A Three-Month Consumption of Eggs Enriched with ω-3, ω-5 and ω-7 Polyunsaturated Fatty Acids Significantly Decreases the Waist Circumference of Subjects at Risk of Developing Metabolic Syndrome: A Double-Blind Randomized Controlled Trial
Abstract
1. Introduction
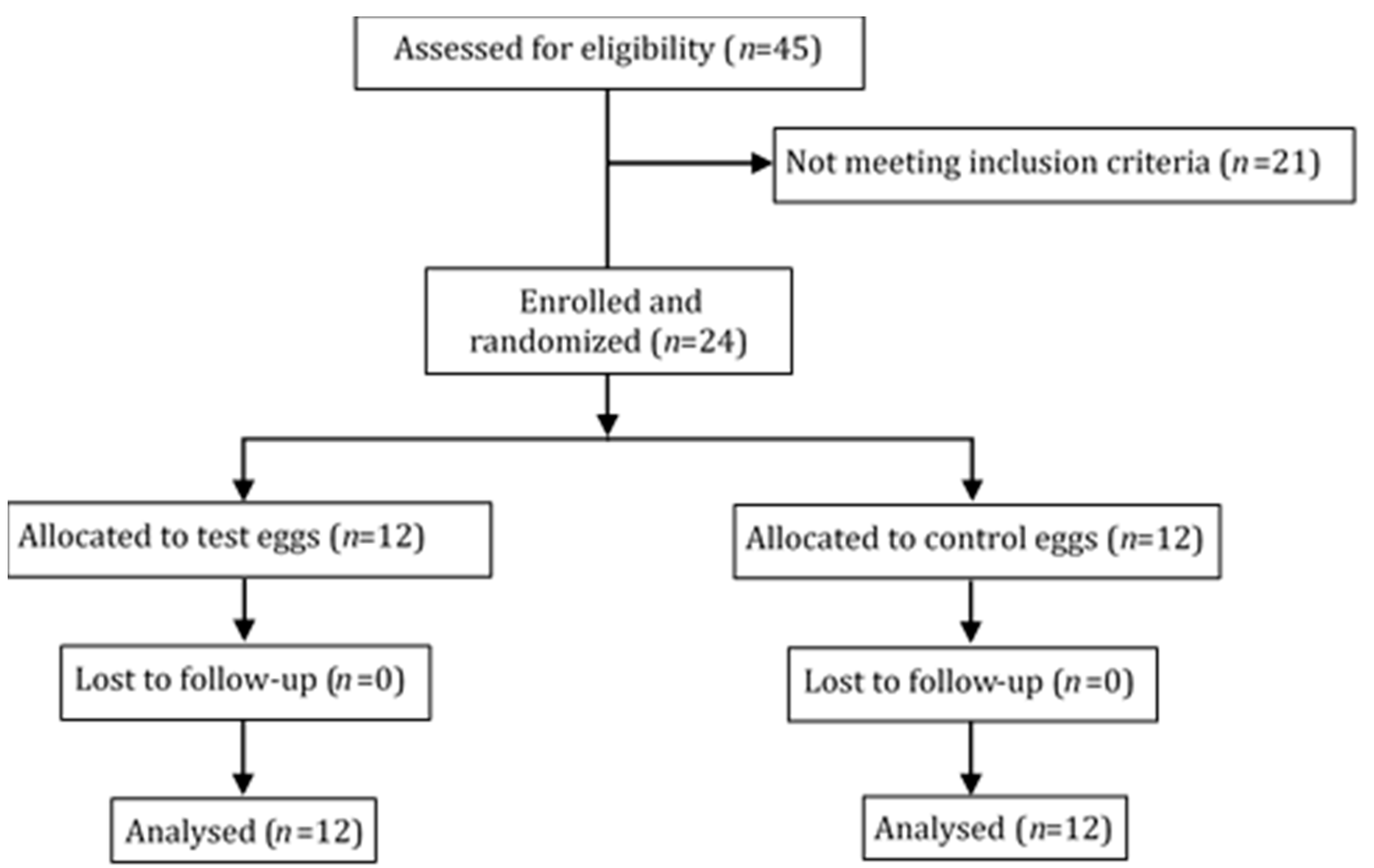
2. Materials and Methods
2.1. Study Population
2.2. Study Design
2.3. Dietary Assessment
2.4. Anthropometric Measurements
2.5. Clinical Investigations
2.6. Measurement of Nitrosylated Hemoglobin and Peripheral Artery Tonometry
2.7. Statistics and Data Analysis
3. Results
3.1. Dietary Analysis and Tolerance
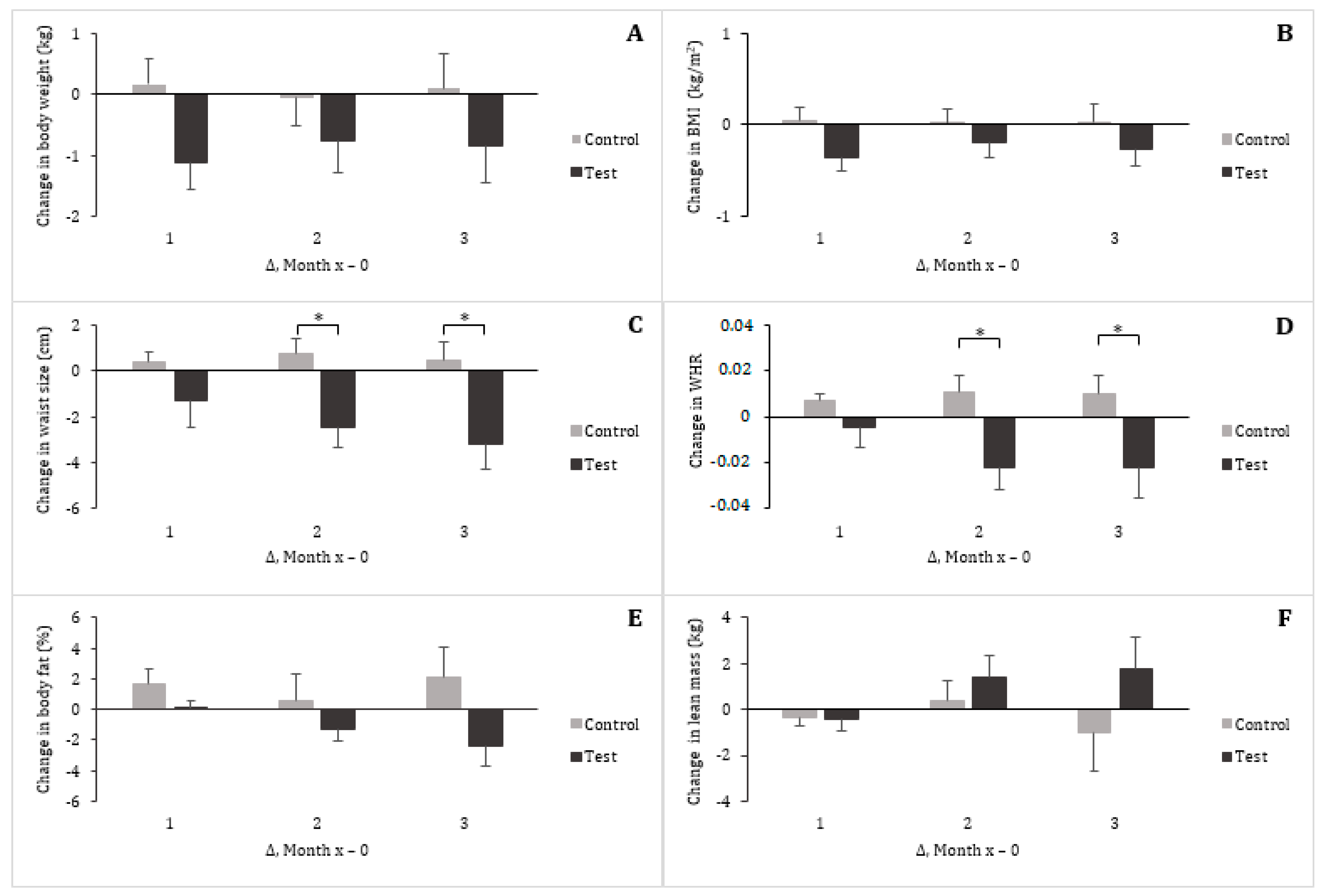
3.2. Vital Signs and Anthropometrics
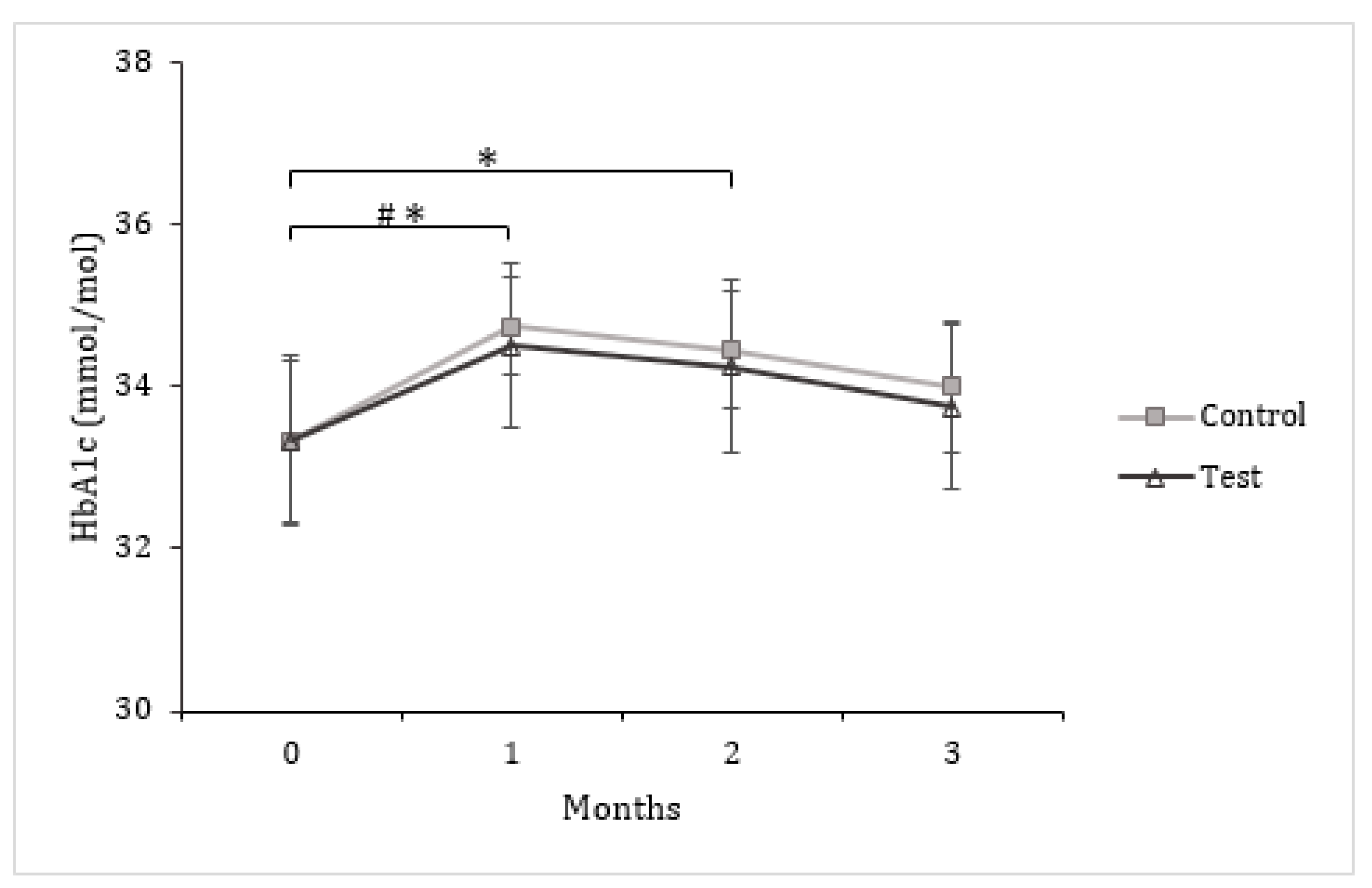
3.3. Insulin and Glucose Metabolism
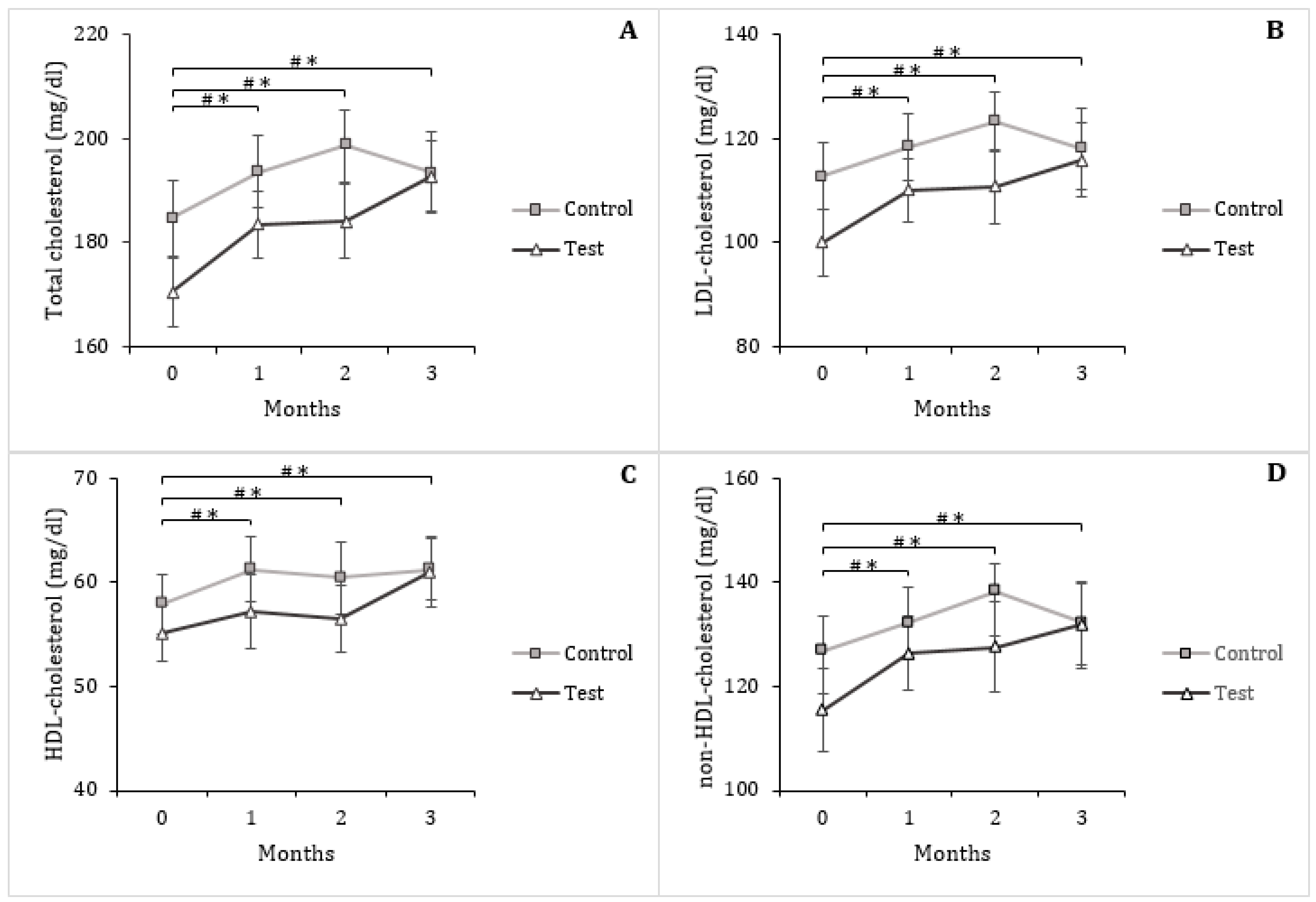
3.4. Serum Triglycerides and Lipoproteins
3.5. Inflammation and Oxidative Stress
3.6. Erythrocyte Level of Nitrosylated Hemoglobin and Vascular Endothelial Function
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Alberti, G.; Zimmet, P.; Shaw, J.; Grundy, S.M. The IDF consensus worldwide definition of the metabolic syndrome. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 1–8. [Google Scholar] [CrossRef]
- Nolan, P.B.; Carrick-Ranson, G.; Stinear, J.W.; Reading, S.A.; Dalleck, L.C. Prevalence of metabolic syndrome and metabolic syndrome components in young adults: A pooled analysis. Prev. Med. Rep. 2017, 7, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-S.; Xun, P.; Iribarren, C.; Van Horn, L.; Steffen, L.; Daviglus, M.L.; Siscovick, D.; Liu, K.; He, K. Intake of fish and long-chain omega-3 polyunsaturated fatty acids and incidence of metabolic syndrome among American young adults: A 25-year follow-up study. Eur. J. Nutr. 2016, 55, 1707–1716. [Google Scholar] [CrossRef]
- Baik, I.; Abbott, R.D.; Curb, J.D.; Shin, C. Intake of fish and n-3 fatty acids and future risk of metabolic syndrome. J. Am. Diet. Assoc. 2010, 110, 1018–1026. [Google Scholar] [CrossRef]
- Nigam, A.; Frasure-Smith, N.; Lespérance, F.; Julien, P. Relationship between n-3 and n-6 plasma fatty acid levels and insulin resistance in coronary patients with and without metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Thorseng, T.; Witte, D.R.; Vistisen, D.; Borch-Johnsen, K.; Bjerregaard, P.; Jørgensen, M.E. The association between n-3 fatty acids in erythrocyte membranes and insulin resistance: The Inuit Health in Transition Study. Int. J. Circumpolar Heal. 2009, 68, 327–336. [Google Scholar] [CrossRef]
- Risérus, U.; Vessby, B.; Ärnlöv, J.; Basu, S. Effects of cis-9,trans-11 conjugated linoleic acid supplementation on insulin sensitivity, lipid peroxidation, and proinflammatory markers in obese men. Am. J. Clin. Nutr. 2004, 80, 279–283. [Google Scholar] [CrossRef]
- Schmitt, B.; Ferrt, C.; Daniel, N.; Weill, P.; Kerhoas, N.; Legrand, P. Effet d’un régime riche en acides gras w3 et en CLA 9-cis, 11-trans sur l’insulinorésistance et les paramètres du diabète de type 2. OCL 2006, 13, 70–75. [Google Scholar]
- Vroegrijk, I.O.C.M.; Van Diepen, J.A.; Berg, S.V.D.; Westbroek, I.; Keizer, H.; Gambelli, L.; Hontecillas, R.; Bassaganya-Riera, J.; Zondag, G.C.M.; Romijn, J.A.; et al. Pomegranate seed oil, a rich source of punicic acid, prevents diet-induced obesity and insulin resistance in mice. Food Chem. Toxicol. 2011, 49, 1426–1430. [Google Scholar] [CrossRef]
