Herbal Remedies and Their Possible Effect on the GABAergic System and Sleep
Abstract
1. Introduction
2. Stages of Sleep
3. The Role of GABAergic Signaling in Sleep Physiology
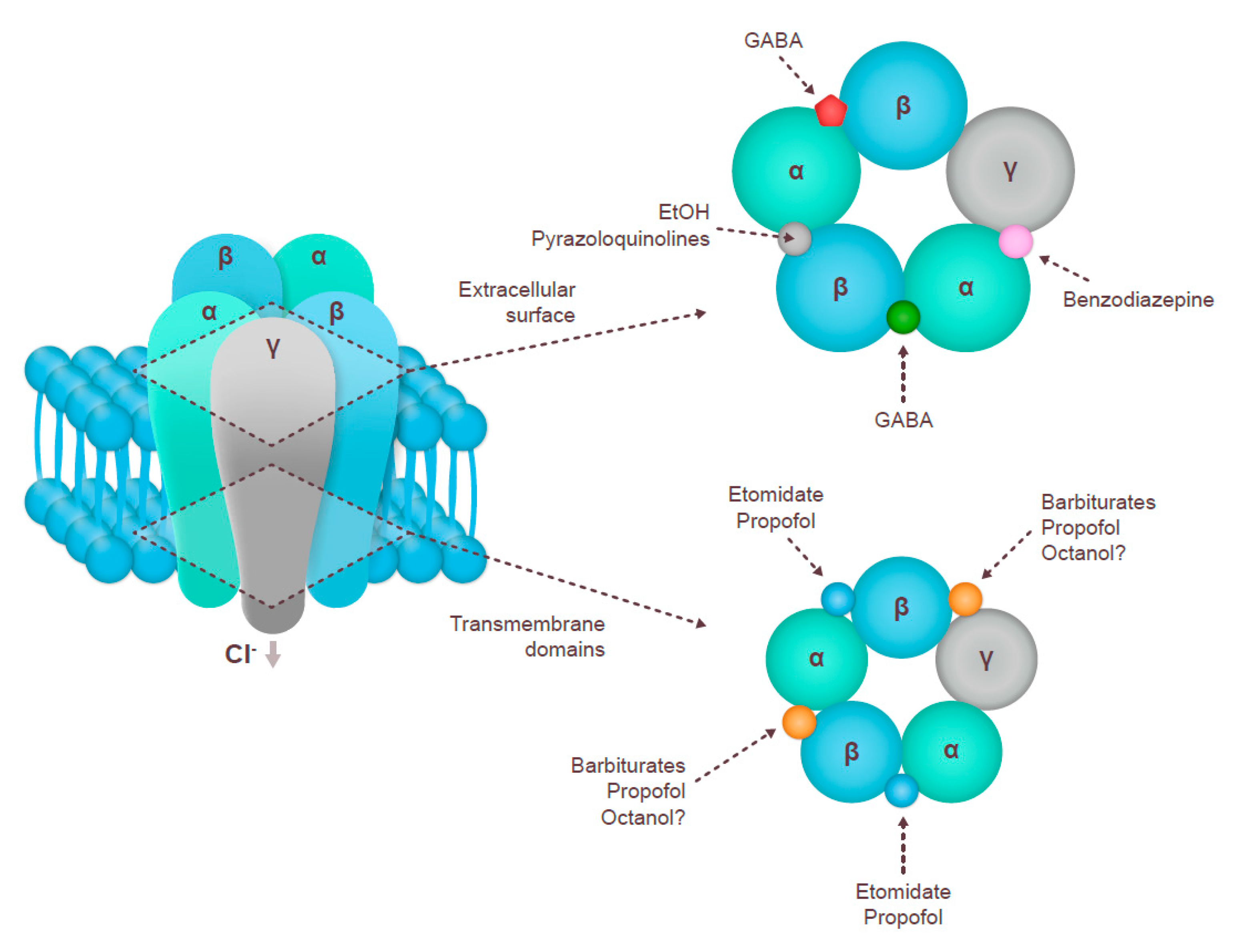
3.1. GABAA Receptor
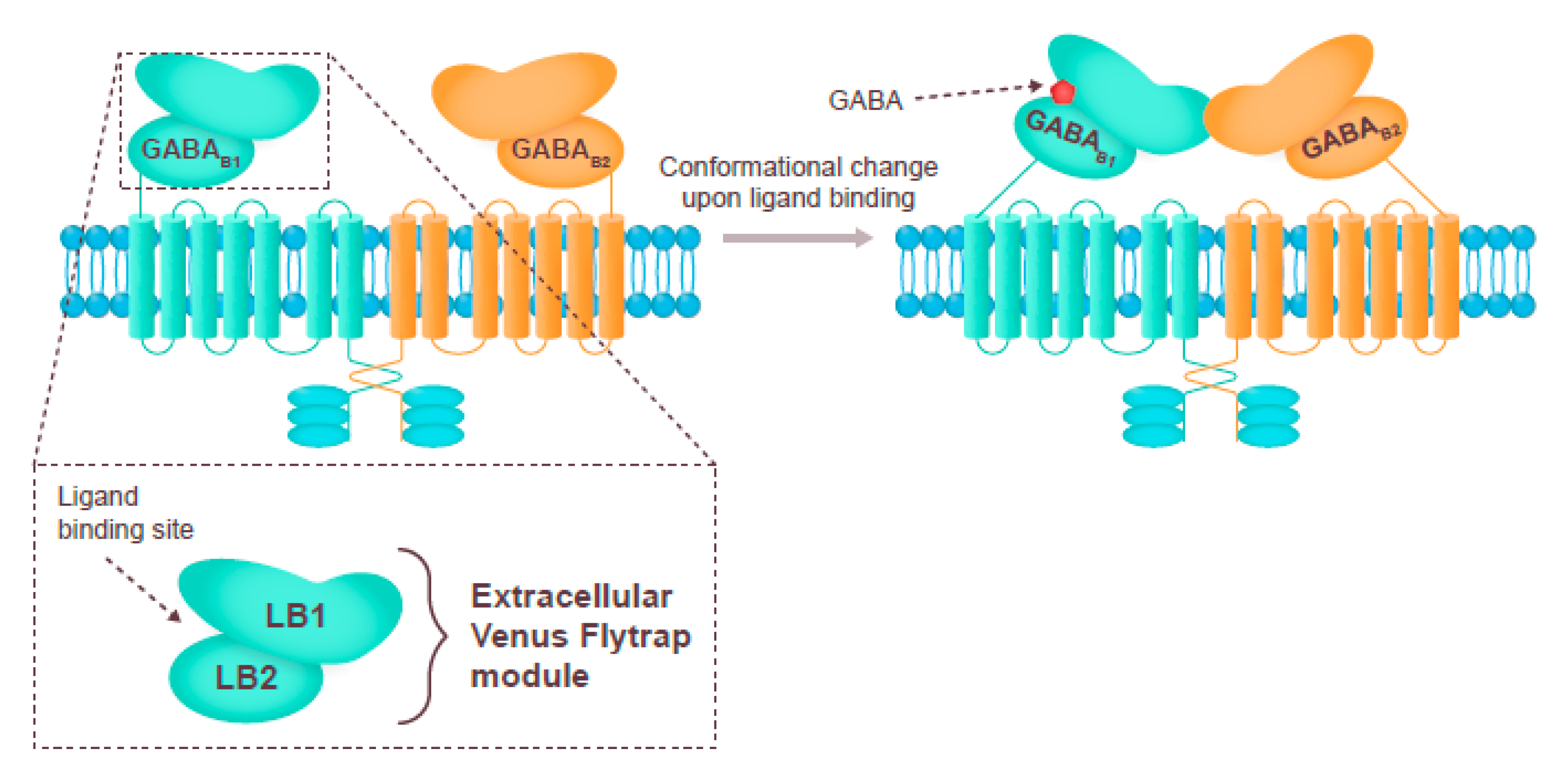
3.2. GABAB Receptor
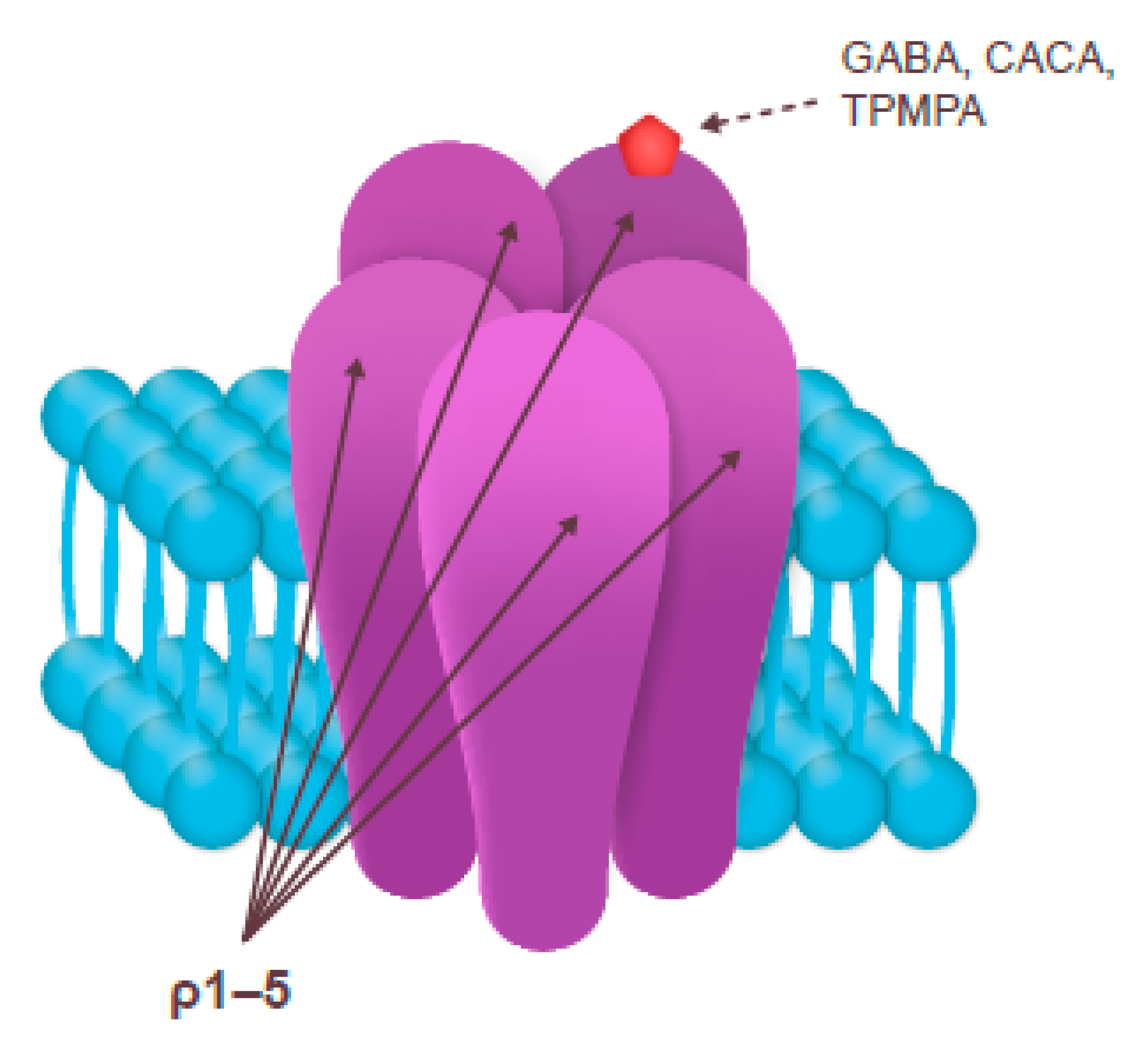
3.3. GABAC Receptor
4. Herbal Remedies Acting on GABA Metabolism and Function
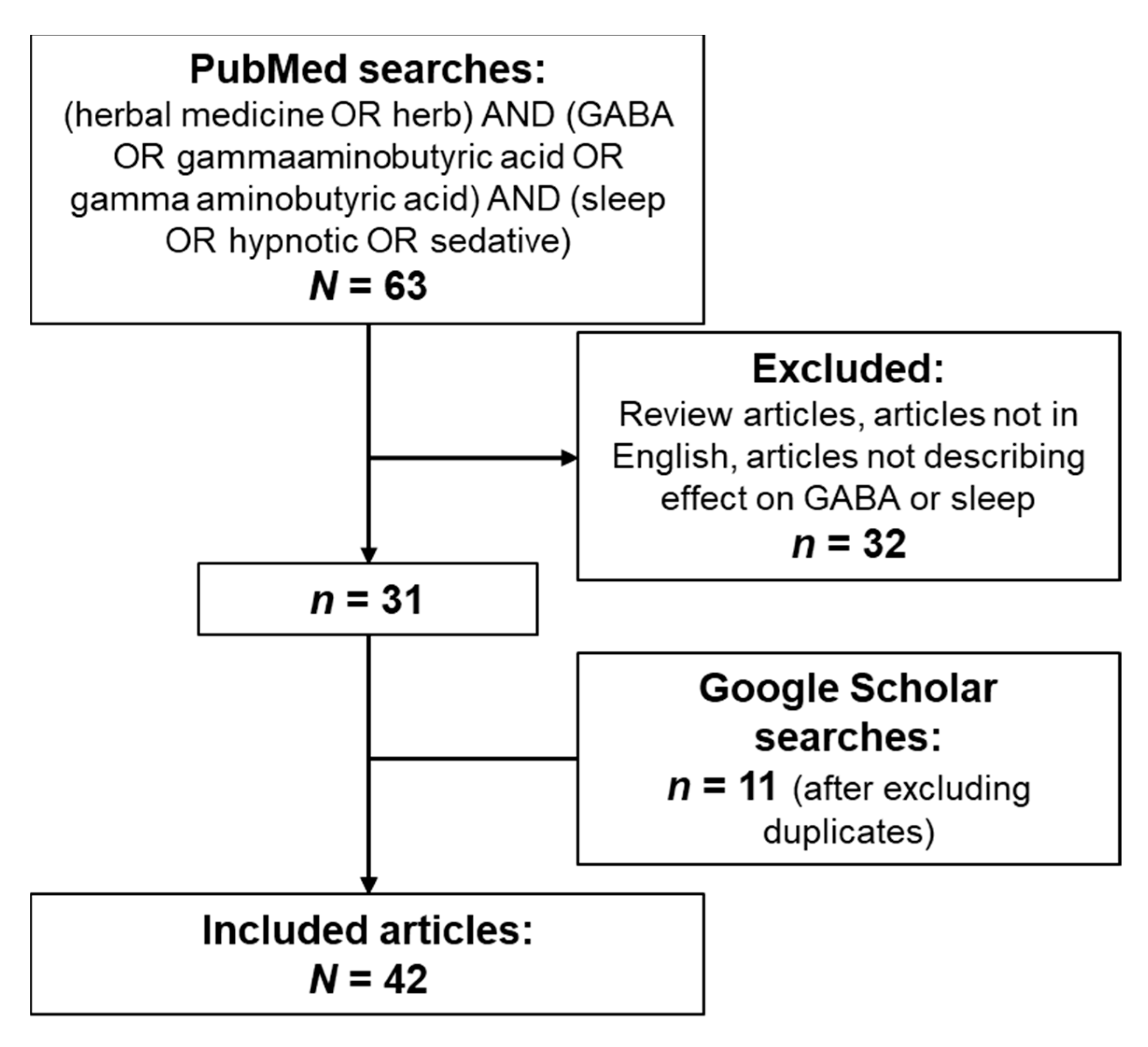
4.1. Systematic Literature Review
4.2. Natural Compounds Acting on GABAA, GABAB, and GABAC
4.3. Other Mechanisms of Action Related to GABA Signaling
5. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Avidan, A.Y.; Neubauer, D.N. Chronic Insomnia Disorder. Continuum (Minneap Minn) 2017, 23, 1064–1092. [Google Scholar] [CrossRef]
- Brown, R.E.; Basheer, R.; McKenna, J.T.; Strecker, R.E.; McCarley, R.W. Control of Sleep and Wakefulness. Physiol. Rev. 2012, 92, 1087–1187. [Google Scholar] [CrossRef] [PubMed]
- Leproult, R.; Van Cauter, E. Role of sleep and sleep loss in hormonal release and metabolism. Endocr. Dev. 2010, 17, 11–21. [Google Scholar] [CrossRef]
- Kripke, D.F. Hypnotic drug risks of mortality, infection, depression, and cancer: But lack of benefit. F1000Research 2016, 5, 918. [Google Scholar] [CrossRef] [PubMed]
- Monograph. Valeriana officinalis. Altern. Med. Rev. 2004, 9, 438–441. [Google Scholar]
- Awad, R.; Levac, D.; Cybulska, P.; Merali, Z.; Trudeau, V.L.; Arnason, J.T. Effects of traditionally used anxiolytic botanicals on enzymes of the gamma-aminobutyric acid (GABA) system. Can. J. Physiol. Pharmacol. 2007, 85, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Schulz, V. Safety of St. John’s Wort extract compared to synthetic antidepressants. Phytomedicine 2006, 13, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Philips. The global pursuit of better sleep health. In Global Sleep Survey; Philips: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Welz, A.N.; Emberger-Klein, A.; Menrad, K. Why people use herbal medicine: Insights from a focus-group study in Germany. BMC Complementary Altern. Med. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, P.; Wu, X.; Zhang, Y.; Cong, D. Effectiveness of Chinese herbal medicine for patients with primary insomnia: A PRISMA-compliant meta-analysis. Medicine (Baltimore) 2019, 98, e15967. [Google Scholar] [CrossRef]
- Frass, M.; Strassl, R.P.; Friehs, H.; Mullner, M.; Kundi, M.; Kaye, A.D. Use and acceptance of complementary and alternative medicine among the general population and medical personnel: A systematic review. Ochsner J. 2012, 12, 45–56. [Google Scholar]
- Ashraf, M.; Saeed, H.; Saleem, Z.; Rathore, H.A.; Rasool, F.; Tahir, E.; Bhatti, T.; Khalid, J.; Bhatti, I.; Tariq, A. A cross-sectional assessment of knowledge, attitudes and self-perceived effectiveness of complementary and alternative medicine among pharmacy and non-pharmacy university students. BMC Complementary Altern. Med. 2019, 19, 95. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Dong, J.W.; Zhao, J.H.; Tang, L.N.; Zhang, J.J. Herbal Insomnia Medications that Target GABAergic Systems: A Review of the Psychopharmacological Evidence. Curr. Neuropharmacol. 2014, 12, 289–302. [Google Scholar] [CrossRef] [PubMed]
