Pre-Exercise Carbohydrate or Protein Ingestion Influences Substrate Oxidation but Not Performance or Hunger Compared with Cycling in the Fasted State
Abstract
1. Introduction
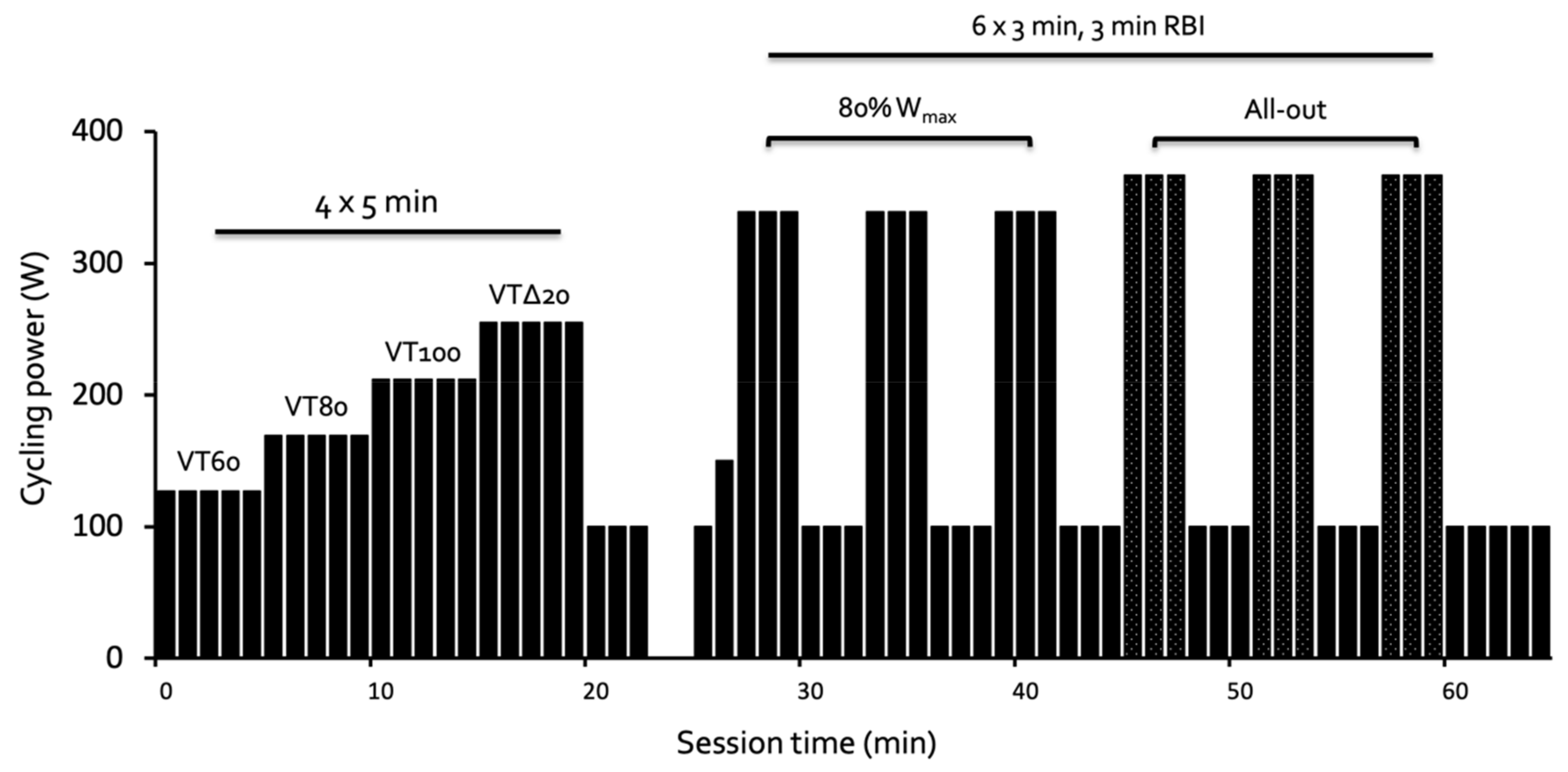
2. Materials and Methods
3. Results
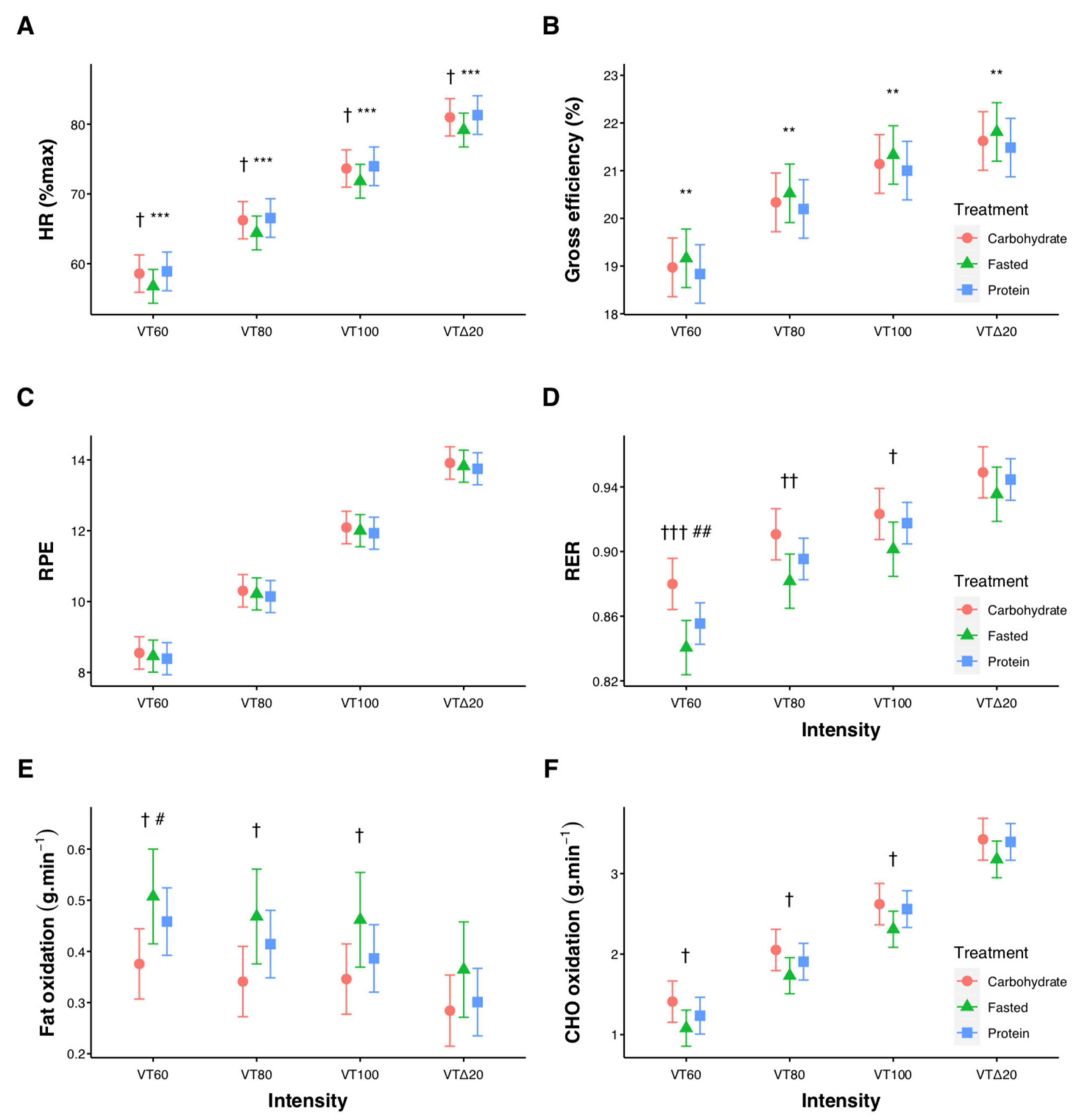
3.1. Submaximal Exercise
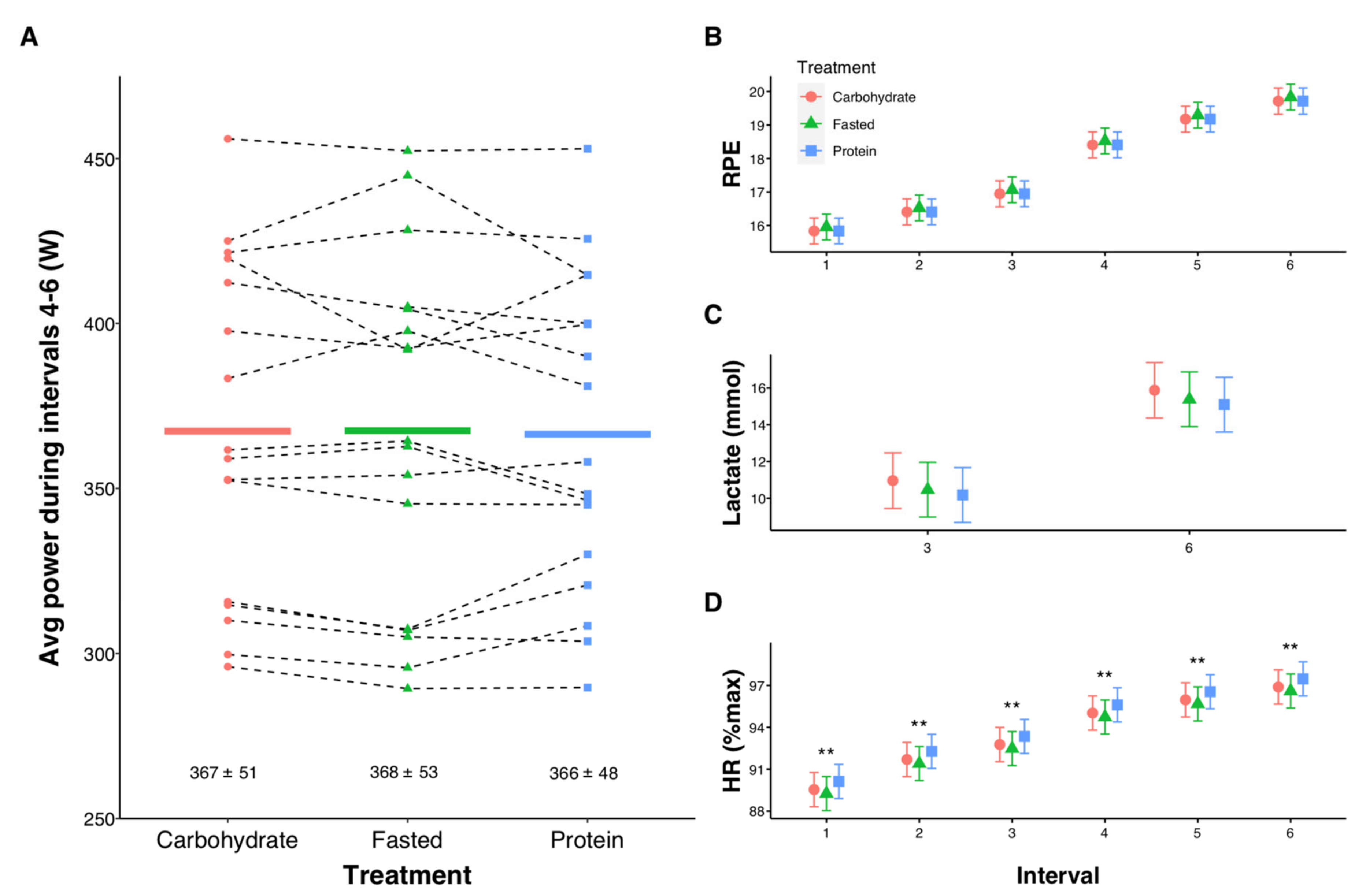
3.2. High-Intensity Exercise
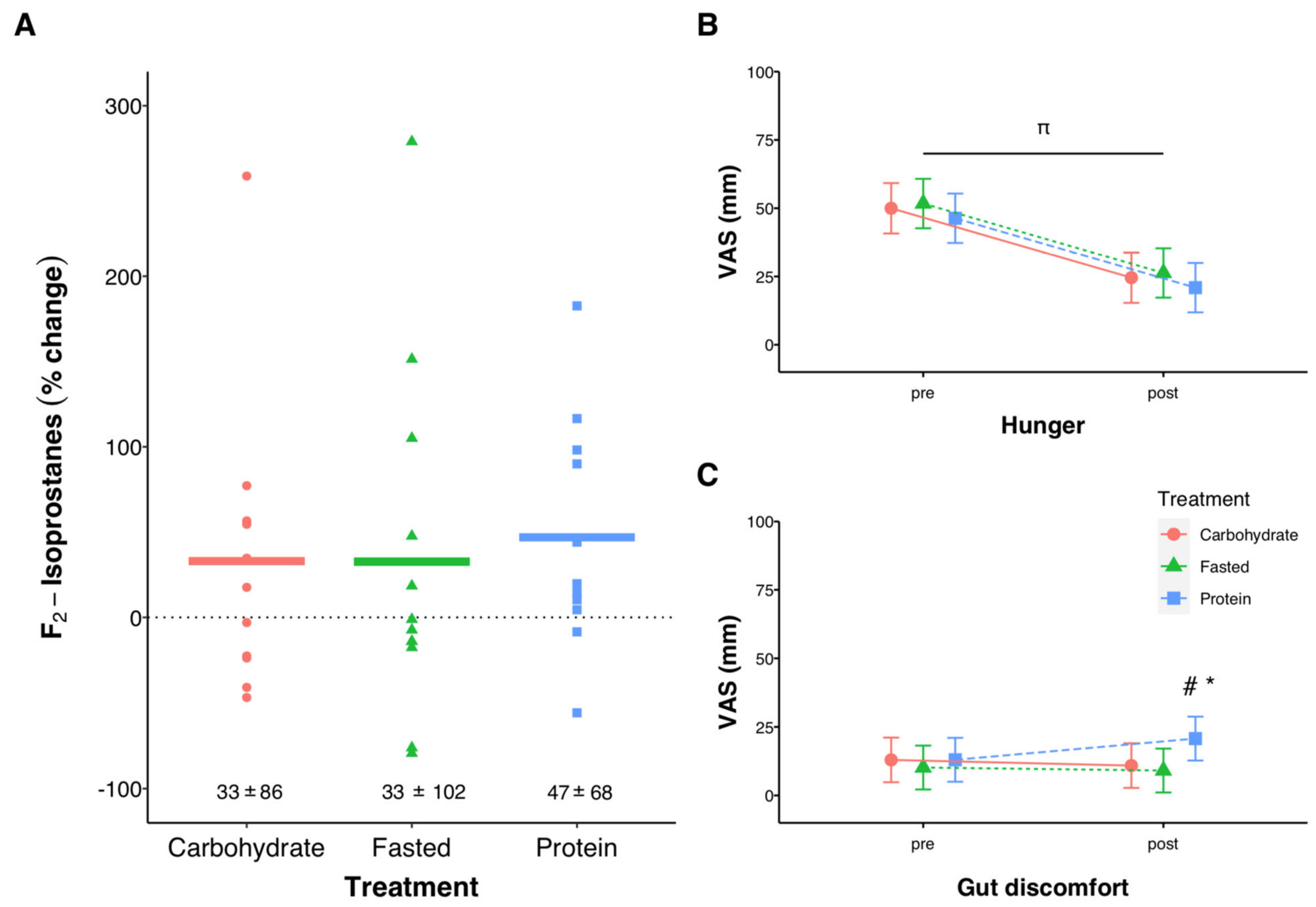
3.3. Pre-Post Exercise
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rothschild, J.; Kilding, A.E.; Plews, D.J. What Should I Eat before Exercise? Pre-Exercise Nutrition and the Response to Endurance Exercise: Current Prospective and Future Directions. Nutrients 2020, 12, 3473. [Google Scholar] [CrossRef] [PubMed]
- Impey, S.G.; Hearris, M.A.; Hammond, K.M.; Bartlett, J.D.; Louis, J.; Close, G.L.; Morton, J.P. Fuel for the work required: A theoretical framework for carbohydrate periodization and the glycogen threshold hypothesis. Sports Med. 2018, 48, 1031–1048. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.F.; Costa, R.R.; Macedo, R.C.O.; Coconcelli, L.; Kruel, L.F.M. Effects of aerobic exercise performed in fasted v. fed state on fat and carbohydrate metabolism in adults: A systematic review and meta-analysis. Br. J. Nutr. 2016, 116, 1153–1164. [Google Scholar] [CrossRef]
- Burke, L.M.; Hawley, J.A.; Jeukendrup, A.; Morton, J.P.; Stellingwerff, T.; Maughan, R.J. Toward a Common Understanding of Diet-Exercise Strategies to Manipulate Fuel Availability for Training and Competition Preparation in Endurance Sport. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, J.A.; Kilding, A.E.; Plews, D.J. Pre-Exercise Nutrition Habits and Beliefs of Endurance Athletes Vary by Sex, Competitive Level, and Diet. J. Am. Coll. Nutr. 2020. [Google Scholar] [CrossRef]
- Rothschild, J.; Kilding, A.E.; Plews, D.J. Prevalence and Determinants of Fasted Training in Endurance Athletes: A Survey Analysis. Int. J. Sport Nutr. Exerc. Metab. 2020, 30, 345–356. [Google Scholar] [CrossRef]
- Impey, S.G.; Smith, D.; Robinson, A.L.; Owens, D.J.; Bartlett, J.D.; Smith, K.; Limb, M.; Tang, J.; Fraser, W.D.; Close, G.L. Leucine-enriched protein feeding does not impair exercise-induced free fatty acid availability and lipid oxidation: Beneficial implications for training in carbohydrate-restricted states. Amino Acids 2015, 47, 407–416. [Google Scholar] [CrossRef]
- Taylor, C.; Bartlett, J.D.; van de Graaf, C.S.; Louhelainen, J.; Coyne, V.; Iqbal, Z.; MacLaren, D.P.; Gregson, W.; Close, G.L.; Morton, J.P. Protein ingestion does not impair exercise-induced AMPK signalling when in a glycogen-depleted state: Implications for train-low compete-high. Eur. J. Appl. Physiol. 2013, 113, 1457–1468. [Google Scholar] [CrossRef]
