Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristic of the Examined Groups
2.2. Collection and Preparation of Samples
2.3. Determination of Mineral Components and Total Antioxidant Status
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Szczudlik, A.; Barcikowska-Kotowicz, M.; Gabryelewicz, T.; Opala, G.; Parnowski, T.; Kuźnicki, J.; Rossa, A.; Sadowska, A. The situation of people suffering from Alzheimer’s disease in Poland; RPO Report; Sytuacja osób chorych na chorobę Alzheimera w Polsce; Polish Alzheimer’s Society: Warsaw, Poland, 2016; Volume 3, pp. 33–34. (In Polish) [Google Scholar]
- Bertram, L.; Lill, C.M.; Tanzi, R.E. The genetics of Alzheimer disease: Back to the future. Neuron 2010, 68, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Glenner, G.G. Alzheimer’s disease. The commonest form of amyloidosis. Arch. Pathol. Lab. Med. 1983, 107, 281–282. [Google Scholar] [PubMed]
- Masters, C.L.; Simms, G.; Weinman, N.A.; Multhaup, G.; McDonald, B.L.; Beyreuther, K. Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl. Acad. Sci. USA 1985, 82, 4245–4249. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.C.; Jiang, Z.F. Accumulated amyloid-beta peptide and hyperphosphorylated tau protein: Relationship and links in Alzheimer’s disease. J. Alzheimers Dis. 2009, 16, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Bartzokis, G. Alzheimer’s disease as homeostatic responses to age-related myelin breakdown. Neurobiol. Aging 2011, 32, 1341–1371. [Google Scholar] [CrossRef]
- Bonda, D.J.; Wang, X.; Perry, G.; Nunomura, A.; Tabaton, M.; Zhu, X.; Smith, M.A. Oxidative stress in Alzheimer disease: A possibility for prevention. Neuropharmacology 2010, 59, 290–294. [Google Scholar] [CrossRef]
- Schrag, M.; Mueller, C.; Zabel, M.; Crofton, A.; Kirsch, W.M.; Ghribi, O.; Squitti, R.; Perry, G. Oxidative stress in blood in Alzheimer’s disease and mild cognitive impairment: A meta-analysis. Neurobiol. Dis. 2013, 59, 100–110. [Google Scholar] [CrossRef]
- Kim, G.H.; Kim, J.E.; Rhie, S.J.; Yoon, S. The role of oxidative stress in neurodegenerative diseases. Exp. Neurobiol. 2015, 24, 325–340. [Google Scholar] [CrossRef]
- Chen, J.; Berry, M.J. Selenium and selenoproteins in the brain and brain diseases. J. Neurochem. 2003, 86, 1–12. [Google Scholar] [CrossRef]
- Benton, D. Selenium intake, mood and other aspects of psychological functioning. Nutr. Neurosci. 2002, 5, 363–374. [Google Scholar] [CrossRef]
