The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement?
Abstract
1. Introduction
2. Physiological Role of Vitamins
3. Alcoholic Liver Disease: Malnutrition and Supplementation
3.1. Vitamin B Group
3.2. Vitamin D Deficiency
3.3. Vitamin E Deficiency
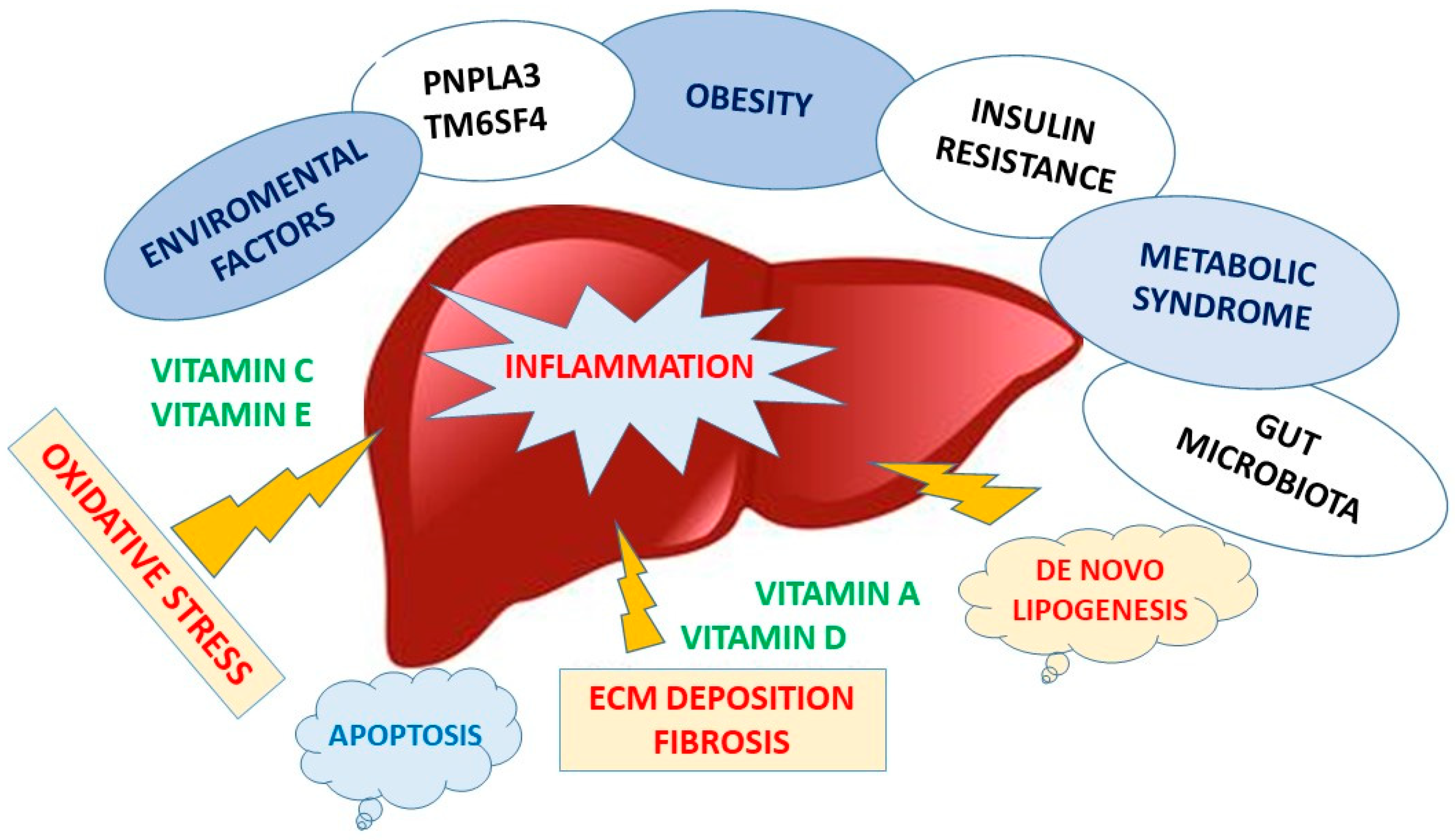
4. Non-Alcoholic Fatty Liver Disease
4.1. Role of Micronutrients in Nafld
4.2. Vitamin C Deficiency
4.3. Vitamin E Deficiency
4.4. Vitamin A Inadequacy
4.5. Vitamin D Deficiency
5. Advanced Liver Disease: Cirrhosis and HCC
5.1. Vitamin A Deficiency
5.2. Vitamin D Deficiency
5.3. Vitamin E Deficiency
5.4. Vitamin K Insufficiency
6. Liver Transplantation
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, J.; Meng, Q.H. Current understanding of the metabolism of micronutrients in chronic alcoholic liver disease. World J. Gastroenterol. 2020, 26, 4567–4578. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice guideline by the American Gastroenterological Association (AGA), American Association for the Study of Liver Diseases (AASLD), and American College of Gastroenterology (AGA). Gastroenterology 2012, 142, 1592–1609. [Google Scholar] [CrossRef]
- Bugianesi, E.; Leone, N.; Vanni, E.; Marchesini, G.; Brunello, F.; Carucci, P.; Musso, A.; De Paolis, P.; Capussotti, L.; Salizzoni, M.; et al. Expanding the natural history of nonalcoholic steatohepatitis: From cryptogenic cirrhosis to hepatocellular carcinoma. Gastroenterology 2002, 123, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Rinella, M.E. Nonalcoholic fatty liver disease: A systematic review. JAMA 2015, 313, 2263–2273. [Google Scholar] [CrossRef] [PubMed]
- Comar, K.M.; Sterling, R.K. Review article: Drug therapy for non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2005, 23, 207–215. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Nikolova, D.; Bjelakovic, M.; Gluud, C. Vitamin D supplementation for chronic liver diseases in adults. Cochrane Database Syst. Rev. 2017, 11, CD011564. [Google Scholar] [CrossRef]
- Saeed, A.; Bartuzi, P.; Heegsma, J.; Dekker, D. Kloosterhuis, N.; de Bruin, A.; Jonker, J.W.; van de Sluis, B.; Faber, K.N. Impaired Hepatic Vitamin A Metabolism in NAFLD Mice Leading to Vitamin A Accumulation in Hepatocytes. Cell. Mol. Gastroenterol. Hepatol. 2021, 11, 309–325. [Google Scholar] [CrossRef]
- Handbook of Vitamins, 4th ed.; Mock, D.M., Zempleni, J., Rucker, R.B., McCormick, D.B., Suttie, J.W., Eds.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Lindschinger, M.; Tatzber, F.; Schimetta, W.; Schmid, I.; Lindschinger, B.; Cvirn, G.; Stanger, O.; Lamont, E.; Wonisch, W. A Randomized Pilot Trial to Evaluate the Bioavailability of Natural versus Synthetic Vitamin B Complexes in Healthy Humans and Their Effects on Homocysteine, Oxidative Stress, and Antioxidant Levels. Oxid. Med. Cell. Longev. 2019, 2019, 6082613. [Google Scholar] [CrossRef]
- Kennedy, D.O. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Stanger, O.; Wonisch, W. Enzymatic and Non-Enzymatic Antioxidative Effects of Folic Acid and Its Reduced Derivates. In Water Soluble Vitamins; Stanger, O., Ed.; Springer: Dordrecht, The Netherlands, 2012; Volume 56. [Google Scholar]
- Frei, B.; Birlouez-Aragon, I.; Lykkesfeldt, J. Authors’ perspective: What is the optimum intake of vitamin C in humans? Crit. Rev. Food Sci. Nutr. 2012, 52, 815–829. [Google Scholar] [CrossRef]
- Kivirikko, K.I.; Myllyla, R. Post-translational processing of procollagens. Ann. N. Y. Acad. Sci. 1985, 460, 187–201. [Google Scholar] [CrossRef]
- Buettner, G.R. The pecking order of free radicals and antioxidants: Lipid peroxidation, α-tocopherol, and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Udomsinprasert, W.; Jittikoon, J. Vitamin D and liver fibrosis: Molecular mechanisms and clinical studies. Biomed. Pharmacother. 2019, 109, 1351–1360. [Google Scholar] [CrossRef]
- Bouillon, R.; In De Groot, L.; Jameson, J. Vitamin D: From Photosynthesis, Metabolism, and Action to Clinical Applications; Saunders: Philadelphia, PA, USA, 2001. [Google Scholar]
- Gascon-Barré, M.; Demers, C.; Mirshahi, A.; Néron, S.; Zalzal, S.; Nanci, A. The normal liver harbors the vitamin D nuclear receptor in nonparenchymal and biliary epithelial cells. Hepatology 2003, 37, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, G.P.; Muller, D.P.; Burroughs, A.K.; Matthews, S.; Kemp, C.; Epstein, O.; Metcalfe, T.A.; Southam, E.; Tazir-Melboucy, M.; Thomas, P.K.; et al. Vitamin E deficiency and its clinical significance in adults with primary biliary cirrhosis and other forms of chronic liver disease. J. Hepatol. 1987, 4, 307–317. [Google Scholar] [CrossRef]
- Mager, D.R.; McGee, P.L.; Furuya, K.N.; Roberts, E.A. Prevalence of vitamin K deficiency in children with mild to moderate chronic liver disease. J. Pediatr. Gastroenterol. Nutr. 2006, 42, 71–76. [Google Scholar] [CrossRef]
- Strople, J.; Lovell, G.; Heubi, J. Prevalence of subclinical vitamin K deficiency in cholestatic liver disease. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 78–84. [Google Scholar] [CrossRef]
- Young, I.S.; Woodside, J.V.J. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.K.; Bataller, R.; Ahn, J.; Kamath, P.S.; Shah, V.H. ACG Clinical Guideline: Alcoholic Liver Disease. Am. J. Gastroenterol. 2018, 113, 175–194. [Google Scholar] [CrossRef]
- Liangpunsakul, S.; Haber, P.; McCaughan, G.W. Alcoholic Liver Disease in Asia, Europe, and North America. Gastroenterology 2016, 150, 1786–1797. [Google Scholar] [CrossRef]
- Wong, M.C.S.; Huang, J.L.W.; George, J.; Huang, J.; Leung, C.; Eslam, M.; Chan, H.L.Y.; Ng, S.C. The changing epidemiology of liver diseases in the Asia-Pacific region. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 57–73. [Google Scholar] [CrossRef] [PubMed]
