Unveiling Metabolic Phenotype Alterations in Anorexia Nervosa through Metabolomics
Abstract
1. Introduction
1.1. Metabolomics: Basic Concepts and Methodological Aspects
1.2. Anorexia Nervosa
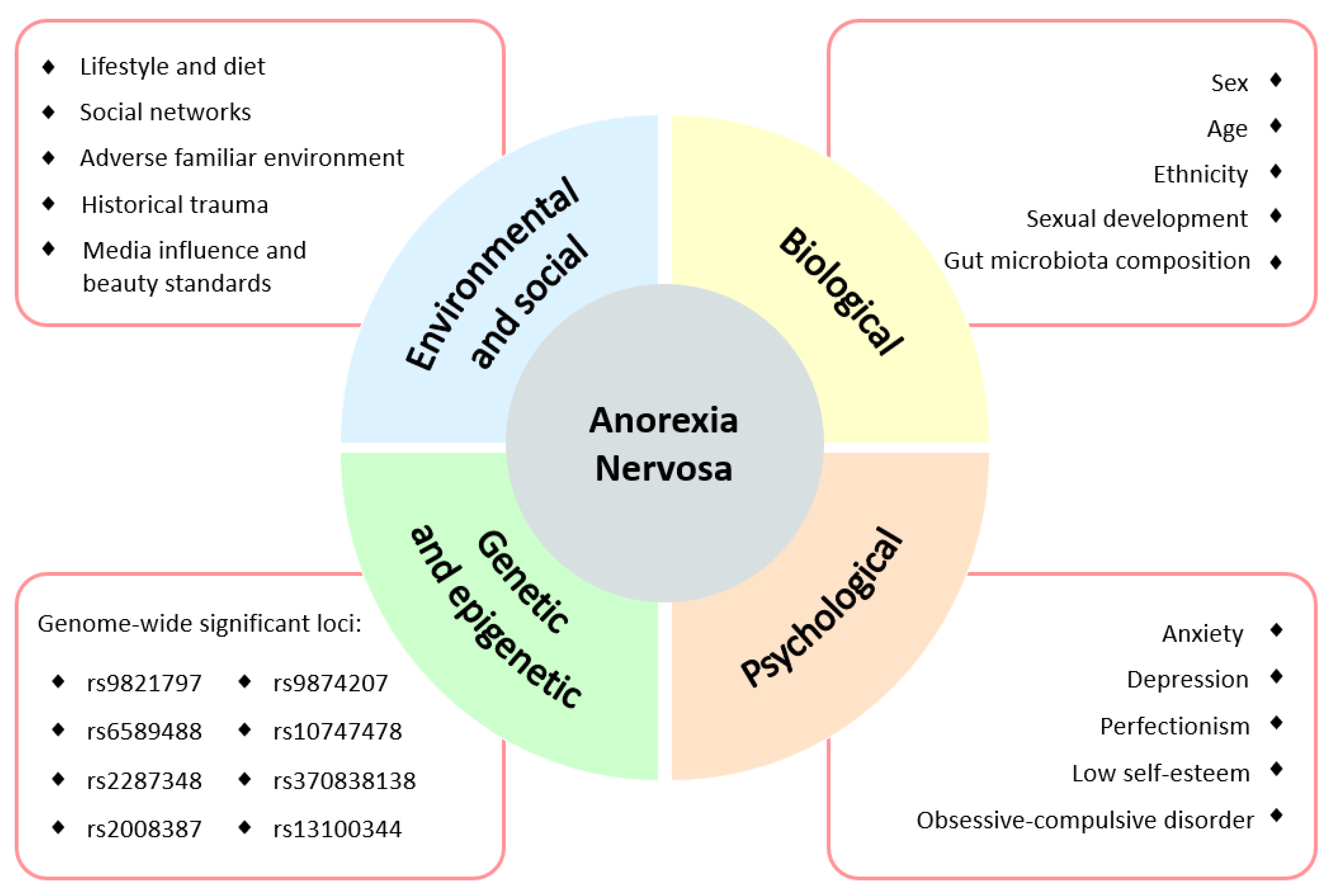
2. Background
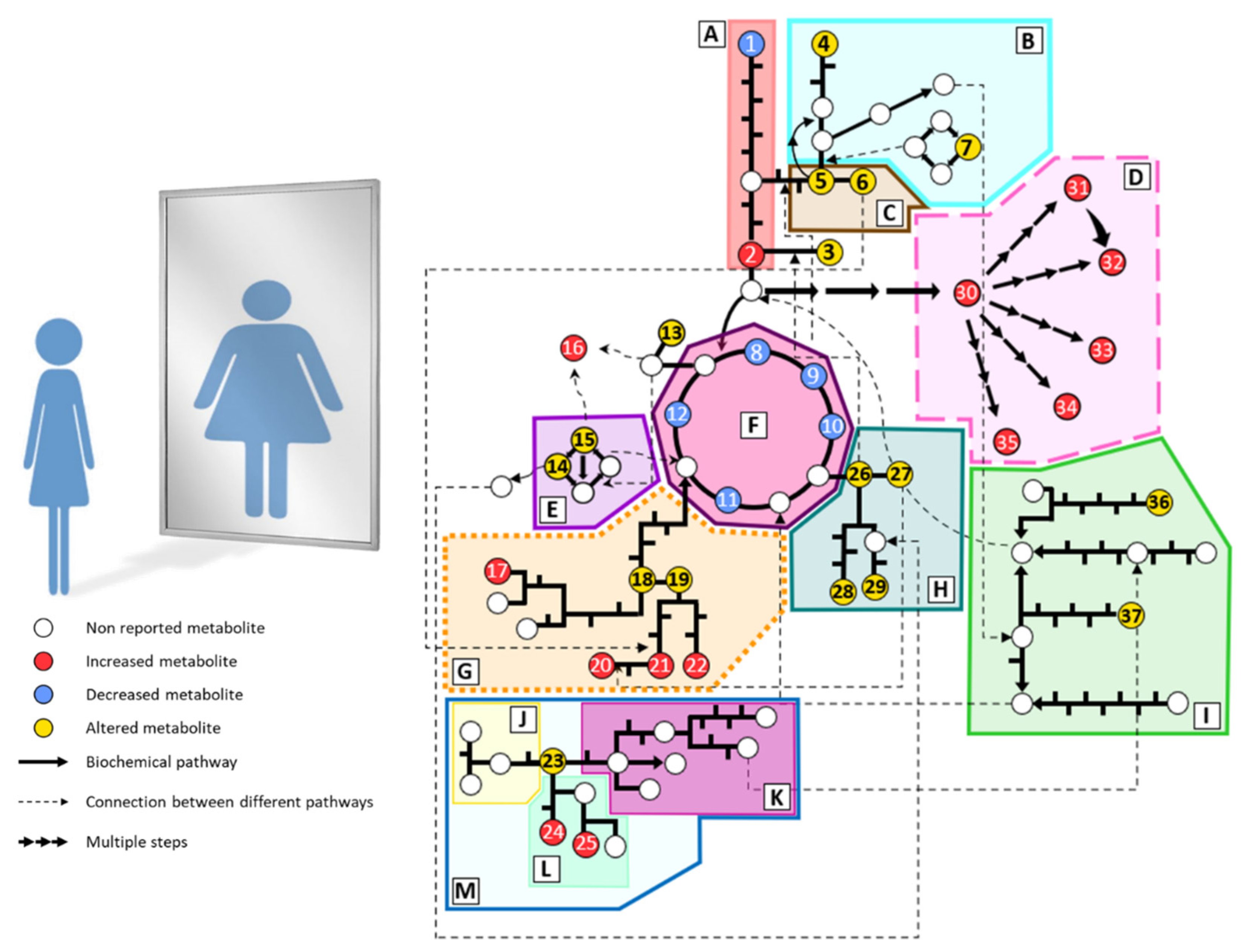
3. Metabolic Alterations in Anorexia Nervosa
3.1. Amino Acids
3.1.1. Plasma and Serum
3.1.2. Feces
3.2. Lipids
3.2.1. Plasma and Serum
3.2.2. Feces
3.3. Sugars
3.3.1. Plasma and Serum
3.3.2. Feces
3.4. Tricarboxylate Cycle
3.4.1. Plasma and Serum
3.4.2. Feces
3.5. Uremic Toxins
3.6. Microbial Metabolites
3.7. Covariates
4. Conclusions and Further Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 1H NMR | proton nuclear magnetic resonance |
| 7β-OH-DHEA | 7β-hydroxy dehydroepiandrosterone |
| AC | acylcarnitines |
| Ala | alanine |
| ALA | alpha-linolenic acid |
| AN | anorexia nervosa |
| AN-BP | anorexia nervosa-binge/purging |
| AN-R | anorexia nervosa-restricting |
| Arg | arginine |
| Asp | aspartate |
| BMI | body-mass index |
| CE-MS | capillary electrophoresis-mass spectrometry |
| DPA | docosapentaenoic acid |
| EPHX2 | epoxide hydrolase 2 |
| EPA | eicosapentaenoic acid |
| FDR | false discovery rate |
| FIA-MS/MS | flow injection analysis-tandem mass spectrometry |
| GABA | γ-amino butyrate |
| GC-MS | gas chromatography-mass spectrometry |
| Gln | glutamine |
| Glu | glutamate |
| Gly | glycine |
| HPLC-MS/MS | high performance liquid chromatography-tandem mass spectrometry |
| Ile | isoleucine |
| LC-MS | liquid chromatography-mass spectrometry |
| LC-MS/MS | liquid chromatography-tandem mass spectrometry |
| Leu | leucine |
| LPC | lysophosphatidylcholines |
| Met | methionine |
| MS | mass spectrometry |
| NMR | nuclear magnetic resonance |
| Orn | ornithine |
| PC | phosphatidylcholines |
| Phe | phenylalanine |
| Pro | proline |
| PUFAs | polyunsaturated fatty acids |
| sEH | soluble epoxide hydrolase |
| Ser | serine |
| SIM | selected ion monitoring |
| SM | sphingomyelins |
| Thr | threonine |
| Trp | tryptophan |
| Tyr | tyrosine |
| UPLC-MS | ultra-performance liquid chromatography-mass spectrometry |
| Val | valine |
References
- González-Riaño, C.; Dudzik, D.; García, A.; Gil-De-La-Fuente, A.; Gradillas, A.; Godzien, J.; López-Gonzálvez, Á.; Rey-Stolle, F.; Rojo, D.; Ruperez, F.J.; et al. Recent Developments along the Analytical Process for Metabolomics Workflows. Anal. Chem. 2020, 92, 203–226. [Google Scholar] [CrossRef]
- Dunn, W.B.; Broadhurst, D.I.; Atherton, H.J.; Goodacre, R.; Griffin, J.L. Systems Level Studies of Mammalian Metabolomes: The Roles of Mass Spectrometry and Nuclear Magnetic Resonance Spectroscopy. Chem. Soc. Rev. 2011, 40, 387–426. [Google Scholar] [CrossRef]
- Dudley, E.; Yousef, M.; Wang, Y.; Griffiths, W.J. 2-Targeted Metabolomics and Mass Spectrometry. Adv. Protein Chem. Struct. Biol. 2010, 80, 45–83. [Google Scholar] [CrossRef]
- Roberts, L.D.; Souza, A.L.; Gerszten, R.E.; Clish, C.B. Targeted Metabolomics. Curr. Protoc. Mol. Biol. 2012, 98, 30.2.1–30.2.24. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Roy, R. Quantitative 1H NMR Spectroscopy. TrAC-Trends Anal. Chem. 2012, 35, 5–26. [Google Scholar] [CrossRef]
