Paleolithic Diet—Effect on the Health Status and Performance of Athletes?
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligiblity Criteria
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
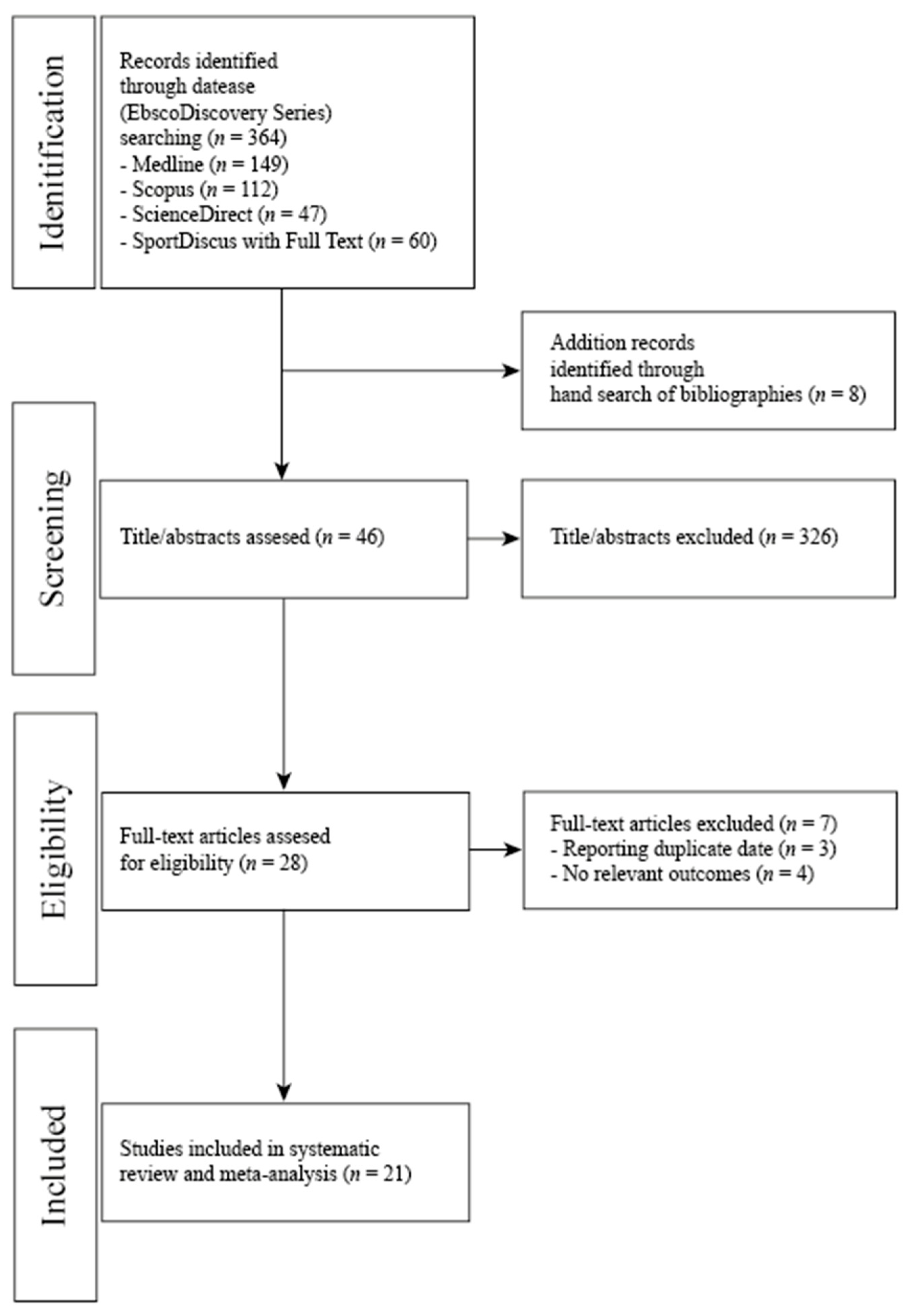
3.1. Characteristics of Included Studies and Search Results
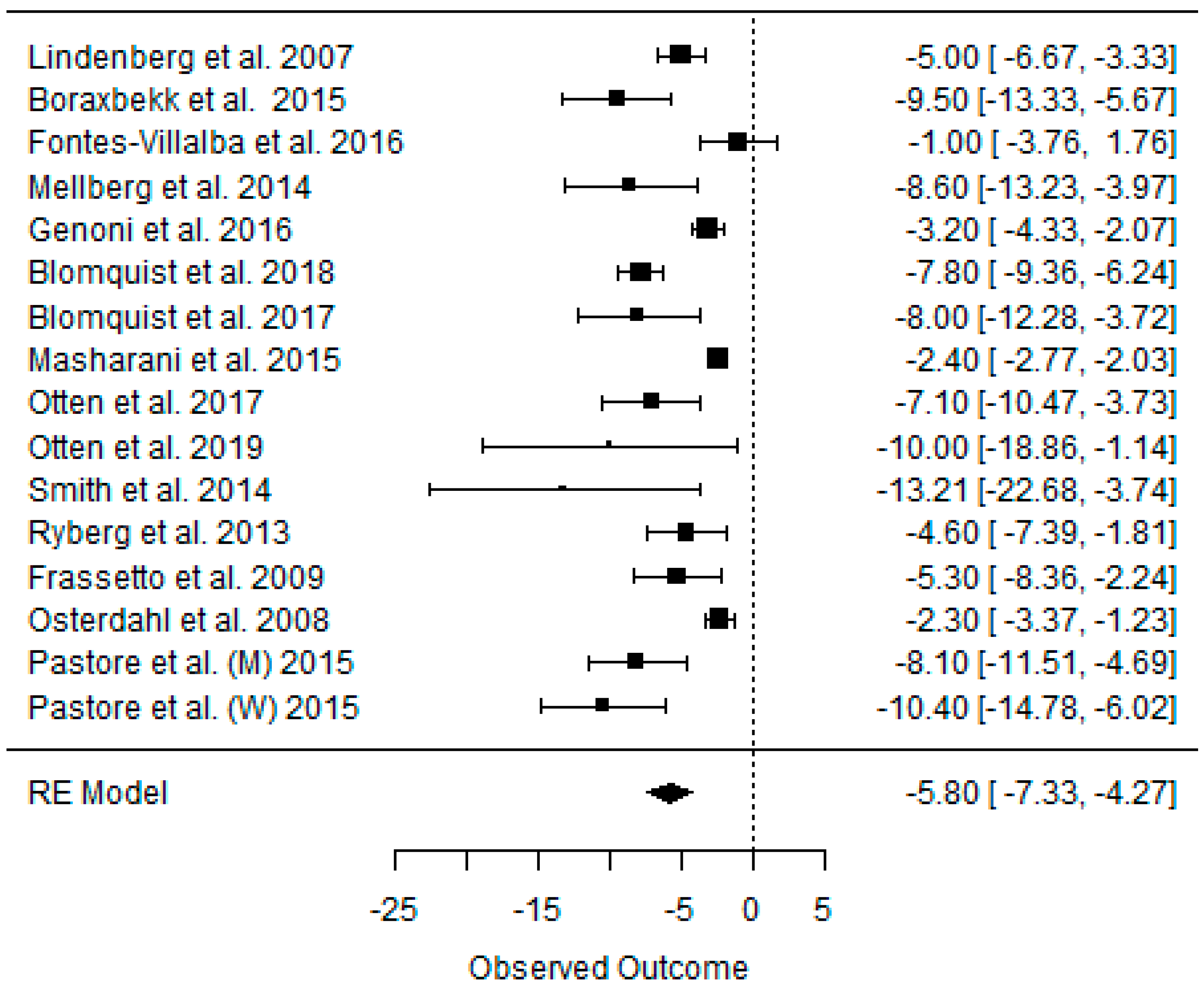
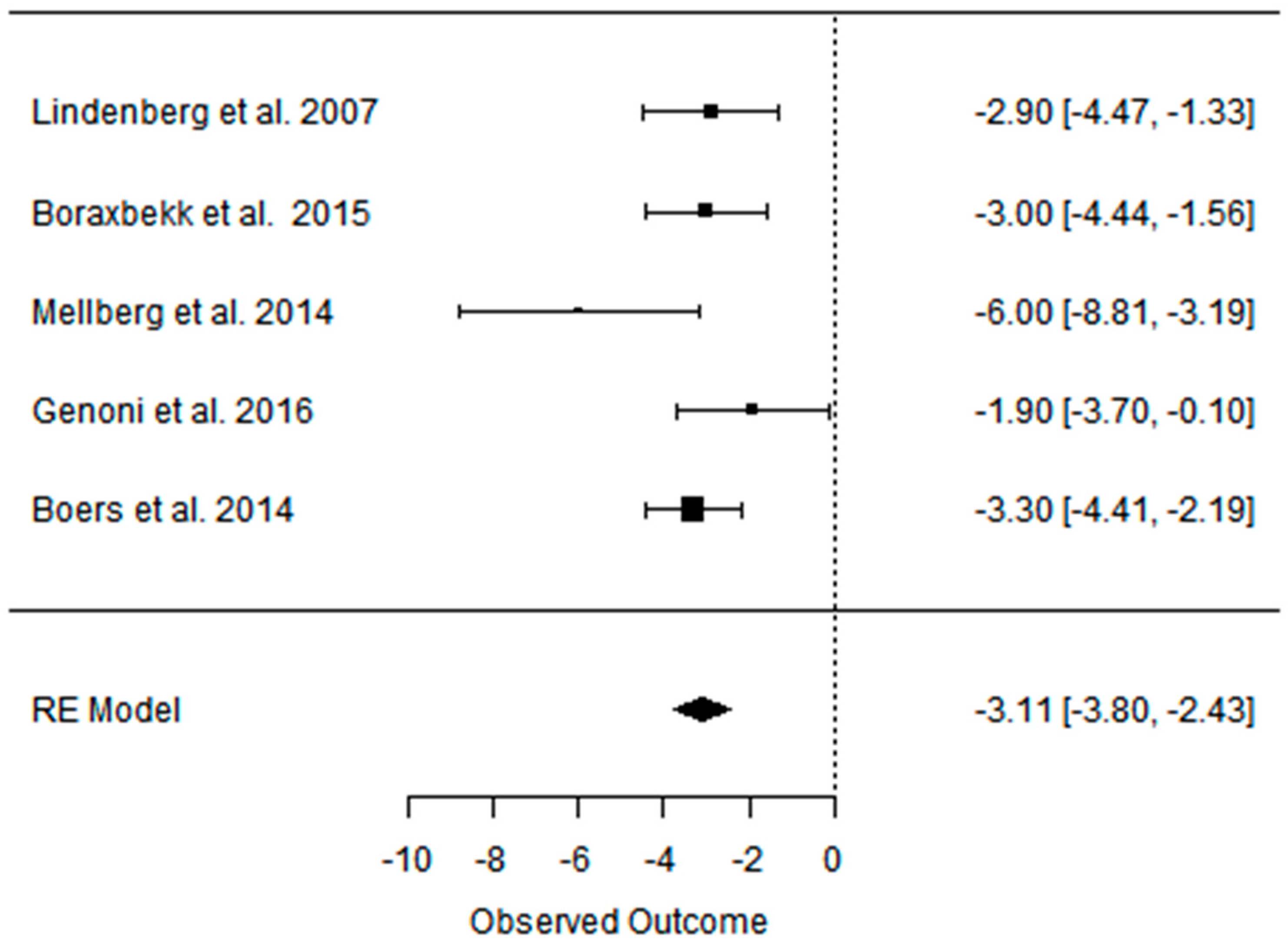
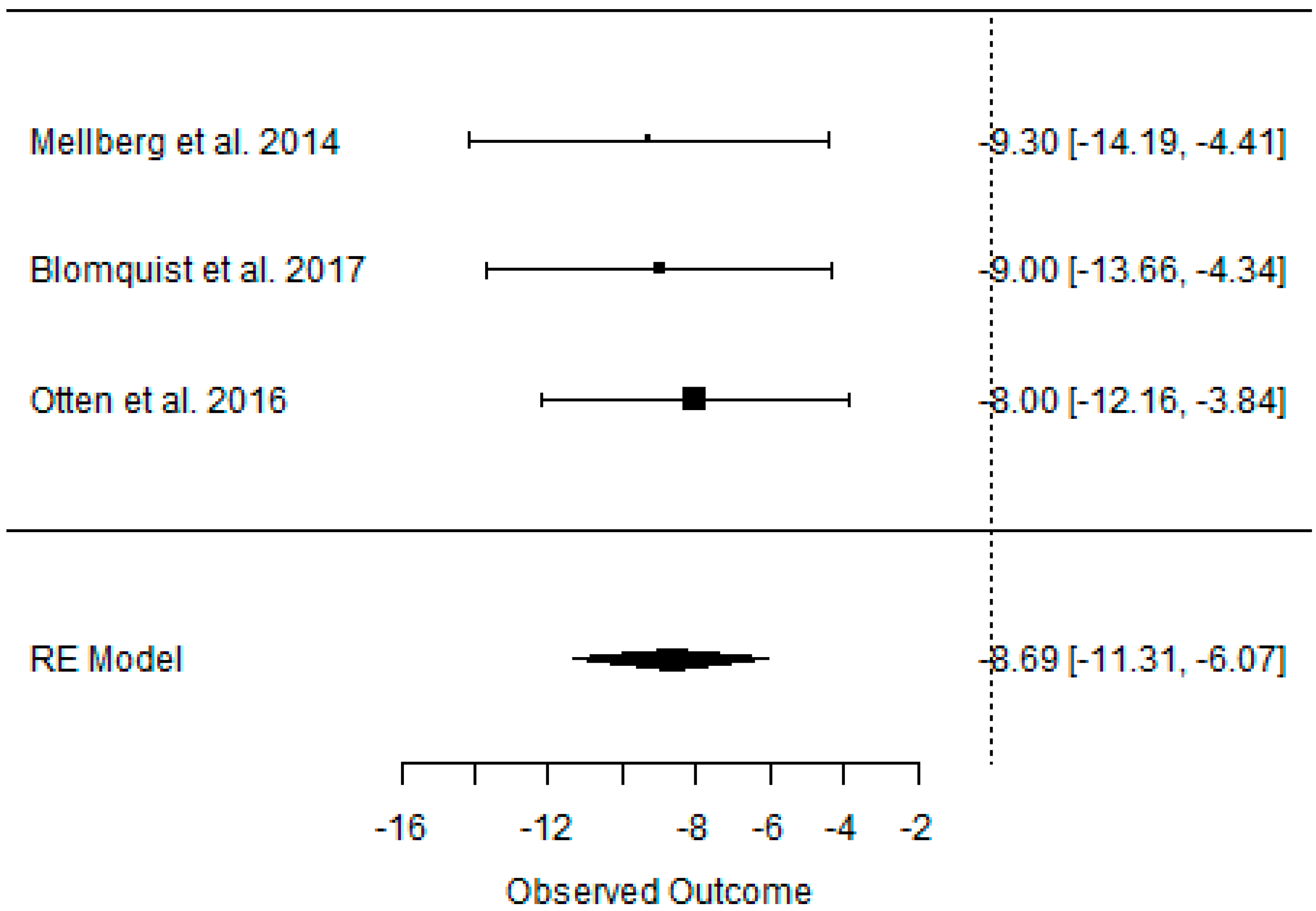
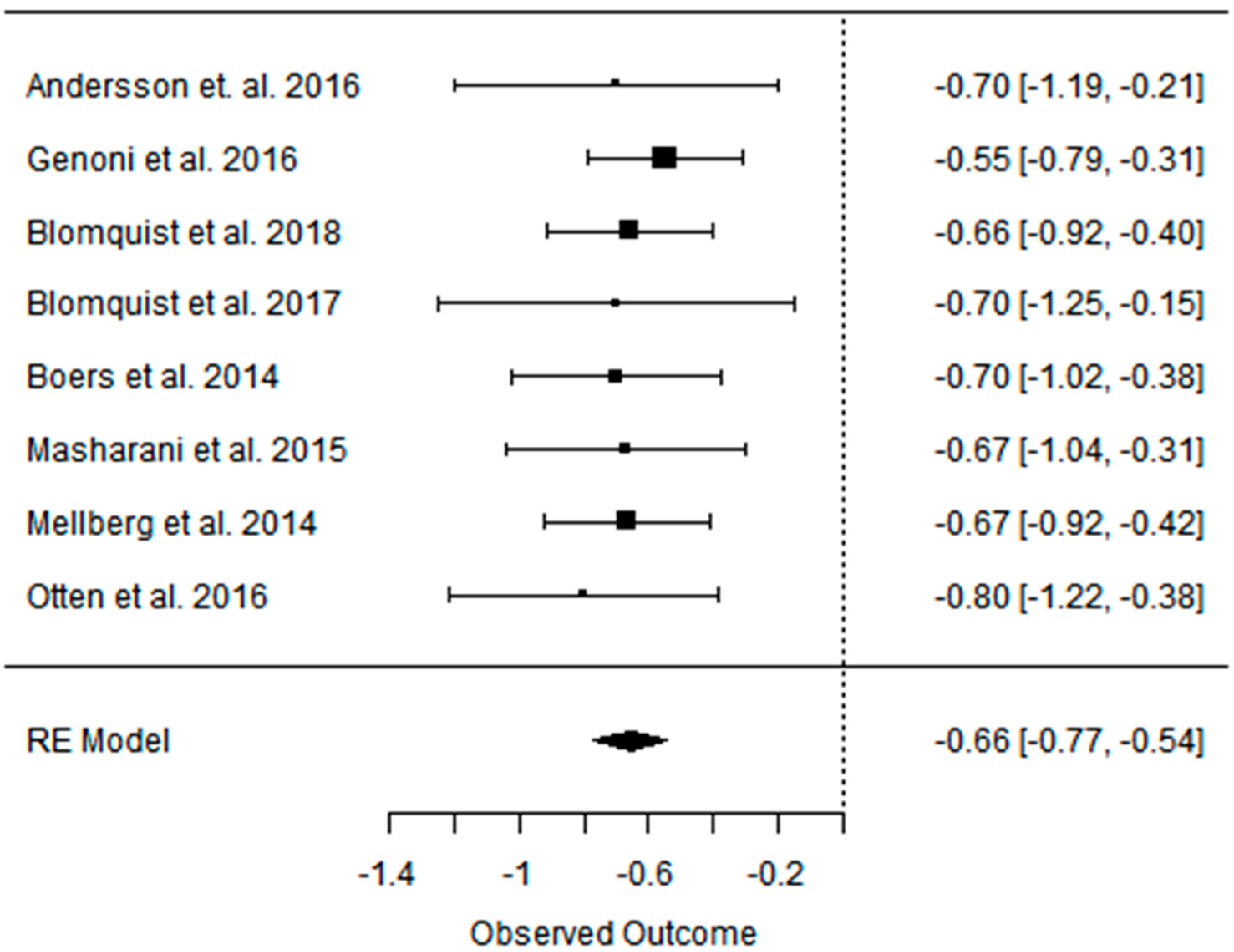
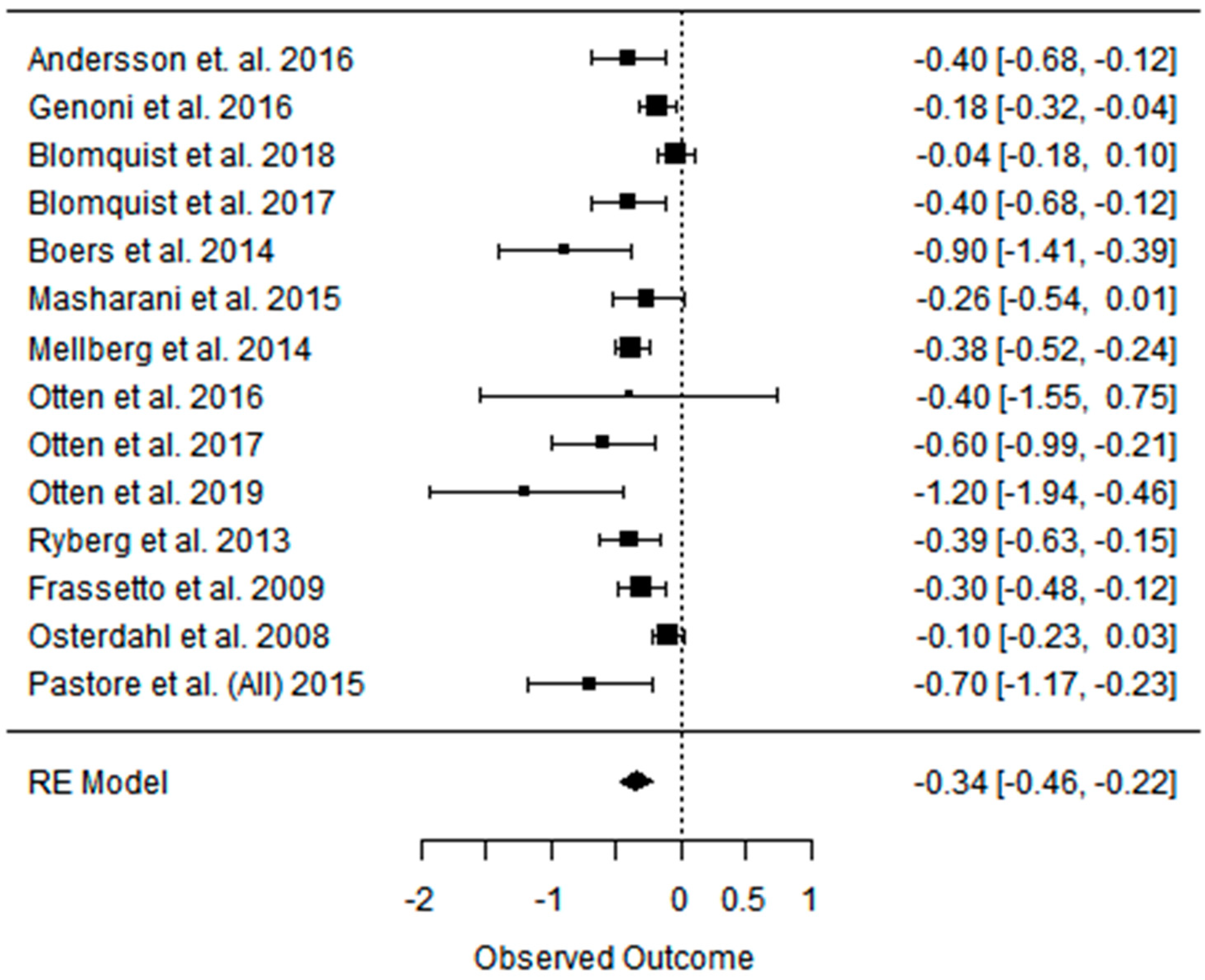
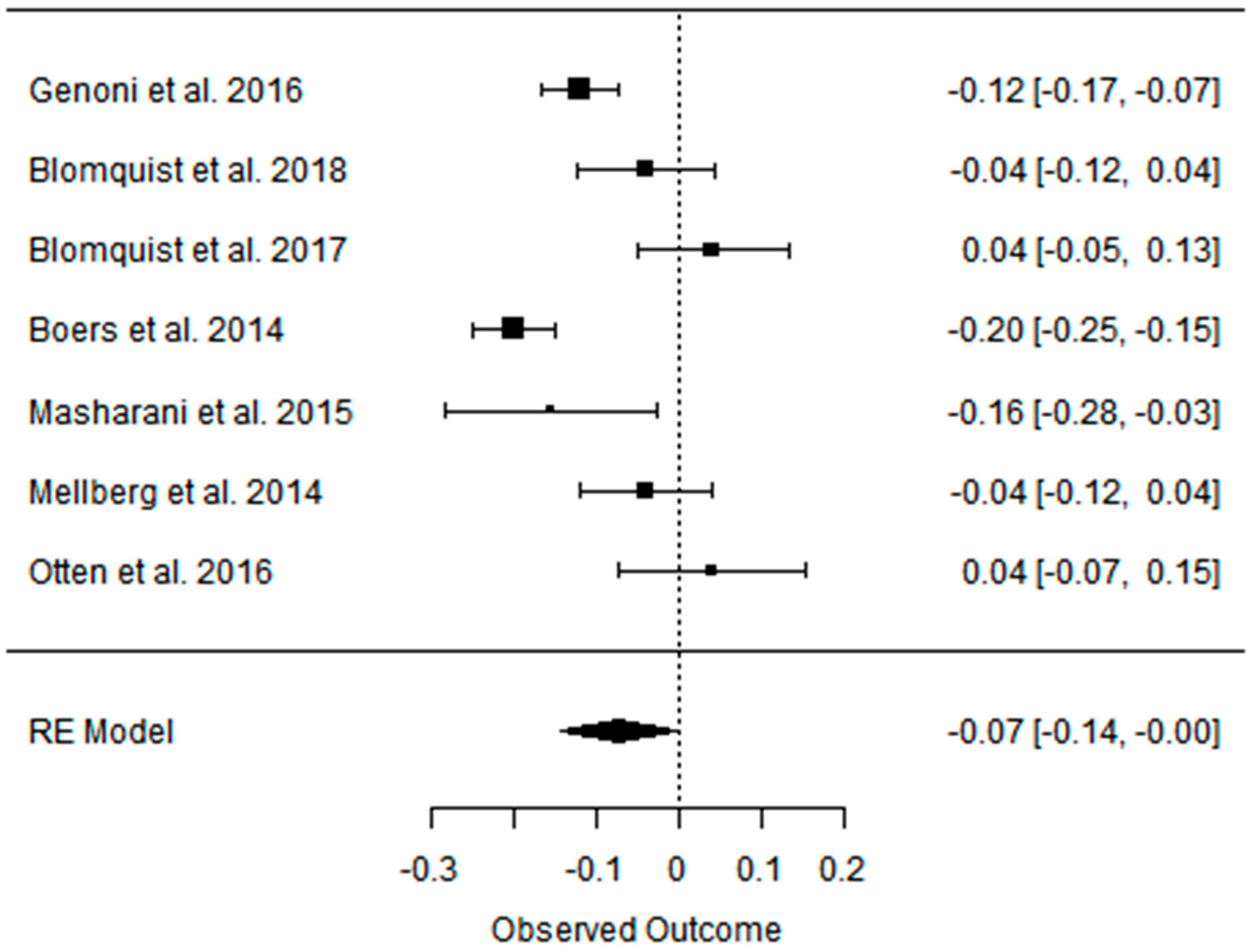
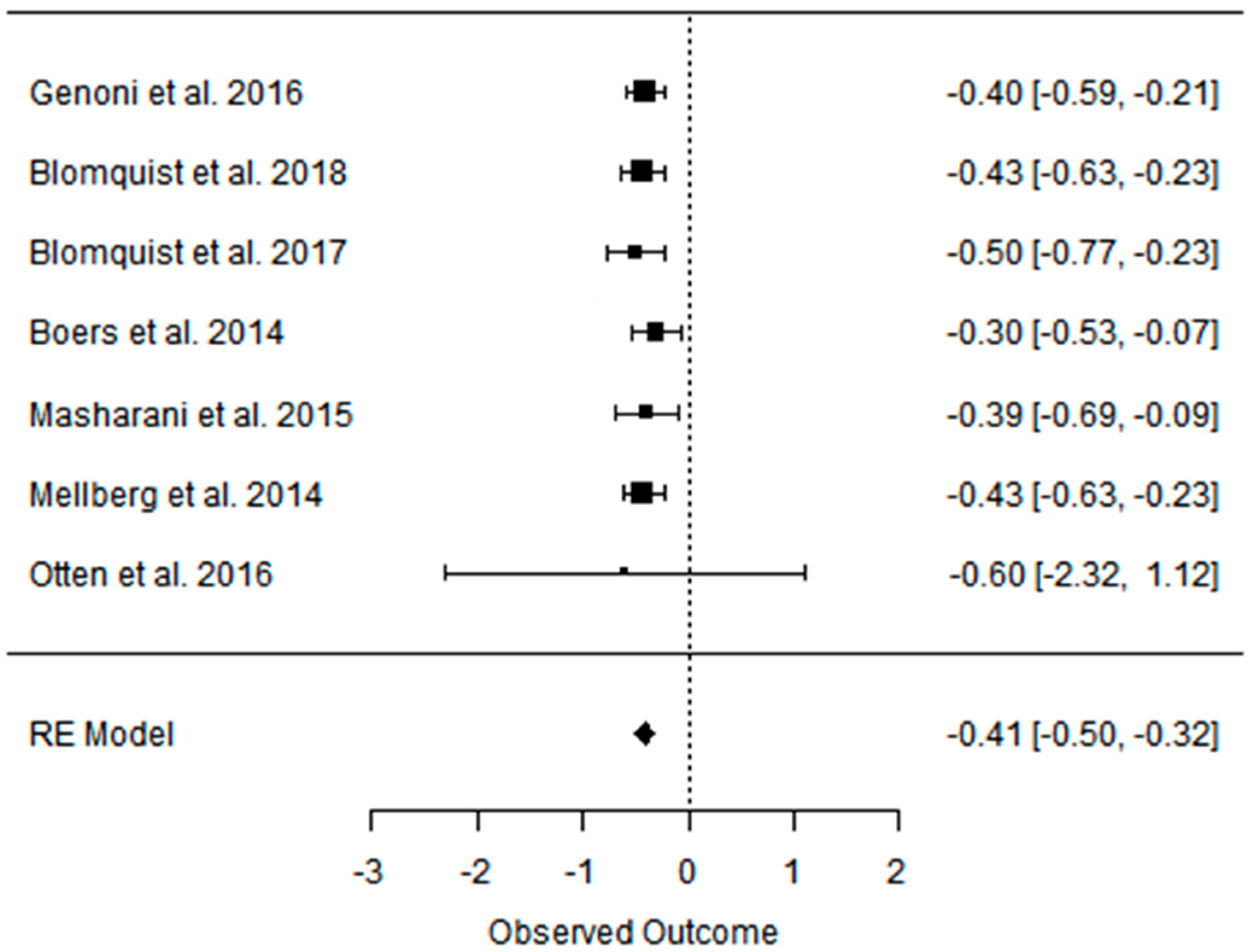
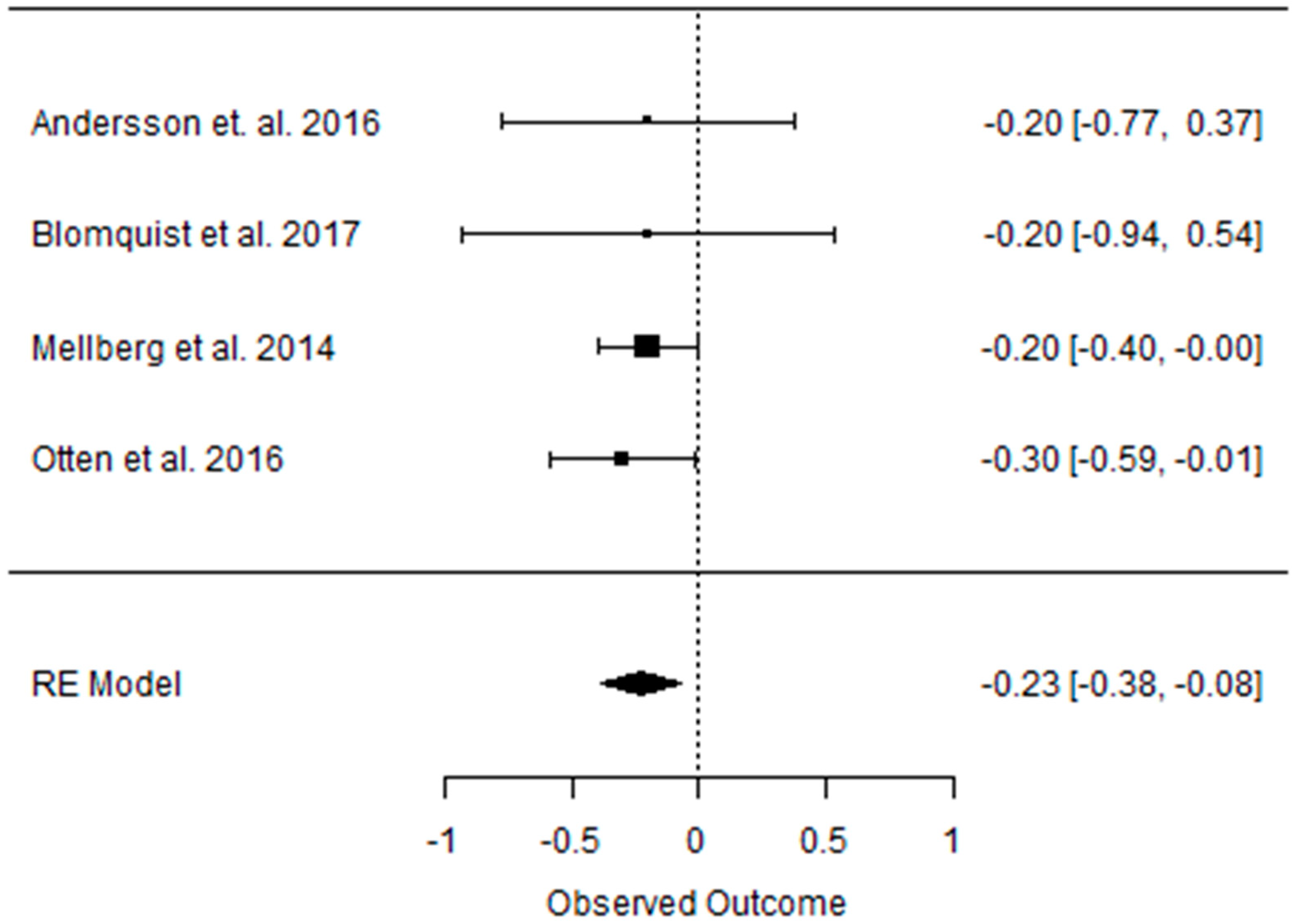
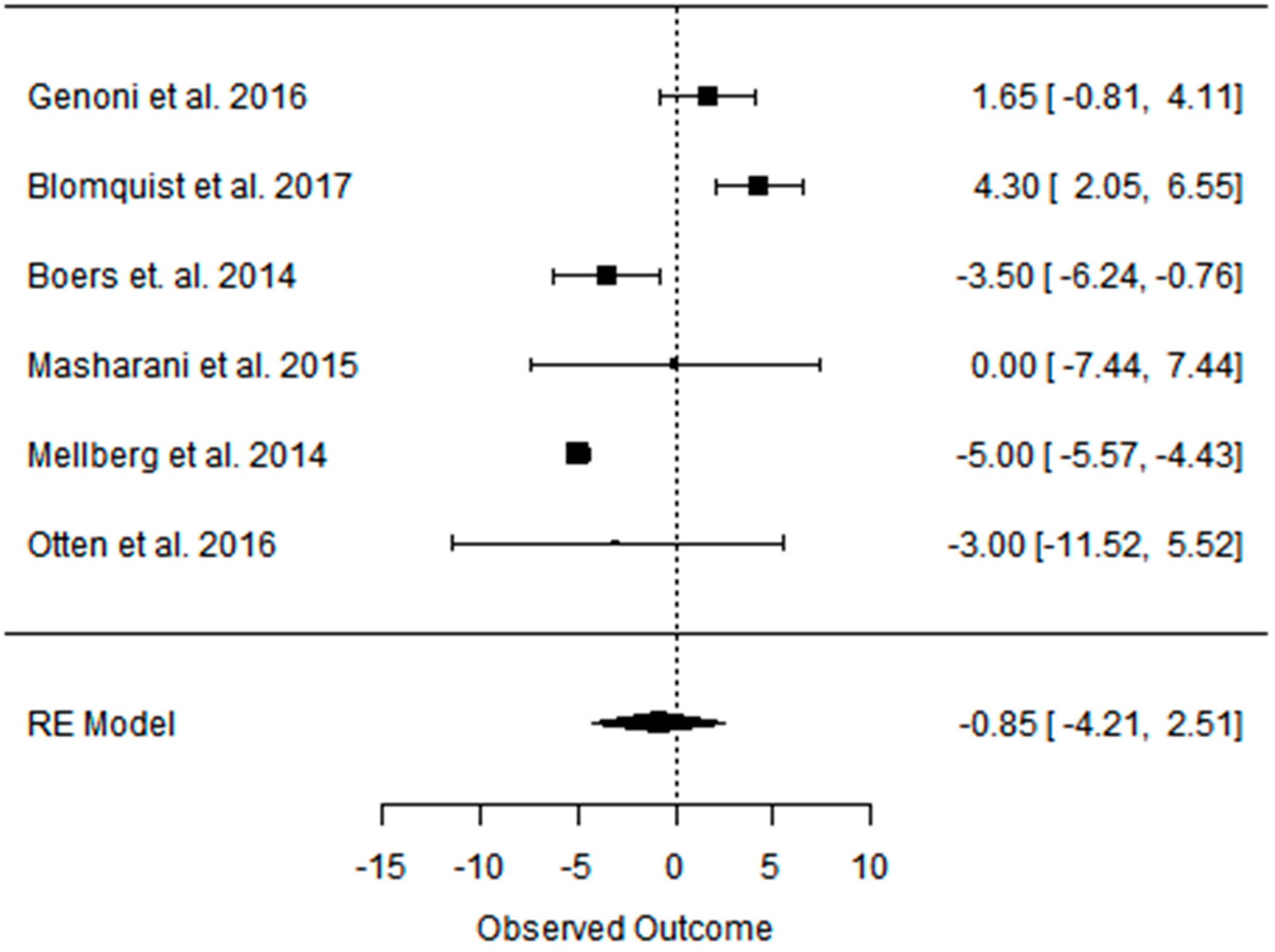
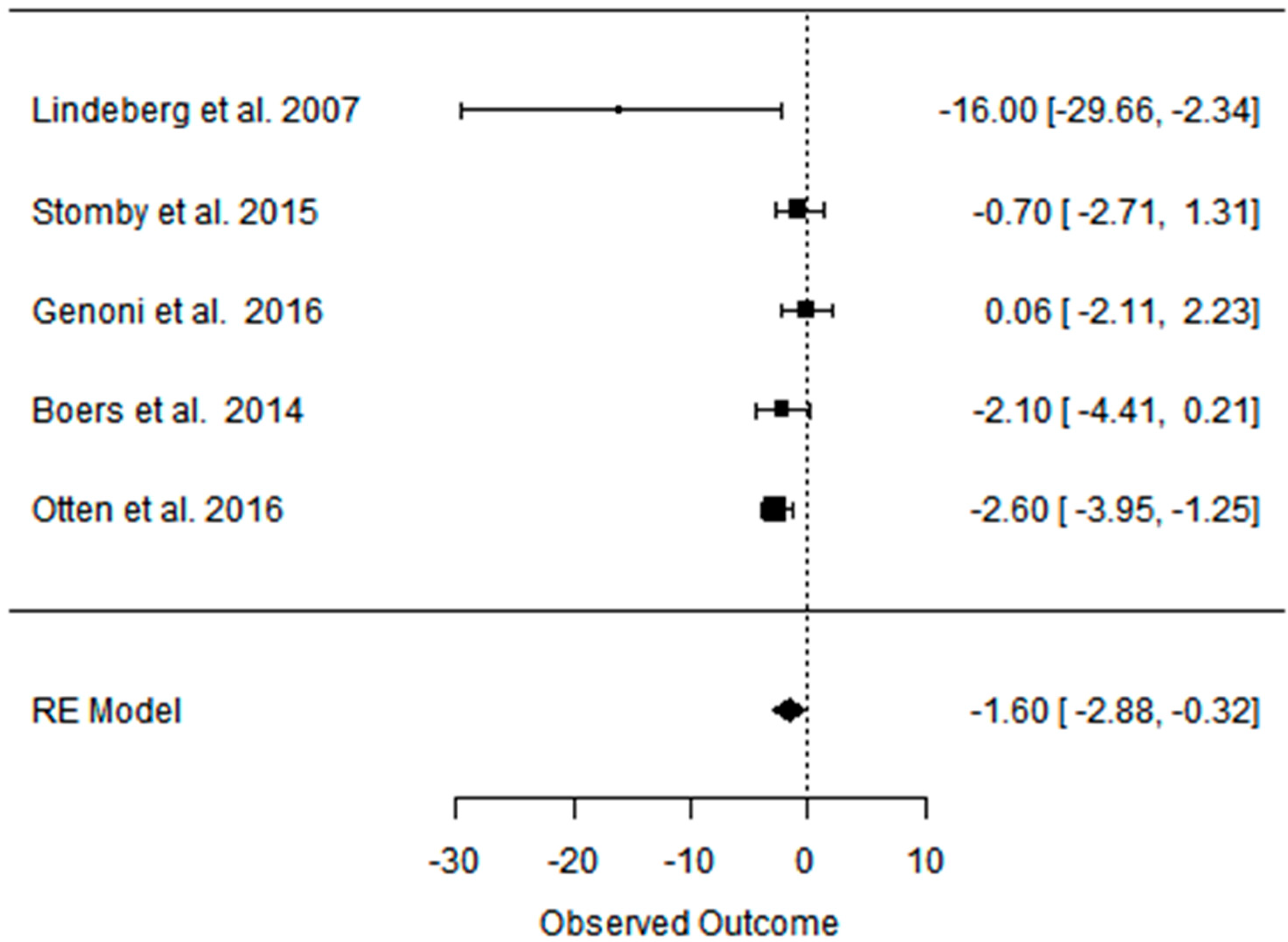
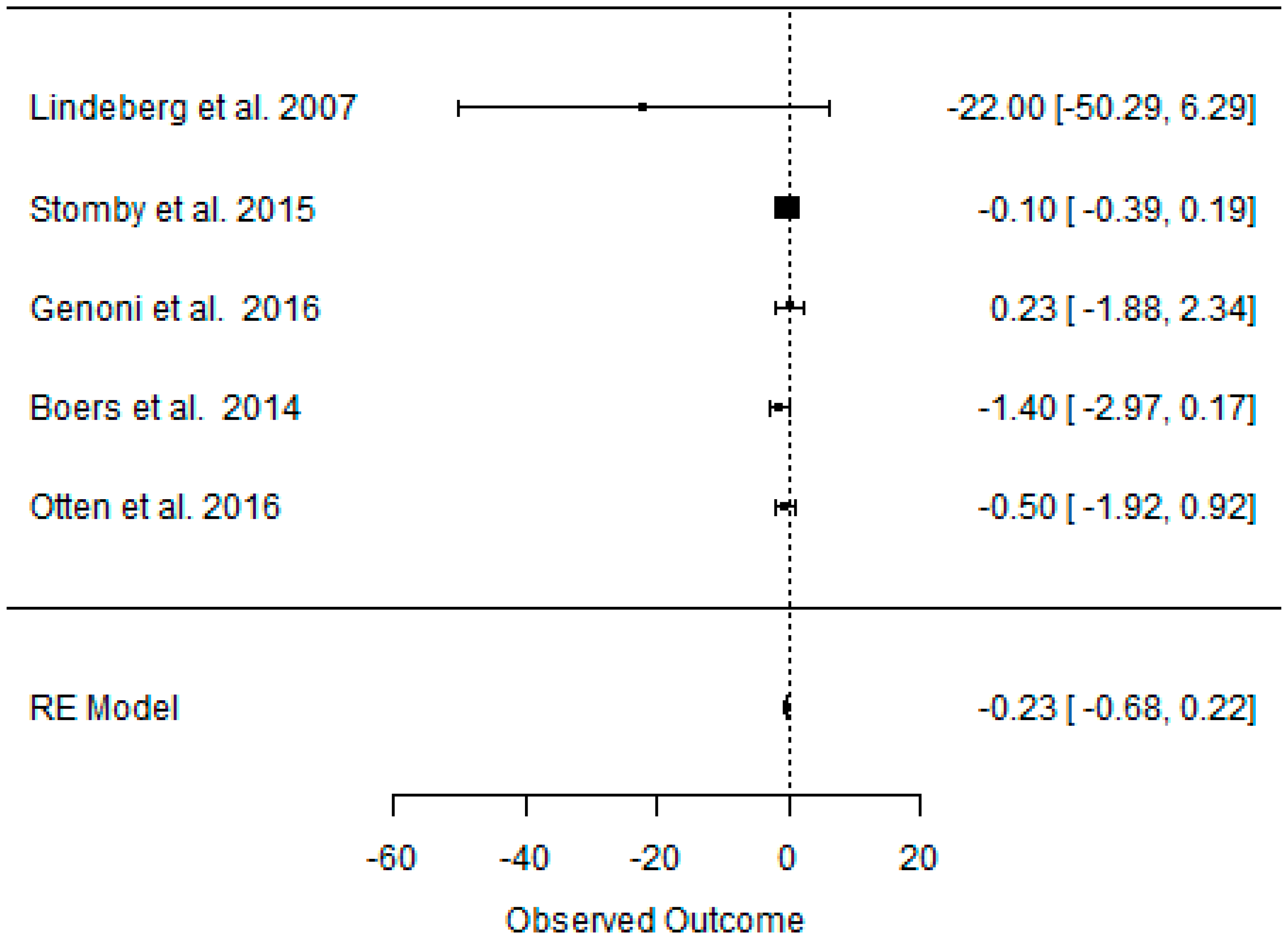
3.2. Effect of the Paleolithic Diet on Body Composition—Anthropometric Parameters (Body Mass, Body Mass Index, Waist Circumference, Fat Mass, and Fat Free Mass) in the Short (up to 6 Months) and Long (over 6 Months) Term
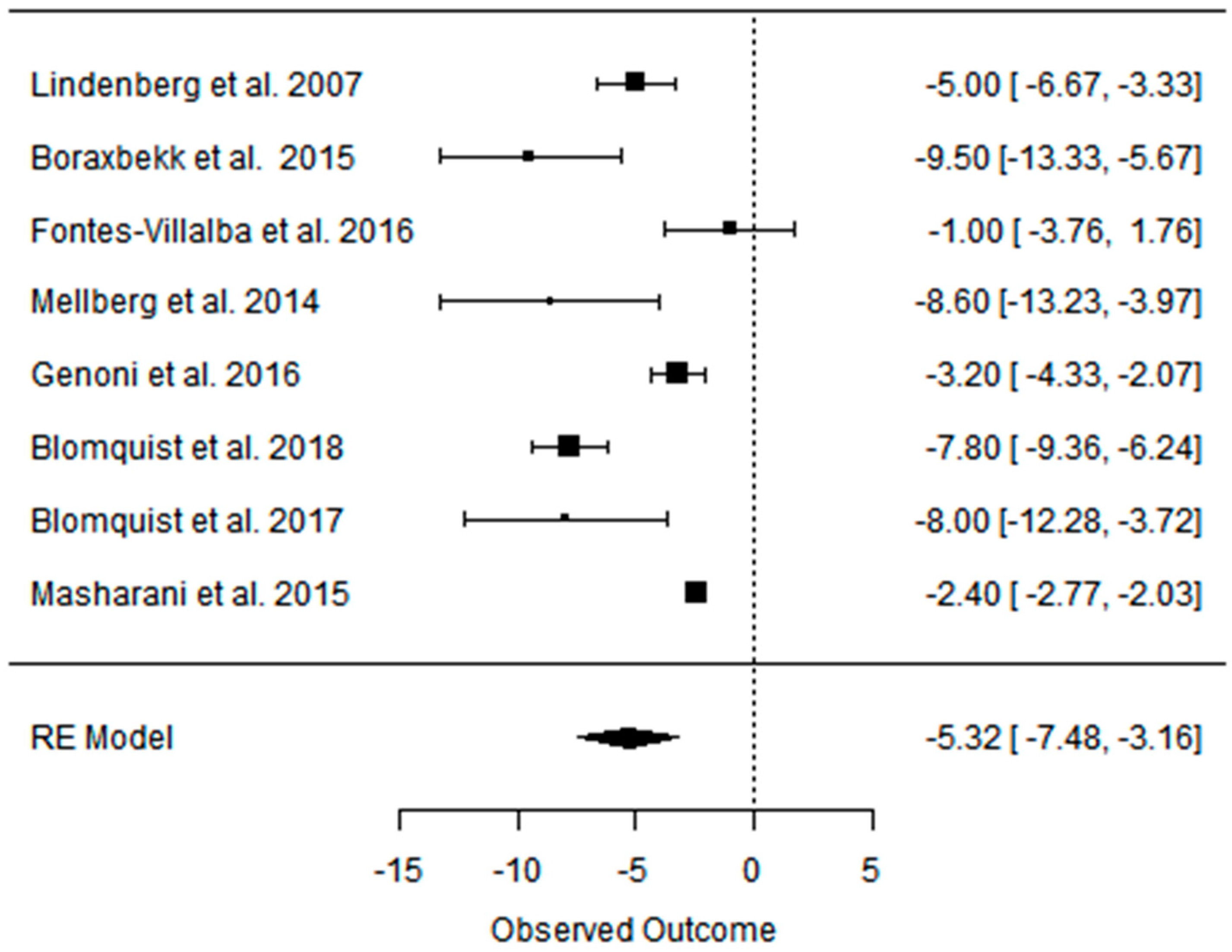
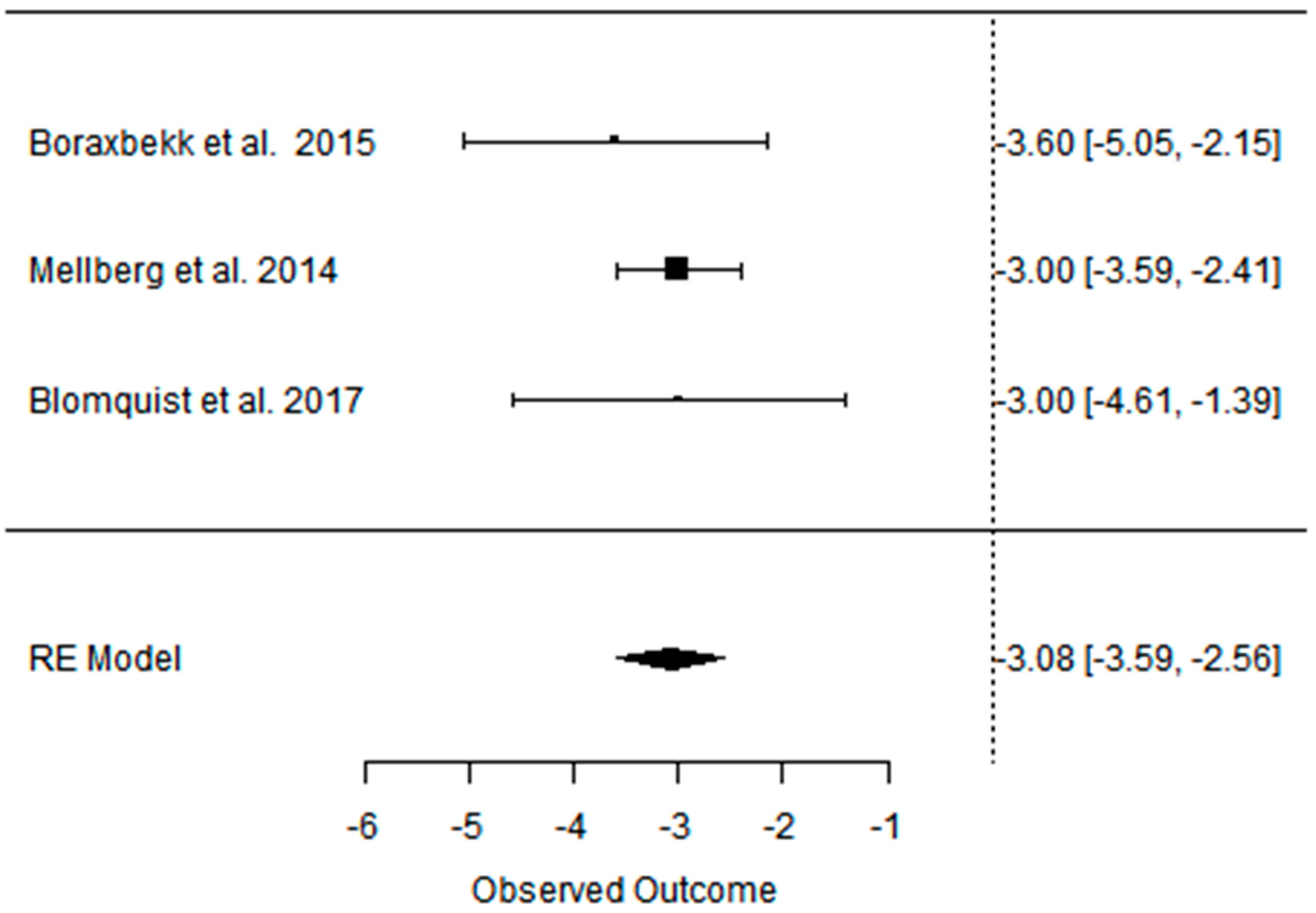
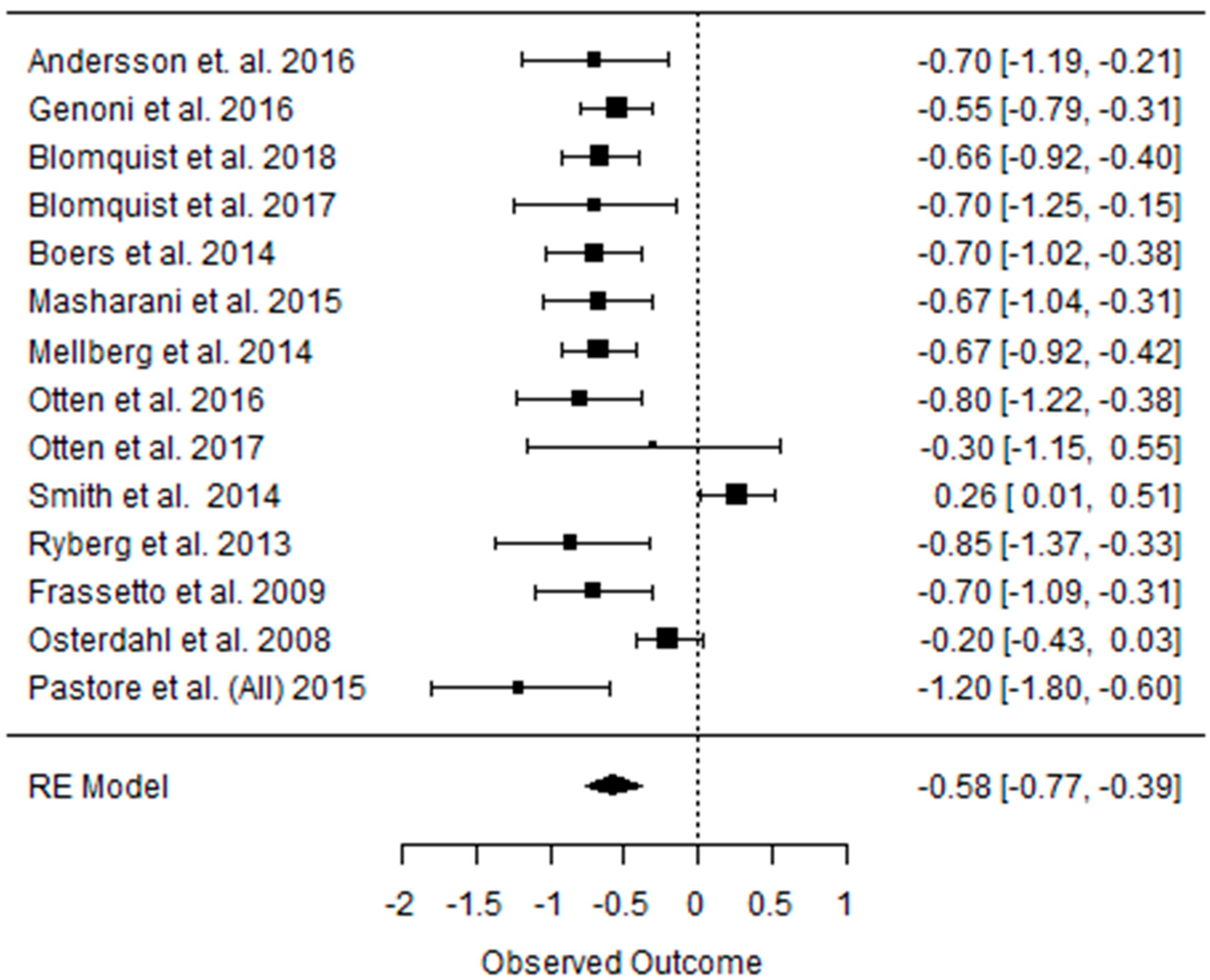
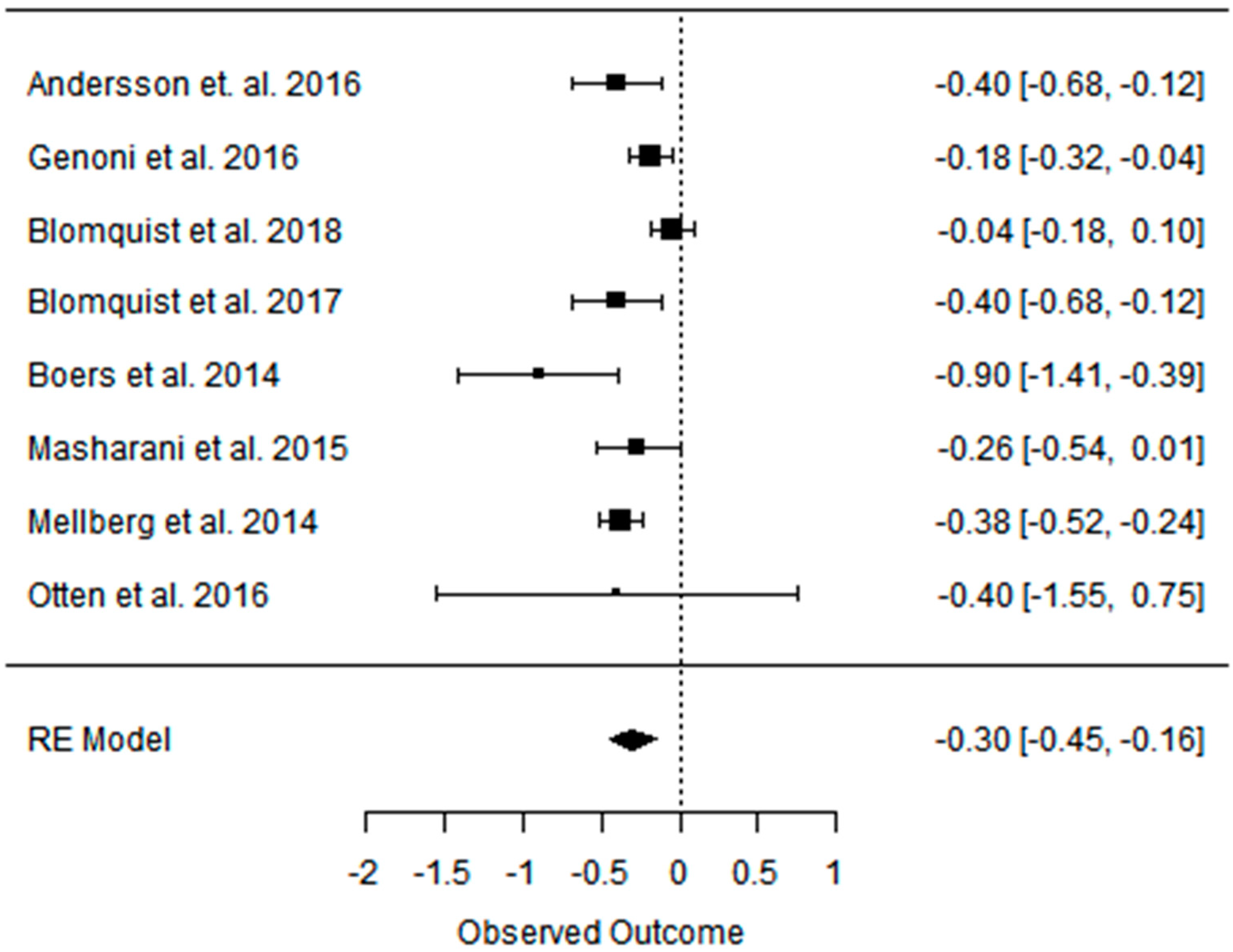
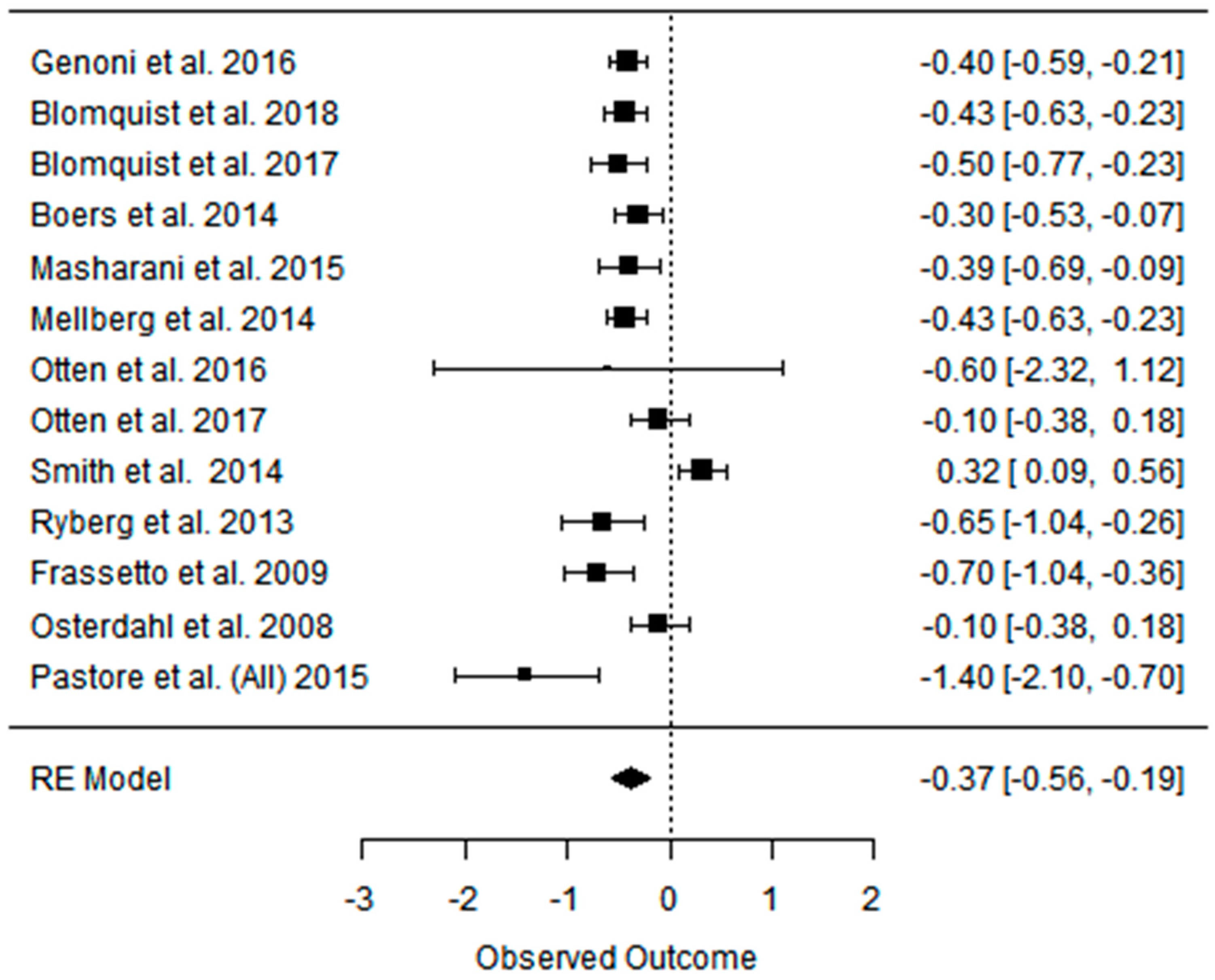
