Heiner Syndrome and Milk Hypersensitivity: An Updated Overview on the Current Evidence
Abstract
1. Introduction
2. Search Methodology and Results
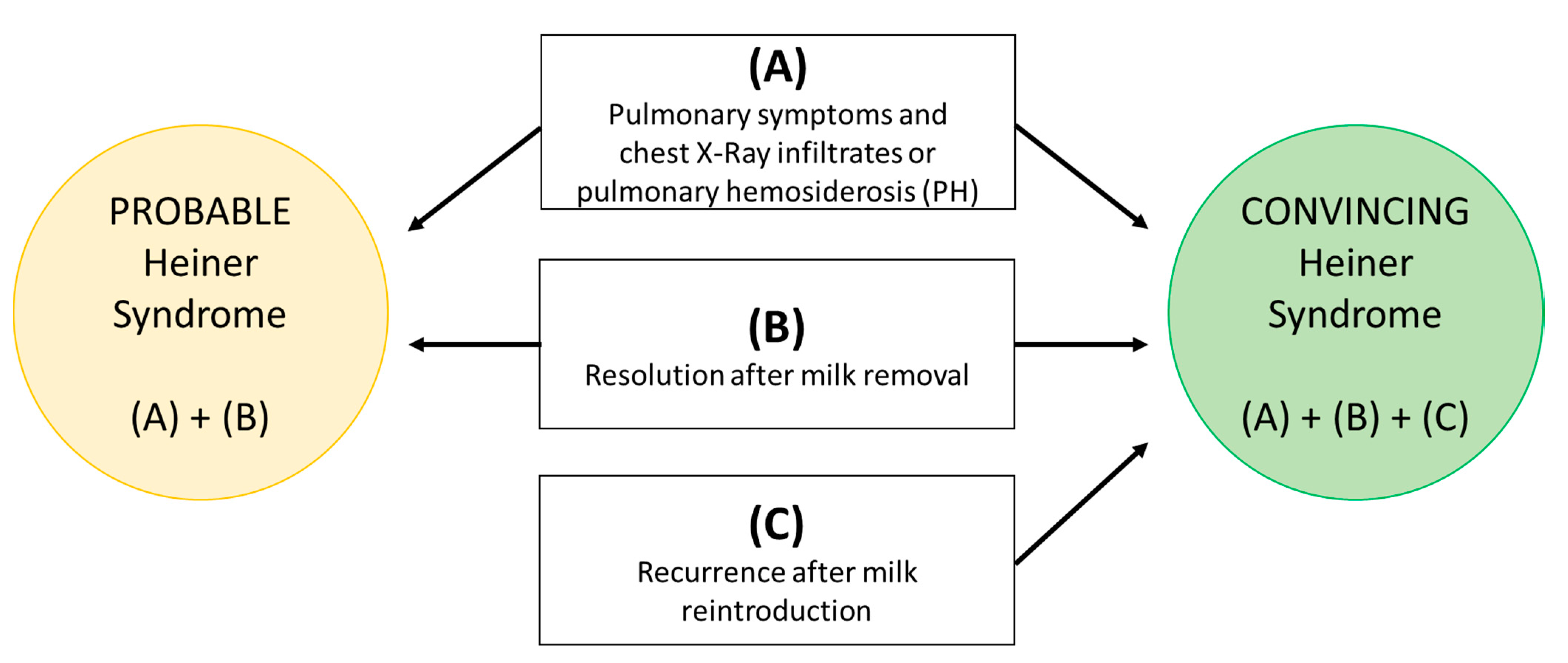
- (A)
- Pulmonary symptoms and XR infiltrates or pulmonary hemosiderosis (PH);
- (B)
- Resolution after milk removal;
- (C)
- Recurrence after milk reintroduction.