- Barhoum, A.; García-Betancourt, M.L.; Rahier, H.; van Assche, G. Physicochemical characterization of nanomaterials: Polymorph, composition, wettability, and thermal stability. In Emerging Applications of Nanoparticles and Architectural Nanostructures: Current Prospects and Future Trends; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 255–278. ISBN 9780128135167. [Google Scholar]
- Goodacre, R.; Vaidyanathan, S.; Dunn, W.B.; Harrigan, G.G.; Kell, D.B. Metabolomics by Numbers: Acquiring and Understanding Global Metabolite Data. Trends Biotechnol. 2004, 22, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Emwas, A.H.M. The Strengths and Weaknesses of NMR Spectroscopy and Mass Spectrometry with Particular Focus on Metabolomics Research. Methods Mol. Biol. 2015, 1277, 161–193. [Google Scholar] [CrossRef]
- Dunn, W.B.; Hankemeier, T. Mass Spectrometry and Metabolomics: Past, Present and Future. Metabolomics 2013, 9, 1–3. [Google Scholar] [CrossRef]
- Fiehn, O. Combining Genomics, Metabolome Analysis, and Biochemical Modelling to Understand Metabolic Networks. Comp. Funct. Genom. 2001, 2, 155–168. [Google Scholar] [CrossRef]
- Hata, K.; Soma, Y.; Yamashita, T.; Takahashi, M.; Sugitate, K.; Serino, T.; Miyagawa, H.; Suzuki, K.; Yamada, K.; Kawamukai, T.; et al. Calibration-Curve-Locking Database for Semi-Quantitative Metabolomics by Gas Chromatography/Mass Spectrometry. Metabolites 2021, 11, 207. [Google Scholar] [CrossRef]
- Liu, X.; Locasale, J.W. Metabolomics—A Primer. Trends Biochem. Sci. 2017, 42, 274. [Google Scholar] [CrossRef]
- Kapoore, R.V.; Vaidyanathan, S. Towards Quantitative Mass Spectrometry-Based Metabolomics in Microbial and Mammalian Systems. Philos. Trans. Ser. A Math. Phys. Eng. Sci. 2016, 374, 1–14. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Peterson, C.B.; Pisetsky, E.M.; Swanson, S.A.; Crosby, R.D.; Mitchell, J.E.; Wonderlich, S.A.; le Grange, D.; Hill, L.; Powers, P.; Crow, S.J. Examining the Utility of Narrowing Anorexia Nervosa Subtypes for Adults. Compr. Psychiatry 2016, 67, 54–58. [Google Scholar] [CrossRef]
- Smink, F.R.; van Hoeken, D.; Hoek, H.W. Epidemiology of Eating Disorders: Incidence, Prevalence and Mortality Rates. Curr. Psychiatry Rep. 2012, 14, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Bulik, C.M.; Flatt, R.; Abbaspour, A.; Carroll, I. Reconceptualizing Anorexia Nervosa. Psychiatry Clin. Neurosci. 2019, 73, 518–525. [Google Scholar] [CrossRef]
- Borgo, F.; Riva, A.; Benetti, A.; Casiraghi, M.C.; Bertelli, S.; Garbossa, S.; Anselmetti, S.; Scarone, S.; Pontiroli, A.E.; Morace, G.; et al. Microbiota in Anorexia Nervosa: The Triangle between Bacterial Species, Metabolites and Psychological Tests. PLoS ONE 2017, 12, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Arcelus, J.; Mitchell, A.J.; Wales, J.; Nielsen, S. Mortality Rates in Patients with Anorexia Nervosa and Other Eating Disorders a Meta-Analysis of 36 Studies. Arch. Gen. Psychiatry 2011, 68, 724–731. [Google Scholar] [CrossRef]
- Chesney, E.; Goodwin, G.M.; Fazel, S. Risks of All-Cause and Suicide Mortality in Mental Disorders: A Meta-Review. World Psychiatry 2014, 13, 153. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Maguire, S.; Palacios, T.; Caterson, I.D. Are the Gut Bacteria Telling Us to Eat or Not to Eat? Reviewing the Role of Gut Microbiota in the Etiology, Disease Progression and Treatment of Eating Disorders. Nutrients 2017, 9, 602. [Google Scholar] [CrossRef]