- Gieske, B.T.; Stecker, R.A.; Smith, C.R.; Witherbee, K.E.; Harty, P.S.; Wildman, R.; Kerksick, C.M. Metabolic impact of protein feeding prior to moderate-intensity treadmill exercise in a fasted state: A pilot study. J. Int. Soc. Sports Nutr. 2018, 15, 56. [Google Scholar] [CrossRef]
- Rowlands, D.S.; Hopkins, W.G. Effect of high-fat, high-carbohydrate, and high-protein meals on metabolism and performance during endurance cycling. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 318–335. [Google Scholar] [CrossRef]
- Willcutts, K.F.; Wilcox, A.; Grunewald, K. Energy metabolism during exercise at different time intervals following a meal. Int. J. Sports Med. 1988, 9, 240–243. [Google Scholar] [CrossRef]
- Aird, T.P.; Davies, R.W.; Carson, B.P. Effects of fasted vs. fed-state exercise on performance and post-exercise metabolism: A systematic review and meta-analysis. Scand J. Med. Sci. Sports 2018, 28, 1476–1493. [Google Scholar] [CrossRef] [PubMed]
- Seiler, S. What is best practice for training intensity and duration distribution in endurance athletes? Int. J. Sports Physiol. Perform. 2010, 5, 276–291. [Google Scholar] [CrossRef]
- Little, J.P.; Chilibeck, P.D.; Ciona, D.; Forbes, S.; Rees, H.; Vandenberg, A.; Zello, G.A. Effect of low- and high-glycemic-index meals on metabolism and performance during high-intensity, intermittent exercise. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Terada, T.; Toghi Eshghi, S.R.; Liubaoerjijin, Y.; Kennedy, M.; Myette-Cote, E.; Fletcher, K.; Boule, N.G. Overnight fasting compromises exercise intensity and volume during sprint interval training but improves high-intensity aerobic endurance. J. Sports Med. Phys. Fit. 2019, 59, 357–365. [Google Scholar] [CrossRef]
- Astorino, T.A.; Sherrick, S.; Mariscal, M.; Jimenez, V.C.; Stetson, K.; Courtney, D. No effect of meal intake on physiological or perceptual responses to self-selected high intensity interval exercise (HIIE). Biol. Sport 2019, 36, 225. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Paschalis, V.; Theodorou, A.A.; Kyparos, A.; Nikolaidis, M.G. Redox basis of exercise physiology. Redox Biol. 2020, 35, 101499. [Google Scholar] [CrossRef]
- Margaritelis, N.V.; Theodorou, A.A.; Paschalis, V.; Veskoukis, A.S.; Dipla, K.; Zafeiridis, A.; Panayiotou, G.; Vrabas, I.S.; Kyparos, A.; Nikolaidis, M.G. Adaptations to endurance training depend on exercise-induced oxidative stress: Exploiting redox interindividual variability. Acta Physiol. 2018, 222. [Google Scholar] [CrossRef]
- Gregersen, S.; Samocha-Bonet, D.; Heilbronn, L.K.; Campbell, L.V. Inflammatory and oxidative stress responses to high-carbohydrate and high-fat meals in healthy humans. J. Nutr. Metab. 2012, 2012, 238056. [Google Scholar] [CrossRef]
