Effects of Two Workload-Matched High-Intensity Interval Training Protocols on Regional Body Composition and Fat Oxidation in Obese Men
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Overview
2.3. Anthropometry and Body Composition
2.4. Peak Oxygen Uptake and Substrate Oxidation during Submaximal Cycling
2.5. Training Protocols
2.6. Dietary Intake and Habitual Physical Activity Assessment
2.7. Statistical Analysis
3. Results
3.1. Energy Intake and Habitual Physical Activity
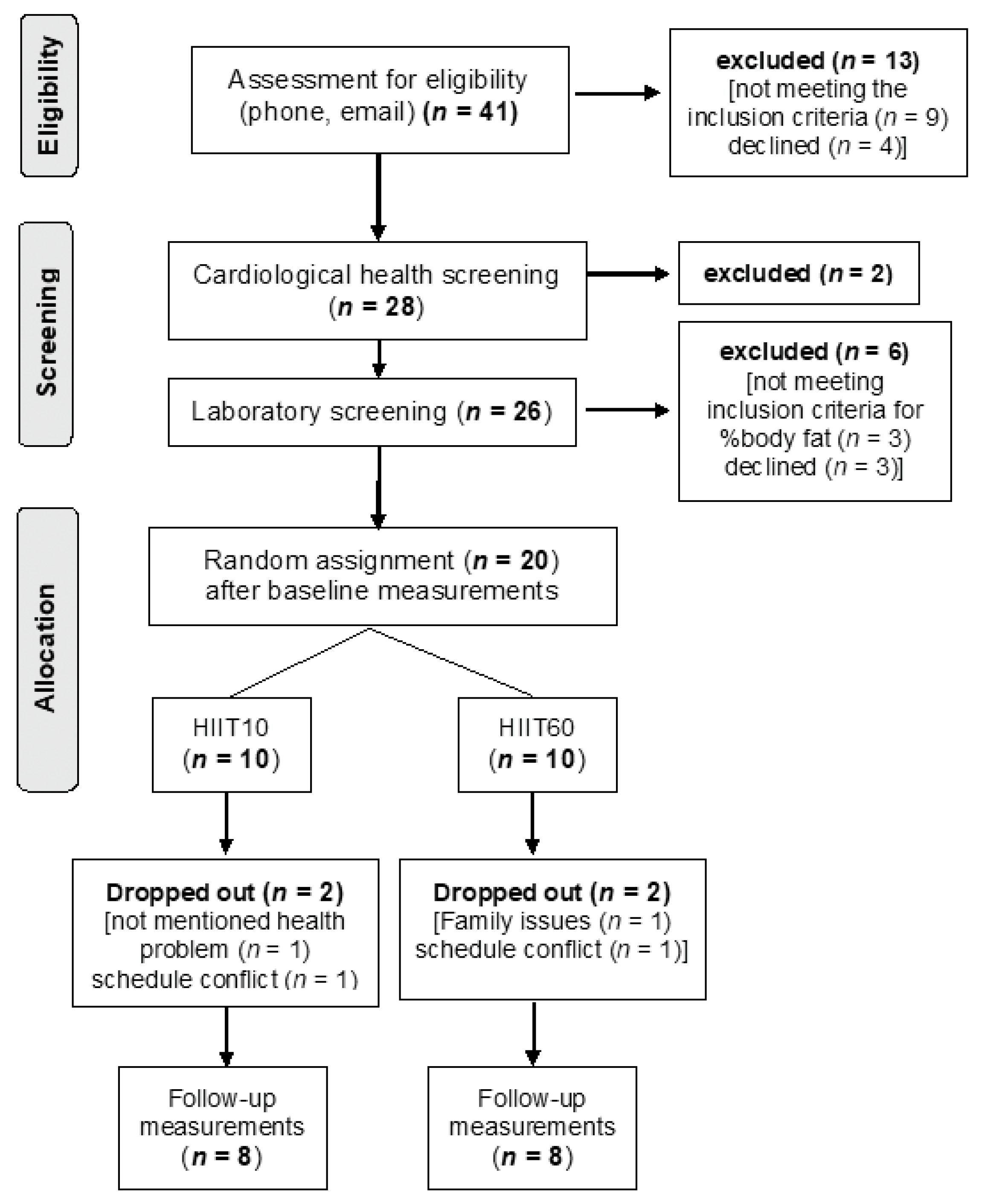
3.2. Exercise Compliance
3.3. Power Output and Energy Expenditure during HIIT Sessions
3.4. Cardiorespiratory Fitness
3.5. Anthropometric and Body Composition Variables
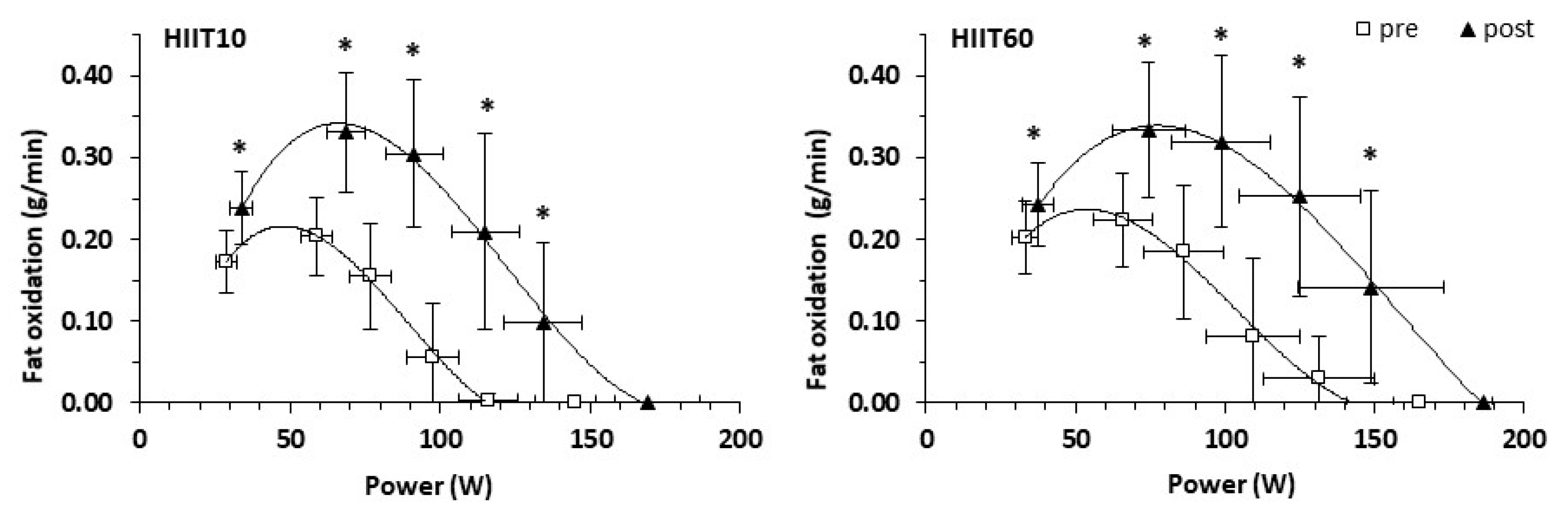
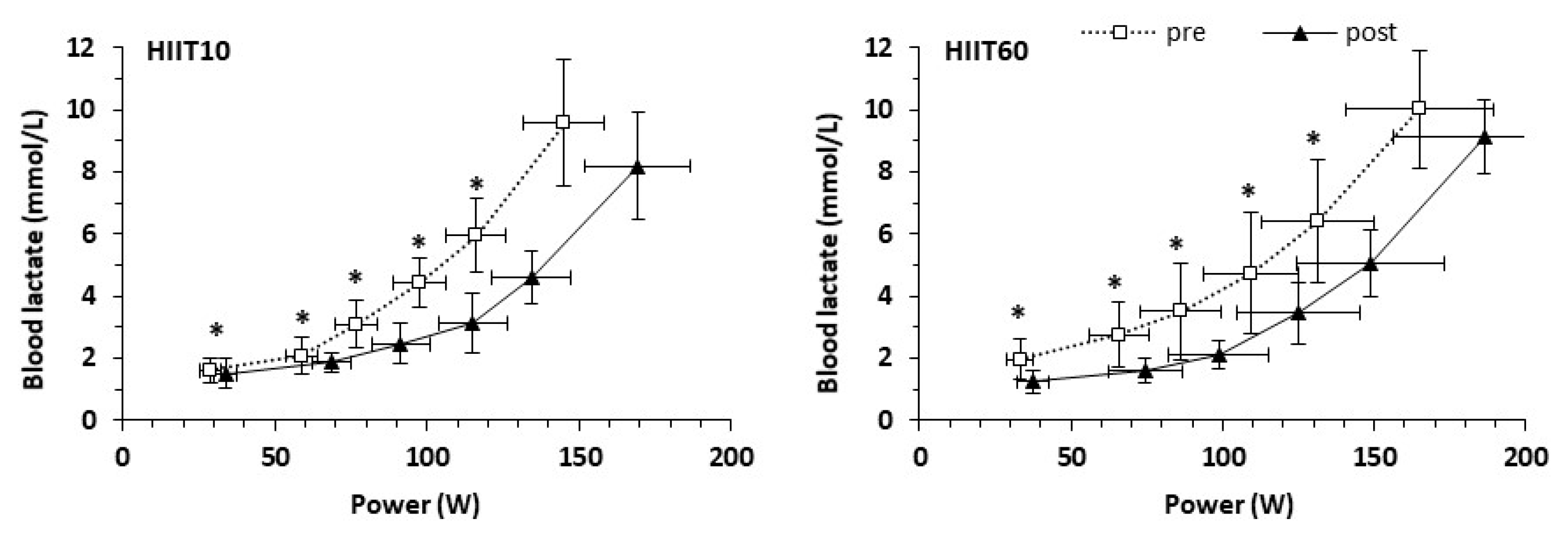
3.6. Substrate Oxidation Indices and Blood Lactate Concentration during Submaximal Exercise
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abarca-Gómez, L.; Abdeen, Z.A.; Hamid, Z.A.; Abu-Rmeileh, N.M.; Acosta-Cazares, B.; Acuin, C.; Adams, R.J.; Aekplakorn, W.; Afsana, K.; Aguilar-Salinas, C.A.; et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- Finkelstein, E.A.; Khavjou, O.A.; Thompson, H.; Trogdon, J.G.; Pan, L.; Sherry, B.; Dietz, W. Obesity and severe obesity forecasts through 2030. Am. J. Prev. Med. 2012, 42, 563–570. [Google Scholar] [CrossRef]
- Manore, M.M.; Larson-Meyer, D.E.; Lindsay, A.R.; Hongu, N.; Houtkooper, L. Dynamic energy balance: An integrated framework for discussing diet and physical activity in obesity prevention—is it more than eating less and exercising more? Nutrients 2017, 9, 905. [Google Scholar] [CrossRef] [PubMed]
- Petridou, A.; Siopi, A.; Mougios, V. Exercise in the management of obesity. Metabolism 2019, 92, 163–169. [Google Scholar] [CrossRef]