- Yeung, W.F.; Chung, K.F.; Poon, M.M.; Ho, F.Y.; Zhang, S.P.; Zhang, Z.J.; Ziea, E.T.; Wong, V.T. Chinese herbal medicine for insomnia: A systematic review of randomized controlled trials. Sleep Med. Rev. 2012, 16, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.L.; Gu, Y.; Wang, W.W.; Lu, L.; Fu, D.L.; Liu, A.J.; Li, H.Q.; Li, J.H.; Lin, Y.; Tang, W.J.; et al. Efficacy and safety of Suanzaoren decoction for primary insomnia: A systematic review of randomized controlled trials. BMC Complementary Altern. Med. 2013, 13, 18. [Google Scholar] [CrossRef] [PubMed]
- Spiegelhalder, K.; Nissen, C.; Riemann, D. Clinical Sleep—Wake Disorders II: Focus on Insomnia and Circadian Rhythm Sleep Disorders. In Sleep-Wake Neurobiology and Pharmacology; Landolt, H.-P., Dijk, D.-J., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 261–276. [Google Scholar] [CrossRef]
- Meolie, A.L.; Rosen, C.; Kristo, D.; Kohrman, M.; Gooneratne, N.; Aguillard, R.N.; Fayle, R.; Troell, R.; Townsend, D.; Claman, D.; et al. Oral nonprescription treatment for insomnia: An evaluation of products with limited evidence. J. Clin. Sleep Med. 2005, 1, 173–187. [Google Scholar] [CrossRef]
- World Health Organization. National Policy on Traditional Medicine and Regulation of Herbal Medicines; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Steiger, A. Sleep and Its Modulation by Substances That Affect GABAA Receptor Function. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 121–146. [Google Scholar] [CrossRef]
- Pal, D.; Mallick, B.N. GABA-ergic Modulation of Pontine Cholinergic and Noradrenergic Neurons for REM Sleep Generation. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 199–212. [Google Scholar] [CrossRef]
- Szabadi, E. Selective targets for arousal-modifying drugs: Implications for the treatment of sleep disorders. Drug Discov. Today 2014, 19, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Szabadi, E. Functional neuroanatomy of the central noradrenergic system. J. Psychopharmacol. 2013, 27, 659–693. [Google Scholar] [CrossRef] [PubMed]
- Reinoso-Suárez, F.; de la Roza, C.; Rodrigo-Angulo, M.L.; de Andrés, I.; Núñez, Á.; Garzón, M. GABAergic Mechanisms in the Ventral Oral Pontine Tegmentum: The REM Sleep—Induction Site—In the Modulation of Sleep—Wake States. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 233–252. [Google Scholar] [CrossRef]
- Möhler, H. Physiology and Pharmacology of the GABA System: Focus on GABA Receptors. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 3–23. [Google Scholar] [CrossRef]
- Gottesmann, C. Function of GABAB and ρ-Containing GABAA Receptors (GABAC Receptors) in the Regulation of Basic and Higher Integrated Sleep-Waking Processes. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 169–188. [Google Scholar] [CrossRef]
- Chebib, M.; Hanrahan, J.R.; Mewett, K.N.; Duke, R.K.; Johnston, G.A.R. Ionotropic GABA Receptors as Therapeutic Targets for Memory and Sleep Disorders. In Annual Reports in Medicinal Chemistry; Elsevier: Amsterdam, The Netherlands, 2004; pp. 13–23. [Google Scholar] [CrossRef]
- Vargas, R. The GABAergic System: An Overview of Physiology, Physiopathology and Therapeutics. Int. J. Clin. Pharmacol. Pharmacother. 2018, 3. [Google Scholar] [CrossRef]
- Wisden, W.; Yu, X.; Franks, N.P. GABA Receptors and the Pharmacology of Sleep. In Sleep-Wake Neurobiology and Pharmacology; Landolt, H.-P., Dijk, D.-J., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 279–304. [Google Scholar] [CrossRef]