- Watson, H.J.; Yilmaz, Z.; Thornton, L.M.; Hübel, C.; Coleman, J.R.I.; Gaspar, H.A.; Bryois, J.; Hinney, A.; Leppä, V.M.; Mattheisen, M.; et al. Genome-Wide Association Study Identifies Eight Risk Loci and Implicates Metabo-Psychiatric Origins for Anorexia Nervosa. Nat. Genet. 2019, 51, 1207–1214. [Google Scholar] [CrossRef]
- Moyano, D.; Vilaseca, M.A.; Artuch, R.; Lambruschini, N. Plasma Amino Acids in Anorexia Nervosa. Eur. J. Clin. Nutr. 1998, 52, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, D.; Tallonneau, I.; Vergès, B. Hypercholesterolaemia in Anorexia Nervosa: Frequency and Changes during Refeeding. Diabetes Metab. 2009, 35, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Ohwada, R.; Hotta, M.; Oikawa, S.; Takano, K. Etiology of Hypercholesterolemia in Patients with Anorexia Nervosa. Int. J. Eat. Disord. 2006, 39, 598–601. [Google Scholar] [CrossRef] [PubMed]
- Winston, A.P. The Clinical Biochemistry of Anorexia Nervosa. Ann. Clin. Biochem. 2012, 49, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Calvo, M.T.; Argente, J. Nutritional and Pubertal Disorders. Endocr. Dev. 2016, 29, 153–173. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.T.; Argente, J. Anorexia Nervosa in Female Adolescents: Endocrine and Bone Mineral Density Disturbances. Eur. J. Endocrinol. 2002, 147, 275–286. [Google Scholar] [CrossRef]
- Argente, J.; Caballo, N.; Barrios, V.; Muñoz, M.T.; Pozo, J.; Chowen, J.A.; Morandé, G.; Hernández, M. Multiple Endocrine Abnormalities of the Growth Hormone and Insulin-like Growth Factor Axis in Patients with Anorexia Nervosa: Effect of Short-and Long Term-Weight Recuperation. J. Clin. Endocrinol. Metab. 1997, 82, 2084–2092. [Google Scholar] [CrossRef][Green Version]
- da Luz Neto, L.M.; de Vasconcelos, F.M.N.; da Silva, J.E.; Pinto, T.C.C.; Sougey, É.B.; Ximenes, R.C.C. Differences in Cortisol Concentrations in Adolescents with Eating Disorders: A Systematic Review. J. De Pediatr. 2019, 95, 18–26. [Google Scholar] [CrossRef]
- Schorr, M.; Miller, K.K. The Endocrine Manifestations of Anorexia Nervosa: Mechanisms and Management. Nat. Rev. Endocrinol. 2017, 13, 174–186. [Google Scholar] [CrossRef]
- Soriano-Guillén, L.; Barrios, V.; Campos-Barros, Á.; Argente, J. Ghrelin Levels in Obesity and Anorexia Nervosa: Effect of Weight Reduction or Recuperation. J. Pediatrics 2004, 144, 36–42. [Google Scholar] [CrossRef]
- Martos-Moreno, G.Á.; Chowen, J.A.; Argente, J. Metabolic Signals in Human Puberty: Effects of over and Undernutrition. Mol. Cell. Endocrinol. 2010, 324, 70–81. [Google Scholar] [CrossRef]
- Argente, J.; Barrios, V.; Chowen, J.; Sinha, M.; Considine, R. Leptin Plasma Levels in Healthy Spanish Children and Adolescents, Children with Obesity, and Adolescents with Anorexia Nervosa and Bulimia Nervosa. J. Pediatrics 1997, 131, 833–838. [Google Scholar] [CrossRef]
- Naureen, F.; Masroor, K.; Khatoon, F.; Ayub, S.; Ahmed, M.I.; Hasnat, A.; Samad, N. Zinc and Tryptophan Levels in Anorexia Nervosa; a Co-Relational Study. Int. J. Biomed. Eng. Sci. 2014, 1, 27–33. [Google Scholar]
