A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS
Abstract
1. Introduction
- -
- hyperandrogenism,
- -
- or its clinical manifestations, such as androgenic baldness, hirsutism,
- -
- menstruation and ovulation disorders,
- -
- enlarged (ovary volume > 10 cm3) or polycystic ovaries (at least 12 follicles) visible in an ultrasound image.
2. Material and Methods
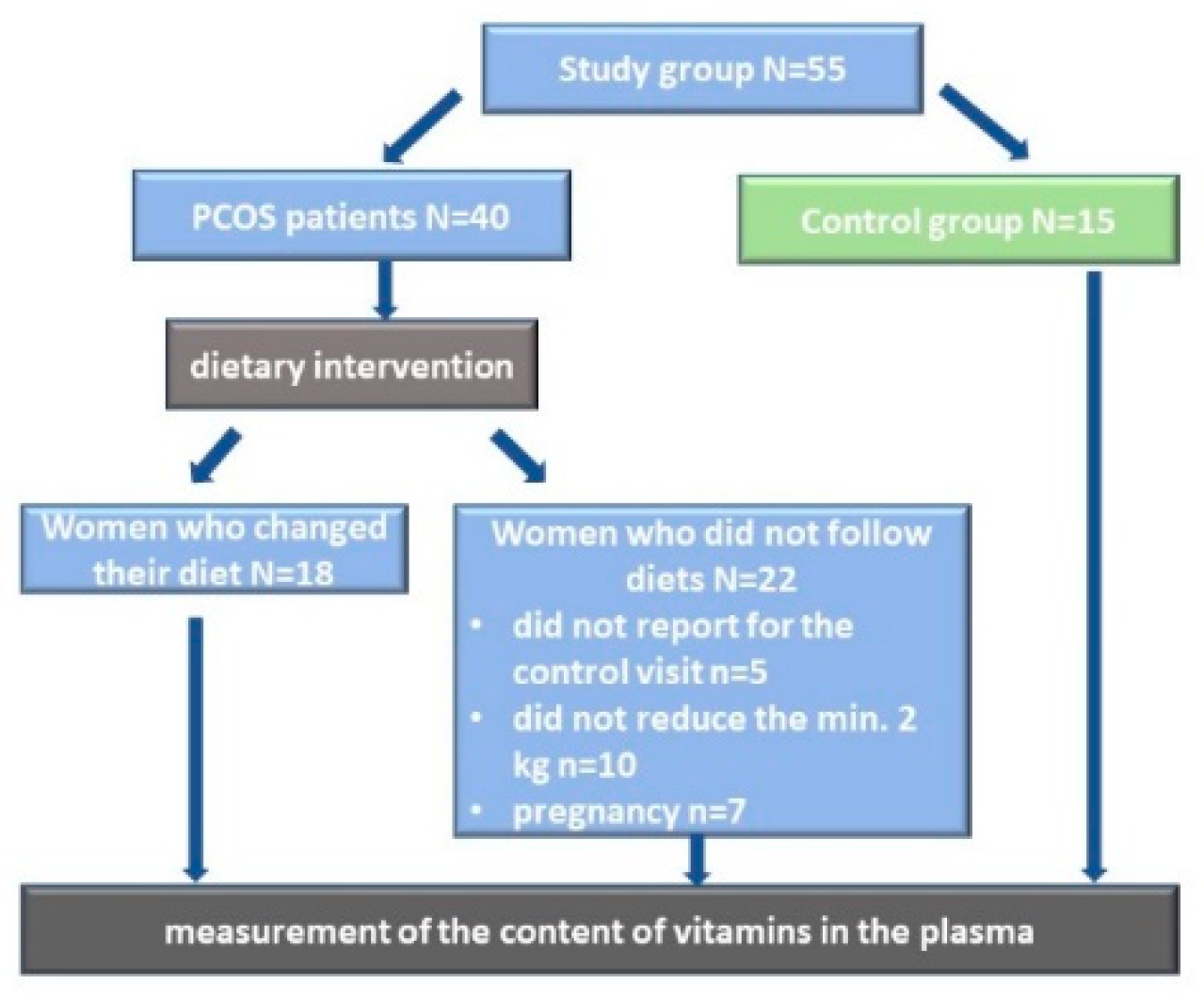
2.1. Study Group
2.2. Quantitative Dietary Assessment
2.3. Dietary Intervention
2.4. Reagents for Biochemical Analyses
2.5. The Analysis of Vitamins Soluble in Water with HPLC
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yildiz, B.O.; Bozdag, G.; Yapici, Z.; Esinler, I.; Yarali, H. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum. Reprod. 2012, 27, 3067–3073. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, E.W.; Tay, C.T.; Hiam, D.S.; Teede, H.J.; Moran, L.J. Comorbidities and complications of polycystic ovary syndrome: An overview of systematic reviews. Clin. Endocrinol. (Oxf.) 2018, 89, 683–699. [Google Scholar] [CrossRef]
- Kim, J.J.; Kim, D.; Yim, J.Y.; Kang, J.H.; Han, K.H.; Kim, S.M.; Choi, Y.M. Polycystic ovary syndrome with hyperandrogenism as a risk factor for non-obese non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2017, 45, 1403–1412. [Google Scholar] [CrossRef]
- Kumarendran, B.; O’Reilly, M.W.; Manolopoulos, K.N.; Toulis, K.A.; Gokhale, K.M.; Sitch, A.J.; Wijeyaratne, C.N.; Coomarasamy, A.; Arlt, W.; Nirantharakumar, K. Polycystic ovary syndrome, androgen excess, and the risk of nonalcoholic fatty liver disease in women: A longitudinal study based on a United Kingdom primary care database. PLoS Med. 2018, 15, e1002542. [Google Scholar] [CrossRef] [PubMed]
- Szczuko, M.; Malarczyk, I.; Zapalowska-Chwyc, M. Improvement in anthropometric parameters after rational dietary intervention in women with polycystic ovary syndrome as the best method to support treatment. Roczniki Państwowego Zakładu Higieny 2017, 68, 409–417. [Google Scholar] [PubMed]
- Greibe, E.; Trolle, B.; Bor, M.V.; Lauszus, F.F.; Nexo, E. Metformin Lowers Serum Cobalamin without Changing Other Markers of Cobalamin Status: A Study on Women with Polycystic Ovary Syndrome. Nutrients 2013, 5, 2475–2482. [Google Scholar] [CrossRef] [PubMed]
- Esmaeilzadeh, S.; Gholinezhad-Chari, M.; Ghadimi, R. The Effect of Metformin Treatment on the Serum Levels of Homocysteine, Folic Acid, and Vitamin B12 in Patients with Polycystic Ovary Syndrome. J. Hum. Reprod. Sci. 2017, 10, 95–101. [Google Scholar]
- Chakraborty, P.; Goswami, S.K.; Rajani, S.; Sharma, S.; Kabir, S.N.; Chakravarty, B.; Jana, K. Recurrent Pregnancy Loss in Polycystic Ovary Syndrome: Role of Hyperhomocysteinemia and Insulin Resistance. PLoS ONE 2013, 8, e64446. [Google Scholar] [CrossRef] [PubMed]
