Beta-Glucans from Fungi: Biological and Health-Promoting Potential in the COVID-19 Pandemic Era
Abstract
:1. Introduction
2. Review
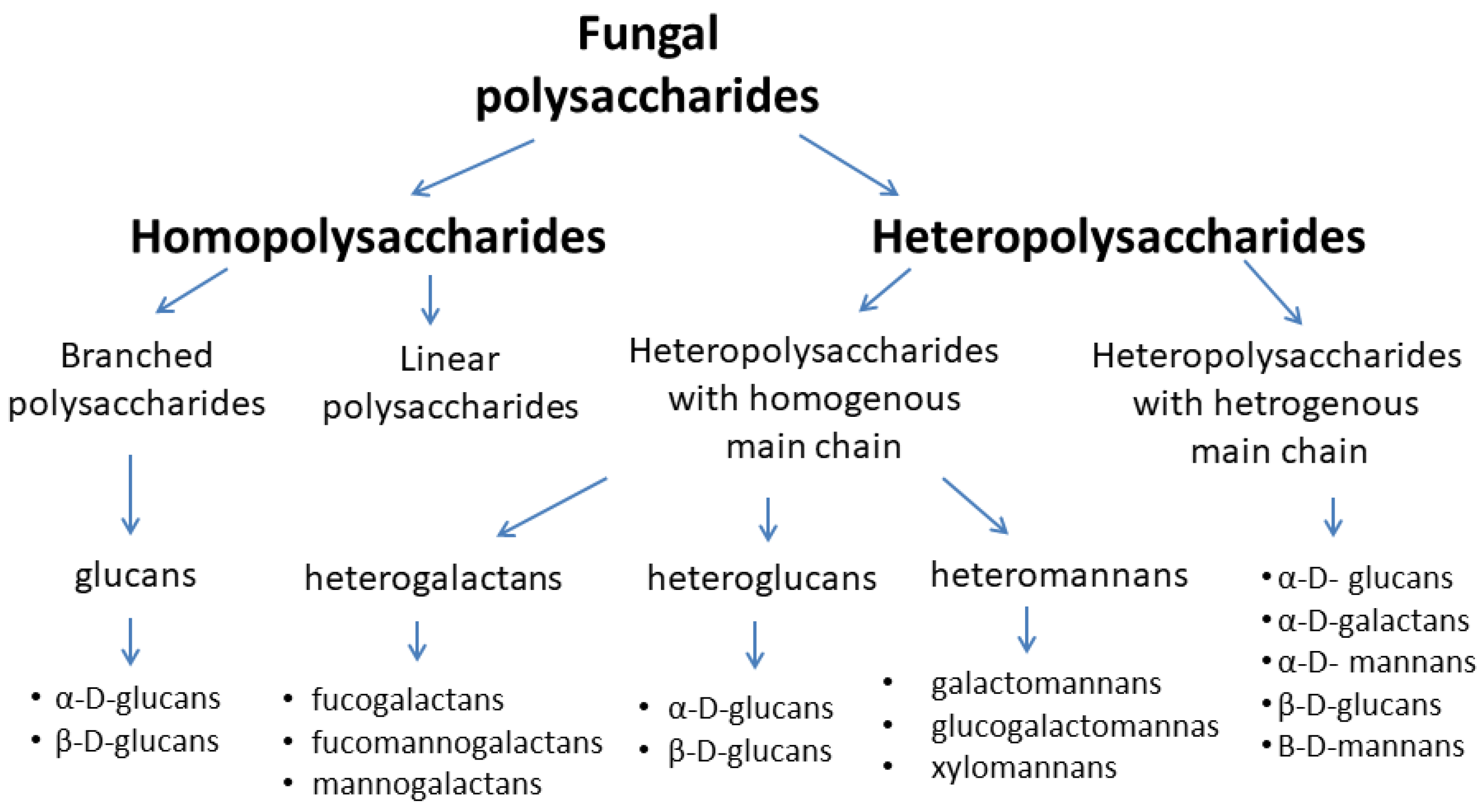
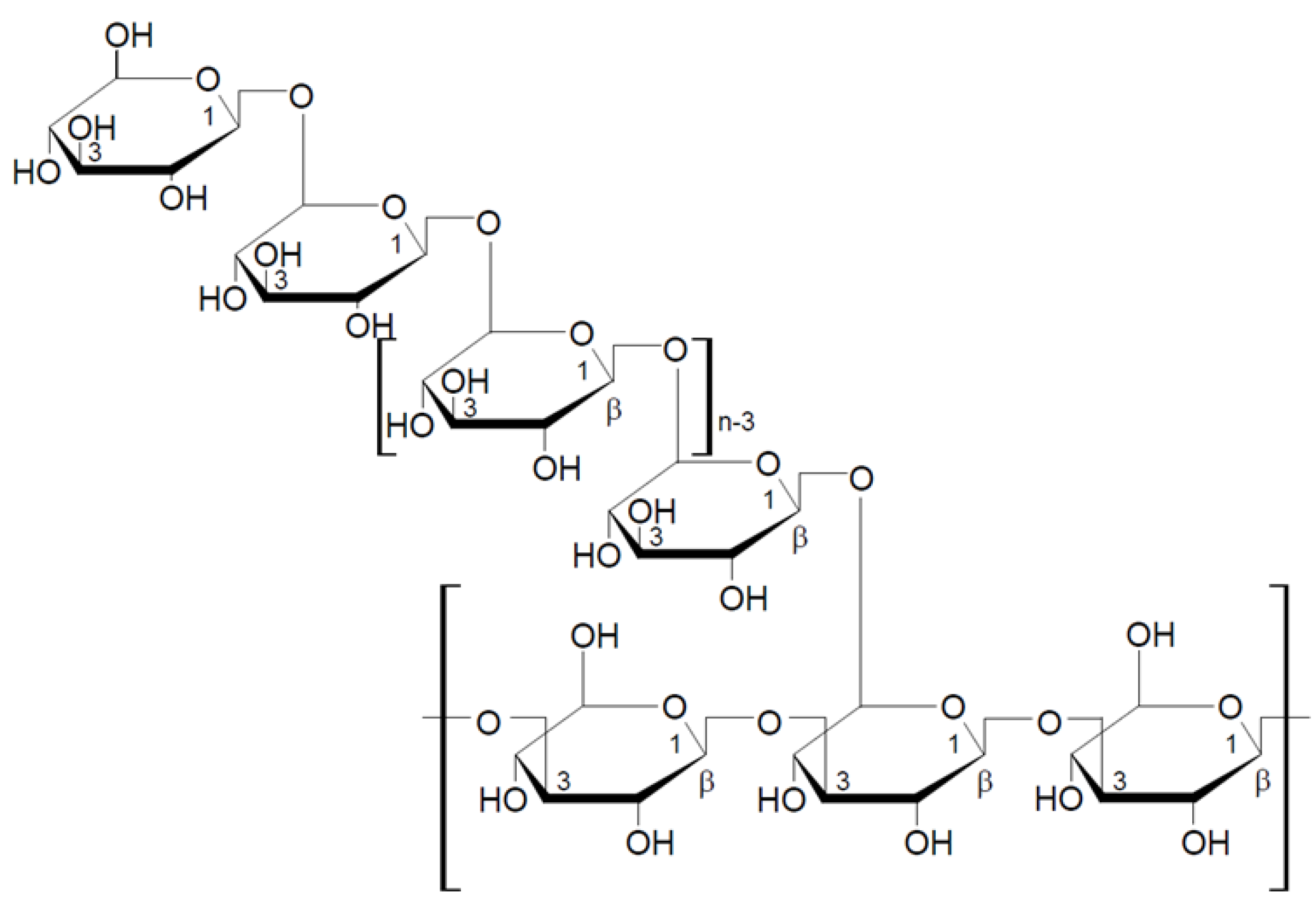
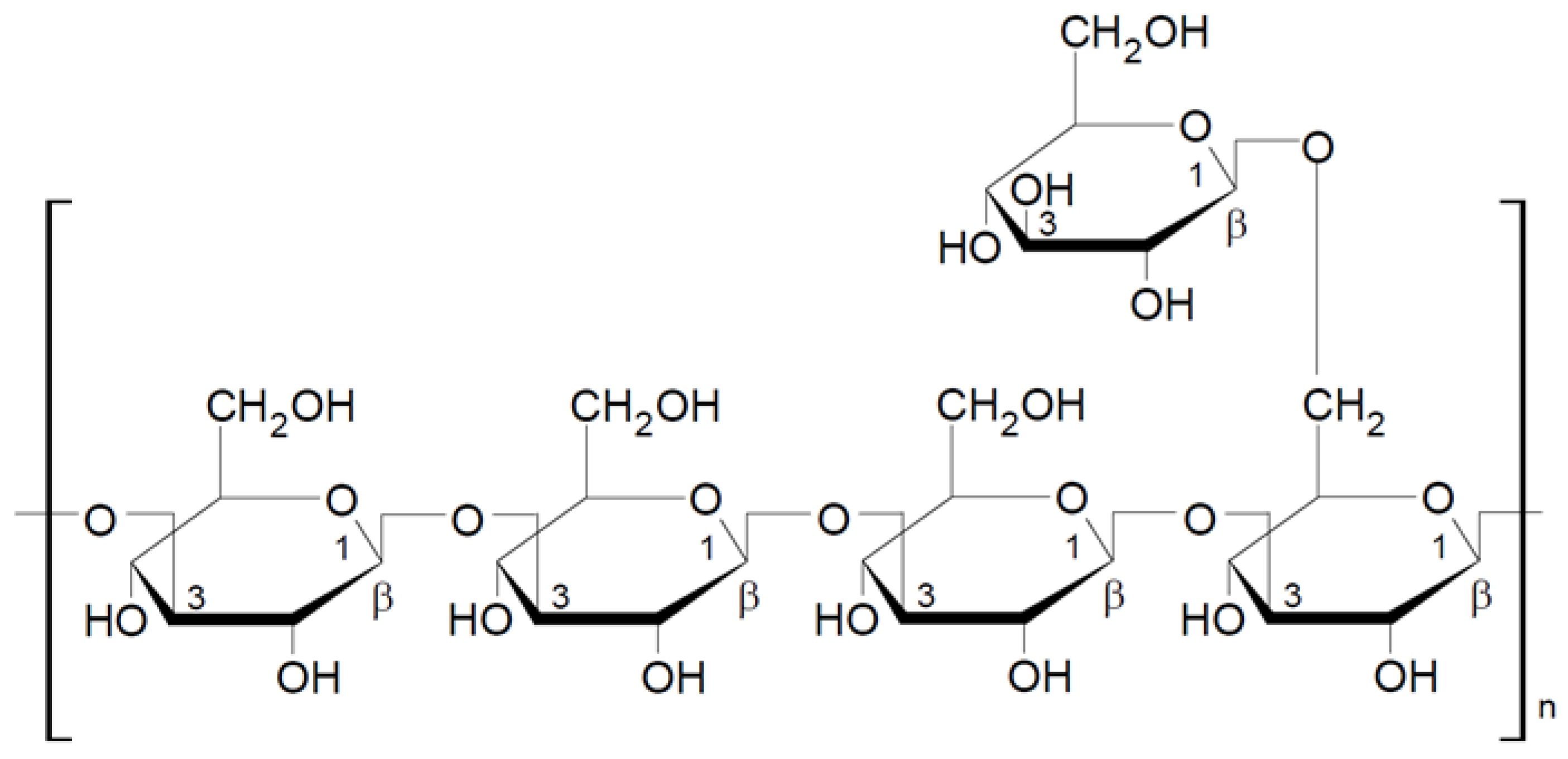
2.1. Classification and Structure of Mushroom Glucans
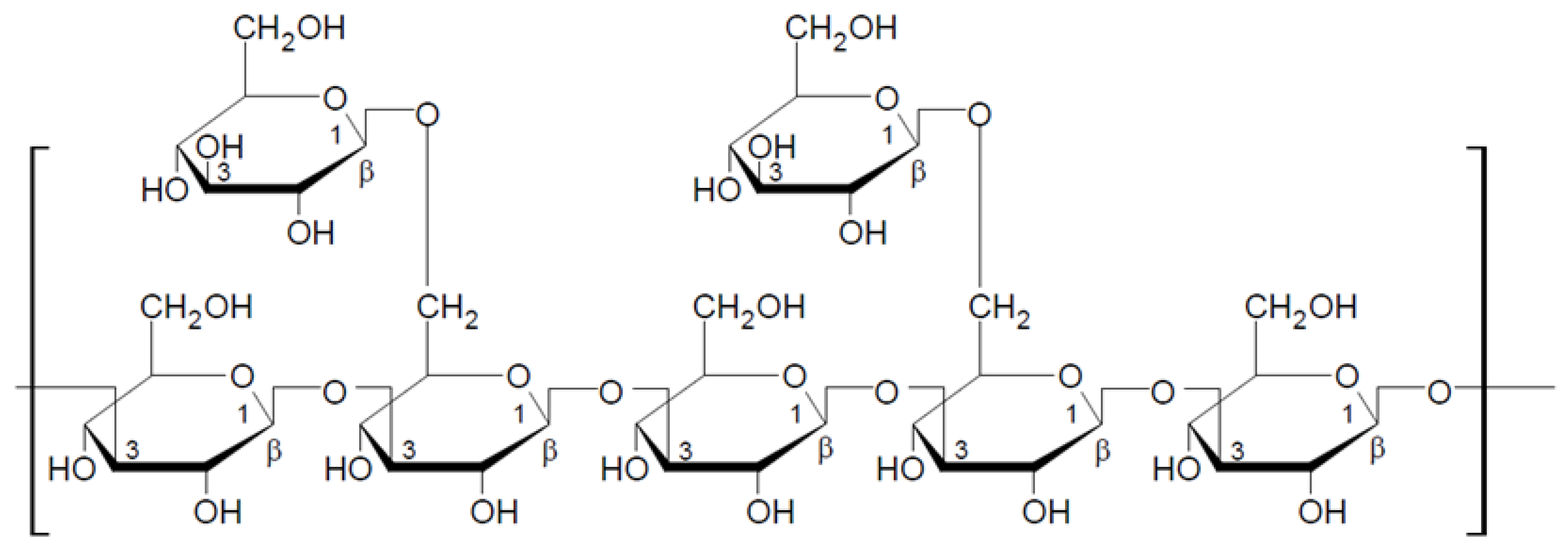
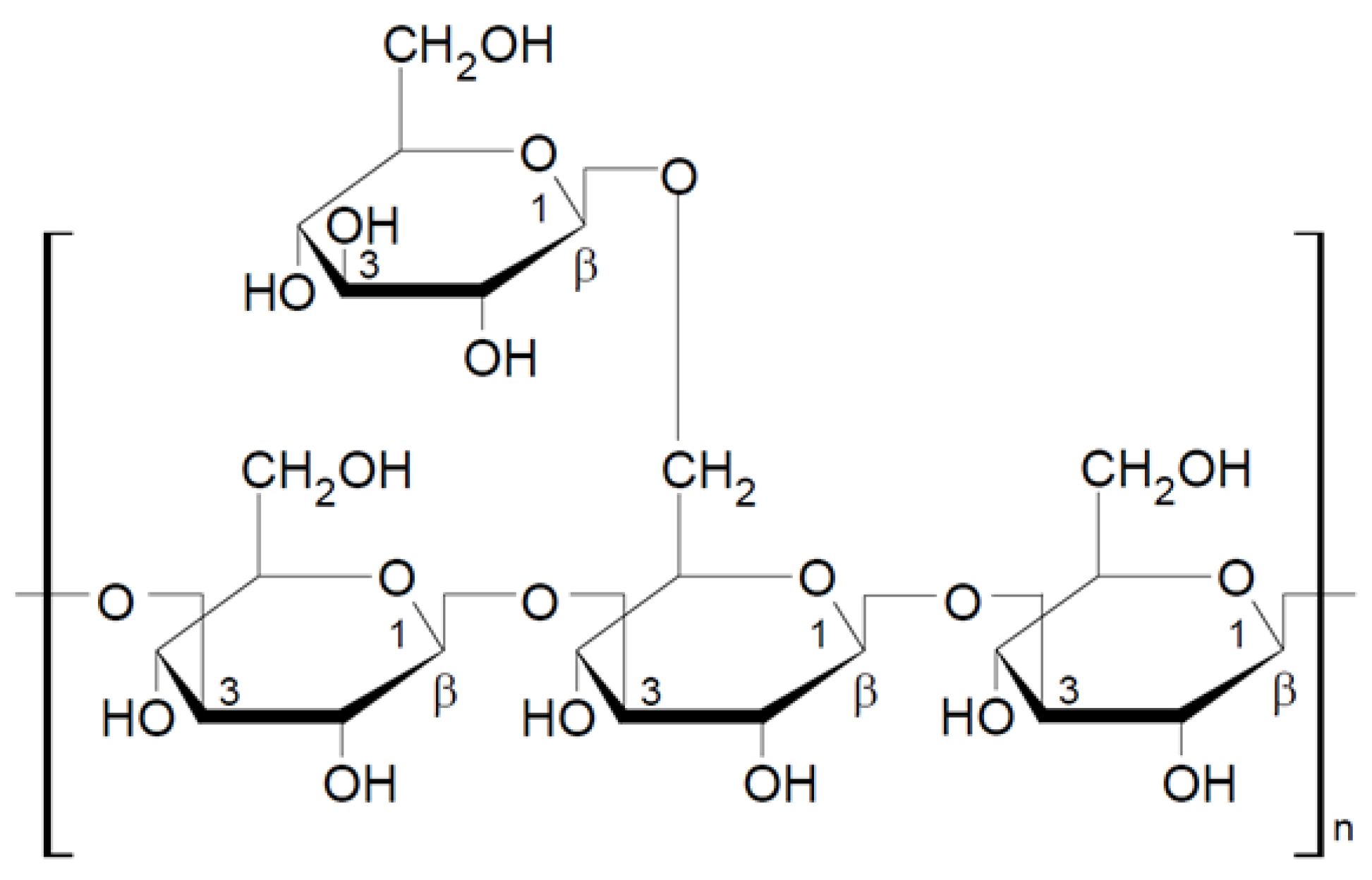
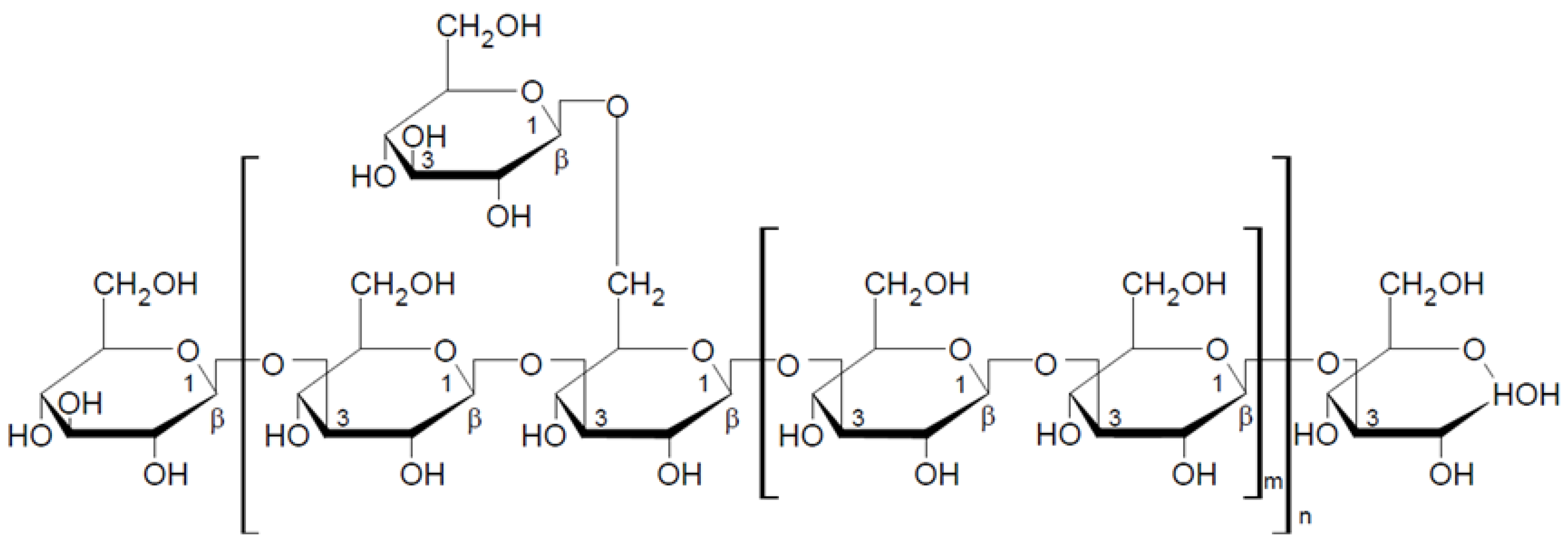
2.1.1. General Characteristic of β-Glucans
2.1.2. Mushroom Beta-Glucans
2.2. Structural Characteristics of Selected Beta-Glucans from Macrofungi
2.2.1. Lentinan
2.2.2. Schizophyllan
2.2.3. Krestin
2.2.4. Grifolan
2.2.5. Pleuran
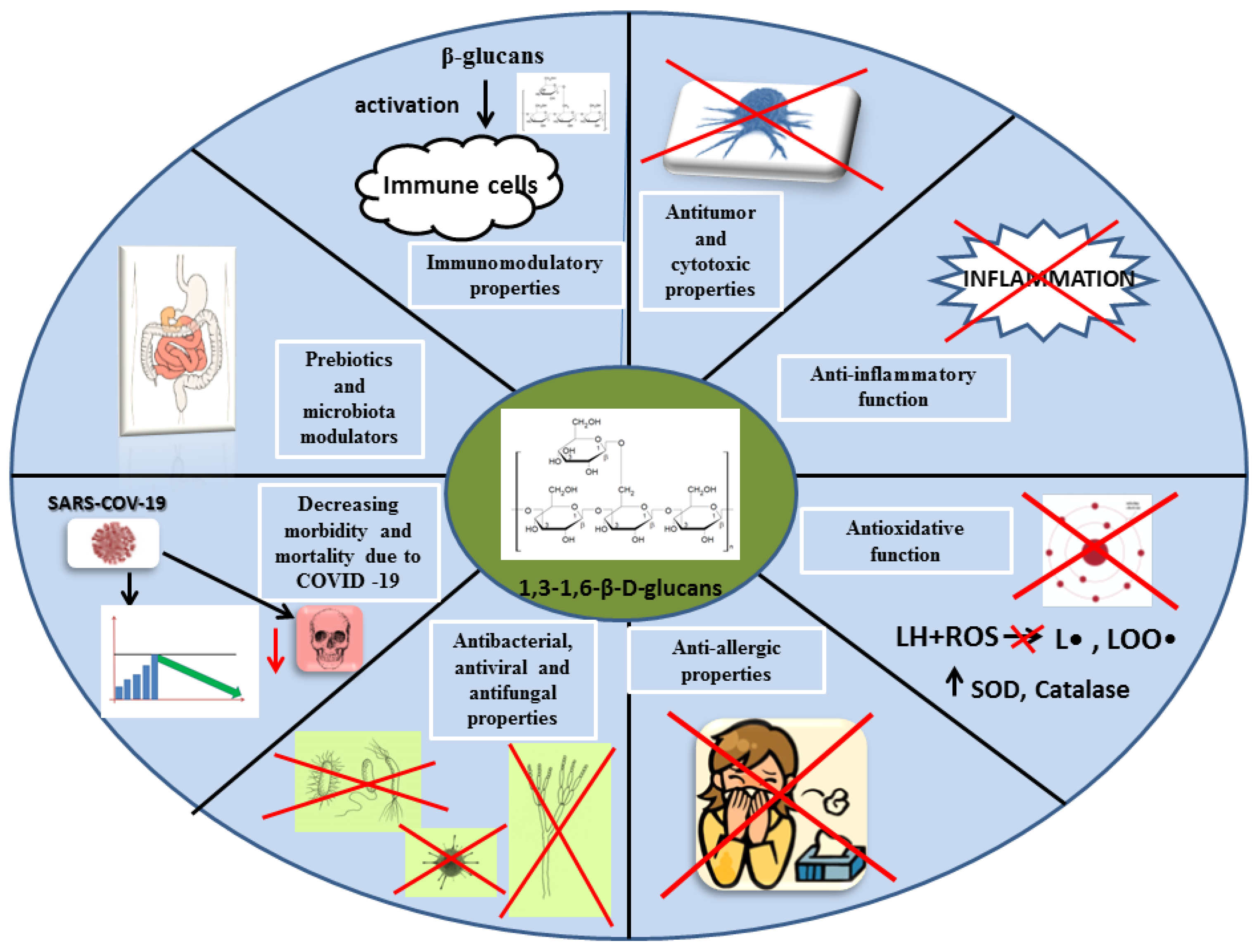
2.3. Mechanisms of Action of Beta-Glucans in the Human Body
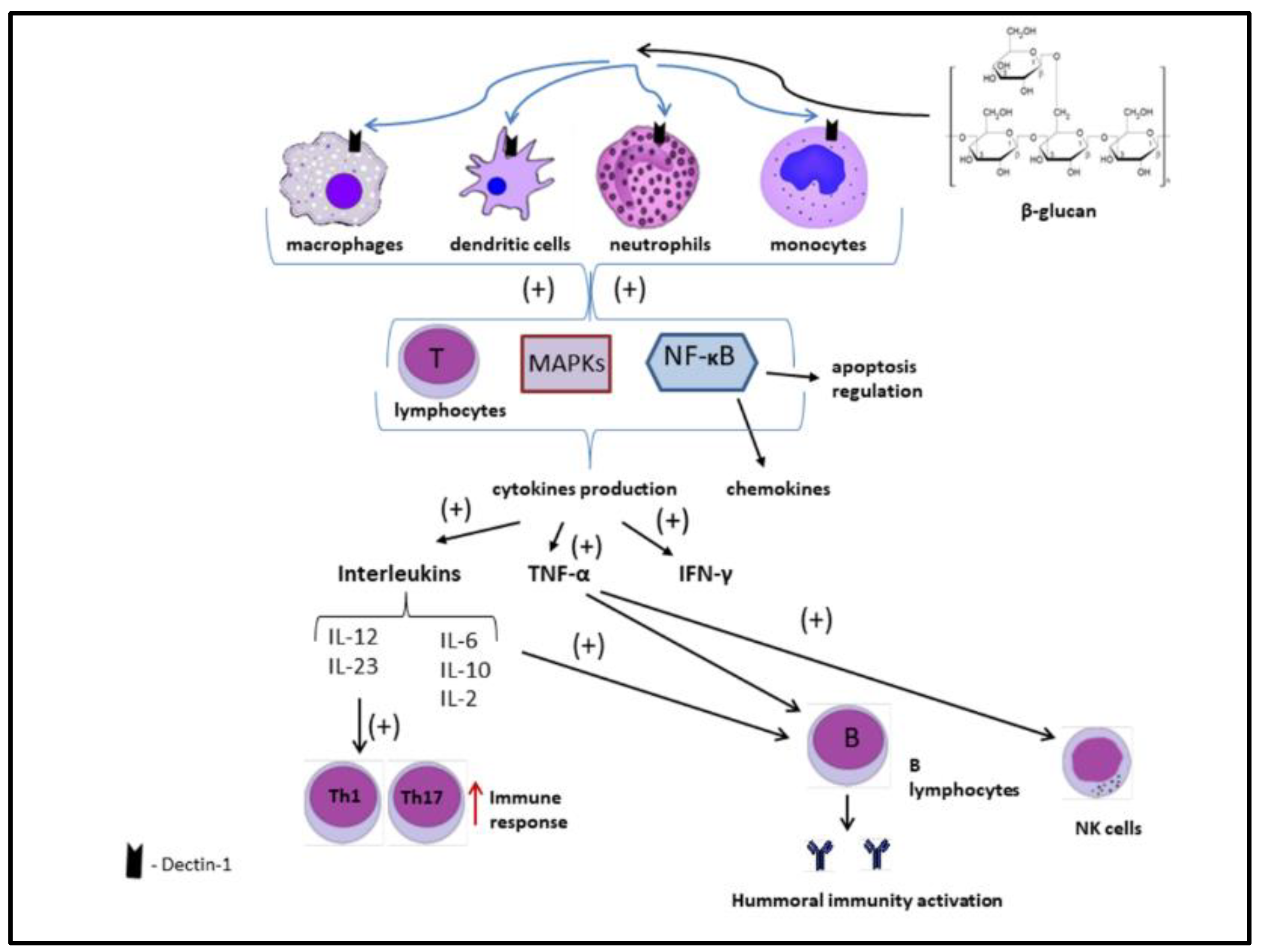
2.3.1. Immunomodulatory Properties of Beta-Glucans
2.3.2. Antitumor and Cytotoxic Properties of Beta-Glucans
2.3.3. Anti-Inflammatory Function of Beta-Glucans
2.3.4. Antioxidant Properties of Beta-Glucans
2.3.5. Beta-Glucans in the Treatment of Allergies
2.3.6. Antibacterial, Antiviral, and Antifungal Properties of Mushroom Beta-Glucans
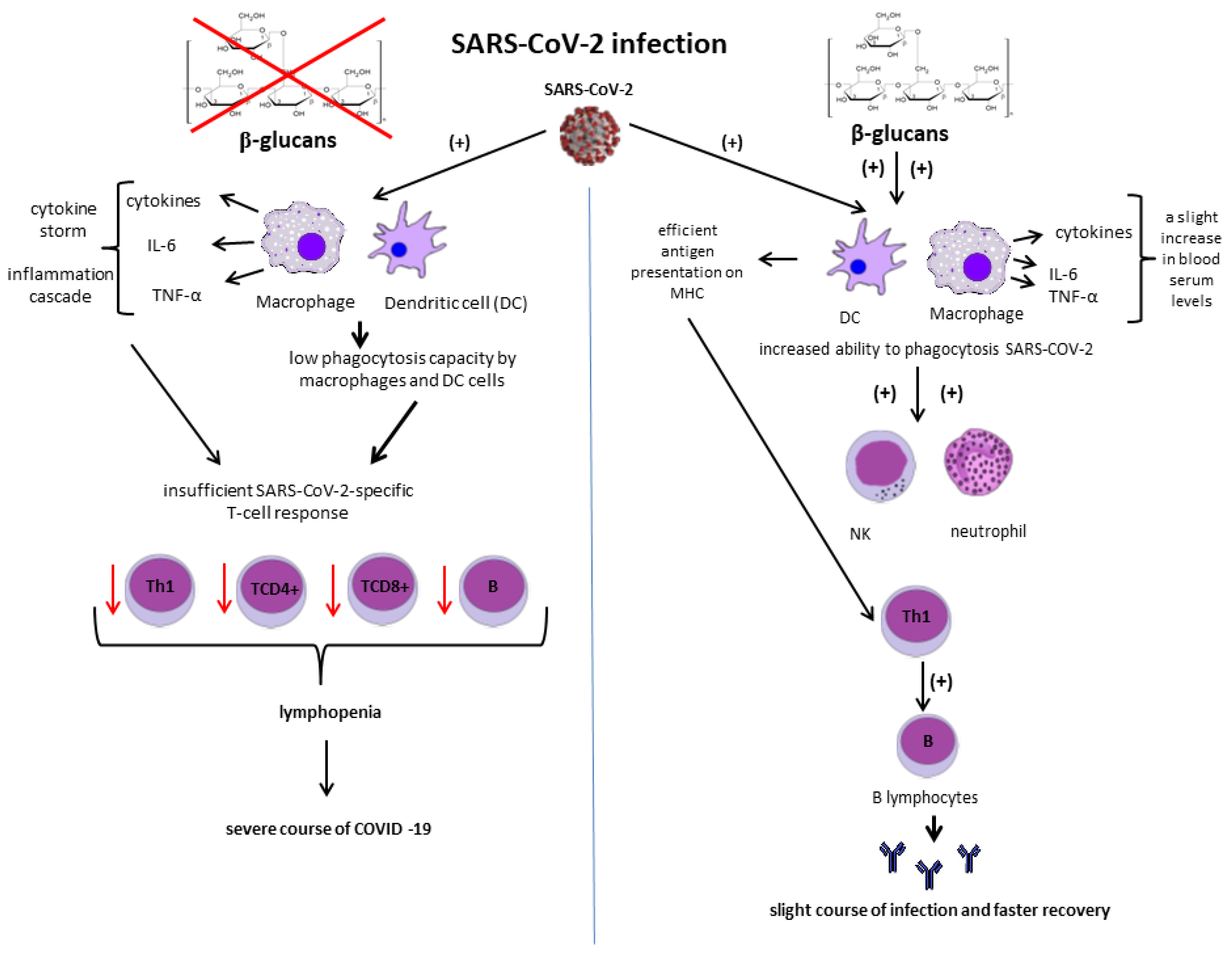
2.3.7. Potential Role for Beta-Glucans in Decreasing Morbidity and Mortality Due to COVID-19
2.3.8. Mushroom Beta-Glucans as Prebiotics and Microbiota Modulators
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peintner, U.; Pöder, R. Ethnomycological remarks on the Iceman’s fungi. In The Iceman and His Natural Environment; Bortenschlager, S., Oeggl, K., Eds.; Springer: Vienna, Austria, 2000; Volume 4, Chapter 12; pp. 143–150. [Google Scholar]
- González, A.; Nobre, C.; Simões, L.S.; Cruz, M.; Loredo, A.; Rodríguez-Jasso, R.M.; Contreras, J.; Texeira, J.; Belmares, R. Evaluation of functional and nutritional potential of a protein concentrate from Pleurotus ostreatus mushroom. Food Chem. 2021, 346, 128884. [Google Scholar] [CrossRef]
- Witkowska, A.M.; Zujko, M.E.; Mirończuk-Chodakowska, I. Comparative study of wild edible mushrooms as sources of antioxidants. Int. J. Med. Mushroom 2011, 13, 335–341. [Google Scholar] [CrossRef]
- Tran, H.B.; Yamamoto, A.; Matsumoto, S.; Ito, H.; Igami, K.; Miyazaki, T.; Kondo, R.; Shimizu, K. Hypotensive effects and angiotensin-converting enzyme inhibitory peptides of reishi (Ganoderma lingzhi) auto-digested extract. Molecules 2014, 19, 13473–13485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Li, J.; Wang, J.; Song, X.; Zhang, J.; Wu, S.; Hu, C.; Gong, Z.; Jia, L. Antihyperlipidaemic and hepatoprotective activities of acidic and enzymatic hydrolysis exopolysaccharides from Pleurotus eryngii SI-04. BMC Complement. Altern. Med. 2017, 17, 403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Wang, C.; Li, J.; Mei, Y.; Liang, Y. Hypoglycemic and Hypolipidemic Effects of Phellinus Linteus Mycelial Extract from Solid-State Culture in A Rat Model of Type 2 Diabetes. Nutrients 2019, 11, 296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, H.C.; Liu, Y.W.; Lum, C.C.; Hsu, K.D.; Lin, S.P.; Hsieh, C.W.; Lin, H.W.; Lu, T.Y.; Cheng, K.C. Ganoderma formosanum exopolysaccharides inhibit tumor growth via immunomodulation. Int. J. Mol. Sci. 2021, 22, 11251. [Google Scholar] [CrossRef]
- Ahmad, M.F.; Ahmad, F.A.; Khan, M.I.; Alsayegh, A.A.; Wahab, S.; Alam, M.I.; Ahmed, F. Ganoderma lucidum: A potential source to surmount viral infections through β-glucans immunomodulatory and triterpenoids antiviral properties. Int. J. Biol. Macromol. 2021, 187, 769–779. [Google Scholar] [CrossRef]
- Moorlag, S.J.C.F.M.; Khan, N.; Novakovic, B.; Kaufmann, E.; Jansen, T.; van Crevel, R.; Divangahi, M.; Netea, M.G. β-Glucan Induces Protective Trained Immunity against Mycobacterium tuberculosis Infection: A Key Role for IL-1. Cell Rep. 2020, 31, 107634. [Google Scholar] [CrossRef]
- Afiukwa, C.A.; Ugwu, O.P.C.; Okoli, S.O.; Idenyi, J.N.; Ossai, E.C. Contents of some vitamins in five edible mushroom verities consumed in Abakaliki Metropolis, Nigeria. Res. J. Pharm. Biol. Chem. Sci. 2013, 4, 805–812. [Google Scholar]
- Dubost, N.J.; Ou, B.; Beeleman, R.B. Quantification of polyphenols and ergothioneine in cultivated mushrooms and correlation to total antioxidant capacity. Food Chem. 2007, 105, 727–735. [Google Scholar] [CrossRef]
- Zięba, P.; Sękara, A.; Bernaś, E.; Krakowska, A.; Sułkowska-Ziaja, K.; Kunicki, E.; Suchanek, M.; Muszyńska, B. Supplementation with magnesium salts-a strategy to increase nutraceutical value of Pleurotus djamor fruiting bodies. Molecules 2021, 26, 3273. [Google Scholar] [CrossRef] [PubMed]
- Cerletti, C.; Esposito, S.; Iacoviello, L. Edible mushrooms and beta-glucans: Impact on human health. Nutrients 2021, 13, 2195. [Google Scholar] [CrossRef]
- Murphy, E.J.; Masterson, C.; Rezoagli, E.; O’Toole, D.; Major, I.; Stack, G.D.; Lynch, M.; Laffey, J.G.; Rowan, N.J. β-Glucan extracts from the same edible shiitake mushroom Lentinus edodes produce differential. In-Vitro immunomodulatory and pulmonary cytoprotective effects- Implications for coronavirus disease (COVID-19) immunotherapies. Sci. Total Environ. 2020, 732, 139330. [Google Scholar] [CrossRef] [PubMed]
- Singdevsachan, S.K.; Mishra, P.A.J.; Baliyarsingh, B.; Tayung, K.; Thatoi, H. Mushroom polysaccharides as potential prebiotics with their antitumor and immunomodulating properties: A review. Bioact. Carbohydr. Diet. Fibre 2016, 7, 1–14. [Google Scholar] [CrossRef]
- He, X.; Wang, X.; Fang, J.; Chang, Y.; Ning, N.; Guo, H.; Huang, L.; Huang, X.; Zhao, Z. Structures, biological activities, and industrial applications of the polysaccharides from Hericium erinaceus (Lion’s Mane) mushroom: A review. Int. J. Biol. Macromol. 2017, 97, 228–237. [Google Scholar] [CrossRef]
- Ruthes, A.C.; Smiderle, F.R.; Iacomini, M. Mushrooms heteropolysaccharides. A review on their sources, structure and biological effects. Carbohydr. Polym. 2016, 136, 358–375. [Google Scholar] [CrossRef] [PubMed]
- Nitschke, J.; Modick, H.; Busch, E.; von Rekowski, R.W.; Altenbach, H.J.; Mölleken, H. A new colorimetric method to quantify β-1,3-1,6-glucans in comparison with total β-1,3-glucans in edible mushrooms. Food Chem. 2011, 127, 791–796. [Google Scholar] [CrossRef]
- Ruthes, A.C.; Smiderle, F.R.; Iacomini, M. D-Glucans from edible mushrooms: A review on the extraction, purification and chemical characterization approaches. Carbohydr. Polym. 2015, 117, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Zhou, W.; Zhang, Y. Fungal polysaccharides. Adv. Pharmacol. 2020, 87, 277–299. [Google Scholar] [CrossRef]
- Synytsya, A.; Míčková, K.; Synytsya, A.; Jablonský, I.; Spěváček, J.; Erban, V.; Kováříková, E.; Čopíková, J. Glucans from fruit bodies of cultivated mushrooms Pleurotus ostreatus and Pleurotus eryngii: Structure and potential prebiotics activity. Carbohydr. Polym. 2009, 76, 548–556. [Google Scholar] [CrossRef]
- Synytsya, A.; Novak, M. Structural analysis of glucans. Ann. Transl. Med. 2014, 2, 17. [Google Scholar] [CrossRef]