- Draganidis, D.; Karagounis, L.G.; Athanailidis, I.; Chatzinikolaou, A.; Jamurtas, A.Z.; Fatouros, I.G. Inflammaging and skeletal muscle: Can protein intake make a difference? J. Nutr. 2016, 146, 1940–1952. [Google Scholar] [CrossRef]
- McAnulty, S.R.; McAnulty, L.S.; Morrow, J.D.; Nieman, D.C.; Owens, J.T.; Carper, C.M. Influence of carbohydrate, intense exercise, and rest intervals on hormonal and oxidative changes. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 478–490. [Google Scholar] [CrossRef]
- Achten, J.; Jeukendrup, A.E. The effect of pre-exercise carbohydrate feedings on the intensity that elicits maximal fat oxidation. J. Sports Sci. 2003, 21, 1017–1025. [Google Scholar] [CrossRef]
- Enevoldsen, L.; Simonsen, L.; Macdonald, I.; Bülow, J. The combined effects of exercise and food intake on adipose tissue and splanchnic metabolism. J. Physiol. 2004, 561, 871–882. [Google Scholar] [CrossRef]
- Davis, J.A.; Whipp, B.J.; Lamarra, N.; Huntsman, D.J.; Frank, M.H.; Wasserman, K. Effect of ramp slope on determination of aerobic parameters from the ramp exercise test. Med. Sci. Sports Exerc. 1982, 14, 339–343. [Google Scholar] [CrossRef] [PubMed]
- McLean, B.D.; Coutts, A.J.; Kelly, V.; McGuigan, M.R.; Cormack, S.J. Neuromuscular, endocrine, and perceptual fatigue responses during different length between-match microcycles in professional rugby league players. Int. J. Sports Physiol. Perform. 2010, 5, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef]
- Lansley, K.E.; Dimenna, F.J.; Bailey, S.J.; Jones, A.M. A ‘new’ method to normalise exercise intensity. Int. J. Sports Med. 2011, 32, 535–541. [Google Scholar] [CrossRef]
- Jeukendrup, A.E.; Wallis, G.A. Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int. J. Sports Med. 2005, 26 (Suppl. 1), S28–S37. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Foster, C.; Florhaug, J.A.; Franklin, J.; Gottschall, L.; Hrovatin, L.A.; Parker, S.; Doleshal, P.; Dodge, C. A new approach to monitoring exercise training. J. Strength Cond. Res. 2001, 15, 109–115. [Google Scholar] [PubMed]
- Nikolaidis, M.G.; Kyparos, A.; Spanou, C.; Paschalis, V.; Theodorou, A.A.; Panayiotou, G.; Grivas, G.V.; Zafeiridis, A.; Dipla, K.; Vrabas, I.S. Aging is not a barrier to muscle and redox adaptations: Applying the repeated eccentric exercise model. Exp. Gerontol. 2013, 48, 734–743. [Google Scholar] [CrossRef]
- Westfall, J.; Kenny, D.A.; Judd, C.M. Statistical power and optimal design in experiments in which samples of participants respond to samples of stimuli. J. Exp. Psychol. Gen. 2014, 143, 2020–2045. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Routledge: Hillside, NJ, USA, 1988. [Google Scholar]
- Oliveira, C.L.P.; Boule, N.G.; Berg, A.; Sharma, A.M.; Elliott, S.A.; Siervo, M.; Ghosh, S.; Prado, C.M. Consumption of a High-Protein Meal Replacement Leads to Higher Fat Oxidation, Suppression of Hunger, and Improved Metabolic Profile After an Exercise Session. Nutrients 2021, 13, 155. [Google Scholar] [CrossRef]
