Nutritional Assessment in Adult Patients with Dysphagia: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Data Extraction
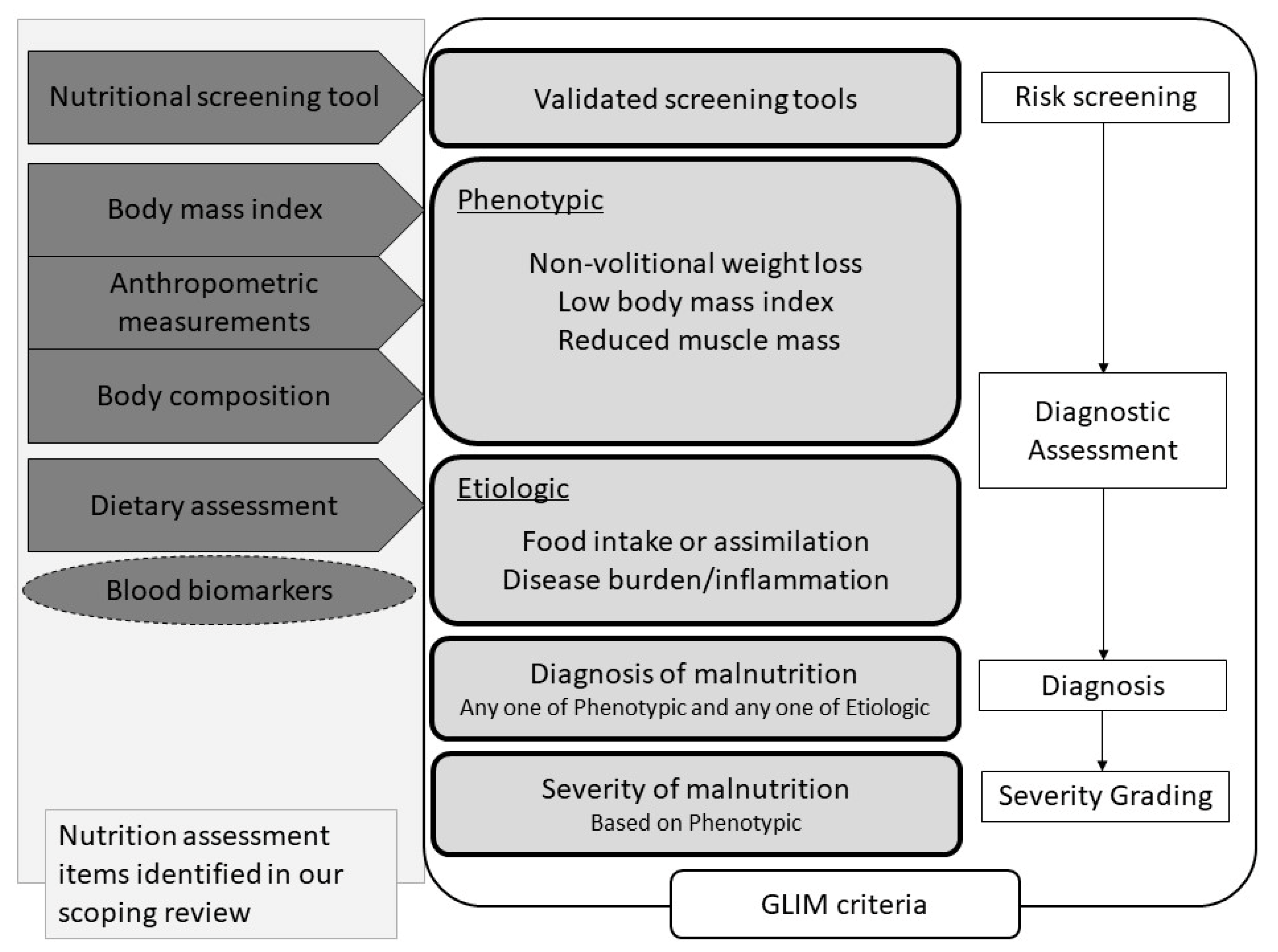
3. Results
3.1. Nutritional Assessment in Adult Patients with Dysphagia
3.2. Blood Biomarkers from Nutritional Assessment in Adult Patients with Dysphagia
3.3. Comparison of Nutritional Assessment Items for Patients with Dysphagia in Acute and Postacute Settings
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cichero, J.A.; Lam, P.; Steele, C.M.; Hanson, B.; Chen, J.; Dantas, R.O.; Duivestein, J.; Kayashita, J.; Lecko, C.; Murray, J.; et al. Development of International Terminology and Definitions for Texture-Modified Foods and Thickened Fluids Used in Dysphagia Management: The IDDSI Framework. Dysphagia 2017, 32, 293–314. [Google Scholar] [CrossRef] [PubMed]
- Pontes, É.S.; Amaral, A.K.; Rêgo, F.L.; Azevedo, E.H.; Silva, P.O. Quality of life in swallowing of the elderly patients affected by stroke. Arq. Gastroenterol. 2017, 54, 27–32. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, Y.W.; Kim, H.S.; Lee, E.H. The mediating and moderating effects of meaning in life on the relationship between depression and quality of life in patients with dysphagia. J. Clin. Nurs. 2019, 28, 2782–2789. [Google Scholar] [CrossRef]
- Thomas, M.N.; Kufeldt, J.; Kisser, U.; Hornung, H.M.; Hoffmann, J.; Andraschko, M.; Werner, J.; Rittler, P. Effects of malnutrition on complication rates, length of hospital stay, and revenue in elective surgical patients in the G-DRG-system. Nutrition 2016, 32, 249–254. [Google Scholar] [CrossRef]
- Allard, J.P.; Keller, H.; Jeejeebhoy, K.N.; Laporte, M.; Duerksen, D.R.; Gramlich, L.; Payette, H.; Bernier, P.; Vesnaver, E.; Davidson, B.; et al. Malnutrition at Hospital Admission-Contributors and Effect on Length of Stay: A Prospective Cohort Study From the Canadian Malnutrition Task Force. JPEN J. Parenter. Enter. Nutr. 2016, 40, 487–497. [Google Scholar] [CrossRef]
- Gallegos, C.; Brito-de la Fuente, E.; Clave, P.; Costa, A.; Assegehegn, G. Nutritional Aspects of Dysphagia Management. Adv. Food Nutr. Res. 2017, 81, 271–318. [Google Scholar] [CrossRef] [PubMed]
- Popman, A.; Richter, M.; Allen, J.; Wham, C. High nutrition risk is associated with higher risk of dysphagia in advanced age adults newly admitted to hospital. Nutr. Diet. 2018, 75, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Blanar, V.; Hodl, M.; Lohrmann, C.; Amir, Y.; Eglseer, D. Dysphagia and factors associated with malnutrition risk: A 5-year multicentre study. J. Adv. Nurs. 2019, 75, 3566–3576. [Google Scholar] [CrossRef] [PubMed]
- Namasivayam, A.M.; Steele, C.M. Malnutrition and Dysphagia in long-term care: A systematic review. J. Nutr. Gerontol. Geriatr. 2015, 34, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Namasivayam-MacDonald, A.M.; Morrison, J.M.; Steele, C.M.; Keller, H. How Swallow Pressures and Dysphagia Affect Malnutrition and Mealtime Outcomes in Long-Term Care. Dysphagia 2017, 32, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Foley, N.C.; Martin, R.E.; Salter, K.L.; Teasell, R.W. A review of the relationship between dysphagia and malnutrition following stroke. J. Rehabil. Med. 2009, 41, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, A.; Momosaki, R.; Kayashita, J.; Fujishima, I. Impact of Multiple Texture-Modified Diets on Oral Intake and Nutritional Status in Older Patients with Pneumonia: A Retrospective Cohort Study. Dysphagia 2020, 35, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, A.; Maeda, K.; Tanaka, K.; Ogawa, M.; Kayashita, J. Texture-modified diets are associated with decreased muscle