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1·9 million participants. Lancet Glob. Health 2018, 6, e1077–e1086. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Gibala, M.J.; McGee, S.L. Metabolic adaptations to short-term high-intensity interval training: A little pain for a lot of gain? Exerc. Sport Sci. Rev. 2008, 36, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Heydari, M.; Freund, J.; Boutcher, S.H. The effect of high-intensity intermittent exercise on body composition of overweight young males. J. Obes. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, G.; Iancu, H.D. High-intensity exercise training does not influence body weight but improves lipid oxidation in obese adults: A 6-week RCT. BMJ. Open Sport Exerc. Med. 2017, 3, e000283. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Berggren, J.R.; Boyle, K.E.; Chapman, W.H.; Houmard, J.A. Skeletal muscle lipid oxidation and obesity: Influence of weight loss and exercise. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E726–E732. [Google Scholar] [CrossRef]
- Perry, C.G.; Heigenhauser, G.J.; Bonen, A.; Spriet, L.L. High-intensity aerobic interval training increases fat and carbohydrate metabolic capacities in human skeletal muscle. Appl. Physiol. Nutr. Metab. 2008, 33, 1112–1123. [Google Scholar] [CrossRef] [PubMed]
- Talanian, J.L.; Holloway, G.P.; Snook, L.A.; Heigenhauser, G.J.; Bonen, A.; Spriet, L.L. Exercise training increases sarcolemmal and mitochondrial fatty acid transport proteins in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E180–E188. [Google Scholar] [CrossRef]
- Venables, M.C.; Achten, J.; Jeukendrup, A.E. Determinants of fat oxidation during exercise in healthy men and women: A cross-sectional study. J. Appl. Physiol. 2005, 98, 160–167. [Google Scholar] [CrossRef]
- Bogdanis, G.C.; Vangelakoudi, A.; Maridaki, M. Peak fat oxidation rate during walking in sedentary overweight men and women. JSSM 2008, 7, 525–531. [Google Scholar]
