Dietary Diversity and Healthy Aging: A Prospective Study
Abstract
1. Introduction
2. Materials and Methods
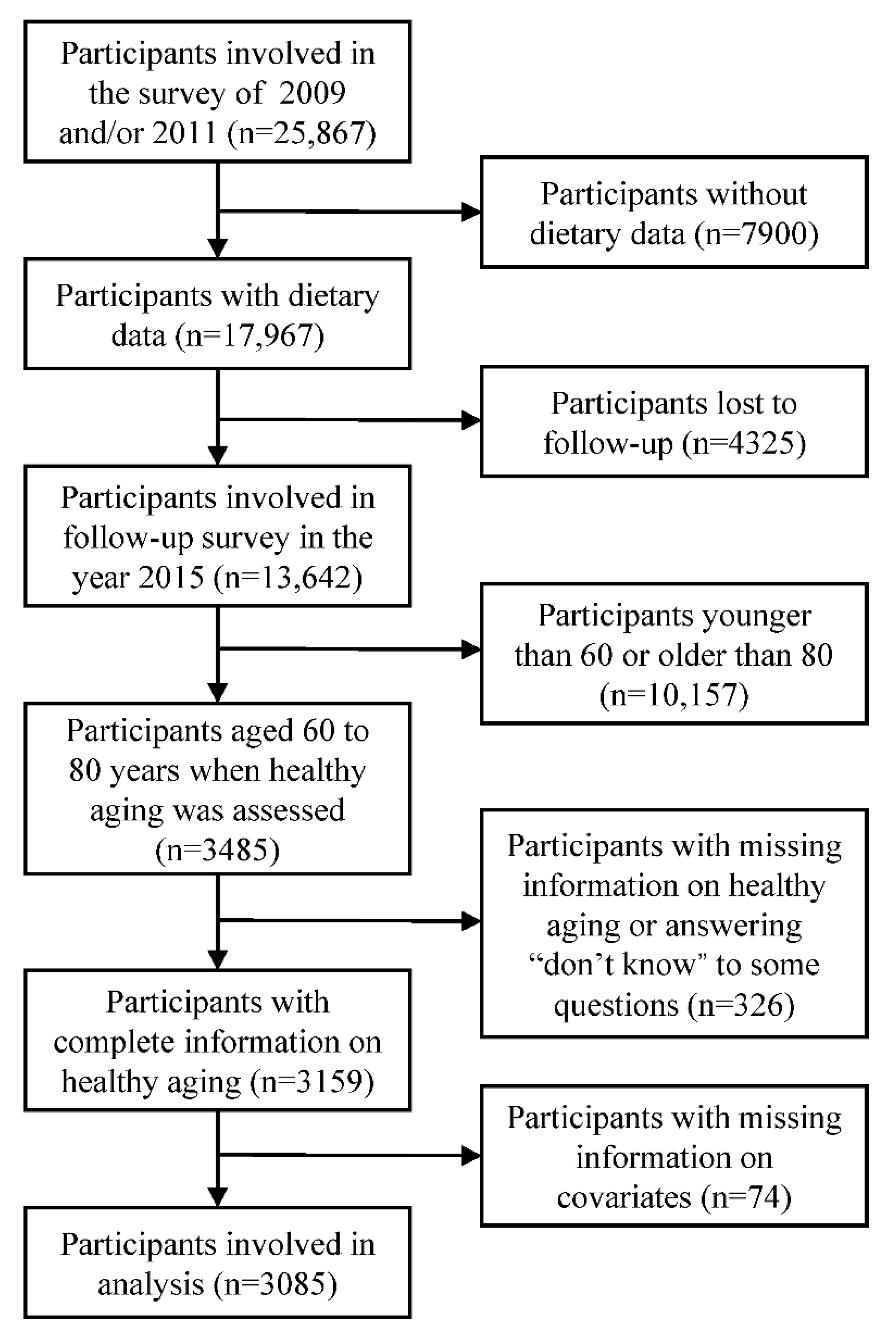
2.1. Study Design and Study Population
2.2. Dietary Survey and Dietary Diversity Score
2.3. Healthy Aging Score
2.4. Self-Reported Life Quality
2.5. Covariates
2.6. Statistics
3. Results
3.1. Baseline Characteristics
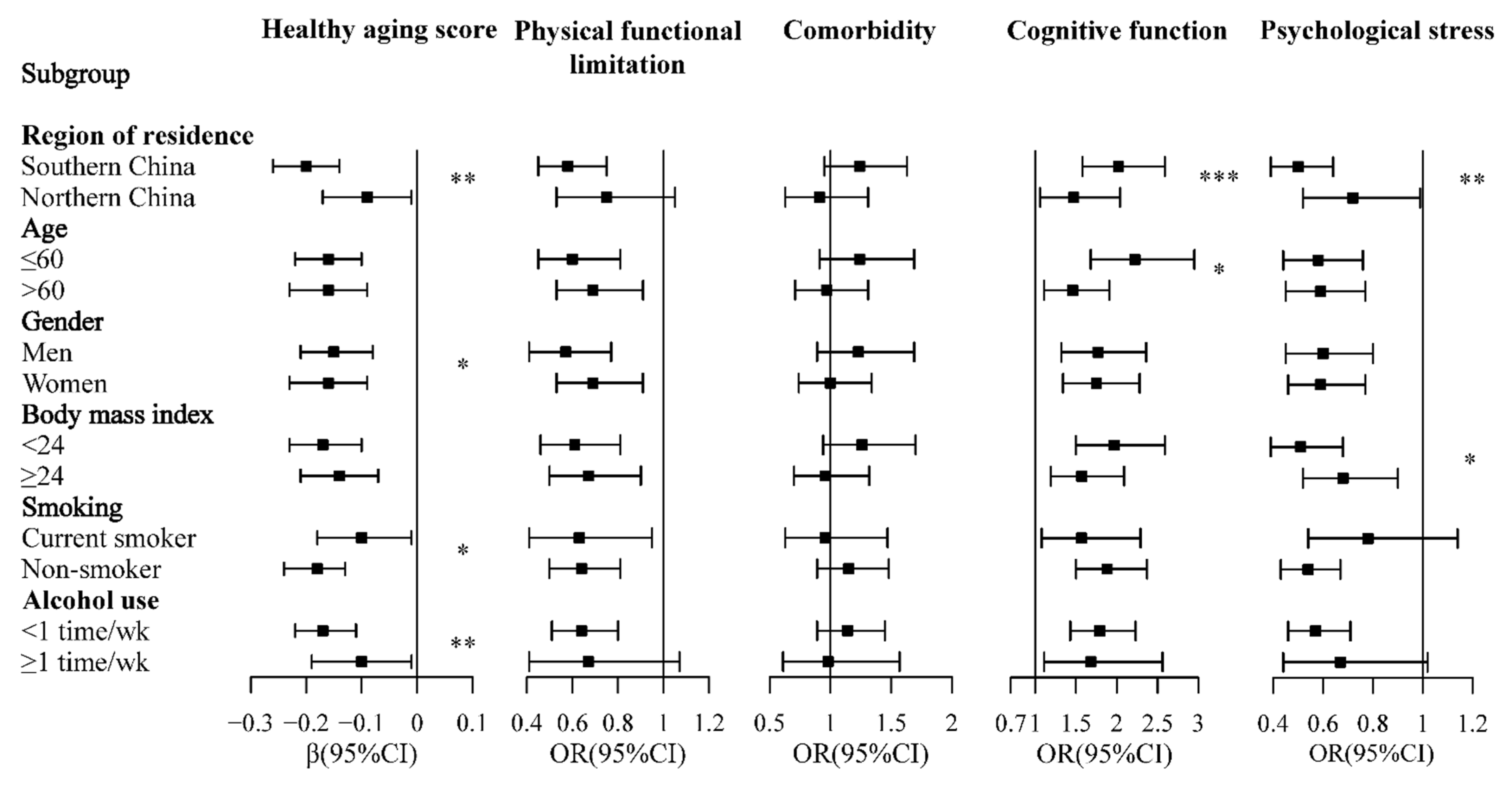
3.2. Dietary Diversity Score and Healthy Aging
3.3. Food Group Consumption and Healthy Aging
3.4. Healthy Aging Score and Self-Reported Life Quality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations, Department of Economic and Social Affairs, Population Division (2020). World Population Ageing 2019 (ST/ESA/SER.A/444). Available online: https://www.un.org/en/development/desa/population/publications/pdf/ageing/WorldPopulationAgeing2019-Report.pdf (accessed on 4 October 2020).
- National Bureau of Statistics. Main Data of the Seventh National Census of China. Available online: http://www.stats.gov.cn/tjsj/zxfb/202105/t20210510_1817176.html (accessed on 20 May 2021).