- Szczuko, M.; Skowronek, M.; Zapałowska-Chwyć, M.; Starczewski, A. Quantitative assessment of nutrition in patients with the polycystic ovary syndrome (PCOS). Roczniki Państwowego Zakładu Higieny 2016, 67, 419–426. [Google Scholar]
- Jarosz, M. Normy żywienia dla populacji Polski [Nutritional Standards for the Population of Poland]; Instytut Żywności i Żywienia: Warszawa, Poland, 2017. (In Polish) [Google Scholar]
- Giorgi, M.G.; Howland, K.; Martin, C.; Bonner, A.B. A Novel HPLC Method for the concurrent analysis and quantitation of seven water—Soluble vitamins in biological fluids (plasma and urine): A validation study and application. Sci. World J. 2012, 2012, 359721. [Google Scholar]
- Chatzimichalakis, P.F.; Samanidou, V.F.; Verpoorte, R.; Papadoyannis, I.N. Development of a validated HPLC method for the determination of B-complex vitamins in pharmaceuticals and biological fluids after solid phase extraction. J. Sep. Sci. 2004, 27, 1181–1188. [Google Scholar] [CrossRef]
- Siji, J. Analysis of Water—Soluble Vitamins from Multivitamin Tablets for Nutrition Labeling; Agilent Application Note; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2011; Publication Number 5990-7950EN. [Google Scholar]
- Szczuko, M.; Hawryłkowicz, V.; Kikut, J.; Drozd, A. The implications of vitamin content in the plasma in reference to the parameters of carbohydrate metabolism and hormone and lipid profiles in PCOS. J. Steroid. Biochem. Mol. Biol. 2020, 198, 105570. [Google Scholar] [CrossRef] [PubMed]
- Olofinnade, A.T.; Onaolapo, A.Y.; Stefanucci, A.; Mollica, A.; Olowe, O.A.; Onaolapo, O.J. Front Cucumeropsis mannii reverses high-fat diet induced metabolic derangement and oxidative stress. Front. Biosci. (Elite Ed.) 2021, 13, 54–76. [Google Scholar]
- Okon, U.A.; Utuk, I.I. Ascorbic acid treatment elevates follicle stimulating hormone and testosterone plasma levels and enhances sperm quality in albino Wistar rats. Niger. Med. J. 2016, 57, 31–36. [Google Scholar] [CrossRef]
- Tu, H.; Li, H.; Wang, Y.; Niyyati, M.; Wang, Y.; Leshin, J.; Levine, M. Low red blood cel vitamin C concentrations induce red blood cell fragility: A link to diabetes via glucose, glucose transporters, and dehydroascorbic acid. EBioMedicine 2015, 2, 1735–1750. [Google Scholar] [CrossRef]
- Szczuko, M.; Zapałowska-Chwyć, M.; Maciejewska, D.; Drozd, A.; Starczewski, A.; Stachowska, E. Significant improvement selected mediators of inflammation in phenotypes of women with PCOS after reduction and low GI diet. Mediat. Inflamm. 2017, 7, 5489523. [Google Scholar] [CrossRef]