- Nutt, D. GABAA receptors: Subtypes, regional distribution, and function. J. Clin. Sleep Med. 2006, 2, S7–S11. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, A.; Wallner, M.; Lindemeyer, A.K.; Olsen, R.W. GABAA Receptor Physiology and Pharmacology. In The Oxford Handbook of Neuronal Ion Channels; Oxford University Press: Oxford, UK, 2018. [Google Scholar] [CrossRef]
- Uusi-Oukari, M.; Korpi, E.R. Regulation of GABAA Receptor Subunit Expression by Pharmacological Agents. Pharmacol. Rev. 2010, 62, 97–135. [Google Scholar] [CrossRef]
- Staner, L.; Cornette, F.; Otmani, S.; Nedelec, J.-F.; Danjou, P. Zolpidem in the Treatment of Adult and Elderly Primary Insomnia Patients. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 383–411. [Google Scholar] [CrossRef]
- Monti, J.M.; Pandi-Perumal, S.R. Eszopiclone: Its use in the treatment of insomnia. Neuropsychiatr. Dis. Treat. 2007, 3, 441–453. [Google Scholar] [PubMed]
- Chinoy, E.D.; Frey, D.J.; Kaslovsky, D.N.; Meyer, F.G.; Wright, K.P., Jr. Age-related changes in slow wave activity rise time and NREM sleep EEG with and without zolpidem in healthy young and older adults. Sleep Med. 2014, 15, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Enna, S.J. GABAB receptor agonists and antagonists: Pharmacological properties and therapeutic possibilities. Expert Opin. Investig. Drugs 2005, 6, 1319–1325. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vienne, J.; Bettler, B.; Franken, P.; Tafti, M. Differential effects of GABAB receptor subtypes, γ-hydroxybutyric Acid, and Baclofen on EEG activity and sleep regulation. J. Neurosci. 2010, 30, 14194–14204. [Google Scholar] [CrossRef] [PubMed]
- Wetter, T.C.; Beitinger, P.A.; Beitinger, M.E.; Wollweber, B. Pathophysiology of Sleep Disorders. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 325–361. [Google Scholar] [CrossRef]
- Landolt, H.-P.; Holst, S.C.; Valomon, A. Clinical and Experimental Human Sleep-Wake Pharmacogenetics. In Sleep-Wake Neurobiology and Pharmacology; Landolt, H.-P., Dijk, D.-J., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 207–241. [Google Scholar] [CrossRef]
- Benarroch, E.E. GABAB receptors: Structure, functions, and clinical implications. Neurology 2012, 78, 578–584. [Google Scholar] [CrossRef]
- Geng, Y.; Bush, M.; Mosyak, L.; Wang, F.; Fan, Q.R. Structural mechanism of ligand activation in human GABA(B) receptor. Nature 2013, 504, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Naffaa, M.M.; Hung, S.; Chebib, M.; Johnston, G.A.R.; Hanrahan, J.R. GABA-ρ receptors: Distinctive functions and molecular pharmacology. Br. J. Pharmacol. 2017, 174, 1881–1894. [Google Scholar] [CrossRef]
- Arnaud, C.; Gauthier, P.; Gottesmann, C. Study of a GABAC receptor antagonist on sleep-waking behavior in rats. Psychopharmacology (Berlin) 2001, 154, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Ulloor, J.; Mavanji, V.; Saha, S.; Siwek, D.F.; Datta, S. Spontaneous REM sleep is modulated by the activation of the pedunculopontine tegmental GABAB receptors in the freely moving rat. J. Neurophysiol. 2004, 91, 1822–1831. [Google Scholar] [CrossRef] [PubMed]
- Kantrowitz, J.; Citrome, L.; Javitt, D. GABA(B) receptors, schizophrenia and sleep dysfunction: A review of the relationship and its potential clinical and therapeutic implications. CNS Drugs 2009, 23, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, P.; Bablon, J.-C.; Da Silva, C. A combination of melatonin, vitamin B6 and medicinal plants in the treatment of mild-to-moderate insomnia: A prospective pilot study. Complementary Ther. Med. 2019, 45, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Cicek, S.S. Structure-Dependent Activity of Natural GABA(A) Receptor Modulators. Molecules 2018, 23, 1512. [Google Scholar] [CrossRef] [PubMed]