- Rehm, J.; Samokhvalov, A.V.; Shield, K.D. Global burden of alcoholic liver diseases. J. Hepatol. 2013, 59, 160–168. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of alcohol-related liver disease. J. Hepatol. 2018, 69, 154–181. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Mendenhall, C.L.; Anderson, S.; Weesner, R.E.; Goldberg, S.J.; Crolic, K.A. Protein-calorie malnutrition associated with alcoholic hepatitis. Veterans Administration Cooperative Study Group on Alcoholic Hepatitis. Am. J. Med. 1984, 76, 211–222. [Google Scholar] [CrossRef]
- Zhou, Z.; Wang, L.; Song, Z.; Saari, J.T.; McClain, C.J.; Kang, Y.J. Zinc supplementation prevents alcoholic liver injury in mice through attenuation of oxidative stress. Am. J. Pathol. 2005, 166, 1681–1690. [Google Scholar] [CrossRef]
- Zaman, S.N.; Tredger, J.M.; Johnson, P.J.; Williams, R. Vitamin B6 concentrations in patients with chronic liver disease and hepatocellular carcinoma. Br. Med. J. 1986, 293, 175. [Google Scholar] [CrossRef]
- Henderson, J.M.; Scott, S.S.; Merrill, A.H.; Hollins, B.; Kutner, M.H. Vitamin B6 repletion in cirrhosis with oral pyridoxine: Failure to improve amino acid metabolism. Hepatology 1989, 9, 582–588. [Google Scholar] [CrossRef]
- Labadarios, D.; Rossouw, J.E.; McConnell, J.B.; Davis, M.; Williams, R. Vitamin B6 deficiency in chronic liver disease--evidence for increased degradation of pyridoxal-5′-phosphate. Gut 1977, 18, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Trépo, E.; Ouziel, R.; Pradat, P.; Momozawa, Y.; Quertinmont, E.; Gervy, C.; Gustot, T.; Degré, D.; Vercruysse, V.; Deltenre, P.; et al. Marked 25-hydroxyvitamin D deficiency is associated with poor prognosis in patients with alcoholic liver disease. J. Hepatol. 2013, 59, 344–350. [Google Scholar] [CrossRef]
- Choi, Y.; Lee, S.; Kim, S.; Lee, J.; Ha, J.; Oh, H.; Lee, Y.; Kim, Y.; Yoon, Y. Vitamin E (α-tocopherol) consumption influences gut microbiota composition. Int. J. Food Sci. Nutr. 2020, 71, 221–225. [Google Scholar] [CrossRef]
- Azzi, A.; Gysin, R.; Kempná, P.; Munteanu, A.; Negis, Y.; Villacorta, L.; Visarius, T.; Zingg, J.M. Vitamin E mediates cell signaling and regulation of gene expression. Ann. N. Y. Acad. Sci. 2004, 1031, 86–95. [Google Scholar] [CrossRef]
- Breen, K.J.; Buttigieg, R.; Iossifidis, S.; Lourensz, C.; Wood, B. Jejunal uptake of thiamin hydrochloride in man: Influence of alcoholism and alcohol. Am. J. Clin Nutr. 1985, 42, 121–126. [Google Scholar] [CrossRef]
- Stickel, F.; Hoehn, B.; Schuppan, D.; Seitz, H.K. Review article: Nutritional therapy in alcoholic liver disease. Aliment. Pharmacol. Ther. 2003, 18, 357–373. [Google Scholar] [CrossRef] [PubMed]
- Galicia-Moreno, M.; Rosique-Oramas, D.; Medina-Avila, Z.; Álvarez-Torres, T.; Falcón, D.; La Tijera, F.H.D.; Béjar, Y.L.; Cordero-Pérez, P.; Muñoz-Espinosa, L.; Pérez-Hernández, J.L.; et al. Behavior of Oxidative Stress Markers in Alcoholic Liver Cirrhosis Patients. Oxidative Med. Cell. Longev. 2016, 2016, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sangeetha, K.; Krishnasamy, N.; Padma, K.; Rajendran, K. Evaluation of Oxidative Stress in Liver Cirrhosis Patients to Early Diagnosis of Minimal Hepatic Encephalopathy. Int. Neuropsychiatr. Dis. J. 2016, 5, 1–9. [Google Scholar] [CrossRef]
- Lai, C.Y.; Cheng, S.B.; Lee, T.Y.; Hsiao, Y.F.; Liu, H.T.; Huang, Y.C. Impact of Glutathione and Vitamin B-6 in Cirrhosis Patients: A Randomized Controlled Trial and Follow-Up Study. Nutrients 2020, 12, 1978. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Liang, M.Y.; Zhang, X.Q. Changes in serum folic acid and vitamin B12 levels in liver cirrhosis and its clinical significance. Zhonghua Nei Ke Za Zhi 1992, 30, 625–627. (In Chinese) [Google Scholar] [PubMed]
- Muro, N.; Bujanda, L.; Sarasqueta, C.; Gil, I.; Hijona, E.; Cosme, Á.; Arenas, J.J.; Etlosegui, M.E.; Sarasola, M.; Calpasoro, J.; et al. Niveles plasmáticos de la vitamina B(12) y ácido fólico en pacientes con hepatopatía crónica [Plasma levels of folate and vitamin B(12) in patients with chronic liver disease]. Gastroenterol. Hepatol. 2010, 33, 280–287. [Google Scholar] [CrossRef]
- Alberino, F.; Gatta, A.; Amodio, P.; Merkel, C.; Pascoli, L.D.; Boffo, G.; Caregaro, L. Nutrition and survival in patients with liver cirrhosis. Nutrition 2001, 17, 445–450. [Google Scholar] [CrossRef]
- Herbert, V.; Zalusky, R.; Davidson, C.S. Correlation of folate deficiency with alcoholism and associated macrocytosis, anemia, and liver disease. Ann. Intern. Med. 1963, 58, 977–988. [Google Scholar] [CrossRef]
- Klipstein, F.A.; Lindenbaum, J. Folate Deficiency In Chronic Liver Disease. Blood 1965, 25, 443–456. [Google Scholar] [CrossRef]
- Majumdar, S.K.; Shaw, G.K.; O’Gorman, P.; Aps, E.J.; Offerman, E.L.; Thomson, A.D. Blood vitamin status (B1, B2, B6, folic acid and B12) in patients with alcoholic liver disease. Int. J. Vit. Am. Nutr. Res. 1982, 52, 266–271. [Google Scholar]
- Kazimierska, E.; Czestochowska, E. Serum homocysteine, vitamin B12 and folic acid concentrations in patients with alcoholic liver cirrhosis. Polski Merkuriusz Lekarski 2003, 15, 140–143. [Google Scholar] [PubMed]
- Lambert, D.; Benhayoun, S.; Adjalla, C.; Gélot, M.M.; Renkes, P.; Gérard, P.; Felden, F.; Belleville, F.; Gaucher, P.; Guéant, J.; et al. Alcoholic cirrhosis and cobalamin metabolism. Digestion 1997, 58, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Ermens, A.A.; Vlasveld, L.T.; Lindemans, J. Significance of elevated cobalamin (vitamin B12) levels in blood. Clin. Biochem. 2003, 36, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Wacker, M.; Holick, M.F. Vitamin D—Effects on skeletal and extraskeletal health and the need for supplementation. Nutrients 2013, 5, 111–148. [Google Scholar] [CrossRef]
- Fleet, J.C.; Kovalenko, P.L.; Li, Y.; Smolinski, J.; Spees, C.; Yu, J.G.; Thomas-Ahner, J.M.; Cui, M.; Neme, A.; Carlberg, C.; et al. Vitamin D Signaling Suppresses Early Prostate Carcinogenesis in TgAPT121 Mice. Cancer Prev. Res. 2019, 12, 343–356. [Google Scholar] [CrossRef]