- Kang, J.; Raines, E.; Rosenberg, J.; Ratamess, N.; Naclerio, F.; Faigenbaum, A. Metabolic responses during postprandial exercise. Res. Sports Med. 2013, 21, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Westerterp, K.R. Diet induced thermogenesis. Nutr. Metab. 2004, 1, 5. [Google Scholar] [CrossRef] [PubMed]
- Crovetti, R.; Porrini, M.; Santangelo, A.; Testolin, G. The influence of thermic effect of food on satiety. Eur. J. Clin. Nutr. 1998, 52, 482–488. [Google Scholar] [CrossRef]
- Welle, S.; Lilavivathana, U.; Campbell, R.G. Increased plasma norepinephrine concentrations and metabolic rates following glucose ingestion in man. Metabolism 1980, 29, 806–809. [Google Scholar] [CrossRef]
- Hulston, C.J.; Venables, M.C.; Mann, C.H.; Martin, C.; Philp, A.; Baar, K.; Jeukendrup, A.E. Training with low muscle glycogen enhances fat metabolism in well-trained cyclists. Med. Sci. Sports Exerc. 2010, 42, 2046–2055. [Google Scholar] [CrossRef] [PubMed]
- Lane, S.C.; Areta, J.L.; Bird, S.R.; Coffey, V.G.; Burke, L.M.; Desbrow, B.; Karagounis, L.G.; Hawley, J.A. Caffeine ingestion and cycling power output in a low or normal muscle glycogen state. Med. Sci. Sports Exerc. 2013, 45, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Knapik, J.J.; Meredith, C.N.; Jones, B.H.; Suek, L.; Young, V.R.; Evans, W.J. Influence of fasting on carbohydrate and fat metabolism during rest and exercise in men. J. Appl. Physiol. 1988, 64, 1923–1929. [Google Scholar] [CrossRef]
- Galloway, S.D.; Lott, M.J.; Toulouse, L.C. Preexercise carbohydrate feeding and high-intensity exercise capacity: Effects of timing of intake and carbohydrate concentration. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 258–266. [Google Scholar] [CrossRef]
- Khong, T.K.; Selvanayagam, V.S.; Hamzah, S.H.; Yusof, A. Effect of quantity and quality of pre-exercise carbohydrate meals on central fatigue. J. Appl. Physiol. 2018, 125, 1021–1029. [Google Scholar] [CrossRef]
- AbuMoh’d, M.F.; Matalqah, L.; Al-Abdulla, Z. Effects of Oral Branched-Chain Amino Acids (BCAAs) Intake on Muscular and Central Fatigue during an Incremental Exercise. J. Hum. Kinet. 2020, 72, 69–78. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, S.F.; Islam, H.; Hazell, T.J. The emerging role of lactate as a mediator of exercise-induced appetite suppression. Am. J. Physiol. Endocrinol. Metab. 2020, 314, E814–E819. [Google Scholar] [CrossRef] [PubMed]