- Schweizer, U.; Schomburg, L.; Savaskan, N.E. The neurobiology of selenium: Lessons from transgenic mice. J. Nutr. 2004, 134, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Crack, P.J.; Cimdins, K.; Ali, U.; Hertzog, P.J.; Iannello, R.C. Lack of glutathione peroxidase-1 exacerbates Abeta -mediated neurotoxicity in cortical neurons. J. Neural. Trans. Vienna. 2006, 113, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Staehelin, H.B. Micronutrients and Alzheimer’s disease. Proc. Nutr. Soc. 2005, 64, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc: Role in immunity, oxidative stress and chronic inflammation. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Bitanihirwe, B.K.; Cunningham, M. Zinc: The brain’s dark horse. Synapse 2009, 63, 1029–1049. [Google Scholar] [CrossRef] [PubMed]
- Markiewicz-Żukowska, R.; Gutowska, A.; Borawska, M.H. Serum zinc concentrations correlate with mental and physical status of nursing home residents. PLoS ONE 2015, 10, e0117257. [Google Scholar] [CrossRef] [PubMed]
- Sensi, S.L.; Granzotto, A.; Siotto, M.; Squitti, R. Copper and zinc dysregulation in Alzheimer’s disease. Trends Pharmacol. Sci. 2018, 39, 1049–1063. [Google Scholar] [CrossRef]
- Pfaender, S.; Grabrucker, A.M. Characterization of biometal profiles in neurological disorders. Metallomics 2014, 6, 960–977. [Google Scholar] [CrossRef]
- Strozyk, D.; Launer, L.J.; Adlard, P.A.; Cherny, R.A.; Tsatsanis, A.; Volitakis, I.; Blennow, K.; Petrovitch, H.; White, L.R.; Bush, A.I. Zinc and copper modulate Alzheimer Abeta levels in human cerebrospinal fluid. Neurobiol. Aging 2009, 30, 1069–1077. [Google Scholar] [CrossRef]
- Cornett, C.R.; Markesbery, W.R.; Ehmann, W.D. Imbalances of trace elements related to oxidative damage in Alzheimer’s disease brain. Neurotoxicology 1998, 19, 339–345. [Google Scholar]
- Perry, G.; Cash, A.D.; Smith, M.A. Alzheimer disease and oxidative stress. J. Biomed. Biotechnol. 2002, 2, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.J.; Zhang, X.; Chen, W.W. Role of oxidative stress in Alzheimer’s disease. Biomed. Rep. 2016, 4, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Feldman, H.H.; Jacova, C.; Dekosky, S.T.; Barberger-Gateau, P.; Cummings, J.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G.; et al. Research criteria for the diagnosis of Alzheimer’s disease: Revising the NINCDS-ADRDA criteria. Lancet Neurol. 2007, 6, 734–746. [Google Scholar] [CrossRef]
- Gronowska-Senger, A. Przewodnik Metodyczny Badań Sposobu Żywienia; Committee of Human Nutrition Science, Polish Academy of Sciences: Warsaw, Poland, 2013. (In Polish) [Google Scholar]
- Dembińska-Kieć, A.; Solnica, B.; Naskalski, J. Diagnostyka Laboratoryjna Z Elementami Biochemii Klinicznej, 4th ed.; Edra Urban & Partner: Wrocław, Poland, 2017; p. 23. (In Polish) [Google Scholar]
- Total Antioxidant Status (TAS). Available online: https://www.randox.com/total-antioxidant-status/ (accessed on 18 December 2020).
- De Benedictis, C.A.; Vilella, A.; Grabrucker, A.M. The role of trace metals in Alzheimer’s disease. In Alzheimer’s Disease; Wisniewski, T., Ed.; Codon Publications: Brisbane, Australia, 2019; Chapter 6; p. 31895516. [Google Scholar]