mass in older adults admitted to a rehabilitation ward. Geriatr. Gerontol. Int. 2018, 18, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Wright, L.; Cotter, D.; Hickson, M.; Frost, G. Comparison of energy and protein intakes of older people consuming a texture modified diet with a normal hospital diet. J. Hum. Nutr. Diet. 2005, 18, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Hudson, H.M.; Daubert, C.R.; Mills, R.H. The interdependency of protein-energy malnutrition, aging, and dysphagia. Dysphagia 2000, 15, 31–38. [Google Scholar] [CrossRef]
- Fujishima, I.; Fujiu-Kurachi, M.; Arai, H.; Hyodo, M.; Kagaya, H.; Maeda, K.; Mori, T.; Nishioka, S.; Oshima, F.; Ogawa, S.; et al. Sarcopenia and dysphagia: Position paper by four professional organizations. Geriatr. Gerontol. Int. 2019, 19, 91–97. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Matsushima, M. Dysphagia Assessed by the 10-Item Eating Assessment Tool Is Associated with Nutritional Status and Activities of Daily Living in Elderly Individuals Requiring Long-Term Care. J. Nutr. Health Aging 2016, 20, 22–27. [Google Scholar] [CrossRef]
- Andrade, P.A.; Santos, C.A.D.; Firmino, H.H.; Rosa, C.O.B. The importance of dysphagia screening and nutritional assessment in hospitalized patients. Einstein 2018, 16, eAO4189. [Google Scholar] [CrossRef] [PubMed]
- Charney, P. Nutrition screening vs nutrition assessment: How do they differ? Nutr. Clin. Pract. 2008, 23, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Correia, M. Nutrition Screening vs Nutrition Assessment: What's the Difference? Nutr. Clin. Pract. 2018, 33, 62–72. [Google Scholar] [CrossRef]
- Reber, E.; Gomes, F.; Vasiloglou, M.F.; Schuetz, P.; Stanga, Z. Nutritional Risk Screening and Assessment. J. Clin. Med. 2019, 8, 65. [Google Scholar] [CrossRef]
- Dent, E.; Hoogendijk, E.O.; Visvanathan, R.; Wright, O.R.L. Malnutrition Screening and Assessment in Hospitalised Older People: A Review. J. Nutr. Health Aging 2019, 23, 431–441. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef]
- Jensen, G.L.; Cederholm, T.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; de Baptista, G.A.; Barazzoni, R.; Blaauw, R.; Coats, A.J.S.; et al. GLIM Criteria for the Diagnosis of Malnutrition: A Consensus Report From the Global Clinical Nutrition Community. JPEN J. Parenter. Enter. Nutr. 2019, 43, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Ueshima, J.; Momosaki, R.; Motokawa, K.; Sonoi, M.; Shimizu, A.; Shirai, Y.; Uno, C.; Kokura, Y.; Shimizu, M.; Nishiyama, A.; et al. Exploring effective nutritional assessment for patients with dysphagia: A scoping review protocol. Available online: https://doi.org/10.6084/m9.figshare.12627416.v1 (accessed on 6 October 2020).
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Munn, Z.; Peters, M.D.J.; Stern, C.; Tufanaru, C.; McArthur, A.; Aromataris, E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 2018, 18, 143. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.; Colquhoun, H.; Kastner, M.; Levac, D.; Ng, C.; Sharpe, J.P.; Wilson, K.; et al. A scoping review on the conduct and reporting of scoping reviews. BMC Med. Res. Methodol. 2016, 16, 15. [Google Scholar] [CrossRef]
- Anderson, S.; Allen, P.; Peckham, S.; Goodwin, N. Asking the right questions: Scoping studies in the commissioning of research on the organisation and delivery of health services. Health Res. Policy Syst. 2008, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.D.J.; Godfrey, C.; McInerney, P.; Munn, Z.; Tricco, A.C.; Khalil, H. Chapter 11: Scoping Reviews (2020 Version). Available online: https://synthesismanual.jbi.global (accessed on 12 June 2020).