- Benke, D.; Barberis, A.; Kopp, S.; Altmann, K.H.; Schubiger, M.; Vogt, K.E.; Rudolph, U.; Mohler, H. GABA A receptors as in vivo substrate for the anxiolytic action of valerenic acid, a major constituent of valerian root extracts. Neuropharmacology 2009, 56, 174–181. [Google Scholar] [CrossRef]
- Patočka, J.; Jakl, J. Biomedically relevant chemical constituents of Valeriana officinalis. J. Appl. Biomed. 2010, 8, 11–18. [Google Scholar] [CrossRef]
- Yuan, C.-S.; Mehendale, S.; Xiao, Y.; Aung, H.H.; Xie, J.-T.; Ang-Lee, M.K. The Gamma-Aminobutyric Acidergic Effects of Valerian and Valerenic Acid on Rat Brainstem Neuronal Activity. Anesth. Analg. 2004, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Mineo, L.; Concerto, C.; Patel, D.; Mayorga, T.; Paula, M.; Chusid, E.; Aguglia, E.; Battaglia, F. Valeriana officinalis Root Extract Modulates Cortical Excitatory Circuits in Humans. Neuropsychobiology 2017, 75, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Khom, S.; Baburin, I.; Timin, E.; Hohaus, A.; Trauner, G.; Kopp, B.; Hering, S. Valerenic acid potentiates and inhibits GABA(A) receptors: Molecular mechanism and subunit specificity. Neuropharmacology 2007, 53, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Alexeev, M.; Grosenbaugh, D.K.; Mott, D.D.; Fisher, J.L. The natural products magnolol and honokiol are positive allosteric modulators of both synaptic and extra-synaptic GABA(A) receptors. Neuropharmacology 2012, 62, 2507–2514. [Google Scholar] [CrossRef] [PubMed]
- Qu, W.M.; Yue, X.F.; Sun, Y.; Fan, K.; Chen, C.R.; Hou, Y.P.; Urade, Y.; Huang, Z.L. Honokiol promotes non-rapid eye movement sleep via the benzodiazepine site of the GABA(A) receptor in mice. Br. J. Pharmacol. 2012, 167, 587–598. [Google Scholar] [CrossRef]
- Squires, R.F.; Ai, J.; Witt, M.R.; Kahnberg, P.; Saederup, E.; Sterner, O.; Nielsen, M. Honokiol and magnolol increase the number of [3H] muscimol binding sites three-fold in rat forebrain membranes in vitro using a filtration assay, by allosterically increasing the affinities of low-affinity sites. Neurochem. Res. 1999, 24, 1593–1602. [Google Scholar] [CrossRef]
- Li, N.; Liu, J.; Wang, M.; Yu, Z.; Zhu, K.; Gao, J.; Wang, C.; Sun, J.; Chen, J.; Li, H. Sedative and hypnotic effects of Schisandrin B through increasing GABA/Glu ratio and upregulating the expression of GABA(A) in mice and rats. Biomed. Pharmacother. 2018, 103, 509–516. [Google Scholar] [CrossRef]
- Wang, M.; Li, N.; Jing, S.; Wang, C.; Sun, J.; Li, H.; Liu, J.; Chen, J. Schisandrin B exerts hypnotic effects in PCPA-treated rats by increasing hypothalamic 5-HT and γ-aminobutyric acid levels. Exp. Ther. Med. 2020, 20, 142. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, X.; Mao, X.; Liu, A.; Liu, Z.; Li, X.; Bi, K.; Jia, Y. Pharmacological evaluation of sedative and hypnotic effects of schizandrin through the modification of pentobarbital-induced sleep behaviors in mice. Eur. J. Pharmacol 2014, 744, 157–163. [Google Scholar] [CrossRef]
- Emadi, F.; Yassa, N.; Hadjiakhoondi, A.; Beyer, C.; Sharifzadeh, M. Sedative effects of Iranian Artemisia annua in mice: Possible benzodiazepine receptors involvement. Pharm. Biol. 2011, 49, 784–788. [Google Scholar] [CrossRef]