- (1)
- Heiner et al., 1960 [3]—In 1960, Heiner [3] (from who the name of the syndrome originates) first reported a chronic respiratory disease associated with multiple CM precipitins in the sera of seven children aged 6 weeks to 17 months. All the patients presented a chronic respiratory disease with maximal severity of clinical manifestations at the time of introduction of raw CM in the diet associated with the other signs and symptoms that define the syndrome, mostly iron deficiency anemia, gastrointestinal signs and symptoms, poor growth and PH documented by gastric or bronchial aspirates. Six out of seven patients overcame their disease by changing the milk content (i.e., milk processing or exclusion of milk proteins) in their diet, by using evaporated or boiled milk (n = 2), extensively hydrolyzed casein formula (n = 1) or soymilk (n = 3). Overall, the milk protein avoidance resulted in the complete disappearance of clinical manifestations and remission of the hematologic status. One patient spontaneously overcame the disease without dietary restriction between 2.5 to 3.5 years (y) of age. When milk was reintroduced after avoidance, two out of six showed a clinical and imaging relapse, and four became tolerant or partially tolerant to milk after intervals without clinical manifestations on a restricted diet ranging from three to six months in duration.Comment: The first description of HS.HSD: Convincing in two cases; probable in four patients.
- (2)
- Holland et al., 1962 [4]—Stimulated by Heiner’s observations, Holland et al. examined serum specimens from 1618 infants and children with the same technique, finding precipitins in 87 of them [4]. Patients of this population showed signs and symptoms suggestive of the syndrome but also different clinical manifestations, such as isolated upper respiratory diseases, hepatosplenomegaly and congenital heart diseases. Only 17 patients were reported to show respiratory signs and symptoms. No data on X-rays were reported. Because of the heterogeneity of the clinical manifestations and the limited number of diagnostic exams in this population, we selected 22/87 patients who improved during the CM diet period. No attempt of reintroduction was performed.Comment: We extrapolated 22 cases with suspected HS from a large and heterogeneous cohort.HSD: Nobody with probable or convincing clinical criteria.
- (3)
- Chang et al., 1969 [5]—Chang described the clinical case of a 9-month-old girl admitted to the hospital for failure to thrive (FTT), anemia and chronic recurrent lung disease starting in the first weeks of age [5]. She underwent a lengthy diagnostic process, until the finding of milk precipitins suggested an HS diagnosis. The patient was then placed on a soymilk diet with clinical resolution. Flare-up signs and symptoms and radiological relapse due to the poor adherence to the diet are mentioned. However, controlled milk reintroduction was not performed.Comment: Single case report. No detailed data are reported about the follow-up.HSD: Probable.
- (4)
- Archer, 1971 [6]—Archer reported the clinical case of a 13-month-old girl with a severe heart failure based on a profound iron deficiency anemia and idiopathic PH, diagnosed by needle biopsy [6]. All of the immunological tests performed in order to investigate a CM sensitization, including serum precipitins, skin prick test (SPT) and immunoglobulin, were negative. Notwithstanding, a milk-free diet was commenced with a good clinical and radiological response. The first relapse during her first week with a childminder was probably due to the inadvertent administration of CM.Comment: The girl was admitted one year before with some symptoms and treated successfully with antibiotics. Results of CM reintroduction doubtful.HSD: Probable.
- (5)
- Boat et al., 1975 [7]—In this study, 6 children with high titers of milk precipitins were identified by screening the sera of 160 children with idiopathic chronic lung disease associated with typical manifestations of milk-induced PH [7]. Elimination of CM from the diet, symptomatic therapy and adenoidectomy (when indicated) resulted in improvement in six out of six patients.Comment: Even if six out of six patients recovered in 5–21 days after milk removal, only one was rechallenged (Patient C) and one (Patient B) developed pneumonia within six months upon CM reintroduction.HSD: Convincing in one case; probable in five patients.
- (6)
- Stafford et al., 1977 [8]—Nine patients with respiratory signs and symptoms and milk precipitins were enrolled in this study in order to elucidate the immunopathologic mechanisms involved in milk-induced PH. No demonstration of a unique immunologic mechanism associated with milk-induced PH in the patients studied [8]. No clinical data about CM withdrawal were reported.Comment: Focus on immunological patho-mechanisms with poor clinical description of the enrolled participants.HSD: Nobody with probable or convincing criteria.
- (7)
- Fossati, 1992 [9]—A 7-year-old girl was admitted to hospital because of anemia, and a PH was diagnosed [9]. Precipitating antibodies were also found. A marked improvement of clinical manifestations and XR results were found after removal of CM from the diet.Comment: Single clinical case report. No data on reintroduction.HSD: Probable.