- Sheng, K.; Wang, C.; Chen, B.; Kang, M.; Wang, M.; Liu, K.; Wang, M. Recent advances in polysaccharides from Lentinus edodes (Berk.): Isolation, structures and bioactivities. Food Chem. 2021, 358, 129883. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, N.; Banerjee, A.; Sarkar, A.; Ghosh, S.; Acharya, K. Mushroom Polysaccharides: A Potent Immune-Modulator. Biointerface Res. Appl. Chem. 2021, 11, 8915–8930. [Google Scholar] [CrossRef]
- Kaur, R.; Sharma, M.; Ji, D.; Xu, M.; Agyei, D. Structural Features, Modification, and Functionalities of Beta-Glucan. Fibers 2020, 8, 1. [Google Scholar] [CrossRef] [Green Version]
- Villares, A.; Mateo-Vivaracho, L.; Guillamon, E. Structural features and healthy properties of polysaccharides occurring in mushrooms. Agriculture 2012, 2, 452–471. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Sathuvan, M.; Zhang, X.; Zhang, W.; Tang, S.; Liu, Y.; Cheong, K.L. Characterization of polysaccharides from different species of brown seaweed using saccharide mapping and chromatographic analysis. BMC Chem. 2021, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xiong, Y.; Bakry, A.M.; Xiong, S.; Yin, T.; Zhang, B.; Huang, J.; Liu, Z.; Huang, Q. Effect of yeast β-glucan on gel properties, spatial structure and sensory characteristics of silver carp surimi. Food Hydrocol. 2019, 88, 256–264. [Google Scholar] [CrossRef]
- Meng, Y.; Lyu, F.; Xu, X. Zhang, L. Recent advances in chain conformation and bioactivities of triple-helix polysaccharides. Biomacromolecules 2020, 21, 1653–1677. [Google Scholar] [CrossRef]
- Wang, Q.; Ellis, P.R. Oat β-glucan: Physico-chemical characteristics in relation to its blood-glucose and cholesterol-lowering properties. Br. J. Nutr. 2014, 112, S4–S13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henrion, M.; Francey, C.; Lê, K.A.; Lamothe, L. Cereal B-Glucans: The Impact of Processing and How It Affects Physiological Responses. Nutrients 2019, 11, 1729. [Google Scholar] [CrossRef] [Green Version]
- Mathews, R.; Kamil, A.; Chu, Y. Global review of heart health claims for oat beta-glucan products. Nutrition 2021, 78, 78–97. [Google Scholar] [CrossRef] [PubMed]
- Boutros, J.A.; Magee, A.S.; Cox, D. Comparison of structural differences between yeast β-glucan sourced from different strains of saccharomyces cerevisiae and processed using proprietary manufacturing processes. Food Chem. 2022, 367, 130708. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Meenu, M.; Liu, H.; Xu, B.A. Concise Review on the Molecular Structure and Function Relationship of β-Glucan. Int. J. Mol. Sci. 2019, 20, 4032. [Google Scholar] [CrossRef] [Green Version]
- Mirończuk-Chodakowska, I.; Witkowska, A.M. Evaluation of Polish wild Mushrooms as Beta-Glucan Sources. Int. J. Environ. Res. Public. Health 2020, 17, 7299. [Google Scholar] [CrossRef]
- Lee, Y.T.; Kim, Y.S. Water-solubility of β-glucans in various edible mushrooms-research note. J. Food Sci. Nutr. 2005, 10, 294–297. [Google Scholar] [CrossRef]
- Ina, K.; Kataoka, T.; Ando, T. The use of lentinan for treating gastric cancer. Anticancer Agents Med. Chem. 2013, 13, 681–688. [Google Scholar] [CrossRef] [Green Version]
- Cognigni, V.; Ranallo, N.; Tronconi, F.; Morgese, F.; Berardi, R. Potential benefit of β-glucans as adjuvant therapy in immuno-oncology: A review. Explor. Target. Antitumor Ther. 2021, 2, 122–138. [Google Scholar] [CrossRef]
- Benson, K.F.; Stamets, P.; Davis, R.; Nally, R.; Taylor, A.; Slater, S.; Jensen, G.S. The mycelium of the Trametes versicolor (Turkey tail) mushroom and its fermented substrate each show potent and complementary immune activating properties in vitro. BMC Complement. Altern. Med. 2019, 19, 342. [Google Scholar] [CrossRef] [Green Version]
- Maehara, Y.; Tsujitani, S.; Saeki, H.; Oki, E.; Yoshinaga, K.; Emi, Y.; Morita, M.; Kohnoe, S.; Kakeji, Y.; Yano, T.; et al. Biological mechanism and clinical effect of protein-bound polysaccharide K (KRESTIN®): Review of development and future perspectives. Surg. Today 2012, 42, 8–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grzybek, J.; Zgórniak-Nowosielska, I.; Kohlmünzer, S. Antitumor and Cytotoxic Activity of Tylopilan, a Fungal Polysaccharide from Tylopilus felleus Fruit Bodies. Planta Med. 1990, 56, 670–671. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, S.; Wang, X.; Zhang, L.; Cheung, P.C.K. Advances in lentinan: Isolation, structure, chain conformation and bioactivities. Food Hydrocol. 2011, 25, 196–206. [Google Scholar] [CrossRef]
- Karácsonyi, Š.; Kuniak, Ľ. Polysaccharides of Pleurotus ostreatus: Isolation and structure of pleuran, an alkali-insoluble β-d-glucan. Carbohydr. Polym. 1994, 24, 107–111. [Google Scholar] [CrossRef]
- Zhang, Y.; Kong, H.; Fang, Y.; Nishinari, K.; Phillips, G.O. Schizophyllan: A review on its structure, properties, bioactivities and recent developments. Bioact. Carbohydr. Diet. Fibre 2013, 1, 53–71. [Google Scholar] [CrossRef]
- Masuda, Y.; Murata, Y.; Hayashi, M.; Nanba, H. Inhibitory effect of MD-Fraction on tumor metastasis: Involvement of NK cell activation and suppression of intercellular adhesion molecule (ICAM)-1 expression in lung vascular endothelial cells. Biol. Pharm. Bull. 2008, 31, 1104–1108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.Y.; Siu, K.C.; Geng, P. Bioactive Ingredients and Medicinal Values of Grifola frondosa (Maitake). Foods 2021, 10, 95. [Google Scholar] [CrossRef]
- Coviello, T.; Palleschi, A.; Grassi, M.; Matricardi, P.; Bocchinfuso, G.; Alhaique, F. Scleroglucan: A Versatile Polysaccharide for Modified Drug Delivery. Molecules 2005, 10, 6–33. [Google Scholar] [CrossRef] [Green Version]
- Chihara, G.; Hamuro, J.; Maeda, Y.; Arai, Y.; Fukuoka, F. Fractionation and purification of the polysaccharides with marked antitumor activity, especially lentinan, from Lentinus edodes (Berk.) Sing. (an edible mushroom). Cancer Res. 1970, 30, 2776–2781. [Google Scholar]
- Giavasis, I. Bioactive fungal polysaccharides as potential functional ingredients in food and nutraceuticals. Curr. Opin. Biotechnol. 2014, 26, 162–173. [Google Scholar] [CrossRef]
- Wu, X.; Zheng, Z.; Guo, T.; Wang, K.; Zhang, Y. Molecular dynamics simulation of lentinan and its interaction with the innate receptor dectin-1. Int. J. Biol. Macromol. 2021, 171, 527–538. [Google Scholar] [CrossRef]
- Wang, K.P.; Zhang, Q.L.; Liu, Y.; Wang, J.; Cheng, Y.; Zhang, Y. Structure and inducing tumor cell apoptosis activity of polysaccharides isolated from Lentinus edodes. J. Agric. Food Chem. 2013, 61, 9849–9858. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Zhou, Y.; Zheng, Z.; Tang, W.; Song, M.; Wang, J.; Wang, K. Lentinan inhibited colon cancer growth by inducing endoplasmic reticulum stress-mediated autophagic cell death and apoptosis. Carbohydr. Polym. 2021, 267, 118154. [Google Scholar] [CrossRef]
- Zi, Y.; Jiang, B.; He, C.; Liu, L. Lentinan inhibits oxidative stress and inflammatory cytokine production induced by benzo(a)pyrene in human keratinocytes. J. Cosmet. Dermatol. 2020, 19, 502–507. [Google Scholar] [CrossRef]