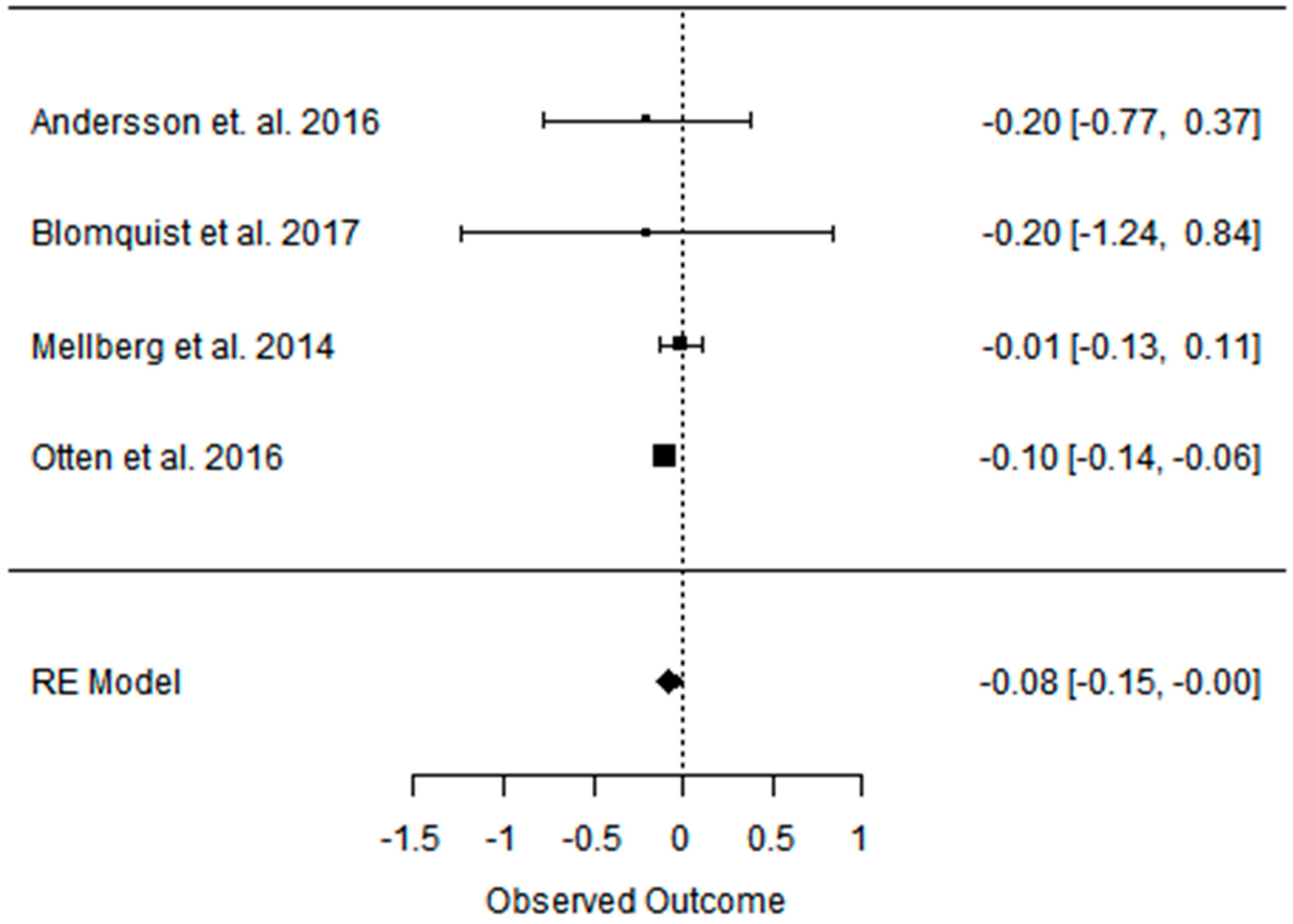
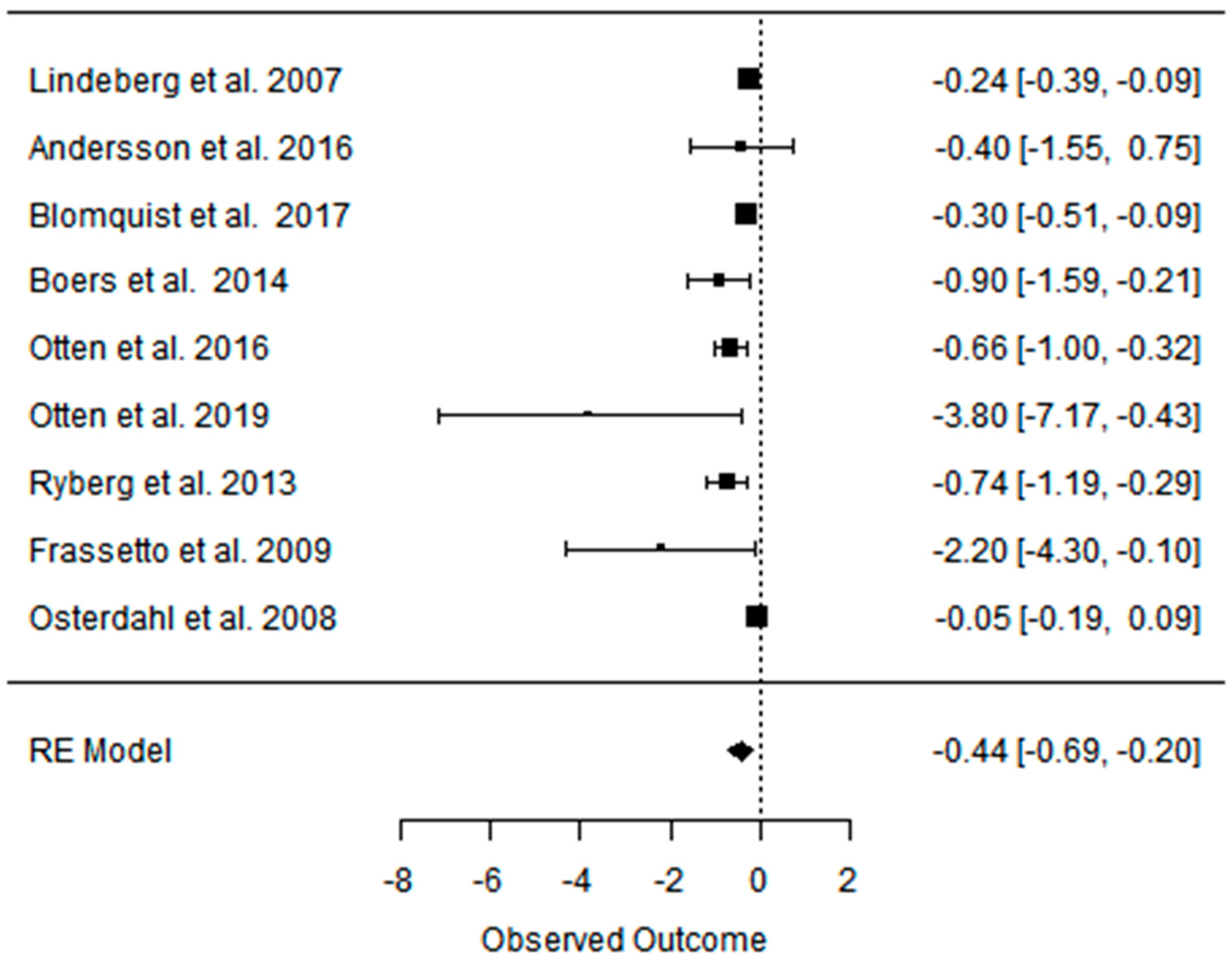
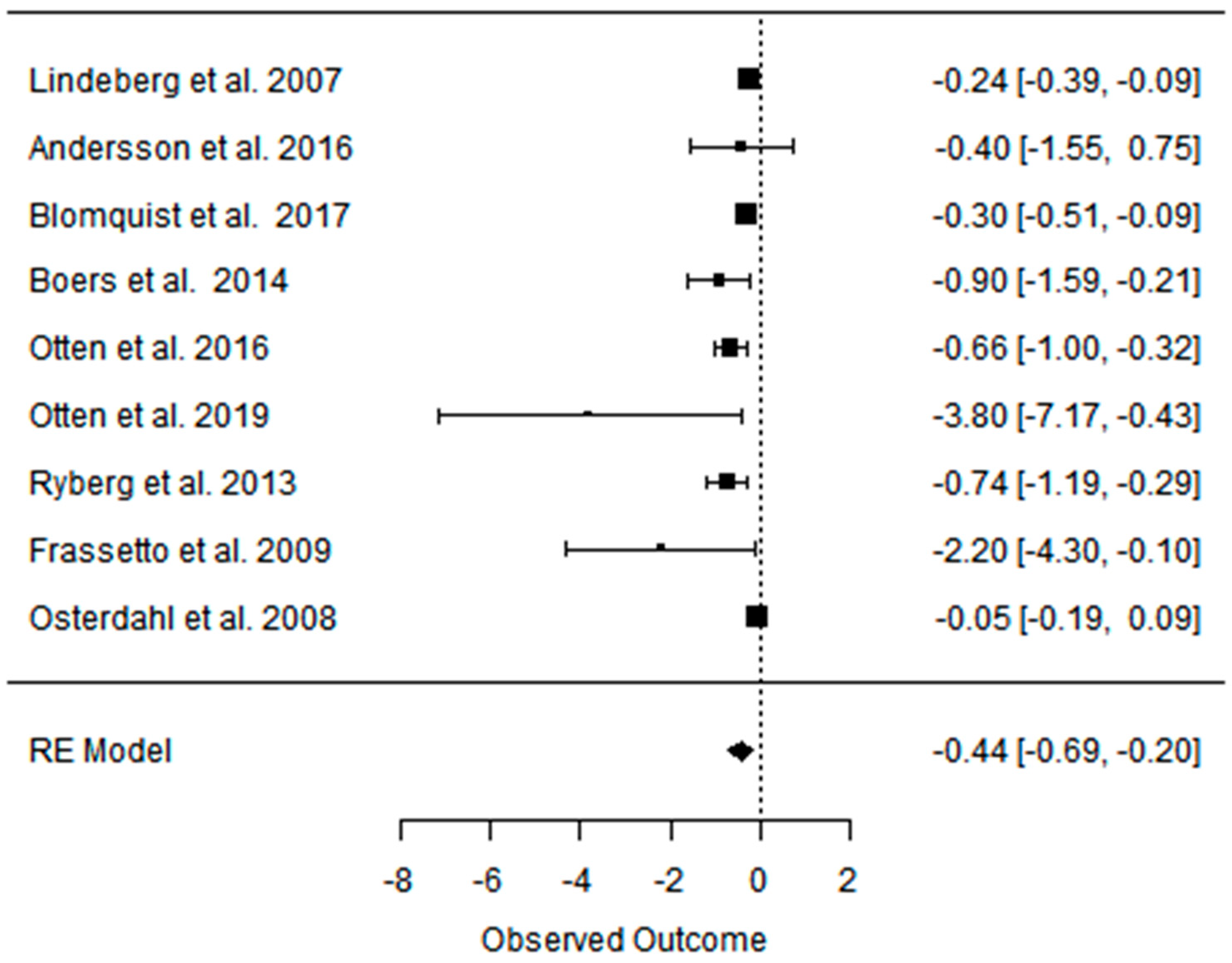
3.3. Effect of the Paleolithic Diet on Lipid Profile (Total Cholesterol, Triglycerydes, HDL-C, and LDL-C in the Short (up to 6 Months) and Long (over 6 Months) Term
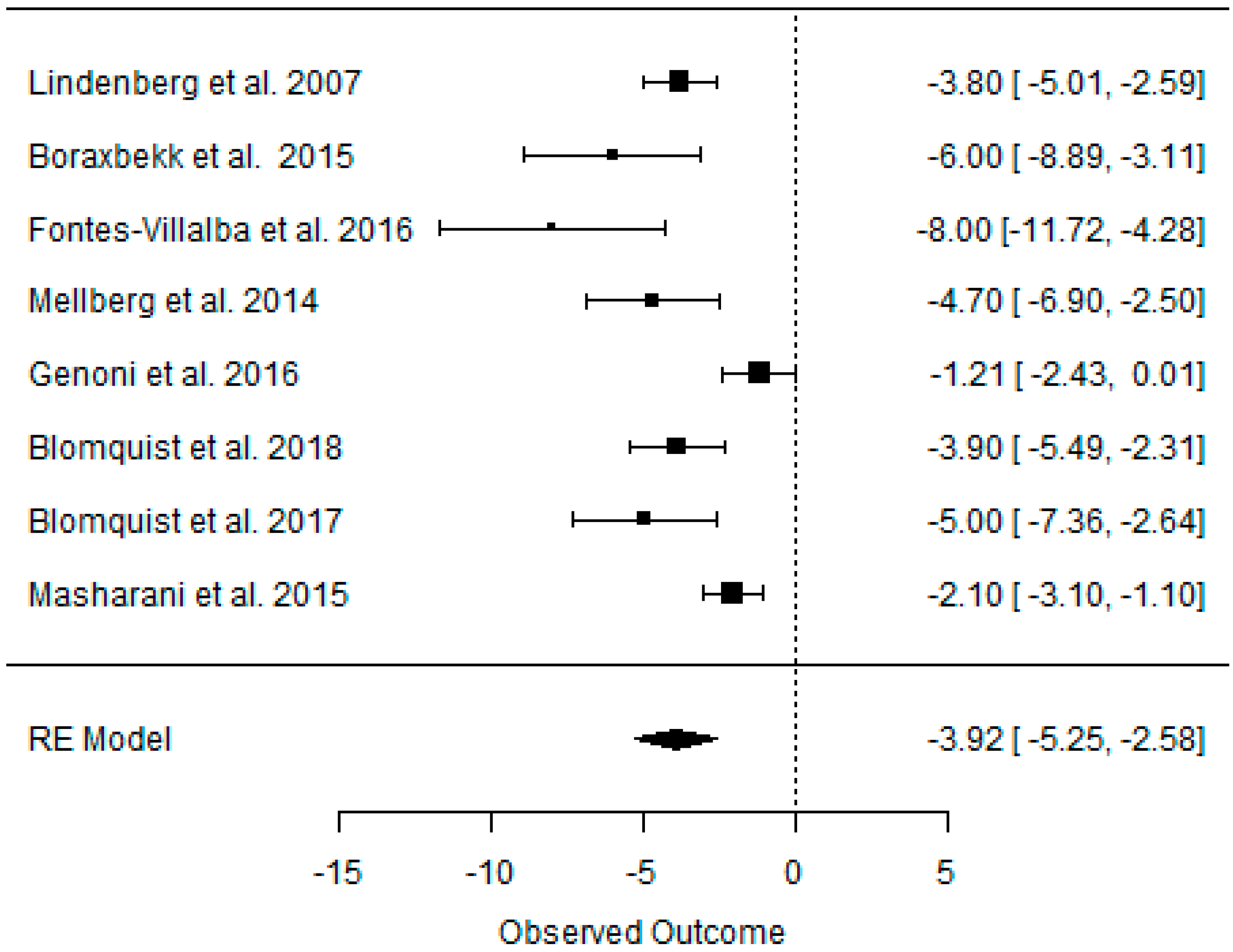
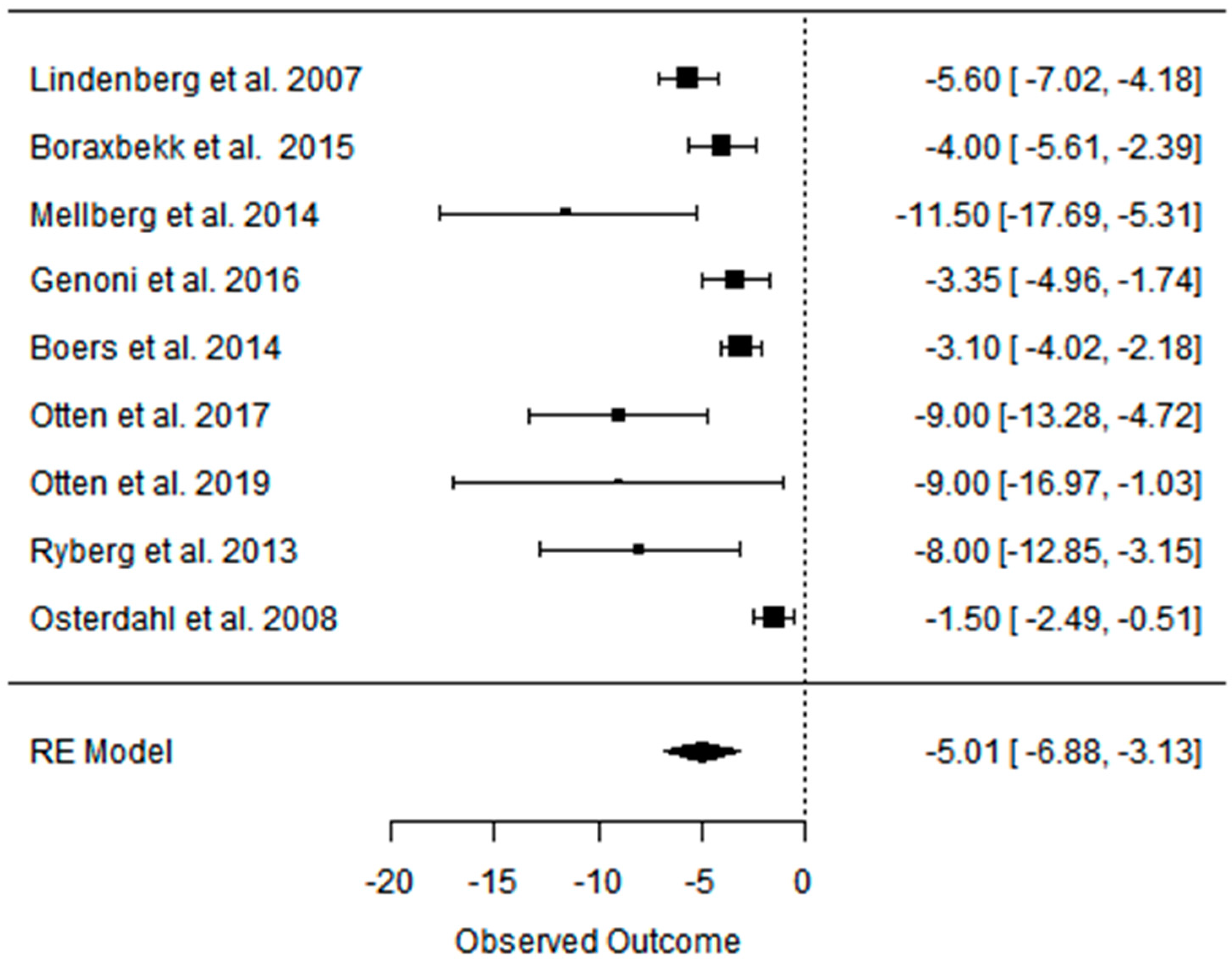
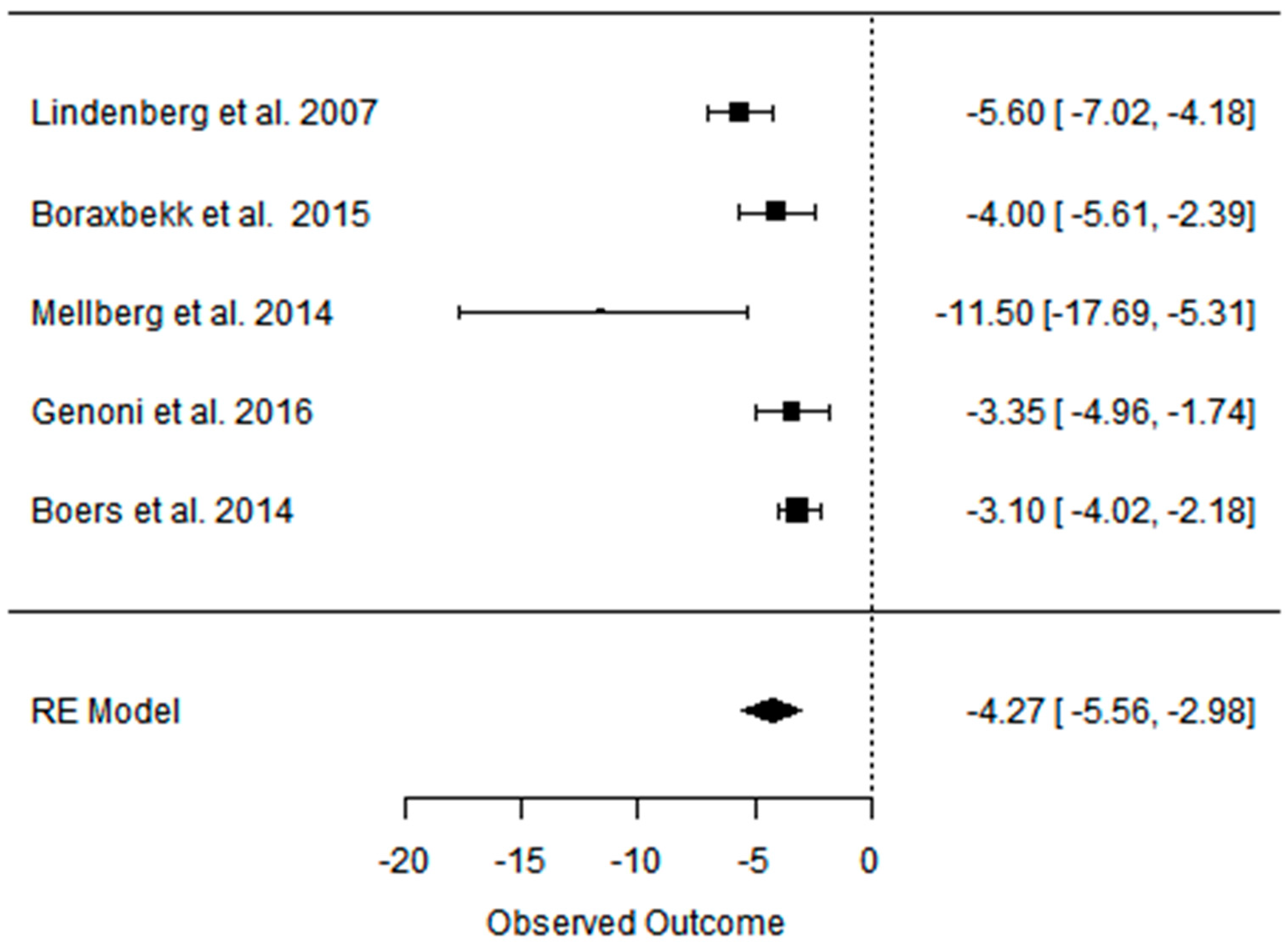
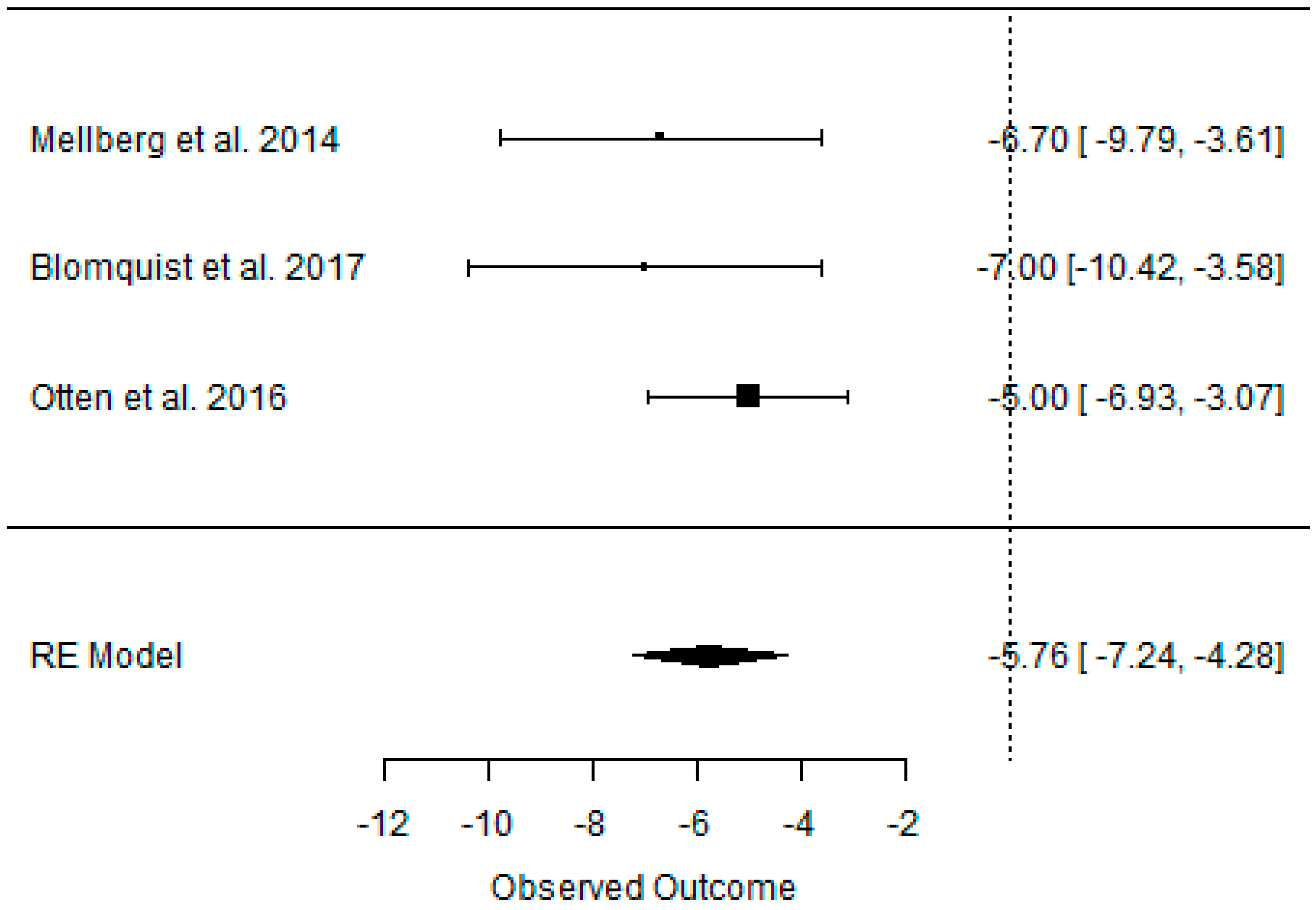
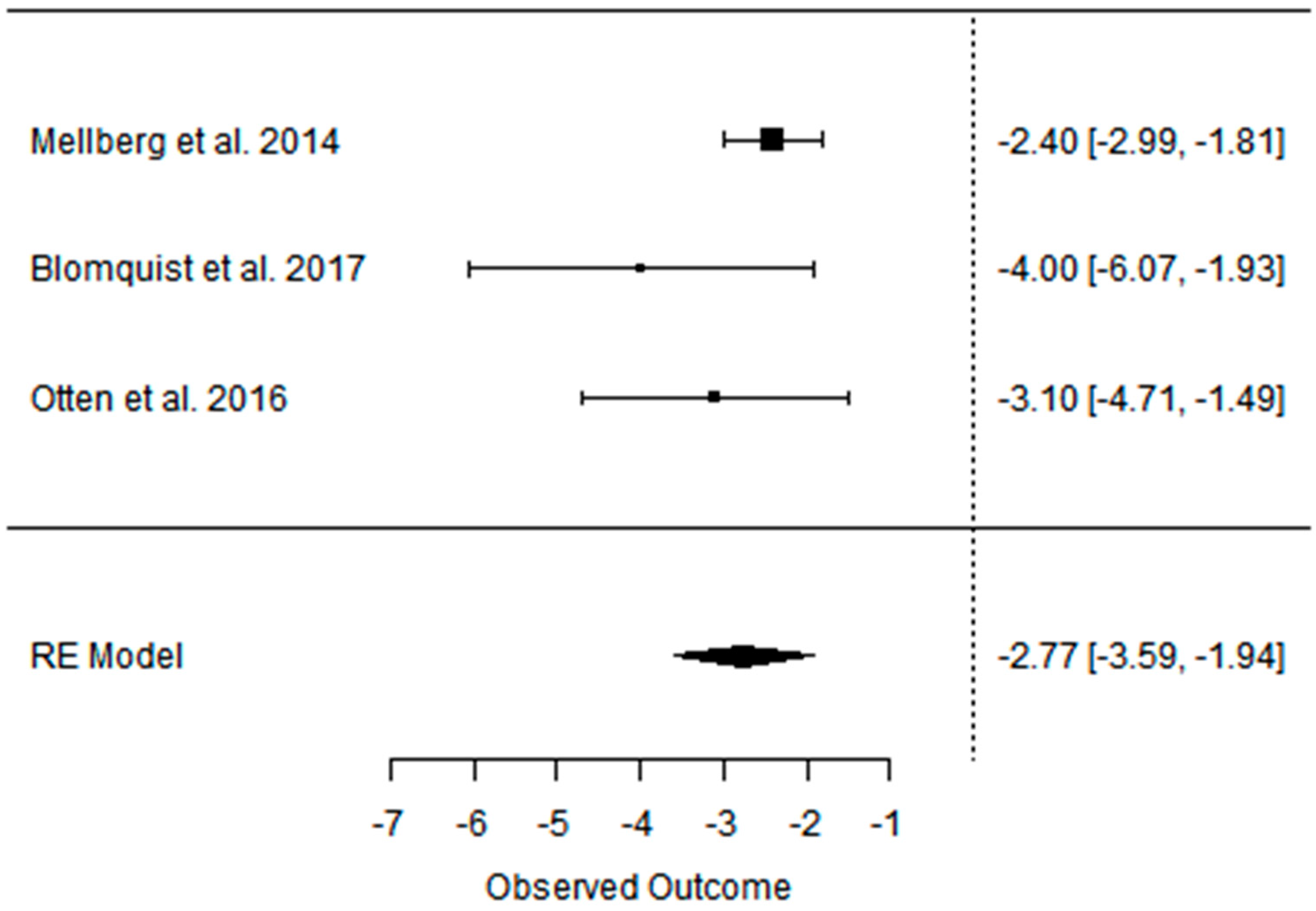
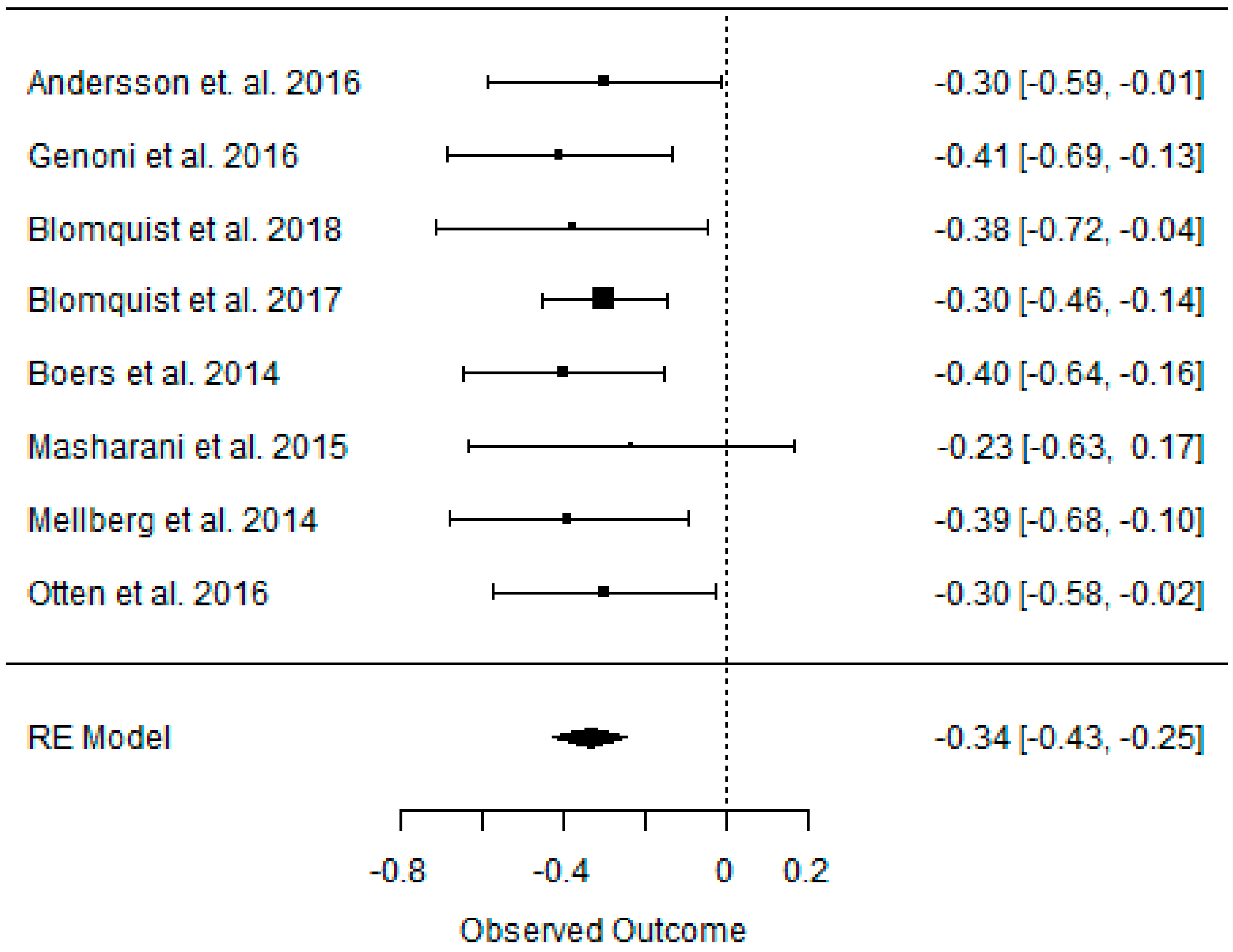
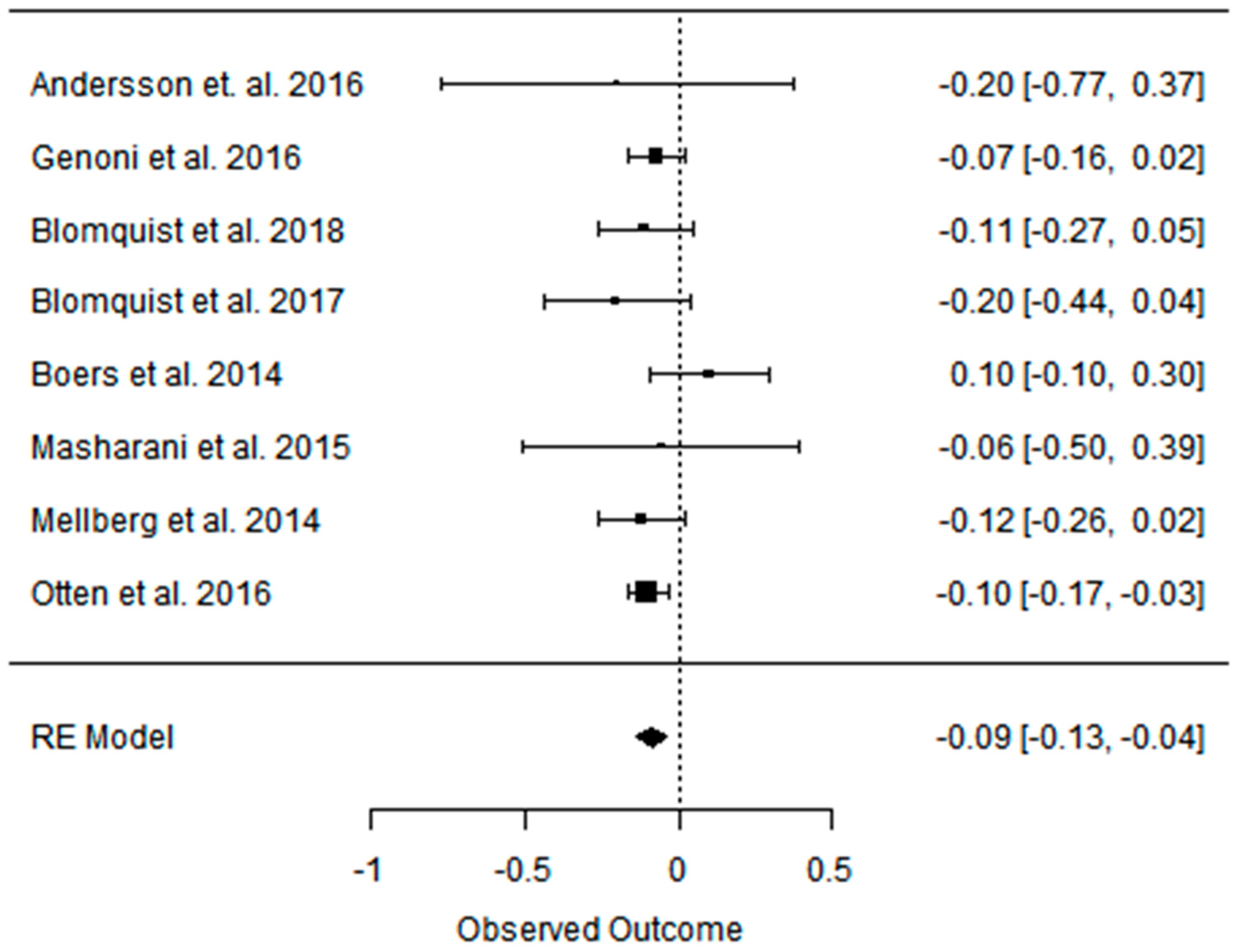
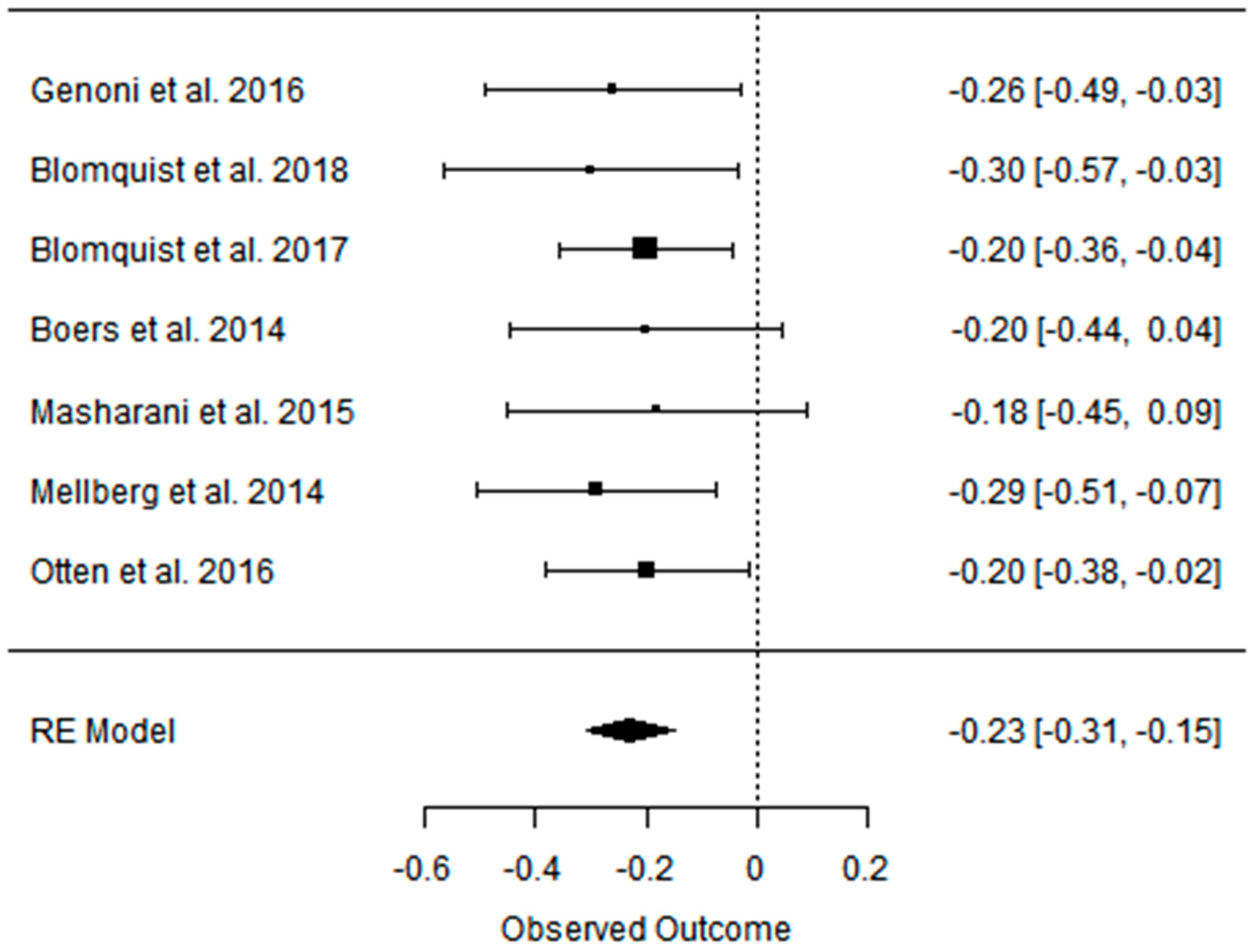
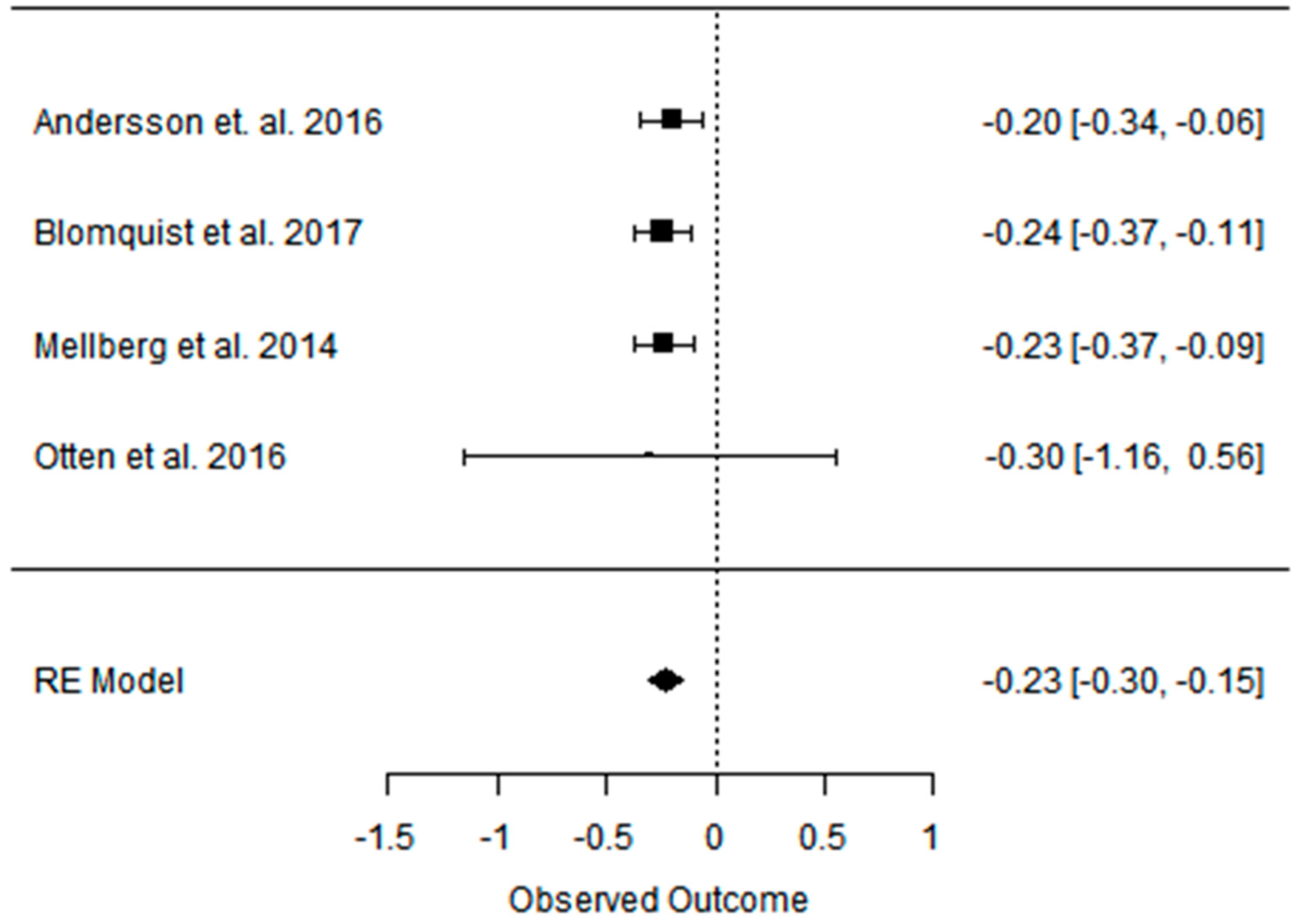
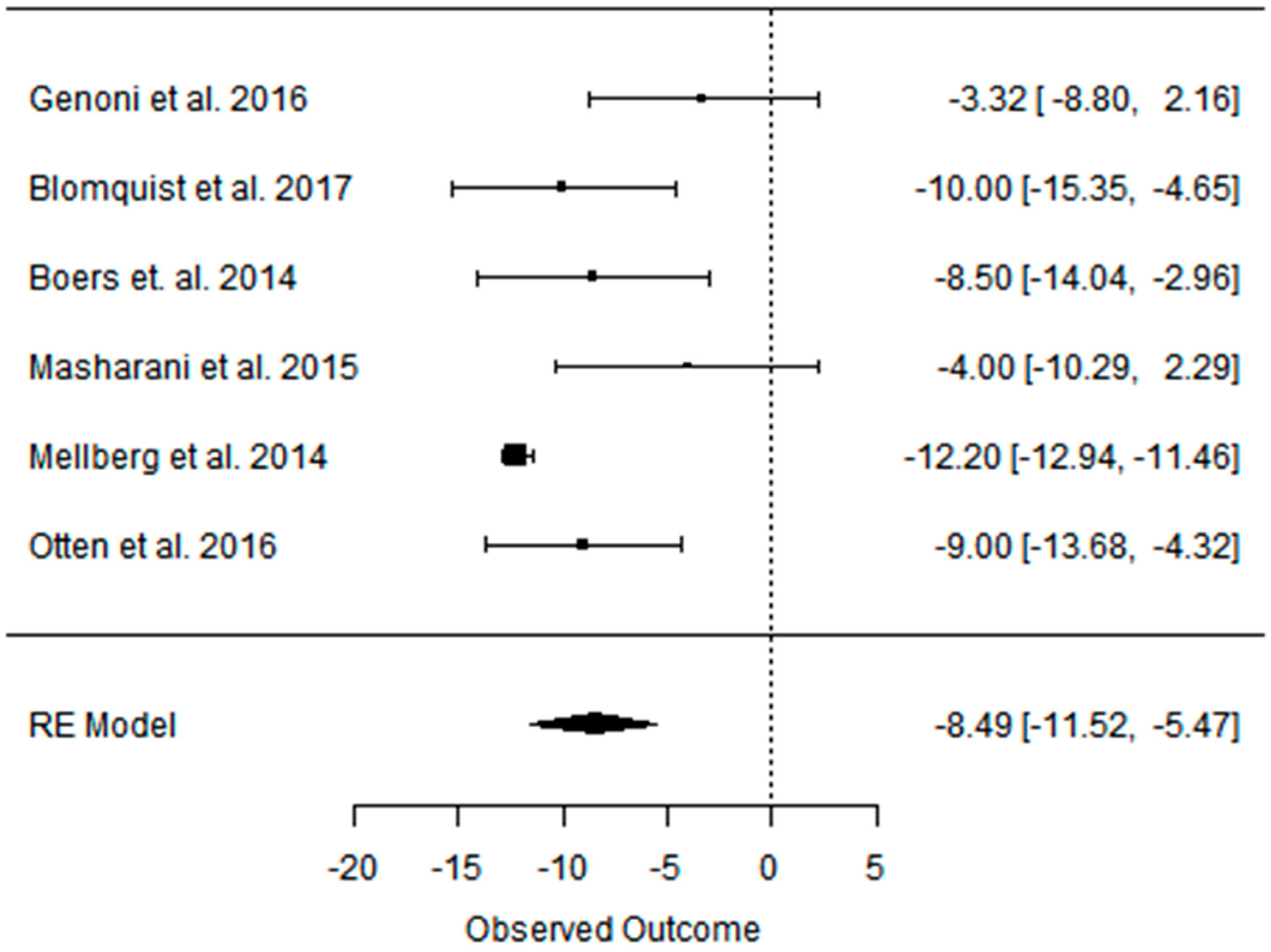
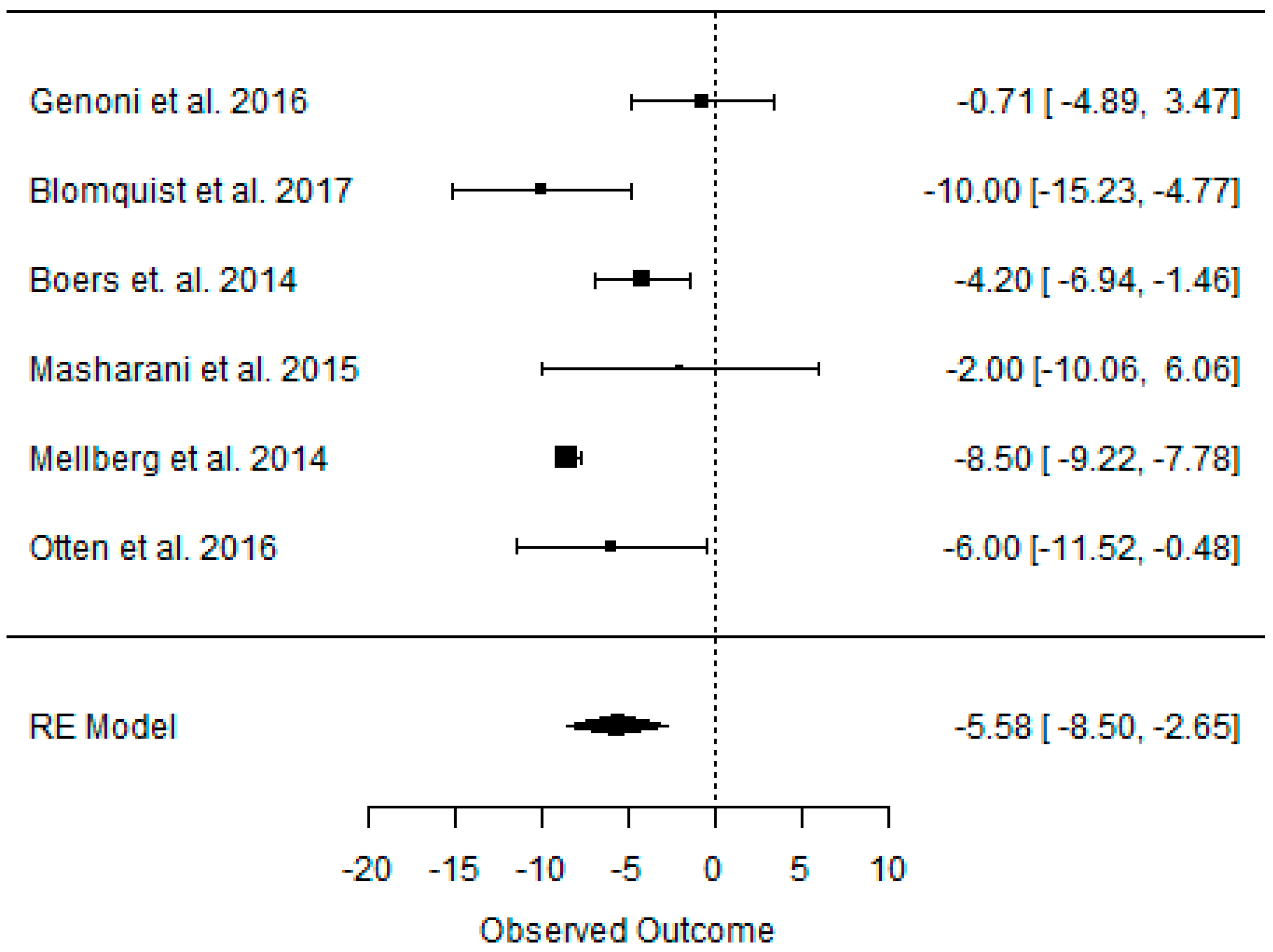
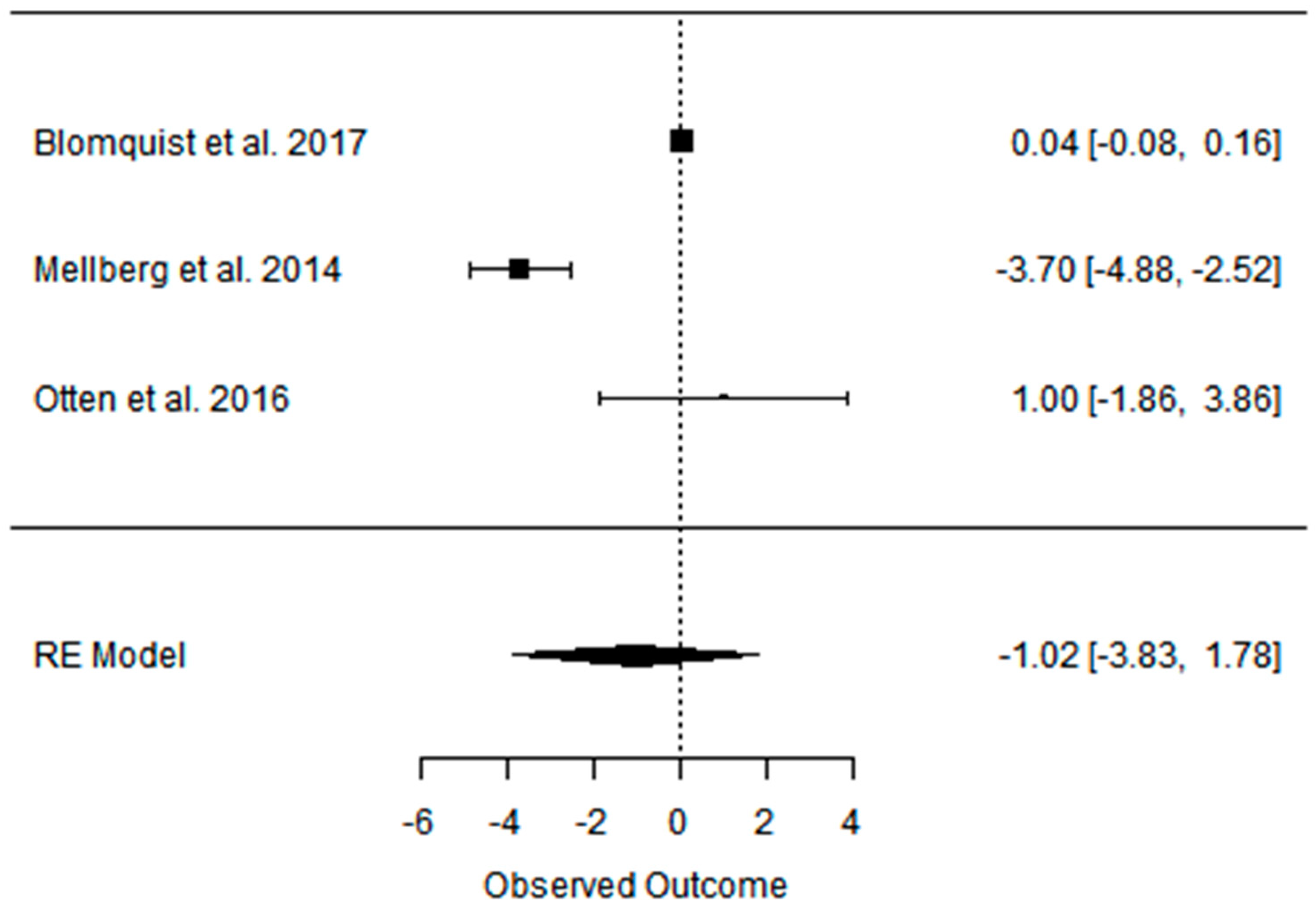
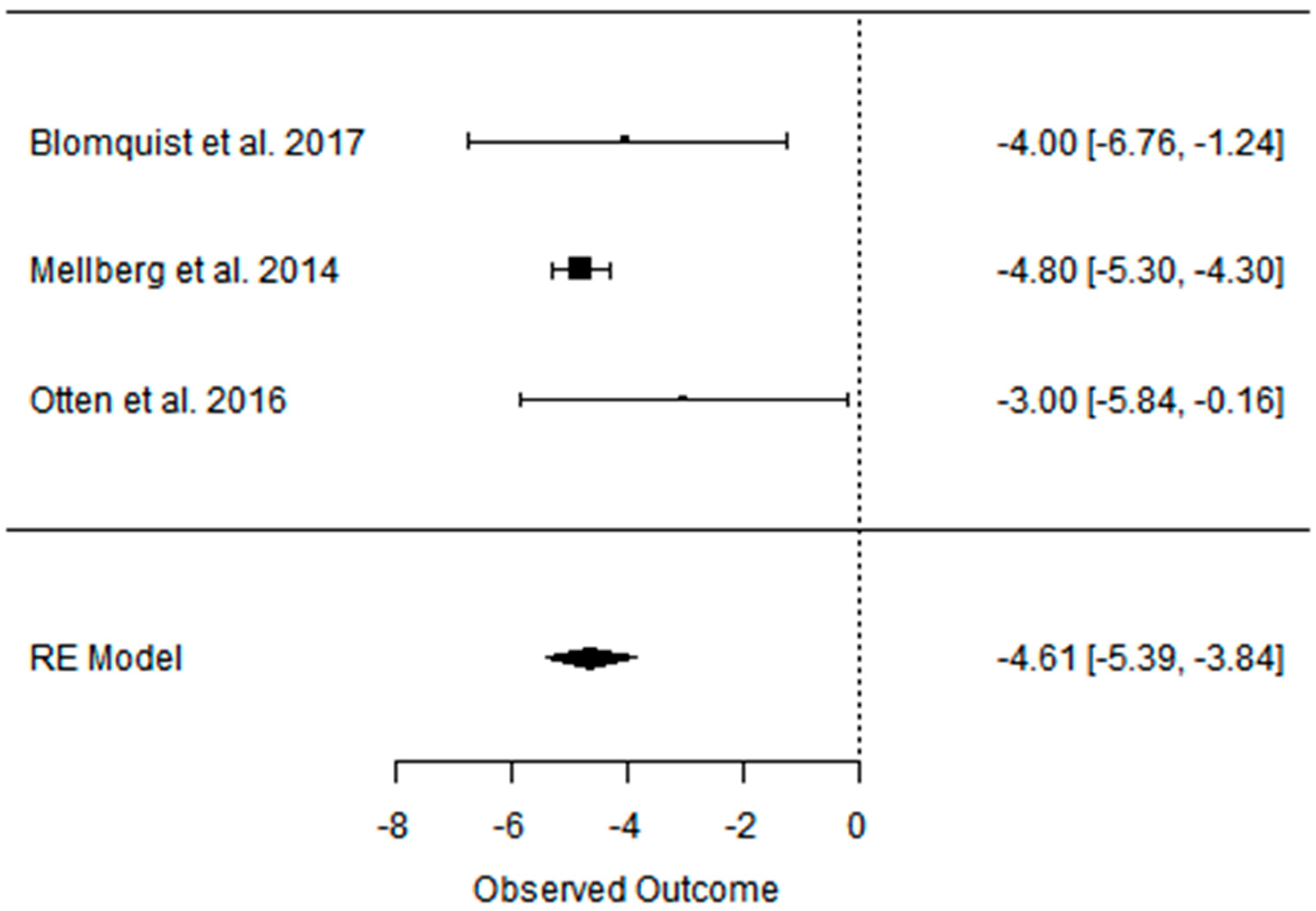
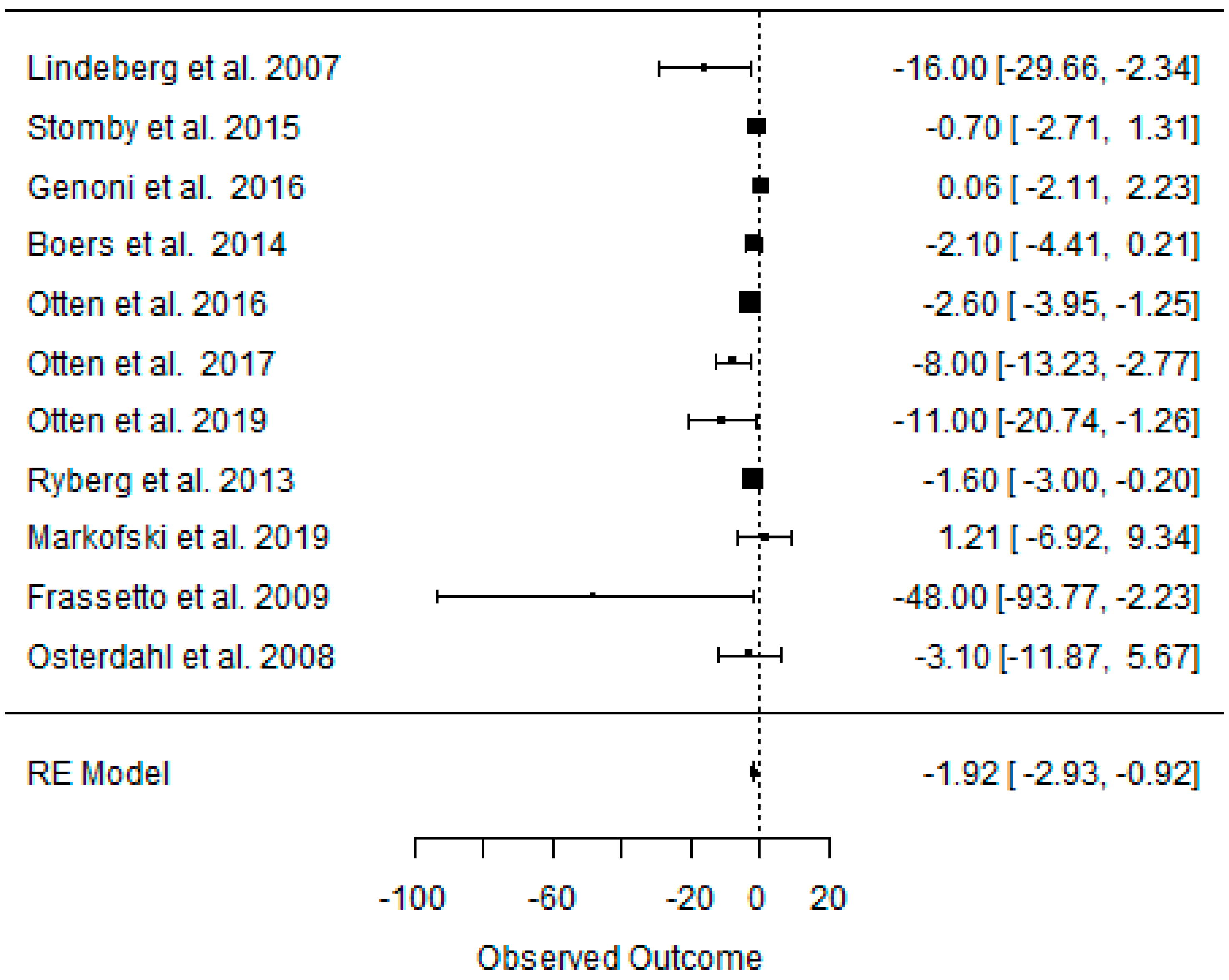
3.4. Effect of the Paleolithic Diet on Blood Pressure (Systolic Blood Pressure, Diastolic Blood Pressure, and Heart Rate) in the Short (up to 6 Months) and Long (over 6 Months) Term
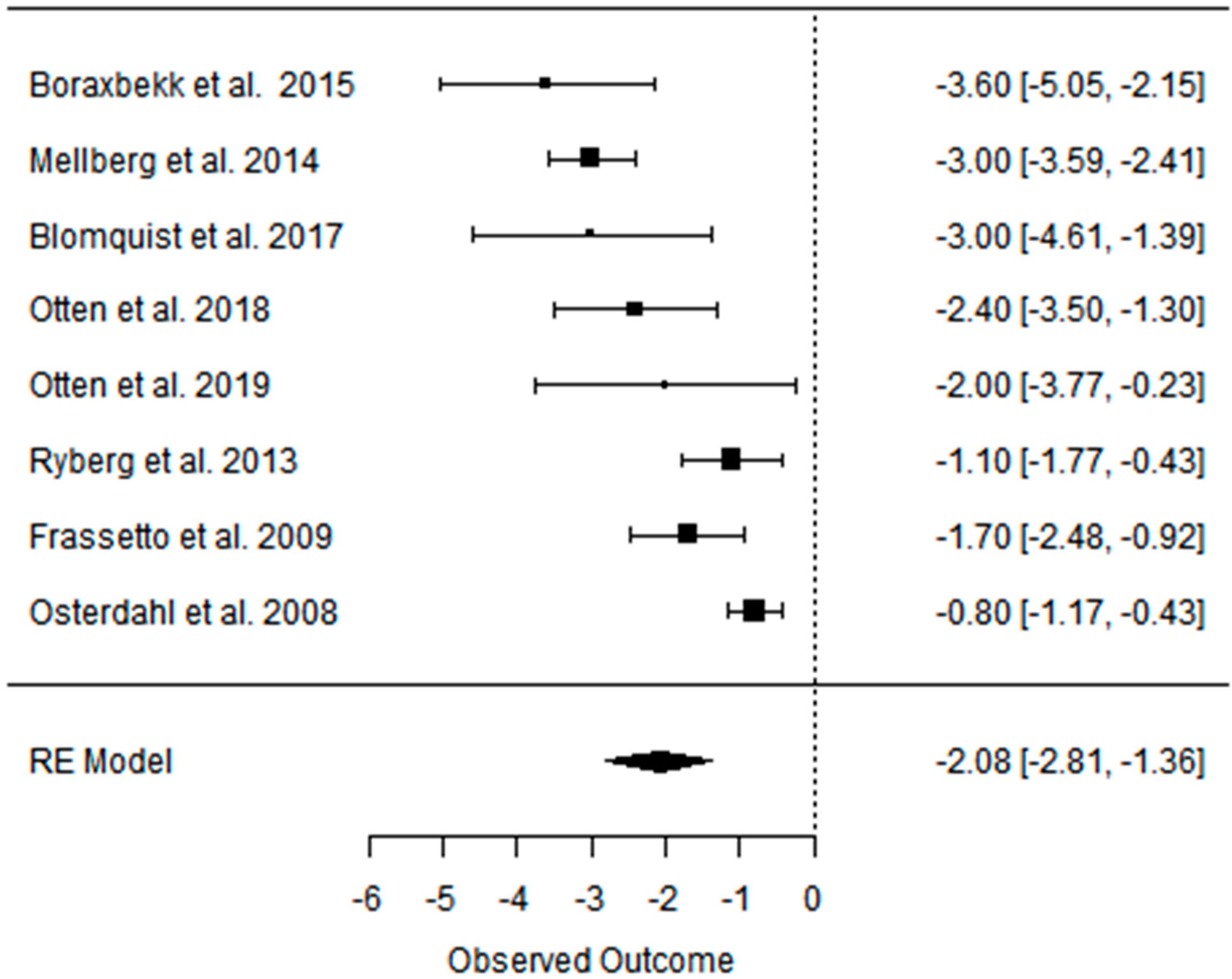
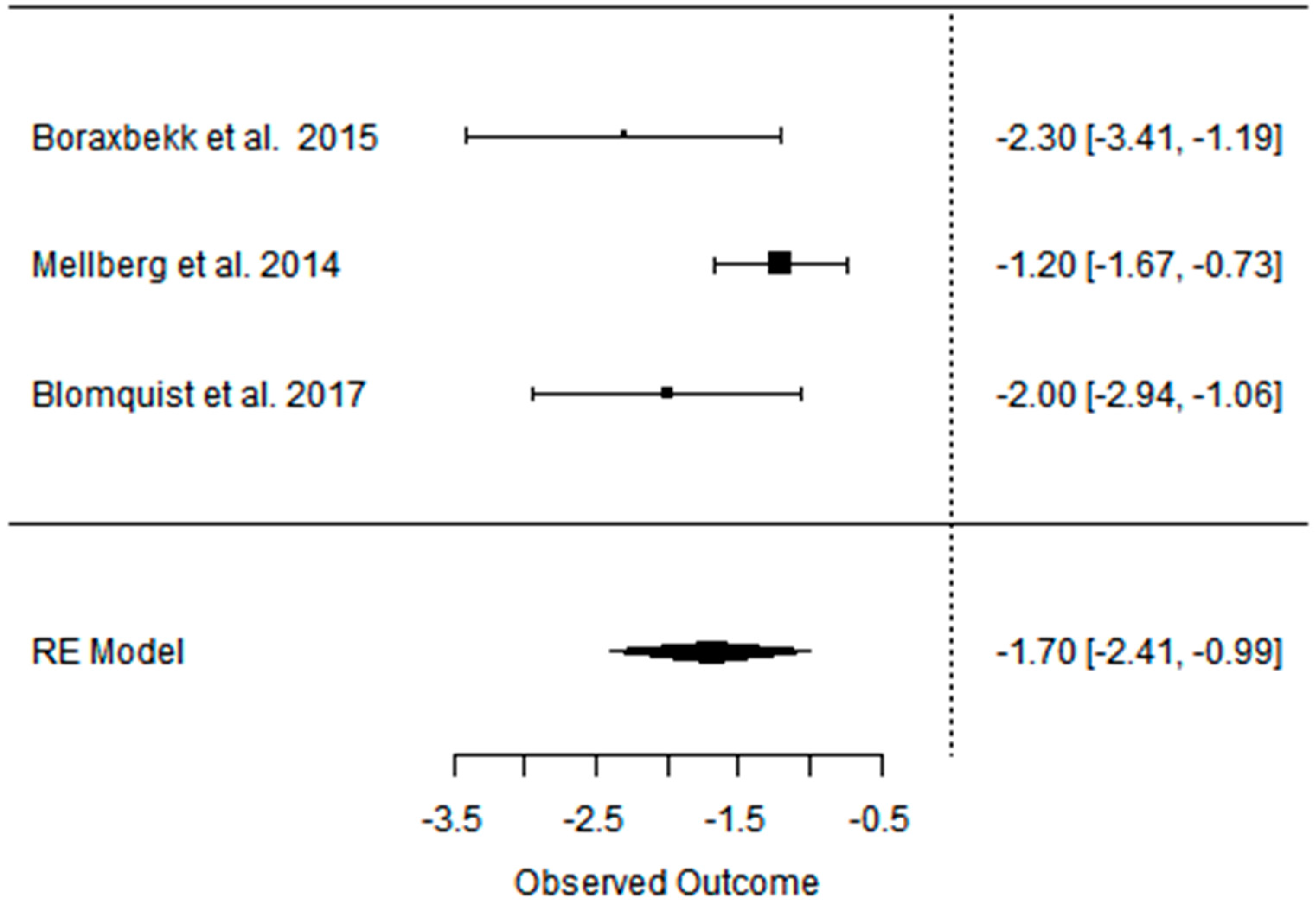