- Fang, E.F.; Xie, C.; Schenkel, J.A.; Wu, C.; Long, Q.; Cui, H.; Aman, Y.; Frank, J.; Liao, J.; Zou, H.; et al. A research agenda for ageing in China in the 21st century (2nd edition): Focusing on basic and translational research, long-term care, policy and social networks. Ageing Res. Rev. 2020, 64, 101174. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Qian, Y.; Dong, Y.; Yang, Z.; Guo, L.; Liu, J.; Shen, Q.; Wang, L. Patterns and changes in life expectancy in China, 1990-2016. PLoS ONE 2020, 15, e0231007. [Google Scholar] [CrossRef]
- Kontis, V.; Bennett, J.E.; Mathers, C.D.; Li, G.; Foreman, K.; Ezzati, M. Future life expectancy in 35 industrialised countries: Projections with a Bayesian model ensemble. Lancet 2017, 389, 1323–1335. [Google Scholar] [CrossRef]
- Salomon, J.A.; Wang, H.; Freeman, M.K.; Vos, T.; Flaxman, A.D.; Lopez, A.D.; Murray, C.J. Healthy life expectancy for 187 countries, 1990–2010: A systematic analysis for the Global Burden Disease Study 2010. Lancet 2012, 380, 2144–2162. [Google Scholar] [CrossRef]
- Zhou, M.G.; Li, Y.C.; Wang, H.D.; Zeng, X.Y.; Wang, L.J.; Liu, S.W.; Liu, Y.N.; Liang, X.F. Analysis on life expectancy and healthy life expectancy in China, 1990-2015. Chin. J. Epidemiol. 2016, 37, 1439–1443. [Google Scholar] [CrossRef]
- Havighurst, R.J. Successful Aging1. Gerontol. 1961, 1, 8–13. [Google Scholar] [CrossRef]
- Aronson, L. Healthy Aging Across the Stages of Old Age. Clin. Geriatr. Med. 2020, 36, 549–558. [Google Scholar] [CrossRef]
- Anton, S.D.; Woods, A.J.; Ashizawa, T.; Barb, D.; Buford, T.W.; Carter, C.S.; Clark, D.J.; Cohen, R.A.; Corbett, D.B.; Cruz-Almeida, Y.; et al. Successful aging: Advancing the science of physical independence in older adults. Ageing Res. Rev. 2015, 24, 304–327. [Google Scholar] [CrossRef] [PubMed]
- Fallon, C.K.; Karlawish, J. Is the WHO Definition of Health Aging Well? Frameworks for “Health” After Three Score and Ten. Am. J. Public Health 2019, 109, 1104–1106. [Google Scholar] [CrossRef]
- World Health Organization. World Report on Ageing and Health 2015. Available online: https://www.who.int/ageing/events/world-report-2015-launch/en/ (accessed on 10 September 2020).
- Young, Y.; Frick, K.D.; Phelan, E.A. Can Successful Aging and Chronic Illness Coexist in the Same Individual? A Multidimensional Concept of Successful Aging. J. Am. Med Dir. Assoc. 2009, 10, 87–92. [Google Scholar] [CrossRef]
- Young, Y.; Fan, M.-Y.; Parrish, J.M.; Frick, K.D. Validation of a Novel Successful Aging Construct. J. Am. Med Dir. Assoc. 2009, 10, 314–322. [Google Scholar] [CrossRef]
- Black, M.; Bowman, M. Nutrition and Healthy Aging. Clin. Geriatr. Med. 2020, 36, 655–669. [Google Scholar] [CrossRef]
- Ademowo, O.S.; Dias, H.K.I.; Pararasa, C.; Griffiths, H.R. Chapter 6—Nutritional Hormesis in a Modern Environment. In The Science of Hormesis in Health and Longevity; Rattan, S.I.S., Kyriazis, M., Eds.; Academic Press: London, UK, 2019; pp. 75–86. ISBN 978-0-12-814253-0. [Google Scholar]
- Milte, C.M.; McNaughton, S.A. Dietary patterns and successful ageing: A systematic review. Eur. J. Nutr. 2016, 55, 423–450. [Google Scholar] [CrossRef] [PubMed]
- United Nations, Food and Agriculture Organization, Nutrition and Consumer Protection Division. Guidelines for Measuring Household and Individual Dietary Diversity. Available online: http://www.fao.org/publications/card/en/c/5aacbe39-068f-513b-b17d-1d92959654ea/ (accessed on 19 November 2019).
- World Health Organization. Healthy Diet. Available online: https://www.who.int/news-room/fact-sheets/detail/healthy-diet (accessed on 22 April 2021).