- Morales, D.; Rutckeviski, R.; Villalva, M.; Abreu, H.; Soler-Rivas, C.; Santoyo, S.; Iacomini, M.; Smiderle, F.R. Isolation and comparison of α- and β-D-glucans from shiitake mushrooms (Lentinula edodes) with different biological activities. Carbohydr. Polym. 2020, 229, 115521. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Jiang, P.; Zhao, J.; Shi, H.; Zhang, P.; Yang, X.; Biazik, J.; Hu, M.; Hua, H.; Ge, X.; et al. β-Glucan from Lentinula edodes prevents cognitive impairments in high-fat diet-induced obese mice: Involvement of colon-brain axis. J. Transl. Med. 2021, 19, 54. [Google Scholar] [CrossRef] [PubMed]
- Kikumoto, S.; Miyajima, T.; Yoshizumi, S.; Fujimoto, S.; Kimura, K. Polysaccharide produced by Schizophyllum commune. 1. Formation and some properties of an extracellular polysaccharide. J. Agric. Chem. Soc. Jpn. 1970, 44, 337–342. [Google Scholar]
- Sutivisedsak, N.; Leathers, T.D.; Price, N.P.J. Production of schizophyllan from distiller’s dried grains with solubles by diverse strains of Schizophyllum commune. Springer Plus 2013, 2, 476. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Yin, C.; Fan, X.; Ma, K.; Yao, F.; Zhou, R.; Shi, D.; Cheng, W.; Gao, H. Characterization of physicochemical and biological properties of Schizophyllum commune polysaccharide extracted with different methods. Int. J. Biol. Macromol. 2020, 156, 1425–1434. [Google Scholar] [CrossRef]
- Thongsiri, C.; Nagai-Yoshioka, Y.; Yamasaki, R.; Adachi, Y.; Usui, M.; Nakashima, K.; Nishihara, T.; Ariyoshi, W. Schizophyllum commune β-glucan: Effect on interleukin-10 expression induced by lipopolysaccharide from periodontopathic bacteria. Carbohydr. Polym. 2021, 253, 117285. [Google Scholar] [CrossRef] [PubMed]
- Lemieszek, M.; Rzeski, W. Anticancer properties of polysaccharides isolated from fungi of the Basidiomycetes class. Contemp. Oncol. 2012, 16, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Gani, A.; Khanday, F.A.; Masoodi, F.A. Biological and pharmaceutical activities of mushroom β-glucan discussed as a potential functional food ingredient. Bioact. Carbohydr. Diet. Fibre 2018, 16, 1–13. [Google Scholar] [CrossRef]
- Yamashita, K.; Ougolkov, A.V.; Nakazato, H.; Ito, K.; Ohashi, Y.; Kitakata, H.; Yasumoto, K.; Omote, K.; Mai, M.; Takahashi, Y.; et al. Adjuvant immunochemotherapy with protein-bound polysaccharide K for colon cancer in relation to oncogenic beta-catenin activation. Dis. Colon. Rectum. 2007, 50, 1169–1181. [Google Scholar] [CrossRef]
- Chang, Y.; Zhang, M.; Jiang, Y.; Liu, Y.; Luo, H.; Hao, C.; Zeng, P.; Zhang, L. Preclinical and clinical studies of Coriolus versicolor polysaccharopeptide as an immunotherapeutic in China. Discov. Med. 2017, 23, 207–219. [Google Scholar]
- Masuda, Y.; Inoue, H.; Ohta, H.; Miyake, A.; Konishi, M.; Nanba, H. Oral administration of soluble β-glucans extracted from Grifola frondosa induces systemic antitumor immune response and decreases immunosuppression in tumor-bearing mice. Int. J. Cancer 2013, 133, 108–119. [Google Scholar] [CrossRef]
- Mao, G.H.; Ren, Y.; Feng, W.W.; Li, Q.; Wu, H.Y.; Jin, D.; Zhao, T.; Xu, C.Q.; Yang, L.Q.; Wu, X.Y. Antitumor and immunomodulatory activity of a water-soluble polysaccharide from Grifola frondosa. Carbohydr. Polym. 2015, 134, 406–412. [Google Scholar] [CrossRef]
- Chen, Y.H.; Lee, C.H.; Hsu, T.H.; Lo, H.C. Submerged-Culture Mycelia and Broth of the Maitake Medicinal Mushroom Grifola frondosa (Higher Basidiomycetes) Alleviate Type 2 Diabetes-Induced Alterations in Immunocytic Function. Int. J. Med. Mushrooms 2015, 17, 541–556. [Google Scholar] [CrossRef]
- Seo, Y.R.; Patel, D.K.; Shin, W.C.; Sim, W.S.; Lee, O.H.; Lim, K.T. Structural Elucidation and Immune-Enhancing Effects of Novel Polysaccharide from Grifola frondosa. Biomed. Res. Int. 2019, 2019, 7528609. [Google Scholar] [CrossRef] [PubMed]
- Jesenak, M.; Majtan, J.; Rennerova, Z.; Kyselovic, J.; Banovcin, P.; Hrubisko, M. Immunomodulatory effect of pleuran (β-glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Int. Immunopharmacol. 2013, 15, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J. Pleuran (β-glucan from Pleurotus ostreatus): An effective nutritional supplement against upper respiratory tract infections? Med. Sport Sci. 2013, 59, 57–61. [Google Scholar] [CrossRef]
- Urbancikova, I.; Hudackova, D.; Majtan, J.; Rennerova, Z.; Banovcin, P.; Jesenak, M. Efficacy of Pleuran (β-Glucan from Pleurotus ostreatus) in the Management of Herpes Simplex Virus Type 1 Infection. Evid. Based. Complement. Altern. Med. 2020, 2020, 8562309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Cui, S.W.; Cheung, P.C.K.; Wang, Q. Antitumor polysaccharides from mushrooms: A review on their isolation process, structural characteristic and antitumor activity. Trends Food Sci. Technol. 2007, 18, 4–19. [Google Scholar] [CrossRef]
- Ping, Z.; Xu, H.; Liu, T.; Huang, J.; Meng, Y.; Xu, X.; Li, W.; Zhang, L. Anti-hepatoma activity of the stiff branched β-d-glucan and effects of molecular weight. J. Mater. Chem. B 2016, 4, 4565–4573. [Google Scholar] [CrossRef]
- Chan, G.C.; Chan, W.K.; Sze, D.M. The effects of beta-glucan on human immune and cancer cells. J. Hematol. Oncol. 2009, 2, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roupas, P.; Keogh, J.; Noakes, M.; Margetts, C.; Taylor, P. The role of edible mushrooms in health. Evaluation of the evidence. J. Funct. Foods 2012, 4, 687–709. [Google Scholar] [CrossRef]
- Gaullier, J.M.; Sleboda, J.; Øfjord, E.S.; Ulvestad, E.; Nurminiemi, M.; Moe, C.; Tor, A.; Gudmundsen, O. Supplementation with a soluble β-glucan exported from Shiitake medicinal mushroom, Lentinus edodes (Berk.) singer mycelium: A crossover, placebo-controlled study in healthy elderly. Int. J. Med. Mushrooms 2011, 13, 319–326. [Google Scholar] [CrossRef] [Green Version]
- Motta, F.; Gershwin, M.E.; Selmi, C. Mushrooms and immunity. J. Autoimmun. 2021, 117, 102576. [Google Scholar] [CrossRef]
- Herre, J.; Gordon, S.; Brown, G.D. Dectin-1 and its role in the recognition of β-glucans by macrophages. Mol. Immunol. 2004, 40, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Marakalala, M.J.; Williams, D.L.; Hoving, J.C.; Engstad, R.; Netea, M.G.; Brown, G.D. Dectin-1 plays a redundant role in the immunomodulatory activities of B-glucan-rich ligands in vivo. Microbes Infect. 2013, 15, 511–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vidya, M.K.; Kumar, V.G.; Sejian, V.; Bagath, M.; Krishnan, G.; Bhatta, R. Toll-like receptors: Significance, ligands, signaling pathways, and functions in mammals. Int. Rev. Immunol. 2018, 37, 20–36. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Toll-like Receptors and Their Crosstalk with Other Innate Receptors in Infection and Immunity. Immunity 2011, 34, 637–650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Seviour, R. Medical importance of fungal β-(1-3), (1-6)-glucans. Mycol. Res. 2007, 111, 635–652. [Google Scholar] [CrossRef]
- Grassin-Delyle, S.; Abrial, C.; Salvator, H.; Brollo, M.; Naline, E.; Devillier, P. The Role of Toll-Like Receptors in the Production of Cytokines by Human Lung Macrophages. J. Innate Immun. 2020, 12, 63–73. [Google Scholar] [CrossRef]