- Huang, D.; Lei, S.; Wu, Y.; Weng, M.; Zhou, Y.; Xu, J.; Xia, D.; Xu, E.; Lai, M.; Zhang, H. Additively protective effects of vitamin D and calcium against colorectal adenoma incidence, malignant transformation and progression: A systematic review and meta-analysis. Clin. Nutr. 2019, 39, 2525–2538. [Google Scholar] [CrossRef] [PubMed]
- Potter, J.J.; Liu, X.; Koteish, A.; Mezey, E. 1,25-dihydroxyvitamin D3 and its nuclear receptor repress human α1 (I) collagen expression and type I collagen formation. Liver Int. 2013, 33, 677–686. [Google Scholar] [CrossRef]
- Anty, R.; Canivet, C.M.; Patouraux, S.; Ferrari-Panaia, P.; Saint-Paul, M.C.; Huet, P.M.; Lebeaupin, C.; Iannelli, A.; Gual, P.; Tran, A. Severe Vitamin D Deficiency May be an Additional Cofactor for the Occurrence of Alcoholic Steatohepatitis. Alcohol. Clin. Exp. Res. 2015, 39, 1027–1033. [Google Scholar] [CrossRef]
- Savić, Ž.; Vračarić, V.; Milić, N.; Nićiforović, D.; Damjanov, D.; Pellicano, R.; Medić-Stojanoska, M.; Abenavoli, L. Vitamin D supplementation in patients with alcoholic liver cirrhosis: A prospective study. Minerva Med. 2018, 109, 352–357. [Google Scholar] [CrossRef]
- Cyrus, T.; Yao, Y.; Rokach, J.; Tang, L.X.; Praticò, D. Vitamin E reduces progression of atherosclerosis in low-density lipoprotein receptor-deficient mice with established vascular lesions. Circulation 2003, 107, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Weinstein, S.J.; Yu, K.; Männistö, S.; Albanes, D. Relationship Between Serum Alpha-Tocopherol and Overall and Cause-Specific Mortality. Circ. Res. 2019, 125, 29–40. [Google Scholar] [CrossRef]
- Vilar-Gomez, E.; Vuppalanchi, R.; Gawrieh, S.; Ghabril, M.; Saxena, R.; Cummings, O.W.; Chalasani, N. Vitamin E Improves Transplant-Free Survival and Hepatic Decompensation Among Patients With Nonalcoholic Steatohepatitis and Advanced Fibrosis. Hepatology 2020, 71, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Soden, J.S.; Devereaux, M.W.; Haas, J.E.; Gumpricht, E.; Dahl, R.; Gralla, J.; Traber, M.G.; Sokol, R.J. Subcutaneous vitamin E ameliorates liver injury in an in vivo model of steatocholestasis. Hepatology 2007, 46, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.A.; Ibrahim, H.M.; Mohamed. A.A.; Tammam, H.G. Vitamin E supplementation ameliorates the hepatotoxicity induced by Tramadol: Toxicological, histological and immunohistochemical study. Toxicol Mech Methods. 2020, 30, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Masalkar, P.D.; Abhang, S.A. Oxidative stress and antioxidant status in patients with alcoholic liver disease. Clin. Chim. Acta 2005, 355, 61–65. [Google Scholar] [CrossRef]
- Ferré, N.; Camps, J.; Prats, E.; Girona, J.; Gómez, F.; Heras, M.; Sim, J.M.; Ribalta, J.; Joven, J. Impaired vitamin E status in patients with parenchymal liver cirrhosis: Relationships with lipoprotein compositional alterations, nutritional factors, and oxidative susceptibility of plasma. Metabolism 2002, 51, 609–615. [Google Scholar]
- Nag, S.; Manna, K.; Saha, M.; Das Saha, K. Tannic acid and vitamin E loaded PLGA nanoparticles ameliorate hepatic injury in a chronic alcoholic liver damage model via EGFR-AKT-STAT3 pathway. Nanomedicine 2020, 15, 235–257. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar]
- Abenavoli, L.; Greco, M.; Milic, N.; Accattato, F.; Foti, D.; Gulletta, E.; Luzza, F. Effect of Mediterranean diet and antioxidant formulation in non-alcoholic fatty liver disease: A randomized study. Nutrients 2017, 9, 870. [Google Scholar] [CrossRef]
- Buzzati, E.; Pinzani, M.; Tsochatzis, E.A. The multiple hit-pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 2016, 65, 1038–1048. [Google Scholar] [CrossRef]
- Tiniakos, D.G.; Vos, M.B.; Brunt, E.M. Nonalcoholic fatty liver disease: Pathology and pathogenesis. Annu. Rev. Pathol. 2010, 5, 145–171. [Google Scholar] [CrossRef] [PubMed]
- Baffy, G.; Brunt, E.M.; Caldwell, S.H. Hepatocellular carcinoma in non-alcoholic fatty liver disease: An emerging menace. J. Hepatol. 2012, 56, 1384–1391. [Google Scholar] [CrossRef]
- Mikolasevic, I.; Filipec-Kanizaj, T.; Mijic, M.; Jakopcic, I.; Milic, S.; Hrstic, I.; Sobocan, N.; Stimac, D.; Burra, P. Nonalcoholic fatty liver disease and liver—Where do we stand? World J. Gastroenterol. 2018, 24, 1491–1506. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Campbell-Sargent, C.; Mirshahi, F.; Rizzo, W.B.; Contos, M.J.; Sterling, R.K.; Sobocan, N.; Stimac, D.; Burrl, P. Nonalcoholic steatohepatitis: Association of insulin resistance and mitochondrial abnormalities. Gastroenterology 2001, 120, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Fabbrini, E.; Sullivan, S.; Klein, S. Obesity and nonalcoholic fatty liver disease: Biochemical, metabolic, and clinical implications. Hepatology 2010, 51, 679–689. [Google Scholar] [CrossRef]
- Eslam, M.; Sanyal, A.J.; George, J.; International Consensus Panel. MAFLD: A consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 2020, 158, 1999–2014. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010, 52, 1836–1846. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Yang, L.; McCall, S.; Huang, J.; Yu, X.X.; Pandey, S.K.; Bhanot, S.; Monia, B.P.; Li, Y.-X.; Diehl, A.M. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology 2007, 45, 1366–1374. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.X.; Ge, C.X.; Qin, Y.T.; Gu, T.T.; Lou, D.S.; Li, Q.; Hu, L.-F.; Feng, J.; Huang, P.; Tan, J. Prolonged PM2.5 exposure elevates risk of oxidative stress-driven nonalcoholic fatty liver disease by triggering increase of dyslipidemia. Free Radic. Biol. Med. 2019, 130, 542–556. [Google Scholar] [CrossRef]