- Deighton, K.; Barry, R.; Connon, C.E.; Stensel, D.J. Appetite, gut hormone and energy intake responses to low volume sprint interval and traditional endurance exercise. Eur. J. Appl. Physiol. 2013, 113, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- Deighton, K.; Zahra, J.C.; Stensel, D.J. Appetite, energy intake and resting metabolic responses to 60 min treadmill running performed in a fasted versus a postprandial state. Appetite 2012, 58, 946–954. [Google Scholar] [CrossRef]
- Rehrer, N.J.; van Kemenade, M.; Meester, W.; Brouns, F.; Saris, W.H. Gastrointestinal complaints in relation to dietary intake in triathletes. Int. J. Sport Nutr. 1992, 2, 48–59. [Google Scholar] [CrossRef]
- Snipe, R.M.J.; Khoo, A.; Kitic, C.M.; Gibson, P.R.; Costa, R.J.S. Carbohydrate and protein intake during exertional heat stress ameliorates intestinal epithelial injury and small intestine permeability. Appl. Physiol. Nutr. Metab. 2017, 42, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Tenforde, A.S.; Barrack, M.T.; Nattiv, A.; Fredericson, M. Parallels with the Female Athlete Triad in Male Athletes. Sports Med. 2016, 46, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Deutz, R.C.; Benardot, D.; Martin, D.E.; Cody, M.M. Relationship between energy deficits and body composition in elite female gymnasts and runners. Med. Sci. Sports Exerc. 2000, 32, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Henriquez-Olguin, C.; Renani, L.B.; Arab-Ceschia, L.; Raun, S.H.; Bhatia, A.; Li, Z.; Knudsen, J.R.; Holmdahl, R.; Jensen, T.E. Adaptations to high-intensity interval training in skeletal muscle require NADPH oxidase 2. Redox Biol. 2019, 24, 101188. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, J.A.; Bishop, D.J. Effects of Dietary Supplements on Adaptations to Endurance Training. Sports Med. 2020, 50, 25–53. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, M.G.; Kyparos, A.; Vrabas, I.S. F2-isoprostane formation, measurement and interpretation: The role of exercise. Prog Lipid Res. 2011, 50, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Kyparos, A.; Paschalis, V.; Theodorou, A.A.; Panayiotou, G.; Zafeiridis, A.; Dipla, K.; Nikolaidis, M.G.; Vrabas, I.S. Reductive stress after exercise: The issue of redox individuality. Redox Biol. 2014, 2, 520–528. [Google Scholar] [CrossRef]
- Braakhuis, A.J.; Hopkins, W.G.; Lowe, T.E. Effect of dietary antioxidants, training, and performance correlates on antioxidant status in competitive rowers. Int. J. Sports Physiol. Perform. 2013, 8, 565–572. [Google Scholar] [CrossRef]
- Coyle, E.F.; Coggan, A.R.; Hemmert, M.K.; Lowe, R.C.; Walters, T.J. Substrate usage during prolonged exercise following a preexercise meal. J. Appl. Physiol. 1985, 59, 429–433. [Google Scholar] [CrossRef]
- Moseley, L.; Lancaster, G.I.; Jeukendrup, A.E. Effects of timing of pre-exercise ingestion of carbohydrate on subsequent metabolism and cycling performance. Eur. J. Appl. Physiol. 2003, 88, 453–458. [Google Scholar] [CrossRef]