- Lukácsi, S.; Nagy-Baló, Z.; Erdei, A.; Sándor, N.; Bajtay, Z. The role of CR3 (CD11b/CD18) and CR4 (CD11c/CD18) in complement-mediated phagocytosis and podosome formation by human phagocytes. Immunol. Lett. 2017, 189, 64–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.S.; Hong, J.T.; Kim, Y.; Han, S.B. Stimulatory Effect of β-glucans on immune cells. Immune Netw. 2011, 11, 191–195. [Google Scholar] [CrossRef] [Green Version]
- Vetvicka, V.; Thornton, B.P.; Wieman, T.J.; Ross, G.D. Targeting of natural killer cells to mammary carcinoma via naturally occurring tumor cell-bound iC3b and beta-glucan-primed CR3 (CD11b/CD18). J. Immunol. 1997, 159, 599–605. [Google Scholar] [PubMed]
- Zani, I.A.; Stephen, S.L.; Mughal, N.A.; Russell, D.; Homer-Vanniasinkam, S.; Wheatcroft, S.B.; Ponnambalam, S. Scavenger Receptor Structure and Function in Health and Disease. Cells 2015, 4, 178–201. [Google Scholar] [CrossRef] [Green Version]
- Rice, P.J.; Kelley, J.L.; Kogan, G.; Ensley, H.E.; Kalbfleisch, J.H.; Browder, I.W.; Williams, D.L. Human monocytes scavenger receptors are pattern recognition receptors for (1-3)-β-D-glucans. J. Leukoc. Biol. 2002, 72, 140–146. [Google Scholar] [PubMed]
- Batbayar, S.; Lee, D.H.; Kim, H.W. Immunomodulation of fungal β-glucan in host defense signaling by dectin-1. Biomol. Ther. 2012, 20, 433–445. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, S.; Balram, A.; Li, W. Convergence: Lactosylceramide-Centric Signaling Pathways Induce Inflammation, Oxidative Stress, and Other Phenotypic Outcomes. Int. J. Mol. Sci. 2021, 22, 1816. [Google Scholar] [CrossRef]
- Nguyen, G.T.; Green, E.R.; Mecsas, J. Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance. Front. Cell Infect. Microbiol. 2017, 7, 373. [Google Scholar] [CrossRef]
- Wu, Y.L.; Han, F.; Luan, S.S.; Ai, R.; Zhang, P.; Li, H.; Chen, L.X. Triterpenoids from Ganoderma lucidum and Their Potential Anti-inflammatory Effects. J. Agric. Food Chem. 2019, 67, 5147–5158. [Google Scholar] [CrossRef]
- Schwartzand, B.; Hadar, Y. Possible mechanisms of action of mushroom-derived glucans on inflammatory bowel disease and associated cancer. Ann. Transl. Med. 2014, 2, 19. [Google Scholar] [CrossRef]
- Bergendiova, K.; Tibenska, E.; Majtan, J. Pleuran (β-glucan from Pleurotus ostreatus) supplementation, cellular immune response and respiratory tract infections in athletes. Eur. J. Appl. Physiol. 2011, 111, 2033–2040. [Google Scholar] [CrossRef]
- Mizuno, M.; Minato, K.; Kawakami, S.; Tatsuoka, S.; Denpo, Y.; Tsuchida, H. Contents of anti-tumor polysaccharides in certain mushrooms and their immunomodulating activities. Food Sci. Technol. Res. 2001, 7, 31–34. [Google Scholar] [CrossRef]
- Pires, A.R.A.; Ruthes, A.C.; Cadena, S.M.S.C.; Acco, A.; Gorin, P.A.J.; Iacomini, M. Cytotoxic effect of Agaricus bisporus and Lactarius rufus β-D-glucans on HepG2 cells. Int. J. Biol. Macromol. 2013, 58, 95–103. [Google Scholar] [CrossRef] [Green Version]
- Mizuno, T. The Extraction and Development of Antitumor-Active Polysaccharides from Medicinal Mushrooms in Japan (Review). Int. J. Med. Mushrooms 1999, 1, 9–29. [Google Scholar] [CrossRef]
- Higashi, D.; Seki, K.; Ishibashi, Y.; Egawa, Y.; Koga, M.; Sasaki, T.; Hirano, K.; Mikami, K.; Futami, K.; Maekawa, T.; et al. The effect of lentinan combination therapy for unresectable advanced gastric cancer. Anticancer Res. 2012, 32, 2365–2368. [Google Scholar]
- Sasaki, S.; Izumi, H.; Morimoto, Y.; Sakurai, K.; Mochizuki, S. Induction of potent cell growth inhibition by schizophyllan/K-ras antisense complex in combination with gemcitabine. Bioorg. Med. Chem. 2020, 28, 115668. [Google Scholar] [CrossRef]
- Yamasaki, A.; Onishi, H.; Imaizumi, A.; Kawamoto, M.; Fujimura, A.; Oyama, Y.; Katano, M. Protein-bound Polysaccharide-K Inhibits Hedgehog Signaling Through Down-regulation of MAML3 and RBPJ Transcription Under Hypoxia, Suppressing the Malignant Phenotype in Pancreatic Cancer. Anticancer Res. 2016, 36, 3945–3952. [Google Scholar]
- Cheung, N.K.; Modak, S.; Vickers, A.; Knuckles, B. Orally administered beta-glucans enhance anti-tumor effects of monoclonal antibodies. Cancer Immunol. Immunother. 2002, 51, 557–564. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, X.D.; Jiang, Z. The application of fungal β-glucans for the treatment of colon cancer. Anticancer Agents Med. Chem. 2013, 13, 725–730. [Google Scholar] [CrossRef] [Green Version]
- Lavi, I.; Nimri, L.; Levinson, D.; Peri, I.; Hadar, Y.; Schwartz, B. Glucans from the edible mushroom Pleurotus pulmonarius inhibit colitis-associated colon carcinogenesis in mice. J. Gastroenterol. 2012, 47, 504–518. [Google Scholar] [CrossRef] [PubMed]
- Turunen, K.T.; Pletsa, V.; Georgiadis, P.; Triantafillidis, J.K.; Karamanolis, D.; Kyriacou, A. Impact of β-glucan on the Fecal Water Genotoxicity of Polypectomized Patients. Nutr. Cancer 2016, 68, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Combination of glucan, resveratrol and vitamin C demonstrates strong anti-tumor potential. Anticancer Res. 2012, 32, 81–87. [Google Scholar]
- Fujimoto, S.; Furue, H.; Kimura, T.; Kondo, T.; Orita, K.; Taguchi, T.; Yoshida, K.; Ogawa, N. Clinical outcome of postoperative adjuvant immunochemotherapy with sizofiran for patients with resectable gastric cancer: A randomised controlled study. Eur. J. Cancer 1991, 27, 1114–1118. [Google Scholar] [CrossRef]
- Chaichian, S.; Moazzami, B.; Sadoughi, F.; Kashani, H.H.; Zaroudi, M.; Asemi, Z. Functional activities of beta-glucans in the prevention or treatment of cervical cancer. J. Ovarian Res. 2020, 13, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roopngam, P.E. Increased Response of Human T-Lymphocytes by Dendritic Cells Pulsed with HPV16E7 and Pleurotus sajor-caju-β-glucan (PBG). Iran. J. Immunol. 2018, 15, 246–255. [Google Scholar] [CrossRef]
- Steimbach, L.; Borgmann, A.V.; Gomar, G.G.; Hoffmann, L.V.; Rutckeviski, R.; de Andrade, D.P.; Smiderle, F.R. Fungal beta-glucans as adjuvants for treating cancer patients–A systematic review of clinical trials. Clin. Nutr. 2021, 40, 3104–3113. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ma, L.; Zhang, L. Molecular basis for Poria cocos mushroom polysaccharide used as an antitumor drug in China. Prog. Mol. Biol. Transl. Sci. 2019, 163, 263–296. [Google Scholar] [CrossRef]
- Vetvicka, V.; Teplyakova, T.V.; Shintyapina, A.B.; Korolenko, T.A. Effects of medicinal fungi-derived β-glucan on tumor progression. J. Fungi 2021, 7, 250. [Google Scholar] [CrossRef]
- Carbonero, E.R.; Gracher, A.H.P.; Komura, D.L.; Marcon, R.; Freitas, C.S.; Baggio, C.H.; Santos, A.R.S.; Torri, G.; Gorin, P.A.J.; Iacomini, M. Lentinus edodes heteroglucan: Antinociceptive and anti-inflamatory effects. Food Chem. 2008, 111, 531–537. [Google Scholar] [CrossRef]
- Komura, D.L.; Carbonero, E.R.; Gracher, A.H.P.; Baggio, C.H.; Freitas, C.S.; Marcon, R.; Santos, A.R.S.; Gorin, P.A.J.; Iacomini, M. Structure of Agaricus spp. fucogalactans and their anti-inflamatory and aninociceptive properties. Bioresour. Technol. 2010, 101, 6192–6199. [Google Scholar] [CrossRef] [PubMed]