- Ngo Njembe, M.T.; Dormal, E.; Gardin, C.; Mignolet, E.; Debier, C.; Larondelle, Y. Effect of the dietary combination of flaxseed and Ricinodendron heudelotii or Punica granatum seed oil on the fatty acid profile of eggs. Food Chem. 2021, 344, 128668. [Google Scholar] [CrossRef] [PubMed]
- Yasar, F.; Nicolas, C.; Dufourny, G. Outil Pour Estimer la Consommation Alimentaire: Photos de Quantification—Composition Nutritionnelle; CIRIHA: Bruxelles, Belgium, 2010. [Google Scholar]
- Lobysheva, I.I.; Van Eeckhoudt, S.; Zotti, F.D.; Rifahi, A.; Pothen, L.; Beauloye, C.; Balligand, J.-L. Heme-nitrosylated hemoglobin and oxidative stress in women consuming combined contraceptives. Clinical application of the EPR spectroscopy. Free. Radic. Biol. Med. 2017, 108, 524–532. [Google Scholar] [CrossRef]
- Axtell, A.L.; Gomari, F.A.; Cooke, J.P. Assessing Endothelial Vasodilator Function with the Endo-PAT 2000. J. Vis. Exp. 2010, 2167, e2167. [Google Scholar] [CrossRef] [PubMed]
- Aparicio-Soto, M.; Sánchez-Hidalgo, M.; Rosillo, M.Á.; Castejón, M.L.; Alarcón-De-La-Lastra, C. Extra virgin olive oil: A key functional food for prevention of immune-inflammatory diseases. Food Funct. 2016, 7, 4492–4505. [Google Scholar] [CrossRef] [PubMed]
- Zambon, A.; Sartore, G.; Passera, D.; Francini-Pesenti, F.; Bassi, A.; Basso, C.; Zambon, S.; Manzato, E.; Crepaldi, G. Effects of hypocaloric dietary treatment enriched in oleic acid on LDL and HDL subclass distribution in mildly obese women. J. Intern. Med. 1999, 246, 191–201. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Giron, M.; Sanchez, F.; Hortelano, P.; Periago, J.; Suarez, M. Effects of dietary fatty acids on lipid metabolism in streptozotocin-induced diabetic rats. Metab. 1999, 48, 455–460. [Google Scholar] [CrossRef]
- Defina, L.F.; Marcoux, L.G.; Devers, S.M.; Cleaver, J.P.; Willis, B.L. Effects of omega-3 supplementation in combination with diet and exercise on weight loss and body composition. Am. J. Clin. Nutr. 2010, 93, 455–462. [Google Scholar] [CrossRef]
- Salim, S.J.; Alizadeh, S.; Djalali, M.; Nematipour, E.; Javanbakht, M.H. Effect of Omega-3 Polyunsaturated Fatty Acids Supplementation on Body Composition and Circulating Levels of Follistatin-Like 1 in Males With Coronary Artery Disease: A Randomized Double-Blind Clinical Trial. Am. J. Men’s Heal. 2017, 11, 1758–1764. [Google Scholar] [CrossRef]
- Crochemore, I.C.; Souza, A.F.; De Souza, A.C.; Rosado, E.L. Omega-3 polyunsaturated fatty acid supplementation does not influence body composition, insulin resistance, and lipemia in women with type 2 diabetes and obesity. Nutr. Clin. Prac. 2012, 27, 553–560. [Google Scholar] [CrossRef]
- Gaullier, J.-M.; Halse, J.; Høivik, H.O.; Høye, K.; Syvertsen, C.; Nurminiemi, M.; Hassfeld, C.; Einerhand, A.; O’Shea, M.; Gudmundsen, O. Six months supplementation with conjugated linoleic acid induces regional-specific fat mass decreases in overweight and obese. Br. J. Nutr. 2007, 97, 550–560. [Google Scholar] [CrossRef]
- Smedman, A.; Vessby, B. Conjugated linoleic acid supplementation in humans—metabolic effects. Lipids 2001, 36, 773–781. [Google Scholar] [CrossRef]