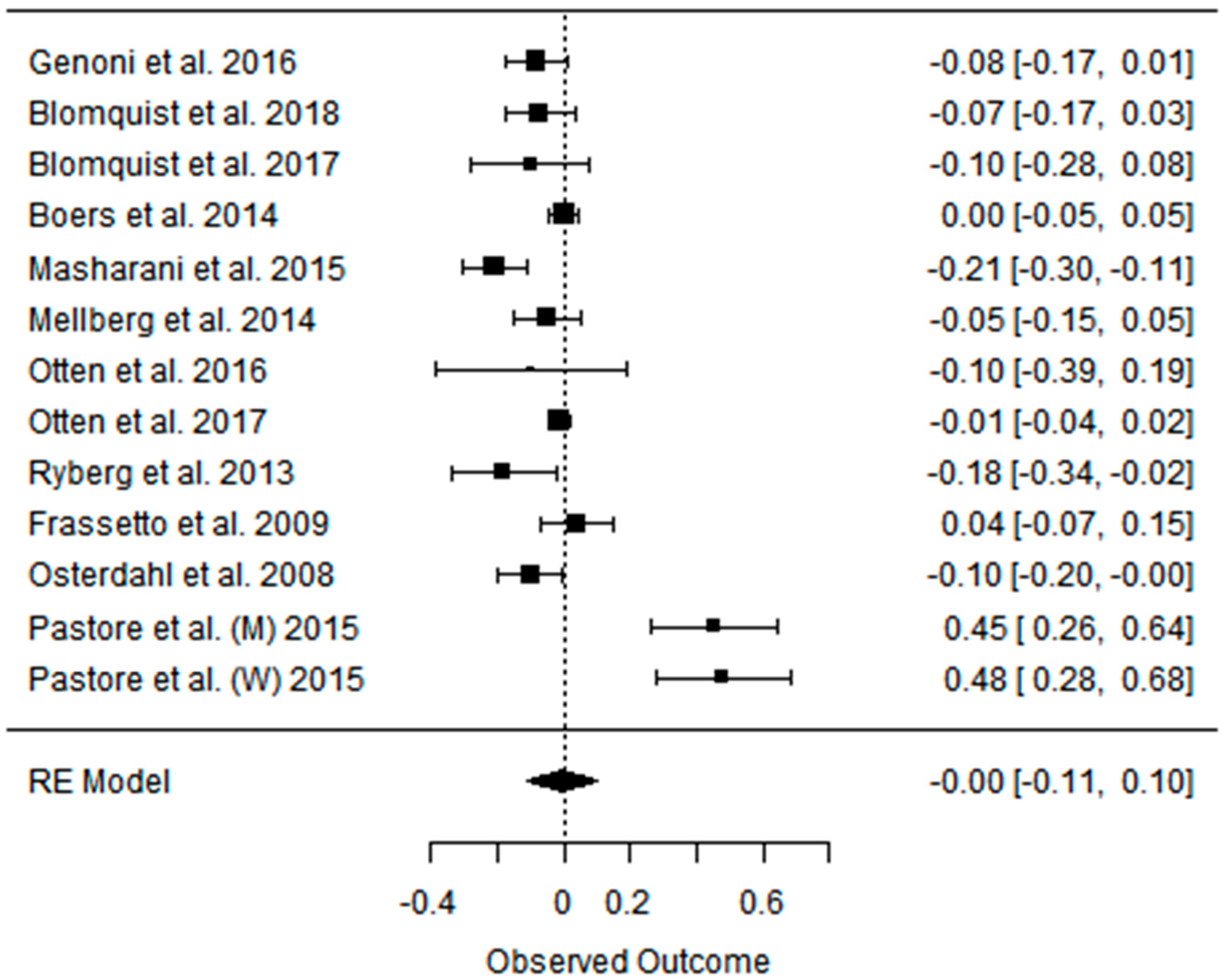
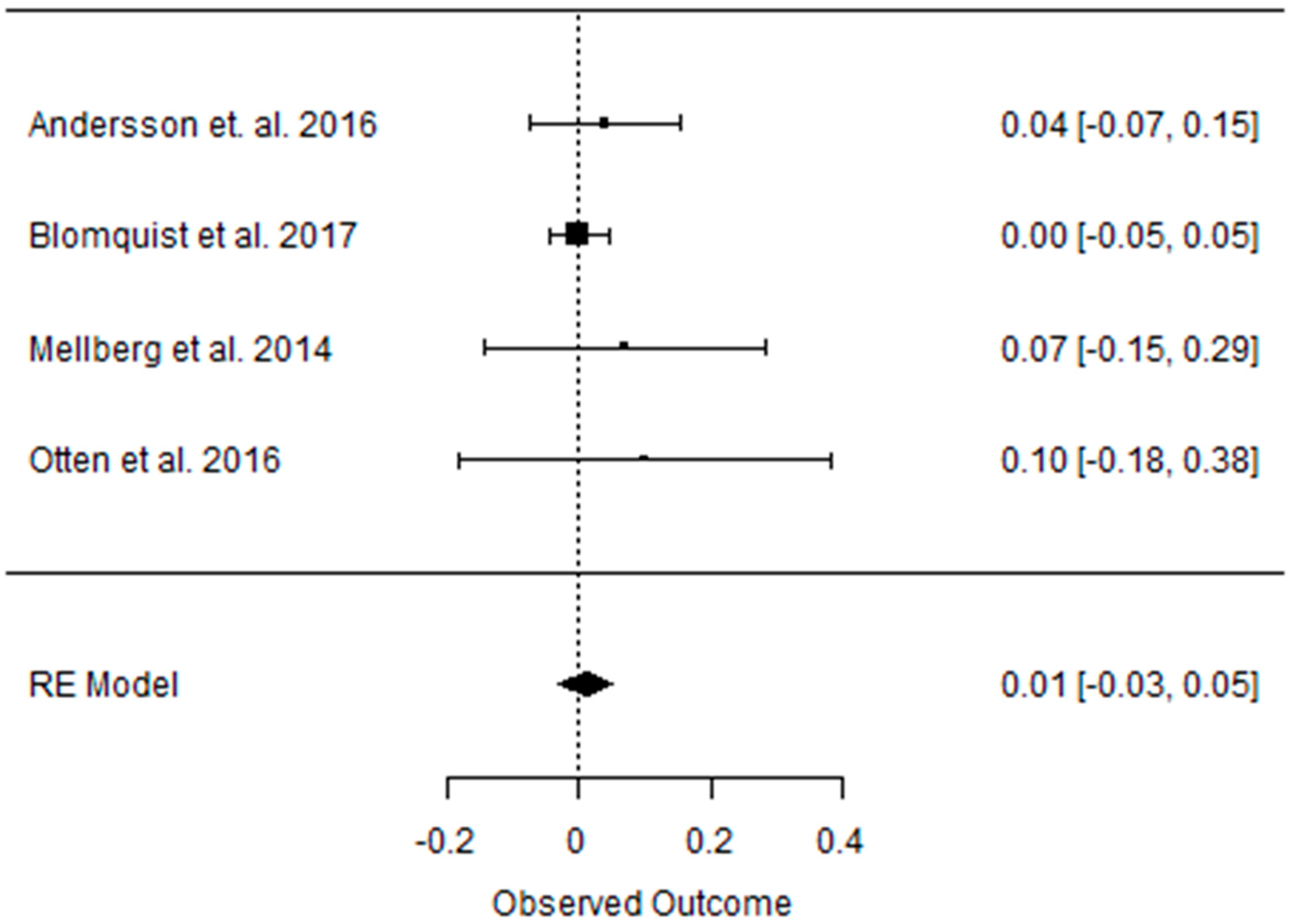
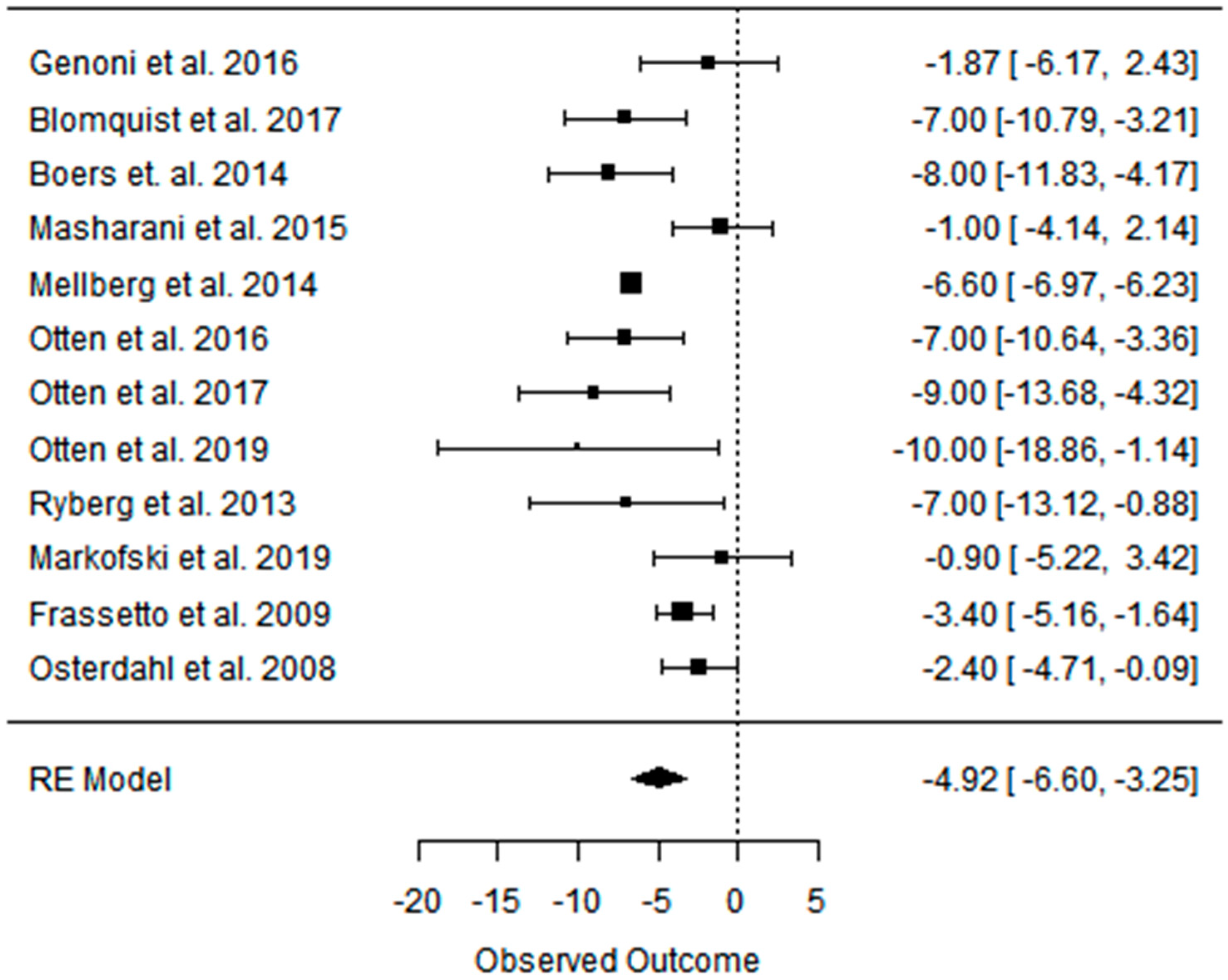
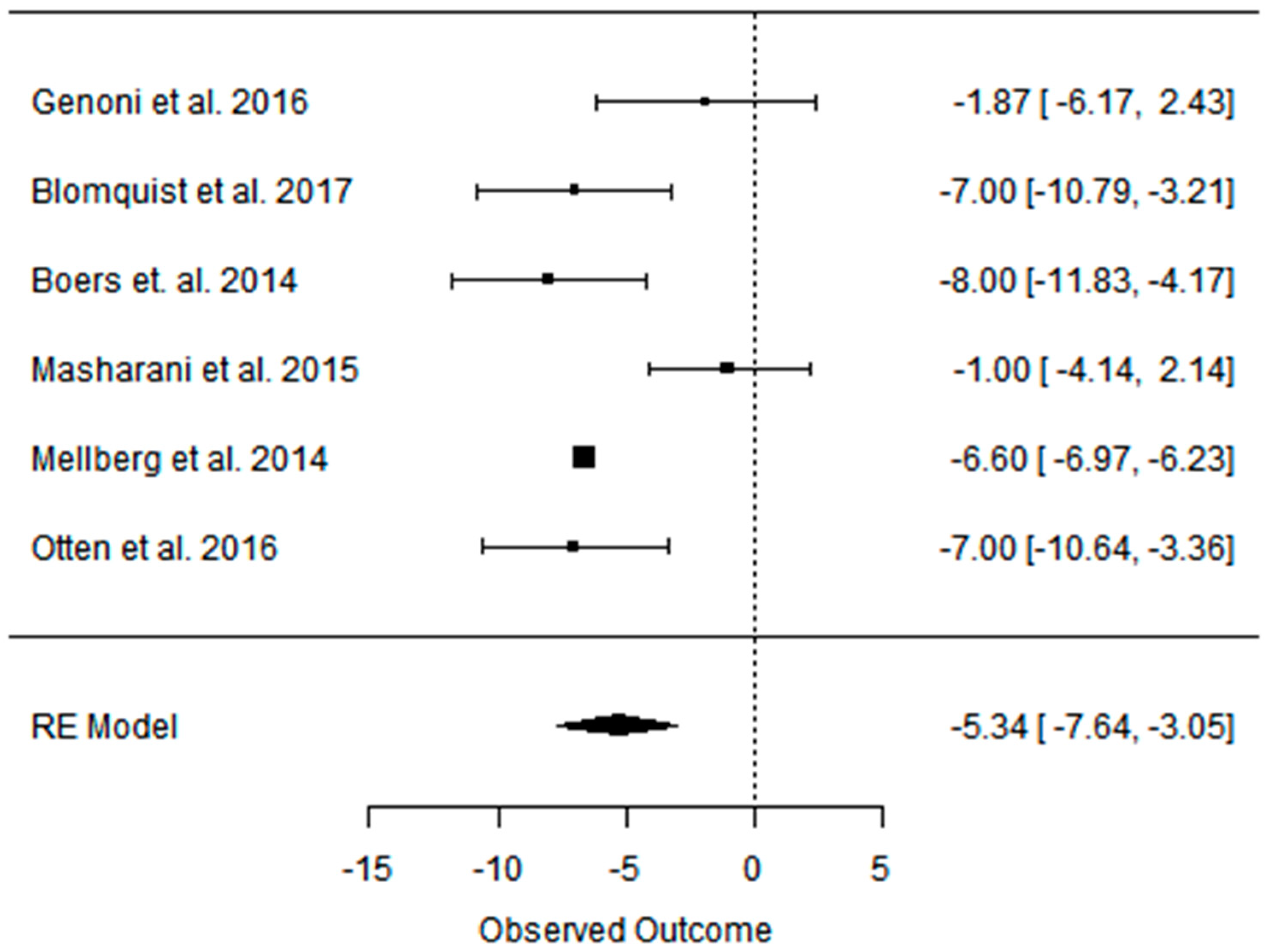
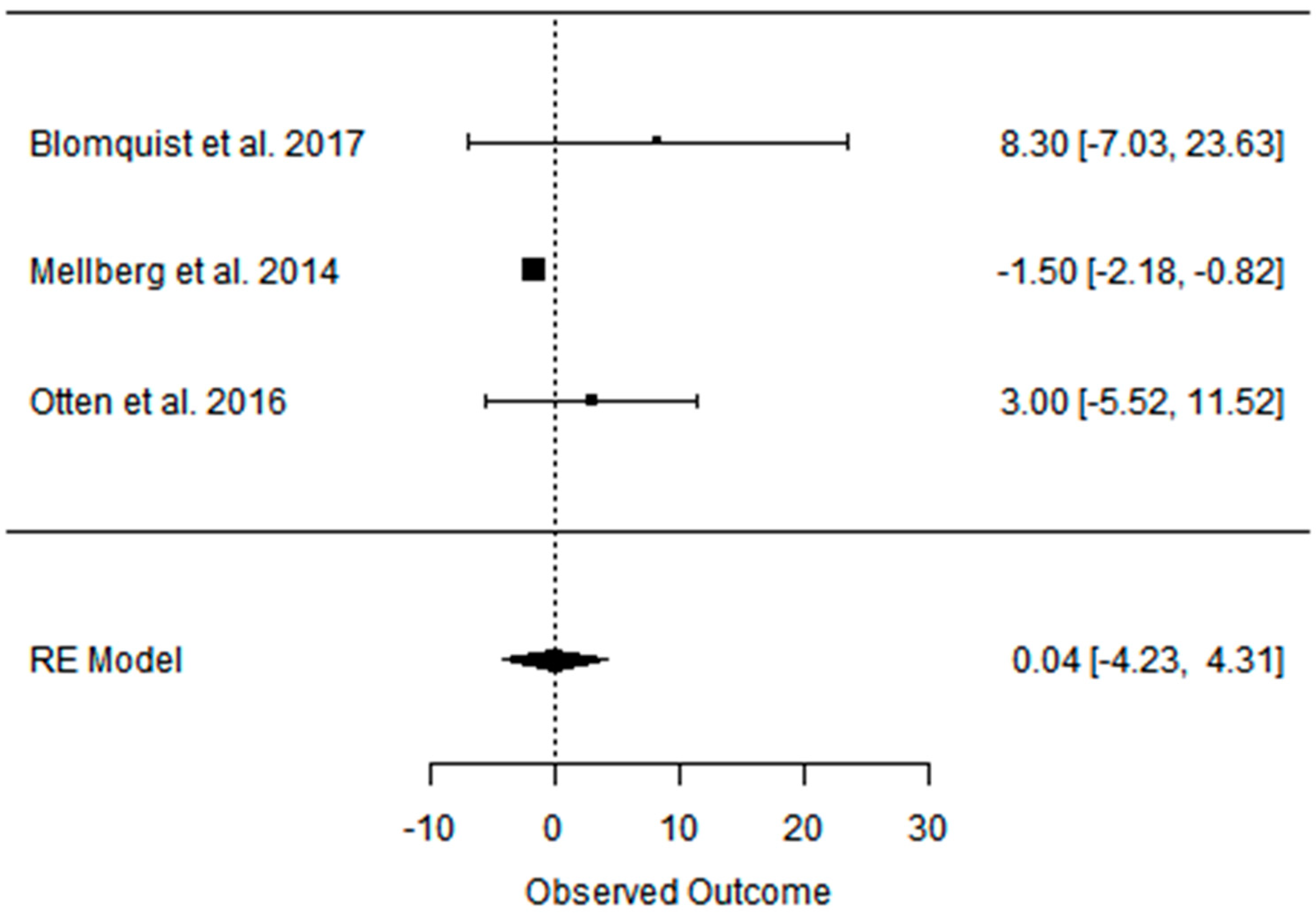
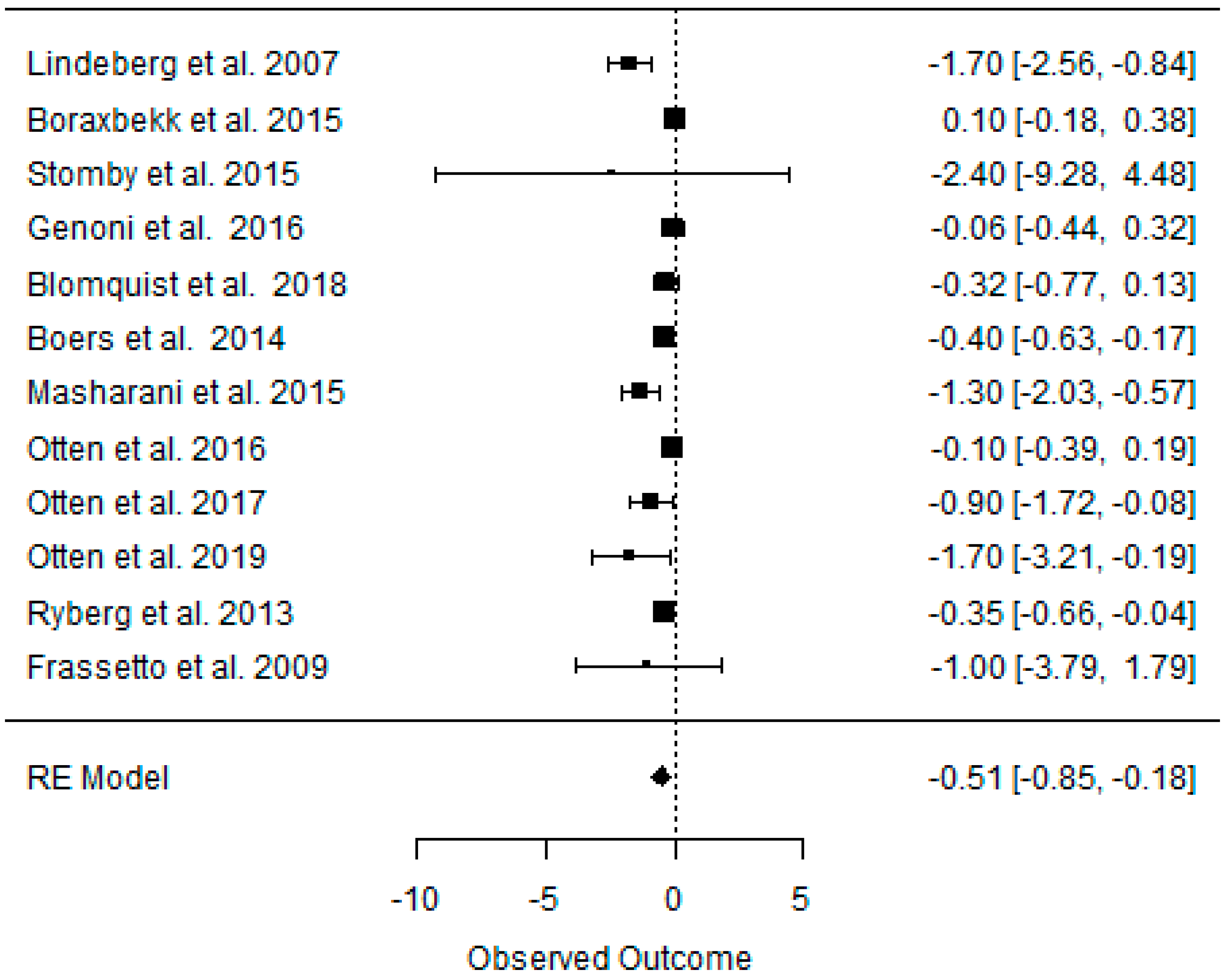
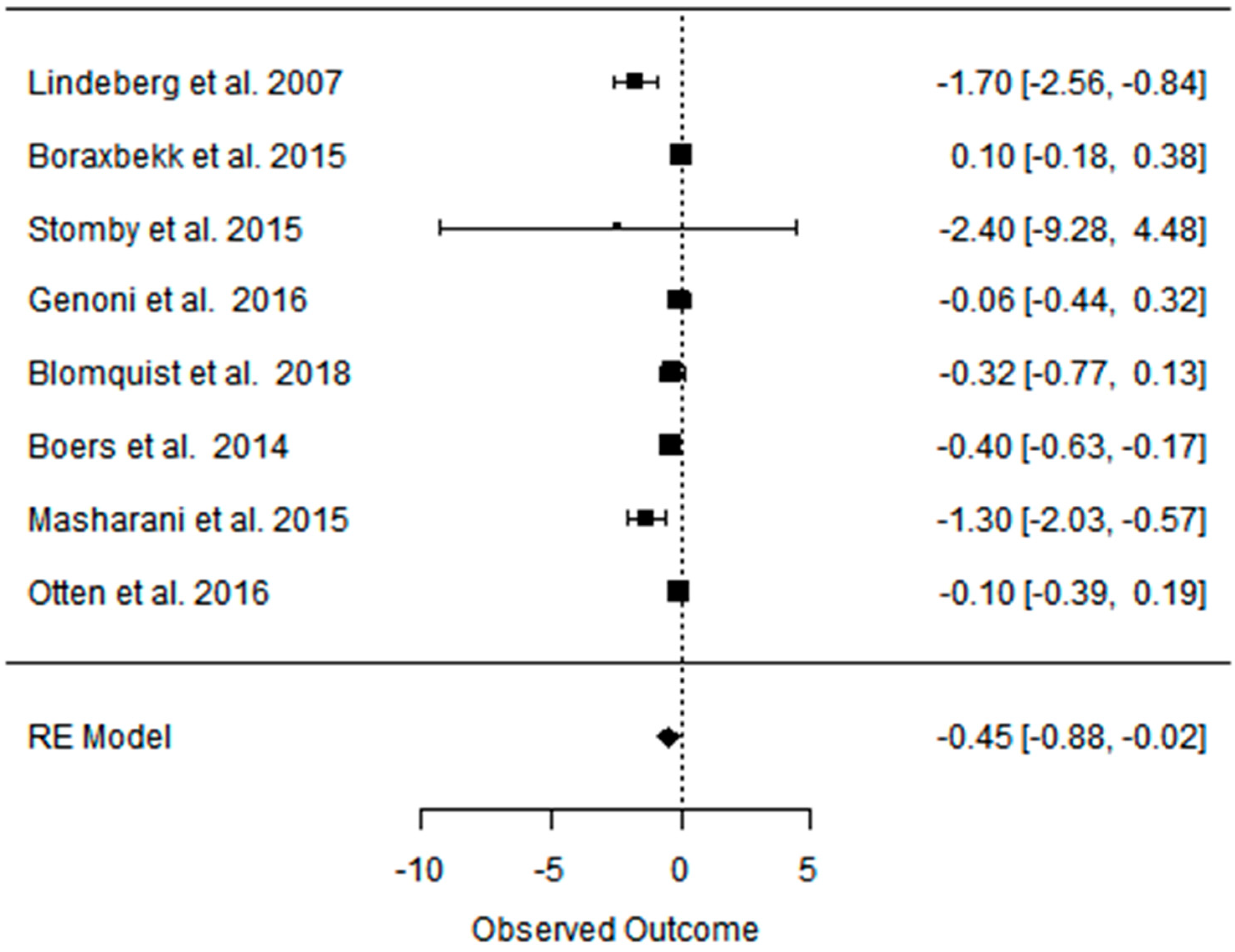
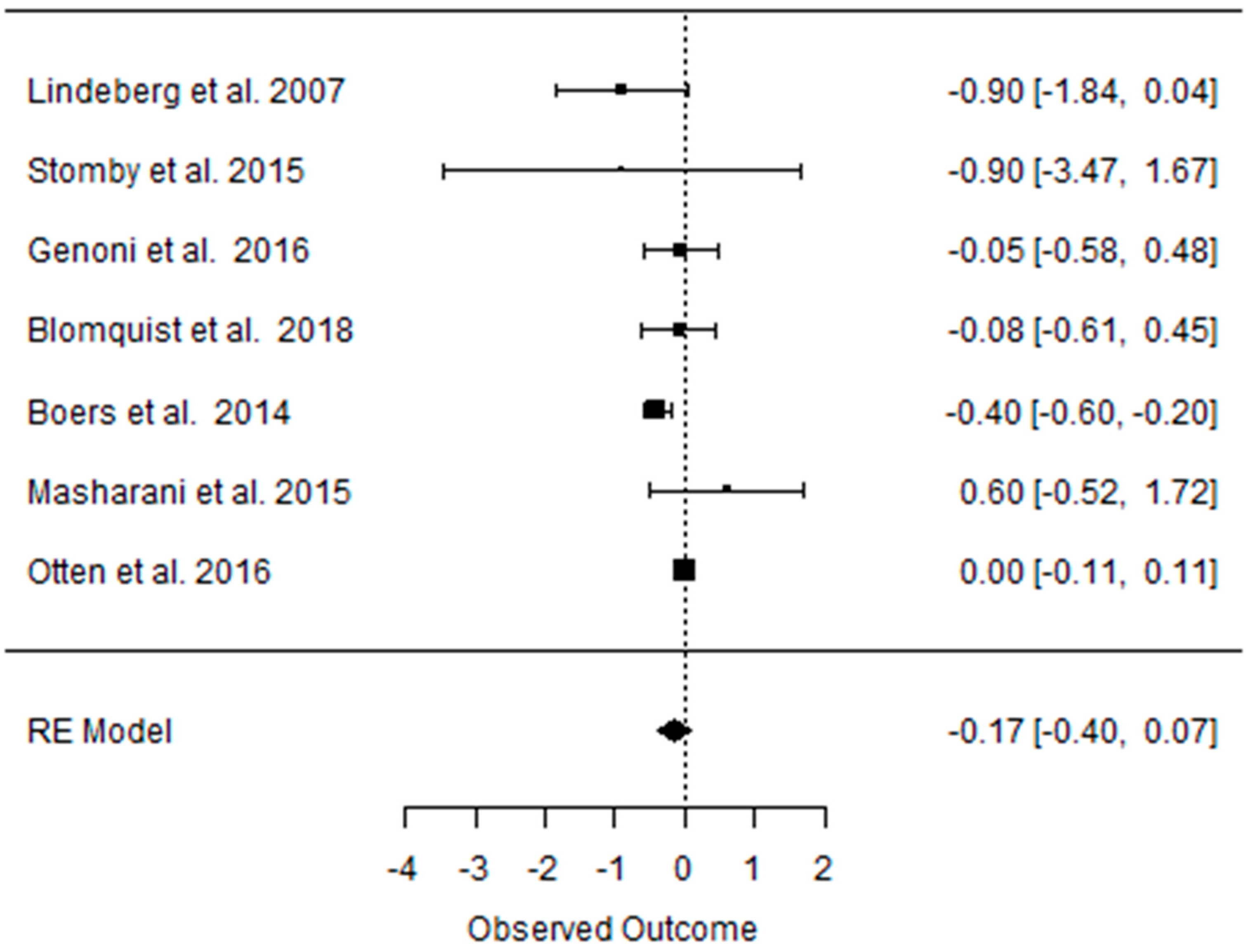
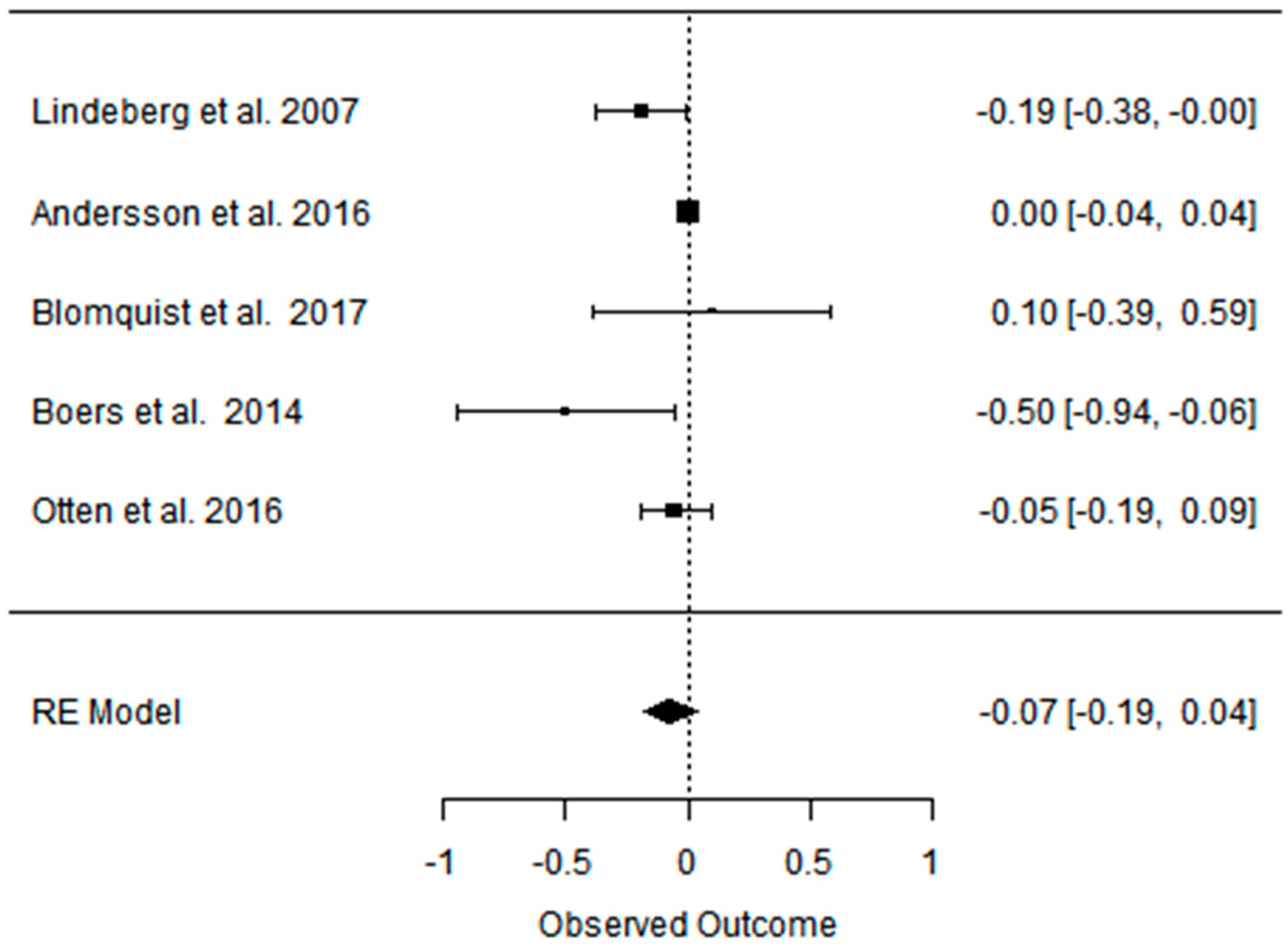
3.5. Effect of the Paleolithic Diet on Carbohydrates Metabolism (Fasting Plasma Glucose, Fasting Plasma Insulin, HOMA-IR, and HbA1c) in the Short (up to 6 Months) and Long (over 6 Months) Term
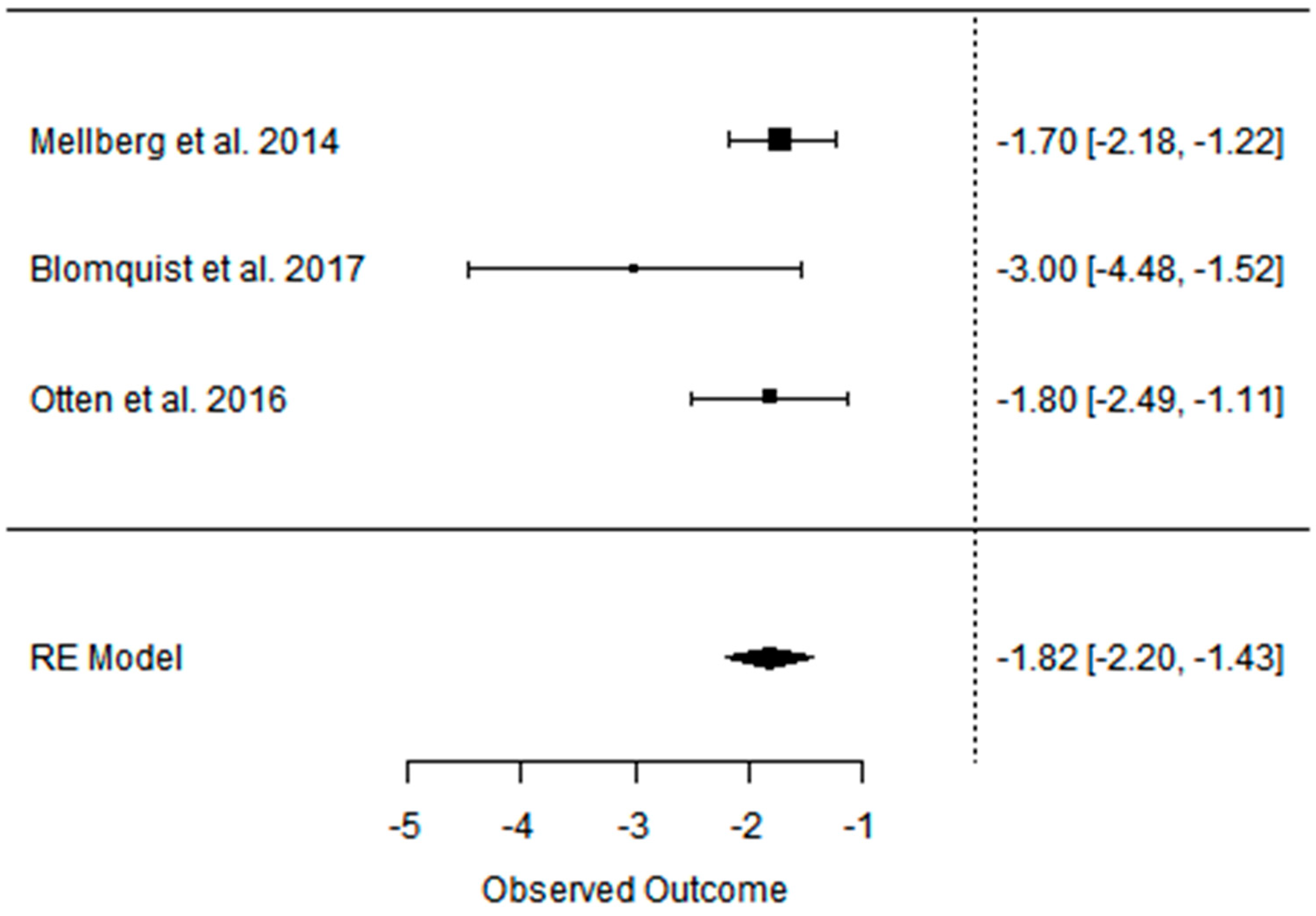
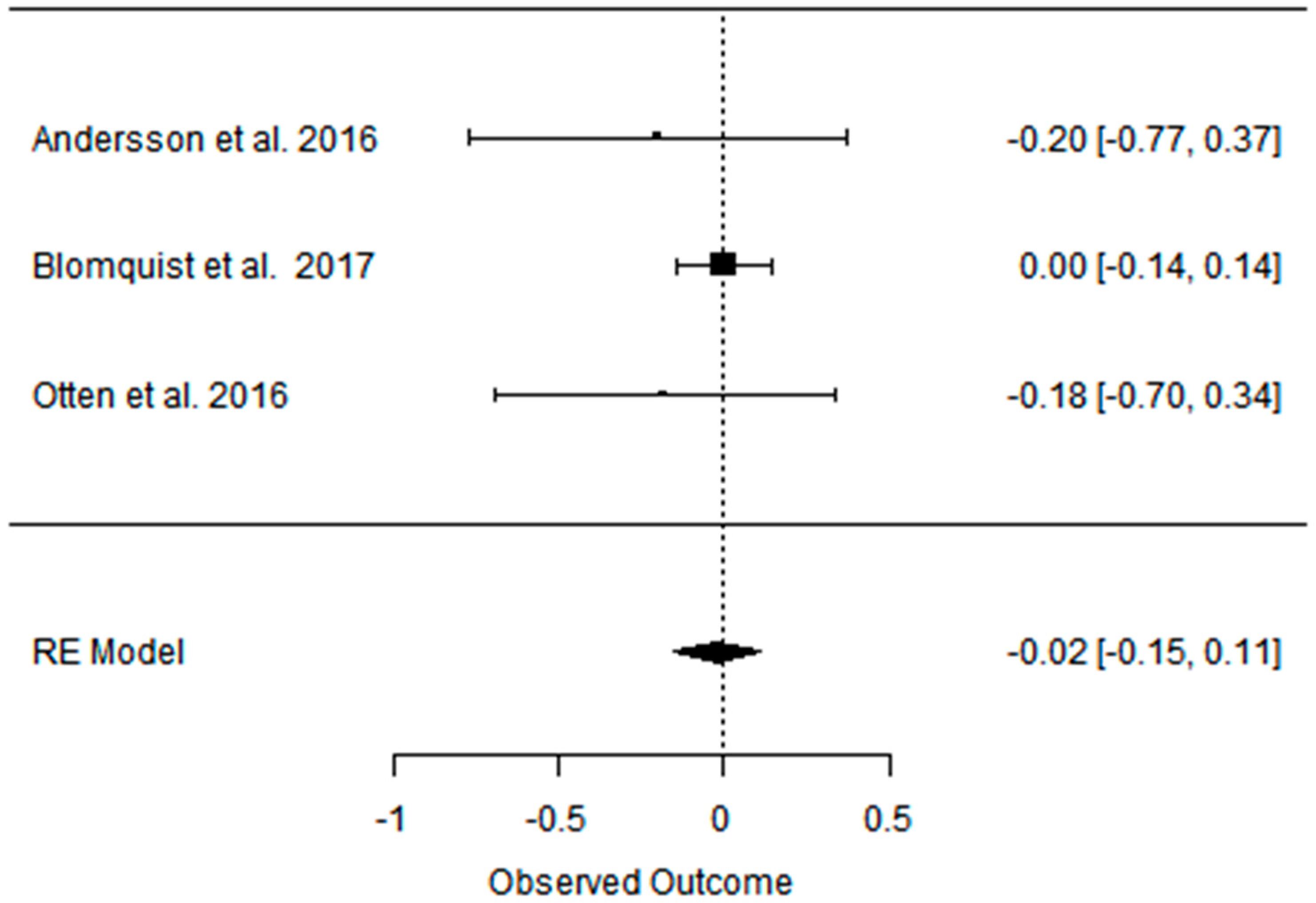
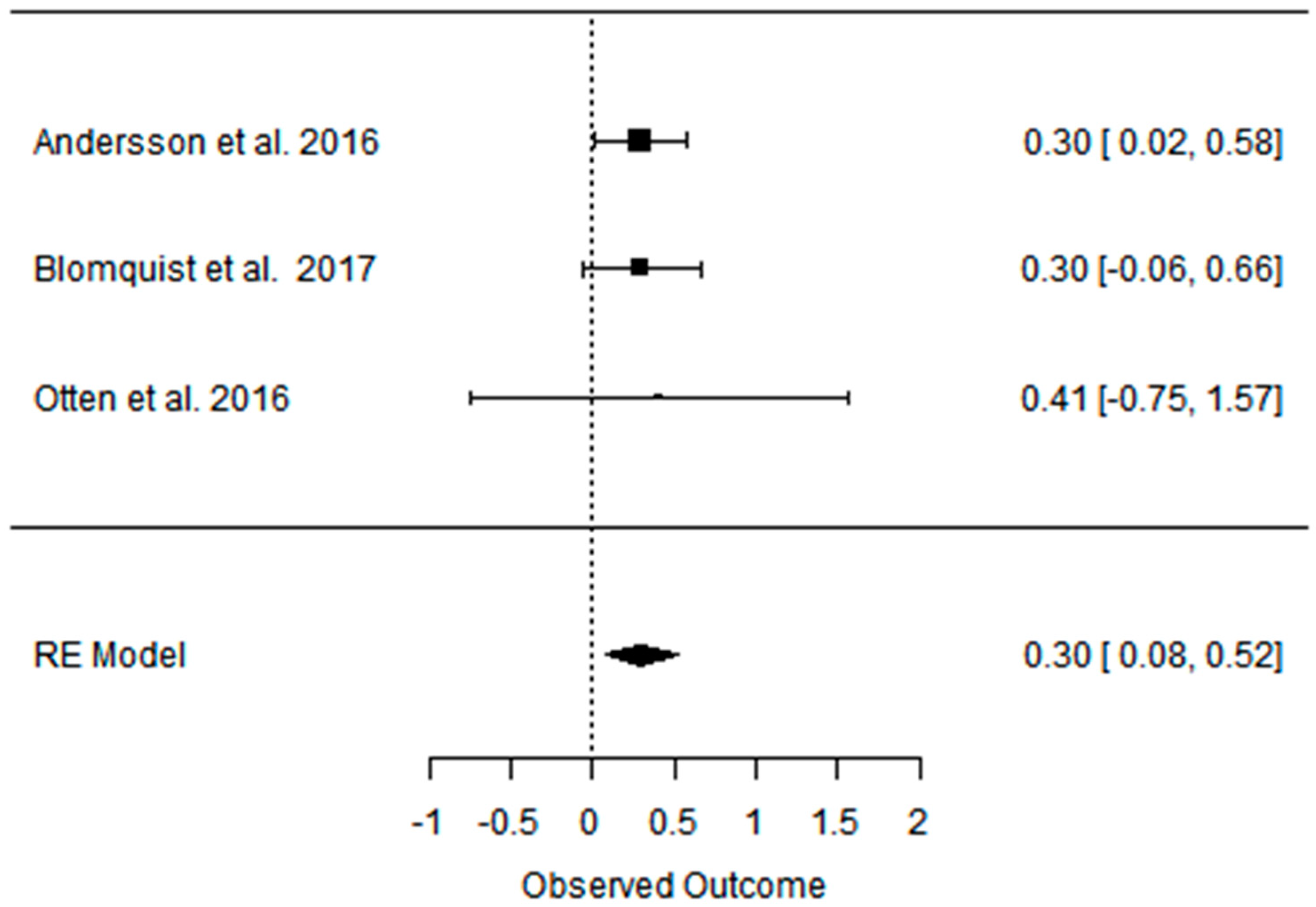
3.6. Effect of the Paleolithic Diet on Physical Capacity (Maximum Oxygen Uptake and Maximum Workload)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A



References
- Eaton, S.B.; Konner, M. Paleolithic nutrition: A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef]
- Milton, K. Hunter-gatherer diets—A different perspective. Am. J. Clin. Nutr. 2000, 71, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Wrangham, R.; Conklin-Brittain, N. Cooking as a biological trait. Comp. Biochem. Physiol. Mol. Integr. Physiol. 2003, 136, 35–46. [Google Scholar] [CrossRef]