- Karppinen, J.E.; Rottensteiner, M.; Wiklund, P.; Hamalainen, K.; Laakkonen, E.K.; Kaprio, J.; Kainulainen, H.; Kujala, U.M. Fat oxidation at rest and during exercise in male monozygotic twins. Eur. J. Appl. Physiol. 2019, 119, 2711–2722. [Google Scholar] [CrossRef] [PubMed]
- Despres, J.P. From syndrome X to cardiometabolic risk: Clinical and public health implications. Proc. Nutr. Soc. 2020, 79, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Schoettl, T.; Fischer, I.P.; Ussar, S. Heterogeneity of adipose tissue in development and metabolic function. J. Exp. Biol. 2018, 221 (Suppl. S1). [Google Scholar] [CrossRef]
- Stavrinou, P.S.; Bogdanis, G.C.; Giannaki, C.D.; Terzis, G.; Hadjicharalambous, M. High-intensity Interval Training Frequency: Cardiometabolic Effects and Quality of Life. Int. J. Sports Med. 2018, 39, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Andreato, L.V.; Esteves, J.V.; Coimbra, D.R.; Moraes, A.J.P.; de Carvalho, T. The influence of high-intensity interval training on anthropometric variables of adults with overweight or obesity: A systematic review and network meta-analysis. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2019, 20, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Tong, T.K.; Zhang, H.; Shi, H.; Liu, Y.; Ai, J.; Nie, J.; Kong, Z. Comparing Time Efficiency of Sprint vs. High-Intensity Interval Training in Reducing Abdominal Visceral Fat in Obese Young Women: A Randomized, Controlled Trial. Front. Physiol. 2018, 9, 1048. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.S.; Hwang, H.; Kim, J.; Park, H.Y.; Lim, K. Effect of interval exercise versus continuous exercise on excess post-exercise oxygen consumption during energy-homogenized exercise on a cycle ergometer. J. Exerc. Nutr. Biochem. 2019, 23, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Germano, M.D.; Sindorf, M.A.G.; Da Silva, C.E.; Evangelista, A.L.; Bocalini, D.S.; Lopes, C.R. High intensity interval training: Cardiorespiratory adaptations, metabolic and performance. Inter. J. Sports Sci. 2015, 5, 240–247. [Google Scholar] [CrossRef]