- Cardoso, B.R.; Ong, T.P.; Jacob-Filho, W.; Jaluul, O.; Freitas, M.I.; Cozzolino, S.M. Nutritional status of selenium in Alzheimer’s disease patients. Br. J. Nutr. 2010, 103, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Vural, H.; Demirin, H.; Kara, Y.; Eren, I.; Delibas, N. Alterations of plasma magnesium, copper, zinc, iron and selenium concentrations and some related erythrocyte antioxidant enzyme activities in patients with Alzheimer’s disease. J. Trace Elem. Med. Biol. 2010, 24, 169–173. [Google Scholar] [CrossRef]
- Xu, J.; Church, S.J.; Patassini, S.; Begley, P.; Kellett, K.A.B.; Vardy, E.R.L.C.; Unwin, R.D.; Hooper, N.M.; Cooper, G.J.S. Plasma metals as potential biomarkers in dementia: A case-control study in patients with sporadic Alzheimer’s disease. Biometals 2018, 31, 267–276. [Google Scholar] [CrossRef]
- González-Domínguez, R.; García-Barrera, T.; Gómez-Ariza, J.L. Homeostasis of metals in the progression of Alzheimer’s disease. Biometals 2014, 27, 539–549. [Google Scholar] [CrossRef]
- Luchsinger, J.A.; Mayeux, R. Dietary factors and Alzheimer’s disease. Lancet Neurol. 2004, 3, 579–587. [Google Scholar] [CrossRef]
- Ceballos-Picot, I.; Merad-Boudia, M.; Nicole, A.; Thevenin, M.; Hellier, G.; Legrain, S.; Berr, C. Peripheral antioxidant enzyme activities and selenium in elderly subjects and in dementia of Alzheimer’s type-place of the extracellular glutathione peroxidase. Free Radic. Biol. Med. 1996, 20, 579–587. [Google Scholar] [CrossRef]
- Smorgon, C.; Mari, E.; Atti, A.R.; Dalla Nora, E.; Zamboni, P.F.; Calzoni, F.; Passaro, A.; Fellin, R. Trace elements and cognitive impairment: An elderly cohort study. Arch. Gerontol. Geriatr. Suppl. 2004, 9, 393–402. [Google Scholar] [CrossRef]
- Berr, C.; Balansard, B.; Arnaud, J.; Roussel, A.M.; Alpérovitch, A. Cognitive decline is associated with systemic oxidative stress: The EVA study—Etude du Vieillissement Arte’riel. J. Am. Geriatr. Soc. 2000, 48, 1285–1291. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Jin, Y.; Hall, K.S.; Liang, C.; Unverzagt, F.W.; Ji, R.; Murrell, J.R.; Cao, J.; Shen, J.; Ma, F.; et al. Selenium level and cognitive function in rural elderly Chinese. Am. J. Epidemiol. 2007, 165, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Akbaraly, N.T.; Hininger-Favier, I.; Carrière, I.; Arnaud, J.; Gourlet, V.; Roussel, A.M.; Berr, C. Plasma selenium over time and cognitive decline in the elderly. Epidemiology 2007, 18, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Shreenath, A.P.; Ameer, M.A.; Dooley, J. Selenium Deficiency. In StatPearls [Internet]. Treasure Island, FL, USA; StatPearls Publishing, 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482260/ (accessed on 15 January 2021).
