Perinatal Dietary Polyunsaturated Fatty Acids in Brain Development, Role in Neurodevelopmental Disorders
Abstract
1. Introduction
2. PUFAs and Brain Development
2.1. Accretion of PUFAs in the Developing Brain
2.2. Needs of LC PUFAs to the Pre and Post-Natal Developing Brain: Transport and Dietary Maternal Supply
2.2.1. Pre-Natal Maternal Supply of LC PUFAs to the Developing Brain via the Placenta and the Blood-Brain Barrier
2.2.2. Post-Natal Maternal Supply of LC PUFAs to the Developing Brain via Breastmilk or Infant Formula
3. Endogenous Production of PUFAs in the Developing Brain
3.1. Expression of Key Enzymes in the Developing Brain
3.2. Gender Differences in Brain PUFAs Accretion and Effect of PUFAs on Sex Determination
4. Mechanisms of Action of PUFAs on Neurodevelopment
4.1. Role of PUFAs in Synaptogenesis and Neuronal Development
4.2. Role of PUFAs in the Regulation of Microglia and Neuroinflammation during Brain Development
5. Role of PUFAs in Neurodevelopmental Diseases
5.1. Early-Life n-3 PUFAs and Cognition in Infants
5.2. Autism Spectrum Disorders (ASD)
5.3. Attention Deficits and Hyperactivity Disorder (ADHD)
5.4. Schizophrenia
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bazinet, R.P.; Layé, S. Polyunsaturated Fatty Acids and Their Metabolites in Brain Function and Disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary Aspects of Diet: The Omega-6/Omega-3 Ratio and the Brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef]
- Weiser, M.J.; Butt, C.M.; Mohajeri, M.H. Docosahexaenoic Acid and Cognition throughout the Lifespan. Nutrients 2016, 8, 99. [Google Scholar] [CrossRef]
- Rodríguez, M.; G. Rebollar, P.; Mattioli, S.; Castellini, C. N-3 PUFA Sources (Precursor/Products): A Review of Current Knowledge on Rabbit. Animals 2019, 9, 806. [Google Scholar] [CrossRef]
- Smink, W.; Gerrits, W.J.J.; Gloaguen, M.; Ruiter, A.; van Baal, J. Linoleic and α-Linolenic Acid as Precursor and Inhibitor for the Synthesis of Long-Chain Polyunsaturated Fatty Acids in Liver and Brain of Growing Pigs. Anim. Int. J. Anim. Biosci. 2012, 6, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.S.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef]
- Layé, S.; Nadjar, A.; Joffre, C.; Bazinet, R.P. Anti-Inflammatory Effects of Omega-3 Fatty Acids in the Brain: Physiological Mechanisms and Relevance to Pharmacology. Pharmacol. Rev. 2018, 70, 12–38. [Google Scholar] [CrossRef] [PubMed]
- De Giuseppe, R.; Roggi, C.; Cena, H. N-3 LC-PUFA Supplementation: Effects on Infant and Maternal Outcomes. Eur. J. Nutr. 2014, 53, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Medicine, I. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; National Academy Press: Washington, DC, USA, 2002; ISBN 978-0-309-08525-0. [Google Scholar]
- Lauterbach, R. EPA+DHA in Prevention of Early Preterm Birth—Do We Know How to Apply It? EBioMedicine 2018, 35, 16–17. [Google Scholar] [CrossRef]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for High-Value Products Towards Human Health and Nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef]
- Jakobsson, A.; Westerberg, R.; Jacobsson, A. Fatty Acid Elongases in Mammals: Their Regulation and Roles in Metabolism. Prog. Lipid Res. 2006, 45, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, A.; Stöhr, H.; White, K.; Weber, B.H. CDNA Cloning, Genomic Structure, and Chromosomal Localization of Three Members of the Human Fatty Acid Desaturase Family. Genomics 2000, 66, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Poulos, A.; Sharp, P.; Johnson, D.; Easton, C. The Occurrence of Polyenoic Very Long Chain Fatty Acids with Greater than 32 Carbon Atoms in Molecular Species of Phosphatidylcholine in Normal and Peroxisome-Deficient (Zellweger’s Syndrome) Brain. Biochem. J. 1988, 253, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Robinson, B.S.; Johnson, D.W.; Poulos, A. Unique Molecular Species of Phosphatidylcholine Containing Very-Long-Chain (C24-C38) Polyenoic Fatty Acids in Rat Brain. Biochem. J. 1990, 265, 763–767. [Google Scholar] [CrossRef]
- Yu, M.; Benham, A.; Logan, S.; Brush, R.S.; Mandal, M.N.A.; Anderson, R.E.; Agbaga, M.-P. ELOVL4 Protein Preferentially Elongates 20:5n3 to Very Long Chain PUFAs over 20:4n6 and 22:6n3. J. Lipid Res. 2012, 53, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Hopiavuori, B.R.; Deák, F.; Wilkerson, J.L.; Brush, R.S.; Rocha-Hopiavuori, N.A.; Hopiavuori, A.R.; Ozan, K.G.; Sullivan, M.T.; Wren, J.D.; Georgescu, C.; et al. Homozygous Expression of Mutant ELOVL4 Leads to Seizures and Death in a Novel Animal Model of Very Long-Chain Fatty Acid Deficiency. Mol. Neurobiol. 2018, 55, 1795–1813. [Google Scholar] [CrossRef] [PubMed]
- Morales, E.; Bustamante, M.; Gonzalez, J.R.; Guxens, M.; Torrent, M.; Mendez, M.; Garcia-Esteban, R.; Julvez, J.; Forns, J.; Vrijheid, M.; et al. Genetic Variants of the FADS Gene Cluster and ELOVL Gene Family, Colostrums LC-PUFA Levels, Breastfeeding, and Child Cognition. PLoS ONE 2011, 6, e17181. [Google Scholar] [CrossRef]
- de Groot, R.H.M.; Emmett, R.; Meyer, B.J. Non-Dietary Factors Associated with n-3 Long-Chain PUFA Levels in Humans—A Systematic Literature Review. Br. J. Nutr. 2019, 121, 793–808. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Shen, J.; Abecasis, G.R.; Kisialiou, A.; Ordovas, J.M.; Guralnik, J.M.; Singleton, A.; Bandinelli, S.; Cherubini, A.; Arnett, D.; et al. Genome-Wide Association Study of Plasma Polyunsaturated Fatty Acids in the InCHIANTI Study. PLoS Genet. 2009, 5, e1000338. [Google Scholar] [CrossRef] [PubMed]
- Muc, M.; Kreiner-Møller, E.; Larsen, J.M.; Birch, S.; Brix, S.; Bisgaard, H.; Lauritzen, L. Maternal Fatty Acid Desaturase Genotype Correlates with Infant Immune Responses at 6 Months. Br. J. Nutr. 2015, 114, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Tan, S.-Y.; Henry, C.J. Long-Chain n-3 PUFA in Vegetarian Women: A Metabolic Perspective. J. Nutr. Sci. 2017, 6, e58. [Google Scholar] [CrossRef]
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar Effects on Infants of N-3 and n-6 Fatty Acids Supplementation to Pregnant and Lactating Women. Pediatrics 2001, 108, E82. [Google Scholar] [CrossRef]
- Chilton, F.H.; Dutta, R.; Reynolds, L.M.; Sergeant, S.; Mathias, R.A.; Seeds, M.C. Precision Nutrition and Omega-3 Polyunsaturated Fatty Acids: A Case for Personalized Supplementation Approaches for the Prevention and Management of Human Diseases. Nutrients 2017, 9, 1165. [Google Scholar] [CrossRef] [PubMed]
- Ameur, A.; Enroth, S.; Johansson, A.; Zaboli, G.; Igl, W.; Johansson, A.C.V.; Rivas, M.A.; Daly, M.J.; Schmitz, G.; Hicks, A.A.; et al. Genetic Adaptation of Fatty-Acid Metabolism: A Human-Specific Haplotype Increasing the Biosynthesis of Long-Chain Omega-3 and Omega-6 Fatty Acids. Am. J. Hum. Genet. 2012, 90, 809–820. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 Polyunsaturated Fatty Acids and Inflammatory Processes: Nutrition or Pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef]
- Dennis, E.A.; Norris, P.C. Eicosanoid Storm in Infection and Inflammation. Nat. Rev. Immunol. 2015, 15, 511–523. [Google Scholar] [CrossRef]
- Bazan, N.G. Docosanoids and Elovanoids from Omega-3 Fatty Acids Are pro-Homeostatic Modulators of Inflammatory Responses, Cell Damage and Neuroprotection. Mol. Aspects Med. 2018, 64, 18–33. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Levy, B.D. Resolvins in Inflammation: Emergence of the pro-Resolving Superfamily of Mediators. J. Clin. Invest. 2018, 128, 2657–2669. [Google Scholar] [CrossRef]
- Sosa-Castillo, E.; Rodríguez-Cruz, M.; Moltó-Puigmartí, C. Genomics of Lactation: Role of Nutrigenomics and Nutrigenetics in the Fatty Acid Composition of Human Milk. Br. J. Nutr. 2017, 118, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Sastry, P.S. Lipids of Nervous Tissue: Composition and Metabolism. Prog. Lipid Res. 1985, 24, 69–176. [Google Scholar] [CrossRef]
- McNamara, R.K.; Carlson, S.E. Role of Omega-3 Fatty Acids in Brain Development and Function: Potential Implications for the Pathogenesis and Prevention of Psychopathology. Prostaglandins Leukot. Essent. Fatty Acids 2006, 75, 329–349. [Google Scholar] [CrossRef] [PubMed]
- Joffre, C.; Dinel, A.L.; Aubert, A.; Fressange-Mazda, C.; Le Ruyet, P.; Layé, S. Impact of Lactobacillus Fermentum and Dairy Lipids in the Maternal Diet on the Fatty Acid Composition of Pups’ Brain and Peripheral Tissues. Prostaglandins Leukot. Essent. Fatty Acids 2016, 115, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.C.; Ward, G.; Ma, Y.C.; Salem, N.; Kim, H.Y. Effect of Docosahexaenoic Acid on the Synthesis of Phosphatidylserine in Rat Brain in Microsomes and C6 Glioma Cells. J. Neurochem. 1998, 70, 24–30. [Google Scholar] [CrossRef]
- Bascoul-Colombo, C.; Guschina, I.A.; Maskrey, B.H.; Good, M.; O’Donnell, V.B.; Harwood, J.L. Dietary DHA Supplementation Causes Selective Changes in Phospholipids from Different Brain Regions in Both Wild Type Mice and the Tg2576 Mouse Model of Alzheimer’s Disease. Biochim. Biophys. Acta 2016, 1861, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Diau, G.-Y. The Influence of Dietary Docosahexaenoic Acid and Arachidonic Acid on Central Nervous System Polyunsaturated Fatty Acid Composition. Prostaglandins Leukot. Essent. Fatty Acids 2007, 77, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Di Miceli, M.; Bosch-Bouju, C.; Layé, S. Polyunsaturated Fatty Acids and Their Derivatives in Neurotransmission and Synapses: A New Hallmark of Synaptopathies. Proc. Nutr. Soc. 2020, 79, 388–403. [Google Scholar] [CrossRef]
- O’brien, J.S. Stability Of The Myelin Membrane. Science 1965, 147, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Poitelon, Y.; Kopec, A.M.; Belin, S. Myelin Fat Facts: An Overview of Lipids and Fatty Acid Metabolism. Cells 2020, 9, 812. [Google Scholar] [CrossRef]
- Cao, D.; Kevala, K.; Kim, J.; Moon, H.-S.; Jun, S.B.; Lovinger, D.; Kim, H.-Y. Docosahexaenoic Acid Promotes Hippocampal Neuronal Development and Synaptic Function. J. Neurochem. 2009, 111, 510–521. [Google Scholar] [CrossRef]
- Kim, H.-Y.; Spector, A.A. N-Docosahexaenoylethanolamine: A Neurotrophic and Neuroprotective Metabolite of Docosahexaenoic Acid. Mol. Aspects Med. 2018, 64, 34–44. [Google Scholar] [CrossRef]
- Makrides, M.; Neumann, M.A.; Byard, R.W.; Simmer, K.; Gibson, R.A. Fatty Acid Composition of Brain, Retina, and Erythrocytes in Breast- and Formula-Fed Infants. Am. J. Clin. Nutr. 1994, 60, 189–194. [Google Scholar] [CrossRef]
- Haggarty, P. Fatty Acid Supply to the Human Fetus. Annu. Rev. Nutr. 2010, 30, 237–255. [Google Scholar] [CrossRef]
- Lauritzen, L.; Hansen, H.S.; Jørgensen, M.H.; Michaelsen, K.F. The Essentiality of Long Chain N-3 Fatty Acids in Relation to Development and Function of the Brain and Retina. Prog. Lipid Res. 2001, 40, 1–94. [Google Scholar] [CrossRef]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Intrauterine Fatty Acid Accretion Rates in Human Brain: Implications for Fatty Acid Requirements. Early Hum. Dev. 1980, 4, 121–129. [Google Scholar] [CrossRef]
- Percy, P.; Percy, A.; Vilbergsson, G.; Månsson, J.E. Polyunsaturated Fatty Acid Accretion in First- and Second-Trimester Human Fetal Brain: Lack of Correlation with Levels in Paired Placental Samples. Biochem. Mol. Med. 1996, 59, 38–43. [Google Scholar] [CrossRef]
- Martinez, M. Developmental Profiles of Polyunsaturated Fatty Acids in the Brain of Normal Infants and Patients with Peroxisomal Diseases: Severe Deficiency of Docosahexaenoic Acid in Zellweger’s and Pseudo-Zellweger’s Syndromes. World Rev. Nutr. Diet. 1991, 66, 87–102. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, R.S.; Luxwolda, M.F.; Offringa, P.J.; Boersma, E.R.; Dijck-Brouwer, D.A.J.; Muskiet, F.A.J. Fetal Intrauterine Whole Body Linoleic, Arachidonic and Docosahexaenoic Acid Contents and Accretion Rates. Prostaglandins Leukot. Essent. Fatty Acids 2012, 86, 13–20. [Google Scholar] [CrossRef]
- Metherel, A.H.; Kitson, A.P.; Domenichiello, A.F.; Lacombe, R.J.S.; Hopperton, K.E.; Trépanier, M.-O.; Alashmali, S.M.; Lin, L.; Bazinet, R.P. Maternal Liver Docosahexaenoic Acid (DHA) Stores Are Increased via Higher Serum Unesterified DHA Uptake in Pregnant Long Evans Rats. J. Nutr. Biochem. 2017, 46, 143–150. [Google Scholar] [CrossRef]
- Metherel, A.H.; Kitson, A.P.; Domenichiello, A.F.; Lacombe, R.J.S.; Hopperton, K.E.; Trépanier, M.-O.; Alashmali, S.M.; Lin, L.; Bazinet, R.P. Docosahexaenoic Acid (DHA) Accretion in the Placenta but Not the Fetus Is Matched by Plasma Unesterified DHA Uptake Rates in Pregnant Long Evans Rats. Placenta 2017, 58, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, A.J.; Crawford, M.A. The Incorporation of Linolenic Aid and Docosahexaenoic Acid into Liver and Brain Lipids of Developing Rats. FEBS Lett. 1972, 26, 127–129. [Google Scholar] [CrossRef]
- Calderon, F.; Kim, H.-Y. Docosahexaenoic Acid Promotes Neurite Growth in Hippocampal Neurons. J. Neurochem. 2004, 90, 979–988. [Google Scholar] [CrossRef]
- Sidhu, V.K.; Huang, B.X.; Kim, H.-Y. Effects of Docosahexaenoic Acid on Mouse Brain Synaptic Plasma Membrane Proteome Analyzed by Mass Spectrometry and (16)O/(18)O Labeling. J. Proteome Res. 2011, 10, 5472–5480. [Google Scholar] [CrossRef] [PubMed]
- Champeil-Potokar, G.; Chaumontet, C.; Guesnet, P.; Lavialle, M.; Denis, I. Docosahexaenoic Acid (22:6n-3) Enrichment of Membrane Phospholipids Increases Gap Junction Coupling Capacity in Cultured Astrocytes. Eur. J. Neurosci. 2006, 24, 3084–3090. [Google Scholar] [CrossRef]
- Rey, C.; Nadjar, A.; Joffre, F.; Amadieu, C.; Aubert, A.; Vaysse, C.; Pallet, V.; Layé, S.; Joffre, C. Maternal N-3 Polyunsaturated Fatty Acid Dietary Supply Modulates Microglia Lipid Content in the Offspring. Prostaglandins Leukot. Essent. Fatty Acids 2018, 133, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.Y.; Gehman, M.F.; Yeh, S.M. Maternal Dietary Fish Oil Enriches Docosahexaenoate Levels in Brain Subcellular Fractions of Offspring. J. Neurosci. Res. 1993, 35, 218–226. [Google Scholar] [CrossRef]
- Brand, A.; Schonfeld, E.; Isharel, I.; Yavin, E. Docosahexaenoic Acid-Dependent Iron Accumulation in Oligodendroglia Cells Protects from Hydrogen Peroxide-Induced Damage. J. Neurochem. 2008, 105, 1325–1335. [Google Scholar] [CrossRef] [PubMed]
- Harel, T.; Quek, D.Q.Y.; Wong, B.H.; Cazenave-Gassiot, A.; Wenk, M.R.; Fan, H.; Berger, I.; Shmueli, D.; Shaag, A.; Silver, D.L.; et al. Homozygous Mutation in MFSD2A, Encoding a Lysolipid Transporter for Docosahexanoic Acid, Is Associated with Microcephaly and Hypomyelination. Neurogenetics 2018, 19, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Madore, C.; Leyrolle, Q.; Morel, L.; Rossitto, M.; Greenhalgh, A.D.; Delpech, J.C.; Martinat, M.; Bosch-Bouju, C.; Bourel, J.; Rani, B.; et al. Essential Omega-3 Fatty Acids Tune Microglial Phagocytosis of Synaptic Elements in the Mouse Developing Brain. Nat. Commun. 2020, 11, 6133. [Google Scholar] [CrossRef] [PubMed]
- Bazinet, R.P.; Metherel, A.H.; Chen, C.T.; Shaikh, S.R.; Nadjar, A.; Joffre, C.; Layé, S. Brain Eicosapentaenoic Acid Metabolism as a Lead for Novel Therapeutics in Major Depression. Brain. Behav. Immun. 2020, 85, 21–28. [Google Scholar] [CrossRef]
- Lapillonne, A.; Moltu, S.J. Long-Chain Polyunsaturated Fatty Acids and Clinical Outcomes of Preterm Infants. Ann. Nutr. Metab. 2016, 69 (Suppl. 1), 35–44. [Google Scholar] [CrossRef]