- Wrangham, R.; Cheney, D.; Seyfarth, R.; Sarmiento, E. Shallow-water habitats as sources of fallback foods for hominins. Am. J. Phys. Anthropol. 2009, 140, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Konner, M.; Eaton, S.B. Paleolithic nutrition: Twenty-five years later. Nutr. Clin. Pract. 2010, 25, 594–602. [Google Scholar] [CrossRef]
- Eaton, S.B.; Eaton, S.B. Paleolithic vs. modern diets-selected pathophysiological implications. Eur. J. Nutr. 2000, 39, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L.; Miller, J.B.; Eaton, S.B.; Mann, N. Macronutrient estimations in hunter-gatherer diets. Am. J. Clin. Nutr. 2000, 72, 1589–1592. [Google Scholar] [CrossRef]
- Ugent, D.; Dillehay, T.; Ramírez, C. Potato remains from a late pleistocene settlement in southcentral Chile. Econ. Bot. 1987, 41, 17–27. [Google Scholar] [CrossRef]
- Mercader, J. Mozambican Grass Seed Consumption during the Middle Stone Age. Science 2009, 326, 1680–1683. [Google Scholar] [CrossRef]
- Henry, A.G.; Brooks, A.S.; Piperno, D.R. Microfossils in calculus demonstrate consumption of plants and cooked foods in Neanderthal diets. Proc. Natl. Acad. Sci. USA 2011, 108, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Henry, A.G.; Ungar, P.S.; Passey, B.H.; Sponheimer, M.; Rossouw, L.; Bamford, M.K.; Sandberg, P.A.; De Ruiter, D.J.; Berger, L.R. The diet of Australopithecus sediba. Nat. Cell Biol. 2012, 487, 90–93. [Google Scholar] [CrossRef]
- Revedin, A.; Aranguren, B.; Becattini, R.; Longo, L.; Marconi, E.; Lippi, M.M.; Skakun, N.; Sinitsyn, A.; Spiridonova, E.; Svoboda, J. Thirty thousand-year-old evidence of plant food processing. Proc. Natl. Acad. Sci. USA 2010, 107, 18815–18819. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.L.; Thompson, A.L. Beyond the Paleolithic prescription: Incorporating diversity and flexibility in the study of human diet evolution. Nutr. Rev. 2013, 71, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Holden, C.; Mace, R. Phylogenetic analysis of the evolution of lactose digestion in adults. Hum. Biol. 1997, 69, 606–628. [Google Scholar] [CrossRef] [PubMed]
- Knight, C. Most people are simply not designed to eat pasta: Evolutionary explanations for obesity in the low carbohydrate diet movement. Public Underst. Sci. 2011, 20, 706–719. [Google Scholar] [CrossRef]