- Cardoso, B.R.; Hare, D.J.; Bush, A.I.; Roberts, B.R. Glutathione peroxidase 4: A new player in neurodegeneration? Mol. Psychiatry 2017, 22, 328–335. [Google Scholar] [CrossRef]
- Zhang, S.; Rocourt, C.; Cheng, W.-H. Selenoproteins and the aging brain. Mech. Ageing Dev. 2010, 131, 253–260. [Google Scholar] [CrossRef]
- Pitts, M.W.; Byrns, C.N.; Ogawa-Wong, A.N.; Kremer, P.; Berry, M.J. Selenoproteins in nervous system development and function. Biol. Trace Elem. Res. 2014, 161, 231–245. [Google Scholar] [CrossRef]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Chen, C.J.; Lai, J.S.; Wu, C.C.; Lin, T.S. Serum selenium in adult Taiwanese. Sci. Total. Environ. 2006, 367, 448–450. [Google Scholar] [CrossRef]
- Arnaud, J.; Akbaraly, T.N.; Hininger, I.; Roussel, A.M.; Berr, C. Factors associated with longitudinal plasma selenium decline in the elderly: The EVA study. J. Nutr. Biochem. 2007, 18, 482–487. [Google Scholar] [CrossRef]
- Letsiou, S.; Nomikos, T.; Panagiotakos, D.; Pergantis, S.A.; Fragopoulou, E.; Antonopoulou, S.; Pitsavos, C.; Stefanadis, C. Serum total selenium status in Greek adults and its relation to age. The ATTICA study cohort. Biol. Trace Elem. Res. 2009, 128, 8–17. [Google Scholar] [CrossRef]
- Planas, M.; Conde, M.; Audivert, S.; Pérez-Portabella, C.; Burgos, R.; Chacón, P.; Rossello, J.; Boada, M.; Tàrraga, L.L. Micronutrient supplementation in mild Alzheimer disease patients. Clin. Nutr. 2004, 23, 265–272. [Google Scholar] [CrossRef]
- Wąsowicz, W.; Gromadzińska, J.; Rydzyński, K.; Tomczak, J. Selenium status of low-selenium area residents: Polish experience. Toxicol. Lett. 2003, 137, 95–101. [Google Scholar] [CrossRef]
- Nogueira, C.W.; Meotti, F.C.; Curte, E.; Pilissão, C.; Zeni, G.; Rocha, J.B. Investigations into the potential neurotoxicity induced by diselenides in mice and rats. Toxicology 2003, 183, 29–37. [Google Scholar] [CrossRef]
- Cabrera, Á.J. Zinc, aging, and immunosenescence: An overview. Pathobiol. Aging Age Relat. Dis. 2015, 5, 25592. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S.; Fitzgerald, J.T.; Hess, J.W.; Kaplan, J.; Pelen, F.; Dardenne, M. Zinc deficiency in elderly patients. Nutrition 1993, 9, 218–224. [Google Scholar]
- Haase, H.; Rink, L. The immune system and the impact of zinc during aging. Immun. Ageing 2009, 6, 9. [Google Scholar] [CrossRef]
- Hagmeyer, S.; Haderspeck, J.C.; Grabrucker, A.M. Behavioral impairments in animal models for zinc deficiency. Front. Behav. Neurosci. 2015, 8, 443. [Google Scholar] [CrossRef]
- Maret, W. The redox biology of redox-inert zinc ions. Free Radic. Biol. Med. 2019, 134, 311–326. [Google Scholar] [CrossRef]