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Sampaio, A.D.S.; Epifanio, M.; Costa, C.A.D.; Bosa, V.L.; Benedetti, F.J.; Sarria, E.E.; Oliveira, S.G.; Mundstock, E.; Mattiello, R. Evidence on nutritional assessment techniques and parameters used to determine the nutritional status of children and adolescents: Systematic review. Cien Saude Colet 2018, 23, 4209–4219. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.J.S.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. J. Cachexia Sarcopenia Muscle 2019, 10, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Bath, P.M.; Lee, H.S.; Everton, L.F. Swallowing therapy for dysphagia in acute and subacute stroke. Cochrane Database Syst. Rev. 2018, 10, CD000323. [Google Scholar] [CrossRef]
- da Silva, A.F.; Moreira, E.A.M.; Barni, G.C.; Panza, V.S.P.; Furkim, A.M.; Moreno, Y.M.F. Relationships between high comorbidity index and nutritional parameters in patients with Oropharyngeal Dysphagia. Clin. Nutr. ESPEN 2020, 38, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Ikenaga, Y.; Nakayama, S.; Taniguchi, H.; Ohori, I.; Komatsu, N.; Nishimura, H.; Katsuki, Y. Factors Predicting Recovery of Oral Intake in Stroke Survivors with Dysphagia in a Convalescent Rehabilitation Ward. J. Stroke Cerebrovasc. Dis. 2017, 26, 1013–1019. [Google Scholar] [CrossRef]
- Kim, J.; Min, Y.W.; Lee, H.; Min, B.H.; Lee, J.H.; Rhee, P.L.; Kim, J.J. Comparative Study of Esophageal Self-expandable Metallic Stent Insertion and Gastrostomy Feeding for Dysphagia Caused by Lung Cancer. Korean J. Gastroenterol. 2018, 71, 124–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Y.J.; Chen, W.X.; Zhang, J.L.; He, F.Y.; Zhu, Z.F.; Zeng, Y.; Yang, F.; Tang, S.C. Cervical oesophagostomy in patients with severe dysphagia following radiotherapy for nasopharyngeal carcinoma. J. Laryngol. Otol. 2014, 128, 142–146. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vilardell, N.; Rofes, L.; Arreola, V.; Martin, A.; Muriana, D.; Palomeras, E.; Ortega, O.; Clavé, P. Videofluoroscopic assessment of the pathophysiology of chronic poststroke oropharyngeal dysphagia. Neurogastroenterol. Motil. 2017, 29, 1–8. [Google Scholar] [CrossRef]
- Masiero, S.; Pierobon, R.; Previato, C.; Gomiero, E. Pneumonia in stroke patients with oropharyngeal dysphagia: A six-month follow-up study. Neurol. Sci. 2008, 29, 139–145. [Google Scholar] [CrossRef]
- Barni, G.C.; Moreira, E.A.M.; da Silva, A.F.; Panza, V.S.P.; de Lima Oliveira, D.; Moreno, Y.M.F.; Furkim, A.M. The relationship of serum 25-hydroxyvitamin D concentration with clinical variables in patients with oropharyngeal dysphagia. Clin. Nutr. ESPEN 2020, 38, 229–235. [Google Scholar] [CrossRef]
- Maeda, K.; Koga, T.; Akagi, J. Interferential current sensory stimulation, through the neck skin, improves airway defense and oral nutrition intake in patients with dysphagia: A double-blind randomized controlled trial. Clin. Interv. Aging 2017, 12, 1879–1886. [Google Scholar] [CrossRef] [PubMed]
- Toh Yoon, E.W.; Hirao, J.; Minoda, N. Outcome of Rehabilitation and Swallowing Therapy after Percutaneous Endoscopic Gastrostomy in Dysphagia Patients. Dysphagia 2016, 31, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Nakadate, A.; Otaka, Y.; Kondo, K.; Yamamoto, R.; Matsuura, D.; Honaga, K.; Muraoka, K.; Akaboshi, K.; Liu, M. Age, Body Mass Index, and White Blood Cell Count Predict the Resumption of Oral Intake in Subacute Stroke Patients. J. Stroke Cerebrovasc. Dis. 2016, 25, 2801–2808. [Google Scholar] [CrossRef]
- Ortega, O.; Sakwinska, O.; Combremont, S.; Berger, B.; Sauser, J.; Parra, C.; Zarcero, S.; Nart, J.; Carrión, S.; Clavé, P. High prevalence of colonization of oral cavity by respiratory pathogens in frail older patients with oropharyngeal dysphagia. Neurogastroenterol. Motil. 2015, 27, 1804–1816. [Google Scholar] [CrossRef]
- Lecleire, S.; Di Fiore, F.; Antonietti, M.; Ben Soussan, E.; Hellot, M.F.; Grigioni, S.; Déchelotte, P.; Lerebours, E.; Michel, P.; Ducrotté, P. Undernutrition is predictive of early mortality after palliative self-expanding metal stent insertion in patients with inoperable or recurrent esophageal cancer. Gastrointest. Endosc. 2006, 64, 479–484. [Google Scholar] [CrossRef]
- Jacobsson, C.; Axelsson, K.; Norberg, A.; Asplund, K.; Wenngren, B.I. Outcomes of individualized interventions in patients with severe eating difficulties. Clin. Nurs. Res. 1997, 6, 25–44. [Google Scholar] [CrossRef]
- Nakazawa, Y.; Kikutani, T.; Igarashi, K.; Yajima, Y.; Tamura, F. Associations between tongue strength and skeletal muscle mass under dysphagia rehabilitation for geriatric out patients. J. Prosthodont. Res. 2020, 64, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Smith, Z.L.; Gonzaga, J.E.; Haasler, G.B.; Gore, E.M.; Dua, K.S. Self-Expanding Metal Stents Improve Swallowing and Maintain Nutrition During Neoadjuvant Therapy for Esophageal Cancer. Dig. Dis. Sci. 2017, 62, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Elmståhl, S.; Bülow, M.; Ekberg, O.; Petersson, M.; Tegner, H. Treatment of dysphagia improves nutritional conditions in stroke patients. Dysphagia 1999, 14, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Bülow, M.; Speyer, R.; Baijens, L.; Woisard, V.; Ekberg, O. Neuromuscular electrical stimulation (NMES) in stroke patients with oral and pharyngeal dysfunction. Dysphagia 2008, 23, 302–309. [Google Scholar] [CrossRef]