- Lankson, H.; Stakkestad, J.A.; Fagertun, H.; Thom, E.; Wadstein, J.; Gudmundsen, O. Conjugated Linoleic Acid Reduces Body Fat Mass in Overweight and Obese Humans. J. Nutr. 2000, 130, 2943–2948. [Google Scholar] [CrossRef] [PubMed]
- Whigham, L.D.; Watras, A.C.; Schoeller, D.A. Efficacy of conjugated linoleic acid for reducing fat mass: A meta-analysis in humans. Am. J. Clin. Nutr. 2007, 85, 1203–1211. [Google Scholar] [CrossRef]
- Sluijs, I.; Plantinga, Y.; De Roos, B.; I Mennen, L.; Bots, M.L. Dietary supplementation with cis-9,trans-11 conjugated linoleic acid and aortic stiffness in overweight and obese adults. Am. J. Clin. Nutr. 2009, 91, 175–183. [Google Scholar] [CrossRef]
- Hargrave, K.M.; Li, C.; Meyer, B.J.; Kachman, S.D.; Hartzell, D.L.; Della-Fera, M.A.; Miner, J.L.; Baile, C.A. Adipose Depletion and Apoptosis Induced by Trans-10, Cis-12 Conjugated Linoleic Acid in Mice. Obes. Res. 2002, 10, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.; Martinez, K.; Schmidt, S.F.; Mandrup, S.; LaPoint, K.; McIntosh, M. Antiobesity mechanisms of action of conjugated linoleic acid. J. Nutr. Biochem. 2010, 21, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Storkson, J.M.; Albright, K.J.; Liu, W.; Pariza, M.W. Evidence that the trans-10,cis-12 isomer of conjugated linoleic acid induces body composition changes in mice. Lipids 1999, 34, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Peláez-Jaramillo, M.J.; Valencia-Enciso, N.; Cárdenas-Mojica, A.A.; Gaete, P.V.; Scher-Nemirovsky, E.A.; Gómez-Arango, L.F.; Colmenares-Araque, D.; Castro-López, C.A.; Betancourt-Villamizar, E.; Jaimes-Madrigal, J.; et al. Impact of a Formulation Containing Unusual Polyunsaturated Fatty Acids, Trace Elements, Polyphenols and Plant Sterols on Insulin Resistance and Associated Disturbances. Diabetes Ther. 2020, 11, 229–245. [Google Scholar] [CrossRef] [PubMed]
- Koba, K.; Imamura, J.; Akashoshi, A.; Kohno-Murase, J.; Nishizono, S.; Iwabuchi, M.; Tanaka, K.; Sugano, M. Genetically Modified Rapeseed Oil Containingcis-9,trans-11,cis-13-Octadecatrienoic Acid Affects Body Fat Mass and Lipid Metabolism in Mice. J. Agric. Food Chem. 2007, 55, 3741–3748. [Google Scholar] [CrossRef]
- Koba, K.; Belury, M.A.; Sugano, M. Potential health benefits of conjugated trienoic acids. Lipid Technol. 2007, 19, 200–203. [Google Scholar] [CrossRef]
- Lai, C.-S.; Tsai, M.-L.; Badmaev, V.; Jimenez, M.; Ho, C.-T.; Pan, M.-H. Xanthigen suppresses preadipocyte differentiation and adipogenesis through down-regulation of PPARγ and C/EBPs and modulation of SIRT-1, AMPK, and FoxO pathways. J. Agric. Food Chem. 2012, 60, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Fuller, N.R.; Caterson, I.D.; Sainsbury, A.; Denyer, G.; Fong, M.; Gerofi, J.; Baqleh, K.; Williams, K.H.; Lau, N.S.; Markovic, T.P. The effect of a high-egg diet on cardiovascular risk factors in people with type 2 diabetes: The Diabetes and Egg (DIABEGG) study—a 3-mo randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 705–713. [Google Scholar] [CrossRef]