- Chinese Nutrition Society. Dietary Guidelines for Chinese (2016); People’s Medical Publishing House Co., Ltd.: Beijing, China, 2016; ISBN 978-7-117-22214-3. [Google Scholar]
- Zhang, J.; Zhao, A.; Wu, W.; Yang, C.; Ren, Z.; Wang, M.; Wang, P.; Zhang, Y. Dietary Diversity Is Associated With Memory Status in Chinese Adults: A Prospective Study. Front. Aging Neurosci. 2020, 12, 580760. [Google Scholar] [CrossRef]
- Tao, L.; Xie, Z.; Huang, T. Dietary Diversity and All-Cause Mortality among Chinese Adults Aged 65 or Older: A Community-Based Cohort Study. Asia Pac J Clin Nutr. 2020, 29, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, R.; Tange, C.; Nishita, Y.; Kato, Y.; Tomida, M.; Imai, T.; Ando, F.; Shimokata, H. Dietary Diversity and All-Cause and Cause-Specific Mortality in Japanese Community-Dwelling Older Adults. Nutrients 2020, 12, 1052. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Du, S.; Zhai, F.; Zhang, B. Cohort Profile: The China Health and Nutrition Survey--monitoring and understanding socio-economic and health change in China, 1989-2011. Int. J. Epidemiol. 2009, 39, 1435–1440. [Google Scholar] [CrossRef]
- Zhai, F.; Guo, X.; Popkin, B.M.; Ma, L.; Wang, Q.; Yu, W.; Jin, S.; Ge, K. Evaluation of the 24-Hour Individual Recall Method in China. Food Nutr. Bull. 1996, 17, 1–17. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, G.; Pan, X. China Food Composition (Book 1), 2nd ed.; Peking University Medical Press: Beijing, China, 2009. [Google Scholar]
- Yang, Y. China Food Composition (Book 2); Peking University Medical Press: Beijing, China, 2004. [Google Scholar]
- Qin, B.; Adair, L.S.; Plassman, B.L.; Batis, C.; Edwards, L.J.; Popkin, B.M.; Mendez, M.A. Dietary Patterns and Cognitive Decline Among Chinese Older Adults. Epidemiology 2015, 26, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y.; Lam, T.-H.; Chan, S.S. Three versions of Perceived Stress Scale: Validation in a sample of Chinese cardiac patients who smoke. BMC Public Health 2010, 10, 513–517. [Google Scholar] [CrossRef]
- Downey, R.G.; King, C.V. Missing Data in Likert Ratings: A Comparison of Replacement Methods. J. Gen. Psychol. 1998, 125, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Venables, W.; Ripley, B. Modern Applied Statistics New York; Fourth, S., Ed.; Springer: New York, NY, USA, 2002; ISBN 978-0-387-21706-2. [Google Scholar]
- Zeileis, A.; Hothorn, T. Diagnostic Checking in Regression Relationships. R News 2002, 2, 7–10. Available online: https://cran.r-project.org/web/packages/lmtest/vignettes/lmtest-intro.pdf (accessed on 15 March 2021).
- Zheng, J.; Zhou, R.; Li, F.; Chen, L.; Wu, K.; Huang, J.; Liu, H.; Huang, Z.; Xu, L.; Yuan, Z.; et al. Association between dietary diversity and cognitive impairment among the oldest-old: Findings from a nationwide cohort study. Clin. Nutr. 2021, 40, 1452–1462. [Google Scholar] [CrossRef]
- Otsuka, R.; Nishita, Y.; Tange, C.; Tomida, M.; Kato, Y.; Nakamoto, M.; Imai, T.; Ando, F.; Shimokata, H. Dietary diversity decreases the risk of cognitive decline among Japanese older adults. Geriatr. Gerontol. Int. 2016, 17, 937–944. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishi, M.; Murayama, H.; Amano, H.; Taniguchi, Y.; Nofuji, Y.; Narita, M.; Matsuo, E.; Seino, S.; Kawano, Y.; et al. Association of dietary variety with body composition and physical function in community-dwelling elderly Japanese. J. Nutr. Heal. Aging 2015, 20, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B.; et al. The Loss of Skeletal Muscle Strength, Mass, and Quality in Older Adults: The Health, Aging and Body Composition Study. Journals Gerontol. Ser. A Boil. Sci. Med Sci. 2006, 61, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Kritchevsky, S.B.; Goodpaster, B.H.; Newman, A.B.; Nevitt, M.; Stamm, E.; Harris, T.B. Leg Muscle Mass and Composition in Relation to Lower Extremity Performance in Men and Women Aged 70 to 79: The Health, Aging and Body Composition Study. J. Am. Geriatr. Soc. 2002, 50, 897–904. [Google Scholar] [CrossRef]