- Bartolome, G.; Prosiegel, M.; Yassouridis, A. Long-term functional outcome in patients with neurogenic dysphagia. NeuroRehabilitation 1997, 9, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, N.; Stegaroiu, R.; Shibata, S.; Ito, K.; Inoue, M.; Ohuchi, A. Changes in the Oral Moisture and the Amount of Microorganisms in Saliva and Tongue Coating after Oral Ingestion Resumption: A Pilot Study. Open Dent. J. 2016, 10, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Lin, X.; Lin, X.J.; Zheng, W.; Zheng, Z.K.; Lin, Z.M.; Chen, J.H. Therapeutic efficacy of neuromuscular electrical stimulation and electromyographic biofeedback on Alzheimer's disease patients with dysphagia. Medicine 2017, 96, e8008. [Google Scholar] [CrossRef] [PubMed]
- Miyake, N.; Kawamoto, K.; Fujiwara, K.; Hasegawa, Y.; Kitano, H. Subglottic laryngeal closure: A unique modified method of laryngotracheal separation to prevent aspiration. Ann. Otol. Rhinol. Laryngol. 2013, 122, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Kishimoto, S.; Sumi, T.; Uchiyama, M.; Ohno, K.; Kobayashi, H.; Kano, M. Improving the Quality of Life of Patients With Severe Dysphagia by Surgically Closing the Larynx. Ann. Otol. Rhinol. Laryngol. 2019, 128, 96–103. [Google Scholar] [CrossRef]
- Carrión, S.; Roca, M.; Costa, A.; Arreola, V.; Ortega, O.; Palomera, E.; Serra-Prat, M.; Cabré, M.; Clavé, P. Nutritional status of older patients with oropharyngeal dysphagia in a chronic versus an acute clinical situation. Clin. Nutr. 2017, 36, 1110–1116. [Google Scholar] [CrossRef]
- Bahat, G.; Tufan, F.; Saka, B.; Akin, S.; Ozkaya, H.; Yucel, N.; Erten, N.; Karan, M.A. Which body mass index (BMI) is better in the elderly for functional status? Arch. Gerontol. Geriatr. 2012, 54, 78–81. [Google Scholar] [CrossRef]
- Cederholm, T.; Bosaeus, I.; Barazzoni, R.; Bauer, J.; Van Gossum, A.; Klek, S.; Muscaritoli, M.; Nyulasi, I.; Ockenga, J.; Schneider, S.M.; et al. Diagnostic criteria for malnutrition—An ESPEN Consensus Statement. Clin. Nutr. 2015, 34, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Kondrup, J.; Allison, S.P.; Elia, M.; Vellas, B.; Plauth, M. ESPEN guidelines for nutrition screening 2002. Clin. Nutr. 2003, 22, 415–421. [Google Scholar] [CrossRef]
- Rubenstein, L.Z.; Harker, J.O.; Salvà, A.; Guigoz, Y.; Vellas, B. Screening for undernutrition in geriatric practice: Developing the short-form mini-nutritional assessment (MNA-SF). J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M366–M372. [Google Scholar] [CrossRef]
- Stratton, R.J.; Hackston, A.; Longmore, D.; Dixon, R.; Price, S.; Stroud, M.; King, C.; Elia, M. Malnutrition in hospital outpatients and inpatients: Prevalence, concurrent validity and ease of use of the 'malnutrition universal screening tool' ('MUST') for adults. Br. J. Nutr. 2004, 92, 799–808. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Riobó Serván, P.; Sierra Poyatos, R.; Soldo Rodríguez, J.; Gómez-Candela, C.; García Luna, P.P.; Serra-Majem, L. Special considerations for nutritional studies in elderly. Nutr. Hosp. 2015, 31 (Suppl. S3), 84–90. [Google Scholar] [CrossRef]
- Zhao, W.T.; Yang, M.; Wu, H.M.; Yang, L.; Zhang, X.M.; Huang, Y. Systematic Review and Meta-Analysis of the Association between Sarcopenia and Dysphagia. J. Nutr. Health Aging 2018, 22, 1003–1009. [Google Scholar] [CrossRef]
- Detsky, A.S.; McLaughlin, J.R.; Baker, J.P.; Johnston, N.; Whittaker, S.; Mendelson, R.A.; Jeejeebhoy, K.N. What is subjective global assessment of nutritional status? JPEN J. Parenter. Enter. Nutr. 1987, 11, 8–13. [Google Scholar] [CrossRef]
- White, J.V.; Guenter, P.; Jensen, G.; Malone, A.; Schofield, M. Consensus statement: Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition: Characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). JPEN J. Parenter. Enter. Nutr. 2012, 36, 275–283. [Google Scholar] [CrossRef]
- Gabay, C.; Kushner, I. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 1999, 340, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Fleck, A. Clinical and nutritional aspects of changes in acute-phase proteins during inflammation. Proc. Nutr. Soc. 1989, 48, 347–354. [Google Scholar] [CrossRef]
- Zhang, Z.; Pereira, S.L.; Luo, M.; Matheson, E.M. Evaluation of Blood Biomarkers Associated with Risk of Malnutrition in Older Adults: A Systematic Review and Meta-Analysis. Nutrients 2017, 9, 829. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.C.; Corkins, M.R.; Malone, A.; Miller, S.; Mogensen, K.M.; Guenter, P.; Jensen, G.L. The Use of Visceral Proteins as Nutrition Markers: An ASPEN Position Paper. Nutr. Clin. Pract. 2020. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, A.; Maeda, K.; Koyanagi, Y.; Kayashita, J.; Fujishima, I.; Mori, N. The Global Leadership Initiative on Malnutrition-Defined Malnutrition Predicts Prognosis in Persons With Stroke-Related Dysphagia. J. Am. Med. Dir. Assoc. 2019, 20, 1628–1633. [Google Scholar] [CrossRef]