- Martinez, M. Tissue Levels of Polyunsaturated Fatty Acids during Early Human Development. J. Pediatr. 1992, 120, S129–S138. [Google Scholar] [CrossRef]
- Larqué, E.; Krauss-Etschmann, S.; Campoy, C.; Hartl, D.; Linde, J.; Klingler, M.; Demmelmair, H.; Caño, A.; Gil, A.; Bondy, B.; et al. Docosahexaenoic Acid Supply in Pregnancy Affects Placental Expression of Fatty Acid Transport Proteins. Am. J. Clin. Nutr. 2006, 84, 853–861. [Google Scholar] [CrossRef]
- Innis, S.M. Polyunsaturated Fatty Acids in Human Milk: An Essential Role in Infant Development. Adv. Exp. Med. Biol. 2004, 554, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M.; Friesen, R.W. Essential N-3 Fatty Acids in Pregnant Women and Early Visual Acuity Maturation in Term Infants. Am. J. Clin. Nutr. 2008, 87, 548–557. [Google Scholar] [CrossRef]
- Gibson, R.A.; Neumann, M.A.; Makrides, M. Effect of Dietary Docosahexaenoic Acid on Brain Composition and Neural Function in Term Infants. Lipids 1996, 31, S177–S181. [Google Scholar] [CrossRef] [PubMed]
- Wierzejska, R.; Jarosz, M.; Wojda, B.; Siuba-Strzelińska, M. Dietary Intake of DHA during Pregnancy: A Significant Gap between the Actual Intake and Current Nutritional Recommendations. Rocz. Panstw. Zakl. Hig. 2018, 69, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, C.E.; Makrides, M.; Yuan, Z.-X.; Horowitz, M.S.; Zamora, D.; Yelland, L.N.; Best, K.; Jensen, J.; Taha, A.Y.; Gibson, R.A. Plasma Oxylipins and Unesterified Precursor Fatty Acids Are Altered by DHA Supplementation in Pregnancy: Can They Help Predict Risk of Preterm Birth? Prostaglandins Leukot. Essent. Fatty Acids 2020, 153, 102041. [Google Scholar] [CrossRef]
- von Schacky, C. Omega-3 Fatty Acids in Pregnancy-The Case for a Target Omega-3 Index. Nutrients 2020, 12, 898. [Google Scholar] [CrossRef]
- Yelland, L.N.; Gajewski, B.J.; Colombo, J.; Gibson, R.A.; Makrides, M.; Carlson, S.E. Predicting the Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation to Reduce Early Preterm Birth in Australia and the United States Using Results of within Country Randomized Controlled Trials. Prostaglandins Leukot. Essent. Fatty Acids 2016, 112, 44–49. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P.; DOMInO Investigative Team. Effect of DHA Supplementation during Pregnancy on Maternal Depression and Neurodevelopment of Young Children: A Randomized Controlled Trial. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef]
- Zhou, S.J.; Best, K.; Gibson, R.; McPhee, A.; Yelland, L.; Quinlivan, J.; Makrides, M. Study Protocol for a Randomised Controlled Trial Evaluating the Effect of Prenatal Omega-3 LCPUFA Supplementation to Reduce the Incidence of Preterm Birth: The ORIP Trial. BMJ Open 2017, 7, e018360. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Best, K.; Yelland, L.; McPhee, A.; Zhou, S.; Quinlivan, J.; Dodd, J.; Atkinson, E.; Safa, H.; van Dam, J.; et al. A Randomized Trial of Prenatal N-3 Fatty Acid Supplementation and Preterm Delivery. N. Engl. J. Med. 2019, 381, 1035–1045. [Google Scholar] [CrossRef]
- Olsen, S.F.; Halldorsson, T.I.; Thorne-Lyman, A.L.; Strøm, M.; Gørtz, S.; Granstrøm, C.; Nielsen, P.H.; Wohlfahrt, J.; Lykke, J.A.; Langhoff-Roos, J.; et al. Plasma Concentrations of Long Chain N-3 Fatty Acids in Early and Mid-Pregnancy and Risk of Early Preterm Birth. EBioMedicine 2018, 35, 325–333. [Google Scholar] [CrossRef]
- Lorentzen, B.; Drevon, C.A.; Endresen, M.J.; Henriksen, T. Fatty Acid Pattern of Esterified and Free Fatty Acids in Sera of Women with Normal and Pre-Eclamptic Pregnancy. Br. J. Obstet. Gynaecol. 1995, 102, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Campbell, F.M.; Bush, P.G.; Veerkamp, J.H.; Dutta-Roy, A.K. Detection and Cellular Localization of Plasma Membrane-Associated and Cytoplasmic Fatty Acid-Binding Proteins in Human Placenta. Placenta 1998, 19, 409–415. [Google Scholar] [CrossRef]
- Campbell, F.M.; Dutta-Roy, A.K. Plasma Membrane Fatty Acid-Binding Protein (FABPpm) Is Exclusively Located in the Maternal Facing Membranes of the Human Placenta. FEBS Lett. 1995, 375, 227–230. [Google Scholar] [CrossRef]
- Gil-Sánchez, A.; Larqué, E.; Demmelmair, H.; Acien, M.I.; Faber, F.L.; Parrilla, J.J.; Koletzko, B. Maternal-Fetal in Vivo Transfer of [13C]Docosahexaenoic and Other Fatty Acids across the Human Placenta 12 h after Maternal Oral Intake. Am. J. Clin. Nutr. 2010, 92, 115–122. [Google Scholar] [CrossRef]
- Biron-Shental, T.; Schaiff, W.T.; Ratajczak, C.K.; Bildirici, I.; Nelson, D.M.; Sadovsky, Y. Hypoxia Regulates the Expression of Fatty Acid-Binding Proteins in Primary Term Human Trophoblasts. Am. J. Obstet. Gynecol. 2007, 197, 516.e1–516.e6. [Google Scholar] [CrossRef] [PubMed]
- Leroy, C.; Tobin, K.A.R.; Basak, S.; Cathrine Staff, A.; Duttaroy, A.K. Fatty Acid-Binding Protein3 Expression in BeWo Cells, a Human Placental Choriocarcinoma Cell Line. Prostaglandins Leukot. Essent. Fatty Acids 2017, 120, 1–7. [Google Scholar] [CrossRef]
- Islam, A.; Kagawa, Y.; Sharifi, K.; Ebrahimi, M.; Miyazaki, H.; Yasumoto, Y.; Kawamura, S.; Yamamoto, Y.; Sakaguti, S.; Sawada, T.; et al. Fatty Acid Binding Protein 3 Is Involved in N-3 and n-6 PUFA Transport in Mouse Trophoblasts. J. Nutr. 2014, 144, 1509–1516. [Google Scholar] [CrossRef] [PubMed]
- Makkar, A.; Mishima, T.; Chang, G.; Scifres, C.; Sadovsky, Y. Fatty Acid Binding Protein-4 Is Expressed in the Mouse Placental Labyrinth, yet Is Dispensable for Placental Triglyceride Accumulation and Fetal Growth. Placenta 2014, 35, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Díaz, P.; Harris, J.; Rosario, F.J.; Powell, T.L.; Jansson, T. Increased Placental Fatty Acid Transporter 6 and Binding Protein 3 Expression and Fetal Liver Lipid Accumulation in a Mouse Model of Obesity in Pregnancy. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R1569–R1577. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.M.; Wadsack, C.; Desoye, G. Placental Fatty Acid Transfer. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Hanebutt, F.L.; Demmelmair, H.; Schiessl, B.; Larqué, E.; Koletzko, B. Long-Chain Polyunsaturated Fatty Acid (LC-PUFA) Transfer across the Placenta. Clin. Nutr. Edinb. Scotl. 2008, 27, 685–693. [Google Scholar] [CrossRef]
- Innis, S.M. Essential Fatty Acid Transfer and Fetal Development. Placenta 2005, 26 (Suppl. A), S70–S75. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Kitson, A.P.; Hopperton, K.E.; Domenichiello, A.F.; Trépanier, M.-O.; Lin, L.E.; Ermini, L.; Post, M.; Thies, F.; Bazinet, R.P. Plasma Non-Esterified Docosahexaenoic Acid Is the Major Pool Supplying the Brain. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Lacombe, R.J.S.; Chouinard-Watkins, R.; Bazinet, R.P. Brain Docosahexaenoic Acid Uptake and Metabolism. Mol. Aspects Med. 2018, 64, 109–134. [Google Scholar] [CrossRef] [PubMed]
- Duttaroy, A.K. Transport of Fatty Acids across the Human Placenta: A Review. Prog. Lipid Res. 2009, 48, 52–61. [Google Scholar] [CrossRef]
- Zhao, Z.; Nelson, A.R.; Betsholtz, C.; Zlokovic, B.V. Establishment and Dysfunction of the Blood-Brain Barrier. Cell 2015, 163, 1064–1078. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.N.; Ma, D.; Shui, G.; Wong, P.; Cazenave-Gassiot, A.; Zhang, X.; Wenk, M.R.; Goh, E.L.K.; Silver, D.L. Mfsd2a Is a Transporter for the Essential Omega-3 Fatty Acid Docosahexaenoic Acid. Nature 2014, 509, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.P.; Wong, B.H.; Chin, C.F.; Galam, D.L.A.; Foo, J.C.; Wong, L.C.; Ghosh, S.; Wenk, M.R.; Cazenave-Gassiot, A.; Silver, D.L. The Lysolipid Transporter Mfsd2a Regulates Lipogenesis in the Developing Brain. PLoS Biol. 2018, 16, e2006443. [Google Scholar] [CrossRef] [PubMed]
- Ben-Zvi, A.; Lacoste, B.; Kur, E.; Andreone, B.J.; Mayshar, Y.; Yan, H.; Gu, C. Mfsd2a Is Critical for the Formation and Function of the Blood-Brain Barrier. Nature 2014, 509, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Andreone, B.J.; Chow, B.W.; Tata, A.; Lacoste, B.; Ben-Zvi, A.; Bullock, K.; Deik, A.A.; Ginty, D.D.; Clish, C.B.; Gu, C. Blood-Brain Barrier Permeability Is Regulated by Lipid Transport-Dependent Suppression of Caveolae-Mediated Transcytosis. Neuron 2017, 94, 581–594.e5. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Sánchez, M.T.; Ruiz-Palacios, M.; Blanco-Carnero, J.E.; Pagan, A.; Hellmuth, C.; Uhl, O.; Peissner, W.; Ruiz-Alcaraz, A.J.; Parrilla, J.J.; Koletzko, B.; et al. Placental MFSD2a Transporter Is Related to Decreased DHA in Cord Blood of Women with Treated Gestational Diabetes. Clin. Nutr. Edinb. Scotl. 2017, 36, 513–521. [Google Scholar] [CrossRef]
- Duttaroy, A.K.; Basak, S. Maternal Dietary Fatty Acids and Their Roles in Human Placental Development. Prostaglandins Leukot. Essent. Fatty Acids 2020, 155, 102080. [Google Scholar] [CrossRef] [PubMed]
- Alakbarzade, V.; Hameed, A.; Quek, D.Q.Y.; Chioza, B.A.; Baple, E.L.; Cazenave-Gassiot, A.; Nguyen, L.N.; Wenk, M.R.; Ahmad, A.Q.; Sreekantan-Nair, A.; et al. A Partially Inactivating Mutation in the Sodium-Dependent Lysophosphatidylcholine Transporter MFSD2A Causes a Non-Lethal Microcephaly Syndrome. Nat. Genet. 2015, 47, 814–817. [Google Scholar] [CrossRef]
- Guemez-Gamboa, A.; Nguyen, L.N.; Yang, H.; Zaki, M.S.; Kara, M.; Ben-Omran, T.; Akizu, N.; Rosti, R.O.; Rosti, B.; Scott, E.; et al. Inactivating Mutations in MFSD2A, Required for Omega-3 Fatty Acid Transport in Brain, Cause a Lethal Microcephaly Syndrome. Nat. Genet. 2015, 47, 809–813. [Google Scholar] [CrossRef]
- Zhou, J.; Chi, X.; Cheng, M.; Huang, X.; Liu, X.; Fan, J.; Xu, H.; Lin, T.; Shi, L.; Qin, C.; et al. Zika Virus Degrades the ω-3 Fatty Acid Transporter Mfsd2a in Brain Microvascular Endothelial Cells and Impairs Lipid Homeostasis. Sci. Adv. 2019, 5. [Google Scholar] [CrossRef]
- Shimomura, I.; Shimano, H.; Horton, J.D.; Goldstein, J.L.; Brown, M.S. Differential Expression of Exons 1a and 1c in MRNAs for Sterol Regulatory Element Binding Protein-1 in Human and Mouse Organs and Cultured Cells. J. Clin. Invest. 1997, 99, 838–845. [Google Scholar] [CrossRef]
- Zhu, H.; Fan, C.; Xu, F.; Tian, C.; Zhang, F.; Qi, K. Dietary Fish Oil N-3 Polyunsaturated Fatty Acids and Alpha-Linolenic Acid Differently Affect Brain Accretion of Docosahexaenoic Acid and Expression of Desaturases and Sterol Regulatory Element-Binding Protein 1 in Mice. J. Nutr. Biochem. 2010, 21, 954–960. [Google Scholar] [CrossRef]
- Chen, Y.; Bang, S.; McMullen, M.F.; Kazi, H.; Talbot, K.; Ho, M.-X.; Carlson, G.; Arnold, S.E.; Ong, W.-Y.; Kim, S.F. Neuronal Activity-Induced Sterol Regulatory Element Binding Protein-1 (SREBP1) Is Disrupted in Dysbindin-Null Mice-Potential Link to Cognitive Impairment in Schizophrenia. Mol. Neurobiol. 2017, 54, 1699–1709. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kang, S.; Ang, M.J.; Kim, J.; Kim, J.C.; Kim, S.-H.; Jeon, T.-I.; Jung, C.; Im, S.-S.; Moon, C. Deficiency of Sterol Regulatory Element-Binding Protein-1c Induces Schizophrenia-like Behavior in Mice. Genes Brain Behav. 2019, 18, e12540. [Google Scholar] [CrossRef]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and Arachidonic Acid Concentrations in Human Breast Milk Worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [CrossRef]
- Duttaroy, A.K. Fetal Growth and Development: Roles of Fatty Acid Transport Proteins and Nuclear Transcription Factors in Human Placenta. Indian J. Exp. Biol. 2004, 42, 747–757. [Google Scholar]
- Farquharson, J.; Cockburn, F.; Patrick, W.A.; Jamieson, E.C.; Logan, R.W. Infant Cerebral Cortex Phospholipid Fatty-Acid Composition and Diet. Lancet Lond. Engl. 1992, 340, 810–813. [Google Scholar] [CrossRef]
- Cacho, N.T.; Lawrence, R.M. Innate Immunity and Breast Milk. Front. Immunol. 2017, 8, 584. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.A. La Lactancia Materna. Una Guía Para la Profesiónmédica, 4th ed.; Mosby/Doyma: Madrid, Spain, 1996. [Google Scholar]
- Marangoni, F.; Agostoni, C.; Lammardo, A.M.; Giovannini, M.; Galli, C.; Riva, E. Polyunsaturated Fatty Acid Concentrations in Human Hindmilk Are Stable throughout 12-Months of Lactation and Provide a Sustained Intake to the Infant during Exclusive Breastfeeding: An Italian Study. Br. J. Nutr. 2000, 84, 103–109. [Google Scholar] [CrossRef]
- Thiemich, M. Ueber Den Einfuss Der Ernahrung Und Lebensweise Auf Die Zusammensetzung Der Frauenmilch. Monatssch Geburtshilfe Gynakol 1899, 9, 504–521. [Google Scholar]
- Helland, I.B.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Fatty Acid Composition in Maternal Milk and Plasma during Supplementation with Cod Liver Oil. Eur. J. Clin. Nutr. 1998, 52, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Connor, W.E.; Lindsey, S. Will Dietary Omega-3 Fatty Acids Change the Composition of Human Milk? Am. J. Clin. Nutr. 1984, 40, 780–785. [Google Scholar] [CrossRef]
- Henderson, R.A.; Jensen, R.G.; Lammi-Keefe, C.J.; Ferris, A.M.; Dardick, K.R. Effect of Fish Oil on the Fatty Acid Composition of Human Milk and Maternal and Infant Erythrocytes. Lipids 1992, 27, 863–869. [Google Scholar] [CrossRef]
- Al, M.D.; van Houwelingen, A.C.; Hornstra, G. Relation between Birth Order and the Maternal and Neonatal Docosahexaenoic Acid Status. Eur. J. Clin. Nutr. 1997, 51, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Neumann, M.; Simmer, K.; Pater, J.; Gibson, R. Are Long-Chain Polyunsaturated Fatty Acids Essential Nutrients in Infancy? Lancet Lond. Engl. 1995, 345, 1463–1468. [Google Scholar] [CrossRef]
- Heird, W.C. The Role of Polyunsaturated Fatty Acids in Term and Preterm Infants and Breastfeeding Mothers. Pediatr. Clin. N. Am. 2001, 48, 173–188. [Google Scholar] [CrossRef]
- Bopp, M.; Lovelady, C.; Hunter, C.; Kinsella, T. Maternal Diet and Exercise: Effects on Long-Chain Polyunsaturated Fatty Acid Concentrations in Breast Milk. J. Am. Diet. Assoc. 2005, 105, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Riva, E.; Giovannini, M.; Pinto, F.; Colombo, C.; Risé, P.; Galli, C.; Marangoni, F. Maternal Smoking Habits Are Associated with Differences in Infants’ Long-Chain Polyunsaturated Fatty Acids in Whole Blood: A Case-Control Study. Arch. Dis. Child. 2008, 93, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Neumann, M.A.; Gibson, R.A. Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Breast Milk Composition. Eur. J. Clin. Nutr. 1996, 50, 352–357. [Google Scholar] [PubMed]
- Salem, N.; Van Dael, P. Arachidonic Acid in Human Milk. Nutrients 2020, 12, 626. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.T.; Palac, H.L.; Baillif, V.; Van Goethem, E.; Dubourdeau, M.; Van Horn, L.; Martin, C.R. Long Chain Fatty Acids and Related Pro-Inflammatory, Specialized pro-Resolving Lipid Mediators and Their Intermediates in Preterm Human Milk during the First Month of Lactation. Prostaglandins Leukot. Essent. Fatty Acids 2017, 121, 1–6. [Google Scholar] [CrossRef]
- Gan, J.; Zhang, Z.; Kurudimov, K.; German, J.B.; Taha, A.Y. Distribution of Free and Esterified Oxylipins in Cream, Cell, and Skim Fractions of Human Milk. Lipids 2020, 55, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Pitino, M.A.; Alashmali, S.M.; Hopperton, K.E.; Unger, S.; Pouliot, Y.; Doyen, A.; O’Connor, D.L.; Bazinet, R.P. Oxylipin Concentration, but Not Fatty Acid Composition, Is Altered in Human Donor Milk Pasteurised Using Both Thermal and Non-Thermal Techniques. Br. J. Nutr. 2019, 122, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Hennebelle, M.; Morgan, R.K.; Sethi, S.; Zhang, Z.; Chen, H.; Grodzki, A.C.; Lein, P.J.; Taha, A.Y. Linoleic Acid-Derived Metabolites Constitute the Majority of Oxylipins in the Rat Pup Brain and Stimulate Axonal Growth in Primary Rat Cortical Neuron-Glia Co-Cultures in a Sex-Dependent Manner. J. Neurochem. 2020, 152, 195–207. [Google Scholar] [CrossRef]