- (8)
- Torres et al., 1996 [10]—This study provides an interesting immunological overview on the HS based on data from a single clinical case of a girl [10]. Specifically, the authors speculate that an inflammatory response occurred after CM intake in the presented clinical case. A 5-day-old female newborn was admitted in an emergency department because of a severe bradycardia due to a myocarditis associated with assessed PH and anemia. Although the total specific IgE and specific IgG to milk proteins were below the detection limits, the patient underwent two oral food challenges (OFC). During the in vivo tests, hematocrit, histamine, tryptase and ECP (eosinophil cationic protein) in blood and BAL, and N-methyl-histamine (NMH) in urine were measured before and at multiple times during the administration of standard formula (first OFC) and non-milk enteral nutrition (second OFC). When the girl was fed with CM, a remarkable increment of all the tested inflammatory mediators was reported; conversely, the hemoglobin level dropped significantly. Then, the same OFC was done with non-milk enteral nutrition with any variation being registered. These data are unfortunately not supported by clinical and radiologic information. Challenge.Comment: Unique case report of PH in which milk OFC induced an increase of inflammatory mediators suggesting a T cell-mediated pathogenesis of HS.HSD: Probable.
- (9)
- Moissidis et al., 2005 [11]—Moissidis et al. reviewed eight cases of children affected by upper respiratory tract symptoms [11]. All cases presented radiological imaging with pulmonary infiltrates, and one had HP (defined as iron-laden macrophages in the bronchoalveolar lavage, gastric washing and open lung biopsy). Seven out of eight had gastrointestinal symptoms. High titers of precipitating antibodies to CM proteins were demonstrated in six out of six patients studied. However, HS was confirmed by the improvement of the clinical and radiological findings after a CM-free diet and relapse when a reintroduction was attempted in three out of six cases.Comment: The most detailed paper on the topic. However, cases were evaluated at different times and under different circumstances; therefore, specific data were not available for each patient.HSD: Convincing in three cases; probable in five patients.
- (10)
- Sigua et al., 2013 [12]—A 12-month-old boy with multifocal pneumonia that was refractory to protracted antibiotic treatment was suspected to suffer from HS [12]. The clinical history showed that the boy underwent a milk-free diet from the first to the tenth month of age because of suspected non-IgE-mediated CM non-bloody diarrhea. HS appeared at CM reintroduction. Serum-precipitating IgG antibodies to all nine CM protein fractions tested were strongly positive. He underwent a strict soy-based diet from 12 months of age with prompt clinical remission and complete resolution of the previously identified pulmonary opacities at a chest X-ray performed at 14 months.Comment: A single patient with HS after a previous history of non-IgE-mediated CM gastrointestinal symptoms.HSD: Probable.
- (11)
- Yavuz et al., 2014 [13]—A 3-year-old boy was referred to the emergency service with respiratory distress and hemoptysis [13]. Because of iron deficiency anemia, a BAL cytological examination was performed in order to confirm a PH. Precipitins were not determined. The patient overcame the disease through a CM avoidance diet. However, a low dose of both prednisolone and azathioprine was also prescribed. Furthermore, authors described that the patient in the next five years had many relapses because of failure to receive the prescribed medications and poor adherence to the diet. Moreover, during a hemoptysis attack, he showed new symptoms, such as edema, hematuria and hypertension. On this occasion, rapidly progressive glomerulonephritis was diagnosed on the basis of the histopathological findings and treated with a combination of cyclophosphamide and methylprednisolone.Comment: In this case report, an elimination diet and drugs were administrated together for an extended period of time, and during the follow-up the compliance was scarce. Therefore, it is difficult to differentiate the effects of each treatment and the actual cause-effect relationship.HSD: Probable.
- (12)
- Mourad et al., 2015 [14]—The clinical case of a 17-month-old boy with idiopathic PH was described by Mourad et al. [14]. BAL demonstrated an abundance of fresh red blood cells and iron-laden macrophages. The CM-specific IgE level was only slightly elevated (1.42 IU/mL). IgG antibody levels to CM proteins were markedly elevated. In spite of the severity of the clinical conditions (i.e., severe anemia and respiratory failure with acidosis), the strict CM-free diet allowed the boy to overcome the disease. Hydrocortisone was also administrated, but it is not clear when it was introduced and for how long. A relapse was reported because the mother, while on raw CM avoidance, started feeding the patient with baked CM products.Comment: Single clinical case report. Hydrocortisone was also used. Controlled reintroduction not performed.HSD: Probable.