- Jaafar, M.H.; Mahadeva, S.; Tan, K.M.; Chin, A.V.; Kamaruzzaman, S.B.; Khor, H.M.; Saedon, N.I.; Tan, M.P. Long-Term Nasogastric Versus Percutaneous Endoscopic Gastrostomy Tube Feeding in Older Asians With Dysphagia: A Pragmatic Study. Nutr. Clin. Pract. 2019, 34, 280–289. [Google Scholar] [CrossRef]
- Furuta, M.; Yokota, T.; Tsushima, T.; Todaka, A.; Machida, N.; Hamauchi, S.; Yamazaki, K.; Fukutomi, A.; Kawai, S.; Kawabata, T.; et al. Comparison of enteral nutrition with total parenteral nutrition for patients with locally advanced unresectable esophageal cancer harboring dysphagia in definitive chemoradiotherapy. Jpn. J. Clin. Oncol. 2019, 49, 910–918. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Takahashi, R.; Murakami, T. The Prevalence and Prognosis of Sarcopenic Dysphagia in Patients Who Require Dysphagia Rehabilitation. J. Nutr. Health Aging 2019, 23, 84–88. [Google Scholar] [CrossRef]
- Chen, H.J.; Chen, J.L.; Chen, C.Y.; Lee, M.; Chang, W.H.; Huang, T.T. Effect of an Oral Health Programme on Oral Health, Oral Intake, and Nutrition in Patients with Stroke and Dysphagia in Taiwan: A Randomised Controlled Trial. Int. J Environ. Res. Public Health 2019, 16, 2228. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Torres, C.A.; Castillo-Martínez, L.; Reyes-Guerrero, R.; Ramos-Vázquez, A.G.; Zavala-Solares, M.; Cassis-Nosthas, L.; Serralde-Zúñiga, A.E. Design and implementation of modified-texture diet in older adults with oropharyngeal dysphagia: A randomized controlled trial. Eur. J. Clin. Nutr. 2019, 73, 989–996. [Google Scholar] [CrossRef]
- Masaki, S.; Kawamoto, T. Comparison of long-term outcomes between enteral nutrition via gastrostomy and total parenteral nutrition in older persons with dysphagia: A propensity-matched cohort study. PLoS ONE 2019, 14, e0217120. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Matsushima, M.; Momosaki, R.; Yoshida, S.; Mutai, R.; Yodoshi, T.; Murayama, S.; Hayashi, T.; Horiguchi, R.; Ichikawa, H. The effects of resistance training of swallowing muscles on dysphagia in older people: A cluster, randomized, controlled trial. Nutrition 2018, 48, 111–116. [Google Scholar] [CrossRef]
- Martín, A.; Ortega, O.; Roca, M.; Arús, M.; Clavé, P. Respond to Letter to the editor: Effect of a Minimal-Massive Intervention in Hospitalized Older Patients with Oropharyngeal Dysphagia: A Proof of Concept Study. J. Nutr. Health Aging 2018, 22, 1019–1020. [Google Scholar] [CrossRef]
- Maeda, K.; Wakabayashi, H.; Shamoto, H.; Akagi, J. Cognitive impairment has no impact on hospital-associated dysphagia in aspiration pneumonia patients. Geriatr. Gerontol. Int. 2018, 18, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, S.; Okamoto, T.; Takayama, M.; Urushihara, M.; Watanabe, M.; Kiriya, Y.; Shintani, K.; Nakagomi, H.; Kageyama, N. Malnutrition risk predicts recovery of full oral intake among older adult stroke patients undergoing enteral nutrition: Secondary analysis of a multicentre survey (the APPLE study). Clin. Nutr. 2017, 36, 1089–1096. [Google Scholar] [CrossRef]
- Santos, C.A.; Fonseca, J.; Carolino, E.; Guerreiro, A.S. Low Serum Chromium Is Rare In Patients That Underwent Endoscopic Gastrostomy For Long Term Enteral Feeding. Arq. Gastroenterol. 2017, 54, 211–216. [Google Scholar] [CrossRef]
- Zanini, M.; Bagnasco, A.; Catania, G.; Aleo, G.; Sartini, M.; Cristina, M.L.; Ripamonti, S.; Monacelli, F.; Odetti, P.; Sasso, L. A Dedicated Nutritional Care Program (NUTRICARE) to reduce malnutrition in institutionalised dysphagic older people: A quasi-experimental study. J. Clin. Nurs. 2017, 26, 4446–4455. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, Y. Factors associated with the level of oral intake in hospitalized older adults with dysphagia: The importance of mental activity. Clin. Nutr. ESPEN 2016, 13, e52–e54. [Google Scholar] [CrossRef]
- van den Berg, M.G.; Kalf, J.G.; Hendriks, J.C.; Takes, R.P.; van Herpen, C.M.; Wanten, G.J.; Drenth, J.P.; Kaanders, J.H.; Merkx, M.A. Normalcy of food intake in patients with head and neck cancer supported by combined dietary counseling and swallowing therapy: A randomized clinical trial. Head Neck 2016, 38 (Suppl. 1), E198–E206. [Google Scholar] [CrossRef]
- Fonseca, J.; Santos, C.A.; Brito, J. Malnutrition and Clinical Outcome of 234 Head and Neck Cancer Patients who Underwent Percutaneous Endoscopic Gastrostomy. Nutr. Cancer 2016, 68, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Li, H.; Lin, R.; Zheng, J.H.; Wei, Y.P.; Li, J.; Chen, P.; Chen, H.Y. Effects of a feeding intervention in patients with Alzheimer's disease and dysphagia. J. Clin. Nurs. 2016, 25, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Crary, M.A.; Carnaby, G.D.; Shabbir, Y.; Miller, L.; Silliman, S. Clinical Variables Associated with Hydration Status in Acute Ischemic Stroke Patients with Dysphagia. Dysphagia 2016, 31, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.A.; Fonseca, J.; Carolino, E.; Guerreiro, A.S. Serum trace elements in dysphagic gastrostomy candidates before endoscopic gastrostomy for long term enteral feeding. Clin. Nutr. 2016, 35, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M.; Higashibeppu, N.; Arioka, Y.; Nakaya, Y. Swallowing rehabilitation with nutrition therapy improves clinical outcome in patients with dysphagia at an acute care hospital. J. Med. Investig. 2014, 61, 353–360. [Google Scholar] [CrossRef]