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, Regional, and National Causes of under-5 Mortality in 2000-15: An Updated Systematic Analysis with Implications for the Sustainable Development Goals. Lancet Lond. Engl. 2016, 388, 3027–3035. [Google Scholar] [CrossRef]
- Hutchinson, E.A.; De Luca, C.R.; Doyle, L.W.; Roberts, G.; Anderson, P.J.; Victorian Infant Collaborative Study Group. School-Age Outcomes of Extremely Preterm or Extremely Low Birth Weight Children. Pediatrics 2013, 131, e1053–e1061. [Google Scholar] [CrossRef] [PubMed]
- Crowther, C.A.; Hiller, J.E.; Doyle, L.W.; Haslam, R.R.; Australasian Collaborative Trial of Magnesium Sulphate (ACTOMg SO4) Collaborative Group. Effect of Magnesium Sulfate given for Neuroprotection before Preterm Birth: A Randomized Controlled Trial. JAMA 2003, 290, 2669–2676. [Google Scholar] [CrossRef]
- Schmidt, B.; Roberts, R.S.; Davis, P.; Doyle, L.W.; Barrington, K.J.; Ohlsson, A.; Solimano, A.; Tin, W.; Caffeine for Apnea of Prematurity Trial Group. Long-Term Effects of Caffeine Therapy for Apnea of Prematurity. N. Engl. J. Med. 2007, 357, 1893–1902. [Google Scholar] [CrossRef]
- Puntis, J.W.L. Nutritional Support in the Premature Newborn. Postgrad. Med. J. 2006, 82, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Dorling, J.; Kempley, S.; Leaf, A. Feeding Growth Restricted Preterm Infants with Abnormal Antenatal Doppler Results. Arch. Dis. Child. Fetal Neonatal Ed. 2005, 90, F359–F363. [Google Scholar] [CrossRef]
- Schanler, R.J.; Shulman, R.J.; Lau, C. Feeding Strategies for Premature Infants: Beneficial Outcomes of Feeding Fortified Human Milk versus Preterm Formula. Pediatrics 1999, 103, 1150–1157. [Google Scholar] [CrossRef]
- Henriksen, C.; Haugholt, K.; Lindgren, M.; Aurvåg, A.K.; Rønnestad, A.; Grønn, M.; Solberg, R.; Moen, A.; Nakstad, B.; Berge, R.K.; et al. Improved Cognitive Development among Preterm Infants Attributable to Early Supplementation of Human Milk with Docosahexaenoic Acid and Arachidonic Acid. Pediatrics 2008, 121, 1137–1145. [Google Scholar] [CrossRef]
- Simmer, K.; Schulzke, S.M.; Patole, S. Long-Chain Polyunsaturated Fatty Acid Supplementation in Preterm Infants. Cochrane Database Syst. Rev. 2008, CD000375. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Collins, C.T.; Davis, P.G.; Doyle, L.W.; Simmer, K.; Colditz, P.B.; Morris, S.; Smithers, L.G.; et al. Neurodevelopmental Outcomes of Preterm Infants Fed High-Dose Docosahexaenoic Acid: A Randomized Controlled Trial. JAMA 2009, 301, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Gould, J.F.; Smithers, L.G.; Makrides, M. The Effect of Maternal Omega-3 (n-3) LCPUFA Supplementation during Pregnancy on Early Childhood Cognitive and Visual Development: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2013, 97, 531–544. [Google Scholar] [CrossRef]
- Innis, S.M. Essential Fatty Acids in Infant Nutrition: Lessons and Limitations from Animal Studies in Relation to Studies on Infant Fatty Acid Requirements. Am. J. Clin. Nutr. 2000, 71, 238S–244S. [Google Scholar] [CrossRef] [PubMed]
- Gould, J.F.; Colombo, J.; Collins, C.T.; Makrides, M.; Hewawasam, E.; Smithers, L.G. Assessing Whether Early Attention of Very Preterm Infants Can Be Improved by an Omega-3 Long-Chain Polyunsaturated Fatty Acid Intervention: A Follow-up of a Randomised Controlled Trial. BMJ Open 2018, 8, e020043. [Google Scholar] [CrossRef]
- Isaacs, E.B.; Ross, S.; Kennedy, K.; Weaver, L.T.; Lucas, A.; Fewtrell, M.S. 10-Year Cognition in Preterms after Random Assignment to Fatty Acid Supplementation in Infancy. Pediatrics 2011, 128, e890–e898. [Google Scholar] [CrossRef]
- Westerberg, A.C.; Schei, R.; Henriksen, C.; Smith, L.; Veierød, M.B.; Drevon, C.A.; Iversen, P.O. Attention among Very Low Birth Weight Infants Following Early Supplementation with Docosahexaenoic and Arachidonic Acid. Acta Paediatr. Oslo Nor. 1992 2011, 100, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Qawasmi, A.; Landeros-Weisenberger, A.; Bloch, M.H. Meta-Analysis of LCPUFA Supplementation of Infant Formula and Visual Acuity. Pediatrics 2013, 131, e262–e272. [Google Scholar] [CrossRef] [PubMed]
- Klevebro, S.; Juul, S.E.; Wood, T.R. A More Comprehensive Approach to the Neuroprotective Potential of Long-Chain Polyunsaturated Fatty Acids in Preterm Infants Is Needed-Should We Consider Maternal Diet and the n-6:N-3 Fatty Acid Ratio? Front. Pediatr. 2019, 7, 533. [Google Scholar] [CrossRef] [PubMed]
- Birch, E.E.; Garfield, S.; Hoffman, D.R.; Uauy, R.; Birch, D.G. A Randomized Controlled Trial of Early Dietary Supply of Long-Chain Polyunsaturated Fatty Acids and Mental Development in Term Infants. Dev. Med. Child Neurol. 2000, 42, 174–181. [Google Scholar] [CrossRef]
- Scott, B.L.; Bazan, N.G. Membrane Docosahexaenoate Is Supplied to the Developing Brain and Retina by the Liver. Proc. Natl. Acad. Sci. USA 1989, 86, 2903–2907. [Google Scholar] [CrossRef] [PubMed]
- Dhopeshwarkar, G.A.; Subramanian, C. Biosynthesis of Polyunsaturated Fatty Acids in the Developing Brain: I. Metabolic Transformations of Intracranially Administered 1-14C Linolenic Acid. Lipids 1976, 11, 67–71. [Google Scholar] [CrossRef]
- Dhopeshwarkar, G.A.; Subramanian, C. Intracranial Conversion of Linoleic Acid to Arachidonic Acid: Evidence for Lack of Delta8 Desaturase in the Brain. J. Neurochem. 1976, 26, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Sanders, T.A.; Naismith, D.J. The Metabolism of Alpha-Linolenic Acid by the Foetal Rat. Br. J. Nutr. 1980, 44, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Sanders, T.A.; Rana, S.K. Comparison of the Metabolism of Linoleic and Linolenic Acids in the Fetal Rat. Ann. Nutr. Metab. 1987, 31, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.P.; Nakamura, M.; Clarke, S.D. Cloning, Expression, and Fatty Acid Regulation of the Human Delta-5 Desaturase. J. Biol. Chem. 1999, 274, 37335–37339. [Google Scholar] [CrossRef]
- Aki, T.; Shimada, Y.; Inagaki, K.; Higashimoto, H.; Kawamoto, S.; Shigeta, S.; Ono, K.; Suzuki, O. Molecular Cloning and Functional Characterization of Rat Delta-6 Fatty Acid Desaturase. Biochem. Biophys. Res. Commun. 1999, 255, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Leonard, A.E.; Kelder, B.; Bobik, E.G.; Chuang, L.T.; Parker-Barnes, J.M.; Thurmond, J.M.; Kroeger, P.E.; Kopchick, J.J.; Huang, Y.S.; Mukerji, P. CDNA Cloning and Characterization of Human Delta5-Desaturase Involved in the Biosynthesis of Arachidonic Acid. Biochem. J. 2000, 347 Pt 3, 719–724. [Google Scholar] [CrossRef]
- Al-Hilal, M.; Alsaleh, A.; Maniou, Z.; Lewis, F.J.; Hall, W.L.; Sanders, T.A.B.; O’Dell, S.D.; MARINA study team. Genetic Variation at the FADS1-FADS2 Gene Locus Influences Delta-5 Desaturase Activity and LC-PUFA Proportions after Fish Oil Supplement. J. Lipid Res. 2013, 54, 542–551. [Google Scholar] [CrossRef]
- Lemaitre, R.N.; Tanaka, T.; Tang, W.; Manichaikul, A.; Foy, M.; Kabagambe, E.K.; Nettleton, J.A.; King, I.B.; Weng, L.-C.; Bhattacharya, S.; et al. Genetic Loci Associated with Plasma Phospholipid N-3 Fatty Acids: A Meta-Analysis of Genome-Wide Association Studies from the CHARGE Consortium. PLoS Genet. 2011, 7, e1002193. [Google Scholar] [CrossRef]
- Deák, F.; Anderson, R.E.; Fessler, J.L.; Sherry, D.M. Novel Cellular Functions of Very Long Chain-Fatty Acids: Insight From ELOVL4 Mutations. Front. Cell. Neurosci. 2019, 13, 428. [Google Scholar] [CrossRef]
- Monroig, O.; Rotllant, J.; Sánchez, E.; Cerdá-Reverter, J.M.; Tocher, D.R. Expression of Long-Chain Polyunsaturated Fatty Acid (LC-PUFA) Biosynthesis Genes during Zebrafish Danio Rerio Early Embryogenesis. Biochim. Biophys. Acta 2009, 1791, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.-H.; Chung, H.-H.; Shu-Chien, A.C. Distinct Developmental Expression of Two Elongase Family Members in Zebrafish. Biochem. Biophys. Res. Commun. 2010, 393, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, S.J.; Cox, S.R.; Shen, X.; Lombardo, M.V.; Reus, L.M.; Alloza, C.; Harris, M.A.; Alderson, H.L.; Hunter, S.; Neilson, E.; et al. Sex Differences in the Adult Human Brain: Evidence from 5216 UK Biobank Participants. Cereb. Cortex N. Y. NY 2018, 28, 2959–2975. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Lombardo, M.V.; Auyeung, B.; Ashwin, E.; Chakrabarti, B.; Knickmeyer, R. Why Are Autism Spectrum Conditions More Prevalent in Males? PLoS Biol. 2011, 9, e1001081. [Google Scholar] [CrossRef] [PubMed]
- Gaub, M.; Carlson, C.L. Gender Differences in ADHD: A Meta-Analysis and Critical Review. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 1036–1045. [Google Scholar] [CrossRef] [PubMed]
- Aleman, A.; Kahn, R.S.; Selten, J.-P. Sex Differences in the Risk of Schizophrenia: Evidence from Meta-Analysis. Arch. Gen. Psychiatry 2003, 60, 565–571. [Google Scholar] [CrossRef]
- Williamson, D.; Johnston, C. Gender Differences in Adults with Attention-Deficit/Hyperactivity Disorder: A Narrative Review. Clin. Psychol. Rev. 2015, 40, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Jäncke, L. Sex/Gender Differences in Cognition, Neurophysiology, and Neuroanatomy. F1000Research 2018, 7. [Google Scholar] [CrossRef]
- Crowe, F.L.; Skeaff, C.M.; Green, T.J.; Gray, A.R. Serum N-3 Long-Chain PUFA Differ by Sex and Age in a Population-Based Survey of New Zealand Adolescents and Adults. Br. J. Nutr. 2008, 99, 168–174. [Google Scholar] [CrossRef]
- Bakewell, L.; Burdge, G.C.; Calder, P.C. Polyunsaturated Fatty Acid Concentrations in Young Men and Women Consuming Their Habitual Diets. Br. J. Nutr. 2006, 96, 93–99. [Google Scholar] [CrossRef]
- Metherel, A.H.; Armstrong, J.M.; Patterson, A.C.; Stark, K.D. Assessment of Blood Measures of N-3 Polyunsaturated Fatty Acids with Acute Fish Oil Supplementation and Washout in Men and Women. Prostaglandins Leukot. Essent. Fatty Acids 2009, 81, 23–29. [Google Scholar] [CrossRef]
- Extier, A.; Langelier, B.; Perruchot, M.-H.; Guesnet, P.; Van Veldhoven, P.P.; Lavialle, M.; Alessandri, J.-M. Gender Affects Liver Desaturase Expression in a Rat Model of N-3 Fatty Acid Repletion. J. Nutr. Biochem. 2010, 21, 180–187. [Google Scholar] [CrossRef]
- Kitson, A.P.; Smith, T.L.; Marks, K.A.; Stark, K.D. Tissue-Specific Sex Differences in Docosahexaenoic Acid and Δ6-Desaturase in Rats Fed a Standard Chow Diet. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2012, 37, 1200–1211. [Google Scholar] [CrossRef]
- Giltay, E.J.; Gooren, L.J.G.; Toorians, A.W.F.T.; Katan, M.B.; Zock, P.L. Docosahexaenoic Acid Concentrations Are Higher in Women than in Men Because of Estrogenic Effects. Am. J. Clin. Nutr. 2004, 80, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Abdelmagid, S.A.; Clarke, S.E.; Roke, K.; Nielsen, D.E.; Badawi, A.; El-Sohemy, A.; Mutch, D.M.; Ma, D.W. Ethnicity, Sex, FADS Genetic Variation, and Hormonal Contraceptive Use Influence Delta-5- and Delta-6-Desaturase Indices and Plasma Docosahexaenoic Acid Concentration in Young Canadian Adults: A Cross-Sectional Study. Nutr. Metab. 2015, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Choi, J.-E.; Park, Y. Low-Linoleic Acid Diet and Oestrogen Enhance the Conversion of α-Linolenic Acid into DHA through Modification of Conversion Enzymes and Transcription Factors. Br. J. Nutr. 2019, 121, 137–145. [Google Scholar] [CrossRef]
- McNamara, R.K.; Able, J.; Jandacek, R.; Rider, T.; Tso, P. Gender Differences in Rat Erythrocyte and Brain Docosahexaenoic Acid Composition: Role of Ovarian Hormones and Dietary Omega-3 Fatty Acid Composition. Psychoneuroendocrinology 2009, 34, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Alessandri, J.-M.; Extier, A.; Al-Gubory, K.H.; Langelier, B.; Baudry, C.; LePoupon, C.; Lavialle, M.; Guesnet, P. Ovariectomy and 17β-Estradiol Alter Transcription of Lipid Metabolism Genes and Proportions of Neo-Formed n-3 and n-6 Long-Chain Polyunsaturated Fatty Acids Differently in Brain and Liver. J. Nutr. Biochem. 2011, 22, 820–827. [Google Scholar] [CrossRef]
- Feltham, B.A.; Balogun, K.A.; Cheema, S.K. Perinatal and Postweaning Diets High in Omega-3 Fatty Acids Have Age- and Sex-Specific Effects on the Fatty Acid Composition of the Cerebellum and Brainstem of C57BL/6 Mice. Prostaglandins Leukot. Essent. Fatty Acids 2019, 148, 16–24. [Google Scholar] [CrossRef]
- Childs, C.E.; Romeu-Nadal, M.; Burdge, G.C.; Calder, P.C. Gender Differences in the N-3 Fatty Acid Content of Tissues. Proc. Nutr. Soc. 2008, 67, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Haven, S.; Lecaj, L.; Borgstrom, M.; Torabi, M.; SanGiovanni, J.P.; Hibbeln, J.R. Brain PUFA Concentrations Are Differentially Affected by Interactions of Diet, Sex, Brain Regions, and Phospholipid Pools in Mice. J. Nutr. 2020, 150, 3123–3132. [Google Scholar] [CrossRef] [PubMed]
- Dervola, K.S.; Roberg, B.A.; Wøien, G.; Bogen, I.L.; Sandvik, T.H.; Sagvolden, T.; Drevon, C.A.; Johansen, E.B.; Walaas, S.I. Marine O-3 Polyunsaturated Fatty Acids Induce Sex-Specific Changes in Reinforcer-Controlled Behaviour and Neurotransmitter Metabolism in a Spontaneously Hypertensive Rat Model of ADHD. Behav. Brain Funct. BBF 2012, 8, 56. [Google Scholar] [CrossRef]
- da Costa, A.E.M.; Gomes, N.S.; Gadelha Filho, C.V.J.; Linhares, M.G.O.E.S.; da Costa, R.O.; Chaves Filho, A.J.M.; Cordeiro, R.C.; Vasconcelos, G.S.; da Silva, F.E.R.; Araujo, T. da S.; et al. Sex Influences in the Preventive Effects of Peripubertal Supplementation with N-3 Polyunsaturated Fatty Acids in Mice Exposed to the Two-Hit Model of Schizophrenia. Eur. J. Pharmacol. 2021, 897, 173949. [Google Scholar] [CrossRef]
- Trivers, R.L.; Willard, D.E. Natural Selection of Parental Ability to Vary the Sex Ratio of Offspring. Science 1973, 179, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Rivers, J.P.; Crawford, M.A. Maternal Nutrition and the Sex Ratio at Birth. Nature 1974, 252, 297–298. [Google Scholar] [CrossRef]
- Austad, S.N.; Sunquist, M.E. Sex-Ratio Manipulation in the Common Opossum. Nature 1986, 324, 58–60. [Google Scholar] [CrossRef]
- Fountain, E.D.; Mao, J.; Whyte, J.J.; Mueller, K.E.; Ellersieck, M.R.; Will, M.J.; Roberts, R.M.; Macdonald, R.; Rosenfeld, C.S. Effects of Diets Enriched in Omega-3 and Omega-6 Polyunsaturated Fatty Acids on Offspring Sex-Ratio and Maternal Behavior in Mice. Biol. Reprod. 2008, 78, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, N.; Cuffe, J.S.M.; Holland, O.J.; Bulmer, A.C.; Hill, M.; Perkins, A.V.; Muhlhausler, B.S.; McAinch, A.J.; Hryciw, D.H. Elevated Maternal Linoleic Acid Reduces Circulating Leptin Concentrations, Cholesterol Levels and Male Fetal Survival in a Rat Model. J. Physiol. 2019, 597, 3349–3361. [Google Scholar] [CrossRef]
- Gulliver, C.E.; Friend, M.A.; King, B.J.; Robertson, S.M.; Wilkins, J.F.; Clayton, E.H. Increased Prostaglandin Response to Oxytocin in Ewes Fed a Diet High in Omega-6 Polyunsaturated Fatty Acids. Lipids 2013, 48, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Abayasekara, D.R.; Wathes, D.C. Effects of Altering Dietary Fatty Acid Composition on Prostaglandin Synthesis and Fertility. Prostaglandins Leukot. Essent. Fatty Acids 1999, 61, 275–287. [Google Scholar] [CrossRef]
- Pratt, N.C.; Huck, U.W.; Lisk, R.D. Offspring Sex Ratio in Hamsters Is Correlated with Vaginal PH at Certain Times of Mating. Behav. Neural Biol. 1987, 48, 310–316. [Google Scholar] [CrossRef]