- Christmass, M.A.; Dawson, B.; Arthur, P.G. Effect of work and recovery duration on skeletal muscle oxygenation and fuel use during sustained intermittent exercise. Eur. J. Appl. Physiol. Occup. Physiol. 1999, 80, 436–447. [Google Scholar] [CrossRef]
- Price, M.; Halabi, K. The effects of work-rest duration on intermittent exercise and subsequent performance. J. Sports Sci. 2005, 23, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Tschakert, G.; Kroepfl, J.; Mueller, A.; Moser, O.; Groeschl, W.; Hofmann, P. How to regulate the acute physiological response to “aerobic” high-intensity interval exercise. J. Sports Sci. Med. 2015, 14, 29–36. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- WHO. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Bazzocchi, A.; Ponti, F.; Albisinni, U.; Battista, G.; Guglielmi, G. DXA: Technical aspects and application. Eur. J. Radiol. 2016, 85, 1481–1492. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.C.; Wallace, J.P.; Eggert, K.E. Predicting max HR and the HR-VO2 relationship for exercise prescription in obesity. Med. Sci. Sports Exerc. 1993, 25, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Pescatello, L.S.; Riebe, D.; Thompson, P.D. (Eds.) ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Jeukendrup, A.E.; Wallis, G.A. Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int. J. Sports Med. 2005, 26 (Suppl. S1), S28–S37. [Google Scholar] [CrossRef]
- Merrill, A.L.; Watt, B.K. Energy Value of Foods: Basis and Derivation; Human Nutrition Research Branch, Agricultural Research Service, US Department of Agriculture: Washington, DC, USA, 1955.