- Rio, A.; Ellis, C.; Shaw, C.; Willey, E.; Ampong, M.A.; Wijesekera, L.; Rittman, T.; Nigel Leigh, P.; Sidhu, P.S.; Al-Chalabi, A. Nutritional factors associated with survival following enteral tube feeding in patients with motor neurone disease. J. Hum. Nutr. Diet 2010, 23, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Obara, H.; Tomite, Y.; Doi, M. Serum trace elements in tube-fed neurological dysphagia patients correlate with nutritional indices but do not correlate with trace element intakes: Case of patients receiving enough trace elements intake. Clin. Nutr. 2008, 27, 587–593. [Google Scholar] [CrossRef]
- Wright, L.; Cotter, D.; Hickson, M. The effectiveness of targeted feeding assistance to improve the nutritional intake of elderly dysphagic patients in hospital. J. Hum. Nutr. Diet 2008, 21, 555–562. [Google Scholar] [CrossRef]
- Hamidon, B.B.; Abdullah, S.A.; Zawawi, M.F.; Sukumar, N.; Aminuddin, A.; Raymond, A.A. A prospective comparison of percutaneous endoscopic gastrostomy and nasogastric tube feeding in patients with acute dysphagic stroke. Med. J. Malaysia 2006, 61, 59–66. [Google Scholar]
- Klose, J.; Heldwein, W.; Rafferzeder, M.; Sernetz, F.; Gross, M.; Loeschke, K. Nutritional status and quality of life in patients with percutaneous endoscopic gastrostomy (PEG) in practice: Prospective one-year follow-up. Dig. Dis. Sci. 2003, 48, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Westergren, A.; Karlsson, S.; Andersson, P.; Ohlsson, O.; Hallberg, I.R. Eating difficulties, need for assisted eating, nutritional status and pressure ulcers in patients admitted for stroke rehabilitation. J. Clin. Nurs. 2001, 10, 257–269. [Google Scholar] [CrossRef]
- Thomas, F.J.; Wiles, C.M. Dysphagia and nutritional status in multiple sclerosis. J. Neurol. 1999, 246, 677–682. [Google Scholar] [CrossRef]
- Norton, B.; Homer-Ward, M.; Donnelly, M.T.; Long, R.G.; Holmes, G.K. A randomised prospective comparison of percutaneous endoscopic gastrostomy and nasogastric tube feeding after acute dysphagic stroke. Bmj 1996, 312, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Smithard, D.G.; O’Neill, P.A.; Parks, C.; Morris, J. Complications and outcome after acute stroke. Does dysphagia matter? Stroke 1996, 27, 1200–1204. [Google Scholar] [CrossRef]
- Håglin, L.; Edström, M.; Bäckman, L.; Forsgren, L. Low level of phosphate in male patients reporting swallowing disturbances in early Parkinson's disease. Clin. Nutr. Exp. 2020, 29, 18–29. [Google Scholar] [CrossRef]
- Morone, G.; Iosa, M.; Paolucci, T.; Muzzioli, L.; Paolucci, S. Relationship Between Body Mass Index and Rehabilitation Outcomes in Subacute Stroke With Dysphagia. Am. J. Phys. Med. Rehabil. 2019, 98, 608–612. [Google Scholar] [CrossRef]
- Nishida, T.; Yamabe, K.; Honda, S. Dysphagia is associated with oral, physical, cognitive and psychological frailty in Japanese community-dwelling elderly persons. Gerodontology 2020, 37, 185–190. [Google Scholar] [CrossRef]
- Akazawa, N.; Okawa, N.; Hino, T.; Tsuji, R.; Tamura, K.; Moriyama, H. Dysphagia is more strongly associated with increased intramuscular adipose tissue of the quadriceps than with loss of muscle mass in older inpatients. Nutr. Res. 2019, 65, 71–78. [Google Scholar] [CrossRef]
- Saito, T.; Hayashi, K.; Nakazawa, H.; Yagihashi, F.; Oikawa, L.O.; Ota, T. A Significant Association of Malnutrition with Dysphagia in Acute Patients. Dysphagia 2018, 33, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Nouvenne, A.; Lauretani, F.; Prati, B.; Cerundolo, N.; Maggio, M.; Meschi, T. Survival in older adults with dementia and eating problems: To PEG or not to PEG? Clin. Nutr. 2016, 35, 1512–1516. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Matsushima, M. Neck Circumference Is Not Associated with Dysphagia but with Undernutrition in Elderly Individuals Requiring Long-term Care. J. Nutr. Health Aging 2016, 20, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Sashika, H.; Matsushima, M. Head lifting strength is associated with dysphagia and malnutrition in frail older adults. Geriatr. Gerontol. Int. 2015, 15, 410–416. [Google Scholar] [CrossRef]
- Gellrich, N.C.; Handschel, J.; Holtmann, H.; Krüskemper, G. Oral cancer malnutrition impacts weight and quality of life. Nutrients 2015, 7, 2145–2160. [Google Scholar] [CrossRef] [PubMed]
- Mittal, B.B.; Pauloski, B.R.; Rademaker, A.W.; Discekici-Harris, M.; Helenowski, I.B.; Mellot, A.; Agulnik, M.; Logemann, J.A. Effect of induction chemotherapy on swallow physiology and saliva production in patients with head and neck cancer: A pilot study. Head Neck 2015, 37, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Zhu, X.; Liang, H.; Huang, H.; Yang, J.; Wang, S. Impact of early enteral nutrition on short term prognosis after acute stroke. J. Clin. Neurosci. 2015, 22, 1473–1476. [Google Scholar] [CrossRef]