- Rantanen, T. Muscle strength, disability and mortality. Scand. J. Med. Sci. Sports 2003, 13, 3–8. [Google Scholar] [CrossRef]
- Evans, W.J.; Paolisso, G.; Abbatecola, A.M.; Corsonello, A.; Bustacchini, S.; Strollo, F.; Lattanzio, F. Frailty and muscle metabolism dysregulation in the elderly. Biogerontology 2010, 11, 527–536. [Google Scholar] [CrossRef]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Lee, J.S.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B.; et al. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The Health, Aging, and Body Composition (Health ABC) Study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [CrossRef]
- McGrath, R.; Stastny, S.; Casperson, S.; Jahns, L.; Roemmich, J.; Hackney, K.J. Daily Protein Intake and Distribution of Daily Protein Consumed Decreases Odds for Functional Disability in Older Americans. J. Aging Health 2020, 32, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Fávaro-Moreira, N.C.; Krausch-Hofmann, S.; Matthys, C.; Vereecken, C.; Vanhauwaert, E.; Declercq, A.; Bekkering, G.E.; Duyck, J. Risk Factors for Malnutrition in Older Adults: A Systematic Review of the Literature Based on Longitudinal Data. Adv. Nutr. 2016, 7, 507–522. [Google Scholar] [CrossRef] [PubMed]
- Donini, L.M.; Scardella, P.; Piombo, L.; Neri, B.; Asprino, R.; Proietti, A.R.; Carcaterra, S.; Cava, E.; Cataldi, S.; Cucinotta, D.; et al. Malnutrition in elderly: Social and economic determinants. J. Nutr. Health Aging 2013, 17, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the elderly: A narrative review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Cano-Ibáñez, N.; Gea, A.; Martínez-González, M.A.; Salas-Salvadó, J.; Corella, D.; Zomeño, M.D.; Romaguera, D.; Vioque, J.; Aros, F.; Warnberg, J.; et al. Dietary Diversity and Nutritional Adequacy among an Older Spanish Population with Metabolic Syndrome in the PREDIMED-Plus Study: A Cross-Sectional Analysis. Nutrients 2019, 11, 958. [Google Scholar] [CrossRef] [PubMed]
- Oldewage-Theron, W.H.; Kruger, R. Food Variety and Dietary Diversity as Indicators of the Dietary Adequacy and Health Status of an Elderly Population in Sharpeville, South Africa. J. Nutr. Elder. 2008, 27, 101–133. [Google Scholar] [CrossRef]
- Meng, S.J.; Yu, L.J. Oxidative Stress, Molecular Inflammation and Sarcopenia. Int. J. Mol. Sci. 2010, 11, 1509–1526. [Google Scholar] [CrossRef] [PubMed]
- Domazetovic, V. Oxidative stress in bone remodeling: Role of antioxidants. Clin. Cases Miner. Bone Metab. 2017, 14, 209–216. [Google Scholar] [CrossRef]
- Mulero, J.; Zafrilla, P.; Martinez-Cacha, A. Oxidative stress, frailty and cognitive decline. J. Nutr. Health Aging 2011, 15, 756–760. [Google Scholar] [CrossRef]
- Sui, S.X.; Williams, L.J.; Holloway-Kew, K.L.; Hyde, N.K.; Pasco, J.A. Skeletal Muscle Health and Cognitive Function: A Narrative Review. Int. J. Mol. Sci. 2020, 22, 255. [Google Scholar] [CrossRef]
- Welch, A.A. Nutritional Influences on Age-Related Skeletal Muscle Loss. Proc. Nutr. Soc. 2014, 73, 16–33. [Google Scholar] [CrossRef]
- Kim, S.; Jazwinski, S.M. The Gut Microbiota and Healthy Aging: A Mini-Review. Gerontology 2018, 64, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Mello, A.M.; Paroni, G.; Daragjati, J.; Pilotto, A. Gastrointestinal Microbiota and Their Contribution to Healthy Aging. Dig. Dis. 2016, 34, 194–201. [Google Scholar] [CrossRef]
- Heiman, M.L.; Greenway, F.L. A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 2016, 5, 317–320. [Google Scholar] [CrossRef]
- Sanchez-Morate, E.; Gimeno-Mallench, L.; Stromsnes, K.; Sanz-Ros, J.; Román-Domínguez, A.; Parejo-Pedrajas, S.; Inglés, M.; Olaso, G.; Gambini, J.; Mas-Bargues, C. Relationship between Diet, Microbiota, and Healthy Aging. Biomed. 2020, 8, 287. [Google Scholar] [CrossRef]