- Szczuko, M.; Zapałowska-Chwyć, M.; Drozd, A.; Maciejewska, D.; Starczewski, A.; Stachowska, E. Effect of IGF-I and TNF-alpha on intensification of steroid pathways in women with PCOS phenotypes are not identical. Enhancement of progesterone pathway in women with PCOS increases the concentration of TNF-alpha. Gynecol. Endocrinol. 2016, 32, 714–717. [Google Scholar] [CrossRef]
- Suzuki, H.; Kunisawa, J. Vitamin-mediated immune regulation in the development of inflammatory diseases. Endocr. Metab. Immune Disord. Drug Targets 2015, 15, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Tangvarasittichai, S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J. Diabetes 2015, 6, 456–480. [Google Scholar] [CrossRef]
- Wanders, D.; Graff, E.C.; White, B.D.; Judd, R.L. Niacin Increases Adiponectin and Decreases Adipose Tissue Inflammation in High Fat Diet-Fed Mice. PLoS ONE 2013, 8, e71285. [Google Scholar] [CrossRef] [PubMed]
- Lavigne, P.M.; Karas, R.H. The current state of niacin in cardiovascular disease prevention. J. Am. Coll. Cardiol. 2013, 61, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Xiang, D.; Zhang, Q.; Wang, Y.T. Effectiveness of niacin supplementation for patients with type 2 diabetes: A meta-analysis of randomized controlled trials. Medicine 2020, 99, e21235. [Google Scholar] [CrossRef] [PubMed]
- Lyubarova, R.; Albers, J.J.; Marcovina, S.M.; Yao, Y.; McBride, R.; Topliceanu, A.; Anderson, T.; Fleg, J.L.; Desvigne-Nickens, P.; Kashyap, M.L.; et al. Effects of Extended-Release Niacin on Quartile Lp-PLA2 Levels and Clinical Outcomes in Statin-treated Patients with Established Cardiovascular Disease and Low Baseline Levels of HDL-Cholesterol: Post Hoc Analysis of the AIM HIGH Trial. J. Cardiovasc. Pharmacol. Ther. 2019, 24, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Nejabati, H.R.; Samadi, N.; Shahnazi, V.; Mihanfar, A.; Fattahi, A.; Latifi, Z.; Bahrami-Asl, Z.; Roshangar, L.; Nouri, M. Nicotinamide and its metabolite N1-Methylnicotinamide alleviate endocrine and metabolic abnormalities in adipose and ovarian tissues in rat model of Polycystic Ovary Syndrome. Chem. Biol. Interact. 2020, 324, 109093. [Google Scholar] [CrossRef]
- Nejabati, H.R.; Schmeisser, K.; Shahnazi, V.; Samimifar, D.; Faridvand, Y.; Bahrami-Asl, Z.; Fathi-Maroufi, N.; Nikanfar, S.; Nouri, M. N1-Methylnicotinamide: An Anti-Ovarian Aging Hormetin? Ageing Res. Rev. 2020, 62, 101131. [Google Scholar] [CrossRef]
- Mogielnicki, A.; Kramkowski, K.; Buczko, W. Nicotinamide and its metabolite—n-methylnicotinamide—merely a vitamin or a new type of medicine to help in the struggle with circulatory system and homeostasis diseases? Kardiol. Pol. 2008, 66 (Suppl. 3), 341–346. [Google Scholar]