- Iannone, L.F.; Preda, A.; Blottière, H.M.; Clarke, G.; Albani, D.; Belcastro, V.; Carotenuto, M.; Cattaneo, A.; Citraro, R.; Ferraris, C.; et al. Microbiota-Gut Brain Axis Involvement in Neuropsychiatric Disorders. Expert Rev. Neurother. 2019, 19, 1037–1050. [Google Scholar] [CrossRef]
- Monteleone, A.M.; Troisi, J.; Serena, G.; Fasano, A.; Grave, R.D.; Cascino, G.; Marciello, F.; Calugi, S.; Scala, G.; Corrivetti, G.; et al. The Gut Microbiome and Metabolomics Profiles of Restricting and Binge-Purging Type Anorexia Nervosa. Nutrients 2021, 13, 507. [Google Scholar] [CrossRef]
- Mörkl, S.; Butler, M.I.; Holl, A.; Cryan, J.F.; Dinan, T.G. Probiotics and the Microbiota-Gut-Brain Axis: Focus on Psychiatry. Curr. Nutr. Rep. 2020, 9, 171–182. [Google Scholar] [CrossRef]
- Seitz, J.; Dahmen, B.; Keller, L.; Herpertz-Dahlmann, B. Gut Feelings: How Microbiota Might Impact the Development and Course of Anorexia Nervosa. Nutrients 2020, 12, 3295. [Google Scholar] [CrossRef]
- MacKay, M.B.; Kravtsenyuk, M.; Thomas, R.; Mitchell, N.D.; Dursun, S.M.; Baker, G.B. D-Serine: Potential Therapeutic Agent and/or Biomarker in Schizophrenia and Depression? Front. Psychiatry 2019, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Billard, J.M. D-Amino Acids in Brain Neurotransmission and Synaptic Plasticity. Amino Acids 2012, 43, 1851–1860. [Google Scholar] [CrossRef] [PubMed]
- Otte, D.M.; Barcena de Arellano, M.L.; Bilkei-Gorzo, A.; Albayram, Ö.; Imbeault, S.; Jeung, H.; Alferink, J.; Zimmer, A. Effects of Chronic D-Serine Elevation on Animal Models of Depression and Anxiety-Related Behavior. PLoS ONE 2013, 8, e67131. [Google Scholar] [CrossRef]
- Fuchs, S.A.; Berger, R.; Klomp, L.W.J.; de Koning, T.J. D-Amino Acids in the Central Nervous System in Health and Disease. Mol. Genet. Metab. 2005, 85, 168–180. [Google Scholar] [CrossRef]
- Zhang, K.; Fujita, Y.; Chang, L.; Qu, Y.; Pu, Y.; Wang, S.; Shirayama, Y.; Hashimoto, K. Abnormal Composition of Gut Microbiota Is Associated with Resilience versus Susceptibility to Inescapable Electric Stress. Transl. Psychiatry 2019, 9, 231. [Google Scholar] [CrossRef]
- Dogan, A.E.; Yuksel, C.; Du, F.; Chouinard, V.-A.; Öngür, D. Brain Lactate and PH in Schizophrenia and Bipolar Disorder: A Systematic Review of Findings from Magnetic Resonance Studies. Neuropsychopharmacology 2018, 43, 1681. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, S.; Löwe, B.; Reas, D.L.; Deter, H.C.; Herzog, W. Long-Term Prognosis in Anorexia Nervosa: Lessons from a 21-Year Follow-up Study. Lancet 2000, 355, 721–722. [Google Scholar] [CrossRef]
- Föcker, M.; Cecil, A.; Prehn, C.; Adamski, J.; Albrecht, M.; Adams, F.; Hinney, A.; Libuda, L.; Bühlmeier, J.; Hebebrand, J.; et al. Evaluation of Metabolic Profiles of Patients with Anorexia Nervosa at Inpatient Admission, Short-and Long-Term Weight Regain—Descriptive and Pattern Analysis. Metabolites 2021, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Föcker, M.; Timmesfeld, N.; Scherag, S.; Knoll, N.; Singmann, P.; Wang-Sattler, R.; Bühren, K.; Schwarte, R.; Egberts, K.; Fleischhaker, C.; et al. Comparison of Metabolic Profiles of Acutely Ill and Short-Term Weight Recovered Patients with Anorexia Nervosa Reveals Alterations of 33 out of 163 Metabolites. J. Psychiatr. Res. 2012, 46, 1600–1609. [Google Scholar] [CrossRef] [PubMed]