- Kavvadias, D.; Abou-Mandour, A.A.; Czygan, F.C.; Beckmann, H.; Sand, P.; Riederer, P.; Schreier, P. Identification of benzodiazepines in Artemisia dracunculus and Solanum tuberosum rationalizing their endogenous formation in plant tissue. Biochem. Biophys. Res. Commun. 2000, 269, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.Z.; Chang, Q.; Zhong, Y.; Xiao, B.X.; Feng, L.; Cao, F.R.; Pan, R.L.; Zhang, Z.S.; Liao, Y.H.; Liu, X.M. Lotus Leaf Alkaloid Extract Displays Sedative-Hypnotic and Anxiolytic Effects through GABAA Receptor. J. Agric. Food Chem. 2015, 63, 9277–9285. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.L.; Wu, B.F.; Shang, J.H.; Zhao, Y.L.; Huang, A.X. Moringa oleifera Lam Seed Oil Augments Pentobarbital-Induced Sleeping Behaviors in Mice via GABAergic Systems. J. Agric. Food Chem. 2020, 68, 3149–3162. [Google Scholar] [CrossRef]
- Shinomiya, K.; Inoue, T.; Utsu, Y.; Tokunaga, S.; Masuoka, T.; Ohmori, A.; Kamei, C. Effects of kava-kava extract on the sleep-wake cycle in sleep-disturbed rats. Psychopharmacology (Berlin) 2005, 180, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Han, H.; Eun, J.S.; Kim, H.C.; Hong, J.T.; Oh, K.W. Sanjoinine A isolated from Zizyphi Spinosi Semen augments pentobarbital-induced sleeping behaviors through the modification of GABA-ergic systems. Biol. Pharm. Bull. 2007, 30, 1748–1753. [Google Scholar] [CrossRef] [PubMed]
- Yi, P.L.; Lin, C.P.; Tsai, C.H.; Lin, J.G.; Chang, F.C. The involvement of serotonin receptors in suanzaorentang-induced sleep alteration. J. Biomed. Sci. 2007, 14, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Appel, K.; Rose, T.; Fiebich, B.; Kammler, T.; Hoffmann, C.; Weiss, G. Modulation of the gamma-aminobutyric acid (GABA) system by Passiflora incarnata L. Phytother. Res. 2011, 25, 838–843. [Google Scholar] [CrossRef]
- Elsas, S.M.; Rossi, D.J.; Raber, J.; White, G.; Seeley, C.A.; Gregory, W.L.; Mohr, C.; Pfankuch, T.; Soumyanath, A. Passiflora incarnata L. (Passionflower) extracts elicit GABA currents in hippocampal neurons in vitro, and show anxiogenic and anticonvulsant effects in vivo, varying with extraction method. Phytomedicine 2010, 17, 940–949. [Google Scholar] [CrossRef]
- Candelario, M.; Cuellar, E.; Reyes-Ruiz, J.M.; Darabedian, N.; Feimeng, Z.; Miledi, R.; Russo-Neustadt, A.; Limon, A. Direct evidence for GABAergic activity of Withania somnifera on mammalian ionotropic GABAA and GABArho receptors. J. Ethnopharmacol. 2015, 171, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Langade, D.; Kanchi, S.; Salve, J.; Debnath, K.; Ambegaokar, D. Efficacy and Safety of Ashwagandha (Withania somnifera) Root Extract in Insomnia and Anxiety: A Double-blind, Randomized, Placebo-controlled Study. Cureus 2019, 11, e5797. [Google Scholar] [CrossRef] [PubMed]
- Fedurco, M.; Gregorová, J.; Šebrlová, K.; Kantorová, J.; Peš, O.; Baur, R.; Sigel, E.; Táborská, E. Modulatory Effects of Eschscholzia californica Alkaloids on Recombinant GABAAReceptors. Biochem. Res. Int. 2015, 2015, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.W.; Peng, C.B.; Pei, Z.; Zhang, M.R.; Yun, T.C.; Yang, Z.M.; Xu, F.P. Effects of tenuifolin on rest/wake behaviour in zebrafish. Exp. Ther. Med. 2020, 19, 2326–2334. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.J.; Wang, G.Y.; Zhang, X.P.; Wang, Q.Q.; Peng, Z.P. Sedative and Hypnotic Effects and Transcriptome Analysis of Polygala tenuifolia in Aged Insomnia Rats. Chin. J. Integr. Med. 2020, 26, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.Y.; Choi, J.H.; Kim, W.; Yoo, K.Y.; Lee, C.H.; Yoon, Y.S.; Won, M.H.; Hwang, I.K. Effects of Melissa officinalis L. (lemon balm) extract on neurogenesis associated with serum corticosterone and GABA in the mouse dentate gyrus. Neurochem. Res. 2011, 36, 250–257. [Google Scholar] [CrossRef]