- Feng, W.; Zhang, Y.; Pan, Y.; Zhang, Y.; Liu, M.; Huang, Y.; Xiao, Y.; Mo, W.; Jiao, J.; Wang, X.; et al. Association of three missense mutations in the homocysteine-related MTHFR and MTRR gene with risk of polycystic ovary syndrome in Southern Chinese women. Reprod. Biol. Endocrinol. 2021, 19, 5. [Google Scholar] [CrossRef]
- Xiong, Y.; Bian, C.; Lin, X.; Wang, X.; Xu, K.; Zhao, X. Methylenetetrahydrofolate reductase gene polymorphisms in the risk of polycystic ovary syndrome and ovarian cancer. Biosci. Rep. 2020, 40, BSR20200995. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; Liu, J.; O’Keefe, J.H. Thiamine and cardiovascular disease: A literature review. Prog. Cardiovasc. Dis. 2018, 61, 27–32. [Google Scholar] [CrossRef]
- Eshak, E.S.; Arafa, A.E. Thiamine deficiency and cardiovascular disorders. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 965–972. [Google Scholar] [CrossRef]
- Jones, M.L.; Buhimschi, I.A.; Zhao, G.; Bartholomew, A.; Smith-Timms, J.; Rood, K.M.; Buhimschi, C.S. Acute Glucose Load, Inflammation, Oxidative Stress, Nonenzymatic Glycation, and Screening for Gestational Diabetes. Reprod. Sci. 2019, 1587–1594. [Google Scholar] [CrossRef]
- Sakasai-Sakai, A.; Takata, T.; Takino, J.I.; Takeuchi, M. The Relevance of Toxic AGEs (TAGE) Cytotoxicity to NASH Pathogenesis: A Mini-Review. Nutrients 2019, 11, 462. [Google Scholar] [CrossRef]
- Xie, F.; Cheng, Z.; Li, S.; Liu, X.; Guo, X.; Yu, P.; Gu, Z. Pharmacokinetic study of benfotiamine and the bioavailability assessment compared to thiamine hydrochloride. J. Clin. Pharmacol. 2014, 54, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Szczuko, M.; Zapałowska-Chwyć, M.; Drozd, A.; Maciejewska, D.; Starczewski, A.; Stachowska, E. Metabolic pathways of oleic and palmitic acid are intensified in PCOS patients with normal androgen levels. Prostaglandins Leukot. Essent. Fat. Acids (PLEFA) 2017, 126, 105–112. [Google Scholar] [CrossRef]
- Pourghasem, S.; Bazarganipour, F.; Taghavi, S.A.; Kutenaee, M.A. The effectiveness of inositol and metformin on infertile polycystic ovary syndrome women with resistant to letrozole. Arch Gynecol. Obstet. 2019, 299, 1193–1199. [Google Scholar] [CrossRef]
- Raj, V.; Ojha, S.; Howarth, F.C.; Belur, P.D.; Subramanya, S.B. Therapeutic potential of benfotiamine and its molecular targets. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 3261–3273. [Google Scholar]
- Majewska, M.; Kozanecka, P.; Szałek, E. Benfotiamine effects in pharmacotherapy of diabetic complications. Farm. Współcz. 2019, 12, 151–157. [Google Scholar]