- (13)
- Alsukhon et al., 2017 [15]—A 2-month-old male with recurrent diarrhea and FTT had persistent cough, tachypnea and high inflammatory markers despite antibiotic therapy for pneumonia [15]. An amino acid-based formula gave improvement in inflammation and respiratory function.Comment: Single case report. Milk reintroduction not performed.HSD: Probable.
- (14)
- Ojuawo et al., 2019 [16]—Ojawo et al. described the clinical case of a 16-week-old boy with FTT, dyspnea and anemia who acceded to the emergency department in Nigeria [16]. Neither antibiotic treatment nor sodium citrate, administered for the suspicion of a renal tubular acidosis, modified his condition. Diagnosis of HS was based on the constellation of clinical features, XR results, and subsequent resolution after stopping CM. Parents on a follow-up visit reported occasional cough and rhinitis whenever CM was reintroduced.Comment: Single case report. No controlled CM reintroduction reported.HSD: Probable.
- (15)
- Koc et al., 2019 [17]—A 6-month-old infant with massive hemoptysis, hematemesis and deep anemia was treated for bronchopneumonia four times [17]. When he was admitted to the emergency department, both chest-X ray and computerized tomography documented many lung opacities and hemosiderin-laden macrophages were found in the patient’s fasting stomach fluid examination, confirming the diagnosis of PH. The boy was discharged with a CM-free diet, with complete clinical and radiological recovery.Comment: No laboratory data were reported, and no milk reintroduction test was reported.HSD: Probable.
- (16)
- Liu et al., 2020 [18]—Liu et al. described a 4-month-old boy with a chronic pulmonary syndrome whose main presenting symptom was a persistent hematochezia since the tenth day of life [18]. Gastrointestinal endoscopic biopsy showed granulation tissue infiltrated by acute and chronic inflammatory cells, including some eosinophils. Additionally, in this case, the improvement of both clinical and radiologic findings after the elimination of milk suggested the diagnosis of HS. In addition to the CM elimination diet, the patient was treated with methylprednisolone (1 mg/kg) and montelukast.Comment: Single case report. No milk reintroduction test reported.HSD: Probable.
3. Discussion
3.1. Age at Onset
3.2. Etiology
3.3. Clinical Characteristics
3.4. Pathogenesis and Immunological Implications
3.5. Pulmonary Modifications and Immunofluorescence Studies
3.6. Diagnostic Criteria
3.7. Differential Diagnosis
3.8. Natural History
3.9. Treatment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heiner, D.C.; Sears, J.W.; Kniker, W.T. Multiple precipitins to cow’s milk in chronic respiratory disease. A syndrome including poor growth, gastrointestinal symptoms, evidence of allergy, iron deficiency anemia, and pulmonary hemosiderosis. Am. J. Dis. Child 1962, 103, 634–654. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.K.; Kniker, W.T.; Cook, C.D.; Heiner, D.C. Cow’s milk-induced pulmonary disease in children. Adv. Pediatr. 1978, 25, 39–57. [Google Scholar]
- Heiner, D.C.; Sears, J.W. Chronic respiratory disease associated with multiple circulating precipitins to cow’s milk. Am. J. Dis. Child. 1960, 100, 500–502. [Google Scholar]