- Richter-Levin, G.; Xu, L. How could stress lead to major depressive disorder? IBRO Rep. 2018, 4, 38–43. [Google Scholar] [CrossRef]
- Lim, L.L.; Chang, W.; Yu, X.; Chiu, H.; Chong, M.-Y.; Kua, E.-H. Depression in Chinese elderly populations. Asia-Pacific Psychiatry 2011, 3, 46–53. [Google Scholar] [CrossRef]
- Novak, M.; Björck, L.; Giang, K.W.; Heden-Ståhl, C.; Wilhelmsen, L.; Rosengren, A. Perceived stress and incidence of Type 2 diabetes: A 35-year follow-up study of middle-aged Swedish men. Diabet. Med. 2012, 30, e8–e16. [Google Scholar] [CrossRef] [PubMed]
- Steptoe, A.; Kivimäki, M. Stress and cardiovascular disease. Nat. Rev. Cardiol. 2012, 9, 360–370. [Google Scholar] [CrossRef]
- Bakkeli, N. Older Adults’ Mental Health in China: Examining the Relationship Between Income Inequality and Subjective Wellbeing Using Panel Data Analysis. J. Happiness Stud. 2020, 21, 1349–1383. [Google Scholar] [CrossRef]
- Chen, S.; Geldsetzer, P.; Bärnighausen, T. The causal effect of retirement on stress in older adults in China: A regression discontinuity study. SSM Popul. Health 2020, 10, 100462. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D.; Moazzami, K.; Wittbrodt, M.T.; Nye, J.A.; Lima, B.B.; Gillespie, C.F.; Rapaport, M.H.; Pearce, B.D.; Shah, A.J.; Vaccarino, V. Diet, Stress and Mental Health. Nutrients 2020, 12, 2428. [Google Scholar] [CrossRef] [PubMed]
- Wade, A.T.; Davis, C.R.; Dyer, K.A.; Hodgson, J.M.; Woodman, R.J.; Keage, H.A.D.; Murphy, K.J. A Mediterranean diet supplemented with dairy foods improves mood and processing speed in an Australian sample: Results from the MedDairy randomized controlled trial. Nutr. Neurosci. 2020, 23, 646–658. [Google Scholar] [CrossRef]
- Parletta, N.; Zarnowiecki, D.; Cho, J.; Wilson, A.; Bogomolova, S.; Villani, A.; Itsiopoulos, C.; Niyonsenga, T.; Blunden, S.; Meyer, B.; et al. A Mediterranean-style dietary intervention supplemented with fish oil improves diet quality and mental health in people with depression: A randomized controlled trial (HELFIMED). Nutr. Neurosci. 2019, 22, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Abshirini, M.; Siassi, F.; Koohdani, F.; Qorbani, M.; Mozaffari, H.; Aslani, Z.; Soleymani, M.; Entezarian, M.; Sotoudeh, G. Dietary total antioxidant capacity is inversely associated with depression, anxiety and some oxidative stress biomarkers in postmenopausal women: A cross-sectional study. Ann. Gen. Psychiatry 2019, 18, 3. [Google Scholar] [CrossRef]
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut Microbiota’s Effect on Mental Health: The Gut-Brain Axis. Clin. Pr. 2017, 7, 131–136. [Google Scholar] [CrossRef]
- Roberts, R.O.; Roberts, L.A.; Geda, Y.E.; Cha, R.H.; Pankratz, V.S.; O’Connor, H.M.; Knopman, D.S.; Petersen, R.C. Relative Intake of Macronutrients Impacts Risk of Mild Cognitive Impairment or Dementia. J. Alzheimer’s Dis. 2012, 32, 329–339. [Google Scholar] [CrossRef]
- Shang, X.; Hill, E.; Li, Y.; He, M. Energy and macronutrient intakes at breakfast and cognitive declines in community-dwelling older adults: A 9-year follow-up cohort study. Am. J. Clin. Nutr. 2021, 113, 1093–1103. [Google Scholar] [CrossRef]
- Sangsefidi, Z.S.; Salehi-Abarghouei, A.; Sangsefidi, Z.S.; Mirzaei, M.; Hosseinzadeh, M. The relation between low carbohydrate diet score and psychological disorders among Iranian adults. Nutr. Metab. 2021, 18, 1–9. [Google Scholar] [CrossRef]
- Daneshzad, E.; Keshavarz, S.; Qorbani, M.; Larijani, B.; Azadbakht, L. Association between a low-carbohydrate diet and sleep status, depression, anxiety, and stress score. J. Sci. Food Agric. 2020, 100, 2946–2952. [Google Scholar] [CrossRef]
- Ozguner, F.; Koyu, A.; Cesur, G. Active smoking causes oxidative stress and decreases blood melatonin levels. Toxicol. Ind. Health 2005, 21, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Doggui, R.; Elsawy, W.; Conti, A.A.; Baldacchino, A. Association between chronic psychoactive substances use and systemic inflammation: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2021, 125, 208–220. [Google Scholar] [CrossRef] [PubMed]