- Schiuma, N.; Costantino, A.; Bartolotti, T.; Dattilo, M.; Bini, V.; Aglietti, M.C.; Renga, M.; Favilli, A.; Falorni, A.; Gerli, S. Micronutrients in support to the one carbon cycle for the modulation of blood fasting homocysteine in PCOS women. J. Endocrinol. Investig. 2020, 43, 779–786. [Google Scholar] [CrossRef]
- Montanino Oliva, M.; Zuev, V.; Lippa, A.; Carra, M.C.; Lisi, F. Efficacy of the synergic action of myoinositol, tyrosine, selenium and chromium in women with PCOS. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8687–8694. [Google Scholar] [CrossRef] [PubMed]
- Le Donne, M.; Metro, D.; Alibrandi, A.; Papa, M.; Benvenga, S. Effects of three treatment modalities (diet, myoinositol or myoinositol associated with D-chiro-inositol) on clinical and body composition outcomes in women with polycystic ovary syndrome. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 2293–2301. [Google Scholar] [CrossRef]
- Gerli, S.; Papaleo, E.; Ferrari, A.; Di Renzo, G.C. Randomized, double blind placebo-controlled trial: Effects of myo-inositol on ovarian function and metabolic factors in women with PCOS. Eur. Rev. Med. Pharmacol. Sci. 2007, 11, 347–354. [Google Scholar]
- Genazzani, A.D.; Prati, A.; Santagni, S.; Ricchieri, F.; Chierchia, E.; Rattighieri, E.; Campedelli, A.; Simoncini, T.; Artini, P.G. Diferential insulin response to myo-onositol administration on obese polycystic ovary syndrome patients. Gynecol. Endocrinol. 2012, 28, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Stracquadanio, M.; Ciotta, L.; Palumbo, M.A. Effects of myo-inositol, gymnemic acid, and L-methylfolate in polycystic ovary syndrome patients. Gynecol. Endocrinol. 2018, 34, 495–501. [Google Scholar] [CrossRef]
- Salehpour, S.; Nazari, L.; Hoseini, S.; Saharkhiz, N.; Ghazi, F.; Sohrabi, M.R. A Potential Therapeutic Role of Myoinositol in the Metabolic and Cardiovascular Profile of PCOS Iranian Women Aged between 30 and 40 Years. Int. J. Endocrinol. 2016, 2016, 7493147. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, F.; Karamali, M.; Shakeri, H.; Asemi, Z. The effects of folate supplementation on inflammatory factors and biomarkers of oxidative stress in overweight and obese women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled clinical trial. Clin. Endocrinol. (Oxf.) 2014, 81, 582–587. [Google Scholar] [CrossRef] [PubMed]
| Parameter | PCOS Patients | Control Group (CG) | p |
|---|---|---|---|
| Age (year) | 32.52 ± 7.12 | 30.23 ± 6.31 | NS |
| Height (m) | 1.67 ± 0.06 | 1.68 ± 0.06 | NS |
| Body mass (kg) | 82.75 ± 15.6 | 62.76 ± 6.67 | 1 × 10−6 |
| BMI (kg/m2) | 29.65 ± 6.76 | 22.22 ± 1.52 | 1 × 10−6 |
| Fat mass (%) | 39.54 ± 8.08 | 25.65 ± 3.96 | 1 × 10−6 |