- Holland, N.H.; Hong, R.; Davis, N.C.; West, C.D. Significance of precipitating antibodies to milk proteins in the serum of infants and children. J. Pediatr. 1962, 61, 181–195. [Google Scholar] [CrossRef]
- Chang, C.H.; Wittig, H.J. Heiner’s syndrome. Radiology 1969, 92, 507–508. [Google Scholar] [CrossRef]
- Archer, J. Idiopathic Pulmonary Hemosiderosis Treated with a Milk-free Diet. Proc. Roy. Soc. Med. 1971, 64, 1217–1218. [Google Scholar] [CrossRef] [PubMed]
- Boat, T.F.; Polmar, S.H.; Whitman, V.; Kleinerman, J.I.; Stern, R.C.; Doeshuk, C.F. Hyperactivity to cow milk in young children with pulmonary hemosiderosis and cor pulmonale secondary to nasopharyngeal obstruction. J. Pediatr. 1975, 87, 23–29. [Google Scholar] [CrossRef]
- Stafford, H.A.; Polmar, S.H.; Boat, T.F. Immunologic Studies in Cow’s Milk-induced Pulmonary Hemosiderosis. Pediatr. Res. 1977, 11, 898–903. [Google Scholar] [CrossRef]
- Fossati, G.; Perri, M.; Careddu, G.; Mirra, N.; Carnelli, V. Pulmonary hemosiderosis induced by cow’s milk proteins: A discussion of a clinical case. Pediatr. Medica Chir. Med Surg. Pediatrics 1992, 14, 203–207. [Google Scholar]
- Torres, M.J.; Girón, M.D.; Corzo, J.L.; Rodriguez, F.; Moreno, F.; Perez, E.; Blanca, M.; Martinez-Valverde, A. Release of in-flammatory mediators after cow’s milk intake in a newborn with idiopathic pulmonary hemosiderosis. J. Allergy Clin. Immunol. 1996, 98, 1120–1123. [Google Scholar] [CrossRef]
- Moissidis, I.; Chaidaroon, D.; Vichyanond, P.; Bahna, S.L. Milk-induced pulmonary disease in infants (Heiner syndrome). Pediatr. Allergy Immunol. 2005, 16, 545–552. [Google Scholar] [CrossRef]
- Sigua, J.A.; Zacharisen, M. Heiner syndrome mimicking an immune deficiency. WMJ Off. Publ. State Med Soc. Wis. 2013, 112, 215–217. [Google Scholar]
- Yavuz, S.; Karabay-Bayazıt, A.; Yılmaz, M.; Gönlüşen, G.; Anarat, A. Crescentic glomerulonephritis in a child with Heiner syndrome. Turk. J. Pediatr. 2014, 56, 661–664. [Google Scholar] [PubMed]
- Mourad, A.A.; Parekh, H.; Bahna, S.L. A 17-month-old patient with severe anemia and respiratory distress. Allergy Asthma Proc. 2015, 36, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Alsukhon, J.; Leonov, A.; Elisa, A.; Koon, G. P327 Food-induced pulmonary hemosiderosis. Ann. Allergy Asthma Immunol. 2017, 119, S77. [Google Scholar] [CrossRef]
- Ojuawo, A.B.; Ojuawo, O.B.; Aladesanmi, A.O.; Adio, M.O.; Abdulkadir, M.B.; Mokuolu, O.A. Heiner Syndrome: An uncommon cause of failure to thrive. Malawi Med. J. 2019, 31, 227–229. [Google Scholar] [CrossRef]
- Koc, A.S.; Sucu, A.; Celik, U. A different clinical presentation of Heiner syndrome: The case of diffuse alveolar hemorrhage causing massive hemoptysis and hematemesis. Respir. Med. Case Rep. 2019, 26, 206–208. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Huang, X.-R.; Zhang, J.-W.; Xiao, Y.-M.; Zhang, T. Hematochezia in a Child with Heiner Syndrome. Front. Pediatr. 2020, 7, 551. [Google Scholar] [CrossRef] [PubMed]
- Heiner, D.C. (University of California Los Angeles, Torrance, CA, USA). Personal Verbal Communication, 2015.