- Ruthes, A.C.; Carbonero, E.R.; Córodva, M.M.; Baggio, C.H.; Santos, A.R.S.; Sassaki, G.L.; Cipriani, T.R.; Gorin, P.A.J.; Iacomini, M. Lactarius rufus (1-3),(1-6)-β-D-glucans: Structure, antinociceptive and anti-inflammatory effects. Carbohydr. Polym. 2013, 94, 129–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smiderle, F.R.; Olsen, L.M.; Carbonero, E.R.; Beggio, C.H.; Freitas, C.S.; Marcon, R.; Santos, A.R.S.; Gorin, P.A.J.; Iacomini, M. Anti-inflammatory and analgesic properties in a rodent model of a (1-3),(1-6)-linked beta-glucan isolated from Pleurotus pulmonarius. Eur. J. Pharmacol. 2008, 597, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Lin, C.; Bian, Z.; Xu, B. An insight into anti-inflammatory effects of fungal beta-glucans. Trends Food Sci. Technol. 2015, 41, 49–59. [Google Scholar] [CrossRef]
- Liu, D.Z.; Liang, H.J.; Chen, C.H.; Su, C.H.; Lee, T.H.; Huang, C.T.; Hou, W.C.; Lin, S.Y.; Zhong, W.B.; Lin, P.J.; et al. Comparative anti-infalmmatory characterization of wild fruiting body, liquid-state fermentation, and solid-state culture of Taiwanofungus camhoratus in microglia and the mechanism of its action. J. Ethnopharmacol. 2007, 113, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Padilha, M.M.; Avila, A.A.L.; Sousa, P.J.C.; Cardoso, L.G.V.; Perazzo, F.F.; Carvalho, J.C.T. Anti-inflammatory activity of aqueous and alkaline extracts from mushrooms (Agaricus blazei Murill). J. Med. Food. 2009, 12, 359–364. [Google Scholar] [CrossRef]
- Piskov, S.; Timchenko, L.; Grimm, W.D.; Rzhepakovsky, I.; Avanesyan, S.; Sizonenko, M.; Kurchenko, V. Effects of Various Drying Methods on Some Physico-Chemical Properties and the Antioxidant Profile and ACE Inhibition Activity of Oyster Mushrooms (Pleurotus ostreatus). Foods 2020, 9, 160. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Dai, L.; Kong, X.; Chen, L. Characterization and in vitro antioxidant activities of polysaccharides from Pleurotus ostreatus. Int. J. Biol. Macromol. 2012, 51, 259–265. [Google Scholar] [CrossRef]
- Telles, C.B.S.; Sabry, D.A.; Alimeda-Lima, J.; Costa, M.S.S.P.; Melo-Silveira, R.F.; Trindade, E.S.; Sassaki, G.L.; Wisbeck, E.; Furlan, S.A.; Leite, E.L.; et al. Sulfation of the extracellular polysaccharide produced by the edible mushroom Pleurotus sajor-caju alters its antioxidant, anticoagulant and antiproliferative properties in vitro. Carbohydr. Polym. 2011, 85, 514–521. [Google Scholar] [CrossRef] [Green Version]
- Vamanu, E. Antioxidant properties of polysaccharides obtained by batch cultivation of Pleurotus ostreatus mycelium. Nat. Prod. Res. 2013, 27, 1115–1118. [Google Scholar] [CrossRef]
- Lai, M.N.; Ng, L.T. Antioxidant and antiedema properties of solid-state cultures honey mushrooms, Armillaria mellea (higher Basidiomycetes), extracts and their polysaccharide and polyphenol contents. Int. J. Med. Mushrooms 2013, 15, 1–8. [Google Scholar] [CrossRef]
- Jhan, M.H.; Yeh, C.H.; Tsai, C.C.; Kao, C.T.; Chang, C.K.; Hsieh, C.W. Enhancing the Antioxidant Ability of Trametes versicolor Polysaccharopeptides by an Enzymatic Hydrolysis Process. Molecules 2016, 21, 1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ker, Y.B.; Chen, K.C.; Chyau, C.C.; Chen, C.C.; Guo, J.H.; Hsieh, C.L.; Wang, H.E.; Peng, C.C.; Chang, C.H.; Peng, R.Y. Antioxidant capability of polysaccharides fractionated from submerge-cultured Agaricus blazei mycelia. J. Agric. Food Chem. 2005, 53, 7052–7058. [Google Scholar] [CrossRef] [PubMed]
- Diallo, I.; Boudard, F.; Morel, S.; Vitou, M.; Guzman, C.; Saint, N.; Michel, A.; Rapior, S.; Traoré, L.; Poucheret, P.; et al. Antioxidant and Anti-Inflammatory Potential of Shiitake Culinary-Medicinal Mushroom, Lentinus edodes (Agaricomycetes), Sporophores from Various Culture Conditions. Int. J. Med. Mushrooms 2020, 22, 535–546. [Google Scholar] [CrossRef]
- Rani, P.; Lal, M.R.; Maheshwari, U.; Krishnan, S. Antioxidant Potential of Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (Higher Basidiomycetes) Cultivated on Artocarpus heterophyllus Sawdust Substrate in India. Int. J. Med. Mushrooms 2015, 17, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Mau, J.L.; Lin, H.C.; Chen, C.C. Antioxidant properties of several medicinal mushrooms. J Agric. Food Chem. 2002, 50, 6072–6077. [Google Scholar] [CrossRef] [PubMed]
- Dore, C.M.; Alves, M.G.; da Santos, M.G.L.; de Souza, L.A.; Baseia, I.G.; Leite, E.L. Antioxidant and anti-inflammatory properties of an extract rich in polysaccharides of the mushroom Polyporus dermoporus. Antioxidants 2014, 3, 730–744. [Google Scholar] [CrossRef]
- Fu, L.; Wang, Y.; Yang, Y.; Hao, L. Evaluation of the antioxidant activity of extracellular polysaccharides from Morchella esculenta. Food Funct. 2013, 4, 871–879. [Google Scholar] [CrossRef]
- Kogan, G.; Pajtinka, M.; Babincova, M.; Miadokova, E.; Rauko, P.; Slamenova, D.; Korolenko, T.A. Yeast cell wall polysaccharides as antioxidants and antimutagenes: Can they fight cancer. Neoplasma 2008, 5, 387–393. [Google Scholar]
- Rutkowski, K.; Sowa, P.; Rutkowska-Talipska, J.; Sulkowski, S.; Rutkowski, R. Allergic diseases: The price of civilisational progress. Postepy Dermatol. Alergol. 2014, 31, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Jesenak, M.; Banovcina, P.; Rennerova, Z.; Majtan, J. β-Glucans in the treatment and prevention of allergic diseases. Allergol. Immunopathol. 2012, 42, 149–156. [Google Scholar] [CrossRef]
- Del Giudice, M.M.; Maiello, N.; Capristo, C.; Alterio, E.; Capasso, M.; Perrone, L.; Ciprandi, G. Resveratrol plus carboxymethyl-β-glucan reduces nasal symptoms in children with pollen-induced allergic rhinitis. Curr. Med. Res. Opin. 2014, 30, 1931–1935. [Google Scholar] [CrossRef] [PubMed]
- Song, H.H.; Chae, H.S.; Oh, S.R.; Lee, H.K.; Chin, Y.W. Anti-inflammatory and anti-allergic effect of Agaricus blazei extract in bone marrow-derived mast cells. Am. J. Chin. Med. 2012, 40, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.K.; Kim, J.W.; Cho, H.R. Effect of β-glucan originated from Aureobasidium pullulans asthma induced by ovalbumin in mouse. Arch. Pharm. Res. 2012, 35, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Burg, A.R.; Quigley, L.; Jones, A.V.; O’Connor, G.M.; Boelte, K.; McVicar, D.W.; Orr, S.J. Orally administered β-glucan attenuates the Th2 response in a model of airway hypersensitivity. Springerplus 2016, 5, 815. [Google Scholar] [CrossRef] [Green Version]
- Jesenak, M.; Hrubisko, M.; Majtan, J.; Rennerova, Z.; Banovcin, P. Anti-allergic effect of pleuran (β-glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Phytother. Res. 2014, 28, 471–474. [Google Scholar] [CrossRef]
- Sarinho, E.; Medeiros, D.; Schor, D.; Rego Silva, A.; Sales, V.; Motta, M.E.; Costa, A.; Azoubel, A.; Rizzo, J.A. Production of interleukin-10 in asthmatic children after beta-1-3-glucan. Allergol. Immunopathol. 2009, 37, 188–192. [Google Scholar] [CrossRef]