- Green, M.P.; Spate, L.D.; Parks, T.E.; Kimura, K.; Murphy, C.N.; Williams, J.E.; Kerley, M.S.; Green, J.A.; Keisler, D.H.; Roberts, R.M. Nutritional Skewing of Conceptus Sex in Sheep: Effects of a Maternal Diet Enriched in Rumen-Protected Polyunsaturated Fatty Acids (PUFA). Reprod. Biol. Endocrinol. RBE 2008, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Marei, W.F.A.; Khalil, W.A.; Pushpakumara, A.P.G.; El-Harairy, M.A.; Abo El-Atta, A.M.A.; Wathes, D.C.; Fouladi-Nashta, A. Polyunsaturated Fatty Acids Influence Offspring Sex Ratio in Cows. Int. J. Vet. Sci. Med. 2018, 6, S36–S40. [Google Scholar] [CrossRef]
- Lammoglia, M.A.; Willard, S.T.; Oldham, J.R.; Randel, R.D. Effects of Dietary Fat and Season on Steroid Hormonal Profiles before Parturition and on Hormonal, Cholesterol, Triglycerides, Follicular Patterns, and Postpartum Reproduction in Brahman Cows. J. Anim. Sci. 1996, 74, 2253–2262. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.S.; Pushpakumara, P.G.A.; Cheng, Z.; Peters, A.R.; Abayasekara, D.R.E.; Wathes, D.C. Effects of Dietary Polyunsaturated Fatty Acids on Ovarian and Uterine Function in Lactating Dairy Cows. Reprod. Camb. Engl. 2002, 124, 119–131. [Google Scholar] [CrossRef]
- Agung, B.; Otoi, T.; Wongsrikeao, P.; Taniguchi, M.; Shimizu, R.; Watari, H.; Nagai, T. Effect of Maturation Culture Period of Oocytes on the Sex Ratio of in Vitro Fertilized Bovine Embryos. J. Reprod. Dev. 2006, 52, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Gerber, A.J.; Peterson, B.S.; Giedd, J.N.; Lalonde, F.M.; Celano, M.J.; White, S.L.; Wallace, G.L.; Lee, N.R.; Lenroot, R.K. Anatomical Brain Magnetic Resonance Imaging of Typically Developing Children and Adolescents. J. Am. Acad. Child Adolesc. Psychiatry 2009, 48, 465–470. [Google Scholar] [CrossRef]
- Dubois, J.; Dehaene-Lambertz, G.; Kulikova, S.; Poupon, C.; Hüppi, P.S.; Hertz-Pannier, L. The Early Development of Brain White Matter: A Review of Imaging Studies in Fetuses, Newborns and Infants. Neuroscience 2014, 276, 48–71. [Google Scholar] [CrossRef]
- Coti Bertrand, P.; O’Kusky, J.R.; Innis, S.M. Maternal Dietary (n-3) Fatty Acid Deficiency Alters Neurogenesis in the Embryonic Rat Brain. J. Nutr. 2006, 136, 1570–1575. [Google Scholar] [CrossRef] [PubMed]
- Yavin, E.; Himovichi, E.; Eilam, R. Delayed Cell Migration in the Developing Rat Brain Following Maternal Omega 3 Alpha Linolenic Acid Dietary Deficiency. Neuroscience 2009, 162, 1011–1022. [Google Scholar] [CrossRef]
- Sakayori, N.; Kikkawa, T.; Tokuda, H.; Kiryu, E.; Yoshizaki, K.; Kawashima, H.; Yamada, T.; Arai, H.; Kang, J.X.; Katagiri, H.; et al. Maternal Dietary Imbalance between Omega-6 and Omega-3 Polyunsaturated Fatty Acids Impairs Neocortical Development via Epoxy Metabolites. Stem Cells Dayt. Ohio 2016, 34, 470–482. [Google Scholar] [CrossRef]
- Wong, C.T.; Ussyshkin, N.; Ahmad, E.; Rai-Bhogal, R.; Li, H.; Crawford, D.A. Prostaglandin E2 Promotes Neural Proliferation and Differentiation and Regulates Wnt Target Gene Expression. J. Neurosci. Res. 2016, 94, 759–775. [Google Scholar] [CrossRef] [PubMed]
- Joardar, A.; Sen, A.K.; Das, S. Docosahexaenoic Acid Facilitates Cell Maturation and Beta-Adrenergic Transmission in Astrocytes. J. Lipid Res. 2006, 47, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Das, S. Docosahexaenoic Acid (DHA) Induced Morphological Differentiation of Astrocytes Is Associated with Transcriptional Upregulation and Endocytosis of Β2-AR. Mol. Neurobiol. 2019, 56, 2685–2702. [Google Scholar] [CrossRef] [PubMed]
- Shinjyo, N.; Piscitelli, F.; Verde, R.; Di Marzo, V. Impact of Omega-6 Polyunsaturated Fatty Acid Supplementation and γ-Aminobutyric Acid on Astrogliogenesis through the Endocannabinoid System. J. Neurosci. Res. 2013, 91, 943–953. [Google Scholar] [CrossRef]
- Cremona, O.; Di Paolo, G.; Wenk, M.R.; Lüthi, A.; Kim, W.T.; Takei, K.; Daniell, L.; Nemoto, Y.; Shears, S.B.; Flavell, R.A.; et al. Essential Role of Phosphoinositide Metabolism in Synaptic Vesicle Recycling. Cell 1999, 99, 179–188. [Google Scholar] [CrossRef]
- Di Paolo, G.; Moskowitz, H.S.; Gipson, K.; Wenk, M.R.; Voronov, S.; Obayashi, M.; Flavell, R.; Fitzsimonds, R.M.; Ryan, T.A.; De Camilli, P. Impaired PtdIns(4,5)P2 Synthesis in Nerve Terminals Produces Defects in Synaptic Vesicle Trafficking. Nature 2004, 431, 415–422. [Google Scholar] [CrossRef]
- Rohrbough, J.; Rushton, E.; Palanker, L.; Woodruff, E.; Matthies, H.J.G.; Acharya, U.; Acharya, J.K.; Broadie, K. Ceramidase Regulates Synaptic Vesicle Exocytosis and Trafficking. J. Neurosci. Off. J. Soc. Neurosci. 2004, 24, 7789–7803. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.E.; Bazan, N.G. Changing Fatty Acid Content of Growth Cone Lipids Prior to Synaptogenesis. J. Neurochem. 1992, 59, 318–325. [Google Scholar] [CrossRef]
- Martin, R.E. Docosahexaenoic Acid Decreases Phospholipase A2 Activity in the Neurites/Nerve Growth Cones of PC12 Cells. J. Neurosci. Res. 1998, 54, 805–813. [Google Scholar] [CrossRef]
- Auestad, N.; Innis, S.M. Dietary N-3 Fatty Acid Restriction during Gestation in Rats: Neuronal Cell Body and Growth-Cone Fatty Acids. Am. J. Clin. Nutr. 2000, 71, 312S–314S. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M.; de La Presa Owens, S. Dietary Fatty Acid Composition in Pregnancy Alters Neurite Membrane Fatty Acids and Dopamine in Newborn Rat Brain. J. Nutr. 2001, 131, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Manabe, S.; Wada, O.; Crawford, M.A. Rapid Incorporation of Docosahexaenoic Acid from Dietary Sources into Brain Microsomal, Synaptosomal and Mitochondrial Membranes in Adult Mice. Int. J. Vitam. Nutr. Res. Int. Z. Vitam.- Ernahrungsforschung J. Int. Vitaminol. Nutr. 1997, 67, 272–278. [Google Scholar]
- Igarashi, M.; Santos, R.A.; Cohen-Cory, S. Impact of Maternal N-3 Polyunsaturated Fatty Acid Deficiency on Dendritic Arbor Morphology and Connectivity of Developing Xenopus Laevis Central Neurons in Vivo. J. Neurosci. Off. J. Soc. Neurosci. 2015, 35, 6079–6092. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.B.; Ménagé, C.; Grégoire, S.; Garcia, T.; Ferveur, J.-F.; Bretillon, L.; Grosjean, Y. Lack of Dietary Polyunsaturated Fatty Acids Causes Synapse Dysfunction in the Drosophila Visual System. PLoS ONE 2015, 10, e0135353. [Google Scholar] [CrossRef]
- Carbone, B.E.; Abouleish, M.; Watters, K.E.; Vogel, S.; Ribic, A.; Schroeder, O.H.-U.; Bader, B.M.; Biederer, T. Synaptic Connectivity and Cortical Maturation Are Promoted by the ω-3 Fatty Acid Docosahexaenoic Acid. Cereb. Cortex N. Y. NY 2020, 30, 226–240. [Google Scholar] [CrossRef] [PubMed]
- Fujita, S.; Ikegaya, Y.; Nishikawa, M.; Nishiyama, N.; Matsuki, N. Docosahexaenoic Acid Improves Long-Term Potentiation Attenuated by Phospholipase A(2) Inhibitor in Rat Hippocampal Slices. Br. J. Pharmacol. 2001, 132, 1417–1422. [Google Scholar] [CrossRef]
- Mazzocchi-Jones, D. Impaired Corticostriatal LTP and Depotentiation Following IPLA2 Inhibition Is Restored Following Acute Application of DHA. Brain Res. Bull. 2015, 111, 69–75. [Google Scholar] [CrossRef]
- Cao, H.; Li, M.-Y.; Li, G.; Li, S.-J.; Wen, B.; Lu, Y.; Yu, X. Retinoid X Receptor α Regulates DHA-Dependent Spinogenesis and Functional Synapse Formation In Vivo. Cell Rep. 2020, 31, 107649. [Google Scholar] [CrossRef]
- Kim, H.-Y.; Moon, H.-S.; Cao, D.; Lee, J.; Kevala, K.; Jun, S.B.; Lovinger, D.M.; Akbar, M.; Huang, B.X. N-Docosahexaenoylethanolamide Promotes Development of Hippocampal Neurons. Biochem. J. 2011, 435, 327–336. [Google Scholar] [CrossRef]
- Lee, J.-W.; Huang, B.X.; Kwon, H.; Rashid, M.A.; Kharebava, G.; Desai, A.; Patnaik, S.; Marugan, J.; Kim, H.-Y. Orphan GPR110 (ADGRF1) Targeted by N-Docosahexaenoylethanolamine in Development of Neurons and Cognitive Function. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Manni, M.M.; Tiberti, M.L.; Pagnotta, S.; Barelli, H.; Gautier, R.; Antonny, B. Acyl Chain Asymmetry and Polyunsaturation of Brain Phospholipids Facilitate Membrane Vesiculation without Leakage. eLife 2018, 7. [Google Scholar] [CrossRef]
- Hashimoto, M.; Hossain, S.; Shido, O. Docosahexaenoic Acid but Not Eicosapentaenoic Acid Withstands Dietary Cholesterol-Induced Decreases in Platelet Membrane Fluidity. Mol. Cell. Biochem. 2006, 293, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Darios, F.; Davletov, B. Omega-3 and Omega-6 Fatty Acids Stimulate Cell Membrane Expansion by Acting on Syntaxin 3. Nature 2006, 440, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Bruno, M.J.; Koeppe, R.E.; Andersen, O.S. Docosahexaenoic Acid Alters Bilayer Elastic Properties. Proc. Natl. Acad. Sci. USA 2007, 104, 9638–9643. [Google Scholar] [CrossRef]
- Tounian, P.; Bellaïche, M.; Legrand, P. ARA or No ARA in Infant Formulae, That Is the Question. Arch. Pediatr. Organe Off. Soc. Francaise Pediatr. 2021, 28, 69–74. [Google Scholar] [CrossRef]
- Koletzko, B.; Bergmann, K.; Brenna, J.T.; Calder, P.C.; Campoy, C.; Clandinin, M.T.; Colombo, J.; Daly, M.; Decsi, T.; Demmelmair, H.; et al. Should Formula for Infants Provide Arachidonic Acid along with DHA? A Position Paper of the European Academy of Paediatrics and the Child Health Foundation. Am. J. Clin. Nutr. 2020, 111, 10–16. [Google Scholar] [CrossRef]
- Brenna, J.T. Long-Chain Polyunsaturated Fatty Acids and the Preterm Infant: A Case Study in Developmentally Sensitive Nutrient Needs in the United States. Am. J. Clin. Nutr. 2016, 103, 606S–615S. [Google Scholar] [CrossRef]
- Crawford, M.A.; Wang, Y.; Forsyth, S.; Brenna, J.T. The European Food Safety Authority Recommendation for Polyunsaturated Fatty Acid Composition of Infant Formula Overrules Breast Milk, Puts Infants at Risk, and Should Be Revised. Prostaglandins Leukot. Essent. Fatty Acids 2015, 102–103, 1–3. [Google Scholar] [CrossRef]
- Hadley, K.B.; Ryan, A.S.; Forsyth, S.; Gautier, S.; Salem, N. The Essentiality of Arachidonic Acid in Infant Development. Nutrients 2016, 8, 216. [Google Scholar] [CrossRef]
- Harauma, A.; Yasuda, H.; Hatanaka, E.; Nakamura, M.T.; Salem, N.; Moriguchi, T. The Essentiality of Arachidonic Acid in Addition to Docosahexaenoic Acid for Brain Growth and Function. Prostaglandins Leukot. Essent. Fatty Acids 2017, 116, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Gordon, S. Macrophages and Microglia in the Nervous System. Trends Neurosci. 1988, 11, 273–277. [Google Scholar] [CrossRef]
- Salter, M.W.; Stevens, B. Microglia Emerge as Central Players in Brain Disease. Nat. Med. 2017, 23, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, A.D.; David, S.; Bennett, F.C. Immune Cell Regulation of Glia during CNS Injury and Disease. Nat. Rev. Neurosci. 2020, 21, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Madore, C.; Leyrolle, Q.; Lacabanne, C.; Benmamar-Badel, A.; Joffre, C.; Nadjar, A.; Layé, S. Neuroinflammation in Autism: Plausible Role of Maternal Inflammation, Dietary Omega 3, and Microbiota. Neural Plast. 2016, 2016, 3597209. [Google Scholar] [CrossRef]
- Thion, M.S.; Ginhoux, F.; Garel, S. Microglia and Early Brain Development: An Intimate Journey. Science 2018, 362, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Askew, K.; Gomez-Nicola, D. A Story of Birth and Death: Insights into the Formation and Dynamics of the Microglial Population. Brain. Behav. Immun. 2018, 69, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Hoeffel, G.; Ginhoux, F. Fetal Monocytes and the Origins of Tissue-Resident Macrophages. Cell. Immunol. 2018, 330, 5–15. [Google Scholar] [CrossRef]
- Mass, E.; Ballesteros, I.; Farlik, M.; Halbritter, F.; Günther, P.; Crozet, L.; Jacome-Galarza, C.E.; Händler, K.; Klughammer, J.; Kobayashi, Y.; et al. Specification of Tissue-Resident Macrophages during Organogenesis. Science 2016, 353. [Google Scholar] [CrossRef]
- Bennett, M.L.; Bennett, F.C.; Liddelow, S.A.; Ajami, B.; Zamanian, J.L.; Fernhoff, N.B.; Mulinyawe, S.B.; Bohlen, C.J.; Adil, A.; Tucker, A.; et al. New Tools for Studying Microglia in the Mouse and Human CNS. Proc. Natl. Acad. Sci. USA 2016, 113, E1738–E1746. [Google Scholar] [CrossRef]
- Sierra, A.; Encinas, J.M.; Deudero, J.J.P.; Chancey, J.H.; Enikolopov, G.; Overstreet-Wadiche, L.S.; Tsirka, S.E.; Maletic-Savatic, M. Microglia Shape Adult Hippocampal Neurogenesis through Apoptosis-Coupled Phagocytosis. Cell Stem Cell 2010, 7, 483–495. [Google Scholar] [CrossRef]
- Bialas, A.R.; Stevens, B. TGF-β Signaling Regulates Neuronal C1q Expression and Developmental Synaptic Refinement. Nat. Neurosci. 2013, 16, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Paolicelli, R.C.; Gross, C.T. Microglia in Development: Linking Brain Wiring to Brain Environment. Neuron Glia Biol. 2011, 7, 77–83. [Google Scholar] [CrossRef]
- Squarzoni, P.; Oller, G.; Hoeffel, G.; Pont-Lezica, L.; Rostaing, P.; Low, D.; Bessis, A.; Ginhoux, F.; Garel, S. Microglia Modulate Wiring of the Embryonic Forebrain. Cell Rep. 2014, 8, 1271–1279. [Google Scholar] [CrossRef] [PubMed]
- Estes, M.L.; McAllister, A.K. Immune Mediators in the Brain and Peripheral Tissues in Autism Spectrum Disorder. Nat. Rev. Neurosci. 2015, 16, 469–486. [Google Scholar] [CrossRef] [PubMed]
- Lenz, K.M.; Nelson, L.H. Microglia and Beyond: Innate Immune Cells As Regulators of Brain Development and Behavioral Function. Front. Immunol. 2018, 9, 698. [Google Scholar] [CrossRef]
- Rey, C.; Nadjar, A.; Buaud, B.; Vaysse, C.; Aubert, A.; Pallet, V.; Layé, S.; Joffre, C. Resolvin D1 and E1 Promote Resolution of Inflammation in Microglial Cells in Vitro. Brain. Behav. Immun. 2016, 55, 249–259. [Google Scholar] [CrossRef]
- Madore, C.; Nadjar, A.; Delpech, J.-C.; Sere, A.; Aubert, A.; Portal, C.; Joffre, C.; Layé, S. Nutritional N-3 PUFAs Deficiency during Perinatal Periods Alters Brain Innate Immune System and Neuronal Plasticity-Associated Genes. Brain. Behav. Immun. 2014, 41, 22–31. [Google Scholar] [CrossRef]
- Labrousse, V.F.; Nadjar, A.; Joffre, C.; Costes, L.; Aubert, A.; Grégoire, S.; Bretillon, L.; Layé, S. Short-Term Long Chain Omega3 Diet Protects from Neuroinflammatory Processes and Memory Impairment in Aged Mice. PLoS ONE 2012, 7, e36861. [Google Scholar] [CrossRef] [PubMed]
- Labrousse, V.F.; Leyrolle, Q.; Amadieu, C.; Aubert, A.; Sere, A.; Coutureau, E.; Grégoire, S.; Bretillon, L.; Pallet, V.; Gressens, P.; et al. Dietary Omega-3 Deficiency Exacerbates Inflammation and Reveals Spatial Memory Deficits in Mice Exposed to Lipopolysaccharide during Gestation. Brain. Behav. Immun. 2018, 73, 427–440. [Google Scholar] [CrossRef]
- Delpech, J.-C.; Thomazeau, A.; Madore, C.; Bosch-Bouju, C.; Larrieu, T.; Lacabanne, C.; Remus-Borel, J.; Aubert, A.; Joffre, C.; Nadjar, A.; et al. Dietary N-3 PUFAs Deficiency Increases Vulnerability to Inflammation-Induced Spatial Memory Impairment. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2015, 40, 2774–2787. [Google Scholar] [CrossRef]
- Fourrier, C.; Remus-Borel, J.; Greenhalgh, A.D.; Guichardant, M.; Bernoud-Hubac, N.; Lagarde, M.; Joffre, C.; Layé, S. Docosahexaenoic Acid-Containing Choline Phospholipid Modulates LPS-Induced Neuroinflammation in Vivo and in Microglia in Vitro. J. Neuroinflammation 2017, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Leyrolle, Q.; Decoeur, F.; Briere, G.; Amadieu, C.; Quadros, A.R.A.A.; Voytyuk, I.; Lacabane, C.; Benmamar-Badel, A.; Bourel, J.; Aubert, A.; et al. Maternal Dietary Omega-3 Deficiency Worsens the Deleterious Effects of Prenatal Inflammation on the Gut-Brain Axis in the Offspring across Lifetime. Neuropsychopharmacology 2020, 46, 579–602. [Google Scholar] [CrossRef] [PubMed]