- Hu, J.Y.; Zhang, D.L.; Liu, X.L.; Li, X.S.; Cheng, X.Q.; Chen, J.; Du, H.N.; Liang, Y. Pathological concentration of zinc dramatically accelerates abnormal aggregation of full-length human Tau and thereby significantly increases Tau toxicity in neuronal cells. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 414–427. [Google Scholar] [CrossRef]
- Cristóvão, J.S.; Santos, R.; Gomes, C.M. Metals and neuronal metal binding proteins implicated in Alzheimer’s disease. Oxid. Med. Cell Longev. 2016, 2016, 9812178. [Google Scholar] [CrossRef]
- Zhu, X.; Lee, H.; Perry, G.; Smith, M.A. Alzheimer disease, the two-hit hypothesis: An update. Biochim. Biophys. Acta 2007, 1772, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, S.A.; Coleman, L.A.; Masters, C.L.; Camakaris, J.; Hill, A.F. Regulation of prion gene expression by transcription factors SP1 and metal transcription factor-1. J. Biol. Chem. 2009, 284, 1291–1301. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, E.M.; Chaney, M.O.; Norris, F.H.; Pascual, R.; Little, S.P. Conservation of the sequence of the Alzheimer’s disease amyloid peptide in dog, polar bear and five other mammals by cross-species polymerase chain reaction analysis. Brain Res. Mol. Brain Res. 1991, 10, 299–305. [Google Scholar] [CrossRef]
- Cherny, R.A.; Atwood, C.S.; Xilinas, M.E.; Gray, D.N.; Jones, W.D.; McLean, C.A.; Barnham, K.J.; Volitakis, I.; Fraser, F.W.; Kim, Y.; et al. Treatment with a copper-zinc chelator markedly and rapidly inhibits beta-amyloid accumulation in Alzheimer’s disease transgenic mice. Neuron 2001, 30, 665–676. [Google Scholar] [CrossRef]
- Bush, A.I. Metal complexing agents as therapies for Alzheimer’s disease. Neurobiol. Aging 2002, 23, 1031–1038. [Google Scholar] [CrossRef]
- Sayre, L.M.; Perry, G.; Harris, P.L.R.; Liu, Y.; Schubert, K.A.; Smith, M.A. In situ oxidative catalysis by neurofibrillary tangles and senile plaques in Alzheimer’s disease: A central role for bound transition metals. J. Neurochem. 2000, 74, 270–279. [Google Scholar] [CrossRef]
- Crouch, P.J.; Hung, L.W.; Adlard, P.A.; Cortes, M.; Lal, V.; Filiz, G.; Perez, K.A.; Nurjono, M.; Caragounis, A.; Du, T.; et al. Increasing Cu bioavailability inhibits a betaoligomers and tau phosphorylation. Proc. Natl. Acad. Sci. USA 2009, 106, 381–386. [Google Scholar] [CrossRef]
- Voss, K.; Harris, C.; Ralle, M.; Duffy, M.; Murchison, C.; Quinn, J.F. Modulation of tau phosphorylation by environmental copper. Transl. Neurodegener. 2014, 3, 24. [Google Scholar] [CrossRef]
- Malavolta, M.; Giacconi, R.; Piacenza, F.; Santarelli, L.; Cipriano, C.; Costarelli, L.; Tesei, S.; Pierpaoli, S.; Basso, A.; Galeazzi, R.; et al. Plasma copper/zinc ratio: An inflammatory/nutritional biomarker as predictor of all-cause mortality in elderly population. Biogerontology 2010, 11, 309–319. [Google Scholar] [CrossRef]