- Yamada, J.; Hamuro, J.; Hatanaka, H.; Hamabata, K.; Kinoshita, S. Alleviation of seasonal allergic symptoms with suprfine β-1,3-glucan: A randomized study. J. Allergy Clin. Immunol. 2007, 119, 1119–1126. [Google Scholar] [CrossRef]
- Mahmood, F.; Hetland, G.; Nentwich, I.; Mirlashari, M.R.; Ghiasvand, R.; Nissen-Meyer, L.S.H. Agaricus blazei-Based Mushroom Extract Supplementation to Birch Allergic Blood Donors: A Randomized Clinical Trial. Nutrients 2019, 11, 2339. [Google Scholar] [CrossRef] [Green Version]
- Xie, Q.M.; Deng, J.F.; Deng, Y.M.; Shao, C.S.; Zhang, H.; Ke, C.K. Effects of Cryptoporus polysaccharide on rat allergic rhinitis associated with inhibitory exotoxin mRNA expression. J. Ethnopharmacol. 2006, 107, 424–430. [Google Scholar] [CrossRef]
- Mothana, R.A.A.; Awadh Ali, N.A.; Jansen, R.; Wegner, U.; Mentel, R.; Lindequist, U. Antiviral lanostanoid triterpenes from the fungus Ganoderma pfeifferi. Fitoterapia 2003, 74, 177–180. [Google Scholar] [CrossRef]
- Battulga, T.; Tumurbaatar, O.; Ganzorig, O.; Ishimura, T.; Kanamoto, T.; Nakashima, H.; Miyazaki, K.; Yoshida, T. Analysis of interaction between sulfated polysaccharides and HIV oligopeptides by surface plasmon resonance. Int. J. Biol. Macromol. 2019, 125, 909–914. [Google Scholar] [CrossRef]
- Seo, D.J.; Choi, C. Antiviral Bioactive Compounds of Mushrooms and Their Antiviral Mechanisms: A Review. Viruses 2021, 13, 350. [Google Scholar] [CrossRef] [PubMed]
- Hetland, G.; Johnson, E.; Eide, D.M.; Grinde, B.; Samuelsen, A.B.; Wiker, H. Antimicrobial effects of β-glucans and pectin and of the Agaricus blazei-based mushroom extract, andosan™ examples of mouse models for pneumococcal-, fecal bacterial-, and mycobacterial infections. In Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2013; pp. 889–898. [Google Scholar]
- Özcan, Ö.; Ertan, F. Beta-glucan Content, Antioxidant and Antimicrobial Activities of Some Edible Mushroom Species. Food Sci. Technol. 2018, 6, 47–55. [Google Scholar] [CrossRef] [Green Version]
- Liang, J.; Melican, D.; Cafro, L.; Palace, G.; Fisette, L.; Armstrong, R.; Patchen, M.L. Enhanced clearance of a multiple antibiotic resistant Staphylococcus aureus in rats treated with PGG-glucan is associated with increased leukocyte counts and increased neutrophil oxidative burst activity. Int. J. Immunopharmacol. 1998, 20, 595–614. [Google Scholar] [CrossRef]
- Persson Waller, K.; Grönlund, U.; Johannisson, A. Intramammary infusion of beta1,3-glucan for prevention and treatment of Staphylococcus aureus mastitis. J. Vet. Med. B Infect. Dis. Vet. Public Health 2003, 50, 121–127. [Google Scholar] [CrossRef]
- Markova, N.; Kussovski, V.; Drandarska, I.; Nikolaeva, S.; Georgieva, N.; Radoucheva, T. Protective activity of lentinan in experimental tuberculosis. Int. Immunopharmacol. 2003, 3, 1557–1562. [Google Scholar] [CrossRef]
- Chang, C.S.; Huang, S.L.; Chen, S.; Chen, S.N. Innate immune responses and efficacy of using mushrooms beta-glucan mixture (MBG) on orange-spotted grouper. Epinephelus coioides, aquaculture. Fish Shellfish. Immunol. 2013, 35, 115–125. [Google Scholar] [CrossRef] [PubMed]
- El-Boshy, M.E.; El-Ashram, A.M.; Abdelhamid, F.M.; Gadalla, H.A. Immunomodulatory effect of dietary Saccharomyces cerevisiae, beta-glucan and laminaran in mercuric chloride treated Nile tilapia (Oreochromis niloticus) and experimentally infected with Aeromonas hydrophila. Fish Shellfish Immunol. 2010, 28, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Glucan supplementation enhances the immune response against an influenza challenge in mice. Ann. Transl. Med. 2015, 3, 22. [Google Scholar] [CrossRef]
- Gordon, M.; Bihari, B.; Goosby, E.; Gorter, R.; Greco, M.; Guralnik, M.; Mimura, T.; Rudnicki, V.; Wong, R.; Kaneko, Y. A placebo controlled trail of the immune modulator, lentinan, in HIV-positive patients: A phase I/II trial. J. Med. 1998, 29, 305–330. [Google Scholar]
- Adotey, G.; Quarco, A.; Holliday, J.C.; Fofie, S.; Saaka, B. Effect of immunomodulating and antiviral agent of medicinal mushrooms (immune Assist 24/7 TM ) on CD+ T-lymphocyte counts of HIV-infected patents. Int. J. Med. Mushrooms 2011, 13, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.Q.; Li, J.W.; Chao, F.; Jin, M.; Wang, X.W.; Shen, Z.Q. Isolation, identification and function of a novel anti-HSV-1 protein from Grifola frondosa. Antiviral Res. 2007, 75, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Rouhier, P.; Kopp, M.; Begot, V.; Bruneteau, M.; Fritig, B. Structural features of fungal β-d-glucans for the efficient inhibition of the initiation of virus infection on Nicotiana tabacum. Phytochemistry 1995, 39, 57–62. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.Y.; Xia, X.M.; Li, P.; Wang, K.Y. Inhibitory effect of sulfated lentinan and lentinan against tobacco mosaic virus (TMV) in tobacco seedlings. Int. J. Biol. Macromol. 2013, 61, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Rincão, V.P.; Yamamoto, K.A.; Ricardo, N.M.P.S.; Soares, S.A.; Meirelles, L.D.P.; Nozawa, C.; Linhares, R.E.C. Polysaccharide and extracts from Lentinula edodes: Structural features and antiviral activity. Virol. J. 2012, 9, 37. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, T.L.; Chen, J.; Hu, Y.; Wang, D.; Fan, Y.; Wang, J.; Abula, S.; Zhang, J.; Qin, T.; Chen, X.; et al. In vitro antiviral activity of sulfated Auricularia auricula polysaccharides. Carbohydr. Polym. 2012, 90, 1254–1258. [Google Scholar] [CrossRef]
- Fan, X.; Hu, H.; Chen, D.; Yu, B.; He, J.; Yu, J.; Luo, J.; Eckhardt, E.; Luo, Y.; Wang, J.; et al. Lentinan administration alleviates diarrhea of rotavirus-infected weaned pigs via regulating intestinal immunity. J. Anim. Sci. Biotechnol. 2021, 12, 43. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.J.; Ferreira, I.C.F.R.; Dias, J.; Teixeira, V.; Martins, A.; Pintado, M. A review on antifungal activity of mushroom (Basidiomycetes) extracts and isolated compounds. Curr. Top. Med. Chem. 2013, 13, 2648–2659. [Google Scholar] [CrossRef] [Green Version]
- Waithaka, P.N.; Gathuru, E.M.; Githaiga, B.M.; Onkoba, K.M. Antimicrobial Activity of Mushroom (Agaricus bisporus) and Fungal (Trametes gibbosa) Extracts from Mushrooms and Fungi of Egerton Main Campus, Njoro Kenya. J. Biomed. Sci. 2017, 6, 3. [Google Scholar] [CrossRef] [Green Version]
- Merel, D.; Savoie, J.M.; Mata, G.; Salmones, D.; Ortega, C.; Atanasova, V.; Chéreau, S.; Monribot-Villanueva, J.L.; Guerrero-Analco, J.A. Methanolic Extracts from Cultivated Mushrooms Affect the Production of Fumonisins B and Fusaric Acid by Fusarium verticillioides. Toxins 2020, 12, 366. [Google Scholar] [CrossRef] [PubMed]
- Minari, M.C.; Rincão, V.P.; Soares, S.A.; Ricardo, N.M.; Nozawa, C.; Linhares, R.E. Antiviral properties of polysaccharides from Agaricus brasiliensis in the replication of bovine herpesvirus 1. Acta Virol. 2011, 55, 255–259. [Google Scholar] [CrossRef] [Green Version]
- Drummond, R.A.; Brown, G.D. The role of Dectin -1 in the host defence against fungal infections. Curr. Opin. Microbiol. 2011, 14, 392–399. [Google Scholar] [CrossRef]