- Ding, S.; Yuan, C.; Si, B.; Wang, M.; Da, S.; Bai, L.; Wu, W. Combined effects of ambient particulate matter exposure and a high-fat diet on oxidative stress and steatohepatitis in mice. PLoS ONE 2019, 14, e0214680. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.M.; Mercante, J.W.; Neish, A.S. Reactive oxygen production induced by the gut microbiota: Pharmacotherapeutic implications. Curr. Med. Chem. 2012, 19, 1519–1529. [Google Scholar] [CrossRef]
- Kamada, N.; Nunez, G. Regulation of the immune system by the resident intestinal bacteria. Gastroenterology 2014, 146, 1477–1488. [Google Scholar] [CrossRef] [PubMed]
- Vajro, P.; Paolella, G.; Fasano, A. Microbiota and gut-liver axis: A mini-review on their influences on obesity and obesity related liver disease. J. Pedeatr. Gastroenterol. Nutr. 2013, 56, 461–468. [Google Scholar] [CrossRef]
- Musso, G.; Gambino, R.; Cassader, M. Obesity, diabetes, and gut microbiota; the hygiene hypothesis expanded? Diabetes Care 2010, 33, 2277–2284. [Google Scholar] [CrossRef]
- Pierri, L.; Saggese, P.; Guercio Nuzio, S.; Troisi, J.; Di Stasi, M.; Poeta, M.; Poeta, M.; Savastano, R.; Marchese, G.; Tarallo, R.; et al. Relations of gut liver axis components and gut microbiota in obese children with fatty liver: A pilot study. Clin. Res. Hepatol. Gastroenterol. 2018, 42, 387–390. [Google Scholar] [CrossRef]
- Valitutti, F.; Cucchiara, S.; Fasano, A. Celiac disease and the microbiome. Nutrients 2019, 11, 2403. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and IBD: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef]
- Prakash, S.; Rodes, L.; Coussa-Charley, M.; Tomaro-Duchesneau, C. Gut microbiota: Next frontier in understanding human health and development of biotherapeutics. Biologics 2011, 5, 71–86. [Google Scholar] [CrossRef]
- Tariq, Z.; Green, J.C.; Hodson, L. Are oxidative stress mechanisms the common denominator in the progression from hepatic steatosis towards non-alcoholic steatohepatitis (NASH). Liver Int. 2014, 34, e180–e190. [Google Scholar] [CrossRef]
- Thakur, V.; Pritchard, M.T.; McMullen, M.R.; Wang, Q.; Nagy, L.E. Chronic ethanol feeding increases activation of NADPH oxidase by lipopolysaccharide in rat Kupffer cells: Role of increased reactive oxygen in LPS-stimulated ERK1/ 2 activation and TNF-alpha production. J. Leukoc. Biol. 2006, 79, 1348–1356. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Colletti, A.; Bellentani, S. Nutraceutical Approach to Non-Alcoholic Fatty Liver Disease (NAFLD): The Available Clinical Evidence. Nutrients 2018, 10, 1153. [Google Scholar] [CrossRef]
- Blanche, C.; Wang, X.D. Non-alcoholic steatohepatitis and hepatocellular carcinoma: Implications for lycopene intervention. Nutrients 2013, 6, 124–162. [Google Scholar] [CrossRef]
- Abenavoli, L.; Milanović, M.; Milić, N.; Luzza, F.; Giuffrè, A.M. Olive oil antioxidants and non-alcoholic fatty liver disease. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 739–749. [Google Scholar] [CrossRef]
- Emma, M.R.; Augello, G.; Di Stefano, V.; Azzolina, A.; Giannitrapani, L.; Montalto, G.; Cervello, M.; Cusimano, A. Potential Uses of Olive Oil Secoiridoids for the Prevention and Treatment of Cancer: A Narrative Review of Preclinical Studies. Int. J. Mol. Sci. 2021, 22, 1234. [Google Scholar] [CrossRef]
- Yubero-Serrano, E.M.; Lopez-Moreno, J.; Gomez-Delgado, F.; Lopez-Miranda, J. Extra virgin olive oil: More than a healthy fat. Eur. J. Clin. Nutr. 2019, 72, 8–17. [Google Scholar] [CrossRef]
- Pang, K.L.; Chin, K.Y. The Biological Activities of Oleocanthal from a Molecular Perspective. Nutrients 2018, 10, 570. [Google Scholar] [CrossRef]
- Cicerale, S.; Lucas, L.J.; Keast, R.S. Antimicrobial, antioxidant and anti-inflammatory phenolic activities in extra virgin olive oil. Curr. Opin. Biotechnol. 2012, 23, 129–135. [Google Scholar] [CrossRef]
- Patti, A.M.; Carruba, G.; Cicero, A.F.G.; Banach, M.; Nikolic, D.; Giglio, R.V.; Terranova, A.; Soresi, M.; Giannitrapani, L.; Montalto, G.; et al. Daily Use of Extra Virgin Olive Oil with High Oleocanthal Concentration Reduced Body Weight, Waist Circumference, Alanine Transaminase, Inflammatory Cytokines and Hepatic Steatosis in Subjects with the Metabolic Syndrome: A 2-Month Intervention Study. Metabolites 2020, 10, 392. [Google Scholar] [CrossRef]
- Priore, P.; Cavallo, A.; Gnoni, A.; Damiano, F.; Gnoni, G.V.; Siculella, L. Modulation of hepatic lipid metabolism by olive oil and its phenols in nonalcoholic fatty liver disease. IUBMB Life 2015, 67, 9–17. [Google Scholar] [CrossRef]
- Molenaar, M.R.; Penning, L.C.; Helms, J.B. Playing Jekyll and Hyde-The Dual Role of Lipids in Fatty Liver Disease. Cells 2020, 9, 2244. [Google Scholar] [CrossRef]
- Saeed, A.; Dullaart, R.P.F.; Schreuder, T.C.M.A.; Blokzijl, H.; Faber, K.N. Disturbed Vitamin A Metabolism in Non-Alcoholic Fatty Liver Disease (NAFLD). Nutrients 2017, 10, 29. [Google Scholar] [CrossRef]
- Coelho, J.M.; Cansanção, K.; Perez, R.M.; Leite, N.C.; Padilha, P.; Ramalho, A.; Peres, W. Association between serum and dietary antioxidant micronutrients and advanced liver fibrosis in non-alcoholic fatty liver disease: An observational study. PeerJ 2020, 8, e9838. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Hodgesm, R.E. Serum levels of vitamin C in relation to dietary and supplemental intake of vitamin C in smokers and nonsmokers. Ann. N. Y. Acad. Sci. 1987, 498, 144–152. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Poulsen, H.E. Is vitamin C supplementation beneficial? Lessons learned from randomised controlled trials. Br. J. Nutr. 2010, 103, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Matos, M.D.; Kawiecki, D.; Ballard, K.; Guo, Y.; Volek, J.S.; Bruno, R.S. Vitamin C status is related to proinflammatory responses and impaired vascular endothelial function in healthy, college-aged lean and obese men. J. Am. Diet. Assoc. 2011, 111, 737–743. [Google Scholar] [CrossRef]