- Agata, H.; Kondo, N.; Fukutomi, O.; Takemura, M.; Tashita, H.; Kobayashi, Y.; Shinoda, S.; Nishida, T.; Shinbara, M.; Orii, T. Pulmonary hemosiderosis with hypersensitivity to buckwheat. Ann. Allergy Asthma Immunol. 1997, 78, 233–237. [Google Scholar] [PubMed]
- Soergel, K.H.; Sommers, S.C. Idiopathic pulmonary haemosiderosis and related syndromes. Am. J. Med. 1962, 32, 499–511. [Google Scholar] [CrossRef]
- Saha, B.K. Idiopathic pulmonary hemosiderosis: A state of the art review. Respir. Med. 2021, 176, 106234. [Google Scholar] [CrossRef] [PubMed]
- Taytard, J.; Nathan, N.; De Blic, J.; Fayon, M.; Epaud, R.; Deschildre, A.; Troussier, F.; Lubrano, M.; Chiron, R.; Reix, P.; et al. New insights into pediatric idiopathic pulmonary hemosiderosis: The French RespiRare® cohort. Orphanet J. Rare Dis. 2013, 8, 161. [Google Scholar] [CrossRef]
- Lee, E.J.; Heiner, D.C. Allergy to Cow Milk. Pediatrics Rev. 1986, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.; Craver, R.D. Cow’s milk-induced pulmonary hemosiderosis. J. State Med Soc. Off. Organ State Med Soc. 1989, 141, 19–22. [Google Scholar]
| Authors, Year | Country | n. Cases | Age at Onset (Months) | Signs/Symptoms | Lung Radiologic Infiltrates | Pulmonary Hemosiderosis | Hemosiderosis Diagnosis | Milk Precipitins | Delayed Hypersensitivity | Allergic Sensitization to Milk [sIgE and/or skin prick test (SPT)] | Improvement Upon Milk Avoidance | Recurrence Upon Milk Reintroduction | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Heiner, 1960 | USA | 7 | 1–17 | chronic cough (7/7); wheezing (7/7); chronic rhinitis (7/7); frequent fever (7/7); frequent earache (6/7); diarrhea (6/7); vomiting (5/7); hemoptysis (4/7) | yes, recurrent | yes (5/7) | gastric or bronchial aspirates (4/7); pulmonary needle biopsy (1/7) | yes | intradermal test (ID) late response pos (4/7) | ID +ve (7/7) | yes (6/6) | yes (2/6) |
| 2 | Holland, 1962 | USA | 22 | 4–12 | respiratory disease; failure to thrive (FTT); anemia; splenomegaly; hepatomegaly | non specified (NS) | NS | NS | yes | NS | NS | yes (22/24 on milk-free diet) | NS |
| 3 | Chang, 1969 | USA | 1 | 9 | FTT ; anemia; chronic recurrent lung disease | yes | NS | NS | yes | NS | NS | yes | doubtful, based on clinical history (CH) |
| 4 | Archer, 1971 | England | 1 | 13 | iron-deficiency anemia; lethargy; pallor; bloody vomit; severe cardiac failure; hemoptysis | yes | yes | needle biopsy | no | NS | SPT –ve | yes | doubtful, based on CH |
| 5 | Boat, 1975 | USA | 6 | 7–48 | idiopathic chronic or recurrent pulmonary disease; upper respiratory symptoms; FTT (3/6); frequent regurgitation and watery stools (1/6). SOF (1/6): Iron deficiency (5/6); Anemia (4/6); Eosinophilia (4/6); right ventricular hypertrophy (3/6); adenoid hypertrophy (3/6) | yes (6/6) | yes (5/6) | gastric washing or bronchoalveolar lavage (BAL) | yes (6/6) | 4 ID delayed response | total IgE –ve (6/6); SPT +ve (5/6) | yes (5/6); 1/6 loss of data | yes (1/6) |
| 6 | Stafford, 1977 | USA | 7 | 8–48 | wheezing (5/9); chronic rhinitis (3/9); large adenoids/tonsils (4/9); anemia (4/9); gastrointestinal symptoms (4/9); eosinophilia (6/9) | yes (7/9) | yes (3/9) | gastric washing and BAL | yes (9/9) | lymphocyte response (3/3) | SPT +ve (6/9), sIgE +ve (5/8) | NS | NS |
| 7 | Fossati, 1992 | Italy | 1 | 7 years | anemia; respiratory symptoms | yes | yes | NS | yes | NS | NS | yes | NS |
| 8 | Torres, 1996 | Spain | 1 | 0 (5 days) | vomiting with blood; respiratory failure; restrictive miocardiopathy; anemia; eosinophilia | yes (chest X-Ray, CXR) | yes | BAL | NS | NS | neg | yes | symptoms not reported |