| Waist circumference (cm) | 99.87 ± 15.65 | 74.75 ± 5.01 | 1 × 10−6 |
| Hip circumference (cm) | 109.45 ± 8.96 | 95.85 ± 4.88 | 1 × 10−6 |
| WHR | 0.92 ± 0.08 | 0.78 ± 0.03 * | 1 × 10−6 |
| Vitamins | PCOS-I N = 40 | PCOS-II N = 18 | CG N = 15 | P PCOS-I vs. PCOS-II | P PCOS-I vs. CG | P PCOS-II vs. CG |
|---|---|---|---|---|---|---|
| C (mg) | 68.53 ± 38.22 | 234.61 ± 87.3 | 101.37 ± 73.04 | 1 × 10−6 | 0.017 | 1 × 10−5 |
| B1 (mg) | 1.21 ± 0.33 | 1.64 ± 0.29 | 1.12 ± 0.32 | 1 × 10−6 | 0.195 | 1 × 10−5 |
| B2 (mg) | 1.33 ± 0.29 | 1.76 ± 0.59 | 1.41 ± 0.39 | 0.0004 | 0.564 | 0.041 |
| niacin (mg) | 15.42 ± 4.18 | 21.2 ± 4.65 | 15.06 ± 4.58 | 1 × 10−6 | 0.771 | 0.0001 |
| B6 (mg) | 2.08 ± 0.63 | 2.64 ± 0.78 | 2.16 ± 0.91 | 0.002 | 0.718 | 0.049 |
| Folates (μg) | 221.2 ± 65.4 | 321.3 ± 72.3 | 262.8 ± 78.56 | 1 × 10−5 | 0.041 | 0.074 * |
| B12 (μg) | 3.30 ± 2.13 | 3.72 ± 1.05 | 3.03 ± 2.34 | 0.323 | 0.651 | 0.168 |
| Vitamin [µg/mL] | PCOS-I N = 40 | PCOS-II N = 18 | CG N = 15 | P PCOS-I vs. PCOS-II | P PCOS-I vs. CG | P PCOS-II vs. CG |
|---|---|---|---|---|---|---|
| C | 1.032 ± 1.236 | 1.006 ± 0.581 | 0.667 ± 0.115 | 0.287 | 0.043 | 0.050 |
| B1 | 0.256 ± 0.275 | 0.336 ± 0.282 | 0.560 ± 0.416 | 0.241 | 0.023 | 0.095 * |
| B2 | 0.004 ± 0.002 | 0.005 ± 0.002 | 0.006 ± 0.004 | 0.279 | 0.112 | 0.428 |
| B3—nicotinic acid | 0.496 ± 0.449 | 0.153 ± 0.367 | 0.062 ± 0.050 | 0.018 | 0.001 | 0.370 |
| PP—niacinamide | 0.721 ± 0.212 | 0.756 ± 0.163 | 0.849 ± 0.201 | 0.418 | 0.173 | 0.292 |
| B5 | 0.479 ± 0.230 | 0.606 ± 0.191 | 0.722 ± 0.192 | 0.126 | 0.002 | 0.116 |
| B6 | 0.568 ± 0.283 | 0.758 ± 0.240 | 0.809 ± 0.217 | 0.046 | 0.042 | 0.770 |
| B7 | 0.253 ± 0.321 | 0.262 ± 0.169 | 0.264 ± 0.256 | 0.189 | 0.674 | 0.419 |
| B9 | 2.016 ± 0.465 | 2.130 ± 0.441 | 2.480 ± 0.616 | 0.385 | 0.035 | 0.083 * |
| B12 | 0.056 ± 0.029 | 0.072 ± 0.073 | 0.080 ± 0.055 | 0.386 | 0.064 | 0.559 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szczuko, M.; Szydłowska, I.; Nawrocka-Rutkowska, J. A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS. Nutrients 2021, 13, 746. https://doi.org/10.3390/nu13030746
Szczuko M, Szydłowska I, Nawrocka-Rutkowska J. A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS. Nutrients. 2021; 13(3):746. https://doi.org/10.3390/nu13030746
Chicago/Turabian StyleSzczuko, Małgorzata, Iwona Szydłowska, and Jolanta Nawrocka-Rutkowska. 2021. "A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS" Nutrients 13, no. 3: 746. https://doi.org/10.3390/nu13030746
APA StyleSzczuko, M., Szydłowska, I., & Nawrocka-Rutkowska, J. (2021). A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS. Nutrients, 13(3), 746. https://doi.org/10.3390/nu13030746