- Meyer, U. Neurodevelopmental Resilience and Susceptibility to Maternal Immune Activation. Trends Neurosci. 2019, 42, 793–806. [Google Scholar] [CrossRef]
- Chang, P.K.-Y.; Khatchadourian, A.; McKinney, R.A.; Maysinger, D. Docosahexaenoic Acid (DHA): A Modulator of Microglia Activity and Dendritic Spine Morphology. J. Neuroinflammation 2015, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Sharon, G.; Sampson, T.R.; Geschwind, D.H.; Mazmanian, S.K. The Central Nervous System and the Gut Microbiome. Cell 2016, 167, 915–932. [Google Scholar] [CrossRef]
- Codagnone, M.G.; Spichak, S.; O’Mahony, S.M.; O’Leary, O.F.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Programming Bugs: Microbiota and the Developmental Origins of Brain Health and Disease. Biol. Psychiatry 2019, 85, 150–163. [Google Scholar] [CrossRef]
- Li, Q.; Leung, Y.O.; Zhou, I.; Ho, L.C.; Kong, W.; Basil, P.; Wei, R.; Lam, S.; Zhang, X.; Law, A.C.K.; et al. Dietary Supplementation with N-3 Fatty Acids from Weaning Limits Brain Biochemistry and Behavioural Changes Elicited by Prenatal Exposure to Maternal Inflammation in the Mouse Model. Transl. Psychiatry 2015, 5, e641. [Google Scholar] [CrossRef] [PubMed]
- Parenti, I.; Rabaneda, L.G.; Schoen, H.; Novarino, G. Neurodevelopmental Disorders: From Genetics to Functional Pathways. Trends Neurosci. 2020. [Google Scholar] [CrossRef]
- Barker, D.J.P. The Origins of the Developmental Origins Theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Susser, E.; Neugebauer, R.; Hoek, H.W.; Brown, A.S.; Lin, S.; Labovitz, D.; Gorman, J.M. Schizophrenia after Prenatal Famine. Further Evidence. Arch. Gen. Psychiatry 1996, 53, 25–31. [Google Scholar] [CrossRef]
- Hsu, M.-C.; Huang, Y.-S.; Ouyang, W.-C. Beneficial Effects of Omega-3 Fatty Acid Supplementation in Schizophrenia: Possible Mechanisms. Lipids Health Dis. 2020, 19. [Google Scholar] [CrossRef] [PubMed]
- Altshuler, G. Some Placental Considerations Related to Neurodevelopmental and Other Disorders. J. Child Neurol. 1993, 8, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Hodyl, N.A.; Aboustate, N.; Bianco-Miotto, T.; Roberts, C.T.; Clifton, V.L.; Stark, M.J. Child Neurodevelopmental Outcomes Following Preterm and Term Birth: What Can the Placenta Tell Us? Placenta 2017, 57, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Shaddy, D.J.; Kerling, E.H.; Gustafson, K.M.; Carlson, S.E. Docosahexaenoic Acid (DHA) and Arachidonic Acid (ARA) Balance in Developmental Outcomes. Prostaglandins Leukot. Essent. Fatty Acids 2017, 121, 52–56. [Google Scholar] [CrossRef]
- Hsieh, A.T.; Anthony, J.C.; Diersen-Schade, D.A.; Rumsey, S.C.; Lawrence, P.; Li, C.; Nathanielsz, P.W.; Brenna, J.T. The Influence of Moderate and High Dietary Long Chain Polyunsaturated Fatty Acids (LCPUFA) on Baboon Neonate Tissue Fatty Acids. Pediatr. Res. 2007, 61, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Birch, E.E.; Carlson, S.E.; Hoffman, D.R.; Fitzgerald-Gustafson, K.M.; Fu, V.L.N.; Drover, J.R.; Castañeda, Y.S.; Minns, L.; Wheaton, D.K.H.; Mundy, D.; et al. The DIAMOND (DHA Intake And Measurement Of Neural Development) Study: A Double-Masked, Randomized Controlled Clinical Trial of the Maturation of Infant Visual Acuity as a Function of the Dietary Level of Docosahexaenoic Acid. Am. J. Clin. Nutr. 2010, 91, 848–859. [Google Scholar] [CrossRef]
- Montgomery, P.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Richardson, A.J. Low Blood Long Chain Omega-3 Fatty Acids in UK Children Are Associated with Poor Cognitive Performance and Behavior: A Cross-Sectional Analysis from the DOLAB Study. PLoS ONE 2013, 8, e66697. [Google Scholar] [CrossRef]
- Kalmijn, S.; van Boxtel, M.P.J.; Ocké, M.; Verschuren, W.M.M.; Kromhout, D.; Launer, L.J. Dietary Intake of Fatty Acids and Fish in Relation to Cognitive Performance at Middle Age. Neurology 2004, 62, 275–280. [Google Scholar] [CrossRef]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 Fatty Acid Addition during Pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef]
- González, F.E.; Báez, R.V. IN TIME: IMPORTANCE OF OMEGA 3 IN CHILDREN’S NUTRITION. Rev. Paul. Pediatr. 2017, 35, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.T.; Janowsky, J.S.; Carroll, R.E.; Taylor, J.A.; Auestad, N.; Montalto, M.B. Formula Supplementation with Long-Chain Polyunsaturated Fatty Acids: Are There Developmental Benefits? Pediatrics 1998, 102, E59. [Google Scholar] [CrossRef] [PubMed]
- Osendarp, S.J.M.; Baghurst, K.I.; Bryan, J.; Calvaresi, E.; Hughes, D.; Hussaini, M.; Karyadi, S.J.M.; van Klinken, B.J.-W.; van der Knaap, H.C.M.; Lukito, W.; et al. Effect of a 12-Mo Micronutrient Intervention on Learning and Memory in Well-Nourished and Marginally Nourished School-Aged Children: 2 Parallel, Randomized, Placebo-Controlled Studies in Australia and Indonesia. Am. J. Clin. Nutr. 2007, 86, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- van der Merwe, L.F.; Moore, S.E.; Fulford, A.J.; Halliday, K.E.; Drammeh, S.; Young, S.; Prentice, A.M. Long-Chain PUFA Supplementation in Rural African Infants: A Randomized Controlled Trial of Effects on Gut Integrity, Growth, and Cognitive Development. Am. J. Clin. Nutr. 2013, 97, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Fransson, G.; Östlund, S.; Areskoug, B.; Gillberg, C. Omega 3/6 Fatty Acids for Reading in Children: A Randomized, Double-Blind, Placebo-Controlled Trial in 9-Year-Old Mainstream Schoolchildren in Sweden. J. Child Psychol. Psychiatry 2017, 58, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Bloch, M.H.; Qawasmi, A. Omega-3 Fatty Acid Supplementation for the Treatment of Children with Attention-Deficit/Hyperactivity Disorder Symptomatology: Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 991–1000. [Google Scholar] [CrossRef]
- Chang, J.P.-C.; Su, K.-P.; Mondelli, V.; Satyanarayanan, S.K.; Yang, H.-T.; Chiang, Y.-J.; Chen, H.-T.; Pariante, C.M. High-Dose Eicosapentaenoic Acid (EPA) Improves Attention and Vigilance in Children and Adolescents with Attention Deficit Hyperactivity Disorder (ADHD) and Low Endogenous EPA Levels. Transl. Psychiatry 2019, 9, 303. [Google Scholar] [CrossRef]
- Helland, I.B.; Smith, L.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Maternal Supplementation with Very-Long-Chain n-3 Fatty Acids during Pregnancy and Lactation Augments Children’s IQ at 4 Years of Age. Pediatrics 2003, 111, e39–e44. [Google Scholar] [CrossRef]
- Hoffman, D.R.; Birch, E.E.; Castañeda, Y.S.; Fawcett, S.L.; Wheaton, D.H.; Birch, D.G.; Uauy, R. Visual Function in Breast-Fed Term Infants Weaned to Formula with or without Long-Chain Polyunsaturates at 4 to 6 Months: A Randomized Clinical Trial. J. Pediatr. 2003, 142, 669–677. [Google Scholar] [CrossRef]
- Birch, E.E.; Hoffman, D.R.; Castañeda, Y.S.; Fawcett, S.L.; Birch, D.G.; Uauy, R.D. A Randomized Controlled Trial of Long-Chain Polyunsaturated Fatty Acid Supplementation of Formula in Term Infants after Weaning at 6 Wk of Age. Am. J. Clin. Nutr. 2002, 75, 570–580. [Google Scholar] [CrossRef]
- Voigt, R.G.; Jensen, C.L.; Fraley, J.K.; Rozelle, J.C.; Brown, F.R.; Heird, W.C. Relationship between Omega3 Long-Chain Polyunsaturated Fatty Acid Status during Early Infancy and Neurodevelopmental Status at 1 Year of Age. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2002, 15, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Fleith, M.; Clandinin, M.T. Dietary PUFA for Preterm and Term Infants: Review of Clinical Studies. Crit. Rev. Food Sci. Nutr. 2005, 45, 205–229. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Is Docosahexaenoic Acid, an n-3 Long-Chain Polyunsaturated Fatty Acid, Required for Development of Normal Brain Function? An Overview of Evidence from Cognitive and Behavioral Tests in Humans and Animals. Am. J. Clin. Nutr. 2005, 82, 281–295. [Google Scholar] [CrossRef]
- Bongiovanni, K.D.; Depeters, E.J.; Van Eenennaam, A.L. Neonatal Growth Rate and Development of Mice Raised on Milk Transgenically Enriched with Omega-3 Fatty Acids. Pediatr. Res. 2007, 62, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, I.; Salem, N. Omega-3 Fatty Acids and Rodent Behavior. Prostaglandins Leukot. Essent. Fatty Acids 2006, 75, 271–289. [Google Scholar] [CrossRef]
- Lafourcade, M.; Larrieu, T.; Mato, S.; Duffaud, A.; Sepers, M.; Matias, I.; De Smedt-Peyrusse, V.; Labrousse, V.F.; Bretillon, L.; Matute, C.; et al. Nutritional Omega-3 Deficiency Abolishes Endocannabinoid-Mediated Neuronal Functions. Nat. Neurosci. 2011, 14, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Moranis, A.; Delpech, J.-C.; De Smedt-Peyrusse, V.; Aubert, A.; Guesnet, P.; Lavialle, M.; Joffre, C.; Layé, S. Long Term Adequate N-3 Polyunsaturated Fatty Acid Diet Protects from Depressive-like Behavior but Not from Working Memory Disruption and Brain Cytokine Expression in Aged Mice. Brain. Behav. Immun. 2012, 26, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Levy, S.E.; Mandell, D.S.; Schultz, R.T. Autism. Lancet Lond. Engl. 2009, 374, 1627–1638. [Google Scholar] [CrossRef]
- Masi, A.; DeMayo, M.M.; Glozier, N.; Guastella, A.J. An Overview of Autism Spectrum Disorder, Heterogeneity and Treatment Options. Neurosci. Bull. 2017, 33, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Happé, F.; Ronald, A.; Plomin, R. Time to Give up on a Single Explanation for Autism. Nat. Neurosci. 2006, 9, 1218–1220. [Google Scholar] [CrossRef]
- Abrahams, B.S.; Geschwind, D.H. Advances in Autism Genetics: On the Threshold of a New Neurobiology. Nat. Rev. Genet. 2008, 9, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Kinney, D.K.; Munir, K.M.; Crowley, D.J.; Miller, A.M. Prenatal Stress and Risk for Autism. Neurosci. Biobehav. Rev. 2008, 32, 1519–1532. [Google Scholar] [CrossRef] [PubMed]
- Samsam, M.; Ahangari, R.; Naser, S.A. Pathophysiology of Autism Spectrum Disorders: Revisiting Gastrointestinal Involvement and Immune Imbalance. World J. Gastroenterol. 2014, 20, 9942–9951. [Google Scholar] [CrossRef]
- Minshew, N.J.; Williams, D.L. The New Neurobiology of Autism: Cortex, Connectivity, and Neuronal Organization. Arch. Neurol. 2007, 64, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Minshew, N.J.; Keller, T.A. The Nature of Brain Dysfunction in Autism: Functional Brain Imaging Studies. Curr. Opin. Neurol. 2010, 23, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, E.; Ariza, J.; Rogers, H.; Noctor, S.C.; Martínez-Cerdeño, V. The Number of Parvalbumin-Expressing Interneurons Is Decreased in the Prefrontal Cortex in Autism. Cereb. Cortex N. Y. NY 2017, 27, 1931–1943. [Google Scholar] [CrossRef]
- Takarae, Y.; Sweeney, J. Neural Hyperexcitability in Autism Spectrum Disorders. Brain Sci. 2017, 7, 129. [Google Scholar] [CrossRef]
- Sun, C.; Zou, M.; Wang, X.; Xia, W.; Ma, Y.; Liang, S.; Hao, Y.; Wu, L.; Fu, S. FADS1-FADS2 and ELOVL2 Gene Polymorphisms in Susceptibility to Autism Spectrum Disorders in Chinese Children. BMC Psychiatry 2018, 18, 283. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.L.; Will, M.J.; Hecht, P.M.; Parker, C.L.; Beversdorf, D.Q. Maternal Diet Rich in Omega-6 Polyunsaturated Fatty Acids during Gestation and Lactation Produces Autistic-like Sociability Deficits in Adult Offspring. Behav. Brain Res. 2013, 238, 193–199. [Google Scholar] [CrossRef]
- El-Ansary, A.; Al-Ayadhi, L. Relative Abundance of Short Chain and Polyunsaturated Fatty Acids in Propionic Acid-Induced Autistic Features in Rat Pups as Potential Markers in Autism. Lipids Health Dis. 2014, 13, 140. [Google Scholar] [CrossRef] [PubMed]
- Alfawaz, H.; Al-Onazi, M.; Bukhari, S.I.; Binobead, M.; Othman, N.; Algahtani, N.; Bhat, R.S.; Moubayed, N.M.S.; Alzeer, H.S.; El-Ansary, A. The Independent and Combined Effects of Omega-3 and Vitamin B12 in Ameliorating Propionic Acid Induced Biochemical Features in Juvenile Rats as Rodent Model of Autism. J. Mol. Neurosci. MN 2018, 66, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Basil, P.; Li, Q.; Gui, H.; Hui, T.C.K.; Ling, V.H.M.; Wong, C.C.Y.; Mill, J.; McAlonan, G.M.; Sham, P.-C. Prenatal Immune Activation Alters the Adult Neural Epigenome but Can Be Partly Stabilised by a N-3 Polyunsaturated Fatty Acid Diet. Transl. Psychiatry 2018, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Tiwari, V.; Singh, M.; Yadav, R.K.; Roy, S.; Devi, U.; Gautam, S.; Rawat, J.K.; Ansari, M.N.; Saeedan, A.S.; et al. Comparative Efficacy of Alpha-Linolenic Acid and Gamma-Linolenic Acid to Attenuate Valproic Acid-Induced Autism-like Features. J. Physiol. Biochem. 2017, 73, 187–198. [Google Scholar] [CrossRef]
- van Elst, K.; Brouwers, J.F.; Merkens, J.E.; Broekhoven, M.H.; Birtoli, B.; Helms, J.B.; Kas, M.J.H. Chronic Dietary Changes in N-6/n-3 Polyunsaturated Fatty Acid Ratios Cause Developmental Delay and Reduce Social Interest in Mice. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2019, 29, 16–31. [Google Scholar] [CrossRef]
- Pietropaolo, S.; Goubran, M.G.; Joffre, C.; Aubert, A.; Lemaire-Mayo, V.; Crusio, W.E.; Layé, S. Dietary Supplementation of Omega-3 Fatty Acids Rescues Fragile X Phenotypes in Fmr1-Ko Mice. Psychoneuroendocrinology 2014, 49, 119–129. [Google Scholar] [CrossRef]
- Bell, J.G.; Sargent, J.R.; Tocher, D.R.; Dick, J.R. Red Blood Cell Fatty Acid Compositions in a Patient with Autistic Spectrum Disorder: A Characteristic Abnormality in Neurodevelopmental Disorders? Prostaglandins Leukot. Essent. Fatty Acids 2000, 63, 21–25. [Google Scholar] [CrossRef]
- Vancassel, S.; Durand, G.; Barthélémy, C.; Lejeune, B.; Martineau, J.; Guilloteau, D.; Andrès, C.; Chalon, S. Plasma Fatty Acid Levels in Autistic Children. Prostaglandins Leukot. Essent. Fatty Acids 2001, 65, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Meguid, N.A.; Atta, H.M.; Gouda, A.S.; Khalil, R.O. Role of Polyunsaturated Fatty Acids in the Management of Egyptian Children with Autism. Clin. Biochem. 2008, 41, 1044–1048. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, G.A.; Al-Ayadhi, L.Y. Reduced Levels of Plasma Polyunsaturated Fatty Acids and Serum Carnitine in Autistic Children: Relation to Gastrointestinal Manifestations. Behav. Brain Funct. BBF 2015, 11, 4. [Google Scholar] [CrossRef]
- Jory, J. Abnormal Fatty Acids in Canadian Children with Autism. Nutr. Burbank Los Angel. Cty. Calif 2016, 32, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Yui, K.; Kawasaki, Y.; Yamada, H. Modulation of Omega-6 Polyunsaturated Fatty Acid-Dependent Signaling and Its Therapeutic Potential for the Core Symptoms in Individuals with Autism Spectrum Disorders. Jpn. J. Biol. Psychiatry 2016, 27, 41–47. [Google Scholar] [CrossRef]
- Mazahery, H.; Stonehouse, W.; Delshad, M.; Kruger, M.C.; Conlon, C.A.; Beck, K.L.; von Hurst, P.R. Relationship between Long Chain N-3 Polyunsaturated Fatty Acids and Autism Spectrum Disorder: Systematic Review and Meta-Analysis of Case-Control and Randomised Controlled Trials. Nutrients 2017, 9, 155. [Google Scholar] [CrossRef]
- Parletta, N.; Niyonsenga, T.; Duff, J. Omega-3 and Omega-6 Polyunsaturated Fatty Acid Levels and Correlations with Symptoms in Children with Attention Deficit Hyperactivity Disorder, Autistic Spectrum Disorder and Typically Developing Controls. PLoS ONE 2016, 11, e0156432. [Google Scholar] [CrossRef] [PubMed]