- Pritchett, K.; Bishop, P.; Pritchett, R.; Kovacs, M.; Davis, J.; Casaru, C.; Green, M. Effects of timing of pre-exercise nutrient intake on glucose responses and intermittent cycling performance. S. Afr. J. Sports Med. 2008, 20, 86–90. [Google Scholar] [CrossRef]
- Smith, G.J.; Rhodes, E.C.; Langill, R.H. The effect of pre-exercise glucose ingestion on performance during prolonged swimming. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 136–144. [Google Scholar] [CrossRef]
- Sherman, W.M.; Brodowicz, G.; Wright, D.A.; Allen, W.K.; Simonsen, J.; Dernbach, A. Effects of 4 h preexercise carbohydrate feedings on cycling performance. Med. Sci. Sports Exerc. 1989, 21, 598–604. [Google Scholar] [CrossRef]
- Jentjens, R.L.; Cale, C.; Gutch, C.; Jeukendrup, A.E. Effects of pre-exercise ingestion of differing amounts of carbohydrate on subsequent metabolism and cycling performance. Eur. J. Appl. Physiol. 2003, 88, 444–452. [Google Scholar] [CrossRef]
- Mears, S.A.; Dickinson, K.; Bergin-Taylor, K.; Dee, R.; Kay, J.; James, L.J. Perception of Breakfast Ingestion Enhances High-Intensity Cycling Performance. Int. J. Sports Physiol. Perform. 2018, 13, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Clark, V.R.; Hopkins, W.G.; Hawley, J.A.; Burke, L.M. Placebo effect of carbohydrate feedings during a 40-km cycling time trial. Med. Sci. Sports Exerc. 2000, 32, 1642–1647. [Google Scholar] [CrossRef]
- Hulston, C.J.; Jeukendrup, A.E. No placebo effect from carbohydrate intake during prolonged exercise. Int. J. Sport Nutr. Exerc. Metab. 2009, 19, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Riddell, M.C.; Partington, S.L.; Stupka, N.; Armstrong, D.; Rennie, C.; Tarnopolsky, M.A. Substrate utilization during exercise performed with and without glucose ingestion in female and male endurance trained athletes. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 407–421. [Google Scholar] [CrossRef]
- Tobias, I.S.; Lazauskas, K.K.; Siu, J.; Costa, P.B.; Coburn, J.W.; Galpin, A.J. Sex and fiber type independently influence AMPK, TBC1D1, and TBC1D4 at rest and during recovery from high-intensity exercise in humans. J. Appl. Physiol. 2020, 128, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Hetlelid, K.J.; Plews, D.J.; Herold, E.; Laursen, P.B.; Seiler, S. Rethinking the role of fat oxidation: Substrate utilisation during high-intensity interval training in well-trained and recreationally trained runners. BMJ Open Sport Exerc. Med. 2015, 1, e000047. [Google Scholar] [CrossRef]
- Rowlands, D.S.; Hopkins, W.G. Effects of high-fat and high-carbohydrate diets on metabolism and performance in cycling. Metabolism 2002, 51, 678–690. [Google Scholar] [CrossRef]