- Burdo, J.; Booij, L.; Kahan, E.; Thaler, L.; Israël, M.; Agellon, L.B.; Nitschmann, E.; Wykes, L.; Steiger, H. Plasma Levels of One-Carbon Metabolism Nutrients in Women with Anorexia Nervosa. Int. J. Eat. Disord. 2020, 53, 1534–1538. [Google Scholar] [CrossRef]
- Bulant, J.; Hill, M.; Velíková, M.; Yamamotová, A.; Martásek, P.; Papežová, H. Changes of BMI, Steroid Metabolome and Psychopathology in Patients with Anorexia Nervosa during Hospitalization. Steroids 2020, 153, 108523. [Google Scholar] [CrossRef]
- Miyata, N.; Hata, T.; Takakura, S.; Yoshihara, K.; Morita, C.; Mikami, K.; Nomoto, K.; Miyazaki, K.; Tsuji, H.; Sudo, N. Metabolomics Profile of Japanese Female Patients with Restricting-Type Anorexia Nervosa. Physiol. Behav. 2021, 228. [Google Scholar] [CrossRef]
- Shih, P.-A.B. Integrating Multi-Omics Biomarkers and Postprandial Metabolism to Develop Personalized Treatment for Anorexia Nervosa. Prostaglandins Other Lipid Mediat. 2017, 132, 69–76. [Google Scholar] [CrossRef]
- Shih, P.; Yang, J.; Morisseau, C.; German, J.; Scott-Van Zeeland, A.; Armando, A.; Quehenberger, O.; Bergen, A.; Magistretti, P.; Berrettini, W.; et al. Dysregulation of Soluble Epoxide Hydrolase and Lipidomic Profiles in Anorexia Nervosa. Mol. Psychiatry 2016, 21, 537–546. [Google Scholar] [CrossRef]
- Prochazkova, P.; Roubalova, R.; Dvorak, J.; Kreisinger, J.; Hill, M.; Tlaskalova-Hogenova, H.; Tomasova, P.; Pelantova, H.; Cermakova, M.; Kuzma, M.; et al. The Intestinal Microbiota and Metabolites in Patients with Anorexia Nervosa. Gut Microbes 2021, 13, 1–25. [Google Scholar] [CrossRef]
- Salehi, A.M.; Nilsson, I.A.; Figueira, J.; Thornton, L.M.; Abdulkarim, I.; Palsson, E.; Bulik, C.M.; Landen, M. Serum Profiling of Anorexia Nervosa. A 1 H NMR-Based Metabolomics Study. Eur. Neropsychopharmacol. 2021, 49, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Dow, M.; Woodside, B.; German, J.B.; Quehenberger, O.; Shih, P.B. Food-Intake Normalization of Dysregulated Fatty Acids in Women with Anorexia Nervosa. Nutrients 2019, 11, 2208. [Google Scholar] [CrossRef]
- Monteleone, P.; Monteleone, A.M.; Troisi, J.; Dalle Grave, R.; Corrivetti, G.; Calugi, S.; Scala, G.; Patriciello, G.; Zanetti, A.; Maj, M. Metabolomics Signatures of Acutely Ill and Short-Term Weight Recovered Women with Anorexia Nervosa. Mol. Psychiatry 2021, 26, 3980–3991. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, A.M.; Troisi, J.; Fasano, A.; Dalle Grave, R.; Marciello, F.; Serena, G.; Calugi, S.; Scala, G.; Corrivetti, G.; Cascino, G.; et al. Multi-Omics Data Integration in Anorexia Nervosa Patients before and after Weight Regain: A Microbiome-Metabolomics Investigation. Clin. Nutr. 2020, 40, 1137–1146. [Google Scholar] [CrossRef]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Food Intake and Starvation Induce Metabolic Changes. In Biochemistry; W H Freeman: New York, NY, USA, 2002. [Google Scholar]
- Žák, A.; Vecka, M.; Tvrzická, E.; Hrubý, M.; Novák, F.; Papežová, H.; Lubanda, H.; Veselá, L.; Staňková, B. Composition of Plasma Fatty Acids and Non-Cholesterol Sterols in Anorexia Nervosa. Physiol. Res. 2005, 54, 443–451. [Google Scholar]