- Sliwinski, S.; Croonenberghs, J.; Christophe, A.; Deboutte, D.; Maes, M. Polyunsaturated Fatty Acids: Do They Have a Role in the Pathophysiology of Autism? Neuro Endocrinol. Lett. 2006, 27, 465–471. [Google Scholar] [PubMed]
- Lyall, K.; Munger, K.L.; O’Reilly, É.J.; Santangelo, S.L.; Ascherio, A. Maternal Dietary Fat Intake in Association with Autism Spectrum Disorders. Am. J. Epidemiol. 2013, 178, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Iosif, A.-M.; Hansen, R.L.; Schmidt, R.J. Maternal Polyunsaturated Fatty Acids and Risk for Autism Spectrum Disorder in the MARBLES High-Risk Study. Autism Int. J. Res. Pract. 2020. [Google Scholar] [CrossRef]
- Politi, P.; Cena, H.; Comelli, M.; Marrone, G.; Allegri, C.; Emanuele, E.; Ucelli di Nemi, S. Behavioral Effects of Omega-3 Fatty Acid Supplementation in Young Adults with Severe Autism: An Open Label Study. Arch. Med. Res. 2008, 39, 682–685. [Google Scholar] [CrossRef]
- De Crescenzo, F.; D’Alò, G.L.; Morgano, G.P.; Minozzi, S.; Mitrova, Z.; Saulle, R.; Cruciani, F.; Fulceri, F.; Davoli, M.; Scattoni, M.L.; et al. Impact of Polyunsaturated Fatty Acids on Patient-Important Outcomes in Children and Adolescents with Autism Spectrum Disorder: A Systematic Review. Health Qual. Life Outcomes 2020, 18, 28. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.; Sears, B.; Rizzo, A.M.; Mariani Cerati, D.; Caprio, M.; Ricordi, C.; Fabbri, A. Omega-3 PUFAs and Vitamin D Co-Supplementation as a Safe-Effective Therapeutic Approach for Core Symptoms of Autism Spectrum Disorder: Case Report and Literature Review. Nutr. Neurosci. 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, C.A.; Meyer, B.J.; Jones, B.; von Hurst, P.R. A Randomised Controlled Trial of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids in the Treatment of Irritability and Hyperactivity among Children with Autism Spectrum Disorder. J. Steroid Biochem. Mol. Biol. 2019, 187, 9–16. [Google Scholar] [CrossRef]
- Cheng, Y.-S.; Tseng, P.-T.; Chen, Y.-W.; Stubbs, B.; Yang, W.-C.; Chen, T.-Y.; Wu, C.-K.; Lin, P.-Y. Supplementation of Omega 3 Fatty Acids May Improve Hyperactivity, Lethargy, and Stereotypy in Children with Autism Spectrum Disorders: A Meta-Analysis of Randomized Controlled Trials. Neuropsychiatr. Dis. Treat. 2017, 13, 2531–2543. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, K.W.; Boone, K.M.; Gracious, B.; Klebanoff, M.A.; Rogers, L.K.; Rausch, J.; Bartlett, C.; Coury, D.L.; Keim, S.A. Effect of Omega-3 and -6 Supplementation on Language in Preterm Toddlers Exhibiting Autism Spectrum Disorder Symptoms. J. Autism Dev. Disord. 2017, 47, 3358–3369. [Google Scholar] [CrossRef]
- Keim, S.A.; Gracious, B.; Boone, K.M.; Klebanoff, M.A.; Rogers, L.K.; Rausch, J.; Coury, D.L.; Sheppard, K.W.; Husk, J.; Rhoda, D.A. ω-3 and ω-6 Fatty Acid Supplementation May Reduce Autism Symptoms Based on Parent Report in Preterm Toddlers. J. Nutr. 2018, 148, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M.; Hollander, E. Evidence That Eicosapentaenoic Acid Is Effective in Treating Autism. J. Clin. Psychiatry 2003, 64, 848–849. [Google Scholar] [CrossRef] [PubMed]
- Meiri, G.; Bichovsky, Y.; Belmaker, R.H. Omega 3 Fatty Acid Treatment in Autism. J. Child Adolesc. Psychopharmacol. 2009, 19, 449–451. [Google Scholar] [CrossRef] [PubMed]
- Ooi, Y.P.; Weng, S.-J.; Jang, L.Y.; Low, L.; Seah, J.; Teo, S.; Ang, R.P.; Lim, C.G.; Liew, A.; Fung, D.S.; et al. Omega-3 Fatty Acids in the Management of Autism Spectrum Disorders: Findings from an Open-Label Pilot Study in Singapore. Eur. J. Clin. Nutr. 2015, 69, 969–971. [Google Scholar] [CrossRef]
- Johnson, C.R.; Handen, B.L.; Zimmer, M.; Sacco, K. Polyunsaturated Fatty Acid Supplementation in Young Children with Autism. J. Dev. Phys. Disabil. 2010, 22, 1–10. [Google Scholar] [CrossRef]
- Amminger, G.P.; Berger, G.E.; Schäfer, M.R.; Klier, C.; Friedrich, M.H.; Feucht, M. Omega-3 Fatty Acids Supplementation in Children with Autism: A Double-Blind Randomized, Placebo-Controlled Pilot Study. Biol. Psychiatry 2007, 61, 551–553. [Google Scholar] [CrossRef]
- Bent, S.; Bertoglio, K.; Ashwood, P.; Bostrom, A.; Hendren, R.L. A Pilot Randomized Controlled Trial of Omega-3 Fatty Acids for Autism Spectrum Disorder. J. Autism Dev. Disord. 2011, 41, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Yui, K.; Koshiba, M.; Nakamura, S.; Kobayashim, Y.; Ohnishi, M. Efficacy of Adding Large Doses of Arachidonic Acid to Docosahexaenoic Acid against Restricted Repetitive Behaviors in Individuals with AutismSpectrum Disorders: A Placebo-Controlled Trial. J. Addict. Res. Ther. 2011, 2–6. [Google Scholar] [CrossRef]
- Yui, K.; Koshiba, M.; Nakamura, S.; Kobayashi, Y. Effects of Large Doses of Arachidonic Acid Added to Docosahexaenoic Acid on Social Impairment in Individuals with Autism Spectrum Disorders: A Double-Blind, Placebo-Controlled, Randomized Trial. J. Clin. Psychopharmacol. 2012, 32, 200–206. [Google Scholar] [CrossRef]
- Bent, S.; Hendren, R.L.; Zandi, T.; Law, K.; Choi, J.-E.; Widjaja, F.; Kalb, L.; Nestle, J.; Law, P. Internet-Based, Randomized, Controlled Trial of Omega-3 Fatty Acids for Hyperactivity in Autism. J. Am. Acad. Child Adolesc. Psychiatry 2014, 53, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.G.; Mellon, M.W.; Katusic, S.K.; Weaver, A.L.; Matern, D.; Mellon, B.; Jensen, C.L.; Barbaresi, W.J. Dietary Docosahexaenoic Acid Supplementation in Children with Autism. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Mankad, D.; Dupuis, A.; Smile, S.; Roberts, W.; Brian, J.; Lui, T.; Genore, L.; Zaghloul, D.; Iaboni, A.; Marcon, P.M.A.; et al. A Randomized, Placebo Controlled Trial of Omega-3 Fatty Acids in the Treatment of Young Children with Autism. Mol. Autism 2015, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Parellada, M.; Llorente, C.; Calvo, R.; Gutierrez, S.; Lázaro, L.; Graell, M.; Guisasola, M.; Dorado, M.L.; Boada, L.; Romo, J.; et al. Randomized Trial of Omega-3 for Autism Spectrum Disorders: Effect on Cell Membrane Composition and Behavior. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2017, 27, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Boone, K.M.; Gracious, B.; Klebanoff, M.A.; Rogers, L.K.; Rausch, J.; Coury, D.L.; Keim, S.A. Omega-3 and -6 Fatty Acid Supplementation and Sensory Processing in Toddlers with ASD Symptomology Born Preterm: A Randomized Controlled Trial. Early Hum. Dev. 2017, 115, 64–70. [Google Scholar] [CrossRef]
- Karhu, E.; Zukerman, R.; Eshraghi, R.S.; Mittal, J.; Deth, R.C.; Castejon, A.M.; Trivedi, M.; Mittal, R.; Eshraghi, A.A. Nutritional Interventions for Autism Spectrum Disorder. Nutr. Rev. 2019. [Google Scholar] [CrossRef]
- Froehlich, T.E.; Lanphear, B.P.; Epstein, J.N.; Barbaresi, W.J.; Katusic, S.K.; Kahn, R.S. Prevalence, Recognition, and Treatment of Attention-Deficit/Hyperactivity Disorder in a National Sample of US Children. Arch. Pediatr. Adolesc. Med. 2007, 161, 857–864. [Google Scholar] [CrossRef]
- Gentile, J.P.; Atiq, R.; Gillig, P.M. Adult ADHD: Diagnosis, Differential Diagnosis, and Medication Management. Psychiatry Edgmont Pa Townsh. 2006, 3, 25–30. [Google Scholar]
- Epstein, J.N.; Loren, R.E.A. Changes in the Definition of ADHD in DSM-5: Subtle but Important. Neuropsychiatry 2013, 3, 455–458. [Google Scholar] [CrossRef]
- Wilens, T.E.; Biederman, J.; Faraone, S.V.; Martelon, M.; Westerberg, D.; Spencer, T.J. Presenting ADHD Symptoms, Subtypes, and Comorbid Disorders in Clinically Referred Adults with ADHD. J. Clin. Psychiatry 2009, 70, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Steinau, S. Diagnostic Criteria in Attention Deficit Hyperactivity Disorder—Changes in DSM 5. Front. Psychiatry 2013, 4. [Google Scholar] [CrossRef]
- Millichap, J.G. Etiologic Classification of Attention-Deficit/Hyperactivity Disorder. Pediatrics 2008, 121, e358–e365. [Google Scholar] [CrossRef]
- Thapar, A.; Cooper, M.; Jefferies, R.; Stergiakouli, E. What Causes Attention Deficit Hyperactivity Disorder? Arch. Dis. Child. 2012, 97, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Crosbie, J.; Wigg, K.; Ickowicz, A.; Pathare, T.; Roberts, W.; Malone, M.; Schachar, R.; Tannock, R.; Kennedy, J.L.; et al. Glutamate Receptor, Ionotropic, N-Methyl D-Aspartate 2A (GRIN2A) Gene as a Positional Candidate for Attention-Deficit/Hyperactivity Disorder in the 16p13 Region. Mol. Psychiatry 2004, 9, 494–499. [Google Scholar] [CrossRef]
- Gizer, I.R.; Ficks, C.; Waldman, I.D. Candidate Gene Studies of ADHD: A Meta-Analytic Review. Hum. Genet. 2009, 126, 51–90. [Google Scholar] [CrossRef]
- Banaschewski, T.; Becker, K.; Scherag, S.; Franke, B.; Coghill, D. Molecular Genetics of Attention-Deficit/Hyperactivity Disorder: An Overview. Eur. Child Adolesc. Psychiatry 2010, 19, 237–257. [Google Scholar] [CrossRef]
- Castellanos, F.X.; Lee, P.P.; Sharp, W.; Jeffries, N.O.; Greenstein, D.K.; Clasen, L.S.; Blumenthal, J.D.; James, R.S.; Ebens, C.L.; Walter, J.M.; et al. Developmental Trajectories of Brain Volume Abnormalities in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. JAMA 2002, 288, 1740–1748. [Google Scholar] [CrossRef] [PubMed]
- Kessler, D.; Angstadt, M.; Welsh, R.C.; Sripada, C. Modality-Spanning Deficits in Attention-Deficit/Hyperactivity Disorder in Functional Networks, Gray Matter, and White Matter. J. Neurosci. Off. J. Soc. Neurosci. 2014, 34, 16555–16566. [Google Scholar] [CrossRef]
- Li, X.; Cao, Q.; Pu, F.; Li, D.; Fan, Y.; An, L.; Wang, P.; Wu, Z.; Sun, L.; Li, S.; et al. Abnormalities of Structural Covariance Networks in Drug-Naïve Boys with Attention Deficit Hyperactivity Disorder. Psychiatry Res. 2015, 231, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-R.; Hsu, S.-F.; Hsu, C.-D.; Hwang, L.-H.; Yang, S.-C. Dietary Patterns and Blood Fatty Acid Composition in Children with Attention-Deficit Hyperactivity Disorder in Taiwan. J. Nutr. Biochem. 2004, 15, 467–472. [Google Scholar] [CrossRef]
- Young, G.S.; Maharaj, N.J.; Conquer, J.A. Blood Phospholipid Fatty Acid Analysis of Adults with and without Attention Deficit/Hyperactivity Disorder. Lipids 2004, 39, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Colter, A.L.; Cutler, C.; Meckling, K.A. Fatty Acid Status and Behavioural Symptoms of Attention Deficit Hyperactivity Disorder in Adolescents: A Case-Control Study. Nutr. J. 2008, 7, 8. [Google Scholar] [CrossRef]
- Irmisch, G.; Richter, J.; Thome, J.; Sheldrick, A.J.; Wandschneider, R. Altered Serum Mono- and Polyunsaturated Fatty Acid Levels in Adults with ADHD. Atten. Deficit Hyperact. Disord. 2013, 5, 303–311. [Google Scholar] [CrossRef]
- Hawkey, E.; Nigg, J.T. Omega-3 Fatty Acid and ADHD: Blood Level Analysis and Meta-Analytic Extension of Supplementation Trials. Clin. Psychol. Rev. 2014, 34, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Sinn, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R. Polyunsaturated Fatty Acids, Cognition and Literacy in Children with ADHD with and without Learning Difficulties. J. Child Health Care Prof. Work. Child. Hosp. Community 2011, 15, 299–311. [Google Scholar] [CrossRef]
- Fuentes-Albero, M.; Martínez-Martínez, M.I.; Cauli, O. Omega-3 Long-Chain Polyunsaturated Fatty Acids Intake in Children with Attention Deficit and Hyperactivity Disorder. Brain Sci. 2019, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Bruno, K.J.; Hess, E.J. Rodent Models of ADHD. Curr. Top. Behav. Neurosci. 2012, 9, 273–300. [Google Scholar] [CrossRef]
- Lou, H.C.; Rosa, P.; Pryds, O.; Karrebaek, H.; Lunding, J.; Cumming, P.; Gjedde, A. ADHD: Increased Dopamine Receptor Availability Linked to Attention Deficit and Low Neonatal Cerebral Blood Flow. Dev. Med. Child Neurol. 2004, 46, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Rosa-Neto, P.; Lou, H.C.; Cumming, P.; Pryds, O.; Karrebaek, H.; Lunding, J.; Gjedde, A. Methylphenidate-Evoked Changes in Striatal Dopamine Correlate with Inattention and Impulsivity in Adolescents with Attention Deficit Hyperactivity Disorder. NeuroImage 2005, 25, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Newcorn, J.; Fowler, J.S.; Telang, F.; Solanto, M.V.; Logan, J.; Wong, C.; Ma, Y.; Swanson, J.M.; et al. Brain Dopamine Transporter Levels in Treatment and Drug Naïve Adults with ADHD. NeuroImage 2007, 34, 1182–1190. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Newcorn, J.; Telang, F.; Solanto, M.V.; Fowler, J.S.; Logan, J.; Ma, Y.; Schulz, K.; Pradhan, K.; et al. Depressed Dopamine Activity in Caudate and Preliminary Evidence of Limbic Involvement in Adults with Attention-Deficit/Hyperactivity Disorder. Arch. Gen. Psychiatry 2007, 64, 932–940. [Google Scholar] [CrossRef]
- Wang, G.-X.; Ma, Y.-H.; Wang, S.-F.; Ren, G.-F.; Guo, H. Association of Dopaminergic/GABAergic Genes with Attention Deficit Hyperactivity Disorder in Children. Mol. Med. Rep. 2012, 6, 1093–1098. [Google Scholar] [CrossRef][Green Version]
- Bello, E.P.; Mateo, Y.; Gelman, D.M.; Noaín, D.; Shin, J.H.; Low, M.J.; Alvarez, V.A.; Lovinger, D.M.; Rubinstein, M. Cocaine Supersensitivity and Enhanced Motivation for Reward in Mice Lacking Dopamine D2 Autoreceptors. Nat. Neurosci. 2011, 14, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Linden, J.; James, A.S.; McDaniel, C.; Jentsch, J.D. Dopamine D2 Receptors in Dopaminergic Neurons Modulate Performance in a Reversal Learning Task in Mice. eNeuro 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Swanson, J.M. Clinical Practice: Adult Attention Deficit-Hyperactivity Disorder. N. Engl. J. Med. 2013, 369, 1935–1944. [Google Scholar] [CrossRef] [PubMed]
- Giros, B.; Jaber, M.; Jones, S.R.; Wightman, R.M.; Caron, M.G. Hyperlocomotion and Indifference to Cocaine and Amphetamine in Mice Lacking the Dopamine Transporter. Nature 1996, 379, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Spielewoy, C.; Roubert, C.; Hamon, M.; Nosten-Bertrand, M.; Betancur, C.; Giros, B. Behavioural Disturbances Associated with Hyperdopaminergia in Dopamine-Transporter Knockout Mice. Behav. Pharmacol. 2000, 11, 279–290. [Google Scholar] [CrossRef]
- Hall, F.S.; Sora, I.; Hen, R.; Uhl, G.R. Serotonin/Dopamine Interactions in a Hyperactive Mouse: Reduced Serotonin Receptor 1B Activity Reverses Effects of Dopamine Transporter Knockout. PLoS ONE 2014, 9, e115009. [Google Scholar] [CrossRef]
- Takamatsu, Y.; Hagino, Y.; Sato, A.; Takahashi, T.; Nagasawa, S.Y.; Kubo, Y.; Mizuguchi, M.; Uhl, G.R.; Sora, I.; Ikeda, K. Improvement of Learning and Increase in Dopamine Level in the Frontal Cortex by Methylphenidate in Mice Lacking Dopamine Transporter. Curr. Mol. Med. 2015, 15, 245–252. [Google Scholar] [CrossRef]
- Cinque, S.; Zoratto, F.; Poleggi, A.; Leo, D.; Cerniglia, L.; Cimino, S.; Tambelli, R.; Alleva, E.; Gainetdinov, R.R.; Laviola, G.; et al. Behavioral Phenotyping of Dopamine Transporter Knockout Rats: Compulsive Traits, Motor Stereotypies, and Anhedonia. Front. Psychiatry 2018, 9, 43. [Google Scholar] [CrossRef]
- Sagvolden, T.; Metzger, M.A.; Schiørbeck, H.K.; Rugland, A.L.; Spinnangr, I.; Sagvolden, G. The Spontaneously Hypertensive Rat (SHR) as an Animal Model of Childhood Hyperactivity (ADHD): Changed Reactivity to Reinforcers and to Psychomotor Stimulants. Behav. Neural Biol. 1992, 58, 103–112. [Google Scholar] [CrossRef]
- Vancassel, S.; Blondeau, C.; Lallemand, S.; Cador, M.; Linard, A.; Lavialle, M.; Dellu-Hagedorn, F. Hyperactivity in the Rat Is Associated with Spontaneous Low Level of N-3 Polyunsaturated Fatty Acids in the Frontal Cortex. Behav. Brain Res. 2007, 180, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Hauser, J.; Makulska-Gertruda, E.; Reissmann, A.; Sontag, T.-A.; Tucha, O.; Lange, K.W. The Effects of Nutritional Polyunsaturated Fatty Acids on Locomotor Activity in Spontaneously Hypertensive Rats. Atten. Deficit Hyperact. Disord. 2014, 6, 61–65. [Google Scholar] [CrossRef]