- Ekblom, B. Energy expenditure at rest and during exercise. In International Textbook of Obesity, 1st ed.; Björntorp, P., Ed.; John Wiley & Sons, Ltd: Chichester, UK, 2001; pp. 145–154. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Laurence Erlbaum Associates Inc.: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Thompson, W.R. Worldwide Survey of Fitness Trends for 2020. ACSM’S Health Fit. J. 2019, 23, 10–18. [Google Scholar] [CrossRef]
- Batacan, R.B., Jr.; Duncan, M.J.; Dalbo, V.J.; Tucker, P.S.; Fenning, A.S. Effects of high-intensity interval training on cardiometabolic health: A systematic review and meta-analysis of intervention studies. Br. J. Sports Med. 2017, 51, 494–503. [Google Scholar] [CrossRef]
- Milanovic, Z.; Sporis, G.; Weston, M. Effectiveness of High-Intensity Interval Training (HIT) and Continuous Endurance Training for VO2max Improvements: A Systematic Review and Meta-Analysis of Controlled Trials. Sports Med. 2015, 45, 1469–1481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tong, T.K.; Kong, Z.; Shi, Q.; Liu, Y.; Nie, J. Exercise training-induced visceral fat loss in obese women: The role of training intensity and modality. Scand. J. Med. Sci. Sports 2021, 31, 30–43. [Google Scholar] [CrossRef] [PubMed]
- LaForgia, J.; Withers, R.T.; Gore, C.J. Effects of exercise intensity and duration on the excess post-exercise oxygen consumption. J. Sports Sci. 2006, 24, 1247–1264. [Google Scholar] [CrossRef]
- Gahreman, D.; Wang, R.; Boutcher, Y.; Boutcher, S. Green Tea, Intermittent Sprinting Exercise, and Fat Oxidation. Nutrients 2015, 7, 5646–5663. [Google Scholar] [CrossRef] [PubMed]
- Islam, H.; Townsend, L.K.; Hazell, T.J. Modified sprint interval training protocols. Part I. Physiological responses. Appl. Physiol. Nutr. Metab. 2017, 42, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, R.A.d.; Cruz, R.S.d.O.; Turnes, T.; Pereira, K.L.; Caputo, F. Relationships between VO2 and blood lactate responses after all-out running exercise. Appl. Physiol. Nutr. Metab. 2015, 40, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Tan, S.J.; Markworth, J.F.; Broadbent, J.A.; Skinner, T.L.; Cameron-Smith, D. Metabolic and hormonal responses to isoenergetic high-intensity interval exercise and continuous moderate-intensity exercise. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E539–E552. [Google Scholar] [CrossRef] [PubMed]
- Moniz, S.C.; Islam, H.; Hazell, T.J. Mechanistic and methodological perspectives on the impact of intense interval training on post-exercise metabolism. Scand. J. Med. Sci. Sports 2020, 30, 638–651. [Google Scholar] [CrossRef] [PubMed]
- Welle, S.; Nair, K.S. Relationship of resting metabolic rate to body composition and protein turnover. Am. J. Physiol. 1990, 258, E990–E998. [Google Scholar] [CrossRef]
- Schoenfeld, B.J. Does exercise-induced muscle damage play a role in skeletal muscle hypertrophy? J. Strength Cond. Res. 2012, 26, 1441–1453. [Google Scholar] [CrossRef]
- Ravussin, E.; Lillioja, S.; Anderson, T.E.; Christin, L.; Bogardus, C. Determinants of 24-hour energy expenditure in man. Methods and results using a respiratory chamber. J. Clin. Investig. 1986, 78, 1568–1578. [Google Scholar] [CrossRef] [PubMed]
- Cava, E.; Yeat, N.C.; Mittendorfer, B. Preserving Healthy Muscle during Weight Loss. Adv. Nutr. 2017, 8, 511–519. [Google Scholar] [CrossRef]