- Osredkar, J.; Sustar, N. Copper and zinc, biological role and significance of copper/zinc imbalance. J. Clinic. Toxicol. 2011, S3, 1. [Google Scholar] [CrossRef]
- Deigendesch, N.; Zychlinsky, A.; Meissner, F. Copper regulates the canonical NLRP3 inflammasome. J. Immunol. 2018, 200, 1607–1617. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, L.F.; Medeiros, M.H.G.; Augusto, O. Oxidative damage and neurodegeneration: What have we learned from transgenic and knockout animals? Quím. Nova 2006, 29, 1352–1360. [Google Scholar] [CrossRef]
- Rodrigues, G.P.; Cozzolino, S.M.F.; Marreiro, D.D.N.; Caldas, D.R.C.; da Silva, K.G.; de Sousa Almondes, K.G.; Neto, J.M.M.; Pimentel, J.A.C.; de Carvalho, C.M.R.G.; Nogueira, N.D.N. Mineral status and superoxide dismutase enzyme activity in Alzheimer’s disease. J. Trace Elem. Med. Biol. 2017, 44, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Summersgill, H.; England, H.; Lopez-Castejon, G.; Lawrence, C.B.; Luheshi, N.M.; Pahle, J.; Mendes, P.; Brough, D. Zinc depletion regulates the processing and secretion of IL-1β. Cell Death Dis. 2014, 5, e1040. [Google Scholar] [CrossRef]
- Platel, K.; Srinivasan, K. Bioavailability of micronutrients from plant foods: An update. Crit. Rev. Food Sci. Nutr. 2016, 56, 1608–1619. [Google Scholar] [CrossRef]
- Margier, M.; Georgé, S.; Hafnaoui, N.; Remond, D.; Nowicki, M.; Du Chaffaut, L.; Amiot, M.J.; Reboul, E. Nutritional composition and bioactive content of legumes: Characterization of pulses frequently consumed in France and effect of the cooking method. Nutrients 2018, 10, 1668. [Google Scholar] [CrossRef]
- Górska-Warsewicz, H.; Rejman, K.; Laskowski, W.; Czeczotko, M. Milk and dairy products and their nutritional contribution to the average Polish diet. Nutrients 2019, 11, 1771. [Google Scholar] [CrossRef]
- Jâkobsone, I.; Kantâne, I.; Zute, S.; Jansone, I.; Bartkeviès, V. Macro-elements and trace elements in cereal grains cultivated in Latvia. Proc. Latv. Acad. Sci. Sect. B 2015, 69, 152–157. [Google Scholar] [CrossRef]
- Yu, L.; Nanguet, A.L.; Beta, T. Comparison of antioxidant properties of refined and whole wheat flour and bread. Antioxidants Basel. 2013, 2, 370–383. [Google Scholar] [CrossRef]
- Sitarz-Palczak, E.; Kalembkiewicz, J.; Galas, D. Evaluation of the content of selected heavy metals in samples of polish honeys. J. Ecol. Eng. 2015, 16, 130–138. [Google Scholar] [CrossRef]
- Jablonska, E.; Gromadzinska, J.; Klos, A.; Bertrandt, J.; Skibniewska, K.; Darago, A.; Wasowicz, W. Selenium, zinc and copper in the Polish diet. J. Food Compos. Anal. 2013, 31, 259–265. [Google Scholar] [CrossRef]
- Cantwell, M.; Elliott, C. Nitrates, nitrites and nitrosamines from processed meat intake and colorectal cancer risk. J. Clin. Nutr. Diet. 2017, 3, 27. [Google Scholar] [CrossRef]