- Fuller, N.R.; Sainsbury, A.; Caterson, I.D.; Denyer, G.; Fong, M.; Gerofi, J.; Leung, C.; Lau, N.S.; Williams, K.H.; Januszewski, A.S.; et al. Effect of a high-egg diet on cardiometabolic risk factors in people with type 2 diabetes: The Diabetes and Egg (DIABEGG) Study—randomized weight-loss and follow-up phase. Am. J. Clin. Nutr. 2018, 107, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Herron, K.L.; Vega-Lopez, S.; Conde, K.; Ramjiganesh, T.; Shachter, N.S.; Fernandez, M.L. Men Classified as Hypo- or Hyperresponders to Dietary Cholesterol Feeding Exhibit Differences in Lipoprotein Metabolism. J. Nutr. 2003, 133, 1036–1042. [Google Scholar] [CrossRef]
- Blesso, C.N.; Fernandez, M.L. Dietary Cholesterol, Serum Lipids, and Heart Disease: Are Eggs Working for or Against You? Nutrients 2018, 10, 426. [Google Scholar] [CrossRef]
- Berger, S.; Raman, G.; Vishwanathan, R.; Jacques, P.F.; Johnson, E.J. Dietary cholesterol and cardiovascular disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2015, 102, 276–294. [Google Scholar] [CrossRef]
- Moriyama, K.; Takahashi, E. Non-HDL Cholesterol is a More Superior Predictor of Small-Dense LDL Cholesterol than LDL Cholesterol in Japanese Subjects with TG Levels <400 mg/dL. J. Atheroscler. Thromb. 2016, 23, 1126–1137. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, B.J.; Rana, J.S.; Stroes, E.S.G.; Després, J.-P.; Shah, P.K.; Kastelein, J.J.P.; Wareham, N.J.; Boekholdt, S.M.; Khaw, K.-T. Beyond low-density lipoprotein cholesterol: Respective contributions of non–high-density lipoprotein cholesterol levels, triglycerides, and the total cholesterol/high-density lipoprotein cholesterol ratio to coronary heart disease risk in apparently healthy men and women. J. Am. Coll. Cardiol. 2009, 55, 35–41. [Google Scholar]
- Varbo, A.; Benn, M.; Tybjærg-Hansen, A.; Jørgensen, A.B.; Frikke-Schmidt, R.; Nordestgaard, B.G. Remnant Cholesterol as a Causal Risk Factor for Ischemic Heart Disease. J. Am. Coll. Cardiol. 2013, 61, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Brunner, F.J.; Waldeyer, C.; Ojeda, F.; Salomaa, V.; Kee, F.; Sans, S.; Thorand, B.; Giampaoli, S.; Brambilla, P.; Tunstall-Pedoe, H.; et al. Application of non-HDL cholesterol for population-based cardiovascular risk stratification: Results from the Multinational Cardiovascular Risk Consortium. Lancet 2019, 394, 2173–2183. [Google Scholar] [CrossRef]
- Caparevic, Z.; Kostic, N.; Ilic, S.; Stojanovic, D.; Ivanović, A.M. Oxidized LDL and C-reactive protein as markers for detection of accelerated atherosclerosis in type 2 diabetes. Med. Pregl. 2006, 59, 160–164. [Google Scholar] [CrossRef][Green Version]
- Hulthe, J.; Fagerberg, B. Circulating Oxidized LDL Is Associated With Subclinical Atherosclerosis Development and Inflammatory Cytokines (AIR Study). Arter. Thromb. Vasc. Biol. 2002, 22, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Holvoet, P.; Lee, D.-H.; Steffes, M.; Gross, M.; Jacobs, D.R. Association Between Circulating Oxidized Low-Density Lipoprotein and Incidence of the Metabolic Syndrome. Obstet. Gynecol. Surv. 2008, 63, 575–576. [Google Scholar] [CrossRef][Green Version]