- Arad, A.D.; DiMenna, F.J.; Thomas, N.; Tamis-Holland, J.; Weil, R.; Geliebter, A.; Albu, J.B. High-intensity interval training without weight loss improves exercise but not basal or insulin-induced metabolism in overweight/obese African American women. J. Appl. Physiol. 2015, 119, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Gillen, J.B.; Percival, M.E.; Ludzki, A.; Tarnopolsky, M.A.; Gibala, M.J. Interval training in the fed or fasted state improves body composition and muscle oxidative capacity in overweight women. Obesity 2013, 21, 2249–2255. [Google Scholar] [CrossRef] [PubMed]
- Nybo, L.; Sundstrup, E.; Jakobsen, M.D.; Mohr, M.; Hornstrup, T.; Simonsen, L.; Bülow, J.; Randers, M.B.; Nielsen, J.J.; Aagaard, P.; et al. High-intensity training versus traditional exercise interventions for promoting health. Med. Sci. Sports Exerc. 2010, 42, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Tjonna, A.E.; Leinan, I.M.; Bartnes, A.T.; Jenssen, B.M.; Gibala, M.J.; Winett, R.A.; Wisloff, U. Low- and high-volume of intensive endurance training significantly improves maximal oxygen uptake after 10-weeks of training in healthy men. PLoS ONE 2013, 8, e65382. [Google Scholar] [CrossRef]
- Foster, C.; Farland, C.V.; Guidotti, F.; Harbin, M.; Roberts, B.; Schuette, J.; Tuuri, A.; Doberstein, S.T.; Porcari, J.P. The Effects of High Intensity Interval Training vs Steady State Training on Aerobic and Anaerobic Capacity. J. Sports Sci. Med. 2015, 14, 747–755. [Google Scholar] [PubMed]
- Joyner, M.J. Physiological limits to endurance exercise performance: Influence of sex. J. Physiol. 2017, 595, 2949–2954. [Google Scholar] [CrossRef]
- Kong, Z.; Sun, S.; Liu, M.; Shi, Q. Short-Term High-Intensity Interval Training on Body Composition and Blood Glucose in Overweight and Obese Young Women. J. Diabetes Res. 2016, 2016, 4073618. [Google Scholar] [CrossRef]
- Dohlmann, T.L.; Hindso, M.; Dela, F.; Helge, J.W.; Larsen, S. High-intensity interval training changes mitochondrial respiratory capacity differently in adipose tissue and skeletal muscle. Physiol. Rep. 2018, 6, e13857. [Google Scholar] [CrossRef] [PubMed]
- MacInnis, M.J.; Gibala, M.J. Physiological adaptations to interval training and the role of exercise intensity. J. Physiol. 2017, 595, 2915–2930. [Google Scholar] [CrossRef] [PubMed]
- Zurbuchen, A.; Lanzi, S.; Voirol, L.; Trindade, C.B.; Gojanovic, B.; Kayser, B.; Bourdillon, N.; Cheneviere, X.; Malatesta, D. Fat Oxidation Kinetics Is Related to Muscle Deoxygenation Kinetics During Exercise. Front. Physiol. 2020, 11, 571. [Google Scholar] [CrossRef]
- Shaw, C.S.; Swinton, C.; Morales-Scholz, M.G.; McRae, N.; Erftemeyer, T.; Aldous, A.; Murphy, R.M.; Howlett, K.F. Impact of exercise training status on the fiber type-specific abundance of proteins regulating intramuscular lipid metabolism. J. Appl. Physiol. 2020, 128, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Burgomaster, K.A.; Howarth, K.R.; Phillips, S.M.; Rakobowchuk, M.; Macdonald, M.J.; McGee, S.L.; Gibala, M.J. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J. Physiol. 2008, 586, 151–160. [Google Scholar] [CrossRef] [PubMed]
| HIIT10 | HIIT60 | |||||||
|---|---|---|---|---|---|---|---|---|
| Week 1 | Week 4 | Week 8 | Week 1 | Week 4 | Week 8 | Effects | p-Value | |
| Energy | 1985 | 1707 | 1852 | 2313 | 2280 | 2327 | Time | 0.480 |