- Barchetta, I.; Angelico, F.; Del Ben, M.; Baroni, M.G.; Pozzilli, P.; Morini, S.; Cavallo, M.G. Strong association between non alcoholic fatty liver disease (NAFLD) and low 25(OH) vitamin D levels in an adult population with normal serum liver enzymes. BMC Med. 2011, 9, 85. [Google Scholar] [CrossRef]
- Maria Del Bas, J.; Rodríguez, B.; Puiggròs, F.; Mariné, S.; Rodríguez, M.A.; Moriña, D.; Armengol, L.; Caimari, A.; Arola, L. Hepatic accumulation of S-adenosylmethionine in hamsters with non-alcoholic fatty liver disease associated with metabolic syndrome under selenium and vitamin E deficiency. Clin. Sci. 2019, 133, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Chongmelaxme, B.; Phisalprapa, P.; Sawangjit, R.; Dilokthornsakul, P.; Chaiyakunapruk, N. Weight Reduction and Pioglitazone are Cost-Effective for the Treatment of Non-Alcoholic Fatty Liver Disease in Thailand. Pharmacoeconomics 2019, 37, 267–278. [Google Scholar] [CrossRef]
- Simon, J.A.; Hudes, E.S.; Tice, J.A. Relation of serum ascorbic acid to mortality among us adults. J. Am. Coll. Nutr. 2001, 20, 255–263. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Bjelakovic, M.; Nagorni, A.; Gluud, C. Meta-analysis: Antioxidant supplements for liver diseases—The Cochrane Hepato-Biliary Group. Aliment. Pharmacol. Ther. 2010, 32, 356–367. [Google Scholar] [CrossRef]
- Valdecantos, M.P.; Perez-Matute, P.; Quintero, P.; Martinez, J.A. Vitamin C, resveratrol and lipoic acid actions on isolated rat liver mitochondria: All antioxidants but different. Redox Rep. 2010, 15, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Rose, F.J.; Webster, J.; Barry, J.B.; Phillips, L.K.; Richards, A.A.; Whitehead, J.P. Synergistic effects of ascorbic acid and thiazolidinedione on secretion of high molecular weight adiponectin from human adipocytes. Diabetes Obes. Metab. 2010, 12, 1084–1089. [Google Scholar] [CrossRef]
- Rezazadeh, A.; Yazdanparast, R.; Molaei, M. Amelioration of diet-induced nonalcoholic steatohepatitis in rats by Mn-salen complexes via reduction of oxidative stress. J. Biomed. Sci. 2012, 19, 26. [Google Scholar] [CrossRef]
- Ipsen, D.H.; Tveden-Nyborg, P.; Lykkesfeldt, J. Does vitamin C deficiency promote fatty liver disease development? Nutrients 2014, 6, 5473–5499. [Google Scholar] [CrossRef] [PubMed]
- Uzun, H.; Zengin, K.; Taskin, M.; Aydin, S.; Simsek, G.; Dariyerli, N. Changes in leptin, plasminogen activator factor and oxidative stress in morbidly obese patients following open and laparoscopic swedish adjustable gastric banding. Obes. Surg. 2004, 14, 659–665. [Google Scholar] [CrossRef]
- Tveden-Nyborg, P.; Lykkesfeldt, J. Does vitamin C deficiency increase lifestyle-associated vascular disease progression? Evidence based on experimental and clinical studies. Antioxid. Redox Signal. 2013, 19, 2084–2104. [Google Scholar] [CrossRef]
- Musso, G.; Gambino, R.; de Michieli, F.; Cassader, M.; Rizzetto, M.; Durazzo, M.; Fagà, E.; Silli, B.; Pagano, G. Dietary habits and their relations to insulin resistance and postprandial lipemia in nonalcoholic steatohepatitis. Hepatology 2003, 37, 909–916. [Google Scholar] [CrossRef]
- Han, J.M.; Jo, A.N.; Lee, S.M.; Bae, H.S.; Jun, D.W.; Cho, Y.K.; Suk, K.T.; Yoon, J.H.; Ahn, S.B.; Kim, S.W.; et al. Associations between intakes of individual nutrients or whole food groups and non-alcoholic fatty liver disease among Korean adults. J. Gastroenterol. Hepatol. 2014, 29, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Da, S.H.; Arendt, B.M.; Noureldin, S.A.; Therapondos, G.; Guindi, M.; Allard, J.P. A cross-sectional study assessing dietary intake and physical activity in Canadian patients with nonalcoholic fatty liver disease vs healthy controls. J. Acad. Nutr. Diet. 2014, 114, 1181–1194. [Google Scholar]
- Madan, K.; Bhardwaj, P.; Thareja, S.; Gupta, S.D.; Saraya, A. Oxidant stress and antioxidant status among patients with nonalcoholic fatty liver disease (NAFLD). J. Clin. Gastroenterol. 2006, 40, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Lei, G.H.; Fu, L.; Zeng, C.; Yang, T.; Peng, S.F. Association between Dietary Vitamin C Intake and Non-Alcoholic Fatty Liver Disease: A Cross-Sectional Study among Middle-Aged and Older Adults. PLoS ONE 2016, 11, e0147985. [Google Scholar] [CrossRef]
- Zhang, Z.C.; Liu, Y.; Xiao, L.L.; Li, S.F.; Jiang, J.H.; Zhao, Y.; Qian, S.-W.; Tang, Q.-Q.; Li, X. Up-regulation of miR-125b by estrogen protects against non-alcoholic fatty liver in female mice. J. Hepatol. 2015, 63, 1466–1475. [Google Scholar] [CrossRef]
- Yoneda, M.; Hasegawa, T.; Sato, K. Vitamin E therapy for NAFLD/NASH. Nutrition 2015, 31, 898–899. [Google Scholar] [CrossRef][Green Version]
- Nobili, V.; Manco, M.; Devito, R.; Di Ciommo, V.; Comparcola, D.; Sartorelli, M.R.; Piemonte, F.; Marcellini, M.; Angulo, P. Lifestyle intervention and antioxidant therapy in children with nonalcoholic fatty liver disease: A randomized, controlled trial. Hepatology 2008, 48, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Yoneda, M.; Nakamura, K.; Makino, I.; Terano, A. Plasma transforming growth factor-beta1 level and efficacy of alpha-tocopherol in patients with non-alcoholic steatohepatitis: A pilot study. Aliment. Pharmacol. Therapeutics. 2001, 15, 1667–1672. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Torgerson, S.; Hayashi, P.; Ward, J.; Schenker, S. Vitamin E and vitamin C treatment improves fibrosis in patients with nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2003, 98, 2485–2490. [Google Scholar] [CrossRef]
- Hadzi-Petrushev, N.; Dimovska, K.; Jankulovski, N.; Mitrov, D.; Mladenov, M. Supplementation with Alpha-Tocopherol and Ascorbic Acid to Nonalcoholic Fatty Liver Disease’s Statin Therapy in Men. Adv. Pharmacol. Sci. 2018, 2018, 4673061. [Google Scholar] [CrossRef]
- Nagashimada, M.; Ota, T. Role of vitamin E in nonalcoholic fatty liver disease. IUBMB Life 2019, 71, 516–522. [Google Scholar] [CrossRef]