- McKenzie, S.; Phillips, S.M.; Carter, S.L.; Lowther, S.; Gibala, M.J.; Tarnopolsky, M.A. Endurance exercise training attenuates leucine oxidation and BCOAD activation during exercise in humans. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E580–E587. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Leese, G.P.; Smith, K.; Watt, P.W.; Nevill, A.; Rooyackers, O.; Wagenmakers, A.J.; Rennie, M.J. Modulation of whole body protein metabolism, during and after exercise, by variation of dietary protein. J. Appl. Physiol. 1998, 85, 1744–1752. [Google Scholar] [CrossRef] [PubMed]
- Lemon, P.W.; Mullin, J.P. Effect of initial muscle glycogen levels on protein catabolism during exercise. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1980, 48, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Larsen, M.S.; Holm, L.; Svart, M.V.; Hjelholt, A.J.; Bengtsen, M.B.; Dollerup, O.L.; Dalgaard, L.B.; Vendelbo, M.H.; van Hall, G.; Moller, N.; et al. Effects of protein intake prior to carbohydrate-restricted endurance exercise: A randomized crossover trial. J. Int. Soc. Sports Nutr. 2020, 17, 7. [Google Scholar] [CrossRef] [PubMed]

| Contrast | Intensity | Heart Rate | Gross Efficiency | RPE | RER | Fat Oxidation (g/min) | Carbohydrate Oxidation (g/min) |
|---|---|---|---|---|---|---|---|
| CARB-FASTED | VT60 | Small | Trivial | Trivial | Large | Large | Medium |
| ES = 0.34 | ES = −0.15 | ES = 0.08 | ES = 1.29 | ES = −0.85 | ES = 0.69 | ||
| [0.07, 0.61] | [−0.32, 0.02] | [−0.23, 0.39] | [0.69, 1.89] | [−1.46, −0.25] | [0.18, 1.21] | ||
| CARB-PROTEIN | VT60 | Trivial | Trivial | Trivial | Large | Medium | Small |
| ES = −0.06 | ES = 0.11 | ES = 0.15 | ES = 0.80 | ES = −0.54 | ES = 0.37 | ||
| [−0.37, 0.25] | [−0.06, 0.28] | [−0.16, 0.45] | [0.28, 1.33] | [−1, −0.07] | [−0.1, 0.84] | ||
| FASTED-PROTEIN | VT60 | Small | Small | Trivial | Small | Small | Small |
| ES = −0.4 | ES = 0.26 | ES = 0.07 | ES = −0.49 | ES = 0.32 | ES = −0.33 | ||
| [−0.55, −0.24] | [0.09, 0.42] | [−0.23, 0.37] | [−1.03, 0.06] | [−0.2, 0.84] | [−0.82, 0.17] | ||
| CARB-FASTED | VT80 | Small | Trivial | Trivial | Large | Large | Medium |
| ES = 0.34 | ES = −0.15 | ES = 0.08 | ES = 0.95 | ES = −0.82 | ES = 0.67 | ||
| [0.07, 0.61] | [−0.32, 0.02] | [−0.23, 0.39] | [0.36, 1.54] | [−1.43, −0.22] | [0.16, 1.19] | ||
| CARB-PROTEIN | VT80 | Trivial | Trivial | Trivial | Medium | Small | Small |
| ES = −0.06 | ES = 0.11 | ES = 0.15 | ES = 0.50 | ES = −0.47 | ES = 0.31 | ||
| [−0.37, 0.25] | [−0.06, 0.28] | [−0.16, 0.45] | [−0.02, 1.02] | [−0.94, −0.01] | [−0.16, 0.77] | ||
| FASTED-PROTEIN | VT80 | Small | Small | Trivial | Small | Small | Small |
| ES = −0.4 | ES = 0.26 | ES = 0.07 | ES = −0.45 | ES = 0.35 | ES = −0.37 | ||
| [−0.55, −0.24] | [0.09, 0.42] | [−0.23, 0.37] | [−0.99, 0.09] | [−0.17, 0.87] | [−0.86, 0.13] | ||
| CARB-FASTED | VT100 | Small | Trivial | Trivial | Medium | Medium | Medium |