| intake | ±514 | ±441 | ±524 | ±796 | ±707 | ±795 | Group | 0.132 |
| (kcal) | Interaction | 0.632 | ||||||
| CHO | 39.8 | 42.5 | 42.2 | 43.4 | 40.3 | 41.5 | Time | 0.990 |
| (%) | ±10.4 | ±11.5 | ±12.3 | ±5.6 | ±3.9 | ±7.4 | Group | 0.947 |
| Interaction | 0.600 | |||||||
| Fat | 41.2 | 38.7 | 40.0 | 40.0 | 40.0 | 40.2 | Time | 0.921 |
| (%) | ±9.7 | ±11.7 | ±10.5 | ±5.9 | ±8.6 | ±11.3 | Group | 0.975 |
| Interaction | 0.914 | |||||||
| Protein | 19.0 | 18.7 | 17.9 | 16.7 | 19.6 | 18.3 | Time | 0.562 |
| (%) | ±5.2 | ±3.8 | ±3.3 | ±3.8 | ±6.9 | ±5.5 | Group | 0.869 |
| Interaction | 0.436 |
| HIIT10 | HΙΤ60 | Pre vs. Post | ||||||
|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre vs. Post Main Effect | Change (90 CI%); Hedges g | Group | Interaction | |
| VO2peak (mL/kg/min) | 29.4 ± 2.4 | 36.0 ± 2.1 | 32.5 ± 5.0 | 38.1 ± 5.5 | p < 0.001, η2 = 0.495 | 6.1 (5.3, 6.8), ES = 1.42 | p = 0.534, η2 = 0.028 | p = 0.233, η2 = 0.104 |
| HRpeak (beats/min)) | 175 ± 10 | 177 ± 7 | 177 ± 9 | 173 ± 8 | p = 0.718, η2 = 0.010 | −1 (−5, 3), ES = 0.11 | p = 0.817, η2 = 0.004 | p = 0.248, η2 = 0.094 |
| Wpeak (W) | 193 ± 16 | 231 ± 9 | 214 ± 32 | 250 ± 40 | p < 0.001, η2 = 0.905 | 37 (32, 42), ES = 1.28 | p = 0.152, η2 = 0.141 | p = 0.141, η2 = 0.007 |
| HIIT10 | HΙΤ60 | Pre vs. Post | ||||||
|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre vs. Post Main Effect | Change (90 CI%); Hedges g | Group | Interaction | |
| Body mass (kg) | 91.94 ± 7.92 | 90.17 ± 7.20 | 94.26 ± 14.10 | 93.88 ± 14.41 | p = 0.043, η2 = 0.262 | −1.12 (−1.88, −0.35), ES = 0.09 | p = 0.765, η2 = 0.007 | p = 0.175, η2 = 0.127 |
| BMI (kg/m2) | 29.8 ± 2.1 | 29.2 ± 1.8 | 30.1 ± 2.6 | 30.0 ± 2.7 | p = 0.037, η2 = 0.274 | −0.36 (−0.62, −0.10), ES = 0.16 | p = 0.655, η2 = 0.015 | p = 0.166, η2 = 0.133 |
| WC (cm) | 103.7 ± 7.3 | 99.9 ± 7.0 | 102.4 ± 9.6 | 100.7 ± 9.8 | p < 0.001, η2 = 0.626 | −2.8 (−3.8, −1.8), ES = 1.64 | p = 0.952, η2 = 0.000 | p = 0.077, η2 = 0.206 |
| Total body fat (%) | 31.5 ± 4.0 | 29.4 ± 4.2 | 32.1 ± 3.9 | 31.0 ± 3.1 | p = 0.043, η2 = 0.262 | −1.6 (−2.7, −0.4), ES = 0.41 | p = 0.577, η2 = 0.023 | p = 0.514, η2 = 0.031 |
| Total fat mass (kg) | 29.12 ± 5.68 | 26.68 ± 5.35 | 30.26 ± 6.15 | 29.07 ± 5.30 | p = 0.002, η2 = 0.495 | −1.81 (−2.63, −0.99), ES = 0.32 | p = 0.534, η2 = 0.028 | p = 0.233, η2 = 0.104 |
| Trunk fat mass (kg) | 17.64 ± 3.06 | 15.87 ± 2.78 | 17.34 ± 3.49 | 16.21 ± 3.26 | p < 0.001, η2 = 0.612 | −1.45 (−1.95, −0.94), ES = 0.46 | p = 0.991, η2 = 0.000 | p = 0.321, η2 = 0.070 |
| Leg fat mass (kg) | 8.86 ± 2.49 | 8.42 ± 2.37 | 9.77 ± 3.12 | 9.81 ± 2.48 | p = 0.235, η2 = 0.099 | −0.20 (−0.48, 0.07), ES = 0.08 | p = 0.393, η2 = 0.053 | p = 0.174, η2 = 0.128 |
| Arm fat mass (kg) | 2.63 ± 0.80 | 2.39 ± 0.71 | 3.15 ± 0.89 | 3.06 ± 0.90 | p = 0.077, η2 = 0.206 | −0.17 (−0.31, −0.02), ES = 0.19 | p = 0.162, η2 = 0.134 | p = 0.413, η2 = 0.048 |
| Lean body mass (kg) | 54.57 ± 4.31 | 55.40 ± 4.69 | 55.87 ± 10.02 | 56.68 ± 10.34 | p < 0.001, η2 = 0.705 | 0.82 (0.59, 1.05), ES = 0.10 | p = 0.748, η2 = 0.008 | p = 0.975, η2 = 0.000 |
| Leg lean mass (kg) | 18.41 ± 1.57 | 19.25 ± 1.87 | 19.29 ± 2.78 | 20.17 ± 3.08 | p < 0.001, η2 = 0.723 | 0.86 (0.63, 1.08), ES = 0.35 | p = 0.463, η2 = 0.039 | p = 0.904, η2 = 0.001 |
| Trunk lean mass (kg) | 29.21 ± 3.39 | 29.26 ± 3.33 | 29.76 ± 6.47 | 29.72 ± 6.47 | p = 0.756, η2 = 0.007 | 0.00 (−0.08, 0.08), ES = 0.00 | p = 0.896, η2 = 0.001 | p = 0.205, η2 = 0.112 |