- West, R. Tobacco smoking: Health impact, prevalence, correlates and interventions. Psychol. Health 2017, 32, 1018–1036. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Zhang, M.; Luo, X.; Zhao, J.; Yin, L.; Pang, C.; Feng, T.; Wang, S.; Wang, B.; Zhang, H.; et al. Secular trend of the leading causes of death in China from 2003 to 2013. Afr. Health Sci. 2017, 17, 532–537. [Google Scholar] [CrossRef][Green Version]
- Griswold, M.G.; Fullman, N.; Hawley, C.; Arian, N.; Zimsen, S.R.M.; Tymeson, H.D.; Venkateswaran, V.; Tapp, A.D.; Forouzanfar, M.H.; Salama, J.S.; et al. Alcohol use and burden for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2018, 392, 1015–1035. [Google Scholar] [CrossRef]
- Jung, M.-H.; Shin, E.-S.; Ihm, S.-H.; Jung, J.-G.; Lee, H.-Y.; Kim, C.-H. The effect of alcohol dose on the development of hypertension in Asian and Western men: Systematic review and meta-analysis. Korean J. Intern. Med. 2020, 35, 906–916. [Google Scholar] [CrossRef]
- Park, H.; Shin, S.K.; Joo, I.; Song, D.S.; Jang, J.W.; Park, J.-W. Systematic Review with Meta-Analysis: Low-Level Alcohol Consumption and the Risk of Liver Cancer. Gut Liver 2020, 14, 792–807. [Google Scholar] [CrossRef]
- Scherübl, H. Alcohol Use and Gastrointestinal Cancer Risk. Visc. Med. 2020, 36, 175–181. [Google Scholar] [CrossRef]
- Tang, D.; Bu, T.; Feng, Q.; Liu, Y.; Dong, X. Differences in Overweight and Obesity between the North and South of China. Am. J. Health Behav. 2020, 44, 780–793. [Google Scholar] [CrossRef]
- Wu, J.; Cheng, X.; Qiu, L.; Xu, T.; Zhu, G.; Han, J.; Xia, L.; Qin, X.; Cheng, Q.; Liu, Q. Prevalence and Clustering of Major Cardiovascular Risk Factors in China. Medicine 2016, 95, e2712. [Google Scholar] [CrossRef]
- SalehiAbargouei, A.; Akbari, F.; Bellissimo, N.; Azadbakht, L. Dietary diversity score and obesity: A systematic review and meta-analysis of observational studies. Eur. J. Clin. Nutr. 2016, 70, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Qorbani, M.; Mahdavi-Gorabi, A.; Khatibi, N.; Ejtahed, H.-S.; Khazdouz, M.; Djalalinia, S.; Sahebkar, A.; Esmaeili-Abdar, M.; Hasani, M. Dietary diversity score and cardio-metabolic risk factors: An updated systematic review and meta-analysis. Eat. Weight. Disord. Stud. Anorex. Bulim. Obes. 2021, 1–16. [Google Scholar] [CrossRef]
| Variables | Dietary Diversity Score a | p | ||
|---|---|---|---|---|
| T1 | T2 | T3 | ||
| Number of participants | 991 | 1076 | 1018 | |
| Age (year) | 62.1(5.6) | 61.6(5.5) | 61.9(5.6) | 0.088 |
| Body mass index (kg/m2) | 23.3(3.8) | 23.9(3.4) | 24.4(3.2) | <0.001 |
| Gender | 0.166 | |||
| Men | 44.1 | 48.2 | 46.7 | |
| Women | 55.9 | 51.8 | 53.3 | |
| Region of residence | <0.001 | |||
| Southern China | 60.9 | 69.1 | 63.0 | |
| Northern China | 39.1 | 30.9 | 37.0 | |
| Residency | <0.001 | |||
| Rural | 84.2 | 65.1 | 36.1 | |
| Urban | 15.8 | 34.9 | 63.9 | |
| Education | <0.001 | |||
| Primary school and below | 80.7 | 61.9 | 35.1 | |
| Middle school and above | 19.3 | 38.1 | 64.9 | |
| Per capita household income | <0.001 | |||
| Low | 54.0 | 32.4 | 11.8 | |
| Middle | 31.1 | 37.7 | 25.3 | |
| High | 14.9 | 29.9 | 62.9 | |
| Marriage status | <0.001 | |||
| Married | 83.9 | 88.4 | 90.2 | |
| Others | 16.1 | 11.6 | 9.8 | |
| Smoking | 0.001 | |||
| Current smoker | 30.1 | 28.0 | 23.1 | |
| Non-smoker | 69.9 | 72.0 | 76.9 | |
| Alcohol use (times/week) | 0.474 | |||
| <1 | 79.8 | 78.0 | 77.8 | |
| ≥1 | 20.2 | 22.0 | 22.2 | |
| Variables | Dietary Diversity Score a | p-Trend | ||
|---|---|---|---|---|
| T1 | T2 | T3 | ||
| Healthy aging score b | ||||
| Crude | Ref | −0.12(−0.16, −0.08) | −0.26(−0.31, −0.22) | <0.001 |
| Model 1 | Ref | −0.06(−0.10, −0.02) | −0.15(−0.20, −0.11) | <0.001 |
| Model 2 | Ref | −0.06(−0.10, −0.02) | −0.16(−0.20, −0.11) | <0.001 |