- Ren, Q. Soluble Epoxide Hydrolase Inhibitor: A Novel Potential Therapeutic or Prophylactic Drug for Psychiatric Disorders. Front. Pharmacol. 2019, 10, 420. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.A.; Hübel, C.; Hindborg, M.; Lindkvist, E.; Kastrup, A.M.; Yilmaz, Z.; Støving, R.K.; Bulik, C.M.; Sjögren, J.M. Increased Lipid and Lipoprotein Concentrations in Anorexia Nervosa: A Systematic Review and Meta-Analysis. Int. J. Eat. Disord. 2019, 52, 611–629. [Google Scholar] [CrossRef]
- Weinbrenner, T.; Züger, M.; Jacoby, G.E.; Herpertz, S.; Liedtke, R.; Sudhop, T.; Gouni-Berthold, I.; Axelson, M.; Berthold, H.K. Lipoprotein Metabolism in Patients with Anorexia Nervosa: A Case–Control Study Investigating the Mechanisms Leading to Hypercholesterolaemia. Br. J. Nutr. 2004, 91, 959–969. [Google Scholar] [CrossRef]
- Breton, J.; Déchelotte, P.; Ribet, D. Intestinal Microbiota and Anorexia Nervosa. Clin. Nutr. Exp. 2019, 28, 11–21. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, Stability and Resilience of the Human Gut Microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef] [PubMed]
- Mörkl, S.; Lackner, S.; Müller, W.; Gorkiewicz, G.; Kashofer, K.; Oberascher, A.; Painold, A.; Holl, A.; Holzer, P.; Meinitzer, A.; et al. Gut Microbiota and Body Composition in Anorexia Nervosa Inpatients in Comparison to Athletes, Overweight, Obese, and Normal Weight Controls. Int. J. Eat. Disord. 2017, 50, 1421–1431. [Google Scholar] [CrossRef] [PubMed]
- Morita, C.; Tsuji, H.; Hata, T.; Gondo, M.; Takakura, S.; Kawai, K.; Yoshihara, K.; Ogata, K.; Nomoto, K.; Miyazaki, K.; et al. Gut Dysbiosis in Patients with Anorexia Nervosa. PLoS ONE 2015, 10, e0145274. [Google Scholar] [CrossRef]
- Kleiman, S.C.; Watson, H.J.; Bulik-Sullivan, E.C.; Huh, E.Y.; Tarantino, L.M.; Bulik, C.M.; Carroll, I.M. The Intestinal Microbiota in Acute Anorexia Nervosa and during Renourishment: Relationship to Depression, Anxiety, and Eating Disorder Psychopathology. Psychosom. Med. 2015, 77, 969–981. [Google Scholar] [CrossRef]
- Mack, I.; Cuntz, U.; Grmer, C.; Niedermaier, S.; Pohl, C.; Schwiertz, A.; Zimmermann, K.; Zipfel, S.; Enck, P.; Penders, J. Weight Gain in Anorexia Nervosa Does Not Ameliorate the Faecal Microbiota, Branched Chain Fatty Acid Profiles, and Gastrointestinal Complaints. Sci. Rep. 2016, 6, 26752. [Google Scholar] [CrossRef]
- Pfleiderer, A.; Lagier, J.C.; Armougom, F.; Robert, C.; Vialettes, B.; Raoult, D. Culturomics Identified 11 New Bacterial Species from a Single Anorexia Nervosa Stool Sample. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1471–1481. [Google Scholar] [CrossRef]
- Armougom, F.; Henry, M.; Vialettes, B.; Raccah, D.; Raoult, D. Monitoring Bacterial Community of Human Gut Microbiota Reveals an Increase in Lactobacillus in Obese Patients and Methanogens in Anorexic Patients. PLoS ONE 2009, 4, e7125. [Google Scholar] [CrossRef]
- Holzer, P.; Farzi, A. Neuropeptides and the Microbiota- Gut-Brain Axis. Adv. Exp. Med. Biol. 2014, 817, 196–219. [Google Scholar] [CrossRef]
- Gribble, F.M.; Reimann, F. Function and Mechanisms of Enteroendocrine Cells and Gut Hormones in Metabolism. Nat. Rev. Endocrinol. 2019, 15, 226–237. [Google Scholar] [CrossRef]
- Kimura, I.; Inoue, D.; Maeda, T.; Hara, T.; Ichimura, A.; Miyauchi, S.; Kobayashi, M.; Hirasawa, A.; Tsujimoto, G. Short-Chain Fatty Acids and Ketones Directly Regulate Sympathetic Nervous System via G Protein-Coupled Receptor 41 (GPR41). Proc. Natl. Acad. Sci. USA 2011, 108, 8030–8035. [Google Scholar] [CrossRef]