- Roeder, A.; Kirschning, C.J.; Rupec, R.A.; Schaller, M.; Weindl, G.; Korting, H.C. Toll-like receptors as key mediators in innate antifungal immunity. Med. Mycol. 2004, 42, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhao, Y.; Zhang, F.; Wang, Q.; Li, T.; Liu, Z.; Wang, J.; Qin, Y.; Zhang, X.; Yan, X.; et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin. Immunol. 2020, 214, 108393. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.S.; Suryaprakash, V.; Senthilkumar, R.; Preethy, S.; Katoh, S.; Ikewaki, N.; Abraham, S.J.K. Role of Immune Dysregulation in Increased Mortality Among a Specific Subset of COVID-19 Patients and Immune-Enhancement Strategies for Combatting Through Nutritional Supplements. Front. Immunol. 2020, 11, 1548. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- van Steenwijk, H.P.; Bast, A.; de Boer, A. Immunomodulating Effects of Fungal Beta-Glucans: From Traditional Use to Medicine. Nutrients 2021, 13, 1333. [Google Scholar] [CrossRef]
- Netea, M.G.; Joosten, L.A.B.; Latz, E.; Mills, K.H.G.; Natoli, G.; Stunnenberg, H.G.; O’Neill, L.A.J.; Xavier, R.J. Trained immunity: A program of innate immune memory in health and disease. Science 2016, 352, aaf1098. [Google Scholar] [CrossRef] [Green Version]
- Geller, A.; Yan, J. Could the Induction of Trained Immunity by β-Glucan Serve as a Defense against COVID-19? Front. Immunol. 2020, 11, 1782. [Google Scholar] [CrossRef]
- dos Santos, J.C.; Barroso de Figueiredo, A.M.; Teodoro Silva, M.V.; Cirovic, B.; de Bree, L.C.J.; Damen, M.S.M.A.; Moorlag, S.J.C.F.M.; Gomes, R.S.; Helsen, M.M.; Oosting, M.; et al. β-Glucan-Induced Trained Immunity Protects against Leishmania braziliensis Infection: A Crucial Role for IL-32. Cell Rep. 2019, 28, 2659–2672.e6. [Google Scholar] [CrossRef] [Green Version]
- Leonhardt, J.; Große, S.; Marx, C.; Siwczak, F.; Stengel, S.; Bruns, T.; Bauer, R.; Kiehntopf, M.; Williams, D.L.; Wang, Z.Q.; et al. Candida albicans β-Glucan Differentiates Human Monocytes into a Specific Subset of Macrophages. Front. Immunol. 2018, 9, 2818. [Google Scholar] [CrossRef]
- Wang, X.; Ha, D.; Yoshitake, R.; Chen, S. White button mushroom interrupts tissue AR-mediated TMPRSS2 expression and attenuates pro-inflammatory cytokines in C57BL/6 mice. NPJ Sci. Food 2021, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Baruah, K.; Cox, E.; Vanrompay, D.; Bossier, P. Structure-Functional Activity Relationship of β-Glucans from the Perspective of Immunomodulation: A Mini-Review. Front. Immunol. 2020, 11, 658. [Google Scholar] [CrossRef] [Green Version]
- Jawhara, S. How to boost the immune defence prior to respiratory virus infections with the special focus on coronavirus infections. Gut Pathog. 2020, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. J. Fungi 2020, 6, 356. [Google Scholar] [CrossRef]
- Ikewaki, N.; Iwasaki, M.; Kurosawa, G.; Rao, K.S.; Lakey-Beitia, J.; Preethy, S.; Abraham, S.J.K. β-Glucans: Wide-spectrum immune-balancing food-supplement-based enteric (β-WIFE) vaccine adjuvant approach to COVID-19. Hum. Vaccines Immunother. 2021, 17, 2808–2813. [Google Scholar] [CrossRef]
- Ikewaki, N.; Iwasaki, M.; Abraham, S.J.K. Biological response modifier glucan through balancing of blood glucose may have a prophylactic potential in COVID-19 patients. J. Diabetes Metab. Disord. 2020, 19, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ikewaki, N.; Rao, K.S.; Archibold, A.D.; Iwasaki, M.; Senthilkumar, R.; Preethy, S.; Katoh, S.; Abraham, S.J.K. Coagulopathy associated with COVID-19–Perspectives and Preventive strategies using a biological response modifier Glucan. Thromb. J. 2020, 18, 27. [Google Scholar] [CrossRef]
- Ikewaki, N.; Fujii, N.; Onaka, T.; Ikewaki, S.; Inoko, H. Immunological actions of Sophy beta-glucan (beta-1,3-1,6 glucan), currently available commercially as a health food supplement. Microbiol. Immunol. 2007, 51, 861–873. [Google Scholar] [CrossRef] [Green Version]
- Shah, V.B.; Williams, D.L.; Keshvara, L. Beta-glucan attenuates TLR2- and TLR4-mediated cytokine production by microglia. Neurosci. Lett. 2009, 458, 111–115. [Google Scholar] [CrossRef] [Green Version]
- Raghavan, K.; Kandaswamy, R.S.; Ikewaki, N.; Iwasaki, M.; Abrahame, S.J.K. Potentials to alleviate coagulopathy and enhance microglial function of beta (β)-glucans, making them worth a clinical study for COVID-19’s neurological sequalae. J. Neurol. Sci. 2021, 427, 117554. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, K.T.; Carbajal, K.S.; Segal, B.M.; Giger, R.J. Neuroinflammation triggered by β-glucan/dectin-1 signaling enables CNS axon regeneration. Proc. Natl. Acad. Sci. USA 2015, 112, 2581–2586. [Google Scholar] [CrossRef] [Green Version]
- Di Pierro, F.; Bertuccioli, A.; Cavecchia, I. Possible therapeutic role of a highly standardized mixture of active compounds derived from cultured Lentinula edodes mycelia (AHCC) in patients infected with 2019 novel coronavirus. Minerva Gastroenterol. Dietol. 2020, 66, 172–176. [Google Scholar] [CrossRef]
- Sevindik, M. Mushrooms as natural antiviral sources and supplements foods against coronavirus (COVID-19). J Bacteriol. Mycol. Open Access 2021, 9, 73–76. [Google Scholar] [CrossRef]
- Mallakpour, S.; Azadi, E.; Hussain, C.M. Chitosan, alginate, hyaluronic acid, gums, and β-glucan as potent adjuvants and vaccine delivery systems for viral threats including SARS-CoV-2: A review. Int. J. Biol. Macromol. 2021, 182, 1931–1940. [Google Scholar] [CrossRef] [PubMed]
- Soares, E.; Groothuismink, Z.M.A.; Boonstra, A.; Borges, O. Glucan particles are a powerful adjuvant for the HBsAg, favoring antiviral immunity. Mol. Pharm. 2019, 16, 1971–1981. [Google Scholar] [CrossRef]
- Jin, J.W.; Tang, S.Q.; Rong, M.Z.; Zhang, M.Q. Synergistic effect of dual targeting vaccine adjuvant with aminated β-glucan and CpG-oligodeoxynucleotides for both humoral and cellular immune responses. Acta Biomater. 2018, 78, 211–223. [Google Scholar] [CrossRef]
- Vancraeyneste, H.; Charlet, R.; Guerardel, Y.; Choteau, L.; Bauters, A.; Tardivel, M.; François, N.; Dubuquoy, L.; Soloviev, D.; Poulain, D.; et al. Short fungal fractions of beta-1,3 glucans affect platelet activation. Am. J. Physiol. Heart Circ. Physiol. 2016, 3113, H725–H734. [Google Scholar] [CrossRef] [PubMed]
- Aida, F.M.N.A.; Shuhaimi, M.; Yazid, M.; Maaruf, A.G. Mushrooms as a potential source of prebiotics: A review. Trend. Food Sci. Technol. 2009, 20, 567–575. [Google Scholar] [CrossRef]
- Yang, S.; Xie, X.; Ma, J.; He, X.; Li, Y.; Du, M.; Li, L.; Yang, L.; Wu, Q.; Chen, W.; et al. Selective Isolation of Bifidobacterium from Human Faeces Using Pangenomics, Metagenomics, and Enzymology. Front. Microbiol. 2021, 12, 649698. [Google Scholar] [CrossRef] [PubMed]
- Mitsou, E.K.; Saxami, G.; Stamoulou, E.; Kerezoudi, E.; Terzi, E.; Koutrotsios, G.; Bekiaris, G.; Zervakis, G.I.; Mountzouris, K.C.; Pletsa, V.; et al. Effects of Rich in Β-Glucans Edible Mushrooms on Aging Gut Microbiota Characteristics: An In Vitro Study. Molecules 2020, 25, 2806. [Google Scholar] [CrossRef] [PubMed]