- Di Nunzio, M.; Valli, V.; Bordoni, A. PUFA and oxidative stress. Differential modulation of the cell response by DHA. Int. J. Food Sci. Nutr. 2016, 67, 834–843. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, A.; Franklin, M.F.; Wahle, K.; Duthie, G.G. Dietary intakes of polyunsaturated fatty acids and indices of oxidative stress in human volunteers. Eur. J. Clin. Nutr. 1999, 53, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Reaven, P.D.; Witztum, J.L. Oxidized low density lipoproteins in atherogenesis: Role of dietary modification. Annu. Rev.Nutr. 1996, 16, 51–71. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Philis-Tsimikas, A.; Alexopoulos, S.; Sigari, F.; Lee, C.; Reaven, P.D. LDL isolated from Greek subjects on a typical diet or from American subjects on an oleate-supplemented diet induces less monocyte chemotaxis and adhesion when exposed to oxidative stress. Arter. Thromb. Vasc. Biol. 1999, 19, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Cao, Y.; Chen, J.-N.; Chen, Z.-Y. Oxidative Stability of Conjugated Linolenic Acids. J. Agric. Food Chem. 2009, 57, 4212–4217. [Google Scholar] [CrossRef]
- Zotti, F.D.; Verdoy, R.; Brusa, D.; Lobysheva, I.I.; Balligand, J.-L. Redox regulation of nitrosyl-hemoglobin in human erythrocytes. Redox Biol. 2020, 34, 101399. [Google Scholar] [CrossRef]
- Feron, O.; Dessy, C.; Moniotte, S.; Desager, J.-P.; Balligand, J.-L. Hypercholesterolemia decreases nitric oxide production by promoting the interaction of caveolin and endothelial nitric oxide synthase. J. Clin. Investig. 1999, 103, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Zotti, F.D.; Lobysheva, I.I.; Balligand, J.-L. Nitrosyl-hemoglobin formation in rodent and human venous erythrocytes reflects NO formation from the vasculature in vivo. PLoS ONE 2018, 13, e0200352. [Google Scholar] [CrossRef] [PubMed]
| Control | Test | |
|---|---|---|
| Fatty acids (mg/egg) | ||
| Lauric acid (C12:0) | 3.93 ± 0.08 | 3.95 ± 0.08 |
| Myristic acid (C14:0) | 9.47 ± 0.21 | 12.04 ± 0.28 |
| Palmitic acid (C16:0) | 993.91 ± 17.29 | 1008.68 ± 16.67 |
| Palmitoleic acid (C16:1c9) | 56.24 ± 2.00 | 51.03 ± 1.81 |
| Stearic acid (C18:0) | 344.47 ± 5.90 | 386.41 ± 5.52 |
| Oleic acid (C18:1c9) | 2689.32 ± 33.29 | 1115.25 ± 25.97 |
| Cis-vaccenic acid (C18:1c11) | 70.96 ± 1.25 | 35.58 ± 1.04 |
| Linoleic acid (C18:2c9,c12) | 543.01 ± 10.58 | 622.70 ± 13.30 |
| Gamma-linolenic acid (C18:3c6,c9,c12) | 3.52 ± 0.13 | 4.20 ± 0.13 |
| Alpha-linolenic acid (C18:3c9,c12,c15) | 14.31 ± 0.34 | 105.19 ± 4.04 |
| Rumenic acid (C18:2c9,t11) | 2.06 ± 0.21 | 595.16 ± 10.64 |
| Punicic acid (C18:3c9,t11,c13) | ND | 321.59 ± 9.25 |
| Dihomo-gamma-linolenic acid (C20:3c8,c11,c14) | 6.50 ± 0.13 | 7.55 ± 0.17 |
| Arachidonic acid (C20:4c5,c8,c11,c14) | 97.89 ± 1.46 | 62.40 ± 1.45 |
| Eicosapentaenoic acid (C20:5c5,c8,c11,c14,c17) | 0.05 ± 0.02 | 2.58 ± 0.11 |
| n-6 Docosapentaenoic acid (C22:5c4,c7,c10,c13,c16) | 23.35 ± 0.70 | 4.47 ± 0.24 |
| Docosahexaenoic acid (C22:6c4,c7,c10,c13,c16,c19) | 37.72 ± 0.69 | 82.81 ± 1.89 |
| Σ SFA | 1378.63 ± 22.40 | 1445.70 ± 22.31 |
| Σ MUFA | 2833.56 ± 35.64 | 1221.60 ± 28.01 |
| Σ n-6 PUFA | 691.28 ± 119.13 | 716.03 ± 138.72 |
| Σ n-3 PUFA | 61.55 ± 1.02 | 230.16 ± 6.15 |
| Total lipids (% by weight of fresh yolk) | 31.08 ± 0.59 | 32.45 ± 0.44 |
| Total cholesterol (mg/egg) | 177.19 ± 5.40 | 182.26 ± 10.13 |
| Control (n = 12) | Test (n = 12) | p-Value a | |||
|---|---|---|---|---|---|
| Mean ± SEM | Range | Mean ± SEM | Range | ||
| Sex (women/men) | 9/3 | - | 8/4 | - | - |
| Age (years) | 51.67 ± 2.39 | 42–71 | 45.58 ± 2.43 | 35–59 | 0.105 |