- Yki-Järvinen, H. Nutritional Modulation of Non-Alcoholic Fatty Liver Disease and Insulin Resistance. Nutrients.. 2015, 7, 9127–9138. [Google Scholar] [CrossRef]
- Sumida, Y.; Yoneda, M. Current and future pharmacological therapies for NAFLD/NASH. J. Gastroenterol. 2018, 53, 362–376. [Google Scholar] [CrossRef]
- Wallert, M.; Börmel, L.; Lorkowski, S. Inflammatory Diseases and Vitamin E-What Do We Know and Where Do We Go? Mol. Nutr. Food Res. 2021, 65, e2000097. [Google Scholar] [CrossRef]
- Sumida, Y.; Naito, Y.; Tanaka, S.; Sakai, K.; Inada, Y.; Taketani, H.; Kanemasa, K.; Yasui, K.; Itoh, Y.; Okanoue, T. Long-term (C 2 year) efficacy of vitamin E for non-alcoholic steatohepatitis. Hepatogastroenterology 2013, 60, 1445–1450. [Google Scholar] [CrossRef]
- Vadarlis, A.; Antza, C.; Bakaloudi, D.R.; Doundoulakis, I.; Kalopitas, G.; Samara, M.; Dardavessis, T.; Maris, T.; Chourdakis, M. Systematic review with meta-analysis: The effect of vitamin E supplementation in adult patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2021, 36, 311–319. [Google Scholar] [CrossRef]
- Miller, E.R., 3rd; Pastor-Barriuso, R.; Dalal, D.; Riemersma, R.A.; Appel, L.J.; Guallar, E. Meta-analysis: High-dosage vitamin E supplementation may increase all-cause mortality. Ann. Intern. Med. 2005, 142, 37–46. [Google Scholar] [CrossRef]
- Batten, M.L.; Imanishi, Y.; Maeda, T.; Tu, D.C.; Moise, A.R.; Bronson, D.; Possin, D.; Van Gelder, R.N.; Baehr, W.; Palczewski, K. Lecithin-retinol Acyltransferase Is Essential for Accumulation of All-trans-Retinyl Esters in the Eye and in the Liver. J. Biol. Chem. 2003, 279, 10422–10432. [Google Scholar] [CrossRef]
- Lim, H.S.; Choi, J.; Lee, B.; Kim, S.G.; Kim, Y.S.; Yoo, J.J. Association between Inflammatory Biomarkers and Nutritional Status in Fatty Liver. Clin. Nutr. Res. 2020, 9, 182–194. [Google Scholar] [CrossRef]
- Pacifico, L.; Osborn, J.F.; Bonci, E.; Pierimarchi, P.; Chiesa, C. Association between vitamin D levels and nonalcoholic fatty liver disease: Potential confounding variables. Mini Rev. Med. Chem. 2019, 19, 310–332. [Google Scholar] [CrossRef]
- Roth, C.L.; Elfers, C.T.; Figlewicz, D.P.; Melhorn, S.J.; Morton, G.J.; Hoofnagle, A.; Yeh, M.M.; Nelson, J.E.; Kowdley, K.V. Vitamin D deficiency in obese rats exacerbates nonalcoholic fatty liver disease and increases hepatic resistin and toll-like receptor activation. Hepatology 2012, 55, 1103–1111. [Google Scholar] [CrossRef]
- Jaruvongvanich, V.; Ahuja, W.; Sanguankeo, A.; Wijarnpreecha, K.; Upala, S. Vitamin D and histologic severity of nonalcoholic fatty liver disease: A systematic review and meta-analysis. Dig. Liver Dis. 2017, 49, 618–622. [Google Scholar] [CrossRef]
- Saberi, B.; Dadabhai, A.S.; Nanavati, J.; Wang, L.; Shinohara, R.T.; Mullin, G.E. Vitamin D levels do not predict the stage of hepatic fibrosis in patients with non-alcoholic fatty liver disease: A PRISMA compliant systematic review and meta-analysis of pooled data. World J. Hepatol. 2018, 10, 142–154. [Google Scholar] [CrossRef]
- Liangpunsakul, S.; Chalasani, N. Serum vitamin D concentrations and unexplained elevation in ALT among US adults. Dig. Dis. Sci. 2011, 56, 2124–2129. [Google Scholar] [CrossRef]
- Cho, Y.H.; Kim, J.W.; Shim, J.O.; Yang, H.R.; Chang, J.Y.; Moon, J.S.; Ko, J.S. Association between vitamin D deficiency and suspected nonalcoholic fatty liver disease in an adolescent population. Pediatr. Gastroenterol. Hepatol. Nutr. 2019, 22, 233–241. [Google Scholar] [CrossRef]
- Leitao, J.; Carvalhana, S.; Silva, A.P.; Velasco, F.; Medeiros, I.; Alves, A.C.; Bourbon, M.; Oliveiros, B.; Carvalho, A.; Cortez-Pinto, H. No evidence for lower levels of serum vitamin D in the presence of hepatic steatosis. A study on the portuguese general population. Int. J. Med. Sci. 2018, 15, 1778–1786. [Google Scholar] [CrossRef]
- Tabrizi, R.; Moosazadeh, M.; Lankarani, K.B.; Akbari, M.; Heydari, S.T.; Kolahdooz, F.; Samimi, M.; Asemi, Z. The effects of vitamin D supplementation on metabolic profiles and liver function in patients with non-alcoholic fatty liver disease: A systematic review and meta-analysis of randomized controlled trials. Diabetes Metab. Syndr. 2017, 11, S975–S982. [Google Scholar] [CrossRef]
- Barchetta, I.; Del Ben, M.; Angelico, F.; Di Martino, M.; Fraioli, A.; La Torre, G.; Saulle, R.; Perri, L.; Morini, S.; Tiberti, C.; et al. No effects of oral vitamin D supplementation on non-alcoholic fatty liver disease in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled trial. BMC Med. 2016, 14, 92. [Google Scholar] [CrossRef]
- Kitson, M.T.; Pham, A.; Gordon, A.; Kemp, W.; Roberts, S.K. High-dose vitamin D supplementation and liver histology in NASH. Gut 2016, 65, 717–718. [Google Scholar] [CrossRef]
- Amiri, H.L.; Agah, S.; Azar, J.T.; Hosseini, S.; Shidfar, F.; Mousavi, S.N.; Blv, M. Effect of daily calcitriol supplementation with and without calcium on disease regression in non-alcoholic fatty liver patients following an energy-restricted diet: Randomized, controlled, double-blind trial. Clin. Nutr. 2017, 36, 1490–1497. [Google Scholar] [CrossRef]
- Della Corte, C.; Carpino, G.; De Vito, R.; De Stefanis, C.; Alisi, A.; Cianfarani, S.; Overi, D.; Mosca, A.; Stronati, L.; Cucchiara, S.; et al. Docosahexanoic Acid Plus Vitamin D Treatment Improves Features of NAFLD in Children with Serum Vitamin D Deficiency: Results from a Single Centre Trial. PLoS ONE 2016, 11, e0168216. [Google Scholar] [CrossRef]
- Lee, S.M.; Jun, D.W.; Cho, Y.K.; Jang, K.S. Vitamin D deficiency in non-alcoholic fatty liver disease: The chicken or the egg? Clin. Nutr. 2017, 36, 191–197. [Google Scholar] [CrossRef]
- Zhang, Z.; Thorne, J.L.; Moore, J.B. Vitamin D and nonalcoholic fatty liver disease. Curr. Opin. Clin. Nutr. Metab. Care 2019, 22, 449–458. [Google Scholar] [CrossRef]
- IARC. 2018. Available online: http://globocan.iarc.fr/ (accessed on 29 May 2018).