- Nikitina, O.; Cherno, N.; Osolina, S.; Naumenko, K. Yeast glucan and glucan-containing mushroom biopolymer complexes–Stimulators of microflora growth. Int. Food Res. J. 2017, 24, 2220–2227. [Google Scholar]
- Zhao, R.; Yang, W.; Pei, F.; Zhao, L.; Hu, Q. In vitro fermentation of six kinds of edible mushrooms and its effects on fecal microbiota composition. LWT-Food Sci. Technol. 2018, 96, 627–635. [Google Scholar] [CrossRef]
- Vamanu, E.; Gatea, F.; Sârbu, I. In Vitro Ecological Response of the Human Gut Microbiome to Bioactive Extracts from Edible Wild Mushrooms. Molecules 2018, 23, 2128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nie, Y.; Lin, Q.; Luo, F. Effects of Non-Starch Polysaccharides on Inflammatory Bowel Disease. Int. J. Mol. Sci. 2017, 18, 1372. [Google Scholar] [CrossRef] [Green Version]
- Gurung, M.; Li, Z.; You, H.; Rodrigues, R.; Jump, D.B.; Morgun, A.; Shulzhenko, N. Role of gut microbiota in type 2 diabetes pathophysiology. EBioMedicine 2020, 51, 102590. [Google Scholar] [CrossRef] [Green Version]
- Guo, W.L.; Pan, Y.Y.; Li, L.; Li, T.T.; Liu, B.; Lv, X.C. Ethanol extract of Ganoderma lucidum ameliorates lipid metabolic disorders and modulates the gut microbiota composition in high-fat diet fed rats. Food Funct. 2018, 9, 3419–3431. [Google Scholar] [CrossRef]
- Diling, C.; Yinrui, G.; Longkai, Q.; Xiaocui, T.; Yadi, L.; Jiaxin, F.; Xiangxiang, Z.; Miao, Z.; Ou, S.; Dongdong, W.; et al. Metabolic regulation of Ganoderma lucidum extracts in high sugar and fat diet-induced obese mice by regulating the gut-brain axis. J. Funct. Foods 2020, 65, 103639. [Google Scholar] [CrossRef]
- Li, M.; Yu, L.; Zhao, J.; Zhang, H.; Chen, W.; Zhai, Q.; Tian, F. Role of dietary edible mushrooms in the modulation of gut microbiota. J. Funct. Foods 2021, 83, 104538. [Google Scholar] [CrossRef]
- Ruthes, A.C.; Cantu-Jungles, T.M.; Cordeiro, L.M.C.; Iacomini, M. Prebiotic potential of mushroom d-glucans: Implications of physicochemical properties and structural features. Carbohydr. Polym. 2021, 262, 117940. [Google Scholar] [CrossRef]
- Muthukumaran, J.; Xiao, J.; Xu, B.A. Critical Review on Health Promoting Benefits of Edible Mushrooms through Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 1934. [Google Scholar] [CrossRef] [Green Version]
| Name of β-Glucan | Abbreviation | Mushroom Species | Glucan Structure | Reference |
|---|---|---|---|---|
| Krestin | PSK PSP | Trametes versicolor | 1,3-β-glucan 1,4-β-glucan multi-sugar–protein complex containing mainly 1,3-β-D-glucans | [39,40] |
| Tylopilan | - | Tylopillus felleus (Bull.) | 1,3-1,6-β-glucan | [41] |
| Lentinan | LNT | Lentinula edodes | 1,3-1,6-β-glucan | [42] |
| Pleuran | - | Pleurotus ostreatus | 1,3-β- glucan with galactose and mannose | [43] |
| Schizophyllan | SPG | Schizophyllum commune | 1,3-1,6-β-glucan | [44] |
| MD-fraction | Grifola frondosa | 1,6-1,3-β-glucan | [45] | |
| Grifolan | GRN | 1,3-1,6-β-glucan | [46] | |
| Scleroglucan | SSG | Sclerotium glucanicum Sclerotium rolfsii | 1,3-1,6-β-glucan | [47] |
| No. | Type of Glucan | Mechanism of Action in Prevention and Treatment of COVID-19 | References |
|---|---|---|---|
| 1. | AFO-202-β–glucan (β-1,3-1,6 glucan from black yeast Aureobasidium pullulans) | Potential as a vaccine adjuvant against COVID-19; Prevention of COVID-19-associated coagulopathy. | [179] |
| 2. | AFO-202 -β-glucans (β-1,3-1,6 glucan from black yeast Aureobasidium pullulans) | Regulation of blood glucose and lipid levels by β-Glucans as an indispensable tool of defense against COVID-19. | [180,181] |
| 3. | AFO-202-β–glucan (β-1,3-1,6 glucan from black yeast Aureobasidium pullulans) | Immune enhancement by decreasing hyper-inflammation factors (IL-6) and minimizing the likelihood of a cytokine storm; increasing IFN-γ, sFAS, and factors like IL-7; and enhancing anti-viral cytotoxic immunity, (T cells, NK cells, macrophages, antibody production by B cells). | [168] |
| 4. | AFO-202-β–glucan (β-1,3-1,6 glucan from black yeast Aureobasidium pullulans) | Immune regulatory and enhancing immune system; Immune stimulator that can activate macrophages and have positive immune actions on B-lymphocytes, natural killer cells, and suppressor T cells in the immune system. | [168,182] |
| 5. | 1,3-β-D-glucan (from Saccharomyces cerevisiae) | Prevention and treatment of excessive microglia activation during chronic inflammation characteristic of COVID-19 course. | [183,184] |
| 6. | 1,3-β-D-glucan (curdlan and fragmentated zymozan- proteoglucan from Saccharomyces cerevisiae) | Potentials to enhance microglial function and regeneration of CNS axons in COVID-19 neurological sequalae. | [184,185] |
| 7. | 1,3-1,6-β-D glucans (from shiitake mushroom Lentinula edodes) | Immunomodulatory and pulmonary cytoprotective effects. | [14] |
| 8. | β-glucans (from mushrooms as Lentinula edodes and Pleurotus ostreatus) | Immunomodulating effects. | [170] |
| 9. | β-glucan (from white button mushroom Agaricus bisporus) | Interrupts AR (androgen receptor)-mediated TMPRSS2 (Transmembrane protease serine 2) expression that is involved in viral entry, through its AR antagonistic activity; Attenuates serum pro-inflammatory cytokines and reduces MDSC (myeloid-derived suppressor cells) counts that are involved in the host response to viral infection, through its immunoregulatory activity). | [175] |
| 10. | 1,4-α–glucan (from Lentinula edodes) | Modulation and activation of NK-cells, T-cells, and γδ-T (gamma delta T). | [186] |
| 11. | β-glucans (from edible and medicinal mushrooms) | Support the immune system before, during, and after COVID-19. | [187] |
| 12. | Aminated β-glucan (AβG) | Potential vaccine adjuvant, immunopotentiator for simulation of antigen-presenting cells for T cells’ activation. | [188,189,190] |
| 13. | β-glucan (from yeast, Saccharomysces cerevisiae) | Decreasing platelet activation by increasing TGF-β1 production. Decreasing the concentration of pro-inflammatory cytokines IL-6, which indirectly activates platelets and thrombin production. Prevention of thrombosis during the course of COVID-19. | [169,177,191] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirończuk-Chodakowska, I.; Kujawowicz, K.; Witkowska, A.M. Beta-Glucans from Fungi: Biological and Health-Promoting Potential in the COVID-19 Pandemic Era. Nutrients 2021, 13, 3960. https://doi.org/10.3390/nu13113960
Mirończuk-Chodakowska I, Kujawowicz K, Witkowska AM. Beta-Glucans from Fungi: Biological and Health-Promoting Potential in the COVID-19 Pandemic Era. Nutrients. 2021; 13(11):3960. https://doi.org/10.3390/nu13113960
Chicago/Turabian StyleMirończuk-Chodakowska, Iwona, Karolina Kujawowicz, and Anna Maria Witkowska. 2021. "Beta-Glucans from Fungi: Biological and Health-Promoting Potential in the COVID-19 Pandemic Era" Nutrients 13, no. 11: 3960. https://doi.org/10.3390/nu13113960
APA StyleMirończuk-Chodakowska, I., Kujawowicz, K., & Witkowska, A. M. (2021). Beta-Glucans from Fungi: Biological and Health-Promoting Potential in the COVID-19 Pandemic Era. Nutrients, 13(11), 3960. https://doi.org/10.3390/nu13113960