- Moghaddam, M.F.; Shamekhi, M.; Rakhshani, T. Effectiveness of Methylphenidate and PUFA for the Treatment of Patients with ADHD: A Double-Blinded Randomized Clinical Trial. Electron. Physician 2017, 9, 4412–4418. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Bryan, J. Effect of Supplementation with Polyunsaturated Fatty Acids and Micronutrients on Learning and Behavior Problems Associated with Child ADHD. J. Dev. Behav. Pediatr. JDBP 2007, 28, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Bélanger, S.A.; Vanasse, M.; Spahis, S.; Sylvestre, M.-P.; Lippé, S.; L’heureux, F.; Ghadirian, P.; Vanasse, C.-M.; Levy, E. Omega-3 Fatty Acid Treatment of Children with Attention-Deficit Hyperactivity Disorder: A Randomized, Double-Blind, Placebo-Controlled Study. Paediatr. Child Health 2009, 14, 89–98. [Google Scholar] [CrossRef]
- Richardson, A.J.; Puri, B.K. A Randomized Double-Blind, Placebo-Controlled Study of the Effects of Supplementation with Highly Unsaturated Fatty Acids on ADHD-Related Symptoms in Children with Specific Learning Difficulties. Prog. Neuropsychopharmacol. Biol. Psychiatry 2002, 26, 233–239. [Google Scholar] [CrossRef]
- Stevens, L.; Zhang, W.; Peck, L.; Kuczek, T.; Grevstad, N.; Mahon, A.; Zentall, S.S.; Arnold, L.E.; Burgess, J.R. EFA Supplementation in Children with Inattention, Hyperactivity, and Other Disruptive Behaviors. Lipids 2003, 38, 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Assareh, M.; Davari Ashtiani, R.; Khademi, M.; Jazayeri, S.; Rai, A.; Nikoo, M. Efficacy of Polyunsaturated Fatty Acids (PUFA) in the Treatment of Attention Deficit Hyperactivity Disorder. J. Atten. Disord. 2017, 21, 78–85. [Google Scholar] [CrossRef]
- Gillies, D.; Sinn, J.K.; Lad, S.S.; Leach, M.J.; Ross, M.J. Polyunsaturated Fatty Acids (PUFA) for Attention Deficit Hyperactivity Disorder (ADHD) in Children and Adolescents. Cochrane Database Syst. Rev. 2012, CD007986. [Google Scholar] [CrossRef]
- Checa-Ros, A.; Haro-García, A.; Seiquer, I.; Molina-Carballo, A.; Uberos-Fernández, J.; Muñoz-Hoyos, A. Early Monitoring of Fatty Acid Profile in Children with Attention Deficit and/or Hyperactivity Disorder under Treatment with Omega-3 Polyunsaturated Fatty Acids. Minerva Pediatr. 2019, 71, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Aman, M.G.; Mitchell, E.A.; Turbott, S.H. The Effects of Essential Fatty Acid Supplementation by Efamol in Hyperactive Children. J. Abnorm. Child Psychol. 1987, 15, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.G.; Llorente, A.M.; Jensen, C.L.; Fraley, J.K.; Berretta, M.C.; Heird, W.C. A Randomized, Double-Blind, Placebo-Controlled Trial of Docosahexaenoic Acid Supplementation in Children with Attention-Deficit/Hyperactivity Disorder. J. Pediatr. 2001, 139, 189–196. [Google Scholar] [CrossRef]
- Hirayama, S.; Hamazaki, T.; Terasawa, K. Effect of Docosahexaenoic Acid-Containing Food Administration on Symptoms of Attention-Deficit/Hyperactivity Disorder—A Placebo-Controlled Double-Blind Study. Eur. J. Clin. Nutr. 2004, 58, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Sinn, N.; Bryan, J.; Wilson, C. Cognitive Effects of Polyunsaturated Fatty Acids in Children with Attention Deficit Hyperactivity Disorder Symptoms: A Randomised Controlled Trial. Prostaglandins Leukot. Essent. Fatty Acids 2008, 78, 311–326. [Google Scholar] [CrossRef]
- Vaisman, N.; Kaysar, N.; Zaruk-Adasha, Y.; Pelled, D.; Brichon, G.; Zwingelstein, G.; Bodennec, J. Correlation between Changes in Blood Fatty Acid Composition and Visual Sustained Attention Performance in Children with Inattention: Effect of Dietary n-3 Fatty Acids Containing Phospholipids. Am. J. Clin. Nutr. 2008, 87, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Ostlund, S.; Fransson, G.; Kadesjö, B.; Gillberg, C. Omega-3/Omega-6 Fatty Acids for Attention Deficit Hyperactivity Disorder: A Randomized Placebo-Controlled Trial in Children and Adolescents. J. Atten. Disord. 2009, 12, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Raz, R.; Carasso, R.L.; Yehuda, S. The Influence of Short-Chain Essential Fatty Acids on Children with Attention-Deficit/Hyperactivity Disorder: A Double-Blind Placebo-Controlled Study. J. Child Adolesc. Psychopharmacol. 2009, 19, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, P.A.; Birberg-Thornberg, U.; Duchén, K.; Landgren, M.; Malmberg, K.; Pelling, H.; Strandvik, B.; Karlsson, T. EPA Supplementation Improves Teacher-Rated Behaviour and Oppositional Symptoms in Children with ADHD. Acta Paediatr. Oslo Nor. 1992 2010, 99, 1540–1549. [Google Scholar] [CrossRef]
- Hariri, M.; Djazayery, A.; Djalali, M.; Saedisomeolia, A.; Rahimi, A.; Abdolahian, E. Effect of N-3 Supplementation on Hyperactivity, Oxidative Stress and Inflammatory Mediators in Children with Attention-Deficit-Hyperactivity Disorder. Malays. J. Nutr. 2012, 18, 329–335. [Google Scholar] [PubMed]
- Manor, I.; Magen, A.; Keidar, D.; Rosen, S.; Tasker, H.; Cohen, T.; Richter, Y.; Zaaroor-Regev, D.; Manor, Y.; Weizman, A. The Effect of Phosphatidylserine Containing Omega3 Fatty-Acids on Attention-Deficit Hyperactivity Disorder Symptoms in Children: A Double-Blind Placebo-Controlled Trial, Followed by an Open-Label Extension. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2012, 27, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R.C. Eicosapentaenoic and Docosahexaenoic Acids, Cognition, and Behavior in Children with Attention-Deficit/Hyperactivity Disorder: A Randomized Controlled Trial. Nutr. Burbank Los Angel. Cty. Calif 2012, 28, 670–677. [Google Scholar] [CrossRef]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R.C. Increased Erythrocyte Eicosapentaenoic Acid and Docosahexaenoic Acid Are Associated With Improved Attention and Behavior in Children With ADHD in a Randomized Controlled Three-Way Crossover Trial. J. Atten. Disord. 2015, 19, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Perera, H.; Jeewandara, K.C.; Seneviratne, S.; Guruge, C. Combined Ω3 and Ω6 Supplementation in Children with Attention-Deficit Hyperactivity Disorder (ADHD) Refractory to Methylphenidate Treatment: A Double-Blind, Placebo-Controlled Study. J. Child Neurol. 2012, 27, 747–753. [Google Scholar] [CrossRef]
- Behdani, F.; Hebrani, P.; Naseraee, A.; Haghighi, M.B.; Akhavanrezayat, A. Does Omega-3 Supplement Enhance the Therapeutic Results of Methylphenidate in Attention Deficit Hyperactivity Disorder Patients? J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2013, 18, 653–658. [Google Scholar]
- Dubnov-Raz, G.; Khoury, Z.; Wright, I.; Raz, R.; Berger, I. The Effect of Alpha-Linolenic Acid Supplementation on ADHD Symptoms in Children: A Randomized Controlled Double-Blind Study. Front. Hum. Neurosci. 2014, 8, 780. [Google Scholar] [CrossRef]
- Widenhorn-Müller, K.; Schwanda, S.; Scholz, E.; Spitzer, M.; Bode, H. Effect of Supplementation with Long-Chain ω-3 Polyunsaturated Fatty Acids on Behavior and Cognition in Children with Attention Deficit/Hyperactivity Disorder (ADHD): A Randomized Placebo-Controlled Intervention Trial. Prostaglandins Leukot. Essent. Fatty Acids 2014, 91, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Bos, D.J.; Oranje, B.; Veerhoek, E.S.; Van Diepen, R.M.; Weusten, J.M.; Demmelmair, H.; Koletzko, B.; de Sain-van der Velden, M.G.; Eilander, A.; Hoeksma, M.; et al. Reduced Symptoms of Inattention after Dietary Omega-3 Fatty Acid Supplementation in Boys with and without Attention Deficit/Hyperactivity Disorder. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2015, 40, 2298–2306. [Google Scholar] [CrossRef] [PubMed]
- Matsudaira, T.; Gow, R.V.; Kelly, J.; Murphy, C.; Potts, L.; Sumich, A.; Ghebremeskel, K.; Crawford, M.A.; Taylor, E. Biochemical and Psychological Effects of Omega-3/6 Supplements in Male Adolescents with Attention-Deficit/Hyperactivity Disorder: A Randomized, Placebo-Controlled, Clinical Trial. J. Child Adolesc. Psychopharmacol. 2015, 25, 775–782. [Google Scholar] [CrossRef]
- Anand, P.; Sachdeva, A. Effect of Poly Unsaturated Fatty Acids Administration on Children with Attention Deficit Hyperactivity Disorder: A Randomized Controlled Trial. J. Clin. Diagn. Res. JCDR 2016, 10, OC01–OC05. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Mohammadbeigi, A.; Sheykholeslam, H.; Moshiri, E.; Dorreh, F. Omega-3 and Zinc Supplementation as Complementary Therapies in Children with Attention-Deficit/Hyperactivity Disorder. J. Res. Pharm. Pract. 2016, 5, 22–26. [Google Scholar] [CrossRef]
- Barragán, E.; Breuer, D.; Döpfner, M. Efficacy and Safety of Omega-3/6 Fatty Acids, Methylphenidate, and a Combined Treatment in Children With ADHD. J. Atten. Disord. 2017, 21, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Kean, J.D.; Sarris, J.; Scholey, A.; Silberstein, R.; Downey, L.A.; Stough, C. Reduced Inattention and Hyperactivity and Improved Cognition after Marine Oil Extract (PCSO-524®) Supplementation in Children and Adolescents with Clinical and Subclinical Symptoms of Attention-Deficit Hyperactivity Disorder (ADHD): A Randomised, Double-Blind, Placebo-Controlled Trial. Psychopharmacology 2017, 234, 403–420. [Google Scholar] [CrossRef]
- Dashti, N.; Hekmat, H.; Soltani, H.R.; Rahimdel, A.; Javaherchian, M. Comparison of Therapeutic Effects of Omega-3 and Methylphenidate (Ritalin(®)) in Treating Children with Attention Deficit Hyperactivity Disorder. Iran. J. Psychiatry Behav. Sci. 2014, 8, 7–11. [Google Scholar] [PubMed]
- Kahn, R.S.; Sommer, I.E.; Murray, R.M.; Meyer-Lindenberg, A.; Weinberger, D.R.; Cannon, T.D.; O’Donovan, M.; Correll, C.U.; Kane, J.M.; van Os, J.; et al. Schizophrenia. Nat. Rev. Dis. Primer 2015, 1, 15067. [Google Scholar] [CrossRef]
- Murray, R.M.; Lewis, S.W. Is Schizophrenia a Neurodevelopmental Disorder? Br. Med. J. Clin. Res. Ed 1987, 295, 681–682. [Google Scholar] [CrossRef]
- Weinberger, D.R. Implications of Normal Brain Development for the Pathogenesis of Schizophrenia. Arch. Gen. Psychiatry 1987, 44, 660–669. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.J.; Féron, F.P.; Burne, T.H.J.; Mackay-Sim, A.; Eyles, D.W. The Neurodevelopmental Hypothesis of Schizophrenia: A Review of Recent Developments. Ann. Med. 2003, 35, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.J.; O’Donovan, M.C.; Thapar, A.; Craddock, N. Neurodevelopmental Hypothesis of Schizophrenia. Br. J. Psychiatry J. Ment. Sci. 2011, 198, 173–175. [Google Scholar] [CrossRef]
- Feigenson, K.A.; Kusnecov, A.W.; Silverstein, S.M. Inflammation and the Two-Hit Hypothesis of Schizophrenia. Neurosci. Biobehav. Rev. 2014, 38, 72–93. [Google Scholar] [CrossRef] [PubMed]
- Bechter, K. Updating the Mild Encephalitis Hypothesis of Schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 42, 71–91. [Google Scholar] [CrossRef] [PubMed]
- Cullen, A.E.; Fisher, H.L.; Roberts, R.E.; Pariante, C.M.; Laurens, K.R. Daily Stressors and Negative Life Events in Children at Elevated Risk of Developing Schizophrenia. Br. J. Psychiatry J. Ment. Sci. 2014, 204, 354–360. [Google Scholar] [CrossRef]
- Manrique-Garcia, E.; Zammit, S.; Dalman, C.; Hemmingsson, T.; Andreasson, S.; Allebeck, P. Cannabis, Schizophrenia and Other Non-Affective Psychoses: 35 Years of Follow-up of a Population-Based Cohort. Psychol. Med. 2012, 42, 1321–1328. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.-Q.; Sun, W.-S.; Liu, B.-X.; Feng, G.-Y.; Yu, L.; Yang, L.; He, G.; Sham, P.; Susser, E.; St. Clair, D.; et al. Prenatal Malnutrition and Adult Schizophrenia: Further Evidence From the 1959-1961 Chinese Famine. Schizophr. Bull. 2009, 35, 568–576. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Chen, G.; Guo, C.; Wen, X.; Song, X.; Zheng, X. Long-Term Effect of Prenatal Exposure to Malnutrition on Risk of Schizophrenia in Adulthood: Evidence from the Chinese Famine of 1959-1961. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2018, 51, 42–47. [Google Scholar] [CrossRef]
- Egbujo, C.N.; Sinclair, D.; Hahn, C.-G. Dysregulations of Synaptic Vesicle Trafficking in Schizophrenia. Curr. Psychiatry Rep. 2016, 18, 77. [Google Scholar] [CrossRef]
- Grace, A.A. Dysregulation of the Dopamine System in the Pathophysiology of Schizophrenia and Depression. Nat. Rev. Neurosci. 2016, 17, 524–532. [Google Scholar] [CrossRef]
- Weinberger, D.R.; Berman, K.F.; Daniel, D.G. Mesoprefrontal Cortical Dopaminergic Activity and Prefrontal Hypofunction in Schizophrenia. Clin. Neuropharmacol. 1992, 15 Pt A (Suppl. 1), 568A–569A. [Google Scholar] [CrossRef]
- Mossaheb, N.; Schloegelhofer, M.; Schaefer, M.R.; Fusar-Poli, P.; Smesny, S.; McGorry, P.; Berger, G.; Amminger, G.P. Polyunsaturated Fatty Acids in Emerging Psychosis. Curr. Pharm. Des. 2012, 18, 576–591. [Google Scholar] [CrossRef]
- Berger, M.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.B.; Berger, G.E.; et al. Relationship Between Polyunsaturated Fatty Acids and Psychopathology in the NEURAPRO Clinical Trial. Front. Psychiatry 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; Toyota, T.; Owada, Y.; Hayashi, T.; Iwayama, Y.; Matsumata, M.; Ishitsuka, Y.; Nakaya, A.; Maekawa, M.; Ohnishi, T.; et al. Fabp7 Maps to a Quantitative Trait Locus for a Schizophrenia Endophenotype. PLoS Biol. 2007, 5, e297. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Kim, H.-J.; Park, J.K.; Kim, J.W.; Chung, J.-H. Association between Polymorphisms of Arachidonate 12-Lipoxygenase (ALOX12) and Schizophrenia in a Korean Population. Behav. Brain Funct. BBF 2010, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Ross, B.M.; Turenne, S.; Moszczynska, A.; Warsh, J.J.; Kish, S.J. Differential Alteration of Phospholipase A2 Activities in Brain of Patients with Schizophrenia. Brain Res. 1999, 821, 407–413. [Google Scholar] [CrossRef]
- Liu, Y.; Jandacek, R.; Rider, T.; Tso, P.; McNamara, R.K. Elevated Delta-6 Desaturase (FADS2) Expression in the Postmortem Prefrontal Cortex of Schizophrenic Patients: Relationship with Fatty Acid Composition. Schizophr. Res. 2009, 109, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Chalon, S.; Vancassel, S.; Zimmer, L.; Guilloteau, D.; Durand, G. Polyunsaturated Fatty Acids and Cerebral Function: Focus on Monoaminergic Neurotransmission. Lipids 2001, 36, 937–944. [Google Scholar] [CrossRef]
- Healy-Stoffel, M.; Levant, B. N-3 (Omega-3) Fatty Acids: Effects on Brain Dopamine Systems and Potential Role in the Etiology and Treatment of Neuropsychiatric Disorders. CNS Neurol. Disord. Drug Targets 2018, 17, 216–232. [Google Scholar] [CrossRef]
- Maekawa, M.; Watanabe, A.; Iwayama, Y.; Kimura, T.; Hamazaki, K.; Balan, S.; Ohba, H.; Hisano, Y.; Nozaki, Y.; Ohnishi, T.; et al. Polyunsaturated Fatty Acid Deficiency during Neurodevelopment in Mice Models the Prodromal State of Schizophrenia through Epigenetic Changes in Nuclear Receptor Genes. Transl. Psychiatry 2017, 7, e1229. [Google Scholar] [CrossRef]
- Fedorova, I.; Alvheim, A.R.; Hussein, N.; Salem, N. Deficit in Prepulse Inhibition in Mice Caused by Dietary N-3 Fatty Acid Deficiency. Behav. Neurosci. 2009, 123, 1218–1225. [Google Scholar] [CrossRef]