- Simbrunner, B.; Semmler, G.; Stadlmann, A.; Scheiner, B.; Schwabl, P.; Paternostro, R.; Bucsics, T.; Bauer, D.; Eigenbauer, E.; Pinter, M.; et al. Vitamin A levels reflect disease severity and portal hypertension in patients with cirrhosis. Hepatol. Int. 2020, 14, 1093–1103. [Google Scholar] [CrossRef]
- Lee, Y.S.; Jeong, W.I. Retinoic acids and hepatic stellate cells in liver disease. J. Gastroenterol. Hepatoloy 2012, 27, 75–79. [Google Scholar] [CrossRef]
- Grunhage, F.; Hochrath, K.; Krawczyk, M.; Höblinger, A.; Obermayer-Pietsch, B.; Geisel, J.; Trauner, M.; Sauerbruch, T.; Lammert, F. Common genetic variation in vitamin D metabolism is associated with liver stiffness. Hepatology 2012, 56, 1883–1891. [Google Scholar] [CrossRef]
- Rocchi, E.; Seium, Y.; Camellini, L.; Casalgrandi, G.; Borghi, A.; D’Alimonte, P.; Cioni, G. Hepatic tocopherol content in primary hepatocellular carcinoma and liver metastases. Hepatology 1997, 26, 67–72. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, W.; Liu, Y.; Gong, W.; Sun, P.; Kong, X.; Yang, M.; Wang, Z. Diagnostic value of prothrombin induced by the absence of vitamin K or antagonist-II (PIVKA-II) for early stage HBV related hepatocellular carcinoma. Infect. Agent Cancer 2017, 12, 47. [Google Scholar] [CrossRef]
- Yang, T.; Xing, H.; Wang, G.; Wang, N.; Liu, M.; Yan, C.; Li, H.; Wei, L.; Li, S.; Fan, Z.; et al. A Novel Online Calculator Based on Serum Biomarkers to Detect Hepatocellular Carcinoma among Patients with Hepatitis, B. Clin Chem. 2019, 65, 1543–1553. [Google Scholar] [CrossRef]
- Li, T.; Yu, Y.; Liu, J.; Tian, X.; Kong, M.; Wu, L.; Tang, S.; Gu, S.; Zhao, J.; Cui, Y.; et al. PIVKA-II level is correlated to development of portal vein tumor thrombus in patients with HBV-related hepatocellular carcinoma. Infect. Agent Cancer 2019, 14, 13. [Google Scholar] [CrossRef]
- Jinghe, X.; Mizuta, T.; Ozaki, I. Vitamin K and hepatocellular carcinoma: The basic and clinic. World J. Clin. Cases 2015, 3, 757–764. [Google Scholar] [CrossRef]
- Kakizaki, S.; Sohara, N.; Sato, K.; Suzuki, H.; Yanagisawa, M.; Nakajima, H.; Takagi, H.; Naganuma, A.; Otsuka, T.; Takahashi, H.; et al. Preventive effects of vitamin K on recurrent disease in patients with hepatocellular carcinoma arising from hepatitis C viral infection. J. Gastroenterol. Hepatol. 2007, 22, 518–522. [Google Scholar] [CrossRef]
- Peres, W.A.; Chaves, G.V.; Gonçalves, J.C.; Ramalho, A.; Coelho, H.S. Vitamin A deficiency in patients with hepatitis C virus-related chronic liver disease. Br. J. Nutr. 2011, 106, 1724–1731. [Google Scholar] [CrossRef]
- He, H.; Mennone, A.; Boyer, J.L.; Cai, S.Y. Combination of retinoicacid and ursodeoxycholic acid attenuates liver injury in bileduct-ligated rats and human hepatic cells. Hepatology 2011, 53, 548–557. [Google Scholar] [CrossRef]
- Leo, M.A.; Lieber, C.S. Alcohol, vitamin A, and beta-carotene: Adverse interactions, including hepatotoxicity and carcinogenicity. Am. J. Clin Nutr. 1999, 69, 1071–1085. [Google Scholar] [CrossRef]
- Geubel, A.P.; De Galocsy, C.; Alves, N.; Rahier, J.; Dive, C. Liver damage caused by therapeutic vitamin A administration: Estimate of dose-related toxicity in 41 cases. Gastroenterology 1991, 100, 1701–1709. [Google Scholar] [CrossRef]
- Petta, S.; Grimaudo, S.; Marco, V.D.; Scazzone, C.; Macaluso, F.S.; Cammà, C.; Cabibi, D.; Pipitone, R.; Craxì, A. Association of vitamin D serum levels and its common genetic determinants, with severity of liver fibrosis in genotype 1 chronic hepatitis C patients. J. Viral Hepat. 2013, 20, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Triantos, C.; Kalafateli, M.; Aggeletopoulou, I.; Diamantopoulou, G.; Spantidea, P.I.; Michalaki, M.; Vourli, G.; Konstantakis, C.; Assimakopoulos, S.F.; Manolakopoulos, S.; et al. Vitamin D-related immunomodulation in patients with liver cirrhosis. Eur. J. Gastroenterol. Hepatol. 2020, 32, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Bitetto, D.; Fattovich, G.; Fabris, C.; Ceriani, E.; Falleti, E.; Fornasiere, E.; Pasino, M.; Ieluzzi, D.; Cussigh, A.; Cmet, S.; et al. Complementary role of vitamin D deficiency and the interleukin-28B rs12979860 C/T polymorphism in predicting antiviral response in chronic hepatitis. Hepatology 2011, 53, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Agmon-Levin, N.; Kopilov, R.; Selmi, C.; Nussinovitch, U.; Sánchez-Castañón, M.; López-Hoyos, M.; Amital, H.; Kivity, S.; Gershwin, E.M.; Shoenfeld, Y. Vitamin D in primary biliary cirrhosis, a plausible marker of advanced disease. Immunol Res. 2015, 61, 141–146. [Google Scholar] [CrossRef]
- Ebadi, M.; Bhanji, R.A.; Mazurak, V.C.; Lytvyak, E.; Mason, A.; Czaja, A.J.; Montano-Loza, A.J. Severe vitamin D deficiency is a prognostic biomarker in autoimmune hepatitis. Aliment. Pharmacol. Ther. 2019, 49, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Basili, S.; Raparelli, V.; Napoleone, L.; Talerico, G.; Corazza, G.R.; Perticone, F.; Sacerdoti, D.; Andriulli, A.; Licata, A.; Pietrangelo, A.; et al. Platelet Count Does Not Predict Bleeding in Cirrhotic Patients: Results from the PRO-LIVER Study. Am. J. Gastroenterol. 2018, 113, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Andriulli, A.; Tripodi, A.; Angeli, P.; Senzolo, M.; Primignani, M.; Giannini, E.G.; Zuin, M. Under the auspices of the Italian Association for the Study of Liver Diseases (AISF) and the Italian Society of Internal Medicine (SIMI). Hemostatic balance in patients with liver cirrhosis: Report of a consensus conference. Dig. Liver Dis. 2016, 48, 455–467. [Google Scholar] [CrossRef]
- Sallah, S.; Bobzien, W. Bleeding problems in patients with liver disease. Ways to manage the many hepatic effects on coagulation. Postgrad. Med. 1999, 106, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Saja, M.F.; Abdo, A.A.; Sanai, F.M.; Shaikh, S.A.; Gader, A.G. The coagulopathy of liver disease: Does vitamin K help? Blood Coagul. Fibrinolysis 2013, 24, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Wang, N.; Yang, Y.; Ma, L.; Du, Q.; Zhang, W.; Tran, A.H.; Jung, H.; Soh, A.; Zheng, Y.; et al. Review on Vitamin K Deficiency and its Biomarkers: Focus on the Novel Application of PIVKA-II in Clinical Practice. Clin. Lab. 2018, 64, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Yan, C.; Cheng, L.; Wang, N.; Dai, S.; Yuan, J.; Lu, W.; Wang, Z.; Han, J.; Zheng, Y.; et al. Clinical application of protein induced by vitamin K antagonist-II as a biomarker in hepatocellular carcinoma. Tumour Biol. 2016, 37, 15447–15456. [Google Scholar] [CrossRef] [PubMed]
- Azuma, K.; Inoue, S. Vitamin K function mediated by activation of steroid and xenobiotic receptor. Clin. Calcium 2009, 19, 1770–1778. (In Japanese) [Google Scholar]
- Tabb, M.M.; Sun, A.; Zhou, C.; Grün, F.; Errandi, J.; Romero, K.; Pham, H.; Inoue, S.; Mallick, S.; Lin, M.; et al. Vitamin K2 regulation of bone homeostasis is mediated by the steroid and xenobiotic receptor SXR. J. Biol. Chem. 2003, 278, 43919–43927. [Google Scholar] [CrossRef]
- Otsuka, M.; Kato, N.; Shao, R.X.; Hoshida, Y.; Ijichi, H.; Koike, Y.; Taniguchi, H.; Moriyama, M.; Shiratori, Y.; Kawabe, T.; et al. Vitamin K2 inhibits the growth and invasiveness of hepatocellular carcinoma cells via protein kinase A activation. Hepatology 2004, 40, 243–251. [Google Scholar] [CrossRef]
- Habu, D.; Shiomi, S.; Tamori, A.; Takeda, T.; Tanaka, T.; Kubo, S.; Nishiguchi, S. Role of vitamin K2 in the development of hepatocellular carcinoma in women with viral cirrhosis of the liver. JAMA 2004, 292, 358–361. [Google Scholar] [CrossRef]