| Body weight (kg) | 76.23 ± 2.64 | 66.4–93.8 | 74.34 ± 4.18 | 59.1–98.1 | 0.402 |
| BMI (kg/m2) | 25.98 ± 0.75 | 21.9–30.3 | 26.04 ± 1.20 | 20.3–35.6 | 0.402 |
| Waist circumference (cm) | 93.38 ± 2.69 | 82–111 | 92.21 ± 2.79 | 83–111 | 0.840 |
| Hip circumference (cm) | 106.38 ± 1.49 | 99–115 | 102.79 ± 2.91 | 92–132 | 0.296 |
| WHR | 0.88 ± 0.03 | 0.8–1.0 | 0.89 ± 0.02 | 0.8–1.1 | 0.564 |
| Body fat (%) | 33.48 ± 1.71 | 22.5–42.3 | 31.83 ± 2.24 | 18.1–48 | 0.624 |
| Lean body mass (kg) | 50.13 ± 2.56 | 42.7–66.6 | 49.81 ± 2.65 | 37.4–64.9 | 1.000 |
| Hemoglobin (g/dL) | 13.89 ± 0.34 | 12.2–15.5 | 14.18 ± 0.49 | 12–16.71 | 0.751 |
| Hematocrit (%) | 41.6 ± 0.89 | 36.1–46.4 | 42.20 ± 1.24 | 36.2–48.2 | 0.773 |
| Red blood cells (106/mm3) | 4.66 ± 0.10 | 4.08–5.22 | 4.75 ± 0.16 | 3.93–5.66 | 0.977 |
| White blood cells (103/mm3) | 5.28 ± 0.27 | 3.95–7.13 | 5.52 ± 0.26 | 3.93–7.27 | 0.507 |
| Platelets (103/mm3) | 226 ± 14.09 | 144–301 | 222.17 ± 15.07 | 144–301 | 0.665 |
| AST (UI/L) | 18.83 ± 1.07 | 14–27 | 21.64 ± 2.13 | 14–35 | 0.477 |
| ALT (UI/L) | 17.25 ± 2.07 | 10–34 | 20.67 ± 2.69 | 10–44 | 0.247 |
| GGT (UI/L) | 15.50 ± 0.97 | 10–20 | 18.17 ± 2.52 | 9–36 | 0.664 |
| Urea (mg/dL) | 30.75 ± 1.50 | 20–38 | 33 ± 2.17 | 23–50 | 0.401 |
| Creatininemia (mg/dL) | 0.79 ± 0.02 | 0.67–0.93 | 0.86 ± 0.04 | 0.67–1.09 | 0.247 |
| eGFR (mL/min) | 92.42 ± 2.28 | 82–104 | 90.75 ± 3.35 | 73–112 | 0.623 |
| Albumin (g/dL) | 4.30 ± 0.09 | 3.90–4.90 | 4.31 ± 0.06 | 3.90–4.60 | 0.640 |
| Total protein (g/dL) | 6.94 ± 0.14 | 6–7.50 | 6.78 ± 0.12 | 6.20–7.30 | 0.271 |
| Months | ||||
|---|---|---|---|---|
| 0 | 1 | 2 | 3 | |
| Energy (kcal/day) | ||||
| Control | 1865.2 ± 159.06 a | 1811.92 ± 146.90 a | 1805.75 ± 145.23 a | 1818.33 ± 161.42 a |
| Test | 1782.45 ± 100.32 a | 1531.36 ± 81.53 a | 1637.36 ± 105.59 a | 1538.27 ± 137.10 a |
| Protein (% of daily energy) | ||||
| Control | 16.00 ± 0.93 a | 17.67 ± 1.24 a | 17.92 ± 1.12 a | 17.50 ± 0.72 a |
| Test | 15.18 ± 1.23 a | 17.27 ± 1.15 a | 15.91 ± 0.94 a | 18.55 ± 1.67 a |
| Carbohydrates (% of daily energy) | ||||
| Control | 43.50 ± 1.92 a | 38.67 ± 1.31 a | 40.75 ± 1.72 a | 40.33 ± 1.68 a |
| Test | 38.27 ± 2.29 a | 39.45 ± 2.18 a | 36.64 ± 2.13 a | 36.09 ± 2.02 a |
| Fiber (g/day) | ||||
| Control | 21.4 ± 1.54 a# | 20.00 ± 16.77 a# | 19.17 ± 15.67 a# | 20.58 ± 1.56 a# |
| Test | 16.45 ± 1.77 a# | 14.18 ± 1.32 a# | 14.09 ± 1.47 a# | 14.09 ± 135 a# |
| Lipids (% of daily energy) | ||||
| Control | 35.62 ± 2.49 a | 36.79 ± 1.74 a | 36.19 ± 1.71 a | 36.06 ± 1.73 a |
| Test | 38.08 ± 2.19 a | 36.58 ± 2.02 a | 40.90 ± 2.17 a | 40.09 ± 1.99 a |
| SFA (% of daily energy) | ||||
| Control | 16.44 ± 1.25 a | 14.80 ± 1.05 a | 14.63 ± 1.22 a | 14.25 ± 0.82 a |
| Test | 15.22 ± 0.88 a | 15.82 ± 1.42 a | 17.59 ± 1.15 a | 16.81 ± 0.83 a |
| MUFA (% of daily energy) | ||||
| Control | 10.29 ± 0.95 a | 12.70 ± 0.80 b | 12.27 ± 0.80 ab | 12.88 ± 0.74 b |
| Test | 13.43 ± 1.51 a | 11.40 ± 0.85 a | 11.86 ± 1.43 a | 11.09 ± 1.03 a |
| PUFA (% of daily energy) | ||||
| Control | 3.40 ± 0.36 a | 4.80 ± 0.45 b | 4.39 ± 0.31 ab | 4.58 ± 0.34 b |
| Test | 4.27 ± 0.62 a | 5.02 ± 0.31 a | 5.00 ± 0.44 a | 5.56 ± 0.54 a |
| Alpha-linolenic acid (mg/day) | ||||
| Control | 593.9 ± 132.63 a | 930.58 ± 136.68 a | 728.33 ± 103.98 a | 679.17 ± 129.75 a |