- Kumari, V.; Soni, W.; Mathew, V.M.; Sharma, T. Prepulse Inhibition of the Startle Response in Men with Schizophrenia: Effects of Age of Onset of Illness, Symptoms, and Medication. Arch. Gen. Psychiatry 2000, 57, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, N.R.; Light, G.A.; Sprock, J.; Calkins, M.E.; Green, M.F.; Greenwood, T.A.; Gur, R.E.; Gur, R.C.; Lazzeroni, L.C.; Nuechterlein, K.H.; et al. Deficient Prepulse Inhibition in Schizophrenia Detected by the Multi-Site COGS. Schizophr. Res. 2014, 152, 503–512. [Google Scholar] [CrossRef]
- Mena, A.; Ruiz-Salas, J.C.; Puentes, A.; Dorado, I.; Ruiz-Veguilla, M.; De la Casa, L.G. Reduced Prepulse Inhibition as a Biomarker of Schizophrenia. Front. Behav. Neurosci. 2016, 10. [Google Scholar] [CrossRef]
- Shioda, N.; Yamamoto, Y.; Watanabe, M.; Binas, B.; Owada, Y.; Fukunaga, K. Heart-Type Fatty Acid Binding Protein Regulates Dopamine D2 Receptor Function in Mouse Brain. J. Neurosci. Off. J. Soc. Neurosci. 2010, 30, 3146–3155. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Kida, H.; Kagawa, Y.; Yasumoto, Y.; Miyazaki, H.; Islam, A.; Ogata, M.; Yanagawa, Y.; Mitsushima, D.; Fukunaga, K.; et al. FABP3 in the Anterior Cingulate Cortex Modulates the Methylation Status of the Glutamic Acid Decarboxylase67 Promoter Region. J. Neurosci. Off. J. Soc. Neurosci. 2018, 38, 10411–10423. [Google Scholar] [CrossRef]
- Ludolph, A.G.; Kassubek, J.; Schmeck, K.; Glaser, C.; Wunderlich, A.; Buck, A.K.; Reske, S.N.; Fegert, J.M.; Mottaghy, F.M. Dopaminergic Dysfunction in Attention Deficit Hyperactivity Disorder (ADHD), Differences between Pharmacologically Treated and Never Treated Young Adults: A 3,4-Dihdroxy-6-[18F]Fluorophenyl-l-Alanine PET Study. NeuroImage 2008, 41, 718–727. [Google Scholar] [CrossRef] [PubMed]
- Heinz, A.; Schlagenhauf, F. Dopaminergic Dysfunction in Schizophrenia: Salience Attribution Revisited. Schizophr. Bull. 2010, 36, 472–485. [Google Scholar] [CrossRef] [PubMed]
- Ducrocq, F.; Walle, R.; Contini, A.; Oummadi, A.; Caraballo, B.; van der Veldt, S.; Boyer, M.-L.; Aby, F.; Tolentino-Cortez, T.; Helbling, J.-C.; et al. Causal Link between N-3 Polyunsaturated Fatty Acid Deficiency and Motivation Deficits. Cell Metab. 2020, 31, 755–772.e7. [Google Scholar] [CrossRef]
- Berland, C.; Montalban, E.; Perrin, E.; Di Miceli, M.; Nakamura, Y.; Martinat, M.; Sullivan, M.; Davis, X.S.; Shenasa, M.A.; Martin, C.; et al. Circulating Triglycerides Gate Dopamine-Associated Behaviors through DRD2-Expressing Neurons. Cell Metab. 2020, 31, 773–790.e11. [Google Scholar] [CrossRef]
- Amminger, G.P.; Schäfer, M.R.; Papageorgiou, K.; Klier, C.M.; Cotton, S.M.; Harrigan, S.M.; Mackinnon, A.; McGorry, P.D.; Berger, G.E. Long-Chain Omega-3 Fatty Acids for Indicated Prevention of Psychotic Disorders: A Randomized, Placebo-Controlled Trial. Arch. Gen. Psychiatry 2010, 67, 146–154. [Google Scholar] [CrossRef]
- Bloemen, O.J.N.; de Koning, M.B.; Schmitz, N.; Nieman, D.H.; Becker, H.E.; de Haan, L.; Dingemans, P.; Linszen, D.H.; van Amelsvoort, T.A.M.J. White-Matter Markers for Psychosis in a Prospective Ultra-High-Risk Cohort. Psychol. Med. 2010, 40, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, N.; Bartholomeusz, C.; Whitford, T.; Hermens, D.F.; Nelson, B.; Rice, S.; Whittle, S.; Pantelis, C.; McGorry, P.; Schäfer, M.R.; et al. White Matter Integrity in Individuals at Ultra-High Risk for Psychosis: A Systematic Review and Discussion of the Role of Polyunsaturated Fatty Acids. BMC Psychiatry 2016, 16, 287. [Google Scholar] [CrossRef]
- Schlögelhofer, M.; Amminger, G.P.; Schaefer, M.R.; Fusar-Poli, P.; Smesny, S.; McGorry, P.; Berger, G.; Mossaheb, N. Polyunsaturated Fatty Acids in Emerging Psychosis: A Safer Alternative? Early Interv. Psychiatry 2014, 8, 199–208. [Google Scholar] [CrossRef]
- McNamara, R.K.; Jandacek, R.; Rider, T.; Tso, P.; Hahn, C.-G.; Richtand, N.M.; Stanford, K.E. Abnormalities in the Fatty Acid Composition of the Postmortem Orbitofrontal Cortex of Schizophrenic Patients: Gender Differences and Partial Normalization with Antipsychotic Medications. Schizophr. Res. 2007, 91, 37–50. [Google Scholar] [CrossRef]
- Khan, M.M.; Evans, D.R.; Gunna, V.; Scheffer, R.E.; Parikh, V.V.; Mahadik, S.P. Reduced Erythrocyte Membrane Essential Fatty Acids and Increased Lipid Peroxides in Schizophrenia at the Never-Medicated First-Episode of Psychosis and after Years of Treatment with Antipsychotics. Schizophr. Res. 2002, 58, 1–10. [Google Scholar] [CrossRef]
- Arvindakshan, M.; Sitasawad, S.; Debsikdar, V.; Ghate, M.; Evans, D.; Horrobin, D.F.; Bennett, C.; Ranjekar, P.K.; Mahadik, S.P. Essential Polyunsaturated Fatty Acid and Lipid Peroxide Levels in Never-Medicated and Medicated Schizophrenia Patients. Biol. Psychiatry 2003, 53, 56–64. [Google Scholar] [CrossRef]
- Kale, A.; Naphade, N.; Sapkale, S.; Kamaraju, M.; Pillai, A.; Joshi, S.; Mahadik, S. Reduced Folic Acid, Vitamin B12 and Docosahexaenoic Acid and Increased Homocysteine and Cortisol in Never-Medicated Schizophrenia Patients: Implications for Altered One-Carbon Metabolism. Psychiatry Res. 2010, 175, 47–53. [Google Scholar] [CrossRef]
- Robinson, D.G.; Gallego, J.A.; John, M.; Hanna, L.A.; Zhang, J.-P.; Birnbaum, M.L.; Greenberg, J.; Naraine, M.; Peters, B.D.; McNamara, R.K.; et al. A Potential Role for Adjunctive Omega-3 Polyunsaturated Fatty Acids for Depression and Anxiety Symptoms in Recent Onset Psychosis: Results from a 16 week Randomized Placebo-Controlled Trial for Participants Concurrently Treated with Risperidone. Schizophr. Res. 2019, 204, 295–303. [Google Scholar] [CrossRef]
- Bozzatello, P.; Brignolo, E.; De Grandi, E.; Bellino, S. Supplementation with Omega-3 Fatty Acids in Psychiatric Disorders: A Review of Literature Data. J. Clin. Med. 2016, 5, 67. [Google Scholar] [CrossRef] [PubMed]
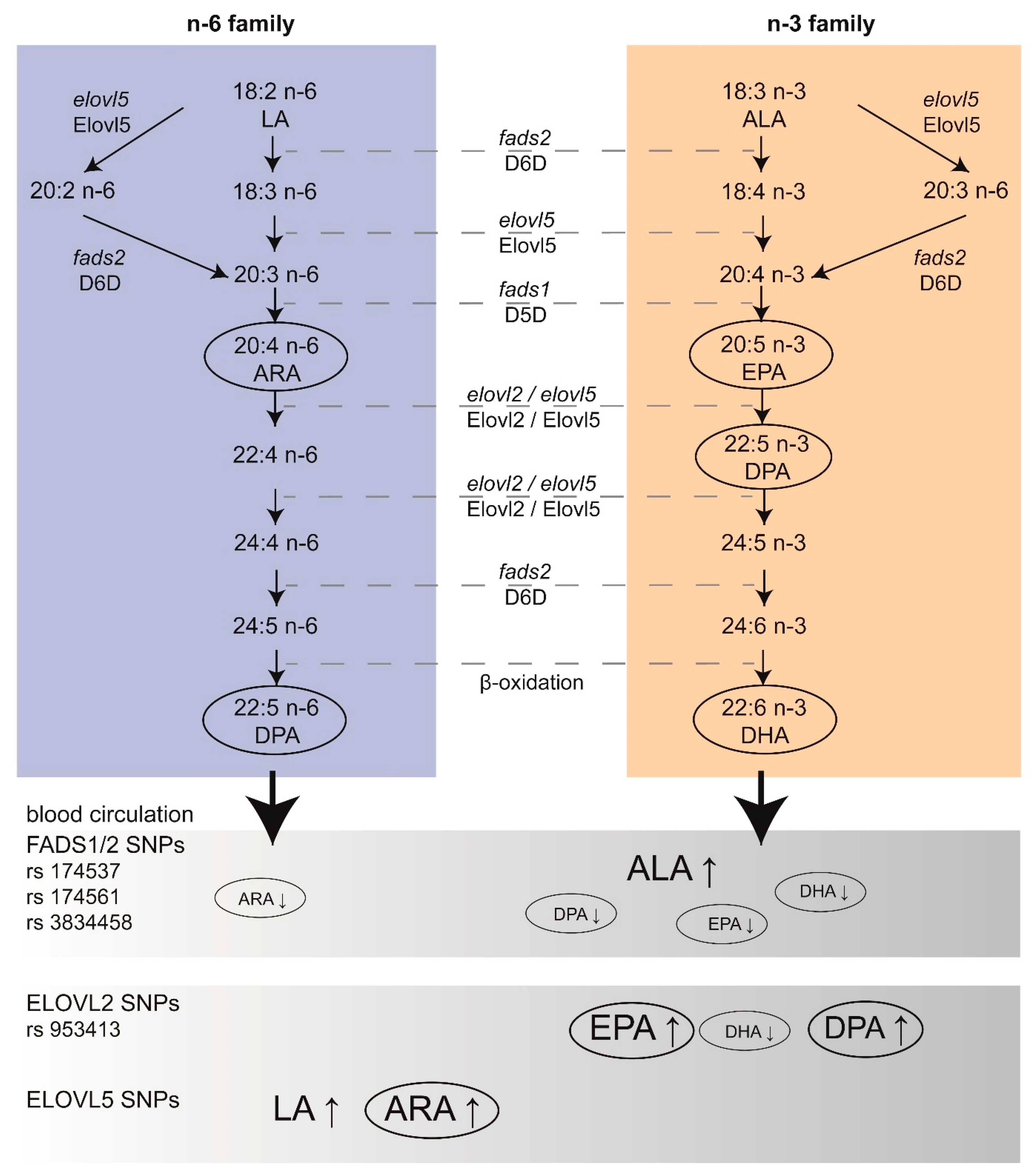

| Study (Year) | Ref. | SNPs | Impact on LC PUFAs Levels | Child Development |
|---|---|---|---|---|
| Morales (2011) | [18] | Mother: fads elovl5 Children: fads elovl5 | Higher colostrum levels of LC n-3 PUFA. | Improved cognition at 14 months. Modification of cognition by breastfeeding. |
| de Groot et al. (2019) | [19] | fads1-fads2 (rs174537, rs175461, rs3834458) elovl2 (rs953413) | Decreased levels of EPA and ARA. Reduced DHA levels. | |
| Tanaka et al. (2009) | [20] | fads (rs174537) elovl2 (rs953413) | Higher ALA, LA and lower ARA, EPA, DPA and DHA levels. Lower DHA and higher DPA and ARA levels. | |
| Al-Hilal et al. (2013) | [151] | fads1-fads2 (rs174537) | Interaction of rs174537 genotype with dietary EPA + DHA supplementation influence D5D and D6D activity. | |
| Lemaitre et al. (2011) | [152] | fads1-fads2 | Increase in ALA levels. Decrease in EPA levels. | |
| elovl2 | Increase in EPA/DPA levels. Decrease in DHA levels. |
| Design | Study (Year) | Ref. | Final Sample Size | Duration | Intervention | Main Outcome(s) |
|---|---|---|---|---|---|---|
| Case reports | Johnson (2003) | [317] | One patient | 4 weeks | EPA 540 mg/day | Significant improvements in symptoms. |
| Infante (2018) | [312] | One patient | 24 months | EPA 204 g/day, DHA 1.2 g/day, vitamin D 25,000 IU/week | Beneficial improvements in psychiatric symptoms. | |
| Open-label trials | Politi (2008) | [310] | 19 interventions | 6 weeks | EPA + DHA 1.86 g/day, vitamin E 10 mg/day | No significant effects. |
| Meiri (2009) | [318] | Nine interventions | 12 weeks | EPA 380 mg/day, DHA 180 mg/day | Some improvements. | |
| Johnson (2010) | [320] | 10 interventions | 3 months | DHA 400 mg/day | No significant effects. | |
| Ooi (2015) | [319] | 41 interventions | 12 weeks | DHA 840 mg/day, EPA 192 mg/day, ARA 66 mg/day, GLA 144 mg/day, vitamin E 60 mg/day, thyme oil mg/day | Improved core symptoms. | |
| Yui (2016) | [304] | 21 controls and 21 interventions | 16 weeks | LA 480 mg/day, ALA 240 mg/kg | Beneficial effects on aberrant behaviours and social responsiveness. | |
| Randomised clinical trials | Amminger (2007) | [321] | Five controls and seven interventions | 6 weeks | EPA 0.84 g/day, DHA 0.7 g/day | Improved hyperactivity and stereotypy. |
| Bent (2011) | [322] | 12 controls and 13 interventions | 12 weeks | EPA 0.7 g/day, DHA 0.46 g/day | No significant effects. | |
| Yui (2011 and 2012) | [323,324] | Six controls and seven interventions | 16 weeks | DHA 0.24 g/day, ARA 0.24 g/day | Improvements in social withdrawal and stereotypy. | |
| Bent (2014) | [325] | 28 controls and 29 interventions | 6 weeks | EPA 0.7 g/day, DHA 0.46 g/day | No significant effects. | |
| Voigt (2014) | [326] | 15 controls and 19 interventions | 6 months | DHA 0.2 g/day | No significant effects. | |
| Mankad (2015) | [327] | 19 controls and 18 interventions | 6 months | EPA + DHA 1.5 g/day | No significant effects. | |
| Parellada (2017) | [328] | 68 patients | 8 weeks | EPA 577.5–693 mg/day, DHA 385–462 mg/day, vitamin E 1.6–2.01 mg/day | Within subject improvements in social motivation and social communication. | |
| Boone (2017) | [329] | 16 controls and 15 interventions | 3 months | EPA 338 mg/day, DHA 225 mg/day, GLA 83 mg/day, n-9 306 mg/day | No significant effects. | |
| Sheppard (2017) | [315] | 12 controls and 12 interventions | 3 months | EPA 338 mg/day, DHA 225 mg/day, GLA 83 mg/day, n-9 306 mg/day | Improved gesture and word use. | |
| Keim (2018) | [316] | 16 controls and 15 interventions | 3 months | EPA 338 mg/day, DHA 225 mg/day, GLA 83 mg/day, n-9 306 mg/day | Improvements in symptoms, but limited to one subscale. | |
| Mazahery (2019) | [313] | 16 controls and 23 interventions | 12 months | DHA 722 mg/day, vitamin D 2000 IU/day | Improved irritability. |
| Design | Study (Year) | Ref. | Final Sample Size | Duration | Intervention | Main Outcome(s) |
|---|---|---|---|---|---|---|
| Open-label trial | Checa-Ros (2019) | [375] | 40 patients | 1 month | EPA 70 mg/day, DHA 250 mg/day, methylphenidate 1 mg/kg/day | Improved attention, improved core symptoms |
| Randomized clinical trials | Aman (1987) | [376] | 31 patients | 4 weeks | LA 2.16 g/day, GLA 270 mg/day | Only minimal effects. |
| Voigt (2001) | [377] | 27 controls and 27 interventions | 4 months | DHA 345 mg/day | No significant effect. | |
| Richardson (2002) | [371] | 14 controls and 15 interventions | 12 weeks | EPA 186 mg/day, DHA 450 mg/day, ALA 96 mg/day, LA 864 mg/day, ARA 42 mg/day, vitamin E 60 IU/day, thyme oil 8 mg/day | General behavioral improvements and small effects. | |
| Stevens (2003) | [372] | 22 controls and 25 interventions | 4 months | EPA 80 mg/day, DHA 480 mg/day, GLA 96 mg/day, ARA 40 mg/day, vitamin E 24 mg/day | No clear benefit but some improvements. | |
| Hirayama (2004) | [378] | 20 controls, 20 interventions | 2 months | DHA 3.6 g/week | Only minimal effects. | |
| Sinn (2007) | [369] | 27 controls and 36 interventions | 15 weeks | EPA 558 mg/day, DHA 174 mg/day, GLA 60 mg/day, vitamin E 10.8 m/day | Improvements in core symptoms. | |
| Sinn (2008) | [379] | 27–38 controls and 72–88 interventions | 15–30 weeks | EPA 558 mg/day, DHA 174 mg/day, GLA 60 mg/day | Improved attention. | |
| Vaisman (2008) | [380] | 21 controls and 21 interventions | 3 months | Fish oil 799 mg/day | Improved attention. | |
| Bélanger (2009) | [370] | 13 controls and 13 interventions | 16 weeks | EPA 20–25 mg/kg/day, DHA 8.5–10.5 mg/kg/day, vitamin E in traces | Improvements in core symptoms. | |
| Johnson (2009) | [381] | 38 controls and 37 interventions | 3–6 months | EPA 558 mg/day, DHA 174 mg/day, GLA 60 mg/day, vitamin E 10.8 mg/day | Improved symptoms in a subgroup. | |
| Raz (2009) | [382] | 31 controls and 32 interventions | 7 weeks | LA 480 mg/day, ALA 120 mg/day, vitamin E 10 mg/day, mineral oil 190 mg/day | No significant effect. | |
| Gustafsson (2010) | [383] | 42 controls and 40 interventions | 15 weeks | EPA 500 mg/day, DHA 2.7 mg/day, vitamin E 10 mg/day | Improved attention. | |
| Hariri (2012) | [384] | 50 controls and 53 interventions | 8 weeks | EPA 635 mg/day, DHA 195 mg/day | Improved symptoms. | |
| Manor (2012) | [385] | 47 controls and 100 interventions | 15–30 weeks | EPA 80 mg/day, DHA 40 mg/day | Improved impulsivity. | |
| Milte (2012) | [386] | 90 patients | 12 months | EPA 264–1109 mg/day, DHA 108–1032 mg/day, LA 1467 mg/day | Improved symptoms in a subgroup. | |
| Milte (2015) | [387] | 87 patients | 12 months | EPA 264–1109 mg/day, DHA 108–1032 mg/day, LA 1467 mg/day | Improved symptoms in a subgroup. | |
| Perera (2012) | [388] | 46 controls and 48 interventions | 6 months | n-3 592.7 m/day, n-6 361.5 mg/day, methylphenidate 0.7–1 mg/kg/day, | Improved symptoms. | |
| Behdani (2013) | [389] | 33 controls and 36 interventions | 8 weeks | EPA 720 mg/day, DHA 480 mg/day, methylphenidate 1 mg/kg/day | No significant effect. | |
| Dashti (2014) | [398] | 28 controls and 28 interventions | 3 days | n-3 1 g/day, methylphenidate 0.9–3 mg/kg/day | Improved symptoms. | |
| Dubnov-Raz (2014) | [390] | Nine controls and eight interventions | 8 weeks | ALA 1 g/day | No significant effect. | |
| Widenhorn-Müller (2014) | [391] | 44 controls and 49 interventions | 16 weeks | EPA 600 mg/day, DHA 120 mg/day | No significant effect | |
| Bos (2015) | [392] | 19 controlsand 19 interventions | 16 weeks | EPA 650 mg/day, DHA 650 mg/day | Improved attention. | |
| Matsudaira (2015) | [393] | 36 controls and 33 interventions | 12 weeks | EPA 558 mg/day, DHA 174 mg/day, GLA 60 mg/day, vitamin E 9.6 mg/day | No significant effect. | |
| Anand (2016) | [394] | 25 controls and 25 interventions | 4 months | EPA 180 mg/day, DHA 120 mg/day | Improved symptoms in a subgroup. | |
| Salehi (2016) | [395] | 50 controls and 50 interventions | 8 weeks | EPA 100–400 mg/day, methylphenidate 0.5–1 mg/kg/day | Improved symptoms. | |
| Assareh (2017) | [373] | 40 patients | 10 weeks | EPA 35 mg/day, DHA 241 mg/day, n-6 LC PUFAs 150 mg/day, methylphenidate 1 mg/kg/day | No significant effect. | |
| Barragán (2017) | [396] | 30 controls and 30 interventions | 12 months | EPA 558 mg/day, DHA 174 mg/day, GLA 60 mg/day, methylphenidate 0.5–1 mg/kg/day | Improved symptoms. | |
| Kean (2017) | [397] | 58 controls and 54 interventions | 14 weeks | EPA 21.9–29.2 mg/day, DHA 16.5–22 mg/day, vitamin E 0.67–0.9 mg/day | Improved hyperactivity and inattention. | |
| Moghaddam (2017) | [368] | 40 patients | 8 weeks | EPA 180 mg/day, DHA 120 mg/day, methylphenidate 1 mg/kg/day | Improvements in symptoms. | |
| Chang (2019) | [270] | 44 controls and 48 interventions | 12 weeks | EPA 1.2 g/day | Improved attention and vigilance in patients with low levels of EPA only. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinat, M.; Rossitto, M.; Di Miceli, M.; Layé, S. Perinatal Dietary Polyunsaturated Fatty Acids in Brain Development, Role in Neurodevelopmental Disorders. Nutrients 2021, 13, 1185. https://doi.org/10.3390/nu13041185
Martinat M, Rossitto M, Di Miceli M, Layé S. Perinatal Dietary Polyunsaturated Fatty Acids in Brain Development, Role in Neurodevelopmental Disorders. Nutrients. 2021; 13(4):1185. https://doi.org/10.3390/nu13041185
Chicago/Turabian StyleMartinat, Maud, Moïra Rossitto, Mathieu Di Miceli, and Sophie Layé. 2021. "Perinatal Dietary Polyunsaturated Fatty Acids in Brain Development, Role in Neurodevelopmental Disorders" Nutrients 13, no. 4: 1185. https://doi.org/10.3390/nu13041185
APA StyleMartinat, M., Rossitto, M., Di Miceli, M., & Layé, S. (2021). Perinatal Dietary Polyunsaturated Fatty Acids in Brain Development, Role in Neurodevelopmental Disorders. Nutrients, 13(4), 1185. https://doi.org/10.3390/nu13041185