- Haruna, Y.; Hasegawa, N.; Imanaka, K.; Kawamoto, S.; Inoue, A. Clinical Impact of Vitamin K Dosing on Sorafenib Treatment for Hepatocellular Carcinoma. J. Cancer 2017, 8, 1988–1994. [Google Scholar] [CrossRef]
- Schreiber, P.W.; Bischoff-Ferrari, H.A.; Boggian, K.; van Delden, C.; Enriquez, N.; Fehr, T.; Garzoni, C.; Hirsch, H.H.; Hirzel, C.; Manuel, O.; et al. Vitamin D status and risk of infections after liver transplantation in the Swiss Transplant Cohort Study. Transpl. Int. 2019, 32, 49–58. [Google Scholar] [CrossRef]
- Bitetto, D.; Fabris, C.; Falleti, E.; Fornasiere, E.; Fumolo, E.; Fontanini, E.; Cussigh, A.; Occhino, G.; Baccarani, U.; Pirisi, M.; et al. Vitamin D and the risk of acute allograft rejection following human liver transplantation. Liver Int. 2010, 30, 417–444. [Google Scholar] [CrossRef]
- Grant, C. A vitamin D protocol post-liver transplantation. J. Am. Assoc. Nurse Pract. 2017, 29, 658–666. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Li, L.; Chen, Y.; Zhang, J.; Zhong, L.; Peng, Z.; Xing, T. Vitamin D supplementation could reduce the risk of acute cellular rejection and infection in vitamin D deficient liver allograft recipients. Int. Immunopharmacol. 2019, 75, 105811. [Google Scholar] [CrossRef]
- Xing, T.; Qiu, G.; Zhong, L.; Ling, L.; Huang, L.; Peng, Z. Calcitriol reduces the occurrence of acute cellular rejection of liver transplants: A prospective controlled study. Pharmazie 2013, 68, 821–826. [Google Scholar] [PubMed]
- Zhang, A.; Wang, Y.; Xie, H.; Zheng, S. Calcitriol inhibits hepatocyte apoptosis in rat allograft by regulating apoptosis-associated genes. Int. Immunopharmacol. 2007, 7, 1122–1128. [Google Scholar] [CrossRef]
- Ninkovic, M.; Love, S.A.; Tom, B.; Alexander, G.J.; Compston, J.E. High prevalence of osteoporosis in patients with chronic liver disease prior to liver transplantation. Calcif. Tissue Int. 2001, 69, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Keane, J.T.; Elangovan, H.; Stokes, R.A.; Gunton, J.E. Vitamin D and the Liver-Correlation or Cause? Nutrients 2018, 10, 496. [Google Scholar] [CrossRef]
- Chaney, A.; Heckman, M.G.; Diehl, N.N.; Meek, S.; Keaveny, A.P. Effectiveness and outcomes of current practice in treating vitamin d deficiency in patients listed for liver transplantation. Endocr. Pract. 2015, 21, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Venu, M.; Martin, E.; Saeian, K.; Gawrieh, S. High prevalence of vitamin A deficiency and vitamin D deficiency in patients evaluated for liver transplantation. Liver Transpl. 2013, 19, 627–633. [Google Scholar] [CrossRef]
- Teriaky, A.; Mosli, M.; Chandok, N.; Al-Judaibi, B.; Marotta, P.; Qumosani, K. Prevalence of fat-soluble vitamin (A, D, and E) and zinc deficiency in patients with cirrhosis being assessed for liver transplantation. Acta Gastroenterol. Belg. 2017, 80, 237–241. [Google Scholar] [PubMed]
- Senzolo, M.; Burra, P.; Cholongitas, E.; Burroughs, A.K. New insights into the coagulopathy of liver disease and liver transplantation. World J. Gastroenterol. 2006, 12, 7725–7736. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.H.; Kim, D.; Cho, H.; Shin, B.S. Ascorbic acid improves thrombotic function of platelets during living donor liver transplantation by modulating the function of the E3 ubiquitin ligases c-Cbl and Cbl-b. J. Int. Med. Res. 2019, 47, 1856–1867. [Google Scholar] [CrossRef]
| Status in Liver Disease | Physiological Role | Potential Role in Liver Disease | |
|---|---|---|---|
| Zinc | ↓ | Neurotransmitter functions, intracellular signaling transduction, inflammatory response, ROS production, immune regulation, wound healing, gene expression | Mitochondrial dysfunction, oxidative injury, glutathione depletion [29] |
| Iron | ↑ | Transportation of oxygen, DNA and ATP synthesis | HSCs activation, liver fibrosis promotion, ferroptosis, increased risk of infections, ROS increased production [29] |
| Copper | ↓/↑ | Bone marrow and CNS homeostasis; co-factor of antioxidant enzymes | Interaction with other trace elements [29] |
| Vitamin B group | ↓ | Pleiotropic co-enzymatic activity, direct precursor for metabolic substrates, antioxidant response | Vitamin B6: limitation of glutathione synthesis affecting antioxidant capability of the liver [30,31,32] |
| Vitamin D | ↓ | Calcium homeostasis immuno-modulating activity | Vitamin D deficiency is associated with poor prognosis and complications of portal hypertension in cirrhosis [33] |
| Vitamin E | ↓ | Antioxidant immuno-modulating activity | Deficiency could increase oxidative stress, modifying the composition of gut microbiota [34] in addition to anti-inflammatory and antioxidant effects and signal transduction of P53, NFkB and Cyclin D1 pathways [35] |
| Antioxidants | Status in Liver Disease | Potential Role in Liver Disease |
|---|---|---|
| Enzymatic | ||
| Superoxide dismutase (SOD) | Possible reduction of inflammatory-induced liver damage [21] | |
| Glutathione | ||
| Non-enzymatic | ||
| Flavonoids | ↓ | Reduction of IL-1α and IFN-γ |
| Lycopene | ↓ | Inhibition of liver diseases, including NAFLD and liver cancer [88] |
| Coenzyme Q10 | ↓ | Reduction of NAFLD degree, transaminases, gamma-GT levels, oxidized LDL levels [87] |
| Olive oil (phenolic component) | ↓ | Reduction of lipogenic pathway and thus attenuation of liver steatosis [95] |
| Vitamin A | ↓ | IFN response modulation Fibrosis development [96,97,98] |
| Vitamin C | ↓ | Possible influence on the progression towards NAFLD [99,100,101] |
| Vitamin D | ↓ | Reduction of fibrosis in HCV-infected patients, reduction of inflammation in NAFLD and HCC proliferation [102] |
| Vitamin E | ↓ | Reduction of LFTs in NAFLD/NASH patients, liver steatosis and lobular inflammation. Possible cirrhosis and HCC reduction if combined with pioglitazone in NAFLD [103,104] |
| Status in Liver Disease | Potential Role in Liver Diseases | |
|---|---|---|
| Vitamin A | ↓ | Fibrosis development, if deficient [148,149] |
| Vitamin D | ↓ | Anti-fibrotic effect [15] Reduction of HCC proliferation [150] |
| Vitamin E | ↓ | Possible HCC reduction [151] |
| Vitamin K | PIVKA-II as a diagnostic marker in HCC patients [152,153,154] Reduction of disease recurrence and long-term survival in patients with HCC after resection or percutaneous local ablation therapy when combined with anti-tumoral agents [155,156] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Licata, A.; Zerbo, M.; Como, S.; Cammilleri, M.; Soresi, M.; Montalto, G.; Giannitrapani, L. The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement? Nutrients 2021, 13, 4014. https://doi.org/10.3390/nu13114014
Licata A, Zerbo M, Como S, Cammilleri M, Soresi M, Montalto G, Giannitrapani L. The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement? Nutrients. 2021; 13(11):4014. https://doi.org/10.3390/nu13114014
Chicago/Turabian StyleLicata, Anna, Maddalena Zerbo, Silvia Como, Marcella Cammilleri, Maurizio Soresi, Giuseppe Montalto, and Lydia Giannitrapani. 2021. "The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement?" Nutrients 13, no. 11: 4014. https://doi.org/10.3390/nu13114014
APA StyleLicata, A., Zerbo, M., Como, S., Cammilleri, M., Soresi, M., Montalto, G., & Giannitrapani, L. (2021). The Role of Vitamin Deficiency in Liver Disease: To Supplement or Not Supplement? Nutrients, 13(11), 4014. https://doi.org/10.3390/nu13114014