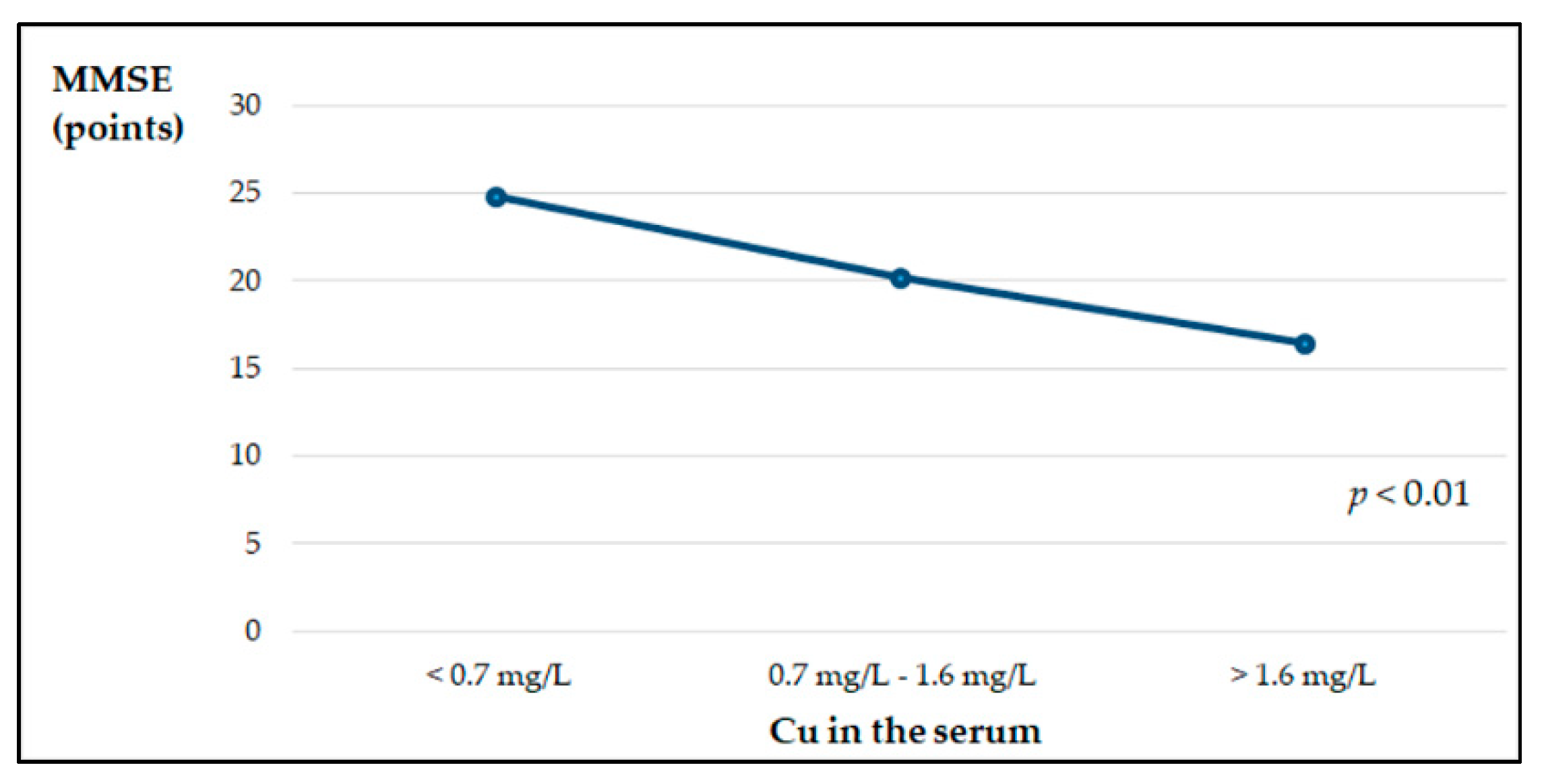

| Variable | Control Group | AD Patients |
|---|---|---|
| Total (n) | 60 | 110 |
| Gender (M/F) | (14/46) | (30/80) |
| Age (years): average ± SD | 67.0 ± 7.9 | 78.0 ± 8.1 |
| Age (years): range | 52–83 | 54–93 |
| BMI (kg/m2): average ± SD | nd | 26.5 ± 4.2 |
| BMI (kg/m2): range | nd | 17.8–40.2 |
| MMSE (points): average ± SD | nd | 20.4 ± 4.3 |
| MMSE (points): range | nd | 11–26 |
| Smoking cigarettes * (n)/no-smoking (n) | nd | 13/97 |
| Alcohol drinking # (n)/no-drinking (n) | nd | 5/105 |
| Variable | Control Group | AD Patients | p-Value | ||
|---|---|---|---|---|---|
| (n = 60) | (n = 110) | ||||
| Average ± SD | |||||
| (min–max) | |||||
| Median (Q1; Q3) | |||||
| Total | Total | ||||
| M (a) | F (b) | M (c) | F (d) | ||
| Se (μg/L) | 79.8 ± 22.0 | 69.1 ± 19.3 | <0.05 | ||
| (44.4–138.4) | (18.27–117.62) | ||||
| 73.3 (65.6; 91.6) | 71.4 (58.1; 79.7) | ||||
| 71.0 ± 13.9 | 81.7 ± 23.1 | 69.5 ± 17.9 | 68.9 ± 19.8 | c vs. d * | |
| (52.4–92.9) | (44.4–138.4) | (41.7–107.5) | (18.3–117.6) | ||
| 72.6 (58.5; 80.2) | 73.3 (66.6; 100.2) | 70.6 (52.1; 81.2) | 72.3 (58.4; 79.4) | ||
| Cu (mg/L) | 1.07 ± 0.3 | 1.03 ± 0.23 | ns | ||
| (0.68–1.94) | (0.64–2.17) | ||||
| 1.02 (0.86; 1.24) | 1.01 (0.87; 1.13) | ||||
| 0.96 ± 0.15 | 1.10 ± 0.30 | 0.90 ± 0.18 | 1.07 ± 0.23 | c vs. d * | |
| (0.70–1.12) | (0.68–1.94) | (0.65–1.27) | (0.64–2.17) | ||
| 0.97 (0.86; 1.09) | 1.02 (0.85; 1.28) | 0.87 (0.76; 1.05) | 1.04 (0.96; 1.16) | ||
| Zn (mg/L) | 0.92 ± 0.23 | 0.75 ± 0.33 | <00000.1 | ||
| (0.56–1.53) | (0.38–3.77) | ||||
| 0.86 (0.75; 1.02) | 0.71 (0.62; 0.83) | ||||
| 0.88 ± 0.27 | 0.92 ± 0.21 | 0.84 ± 0.61 | 0.71 ± 0.14 | c vs. d * | |
| (0.56–1.47) | (0.62–1.53) | (0.38–3.77) | (0.48–1.12) | ||
| 0.84 (0.73; 0.93) | 0.88 (0.75; 1.03) | 0.75 (0.60; 0.90) | 0.70 (0.63; 0.81) | ||
| Cu:Zn molar ratio | 1.24 ± 0.34 | 1.52 ± 0.47 | <0.001 | ||
| (0.60–2.07) | (0.23–3.52) | ||||
| 1.20 (1.01; 1.48) | 1.44 (1.26; 1.82) | ||||
| 1.21 ± 0.39 | 1.25 ± 0.34 | 1.31 ± 0.48 | 1.59 ± 0.45 | b vs. d * c vs. d * | |
| (0.60–1.93) | (0.70–2.07) | (0.23–2.86) | (0.81–3.52) | ||
| 1.23 (1.00; 1.34) | 1.16 (1.01; 1.53) | 1.33 (0.99; 1.50) | 1.58 (1.33; 1.84) | ||
| TAS (mmol/L) | 1.43 ± 0.67 | 1.11 ± 0.42 | <0.01 | ||
| (0.63–3.50) | (0.27–2.96) | ||||
| 1.31 (0.99; 1.70) | 1.10 (0.83; 1.33) | ||||
| 1.88 ± 0.84 | 1.32 ± 0.59 | 1.17 ± 0.36 | 1.09 ± 0.44 | a vs. b * a vs. c * b vs. d * | |
| (1.06–3.47) | (0.63–3.50) | (0.45–1.84) | (0.27–2.96) | ||
| 1.69 (1.16; 2.41) | 1.26 (0.94; 1.46) | 1.23 (0.93; 1.43) | 1.09 (0.79; 1.27) | ||
| Independent Variables | β Coefficient (SE) | Significance Level | Adjusted R2 |