- Murray, B.J.; Cowen, P.J.; Sharpley, A.L. The effect of Li 1370, extract of Ginkgo biloba, on REM sleep in humans. Pharmacopsychiatry 2001, 34, 155–157. [Google Scholar] [CrossRef] [PubMed]
- Sharpley, A.L.; McGavin, C.L.; Whale, R.; Cowen, P.J. Antidepressant-like effect of Hypericum perforatum (St John’s wort) on the sleep polysomnogram. Psychopharmacology (Berlin) 1998, 139, 286–287. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.A.; Cury, T.C.; Cassettari, B.O.; Takahira, R.K.; Florio, J.C.; Costa, M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT(1A)-receptors and reduces cholesterol after repeated oral treatment. BMC Complementary Altern. Med. 2013, 13, 42. [Google Scholar] [CrossRef] [PubMed]
- Egashira, N.; Nogami, A.; Iwasaki, K.; Ishibashi, A.; Uchida, N.; Takasaki, K.; Mishima, K.; Nishimura, R.; Oishi, R.; Fujiwara, M. Yokukansan enhances pentobarbital-induced sleep in socially isolated mice: Possible involvement of GABA(A)-benzodiazepine receptor complex. J. Pharmacol. Sci. 2011, 116, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Ozone, M.; Shimazaki, H.; Ichikawa, H.; Shigeta, M. Efficacy of yokukansan compared with clonazepam for rapid eye movement sleep behaviour disorder: A preliminary retrospective study. Psychogeriatrics 2020, 20, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Yi, P.L.; Tsai, C.H.; Chen, Y.C.; Chang, F.C. Gamma-aminobutyric acid (GABA) receptor mediates suanzaorentang, a traditional Chinese herb remedy, -induced sleep alteration. J. Biomed. Sci. 2007, 14, 285–297. [Google Scholar] [CrossRef]
- Pagel, J.F.; Kram, G. Insomnia: Differential Diagnosis and Current Treatment Approach. In GABA and Sleep: Molecular, Functional and Clinical Aspects; Monti, J.M., Pandi-Perumal, S.R., Möhler, H., Eds.; Springer: Basel, Switzerland, 2010; pp. 363–381. [Google Scholar] [CrossRef]
- Chen, H.Y.; Chang, S.S.; Chan, Y.C.; Chen, C.Y. Discovery of novel insomnia leads from screening traditional Chinese medicine database. J. Biomol. Struct. Dyn. 2014, 32, 776–791. [Google Scholar] [CrossRef]
- Kim, J.; Han, M.; Jeon, W.K. Acute and Subacute Oral Toxicity of Mumefural, Bioactive Compound Derived from Processed Fruit of Prunus mume Sieb. et Zucc., in ICR Mice. Nutrients 2020, 12, 1328. [Google Scholar] [CrossRef]
- Srivastava, J.K.; Shankar, E.; Gupta, S. Chamomile: A herbal medicine of the past with bright future. Mol. Med. Rep. 2010, 3, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Savage, K.; Firth, J.; Stough, C.; Sarris, J. GABA-modulating phytomedicines for anxiety: A systematic review of preclinical and clinical evidence. Phytother. Res. 2018, 32, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, M.K.; Kaul, S.C.; Wadhwa, R.; Yanagisawa, M.; Urade, Y. Triethylene glycol, an active component of Ashwagandha (Withania somnifera) leaves, is responsible for sleep induction. PLoS ONE 2017, 12, e0172508. [Google Scholar] [CrossRef] [PubMed]
- Lozada-Lechuga, J.; Villarreal, M.L.; Fliniaux, M.A.; Bensaddek, L.; Mesnard, F.; Gutierrez Mdel, C.; Cardoso-Taketa, A.T. Isolation of jacaranone, a sedative constituent extracted from the flowers of the Mexican tree Ternstroemia pringlei. J. Ethnopharmacol 2010, 127, 551–554. [Google Scholar] [CrossRef]