| Arm lean mass (kg) | 6.95 ± 0.89 | 6.88 ± 0.93 | 6.81 ± 0.95 | 6.80 ± 0.96 | p = 0.633, η2 = 0.017 | −0.04 (−0.17, 0.09), ES = 0.04 | p = 0.807, η2 = 0.004 | p = 0.748, η2 = 0.008 |
| Head mass (kg) | 5.24 ± 0.24 | 5.13 ± 0.38 | 5.21 ± 0.35 | 5.21 ± 0.32 | p = 0.251, η2 = 0.093 | −0.06 (−0.02, 0.14), ES = 0.17 | p = 0.869, η2 = 0.002 | p = 0.262, η2 = 0.089 |
| Total body BMC (kg) | 3.01 ± 0.30 | 2.96 ± 0.33 | 2.92 ± 0.33 | 2.91 ± 0.32 | p = 0.409, η2 = 0.049 | −0.03 (−0.05, 0.00), ES = 0.08 | p = 0.661, η2 = 0.014 | p = 0.529, η2 = 0.029 |
| HIIT10 | HIIT60 | Pre-Post Change | ||||||
|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | (90% CI) | Effects | p-Value; ES | η2 | |
| PFO | 0.20 | 0.33 | 0.24 | 0.34 | 0.12 | Time (pre vs. post) | <0.001; ES: 1.65 | 0.699 |
| (g/min) | ±0.05 | ±0.08 | ±0.06 | ±0.09 | (0.08, 0.15) | Group | 0.536 | 0.028 |
| Time × Group | 0.525 | 0.030 | ||||||
| PFO | 3.72 | 6.11 | 4.23 | 6.19 | 2.18 | Time (pre vs. post) | <0.001; ES: 1.72 | 0.670 |
| (mg/min | ±0.74 | ±1.24 | ±0.82 | ±1.92 | (1.54, 2.82) | Group | 0.54 | 0.027 |
| /kg LBM) | Time × Group | 0.597 | 0.020 | |||||
| Fatpeak | 33.2 | 36.6 | 34.1 | 38.6 | 3.9 | Time (pre vs. post) | 0.039; ES: 0.68 | 0.270 |
| (%VO2peak) | ±5.5 | ±7.1 | ±3.8 | ±6.5 | (1.2, 6.7) | Group | 0.547 | 0.026 |
| Time × Group | 0.783 | 0.006 | ||||||
| Fatpeak | 60.1 | 62.2 | 58.7 | 63.6 | 3.5 | Time (pre vs. post) | 0.011; ES: 0.10 | 0.383 |
| (%HRpeak) | ±5.8 | ±5.3 | ±7.3 | ±4.4 | (1.5, 5.5) | Group | 0.989 | <0.001 |
| Time × Group | 0.263 | 0.089 | ||||||
| COP | 29.8 | 40.8 | 32.3 | 36.9 | 7.8 | Time (pre vs. post) | 0.023; ES: 1.01 | 0.363 |
| (%VO2peak) | ±6.2 | ±6.9 | ±6.2 | ±10.4 | (3.2, 12.4) | Group | 0.793 | 0.006 |
| Time × Group | 0.301 | 0.089 | ||||||
| PCHO | 2.42 | 2.93 | 2.41 | 3.11 | 0.61 | Time (pre vs. post) | <0.001; ES = 1.03 | 0.620 |
| (g/min) | ±0.45 | ±0.68 | ±0.62 | ±0.52 | (0.32, 0.89) | Group | 0.732 | 0.009 |
| Time × Group | 0.466 | 0.038 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsirigkakis, S.; Mastorakos, G.; Koutedakis, Y.; Mougios, V.; Nevill, A.M.; Pafili, Z.; Bogdanis, G.C. Effects of Two Workload-Matched High-Intensity Interval Training Protocols on Regional Body Composition and Fat Oxidation in Obese Men. Nutrients 2021, 13, 1096. https://doi.org/10.3390/nu13041096
Tsirigkakis S, Mastorakos G, Koutedakis Y, Mougios V, Nevill AM, Pafili Z, Bogdanis GC. Effects of Two Workload-Matched High-Intensity Interval Training Protocols on Regional Body Composition and Fat Oxidation in Obese Men. Nutrients. 2021; 13(4):1096. https://doi.org/10.3390/nu13041096
Chicago/Turabian StyleTsirigkakis, Spyridon, George Mastorakos, Yiannis Koutedakis, Vassilis Mougios, Alan M. Nevill, Zoe Pafili, and Gregory C. Bogdanis. 2021. "Effects of Two Workload-Matched High-Intensity Interval Training Protocols on Regional Body Composition and Fat Oxidation in Obese Men" Nutrients 13, no. 4: 1096. https://doi.org/10.3390/nu13041096
APA StyleTsirigkakis, S., Mastorakos, G., Koutedakis, Y., Mougios, V., Nevill, A. M., Pafili, Z., & Bogdanis, G. C. (2021). Effects of Two Workload-Matched High-Intensity Interval Training Protocols on Regional Body Composition and Fat Oxidation in Obese Men. Nutrients, 13(4), 1096. https://doi.org/10.3390/nu13041096