- Perry, G.H.; Dominy, N.J.; Claw, K.G.; Lee, A.S.; Fiegler, H.; Redon, R.; Werner, J.; Villanea, F.A.; Mountain, J.L.; Misra, R.; et al. Diet and the evolution of human amylase gene copy number variation. Nat. Genet. 2007, 39, 1256–1260. [Google Scholar] [CrossRef] [PubMed]
- De Menezes, E.V.A.; Sampaio, H.A.D.C.; Carioca, A.A.F.; Parente, N.A.; Brito, F.O.; Moreira, T.M.M.; De Souza, A.C.C.; Arruda, S.P.M. Influence of Paleolithic diet on anthropometric markers in chronic diseases: Systematic review and meta-analysis. Nutr. J. 2019, 18, 41. [Google Scholar] [CrossRef]
- Frassetto, L.A.; Schloetter, M.; Mietus-Snyder, M.; Morris, R.C.; Sebastian, A. Metabolic and physiologic improvements from consuming a Paleolithic, hunter-gatherer type diet. Eur. J. Clin. Nutr. 2009, 63, 947–955. [Google Scholar] [CrossRef]
- Jabr, F. How to Really Eat Like a Hunter-Gatherer: Why the Paleo Diet Is Half-Baked. Scientific American. 2013. Available online: https://www.scientificamerican.com/article/why-paleo-diet-half-baked-how-hunter-gatherer-really-eat/ (accessed on 24 February 2021).