- Khosravi, M.; Khakpour, S.; Adibi, L.; Jahromy, M.H. A Study of the Effect of Citrus aurantium L. Essential Oil on Anxiety and Its Interaction with GABAergic Pathways in Male Mice. J. Behav. Brain Sci. 2014, 4, 470–476. [Google Scholar] [CrossRef]
- Romero, K.; Goparaju, B.; Russo, K.; Westover, M.B.; Bianchi, M.T. Alternative remedies for insomnia: A proposed method for personalized therapeutic trials. Nat. Sci. Sleep 2017, 9, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Leach, M.J.; Page, A.T. Herbal medicine for insomnia: A systematic review and meta-analysis. Sleep Med. Rev. 2015, 24, 1–12. [Google Scholar] [CrossRef]
- Liu, L.; Liu, C.; Wang, Y.; Wang, P.; Li, Y.; Li, B. Herbal Medicine for Anxiety, Depression and Insomnia. Curr. Neuropharmacol. 2015, 13, 481–493. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Zheng, X.; Qu, L.; Zhang, H.; Yuan, H.; Hui, J.; Mi, Y.; Ma, P.; Fan, D. Ginsenoside Rg5/Rk1 ameliorated sleep via regulating the GABAergic/serotoninergic signaling pathway in a rodent model. Food Funct. 2020, 11, 1245–1257. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, P.A.; Adosraku, R.K. An HPLC method for the direct assay of the serotonin precursor, 5-hydroxytrophan, in seeds of Griffonia simplicifolia. Phytochem. Anal. 2002, 13, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Gray, M.A.; Oliver, C.; Liley, D.T.; Harrison, B.J.; Bartholomeusz, C.F.; Phan, K.L.; Nathan, P.J. The acute effects of L-theanine in comparison with alprazolam on anticipatory anxiety in humans. Hum. Psychopharmacol. 2004, 19, 457–465. [Google Scholar] [CrossRef] [PubMed]
- López, V.; Nielsen, B.; Solas, M.; Ramírez, M.J.; Jäger, A.K. Exploring Pharmacological Mechanisms of Lavender (Lavandula angustifolia) Essential Oil on Central Nervous System Targets. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.M.; Shimizu, M.; Lee, C.J.; Han, D.S.; Jung, C.K.; Jo, J.H.; Kim, Y.M. Hypnotic effects and binding studies for GABA(A) and 5-HT(2C) receptors of traditional medicinal plants used in Asia for insomnia. J. Ethnopharmacol. 2010, 132, 225–232. [Google Scholar] [CrossRef] [PubMed]
| Latin and Common Name | Known Chemical Components | Known Effect on Sleep | Target | Model | References |
|---|---|---|---|---|---|
| Individual Plants | |||||
| Valeriana officinalis L. (Valerian) | Alkaloids, terpenes, organic acids and their derivatives, valepotriates, and flavones | Reduces sleep latency, improves subjective measures | GABAA receptor | In vitro studies; clinical studies | [47,48,49,50,51] |
| Magnolia sp. | Magnolol and honokiol | Promotes REM sleep | GABAA receptor | In vitro studies; i.p. administration in mice | [52,53,54] |
| Schisandra chinensis (Turcz.) Baill. (Chinese magnolia-vine, Magnolia berry) | Schizandrin B | Promotes sleep | GABAA receptor | i.p. administration in mice and male rats | [55,56,57] |
| Artemisia sp. | Benzodiazepines | Reduces sleep latency | GABAA receptor | In vitro studies; i.p. administration in male mice | [58,59] |
| Nelumbo nucifera Gaertn. (Lotus) | Nuciferine, alkaloids | Promotes sleep | GABAA receptor | In vitro studies | [60] |
| Moringa oleifera Lam. (Drumstick tree) | Oleic acid, β-Sitosterol, and Stigmasterol | Increases sleep quality | GABAA receptor | p.o. administration in male mice | [61] |