| Test | 697.81 ± 119.61 a | 675 ± 87.35 a | 629.73 ± 100.52 a | 737.82 ± 168.48 a |
| Rumenic acid (mg/day) | ||||
| Control | 125.9 ± 19.23 a | 116.08 ± 28.92 a## | 108.75 ± 19.82 a## | 117.58 ± 17.81 a## |
| Test | 92.64 ± 19.93 a | 1167.27 ± 116.94 b## | 1106.55 ± 131.88 b## | 1187.73 ± 112.41 b## |
| Punicic acid (mg/day) | ||||
| Control | 0.00 ± 0.00 a | 0.00 ± 0.00 a### | 0.00 ± 0.00 a### | 0.00 ± 0.00 a### |
| Test | 0.00 ± 0.00 a | 643.18 ± 18.51 b### | 643.18 ± 18.51 b### | 643.18 ± 18.51 b### |
| Docosahexaenoic acid (mg/day) | ||||
| Control | 148.20 ± 63.85 a | 216.17 ± 51.55 a | 230.42 ± 59.12 a | 265.50 ± 126.67 a |
| Test | 101.55 ± 37.62 a | 189.64 ± 14.02 ab | 278.27 ± 72.18 b | 232.73 ± 34.41 b |
| Cholesterol (mg/day) | ||||
| Control | 284.30 ± 38.36 a | 621.50 ± 34.93 b | 581.08 ± 19.91 b | 593.5 ± 17.38 b |
| Test | 262.45 ± 38.25 a | 571 ± 21.43 b | 589.09 ± 54.26 b | 573.45 ± 23.66 b |
| Control (n = 12) | Test (n = 12) | |||||
|---|---|---|---|---|---|---|
| Month 0 | Month 3 | ∆, Month 3-0 | Month 0 | Month 3 | ∆, Month 3-0 | |
| Resting heart rate (beats/min) | 68.33 ± 2.23 | 66.50 ± 2.22 | −1.83 ± 2.22 | 66.25 ± 2.51 | 69.50 ± 2.24 | 3.25 ± 2.45 |
| Systolic blood pressure (mmHg) | 128.33 ± 4.80 | 122.33 ± 3.17 | −6.00 ± 3.01 | 122.33 ± 4.76 | 118.17 ± 5.59 | −4.17 ± 4.39 |
| Diastolic blood pressure (mmHg) | 78.50 ± 3.27 | 76.25 ± 1.85 | −2.25 ± 2.58 | 77.08 ± 3.44 | 73.33 ± 3.57 | −3.75 ± 2.45 |
| Weight (kg) | 76.23 ± 2.64 | 76.32 ± 2.76 | 0.09 ± 0.58 | 74.34 ± 4.18 | 73.48 ± 4.01 | −0.86 ± 0.58 |
| BMI (kg/m2) | 25.98 ± 0.75 | 26.01 ± 0.79 | 0.03 ± 0.19 | 26.04 ± 1.20 | 25.78 ± 1.16 | −0.26 ± 0.19 |
| Waist circumference (cm) a | 93.38 ± 2.69 | 93.88 ± 2.69 | 0.50 ± 0.79 | 92.21 ± 2.79 | 89.04 ± 2.60 | −3.17 ± 1.12 ** |
| Hip circumference (cm) | 106.38 ± 1.49 | 105.63 ± 1.44 | −0.75 ± 0.53 | 102.79 ± 2.91 | 101.83 ± 2.57 | −0.96 ± 1.11 |
| WHR | 0.88 ± 0.03 | 0.89 ± 0.02 | 0.01 ± 0.01 | 0.90 ± 0.02 | 0.88 ± 0.03 | −0.02 ± 0.01 |
| Body fat (%) | 33.48 ± 1.71 | 35.62 ± 2.30 | 2.14 ± 1.95 | 31.83 ± 2.24 | 29.48 ± 2.64 | −2.35 ±1.37 |
| Lean mass (kg) | 50.13 ± 2.56 | 49.08 ± 2.43 | −1.04 ± 1.64 | 49.81 ± 2.65 | 51.54 ± 3.00 | 1.73 ± 1.44 |
| Fasting blood glucose (mg/dL) a | 86.75 ± 1.44 | 86.42 ± 2.90 | −0.33 ± 2.43 | 87.25 ± 2.69 | 89.42 ± 2.42 | 2.17 ± 2.77 |
| Fasting insulin (mUI/mL) a | 6.47 ± 0.66 | 5.76 ± 0.54 | −0.71 ± 0.48 | 6.24 ± 0.82 | 7.38 ± 1.00 | 1.13 ± 0.90 |
| HOMA-IR | 1.40 ± 0.16 | 1.25 ± 0.14 | −0.15 ± 0.12 | 1.38 ± 0.20 | 1.68 ± 0.27 | 0.30 ± 0.25 |
| QUICKI | 0.37 ± 0.03 | 0.38 ± 0.01 | 0.01 ± 0.004 | 0.38 ± 0.04 | 0.36 ± 0.01 | −0.01 ± 0.01 |
| HbA1c (mmol/mol) a | 33.33 ± 1.00 | 34.00 ± 0.82 | 0.67 ± 0.31 | 33.33 ± 1.05 | 33.75 ± 1.02 | 0.42 ± 0.29 |
| Triglycerides (mg/dL) | 70.17 ± 5.74 | 70.83 ± 6.80 | 0.67 ± 7.29 | 77.25 ± 9.98 | 79.17 ± 11.82 | 1.92 ± 9.33 |
| Total cholesterol (mg/dL) | 184.67 ± 7.21 | 193.50 ± 7.95 | 8.83 ± 4.44 ** | 170.50 ± 6.66 | 192.83 ± 6.67 | 22.33 ± 5.46 ** |
| LDL-cholesterol (mg/dL) | 112.67 ± 6.39 | 118.08 ± 7.88 | 5.42 ± 3.60 ** | 100.00 ± 6.29 | 116.00 ± 7.06 | 16.00 ± 4.51 ** |
| HDL-cholesterol (mg/dL) | 57.92 ± 2.87 | 61.25 ± 2.92 | 3.33 ± 1.29 ** | 55.08 ± 2.70 | 61.00 ± 3.36 | 5.92 ± 1.64 ** |