- Qiu, J.; Liu, R.; Ma, Y.; Li, Y.; Chen, Z.; He, H.; Chen, J.; Tong, L.; Huang, C.; You, Q. Lipopolysaccharide-Induced Depression-Like Behaviors Is Ameliorated by Sodium Butyrate via Inhibiting Neuroinflammation and Oxido-Nitrosative Stress. Pharmacology 2020, 105, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, F.A.; Lin, C.L.; Crusio, W.E.; Akbarian, S. Antidepressant-Like Effects of the Histone Deacetylase Inhibitor, Sodium Butyrate, in the Mouse. Biol. Psychiatry 2007, 62, 55–64. [Google Scholar] [CrossRef] [PubMed]
- van de Wouw, M.; Boehme, M.; Lyte, J.M.; Wiley, N.; Strain, C.; O’Sullivan, O.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Short-Chain Fatty Acids: Microbial Metabolites That Alleviate Stress-Induced Brain–Gut Axis Alterations. J. Physiol. 2018, 596, 4923–4944. [Google Scholar] [CrossRef]
- Han, A.; Sung, Y.; Chung, S.; Kwon, M. Possible Additional Antidepressant-like Mechanism of Sodium Butyrate: Targeting the Hippocampus. Neuropharmacology 2014, 81, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Hoyles, L.; Snelling, T.; Umlai, U.-K.; Nicholson, J.K.; Carding, S.R.; Glen, R.C.; McArthur, S. Microbiome–Host Systems Interactions: Protective Effects of Propionate upon the Blood–Brain Barrier. Microbiome 2018, 6, 55. [Google Scholar] [CrossRef]
- Ferreira, D.L.S.; Hübel, C.; Herle, M.; Abdulkadir, M.; Loos, R.J.F.; Bryant-Waugh, R.; Bulik, C.M.; de Stavola, B.L.; Lawlor, D.A.; Micali, N. Associations between Blood Metabolic Profile at 7 Years Old and Eating Disorders in Adolescence: Findings from the Avon Longitudinal Study of Parents and Children. Metabolites 2019, 9, 191. [Google Scholar] [CrossRef]
| Methodology | Instrumental Analysis | Sample | Study Design | Findings | Ref. |
|---|---|---|---|---|---|
| Targeted | FIA-MS/MS | Serum | Evaluation of the metabolic profile of patients during weight recovery. Female adolescents. Healthy controls (n= 25) AN patients at inpatient admission (n = 35) Short-term weight recovery (n = 26) Long-term weight recovery (n = 22) |
| [47] |
| Targeted | FIA-MS/MS | Serum | Comparison of the metabolic profile of acute patients and short-term weight recovered patients. Young females. Healthy controls (n = 16) Acute AN patients (n = 29) Short-term weight recovery (n = 29) |
| [48] |
| Targeted | LC-MS/MS | Plasma | Analysis of one-carbon metabolism in AN-R and AN-BP patients, in recovered AN patients, and healthy controls. Young women. No eating-disorder history (controls) (n = 36) AN-R patients (n = 30) AN-BP patients (n = 23) AN remitted patients (n = 40; 36 with AN-BP history, and 9 with AN-R history) |
| [49] |
| Targeted | GC-MS (SIM) | Serum | Changes in BMI, and psychopathology and steroid metabolome profiling in AN patients before and after hospitalization. Young women. AN patients (n = 33) |
| [50] |
| Untargeted Targeted | UPLC-MS CE-MS LC-MS/MS | Serum | Comparison of the metabolic profile of AN-R patients with age-matching healthy controls. Young females. Healthy controls (n = 10) AN-R patients (n = 10) |
| [51] |