| Piper methysticum L. (Kava-kava) | Kavapyrones | Decreases sleep latency; no effect on NREM sleep | GABAA receptor (not benzodiazepine site) | p.o. administration in mice | [62] |
| Zizyphus jujube (Jujube, or red date) | Sanjoinine A, suanzaorentang | Improves sleep quality, prolonging sleep time and increasing NREM sleep | GABAA receptor; activation of GABA synthesis through enhanced expression of GAD; serotonin receptors | i.p. and p.o. administration in male rats | [63,64] |
| Passiflora incarnata (Passionflower) | Apigenin, alkaloids, flavones | Reduces sleep latency, increases sleep duration | GABAA and GABAB receptors, (and possibly GABAC receptor) | In vitro studies; p.o. administration in mice | [65,66] |
| Withania somnifera L. (Indian ginseng) | Withanolide A, withaferin A | Reduces sleep latency, improves sleep quality | GABAA and GABAC receptors | In vitro studies; clinical studies | [67,68] |
| Eschscholzia californica Cham. (Californian poppy) | Alkaloids | Improves sleep latency and duration | GABAA receptor; serotonin receptor | In vitro studies | [69] |
| Polygala tenuifolia Willd. (Yuan Zhi) | Tenufolin | Increases sleep duration | Increases the levels of GABA and GABA transporter 1 | Zebrafish and rats | [70,71] |
| Melissa officinalis L. (Lemon balm) | Rosmarinic acid | Improves sleep quality | Decreases the level of GABA transaminase | In vitro studies; i.p. administration in mice | [72] |
| Ginkgo biloba L. (Ginkgo) | Ginkgotoxin, flavonoids, terpenoids | Improves subjective sleep quality measures | Inhibition of GAD activity | Clinical studies | [73] |
| Hypericum perforatum L. (St John’s Wort) | Hypericin, pseudohypericin, hyperoside, among others | Increases REM latency and deep sleep | Inhibition of GAD and GABA transporter activity | Clinical studies | [74] |
| Citrus aurantium L. (bitter orange) | Limonene, β-myrcene | Increases sleep duration | Serotonergic system; proposed interaction with GABA receptor binders, such as diazepam | p.o administration in male mice | [75] |
| Plant mixes | |||||
| Yokukansan (Atractylodes lancea rhizoma, Poria sclerotium, Cnidium rhizoma, Japanese Angelica radix, Bupleurum radix, Glycyrrhiza radix, and Uncaria thorn) | Various | Decreases sleep latency, improves dream content in the REM behavior disorder | GABAA receptor | p.o. administration in male mice; clinical studies | [76,77] |
| Suanzaorentang, a traditional Chinese medicine | Various | Increases NREM, no effect on REM sleep | GABAA receptor; serotonergic system | Clinical studies | [64,78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruni, O.; Ferini-Strambi, L.; Giacomoni, E.; Pellegrino, P. Herbal Remedies and Their Possible Effect on the GABAergic System and Sleep. Nutrients 2021, 13, 530. https://doi.org/10.3390/nu13020530
Bruni O, Ferini-Strambi L, Giacomoni E, Pellegrino P. Herbal Remedies and Their Possible Effect on the GABAergic System and Sleep. Nutrients. 2021; 13(2):530. https://doi.org/10.3390/nu13020530
Chicago/Turabian StyleBruni, Oliviero, Luigi Ferini-Strambi, Elena Giacomoni, and Paolo Pellegrino. 2021. "Herbal Remedies and Their Possible Effect on the GABAergic System and Sleep" Nutrients 13, no. 2: 530. https://doi.org/10.3390/nu13020530
APA StyleBruni, O., Ferini-Strambi, L., Giacomoni, E., & Pellegrino, P. (2021). Herbal Remedies and Their Possible Effect on the GABAergic System and Sleep. Nutrients, 13(2), 530. https://doi.org/10.3390/nu13020530