| LDL/HDL | 1.99 ± 0.14 | 1.97 ± 0.15 | −0.03 ± 0.05 | 1.90 ± 0.19 | 2.01 ± 0.20 | 0.10 ± 0.07 |
| non-HDL-cholesterol (mg/dL) | 126.75 ± 6.66 | 132.25 ± 7.38 | 5.50 ± 3.73 ** | 115.42 ± 8.09 | 131.83 ± 8.28 | 16.42 ± 4.53 ** |
| Control (n = 12) | Test (n = 12) | |||||
|---|---|---|---|---|---|---|
| Month 0 | Month 3 | ∆, Month 3-0 | Month 0 | Month 3 | ∆, Month 3-0 | |
| hs-CRP (mg/dL) | 0.23 ± 0.06 # | 0.19 ± 0.04 | −0.03 ± 0.06 | 0.09 ± 0.02# | 0.21 ± 0.06 | 0.11 ± 0.06 |
| IL-6 (pg/mL) | 1.29 ± 0.21 | 1.29 ± 0.24 | 0.00 ± 0.27 | 1.20 ± 0.16 | 1.69 ± 0.36 | 0.49 ± 0.36 |
| TNF-α (pg/mL) | 0.87 ± 0.08 | 0.86 ± 0.07 | −0.01 ± 0.04 | 0.79 ± 0.05 | 0.85 ± 0.05 | 0.06 ± 0.03 |
| Ox-LDL (U/L) | 55.66 ± 4.14 | 59.13 ± 3.75 | 3.47 ± 3.51 | 51.89 ± 4.29 * | 63.06 ± 3.94 * | 11.16 ± 9.81 |
| HbNO (nmol/L) | 102.50 ± 10.37 | 78.44 ± 13.59 | −24.06 ± 17.48 | 110.42 ± 18.29 | 83.86 ± 16.39 | −26.56 ± 14.33 |
| RHI | 2.30 ± 0.18 | 2.17 ± 0.17 | −0.18 ± 0.20 | 2.32 ± 0.19 | 1.98 ± 0.19 | −0.34 ± 0.12 |
| LnRHI | 0.81 ± 0.08 | 0.74 ± 0.09 | −0.09 ± 0.11 | 0.81 ± 0.08 | 0.63 ± 0.10 | −0.17 ± 0.06 |
| AI (%) | 18.74 ± 6.20 | 20.50 ± 8.12 | 1.76 ± 4.33 | 12.60 ± 6.33 | 14.94 ± 5.66 | 2.33 ± 2.66 |
| AI@75bpm (%) | 8.92 ± 6.21 | 12.95 ± 9.01 | 4.03 ± 5.39 | 4.64 ± 5.66 | 7.34 ± 5.22 | 2.69 ± 2.53 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngo Njembe, M.T.; Pachikian, B.; Lobysheva, I.; Van Overstraeten, N.; Dejonghe, L.; Verstraelen, E.; Buchet, M.; Rasse, C.; Gardin, C.; Mignolet, E.; et al. A Three-Month Consumption of Eggs Enriched with ω-3, ω-5 and ω-7 Polyunsaturated Fatty Acids Significantly Decreases the Waist Circumference of Subjects at Risk of Developing Metabolic Syndrome: A Double-Blind Randomized Controlled Trial. Nutrients 2021, 13, 663. https://doi.org/10.3390/nu13020663
Ngo Njembe MT, Pachikian B, Lobysheva I, Van Overstraeten N, Dejonghe L, Verstraelen E, Buchet M, Rasse C, Gardin C, Mignolet E, et al. A Three-Month Consumption of Eggs Enriched with ω-3, ω-5 and ω-7 Polyunsaturated Fatty Acids Significantly Decreases the Waist Circumference of Subjects at Risk of Developing Metabolic Syndrome: A Double-Blind Randomized Controlled Trial. Nutrients. 2021; 13(2):663. https://doi.org/10.3390/nu13020663
Chicago/Turabian StyleNgo Njembe, Monique T., Barbara Pachikian, Irina Lobysheva, Nancy Van Overstraeten, Louis Dejonghe, Eleonore Verstraelen, Marine Buchet, Catherine Rasse, Cécile Gardin, Eric Mignolet, and et al. 2021. "A Three-Month Consumption of Eggs Enriched with ω-3, ω-5 and ω-7 Polyunsaturated Fatty Acids Significantly Decreases the Waist Circumference of Subjects at Risk of Developing Metabolic Syndrome: A Double-Blind Randomized Controlled Trial" Nutrients 13, no. 2: 663. https://doi.org/10.3390/nu13020663
APA StyleNgo Njembe, M. T., Pachikian, B., Lobysheva, I., Van Overstraeten, N., Dejonghe, L., Verstraelen, E., Buchet, M., Rasse, C., Gardin, C., Mignolet, E., Balligand, J.-L., & Larondelle, Y. (2021). A Three-Month Consumption of Eggs Enriched with ω-3, ω-5 and ω-7 Polyunsaturated Fatty Acids Significantly Decreases the Waist Circumference of Subjects at Risk of Developing Metabolic Syndrome: A Double-Blind Randomized Controlled Trial. Nutrients, 13(2), 663. https://doi.org/10.3390/nu13020663