| ES = 0.34 | ES = −0.15 | ES = 0.08 | ES = 0.71 | ES = −0.75 | ES = 0.66 | ||
| [0.07, 0.61] | [−0.32, 0.02] | [−0.23, 0.39] | [0.13, 1.3] | [−1.35, −0.15] | [0.14, 1.17] | ||
| CARB-PROTEIN | VT100 | Trivial | Trivial | Trivial | Trivial | Small | Trivial |
| ES = −0.06 | ES = 0.11 | ES = 0.15 | ES = 0.19 | ES = −0.26 | ES = 0.13 | ||
| [−0.37, 0.25] | [−0.06, 0.28] | [−0.16, 0.45] | [−0.33, 0.7] | [−0.73, 0.2] | [−0.34, 0.59] | ||
| FASTED-PROTEIN | VT100 | Small | Small | Trivial | Medium | Small | Medium |
| ES = −0.4 | ES = 0.26 | ES = 0.07 | ES = −0.53 | ES = 0.49 | ES = −0.53 | ||
| [−0.55, −0.24] | [0.09, 0.42] | [−0.23, 0.37] | [−1.07, 0.01] | [−0.03, 1.01] | [−1.02, −0.03] | ||
| CARB-FASTED | VTΔ20 | Small | Trivial | Trivial | Small | Medium | Medium |
| ES = 0.34 | ES = −0.15 | ES = 0.08 | ES = 0.44 | ES = −0.52 | ES = 0.53 | ||
| [0.07, 0.61] | [−0.32, 0.02] | [−0.23, 0.39] | [−0.14, 1.03] | [−1.13, 0.09] | [0, 1.05] | ||
| CARB-PROTEIN | VTΔ20 | Trivial | Trivial | Trivial | Trivial | Trivial | Trivial |
| ES = −0.06 | ES = 0.11 | ES = 0.15 | ES = 0.14 | ES = −0.11 | ES = 0.07 | ||
| [−0.37, 0.25] | [−0.06, 0.28] | [−0.16, 0.45] | [−0.37, 0.66] | [−0.58, 0.36] | [−0.4, 0.54] | ||
| FASTED-PROTEIN | VTΔ20 | Small | Small | Trivial | Small | Small | Small |
| ES = −0.4 | ES = 0.26 | ES = 0.07 | ES = −0.30 | ES = 0.41 | ES = −0.46 | ||
| [−0.55, −0.24] | [0.09, 0.42] | [−0.23, 0.37] | [−0.84, 0.24] | [−0.11, 0.94] | [−0.96, 0.04] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rothschild, J.A.; Kilding, A.E.; Broome, S.C.; Stewart, T.; Cronin, J.B.; Plews, D.J. Pre-Exercise Carbohydrate or Protein Ingestion Influences Substrate Oxidation but Not Performance or Hunger Compared with Cycling in the Fasted State. Nutrients 2021, 13, 1291. https://doi.org/10.3390/nu13041291
Rothschild JA, Kilding AE, Broome SC, Stewart T, Cronin JB, Plews DJ. Pre-Exercise Carbohydrate or Protein Ingestion Influences Substrate Oxidation but Not Performance or Hunger Compared with Cycling in the Fasted State. Nutrients. 2021; 13(4):1291. https://doi.org/10.3390/nu13041291
Chicago/Turabian StyleRothschild, Jeffrey A., Andrew E. Kilding, Sophie C. Broome, Tom Stewart, John B. Cronin, and Daniel J. Plews. 2021. "Pre-Exercise Carbohydrate or Protein Ingestion Influences Substrate Oxidation but Not Performance or Hunger Compared with Cycling in the Fasted State" Nutrients 13, no. 4: 1291. https://doi.org/10.3390/nu13041291
APA StyleRothschild, J. A., Kilding, A. E., Broome, S. C., Stewart, T., Cronin, J. B., & Plews, D. J. (2021). Pre-Exercise Carbohydrate or Protein Ingestion Influences Substrate Oxidation but Not Performance or Hunger Compared with Cycling in the Fasted State. Nutrients, 13(4), 1291. https://doi.org/10.3390/nu13041291