|---|---|---|---|
| Selenium | |||
| White bread | 0.371 (0.136) | 0.0088 | 0.22 |
| Wholegrain bread | 0.292 (0.141) | 0.0434 | |
| Butter | 0.295 (0.144) | 0.0454 | |
| Coffee | 0.198 (0.125) | 0.1210 | |
| Cheese | 0.148 (0.122) | 0.2306 | |
| Tinned fish | 0.137 (0.117) | 0.2485 | |
| Legumes | −0.308 (0.125) | 0.0169 | |
| Tea | −0.226 (0.127) | 0.0811 | |
| Sausages | −0.175 (0.124) | 0.1635 | |
| Jam | −0.142 (0.124) | 0.2581 | |
| Copper | |||
| Cottage cheese | 0.360 (0.112) | 0.0025 | 0.38 |
| Legumes | 0.347 (0.133) | 0.0124 | |
| Sausages | 0.228 (0.113) | 0.0501 | |
| Wholegrain bread | 0.259 (0.141) | 0.0729 | |
| Eggs | 0.165 (0.116) | 0.1624 | |
| Butter | 0.173 (0.131) | 0.1925 | |
| Fish | 0.156 (0.119) | 0.1961 | |
| White bread | 0.136 (0.125) | 0.2811 | |
| Meat | −0.490 (0.119) | 0.0002 | |
| Honey | −0.543 (0.152) | 0.0009 | |
| Grits, rice | −0.378 (0.125) | 0.0040 | |
| Margarine | −0.335 (0.133) | 0.0152 | |
| Coffee | −0.261 (0.113) | 0.0254 | |
| Vegetable oil | −0.205 (0.110) | 0.0681 | |
| Tinned fish | −0.143 (0.108) | 0.1920 | |
| Zinc | |||
| Flour products | 0.278 (0.129) | 0.0358 | 0.26 |
| Honey | 0.266 (0.129) | 0.0434 | |
| Poultry | 0.229 (0.123) | 0.0692 | |
| Meat | 0.220 (0.123) | 0.0791 | |
| Jam | 0.191 (0.129) | 0.1433 | |
| Tea | 0.148 (0.119) | 0.2170 | |
| Cakes | −0.376 (0.117) | 0.0023 | |
| Sausages | −0.298 (0.130) | 0.0264 | |
| Raw vegetables | −0.183 (0.122) | 0.1390 | |
| Cottage cheese | −0.146 (0.120) | 0.2297 | |
| Total antioxidant status | |||
| Flour products | 0.378 (0.121) | 0.0028 | 0.15 |
| Ham | 0.189 (0.128) | 0.1453 | |
| Poultry | 0.119 (0.126) | 0.3492 | |
| Offal | −0.241 (0.118) | 0.0457 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socha, K.; Klimiuk, K.; Naliwajko, S.K.; Soroczyńska, J.; Puścion-Jakubik, A.; Markiewicz-Żukowska, R.; Kochanowicz, J. Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease. Nutrients 2021, 13, 287. https://doi.org/10.3390/nu13020287
Socha K, Klimiuk K, Naliwajko SK, Soroczyńska J, Puścion-Jakubik A, Markiewicz-Żukowska R, Kochanowicz J. Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease. Nutrients. 2021; 13(2):287. https://doi.org/10.3390/nu13020287
Chicago/Turabian StyleSocha, Katarzyna, Katarzyna Klimiuk, Sylwia K. Naliwajko, Jolanta Soroczyńska, Anna Puścion-Jakubik, Renata Markiewicz-Żukowska, and Jan Kochanowicz. 2021. "Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease" Nutrients 13, no. 2: 287. https://doi.org/10.3390/nu13020287
APA StyleSocha, K., Klimiuk, K., Naliwajko, S. K., Soroczyńska, J., Puścion-Jakubik, A., Markiewicz-Żukowska, R., & Kochanowicz, J. (2021). Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease. Nutrients, 13(2), 287. https://doi.org/10.3390/nu13020287