- Medin, J.; Windahl, J.; von Arbin, M.; Tham, K.; Wredling, R. Eating difficulties among patients 3 months after stroke in relation to the acute phase. J. Adv. Nurs. 2012, 68, 580–589. [Google Scholar] [CrossRef] [PubMed]
- Terré, R.; Mearin, F. Prospective evaluation of oro-pharyngeal dysphagia after severe traumatic brain injury. Brain Inj. 2007, 21, 1411–1417. [Google Scholar] [CrossRef]
- Bertoli, S.; Cardinali, S.; Veggiotti, P.; Trentani, C.; Testolin, G.; Tagliabue, A. Evaluation of nutritional status in children with refractory epilepsy. Nutr. J. 2006, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Penhavel, F.A.; Waitzberg, D.L.; Trevenzol, H.P.; Alves, L.; Zilberstein, B.; Gama-Rodrigues, J. Pre-and postoperative nutritional evaluation in patients with chagasic megaesophagus. Nutr. Hosp. 2004, 19, 89–94. [Google Scholar] [PubMed]
- Reyes, A.L.; Cash, A.J.; Green, S.H.; Booth, I.W. Gastrooesophageal reflux in children with cerebral palsy. Child Care Health Dev. 1993, 19, 109–118. [Google Scholar] [CrossRef]
- Topf, M.C.; Magaña, L.C.; Salmon, K.; Hamilton, J.; Keane, W.M.; Luginbuhl, A.; Curry, J.M.; Cognetti, D.M.; Boon, M.; Spiegel, J.R. Safety and efficacy of functional laryngectomy for end-stage dysphagia. Laryngoscope 2018, 128, 597–602. [Google Scholar] [CrossRef]
- Rogus-Pulia, N.; Rusche, N.; Hind, J.A.; Zielinski, J.; Gangnon, R.; Safdar, N.; Robbins, J. Effects of Device-Facilitated Isometric Progressive Resistance Oropharyngeal Therapy on Swallowing and Health-Related Outcomes in Older Adults with Dysphagia. J. Am. Geriatr. Soc. 2016, 64, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, E.; Kojima, T.; Kaneko, K.; Minashi, K.; Onozawa, M.; Nihei, K.; Fuse, N.; Yano, T.; Yoshino, T.; Tahara, M.; et al. Efficacy of concurrent chemoradiotherapy as a palliative treatment in stage IVB esophageal cancer patients with dysphagia. Jpn. J. Clin. Oncol. 2011, 41, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Martino, R.; Beaton, D.; Diamant, N.E. Using different perspectives to generate items for a new scale measuring medical outcomes of dysphagia (MOD). J. Clin. Epidemiol. 2009, 62, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Benfer, K.A.; Weir, K.A.; Ware, R.S.; Davies, P.S.W.; Arvedson, J.; Boyd, R.N.; Bell, K.L. Parent-reported indicators for detecting feeding and swallowing difficulties and undernutrition in preschool-aged children with cerebral palsy. Dev. Med. Child Neurol. 2017, 59, 1181–1187. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.M.; Corredor, J.; Fisher-Medina, J.; Cohen, J.; Rabinowitz, S. Diagnosis and treatment of feeding disorders in children with developmental disabilities. Pediatrics 2001, 108, 671–676. [Google Scholar] [CrossRef]
- Motil, K.J.; Schultz, R.J.; Browning, K.; Trautwein, L.; Glaze, D.G. Oropharyngeal dysfunction and gastroesophageal dysmotility are present in girls and women with Rett syndrome. J. Pediatr. Gastroenterol. Nutr. 1999, 29, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Ferrero López, M.I.; De la Rubia Ortí, J.E.; Castellano Vela, E.; González Monte, C.; Sanchis-Bayarri Bernal, V.; Navarro Sanz, R. Factors associated with mortality in patients with dysphagia help in making dietary and nutritional choices. Nutr. Hosp. 2014, 31, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Motsch, C.; Hackelsberger, A.; Nebelung, K. Percutaneous endoscopic gastrostomy in patients with ENT tumors. Hno 1998, 46, 925–931. [Google Scholar] [CrossRef]
- Fujishima, I. Nutritional Management in Home Care: Including Eating Disorder and Dysphagia Assessments. Jpn. Med. Assoc. J. 2015, 58, 31–35. [Google Scholar]

| Broad Category | Subcategory | Number of Articles | Disease, (n) | First Author | Year |
|---|---|---|---|---|---|
| Body mass index | Body mass index | 9 [35,36,41,42,43,44,45,46,47] | Stroke (5), HNC (2), Amyotrophic lateral sclerosis (2), Pulmonary disease (2), Cardiovascular disease (2), Firearm injury (2), Cervical trauma (2), Diabetes mellitus (2), Dyslipidemia (2), Hypertension (2), Machado–Joseph disease (2), Meyge’s syndrome (2), Rubinstein–Taybi syndrome (2), Parkinson’s disease (2), Alzheimer’s disease (2), Dementia, Esophageal cancer, Brain tumor, Myelitis, Huntington’s disease, Progressive supranuclear palsy, Trigeminal neuropathy, Traumatic brain injury, Presbyphagia | da Silva AF, Barni GC | 2020 |
| Maeda K, Ikenaga Y | 2017 | ||||
| Toh Yoon EW, Nakadate A | 2016 | ||||
| Ortega O | 2015 | ||||
| Lecleire S | 2006 | ||||
| Jacobsson C | 1997 | ||||
| Nutritional screening tool | MNA-SF | 4 [39,42,45,48] | Stroke | Nakazawa Y | 2020 |
| Vilardell N, Maeda K | 2017 | ||||
| Ortega O | 2015 | ||||
| Anthropometric measurements | Weight | 5 [37,38,46,49,50] | Esophageal cancer (2), Stroke, HNC, Lung cancer | Kim J | 2018 |
| Smith ZL | 2017 | ||||
| Wang YJ | 2014 | ||||
| Lecleire S | 2006 | ||||
| Elmståhl S | 1999 | ||||
| TSFT | 1 [47] | Stroke, brain tumor | Jacobsson C | 1997 | |
| MAMC | 1 [47] | Stroke, brain tumor | Jacobsson C | 1997 | |
| Body composition | SMM (BIA) | 1 [48] | Nakazawa Y | 2020 | |