| Untargeted Targeted | GC-MS HPLC-MS/MS | Plasma | Multiomics study of AN (genomics, proteomics, and metabolomics). Multiplatform metabolomics study of the lipidome and eicosanoid metabolome of acute AN patients, recovered patients, and healthy controls. Healthy controls (n= 36 for PUFAs analysis, n = 38 for eicosanoid analysis) Ill AN patients (n= 30 for PUFAs analysis, n = 10 for eicosanoid analysis) Recovered AN patients (n= 30 for PUFAs analysis, n = 10 for eicosanoid analysis) |
| [52] |
| Untargeted Targeted | GC-MS HPLC-MS/MS | Plasma | Evaluation of the lipidomic profile of AN patients compared to healthy controls and recovered patients. Young females. Healthy controls (n = 36) Ill AN patients (n = 30) Recovered AN patients (n = 30) |
| [53] |
| Untargeted Targeted | 1H NMR MS (SIM) | Feces | Multiomics approach for analyzing the intestinal microbiota and its metabolites in patients before and after treatment compared to healthy controls. Young females. Healthy controls (n = 67) AN-R patients (n = 59) |
| [54] |
| Untargeted | 1H NMR | Serum | Metabolome profiling of acute AN patients, recovered patients, and healthy controls. Young women. Healthy controls (n = 65) AN patients (n = 65) Recovered AN patients (n = 65) |
| [55] |
| Untargeted | GC-MS | Plasma | Evaluation of the fatty acid profile after renourishment therapy in AN patients. Young women. Healthy controls (n= 47) Ill AN patients (n = 30) Recovered AN patients (n = 20) |
| [56] |
| Untargeted | GC-MS | Feces | Comparison of metabolic profiles of patients in an acute state, after recovery, and healthy controls. Young females. Healthy controls (n = 20) AN patients (n = 24; 18 with AN-R, 6 with AN-BP) AN patients after short-term weight restoration (n = 16) |
| [57] |
| Untargeted | GC-MS | Feces | Analysis of the microbiome and the metabolome of AN patients before and after treatment compared to healthy controls. Young females. Healthy controls (n = 20) AN patients before treatment (n = 21; 16 with AN-R and 5 AN-BP) AN patients after short-term weight restoration (n = 16) |
| [58] |
| Untargeted | GC-MS | Feces | Evaluation of the microbiome and the metabolic profile of AN-R and AN-BP patients and healthy controls. Young females. Healthy controls (n = 20) AN-R patients (n = 17) AN-BP patients (n = 6) |
| [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayo-Martínez, L.; Rupérez, F.J.; Martos-Moreno, G.Á.; Graell, M.; Barbas, C.; Argente, J.; García, A. Unveiling Metabolic Phenotype Alterations in Anorexia Nervosa through Metabolomics. Nutrients 2021, 13, 4249. https://doi.org/10.3390/nu13124249
Mayo-Martínez L, Rupérez FJ, Martos-Moreno GÁ, Graell M, Barbas C, Argente J, García A. Unveiling Metabolic Phenotype Alterations in Anorexia Nervosa through Metabolomics. Nutrients. 2021; 13(12):4249. https://doi.org/10.3390/nu13124249
Chicago/Turabian StyleMayo-Martínez, Laura, Francisco J. Rupérez, Gabriel Á. Martos-Moreno, Montserrat Graell, Coral Barbas, Jesús Argente, and Antonia García. 2021. "Unveiling Metabolic Phenotype Alterations in Anorexia Nervosa through Metabolomics" Nutrients 13, no. 12: 4249. https://doi.org/10.3390/nu13124249
APA StyleMayo-Martínez, L., Rupérez, F. J., Martos-Moreno, G. Á., Graell, M., Barbas, C., Argente, J., & García, A. (2021). Unveiling Metabolic Phenotype Alterations in Anorexia Nervosa through Metabolomics. Nutrients, 13(12), 4249. https://doi.org/10.3390/nu13124249