| 9 | Moissidis, 2005 | USA | 8 | 4–29 | cough (7/9); wheezing (3/9); dyspnea (1/9); hemoptysis (2/9); nasal congestion (3/9); recurrent otitis media (OM) (3/9); recurrent fever (4/9); gastrointestinal symptoms (5/9); in 7/8; hematochezia (1/9); FTT (2/9); eosinophilia in 5/8 | yes (9/9) | yes (1/9) | NS | yes (6/6) | NS | sIgE (1/3), SPT (1/2) | yes (8/9) | positive challenge (3/3) |
| 10 | Sigua, 2013 | USA | 1 | 12 | persistent cough; progressive anorexia; intermittent fever; weight loss; iron deficiency anemia | yes (CXR) | no | BAL | yes | NS | sIgE –ve | yes | doubtful, based on CH |
| 11 | Yavuz, 2014 | Turkey | 1 | 36 | respiratory distress; hemoptysis; recurrent bronchitis; FTT; iron deficiency anemia; eosinophilia; increased inflammatory index; | yes (CXR, CT) | yes | BAL | NS | NS | sIgE +ve | yes | doubtful, based on CH |
| 12 | Mourad, 2015 | USA | 1 | 17 | severe anemia; respiratory distress | yes (CXR) | yes | BAL | IgG | NS | sIgE +ve | yes | doubtful, based on CH |
| 13 | Alsukhon, 2017 | USA | 1 | 2 | FTT; recurrent diarrhea; persistent cough; tachypnea; high inflammatory markers | yes (CXR) | NS | NS | IgG4 | NS | sIgE –ve | yes | NS |
| 14 | Ojuawo, 2019 | Nigeria | 1 | 4 | FTT; cough; dyspnea; wheeze; rhinitis; gastrointestinal symptoms; anemia | yes (CXR) | NS | NS | NS | NS | NS | yes | NS doubtful, based on CH |
| 15 | Koc, 2019 | Turkey | 1 | 6 | massive hemoptysis; hematemesis; deep anemia | yes (CXR, CT) | yes | gastric washing | NS | NS | NS | yes | NS |
| 16 | Liu, 2020 | China | 1 | 4 | respiratory failure; hematochezia; diarrhea; elevated WBC and C-reactive protein | yes (CXR, CT) | no | sputum or fasting gastric fluid | NS | NS | –ve | yes | NS |
| HS | IPH | |
|---|---|---|
| Age | infants or young children | older children/adults |
| Hemosiderosis | often | always |
| GI symptoms | often | rarely |
| Precipitins | yes | no |
| Response to diet | yes | no |
| Prognosis | good | variable |
| Pros | Cons |
|---|---|
| Multiorgan involvement (in particular lung and GI) | Absence of case–control studies |
| Detection of precipitating antibodies | Precipitating antibodies not pathognomonic |
| Scarce response to non anti-inflammatory drugs | In most cases the additional administration of anti-inflammatory drugs probably resolved hypersensitivity pneumonia or PH |
| Clinical improvement after milk removal | The presence of milk in pulmonary infiltrates reported only in one case |
| Symptoms’ reoccurrence after milk reintroduction | Confirmatory challenge not provided in most cases and/or not adequately performed |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arasi, S.; Mastrorilli, C.; Pecoraro, L.; Giovannini, M.; Mori, F.; Barni, S.; Caminiti, L.; Castagnoli, R.; Liotti, L.; Saretta, F.; et al. Heiner Syndrome and Milk Hypersensitivity: An Updated Overview on the Current Evidence. Nutrients 2021, 13, 1710. https://doi.org/10.3390/nu13051710
Arasi S, Mastrorilli C, Pecoraro L, Giovannini M, Mori F, Barni S, Caminiti L, Castagnoli R, Liotti L, Saretta F, et al. Heiner Syndrome and Milk Hypersensitivity: An Updated Overview on the Current Evidence. Nutrients. 2021; 13(5):1710. https://doi.org/10.3390/nu13051710
Chicago/Turabian StyleArasi, Stefania, Carla Mastrorilli, Luca Pecoraro, Mattia Giovannini, Francesca Mori, Simona Barni, Lucia Caminiti, Riccardo Castagnoli, Lucia Liotti, Francesca Saretta, and et al. 2021. "Heiner Syndrome and Milk Hypersensitivity: An Updated Overview on the Current Evidence" Nutrients 13, no. 5: 1710. https://doi.org/10.3390/nu13051710
APA StyleArasi, S., Mastrorilli, C., Pecoraro, L., Giovannini, M., Mori, F., Barni, S., Caminiti, L., Castagnoli, R., Liotti, L., Saretta, F., Marseglia, G. L., & Novembre, E. (2021). Heiner Syndrome and Milk Hypersensitivity: An Updated Overview on the Current Evidence. Nutrients, 13(5), 1710. https://doi.org/10.3390/nu13051710