| Percent body fat | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Lean body mass | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Dietary assessment | Food intake level | 2 [51,52] | Stroke, Brain trauma, Encephalitis, Central pontine myelinolysis, Neoplasm | Bülow M | 2008 |
| Bartolome G | 1997 | ||||
| Food Frequency Questionnaire | 1 [41] | Parkinson’s disease, Alzheimer’s disease, Huntington’s disease, Amyotrophic lateral sclerosis, Machado–Joseph disease, Meyge’s syndrome, Progressive supranuclear palsy, Stroke, Trigeminal neuropathy, Myelitis, Rubinstein–Taybi syndrome, Firearm injury, Cervical trauma, Presbyphagia, Diabetes mellitus, Hypertension, Dyslipidemia, Cardiovascular disease | Barni GC | 2020 | |
| Energy intake | 1 [42] | Maeda K | 2017 | ||
| Period to meal resumption and dietary form | 1 [53] | Kishimoto N | 2016 | ||
| Daily food intake | 1 [40] | Stroke | Masiero, S | 2008 | |
| Others | MNA | 1 [54] | Alzheimer’s Disease | Tang Y | 2017 |
| O-PNI | 1 [43] | Toh Yoon EW | 2016 |
| Broad Category | Subcategory | Number of Articles | Disease, (n) | First Author | Year |
|---|---|---|---|---|---|
| Blood biomarkers | Albumin | 9 [43,44,46,47,49,50,54,55,56] | Stroke (3), Esophageal cancer (2), Oropharyngeal cancer, Alzheimer’s disease, Gaucher disease, Niemann–Pick disease, High cervical spinal cord injury, Brain tumor | Kimura Y | 2019 |
| Smith ZL, Tang, Y | 2017 | ||||
| Toh Yoon EW, Nakadate A | 2016 | ||||
| Miyake N | 2013 | ||||
| Lecleire S | 2006 | ||||
| Elmståhl S | 1999 | ||||
| Jacobsson C | 1997 | ||||
| Hemoglobin | 1 [54] | Alzheimer’s disease | Tang Y | 2017 | |
| Total protein | 1 [55] | Gaucher disease, Niemann–Pick disease, High cervical spinal cord injury, Oropharyngeal cancer | Miyake N | 2013 | |
| Transferrin | 1 [47] | Stroke, Brain tumor | Jacobsson C | 1997 | |
| Lymphocytes | 1 [56] | Kimura Y | 2019 | ||
| Pre-albumin | 1 [47] | Stroke, Brain tumor | Jacobsson C | 1997 | |
| C-reactive protein | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Ceruloplasmin | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Transthyretin | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Retinol-binding protein | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Total iron-binding capacity | 1 [50] | Stroke | Elmståhl S | 1999 | |
| Orosomucoid | 1 [50] | Stroke | Elmståhl S | 1999 |
| Acute Setting, (n) | Post-Acute Setting, (n) | |
|---|---|---|
| Disease | Parkinson’s Disease, Alzheimer’s Disease, Huntington’s Disease, Amyotrophic Lateral Sclerosis, Stroke, Machado–Joseph Disease, Meige Syndrome, Rubinstein–Taybi Syndrome, Progressive supranuclear palsy, Trigeminal neuropathy, Traumatic brain injury, Firearm Injury, Myelitis, Cervical Trauma, Systemic arterial hypertension, Pneumonia, Diabetes mellitus, Dyslipidemia, Cardiovascular disease, Chronicobstructive pulmonary disease, Presbyphagia, Gaucher disease, Niemann-Pick disease, High cervical spinal cord injury, Oropharyngeal cancer, Head and Neck cancer, Esophageal cancer, Brain tumor, Lung cancer, Nasopharyngeal carcinoma | Stroke, Brain trauma, Encephalitis, Central pontine myelinolysis, Neoplasm, Alzheimer’s Disease |
| Body mass index | Body mass index (6) | Body mass index (3) |
| Nutritional screening tool | MNA-SF (2) | MNA-SF (2) |
| Anthropometric measurements | Weight (5), TSFT (1), MAMC (1) | none |
| Body composition | Percentage body fat (1), Lean body mass (1) | SMM (BIA) (1) |
| Dietary assessment | Food Frequency Questionnaire (1), Energy intake (1), Period to meal resumption and dietary form (1), Daily food intake (1) | Food intake level (2) |
| Blood biomarkers | Albumin (6), Total protein (1), Transferrin (1), Lymphocytes (1), Pre-albumin (1), C-reactive protein (1), Ceruloplasmin (1), Transthyretin (1), Retinol-binding protein (1), TIBC (1), Orosomucoid (1) | Albumin (3), Hemoglobin (1) |
| Others | none | MNA (1), O-PNI (1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueshima, J.; Momosaki, R.; Shimizu, A.; Motokawa, K.; Sonoi, M.; Shirai, Y.; Uno, C.; Kokura, Y.; Shimizu, M.; Nishiyama, A.; et al. Nutritional Assessment in Adult Patients with Dysphagia: A Scoping Review. Nutrients 2021, 13, 778. https://doi.org/10.3390/nu13030778
Ueshima J, Momosaki R, Shimizu A, Motokawa K, Sonoi M, Shirai Y, Uno C, Kokura Y, Shimizu M, Nishiyama A, et al. Nutritional Assessment in Adult Patients with Dysphagia: A Scoping Review. Nutrients. 2021; 13(3):778. https://doi.org/10.3390/nu13030778
Chicago/Turabian StyleUeshima, Junko, Ryo Momosaki, Akio Shimizu, Keiko Motokawa, Mika Sonoi, Yuka Shirai, Chiharu Uno, Yoji Kokura, Midori Shimizu, Ai Nishiyama, and et al. 2021. "Nutritional Assessment in Adult Patients with Dysphagia: A Scoping Review" Nutrients 13, no. 3: 778. https://doi.org/10.3390/nu13030778
APA StyleUeshima, J., Momosaki, R., Shimizu, A., Motokawa, K., Sonoi, M., Shirai, Y., Uno, C., Kokura, Y., Shimizu, M., Nishiyama, A., Moriyama, D., Yamamoto, K., & Sakai, K. (2021). Nutritional Assessment in Adult Patients with Dysphagia: A Scoping Review. Nutrients, 13(3), 778. https://doi.org/10.3390/nu13030778