- Zuk, M. Paleofantasy: What Evolution Really Tells Us about Sex, Diet, and How We Live; WW Norton & Co.: New York, NY, USA, 2013; pp. 52–65. [Google Scholar]
- Eaton, S.B.; Konner, M.; Shostak, M. Stone Agers in the fast lane: Chronic degenerative diseases in evolutionary perspective. Am. J. Med. 1988, 84, 739–749. [Google Scholar] [CrossRef]
- Cohen, M. Health and the Rise of Civilization; Yale University Press: New Haven, CT, USA, 1989; pp. 131–141. [Google Scholar]
- Jansson, B. Dietary, total body, and intracellular potassium-to-sodium ratios and their influence on cancer. Cancer Detect. Prev. 1990, 14, 563–565. [Google Scholar]
- Eaton, S.; Cordain, L. Evolutionary Aspects of Diet: Old Genes, New Fuels. World Rev. Nutr. Diet. 1997, 81, 26–37. [Google Scholar] [CrossRef]
- O’Keefe, H.; Cordain, L. Cardiovascular disease resulting from a diet and lifestyle at odds with our Paleolithic genome: How to become a 21st-century hunter-gatherer. Mayo Clin. Proc. 2004, 79, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, A.; Frassetto, L.A.; Sellmeyer, D.E.; Merriam, R.L.; Morris, R.C., Jr. Estimation of the net acid load of the diet of ancestral preagricultural Homo sapiens and their hominid ancestors. Am. J. Clin. Nutr. 2002, 76, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Lindeberg, S.; Jonsson, T.; Granfeldt, Y.; Borgstrand, E.; Soffman, J.; Sjostrom, K.; Ahren, B. A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischemic heart disease. Diabetologia 2007, 50, 1795–1807. [Google Scholar] [CrossRef] [PubMed]
- Irish, A.K.; Erickson, C.M.; Wahls, T.L.; Snetselaar, L.G.; Darling, W.G. Randomized control trial evaluation of a modifed Paleolithic dietary intervention in the treatment of relapsing-remitting multiple sclerosis: A pilot study. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 1–18. [Google Scholar]
- Kidd, P.M. Neurodegeneration from mitochondrial insufficiency: Nutrients, stem cells, growth factors, and prospects for brain rebuilding using integrative management. Altern. Med. Rev. J. Clin. Ther. 2005, 10, 268–293. [Google Scholar]
- Torkildsen, Ø.; Løken-Amsrud, K.I.; Wergeland, S.; Holmøy, T.; Myhr, K.-M. Fat-soluble vitamins as disease modulators in multiple sclerosis. Acta Neurol. Scand. 2013, 196, 16–23. [Google Scholar] [CrossRef]
- Sanoobar, M.; Dehghan, P.; Khalili, M.; Azimi, A.; Seifar, F. Coenzyme Q10 as a treatment for fatigue and depression in multiple sclerosis patients: A double blind randomized clinical trial. Nutr. Neurosci. 2016, 19, 138–143. [Google Scholar] [CrossRef]
- Eaton, S.B.; Eaton, S.B., III; Sinclair, A.J.; Cordain, L.; Mann, N.J. Dietary intake of long-chain polyunsaturated fatty acids during the Paleolithic. World. Rev. Nutr. Diet. 1998, 83, 12–23. [Google Scholar]
- Kuipers, R.S.; Luxwolda, M.F.; Dijck-Brouwer, D.A.J.; Eaton, S.B.; Crawford, M.A.; Cordain, L.; Muskiet, F.A.J. Estimated macronutrient and fatty acid intakes from an East African Paleolithic diet. Br. J. Nutr. 2010, 104, 1666–1687. [Google Scholar] [CrossRef]
- Cordain, L.; Friel, J. The Paleo Diet for Athletes: The Ancient Nutritional Formula for Peak Athletic Performance; Rodale Books: Emmaus, PA, USA, 2012. [Google Scholar]
- Holt, S.H.; Miller, J.C.; Petocz, P.; Farmakalidis, E. A satiety index of common foods. Eur. J. Clin. Nutr. 1995, 49, 675–690. [Google Scholar]
- Ryberg, M.; Sandberg, S.; Mellberg, C.; Stegle, O.; Lindahl, B.; Larsson, C.; Hauksson, J.; Olsson, T. A Palaeolithic-type diet causes strong tissue-specific effects on ectopic fat deposition in obese postmenopausal women. J. Intern. Med. 2013, 274, 67–76. [Google Scholar] [CrossRef]
- Accurso, A.; Bernstein, R.K.; Dahlqvist, A.; Draznin, B.; Feinman, R.D.; Fine, E.J.; Gleed, A.; Jacobs, D.B.; Larson, G.; Lustig, R.H.; et al. Dietary carbohydrate restriction in type 2 diabetes mellitus and metabolic syndrome: Time for a critical appraisal. Nutr. Metab. 2008, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Pastore, R.L.; Brooks, J.T.; Carbone, J.W. Paleolithic nutrition improves plasma lipid concentrations of hypercholesterolemic adults to a greater extent than traditional heart-healthy dietary recommendations. Nutr. Res. 2015, 35, 474–479. [Google Scholar] [CrossRef]
- Boraxbekk, C.-J.; Stomby, A.; Ryberg, M.; Lindahl, B.; Larsson, C.; Nyberg, L.; Olsson, T. Diet-Induced Weight Loss Alters Functional Brain Responses during an Episodic Memory Task. Obes. Facts 2015, 8, 261–272. [Google Scholar] [CrossRef]
- Otten, J.; Mellberg, C.; Ryberg, M.; Sandberg, S.; Kullberg, J.; Lindahl, B.; Larsson, C.; Hauksson, J.; Olsson, T. Strong and persistent effect on liver fat with a Paleolithic diet during a two-year intervention. Int. J. Obes. 2016, 40, 747–753. [Google Scholar] [CrossRef]
- Otten, J.; Stomby, A.; Waling, M.; Isaksson, A.; Tellström, A.; Lundin-Olsson, L.; Brage, S.; Ryberg, M.; Svensson, M.; Olsson, T. Benefits of a Paleolithic diet with and without supervised exercise on fat mass, insulin sensitivity, and glycemic control: A randomized controlled trial in individuals with type 2 diabetes. Diabetes Metab. Res. Rev. 2017, 33, e2828. [Google Scholar] [CrossRef] [PubMed]
- Otten, J.; Stomby, A.; Waling, M.; Isaksson, A.; Söderström, I.; Ryberg, M.; Svensson, M.; Hauksson, J.; Olsson, T. A heterogeneous response of liver and skeletal muscle fatt the combination of a Paleolithic diet and exercise in obese individuals with type 2 diabetes: A randomized controlled trial. Diabetes 2018, 61, 1548–1559. [Google Scholar]
- Otten, J.; Andersson, J.; Ståhl, J.; Stomby, A.; Saleh, A.; Waling, M.; Ryberg, M.; Hauksson, J.; Svensson, M.; Johansson, B.; et al. Exercise Training Adds Cardiometabolic Benefits of a Paleolithic Diet in Type 2 Diabetes Mellitus. J. Am. Heart Assoc. 2019, 8, e010634. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.; Mellberg, C.; Otten, J.; Ryberg, M.; Rinnström, D.; Larsson, C.; Lindahl, B.; Hauksson, J.; Johansson, B.; Olsson, T. Left ventricular remodeling changes without concomitant loss of myocardial fat after long-term dietary intervention. Int. J. Cardiol. 2016, 216, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Stomby, A.; Simonyte, K.; Mellberg, C.; Ryberg, M.; Stimson, R.H.; Larsson, C.; Lindahl, B.; Andrew, R.; Walker, B.R.; Olsson, T. Diet-induced weight loss has chronic tissue-specific effects on glucocorticoid metabolism in overweight postmenopausal women. Int. J. Obes. 2015, 39, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Blomquist, C.; Alvehus, M.; Burén, J.; Ryberg, M.; Larsson, C.; Lindahl, B.; Mellberg, C.; Söderström, I.; Chorell, E.; Olsson, T. Attenuated Low-Grade Inflammation Following Long-Term Dietary Intervention in Postmenopausal Women with Obesity. Obesesity 2017, 25, 892–900. [Google Scholar] [CrossRef]
- Blomquist, C.; Chorell, E.; Ryberg, M.; Mellberg, C.; Worrsjö, E.; Makoveichuk, E.; Larsson, C.; Lindahl, B.; Olivecrona, G.; Olsson, T. Decreased lipogenesis-promoting factors in adipose tissue in postmenopausal women with overweight on a Paleolithic-type diet. Eur. J. Nutr. 2018, 57, 2877–2886. [Google Scholar] [CrossRef] [PubMed]
- Mellberg, C.; Sandberg, S.; Ryberg, M.; Eriksson, M.; Brage, S.; Larsson, C.; Olsson, T.; Lindahl, B. Long-term effects of a Palaeolithic-type diet in obese postmenopausal women: A 2-year randomized trial. Eur. J. Clin. Nutr. 2014, 68, 350–357. [Google Scholar] [CrossRef]
- Stomby, A.; Otten, J.; Ryberg, M.; Nyberg, L.; Olsson, T.; Boraxbekk, C.-J. A Paleolithic Diet with and without Combined Aerobic and Resistance Exercise Increases Functional Brain Responses and Hippocampal Volume in Subjects with Type 2 Diabetes. Front. Aging Neurosci. 2017, 9, 391. [Google Scholar] [CrossRef]
- Fontes-Villalba, M.; Lindeberg, S.; Granfeldt, Y.; Knop, F.K.; Memon, A.A.; Carrera-Bastos, P.; Picazo, Ó.; Chanrai, M.; Sunquist, J.; Sundquist, K.; et al. Palaeolithic diet decreases fasting plasma leptin concentrations more than a diabetes diet in patients with type 2 diabetes: A randomised cross-over trial. Cardiovasc. Diabetol. 2016, 15, 80. [Google Scholar] [CrossRef]
- Masharani, U.; Sherchan, P.; Schloetter, M.; Stratford, S.; Xiao, A.; Sebastian, A.; Nolte Kennedy, M.; Frassetto, L. Metabolic and physiologic effects from consuming a hunter-gatherer (Paleolithic)-type diet in type 2 diabetes. Eur. J. Clin. Nutr. 2015, 69, 944–948. [Google Scholar] [CrossRef]
- Boers, I.; Muskiet, F.A.; Berkelaar, E.; Schut, E.; Penders, R.; Hoenderdos, K.; Wichers, H.J.; Jong, M.C. Favourable effects of consuming a Palaeolithic-type diet on characteristics of the metabolic syndrome: A randomized controlled pilot-study. Lipids Health Dis. 2014, 13, 160. [Google Scholar] [CrossRef]
- Genoni, J.L.; Lyons-Wall, P.; Devine, A. Cardiovascular, metabolic effects and dietary composition of ad- libitum Paleolithic vs. Australian Guide to Healthy Eating Diets in Healthy Women: A 4-week randomized trial. Nutrients 2016, 8, 314. [Google Scholar] [CrossRef]
- Smith, M.M.; Trexler, E.T.; Sommer, A.J.; Starkoff, B.E.; Devor, S.T. Unrestricted Paleolithic diet is associated with unfavorable changes to blood lipids in healthy subjects. Int. J. Exerc. Sci. 2014, 7, 128–139. [Google Scholar]
- Österdahl, M.; Kocturk, T.; Koochek, A.; Wändell, P. Effects of a short-term intervention with a Paleolithic diet in healthy volunteers. Eur. J. Clin. Nutr. 2008, 62, 682–685. [Google Scholar] [CrossRef]
- Bolla, A.M.; Caretto, A.; Laurenzi, A.; Scavini, M.; Piemonti, L. Low-Carb and Ketogenic Diets in Type 1 and Type 2 Diabetes. Nutrients 2019, 11, 962. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-K.; Borer, K.; Lin, P.-J. Low-Carbohydrate-High-Fat Diet: Can it Help Exercise Performance? J. Hum. Kinet. 2017, 56, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of vary-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Retelny, V.S. Can Extremely Fat-Restricted or High-Fat Diets Be Effective-and Safe-for Weight Loss? Food Nutr. 2015. Available online: www.foodandnutrition.org/January-February-2016/Oil-Free-Plant-based-Diet-Versus-Ketogenic-Diet/ (accessed on 21 June 2017).
- Burke, L.M.; Hawley, J.A. Effects of short-term fat adaptation on metabolism and performance of prolonged exercise. Med. Sci. Sports Exerc. 2002, 13, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zinn, C.; Wood, M.; Williden, M.; Chatterton, S.; Maunder, E. Ketogenic diet benefits body composition and well-being but not performance in a pilot case study of New Zealand endurance athletes. J. Int. Soc. Sports Nutr. 2017, 14, 1–9. [Google Scholar] [CrossRef]
- Walker, J.L.; Heigenhauser, G.J.F.; Hultman, E.; Spriet, L.L. Dietary carbohydrate, muscle glycogen content, and endurance performance in well-trained women. J. Appl. Physiol. 2000, 88, 2151–2158. [Google Scholar] [CrossRef]
- Rhyu, H.-S.; Cho, S.-Y. The effect of weight loss by ketogenic diet on the body composition, performance-related physical fitness factors and cytokines of Taekwondo athletes. J. Exerc. Rehabil. 2014, 10, 326–331. [Google Scholar] [CrossRef]
- Volek, J.S.; Freidenreich, D.J.; Saenz, C.; Kunces, L.J.; Creighton, B.C.; Bartley, J.M. Metabolic characteristics of keto-adapted ultra- endurance runners. Metabolism 2016, 65, 100–110. [Google Scholar] [CrossRef]
- Zajac, A.; Poprzecki, S.; Maszczyk, A.; Czuba, M.; Michalczyk, M.; Zydek, G. The Effects of a Ketogenic Diet on Exercise Metabolism and Physical Performance in Off-Road Cyclists. Nutrients 2014, 6, 2493–2508. [Google Scholar] [CrossRef] [PubMed]
- Bujko, J.; Kowalski, Ł.M. The question of ergogenic potential of the Paleolithic diet. Trends Sport Sci. 2014, 21, 213–219. [Google Scholar]
- Popp, C.J.; Brown, M.M.B.; Bridges, W.C.; Jesch, E.D. The Effectiveness of MyPlate and Paleolithic-based Diet Recommendations, both with and without Exercise, on Aerobic Fitness, Muscular Strength and Anaerobic Power in Young Women: A Randomized Clinical Trial. Int. J. Exerc. Sci. 2018, 11, 921–933. [Google Scholar] [PubMed]
- Manousou, S.; Stål, M.; Larsson, C.; Mellberg, C.; Lindahl, B.; Eggertsen, R.; Hulthén, L.; Olsson, T.; Ryberg, M.; Sandberg, S.; et al. A Paleolithic-type diet results in iodine deficiency: A 2-year randomized trial in postmenopausal obese women. Eur. J. Clin. Nutr. 2018, 72, 124–129. [Google Scholar] [CrossRef]
- Tufanaru, C.; Munn, Z.; Stephenson, M.; Aromataris, E. Fixed or random effects meta-analysis? Common methodological issues in systematic reviews of effectiveness. Int. J. Evid. Based Health 2015, 13, 196–207. [Google Scholar] [CrossRef]
- Markofski, M.M.; Jennings, K.; Dolan, C.; Davies, N.A.; LaVoy, E.C.; Ryan, E.J.; Carrillo, A.E. Single-Arm 8-Week Ad Libitum Self-Prepared Paleo Diet Reduces Cardiometabolic Disease Risk Factors in Overweight Adults. Am. J. Lifestyle Med. 2019. [Google Scholar] [CrossRef]
- Bisht, B.; Darling, W.G.; White, E.C.; White, K.A.; Shivapour, E.T.; Zimmerman, M.B.; Wahls, T.L. Effects of a multimodal intervention on gait andbalance of subjects with progressive multiple sclerosis: A prospective longitudinal pilot study. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 79–93. [Google Scholar]
- Melnik, B.C.; John, S.M.; Schmitz, G. Over-stimulation of insulin/IGF-1 signaling by western diet may promote diseases of civilization: Lessons learnt from laron syndrome. Nutr. Metab. 2011, 8, 41. [Google Scholar] [CrossRef]
- Melnik, B.C.; John, S.M.; Carrera-Bastos, P.; Cordain, L. The impact of cow’s milk-mediated mTORC1-signaling in the initiation and progression of prostate cancer. Nutr. Metab. 2012, 9, 74–93. [Google Scholar] [CrossRef]
- Ghaedi, E.; Mohammadi, M.; Mohammadi, H.; Ramezani-Jolfaie, N.; Malekzadeh, J.; Hosseinzadeh, M.; Salehi-Abargouei, A. Effects of a Paleolithic Diet on Cardiovascular Disease Risk Factors: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2019, 10, 634–646. [Google Scholar] [CrossRef]
- Manheimer, E.W.; van Zuuren, E.J.; Fedorowicz, Z.; Pijl, H. Paleolithic nutrition for metabolic syndrome: Systematic review and meta-analysis. Am. J. Clin. Nutr. 2015, 102, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, T.; Granfeldt, Y.; Lindeberg, S.; Hallberg, A.-C. Subjective satiety and other experiences of a Paleolithic diet compared to a diabetes diet in patients with type 2 diabetes. Nutr. J. 2013, 12, 105. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, T.; Granfeldt, Y.; Ahrén, B.; Branell, U.-C.; Pålsson, G.; Hansson, A.; Söderström, M.; Lindeberg, S. Beneficial effects of a Paleolithic diet on cardiovascular risk factors in type 2 diabetes: A randomized cross-over pilot study. Cardiovasc. Diabetol. 2009, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Bueno, N.B.; de Melo, I.S.V.; de Oliveira, S.L.; da Rocha Ataide, T. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomized controlled trials. Br. J. Nutr. 2013, 110, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.; Swendener, A.; Shaw, B.; Shaw, I. Comparison of anthropometric and metabolic responses to a short-term carbohydrate restricted diet and exercise versus a traditional diet and exercise. Afr. J. Phys. Health Educ. Recreat. Dance 2010, 16, 535–544. [Google Scholar] [CrossRef]
- Hession, M.; Rolland, C.; Kulkarni, U.; Wise, A.; Broom, J. Systematic review of randomized controlled trials of low-carbohydrate vs. low-fat/low-calorie diets in the management of obesity and its comorbidities. Obes. Rev. 2009, 10, 36–50. [Google Scholar] [CrossRef]
- Hu, T.; Mills, K.T.; Yao, L.; Demanelis, K.; Eloustaz, M.; Yancy, W.J., Jr.; Kelly, T.N.; He, J.; Bazzano, L.A. Effects of low-carbohydrate diets versus low-fat diets on metabolic risk factors: A meta-analysis of randomized controlled clinical trials. Am. J. Epidemiol. 2012, 176, 44–54. [Google Scholar] [CrossRef]
- Palmer, B.F.; Clegg, D.J. Achieving the Benefits of a High-Potassium, Paleolithic Diet, Without the Toxicity. Mayo Clin. Proc. 2016, 91, 496–508. [Google Scholar] [CrossRef]
- Jamka, M.; Kulczynski, B.; Juruć, A.; Gramza-Michałowska, A.; Stokes, C.S.; Walkowiak, J. The Effect of the Paleolithic Diet vs. Healthy Diets on Glucose and Insulin Homeostasis: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Med. 2020, 9, 296. [Google Scholar] [CrossRef]
- Dinu, M.; Pagliai, G.; Angelino, D.; Rosi, A.; Dall’Asta, M.; Bresciani, L.; Ferraris, C.; Guglielmetti, M.; Godos, J.; Del Bo’, C.; et al. Effects of Popular Diets on Anthropometric and Cardiometabolic Parameters: An Umbrella Review of Meta-Analyses of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 815–833. [Google Scholar] [CrossRef]
- Paoli, A.; Grimaldi, K.; D’Agostino, D.; Cenci, L.; Moro, T.; Bianco, A.; Palma, A. Ketogenic diet does not affect strength performance in elite artistic gymnasts. J. Int. Soc. Sports Nutr. 2012, 9, 34. [Google Scholar] [CrossRef]
- Michalczyk, M.; Zajac, A.; Mikolajec, K.; Zydek, G.; Langfort, J. No Modification in Blood Lipoprotein Concentration but Changes in Body Composition After 4 Weeks of Low Carbohydrate Diet (LCD) Followed by 7 Days of Carbohydrate Loading in Basketball Players. J. Hum. Kinet. 2018, 65, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Antonio, J.; Ellerbroek, A.; Silver, T.; Vargas, L.; Peacock, C.A. The effects of a high protein diet on indices of health and body composition—A crossover trial in resistance-trained men. J. Int. Soc. Sports Nutr. 2016, 13, 41–54. [Google Scholar] [CrossRef]
- Kang, J.; Ratamess, N.A.; Faigenbaum, A.D.; Bush, J.A. Ergogenic Properties of Ketogenic Diets in Normal-Weight Individuals: A Systematic Review. J. Am. Coll. Nutr. 2020, 39, 665–675. [Google Scholar] [CrossRef]
- Paoli, A.; Bianco, A.; Grimaldi, K.A. The ketogenic diet and sport: A possible marriage? Exerc. Sport Sci. Rev. 2015, 43, 153–162. [Google Scholar] [CrossRef]
- Bazzano, L.A.; Hu, T.; Reynolds, K.; Yao, L.; Bunol, C.; Liu, Y.; Chen, C.S.; Klag, M.J.; Whelton, P.K.; He, J. Effects of low-carbohydrate and low-fat diets: A randomized trial. Ann. Intern. Med. 2014, 161, 309–318. [Google Scholar] [CrossRef] [PubMed]
- McSwiney, F.T.; Wardrop, B.; Hyde, P.N.; Lafountain, R.A.; Volek, J.S.; Doyle, L. Keto-adaptation enhances exercise performance and body composition responses to training in endurance athletes. Metabolism 2018, 8, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, J.C.; Wood, R.J.; Davidson, P.W.; Collins, S.M.; Matthews, T.D.; Gregory, S.M.; Paolone, V.J. Effects of a Short-Term Carbohydrate-Restricted Diet on Strength and Power Performance. J. Strength Cond. Res. 2013, 27, 2255–2262. [Google Scholar] [CrossRef]
- McSwiney, F.T.; Fusco, B.; McCabe, L.; Lombard, A.; Crowley, P.; Walsh, J.; Hone, M.; Egan, B. Changes in body composition and substrate utilization after a short- term ketogenic diet in endurance- trained males. Biol. Sport 2021, 38, 145–152. [Google Scholar] [CrossRef]
- Vargas, S.; Romance, R.; Petro, J.L.; Bonilla, D.A.; Galancho, I.; Espinar, S.; Kreider, R.B.; Benitez-Pores, J. efficacy of ketogenic diet on body composition during resistance training in trained men: A randomized controlled trail. J. Int. Soc. Sports Nutr. 2018, 15, 31. [Google Scholar] [CrossRef]
- Phinney, S.D. Ketogenic diets and physical performance. Nutr. Metab. 2004, 1, 2. [Google Scholar] [CrossRef]
- Waldman, H.S.; Krings, B.M.; Basham, S.A.; Smith, J.E.W.; Fountain, B.J.; McAllister, M.J. Effects of a 15-Day Low Carbohydrate, High-Fat Diet in Resistance-Trained Men. J. Strength Cond. Res. 2018, 32, 3103–3111. [Google Scholar] [CrossRef]
- Bergman, B.C.; Butterfield, G.E.; Wolfel, E.E.; Lopaschuk, G.D.; Casazza, G.A.; Horning, M.A.; Brooks, G.A. Muscle net glucose uptake and glucose kinetics after endurance training in men. Am. J. Physiol. Content 1999, 277, E81–E92. [Google Scholar] [CrossRef]
- Westman, E.C.; Feinman, R.D.; Mavropoulos, J.C.; Vernon, M.C.; Volek, J.S.; Wortman, J.A.; Yancy, W.S.; Phinney, S.D. Low-carbohydrate nutrition and metabolism. Am. J. Clin. Nutr. 2007, 86, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.M.; Van Loon, L.J.C.; Hawley, J.A. Postexercise muscle glycogen resynthesis in humans. J. Appl. Physiol. 2017, 122, 1055–1067. [Google Scholar] [CrossRef]
- Philp, A.; Hargreaves, M.; Baar, K. More than a store: Regulatory roles for glycogen in skeletal muscle adaptation to exercise. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E1343–E1351. [Google Scholar] [CrossRef]
- Phillips, S.M. A Brief Review of Critical Processes in Exercise-Induced Muscular Hypertrophy. Sports Med. 2014, 44, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Creer, A.; Gallagher, P.; Slivka, D.; Jemiolo, B.; Fink, W.; Trappe, S. Influence of muscle glycogen availability on ERK1/2 and Akt signaling after resistance exercise in human skeletal muscle. J. Appl. Physiol. 2005, 99, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.P.; Hennessy, E. A review of the ketogenic diet for endurance athletes: Performance enhancer or placebo effect? J. Int. Soc. Sports Nutr. 2020, 17, 33. [Google Scholar] [CrossRef]