| Physical functional limitation c | ||||
| Crude | Ref | 0.77(0.65, 0.90) | 0.58(0.49, 0.68) | <0.001 |
| Model 1 | Ref | 0.87(0.73, 1.04) | 0.64(0.52, 0.79) | <0.001 |
| Model 2 | Ref | 0.86(0.73, 1.03) | 0.64(0.52, 0.78) | <0.001 |
| Comorbidity c | ||||
| Crude | Ref | 1.07(0.90, 1.27) | 1.26(1.06, 1.50) | 0.010 |
| Model 1 | Ref | 1.09(0.91, 1.31) | 1.19(0.97, 1.47) | 0.101 |
| Model 2 | Ref | 1.01(0.84, 1.22) | 1.09(0.88, 1.35) | 0.441 |
| Cognitive function c | ||||
| Crude | Ref | 1.58(1.34, 1.85) | 2.88(2.44, 3.39) | <0.001 |
| Model 1 | Ref | 1.25(1.05, 1.48) | 1.80(1.48, 2.19) | <0.001 |
| Model 2 | Ref | 1.23(1.04, 1.46) | 1.77(1.46, 2.15) | <0.001 |
| Psychological stress c | ||||
| Crude | Ref | 0.56(0.48, 0.66) | 0.42(0.36, 0.50) | <0.001 |
| Model 1 | Ref | 0.63(0.53, 0.74) | 0.59(0.48, 0.71) | <0.001 |
| Model 2 | Ref | 0.63(0.53, 0.75) | 0.59(0.49, 0.72) | <0.001 |
| Food Groups | Number of Participants | Median (IQR) | Crude | Adjusted | ||
|---|---|---|---|---|---|---|
| β (95%CI) | p-Trend | β (95%CI) | p-Trend | |||
| Staple foods | <0.001 | 0.060 | ||||
| Low | 1028 | 130.8 (33.2) | Ref | Ref | ||
| Middle | 1028 | 178.2 (20.5) | 0.05 (0.01, 0.10) | 0 (−0.04, 0.04) | ||
| High | 1029 | 228.2 (41.5) | 0.12 (0.08, 0.16) | 0.04 (0, 0.08) | ||
| Soybeans and nuts | 0.180 | 0.995 | ||||
| Low | 1028 | 0 (2.2) | Ref | Ref | ||
| Middle | 1028 | 8.4 (4.1) | −0.05 (−0.09, −0.01) | −0.03 (−0.07, 0.01) | ||
| High | 1029 | 21.2 (13.5) | −0.03 (−0.08, 0.01) | 0 (−0.04, 0.04) | ||
| Vegetables | 0.585 | 0.167 | ||||
| Low | 1028 | 95.8 (33.5) | Ref | Ref | ||
| Middle | 1028 | 149.6 (26.2) | 0 (−0.05, 0.04) | −0.01 (−0.05, 0.02) | ||
| High | 1029 | 225.0 (70.2) | −0.01 (−0.06, 0.03) | −0.03 (−0.07, 0.01) | ||
| Fruits | <0.001 | <0.001 | ||||
| Non-consumer | 1482 | 0 | Ref | Ref | ||
| Low | 802 | 31.2 (20.2) | −0.10 (−0.14, −0.06) | −0.05 (−0.09, −0.01) | ||
| High | 801 | 88.1 (54.0) | −0.13 (−0.18, −0.09) | −0.09 (−0.13, −0.05) | ||
| Meat and poultry | <0.001 | 0.047 | ||||
| Low | 1028 | 16.1 (24.0) | Ref | Ref | ||
| Middle | 1028 | 42.7 (12.2) | −0.08 (−0.13, −0.04) | −0.04 (−0.08, 0) | ||
| High | 1029 | 76.9 (30.6) | −0.13 (−0.17, −0.08) | −0.04 (−0.09, 0) | ||
| Aquatic products | <0.001 | <0.001 | ||||
| Non-consumer | 1436 | 0 | Ref | Ref | ||
| Low | 824 | 13.8 (8.9) | −0.12 (−0.16, −0.07) | −0.05 (−0.09, −0.01) | ||
| High | 825 | 38.0 (21.7) | −0.17 (−0.21, −0.13) | −0.10 (−0.14, −0.06) | ||
| Eggs | 0.006 | 0.113 | ||||
| Low | 1028 | 0(5.1) | Ref | Ref | ||
| Middle | 1028 | 14.6 (5.7) | −0.02 (−0.06, 0.02) | 0.01 (−0.03, 0.05) | ||
| High | 1029 | 30.7 (15.3) | −0.06 (−0.10, −0.02) | −0.03 (−0.07, 0.01) | ||
| Milk and dairy products | <0.001 | 0.054 | ||||
| Non-consumer | 2493 | 0 | Ref | Ref | ||
| Low | 296 | 45.9 (30.3) | −0.13 (−0.19, −0.06) | −0.08 (−0.14, −0.02) | ||
| High | 296 | 126.2 (65.4) | −0.13 (−0.19, −0.07) | −0.05 (−0.11, 0.01) | ||
| Self-Reported Life Quality | Number of Participants | Crude | Adjusted | ||
|---|---|---|---|---|---|
| OR (95%CI) | p | OR (95%CI) | p | ||
| Fair | 1148 | Ref | Ref | ||
| Good | 1763 | 0.38 (0.32, 0.44) | <0.001 | 0.38 (0.32, 0.45) | <0.001 |
| Poor | 168 | 4.15 (3.05, 5.66) | <0.001 | 4.41 (3.12, 6.25) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Zhao, A. Dietary Diversity and Healthy Aging: A Prospective Study. Nutrients 2021, 13, 1787. https://doi.org/10.3390/nu13061787
Zhang J, Zhao A. Dietary Diversity and Healthy Aging: A Prospective Study. Nutrients. 2021; 13(6):1787. https://doi.org/10.3390/nu13061787
Chicago/Turabian StyleZhang, Jian, and Ai Zhao. 2021. "Dietary Diversity and Healthy Aging: A Prospective Study" Nutrients 13, no. 6: 1787. https://doi.org/10.3390/nu13061787
APA StyleZhang, J., & Zhao, A. (2021). Dietary Diversity and Healthy Aging: A Prospective Study. Nutrients, 13(6), 1787. https://doi.org/10.3390/nu13061787







