole, A.; Stefanowicz, F.; Robson, G.; Ross, D.P.; Young, D.; Ireland, A.; Forrest, E.; et al. A prospective evaluation of thiamine and magnesium status in relation to clinicopathological characteristics and 1-year mortality in patients with alcohol withdrawal syndrome. J. Transl. Med. 2019, 17, 384. [Google Scholar] [CrossRef]
- Maguire, D.; Ross, D.P.; Talwar, D.; Forrest, E.; Naz Abbasi, H.; Leach, J.P.; Woods, M.; Zhu, L.Y.; Dickson, S.; Kwok, T.; et al. Low serum magnesium and 1-year mortality in alcohol withdrawal syndrome. Eur. J. Clin. Investig. 2019, 49, e13152. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vanoni, F.O.; Milani, G.P.; Agostoni, C.; Treglia, G.; Faré, P.B.; Camozzi, P.; Lava, S.A.G.; Bianchetti, M.G.; Janett, S. Magnesium Metabolism in Chronic Alcohol-Use Disorder: Meta-Analysis and Systematic Review. Nutrients 2021, 13, 1959. https://doi.org/10.3390/nu13061959
Vanoni FO, Milani GP, Agostoni C, Treglia G, Faré PB, Camozzi P, Lava SAG, Bianchetti MG, Janett S. Magnesium Metabolism in Chronic Alcohol-Use Disorder: Meta-Analysis and Systematic Review. Nutrients. 2021; 13(6):1959. https://doi.org/10.3390/nu13061959
Chicago/Turabian StyleVanoni, Flora O., Gregorio P. Milani, Carlo Agostoni, Giorgio Treglia, Pietro B. Faré, Pietro Camozzi, Sebastiano A. G. Lava, Mario G. Bianchetti, and Simone Janett. 2021. "Magnesium Metabolism in Chronic Alcohol-Use Disorder: Meta-Analysis and Systematic Review" Nutrients 13, no. 6: 1959. https://doi.org/10.3390/nu13061959
APA StyleVanoni, F. O., Milani, G. P., Agostoni, C., Treglia, G., Faré, P. B., Camozzi, P., Lava, S. A. G., Bianchetti, M. G., & Janett, S. (2021). Magnesium Metabolism in Chronic Alcohol-Use Disorder: Meta-Analysis and Systematic Review. Nutrients, 13(6), 1959. https://doi.org/10.3390/nu13061959