- Bartlett, J.D.; Hawley, J.A.; Morton, J.P. Carbohydrate availability and exercise training adaptation: Too much of a good thing? Eur. J. Sport Sci. 2015, 15, 3–12. [Google Scholar] [CrossRef]
- Phinney, S.; Bistrian, B.; Evans, W.; Gervino, E.; Blackburn, G. The human metabolic response to chronic ketosis without caloric restriction: Preservation of submaximal exercise capability with reduced carbohydrate oxidation. Metabolism 1983, 32, 769–776. [Google Scholar] [CrossRef]
- Shaw, D.M.; Merien, F.; Braakhuis, A.; Maunder, E.D.; Dulson, D.K. Effect of a Ketogenic Diet on Submaximal Exercise Capacity and Efficiency in Runners. Med. Sci. Sports Exerc. 2019, 51, 2135–2146. [Google Scholar] [CrossRef] [PubMed]
| Study and Year | Type of Diets/ Type of Study | Subjects (n)/Participants Characteristic/Age/ Duration of Intervention (Time) | Effect on Body Composition | Effect on Lipid Profile | Effect on Blood Pressure | Effect on Carbohydrates Metabolism | Effect on Athletic Performance |
|---|---|---|---|---|---|---|---|
| Andersson et al. 2016 [45] | PD vs. CD (NNR) ad libitum RCT, parallel | [70] healthy postmenopausal women with body mass index (BMI) ≥ 27 kg/m2 (overweigh/obese) long-term (24 months) | Partially positive: Both diet groups decreased their BM and BMI at 6 and 24 months without significant differences between groups. | No effect: No difference in cholesterol levels over time or between groups. | Partially positive: BP was reduced at 6 months but returned to baseline levels at 24 months. | No effect: No differences were observed over time or between groups regarding fasting glucose, insulin concentrations and HOMA-IR. | Not analyzed |
| Boers et al. 2014 [53] | PD vs. CD (Dutch Health Cuncil) isocaloric | [32] men (n = 9) and women (n = 25) with at least two characteristics of the metabolic syndrome PD: 52 ± 10.2 CD: 55 ± 9 short-term (2 week) | Partially positive: In both groups, change was observed in WC. | Positive: Lower TC and TG and a higher mean HDL. The TC/HDL and TG/HDL ratios were lower in the PD compared to reference. | Positive: Lower SBP, DBP. | Positive: decreased fasting plasma insulin and HOMA-IR. | Not analyzed |
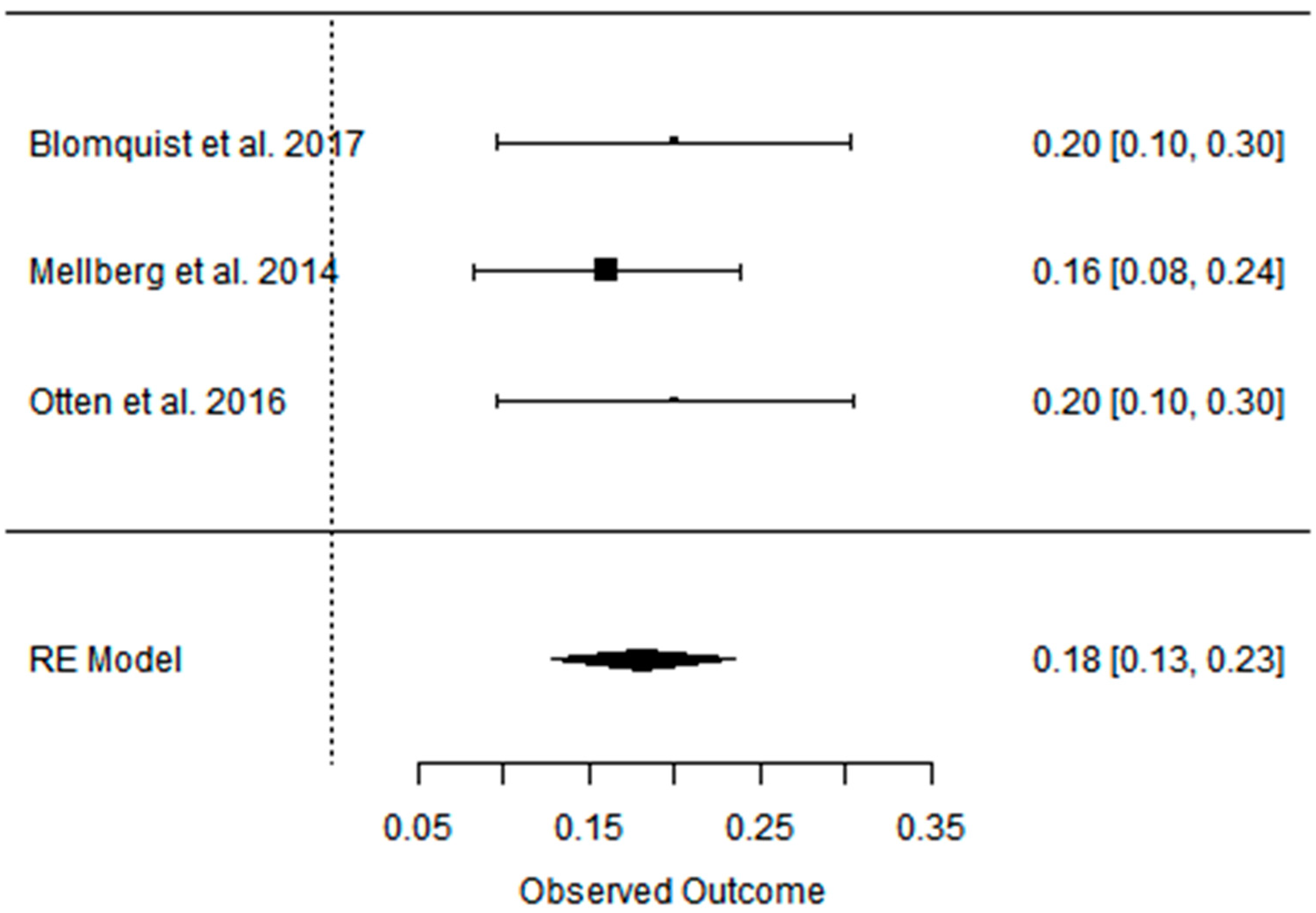
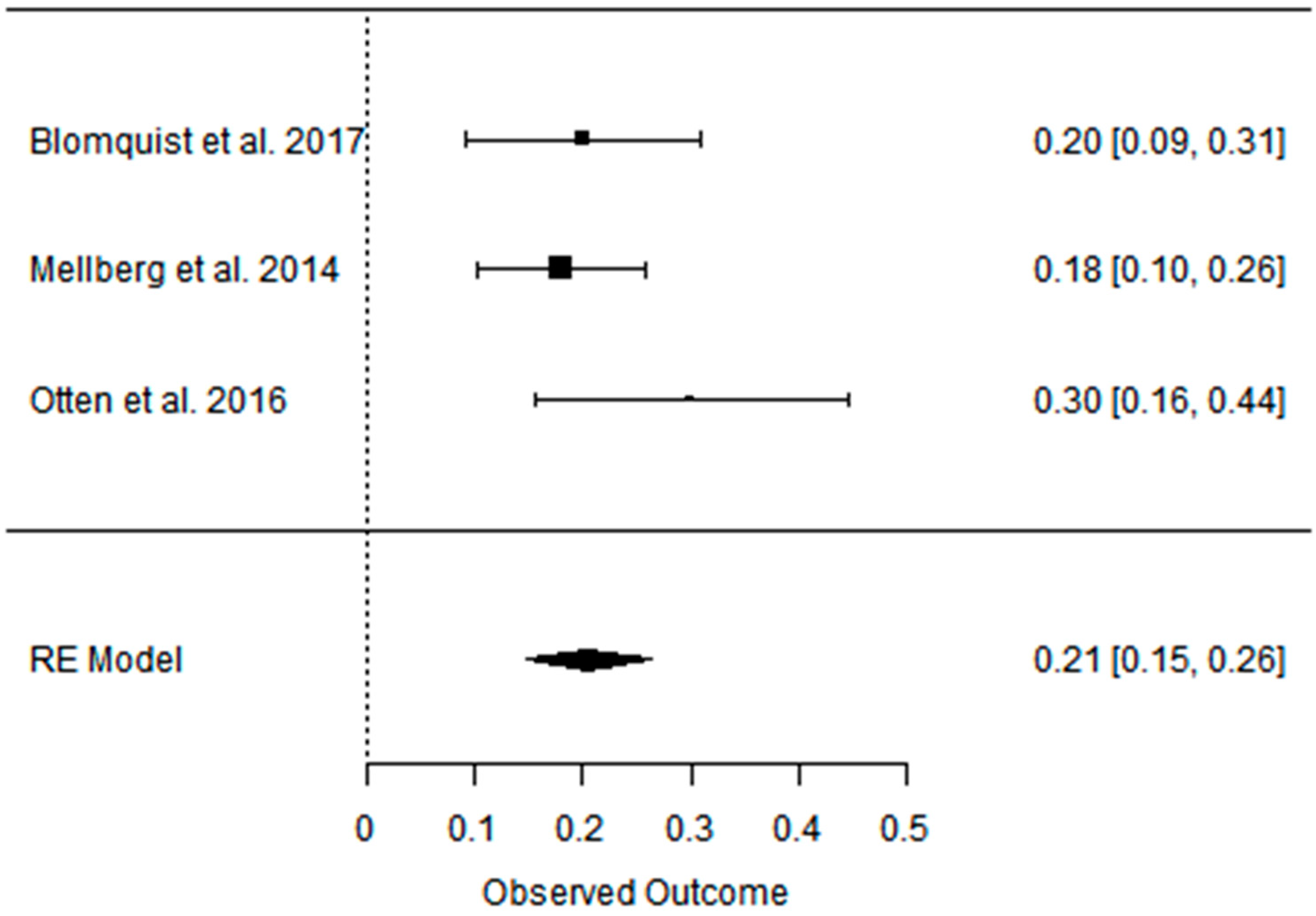
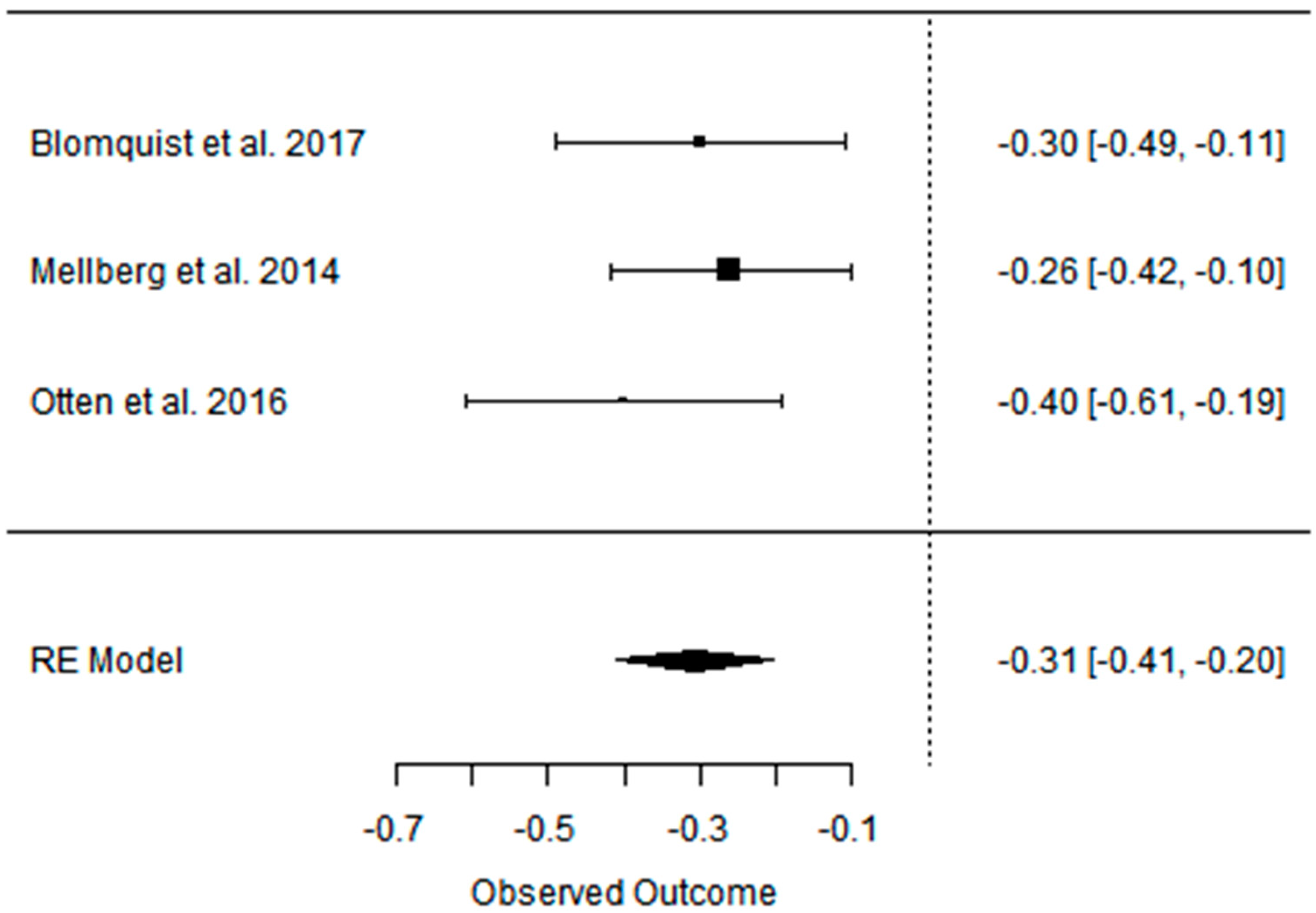
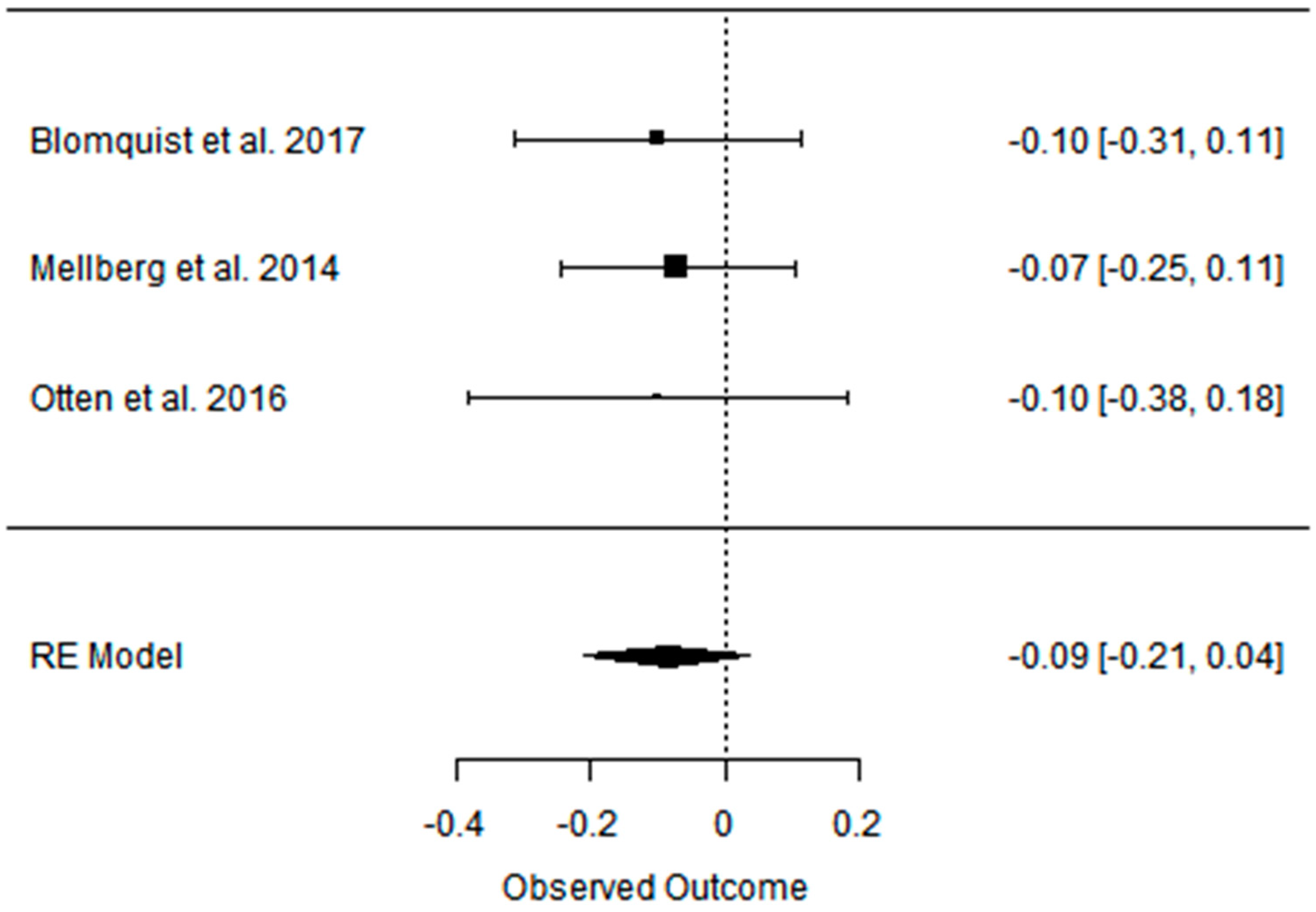
| Blomquist et al. 2017 [47] | PD vs. CD ad libitum RCT, parallel | [69] postmenopausal women with over- weight PD: 60 ± 5.6 CD: 61 ± 7 short-term (6 months); long-term (24 months) | Partially positive: Android fat decreased significantly more in the PD group during the first 6 months with weight maintenance at 24 months in both groups. | Positive: HDL levels increased in both groups between 6 and 24 months, LDL and TG levels decreased significantly in the PD group after 24 months. | Partially positive: In both groups, blood pressure decreased at 6 months, and the effect on DBP remained after 24 months in the PD group. | Partially positive: HOMA-IR decreased significantly in 6 months for PD group, rest of effect statistically insignificant. | Not analyzed |
| Blomquist et al. 2018 [48] | PD vs. CD ad libitum RCT, parallel | [58] postmenopausal women with over-weight/healthy PD: 60 ± 5.5 CD: 62 ± 5.7 short-term (6 months) | Positive: significantly larger reductions in body weight and SAG in PD compared to the CD group. | Partially positive: TG decreased significantly more in the PD group compared to the CD group. TC levels and LDL decreased in both groups, without differences between groups. The levels of HDL and FFA remained stable in both groups. | Not analyzed | Positive: The PD led to improved insulin sensitivity. The HOMA-IR index decreased significantly in the PD group, without significant difference between diet groups. | Not analyzed |
| Boraxbekk et al. 2015 [40] | PD vs. CD (NNR) ad libitum RCT, parallel | [20] overweight or obese postmenopausal women PD = 61 ± 1.6 CD = 61.6 ± 1.7 short-term (6 months) | Partially positive: PD and NNR improved anthropometric measurements without significant differences between groups. | Partially positive: Levels of FFA in plasma decreased in both groups. | Not analyzed | No effects: no changes in plasma glucose, insulin, or HOMA-IR. | Not analyzed |
| Fontes- Villalba et al. 2016 [51] | PD vs. CD (diabetes diet) ad libitum RCT, crossover | [13] patients with type 2 diabetes/ PD: 66 ± 6 CD: 63 ± 6 short-term (12 weeks) | Positive: weight loss was significantly greater after the PD than the diabetes diet | Not analyzed | Not analyzed | No effects: did not change fasting levels of insulin. | Not analyzed |
| Frassetto et al. 2009 [18] | 3 days, three ramp-up diets of increasing potassium and fiber for 7 days, then a PD for 10 days; isocaloric RCT | [9] nonobese sedentary healthy volunteers/healthy population in physical activity 38 ± 12 short-term (10 day) | No effects: Not changed | Positive: large significant reductions in TC, LDL, and TG. No significant change occurred in HDL. | Positive: significant reductions in BP associated with improved arterial distensibility. | Positive: significant reduction in plasma insulin vs. time AUC during the OGTT. | Not analyzed |
| Genoni et al. 2016 [54] | PD vs. CD (AGHE) ad libitum RCT, parallel | [39] healthy women with BMI 27 ± 4 kg/m2 47 ± 13 short-term (4 weeks) | Positive: Significantly greater BM loss and WC occurred in the PD. | Partially positive: In both dietary groups experienced within group reductions to TC and LDL. No significant differences in these changes between the dietary groups. | No effect: No significant differences. | No effect: No significant differences between dietary groups in biomarkers of metabolism (fasting glucose and insulin). | Not analyzed |
| Lindeberg et al. 2007 [28] | PD vs. CD (Mediterranean) ad libitum RCT, parallel | [29] ischaemic heart disease plus either glucose intolerance or type 2 diabetes PD: 65 ± 10 CD: 57 ± 7 short-term (12 weeks) | Positive: weight loss and a decrease in WC. | Not analyzed | Not analyzed | Positive: improving glucose tolerance. | Not analyzed |
| Masharani et al. 2015 [52] | PD vs. CD (ADA) isocaloric RCT, parallel | [24] type 2 diabetes patients PD: 58 ± 8 CD: 56 ± 13 short-term (2 weeks) | Partially positive: The average BM changes were similar in both groups without caloric restriction. | Positive: The PD group had statistically significant declines in TC, HDL, and LDL. | No effects: The mean BP did not significantly change in any of the two groups. | Positive: PD group had greater benefits on glucose control, with significant improvement in insulin sensitivity. | Not analyzed |
| Mellberg et al. 2014 [49] | PD vs. CD (NNR) ad libitum RCT, parallel | [69], (after 6 m. 61, after 2 y: 49) overweight postmenopausal women PD: 59.9 ± 5.5 CD: 60.3 ± 5.9 short-term (6 months); long-term (24 months) | Partially positive: Both groups significantly decreased FM at 6 months and 24 months, with a more pronounced loss in the PD at 6 months but not at 24 months. WC and SAG also decreased in both the groups, with a more pronounced decrease in the PD at 6 months. | Partially positive: TG levels decreased significantly more at 6 and 24 months in the PD than in the NNR. LDL and TC decreased at both 6 and 24 months, HDL increased in 24 months. | Positive: decreased in both 6 and 24 mons: DBP, SBP, HR. | No effect: No differences were measured over time or between groups with regard to fasting glucose and fasting insulin concentrations and tissue plasminogen activator activity. | Not analyzed |
| Otten et. al. 2016 [41] | PD vs. CD (LFD) ad libitum RCT | [41] healthy, overweight/obese postmenopausal women PD: 61 ± 6 CD: 66 ± 2 short-term (6 months); long-term (24 months) | Partially positive: Both diet groups decreased their BM, BMI, WC, and FM. At 6 months, the PD group showed a greater effect than the LFD group. The LFD lost less LBM compared with the PD. At 24 months, there were no significant differences in body composition between diet groups except for the better preservation of LBM in the LFD group. | Partially positive: TGs, TC, and LDL improved significantly more in the PD group during the first 6 months of the study. At 24 months, both study groups showed an improvement of HDL. | Partially positive: SBP improved in both study groups at 6 months. DBP improved only in the PD group. | Partially positive: HOMA-IR improved significantly after 6 months. Between 6 and 24 months, hepatic insulin sensitivity deteriorated significantly in the PD group with a similar trend in the LFD group. | Not analyzed |
| Österdahl et al. 2008 [56] | PD vs. CD (normal diet) ad libitum Short communication | [14] healthy volunteers 30 ± 10 short-term (3 weeks) | Positive: Mean BM and WC decreased. | No effects: Not changed | Partially positive: Decreased SBP | No effects: Not changed | Not analyzed |
| Pastore et. al. 2015 [39] | PD vs. CD (AHA) ad libitum 4 months CD, followed by 4 months PD; RCT | [20] hypercholesterolemic patients 53 ± 7 short-term (4 months) | Positive: PD induced a significant BM loss, compared with AHA. | Positive: PD significantly lowered mean TC, LDL, and TG and increased HDL. | Not analyzed | Not analyzed | Not analyzed |
| Stomby et al. 2015 [46] | PD vs. CD (NNR) ad libitum RCT, parallel | [49] overweight and obese postmenopausal women short-term (6 months) long-term (24 months) | Partially positive: At 6 months, the PD group had a greater reduction of BM, BMI, and FM. At 24 months, there were no significant differences in anthropometric measurements between the groups. | Partially positive: TC decreased after 6 months but was unaltered at 24 months compared with baseline. TGs and LDL decreased throughout the intervention, whereas HDL increased after 24 months. There were no group differences in blood lipids. | Partially positive: SBP and DBP decreased at 6 months but had increased to baseline levels after 24 months. There were no group differences in blood pressure. | Partially positive: Fasting serum insulin and HOMA-IR decreased at 6 months but was unaltered after 24 months. There were no group differences, fasting serum insulin, and HOMA-IR. | Not analyzed |
| Otten et al. 2017 [42] | PD vs. PD-EX (PD-EX: a combination of aerobic exercise and resistance training in 1 h sessions three times weekly) ad libitum | [29] individuals with type 2 diabetes with BMI 25–40 kg/m2 short-term (12 weeks) | Positive: Both groups showed decreases in BM, FM and WC, without differences between intervention groups. Male participants decreased their WC more in the PD group compared to the PD-EX. Males in the PD-EX group retained more LBM than males in the PD group. | Partially positive: TG decreased in both study groups, while the HDL and LDL levels remained unchanged throughout the intervention. | Positive: Blood pressure decreased during the study in both intervention groups without any group difference. | Positive: Insulin sensitivity and glycemic control improved in both groups, without a difference between groups. The HOMA-IR and revised QUICKI improved in both intervention groups, and the HbA1c decreased during the study in both the PD group and the PD-EX group. | Partially positive The VO2max and the ergometer cycling workload increased during the study in the PD-EX group, but not in the PD group. Resting HR decreased more in the PD-EX group than the PD group. |
| Otten et al. 2018 [43] | PD vs. PD-EX (PD-EX: aerobic exercise/resistance training in 1 h sessions 3 times weekly) ad libitum RCT | [32] individuals with type 2 diabetes with BMI 25–40 kg/m2 short-term (12 weeks) | Positive: Both study groups showed a BM, BMI, FM loss. | Positive: TG decreased in both gropus | Not analyzed | Positive: Both groups improved their peripheral and adipose tissue insulin sensitivity, but not their hepatic insulin sensitivity. | Partially positive: The VO2max increased in the PD-EX group only. |
| Otten et al. 2019 [44] | PD vs. PD-EX (exercise training 3 h per week) ad libitum | [22] overweight and obese subjects with type 2 diabetes mellitus PD: 59 PD-EX: 61 short-term (12 weeks) | Positive: significant decreases in both groups in terms of BM, BMI, WC | Partially positive: The PD-EX group showed significant decreases in myocardial TG levels. These variables were unchanged in the PD group. There were significant decreases in both groups in terms of fasting triglycerides. | Positive: significant decreases in both groups in terms of SBP and DBP. | Positive: significant decreases in both groups in terms of fasting glucose, HbA1c, fasting insulin, and HOMA-IR. | Partially positive The VO2max increased significantly in both groups, although the increase in the PD-EX group was more pronounced. Mean resting HR decreased and the W max. increased significantly in the PD-EX group, while no changes were seen for these measures in the PD group. |
| Markofski et al. 2019 [71] | PD pre vs. post ad libitum | [7] overweight, physically inactive but otherwise healthy adults 32.7 ± 4.9 short-term (8 weeks) | Positive: time effect pre- to post-intervention for BM and BMI. | Not analyzed | No effect: SBP and DPB were unchanged following the PD intervention. | Not described | Not analyzed |
| Ryberg et al. 2013 [37] | PD pre vs. post ad libitum higher energy intake before intervention | [10] healthy, nonsmoking postmenopausal women with BMI > 27 kg/m2 short-term (5 weeks) | Positive: BMI, waist and hip circumference, waist/hip ratio, and SAG also decreased significantly. | Positive: TC, TG, HDL, LDL, and LDL/HDL decreased significantly. | Positive: DBP and resting HR decreased significantly. | Partially positive: Fasting serum glucose and HOMA indices decreased significantly. Insulin sensitivity did not change. | Not analyzed |
| Smith et al. 2014 [55] | PD pre vs. post subjects completed a CrossFit-based exercise program while adhering to the Paleo diet. ad libitum | [44] healthy population F: 31.2 M: 33.5 short-term (10 weeks) | Positive: FM percentage decreased significantly, as did BM. | Partially positive: A significant increase in non-HDL, LDL, TC/HDL, and TC in healthy subjects following a PD. Deleterious changes were found in those with optimal HDL, non-HDL, TC/HDL, and LDL whereas those within sub-optimal stratifications showed no significant change. | Not analyzed | Not analyzed | Positive: significantly increasing the VO2max, a common measure of cardiorespiratory fitness |
| BM (kg) | BMI (kg/m2) | WC (cm) | FM (kg) | FM (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| beta | −5.798 | −5.321 | −3.916 | −2.084 | −3.076 | −1.697 | −5.007 | −4.271 | −3.114 | −4.510 | −4.125 | −2.136 | −2.391 | −2.557 | −0.281 |
| 95%HCI | −4.269 | −3.163 | −2.579 | −1.361 | −2.560 | −0.989 | −3.132 | −2.977 | −2.429 | −1.572 | 0.391 | 1.234 | −1.158 | 0.128 | 1.292 |
| 95%LCI | −7.328 | −7.478 | −5.254 | −2.807 | −3.592 | −2.406 | −6.882 | −5.564 | −3.798 | −7.448 | −8.642 | −5.506 | −3.625 | −5.242 | −1.866 |
| p value | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.003 | 0.073 | 0.214 | 0.000 | 0.062 | 0.722 |
| K | 16 | 8 | 8 | 8 | 3 | 3 | 9 | 5 | 5 | 3 | 2 | 2 | 4 | 2 | 2 |
| BM (kg) | BMI (kg/m2) | WC (cm) | FM (kg) | FM (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| Beta | −8.690 | −8.690 | −5.760 | −2.765 | −2.765 | −1.815 | −12.097 | −12.097 | −10.874 | −5.464 | −5.464 | −4.451 | −2.722 | −2.722 | −2.607 |
| 95%HCI | −6.070 | −6.070 | −4.283 | −1.937 | −1.937 | −1.435 | −7.624 | −7.624 | −7.640 | −3.441 | −3.441 | −2.889 | −1.323 | −1.323 | −0.762 |
| 95%LCI | −11.310 | −11.310 | −7.236 | −3.594 | −3.594 | −2.196 | −16.570 | −16.570 | −14.108 | −7.488 | −7.488 | −6.013 | −4.120 | −4.120 | −4.452 |
| p value | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.006 |
| k | 3 | 3 | 3 | 3 | 3 | 3 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| TC (mg/dL) | TG (mg/dL) | HDL-C (mg/dL) | LDL-C (mg/dL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PDs | CD |
| Beta | −0.576 | −0.657 | −0.336 | −0.341 | −0.302 | −0.089 | −0.002 | −0.078 | −0.072 | −0.373 | −0.408 | −0.227 |
| 95%HCI | −0.387 | −0.544 | −0.246 | −0.222 | −0.156 | −0.044 | 0.104 | −0.017 | −0.003 | −0.189 | −0.318 | −0.147 |
| 95%LCI | −0.766 | −0.770 | −0.427 | −0.460 | −0.448 | −0.134 | −0.108 | −0.139 | −0.142 | −0.557 | −0.498 | −0.308 |
| p value | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.971 | 0.012 | 0.042 | 0.000 | 0.000 | 0.000 |
| k | 14 | 8 | 8 | 14 | 8 | 8 | 13 | 7 | 7 | 13 | 7 | 7 |
| TC (mg/dL) | TG (mg/dL) | HDL-C (mg/dL) | LDL-C (mg/dL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| Beta | −0.228 | −0.228 | 0.010 | −0.225 | −0.225 | −0.077 | 0.181 | 0.181 | 0.205 | −0.307 | −0.307 | −0.085 |
| 95%HCI | −0.076 | −0.076 | 0.051 | −0.148 | −0.148 | −0.004 | 0.235 | 0.235 | 0.263 | −0.203 | −0.203 | 0.037 |
| 95%LCI | −0.381 | −0.381 | −0.031 | −0.302 | −0.302 | −0.151 | 0.128 | 0.128 | 0.147 | −0.412 | −0.412 | −0.208 |
| p value | 0.003 | 0.003 | 0.639 | 0.000 | 0.000 | 0.040 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.172 |
| k | 4 | 4 | 4 | 4 | 4 | 4 | 3 | 3 | 3 | 3 | 3 | 3 |
| SBP (mmHg) | DBP (mmHg) | HR (bpm) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| Beta | −6.926 | −8.493 | −5.579 | −4.922 | −5.345 | −0.848 | −3.021 | −2.200 | −3.200 |
| 95%HCI | −4.484 | −5.467 | −2.654 | −3.247 | −3.047 | 2.513 | 0.906 | −1.729 | −2.785 |
| 95%LCI | −9.367 | −11.519 | −8.504 | −6.597 | −7.643 | −4.210 | −6.948 | −2.671 | −3.615 |
| p value | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.621 | 0.132 | 0.000 | 0.000 |
| k | 12 | 6 | 6 | 12 | 6 | 6 | 4 | 1 | 1 |
| SBP (mmHg) | DBP (mmHg) | |||||
|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD |
| Beta | −1.021 | −1.021 | 0.828 | −4.613 | −4.613 | 0.040 |
| 95%HCI | 1.785 | 1.785 | 2.479 | −3.837 | −3.837 | 4.315 |
| 95%LCI | −3.827 | −3.827 | −0.822 | −5.389 | −5.389 | −4.234 |
| p value | 0.476 | 0.476 | 0.325 | 0.000 | 0.000 | 0.985 |
| k | 3 | 3 | 3 | 3 | 3 | 3 |
| fP Glucose (mmol/L) | fP Insulin (mmol/L) | HOMA-IR | HbA1c (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| beta | −0.511 | −0.450 | −0.167 | −1.922 | −1.600 | −0.230 | −0.442 | −0.408 | −0.074 | −0.404 | −0.178 | −0.122 |
| 95%HCI | −0.176 | −0.025 | 0.068 | −0.916 | −0.323 | 0.224 | −0.196 | −0.185 | 0.037 | −0.019 | −0.028 | 0.021 |
| 95%LCI | −0.847 | −0.876 | −0.40 | −2.928 | −2.878 | −0.684 | −0.687 | −0.631 | −0.185 | −0.831 | −0.328 | −0.265 |
| p value | 0.003 | 0.050 | 0.165 | 0.000 | 0.011 | 0.267 | 0.000 | 0.000 | 0.157 | 0.063 | 0.020 | 0.095 |
| k | 12 | 8 | 7 | 11 | 5 | 5 | 9 | 5 | 5 | 3 | 2 | 2 |
| fP Glucose (mmol/L) | fP Insulin (mmol/L) | HOMA-IR | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | CD | PDs | PD | CD | PDs | PD | CD |
| beta | −0.113 | −0.113 | 0.002 | −0.240 | −0.240 | 0.351 | −0.022 | −0.022 | 0.304 |
| 95%HCI | 0.169 | 0.169 | 0.115 | 0.304 | 0.304 | 1.186 | 0.109 | 0.109 | 0.523 |
| 95%LCI | −0.396 | −0.396 | −0.111 | −0.784 | −0.784 | −0.484 | −0.153 | −0.153 | 0.085 |
| p value | 0.431 | 0.431 | 0.973 | 0.387 | 0.387 | 0.410 | 0.741 | 0.741 | 0.007 |
| k | 2 | 2 | 2 | 2 | 2 | 2 | 3 | 3 | 3 |
| VO2max (mL/kg/min) | VO2max (L/min) | Maximum Workload (W max) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Indicator | PDs | PD | PD-EX | PDs | PD | PD-EX | PDs | PD | PD-EX |
| Beta | 2.068 | 1.921 | 3.484 | 0.127 | 0.000 | 0.400 | 0.000 | 0.000 | 30.000 |
| 95%HCI | 2.853 | 2.725 | 4.940 | 0.400 | 0.112 | 0.754 | 0.112 | 0.112 | 56.575 |
| 95%LCI | 1.282 | 1.116 | 2.028 | −0.146 | −0.112 | 0.046 | −0.112 | −0.112 | 3.425 |
| p value | 0.000 | 0.000 | 0.000 | 0.362 | 1.000 | 0.027 | 1.000 | 1.000 | 0.027 |
| k | 3 | 2 | 2 | 2 | 1 | 1 | 1 | 1 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frączek, B.; Pięta, A.; Burda, A.; Mazur-Kurach, P.; Tyrała, F. Paleolithic Diet—Effect on the Health Status and Performance of Athletes? Nutrients 2021, 13, 1019. https://doi.org/10.3390/nu13031019
Frączek B, Pięta A, Burda A, Mazur-Kurach P, Tyrała F. Paleolithic Diet—Effect on the Health Status and Performance of Athletes? Nutrients. 2021; 13(3):1019. https://doi.org/10.3390/nu13031019
Chicago/Turabian StyleFrączek, Barbara, Aleksandra Pięta, Adrian Burda, Paulina Mazur-Kurach, and Florentyna Tyrała. 2021. "Paleolithic Diet—Effect on the Health Status and Performance of Athletes?" Nutrients 13, no. 3: 1019. https://doi.org/10.3390/nu13031019
APA StyleFrączek, B., Pięta, A., Burda, A., Mazur-Kurach, P., & Tyrała, F. (2021). Paleolithic Diet—Effect on the Health Status and Performance of Athletes? Nutrients, 13(3), 1019. https://doi.org/10.3390/nu13031019






